Abstract
While feeding, foragers can alter their environment. Such alteration constitutes ecological niche construction (ENC) if it enables future benefits for the constructor and conspecific individuals. The environmental modification may also affect non‐constructing, bystander species, especially if they share resources with constructor species. If so, ENC could confer the constructor species a competitive advantage by both enhancing its foraging returns and reducing those of bystander species.
Expectations – (E1) ENC frequency should vary positively with the recent and current density of the constructor species, and (E2) constructors should use modifications disproportionately. In contrast, bystanders should (E3) experience intensified competition for the affected resource, and (E4) exhibit diverse, possibly mitigating, responses to ENC, depending on opportunity and relative benefits.
We investigated these expectations in Argentina for competition for Fuchsia magellanica nectar between an invasive bumble bee Bombus terrestris (terr: putative constructor), which often bites holes at the bases of floral tubes to rob nectar, and native B. dahlbomii (dahl: bystander), which normally accesses Fuchsia nectar through the flower mouth (front visits). Robbing holes constitute ENC, as they persist until the 7‐day flowers wilt. The dynamics of the incidence of robbed flowers, abundance of both bees and the number and types of their flower visits (front or robbing) were characterised by alternate‐day surveys of plants during 2.5 months.
After initially accessing Fuchsia nectar via front visits, terr switched to robbing and its abundance on Fuchsia increased 20‐fold within 10 days (E2). Correspondingly, the incidence of robbed flowers varied positively with recent and past terr abundance (E1). In contrast, dahl abundance remained low and varied negatively with the incidence of robbed flowers (E3). When terr ceased visiting Fuchsia, dahl abundance increased sixfold within 10 days (E3), possibly because many dahl previously had avoided competition with terr by feeding on other plant species (E4). While terr was present, dahl on Fuchsia used front visits (tolerance) or used existing robbing holes (adoption: E4). The diverse dahl responses suggest partial compensation for competition with terr.
ENC alters competitive asymmetry, favouring constructor species. However, bystander responses can partially offset this advantage, perhaps facilitating coexistence.
Keywords: bumble bee, ecological niche construction, exploitation competition, invasive species, nectar robbing, southern South America
Nectar robbing by flower‐biting bumble bees constitutes ecological niche construction if robbing holes persist and intensify nectar competition experienced by non‐biting, bystander flower visitors. In response, bystanders can tolerate robbing, switch plant species or adopt robbing, perhaps partially compensating for exploitative competition associated with ecological niche construction.

Resumen
Los animales pueden alterar su ambiente como consecuencia del forrajeo. Esta alteración constituye una forma de construcción de nicho ecológico (CNE) cuando la misma promueve beneficios para el constructor y otros individuos de la misma especie. Esta modificación ambiental puede también afectar otras especies no involucradas directamente en la CNE, especialmente cuando comparten recursos con la especie constructora. En este caso, la CNE puede conferir una ventaja competitiva a la especie constructora, incrementado sus beneficios alimenticios en detrimento de los de las especies no constructoras.
Expectativas – (E1) La frecuencia de eventos de CNE debería variar positivamente con la densidad presente y pasada de la especie constructora, y (E2) los individuos de la especie constructora deberían usar las modificaciones asociadas a la CNE en forma diferencial. Por el contrario, los individuos de las otras especies que usen el mismo recurso deberían (E3) experimentar una intensificación de la competencia, y (E4) exhibir una diversidad de respuestas, algunas que atenúen las consecuencias negativas de la CNE, lo que va a depender de la oportunidad y de los beneficios relativos.
En Argentina, investigamos el cumplimiento de estas expectativas en la competencia por el néctar de Fuchsia magellanica entre el abejorro invasor, Bombus terrestris (terr: la especie constructora), quien frecuentemente agujerea la base de tubos florales para robar néctar, y el abejorro nativo B. dahlbomii (dahl: la especie afectada), quien normalmente accede al néctar de Fuchsia en forma legítima a través de la apertura de la flor (visitas frontales). Los agujeros realizados por terr constituyen un caso de CNE, ya que persisten a lo largo de la vida de la vida de la flor (aprox. 7‐day). En este trabajo relevamos la incidencia de flores robadas, la abundancia de las dos especies de abejas, y el número y tipo de sus visitas florales (frontales o de robo) día por medio durante 2.5 meses.
Inicialmente terr accedió las flores de Fuchsia frontalmente, pero más tarde cambió su comportamiento convirtiéndose en un robador casi exclusivo a lo largo de un período de 10 días en que su abundancia se incrementó 20 veces (E2). En consecuencia, la incidencia de flores robadas varió positivamente con la abundancia de terr presente y pasada (E1). Por el contrario, la abundancia de dahl fue baja y varió negativamente con la incidencia de flores robadas (E3). En particular, la abundancia de dahl se incrementó seis veces en un período de 10 días una vez que terr cesó de visitar las flores de Fuchsia (E3), posiblemente porque dahl evitó la competencia con terr forrajeando en otras especies de plantas (E4). Cuando terr estuvo presente, dahl visitó las flores de Fuchsia frontalmente (tolerancia) o usó los agujeros existentes (adopción de comportamiento robador: E4). Estas respuestas diversas de dahl sugieren una compensación parcial de los efectos negativos de la competencia con terr.
La CNE incrementa la asimetría de la competencia, favoreciendo la especie constructora. Sin embargo, las diferentes respuestas de las especies afectadas pueden compensar parcialmente esta desventaja competitiva, tal vez facilitando la coexistencia.
1. INTRODUCTION
In the process of accessing a resource, consumers can cause lingering changes to their abiotic or biotic environment that enhance future resource consumption by themselves and conspecifics, resulting in ecological niche construction (ENC; Kylafis & Loreau, 2011). ENC can enable ongoing access to a resource that would otherwise be unattainable by the constructor species and/or improve its foraging efficiency. ENC also has indirect benefits if it increases the constructor's competitiveness for shared resources with non‐constructing ‘bystander’ species (Kylafis & Loreau, 2011). Thus, ENC can have diverse ecological consequences for individual performance and population dynamics of constructor and bystander species, and hence for community structure (see Bråthen & Ravolainen, 2015; Isbell & Loreau, 2014; Kylafis & Loreau, 2008, 2011). Depending on the magnitude, consistency and duration of the ecological effects of ENC, it could also impose selection on traits of the affected species, perhaps precipitating a between‐generation evolutionary response known simply as niche construction (Barker & Odling‐Smee, 2014; Matthews et al., 2014; Odling‐Smee et al., 2013). Regardless of whether adaptation occurs, ENC could be ecologically relevant for many organisms.
Explicit consideration of ENC has focused on its population and community consequences (Bråthen & Ravolainen, 2015; Kylafis & Loreau, 2011), rather than the responses by individuals of constructor and bystander species that generate those consequences. Bystander species could be either beneficiaries of the constructor species if ENC improves bystander access to resources, or competitors if ENC hampers bystander consumption of a shared resource. Competitor bystanders could respond behaviourally to ENC in three ways (Figure 1; see Irwin et al., 2010). Two responses are typical reactions to competitive depletion of a shared resource (Wisheu, 1998). In the absence of reasonable alternatives, bystanders experiencing exploitation competition must tolerate the reduced resource availability and its consequences. If instead alternatives exist and offer greater foraging returns than the resource shared with constructor individuals, avoiding competition by a dietary switch (i.e. resource partitioning; Bolnick et al., 2003; Kotler & Brown, 2007) may be more profitable than tolerance. The third option is specific to interaction with a niche constructor, namely for a bystander to adopt the environmental modification opportunistically and switch its mode of foraging on the shared resource. Whether individual bystanders tolerate, avoid or adopt ENC will depend on the relative foraging efficiencies associated with these alternative (see Dedej & Delaplane, 2005; Higginson & Ruxton, 2015; Lichtenberg et al., 2018, 2020). These responses need not be fixed (e.g. Bronstein et al., 2017; Lichtenberg et al., 2020), but instead likely vary with the prevailing frequency of ENC, as determined by the persistence of individual environmental modifications, the past and current densities of the niche constructor and the replenishment of unmodified conditions (see Jones et al., 1997).
