Abstract
A gas chromatography coupled with tandem mass spectrometry (GC-MS/MS) method is developed to determine 18 representative polycyclic aromatic hydrocarbons (PAHs) in cosmetics, including Benzo[a]pyrene (BaP) and others. The method offers high sensitivity and selectivity under selected reaction monitoring (SRM) mode to satisfy the requirements of both quantitation and qualitation. The extraction solvent system used in this study is acetone/hexane 1:1 (v/v) and other purification procedure is unnecessary. The linearities of 18 PAHs are validated in different concentration in the range of 0.25–20 ng/mL individually with coefficient correlation (r) higher than 0.996. The recoveries for spiking 3 different concentrations are from 87.40% to 120.44% for 18 PAHs and the coefficient of variation (CV) are below 12.32%. Limit of quantification (LOQ) of 18 PAHs is in the range of 0.05–0.2 mg/kg. A matrix enhancement effect is observed and can be compensated with deuterated internal standard. The method has been successfully applied to 73 samples, over 40 of them are lipsticks. The results show none of the samples detect Benzo[a]pyrene (BaP) and Dibenzo[a,h]anthracene (DBA), both are classified as the most carcinogenic. 8 PAHs are detected and the average value between 0.08 and 0.27 mg/kg. This study offers a sensitive and simple method to analyze 18 representative PAHs successfully and can be applied to cosmetic products and raw materials.
Keywords: Cosmetics, Polycyclic aromatic hydrocarbons, GC-MS/MS
1. Introduction
Some cosmetic ingredients, including mineral oil, vaseline, petrolatum, ozokerite and others, have various applications and different consistencies and often use as antistatic agents, plasticizers, solvents and viscosity regulators. Most of them are lipophilic, by forming a water-repellant film and creating an effective barrier against the evaporation of the skin’s moisture, can be used as moisturizing agents [1]. However, these ingredients mainly come from petroleum distillation. If the refining procedure is under incomplete combustion, polycyclic aromatic hydrocarbons (PAHs) may enter final products through manufacturing process and spread out the safety of products further.
Polycyclic aromatic hydrocarbons (PAHs), a group of several hundred chemically related hydrophobic compounds, can be derived from natural or anthropogenic sources like forest fires, volcanoes eruption, man-made incomplete combustion of organic materials such as coal, wood and petroleum or automotive emissions and smoking. They are commonly detected in air, water, food and soils. They composed of at least two or more fused conjugated benzene rings, with a pair of carbon atoms shared between rings in their molecules [2–5]. In general, PAHs have low vapor pressure and tend to decrease with increasing molecular weight. Some of them are present at ambient temperature in air as gas or associated with particles [6]. In addition, these compounds are highly lipophilic and possess high solubility in organic solvents. Routes of exposure to PAHs include ingestion, inhalation and dermal contact. Short term exposure to PAHs cause harmful effects, such as skin irritation and inflammation [7]. These compounds receive more attention in recent years because their reactive metabolites, such as epoxides and dihydrodiols, can bind to cellular proteins and DNA to form an adduct and chelate at a critical site to the regulation of cell differentiation or growth, for example in cell cycle control and DNA repair. Therefore, some of them considered as carcinogens [8]. Some long-term studies have shown an increased risk of predominantly skin and lung, but also bladder and gastrointestinal cancers [9–11]. The International Agency for Research on Cancer (IARC) lists a number of PAHs, Benzo[a]pyrene (BaP), Dibenzo[a,h]anthracene (DBA), Benzo[a]anthracene (BaA), Chrysene (Chr), Benzo[b]fluoranthene (BbFL), Benzo[k]fluoranthene (BkFL), Indeno[1,2,3-cd] pyrene (InP) and Benzo[g,h,i]perylene (BghiP) as known, probably or possibly carcinogens [12]. The US EPA also listed 16 PAHs as priority pollutant and 7 of them are in consist with IARC, except BghiP, considered as probable human carcinogens [13]. Moreover, the Regulation (EU) No.1223/2009 listed BaA, BaP, BbFL, Benzo[e]pyrene (BeP), BkFL, Benzo[j]fluoranthene (BjFL), Chr and DBA are prohibited substances in cosmetic products and also set the content of BaP in raw material such as paraffin waxes and creosote oil should be less than 0.005% [14]. In addition, the Regulation (EU) No.1272/2013, amending Annex XVII to Regulation (EC) No.1907/2006 of the European parliament and of the council on the registration, evaluation, authorisation and restriction of chemicals (REACH), extending 8 PAHs restriction, which in accordance with the Regulation (EU) No.1223/2009, to rubber and plastic components that comes into direct as well as prolonged or short-term repetitive contact with the human skin or the oral cavity under normal or reasonably foreseeable conditions of use, demonstrating more official concern in PAHs [15]. In this study, we select 18 representative PAHs, structures and abbreviations shown in Fig. 1, are outlined as priority pollutant by US EPA, classified with more carcinogenicity by IARC and listed as prohibited substances in cosmetic regulation by EU, expecting to set up a analytical method for cosmetic analysis. However, many previous studies focus on soil sediments, water, ambient air and food [16–20], only few studies are investigated in cosmetics [21,22]. Common analytical techniques employed for PAHs is HPLC, may combined with UV, fluorescence or diode array detector (DAD) [23,24]. However, trace amount of PAHs identification is easily interfered by sample matrice and other components if only based on retention time. Gas chromatography combined single quadrupolemass spectrometry or triple quadrupole mass spectrometry also had been employed for PAH determination in recent years [25,26]. In view of the sensitivity and specificity offered by triple quadrupole is still better, we employed gas chromatograph/tandem mass spectrometer (GC-MS/MS) with SRM mode to quantify trace amount of PAHs from mineral-based cosmetic products.
