Abstract
Cancer immunotherapy has emerged as one of the most powerful anticancer therapies. However, the details on the interaction between tumors and the immune system are complicated and still poorly understood. Optical fluorescence imaging is a technique that allows for the visualization of fluorescence-labeled immune cells and monitoring of the immune response during immunotherapy. To this end, near-infrared (NIR) light has been adapted for optical fluorescence imaging because it is relatively safe and simple without hazardous ionizing radiation and has relatively deeper tissue penetration into living organisms than visible fluorescence light. In this review, we discuss state-of-the-art NIR optical imaging techniques in cancer immunotherapy to observe the dynamics, efficacy, and responses of the immune components in living organisms. The use of bioimaging labeling techniques will give us an understanding of how the immune system is primed and ultimately developed.
Keywords: immunotherapy; cell engineering; biomarkers, tumor; adjuvants, pharmaceutic
Introduction
Cancer immunotherapy has emerged as one of the most powerful anti-cancer therapies. However, the details on the interaction between tumors and the immune system are complicated and still poorly understood.1 In this respect, current cancer immunotherapy is only effective for a relatively small number of patients. To overcome this limitation, understanding how the immune system combats cancer along with elucidating the mechanism of action is of significant importance. Thus, bioimaging of the proliferation, migration, differentiation, and activation of immune cells can serve as a powerful tool for visualizing various biological entities of immunological diseases and monitoring the progress of pathological conditions.2 Among many bioimaging modalities, optical fluorescence imaging in the near-infrared (NIR) window (650–1700 nm) has frequently been used in preclinical and clinical applications because it is relatively safe and simple without hazardous ionizing radiation and has relatively deeper tissue penetration into living organisms than visible fluorescence (figure 1A). Particularly, NIR fluorescence imaging in the second NIR window (NIR-II; >1000 nm) has recently captured the attention of many researchers in molecular cancer imaging due to the significant reduction in autofluorescence and tissue scattering, allowing deeper and higher resolution intraoperative imaging.3 These unique features permit the non-invasive imaging and tracking of immune cells in living organisms.
Figure 1.
Near-infrared (NIR) immunofluorescence imaging. (A) The biological window for optical fluorescence imaging. The NIR region has low absorption, scattering, and autofluorescence in tissue. Adapted from Ref. 39 with permission from Elsevier. (B) Schematic illustration of an optical imaging system and (C) light delivery method for minimal invasive optical imaging and photoimmunotherapy. Adapted from Refs 4 and 5 with permission from Springer Nature. LP, longpass; BP, bandpass; ROI, region of interest; RF, radio frequency; LED, light emitting diode. (D) Schematic representation of the tracking and monitoring of immune components. The tracking method is divided into ex vivo and in vivo labeling. Figures were created with Biorender.com.
In cancer immunotherapy, optical fluorescence imaging is used to visualize fluorescence-labeled immune cells and/or monitor immune responses under reflectance-based imaging and/or microscopic imaging systems. As illustrated in figure 1B, an NIR fluorescence imaging system requires excitation light sources, appropriate absorbance/emission filters, and sensitive charge-coupled device cameras.3 In addition, since cell-based treatment can be applied anywhere in the body, we frequently need to approach the macroscopic level of the area of interest to collect an image exclusively. To this end, optical fiber-based minimally invasive fluorescence imaging systems and light delivery methods should be considered (figure 1C).4 5
There are two categories in immunotherapy for which we can use optical imaging: tracking and monitoring of immune cells (figure 1D). For tracking purposes, immune cells can be labeled with small-molecule fluorophores or nanoparticles (NPs) in either ex vivo or in vivo situations to observe the behavior of immune components. Ex vivo labeling is not limited to therapeutic cells but can be applicable to any type of cells and immunological agents including vaccines. The fluorophores chemically and/or physically bind to the membrane proteins of isolated cells or are transported into the isolated cell by diffusion, endocytosis, or active transport.6 In contrast to ex vivo labeling, in vivo labeling should use specific targetability of exogenously administered fluorophores that can target specific surface receptors or membrane transporters of cells. To monitor and assess immune responses further, activatable fluorophores have also been explored. The diminished fluorescence signal of activatable fluorophores at normal conditions can be recovered (turned-on) via biological reactions such as enzymatic cleavage and oxidation.7 This strategy can minimize the background signals resulting in increased signal-to-background ratio as well as high specificity and sensitivity. This article provides a comprehensive review of state-of-the-art NIR optical imaging techniques used in cancer immunotherapy to monitor immune components and accompanying immune responses.
Labeling tools for NIR fluorescence imaging
NIR fluorescence imaging offers the desired spatial resolution required for the observation of immune components in a real-time setting. However, a selection of fluorophores is often crucial because the performance of labeling agents depends strongly on their physicochemical and optical properties (ie, hydrodynamic diameter (HD), molecular weight (MW), absorption/emission wavelengths, surface charges, pKa, hydrophobicity, plasma protein binding, and photostability).8–10 The ideal imaging probe for labeling the immune components of interest should have the following important properties: (1) high imaging specificity for tracking the desired immune components, (2) high imaging sensitivity for detection (reducing non-specific uptake), (3) non-immunogenicity, and (4) minimal toxicity.11
Currently available optical fluorescence probes for use in cell-based therapy are summarized in table 1. Transfection of cells with fluorescent proteins, such as biological green fluorescent protein, allows for longitudinal tracking of in vivo cell proliferation.12 However, it can be difficult to reach enough fluorescence density in a short period of time after injection, and the use of virus infection limits their transition to the clinic. Here, for this reason, we limit our discussion to small molecule and nanosized fluorophores along with their physicochemical characteristics and biomedical applications. Further detailed labeling mechanisms and strategies will be discussed in NIR Fluorescence Imaging of Immune System section.
Table 1.
Representative NIR fluorophores for ex vivo and in vivo labeling
| Class | Name | Type | Targets | Labeling mechanism | Reference(s) |
| Small-molecule fluorophores | DiX series | Heptamethine cyanine | Cell membrane | Membrane insertion | 14 50 |
| CIR38M | Heptamethine cyanine | Amine group on cell membrane protein | NHS ester amine reaction | 55 | |
| IRDye800CW | Heptamethine cyanine | Biomarker PD-L1 | Antibody affinity | 74 | |
| ESNF13 | Pentamethine cyanine | Mitochondria | Structure inherent targeting | 2 19 | |
| SH1 | Heptamethine cyanine | Monocyte in bone marrow | Structure inherent targeting | 20 | |
| CTNF126 | Heptamethine cyanine | Lysosome | Structure inherent targeting | 52 | |
| CDg16 | Aminoacridine | Activated macrophage (M2) | Structure inherent targeting | 78 | |
| CDnir7 | Heptamethine cyanine | Macrophage | Structure inherent targeting | 76 | |
| Nanoparticles | Quantum dot | Semiconductor crystal | Biomarker protein | Antibody affinity | 41 |
| Rare-earth nanoparticle | Rare-earth atoms | Biomarker protein | Antibody affinity | 79 |
NHS, N-hydroxysuccinimide; NIR, near-infrared; PD-L1, programmed cell death ligand-1.
Small-molecule fluorophores
Small-molecule fluorophores (a.k.a., fluorescence dyes or organic dyes) have several advantages such as a well-established synthetic route, well-defined molecular structure, good biocompatibility, and easy metabolism.13 Small-molecule fluorophores including polymethine cyanines, porphyrin analogs, xanthene-based dyes, and squaraine derivatives have been designed to visualize key components of immune systems in cell-based therapy. There are commercially available fluorescence dyes for ex vivo cell labeling, including chloromethyl fluorescein diacetate (CMFDA and CellTracker) and long alkyl chain cyanine dyes (lipophilic tracers: DiI, DiO, DiD, DiA, and DiR).14 The cell labeling mechanism with fluorophores is known to be based on a strong interaction with cellular components such as the insertion of fluorophores into bulk lipid membranes or covalent bonding on the cell membrane. However, the addition of cell tracking dyes can change cellular mechanical properties such as rigidity and adhesion due to the increases in the lateral interaction among phospholipid chains and amphiphilic dye molecules, which might interfere with cellular mobility and immunogenicity.15
Recently, simple cyanine-based fluorophores have been developed to target cells, organs, and tumors.16–18 The newly designed fluorophores have a native cellular targeting property resulting in no need for further chemical conjugation of targeting moieties, called the ‘structure-inherent targeting (SIT)’ strategy. The SIT fluorophores have been applied for longitudinal monitoring of cell proliferation and differentiation,2 real-time tracking of ex vivo expanded natural killer (NK) cells,19 and tumor-associated macrophage targeting.20 In addition, in vivo imaging is possible with cells labeled with fluorophores which have absorption/emission in the NIR window due to NIR light’s ability to overcome the penetration limitation, which is exhibited by light from the visible region.