FIGURE 1.

General effects of an ecological niche constructor on the environment and potential behavioural responses of bystander organisms
A widespread case of ENC, known as nectar robbing (Irwin et al., 2010), occurs when a short‐tongued flower visitor (primary robber; Inouye, 1980) bites holes at the bases of tubular flowers to ingest nectar that it cannot access readily by probing ‘legitimately’ through flower mouths (e.g. Navarro, 1999; Pyke, 1982; Varma & Sinu, 2019). Primary robbing constitutes ENC (Newman & Thomson, 2005) for several reasons. First, primary robbing can increase the robber's foraging returns compared to probing legitimately (Lichtenberg et al., 2018; Pyke, 1982). Second, by extracting more nectar by robbing than by visiting legitimately, a flower‐biting (constructor) species depletes resources for legitimate (bystander) visitors (González‐Gómez & Valdivia, 2005; Maloof & Inouye, 2000). Third, damaged floral tubes do not heal, so holes persist until the flower wilts. These holes provide continued nectar access for later visiting secondary robbers, either the original primary robbers or adopting bystanders of the same or difference species, that capitalise on the benefits of the holes without creating them (Bronstein et al., 2017; Irwin et al., 2010). Although interactions of nectar robbers and affected plants are well‐studied, competition between robbing and legitimate flower visitors has received limited attention (Irwin et al., 2010).
We considered competition between Bombus dahlbomii Guérin‐Méneville (hereafter dahl), the only native bumble bee in Patagonia, and B. terrestris Linnaeus (hereafter terr), a recent invader, for nectar of Fuchsia magellanica Lam. (Onagraceae; hereafter Fuchsia; Figure S1). Terr is short‐tongued (mean ± SE for workers = 6.3 ± 0.5 mm; Goulson et al., 2008) and a notorious primary and secondary nectar robber of long‐tubed flowers throughout its native and invasive ranges (Goulson, 2010), including those of Fuchsia (Figure S1c,d; Valdivia et al., 2016; Stanley & Cosnett, 2021). Thus, terr is a putative niche constructor. In contrast, dahl is likely a bystander species in this case, as its relatively long proboscis (11.10 ± 0.17 mm; Madjidian et al., 2008) is similar in length to the basal nectar tube of Fuchsia flowers (range = 7–15 mm, Berry, 1989; Figure S1d). Using daily observations during 11 weeks, we characterised the dynamics of resources (Fuchsia flower abundance), environmental modification (robbed flowers) and the densities and behaviours of terr and dahl. These observations allowed assessment of the expectations that:
Environmental modification (robbed flowers) varies positively with past and present densities of the putative constructor species;
The constructor species benefits from its environmental modification by improving its foraging efficiency and disproportionately increases its use of the affected resource;
The bystander species experiences competition with the constructor species for the modified resource; and
The bystander species experiences increased interspecific competition owing to ENC and exhibits diverse, possibly mitigating, responses to the associated environmental modification.
As we observed bees only visiting Fuchsia flowers, tolerance and adoption of the constructor's environmental modification could be detected directly, whereas avoidance could only be inferred.
2. MATERIALS AND METHODS
2.1. Study species and site
Fuchsia is a shrub native to temperate regions of Chile and Argentina along the Andes Mountains (Berry, 1989). It flowers continuously for up to 6 months during austral summer (Dzendoletas et al., 2003). Individual Fuchsia flowers are pendent with a basal nectar tube, flaring red sepals and a distal purple corolla tube formed by separate overlapping petals (Figure S1a). They remain open for a mean ± SD of 7.1 ± 1.6 days (median = 7 days), producing copious nectar (volume ± SD = 0.563 ± 0.301 μl/hr, concentration = 21.8 ± 5.1%; Rosenberger, N.M., unpubl. data). Native Fuchsia pollinators include dahl and the hummingbird Sephanoides sephaniodes Lesson (Traveset et al., 1998; Valdivia et al., 2016). We do not consider S. sephaniodes further, as it represented only 2.8% of flower visitors recorded during 129.5 hr of observation.
This study was conducted at Puerto Blest, Parque Nacional Nahuel Huapi, Río Negro, Argentina (−41.033333, −71.816667), at the end of the western arm of Lago Nahuel Huapi near the Andean continental divide. Puerto Blest is located in Valdivian temperate rainforest dominated by Nothofagus dombeyi (Mirb.) Oerst., which also includes various herbaceous species and hummingbird‐pollinated shrubs (Aizen & Rovere, 2010; Dzendoletas et al., 2003). Other nectar‐producing, native species that flower during the study period at Puerto Blest include Berberis darwinii, Campsidium valdivianum, Dasyphyllum diacanthoides, Desfontainia spinosa, Discaria chacaye, Escallonia rubra, Fragaria chiloensis, Gaultheria mucronata and Luma apiculata (Dzendoletas et al., 2003). The Administración de Parques Nacionales Argentinas permitted this study under project permit 1430, and no ethics approval was required as animal visitors were not handled or captured.
2.2. Plant surveys
This study considered daily records of dahl and terr visiting flowers of 22 Fuchsia plants. We selected these plants haphazardly throughout the study area to represent habitat variation, including four along the beach of Lago Nahuel Huapi, four in N. dombeyi forest, 10 along forest streams and four in an open, disturbed area around the Puerto Blest Hotel. The study plants varied in size, with a maximum of 34–510 flowers open simultaneously. Maximum display size did not differ significantly among habitats (F 3,18 = 1.46, p > 0.25; GLM with negative binomial distribution).
Two observers sampled plants daily from 1 December 2015 to 8 April 2016 between 07:00 hr and 20:00 hr. Dahl and terr were first observed visiting Fuchsia flowers on 9 and 28 December respectively; however, our analyses consider observations from only 20 January to 8 April 2016 when bumble bees visited Fuchsia flowers consistently. Individual plants were surveyed on alternate days, with half the plants observed each day (see Table S1). For analyses of bee abundance and visit behaviour, we excluded surveys for 21 January, 17 March and 7 April, when no bees were observed owing to inclement weather.
While surveying a plant, an observer counted all open flowers and observed visits to some or all of them for 10 min (i.e. total 110 observation min per day). When floral display size exceeded 30 flowers, a subset of observation flowers was selected on one or more branches. For each visitor to observation flowers, the observer recorded its species and the numbers of flowers visited by probing the distal end of the floral tube (front visit) or through a hole in the side of the floral tube (tube visit or robbing). For tube visits, observers also recorded whether the visitor created the hole (primary robbing) or used an existing hole (secondary robbing). Observation of a visitor ceased when it left the observation flowers or the 10‐min survey ended.
Additional flowers were monitored to quantify the proportion of robbed flowers (i.e. extent of ENC). Every 4 days, an observer haphazardly selected five mature flower buds per plant and attached a uniquely numbered jeweller's tag to each flower's pedicle. Every sampling day thereafter until wilting, the nectar tubes of tagged flowers were inspected for the presence of holes caused by primary robbing.
2.3. Data analysis
We primarily report analyses that assessed statistical effects on daily variation of three dependent variables—the per‐plant proportion of robbed tagged flowers, the numbers of bees of both species visiting flowers during 10‐min observations of individual plants (local bee abundance) and the numbers of front or robbing flower visits by those bees (flower visitation). A fourth analysis, described and presented in Appendix S1, considered effects on the proportion of dahl that engaged solely in front or robbing visits. These analyses involved GLMMs (Stroup, 2013), as implemented in the glimmix procedure of SAS/STAT 14.2 (SAS/STAT® 14.2 User's Guide, 2016; robbed flowers and visit types) or the glmmTMB procedure (version 1.0.2.9000; Brooks et al., 2017) of R (version 4.0.2; R Core Team, 2020; bee abundance). Each analysis included bee species (except robbed flowers and dahl visit type) and habitat as fixed factors, as well as relevant continuous covariates (see below and Table S2). General among‐plant variation was assessed by including plant as a random factor. We also accounted for correlated responses arising from repeated measurement of sampling units (robbed flowers—study plant; bee abundance—bee species per plant; flower visits—bee species per observation period). Specifically, the temporal covariance between pairs of observations within sampling units was modelled as a declining exponential function of the intervening interval (Stroup, 2013). Sample date was not additionally included as an independent variable, as the relevant influences involved the daily dynamics of flower abundance and bee abundance and behaviour, not the specific date.