Fig. 1.
Chemical structures of the 18 PAHs.
2. Materials and methods
2.1. Reagents and solutions
Analytical grade (99.9% pure) acetone and hexane are purchased from Merck Millipore (Massachusetts, USA). 18 PAH standard mixture solution (Acenaphthene (AcP), Acenaphthylene (AcPy), Anthracene (Ant), Benzo[a]anthracene (BaA), Benzo[a]pyrene (BaP), Benzo[e]pyrene (BeP), Benzo[b]fluoranthene (BbFL), Benzo[g,h,i]perylene (BghiP), Benzo[j]fluoranthene (BjFL), Benzo[k]fluoranthene (BkFL), Chrysene (Chr), Dibenzo[a,h]anthracene (DBA), Fluoranthene (FL), Fluorene (Flu), Indeno [1,2,3-cd]pyrene (InP), Naphthalene (NaP), Phenanthrene (Phe) and Pyrene (Pyr)) are obtained from Sigma–Aldrich (Missouri, USA). Powdered Benzo[e]pyrene-d12 (BeP-d12) perdeuterated internal standard is purchased from Sigma–Aldrich (Missouri, USA). 16 PAH internal standard mixture solution (1000 μg/mL in toluene) (AcP-d10, Acpy-d8, Ant-d10, BaA-d12, Bap-d12, BbFL-d12, BghiP-d12, BkFL-d12, Chr-d12, DBA-d14, Flu-d10, FL-d10, InP-d12, NaP-d8, Phe-d10, Pyr-d10) are from Chiron (Trondheim, Norway). Disposable Nylone filters (0.22 μm) are provided by Merck Millipore (Massachusetts, USA).
2.2. GC-MS/MS instrumentation and condition
The gas chromatography coupled with tandem mass spectrometry consisted of an Agilent 7890 GC system (Agilent Technologies, Santa Clara, CA, USA) connected to a triple quadrupole mass spectrometer 7000C (Agilent Technologies, Santa Clara, CA, USA). Data analysis is performed by Agilent Mass Hunter Quantitative Analysis software (Version: B.07.01/ Build 7.1.524.0). GC analysis is performed on a DB-EUPAH column (20 m length × 0.18 mm i.d., 0.14 μm film thickness). The column temperature is initially held at 50 °C for 2min, raised to 150°C at the rate of 25 °C/min and held 3 min, then to 165 °C at the rate of 5 °C/min and held 3 min, following to 175 °C at the rate of 10 °C/min and held 5 min, then to 225 °C at the rate of 25 °C/min and held 5 min, following to 265 °C at the rate of 20 °C/min held 10 min, and to 300 °C at the rate of 5 °C/min, finally to 320 °C at the rate of 10 °C/min, held at final temperature for 5 min. Helium gas is used as carrier gas with the constant flow rate of 1.0mL/min. Total running time is 60 min. The injection volume is 2 uL in a splitless mode. The mass spectrometer is operated in electron impact ionization mode with electron energy of 70 eV. Temperatures of ion source, injector and transfer line temperature are set at 340 °C, 340 °C and 280 °C respectively. A solvent delay fixed at 5 min and the analytes are separated into 10 discrete time segments for SRM monitoring with dwell time of 5 ms. (4–7 min for NaP, 7–10.25 min for AcPy and AcP, 10.25–14 min for Flu, 14–20 min for Phe and Ant, 20–27 min for FL and Pyr, 27–32.5 min for BaA and Chr, 32.5–36 min for BbFL, BkFL and BjFL, 36–40 min for BeP and BaP, 40–46 min for InP and DBA, 46–60 min for BghiP). The gain factor of detector setting is fixed at 10. Table 1 shows the retention time and analyte-dependent ions and collision energy used for analysis.
Table 1.
GC–MS/MS parameters for individual PAHs and perdeuterated internal standard.