For non-invasive imaging, the current focus is geared towards the development of NIR-II fluorophores, which yield high contrast to background ratios due to minimal tissue scattering and autofluorescence.21 22 Several types of NIR-II fluorophores including FD-1080,23 Flav7,24 25 and donor–acceptor–donor (D-A-D) structures with a benzothiadiazole core26 27 have been explored. The D-A-D structure expands semiconducting polymers, which can form self-assembling NPs with amphiphilic polymers. In addition to NIR-II fluorophores, the long non-negligible emission tail of existing NIR-I dyes (eg, indocyanine green, ICG) allows them to be repurposed for NIR-II fluorescence imaging, which yields even higher signal intensities than commercially available NIR-II dyes (eg, IR-E1050).28–30 In this way, the clinically approved fluorophore ICG has been demonstrated for NIR-II imaging in preclinical and clinical settings.31 32 Recently, Kang et al repurposed heptamethine fluorophores to NIR-II tail imaging and demonstrated the ability to target tumor-associated immune cells with high sensitivity and accuracy.20
Fluorescent nanoparticles
In contrast to small-molecule fluorophores, NPs hold great promise to overcome the low sensitivity, chemical degradation, and photobleaching/quenching of organic fluorophores in cell labeling. NPs can be further tuned in their optical, electrical, magnetic, and biological properties and carry large payloads along with contrast agents.33 Although this approach has several notable disadvantages including the complexity of their design, high cost, difficulty in large-scale production, and, most importantly, the unknown long-term toxicity to biological systems,34 NPs are generally more amenable to broader approaches for bioimaging and tissue targetability compared with small molecules.35 Ex vivo labeling with NPs mostly relies on naturally high endocytosis activity of cells, especially macrophages.36 In addition to endocytosis, several techniques such as electroporation, microinjection, and transfection have been developed.37 38
For in vivo labeling, NPs often need to be conjugated with specific targeting moieties such as antibodies, peptides, aptamers, and so on, to label immune cells selectively after administration. Despite many NPs having been described, they often have different in vivo behaviors and biological properties which are caused by the complexity of biological environments and the diversity of NPs. Thus, the physicochemical properties of NPs, such as HD, MW, shape, composition, hydrophilicity/lipophilicity, and surface characteristics, should be considered when selecting NPs for in vivo labeling.9 Additionally, the photophysical properties of NPs, including absorption/emission spectra, extinction coefficient, quantum yield, plasma protein binding, and photostability, can be another important factor for the selection of the right fluorophores.39 40
As such, fluorescent quantum dots (QDs) have been widely used for biomedical assays, imaging, and lymphatic mapping because they have shown superior optical properties and stability, and tunable wavelengths by size.41 However, the potential toxicity of heavy metal-cored QDs by disruption of mitochondrial function can lead to DNA damage, which presents a major obstacle to their clinical translation.42 On the other hand, semiconducting polymers are emerging fluorescent organic nanomaterials with photoconversion properties that allow for not only photodynamic therapy (PDT) and photothermal therapy (PTT) but also to serve as optical transducers for remote regulation of biological actions in living animals.43 In addition, hydrophobic small-molecule fluorophores can also form NPs with semiconducting polymers via self-assembly or chemical conjugation to improve the water solubility and prolong the blood circulation time of semiconducting polymers.44
NIR fluorescence imaging of immune system
NIR fluorescence imaging in cancer immunotherapy is majorly focused on the visualization of fluorescence-labeled immune cells or immunological agents such as vaccines to track and/or monitor them. In this section, we discuss the real-time tracking method of immune cells and vaccines by labeling with selected NIR fluorophores in either ex vivo or in vivo.
“Ex vivo” labeling for immune cells
To better understand the function and underlying mechanisms of immune responses, real-time in vivo monitoring and localization of fluorescence-labeled immune cells of interest have been actively investigated. The effector cells include T lymphocytes (ie, CD4+ and CD8+ T cells), NK cells, and dendritic cells (DCs).45 As a cancer treatment, effector cells can be amplified and modified to recognize tumor antigens ex vivo before transfusion back into the host to kill tumor cells selectively.46 Most studies focus on evaluating the migration and function of cytotoxic T lymphocytes in a model of adoptive transfer immunotherapy or Chimeric antigen receptor (CAR) T cell therapy, and the effector cells are detected by flow cytometry and microscopy days after labeling.47
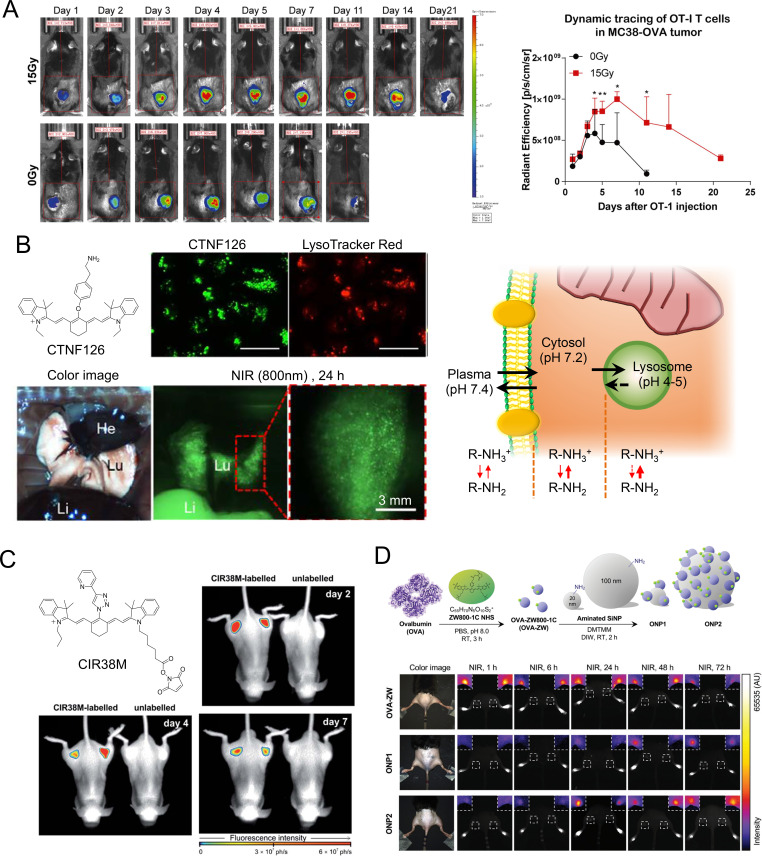
The exogenous labeling is generally performed by incubating cells with lipophilic fluorophores.48 There are two primary mechanisms of entry into the cell: (1) direct fusion with the plasma membrane and (2) endocytosis.6 The choice of labeling method can be determined based on the final goal of the study. For example, commercially available lipophilic tracers (eg, DiI, DiO, DiD, DiA, and DiR) contain two long alkyl chains which can be easily captured by the phospholipid bilayer of the plasma membrane in an aqueous medium.49 Therefore, this method is frequently used for T cell tracking in the context of cancer immunotherapy in preclinical models. As shown in figure 2A, DiR-labeled OT-I T cells accumulated specifically in tumorous tissues 24 hours after the adoptive transfer of T cells, which peaked on day 4 and persisted up to day 21 in mice.50
Figure 2.
