Abstract
Bronchiolitis obliterans syndrome (BOS) may develop after either lung or haematopoietic stem cell transplantation (HSCT), with similarities in histopathological features and clinical manifestations. However, there are differences in the contributory factors and clinical trajectories between the two conditions. BOS after HSCT occurs due to systemic graft-versus-host disease (GVHD), whereas BOS after lung transplantation is limited to the lung allograft. BOS diagnosis after HSCT is more challenging, as the lung function decline may occur due to extrapulmonary GVHD, causing sclerosis or inflammation in the fascia or muscles of the respiratory girdle. Treatment is generally empirical with no established effective therapies. This review provides rare insights and commonalities of both conditions, which are not well elaborated elsewhere in contemporary literature, and highlights the importance of cross disciplinary learning from experts in other transplant modalities. Treatment algorithms for each condition are presented, based on the published literature and consensus clinical opinion. Immunosuppression should be optimised, and other conditions or contributory factors treated where possible. When initial treatment fails, the ultimate therapeutic option is lung transplantation (or re-transplantation in the case of BOS after lung transplantation) in carefully selected candidates. Novel therapies under investigation include aerosolised liposomal cyclosporine, Janus kinase inhibitors, antifibrotic therapies and (in patients with BOS after lung transplantation) B-cell-directed therapies. Effective novel treatments that have a tangible impact on survival and thereby avoid the need for lung transplantation or re-transplantation are urgently required.
Short abstract
A review focusing on novel treatment approaches for bronchiolitis obliterans syndrome, to reverse the pathological changes and thereby impact tangibly on survival or need for subsequent lung transplantation, and improve patients’ quality of life https://bit.ly/3lCIB0x
Introduction
Pulmonary complications such as bronchiolitis obliterans syndrome (BOS) are common after lung or haematopoietic stem cell transplantation (HSCT) [1]. BOS is characterised by a progressive obstructive ventilatory defect due to the development of obliterative bronchiolitis [1–7]. Hitherto, the rarity of obliterative bronchiolitis delayed the understanding of its disease pathogenesis [8]. However, the emergence of lung transplant and HSCT as therapeutic modalities has led to an increased interest in this rare condition, providing much of the literature regarding the risk factors, natural history, therapeutic responsiveness and outcomes [9, 10]. While microvascular ischaemia likely plays a role in pathogenesis, the driving force is almost certainly immune related; for this reason, the twin pathways of “graft versus host” and “host versus graft” need to be compared.
BOS after lung transplantation is the most common phenotype of chronic lung allograft dysfunction (CLAD), developing in up to 50% of recipients by the fifth post-transplant year and is the major cause of late post-transplant mortality [11–13]. BOS after HSCT is less common, affecting 2–10% of recipients within 5 years [14–20]. BOS is the lung manifestation of multisystemic graft-versus-host disease (GVHD), with a prevalence of 14% in patients with chronic GVHD [21]. BOS negatively impacts prognosis after lung transplantation and HSCT [11–13, 18, 21–23]. However, as data required to confirm evidence-based therapeutic recommendations are limited, treatment is generally consensus driven [24].
The aims of the current article are to describe the characteristics of BOS after lung transplantation and HSCT, provide a viewpoint on diagnosis and management of these conditions based on expert opinion and a review of the literature, and describe research into new treatment strategies.
Methods
A videoconference of international experts (adult and paediatric transplant physicians, pulmonologists and haematologists), facilitated by a professional moderator, was held on 18 December 2020. This review summarises the consensus opinions of the group on BOS after lung transplantation and HSCT, supported by published literature.
What are CLAD-BOS and chronic GVHD-BOS?
CLAD
The International Society for Heart and Lung Transplantation (ISHLT) consensus statement defines CLAD as a persistent decline in pulmonary function, characterised by a decrease of ≥20% in forced expiratory volume in 1 s (FEV1) for >3 months from post-transplant baseline, after excluding other potential causes (table 1) [3]. The baseline value is calculated from the mean of the two best post-operative FEV1 measurements taken ≥3 weeks apart. CLAD severity is staged based on the current FEV1 relative to baseline [3].
TABLE 1.
| CLAD-BOS# after lung transplantation [3] | GVHD-BOS [41] |
| Persistent (present for >3 weeks) decrease in FEV1 of ≥20% from the reference baseline value¶ after exclusion of other possible causes | FEV1 <75% of predicted with ≥10% decline over <2 years; FEV1 should not correct to >75% predicted with albuterol and absolute decline for corrected values should remain ≥10% over 2 years |
| Obstruction on spirometry (FEV1/FVC ratio of <0.7) | FEV1/vital capacity ratio of <0.7 or the fifth percentile of predicted+ |
| No evidence of restriction§ | Absence of respiratory tract infection documented with investigations and directed by clinical symptomsƒ |
| No CT evidence of pulmonary or pleural fibrosis## | One of the two supporting features of BOS: 1) evidence of air trapping on expiratory CT, or small airway thickening or bronchiectasis by high-resolution chest CT, or 2) evidence of air trapping by pulmonary function testing¶¶ |
BOS: bronchiolitis obliterans syndrome; HSCT: haematopoietic stem cell transplant; CLAD: chronic lung allograft dysfunction; GVHD: graft-versus-host disease; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; CT: computed tomography. #: for diagnostic criteria in other CLAD phenotypes, see Verleden et al. [3]. ¶: baseline value is calculated as the mean of the best two post-operative FEV1 measurements taken >3 weeks apart. +: vital capacity included FVC or slow vital capacity, whichever is greater; the fifth percentile of predicted is the lower limit of the 90% confidence interval; use lower limits of normal for paediatric or elderly patients. §: reduction in total lung capacity of ≥10% from baseline. ƒ: for example, chest radiographs, CT scans, microbiological cultures (sinus aspiration, upper respiratory tract viral screen, sputum culture, bronchoalveolar lavage). ##: parenchymal opacities and/or pleural thickening indicative of pulmonary and/or pleural fibrosis and likely to cause a restrictive physiology. ¶¶: residual volume >120% predicted or residual volume/total lung capacity elevated outside the 90% confidence interval.
Restrictive, obstructive, mixed or undefined clinical phenotypes of CLAD are defined based on the predominant ventilatory pattern, total lung capacity (TLC) and presence/absence of opacities on chest computed tomography (CT) scan (supplementary table S1) [3, 25]. Pulmonary function changes characteristic of the restrictive allograft syndrome (RAS) phenotype are declining TLC and a FEV1/forced vital capacity (FVC) ratio >0.7, whereas BOS is characterised by stable or increasing TLC and declining FEV1/FVC indicative of obstruction and hyperinflation [26]. The presence of multi-lobar persistent parenchymal and/or pleural opacities on CT (or chest radiograph (CXR) if CT is not available) is required for RAS diagnosis [3, 25].
∼65–70% of CLAD patients present predominantly with the BOS phenotype, and 10–35% have the RAS phenotype [12, 26–29], although the proportion with RAS appeared lower in a more recent study based on the latest ISHLT consensus criteria [29]. This is partly because mixed phenotype is now considered a different entity, whereas RAS and mixed were previously grouped together. Using these criteria, Levy and colleagues estimated that ∼5% of patients present with mixed and ∼10% with undefined phenotype [29], whereas Verleden and colleagues estimated that the undefined phenotype was less frequent (2%); however, raw data were not independently verified. Overall survival and graft survival are worse in those with RAS or mixed phenotype than the BOS or undefined phenotype [12, 26, 27, 29–31]. Survival differences between CLAD phenotypes are unaffected by age, sex, native lung disease and cytomegalovirus (CMV) serostatus mismatch [29]. However, allograft survival among all patients with CLAD is generally worse in those with CMV serostatus mismatch and in those failing to achieve predicted FEV1 and FVC post-operatively [12].
Phenotypes are not static, and patients can progress from the BOS to RAS phenotype, and more rarely from RAS to BOS [26, 32]. A patient who transitions from BOS to RAS will meet the criteria for the mixed phenotype [3, 25], so it is more accurate to describe the phenotypic change as BOS-to-mixed. Survival in BOS-to-RAS patients is worse than in BOS, but comparable to or better than in RAS patients [31, 33].
Risk factors for BOS and RAS include non-adherence to or suboptimal immunosuppressive regimens, acute cellular rejection (ACR) episodes, lymphocytic bronchiolitis, community-acquired respiratory viral (CARV) infection, donor-specific antibodies, air pollution, gastro-oesophageal reflux (GERD), Pseudomonas aeruginosa or Aspergillus fumigatus colonisation and CMV mismatch [31, 34, 35], although more RAS patients seem to develop human leukocyte antigen (HLA) antibodies compared with BOS patients [36]. Additionally, the development of RAS, but not BOS, appears to be delayed after living donor lung lobar versus cadaveric donor transplantation [37].
GVHD-BOS
GVHD-BOS usually develops between 100 days and 2 years of HSCT, but onset beyond 5–6 years post-HSCT has been noted, usually in patients experiencing an extrapulmonary GVHD flare [7, 16, 17]. Risk factors for GVHD-BOS are impaired lung function before and early after HSCT, a myeloablative/busulfan-containing conditioning regimen, CMV seropositivity, pre-transplant history of pulmonary disease, female donor, unrelated donor and prior acute GVHD; receipt of antithymocyte globulin, which decreases chronic GVHD risk, reduces the risk of BOS [17, 18, 38].
There is a need for clinical biomarkers at 80–100 days post-HSCT, because declining lung function at this time-point is a significant risk factor for later BOS development. Beyond chest irradiation and FEV1, forced expiratory flow between 25% and 75% of maximum has emerged as an important biomarker for the early detection of at-risk patients [38, 39].
Patients may present with persistent cough or dyspnoea-on-exertion, while asymptomatic patients may be identified during routine monitoring of pulmonary function [7]. The nonspecific symptomatology of GVHD-BOS may contribute to diagnostic delays [40]. Unlike CLAD, which affects only the lungs, GVHD is a systemic condition, so BOS after HSCT usually occurs in association with signs/symptoms affecting other organ systems, such as the skin, nails, eyes, mouth, hair, genitals, joints, liver and haematopoietic systems, with clinical manifestations of fatigue or decreased endurance at the outset [7, 41, 42]. These often present before GVHD-BOS diagnosis [7]. Overall, the diagnostic features of GVHD-BOS are similar to those of CLAD, with primary pulmonary function impairment [41].
