Abstract
Background:
Air dispersal of respiratory viruses other than SARS-CoV-2 has not been systematically reported. The incidence and factors associated with air dispersal of respiratory viruses are largely unknown.
Methods:
We performed air sampling by collecting 72,000 L of air over 6 hours for pediatric and adolescent patients infected with parainfluenza virus 3 (PIF3), respiratory syncytial virus (RSV), rhinovirus, and adenovirus. The patients were singly or 2-patient cohort isolated in airborne infection isolation rooms (AIIRs) from December 3, 2021, to January 26, 2022. The viral load in nasopharyngeal aspirates (NPA) and air samples were measured. Factors associated with air dispersal were investigated and analyzed.
Results:
Of 20 singly isolated patients with median age of 30 months (range, 3 months–15 years), 7 (35%) had air dispersal of the viruses compatible with their NPA results. These included 4 (40%) of 10 PIF3-infected patients, 2 (66%) of 3 RSV-infected patients, and 1 (50%) of 2 adenovirus-infected patients. The mean viral load in their room air sample was 1.58×103 copies/mL. Compared with 13 patients (65%) without air dispersal, these 7 patients had a significantly higher mean viral load in their NPA specimens (6.15×107 copies/mL vs 1.61×105 copies/mL; P < .001). Another 14 patients were placed in cohorts as 7 pairs infected with the same virus (PIF3, 2 pairs; RSV, 3 pairs; rhinovirus, 1 pair; and adenovirus, 1 pair) in double-bed AIIRs, all of which had air dispersal. The mean room air viral load in 2-patient cohorts was significantly higher than in rooms of singly isolated patients (1.02×104 copies/mL vs 1.58×103 copies/mL; P = .020).
Conclusion:
Air dispersal of common respiratory viruses may have infection prevention and public health implications.
The transmission of respiratory viruses by droplet or contact routes in healthcare settings is a principal dogma of infection prevention. The transmission-based precautions as illustrated in the recommendations for isolation precautions in hospitals by the Centers for Disease Control and Prevention (CDC) were based on this dogma since 1996. 1,2 However, transmission of respiratory viruses by airborne route has been implicated in community settings over the past decades, including transmission of influenza A in a commercial airliner 3 or within household, 4 and transmission of rhinovirus among game card players. 5 Spread of respiratory syncytial virus (RSV) by aerosol was also suggested in the healthcare setting. 6 During the outbreak of severe acute respiratory syndrome (SARS) in 2003 by SARS coronavirus 1 (SARS-CoV-1), airborne transmission of SARS-CoV-1 was observed in both community and healthcare settings. 7,8 With the emergence of coronavirus disease 2019 (COVID-19) due to SARS coronavirus 2 (SARS-CoV-2), airborne transmission has been increasingly reported in the healthcare and community settings. 9–14
Since we have been performing air sampling to detect of SARS-CoV-2 RNA in the airborne infection isolation room (AIIR) of hospitals and community treatment facilities during the COVID-19 pandemic, 15–18 we would like to know whether air dispersal also occurs in patients infected with common respiratory viruses other than SARS-CoV-2. Here, we performed room air sampling of pediatric and adolescent patients with laboratory-confirmed respiratory viral infection. These findings may have implications in infection prevention and public health measures.