Ex vivo labeling for immune cells and vaccines. (A) Fluorescence imaging from DiR-labeled OT-I T cell injected mice (left panel). Representative fluorescence signals from DiR-labeled cells relative to background fluorescence signals (right panel). Reproduced from Ref. 50 under (CC BY 4.0). (B) Longitudinal cell tracking process of near-infrared (NIR) fluorophores and lysosomal sequestration of CTNF126. Reproduced from Ref. 52 with permission from John Wiley and Sons. (C) Long-term longitudinal tracking of CIR38M-labeled CD4+ T cells in vivo. Reproduced from Ref. 55 under (CC BY 3.0). (D) Preparation of model vaccines using NIR fluorescent ZW800-1C and quantitative image analysis of vaccine trafficking in the draining lymph nodes. Reproduced from Ref. 66 with permission from John Wiley and Sons.
Of note, tagging the cell membrane can interfere with cellular mobility, while staining mitochondria can affect the membrane potential.15 51 To minimize such potential alterations of cellular activities, Choi and Henary designed lysosome-targeted NIR fluorophores including CTNF126 by adding a primary amine docking moiety on the heptamethine core (figure 2B).52 The primary amine linker can be ionized to a cation which allows cellular membrane permeation, after which the dye is rapidly sequestered by efficient intracellular fixations via a reductive amination reaction by formalin.52 CTNF126-labeled human prostate cancer cells (PC3) were administered to mice, and their fluorescence was observed longitudinally in living organisms.52–54 CTNF126 has a high extinction coefficient, quantum yield, and photostability, which is useful for sequential cell tracking in the body or under microscopy. Another labeling technique is to make a covalent bond to the cell membrane using the active NHS ester form of fluorophores. Mellanby et al designed a tricarbocyanine N-triazole-based NIR fluorophore CIR38M as a non-transferable marker to monitor the fate of therapeutic T cells non-invasively in vivo (figure 2C).55 CIR38M, which exhibits improved brightness and photostability, can label small populations of T cells with high sensitivity (around 4000 cells), extending the detectability of cells in vivo to over 7 days post-transfer. No functional alterations were observed in T cells with this method.
NIR-fluorescent NPs have also been investigated for ex vivo cell labeling, mainly relying on endocytosis as the labeling technique. Fuller et al proved that silica NPs coated with cationic polymers enabled them to escape from the endosomes and enter the cytoplasm of the subject cells.56 Zhao et al labeled bone marrow-derived macrophages (BMMs) with NIR light-activated phthalocyanine-loaded lipid NPs, with no impairment in cellular functioning.57 Engineered BMMs were observed to efficiently home to primary and bone metastatic tumors by NIR fluorescence imaging. In addition, chemotherapeutic agents inside engineered BMMs underwent NIR-triggered release, then effectively killed primary tumors via chemotherapy and induced immunogenic cancer cell death. Heo et al synthesized poly(lactic-co-glycolic acid) (PLGA) NPs containing tumor model antigen ovalbumin (OVA) and ICG to deliver tumor antigen-specific information to DCs ex vivo and track the migration of DCs in vivo.58 PLGA NP-treated DCs were identified similarly to prime CD8+ T cells via cross-presentation by cotreating them with another PLGA NP containing small interfering RNA and an immune modifier R837. The migration of DCs to lymph nodes was detected by in vivo NIR imaging for 48 hours. In addition, Xiang et al demonstrated that tumor antigen-presenting DCs labeled with upconversion nanoparticles emit high-energy photons in the draining lymph nodes.59 In a similar study, Lim and colleagues labeled human NK cells with antibody-coated QDs to show the therapeutic effect of intratumorally injected NK cells using NIR imaging.41
“Ex vivo” labeling for vaccines
Vaccines have been a successful and effective medical intervention, representing one of the most important applications of immunology to prevent infectious diseases.60 61 The same strategy has been used to enhance immune responses against cancer. Cancer vaccines are designed to train the immune system to recognize cancer, thereby treating active cancer or preventing future cancer.62 63 However, the dynamics, efficacy, and responses of many vaccines are not fully characterized due to a lack of an optimal imaging tool. Therefore, longitudinal, non-invasive, and reliable imaging methods are urgently needed.
Lindsay et al directly labeled the yellow fever prME mRNA as a model vaccine with an orthogonal dual positron emission tomography (PET)-NIR probe (DOTA-Cu64 DyLight 680) and observed the dynamics of the mRNA vaccine at the injection site and in the draining lymph nodes using PET-CT and NIR imaging after intramuscular administration to monkeys.64 The mRNA-positive lymph node was able to be selectively extracted during necropsy with a portable NIR camera at 28 hours postinjection. This approach may reveal spatiotemporal determinants of vaccine efficacy in preclinical and translational studies employing large mammals.
Separately, model antigen OVA-conjugated ICG was investigated as a photosensitizing molecule, where ICG-OVA nanovaccines appeared in the inguinal lymph node at 0.5 hour post-intradermal injection and accumulated over time up to 9-hour postinjection.65 Katagiri et al reported a real-time trafficking method of a model vaccine labeled with zwitterionic NIR fluorophore ZW800-1C, which enabled the determination of the longitudinal fate of vaccines after intradermal administration (figure 2D).66 Of note, zwitterionic NIR fluorophores show minimal interactions with biological tissues and do not interfere with the interactions between vaccines and immune cells due to their non-sticky properties resulting from their balanced charges and hydrophilicity.67–72 This combination of zwitterionic NIR fluorophores and NIR imaging can be useful for the optimization of vaccine design as well as the safety evaluation of clinical vaccine candidates. Optical imaging techniques will be increasingly used to uncover how immune systems and vaccine molecules interact, resulting in the development of more effective cancer vaccines.
“In vivo” labeling for immune cells
In the current state of technology, the ex vivo labeling of lymphocytes (eg, CAR-T cells) from isolated cells is prevalent for NIR imaging.73 There are, however, significant limitations to this method as the isolation and labeling procedures have an unfavorable effect on lymphocytes. Therefore, a new methodology is highly desired to indirectly label sensitive immune cells including T and B cells in vivo with an injectable probe.35 Chatterjee et al labeled anti-programmed cell death ligand-1 (anti-PD-L1) antibody MPDL3280A (atezolizumab) with IRDye800CW or the radioactive isotope indium-111 (111In).74 Imaging PD-L1 expression was validated in orthotopic and subcutaneous triple-negative breast cancer and non-small cell lung cancer-bearing xenografts with varying levels of expression of PD-L1. The results were consistent between data from optical imaging and single-photon emission computed tomography (SPECT)/CT regarding tissue antibody deposits. It is theoretically possible to label any monoclonal antibodies (mAbs) or peptides with a fluorescent agent for immune cell imaging. However, according to Connor et al, IRDye800CW labeling via NHS chemistry results in significantly altered pharmacokinetics and tissue distribution of the fluorophore-labeled mAb compared with conventional radiolabeling.75
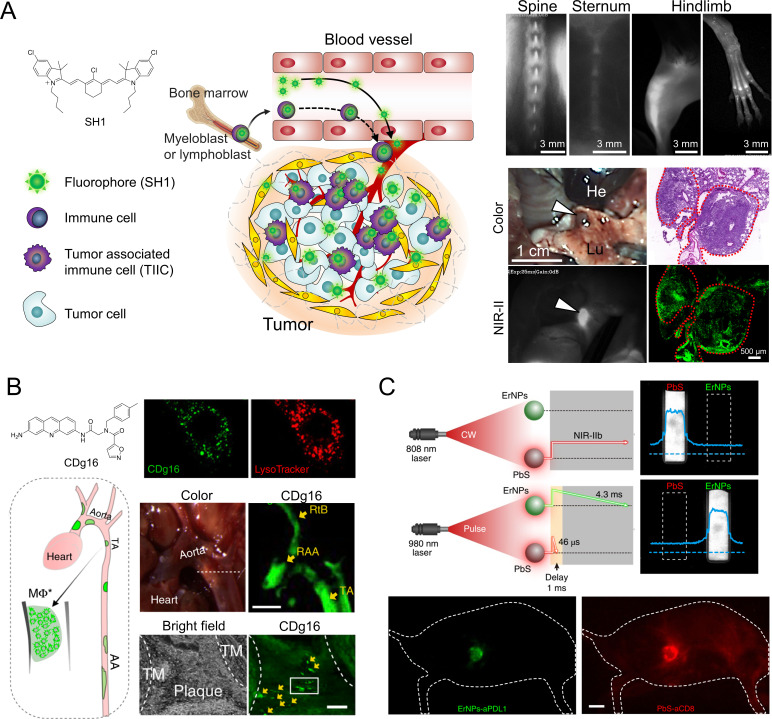
Since no chemical conjugation of targeting moieties is needed, “structure-inherent targeting (SIT)” is gaining more attention since it was first introduced in 2015.17 The SIT strategy suggests that bone-marrow-derived and/or tissue-resident and tumor-associated immune cells can be targeted using heptamethine cyanine-based fluorophores without further modification and/or conjugation of targeting moieties (figure 3A).20 Kang et al reported immune cell targeting in bone marrow with NIR-II fluorophore SH1 as an SIT fluorophore without targeting ligands. SH1 provides ubiquitous tumor targetability in pancreatic, breast, and lung cancer models with a high tumor-to-background ratio (TBR) via a tumor-associated immune cell-mediated targeting mechanism.