The US National Institutes of Health definition is shown in table 1 [41]. These features include FEV1/vital capacity (forced or slow, whichever is greater) <0.7 or the 5th percentile of predicted and FEV1 <75% of predicted and with ≥10% decline over <2 years in the absence of an infective cause, and evidence of air trapping or other signs of BOS (small airway thickening or bronchiectasis) on CT or evidence of gas trapping on lung function testing.
Unlike CLAD, specific GVHD-BOS phenotypes have not been defined [43]. However, Bergeron et al. [44] proposed two different lung function patterns in patients with GVHD-BOS. The first is a typical obstructive defect characterised by FEV1/FVC ratio <0.7, and the second is atypical with FEV1 <80% and FVC <80% but normal lung capacity, such that the FEV1/FVC ratio was >0.7. Outcomes did not differ between the two groups, but patients with the typical pattern had fewer centrilobular nodules on CT [44]. These data need to be further explored as they could also represent a restrictive GVHD lung disease or a subset of patients with BOS who do not demonstrate obstruction due to extrapulmonary constraints (e.g. sclerotic GVHD of the respiratory girdle).
The trajectory of FEV1 decline in patients with GVHD-BOS is heterogeneous and deterioration may happen rapidly [45, 46]. Studies have shown that patients with a rapid decline in lung function (25% FEV1 decline) during the first 3 months of GVHD-BOS, those with poorer FVC at diagnosis of GVHD-BOS or those with nontuberculous mycobacteria in bronchoalveolar lavage (BAL) culture have worse survival than those who do not [14, 45–47].
Pathophysiology and aetiology
CLAD
The temporal relationship between the development of CLAD and infectious diseases (e.g. chronic pulmonary P. aeruginosa and CARV) supports the concept that one pathway of CLAD is a microbe–allograft–host interaction, whereby the infectious pathogen causes allograft cells to release chemokines that recruit host leukocytes to the site that then recognise the airways as non-self [48, 49]. Other pathways involve endogenous molecules released by injury to small airways or their microvasculature following diverse injuries including ACR [50]. The influx of leukocytes precipitates an allo-response causing graft dysfunction [48, 49]. Thus, CLAD is a host-versus-graft (rather than a graft-versus-host) disease. The inescapable conclusion is that both are immune-regulated phenomena, which may explain why BOS is common after lung transplantation and HSCT.
Dysfunction of the regulatory mechanisms and induction of an acute inflammatory response generate a positive feedback loop and amplify the immune response, causing the pathological process to transition from an acute to a chronic response [50]. Humoral (adaptive) immune activation may be a contributing mechanism determining CLAD-RAS phenotype development [51]. Continuous exposure of the airway epithelium to inflammatory processes can lead to fibroblast recruitment and eventually extracellular matrix remodelling [25]. Acute fibrinous and organising pneumonia appears to be an early event in this process [25, 52].
The histological features of CLAD can be heterogeneous. The dominant and most frequent finding is obliterative bronchiolitis. Other common findings include variable grades of ACR including perivascular and/or peribronchiolar lymphocytic infiltrates, pleuro-parenchymal fibro-elastosis, and microvascular damage [53].
GVHD-BOS
The early processes leading to BOS in GVHD differ from those in CLAD, but the eventual histological changes are relatively similar. As described earlier, BOS after HSCT is a manifestation of chronic GVHD. While less is known about the aetiology of BOS after HSCT, chronic GVHD is caused by central tolerance failure and B-cell and auto-antibody production [40, 54]. T-cells play a major role in the initiation of GVHD; the subset of T-cells that are primarily responsible for the development of pulmonary GVHD are not characterised, although CD4+ T-helper 17 cells are likely involved [40].
The pathology of GVHD-BOS is less well defined than BOS after lung transplantation due to a lower volume of surgical lung biopsies and lower autopsy rates. One study showed two distinct patterns: constrictive bronchiolitis obliterans (CBO) and lymphocytic bronchiolitis [55]. CBO demonstrates marked bronchiolar narrowing with fibrous lesions and hyperplasia of the epithelium, whereas in lymphocytic bronchiolitis, fibrosis is absent, and there is bronchiolar dilatation, and epithelial thinning, necrosis or disappearance [56]. Patients with lymphocytic bronchiolitis tend to have better survival than those with CBO [55].
The pattern of morphological changes on pulmonary micro-CT is similar in patients with obstructive CLAD and those with GVHD-BOS [57], with both having a reduced number of terminal bronchioles [57].
Diagnosis
Early diagnosis of CLAD may be more likely than GVHD-BOS, since pulmonary function is monitored more frequently in lung transplant recipients. In contrast, the major concern after HSCT is haematological malignancy relapse, hence pulmonary function testing is generally less frequent and sporadic [45]. As a result, many HSCT patients potentially miss the opportunity for early intervention for BOS [45].
Current guidelines recommend implementing early investigations for CLAD as soon as the condition is suspected (i.e. ≥10% reduction in baseline FEV1) [3]. Spirometry is a key monitoring and diagnostic tool in both lung transplant or HSCT recipients with suspected pulmonary complications, but extrapulmonary manifestations may affect lung function tests in GVHD-BOS patients. Home spirometry may detect early pulmonary function decline but is not a substitute for office testing [58] and requires periodical calibration of the home spirometer against the laboratory spirometer and assessment of the patient's technique. Home spirometry may be a useful way to limit clinic visits (e.g. during the SARS-CoV-2 pandemic).
Once lung function decline is confirmed, possible causes should be investigated using bronchoscopy with visual airway inspection, transbronchial biopsies where indicated, BAL, TLC testing (e.g. plethysmography) and CT imaging (supplementary table S2) [3]. Lung biopsy (usually by the transbronchial route) is considered the gold standard diagnostic modality to rule out other causes of FEV1 decline, such as ACR. However, the risks of biopsy (bleeding and pneumothorax) may outweigh the benefits in some patients, particularly those with GVHD-BOS, in whom noninvasive methods play an important role in diagnosis [3, 59–61].
Small airway brushings may detect a lymphocytic gene expression signature in patients with CLAD, which may not be apparent in transbronchial biopsies [62]. Functional magnetic resonance imaging (MRI) can assess regional changes in lung function assisting early CLAD detection [31] but is expensive and not universally available [59].
CT or CXR are commonly used to identify opacities, but CT is preferred due to greater sensitivity and specificity than CXR and better visualisation of changes in lung parenchyma and small airways. The presence of air trapping, and especially its increase, as seen on high-resolution chest CT during inspiration and expiration is supportive of BOS diagnosis [63, 64]. Radiation exposure is higher with CT than conventional CXR but can be substantially reduced by low-radiation protocols. While the initial use of CXR followed by CT in patients with suspicious CXR findings may be optimal, air trapping can limit the utility of CXR in GVHD-BOS patients [59].
Typical chest CT findings in CLAD (RAS/mixed phenotype) are opacities (ground glass appearance, consolidation, small linear or reticular densities) and/or increased pleural thickening indicative of fibrosis [3]. Centrilobular opacities, air trapping and bronchial wall thickening may be present on CT of patients with BOS after lung transplantation or GVHD, but pleural thickening is absent or rare after GVHD [64].
An emerging modality in BOS diagnosis and assessment is parametric response mapping (PRM), in which expiratory and inspiratory CT scans undergo voxel-based analysis [65, 66]. In patients with BOS after lung transplantation, PRM correlates with FEV1 decline (at least in patients without a restrictive pattern), whereas air trapping does not [66]. In patients with BOS after HSCT, PRM can detect BOS even in those with concurrent infection [65]. PRM is useful for monitoring BOS progression after either type of transplant [65, 66], but further data are required.
MRI has been investigated for morphological assessment of transplanted lungs, and some MRI parameters may be early markers of CLAD [67, 68], but further data are needed before MRI is routinely used for BOS assessment.
Perhaps the most innovative strategy to monitor ACR and antibody-mediated rejection revolves around the understanding that these responses cause cell death and the release of donor-derived cell-free DNA into the circulation, which may be used as a noninvasive, quantitative marker to track the events leading to CLAD [69].
Artificial intelligence (AI) is another innovation that can be applied to understanding rare diseases. One example is the identification of lung GVHD after HSCT by quantitative imaging [70]. AI has the potential to increase our understanding of the similarities and differences between CLAD-BOS and GVHD-BOS.
Current treatment approaches
CLAD-BOS
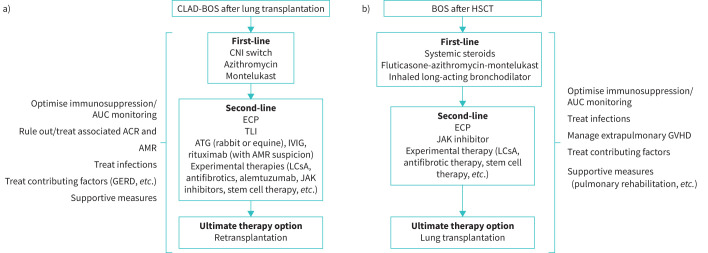
Initially, any precipitating or underlying conditions should be identified and treated, including ACR, infection, lymphocytic bronchiolitis, GERD or others [3], and the patient's maintenance immunosuppressive regimen should be optimised (figure 1a). A consecutive case series indicated that early management of BOS, when it is less severe, may be efficacious in stabilising declining lung function parameters at a higher plateau. Based on their years of clinical practice experience, the authors agreed that early introduction of therapies may mitigate progression to more severe BOS [71]. If ACR is diagnosed, it should be treated as per standard protocols avoiding prolonged courses of high-dose steroids [3, 24]. There is evidence suggesting that the prevalence of BOS after lung transplantation is lower in patients receiving tacrolimus versus cyclosporine [72] and that switching from cyclosporine to tacrolimus stabilises lung function [24, 73].
FIGURE 1.
