Abstract
An efficient method is described for the generation of site-specific chromosomal integrations in Lactobacillus acidophilus and Lactobacillus gasseri. The strategy is an adaptation of the lactococcal pORI system (K. Leenhouts, G. Venema, and J. Kok, Methods Cell Sci. 20:35–50, 1998) and relies on the simultaneous use of two plasmids. The functionality of the integration strategy was demonstated by the insertional inactivation of the Lactobacillus acidophilus NCFM lacL gene encoding β-galactosidase and of the Lactobacillus gasseri ADH gusA gene encoding β-glucuronidase.
Lactobacillus acidophilus and related lactobacilli represent an important group of lactic acid bacteria which are commonly found in association with the human gastrointestinal tract and are delivered as health-promoting bacteria in foods and probiotics. Recent research efforts have focused on expanding the use of probiotic lactobacilli to include biotechnological applications such as enzyme and vaccine delivery systems (reviewed in reference 12). The results to date are promising, but successful implementation of this technology depends on the ability to identify and genetically engineer proper candidate strains. Unfortunately, efficient integration systems are not available for many important lactobacilli. In order to study gene function and chromosomally stabilize expression cassettes, an integration strategy is needed for thermophilic lactobacilli that is independent of transformation frequency, based on a broad-host-range replicon, and stably maintains integrants at the desired temperature (37°C).
Conditionally replicating plasmids based on the broad-host-range lactococcal pWV01 replicon have been used to make a variety of integrations into lactococcal chromosomes. Law et al. (4) described an integration strategy for Lactococcus that utilizes pWV01-derived vectors from which the repA gene has been removed. These “Ori+” integration vectors replicate only in strains providing repA in trans. One method to establish these vectors in a desired host strain is to supply repA from a second temperature-sensitive helper plasmid. When a DNA fragment with homology to the host chromosome has been cloned on the Ori+ vector, a subsequent temperature shift selects for loss of the helper plasmid and integration of Ori+-plasmid DNA. Despite the potential usefulness of this system, its application to thermophilic lactobacilli was still limited by the low functional temperature range of the available helper plasmid, pVE6007.
Creation and temperature sensitivity of plasmid pTRK669.
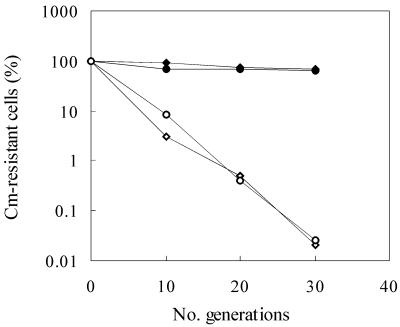
In order to increase the host range of this system to include thermophilic lactobacilli, we sought to combine the useful features of the Ori+ vectors with a more suitable pWV01-based helper plasmid. Previous experiments in our laboratory have indicated that the wild-type pWV01 replicon, while not completely temperature sensitive, is moderately unstable at temperatures of >42°C in thermophilic lactobacilli, similar to what has been reported for the related plasmid, pE194, in Bacillus subtilis (14). While not suitable for performing traditional temperature-sensitive integration experiments, this feature made it possible to adapt the two-plasmid lactococcal integration strategy for use in Lactobacillus. A helper plasmid, based on pGK12, was created to provide repA in trans for the replication of pORI28-based plasmids (7). This helper plasmid, pTRK669, retains the pWV01 replicon including the origin of replication and repAC genes as well as the cat gene (Table 1). The erythromycin resistance (Emr) marker was removed in order to make the new plasmid compatible with pORI28, which harbors its own Emr gene. Plasmids pGK12 and pTRK669 were transformed into L. acidophilus, where their stability was monitored during growth at 37 and 43°C in the absence of antibiotic selection (Fig. 1). The results indicate that both plasmids are stable at 37°C but not at 43°C. An almost 4-log reduction of plasmid-containing cells was observed at 43°C over the course of 30 generations. Similar results were obtained using L. gasseri cells (data not shown). The ability of pTRK669 to supply RepA for the replication of pORI28 was tested by transferring pORI28 into L. acidophilus and L. gasseri cells with or without pTRK669. Emr clones could only be recovered from cells which also carried pTRK669, indicating that replication of pORI28 was supported by pTRK669.
TABLE 1.