Methods
Setting
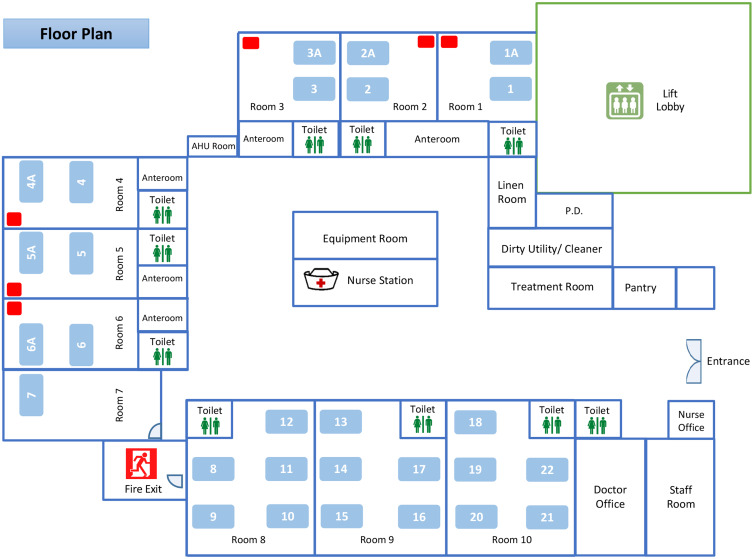
This study was conducted in a pediatric ward of Queen Mary Hospital, a 1,700-bed, university-affiliated, teaching hospital in Hong Kong. The pediatric ward contains 28 beds arranged as 6 double-bed AIIRs (room 1–6), 1 single-bed (room 7), and three 5-bed cubicles (room 8–10) without pressure difference between the cubicles and the common area (Fig. 1). The air changes per hour in the AIIRs and the cubicles are 12 and 6, respectively. The temperature and humidity of the AIIRs are set at 22oC and 65%, respectively. The AIIR is prioritized to care for patients aged ≤17 years and infected with pathogens of airborne transmission. Other patients who are aged ≤17 years and with fever and respiratory symptoms will also be admitted through the emergency department to this pediatric ward. This study was approved by the Institutional Review Board of The University of Hong Kong/Hospital Authority Hong Kong West Hospital Cluster.
Fig. 1.
The floor plan of a pediatric ward of Queen Mary Hospital. Note. The pediatric ward contains 28 beds in 6 double-bed airborne infection isolation rooms (AIIRs) (bed numbers 1, 1A, 2, 2A, 3, 3A, 4, 4A, 5, 5A, 6, and 6A), 1 single-bed room (bed number 7), and three 5-bed cubicles (bed numbers 8–22) without pressure difference between the cubicles and the common area. The air sampler is denoted as a red rectangle placed at the corner of the AIIRs at a distance >2 m from the patient’s head.
Microbiological diagnosis of patients with respiratory symptoms
Upon admission, nasopharyngeal aspirates (NPA) were collected for rapid molecular diagnostic test. The NPA in viral transport medium (VTM) were simultaneously tested for 23 pathogens using the BIOFIRE FILMARRAY Respiratory 2.1 plus Panel (bioMérieux, Marcy l’Étoile, France). 19
Collection of air sample from patients with respiratory viral infection
Patients infected by a single virus detected by BIOFIRE FILMARRAY Respiratory 2.1 plus Panel were eligible for this study. Patients with newly diagnosed respiratory viral infection and singly isolated in AIIRs were selected. If >1 eligible patient was available on the day of air sample collection, only 1 patient was chosen at random. In addition, air sampling in double-bed AIIRs with cohort patients was performed. Repeated air samples for the same cohort of patients may be performed to monitor the change in viral load during hospitalization. Verbal consent was obtained from each patient or their parent.
We collected the air samples using an AerosolSense Sampler (Thermo Fisher Scientific, MA) as previously described. 17 Briefly, the air sample was collected through an omnidirectional inlet and was directed toward the collection substrate through an accelerating slit impactor at a flow rate of 200 L per minute for 6 hours. Air samples of 72,000 L were collected. The samples were sent to the microbiology laboratory within 30 minutes for further processing. The air sampler was placed at the corner of the AIIR at a distance >2 m from the patient’s head (Fig. 1).
Viral load assessment of air and clinical samples
Upon receiving the air samples, the collection substrate was immersed in 2 mL VTM, and 1 mL medium was used for total nucleic acid extraction using the eMAG extraction system (bioMérieux, Marcy-l’Etoile, France) following the manufacturer’s instructions. Quantifications of viral RNA or DNA in the air samples were performed using in-house real-time reverse transcription polymerase chain reaction (RT-PCR) as previously described. 20–22 The choice of in-house RT-PCR depended on the virological finding of clinical sample.
For the NPA specimens, total nucleic acid extraction was performed using 250 µL of the specimen. RT-PCRs for specific respiratory viruses were performed for viral load assay as described above.