| PAH | Retention time (min) | Precursor ion (m/z) | Collision energy (eV) | Product ion (m/z) |
|---|---|---|---|---|
| NaP | 5.98 | 128 | 20 | 102 |
| 128 | 25 | 78 | ||
| NaP-d8 | 5.96 | 136 | 15 | 108 |
| 134 | 15 | 82 | ||
| AcPy | 8.97 | 152 | 20 | 151 |
| 152 | 35 | 150 | ||
| AcPy-D8 | 8.95 | 160 | 20 | 158 |
| 158 | 15 | 156 | ||
| AcP | 9.42 | 154 | 20 | 153 |
| 153 | 20 | 152 | ||
| AcP-d10 | 9.29 | 164 | 15 | 162 |
| 162 | 20 | 160 | ||
| Flu | 11.22 | 166 | 20 | 165 |
| 165 | 25 | 164 | ||
| Flu-d10 | 11.01 | 176 | 15 | 174 |
| 174 | 15 | 172 | ||
| Phe | 16.74 | 178 | 35 | 176 |
| 178 | 25 | 152 | ||
| Phe-d10 | 16.59 | 188 | 25 | 160 |
| 94 | 10 | 80 | ||
| Ant | 16.94 | 178 | 35 | 176 |
| 178 | 25 | 152 | ||
| Ant-d10 | 16.81 | 188 | 25 | 160 |
| 94 | 10 | 80 | ||
| FL | 23.88 | 202 | 25 | 201 |
| 202 | 40 | 200 | ||
| FL-d10 | 23.81 | 212 | 40 | 208 |
| 212 | 25 | 210 | ||
| Pyr | 24.87 | 202 | 25 | 201 |
| 202 | 40 | 200 | ||
| Pyr-d10 | 24.78 | 212 | 40 | 208 |
| 212 | 25 | 210 | ||
| BaA | 30.31 | 228 | 35 | 226 |
| 113 | 15 | 112 | ||
| BaA-d12 | 30.23 | 240 | 35 | 236 |
| 240 | 20 | 238 | ||
| Chr | 30.55 | 228 | 20 | 227 |
| 228 | 35 | 226 | ||
| Chr-d12 | 30.46 | 240 | 20 | 236 |
| 240 | 35 | 238 | ||
| BbFL | 34.79 | 252 | 40 | 250 |
| 126 | 15 | 113 | ||
| BbFL-d12 | 34.65 | 264 | 35 | 260 |
| 132 | 10 | 118 | ||
| BkFL | 34.93 | 252 | 40 | 250 |
| 126 | 15 | 113 | ||
| BkFL-d12 | 34.80 | 264 | 35 | 260 |
| 132 | 10 | 118 | ||
| BjFL | 35.08 | 252 | 40 | 250 |
| 125 | 15 | 124 | ||
| aBbFL-d12 | 34.65 | 264 | 35 | 260 |
| 132 | 10 | 118 | ||
| BeP | 36.90 | 252 | 45 | 250 |
| 250 | 45 | 248 | ||
| BeP-12 | 36.70 | 264 | 50 | 260 |
| 132 | 10 | 118 | ||
| BaP | 37.24 | 252 | 40 | 250 |
| 126 | 15 | 113 | ||
| BaP-d12 | 37.06 | 264 | 40 | 260 |
| 263 | 40 | 259 | ||
| InP | 44.93 | 276 | 50 | 274 |
| 138 | 20 | 137 | ||
| InP-d12 | 44.77 | 144 | 15 | 142 |
| 144 | 10 | 130 | ||
| DBA | 45.13 | 278 | 45 | 276 |
| 139 | 20 | 138 | ||
| DBA-d14 | 44.93 | 292 | 30 | 288 |
| 146 | 10 | 144 | ||
| BghiP | 46.61 | 276 | 50 | 274 |
| 138 | 20 | 137 | ||
| BghiP-d12 | 46.45 | 144 | 15 | 142 |
| 144 | 10 | 130 |
BbFL-d12 was also used as internal standard for BjFL.
2.3. PAH standards, internal standards solution preparation
A stock solution of individual PAHs is prepared by dissolving each PAH compound 50 mg in 50 mL acetone/hexane 1:1(v/v) (1000 μg/mL). The stock solution is further diluted to obtain a working solution at 10 μg/mL 1 mL 16 PAHs internal standard mixture solution (1000 μg/mL) and 5 mg BeP-d12 are also dissolved by the same solvent system at 10 mL and 50 mL respectively, both with concentration at 100 μg/mL. The internal standard mixture solution is further diluted to 200 ng/ mL and are stored in the dark at 4 °C.
2.4. Sample preparation
Totally 73 samples are collected randomly as testing samples. 9 of them are raw materials (Mineral oil, paraffin, petrolatum and Wax) and over 40 of the mare lipsticks. Each sample weighed 1 g accurately, treated with 200 ng/mL internal standard mixture solution for 1 mL and added acetone/hexane 1:1(v/v) 10 mL. After ultrasonication for 30 min, make up to 20 mL. Afterwards, additional 10 times dilution procedure done by the same extraction solvent and made final solution with internal standard at final concentration at 1 ng/mL. The final solution is filtered by a 0.22 μm nylon filter and transferred into a vial.
Sample preparation for recovery test is similar. 18 PAHs standard solution in 3 different concentrations in the range of 0.05–4 mg/kg are spiked individually into 1 g homogenized blank sample along with 1 mL of 200 ng/mL internal standard mixture solution respectively. Through similar dilution procedure described above, obtaining test solutions with 1 ng/mL internal standards to determine extraction recovery.