Figure 3.
In vivo labeling for immune cells. (A) Tumor-associated immune cell‐mediated orthotopic tumor imaging with SH1 NIR fluorophores. Reproduced from Ref. 20 with permission from John Wiley and Sons. (B) Activated macrophage staining with CDg16 for the detection of atherosclerotic plaques. Reproduced from Ref. 78 under (CC BY 4.0). (C) In vivo duplex NIR-II imaging of immune responses using ErNPs-aPDL1 (green color) and PbS-aCD8 (red color). AA, abdominal aorta; He, heart; Lu, lung; NIR, near-infrared; RAA, the root of aorta arch; RtB; right brachiocephalic artery; TA, thoracic aorta; TM, tunica intima. aPDL1, anti-programmed cell death-ligand 1; ErNPs, erbium-based nanoparticles; PbS, lead sulfide. Reproduced from Ref. 79 with permission from Springer Nature.
Moreover, Chang and coworkers have explored a diversity-oriented fluorescence library (DOFL) approach to find macrophage-targeted NIR fluorophores.76–78 Using DOFL screening, CDnir7 based on heptamethine was discovered to selectively detect macrophages and visualize inflammation in vivo using the IVIS spectrum. They also discovered an activated macrophage probe CDg16 having a selective cellularization mechanism through the SLC18B1 transporter. The CDg16 staining effectively and selectively visualizes inflammatory atherosclerotic plaques ex vivo (figure 3B). However, CDg16 emits green fluorescence light, which is not viable for in vivo imaging due to the high tissue absorption and scattering as well as elevated autofluorescence.7
Antibody-nanoparticle conjugates have been demonstrated for the in vivo labeling of immune cells. Dai and colleagues demonstrated duplex imaging of PD-L1 and cytotoxic T lymphocytes by using two biocompatible NPs, anti-PD-L1 antibody-conjugated rare-earth erbium-based NPs (ErNPs) and anti-CD8α conjugated lead sulfide QDs, respectively (figure 3C).79 ErNPs exhibiting downconversion luminescence at ~1600 nm achieved an improved TBR. They demonstrated that in vivo NIR-IIb molecular imaging of PD-L1 and CD8 revealed the activation of cytotoxic CD8+ T lymphocytes in the tumor microenvironment in response to immunotherapy.
Monitoring immune response with activatable probes
In most applications, “always-on” imaging probes have been used to monitor the interaction of immune cells. However, the detected signals from these fluorophores do not correlate well with biomarker expression levels relevant to immunotherapeutic responses. Activatable probes that can only activate in the intended biomarker’s presence have a minimal background and increased specificity compared with always-on probes.80 This means that activatable probes can be used as real-time in vivo imaging agents for monitoring immunotherapeutic responses. To monitor immune response with activatable probes, internal microenvironment stimuli such as pH, enzymes, redox potential, and oxidative stress have been considered.9
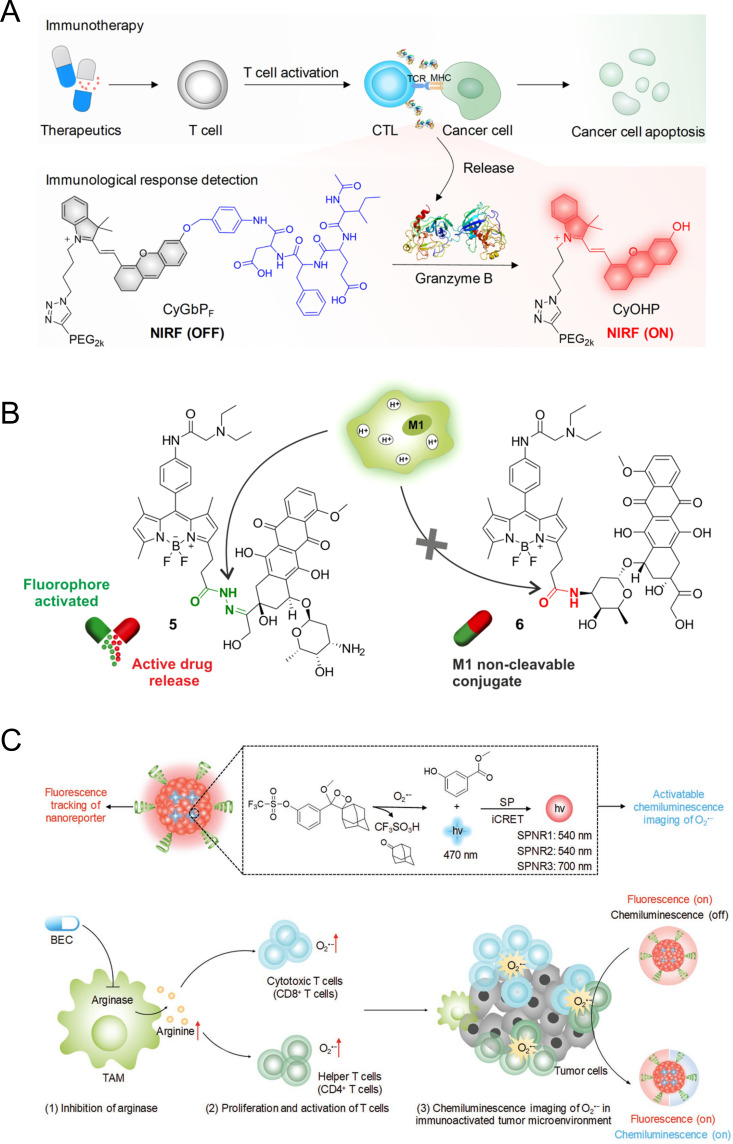
Cancer immunotherapy entails activating and differentiating naive T cells, which are then differentiated in the body to cytotoxic T-lymphocytes (CTLs) after contact with antigen-presenting cells. Granzyme B (GranB) can be used as a biomarker for immune activation because it is released mainly by the CTLs to kill cancer cells. Pu and colleagues synthesized two NIR macromolecular reporters caged by two different peptide substrates: CyGbPF and CyGbPP, containing N-acetyl-Ile-Glu-Phe-Asp (IEFD) and N-acetyl-IleGlu-Pro-Asp (IEPD), respectively, which are responsive to GranB for real-time in vivo evaluation of immunotherapy (figure 4A).81 The reporters are initially non-fluorescent due to the diminished electron-donating ability of the oxygen atom in CyOH. However, on cleavage of the peptide-caged moiety by GranB released from CTLs in the tumor microenvironment, CyGbPF or CyGbPP are converted into CyOHP which exhibits an enhanced NIR fluorescence signal. In this way, turn-on signal intensity was linked to the level of GranB expression, which allows for in situ assessment of immunotherapeutic response in living animals.