Treatment approach for BOS after a) lung transplantation and b) HSCT. ACR: acute cellular rejection; AMR: antibody-mediated rejection; ATG: antithymocyte globulin; AUC: area under the plasma concentration–time curve; BOS: bronchiolitis obliterans; CNI: calcineurin inhibitor; CLAD: chronic lung allograft dysfunction; ECP: extracorporeal photopheresis; GERD: gastro-oesophageal reflux disease; GVHD: graft-versus-host disease; HSCT: haematopoietic stem cell transplant; IVIG: intravenous immunoglobulin; JAK: Janus kinase; LCsA: aerosolised liposomal cyclosporine; TLI: total lymphoid irradiation.
Among available treatments, the most evidence exists for azithromycin [74–81], including data from a randomised, placebo-controlled trial [82]. In observational studies, azithromycin was associated with a FEV1 increase of 16–18% predicted [74, 75, 78], and an absolute FEV1 increase of 0.11–0.86 L [74, 78–80]. However, response rates were only 29–50% in these studies, indicating that ≥50% of patients showed no improvement [74, 75, 78–81]. Factors associated with a greater likelihood of response were airway neutrophilia (detected by BAL) [79, 81] and early treatment initiation [76]. In the randomised study, nine out of 23 patients on azithromycin (39%), but none of those in the placebo group, responded to treatment with a FEV1 increase of ≥10% predicted (p=0.002). The difference in FEV1 between the azithromycin and placebo groups was 0.278 L (p<0.001) [82]. Where safety data were reported, azithromycin was generally well tolerated [75, 82]. One patient developed laryngeal oedema (serious) that resolved after azithromycin discontinuation [75]. The most frequent adverse effects were gastrointestinal disorders. The recent ISHLT consensus states that azithromycin should be initiated as early as possible, even before any definite BOS diagnosis has been made [3], based on its effects on lung function [76, 77, 81]. The optimal dosage and duration of azithromycin have not been established [78, 80, 81]. BOS may occur despite long-term maintenance azithromycin.
The leukotriene antagonist montelukast has also been investigated for BOS treatment [83–85]. Montelukast is an oral treatment for persistent asthma [86], showing antifibrotic effects in animal models of BOS after lung transplantation [87]. In a pilot study of 11 patients with low neutrophilia on BAL (<15%) who were expected to be poor responders to azithromycin, montelukast slowed FEV1 decline [85]. Similarly, attenuation of FEV1 decline was seen in a retrospective study of 153 BOS patients, which also showed significantly longer survival in patients with response to montelukast than in those who did not respond [83]. However, a randomised, placebo-controlled trial failed to demonstrate any effect of montelukast on FEV1 or graft survival in BOS patients, although benefit was seen in patients with early-stage BOS [84]. As this study only included 15 patients in each treatment arm, further research in larger patient cohorts is warranted.
Second-line options include extracorporeal photopheresis (ECP) or total lymphoid irradiation (TLI) [3]. ECP slows the rate of FEV1 decline in patients with CLAD-BOS [88] and is probably more effective in patients with the BOS than the RAS phenotype [89]. Some data support an improvement in survival among patients with CLAD receiving ECP, but overall, the evidence supporting ECP in BOS is considered to be Class IIB, level C [90]. Moreover, ECP is expensive, not universally available and potentially burdensome for some patients. It requires secure venous access and multiple treatment sessions initially over a short period [90]. On the other hand, ECP is generally well tolerated, with no major adverse effects reported in a large patient series [90].
TLI has been shown to slow the rate of FEV1 decline in BOS patients, including those not responding to azithromycin [91, 92], but studies to date have been small and observational [90]. In addition, not all patients are able to complete the required number of treatment sessions due to bone marrow suppression or infection [91].
Re-transplantation remains an option in carefully selected patients with BOS who fail available first- and second-line treatment options.
GVHD-BOS
Other manifestations of chronic GVHD or non-infectious complications have been described, including interstitial lung diseases, but specific management of these conditions differs from the approach to BOS [7].
The first step in managing BOS after HSCT is to ensure that comorbidities and potential precipitating factors (e.g. GERD) are managed, and that immunosuppressive treatment is optimised [60]. Treatment of BOS is often additive to that targeting the extrapulmonary manifestations of GVHD; therefore, clinicians should manage the immunosuppression as a whole (figure 1b). Long-term corticosteroids are not considered beneficial for BOS after HSCT but may be part of therapy for GVHD.
Currently, there is limited evidence to guide treatment of BOS after HSCT [93]. The European Society for Blood and Marrow Transplantation recommends the combination of fluticasone, azithromycin and montelukast (FAM), with a steroid pulse and rapid taper over 1 month (class 2A evidence) [93]. This recommendation was based on data from a non-randomised study in 36 patients with BOS after HSCT who received FAM plus a steroid burst (planned to be 4 weeks total of prednisone commencing at 1 mg·kg−1·day−1 and initiating taper after 1 week) [94]. Notably, most patients were on standard treatment for chronic GVHD (i.e. sirolimus or a calcineurin inhibitor), which likely contributed to BOS control. After 3 months, only 6% of patients met the definition of treatment failure (FEV1 decline of ≥10%) and 17% met the definition at 6 months [94]. Patient-reported outcomes showed significant improvements from baseline at 3 months, including in social functioning, mental and emotional well-being, the 6-min walk test and symptom severity. FAM was generally well tolerated, and only one patient discontinued treatment because of adverse events [94].
The data for azithromycin alone are less robust [95]. While one study suggested that azithromycin alone as a preventive strategy for BOS may be linked to relapse, more recent larger studies did not show increased risk of relapse with azithromycin post-GVHD, although increased risk of secondary cancers with higher use of concomitant steroids was observed [96, 97]. A combination of inhaled long-acting bronchodilator plus inhaled corticosteroid has been shown to improve FEV1 at 1 month (by 200 mL and 12%) in newly diagnosed patients with mild-to-moderate BOS, without the use of systemic corticosteroids [98]. Patients may also benefit from supportive care including prophylaxis for infection, pulmonary rehabilitation, nutritional support and treatment for GERD [60, 99].
There are no standard second-line treatment approaches to lung chronic GVHD, so these are determined by national, local or institutional guidelines [93]. Some retrospective data support the use of ECP, with ECP improving survival in HSCT with BOS without significantly impacting pulmonary function [100]. Given the overall paucity of data, enrolment in a clinical trial should be prioritised as a second-line treatment approach [60, 93].
In the USA, other agents for chronic GVHD are being explored: the Bruton's tyrosine kinase inhibitor ibrutinib is approved for the second-line treatment of chronic GVHD [101], as is the Janus kinase (JAK) inhibitor ruxolitinib, but there are no data regarding their efficacy for BOS [102]. One trial evaluated the use of a tumour necrosis factor-α inhibitor, etanercept, and showed benefit (i.e. ≥10% improvement in absolute FEV1 or FVC) in one-third of patients with BOS [103].
As with CLAD, the ultimate treatment option is a lung transplant [104].
Future directions
Hypothesis-driven novel approaches
Any novel therapeutic approach for BOS management is driven by the understanding of the disease pathogenesis of obliterative bronchiolitis. In this regard, the immunogenicity of the allograft is considered to be important, as obliterative bronchiolitis develops due to an injury–response mechanism, where the small airways are targeted as non-self after upregulation of HLA on respiratory epithelial cells [105]. Early studies showed that bronchiolar epithelial cells from lobectomy samples of former smokers expressed both Class 1 and Class 2 HLAs, as did small airways in explants from patients undergoing re-transplantation for obliterative bronchiolitis [106, 107]. Despite this promising groundwork, the potential role of HLA presentation on small airway epithelia has only recently been reconsidered with the acceptance of the relevance of antibody-mediated rejection as a cause of allograft injury [108]. Indeed, the presence of donor-specific antibodies against Class 2 HLAs, especially those against HLA-DQ2, is considered a major risk factor for CLAD development [109].
Ischaemia plays an important role in scar formation after injury, and damage to the “watershed” microvasculature of the terminal bronchioles appears to compound the geometric factors which determine why small airways bear the brunt of the rejection response [110]. Obliterative bronchiolitis lesions seen on micro-CT scans are essentially scars at focal segments of small airways where the remnants of the external elastic lamina are best visualised on histopathology by elastin van Gieson staining [4]. While the upstream bronchiole may appear normal or simply show thickening of the basement membrane, the acinus subtended by the affected bronchiole is excluded from gas exchange [4]. Evidence for an immunological aetiology for BOS is supported by a retrospective analysis of transbronchial lung biopsies, where the severity of lymphocytic bronchiolitis was strongly correlated with the time to develop BOS [111].
Understanding the fundamental pathogenesis of BOS and applying this knowledge in clinical practice is essential to mitigate the risk of BOS after lung transplantation or HSCT. Since HLA presentation is thought to be the main target, efforts should be directed towards effective prevention of trigger events which upregulate antigen presentation and damage small airways. Trigger events probably include T-cell-mediated ACR, CARV infections with lower respiratory tract tropism (such as respiratory syncytial virus), certain coronavirus infections, bacterial infections (such as P. aeruginosa, mycoplasma and chlamydia) and inflammation from aspiration of gastric contents [112]. The possibility of autoimmunity to cryptic self-antigens liberated by these triggers is supported by the presence of self-antigens in microvesicles and the development of autoantibodies against Type V collagen and K-α1 tubulin [113, 114].
Novel therapies will need to embrace these concepts to successfully prevent tissue damage and modify aberrant repair. A multifaceted approach that includes risk factor management for the triggers outlined above is essential, starting with a global response to ameliorate the toxic effects of air pollution on small airways and protect airways from direct/indirect exposure to cigarette smoke and e-cigarette emissions [115].
In line with these hypotheses, several agents have been or are being investigated for the treatment of BOS after lung transplantation or HSCT, as discussed below.