Epidemiological characteristics of patients with air dispersal of respiratory viruses
The demographics, clinical symptoms, presence of underlying diseases, and the medical treatment among patients with or without detectable viral genome by air samples were analyzed. The use of surgical mask by patients during air sample collection was recorded. A case–control analysis was performed to analyze the factors associated with air dispersal of respiratory viruses. Case and control were defined as patients with or without air dispersal of respiratory viruses, respectively.
Statistical analysis
The factors associated with air dispersal of respiratory viruses were analyzed using the Student t test or Fisher’s exact test where appropriate. A 2-sided P value <.05 was considered statistically significant.
Results
Setting
Between December 3, 2021, and January 26, 2022, air sampling was conducted on 30 working days in the pediatric ward for 34 patients. Their NPA revealed parainfluenza virus 3 (PIF3) in 14 patients (41.1%), RSV in 9 patients (26.5%), human rhinovirus/enterovirus in 7 patients (20.6%), and adenovirus in 4 patients (11.8%). Of these 34 patients, 20 patients were singly isolated in AIIRs and another 14 patients shared double-bed AIIRs in which the patients were place near the air supply.
Epidemiological characteristics of patients with air dispersal of respiratory viruses
Of 20 singly isolated patients, 9 (45%) were male. The median age was 30 months (range, 3 months–15 years). Their NPA revealed PIF3 in 10 patients (50%), human rhinovirus/enterovirus in 5 patients (25%), RSV in 3 patients (15%), and adenovirus in 2 patients (10%). Rhinovirus-specific RT-PCR confirmed that the 5 patients with human rhinovirus/enterovirus detection had rhinovirus in their NPA specimens. Of 20 patients, 7 (35%) had air dispersal of the same respiratory viruses (Table 1). None of these 20 patients wore a surgical mask during air sampling. In the case–control analysis, case patients had a significantly higher mean viral load in the NPA than the controls (Table 2). Of 7 patients with air dispersal of respiratory viruses, the mean viral load in the air samples was 1.58 ×103 copies/mL (range, 63–7.60×103 copies/mL).
Table 1.
Epidemiological Characteristics of Patients Who Were Singly Isolated in Airborne Infection Isolation Room With Respiratory Tract Infection Associated With Detectable Viral Genome by Air Sampler
| Patient | Sex/age | Symptoms (Underlying Disease, If Any) | Respiratory Virus | Viral Load in NPA (Date of Collection) | Viral Load in Air (Date of Collection) a |
Treatment |
|---|---|---|---|---|---|---|
| 1 | M/3 y | Fever, cough, SOB (asthma) | PIF | 2.87×107 copy/mL (2 Dec 2021) |
763 copy/mL (6 Dec 2021) b |
Salbutamol puff, prednisolone, paracetamol, chlorphenamine |
| 2 | F/3 y | Fever, cough, RN | PIF | 9.26×106 copy/mL (11 Dec 2021) |
162 copy/mL (13 Dec 2021) b |
Amoxicillin-clavulanate, paracetamol |
| 3 | F/3 mo | Cough | PIF | 9.76×107 copy/mL (20 Dec 2021) |
1,435 copy/mL (22 Dec 2021) b |
Chlorphenamine |
| 4 | M/31 mo | Fever, cough, SOB (cyclical neutropenia, asthma) | PIF | 3.95×107 copy/mL (26 Dec 2021) |
63 copy/mL (28 Dec 2021) b |
Salbutamol puff, prednisolone |
| 5 | M/29 mo | Fever, cough, RN | RSV | 1.64×108 copy/mL (28 Dec 2021) |
619 copy/mL (29 Dec 2021) b |
Paracetamol |
| 6 | F/20 mo | Fever, SOB | RSV | 8.76×107 copy/mL (9 Jan 2022) |
382 copy/mL (17 Jan 2022) b |
Salbutamol puff |
| 7 | M/12 mo | Fever | Adenovirus | 3.92×105 copy/mL (7 Dec 2021) |
7,602 copy/mL (8 Dec 2021) c |
Paracetamol |
Note. NPA, nasopharyngeal aspirates; PIF, parainfluenza virus; RN, running nose; RSV, respiratory syncytial virus; SOB, shortness of breath.
