Abstract
The diversity of the endophytic Nostoc symbionts of two thalloid bryophytes, the hornwort Anthoceros fusiformis and the liverwort Blasia pusilla, was examined using the tRNALeu (UAA) intron sequence as a marker. The results confirmed that many different Nostoc strains are involved in both associations under natural conditions in the field. The level of Nostoc diversity within individual bryophyte thalli varied, but single DNA fragments were consistently amplified from individual symbiotic colonies. Some Nostoc strains were widespread and were detected from thalli collected from different field sites and different years. These findings indicate a moderate level of spatial and temporal continuity in bryophyte-Nostoc symbioses.
Bryophytes are spore plants and include three main groups: mosses, liverworts, and hornworts. Previously they were all regarded as primitive plants, phylogenetically intermediate between green algae and vascular plants, but ultrastructural, chemical, and molecular data show that all three bryophyte groups clearly represent independent lineages with ancient histories.
Two species of thalloid liverworts (Blasia pusilla and Cavicularia densa) and all hornworts (e.g., Anthoceros, Notothylas, and Phaeoceros spp.) form stable symbioses with nitrogen-fixing cyanobacteria of the genus Nostoc (1, 16). In these endophytic associations, the symbiotic Nostoc colonies are housed in special cavities, or domatia, located on the ventral surface of the gametophyte. The bryophyte produces the symbiotic structures before infection, and motile Nostoc filaments, or hormogonia, enter these structures through stomata or slime pores (4, 18). As the symbiosis is restricted to the gametophyte, each bryophyte generation needs to be infected by new cyanobacteria. This is significant because bryophytes with symbiotic cyanobacteria tend to be pioneer species, which live in temporary habitats. Overwintering gametophytes, even in temperate climates, tend to show much dieback. In the spring there is rapid recolonization via spores and later via two types of asexual gemmae as well (5, 19).
The hornwort-Nostoc and Blasia-Nostoc associations are easily subjected to experimental manipulations, and they have often been used as model systems to define properties of symbiotically associated Nostoc spp. Both partners can be cultured separately, and reestablishment of the symbiosis can be studied under controlled conditions (6, 7, 9, 18, 21). Although many studies have been performed with these systems, relatively few have addressed questions of cyanobiont identity and specificity. Early studies relied on phenotypic characters for identification of the cyanobiont (4, 8, 10, 18), but recently, molecular methods have also been used (21). Cyanobacterial symbionts from the hornwort Phaeoceros laevis were compared using different PCR amplification techniques, including those employing either short arbitrary primers or primers specific for the regions flanking the 16S-23S rRNA internal transcriber spacer. The results showed a great diversity of cyanobionts, mostly Nostoc strains, even within one bryophyte thallus. Particular cyanobacterial strains seemed to be quite limited in their distribution, because individual strains were never found at more than one field site (21).
In the present work, we examined the genetic diversity of symbiotic cyanobacteria in natural populations of two species of thalloid bryophytes, Anthoceros fusiformis and B. pusilla. Several cyanobacterial colonies from each bryophyte thallus were used directly in PCRs with primers specific for the tRNALeu gene. This intron has been widely used to examine questions of diversity and phylogeny in cyanobacteria (3, 11, 12, 13, 14) and in diverse plant groups, including liverworts (22), making it an appropriate tool for our work.
Biological material.