Bacterial strains and plasmids
| Strain or plasmid | Origin or relevant characteristics | Source or reference |
|---|---|---|
| Strains | ||
| L. acidophilus | ||
| NCFM | Human intestinal isolate | 1 |
| NCK1398 | NCFM lacL::pTRK670 | This study |
| L. gasseri | ||
| ADH | Human intestinal isolate (called strain MS02) | 2 |
| NCK1423 | ADH gusA::pTRK685 | This study |
| E. coli | ||
| DH5α | F− φ80dlacZΔM15 Δ(lacZYA-argF) U169 endA1 recA1 hsdR17(rK− mK+) deoR | Gibco-BRLa |
| EC1000 | RepA+ MC1000, Kmr, carrying a single copy of the pWV01 repA gene in the glgB gene; host for pOR128-based plasmids | 4 |
| Plasmids | ||
| pGK12 | ori (pWV01), Emr Cmr, RepA+, gram-positive shuttle vector | 3 |
| pTRK669 | ori (pWV01), Cmr, RepA+ | This study |
| pORI28 | Emr, ori (pWV01), replicates only with repA provided in trans | 4 |
| pTRK670 | 2.6 kb; pORI28 with 945-bp internal L. acidophilus NCFM lacL fragment | This study |
| pTRK685 | 2.4 kb; pORI28 with 777-bp internal L. gasseri ADH gusA fragment | This study |
Gibco-BRL, Grand Island, N.Y.
FIG. 1.
Stability of pGK12 (♦) and pTRK669 (●) in L. acidophilus NCFM at 37°C (closed symbols) and 43°C (open symbols). The percentage of Cmr cells in each culture was determined by plating on MRS versus MRS plus chloramphenicol.
Inactivation of the L. acidophilus lacL gene.
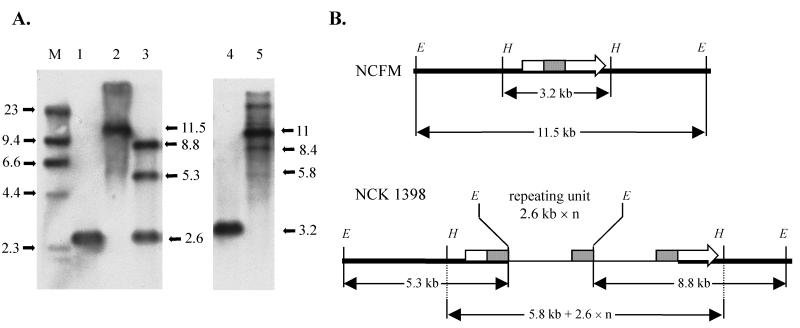
In order to verify the functionality of the integration system, plasmid pTRK670 was designed to disrupt the L. acidophilus lacL gene, encoding β-galactosidase. The primers bgalF (5′-GACTGGATCCTGCCGAACGAGCCATGTATG-3′) and bgalR (5′-GACTGAATTCCCGGCATAAGATTCGTTTCC-3′), based on the previously reported lacL gene of L. acidophilus JCM 1229 (GenBank no. AB004867), were used to amplify a 945-bp internal region of lacL from L. acidophilus NCFM. This fragment was then cloned via the BamHI/EcoRI sites (underlined) into pORI28, an Ori+ RepA− integration plasmid (4). DNA sequencing confirmed that the sequence of the NCFM fragment was 100% identical to the JCM1229 lacL sequence. Plasmid pTRK670 was then introduced by electroporation into L. acidophilus(pTRK669). One Emr, chloramphenicol-resistant (Cmr) transformant, carrying both plasmids, was propagated overnight once at 37°C in MRS broth plus 5 μg of erythromycin and 5 μg of chloramphenicol per ml. The culture was then transferred three times at 43°C (1% inoculum) in MRS broth plus 5 μg of erythromycin/ml (ca. 30 generations). After this enrichment, the culture was plated with a Whitley Automatic Spiral Plater (Don Whitley Scientific Limited, West Yorkshire, England) on MRS–X-Gal (5-bromo-4-chloro-3-indolyl-β-d- galactopyranoside)–galactose plates with either erythromycin, chloramphenicol, or no antibiotic, and incubated for 48 h at 37°C. The results of the plating indicated that the final culture contained ∼107 Emr cells per ml and <10−7 Cmr cells per ml. All of the recovered colonies were white, indicating the loss of β-galactosidase activity. Conversely, a control culture, which had been enriched at 37°C instead of at 43°C, still contained a large percentage of LacL+, Cmr cells. One Emr Cms clone, designated NCK1398 was chosen for further confirmation. To confirm the loss of β-galactosidase activity, ONPG (o-nitrophenyl-β-d-galactopyranoside) assays (11) were performed on log-phase cultures of L. acidophilus NCFM and NCK1398. In order to induce β-galactosidase production, cultures were grown with galactose as the sole carbon source. Upon analysis, no measurable β-galactosidase activity could be detected from NCK1398 compared to 2,652 ± 167 U from NCFM. In order to demonstrate that the disruption of β-galactosidase was the result of integration of pTRK670 in the lacL gene, Southern hybridizations were performed using the 945-bp lacL fragment as a probe (Fig. 2). The lacL probe hybridized to