The analysis of daily variation in the proportion of robbed flowers per plant addressed Expectation 1, that the putative constructor species, terr, modified the foraging environments of both bee species (see Table S2). If so, the proportion of robbed flowers should have varied positively with total terr abundance, as measured by the daily sum of terr counts for all study plants. Given the average 7‐day life span of individual flowers, both current and recent total terr abundances are relevant. As a measure of past abundance, we used the average terr count for the preceding two samples of a plant (i.e. 2 and 4 days prior). We also included total current flower number on all study plants as a measure of overall flower abundance. This analysis involved a quasi‐binomial distribution (logit link function) to account for overdispersion in the dependent variable.
The analysis of daily variation in local bee abundance simultaneously assessed numerical responses of both bee species to the current total abundance of each species (i.e. among all study plants), resource availability (open flowers per plant) and environmental modification (proportion of robbed flowers per plant; Table S2). We interpret negative effects of conspecific and heterospecific total abundance as intra‐ and interspecific competition respectively (Expectations 2 and 3). Negative or positive effects of the proportion of robbed flowers similarly indicate competitive or facilitative effects of environmental modification respectively (Expectations 2 and 3). Note that considering the effect of total terr abundance on the proportion of robbed flowers (above) and the effect of the proportion of robbed flowers on local terr abundance does not involve circularity because of the contrasting scope of analysis (total versus local bee abundance) and the fact that almost all robbing bees used existing floral holes that may have been created up to 6 days earlier. The distribution of local abundance of each bee species included more zeroes than expected for a negative binomial distribution. Therefore, we used the glmmTMB procedure to assess simultaneously the (possibly different) influences on: the probability that bees were not available to be detected during an observation period (zero‐inflation component; binary distribution, logit link function); and the number of bees recorded visiting the observation flowers (possibly 0) if they were available on the plant (conditional component; negative binomial distribution, ln link function).
The analysis of daily variation in the numbers of front and tube visits to observation flowers by individual bees during 10‐min observations assessed the effects of environmental modification (proportion of robbed flowers per plant), the current local (per plant) and total (all plants) densities of both bee species, and resource availability (open flowers per plant; Table S2). The use of tube visits should vary positively with the proportion of robbed flowers to the extent that perforated flowers increase a bee's foraging efficiency. This visit type should be especially prevalent for the constructor species, terr, given its relatively short tongue (Expectation 2). For the bystander species, dahl, the use of front versus tube visits indicates whether individuals tolerated or adopted the constructor's environmental modification respectively (Expectation 4). The visitation analysis considered only the 97.5% of the 1,313 observed bees that exclusively used one behaviour or the other, with visit type included as a categorical factor. The remaining 33 bees (5 terr and 27 dahl) used front and tube visits interchangeably to extract nectar from individual plants. This analysis involved a log‐normal distribution and identity link function.
The three analyses incorporated two common features concerning the handling of independent variables. First, all analyses initially included pairwise interactions between independent variables to account for heterogeneous effects. As sampling did not involve a balanced, fully crossed design, collinearity is likely between interactions and their constituent variables, hampering characterisation of statistical effects. Therefore, we used backward elimination to exclude interactions that did not statistically influence (i.e. α > 0.05) variation in the dependent variable (Kutner et al., 2005). Second, as independent variables, bee abundance (+1) and flower counts were ln‐transformed for all analyses. Consequently, the analyses of bee abundance (conditional component only) and visit behaviour considered linear ln–ln relations (power functions) for the effects of these independent variables. In these cases, a partial regression coefficient of β = 1 indicates proportional variation of the dependent variable with an independent variable, β < 1 indicates a decelerating relation and β > 1 indicates an accelerating relation.
To illustrate the relations of dependent variables to independent variables, we present partial effects that account for variation in other independent variables in a final statistical model. For categorical independent variables, we present back‐transformed marginal means (Milliken & Johnson, 1984) for categories represented by >4 observations, unless indicated otherwise. We illustrate the effect of a focal continuous independent variable, Xf , as the associated variation in adjusted values of the dependent variable. For observation i, this value is
where g′ is the inverse link function, β 0 is the intercept, βf and βj are the partial regression coefficients for the focal and jth non‐focal independent variables, is the mean of the jth non‐focal independent variable and ei is the observation's residual.2322
3. RESULTS
3.1. Dynamics of flowering, bee visitation and robbed flowers
The daily surveys revealed differing phenologies for Fuchsia and its dahl and terr visitors (Figure 2). During the last third of January, relatively few Fuchsia flowers were open (Figure 2a) and both bee species visited them relatively infrequently, with dahl being about 60% more abundant than terr (Figure 2b,c). Throughout this period, all Fuchsia flowers were intact (Figure 2d) and bees of both species used only front visits to ingest nectar (Figure 2e,f).
FIGURE 2.

Daily variation of (a) overall flowering by 22 Fuchsia magellanica plants, (b, c) Bombus terrestris and B. dahlbomii abundance (bees per plant per 10‐min observation), (d) the proportion of robbed flowers per plant and (e) the numbers and (f) proportions of nectar‐robbing flower visits by B. terrestris and B. dahlbomii per observation period. Panels b–d depict marginal (±SE) and predicted means (solid line) based on the respective statistical analyses (which did not explicitly include date). Gaps in lines in panels e and f indicate days when no bees of the associated species were observed visiting Fuchsia flowers
On 2 February, terr began robbing Fuchsia flowers and within 3 days it robbed almost exclusively until it ceased visiting Fuchsia (Figure 2f, red line). During the first 10 days of February, mean terr abundance per Fuchsia plant increased almost 20‐fold (Figure 2b), 72 times faster than the concurrent 27% increase in Fuchsia flowering (Figure 2a). In contrast, after a slight peak on 2 February, dahl abundance per plant declined during the first 10 days of February (Figure 2c). Both terr abundance and the proportion of robbed flowers peaked on February 10 (Figure 2b,d), one month before peak Fuchsia flowering (Figure 2a). Terr abundance then generally declined until late March, after which terr was not observed visiting Fuchsia (Figure 2b). The proportion of robbed flowers declined from 0.8 to 0.2 from 10 February to 29 February. It then rose again to an average of about 0.5 in mid‐March before declining (Figure 2d), roughly paralleling the phenology of flowering (Figure 2a). On the final sampling day (8 April), no observation flowers had been robbed, despite moderate dahl abundance (Figure 2c). The lagged persistence of robbed flowers until 8 days after terr ceased visiting is consistent with the average longevity of Fuchsia flowers.
Throughout February and the first 3 weeks of March, dahl abundance per plant remained relatively constant, compared terr abundance (Figure 2b,c). Dahl was first observed using robbing holes 5 days after terr, but this behaviour by dahl was not observed daily for another 6 days (Figure 2e,f), after the availability of robbed flowers peaked (Figure 2d). Dahl mostly used front visits to access Fuchsia nectar, except during the first 2 weeks of March (Figure 2f). During the last 10 days of March, when Fuchsia flowering declined and terr was rarely observed visiting Fuchsia, dahl abundance increased sixfold and more dahl than terr were observed using robbed flowers (Figure 2e, compare blue and red lines). During April, dahl abundance declined with flower availability (Figure 2a,c).