72,000 L of air was collected over a 6-h period for each air sample. All patients did not wear surgical mask during air sample collection. During the viral load assay for air samples, the collection substrate was immersed in 2 mL of viral transport medium. Therefore, the viral load in air is expressed as the copy of viral genome per mL of viral transport medium.
Detectable viral RNA in air.
Detectable viral DNA in air.
Table 2.
Case–Control Analysis of Patients With or Without Air Dispersal of Respiratory Viruses During Respiratory Tract Infection
| Variable | Patients With Air Dispersal of Respiratory Viruses (n = 7), No. (%) a | Patients Without Air Dispersal of Respiratory Viruses (n = 13), No. (%) a | P Value |
|---|---|---|---|
| Age, mean mo ± SD | 21±14 | 48±57 | .312 |
| Sex, male | 4 (57.1) | 5 (38.5) | .642 |
| Respiratory viruses | |||
| Parainfluenza virus 3 | 4 (57.1) | 6 (46.2) | 1 |
| Respiratory syncytial virus | 2 (28.6) | 1 (7.7) | .270 |
| Rhinovirus | 0 | 5 (38.5) | .114 |
| Adenovirus | 1 (14.3) | 1 (7.7) | .158 |
| Symptoms | |||
| Fever | 6 (85.7) | 5 (38.5) | .070 |
| Cough | 5 (71.4) | 4 (30.8) | .160 |
| Running nose | 2 (28.6) | 4 (30.8) | 1 |
| SOB | 3 (42.9) | 2 (15.4) | .290 |
| Viral load of NPA | |||
| Mean copy/mL | 6.15×107 | 1.61×105 | <.001 |
| ≥5 log10 b | 7 (100) | 5 (38.5) | .015 |
| Day of air sampling after NPA collection (mean ± SD) c | 2.57±1.68 | 2.23 ± 1.93 | .713 |
| Use of medication | |||
| Salbutamol | 3 (42.9) | 3 (23.1) | .613 |
| Corticosteroid (inhaled or oral) | 2 (28.6) | 2 (15.4) | .587 |
Note. NPA, nasopharyngeal aspirates; SD, standard deviation; SOB, shortness of breath.
Units unless otherwise indicated.
Viral load of NPA ≥ 5 log10 indicates high viral load in the clinical specimens.
Each patient had one air sample collection during hospitalization.
Of another 14 patients shared double-bed AIIRs, 6 (42.9%) were male. The median age was 15 months (range, 65 days–10 years). These 14 patients were grouped into 7 pairs with the same virological diagnosis in each double-bed AIIR (Table 3). Of these 14 patients, the mean viral load of respiratory viruses in their NPA was 4.64×107 copies/mL (range, 5.33×103 to 1.68×108 copies/mL). Except for a 10-year-old girl with RSV infection, all patients in the double-bed AIIRs did not wear surgical mask during air sampling. All air samples were positive, with a mean viral load of 1.02×104 copies/mL (range, 10–4.99×104 copies/mL). The mean viral load in air samples was significantly higher in AIIRs housing 2 patients than in AIIRs for singly isolated patients (1.02×104 copies/mL vs 1.58×103 copies/mL; P = .020).
Table 3.