an EcoRI fragment of ca. 11.5 kb in L. acidophilus NCFM. In NCK1398, this band disappeared and was replaced by junction fragments of ca. 5.3 and 8.8 kb due to the presence of a single EcoRI site present in the pTRK670 plasmid sequence. In addition to the two junction fragments, another band was observed that corresponded to the amplified copies of the 2.6-kb pTRK670 sequence. Further confirmation was obtained by hybridization to HindIII digestions because no HindIII sites were present within the pTRK670 sequence. The lacL probe hybridized to an ca. 3.2-kb fragment in L. acidophilus NCFM. In NCK1398, this band disappeared and was replaced with bands of ca. 5.8, 8.4, and 11.0 kb, corresponding to insertions of one, two, and three plasmid copies, respectively. These results were identical in each of the clones tested, indicating that each individual clone gave rise to a mixed population of cells harboring different numbers of plasmid copies. This variation in plasmid copy number has been observed in other lactic acid bacteria (9, 10). Leenhouts et al. (8) reported L. lactis clones that had generated one, two, or three chromosomal plasmid copies after the insertion of a pORI-type plasmid. This amplification of plasmid sequences has generally been attributed to recombinatory activity between the flanking DNA regions of homology that result from campbell-like integration and is influenced by a number of factors, including the nature and location of the insertion event.
FIG. 2.
(A) Southern hybridization analysis of L. acidophilus NCFM and NCK1398. Chromosomal DNA was digested with EcoRI (lanes 1, 2, and 3) or HindIII (lanes 4 and 5) and hybridized with the 945-bp internal lacL fragment as a probe. Lane 1, pTRK670; lanes 2 and 4, NCFM; lanes 3 and 5, NCK1398; M, digoxigenin-labeled λ-HindIII marker. DNA sizes are indicated in kilobases. (B) Schematic representation of the relevant regions of the NCFM and NCK1398 chromosomes. Chromosomal DNA is represented by a heavy line, plasmid DNA is represented by a thin line, the lacL gene is represented by an arrow, and the internal lacL fragment is represented by a solid box. The EcoRI (E) and HindIII (H) sites are indicated. The repeating unit represents the plasmid DNA present in various copies (“n”). The diagram is not drawn to scale.
Inactivation of the L. gasseri gusA gene.
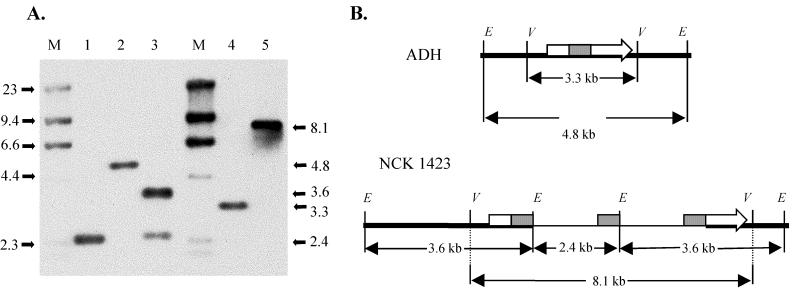
The complete sequence of the L. gasseri ADH gusA gene, encoding β-glucuronidase, has been reported recently (13). A 777-bp internal region of the gusA gene was amplified by PCR using the primers GUS1F (5′-GACTTCTAGAACAGTTGACGAATACACAGAT-3′) and GUS1R (5′-GTGAATTCAGGCGATGAGAAGAAGATAATG-3′). This fragment was then cloned into pORI28 as an XbaI/EcoRI fragment using the sites added to the 5′ ends of the primers (underlined). The resulting plasmid, pTRK685, was then transferred by electroporation into L. gasseri ADH(pTRK669), and the integration strategy was carried out as described above. The results were similar to those obtained for lacL; only Emr Cms colonies were obtained, and disruption of the gusA gene was evidenced by the formation of only white colonies. One clone, designated NCK1423, was subjected to further testing. Confirmation of the GusA− phenotype was obtained by performing β-glucuronidase assays (13) on stationary-phase L. gasseri ADH and NCK1423 cells. No measurable β-glucuronidase activity could be detected with NCK1423 compared to 97.2 ± 11 U in ADH. Integration of plasmid DNA into the gusA gene was confirmed by Southern hybridization (Fig. 3). The results of the Southern hybridization revealed that integration had occurred within the gusA gene. However, unlike the previous integration into L. acidophilus lacL, the number of plasmid DNA sequences integrated into the gusA gene appeared to be fixed at two copies.