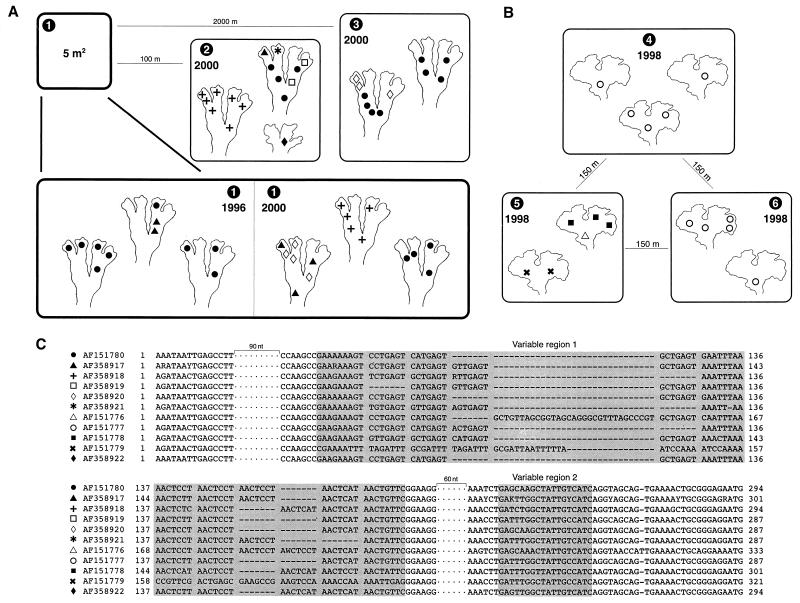
The B. pusilla L. specimens were collected in 1996 (one site) and 2000 (three sites, including the original 1996 site) in Saarijärvi in central Finland. The specimens from sites 1 to 3 were collected from mineral soil along road banks (at 62°47′N, 25°3′E, at an elevation of 140 to 170 m). The sites included Kalmari Village, Impivaara (site 1), Soljala (site 2), and Virranperä (site 3) (Fig. 1A). From each field site, two or three Blasia thalli were analyzed (Fig. 1A). At one site (Fig. 1A, site 2), thalli of Peltigera didactyla (With.) J. R. Laundon, a small cyanobacterial lichen, were found growing within a few centimeters of Blasia thalli. Specimens of this lichen were also collected for analysis. The A. fusiformis Austin specimens from sites 4, 5, and 6 were collected in 1998 from sandy soil along a creek bank (at 44°16′N, 124°06′W, at an elevation of 15 to 20 m) located in Lane County, Oreg. (Cummins Creek Wilderness, at an outlet of Gwynn Creek) (Fig. 1B). Two or three Anthoceros thalli from each site were analyzed (Fig. 1B). In the laboratory, the bryophyte thalli were rinsed with sterile water, and individual colonies of symbiotic cyanobacteria were collected into 10 μl of sterile water by using sterile scalpels. Amplification and sequencing were performed as previously described by Paulsrud and Lindblad (12) for samples collected in 1996 and 1998 or by Paulsrud et al. (15) for samples collected in 2000. In agreement with previous work using the same approach on both cycad and lichen symbioses (3, 12, 13, 14), differences in the nucleotide sequences were principally restricted to the intron's first variable region which could be divided into blocks of 7 nucleotides. The differences in intron size may be attributable to loss or acquisition of such blocks (Fig. 1C).
FIG. 1.
Schematic representation of the biological material examined, with the respective tRNALeu (UAA) intron sequences found in this study. The collection sites are numbered from 1 to 6, and the collection years are shown. Individual cyanobacterial colonies are indicated with a symbol, and each symbol represents a specific nucleotide sequence. These sequences can be accessed by the GenBank numbers indicated next to the symbols in panel C. (A) B. pusilla thalli collected in 1996 (one site) and 2000 (three different sites, including the site from 1996) in central Finland. Site 2 also includes the Peltigera didactyla thallus (lower right schematic). (B) A. fusiformis thalli collected in 1998 at three different sites (all within 150 m of each other) at one location in Oregon. (C) Alignment of the tRNALeu (UAA) intron sequences obtained in the present work. From the conserved sequence of the intron, two DNA stretches of 90 and 60 nucleotides each were removed. The two highly variable regions are indicated by shading. When sequences from different specimens differed, ambiguous bases were introduced in accordance with the International Union of Pure and Applied Chemistry nucleotide code.
Genetic diversity.
The sequence data from the different cyanobacterial symbionts confirm that many different Nostoc strains are involved in Nostoc-Anthoceros and Nostoc-Blasia associations in the field. A similar result was previously reported by West and Adams for the Nostoc-Phaeoceros association (21). In addition to these two studies, in which molecular methods were used for identifying cyanobacterial strains in the field, several laboratory experiments have also demonstrated a potential for high cyanobiont diversity in bryophyte symbioses (6, 7, 18, 21). The bryophyte primordia, whether originating from germinating spores or gemmae, acquire compatible cyanobionts from their surroundings. The symbiotic cyanobacteria can potentially come from several different sources, including populations of free-living Nostocs in soil, neighboring bryophytes, or other symbiotic associations, such as cyanolichens. The relative importance of each of these sources may differ, offering a promising field for future investigations.