Epidemiological Characteristics of Patients Under Cohort Nursing in Airborne Infection Isolation Room With Respiratory Tract Infection Associated With Detectable Viral Genome by Air Sampler
| Pair of Patients (Episode of Air Sampling) | Sex/Age | Symptoms (Underlying Disease, If Any) | Respiratory Virus | Viral Load in NPA (Date of Collection) |
Viral Load in Air (Date of Collection) a |
Treatment [Remark] |
|---|---|---|---|---|---|---|
| 1 (1) | F/65 d | Cough (Down syndrome) | RSV | 3.47×107 copy/mL (23 Dec 2021) |
10 copy/mL (30 Dec 2021) b |
Salbutamol puff, paracetamol |
| 1 (1) | F/10 y | Seizure (epilepsy) | RSV | 4.10×107 copy/mL (29 Dec 2021) |
Nil [wearing mask in AIIR] | |
| 2 (2) | F/5 mo | Fever, cough, RN | RSV | 8.30×105 copy/mL (6 Jan 2022) |
5,745 copy/mL (11 Jan 2022) b |
Salbutamol puff, amoxicillin-clavulanate |
| 2 (2) | F/10 mo | Fever, cough, RN | RSV | 1.97×107 copy/mL (11 Jan 2022) |
Amoxicillin-clavulanate | |
| 3 (3–5) | F/5 mo | Fever, cough, RN | RSV | 5.42×107 copy/mL (6 Jan 2022) |
4.99×105 copy/mL (12 Jan 2022) b ; 691 copy/mL (13 Jan 2022) b ; 474 copy/mL (14 Jan 2022) b |
Salbutamol puff, amoxicillin-clavulanate |
| 3 (3–5) | F/20 mo | Fever, SOB | RSV | 8.76×107 copy/mL (9 Jan 2022) |
Salbutamol puff | |
| 4 (6–7) | M/6 y | Fever, cough (cerebral palsy) | PIF | 1.68×108 copy/mL (4 Jan 2022) | 1.42 ×105 copy/mL (4 Jan 2022) b ; 9,188 copy/mL (5 Jan 2022) b |
Salbutamol puff, paracetamol |
| 4 (6–7) | M/24 mo | Fever (DD) | PIF | 3.49×107 copy/mL (3 Jan 2022) |
Paracetamol | |
| 5 (8) | F/4 mo | Cough | PIF | 2.07×104 copy/mL (20 Jan 2022) | 1.27×105 copy/mL (21 Jan 2022) b |
Salbutamol puff |
| 5 (8) | M/3 y | Fever, cough | PIF | 8.21×107 copy/mL (20 Jan 2022) |
Nil | |
| 6 (9) | M/18 mo | Cough, RN, SOB | Rhinovirus | 6.17×105 copy/mL (18 Jan 2022) | 54 copy/mL (20 Jan 2022) b |
Salbutamol puff, paracetamol |
| 6 (9) | M/11 mo | Vomiting | Rhinovirus | 3.38×107 copy/mL (18 Jan 2022) |
Nil | |
| 7 (10) | F/4 y | Fever, vomiting | Adenovirus | 9.15×107 copy/mL (24 Jan 2022) | 5,150 copy/mL (25 Jan 2022) c |
Paracetamol |
| 7 (10) | M/9 mo | Cough, RN, SOB (CDH) | Adenovirus | 5.33×103 copy/mL (23 Jan 2022) |
Salbutamol puff |
Note. AIIR, airborne infection isolation room; CDH, congenital heart disease; DD, developmental delay; NPA, nasopharyngeal aspirates; PIF, parainfluenza virus; RN, running nose; RSV, respiratory syncytial virus; SOB, shortness of breath.
72,000 L of air was collected over a 6-h period for each air sample. Except for a 10-year-old girl, all patients did not wear surgical masks during the air sample collection. During the viral load assay for air samples, the collection substrate was immersed in 2 mL viral transport medium. Therefore, the viral load in air is expressed as the copy of viral genome per mL of viral transport medium.
Detectable viral RNA in air.
Detectable viral DNA in air.
Discussion
Air dispersal of respiratory viruses including PIF3, RSV, rhinovirus, and adenovirus were documented by the detection of viral load in the 72,000 L of air samples collected inside the AIIRs occupied by patients with symptomatic infections. In addition to the previous reports of airborne transmission of respiratory viruses, 5,6 air dispersal of PIF3 was also recognized. Instead of collecting the exhaled air from the individual patients 23–26 or performing the air sampling in the settings of emergency room or outpatient clinics with various confounding factors in the environment, 27,28 this study is the first to demonstrate air dispersal in singly isolated patients with environmental control of air change, flow, temperature, and humidity in the AIIRs. Of 20 infected patients singly isolated in AIIRs, air dispersal was detected in 35%. The presence of air dispersal was only associated with the viral load in NPA but was not related to demographic characteristics, clinical symptoms, or use of bronchodilator and inhaled corticosteroids among the singly isolated patients. In addition, the mean viral load of respiratory viruses in room air sample was significantly higher in the AIIR caring for 2 patients with the same viral etiology than that in the AIIR caring for a single patient. This finding suggests that the burden of viral load among symptomatic infected cases was associated with the air dispersal of respiratory viruses.