Figure 4.
Monitoring immune response with activatable probes. (A) Fluorescence imaging mechanism of granzyme B activatable probe. Reproduced from Ref. 81 with permission from American Chemical Society. (B) Cell tracking mechanism based on BODIPY−prodrug M1 activatable conjugate. Reproduced from 82 under (CC-BY). (C) Illustration of the mechanism of superoxide anion (O2·−) activated chemiluminescence SPNR for imaging of immune activation. Reproduced from Ref. 84 with permission from John Wiley and Sons. CTL, cytotoxic T-lymphocyte; MHC, major histocompatability complex; NIR, near-infrared; SPNR, superoxide anion activatable NIR chemiluminescent reporter; TCR, T-cell receptor; BEC, S-(2-boronoethyl)-L-cysteine; TAM, tumor-associated macrophage.
Separately, Fernandez et al demonstrated the discrimination of subpopulations of macrophages (ie, proinflammatory M1 vs anti-inflammatory M2 macrophages) (figure 4B).82 A BODIPY fluorophore was conjugated with prodrug (doxorubicin, DOX) through an acid-labile hydrazone bond. Under physiological conditions, fluorophore-prodrug conjugates display a relatively weak fluorescence intensity, but in mildly acidic environments (pH 4.5–6.5), the vulnerable hydrazone linkage is hydrolyzed, resulting in the release of cytotoxic DOX and the turning on of pH-dependent BODIPY fluorescence. Among the different subpopulations of macrophages, M1 macrophages contain intracellular acidic phagosomes which present pH values between 4.5 (late phagosomes) and 6.5 (early phagosomes). Thus, fluorophore-prodrug exhibits dose-dependent turn-on fluorescence response and cytotoxicity in lipopolysaccharide-induced proinflammatory M1 macrophages, while no response is observed in anti-inflammatory M2 macrophages. This result suggested that fluorogenic reaction-based prodrug conjugates allow for the real-time monitoring of targeted therapies such as prodrug activation and intracellular trafficking.83
Cui et al reported a series of superoxide anion (O2·−) activatable NIR chemiluminescent reporters (SPNRs) to detect O2·− for real-time in vivo NIR imaging of drug-induced cancer immune activation (figure 4C).84 SPNRs consist of a semiconducting polymer with emission at ~700 nm and a dioxetane derivative as an O2·− responsive chemiluminescence substrate. On specific reaction with O2·−, the sulfonate ester of caged dioxetanes within SPNRs was specifically cleaved to induce the deprotection of trifluoromethanesulfonate groups, resulting in the formation of highly energetic and unstable phenolate-dioxetane derivatives. Subsequently, intraparticle chemiluminescence resonance energy transfer from the unstable derivatives to the semiconducting polymer occurs, leading to the generation of chemiluminescence signals at 700 nm. Since cytotoxic T cells have higher endogenous O2·− level than cancer and normal cells, SPNR was able to turn on the chemiluminescence signals to report the O2·− level depending on the populations of activated cytotoxic T cells and helper T cells during the cancer immunotherapy.
Potential applications of immune cell-targeted imaging in cancer immunotherapy
NIR fluorophores for immune cell-targeted imaging could be used for a theranostic approach. An ideal cancer therapy would directly destroy cancer cells and activate the local host immune response to eliminate remaining cancer cells.85 To this end, many efforts have been made to minimize off-target effects in the immune system. One of such therapeutic modalities includes PDT, in which a photosensitive agent is activated through light.86 In addition to its own targetability, delivering light to a targeted tumor adds an additional layer of specificity which prevents normal cells from being severely damaged. However, PDT predominantly induces apoptosis of cancer cells which could negatively result in tolerogenic cell death and tolerance to these cells.87 On the other hand, PTT converts light energy into heat to induce direct cancer cell death at the target site and has proven effective in both preclinical and clinical settings.4 Both PDT and PTT modalities target cancer cells, not the immune system.
Another significant approach is immunomodulation with an immune cell-targeted imaging probe. An important feature of host immune activation induced by cancer immunotherapy is the repriming of polyclonal CD8+ T cells against various released cancer antigens. Therefore, the development of theranostic agents which target immune cells is useful for both imaging and inducing reproducible priming of T cells on NIR irradiation, presenting a new avenue for cancer immunotherapy.47 Currently, there is a paucity of theranostic agents for cancer imaging and immunotherapy. As reviewed in the “Ex vivo” labeling for immune cells section, most current efforts focus on ex vivo labeling of innate immune cells including macrophages or DCs with NPs. For clinical applications and better efficacy, in vivo labeling of a wide array of immune cells and reproducible control of activation of these cells is desired. Such an agent could potentially induce a large enough robust immune response to take on the bulk of cancer cells within an established tumor and long-term regression to resolve the historical issues in cancer therapy.
Immune cell-targeted imaging could further resolve one of the crucial issues in cancer immunotherapy, named “pseudoprogression.” Pseudoprogression refers to an increase in the size of a lesion or the number of metastatic lesions observed in MRI or CT scans because of infiltration of immune cells, such as T or B lymphocytes, although the treatment is actually working and the condition is improving.88 For this reason, pseudoprogression often leads to premature cessation of efficacious treatment owing to the false judgment of progression.89 Even though several methods to diagnose pseudoprogression after immunotherapy have been introduced, such as biopsies of enlarged lesions or new lesions, radiographic follow-up, MRI+PET CT, ultrasonography, circulating tumor DNA analysis, and serum interleukin 8 level detection, the diagnostic accuracy is still controversial.90–93 In addition, there are many studies on pseudoprogression, but there is still no consensus on its exact molecular mechanism. The combination of immune cell targeting fluorophores and NIR imaging might identify the degree of uptake for immune cells, which is a new way to differentiate pseudoprogression from true progression. Such differential theranostics might provide more accurate diagnosis and treatment plans to physicians, for example, avoiding premature discontinuation of immunotherapy or allowing for early initiation of other treatments, including personalized therapy.
Conclusions and perspectives
Optical imaging techniques provide high-resolution, simple, non-hazardous, and economical advantages over the other bioimaging modalities. For these reasons, optical imaging is the most widely used molecular and cellular imaging technique in preclinical applications.1 In particular, NIR imaging designed to monitor anticancer immune responses has made significant contributions and will continue to provide important insights to cancer immunotherapy communities.
Although NIR imaging shows many advantages including its non-toxic, low-cost, real-time, and high-resolution capabilities, as well as suitability for preclinical research, it is limited in human subjects due to low light penetration in thick layers of tissue and complications related to clinical imaging.48 Specifically, high resolution (ie, to the single immune cell level), real-time live imaging with deep penetration depth is desirable for longitudinal analysis of the dynamics of immune cell interactions. To this end, there is a significant interest in the development of NIR-II fluorophores for cancer immunotherapy as discussed in the “Ex vivo” labeling for immune cells section.94 95
Another direction includes afterglow technology, which provides ultralong-lived emitting probes after switching off the excitation source.96–98 Afterglow probes trap irradiated photoenergy and then slowly release the stored energy by photonic emission on physical activations. These real-time, light-excitation-free ultrasensitive probes can circumvent the interference of tissue autofluorescence and the limitation of tissue penetration depth (up to 5 cm). However, both technologies lack highly sensitive and biocompatible imaging probes, which significantly impedes their clinical application. The development of non-toxic, efficacious agents for NIR-II and afterglow imaging is warranted to broaden the application of optical imaging in cancer immunotherapy. Alternatively, multimodal imaging probes for both optical and nuclear or magnetic imaging have also been employed to overcome the limitations of each imaging modality and may be able to advance imaging of the immune system for future clinical use.
Acknowledgments
We thank Wesley R Stiles for manuscript editing.