None of the analyzed colonies within the bryophyte thalli contained a mixture of different intron sequences. The development of individual symbiotic colonies seems to become stabilized at an early stage, and new Nostoc strains do not seem to enter cavities after these have acquired some specific strain. Conversely, different Nostoc strains were often found in different symbiotic cavities of individual gametophytes. This was true for both bryophyte species; the apparent difference in the level of diversity between the species may well have been a sampling effect. The level of Nostoc diversity within the bryophytes was not consistent; some gametophytes seemed to contain only one Nostoc strain (e.g., Fig. 1A, the GenBank AF358918 sequence, sites 1 and 2, and Fig. 1B, the AF151777 sequence, sites 4 and 6), while others housed several different strains (e.g., Fig. 1A, site 2, and Fig. 1B, site 5). In the cases where only one Nostoc strain was found within a gametophyte, all the symbionts may well have originated from the same source. It is possible that during growth of the bryophyte, mature cyanobacterial colonies released hormogonia, which then infected new symbiotic cavities at the thallus apex. Conversely, in cases where several Nostoc strains were detected in single thalli, a mixture of symbiotically competent cyanobacteria must have existed during thallus growth. The assemblage of competent Nostoc strains in such cases must also have coexisted for some time, as the distribution of specific strains within individual Blasia thalli did not correlate with the developmental sequence of the symbiotic structures. In these cases, random infection by hormogonia from old cyanobacterial colonies may have acted to maintain cyanobacterial diversity within growing thalli, providing that these had initially been infected by more than one Nostoc strain.
The finding that some symbiotic Nostoc strains were shared by bryophytes growing 2,000 m apart differed from the results of West and Adams (21), who detected a more localized distribution of Nostoc strains in Phaeoceros laevis. However, one cannot directly compare the results of these studies, as different molecular methods with different levels of resolution were used. The specimens of Anthoceros and Blasia in this study were collected from different parts of the world. Thus, the absence of overlap in the Nostoc strains of these two species was probably due more to spatial isolation than to differences in cyanobacterial specificity. This hypothesis is supported by the fact that some Nostoc strains identified in the present study had previously been detected in lichens. From Blasia, the GenBank AF358920 sequence was identical to the AF055661 sequence from the lichen Peltigera neopolydactyla from central Finland, and the AF358918 sequence was 3 nucleotides different from the AF176612 sequence from the lichen Peltigera neopolydactyla from North America. Also, the AF358922 sequence from the lichen Peltigera didactyla was 2 nucleotides different from the AF176613 sequence from Peltigera britannica from North America.
Regarding their patterns of cyanobacterial specificity, the bryophytes seem to have several features in common with the cycads from which several Nostoc strains have been amplified from individual host plants, but specific coralloid roots always contained single strains (3). A similar pattern has also been detected in the cyanolichen Peltigera venosa, which may have different Nostoc strains in different colonies from a single thallus (14). Other cyanolichens have generally shown higher levels of specificity (12, 13, 14).
One site in central Finland where Blasia was found was sampled first in August 1996 and again in August 2000 (Fig. 1A, site 1). This site revealed temporal stability in the cyanobiont composition of the bryophyte population: both Nostoc strains found in 1996 were also detected in 2000. The sampling dates were separated by three full gametophyte generations.
Although further studies are still needed, our results indicate that there is a moderate level of spatial and temporal continuity in the Blasia-Nostoc symbiosis. This finding is understandable in light of what is known about the seasonality and reproductive biology of B. pusilla. Most symbiotic bryophytes are pioneer species, exploiting temporary niches (19, 20). The ability of Blasia to effectively colonize temporary habitats depends partly on the fact that the gametophytes can produce not only sporophytes and sexual spores but also two types of asexual propagules. Large stellate gemmae are produced on the dorsal epidermis, while smaller ovoid gemmae are produced in long-necked flask-shaped receptacles (2, 5, 17, 19). It is of particular interest that the apically derived stellate gemmae reach a complex level of development before they detach and germinate. In fact, they are essentially miniature Blasia gametophytes, equipped with two lobes and two auricles, regularly containing symbiotic Nostoc colonies (5, 17). With the help of these symbiotic diaspores, the whole symbiotic consortium, consisting of the bryophyte and cyanobiont, can disperse together. Therefore, unlike the other bryophyte symbioses, the Blasia-Nostoc association has the potential for maintaining specific symbiotic consortia over time and space. To our knowledge, the potential significance of this special mode of propagation has not been recognized in previous literature on cyanobacterial symbioses.
Acknowledgments
This study was financially supported by Anna and Gunnar Vidfelt's Foundation (P.P.), the Swedish Natural Science Research Council (P.L.), and the Academy of Finland (J.R.).