Aerosolised liposomal cyclosporine
Aerosolised cyclosporine delivers immunosuppressive therapy directly to the allograft, where it acts locally at the immune activation site, limiting systemic exposure [116]. A liposomal cyclosporine formulation for aerosolised delivery (Zambon), currently undergoing phase III development, has been granted orphan drug status by the US Food and Drug Administration [117]. This formulation showed good lung deposition after nebuliser administration in lung transplant recipients [116]. In a randomised study, adding aerosolised liposomal cyclosporine to standard care improved or stabilised a range of lung function parameters and was significantly more effective than standard care alone [118], but this study was open-label without placebo control. Treatment was well tolerated, with no increase in serious adverse events with aerosolised liposomal cyclosporine versus standard care alone; adverse events included conjunctivitis, pharyngitis and productive cough [118].
The Boston clinical trial programme is investigating aerosolised liposomal cyclosporine in patients with BOS after lung transplantation or HSCT (table 2); results will become available over the next 2–3 years.
TABLE 2.
Clinical trials with aerosolised liposomal cyclosporine
| Study | Clinical trial registration number | Phase | Design (n) | Treatments | Primary end-point | Completion date |
| BOS after single or double lung transplantation | ||||||
| Iacono et al. 2019 (single or double LTx) [118] | NCT01650545 | IIb | Open-label, parallel (21) | LCsA 5 or 10 mg+SOC versus SOC | 1) A composite of BOS PFS, defined as time from randomisation to ≥20% decline in FEV1, re-transplantation or death, whichever occurred first (prolonged mechanical ventilation and irreversible respiratory failure equivalent to ≥20% decline of FEV1), and 2) BOS grade progression by grade changes from randomisation to study completion |
Sep 2017 |
| BOSTON-1 (single LTx) | NCT03657342 | III | Randomised, single-blind (110) | LCsA 5 mg+SOC versus SOC | Mean change in FEV1 from baseline to week 48 | July 2023 |
| BOSTON-2 (double LTx) | NCT03656926 | III | Randomised, single-blind (152) | LCsA 10 mg+SOC versus SOC | Mean change in FEV1 from baseline to week 48 | July 2023 |
| BOSTON-3 (OLE for BOSTON-1 and -2) | NCT04039347 | III | Open-label (220) | LCsA 5 mg or 10 mg | Mean change in FEV1 from baseline to week 24 | Apr 2024 |
| BOS after HSCT | ||||||
| BOSTON-4 | NCT04107675 | II | Randomised, single-blind (24) | LCsA 2.5, 5 or 10 mg versus placebo | Safety and tolerability | May 2022 |
BOS: bronchiolitis obliterans syndrome; LTx: lung transplantation; LCsA: aerosolised liposomal cyclosporine; SOC: standard of care; OLE: open-label extension; PFS: progression-free survival; FEV1: forced expiratory volume in 1 s; HSCT: haematopoietic stem cell transplant.
JAK inhibitors
JAK inhibitors are used for the treatment of solid tumours and are in development for autoimmune conditions [119]. Ruxolitinib (Incyte, Novartis, Wilmington, DE, USA) is approved for the use of steroid-refractory GVHD in the USA [102] and is the most studied agent from this class for the treatment of BOS after HSCT or bone marrow transplantation. To date, published data are limited to small case series or case studies in adults or children with steroid-resistant BOS [120–123]. However, some studies have suggested a potential role for these agents in the treatment of BOS [120–123]. An open-label phase II study (NCT03674047) is underway in the USA in patients with newly diagnosed or established BOS after HSCT (completion expected in March 2023).
B-cell-directed therapies
B-cells are important mediators of chronic GVHD [124, 125] and are implicated in the development of CLAD [126]; thus, treatments inhibiting B-cell activation may be useful for BOS treatment after lung transplantation or HSCT. However, B-cell-directed therapies tend to be associated with significant safety concerns, including cytopenias, immunosuppression and infections [127, 128].
Rituximab is a chimeric human/mouse monoclonal antibody against CD20 that has cytolytic activity against B-cells and is approved for the treatment of B-cell lymphomas [127]. Small case series suggest that rituximab may be effective in improving lung function in patients with BOS after lung transplantation with concurrent antibody-mediated rejection [129–131]. There are less data regarding its use in patients with BOS after HSCT. One report of three cases described lung function stabilisation in only one patient, but the authors acknowledged that rituximab was only initiated in severely ill patients, and its efficacy if used at an earlier stage of BOS could not be precluded [131].
Alemtuzumab is an anti-CD52 monoclonal antibody approved for use in B-cell lymphomas [128]. A database analysis suggested that using alemtuzumab as part of the induction regimen reduced the 5-year risk of BOS after lung transplantation compared with basiliximab-based induction or no induction [132]. Two retrospective studies and a case series (n=10) reported slowing or reversal of lung function decline with alemtuzumab in a high proportion of patients with BOS after lung transplantation [133–135]. Alemtuzumab appeared to be similarly effective to ECP [134]. Patients with early-stage BOS were more likely to respond to alemtuzumab than those with late-stage BOS [132]. Notably, this strategy would mitigate anticancer and anti-infection control in patients with BOS after HSCT and would thus be associated with higher risk.
Antifibrotic treatments
The antifibrotic agents pirfenidone and nintedanib are used in the treatment of chronic fibrosing pulmonary conditions, such as idiopathic pulmonary fibrosis [136, 137], leading to speculation that they may ameliorate the fibrotic changes in BOS after transplantation. Preclinical studies demonstrated antifibrotic effects with pirfenidone in animal models of BOS or post-transplant pulmonary complications [138–141]. Preliminary data from a case series of 11 RAS patients showed that pirfenidone stabilised lung function during long-term treatment and provided a bridge to a second lung transplant in three patients (27%) [142]. Published data with nintedanib are limited to case reports: one showed a clinical benefit in a patient with BOS after HSCT [143], and another showed no benefit in a patient with BOS after lung transplantation [144].
Clinical trials with both agents are underway in post-transplant patients with BOS (table 3). The results of the EPOS trial (pirfenidone versus placebo) were negative [145]. Another small phase II study (PIRCLAD; NCT03359863) is also investigating pirfenidone in patients with RAS (n=10), with completion expected in October 2021. A potential role for mesenchymal stem cell therapy is being explored in a randomised controlled study in Australia, but results are not yet available.
TABLE 3.
Clinical trials with antifibrotic treatments
| Clinical trial registration number (name) | Patients (target n) | Phase | Design | Treatments | Primary end-point | Completion date |
| Nintedanib | ||||||
| NCT03805477 | BOS after HSCT (40) | II | Open-label | Nintedanib 150 mg twice daily | Adverse events leading to treatment interruption or discontinuation | Feb 2021 |
| NCT03283007 (INFINITY study) | Grade 1–2 BOS after LTx (80) | III | Randomised, quadruple-blind | Nintedanib 150 mg twice daily versus placebo | Reduction in the rate of FEV1 decline from baseline to month 6 | Jun 2023 |
| Pirfenidone | ||||||
| NCT03315741 | BOS after HSCT (30) | I | Open-label | Pirfenidone ≤2403 mg·day−1 | Number of patients requiring a dose reduction for >21 days due to adverse events | Feb 2022 |
| NCT03473340 (STOP-CLAD) | CLAD after LTx (60) | II | Randomised, double-blind | Pirfenidone 801–2403 mg·day−1 versus placebo | Per cent change in functional small airways disease as measured by parametric response mapping (HRCT) at week 24 | Mar 2022 |
| NCT02262299 (EPOS) | Grade 1–3 BOS after LTx (90) | II/III | Randomised, double-blind | Pirfenidone 801–2403 mg·day−1 versus placebo | Change in FEV1 decline from baseline to month 6 | Dec 2019 |
BOS: bronchiolitis obliterans syndrome; HSCT: haematopoietic stem cell transplant; LTx: lung transplantation; FEV1: forced expiratory volume in 1 s; CLAD: chronic lung allograft dysfunction; HRCT: high-resolution computed tomography.
Conclusions
Management strategies for BOS after lung transplantation or HSCT are similar, although a key difference is the need to manage systemic GVHD in HSCT patients. Currently, treatment options for either condition are limited and novel treatments are urgently needed, particularly treatments which not only slow or stabilise disease progression but also potentially reverse the pathological changes and thereby provide clinically meaningful survival and quality of life outcomes.
Supplementary material
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material 00185-2022.SUPPLEMENT (308.7KB, pdf)
Acknowledgements
We would like to thank Catherine Rees of Springer Healthcare Communications who wrote the outline and first draft of this manuscript. This medical writing assistance was funded by Zambon.
Provenance: Submitted article, peer reviewed.
Author contributions: A.R. Glanville contributed to the concept and design of the manuscript, chaired the panel discussion, and manuscript drafting and approval. All authors contributed to the panel discussion and manuscript review.
Conflict of interest: A.R. Glanville has received an honorarium for an advisory role from Zambon and is chair of the Zambon DSMB for the Boston trials.
Conflict of interest: G.M. Verleden has received an honorarium for an advisory role from Zambon.
Conflict of interest: M. Perch has received an honorarium for an advisory role from Zambon, a research grant (institutional) from Roche, speaker fees from Novartis, GSK and Therakos, and other financial support from Boehringer.
Conflict of interest: E.D. Lease has received an honorarium for an advisory role from Zambon.
Conflict of interest: G-S. Cheng has received an honorarium for an advisory role from Zambon.
Conflict of interest: A. Bergeron has received an honorarium for an advisory role from Zambon.
Conflict of interest: C. Benden has received an honorarium for an advisory role from Zambon and speaker fees from Therakos.
Conflict of interest: J. Gottlieb has received an honorarium for advisory roles from Zambon, research grants from Zambon and Deutsche Forschungsgemeinschaft, and advisory funding from Theravance, Merck and Altara.
Conflict of interest: J.L. Todd has received an honorarium for an advisory role from Zambon, Altavant and Natera, and research grants (institutional) from Boehringer Ingelheim, AstraZeneca and CareDx.
Conflict of interest: K.M. Williams has received an honorarium for an advisory role from Zambon.