2.5. Method validation and matrix effect
Validation of this method is performed by assessment of the linearity, precision, accuracy, and limit of quantification (LOQ). The linearity of the method is evaluated by performing duplicate analysis in the range of 0.25–20 ng/mL of 18 PAHs standard solutions containing an internal standard mixture (1 ng/mL). The calibration curve employed by calculating integrated peak area ratio of the target ion to respective internal standard, except BjFL referred to BbFL internal standard, and is considered acceptable when the correlation coefficient (r) is > 0.995. Precision is assessed by analyzing 3 repetitions of the same standard solutions above on the same day (n = 3) and over three consecutive days (n = 9). Accuracy is assessed by recovery studies and the extraction procedure already described above. An accuracy between 80% and 120% recovery from nominal concentration considered to be acceptable. The limit of quantitation (LOQ) of the method is determined by the signal measured at the lowest point of the calibration curve, which produced a peak area 10 times greater than noise in repetition. Matrix effects are determined by comparing solvent-only and matrix-matched calibration curves in terms of slope ratios = 100% × (1 – slopesolvent/slopematrix) [27].
3. Results and discussion
3.1. Method development
In view of cosmetic ingredients are complicated and with high proportion of oil-based substances, we need to select a simple and easily prepared solvent system to obtain optimal extraction effect. By referring the study of Haleyura N., employing acetone/hexane 1:1(v/v) in the extraction of PAHs in aged and contaminated soil [28], the solvent system is also applied and selected for our study. In addition, in order to minimize matrix interference and maintain sufficient sensitivity and resolution simultaneously, we employ perdeuterated internal standard and additional 10 times dilution in sample preparation procedure, which make peak identification much more clearly and reduce peak tailing, optimizing the effectiveness of quantification. A comparision of SRM chromatogram between with and without further 10 times dilution for BaP with BaP-d12 are shown in Fig. 2 respectively and other PAHs in the study have similar tendency. (data not shown).
Fig. 2.
MRM chromatogram of Benzo[a]pyrene (BaP) and Benzo[a]pyrene-d12 (BaP-d12) (both shown as green peak) (A) without further dilution (B) with further dilution 10x.
3.2. GC-MS/MS determination
The temperature gradient is optimized with the purpose of obtain the highest resolution between peaks. The procedure is performed in full scan mode by a 10 ng/mL 18 PAHs standard mixture. According to the European guidelines EC/657/2002 [29], each analyte earn at least 4 identification points (IPs) in the study using GC-MS/MS technique. The MS/MS acquisition parameters of each PAHs standards and internal standards are done through SRM mode with a 1 ng/mL of 18 PAH standard solution containing same concentration of internal standard. The developed mass spectrometric condition in this study conformed to the EU confirmation criteria. The chromatogram of 18 PAHs in 1 ng/mL are shown in Fig. 3.
Fig. 3.
Total ion chromatogram of standard solution mixture of 18 PAHs. NaP=Naphthalene; AcPy = Acenaphthylene; AcP=Acenaphthene; Flu = Fluorene; Phe = Phenanthrene; Ant = Anthracene; FL= Fluoranthene; Pyr = Pyrene; BaA= Benzo [a]anthracene; Chr = Chrysene; BbFL = Benzo[b]fluoranthene; BkFL=Benzo[k]fluoranthene; BjFL = Benzo[j]fluoranthene; BeP= Benzo[e]pyrene; BaP= Benzo[a]pyrene; InP= Indeno[1,2,3-cd]pyrene; DBA = Dibenzo[a,h]anthracene; BghiP = Benzo [g,h,i]perylene.
3.3. Method validation and matrix effect
3.3.1. Linearity
The linearity of 18 PAHs standard curves are evaluated at the concentration range of 0.25–20 ng/mL (0.25, 0.5, 1, 2.5, 5, 10 and 20 ng/mL) with internal standard at 1 ng/mL in duplicates. Good linearities are achieved at the concentrations of 1–20 ng/ mL for Indeno [1,2,3-cd]pyrene, 0.5–10 ng/mL for Anthracene (Ant), Benzo[b]fluoranthene (BbFL), Benzo[g,h,i]perylene (BghiP), Benzo[j]fluoranthene (BjFL), Benzo[k]fluoranthene (BkFL), Dibenzo[a,h]anthracene (DBA), Fluorene (Flu) and Naphthalene (NaP), and 0.25–10 ng/mL for other 9 PAHs in the study. All r value of regression in individual compound are higher than 0.996.
3.3.2. Precision
Based on each PAH compound’s linearity range determined above, different concentrations of PAHs standard solutions are measured and calculated for the interday (5 times each day and a successive three-day) and intraday (5 times on the same day) accuracies. The coefficient of variation (CV) of interday and intraday analysis is ranged from 0.89% to 12.37% and 0.38%–11.90%, respectively. These values reflect the good precision. The linear range, regression coefficient (r) and the coefficient of variation (CV) of interday and intraday analysis of each PAH compound are shown in Table 2.
Table 2.
Linear range, correlation coefficient (r) and coefficient of variation (CV) of Intraday and interday analysis of 18 PAHs.