3.2. Incidence of robbed flowers
Consistent with terr's reputation as a primary nectar robber and Expectation 1, its abundance had diverse, interacting effects on the daily proportion of robbed flowers (Table 1; Table S3; Figure 3). Of particular relevance, the incidence of robbed flowers varied positively and synergistically with recent and current total terr abundance (Figure 3a). In addition, the effects of recent and current terr abundance each varied with total flower availability, being strongest when relatively few flowers were open (Figure 3b,c) as robbing began during early February (see Figure 2a). In contrast, during peak flowering, the proportion of robbed flowers varied largely independently of recent and current terr abundance (Figure 3b,c). The incidence of robbed flowers did not differ statistically among habitats, but it varied somewhat among plants within habitats (Table 1). Unsurprisingly, given the longevity of individual flowers, the proportion of robbed flowers correlated strongly between sampling days (Table 1; e.g. r ≈ 0.75 between successive samples).
TABLE 1.
Overall results of GLMMs evaluating effects on the proportion of robbed flowers, the local abundance of each bumble bee species and flower visits by individual bees. The analysis of local bee abundance involved a zero‐inflated negative binomial distribution, allowing joint assessment of effects on bee presence on sample plants and the number that visited observation flowers. Local bee abundance, open flowers and observed flowers refer to counts on individual plants during individual surveys, whereas total abundance and open flowers refer to the sum over all observation plants per sampling day
| Effect | Dependent variable | |||
|---|---|---|---|---|
| Robbed flowers a | Local bee abundance | Flower visits d | ||
| Presence b | Number observed c | |||
| Species | = 1.32 | = 54.90*** | F 1,1238 = 4.52* | |
| Habitat | F 3,17.2 = 1.48 | = 3.74 | = 19.27*** | F 3,34.08 = 1.16 |
| Species × Habitat | = 16.30*** | F 1,1205 = 1.90 | ||
| ln(Total dahl abundance + 1) | = 0.90 | = 25.37*** | F 1,579.9 = 25.06*** | |
| Species × ln(Total dahl abundance + 1) | = 4.22* | = 47.74*** | ||
| ln(Local dahl abundance + 1) | ||||
| ln(Total terr abundance + 1) | F 1,600.5 = 16.30*** | = 114.1*** | F 1,556.8 = 6.10* | |
| Species × ln(Total terr abundance + 1) | = 152.6*** | |||
| ln(Past total terr abundance + 1) | F 1,559.0 = 13.71*** | |||
|
ln(Total terr abundance + 1) × ln(Past total terr abundance + 1) |
F 1,636.9 = 6.89*** | |||
| Proportion robbed flowers | = 8.92** | = 1.61 | F 1,573.6 = 10.35** | |
| Species × Prop. robbed flowers | = 8.35** | = 4.15* | ||
| Visit type | F 1,1142 = 1.02 | |||
| Species × Visit type | F 1,1209 = 3.95* | |||
| Habitat × Visit type | F 3,1170 = 2.29 | |||
| Species × Habitat × Visit type | F 3,1218 = 3.35* | |||
| Visit type × Prop. robbed flowers | F 1,958.5 = 11.96*** | |||
| ln(Total open flowers) | F 1,524.3 = 28.86*** | |||
| ln(Open flowers per plant) | = 5.39* | = 19.68*** | ||
| ln(Observed flowers) | F 1,579.9 = 25.06*** | |||
|
ln(Total terr abundance + 1) × ln(Total open flowers) |
F 1,590.9 = 18.17*** | |||
|
ln(Past total terr abundance + 1) × ln(Total open flowers) |
F 1,568.5 = 13.41*** | |||
| Among‐plant variance | = 4.65* | = 34.5*** | = 21.69*** | |
| Temporal autocorrelation | = 338.2*** | = 11.3** | = 17.89*** | |
Overdispersed binomial distribution, logit link function.
Binary distribution, logit link function.
Negative binomial distribution, ln link function.
Log‐normal distribution, identity link function.
p < 0.05; **p < 0.01; ***p < 0.001.
FIGURE 3.

Partial effects on the daily incidence of nectar‐robbed Fuchsia magellanica flowers, including pairwise interactions between the daily past and current total abundances of Bombus terrestris and the total number of open flowers on all study plants. See Tables 1 and S3 for statistical details
3.3. Local bee abundance
Two processes determined the local (per plant) abundance of bees on individual plants during observation periods, as indicated by the improved fit of an analysis that accounted for zero inflation (∆AIC = 32.1). The first process governed the presence of bees of a given species on a plant during an observation period, which varied with four detected influences (Table 1; Table S4). Bee presence differed consistently among plants, but not among habitats (Table 1). Unsurprisingly, bees were least likely to be present when plants displayed few flowers (Figure S2a; Table S4), which primarily occurred early during the study. Bee presence also varied with overall dahl abundance and the proportion of robbed flowers per plant, with contrasting relations for the two bee species (Table S4). Specifically, the presence of dahl per plant varied positively with total dahl abundance, but independently of the proportion of robbed flowers, whereas terr presence varied independently of dahl abundance, but positively with the availability of robbed flowers (Figure 4a; Figure S2b). The latter association is consistent with Expectation 2. Given these effects, the per‐plant presence of both species did not vary statistically with overall terr abundance; however, indirect effects are likely, given the influence of total terr abundance on the proportion of robbed flowers (Figure 3).
FIGURE 4.

Partial effects on the marginal mean (±SE) numbers of Bombus dahlbomii and B. terrestris visiting Fuchsia magellanica plants during 10‐min surveys, including: (a) the relation of bee presence on study plants to the proportion of robbed flowers; and the relations of observed bee numbers when bees were present to (b) the number of open flowers per plant, (c, d) the total numbers of B. terrestris and B. dahlbomii observed daily on all study plants, (e) the daily proportion of robbed flowers per plant and (f) habitat. See Tables 1 and S4 for statistical details and Figure S2 for additional effects on bee presence
The second process affecting local bee abundance determined the number of bees seen visiting observation flowers if they were present on a plant (Table 1). The observed bee number correlated strongly between sampling days for each species (Table 1; e.g. r ≈ 0.92 between successive samples). In addition, more bees visited observation flowers on plants with larger displays (Figure 4b; Table S4). Similarly, local bee abundance increased in proportion to total conspecific abundance for both species (terr – Figure 4c, red line; test of β = 1, t 1525 = 1.35, p > 0.15; dahl – Figure 4d, blue line; t 1525 = 1.52, p > 0.1). In contrast, the local abundance of both species varied independently of the total abundance of the other species (Figure 4c,d; Table S4). The proportion of robbed flowers on a plant did not affect local terr abundance statistically, whereas fewer dahl were observed on plants with high proportions of robbed flowers (Figure 4e; Table S4), signalling a competitive effect (Expectation 3). In addition to the preceding overall effects, local abundance of the two bee species varied heterogeneously among habitats (Table 1), with more dahl than terr observed visiting Fuchsia in disturbed and stream sites, but not in beach or forest sites (Figure 4f). Together these influences on bee presence and conditional abundance accounted for most daily variation in local bee abundance (compare means and solid lines in Figure 2b,c).
3.4. Flower visits
Of the 4730 flower visits observed during flower sampling, 53.7% involved robbing. Among the 2,542 robbing visits, only 1.3% involved primary robbing, including 28 visits by 20 terr and four by two dahl. Primary robbing by dahl was observed late during the sampling period (28 and 29 March), after terr ceased visiting Fuchsia flowers. Whereas 96.7% of the 519 terr observed visited only robbed flowers (Figure 2f), 31.8% of the 695 dahl used only front visits, 64.2% only robbed and 4.0% used both visit types, indicating both tolerance and opportunistic adoption of robbed flowers (Expectation 4). The average proportion of robbing dahl largely equalled the proportion of robbed flowers (Figure S3c), especially on plants with few terr (Figure S3d; see Appendix S1 and Table S5).