Support statement: Zambon provided financial support for the expert panel meeting and manuscript development. Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Kotloff RM, Ahya VN, Crawford SW. Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2004; 170: 22–48. doi: 10.1164/rccm.200309-1322SO [DOI] [PubMed] [Google Scholar]
- 2.Krishna R, Anjum F, Oliver TI. Bronchiolitis Obliterans. Treasure Island, FL, StatPearls, 2020. [PubMed] [Google Scholar]
- 3.Verleden GM, Glanville AR, Lease ED, et al. Chronic lung allograft dysfunction: definition, diagnostic criteria, and approaches to treatment – a consensus report from the Pulmonary Council of the ISHLT. J Heart Lung Transplant 2019; 38: 493–503. doi: 10.1016/j.healun.2019.03.009 [DOI] [PubMed] [Google Scholar]
- 4.Verleden SE, Vasilescu DM, Willems S, et al. The site and nature of airway obstruction after lung transplantation. Am J Respir Crit Care Med 2014; 189: 292–300. doi: 10.1164/rccm.201310-1894OC [DOI] [PubMed] [Google Scholar]
- 5.Williams KM, Chien JW, Gladwin MT, et al. Bronchiolitis obliterans after allogeneic hematopoietic stem cell transplantation. JAMA 2009; 302: 306–314. doi: 10.1001/jama.2009.1018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barker AF, Bergeron A, Rom WN, et al. Obliterative bronchiolitis. N Engl J Med 2014; 370: 1820–1828. doi: 10.1056/NEJMra1204664 [DOI] [PubMed] [Google Scholar]
- 7.Bergeron A, Cheng GS. Bronchiolitis obliterans syndrome and other late pulmonary complications after allogeneic hematopoietic stem cell transplantation. Clin Chest Med 2017; 38: 607–621. doi: 10.1016/j.ccm.2017.07.003 [DOI] [PubMed] [Google Scholar]
- 8.Turton CW, Williams G, Green M. Cryptogenic obliterative bronchiolitis in adults. Thorax 1981; 36: 805–810. doi: 10.1136/thx.36.11.805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Burke CM, Theodore J, Dawkins KD, et al. Post-transplant obliterative bronchiolitis and other late lung sequelae in human heart-lung transplantation. Chest 1984; 86: 824–829. doi: 10.1378/chest.86.6.824 [DOI] [PubMed] [Google Scholar]
- 10.Ralph DD, Springmeyer SC, Sullivan KM, et al. Rapidly progressive air-flow obstruction in marrow transplant recipients. Possible association between obliterative bronchiolitis and chronic graft-versus-host disease. Am Rev Respir Dis 1984; 129: 641–644. [PubMed] [Google Scholar]
- 11.Kulkarni HS, Cherikh WS, Chambers DC, et al. Bronchiolitis obliterans syndrome-free survival after lung transplantation: an International Society for Heart and Lung Transplantation Thoracic Transplant Registry analysis. J Heart Lung Transplant 2019; 38: 5–16. doi: 10.1016/j.healun.2018.09.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nykanen A, Raivio P, Perakyla L, et al. Incidence and impact of chronic lung allograft dysfunction after lung transplantation – single-center 14-year experience. Scand Cardiovasc J 2020; 54: 192–199. doi: 10.1080/14017431.2020.1726444 [DOI] [PubMed] [Google Scholar]
- 13.Thabut G, Mal H. Outcomes after lung transplantation. J Thorac Dis 2017; 9: 2684–2691. doi: 10.21037/jtd.2017.07.85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kwok WC, Liang BM, Lui MMS, et al. Rapid versus gradual lung function decline in bronchiolitis obliterans syndrome after haematopoietic stem cell transplantation is associated with survival outcome. Respirology 2019; 24: 459–466. doi: 10.1111/resp.13472 [DOI] [PubMed] [Google Scholar]
- 15.Ciki K, Dogru D, Kuskonmaz B, et al. Pulmonary complications following hematopoietic stem cell transplantation in children. Turk J Pediatr 2019; 61: 59–60. doi: 10.24953/turkjped.2019.01.010 [DOI] [PubMed] [Google Scholar]
- 16.Diab M, ZazaDitYafawi J, Soubani AO. Major pulmonary complications after hematopoietic stem cell transplant. Exp Clin Transplant 2016; 14: 259–270. [DOI] [PubMed] [Google Scholar]
- 17.Gazourian L, Rogers AJ, Ibanga R, et al. Factors associated with bronchiolitis obliterans syndrome and chronic graft-versus-host disease after allogeneic hematopoietic cell transplantation. Am J Hematol 2014; 89: 404–409. doi: 10.1002/ajh.23656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pham J, Rangaswamy J, Avery S, et al. Updated prevalence, predictors and treatment outcomes for bronchiolitis obliterans syndrome after allogeneic stem cell transplantation. Respir Med 2020; 177: 106286. doi: 10.1016/j.rmed.2020.106286 [DOI] [PubMed] [Google Scholar]
- 19.Walther S, Rettinger E, Maurer HM, et al. Long-term pulmonary function testing in pediatric bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Pediatr Pulmonol 2020; 55: 1725–1735. doi: 10.1002/ppul.24801 [DOI] [PubMed] [Google Scholar]
- 20.Arai S, Arora M, Wang T, et al. Increasing incidence of chronic graft-versus-host disease in allogeneic transplantation: a report from the Center for International Blood and Marrow Transplant Research. Biol Blood Marrow Transplant 2015; 21: 266–274. doi: 10.1016/j.bbmt.2014.10.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Au BK, Au MA, Chien JW. Bronchiolitis obliterans syndrome epidemiology after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2011; 17: 1072–1078. doi: 10.1016/j.bbmt.2010.11.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Finlen Copeland CA, Snyder LD, Zaas DW, et al. Survival after bronchiolitis obliterans syndrome among bilateral lung transplant recipients. Am J Respir Crit Care Med 2010; 182: 784–789. doi: 10.1164/rccm.201002-0211OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rhee CK, Ha JH, Yoon JH, et al. Risk factor and clinical outcome of bronchiolitis obliterans syndrome after allogeneic hematopoietic stem cell transplantation. Yonsei Med J 2016; 57: 365–372. doi: 10.3349/ymj.2016.57.2.365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Meyer KC, Raghu G, Verleden GM, et al. An international ISHLT/ATS/ERS clinical practice guideline: diagnosis and management of bronchiolitis obliterans syndrome. Eur Respir J 2014; 44: 1479–1503. doi: 10.1183/09031936.00107514 [DOI] [PubMed] [Google Scholar]
- 25.Glanville AR, Verleden GM, Todd JL, et al. Chronic lung allograft dysfunction: definition and update of restrictive allograft syndrome – a consensus report from the Pulmonary Council of the ISHLT. J Heart Lung Transplant 2019; 38: 483–492. doi: 10.1016/j.healun.2019.03.008 [DOI] [PubMed] [Google Scholar]
- 26.Sato M, Waddell TK, Wagnetz U, et al. Restrictive allograft syndrome (RAS): a novel form of chronic lung allograft dysfunction. J Heart Lung Transplant 2011; 30: 735–742. doi: 10.1016/j.healun.2011.01.712 [DOI] [PubMed] [Google Scholar]
- 27.Verleden GM, Vos R, Verleden SE, et al. Survival determinants in lung transplant patients with chronic allograft dysfunction. Transplantation 2011; 92: 703–708. doi: 10.1097/TP.0b013e31822bf790 [DOI] [PubMed] [Google Scholar]
- 28.Woodrow JP, Shlobin OA, Barnett SD, et al. Comparison of bronchiolitis obliterans syndrome to other forms of chronic lung allograft dysfunction after lung transplantation. J Heart Lung Transplant 2010; 29: 1159–1164. doi: 10.1016/j.healun.2010.05.012 [DOI] [PubMed] [Google Scholar]
- 29.Levy L, Huszti E, Renaud-Picard B, et al. Risk assessment of chronic lung allograft dysfunction phenotypes: validation and proposed refinement of the 2019 International Society for Heart and Lung Transplantation classification system. J Heart Lung Transplant 2020; 39: 761–770. doi: 10.1016/j.healun.2020.04.012 [DOI] [PubMed] [Google Scholar]
- 30.Kneidinger N, Milger K, Janitza S, et al. Lung volumes predict survival in patients with chronic lung allograft dysfunction. Eur Respir J 2017; 49: 1601315. doi: 10.1183/13993003.01315-2016 [DOI] [PubMed] [Google Scholar]
- 31.Moher Alsady T, Voskrebenzev A, Greer M, et al. MRI-derived regional flow-volume loop parameters detect early-stage chronic lung allograft dysfunction. J Magn Reson Imaging 2019; 50: 1873–1882. doi: 10.1002/jmri.26799 [DOI] [PubMed] [Google Scholar]
- 32.Van Herck A, Verleden SE, Sacreas A, et al. Validation of a post-transplant chronic lung allograft dysfunction classification system. J Heart Lung Transplant 2019; 38: 166–173. doi: 10.1016/j.healun.2018.09.020 [DOI] [PubMed] [Google Scholar]
- 33.Verleden SE, Von Der Thusen J, Van Herck A, et al. Identification and characterization of chronic lung allograft dysfunction patients with mixed phenotype: a single-center study. Clin Transplant 2020; 34: e13781. doi: 10.1111/ctr.13781 [DOI] [PubMed] [Google Scholar]
- 34.Peghin M, Los-Arcos I, Hirsch HH, et al. Community-acquired respiratory viruses are a risk factor for chronic lung allograft dysfunction. Clin Infect Dis 2019; 69: 1192–1197. doi: 10.1093/cid/ciy1047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Verleden SE, Ruttens D, Vandermeulen E, et al. Bronchiolitis obliterans syndrome and restrictive allograft syndrome: do risk factors differ? Transplantation 2013; 95: 1167–1172. doi: 10.1097/TP.0b013e318286e076 [DOI] [PubMed] [Google Scholar]