| PAH | Linear Range (ng/mL) | r | CV (%) | |
|---|---|---|---|---|
|
| ||||
| Intradaya | Interdayb | |||
| Naphthaleneb | 0.50–10 | 0.999 | 1.03–3.69 | 1.23–4.78 |
| Acenaphthylene | 0.25–10 | 1.000 | 1.25–3.72 | 1.43–4.79 |
| Acenaphthene | 0.25–10 | 1.000 | 0.59–2.75 | 1.20–5.38 |
| Fluorene | 0.50–10 | 1.000 | 0.44–1.97 | 1.15–2.65 |
| Phenanthrene | 0.25–10 | 1.000 | 0.41–3.31 | 0.89–6.85 |
| Anthracene | 0.50–10 | 0.999 | 0.50–1.52 | 1.21–6.07 |
| Fluoranthene | 0.25–10 | 1.000 | 0.51–2.49 | 0.92–3.40 |
| Pyrene | 0.25–10 | 1.000 | 0.43–3.04 | 1.03–5.29 |
| Benzo[a]anthracene | 0.25–10 | 1.000 | 0.88–1.84 | 0.98–2.71 |
| Chrysene | 0.25–10 | 1.000 | 0.42–2.54 | 1.12–4.90 |
| Benzo[b]fluoranthene | 0.50–10 | 1.000 | 0.83–1.81 | 1.32–2.84 |
| Benzo[k]fluoranthene | 0.50–10 | 0.999 | 2.73–7.49 | 3.46–12.37 |
| Benzo[j]fluoranthene | 0.50–10 | 0.999 | 0.38–3.43 | 2.05–8.80 |
| Benzo[e]pyrene | 0.25–10 | 1.000 | 0.48–1.53 | 1.38–9.07 |
| Benzo[a]pyrene | 0.25–10 | 0.999 | 1.95–4.55 | 3.13–7.14 |
| Indeno[1,2,3-cd]pyrene | 1–20 | 0.997 | 3.67–7.17 | 3.46–6.80 |
| Dibenzo(a,h)anthracene | 0.50–10 | 0.997 | 3.64–11.90 | 4.08–12.69 |
| Benzo(g,h,i)perylene | 0.50–10 | 0.999 | 1.50–5.0a | 4.82–8.28 |
CV = coefficient of variation.
n = 3, Repeat 5 times on the same day.
n = 9, Repeat 5 times each day and a successive three-day.
3.3.3. Recoveries
To determine the recoveries of the developed method, standard solutions in 3 different concentrations (0.2, 1, and 4 mg/ kg for InP, 0.1, 0.5 and 2 mg/kg for Ant, BbFL, BghiP, BjFL, BkFL, DBA, Flu, NaP and 0.05, 0.2, and 2 mg/kg for others) with internal standard at 0.2 mg/kg are spiked individually into blank samples in triplicate. The data showed the recoveries range from 87.4 to 120.4% as shown in Table 3. The coefficient of variation (CV) are below 12.32%. In general, the recovery results are satisfactory and indicates the procedure of extraction and dilution is acceptable.
Table 3.
Recoveries (%) and coefficient of variation (CV) of 18 PAHs.
| PAH | Spiking level (ug/g) | Recovery (%) | CV (%) |
|---|---|---|---|
| NaP | 0.10 | 101.36 | 9.62 |
| 0.50 | 102.89 | 1.63 | |
| 2.00 | 92.42 | 2.13 | |
| AcPy | 0.05 | 96.73 | 9.54 |
| 0.20 | 98.76 | 1.57 | |
| 2.00 | 95.06 | 1.93 | |
| AcP | 0.05 | 98.9 | 4.22 |
| 0.20 | 101.58 | 1.19 | |
| 2.00 | 100.63 | 1.00 | |
| Flu | 0.10 | 117.92 | 2.05 |
| 0.20 | 109.61 | 0.99 | |
| 2.00 | 98.72 | 1.10 | |
| Phe | 0.05 | 93.47 | 7.39 |
| 0.20 | 103.17 | 3.06 | |
| 2.00 | 99.39 | 0.95 | |
| Ant | 0.10 | 115.72 | 3.00 |
| 0.20 | 109.23 | 2.12 | |
| 2.00 | 96.32 | 2.73 | |
| FL | 0.05 | 92.82 | 2.83 |
| 0.20 | 101.53 | 3.16 | |
| 2.00 | 97.52 | 0.65 | |
| Pyr | 0.05 | 119.88 | 4.08 |
| 0.20 | 105.75 | 2.95 | |
| 2.00 | 95.42 | 1.01 | |
| BaA | 0.05 | 110.18 | 2.25 |
| 0.20 | 104.88 | 1.64 | |
| 2.00 | 95.96 | 0.78 | |
| Chr | 0.05 | 106.50 | 1.64 |
| 0.20 | 103.94 | 3.09 | |
| 2.00 | 98.47 | 1.45 | |
| BbFL | 0.10 | 106.85 | 1.19 |
| 0.50 | 100.56 | 1.73 | |
| 2.00 | 96.59 | 1.49 | |
| BkFL | 0.10 | 104.15 | 7.39 |
| 0.50 | 102.20 | 6.58 | |
| 2.00 | 96.30 | 1.60 | |
| BjFL | 0.10 | 116.37 | 1.84 |
| 0.50 | 100.23 | 3.40 | |
| 2.00 | 94.38 | 1.72 | |
| BeP | 0.05 | 115.51 | 1.60 |
| 0.20 | 103.70 | 2.16 | |
| 2.00 | 95.68 | 1.18 | |
| BaA | 0.05 | 113.20 | 3.23 |
| 0.20 | 106.98 | 1.77 | |
| 2.00 | 99.18 | 0.67 | |
| InP | 0.20 | 94.72 | 12.32 |
| 1.00 | 93.20 | 6.14 | |
| 4.00 | 87.40 | 5.83 | |
| DBA | 0.10 | 114.63 | 2.45 |
| 0.50 | 97.76 | 2.39 | |
| 2.00 | 89.07 | 2.66 | |
| BghiP | 0.10 | 120.44 | 2.03 |
| 0.50 | 99.18 | 3.86 | |
| 2.00 | 89.37 | 3.13 |
n = 3.