The number of flowers visited per bee during individual survey periods was subject to diverse influences (Table 1; Table S6, Figure 5). Unsurprisingly, the recorded flower visits per bee varied positively with the number of flowers observed (Figure 5a). Overall, bees generally visited more flowers when terr was abundant (Figure 5b) and on plants with few dahl during a survey (Figure 5c). Front‐visiting bees probed fewer observation flowers on plants with a high proportion of robbed flowers, whereas this proportion did not influence the number of flowers visited by robbing bees (Figure 5d). On average, dahl individuals generally visited equivalent numbers of observation flowers whether using front or tube visits, except on beach plants where they probed more flowers while front visiting (Figure 5e). In contrast, terr generally probed fewer flowers while front visiting than while robbing, especially on plants at the beach and disturbed sites (Figure 5f). The number of flowers visited also varied statistically among plants within habitats and correlated positively among bees within observation periods (Table 1).
FIGURE 5.
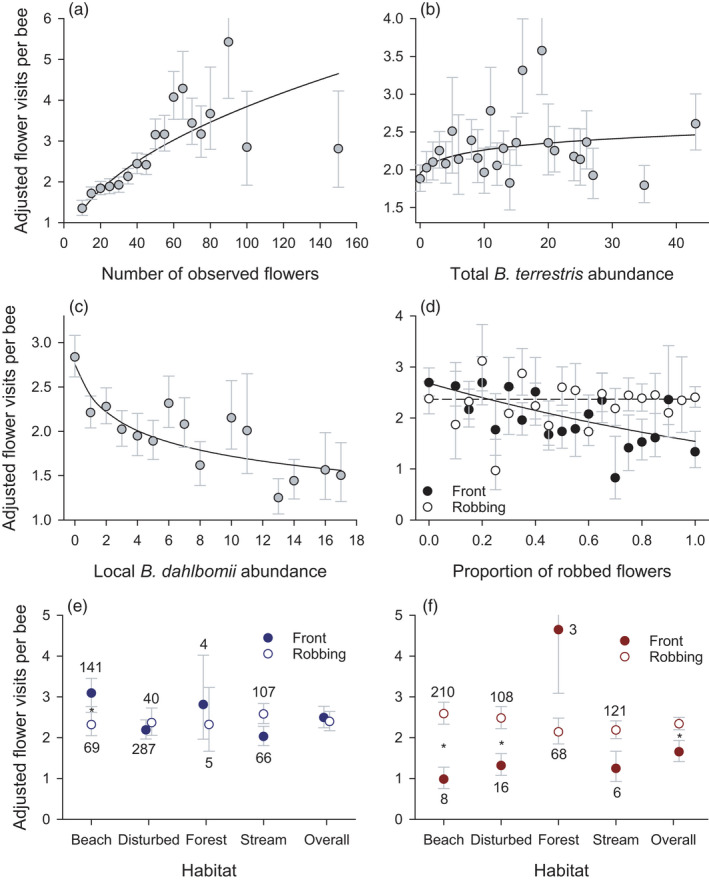
Partial effects of independent variables on marginal mean (±SE) visitation of Fuchsia magellanica flowers by Bombus dahlbomii and B. terrestris during 10‐min surveys, including: (a) the number of observed flowers per plant; (b) the daily total number of B. terrestris observed on all survey plants; (c) the number of B. dahlbomii observed per‐plant survey; (d) interacting effects of visit type and the proportion of robbed flowers per plant; and (e, f) the interacting effects of visit type and habitat for B. dahlbomii and B. terrestris respectively. For statistical details, see Tables 1 and S6
4. DISCUSSION
4.1. Species roles and ecological niche construction
Terr and dahl played contrasting roles as Fuchsia visitors, which affected their competition for nectar. Several lines of evidence identify terr as the ecological niche constructor and dahl as a bystander. First, although primary robbing was observed rarely, most primary robbers were terr and the observations of two dahl biting flowers occurred late during the study. Second, terr switched to robbing first and was responsible for almost all robbing visits observed as the proportion of robbed flowers peaked during the following two weeks (Figure 2d,e). Finally, the occurrence of robbed flowers varied with the past and current abundance of terr (Figure 3; Expectation 1) and the creation of robbed flowers essentially ended when terr ceased visiting Fuchsia. After that, dahl rarely used robbed flowers (Figure 2e, blue line), despite visiting Fuchsia much more frequently than when terr was present (Figure 2c).
The observed dynamics of terr and dahl abundance and behaviour demonstrate that nectar robbing constitutes ENC (see Kylafis & Loreau, 2011; Newman & Thomson, 2005). As summarised in Figure 1, the environmental modification (nectar‐access holes in Fuchsia flowers) created by the biting constructor species, terr, persisted and increased its resource availability and competitiveness, motivating behavioural responses by the bystander species, dahl. Although the constructor species did not initially rob Fuchsia flowers, once robbing began, it used this foraging mode almost exclusively (Figure 2f, red line). By doing so, individuals of the constructor species benefitted from increased visitation of observation flowers (Figure 2f), likely enhancing their nectar intake per flower (Expectation 2). Furthermore, the inferred presence of constructors on Fuchsia varied positively with the frequency of its existing environmental modifications (Figure 4a, red symbols and line). ENC also enhanced the constructor's competitive advantage (Expectation 3), as demonstrated by the negative relation of the number of bystanders (dahl) visiting Fuchsia to existing environmental modifications (Figure 4e, blue symbols and line). This competition prompted diverse responses by the bystander species (Expectation 4). Dahl initially tolerated niche construction, continuing to probe the mouths of Fuchsia flowers to ingest nectar (Figure 2f, blue line). Only after ENC peaked during mid‐February (Figure 2d) did an increasing proportion of dahl individuals adopt the environmental alteration (Figure 2f). By doing so, they may have partially compensated for competition with the constructor species, as robbing bees visited more flowers per observation than front‐visiting individuals when most flowers had been robbed (Figure 5d). In addition, many unobserved individuals of the bystander species may have avoided competition for Fuchsia nectar during peak terr abundance by visiting other plant species (see below). We now consider insights revealed by the terr–dahl–Fuchsia interaction concerning the role of ENC in competition between constructor and bystander species.
4.2. Ecological niche construction and competition asymmetry
The extent to which a constructor's environmental modification improves its own resource access and foraging efficiency should also intensify its exploitation competition with other species vying for the same resource (Expectations 2 and 3). Thus, ENC should make interspecific competition more asymmetrical, favouring the constructor species. This expectation is evident in the apparent competitive release of terr and dahl as ENC began and ended respectively. Competitive release is evident when relaxation of interspecific competition increases a species’ morphological or behavioural variation and/or its density or habitat use (Terborgh & Faaborg, 1973; Yoder et al., 2010; for Bombus examples, see Inouye, 1978; Laverty & Plowright, 1985).
The first relevant event occurred when terr began ENC and abruptly switched from front to robbing visits in early February (Figure 2f). Initially (late January), terr used only front visits to access Fuchsia nectar. In doing so, it was probably at a competitive disadvantage to dahl, which has a longer proboscis and so could access more of the nectar column in Fuchsia flowers (see Newman & Thomson, 2005). The resulting limitation of accessible nectar likely prompted some terr individuals to begin robbing, allowing them to ingest more nectar per flower, alleviating their competitive disadvantage (see Dedej & Delaplane, 2005; Lichtenberg et al., 2018; Pyke, 1982). Correspondingly, terr abundance as a Fuchsia visitor increased 20‐fold in just 10 days (Figure 2b), indicating extreme competitive release, perhaps aided by social transmission of nectar‐robbing behaviour (see Leadbeater & Chittka, 2008). This increase was roughly 13 times faster than the maximal growth of terr colonies (see Duchateau & Velthuis, 1988), so it must have predominantly involved recruitment of individuals that previously fed on other plant species. Simultaneously, use of Fuchsia by dahl declined, so terr became the overwhelmingly dominant consumer of Fuchsia nectar (compare Figure 2b,c). Thus, through their environmental modification, primary‐robbing terr individuals increased their own foraging efficiency, created foraging opportunities for secondary‐robbing conspecifics (Figure 4a, red symbols and line) that had previously rejected Fuchsia as a profitable resource, and reduced the suitability of Fuchsia for dahl (Figure 4e, blue symbols and line).