- 36.Verleden SE, Vanaudenaerde BM, Emonds MP, et al. Donor-specific and -nonspecific HLA antibodies and outcome post lung transplantation. Eur Respir J 2017; 50: 1701248. doi: 10.1183/13993003.01248-2017 [DOI] [PubMed] [Google Scholar]
- 37.Sugimoto S, Yamamoto H, Kurosaki T, et al. Impact of chronic lung allograft dysfunction, especially restrictive allograft syndrome, on the survival after living-donor lobar lung transplantation compared with cadaveric lung transplantation in adults: a single-center experience. Surg Today 2019; 49: 686–693. doi: 10.1007/s00595-019-01782-0 [DOI] [PubMed] [Google Scholar]
- 38.Jamani K, He Q, Liu Y, et al. Early post-transplantation spirometry is associated with the development of bronchiolitis obliterans syndrome after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2020; 26: 943–948. doi: 10.1016/j.bbmt.2019.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bergeron A, Chevret S, Peffault de Latour R, et al. Noninfectious lung complications after allogeneic haematopoietic stem cell transplantation. Eur Respir J 2018; 51: 1702617. doi: 10.1183/13993003.02617-2017 [DOI] [PubMed] [Google Scholar]
- 40.Gronningsaeter IS, Tsykunova G, Lilleeng K, et al. Bronchiolitis obliterans syndrome in adults after allogeneic stem cell transplantation-pathophysiology, diagnostics and treatment. Expert Rev Clin Immunol 2017; 13: 553–569. doi: 10.1080/1744666X.2017.1279053 [DOI] [PubMed] [Google Scholar]
- 41.Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 2015; 21: 389–401.e381. doi: 10.1016/j.bbmt.2014.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cuvelier GDE, Nemecek ER, Wahlstrom JT, et al. Benefits and challenges with diagnosing chronic and late acute GVHD in children using the NIH consensus criteria. Blood 2019; 134: 304–316. doi: 10.1182/blood.2019000216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chambers DC. Bronchiolitis obliterans syndrome 'endotypes’ in haematopoietic stem cell transplantation. Respirology 2019; 24: 408–409. doi: 10.1111/resp.13498 [DOI] [PubMed] [Google Scholar]
- 44.Bergeron A, Godet C, Chevret S, et al. Bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: phenotypes and prognosis. Bone Marrow Transplant 2013; 48: 819–824. doi: 10.1038/bmt.2012.241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cheng GS, Storer B, Chien JW, et al. Lung function trajectory in bronchiolitis obliterans syndrome after allogeneic hematopoietic cell transplant. Ann Am Thorac Soc 2016; 13: 1932–1939. doi: 10.1513/AnnalsATS.201604-262OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Clark JG, Crawford SW, Madtes DK, et al. Obstructive lung disease after allogeneic marrow transplantation. Clinical presentation and course. Ann Intern Med 1989; 111: 368–376. doi: 10.7326/0003-4819-111-5-368 [DOI] [PubMed] [Google Scholar]
- 47.Yomota M, Yanagawa N, Sakai F, et al. Association between chronic bacterial airway infection and prognosis of bronchiolitis obliterans syndrome after hematopoietic cell transplantation. Medicine 2019; 98: e13951. doi: 10.1097/MD.0000000000013951 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Belperio J, Palmer SM, Weigt SS. Host-pathogen interactions and chronic lung allograft dysfunction. Ann Am Thorac Soc 2017; 14: S242–S246. doi: 10.1513/AnnalsATS.201606-464MG [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gregson AL. Infectious triggers of chronic lung allograft dysfunction. Curr Infect Dis Rep 2016; 18: 21. doi: 10.1007/s11908-016-0529-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Royer PJ, Olivera-Botello G, Koutsokera A, et al. Chronic lung allograft dysfunction: a systematic review of mechanisms. Transplantation 2016; 100: 1803–1814. doi: 10.1097/TP.0000000000001215 [DOI] [PubMed] [Google Scholar]
- 51.Misumi K, Wheeler DS, Aoki Y, et al. Humoral immune responses mediate the development of a restrictive phenotype of chronic lung allograft dysfunction. JCI Insight 2020; 5: e136533. doi: 10.1172/jci.insight.136533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Vanstapel A, Verleden SE, Weynand B, et al. Late-onset ‘acute fibrinous and organising pneumonia’ impairs long-term lung allograft function and survival. Eur Respir J 2020; 56: 1902292. doi: 10.1183/13993003.02292-2019 [DOI] [PubMed] [Google Scholar]
- 53.von der Thusen JH, Vandermeulen E, Vos R, et al. The histomorphological spectrum of restrictive chronic lung allograft dysfunction and implications for prognosis. Mod Pathol 2018; 31: 780–790. doi: 10.1038/modpathol.2017.180 [DOI] [PubMed] [Google Scholar]
- 54.Cooke KR, Luznik L, Sarantopoulos S, et al. The biology of chronic graft-versus-host disease: a task force report from the National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease. Biol Blood Marrow Transplant 2017; 23: 211–234. doi: 10.1016/j.bbmt.2016.09.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Holbro A, Lehmann T, Girsberger S, et al. Lung histology predicts outcome of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013; 19: 973–980. doi: 10.1016/j.bbmt.2013.03.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Meignin V, Thivolet-Bejui F, Kambouchner M, et al. Lung histopathology of non-infectious pulmonary complications after allogeneic haematopoietic stem cell transplantation. Histopathology 2018; 73: 832–842. doi: 10.1111/his.13697 [DOI] [PubMed] [Google Scholar]
- 57.Verleden SE, McDonough JE, Schoemans H, et al. Phenotypical diversity of airway morphology in chronic lung graft vs. host disease after stem cell transplantation. Mod Pathol 2019; 32: 817–829. doi: 10.1038/s41379-019-0203-2 [DOI] [PubMed] [Google Scholar]
- 58.Kugler C, Fuehner T, Dierich M, et al. Effect of adherence to home spirometry on bronchiolitis obliterans and graft survival after lung transplantation. Transplantation 2009; 88: 129–134. doi: 10.1097/TP.0b013e3181aad129 [DOI] [PubMed] [Google Scholar]
- 59.Tian D, Huang H, Wen HY. Noninvasive methods for detection of chronic lung allograft dysfunction in lung transplantation. Transplant Rev 2020; 34: 100547. doi: 10.1016/j.trre.2020.100547 [DOI] [PubMed] [Google Scholar]
- 60.Williams KM. How I treat bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Blood 2017; 129: 448–455. doi: 10.1182/blood-2016-08-693507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kavaliunaite E, Aurora P. Diagnosing and managing bronchiolitis obliterans in children. Expert Rev Respir Med 2019; 13: 481–488. doi: 10.1080/17476348.2019.1586537 [DOI] [PubMed] [Google Scholar]
- 62.Dugger DT, Fung M, Hays SR, et al. Chronic lung allograft dysfunction small airways reveal a lymphocytic inflammation gene signature. Am J Transplant 2021; 21: 362–371. doi: 10.1111/ajt.16293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bankier AA, Van Muylem A, Knoop C, et al. Bronchiolitis obliterans syndrome in heart-lung transplant recipients: diagnosis with expiratory CT. Radiology 2001; 218: 533–539. doi: 10.1148/radiology.218.2.r01fe09533 [DOI] [PubMed] [Google Scholar]
- 64.Gunn ML, Godwin JD, Kanne JP, et al. High-resolution CT findings of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. J Thorac Imaging 2008; 23: 244–250. doi: 10.1097/RTI.0b013e3181809df0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Galban CJ, Boes JL, Bule M, et al. Parametric response mapping as an indicator of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014; 20: 1592–1598. doi: 10.1016/j.bbmt.2014.06.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Verleden SE, Vos R, Vandermeulen E, et al. Parametric response mapping of bronchiolitis obliterans syndrome progression after lung transplantation. Am J Transplant 2016; 16: 3262–3269. doi: 10.1111/ajt.13945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Fishman EF, Quirk JD, Sweet SC, et al. What makes a good pediatric transplant lung: insights from in vivo lung morphometry with hyperpolarized 3He magnetic resonance imaging. Pediatr Transplant 2017; 21: e12886. doi: 10.1111/petr.12886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Renne J, Lauermann P, Hinrichs JB, et al. Chronic lung allograft dysfunction: oxygen-enhanced T1-mapping MR imaging of the lung. Radiology 2015; 276: 266–273. doi: 10.1148/radiol.15141486 [DOI] [PubMed] [Google Scholar]