3.3.4. Matrix effect
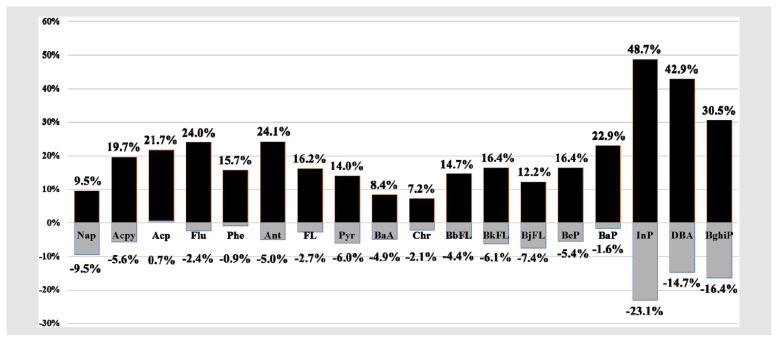
1. Matrix effect results list in Table 4, which is calculated as the difference between the slope of the matrix-matched and solvent-only calibration curves divided by the slope of matrix-matched calibration curve [27]. The results present a significant signal increase in varying degrees of all PAHs in the study, suggesting there is a matrix enhancement effect in PAH analysis by gas chromatography tandem mass spectrometry in cosmetics. Matrix effect is a phenomenon commonly encountered in gas chromatography and several approaches have been proposed to eliminate the enhancement effect, including additional clean-up procedure to remove matrix components, different injector types and matrix-matched standardizations [30]. In this study, by adding final concentration 1 ng/mL internal standard mixture solution, a correction effect has observed, showing a enhancement or suppression under 10% in most PAH compounds, only BghiP, DBA and InP presenting a signal suppression between 10 and 20%, which showing a dramatically (from 30 to 50%) enhancement without internal standard correction. This observation suggests internal standard could increase the accuracy of quantification in this method.
Table 4.
Matrix effects (%) correction by internal standard. *Internal standard mixture solution concentration: 200 ng/mL, *Matrix effect (%) of 18 PAH without internal standard correction in black bars, and correction effect shown in gray bars.
3.3.5. Limit of quantitation
Limit of quantitation (LOQ) in the study is determined by the signal measured at the lowest point of the calibration curve, which produce a peak area 10 times greater than noise in repetition. The LOQs of 18 PAHs range from 0.05 to 0.2 mg/kg (shown in Table 5).
Table 5.
Limit of quantitation (LOQ) of 18 PAHs.
| PAH | LOQ (mg/kg) |
|---|---|
| Acenaphthene (AcP) | 0.05 |
| Acenaphthylene (ACPy) | 0.05 |
| Benzo[a]anthracene (BaA) | 0.05 |
| Benzo[a]pyrene (BaP) | 0.05 |
| Benzo[e]pyrene (BeP) | 0.05 |
| Chrysene (Chr) | 0.05 |
| Fluoranthene (FL) | 0.05 |
| Phenanthrene (Phe) | 0.05 |
| Pyrene (Pyr) | 0.05 |
| Anthracene (Ant) | 0.1 |
| Benzo[b]fluoranthene (BbFL) | 0.1 |
| Benzo[g,h,i]perylene (BghiP) | 0.1 |
| Benzo[k]fluoranthene (BkFL) | 0.1 |
| Benzo[j]fluoranthene (BjFL) | 0.1 |
| Dibenzo[a,h]anthracene (DBA) | 0.1 |
| Fluorene (Flu) | 0.1 |
| Naphthalene (NaP) | 0.1 |
| Indeno[1,2,3-cd]pyrene (InP) | 0.2 |
3.4. Application to real samples
The method in this study is applied to 64 commercial cosmetic products with ingredients from petroleum refining and 9 raw materials can be used in cosmetic manufacturing, totally 73 samples. In view of lip cosmetics are considered as most probable to enter human body by dietary and generally considered containing a relatively high proportion of mineral oil or petrolatum, more than half (41 samples) are lipsticks and we select mineral oil, wax, paraffin and petrolatum as raw materials in our study. The results shows none of samples detect BaP and DBA, both are considered with more carcinogenicity. 8 PAHs in trace amount are detected shown in Table 6. The average value range from 0.08 to 0.27 mg/kg. There are 14 lipsticks detect PAHs, one of them have the highest PAH value of our study, with 1.3 mg/kg of BjFL. The data shows 5 of them detect with multiple PAHs and are list in Table 7. When it comes to raw materials, we find a petrolatum detects BghiP at 0.4 mg/kg and Flu at 0.08 mg/kg, another petrolatum detects trace amount of NaP at 0.1 mg/kg. Others detect none of the representative PAHs in this study. Currently, there is no limit value for PAHs in cosmetic products. If taking limit value set primarily in consumer products in REACH annex XVII amended in 2013, with total amount not extending 0.5 mg/kg of 8 PAHs (BaA, BaP, BbFL, BeP, BkFL, BjFL, Chr and DBA) as a reference [15], almost all samples in the study can comply with it.