The finding of air dispersal of respiratory viruses may have implications in infection prevention. Given the mean viral load in air samples of 1.58×103 copies/mL among the singly isolated patients, the total number of viral copies was 3.16×103 over a collection time of 6 hours because the collection substrate was immersed in 2 mL VTM. Assuming that the rate of air dispersal of respiratory viruses is static, 9 copies of viral genome were dispersed in the air per minute, which is comparable with the amount of air dispersal of SARS-CoV-2 RNA using the same air sampler in the same setting of AIIR. 17 The infectious dose of respiratory viruses demonstrated in human volunteer studies by aerosol exposure varied from 0.68 median tissue culture infectious dose (TCID50) for rhinovirus, to 0.5 TCID50 for adenovirus, to 30–40 TCID50 for RSV. 29 Using the correlation of 1 TCID50 to 103 copies/mL, 30 we estimated the infectious doses of rhinovirus (6.8×102 copies/mL), adenovirus (5.0×102 copies/mL), and RSV (3.0×104 copies/mL). Considering the air sampling collection for 6 hours in AIIR, we translated the infectious dose in the room air in the AIIR to the number of viral copies for rhinovirus (1.36×103 copies), adenovirus (1.0×103 copies), and RSV (6.0×104 copies). Based on these findings, the maximum viral copies detected in 72,000 L of air in 6 hours in the AIIRs singly isolated for RSV was 1.24×103 copies, which may be lower than the infectious dose. However, our study was conducted in the AIIRs with 12 air changes per hour, which may have facilitated the dilution of infectious virus-laden particle in the air. Presumably, the viral copies in the air may be double in the general ward setting, which has 6 air changes per hour. Thus, outbreaks of respiratory viruses would be very common in general ward during the winter season when respiratory viruses are highly prevalent in patients with mild or no symptoms. Further investigation is needed to understand the degree of air dispersal of viral genome in the general ward setting with air ventilation of 6 air changes per hour, as well as in the community setting with poor indoor air dilution, which is also a risk factor for SARS-CoV-2 transmission. 31,32 Although the clinical significance of airborne transmission of respiratory viruses other than SARS-CoV-2 remains to be determined, the enforcement of infection control practice in the hospitals, including hand hygiene and universal masking, has successfully prevented nosocomial transmission of respiratory viruses and SARS-CoV-2 before the emergence of the omicron BA.2 variant. 33,34
This study had several limitations. We did not perform viral culture of the air samples. The demonstration of viral DNA or RNA may not correlate with the presence or level of viable virus. These sophisticated experiments have been performed in the investigation of airborne transmission of RSV. 6 We could not include influenza A virus in this study because universal masking and enhancement of hand hygiene practice likely minimized the influenza activity in both community and hospital settings since the outbreak of COVID-19. 34,35 The relative location of patients to the air sampler may have varied over the sampling time; some of our patients were pediatric cases who may have moved around in the bed. The time lag from the collection of NPA to air samples may affect the correlation of viral loads between the clinical and air samples. In addition, our study was not adequately powered to measure all factors associated with air dispersal of respiratory viruses. However, given the small sample size, our findings clearly demonstrate that the viral load of the patient is an important factor. Further study to investigate the phenomenon of air dispersal of respiratory viruses is warranted.
Acknowledgment
We thank the frontline staff of pediatric ward of Queen Mary Hospital for facilitating this study.
Financial support
This study was supported by the Health and Medical Research Fund (HMRF) Commissioned Research on Control of Infectious Disease (Phase IV), CID-HKU1-16, Food and Health Bureau, Hong Kong SAR Government.
Conflict of interest
All authors report no conflicts of interest relevant to this article.
