Conventional models for drug screening
The increasing cost of drug safety is attributed to the inappropriate pre-clinical models with unrepresentative pharmacokinetic and pharmacodynamic profiles that blur drug efficacy and toxicity. Moreover, the commercial drugs designed by impromper disease models could cause potential multi-organ side effects. Even worse, in some cases, unqualified drug screening is the cause of unexpected hospitalization in clinical. These urgent yet unsolved issues push scholars to reform monolayer cell culture and traditional animal models for drug validation.1 Then, in vitro three-dimensional (3D) cell culture models followed, but these are mainly limited to cell capsulation in defined hydrogels, spheroid systems, and engineered scaffolds. Not surprisingly, the 3D cell aggregates are still far from real organs, invalidating the drug kinetics.
Challenging unsolved scientific problems leads to the progress of science. As a 3D model formed by stem cells self-assembling into mini-organs, organoids are the most in vivo-like models ever reported. Thus far, various tissue organoids have been engineered to recapitulate human organs’ biological behavior and histopathologic properties. Despite providing valuable information for exploring disease development, the spatiotemporally separated organs are cultured in a static phase, which fails to simulate the dynamic physiology of the organs. For instance, the skin-barrier function, lung respiratory mechanics, muscle contraction, and kidney perfusion could severely affect the diffusion and transportation kinetics of the drugs.2 However, all organs are independently involved in drug screening and developmental principles without “organ–organ interaction” by body fluids or blood, which is vital for tissue functions.
Microfluidic organ-on-a-chip models
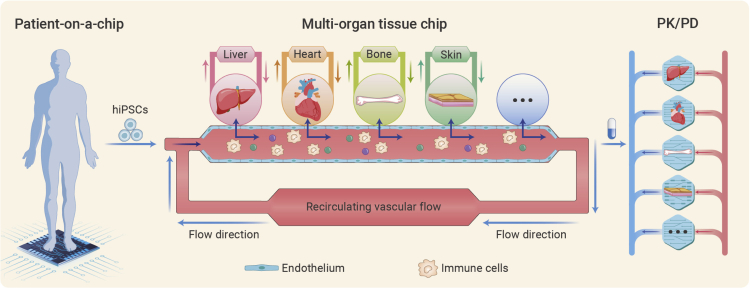
The emerging field of tissue engineering microfluidics has brought about the development of organs on a chip (OOC) or organ chips. Organ chips have emerged with the elusive goal of growing different organs on a chip in vitro for human physiology studies, such as drug screening in the pharmaceutical field and genetic assessment for disease development.3 Different from static organoid cultures, the orgam on a chip permits body-fluid perfusion, facilitating dynamic nutrient exchange and physiochemical signal transmission (shear stress, mechanical forces, bioelectrical stimulation), which better simulates the physiological conditions in living tissues. However, the organ phenotype during its maturation can be affected not only by the microenvironmental factors but also by local stromal or circulating immune cells, metabolites, cytokines, or nutrient gradients. Coupling multiple mature tissues on a microfluidic chip by blood perfusion and maintaining tissue phenotypes is still in its infancy. Consequently, the ultimate goal of creating “body on a chip” has not been fully achieved, though it is still on its way forward. Recently, in a work published in Nature Biomedical Engineering, Gordana Vunjak-Novakovic and colleagues proposed a brand-new tissue chip system to model whole-body physiology. The induced mature heart, liver, bone, and skin tissues were integrated into a relatively complex human body physiology with preserved organ phenotypes instead of compiling several single tissue units by simply sharing the culture media (Figure 1).4 In this multi-organ chip system, the team cultured each tissue within its unique microenvironment by separating them with an endothelium layer and simultaneously linking the mature tissues to the main bloodstream to make them better communicate in an in vivo-like manner.
Figure 1.
Multi-organ tissue chip system
Left: illustration of patient-on-a-chip model. Middle: the patient-derived human induced pluripotent stem cell-induced mature tissue niches are linked by recirculating vascular flow to allow for the recapitulation of interdependent organ functions. Each individual tissue is cultured in its compartment and is separated from the recirculating vascular flow by a selectively permeable endothelial barrier. Right: the interlinked tissues recapitulated the pharmacokinetic and pharmacodynamic profiles of doxorubicin in humans.
Multi-organ chips as potential patient-specific models
The multi-organ tissue chip system was created with the aim of physiological communication between engineered mature tissues to establish the complexity of multi-organ metabolism and pharmacokinetics/pharmacodynamics of drugs. To this end, a selective permeable endothelial barrier with 20 μm pores formed gap junctions under shear stress, permitting cytokine, exosome, and cell exchange in the exterior blood flow. Then, the organ–organ communication was evaluated by exosome uptake and immune cell infiltration studies. The conventional multi-organ tissue chip without external fluid flow, however, showed that the injured tissue targeted immune cell accumulation, further confirming the communication efficacy of the engineered tissue chip.
Organ communication on the chip provides better emulation of disease development in living tissues.5 Gene Ontology analysis showed that all connected tissues maintained their phenotypes and function stability during the 4 weeks of maturation, contributing a more solid basis for the subsequent pharmacokinetics/pharmacodynamics of the model drug for its organ toxicity assessment (Figure 1). These conditions allowed recapitulation of clinical pharmacokinetics/pharmacodynamics profiles of doxorubicin and microRNA biomarkers of cardiotoxicity. Apart from the groundbreaking advance of linking the mature tissue by the external blood flow, another elusive advantage of the presented technique is that all the human tissues were engineered using human induced pluripotent stem cells from a single donor, thereby demonstrating the potential tissue chip’s utility for “patient-on-a-chip” models.
Nonetheless, some concerns remain. As only four tissues are involved in this engineered chip, it is limited to systematically studying the drug action on the whole human physiology. Furthermore, future work should be directed toward establishing an internal circulation system that provides the fluid flow for the engineered multi-organs. Nevertheless, we believe that the groundbreaking advance reported in this study sheds light on the patient on a chip goal and will find its way to clinical applications soon.
Acknowledgments
This work was supported by the Natural Science Foundation of Zhejiang Province (BY22H181190).
Declaration of interests
The authors declare no competing interests.
Published Online: July 6, 2022
Contributor Information
Chunhai Ke, Email: kchnb@163.com.
Wenguo Cui, Email: wgcui80@hotmail.com.
References
- 1.Prasad V., Mailankody S. Research and development spending to bring a single cancer drug to market and revenues after approval. JAMA. Intern. Med. 2017;177:1569–1575. doi: 10.1001/jamainternmed.2017.3601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Clevers H. Modeling development and disease with organoids. Cell. 2016;165:1586–1597. doi: 10.1016/j.cell.2016.05.082. [DOI] [PubMed] [Google Scholar]
- 3.Liu H., Wang Y., Cui K., et al. Advances in hydrogels in organoids and Organs-on-a-Chip. Adv. Mater. 2019;31 doi: 10.1002/adma.201902042. [DOI] [PubMed] [Google Scholar]
- 4.Ronaldson-Bouchard K., Teles D., Yeager K., et al. A multi-organ chip with matured tissue niches linked by vascular flow. Nat. Biomed. Eng. 2022;6:351–371. doi: 10.1038/s41551-022-00882-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vunjak-Novakovic G., Ronaldson-Bouchard K., Radisic M. Organs-on-a-chip models for biological research. Cell. 2021;184:4597–4611. doi: 10.1016/j.cell.2021.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]