Table 6.
Quantitation result in cosmetics samples.
| PAH | Samples detected | Detection Rate (%) | Concentration detected or range (mg/kg) | Average (mg/kg) |
|---|---|---|---|---|
| Phe | 11 | 15.1 | 0.07–0.4 | 0.14 |
| NaP | 10 | 13.7 | 0.1–1.17 | 0.3 |
| AcPy | 6 | 8.2 | 0.07–0.94 | 0.27 |
| Pyr | 3 | 4.1 | 0.08, 0.17, 0.46 | 0.24 |
| Chr | 2 | 2.7 | 0.08, 0.13 | 0.11 |
| FL | 2 | 2.7 | 0.06, 0.09 | 0.08 |
| BghiP | 1 | 1.4 | 0.41 | – |
| BjFL | 1 | 1.4 | 1.31 | – |
Table 7.
Quantitation result in samples with multiple PAHs.
| NaP (mg/kg) | Phe (mg/kg) | Chr (mg/kg) | FL (mg/ kg) | AcPy (mg/kg) | Pyr (mg/kg) | |
|---|---|---|---|---|---|---|
| Sample A | 0.13 | – | 0.08 | – | – | – |
| Sample B | 0.23 | 0.12 | – | 0.09 | – | – |
| Sample C | – | – | – | – | 0.08 | 0.46 |
| Sample D | 0.48 | 0.09 | 0.13 | – | – | – |
| Sample E | – | 0.09 | – | 0.06 | – | – |
4. Conclusions
Our method offer a sensitive way to analyze 18 representative PAHs by GC–MS/MS in cosmetics. The sample preparation procedure is simple and provides good recovery and precision. Moreover, the method can detect all target compounds under SRM mode in a single run with high sensitivity and selectivity. In summary, the GC-MS/MS method can be applied in routine PAHs analysis and provide a reference for laboratories and manufactures.
References
- 1. Petry T, Bury D, Fautz R, Hauser M, Huber B, Markowetz A, et al. Review of data on the dermal penetration of mineral oils and waxes used in cosmetic applications. Toxicol Lett. 2017;280:70–8. doi: 10.1016/j.toxlet.2017.07.899. [DOI] [PubMed] [Google Scholar]
- 2.United States Geological Survey (USGS) Methods of analysis by the U.S. Geological Survey National Water Quality Laboratory: determination of polycyclic aromatic hydrocarbon compounds in sediment by gas chromatography/mass spectrometry. Water-Resources Investigations Report 2003-4318. [Accessed 27 July 2018]. https://pubs.er.usgs.gov/publication/wri034318 .
- 3. Abdel-Shafy HI, Mansour MSM. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt J Pet. 2016;25:107–23. [Google Scholar]
- 4. Srogi K. Monitoring of environmental exposure to polycyclic aromatic hydrocarbons: a review. Environ Chem Lett. 2007;5:169–95. doi: 10.1007/s10311-007-0095-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hyötyläinen T, Oikari A. Bioaccumulation of PAHs from creosote-contaminated sediment in a laboratory-exposed freshwater oligochaete, Lumbriculus variegatus. Chemosphere. 2004;57:159–64. doi: 10.1016/j.chemosphere.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 6. Sakai R, Siegmann HC, Sato H, Voorhees AS. Particulate matter and particle-attached polycyclic aromatic hydrocarbons in the indoor and outdoor air of Tokyo measured with personal monitors. Environ Res. 2002;89:66–71. doi: 10.1006/enrs.2002.4355. [DOI] [PubMed] [Google Scholar]
- 7. Rengarajan T, Rajendran P, Nandakumar N, Lokeshkumar B, Rajendran P, Nishigaki I. Exposure to polycyclic aromatic hydrocarbons with special focus on cancer. Asian Pac J Trop Biomed. 2015;5(3):182–9. [Google Scholar]
- 8. Ramesh A, Archibong AE, Niaz MS. Ovarian susceptibility to benzo[a]pyrene: tissue burden of metabolites and DNA adducts in F-344 rats. J Toxicol Environ Health. 2010;73(23):1611–25. doi: 10.1080/15287394.2010.514225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lippman SM, Hawk ET. Cancer prevention: from 1727 to milestones of the past 100 years. Cancer Res. 2009;69(13):5269–84. doi: 10.1158/0008-5472.CAN-09-1750. [DOI] [PubMed] [Google Scholar]
- 10. Moorthy B, Chu C, Carlin DJ. Polycyclic aromatic hydrocarbons: from metabolism to lung cancer. Toxicol Sci. 2015;145(1):5–15. doi: 10.1093/toxsci/kfv040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Denissenko MF, Pao A, Tang MS, Pfeifer GP. Preferential formation of benzo[a]pyrene adducts at lung cancer mutational hotspots in P53. Science. 1996;274:430–2. doi: 10.1126/science.274.5286.430. [DOI] [PubMed] [Google Scholar]
- 12.International Agency for Research on Cancer (IARC) IARC monographs on the evaluation of carcinogenic risks to humans. [Accessed 27 July 2018]. https://monographs.iarc.fr/agents-classified-bythe-iarc/
- 13.The United States Environmental Protection Agency. Priority Pollutant List. [Accessed 27 July 2018]. https://www.epa.gov/sites/production/files/2015-09/documents/priority-pollutant-list-epa.pdf .