REFERENCES
- 1.Adams D G. Symbiotic interactions. In: Whitton B A, Potts M, editors. The ecology of cyanobacteria. Dordrecht, The Netherlands: Kluwer Academic Publishers; 2000. pp. 523–561. [Google Scholar]
- 2.Bartholomew S E. The sporeling development of Blasia pusilla L. J Hattori Bot Lab. 1986;60:255–261. [Google Scholar]
- 3.Costa J-L, Paulsrud P, Lindblad P. Cyanobiont diversity within coralloid roots of selected cycad species. FEMS Microbiol Ecol. 1999;28:85–91. [Google Scholar]
- 4.Duckett J G, Prasad A K, Davies D A, Walker S. A cytological analysis of the Nostoc-bryophyte relationship. New Phytol. 1977;79:349–362. [Google Scholar]
- 5.Duckett J G, Renzaglia K S. The reproductive biology of the liverwort Blasia pusilla L. J Bryol. 1993;17:541–552. [Google Scholar]
- 6.Enderlin C S, Meeks J C. Pure culture and reconstitution of the Anthoceros-Nostoc symbiotic association. Planta. 1983;158:157–165. doi: 10.1007/BF00397709. [DOI] [PubMed] [Google Scholar]
- 7.Kimura J, Nakano T. Reconstitution of a Blasia-Nostoc symbiotic association under axenic conditions. Nova Hedwigia. 1990;50:191–200. [Google Scholar]
- 8.Lhotsky S. The assimilation of free nitrogen in symbiotic Cyanophyceae. Stud Bot Cechoslov. 1946;7:20–35. [Google Scholar]
- 9.Meeks J C. Symbiosis between nitrogen fixing cyanobacteria and plants. BioScience. 1998;48:266–276. [Google Scholar]
- 10.Pankov H, Martens B. Über Nostoc sphaericum Vauch. Arch Microbiol. 1964;48:203–212. [Google Scholar]
- 11.Paquin B, Kathe S D, Nierzwicki-Bauer S A, Shub D A. Origin and evolution of group I introns in cyanobacterial tRNA genes. J Bacteriol. 1997;179:6798–6806. doi: 10.1128/jb.179.21.6798-6806.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Paulsrud P, Lindblad P. Sequence variation of the tRNALeu intron as a marker for genetic diversity and specificity of symbiotic cyanobacteria in some lichens. Appl Environ Microbiol. 1998;64:310–315. doi: 10.1128/aem.64.1.310-315.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Paulsrud P, Rikkinen J, Lindblad P. Cyanobiont specificity in some Nostoc-containing lichens and in a Peltigera aphthosa photosymbiodeme. New Phytol. 1998;139:517–524. [Google Scholar]
- 14.Paulsrud P, Rikkinen J, Lindblad P. Spatial patterns of the photobiont diversity in some Nostoc-containing lichens. New Phytol. 2000;146:291–299. doi: 10.1046/j.1469-8137.2000.00647.x. [DOI] [PubMed] [Google Scholar]
- 15.Paulsrud, P., J. Rikkinen, and P. Lindblad. Field investigations on cyanobacterial specificity in Peltigera aphthosa (L.) Willd. New Phytol., in press. [DOI] [PubMed]
- 16.Rai A N, Söderback E, Bergman B. Cyanobacterium-plant symbioses. New Phytol. 2000;147:449–481. doi: 10.1046/j.1469-8137.2000.00720.x. [DOI] [PubMed] [Google Scholar]
- 17.Renzaglia K S. A comparative developmental investigation of the gametophyte generation in the Metzgeriales (Hepatophyta) Bryophyt Bibl. 1982;24:1–253. [Google Scholar]
- 18.Rodgers G A, Stewart W D. The cyanophyte-hepatic symbiosis. I. Morphology and physiology. New Phytol. 1977;78:441–458. [Google Scholar]
- 19.Schuster R M. The Hepaticae and Anthocerotae of North America east of the hundredth meridian. V. Chicago, Ill: Field Museum of Natural History; 1992. [Google Scholar]
- 20.Schuster R M. The Hepaticae and Anthocerotae of North America east of the hundredth meridian. VI. Chicago, Ill: Field Museum of Natural History; 1992. [Google Scholar]
- 21.West N J, Adams D G. Phenotypic and genotypic comparison of symbiotic and free-living cyanobacteria from a single field site. Appl Environ Microbiol. 1997;63:4479–4484. doi: 10.1128/aem.63.11.4479-4484.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wheeler J A. Molecular phylogenetic reconstructions of the marchantioid liverwort radiation. Bryologist. 2000;103:314–333. [Google Scholar]