References
- 1. Guideline for isolation precautions in hospitals. Part II. Recommendations for isolation precautions in hospitals. Hospital Infection Control Practices Advisory Committee. Am J Infect Control 1996;24:32–52. [PubMed] [Google Scholar]
- 2. Siegel JD, Emily Rhinehart E, Jackson M, et al. 2007. Guideline for isolation precautions: preventing transmission of infectious agents in healthcare settings. Centers for Disease Control and Prevention website. https://www.cdc.gov/infectioncontrol/pdf/guidelines/isolation-guidelines-H.pdf. Updated July 2019. Accessed April 15, 2022.
- 3. Moser MR, Bender TR, Margolis HS, Noble GR, Kendal AP, Ritter DG. An outbreak of influenza aboard a commercial airliner. Am J Epidemiol 1979;110:1–6. [DOI] [PubMed] [Google Scholar]
- 4. Cowling BJ, Ip DK, Fang VJ, et al. Aerosol transmission is an important mode of influenza A virus spread. Nat Commun 2013;4:1935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Dick EC, Jennings LC, Mink KA, Wartgow CD, Inhorn SL. Aerosol transmission of rhinovirus colds. J Infect Dis 1987;156:442–448. [DOI] [PubMed] [Google Scholar]
- 6. Kulkarni H, Smith CM, Lee Ddo H, Hirst RA, Easton AJ, O’Callaghan C. Evidence of respiratory syncytial virus spread by aerosol. time to revisit infection control strategies? Am J Respir Crit Care Med 2016;194:308–316. [DOI] [PubMed] [Google Scholar]
- 7. Yu IT, Li Y, Wong TW, et al. Evidence of airborne transmission of the severe acute respiratory syndrome virus. N Engl J Med 2004;350:1731–1739. [DOI] [PubMed] [Google Scholar]
- 8. Booth TF, Kournikakis B, Bastien N, et al. Detection of airborne severe acute respiratory syndrome (SARS) coronavirus and environmental contamination in SARS outbreak units. J Infect Dis 2005;191:1472–1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Birgand G, Peiffer-Smadja N, Fournier S, Kerneis S, Lescure FX, Lucet JC. Assessment of air contamination by SARS-CoV-2 in hospital settings. JAMA Netw Open 2020;3:e2033232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Cheng VC, Fung KS, Siu GK, et al. Nosocomial outbreak of coronavirus disease 2019 by possible airborne transmission leading to a superspreading event. Clin Infect Dis 2021;73:e1356–e1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Li Y, Qian H, Hang J, et al. Probable airborne transmission of SARS-CoV-2 in a poorly ventilated restaurant. Build Environ 2021;196:107788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Cheng P, Luo K, Xiao S, et al. Predominant airborne transmission and insignificant fomite transmission of SARS-CoV-2 in a two-bus COVID-19 outbreak originating from the same presymptomatic index case. J Hazard Mater 2022;425:128051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Scientific brief: SARS-CoV-2 transmission. Centers for Disease Control and Prevention website. https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/sars-cov-2-transmission.html. Updated May 7, 2021. Accessed April 10, 2022. [PubMed]
- 14.Coronavirus disease (COVID-19): How is it transmitted? World Health Organization website. https://www.who.int/news-room/questions-and-answers/item/coronavirus-disease-covid-19-how-is-it-transmitted. Published December 23, 2021. Accessed April 10, 2022.
- 15. Cheng VCC, Wong SC, Chen JHK, et al. Escalating infection control response to the rapidly evolving epidemiology of the coronavirus disease 2019 (COVID-19) due to SARS-CoV-2 in Hong Kong. Infect Control Hosp Epidemiol 2020;41:493–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cheng VC, Wong SC, Chan VW, et al. Air and environmental sampling for SARS-CoV-2 around hospitalized patients with coronavirus disease 2019 (COVID-19). Infect Control Hosp Epidemiol 2020;41:1258–1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Wong SC, Yuen LL, Chan VW, et al. Airborne transmission of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): What is the implication of hospital infection control? Infect Control Hosp Epidemiol 2021. doi: 10.1017/ice.2021.318. [DOI] [PMC free article] [PubMed]
- 18. Wong SC, Leung M, Tong DW, et al. Infection control challenges in setting up community isolation and treatment facilities for patients with coronavirus disease 2019 (COVID-19): implementation of directly observed environmental disinfection. Infect Control Hosp Epidemiol 2021;42:1037–1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.BIOFIRE FILMARRAY respiratory panel. bioMérieux website. https://www.biomerieux-diagnostics.com/filmarrayr-respiratory-panel. Accessed June 4, 2022.