The second case of apparent competitive release involved dahl. During February and March, dahl abundance should generally increase in the absence of terr, owing to colony growth. For example, during February 1994 and 1995, before terr invaded western Argentina, Aizen (2001) observed 5‐ and 3.5‐fold increases in dahl abundance on Alstroemeria aurea in a drier forest 52 km east of Puerto Blest. In contrast, during our study, dahl abundance on Fuchsia remained consistently low throughout February and the first 3 weeks of March. However, this changed during the 10 days after 20 March, when terr largely ceased visiting Fuchsia (Figure 2c), probably owing to the end of its colony cycle, which typically lasts about 10 weeks (Gosterit, 2011). In response, dahl abundance increased sixfold, and it became more abundant than previously during the study. Like the earlier spike in terr abundance, the increase in observed dahl abundance occurred too rapidly to have resulted solely from colony growth. Rather, it likely involved recruitment of individuals that had previously avoided reduced resource availability associated with competition with a constructor and were instead visiting other plant species.
These results illustrate contrasting effects of ENC in these competitive‐release episodes: the constructor species’ environmental modification triggered its own release, whereas the absence of the constructor and its modifications promoted the release of the bystander species. Despite the observational nature of this study, the synchrony of these numerical events with the dynamics of the constructor's environmental modification strongly imply that ENC asymmetrically altered competition between constructor and bystander species to the advantage of the constructor.
4.3. Bystander responses
This study illustrates that ENC motivates a suite of behavioural responses by bystander species as they adjust to the associated environmental modification (Figure 1; Expectation 4). Specifically, direct evidence demonstrates that some dahl tolerated robbed flowers, continuing front visits of Fuchsia flowers, whereas others adopted the environmental modification and switched to secondary robbing (Figure 2e,f; also see, Lichtenberg et al., 2020; Newman & Thomson, 2005). That 96% of observed dahl used either front or robbing visits, rather than both, after robbed flowers became available, but the proportion of dahl using robbing visits varied extensively during February and March (Figure 2d) suggests that both foraging modes were similarly rewarding (Bronstein et al., 2017). In addition, the spike in dahl observed visiting Fuchsia after terr ceased visiting (Figure 2c) suggests that many dahl had previously avoided the environmental modification by visiting other plant species.
Apparent co‐occurrence of the three foraging responses to environmental modification suggests that dahl individuals assorted themselves among tolerance (front visits of Fuchsia), adoption (tube visits of Fuchsia) and avoidance (visits to species other than Fuchsia) in proportions that rendered all options equally rewarding, as in an ideal‐free distribution (Dreisig, 1995; Fretwell & Lucas, 1970; Hamilton, 2010; Pleasants, 1981). Two observations support this conclusion. First, in contrast to the universal persistent switch from front visits to robbing by terr, the proportion of dahl that robbed Fuchsia generally increased and then declined during the study in concert with variation in factors that could affect resource dynamics (Appendix S1, Figure S3). Importantly, the average proportion of robbing dahl generally equalled the opportunity for secondary robbing, as governed by the proportion of robbed flowers (Figure S3c), in accordance with the input‐matching rule of the ideal‐free distribution (Houston, 2008). Second, the inferred rapid shift of dahl to Fuchsia when competition relaxed indicates awareness of the change in resource status, probably gained by sampling (Dall & Cuthill, 1997).
According to this interpretation, the various behavioural responses adopted by the bystander species partially compensated for the negative impact of competition with a niche constructor compared to its effect if adoption and avoidance had not been possible. Such compensation is possible because, like most bumble bees (Goulson, 2010), dahl is a behaviourally flexible ecological generalist (Abrahamovich et al., 2001; Aizen et al., 2002). In contrast, for bystanders with a limited behavioural repertoire, adoption may not be a feasible response to competition with a niche constructor. Similarly, bystanders with limited ecological range or those in environments with few suitable alternative resources may be unable to avoid constructor competition.
4.4. Population and evolutionary implications
Foraging‐based ENC can invoke diverse consequences for populations of constructor and bystander consumers and the affected food species that are the object of construction. Whereas ENC should generally enhance constructor abundance and distribution (Krakauer et al., 2009; Kylafis & Loreau, 2008), its effects on bystander populations and affected food species, including demographic consequences, can range from negative to positive (Kylafis & Loreau, 2011; Maloof & Inouye, 2000). For bystander species, positive effects (facilitation) arise if environmental modification creates new foraging opportunities, whereas negative effects result from increased exploitation competition. If such effects occur persistently within and among generations and differentially affect individuals with particular heritable phenotypes, they could also precipitate evolutionary responses (Barker & Odling‐Smee, 2014; Odling‐Smee et al., 2013).
In the specific case of ENC by primary nectar robbing, the population consequences for constructor and bystander species are probably straightforward. For social species, including bumble bees, colony growth and production of sexual individuals vary positively with nectar intake (Requier et al., 2020; Rotheray et al., 2017). Consequently, populations of constructor species and facilitated bystanders should benefit from the actions of primary nectar robbers, whereas those of bystander species that experience increased competition should suffer. Thus, dahl populations likely suffer from competition with nectar‐robbing terr. Unfortunately, this expectation can no longer be tested just 5 years after our study, as dahl populations have been largely extirpated from southern South America, apparently owing to a more severe environmental impact of invading terr, specifically its role as a parasite vector (Arbetman et al., 2013; Schmid‐Hempel et al., 2014). In more persistent bee assemblages, competitive effects of ENC associated with nectar robbing should depend on the fraction of local plant species visited by both constructor and bystander species.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
AUTHORS' CONTRIBUTIONS
N.M.R. and M.A.A. conceived and developed the study design; N.M.R. and R.G.D. collected the data; N.M.R. and L.D.H. developed and implemented the statistical analyses; N.M.R. wrote the first draft of the manuscript and all authors contributed substantially to the final manuscript. All authors gave final approval of the accepted manuscript.
Supporting information
Supplementary Material
ACKNOWLEDGEMENTS
The authors thank the Administración de Parques Nacionales Argentinas for use of the Estación Biológica during data collection. They also thank Agustín Sáez, Carolina Morales and Marina Arbetman for help during the field season and feedback, Sara Reverté for field assistance and Soledad Hourmilougue for logistical support and field assistance. Comments by David Inouye and two anonymous reviewers improved the manuscript. The research was supported financially by a National Geographic Society Young Explorers Grant (9823‐15: N.M.R.), the National Fund for Research of Argentina (PICT 2015‐2333: M.A.A.), the University of Montana (Davidson Honors College Undergraduate Student/Faculty Research Award with R. Callaway; Global Leadership Initiative ‘Beyond the Classroom’ Scholarship; Davidson Honors College Travel Abroad Scholarship: R.G.D.) and a Discovery Grant from the Natural Sciences and Engineering Research Council of Canada (RGPIN/107375‐2012: L.D.H.).
Rosenberger, N. M. , Aizen, M. A. , Dickson, R. G. , & Harder, L. D. (2022). Behavioural responses by a bumble bee to competition with a niche‐constructing congener. Journal of Animal Ecology, 91, 580–592. 10.1111/1365-2656.13646
Handling Editor Andrew Jackson
DATA AVAILABILITY STATEMENT
Data used for this publication are available on Dryad Digital Repository https://doi.org/10.5061/dryad.rbnzs7hcj (Rosenberger et al., 2021).