FIG. 3.
(A) Southern hybridization analysis of L. gasseri ADH and NCK1423. Chromosomal DNA was digested with EcoRI (lanes 1, 2, and 3) or EcoRV (lanes 4 and 5) and hybridized with the 777-bp internal gusA fragment as a probe. Lane 1, pTRK685; lanes 2 and 4, ADH; lanes 3 and 5, NCK1423; M, digoxigenin-labeled λ-HindIII marker. DNA sizes are indicated in kilobases. (B) Schematic representation of the relevant regions of the ADH and NCK1423 chromosomes. Chromosomal DNA is represented by a heavy line, plasmid DNA is represented by a thin line, the gusA gene is represented by an arrow, and the internal gusA fragment is represented by a solid box. The EcoRI (E) and EcoRV (V) sites are indicated. The diagram is not drawn to scale.
Stability of integrations.
To determine the stability of the integrated plasmids, NCK1398 and NCK1423 were propagated in MRS broth in the absence of antibiotics for ca. 50 generations and plated on media with the appropriate chromogenic substrate, i.e., either X-Gal or X-Gluc (5-bromo-4-chloro-3-indolyl-β-d-glucuronic acid). Loss of the integrated plasmid was assessed by restoration of the LacL+ or GusA+ phenotypes, as indicated by the formation of blue colonies. After 50 generations, the percentages of revertants were 5.36% for lacL and 0.72% for gusA, corresponding to frequencies of 0.11% and 0.01% per generation, respectively. These results indicated that, while plasmid DNA is stably integrated in both strains, the integrated DNA is ca. 10-fold more stable in the gusA gene of L. gasseri NCK1423.
Advantages of the system.
The advantages of the method reported here over other integration strategies for lactobacilli are that (i) it is not dependent on transformation efficiency, (ii) it allows growth of mutant strains at preferred growth temperatures, (iii) it allows efficient recovery of stable integrants, and (iv) it is likely to function across a broad range of species. While this study has focused on the use of a single integration event to insertionally inactivate functional genes, a number of other applications are envisioned. For example, this strategy can be easily modified to construct and integrate expression cassettes into preferred sites in the chromosome. However, the primary disadvantage of the current design is that plasmid DNA and antibiotic markers remain integrated in the chromosome, leading to potential complications in food-grade applications. Recently, the pORI system has been used to create unlabeled gene replacements in L. lactis and B. subtilis (7) and to create a food-grade multiple-copy integration system (6) that has been used to overexpress proteins in Lactococcus (5). It is expected that the protocol described here could be easily modified to facilitate similar strategies in lactobacilli.
Acknowledgments
This research was supported by The Southeast Dairy Foods Research Center; Dairy Management, Inc.; Rhodia, Inc.; and the North Carolina Dairy Foundation. W. Michael Russell was supported in part by a U.S. Department of Education GAANN Biotechnology Fellowship.
We thank Jan Kok, University of Groningen, for kindly providing pORI28 and E. coli EC1000. We also thank Evelyn Durmaz, Eric Altermann, Olivia McAuliffe, and Michael Callanan for their help and for critical review of the manuscript.
Footnotes
Paper no. FSR01–7 of the Journal Series of the Department of Food Science, North Carolina State University, Raleigh, NC 27695-7624.