Footnotes
Contributors: HSC provided direction and guidance throughout the preparation of this manuscript. HK drafted the manuscript and the figures. All authors reviewed and edited the manuscript.
Funding: This study was supported by NHLBI No: R01HL143020, the Creative Materials Discovery Program through the National Research Foundation of Korea (2019M3D1A1078938), and ZTI Biosciences (2020A015360). The content expressed is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Competing interests: None declared.
Provenance and peer review: Commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
Not applicable.
References
- 1. Lucignani G, Ottobrini L, Martelli C, et al. Molecular imaging of cell-mediated cancer immunotherapy. Trends Biotechnol 2006;24:410–8. 10.1016/j.tibtech.2006.07.003 [DOI] [PubMed] [Google Scholar]
- 2. Kim SH, Park G, Hyun H, et al. Near-infrared lipophilic fluorophores for tracing tissue growth. Biomed Mater 2013;8:014110. 10.1088/1748-6041/8/1/014110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Choi HS, Kim HK. Multispectral image-guided surgery in patients. Nat Biomed Eng 2020;4:245–6. 10.1038/s41551-020-0536-7 [DOI] [PubMed] [Google Scholar]
- 4. Li X, Lovell JF, Yoon J, et al. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat Rev Clin Oncol 2020;17:657–74. 10.1038/s41571-020-0410-2 [DOI] [PubMed] [Google Scholar]
- 5. Rho S, Stillwell R, Fay P. Optically-enhanced wireless breast lesion localization device for use during lumpectomy. SPIE BiOS 2022;11949. 10.1117/12.2606061 [DOI] [Google Scholar]
- 6. Rennick JJ, Johnston APR, Parton RG. Key principles and methods for studying the endocytosis of biological and nanoparticle therapeutics. Nat Nanotechnol 2021;16:266–76. 10.1038/s41565-021-00858-8 [DOI] [PubMed] [Google Scholar]
- 7. Owens EA, Henary M, El Fakhri G, et al. Tissue-Specific near-infrared fluorescence imaging. Acc Chem Res 2016;49:1731–40. 10.1021/acs.accounts.6b00239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Owens EA, Lee S, Choi J, et al. NIR fluorescent small molecules for intraoperative imaging. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2015;7:828–38. 10.1002/wnan.1337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kang H, Hu S, Cho MH, et al. Theranostic nanosystems for targeted cancer therapy. Nano Today 2018;23:59–72. 10.1016/j.nantod.2018.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Choi HS, Liu W, Liu F, et al. Design considerations for tumour-targeted nanoparticles. Nat Nanotechnol 2010;5:42–7. 10.1038/nnano.2009.314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Nguyen PK, Lan F, Wang Y, et al. Imaging: guiding the clinical translation of cardiac stem cell therapy. Circ Res 2011;109:962–79. 10.1161/CIRCRESAHA.111.242909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Arnhold S, Lenartz D, Kruttwig K, et al. Differentiation of green fluorescent protein-labeled embryonic stem cell-derived neural precursor cells into Thy-1-positive neurons and glia after transplantation into adult rat striatum. J Neurosurg 2000;93:1026–32. 10.3171/jns.2000.93.6.1026 [DOI] [PubMed] [Google Scholar]
- 13. Dai H, Wang X, Shao J, et al. NIR-II organic Nanotheranostics for precision Oncotherapy. Small 2021;17:e2102646. 10.1002/smll.202102646 [DOI] [PubMed] [Google Scholar]
- 14. Godement P, Vanselow J, Thanos S, et al. A study in developing visual systems with a new method of staining neurones and their processes in fixed tissue. Development 1987;101:697–713. 10.1242/dev.101.4.697 [DOI] [PubMed] [Google Scholar]
- 15. Lulevich V, Shih Y-P, Lo SH, et al. Cell tracing dyes significantly change single cell mechanics. J Phys Chem B 2009;113:6511–9. 10.1021/jp8103358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hyun H, Owens EA, Wada H, et al. Cartilage-Specific near-infrared fluorophores for biomedical imaging. Angew Chem Int Ed Engl 2015;54:8648–52. 10.1002/anie.201502287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Hyun H, Park MH, Owens EA, et al. Structure-inherent targeting of near-infrared fluorophores for parathyroid and thyroid gland imaging. Nat Med 2015;21:192–7. 10.1038/nm.3728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Hyun H, Wada H, Bao K, et al. Phosphonated near-infrared fluorophores for biomedical imaging of bone. Angew Chem Int Ed Engl 2014;53:10668–72. 10.1002/anie.201404930 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Uong TNT, Lee K-H, Ahn S-J, et al. Real-time tracking of Ex Vivo-expanded natural killer cells toward human triple-negative breast cancers. Front Immunol 2018;9:825. 10.3389/fimmu.2018.00825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kang H, Shamim M, Yin X, et al. Tumor-associated immune-cell-mediated tumor-targeting mechanism with NIR-II fluorescence imaging. Adv Mater 2022;34:2106500. 10.1002/adma.202106500 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hong G, Antaris AL, Dai H. Near-Infrared fluorophores for biomedical imaging. Nat Biomed Eng 2017;1:0010. 10.1038/s41551-016-0010 [DOI] [Google Scholar]
- 22. Zhu S, Tian R, Antaris AL, et al. Near-Infrared-II molecular dyes for cancer imaging and surgery. Adv Mater 2019;31:e1900321:1900321. 10.1002/adma.201900321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Li B, Lu L, Zhao M, et al. An efficient 1064 nm NIR-II excitation fluorescent molecular dye for deep-tissue high-resolution dynamic bioimaging. Angew Chem Int Ed Engl 2018;57:7483–7. 10.1002/anie.201801226 [DOI] [PubMed] [Google Scholar]
- 24. Cosco ED, Caram JR, Bruns OT, et al. Flavylium Polymethine fluorophores for near- and Shortwave infrared imaging. Angew Chem Int Ed Engl 2017;56:13126–9. 10.1002/anie.201706974 [DOI] [PubMed] [Google Scholar]
- 25. Cosco ED, Spearman AL, Ramakrishnan S, et al. Shortwave infrared polymethine fluorophores matched to excitation lasers enable non-invasive, multicolour in vivo imaging in real time. Nat Chem 2020;12:1123–30. 10.1038/s41557-020-00554-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Yang Q, Ma Z, Wang H, et al. Rational design of molecular fluorophores for biological imaging in the NIR-II window. Adv Mater 2017;29. 10.1002/adma.201605497. [Epub ahead of print: 24 01 2017]. [DOI] [PubMed] [Google Scholar]
- 27. Wan H, Yue J, Zhu S, et al. A bright organic NIR-II nanofluorophore for three-dimensional imaging into biological tissues. Nat Commun 2018;9:1171. 10.1038/s41467-018-03505-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Carr JA, Franke D, Caram JR, et al. Shortwave infrared fluorescence imaging with the clinically approved near-infrared dye indocyanine green. Proc Natl Acad Sci U S A 2018;115:4465–70. 10.1073/pnas.1718917115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Bai L, Hu Z, Han T, et al. Super-stable cyanine@albumin fluorophore for enhanced NIR-II bioimaging. Theranostics 2022;12:4536–47. 10.7150/thno.71443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Tian R, Feng X, Wei L, et al. A genetic engineering strategy for editing near-infrared-II fluorophores. Nat Commun 2022;13:2853. 10.1038/s41467-022-30304-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Cai Z, Zhu L, Wang M, et al. NIR-II fluorescence microscopic imaging of cortical vasculature in non-human primates. Theranostics 2020;10:4265–76. 10.7150/thno.43533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Hu Z, Fang C, Li B, et al. First-in-human liver-tumour surgery guided by multispectral fluorescence imaging in the visible and near-infrared-I/II windows. Nat Biomed Eng 2020;4:259–71. 10.1038/s41551-019-0494-0 [DOI] [PubMed] [Google Scholar]