REFERENCES
- Abrahamovich, A. H. , Tellería, M. C. , & Díaz, N. B. (2001). Bombus species and their associated flora in Argentina. Bee World, 82, 76–87. 10.1080/0005772X.2001.11099505 [DOI] [Google Scholar]
- Aizen, M. A. (2001). Flower sex ratio, pollinator abundance, and the seasonal pollination dynamics of a protandrous plant. Ecology, 82, 127–144. 10.1890/0012-9658(2001)082%5B0127:FSRPAA%5D2.0.CO;2 [DOI] [Google Scholar]
- Aizen, M. A. , & Rovere, A. E. (2010). Reproductive interactions mediated by flowering overlap in a temperate hummingbird‐plant assemblage. Oikos, 119, 696–706. 10.1111/j.1600-0706.2009.17762.x [DOI] [Google Scholar]
- Aizen, M. A. , Vázquez, D. P. , & Smith‐Ramírez, C. (2002). Historia natural y conservación de los mutualismos planta‐animal del bosque templado de Sudamérica austral. Revista Chilena de Historia Natural, 75, 79–97. 10.4067/S0716-078X2002000100008 [DOI] [Google Scholar]
- Arbetman, M. P. , Meeus, I. , Morales, C. L. , Aizen, M. A. , & Smagghe, G. (2013). Alien parasite hitchhikes to Patagonia on invasive bumblebee. Biological Invasions, 15, 489–494. 10.1007/s10530-012-0311-0 [DOI] [Google Scholar]
- Barker, G. , & Odling‐Smee, J. (2014). Integrating ecology and evolution: Niche construction and ecological engineering. In Barker G., Desjardins E., & Pearce T. (Eds.). Entangled life: Organism and environment in the biological and social sciences (pp. 187–211). Springer. [Google Scholar]
- Berry, P. E. (1989). A systematic revision of Fuchsia Sect. Quelusia (Onagraceae). Annals of the Missouri Botanical Garden, 76, 532–584. 10.2307/2399499 [DOI] [Google Scholar]
- Bolnick, D. I. , Svanbäck, R. , Fordyce, J. A. , Yang, L. H. , Davis, J. M. , Hulsey, C. D. , & Forister, M. L. (2003). The ecology of individuals: Incidence and implications of individual specialization. The American Naturalist, 161, 1–28. 10.1086/343878 [DOI] [PubMed] [Google Scholar]
- Bråthen, K. A. , & Ravolainen, V. T. (2015). Niche construction by growth forms is as strong a predictor of species diversity as environmental gradients. Journal of Ecology, 103, 701–713. 10.1111/1365-2745.12380 [DOI] [Google Scholar]
- Bronstein, J. L. , Barker, J. L. , Lichtenberg, E. M. , Richardson, L. L. , & Irwin, R. E. (2017). The behavioral ecology of nectar robbing: Why be tactic constant? Current Opinion in Insect Science, 21, 14–18. 10.1016/j.cois.2017.05.013 [DOI] [PubMed] [Google Scholar]
- Brooks, M. E. , Kristensen, K. , Benthem, K. J. , Magnusson, A. , Berg, C. W. , Nielsen, A. , Skaug, H. J. , Mächler, M. , & Bolker, B. M. (2017). glmmTMB balances speed and flexibility among packages for zero‐inflated generalized linear mixed modeling. The R Journal, 9, 378–400. 10.32614/RJ-2017-066 [DOI] [Google Scholar]
- Dall, S. R. X. , & Cuthill, I. C. (1997). The information costs of generalism. Oikos, 80, 197–202. 10.2307/3546535 [DOI] [Google Scholar]
- Dedej, S. , & Delaplane, K. S. (2005). Net energetic advantage drives honey bees (Apis mellifera L) to nectar larceny in Vaccinium ashei Reade. Behavioral Ecology and Sociobiology, 57, 398–403. 10.1007/s00265-004-0852-z [DOI] [Google Scholar]
- Dreisig, H. (1995). Ideal free distributions of nectar foraging bumblebees. Oikos, 72, 161–172. 10.2307/3546218 [DOI] [Google Scholar]
- Duchateau, M. J. , & Velthuis, H. H. W. (1988). Development and reproductive strategies in Bombus terrestris colonies. Behaviour, 107, 186–207. 10.1163/156853988X00340 [DOI] [Google Scholar]
- Dzendoletas, M. A. , Harvylenko, M. , & Crivelli, E. (2003). Fenología de plantas en Puerto Blest, Parque Nacional Nahuel Huapi, Patagonia, Argentina. Ecología, 17, 87–98. [Google Scholar]
- Fretwell, S. D. , & Lucas, H. L. Jr (1970). On territorial behavior and other factors influencing habitat distribution in birds. Acta Biotheoretica, 19, 16–36. 10.1007/BF01601955 [DOI] [Google Scholar]
- González‐Gómez, P. L. , & Valdivia, C. E. (2005). Direct and indirect effects of nectar robbing on the pollinating behavior of Patagona gigas (Trochilidae). Biotropica, 37, 693–696. 10.1111/j.1744-7429.2005.00088.x [DOI] [Google Scholar]
- Gosterit, A. (2011). Effect of different reproductive strategies on colony development characteristics in Bombus terrestris L. Journal of Apicultural Science, 55, 45–51. [Google Scholar]
- Goulson, D. (2010). Bumblebees: Behaviour, ecology, and conservation (2nd ed.). Oxford University Press. [Google Scholar]
- Goulson, D. , Lye, G. C. , & Darvill, B. (2008). Diet breadth, coexistence and rarity in bumblebees. Biodiversity and Conservation, 17, 3269–3288. 10.1007/s10531-008-9428-y [DOI] [Google Scholar]
- Hamilton, I. M. (2010). Foraging theory. In Westneat D. F. & Fox C. W. (Eds.), Evolutionary behavioral ecology (pp. 177–193). Oxford University Press. [Google Scholar]
- Higginson, A. D. , & Ruxton, G. D. (2015). Foraging mode switching: The importance of prey distribution and foraging currency. Animal Behaviour, 105, 121–137. 10.1016/j.anbehav.2015.04.014 [DOI] [Google Scholar]
- Houston, A. I. (2008). Matching and ideal free distributions. Oikos, 117, 978–983. 10.1111/j.0030-1299.2008.16041.x [DOI] [Google Scholar]
- Inouye, D. W. (1978). Resource partitioning in bumblebees: Experimental studies of foraging behavior. Ecology, 59, 672–678. 10.2307/1938769 [DOI] [Google Scholar]
- Inouye, D. W. (1980). The terminology of floral larceny. Ecology, 61, 1251–1253. 10.2307/1936841 [DOI] [Google Scholar]
- Irwin, R. E. , Bronstein, J. L. , Manson, J. S. , & Richardson, L. (2010). Nectar robbing: Ecological and evolutionary perspectives. Annual Review of Ecology, Evolution, and Systematics, 41, 271–292. 10.1146/annurev.ecolsys.110308.120330 [DOI] [Google Scholar]
- Isbell, F. , & Loreau, M. (2014). Sustainability of human ecological niche construction. Ecology and Society, 19, 45. 10.5751/ES-06395-190145 [DOI] [Google Scholar]
- Jones, C. G. , Lawton, J. H. , & Shachak, M. (1997). Positive and negative effects of organisms as physical ecosystem engineers. Ecology, 78, 1946–1957. 10.1890/0012-9658(1997)078%5B1946:PANEOO%5D2.0.CO;2 [DOI] [Google Scholar]
- Kotler, B. P. , & Brown, J. S. (2007). Community ecology. In Stephens D. W., Brown J. S., & Ydenberg R. C. (Eds.), Foraging: Behavior and ecology (pp. 397–434). University of Chicago Press. [Google Scholar]
- Krakauer, D. C. , Page, K. M. , & Erwin, D. H. (2009). Diversity, dilemmas, and monopolies of niche construction. The American Naturalist, 173, 26–40. 10.1086/593707 [DOI] [PubMed] [Google Scholar]
- Kutner, M. G. , Nachtscheim, C. J. , Neter, J. , & Li, W. (2005). Applied linear statistical models (5th ed.). McGraw‐Hill Irwin. [Google Scholar]