- 69.Keller M, Agbor-Enoh S. Donor-derived cell-free DNA for acute rejection monitoring in heart and lung transplantation. Curr Transplant Rep 2021; 8: 351–358. doi: 10.1007/s40472-021-00349-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sharifi H, Lai YK, Guo H, et al. Machine learning algorithms to differentiate among pulmonary complications after hematopoietic cell transplant. Chest 2020; 158: 1090–1103. doi: 10.1016/j.chest.2020.02.076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ross DJ, Lewis MI, Kramer M, et al. FK 506 ‘rescue’ immunosuppression for obliterative bronchiolitis after lung transplantation. Chest 1997; 112: 1175–1179. doi: 10.1378/chest.112.5.1175 [DOI] [PubMed] [Google Scholar]
- 72.Treede H, Glanville AR, Klepetko W, et al. Tacrolimus and cyclosporine have differential effects on the risk of development of bronchiolitis obliterans syndrome: results of a prospective, randomized international trial in lung transplantation. J Heart Lung Transplant 2012; 31: 797–804. doi: 10.1016/j.healun.2012.03.008 [DOI] [PubMed] [Google Scholar]
- 73.Borro JM, Bravo C, Sole A, et al. Conversion from cyclosporine to tacrolimus stabilizes the course of lung function in lung transplant recipients with bronchiolitis obliterans syndrome. Transplant Proc 2007; 39: 2416–2419. doi: 10.1016/j.transproceed.2007.06.071 [DOI] [PubMed] [Google Scholar]
- 74.Gerhardt SG, McDyer JF, Girgis RE, et al. Maintenance azithromycin therapy for bronchiolitis obliterans syndrome: results of a pilot study. Am J Respir Crit Care Med 2003; 168: 121–125. doi: 10.1164/rccm.200212-1424BC [DOI] [PubMed] [Google Scholar]
- 75.Gottlieb J, Szangolies J, Koehnlein T, et al. Long-term azithromycin for bronchiolitis obliterans syndrome after lung transplantation. Transplantation 2008; 85: 36–41. doi: 10.1097/01.tp.0000295981.84633.bc [DOI] [PubMed] [Google Scholar]
- 76.Jain R, Hachem RR, Morrell MR, et al. Azithromycin is associated with increased survival in lung transplant recipients with bronchiolitis obliterans syndrome. J Heart Lung Transplant 2010; 29: 531–537. doi: 10.1016/j.healun.2009.12.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Li D, Duan Q, Weinkauf J, et al. Azithromycin prophylaxis after lung transplantation is associated with improved overall survival. J Heart Lung Transplant 2020; 39: 1426–1434. doi: 10.1016/j.healun.2020.09.006 [DOI] [PubMed] [Google Scholar]
- 78.Verleden GM, Dupont LJ. Azithromycin therapy for patients with bronchiolitis obliterans syndrome after lung transplantation. Transplantation 2004; 77: 1465–1467. doi: 10.1097/01.TP.0000122412.80864.43 [DOI] [PubMed] [Google Scholar]
- 79.Verleden GM, Vanaudenaerde BM, Dupont LJ, et al. Azithromycin reduces airway neutrophilia and interleukin-8 in patients with bronchiolitis obliterans syndrome. Am J Respir Crit Care Med 2006; 174: 566–570. doi: 10.1164/rccm.200601-071OC [DOI] [PubMed] [Google Scholar]
- 80.Yates B, Murphy DM, Forrest IA, et al. Azithromycin reverses airflow obstruction in established bronchiolitis obliterans syndrome. Am J Respir Crit Care Med 2005; 172: 772–775. doi: 10.1164/rccm.200411-1537OC [DOI] [PubMed] [Google Scholar]
- 81.Vos R, Vanaudenaerde BM, Ottevaere A, et al. Long-term azithromycin therapy for bronchiolitis obliterans syndrome: divide and conquer? J Heart Lung Transplant 2010; 29: 1358–1368. doi: 10.1016/j.healun.2010.05.023 [DOI] [PubMed] [Google Scholar]
- 82.Corris PA, Ryan VA, Small T, et al. A randomised controlled trial of azithromycin therapy in bronchiolitis obliterans syndrome (BOS) post lung transplantation. Thorax 2015; 70: 442–450. doi: 10.1136/thoraxjnl-2014-205998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Vos R, Eynde RV, Ruttens D, et al. Montelukast in chronic lung allograft dysfunction after lung transplantation. J Heart Lung Transplant 2019; 38: 516–527. doi: 10.1016/j.healun.2018.11.014 [DOI] [PubMed] [Google Scholar]
- 84.Ruttens D, Verleden SE, Demeyer H, et al. Montelukast for bronchiolitis obliterans syndrome after lung transplantation: a randomized controlled trial. PLoS ONE 2018; 13: e0193564. doi: 10.1371/journal.pone.0193564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Verleden GM, Verleden SE, Vos R, et al. Montelukast for bronchiolitis obliterans syndrome after lung transplantation: a pilot study. Transpl Int 2011; 24: 651–656. doi: 10.1111/j.1432-2277.2011.01248.x [DOI] [PubMed] [Google Scholar]
- 86.Chauhan BF, Jeyaraman MM, Singh Mann A, et al. Addition of anti-leukotriene agents to inhaled corticosteroids for adults and adolescents with persistent asthma. Cochrane Database Syst Rev 2017; 3: CD010347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Tu ZL, Zhou ZY, Xu HC, et al. LTB4 and montelukast in transplantation-related bronchiolitis obliterans in rats. J Cardiothorac Surg 2017; 12: 43. doi: 10.1186/s13019-017-0605-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Morrell MR, Despotis GJ, Lublin DM, et al. The efficacy of photopheresis for bronchiolitis obliterans syndrome after lung transplantation. J Heart Lung Transplant 2010; 29: 424–431. doi: 10.1016/j.healun.2009.08.029 [DOI] [PubMed] [Google Scholar]
- 89.Greer M, Dierich M, De Wall C, et al. Phenotyping established chronic lung allograft dysfunction predicts extracorporeal photopheresis response in lung transplant patients. Am J Transplant 2013; 13: 911–918. doi: 10.1111/ajt.12155 [DOI] [PubMed] [Google Scholar]
- 90.Benden C, Haughton M, Leonard S, et al. Therapy options for chronic lung allograft dysfunction-bronchiolitis obliterans syndrome following first-line immunosuppressive strategies: a systematic review. J Heart Lung Transplant 2017; 36: 921–933. doi: 10.1016/j.healun.2017.05.030 [DOI] [PubMed] [Google Scholar]
- 91.Fisher AJ, Rutherford RM, Bozzino J, et al. The safety and efficacy of total lymphoid irradiation in progressive bronchiolitis obliterans syndrome after lung transplantation. Am J Transplant 2005; 5: 537–543. doi: 10.1111/j.1600-6143.2004.00709.x [DOI] [PubMed] [Google Scholar]
- 92.Verleden GM, Lievens Y, Dupont LJ, et al. Efficacy of total lymphoid irradiation in azithromycin nonresponsive chronic allograft rejection after lung transplantation. Transplant Proc 2009; 41: 1816–1820. doi: 10.1016/j.transproceed.2009.03.070 [DOI] [PubMed] [Google Scholar]
- 93.Penack O, Marchetti M, Ruutu T, et al. Prophylaxis and management of graft versus host disease after stem-cell transplantation for haematological malignancies: updated consensus recommendations of the European Society for Blood and Marrow Transplantation. Lancet Haematol 2020; 7: e157–e167. doi: 10.1016/S2352-3026(19)30256-X [DOI] [PubMed] [Google Scholar]
- 94.Williams KM, Cheng GS, Pusic I, et al. Fluticasone, azithromycin, and montelukast treatment for new-onset bronchiolitis obliterans syndrome after hematopoietic cell transplantation. Biol Blood Marrow Transplant 2016; 22: 710–716. doi: 10.1016/j.bbmt.2015.10.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Yadav H, Peters SG, Keogh KA, et al. Azithromycin for the treatment of obliterative bronchiolitis after hematopoietic stem cell transplantation: a systematic review and meta-analysis. Biol Blood Marrow Transplant 2016; 22: 2264–2269. doi: 10.1016/j.bbmt.2016.08.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Bergeron A, Chevret S, Granata A, et al. Effect of azithromycin on airflow decline-free survival after allogeneic hematopoietic stem cell transplant: the ALLOZITHRO randomized clinical trial. JAMA 2017; 318: 557–566. doi: 10.1001/jama.2017.9938 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cheng GS, Bondeelle L, Gooley T, et al. Azithromycin use and increased cancer risk among patients with bronchiolitis obliterans after hematopoietic cell transplantation. Biol Blood Marrow Transplant 2020; 26: 392–400. doi: 10.1016/j.bbmt.2019.10.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bergeron A, Chevret S, Chagnon K, et al. Budesonide/formoterol for bronchiolitis obliterans after hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2015; 191: 1242–1249. doi: 10.1164/rccm.201410-1818OC [DOI] [PubMed] [Google Scholar]
- 99.Tran J, Norder EE, Diaz PT, et al. Pulmonary rehabilitation for bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2012; 18: 1250–1254. doi: 10.1016/j.bbmt.2012.01.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hefazi M, Langer KJ, Khera N, et al. Extracorporeal photopheresis improves survival in hematopoietic cell transplant patients with bronchiolitis obliterans syndrome without significantly impacting measured pulmonary functions. Biol Blood Marrow Transplant 2018; 24: 1906–1913. doi: 10.1016/j.bbmt.2018.04.012 [DOI] [PubMed] [Google Scholar]
- 101.US Food and Drug Administration . Imbruvica (ibrutinib). Prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2020/205552s033,210563s010lbl.pdf. Date last updated: December 2020. Date last accessed: 5 April 2022.
- 102.US Food and Drug Administration . Jakafi (ruxolitinib). Prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2020/202192Orig1s019Rpllbl.pdf. Date last updated: January 2020. Date last accessed: 5 April 2022.