- 33. Choi HS, Frangioni JV. Nanoparticles for biomedical imaging: fundamentals of clinical translation. Mol Imaging 2010;9:291–310. 10.2310/7290.2010.00031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Soares S, Sousa J, Pais A, et al. Nanomedicine: principles, properties, and regulatory issues. Front Chem 2018;6:360. 10.3389/fchem.2018.00360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Ji Y, Jones C, Baek Y, et al. Near-Infrared fluorescence imaging in immunotherapy. Adv Drug Deliv Rev 2020;167:121–34. 10.1016/j.addr.2020.06.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Weissleder R, Nahrendorf M, Pittet MJ. Imaging macrophages with nanoparticles. Nat Mater 2014;13:125–38. 10.1038/nmat3780 [DOI] [PubMed] [Google Scholar]
- 37. Sun C, Cao Z, Wu M, et al. Intracellular tracking of single native molecules with electroporation-delivered quantum dots. Anal Chem 2014;86:11403–9. 10.1021/ac503363m [DOI] [PubMed] [Google Scholar]
- 38. Damalakiene L, Karabanovas V, Bagdonas S, et al. Intracellular distribution of nontargeted quantum dots after natural uptake and microinjection. Int J Nanomedicine 2013;8:555–68. 10.2147/IJN.S39658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Son J, Yi G, Yoo J, et al. Light-responsive nanomedicine for biophotonic imaging and targeted therapy. Adv Drug Deliv Rev 2019;138:133–47. 10.1016/j.addr.2018.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Owens EA, Lee S, Choi J, et al. NIR fluorescent small molecules for intraoperative imaging. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2015;7:828–38. 10.1002/wnan.1337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Lim YT, Cho MY, Noh Y-W, et al. Near-infrared emitting fluorescent nanocrystals-labeled natural killer cells as a platform technology for the optical imaging of immunotherapeutic cells-based cancer therapy. Nanotechnology 2009;20:475102. 10.1088/0957-4484/20/47/475102 [DOI] [PubMed] [Google Scholar]
- 42. Kang H, Mintri S, Menon AV, et al. Pharmacokinetics, pharmacodynamics and toxicology of theranostic nanoparticles. Nanoscale 2015;7:18848–62. 10.1039/C5NR05264E [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Li J, Pu K. Semiconducting polymer nanomaterials as near-infrared photoactivatable Protherapeutics for cancer. Acc Chem Res 2020;53:752–62. 10.1021/acs.accounts.9b00569 [DOI] [PubMed] [Google Scholar]
- 44. Kang H, Rho S, Stiles WR, et al. Size‐dependent EPR effect of polymeric nanoparticles on tumor targeting. Adv Healthc Mater 2020;9:1901223. 10.1002/adhm.201901223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Restifo NP, Dudley ME, Rosenberg SA. Adoptive immunotherapy for cancer: harnessing the T cell response. Nat Rev Immunol 2012;12:269–81. 10.1038/nri3191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Fischbach MA, Bluestone JA, Lim WA. Cell-Based therapeutics: the next Pillar of medicine. Sci Transl Med 2013;5:ps177. 10.1126/scitranslmed.3005568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Monaco H, Yokomizo S, Choi HS, et al. Quickly evolving near‐infrared photoimmunotherapy provides multifaceted approach to modern cancer treatment. View 2022;3:20200110. 10.1002/VIW.20200110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Galli F, Varani M, Lauri C, et al. Immune cell labelling and tracking: implications for adoptive cell transfer therapies. EJNMMI Radiopharm Chem 2021;6:7. 10.1186/s41181-020-00116-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Hu F, Liu B. Organelle-specific bioprobes based on fluorogens with aggregation-induced emission (AIE) characteristics. Org Biomol Chem 2016;14:9931–44. 10.1039/c6ob01414c [DOI] [PubMed] [Google Scholar]
- 50. Lai J-Z, Zhu Y-Y, Ruan M, et al. Local irradiation sensitized tumors to adoptive T cell therapy via enhancing the Cross-Priming, homing, and cytotoxicity of antigen-specific CD8 T cells. Front Immunol 2019;10:2857. 10.3389/fimmu.2019.02857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Frye LD, Edidin M. The rapid intermixing of cell surface antigens after formation of mouse-human heterokaryons. J Cell Sci 1970;7:319–35. 10.1242/jcs.7.2.319 [DOI] [PubMed] [Google Scholar]
- 52. Park GK, Lee JH, Levitz A, et al. Lysosome-Targeted bioprobes for sequential cell tracking from macroscopic to microscopic scales. Adv Mater 2019;31:e1806216. 10.1002/adma.201806216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Kim SH, Park JH, Kwon JS, et al. NIR fluorescence for monitoring in vivo scaffold degradation along with stem cell tracking in bone tissue engineering. Biomaterials 2020;258:120267. 10.1016/j.biomaterials.2020.120267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Kim SH, Kwon JS, Cho JG, et al. Non-invasive in vivo monitoring of transplanted stem cells in 3D-bioprinted constructs using near-infrared fluorescent imaging. Bioeng Transl Med 2021;6:e10216. 10.1002/btm2.10216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Mellanby RJ, Scott JI, Mair I, et al. Tricarbocyanine N-triazoles: the scaffold-of-choice for long-term near-infrared imaging of immune cells in vivo. Chem Sci 2018;9:7261–70. 10.1039/c8sc00900g [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Fuller JE, Zugates GT, Ferreira LS, et al. Intracellular delivery of core-shell fluorescent silica nanoparticles. Biomaterials 2008;29:1526–32. 10.1016/j.biomaterials.2007.11.025 [DOI] [PubMed] [Google Scholar]
- 57. Huang Y, Guan Z, Dai X, et al. Engineered macrophages as near-infrared light activated drug vectors for chemo-photodynamic therapy of primary and bone metastatic breast cancer. Nat Commun 2021;12:4310. 10.1038/s41467-021-24564-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Heo MB, Lim YT. Programmed nanoparticles for combined immunomodulation, antigen presentation and tracking of immunotherapeutic cells. Biomaterials 2014;35:590–600. 10.1016/j.biomaterials.2013.10.009 [DOI] [PubMed] [Google Scholar]
- 59. Xiang J, Xu L, Gong H, et al. Antigen-Loaded upconversion nanoparticles for dendritic cell stimulation, tracking, and vaccination in dendritic cell-based immunotherapy. ACS Nano 2015;9:6401–11. 10.1021/acsnano.5b02014 [DOI] [PubMed] [Google Scholar]
- 60. Finco O, Rappuoli R. Designing vaccines for the twenty-first century society. Front Immunol 2014;5:12. 10.3389/fimmu.2014.00012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Ulmer JB, Valley U, Rappuoli R. Vaccine manufacturing: challenges and solutions. Nat Biotechnol 2006;24:1377–83. 10.1038/nbt1261 [DOI] [PubMed] [Google Scholar]
- 62. Paston SJ, Brentville VA, Symonds P, et al. Cancer vaccines, adjuvants, and delivery systems. Front Immunol 2021;12:627932. 10.3389/fimmu.2021.627932 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Saxena M, van der Burg SH, Melief CJM, et al. Therapeutic cancer vaccines. Nat Rev Cancer 2021;21:360–78. 10.1038/s41568-021-00346-0 [DOI] [PubMed] [Google Scholar]
- 64. Lindsay KE, Bhosle SM, Zurla C, et al. Visualization of early events in mRNA vaccine delivery in non-human primates via PET-CT and near-infrared imaging. Nat Biomed Eng 2019;3:371–80. 10.1038/s41551-019-0378-3 [DOI] [PubMed] [Google Scholar]
- 65. Cao FQ, Yan MM, Liu YJ, et al. Photosensitizer-induced self-assembly of antigens as nanovaccines for cancer immunotherapy. Biomater Sci 2018;6:473–7. 10.1039/c7bm01082f [DOI] [PubMed] [Google Scholar]