- Kylafis, G. , & Loreau, M. (2008). Ecological and evolutionary consequences of niche construction for its agent. Ecology Letters, 11, 1072–1081. 10.1111/j.1461-0248.2008.01220.x [DOI] [PubMed] [Google Scholar]
- Kylafis, G. , & Loreau, M. (2011). Niche construction in the light of niche theory. Ecology Letters, 14, 82–90. 10.1111/j.1461-0248.2010.01551.x [DOI] [PubMed] [Google Scholar]
- Laverty, T. M. , & Plowright, R. C. (1985). Competition between hummingbirds and bumble bees for nectar in flowers of Impatiens biflora . Oecologia, 66, 25–32. 10.1007/BF00378548 [DOI] [PubMed] [Google Scholar]
- Leadbeater, E. , & Chittka, L. (2008). Social transmission of nectar‐robbing behaviour in bumble‐bees. Proceedings of the Royal Society B: Biological Sciences, 275, 1669–1674. 10.1098/rspb.2008.0270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lichtenberg, E. M. , Irwin, R. E. , & Bronstein, J. L. (2018). Costs and benefits of alternative food handling tactics help explain facultative exploitation of pollination mutualisms. Ecology, 99, 1815–1824. 10.5281/zenodo.1243208 [DOI] [PubMed] [Google Scholar]
- Lichtenberg, E. M. , Richman, S. K. , Irwin, R. E. , & Bronstein, J. L. (2020). Competition for nectar resources does not affect bee foraging tactic constancy. Ecological Entomology, 45, 904–909. 10.1002/ecy.2395 [DOI] [Google Scholar]
- Madjidian, J. A. , Morales, C. L. , & Smith, H. G. (2008). Displacement of a native by an alien bumblebee: Lower pollinator efficiency overcome by overwhelmingly higher visitation frequency. Oecologia, 156, 835–845. 10.1007/s00442-008-1039-5 [DOI] [PubMed] [Google Scholar]
- Maloof, J. E. , & Inouye, D. W. (2000). Are nectar robbers cheaters or mutualists? Ecology, 81, 2651–2661. 10.1890/0012-9658(2000)081%5B2651:ANRCOM%5D2.0.CO;2 [DOI] [Google Scholar]
- Matthews, B. , De Meester, L. , Jones, C. G. , Ibelings, B. W. , Bouma, T. J. , Nuutinen, V. , van de Koppel, J. , & Odling‐Smee, J. (2014). Under niche construction: An operational bridge between ecology, evolution, and ecosystem science. Ecological Monographs, 84, 245–263. 10.1890/13-0953.1 [DOI] [Google Scholar]
- Milliken, G. A. , & Johnson, D. H. (1984). Analysis of messy data. Designed experiments (Vol. 1). Van Nostrand Reinhold. [Google Scholar]
- Navarro, L. (1999). Pollination ecology and effect of nectar removal in Macleania bullata (Ericaceae). Biotropica, 31, 618–625. 10.1111/j.1744-7429.1999.tb00410.x [DOI] [Google Scholar]
- Newman, D. A. , & Thomson, J. D. (2005). Effects of nectar robbing on nectar dynamics and bumblebee foraging strategies in Linaria vulgaris (Scrophulariaceae). Oikos, 110, 309–320. 10.1111/j.0030-1299.2005.13884.x [DOI] [Google Scholar]
- Odling‐Smee, J. , Erwin, D. H. , Palkovacs, E. P. , Feldman, M. W. , & Laland, K. N. (2013). Niche construction theory: A practical guide for ecologists. Quarterly Review of Biology, 88, 3–28. 10.1086/669266 [DOI] [PubMed] [Google Scholar]
- Pleasants, J. M. (1981). Bumblebee response to variation in nectar availability. Ecology, 62, 1648–1661. 10.2307/1941519 [DOI] [Google Scholar]
- Pyke, G. H. (1982). Local geographic distributions of bumblebees near Crested Butte, Colorado: Competition and community structure. Ecology, 63, 555–573. 10.2307/1938970 [DOI] [PubMed] [Google Scholar]
- R Core Team (2020). R: A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.R‐project.org/ [Google Scholar]
- Requier, F. , Jowanowitsch, K. K. , Kallnik, K. , & Steffan‐Dewenter, I. (2020). Limitation of complementary resources affects colony growth, foraging behavior, and reproduction in bumble bees. Ecology, 101, e02946. 10.1002/ecy.2946 [DOI] [PubMed] [Google Scholar]
- Rosenberger, N. M. , Aizen, M. A. , Dickson, R. G. , & Harder, L. D. (2021). Data from: Behavioral responses by a bumble bee to competition with a niche‐constructing congener. Dryad Digital Repository, 10.5061/dryad.rbnzs7hcj [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rotheray, E. L. , Osborne, J. L. , & Goulson, D. (2017). Quantifying the food requirements and effects of food stress on bumble bee colony development. Journal of Apicultural Research, 56, 288–299. 10.1080/00218839.2017.1307712 [DOI] [Google Scholar]
- SAS Institute Inc . (2016). SAS/STAT® 14.2 user's guide. SAS Institute Inc. [Google Scholar]
- Schmid‐Hempel, R. , Eckhardt, M. , Goulson, D. , Heinzmann, D. , Lange, C. , Plischuk, S. , Escudero, L. R. , Salathé, R. , Scriven, J. J. , & Schmid‐Hempel, P. (2014). The invasion of southern South America by imported bumblebees and associated parasites. Journal of Animal Ecology, 83, 823–837. 10.1111/1365-2656.12185 [DOI] [PubMed] [Google Scholar]
- Stanley, D. A. , & Cosnett, E. (2021). Catching the thief: Nectar robbing behaviour by bumblebees on naturalised Fuchsia magellanica in Ireland. Journal of Pollination Ecology, 29, 240–248. 10.26786/1920-7603(2021)620 [DOI] [Google Scholar]
- Stroup, W. W. (2013). Generalized linear mixed models: Modern concepts, methods and applications. CRC Press. [Google Scholar]
- Terborgh, J. , & Faaborg, J. (1973). Turnover and ecological release in the avifauna of Mona Island, Puerto Rico. The Auk, 90, 759–779. 10.2307/4084357 [DOI] [Google Scholar]
- Traveset, A. , Willson, M. F. , & Sabag, C. (1998). Effect of nectar‐robbing birds on fruit set of Fuchsia magellanica in Tierra Del Fuego: A disrupted mutualism. Functional Ecology, 12, 459–464. 10.1046/j.1365-2435.1998.00212.x [DOI] [Google Scholar]
- Valdivia, C. E. , Carroza, J. P. , & Orellana, J. I. (2016). Geographic distribution and trait‐mediated causes of nectar robbing by the European bumblebee Bombus terrestris on the Patagonian shrub Fuchsia magellanica . Flora, 225, 30–36. 10.1016/j.flora.2016.09.010 [DOI] [Google Scholar]
- Varma, S. , & Sinu, P. A. (2019). Nectar robbing in bellflower (Sesamum radiatum) benefited pollinators but unaffected maternal function of plant reproduction. Scientific Reports, 9, 8357. 10.1038/s41598-019-44741-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wisheu, I. C. (1998). How organisms partition habitats: Different types of community organization can produce identical patterns. Oikos, 83, 246–258. 10.2307/3546836 [DOI] [Google Scholar]
- Yoder, J. B. , Clancey, E. , Des roches, S. , Eastman, J. M. , Gentry, L. , Godsoe, W. , Hagey, T. J. , Jochimsen, D. , Oswald, B. P. , Robertson, J. , Sarver, B. A. J. , Schenk, J. J. , Spear, S. F. , & Harmon, L. J. (2010). Ecological opportunity and the origin of adaptive radiations. Journal of Evolutionary Biology, 23, 1581–1596. 10.1111/j.1420-9101.2010.02029.x [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material
Data Availability Statement
Data used for this publication are available on Dryad Digital Repository https://doi.org/10.5061/dryad.rbnzs7hcj (Rosenberger et al., 2021).