- 20. To KK, Lu L, Yip CC, et al. Additional molecular testing of saliva specimens improves the detection of respiratory viruses. Emerg Microbes Infect 2017;6:e49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Wong SSY, Yip CCY, Sridhar S, et al. Comparative evaluation of a laboratory-developed real-time PCR assay and RealStar Adenovirus PCR Kit for quantitative detection of human adenovirus. Virol J 2018;15:149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Dupouey J, Ninove L, Ferrier V, et al. Molecular detection of human rhinoviruses in respiratory samples: a comparison of Taqman probe-, SYBR green I- and BOXTO-based real-time PCR assays. Virol J 2014;11:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Fabian P, Brain J, Houseman EA, Gern J, Milton DK. Origin of exhaled breath particles from healthy and human rhinovirus-infected subjects. J Aerosol Med Pulm Drug Deliv 2011;24:137–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Gralton J, Tovey ER, McLaws ML, Rawlinson WD. Respiratory virus RNA is detectable in airborne and droplet particles. J Med Virol 2013;85:2151–2159. [DOI] [PubMed] [Google Scholar]
- 25. Yan J, Grantham M, Pantelic J, et al. EMIT Consortium. Infectious virus in exhaled breath of symptomatic seasonal influenza cases from a college community. Proc Natl Acad Sci U S A 2018;115:1081–1086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Leung NHL, Chu DKW, Shiu EYC, et al. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med 2020;26:676–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Tseng CC, Chang LY, Li CS. Detection of airborne viruses in a pediatrics department measured using real-time qPCR coupled to an air-sampling filter method. J Environ Health 2010;73:22–28. [PubMed] [Google Scholar]
- 28. Lindsley WG, Blachere FM, Davis KA, et al. Distribution of airborne influenza virus and respiratory syncytial virus in an urgent care medical clinic. Clin Infect Dis 2010;50:693–698. [DOI] [PubMed] [Google Scholar]
- 29. Karimzadeh S, Bhopal R, Nguyen Tien H. Review of infective dose, routes of transmission and outcome of COVID-19 caused by the SARS-COV-2: comparison with other respiratory viruses. Epidemiol Infect 2021;149:e96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lam AH, Leung KY, Zhang R, et al. Evaluation of an antigen detection rapid diagnostic test for detection of SARS-CoV-2 in clinical samples. COVID 2021;1:775–783. [Google Scholar]
- 31. Wong SC, Chen H, Lung DC, Ho PL, Yuen KY, Cheng VC. To prevent SARS-CoV-2 transmission in designated quarantine hotel for travelers: Is the ventilation system a concern? Indoor Air 2021;31:1295–1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Wong SC, Au AK, Chen H, et al. Transmission of omicron (B.1.1.529) SARS-CoV-2 variant of concern in a designated quarantine hotel for travelers: a challenge of elimination strategy of COVID-19. Lancet Reg Health West Pac 2022;18:100360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wong SC, AuYeung CH, Lam GK, et al. Is it possible to achieve 100 percent hand hygiene compliance during the coronavirus disease 2019 (COVID-19) pandemic? J Hosp Infect 2020;105:779–781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Wong SC, Lam GK, AuYeung CH, et al. Absence of nosocomial influenza and respiratory syncytial virus infection in the coronavirus disease 2019 (COVID-19) era: implication of universal masking in hospitals. Infect Control Hosp Epidemiol 2021;42:218–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Cheng VC, Wong SC, Chuang VW, et al. The role of community-wide wearing of face mask for control of coronavirus disease 2019 (COVID-19) epidemic due to SARS-CoV-2. J Infect 2020;81:107–114. [DOI] [PMC free article] [PubMed] [Google Scholar]