REFERENCES
- 1.Barefoot S F, Klaenhammer T R. Detection and activity of lactacin B, a bacteriocin produced by Lactobacillus acidophilus. Appl Environ Microbiol. 1983;45:1808–1815. doi: 10.1128/aem.45.6.1808-1815.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kleeman E G, Klaenhammer T R. Adherence of Lactobacillus spp. to human fetal intestinal cells. J Dairy Sci. 1982;65:2063–2069. doi: 10.3168/jds.S0022-0302(82)82462-4. [DOI] [PubMed] [Google Scholar]
- 3.Kok J, van der Vossen J M B M, Venema G. Construction of plasmid cloning vectors for lactic streptococci which also replicate in Bacillus subtilis and Escherichia coli. Appl Environ Microbiol. 1984;48:726–732. doi: 10.1128/aem.48.4.726-731.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Law J, Buist G, Haandrikman A, Kok J, Venema G, Leenhouts K. A system to generate chromosomal mutations in Lactococcus lactis which allows fast analysis of targeted genes. J Bacteriol. 1995;177:7011–7018. doi: 10.1128/jb.177.24.7011-7018.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Leenhouts K, Bolhuis A, Boot J, Deutz I, Toonen M, Venema G, Kok J, Ledeboer A. Cloning, expression, and chromosomal stabilization of the Propionibacterium shermanii proline iminopeptidase gene (pip) for food-grade application in Lactococcus lactis. Appl Environ Microbiol. 1998;64:4736–4742. doi: 10.1128/aem.64.12.4736-4742.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Leenhouts K, Bolhuis A, Venema G, Kok J. Construction of a food-grade multiple-copy integration system for Lactococcus lactis. Appl Microbiol Biotechnol. 1998;49:417–423. doi: 10.1007/s002530051192. [DOI] [PubMed] [Google Scholar]
- 7.Leenhouts K, Buist G, Bolhuis A, ten Berge A, Kiel J, Mierau I, Dabrowska M, Venema G, Kok J. A general system for generating unlabeled gene replacements in bacterial chromosomes. Mol Gen Genet. 1996;253:217–224. doi: 10.1007/s004380050315. [DOI] [PubMed] [Google Scholar]
- 8.Leenhouts K J, Kok J, Venema G. Lactococcal plasmid pWV01 as an integration vector for lactococci. Appl Environ Microbiol. 1991;57:2562–2567. doi: 10.1128/aem.57.9.2562-2567.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Leer R J, Christiaens H, Verstraete W, Peters L, Posno M, Pouwels P H. Gene disruption in Lactobacillus plantarum strain 80 by site-specific recombination: isolation of a mutant strain deficient in conjugated bile salt hydrolase activity. Mol Gen Genet. 1993;239:269–272. doi: 10.1007/BF00281627. [DOI] [PubMed] [Google Scholar]
- 10.Leloup L, Ehrlich S D, Zagorec M, Moreldeville F. Single crossover integration in the Lactobacillus sake chromosome and insertional inactivation of the ptsI and lacL Genes. Appl Environ Microbiol. 1997;63:2117–2123. doi: 10.1128/aem.63.6.2117-2123.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O'Sullivan D, Walker S, West S, Klaenhammer T. Development of an expression strategy using a lytic phage to trigger explosive plasmid amplification and gene expression. Bio/Technology. 1996;14:82–87. doi: 10.1038/nbt0196-82. [DOI] [PubMed] [Google Scholar]
- 12.Rush C M, Mercenier A, Pozzi G. Expression of vaccine antigens in Lactobacillus. In: Pozzi G, Wells J M, editors. Gram-positive bacteria as vaccine vehicles for mucosal immunization. Georgetown, Tex: Springer-Landes Bioscience; 1997. pp. 107–144. [Google Scholar]
- 13.Russell W M, Klaenhammer T R. Identification and cloning of gusA, encoding a new beta-glucuronidase from Lactobacillus gasseri ADH. Appl Environ Microbiol. 2001;67:1253–1261. doi: 10.1128/AEM.67.3.1253-1261.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Youngman P, Poth H, Green B, York K, Olmedo G, Smith K. Methods for genetic manipulation, cloning, and functional analysis of sporulation genes in Bacillus subtilis. In: Smith I, Slepecky R A, Setlow P, editors. Regulation of procaryotic development. Washington, D.C.: American Society for Microbiology; 1989. pp. 65–87. [Google Scholar]