- 103.Yanik GA, Mineishi S, Levine JE, et al. Soluble tumor necrosis factor receptor: Enbrel (etanercept) for subacute pulmonary dysfunction following allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2012; 18: 1044–1054. doi: 10.1016/j.bbmt.2011.11.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Greer M, Berastegui C, Jaksch P, et al. Lung transplantation after allogeneic stem cell transplantation: a pan-European experience. Eur Respir J 2018; 51: 1701330. doi: 10.1183/13993003.01330-2017 [DOI] [PubMed] [Google Scholar]
- 105.Burke CM, Glanville AR, Theodore J, et al. Lung immunogenicity, rejection, and obliterative bronchiolitis. Chest 1987; 92: 547–549. doi: 10.1378/chest.92.3.547 [DOI] [PubMed] [Google Scholar]
- 106.Glanville AR, Tazelaar HD, Theodore J, et al. The distribution of MHC class I and II antigens on bronchial epithelium. Am Rev Respir Dis 1989; 139: 330–334. doi: 10.1164/ajrccm/139.2.330 [DOI] [PubMed] [Google Scholar]
- 107.Taylor PM, Rose ML, Yacoub MH. Expression of MHC antigens in normal human lungs and transplanted lungs with obliterative bronchiolitis. Transplantation 1989; 48: 506–510. doi: 10.1097/00007890-198909000-00030 [DOI] [PubMed] [Google Scholar]
- 108.Levine DJ, Glanville AR, Aboyoun C, et al. Antibody-mediated rejection of the lung: a consensus report of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant 2016; 35: 397–406. doi: 10.1016/j.healun.2016.01.1223 [DOI] [PubMed] [Google Scholar]
- 109.Tikkanen JM, Singer LG, Kim SJ, et al. De novo DQ donor-specific antibodies are associated with chronic lung allograft dysfunction after lung transplantation. Am J Respir Crit Care Med 2016; 194: 596–606. doi: 10.1164/rccm.201509-1857OC [DOI] [PubMed] [Google Scholar]
- 110.Luckraz H, Goddard M, McNeil K, et al. Microvascular changes in small airways predispose to obliterative bronchiolitis after lung transplantation. J Heart Lung Transplant 2004; 23: 527–531. doi: 10.1016/j.healun.2003.07.003 [DOI] [PubMed] [Google Scholar]
- 111.Glanville AR, Aboyoun CL, Havryk A, et al. Severity of lymphocytic bronchiolitis predicts long-term outcome after lung transplantation. Am J Respir Crit Care Med 2008; 177: 1033–1040. doi: 10.1164/rccm.200706-951OC [DOI] [PubMed] [Google Scholar]
- 112.Blondeau K, Mertens V, Vanaudenaerde BA, et al. Gastro-oesophageal reflux and gastric aspiration in lung transplant patients with or without chronic rejection. Eur Respir J 2008; 31: 707–713. doi: 10.1183/09031936.00064807 [DOI] [PubMed] [Google Scholar]
- 113.Emtiazjoo AM, Wilkes DS. Humoral immunity and the development of obliterative bronchiolitis after lung transplantation: is there a link? Am J Respir Cell Mol Biol 2013; 48: 145–149. doi: 10.1165/rcmb.2012-0349RT [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Milross L, Hachem R, Levine D, et al. Lung autoantibodies: Ready for prime time? J Heart Lung Transplant 2017; 37: 179–181. doi: 10.1016/j.healun.2017.10.026 [DOI] [PubMed] [Google Scholar]
- 115.Verleden SE, Scheers H, Nawrot TS, et al. Lymphocytic bronchiolitis after lung transplantation is associated with daily changes in air pollution. Am J Transplant 2012; 12: 1831–1838. doi: 10.1111/j.1600-6143.2012.04134.x [DOI] [PubMed] [Google Scholar]
- 116.Behr J, Zimmermann G, Baumgartner R, et al. Lung deposition of a liposomal cyclosporine A inhalation solution in patients after lung transplantation. J Aerosol Med Pulm Drug Deliv 2009; 22: 121–130. doi: 10.1089/jamp.2008.0714 [DOI] [PubMed] [Google Scholar]
- 117.AdisInsight. Drug profile: Ciclosporin liposomal – Breath Therapeutics. 30 April 2020. Springer Nature, Berlin, 2020. [Google Scholar]
- 118.Iacono A, Wijesinha M, Rajagopal K, et al. A randomised single-centre trial of inhaled liposomal cyclosporine for bronchiolitis obliterans syndrome post-lung transplantation. ERJ Open Res 2019; 5: 00167–2019. doi: 10.1183/23120541.00167-2019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Hosseini A, Gharibi T, Marofi F, et al. Janus kinase inhibitors: a therapeutic strategy for cancer and autoimmune diseases. J Cell Physiol 2020; 235: 5903–5924. doi: 10.1002/jcp.29593 [DOI] [PubMed] [Google Scholar]
- 120.Streiler C, Shaikh F, Davis C, et al. Ruxolitinib is an effective steroid sparing agent in bronchiolitis obliterans due to chronic graft-versus-host-disease. Bone Marrow Transplant 2020; 55: 1194–1196. doi: 10.1038/s41409-019-0662-6 [DOI] [PubMed] [Google Scholar]
- 121.Schoettler M, Duncan C, Lehmann L, et al. Ruxolitinib is an effective steroid sparing agent in children with steroid refractory/dependent bronchiolitis obliterans syndrome after allogenic hematopoietic cell transplantation. Bone Marrow Transplant 2019; 54: 1158–1160. doi: 10.1038/s41409-019-0450-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Uygun V, Karasu G, Daloglu H, et al. Ruxolitinib salvage therapy is effective for steroid-refractory graft-versus-host disease in children: a single-center experience. Pediatr Blood Cancer 2020; 67: e28190. doi: 10.1002/pbc.28190 [DOI] [PubMed] [Google Scholar]
- 123.Meng GQ, Wang YN, Wang JS, et al. Ruxolitinib treatment for bronchiolitis obliterans syndrome following hematopoietic stem cell transplant in a patient with primary hemophagocytic lymphohistiocytosis. Chin Med J 2021; 134: 1624–1625. doi: 10.1097/CM9.0000000000001324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.McManigle W, Youssef A, Sarantopoulos S. B cells in chronic graft-versus-host disease. Hum Immunol 2019; 80: 393–399. doi: 10.1016/j.humimm.2019.03.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Sarantopoulos S, Stevenson KE, Kim HT, et al. Altered B-cell homeostasis and excess BAFF in human chronic graft-versus-host disease. Blood 2009; 113: 3865–3874. doi: 10.1182/blood-2008-09-177840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Smirnova NF, Conlon TM, Morrone C, et al. Inhibition of B cell-dependent lymphoid follicle formation prevents lymphocytic bronchiolitis after lung transplantation. JCI Insight 2019; 4: e123971. doi: 10.1172/jci.insight.123971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.US Food and Drug Administration . Rituxan (rituximab). Prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2020/103705s5460lbl.pdf Date last updated: September 2019. Date last accessed: 5 April 2022.
- 128.US Food and Drug Administration . Campath (alemtuzumab). Prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2020/103948s5181lbl.pdf. Date last updated: October 2020. Date last accessed: 5 April 2022.
- 129.Brownback KR, Thomas LA, McGuirk JP, et al. Effect of rituximab on pulmonary function in bronchiolitis obliterans syndrome due to graft-versus-host-disease. Lung 2017; 195: 781–788. doi: 10.1007/s00408-017-0051-0 [DOI] [PubMed] [Google Scholar]
- 130.Yamanashi K, Chen-Yoshikawa TF, Hamaji M, et al. Outcomes of combination therapy including rituximab for antibody-mediated rejection after lung transplantation. Gen Thorac Cardiovasc Surg 2020; 68: 142–149. doi: 10.1007/s11748-019-01189-1 [DOI] [PubMed] [Google Scholar]
- 131.Lorillon G, Robin M, Meignin V, et al. Rituximab in bronchiolitis obliterans after haematopoietic stem cell transplantation. Eur Respir J 2011; 38: 470–472. doi: 10.1183/09031936.00003711 [DOI] [PubMed] [Google Scholar]
- 132.Furuya Y, Jayarajan SN, Taghavi S, et al. The impact of alemtuzumab and basiliximab induction on patient survival and time to bronchiolitis obliterans syndrome in double lung transplantation recipients. Am J Transplant 2016; 16: 2334–2341. doi: 10.1111/ajt.13739 [DOI] [PubMed] [Google Scholar]
- 133.Ensor CR, Rihtarchik LC, Morrell MR, et al. Rescue alemtuzumab for refractory acute cellular rejection and bronchiolitis obliterans syndrome after lung transplantation. Clin Transplant 2017; 31: e12899. doi: 10.1111/ctr.12899 [DOI] [PubMed] [Google Scholar]
- 134.Moniodis A, Townsend K, Rabin A, et al. Comparison of extracorporeal photopheresis and alemtuzumab for the treatment of chronic lung allograft dysfunction. J Heart Lung Transplant 2018; 37: 340–348. doi: 10.1016/j.healun.2017.03.017 [DOI] [PubMed] [Google Scholar]
- 135.Reams BD, Musselwhite LW, Zaas DW, et al. Alemtuzumab in the treatment of refractory acute rejection and bronchiolitis obliterans syndrome after human lung transplantation. Am J Transplant 2007; 7: 2802–2808. doi: 10.1111/j.1600-6143.2007.02000.x [DOI] [PubMed] [Google Scholar]
- 136.US Food and Drug Administration . Esbriet (pirfenidone). Prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2019/022535s012,208780s002lbl.pdf Date last updated: July 2019. Date last accessed: 5 April 2022.
- 137.US Food and Drug Administration . Ofev (nintedanib capsules). Prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2020/205832s014lbl.pdf Date last updated: October 2010. Date last accessed: 5 April 2022.
- 138.Dosanjh A, Ikonen T, Wan B, et al. Pirfenidone: a novel anti-fibrotic agent and progressive chronic allograft rejection. Pulm Pharmacol Ther 2002; 15: 433–437. doi: 10.1006/pupt.2002.0367 [DOI] [PubMed] [Google Scholar]
- 139.Du J, Paz K, Flynn R, et al. Pirfenidone ameliorates murine chronic GVHD through inhibition of macrophage infiltration and TGF-beta production. Blood 2017; 129: 2570–2580. doi: 10.1182/blood-2017-01-758854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.McKane BW, Fernandez F, Narayanan K, et al. Pirfenidone inhibits obliterative airway disease in a murine heterotopic tracheal transplant model. Transplantation 2004; 77: 664–669. doi: 10.1097/01.TP.0000113162.48048.AA [DOI] [PubMed] [Google Scholar]
- 141.von Suesskind-Schwendi M, Heigel E, Pfaehler S, et al. Protective function of pirfenidone and everolimus on the development of chronic allograft rejection after experimental lung transplantation. Histol Histopathol 2016; 31: 793–805. [DOI] [PubMed] [Google Scholar]
- 142.Vos R, Wuyts WA, Gheysens O, et al. Pirfenidone in restrictive allograft syndrome after lung transplantation: a case series. Am J Transplant 2018; 18: 3045–3059. doi: 10.1111/ajt.15019 [DOI] [PubMed] [Google Scholar]
- 143.Tang W, Yu T, Dong T, et al. Nintedanib in bronchiolitis obliterans syndrome after allogeneic hematopoietic stem cell transplantation. Chest 2020; 158: e89–e91. doi: 10.1016/j.chest.2020.05.557 [DOI] [PubMed] [Google Scholar]
- 144.Pluchart H, Chanoine S, Briault A, et al. Restrictive allograft dysfunction after lung transplantation: is there a place for nintedanib? A case report. Fundam Clin Pharmacol 2020; 34: 408–411. doi: 10.1111/fcp.12522 [DOI] [PubMed] [Google Scholar]
- 145.Perch M, Besa V, Corris PA, et al. A European multi-center, randomized, double-blind trial of pirfenidone in bronchiolitis-obliterans-syndrome Grade 1-3 in lung transplant recipients (European Trial of Pirfenidone in BOS (EPOS)) [abstract 2]. J Heart Lung Transplant 2020; 39: S12. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material 00185-2022.SUPPLEMENT (308.7KB, pdf)