- 14.Official Journal of the European Union. Regulation (EC) No 1223/2009 of the European parliament and of the council of 30 November 2009 on cosmetic products. 2009. [Accessed 27 July 2018]. https://eurlex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32009R1223 .
- 15.Official Journal of the European Union. Commission regulation (EU) No 1272/2013 of 6 December 2013 amending annex XVII to regulation (EC) No 1907/2006 of the European parliament and of the council on the registration, evaluation, authorisation and restriction of chemicals (REACH) as regards polycyclic aromatic hydrocarbons. 2013. [Accessed 27 July 2018]. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32013R1272 .
- 16. Dong CD, Chen CF, Chen CW. Determination of polycyclic aromatic hydrocarbons in industrial harbor sediments by GC-MS. Int J Environ Res Public Health. 2012;9(6):2175–88. doi: 10.3390/ijerph9062175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Song YF, Jing X, Fleischmann S, Wilke BM. Comparative study of extraction methods for the determination of PAHs from contaminated soils and sediments. Chemosphere. 2002;48(9):993–1001. doi: 10.1016/s0045-6535(02)00180-7. [DOI] [PubMed] [Google Scholar]
- 18. Šimko P. Determination of polycyclic aromatic hydrocarbons in smoked meat products and smoke flavouring food additives. J Chromatogr B. 2002;770(1–2):3–18. doi: 10.1016/s0378-4347(01)00438-8. [DOI] [PubMed] [Google Scholar]
- 19. Liu Y, Zhu L, Shen X. Polycyclic aromatic hydrocarbons (PAHs) in indoor and outdoor air of hangzhou. Environ Sci Technol. 2001;35(5):840–4. doi: 10.1021/es001354t. [DOI] [PubMed] [Google Scholar]
- 20. Garcıa-Falcon MS, Cancho-Grande B, Simal-Gándara J. Stirring bar sorptive extraction in the determination of PAHs in drinking waters. Water Res. 2004;38(7):1679–84. doi: 10.1016/j.watres.2003.12.034. [DOI] [PubMed] [Google Scholar]
- 21. Monarca S. Polycyclic aromatic hydrocarbons in petroleum products for medicinal and cosmetic uses — analytical procedure. Sci Total Environ. 1980;14(3):233–43. doi: 10.1016/0048-9697(80)90025-x. [DOI] [PubMed] [Google Scholar]
- 22. Lim HH, Shin HS. Identification and quantification of phthalates, PAHs, amines, phenols, and metals in tattoo. Bull Korean Chem Soc. 2015;36:2039–50. [Google Scholar]
- 23.The United States Environmental Protection Agency. Method 1654 revision A: PAH content of oil by HPLC/UV. 1992. [Accessed 27 July 2018]. https://www.epa.gov/sites/production/files/2015-10/documents/method_1654a_1992.pdf .
- 24. Janoszka B. HPLC-fluorescence analysis of polycyclic aromatic hydrocarbons (PAHs) in pork meat and its gravy fried without additives and in the presence of onion and garlic. Food Chem. 2011;126(3):1344–53. [Google Scholar]
- 25. Katerina M, Sorenson WR, Jana H. Determination of polycyclic aromatic hydrocarbons (PAHs) in seafood using gas chromatography-mass spectrometry: collaborative study. J AOAC Int. 2015;98:477–505. doi: 10.5740/jaoacint.15-032. [DOI] [PubMed] [Google Scholar]
- 26. Shang D, Kim M, Haberl M. Rapid and sensitive method for the determination of polycyclic aromatic hydrocarbons in soils using pseudo multiple reaction monitoring gas chromatography/tandem mass spectrometry. J Chromatogr A. 2014;1334:118–25. doi: 10.1016/j.chroma.2014.01.074. [DOI] [PubMed] [Google Scholar]
- 27. Walorczyk S. Validation and use of a QuEChERS-based gas chromatographicetandem mass spectrometric method for multiresidue pesticide analysis in blackcurrants including studies of matrix effects and estimation of measurement uncertainty. Talanta. 2014;120:106–13. doi: 10.1016/j.talanta.2013.11.087. [DOI] [PubMed] [Google Scholar]
- 28. Haleyura N, Shahsavaria E, Mansura AA, Koshlafa E, Morrison PD, Osborn AM, et al. Comparison of rapid solvent extraction systems for the GC–MS/MS characterization of polycyclic aromatic hydrocarbons in aged, contaminated soil. MethodsX. 2016;3:364–70. doi: 10.1016/j.mex.2016.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Official Journal of the European Communities. 2002/657/EC: commission Decision of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. 2002. [Accessed 27 July 2018]. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32002D0657 .
- 30. Kwon H, Lehotay SJ, Geis-Asteggiante L. Variability of matrix effects in liquid and gas chromatography–mass spectrometry analysis of pesticide residues after QuEChERS sample preparation of different food crops. J Chromatogr A. 2012;1270:235–45. doi: 10.1016/j.chroma.2012.10.059. [DOI] [PubMed] [Google Scholar]