- 66. Katagiri W, Lee JH, Tétrault M-A, et al. Real-Time imaging of vaccine biodistribution using zwitterionic NIR nanoparticles. Adv Healthc Mater 2019;8:e1900035. 10.1002/adhm.201900035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Choi HS, Gibbs SL, Lee JH, et al. Targeted zwitterionic near-infrared fluorophores for improved optical imaging. Nat Biotechnol 2013;31:148–53. 10.1038/nbt.2468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Choi HS, Nasr K, Alyabyev S, et al. Synthesis and in vivo fate of zwitterionic near-infrared fluorophores. Angew Chem Int Ed Engl 2011;50:6258–63. 10.1002/anie.201102459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Hyun H, Henary M, Gao T, et al. 700-nm zwitterionic near-infrared fluorophores for Dual-Channel image-guided surgery. Mol Imaging Biol 2016;18:52–61. 10.1007/s11307-015-0870-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Kim H, Cho MH, Choi HS, et al. Zwitterionic near-infrared fluorophore-conjugated epidermal growth factor for fast, real-time, and target-cell-specific cancer imaging. Theranostics 2019;9:1085–95. 10.7150/thno.29719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Kim H, Choi HS, Eom JB, et al. Mini-Platform for Off-On near-infrared fluorescence imaging using Peptide-Targeting ligands. Bioconjug Chem 2020;31:721-728. 10.1021/acs.bioconjchem.9b00844 [DOI] [PubMed] [Google Scholar]
- 72. Kim KS, Kim YS, Bao K, et al. Bioimaging of botulinum toxin and hyaluronate hydrogels using zwitterionic near-infrared fluorophores. Biomater Res 2017;21:15. 10.1186/s40824-017-0102-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Brown CE, Mackall CL. CAR T cell therapy: inroads to response and resistance. Nat Rev Immunol 2019;19:73–4. 10.1038/s41577-018-0119-y [DOI] [PubMed] [Google Scholar]
- 74. Chatterjee S, Lesniak WG, Gabrielson M, et al. A humanized antibody for imaging immune checkpoint ligand PD-L1 expression in tumors. Oncotarget 2016;7:10215–27. 10.18632/oncotarget.7143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Conner KP, Rock BM, Kwon GK, et al. Evaluation of near infrared fluorescent labeling of monoclonal antibodies as a tool for tissue distribution. Drug Metab Dispos 2014;42:1906–13. 10.1124/dmd.114.060319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Kang N-Y, Park S-J, Ang XWE, et al. A macrophage uptaking near-infrared chemical probe CDnir7 for in vivo imaging of inflammation. Chem Commun 2014;50:6589–91. 10.1039/c4cc02038c [DOI] [PubMed] [Google Scholar]
- 77. Yoo JS, Lee S-C, Jow ZY, et al. A macrophage-specific fluorescent probe for intraoperative lymph node staging. Cancer Res 2014;74:44–55. 10.1158/0008-5472.CAN-13-2129 [DOI] [PubMed] [Google Scholar]
- 78. Park S-J, Kim B, Choi S, et al. Imaging inflammation using an activated macrophage probe with Slc18b1 as the activation-selective gating target. Nat Commun 2019;10:1111. 10.1038/s41467-019-08990-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Zhong Y, Ma Z, Wang F, et al. In vivo molecular imaging for immunotherapy using ultra-bright near-infrared-IIb rare-earth nanoparticles. Nat Biotechnol 2019;37:1322–31. 10.1038/s41587-019-0262-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Zhang C, Pu K. Molecular and nanoengineering approaches towards activatable cancer immunotherapy. Chem Soc Rev 2020;49:4234–53. 10.1039/c9cs00773c [DOI] [PubMed] [Google Scholar]
- 81. He S, Li J, Lyu Y, et al. Near-Infrared fluorescent macromolecular reporters for real-time imaging and urinalysis of cancer immunotherapy. J Am Chem Soc 2020;142:7075–82. 10.1021/jacs.0c00659 [DOI] [PubMed] [Google Scholar]
- 82. Fernandez A, Vermeren M, Humphries D, et al. Chemical Modulation of in Vivo Macrophage Function with Subpopulation-Specific Fluorescent Prodrug Conjugates. ACS Cent Sci 2017;3:995–1005. 10.1021/acscentsci.7b00262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Lee MH, Sharma A, Chang MJ, et al. Fluorogenic reaction-based prodrug conjugates as targeted cancer theranostics. Chem Soc Rev 2018;47:28–52. 10.1039/c7cs00557a [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Cui D, Li J, Zhao X, et al. Semiconducting polymer Nanoreporters for near-infrared chemiluminescence imaging of Immunoactivation. Adv Mater 2020;32:e1906314. 10.1002/adma.201906314 [DOI] [PubMed] [Google Scholar]
- 85. Kobayashi H, Furusawa A, Rosenberg A, et al. Near-infrared photoimmunotherapy of cancer: a new approach that kills cancer cells and enhances anti-cancer host immunity. Int Immunol 2021;33:7–15. 10.1093/intimm/dxaa037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Kwiatkowski S, Knap B, Przystupski D, et al. Photodynamic therapy - mechanisms, photosensitizers and combinations. Biomed Pharmacother 2018;106:1098–107. 10.1016/j.biopha.2018.07.049 [DOI] [PubMed] [Google Scholar]
- 87. Falk-Mahapatra R, Gollnick SO. Photodynamic therapy and immunity: an update. Photochem Photobiol 2020;96:550–9. 10.1111/php.13253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Chiou VL, Burotto M. Pseudoprogression and immune-related response in solid tumors. J Clin Oncol 2015;33:3541–3. 10.1200/JCO.2015.61.6870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Jia W, Gao Q, Han A, et al. The potential mechanism, recognition and clinical significance of tumor pseudoprogression after immunotherapy. Cancer Biol Med 2019;16:655–70. 10.20892/j.issn.2095-3941.2019.0144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Sanmamed MF, Perez-Gracia JL, Schalper KA, et al. Changes in serum interleukin-8 (IL-8) levels reflect and predict response to anti-PD-1 treatment in melanoma and non-small-cell lung cancer patients. Ann Oncol 2017;28:1988–95. 10.1093/annonc/mdx190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Balaña C, Capellades J, Pineda E, et al. Pseudoprogression as an adverse event of glioblastoma therapy. Cancer Med 2017;6:2858–66. 10.1002/cam4.1242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Kebir S, Rauschenbach L, Galldiks N, et al. Dynamic O-(2-[18F]fluoroethyl)-L-tyrosine PET imaging for the detection of checkpoint inhibitor-related pseudoprogression in melanoma brain metastases. Neuro Oncol 2016;18:1462–4. 10.1093/neuonc/now154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Costa LB, Queiroz MA, Barbosa FdeG, et al. Pseudoprogression on PSMA PET imaging of a mCRPC patient under anti-PD1 treatment. Eur J Nucl Med Mol Imaging 2019;46:1576–7. 10.1007/s00259-019-04328-1 [DOI] [PubMed] [Google Scholar]
- 94. Wang F, Wan H, Ma Z, et al. Light-sheet microscopy in the near-infrared II window. Nat Methods 2019;16:545–52. 10.1038/s41592-019-0398-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Wang F, Ma Z, Zhong Y, et al. In vivo NIR-II structured-illumination light-sheet microscopy. Proc Natl Acad Sci U S A 2021;118. 10.1073/pnas.2023888118. [Epub ahead of print: 09 02 2021]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. He S, Xie C, Jiang Y, et al. An organic Afterglow Protheranostic Nanoassembly. Adv Mater 2019;31:e1902672. 10.1002/adma.201902672 [DOI] [PubMed] [Google Scholar]
- 97. Miao Q, Xie C, Zhen X, et al. Molecular afterglow imaging with bright, biodegradable polymer nanoparticles. Nat Biotechnol 2017;35:1102–10. 10.1038/nbt.3987 [DOI] [PubMed] [Google Scholar]
- 98. Jiang Y, Huang J, Zhen X, et al. A generic approach towards afterglow luminescent nanoparticles for ultrasensitive in vivo imaging. Nat Commun 2019;10:2064. 10.1038/s41467-019-10119-x [DOI] [PMC free article] [PubMed] [Google Scholar]