Abstract
Background
Curcumin shows great effects of inhibiting tumor cell proliferation, inducing apoptosis, inhibiting tumor metastasis, and inhibiting angiogenesis on a variety of tumors. However, the biological activity and possible mechanisms of curcumin in the treatment of retinoblastoma have not been fully elucidated. This study explored the potential therapeutic targets and pharmacological mechanisms of curcumin against retinoblastoma based on network pharmacology and molecular docking.
Methods
The genes corresponding to curcumin targets were screened from the HERB, PharmMapper, and SwissTargetPrediction databases. Protein-protein interaction (PPI) networks were constructed for the intersecting targets in the STRING database. Cytoscape 3.7.0 was used for network topology analysis and screening of important targets. R 4.1.0 software was used for Gene Ontology (GO) function enrichment and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis of intersection targets. The molecular structures of curcumin and core target proteins were obtained from PubChem and PDB databases, and the two were preprocessed and molecularly docked using AutoDockTools and PyMOL software.
Results
Through network data mining, we obtained 504 curcumin targets and 966 retinoblastoma disease targets, and 44 potential targets for curcumin treatment of retinoblastoma were obtained by mapping. Three core targets were obtained from network topology analysis. 462 biological processes, 21 cellular compositions, and 34 molecular functions were obtained by GO enrichment analysis. KEGG pathway analysis revealed 94 signaling pathways, mainly involving chemical carcinogenesis-receptor activation, chemical carcinogenesis-reactive oxygen species, viral carcinogenesis, Th17 cell differentiation, etc. The molecular docking results indicated that the binding energy of curcumin to the core targets was less than 0 kJ mol−1, among which the binding energy of RB1 and CDKN2A to curcumin was less than −5 kJ mol−1 with significant binding activity.
Conclusion
Based on molecular docking technology and network pharmacology, we initially revealed that curcumin exerts its therapeutic effects on retinoblastoma with multitarget, multipathway, and multibiological functions, providing a theoretical basis for subsequent studies.
1. Introduction
Retinoblastoma is the most common intraocular malignancy in children, which accounts for 2.5% to 4% of all childhood cancers and has an incidence of 1/18,000 to 1/14,000 [1]. Poor treatment may lead to blindness or death. In order to realize early diagnosis and treatment of retinoblastoma and to reduce the mortality of retinoblastoma, it is particularly important to find the key genes for retinoblastoma. For patients with retinoblastoma, the development of new natural compounds and the search for new therapeutic targets are necessary.
Curcumin is a yellow phenolic pigment with low toxicity, wide medicinal source and low price extracted from the rhizome of Curcuma longa of the ginger family, such as turmeric and tulip, which has a wide application prospect and value in clinical treatment [2, 3]. Curcumin shows great effects of inhibiting tumor cell proliferation, inducing apoptosis, inhibiting tumor metastasis, and inhibiting angiogenesis on a variety of tumors [4–6]. Domestic and foreign scholars have carried out some studies on the effect of curcumin on retinoblastoma [7]. However, traditional Chinese medicine treatment of diseases is based on the mechanism of action of multitarget and multipathway, and there is a relative lack of research in systematically elucidating the action targets and molecular mechanisms of curcumin in the treatment of retinoblastoma, which requires the application of big data to explore the existing target pathways related to curcumin and retinoblastoma.
Network pharmacology is a systems biology approach to study the development of diseases, understand drug-organism interactions, and guide new drug discovery. Therefore, this study aims to investigate the targets and pathways of curcumin and its possible molecular mechanisms of action in the treatment of retinoblastoma by using network pharmacology and molecular docking methods and to provide a reference basis for subsequent studies. The study scheme design is shown in Figure 1.
Figure 1.
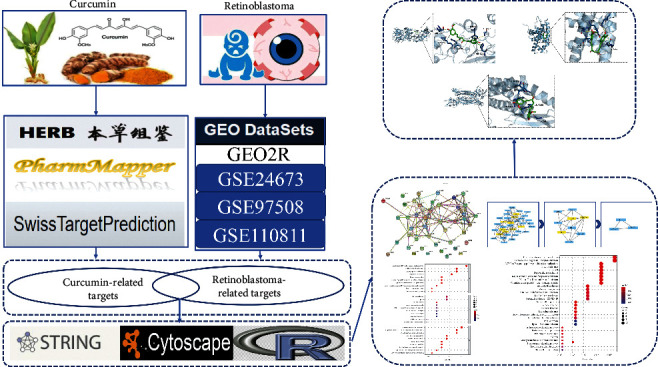
The idea and process of this research.
2. Materials and Methods
2.1. Screening of Potential Targets of Curcumin for the Treatment of Retinoblastoma
We obtained validated and predicted targets for curcumin via the HERB (http://herb.ac.cn/), PharmMapper (http://www.lilab-ecust.cn/pharmmapper/), and SwissTargetPrediction (http://www. swisstargetprediction.ch/?) databases. Download microarray data GSE24673, GSE97508, and GSE110811 for retinoblastoma from the GEO database (https://www.ncbi.nlm.nih.gov/) and screen for differentially expressed genes using the GEO2R online tool (adj.P < 0.05, |logFC|>2) (Table 1). The above retrieved curcumin action targets were mapped to retinoblastoma disease targets to obtain potential targets of curcumin for retinoblastoma treatment.
Table 1.
Description of datasets in this study.
2.2. Protein-Protein Interaction Network Construction, Analysis, and Core Gene Screening
Protein-protein interaction (PPI) networks were constructed using the online database STRING (https://string-db.org/), and TSV format files were saved, which were imported into Cytoscape 3.7.0 software, and the Cyot NCA plug-in was used to further analyze the topology of the regulatory network and screen out the core target genes.
2.3. Enrichment Analysis
To explore the core functional and biological pathways associated with curcumin treatment of retinoblastoma, we performed Gene Ontology (GO) function enrichment and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis using the ClusterProfiler package in R 4.1.0 software. The GO functional enrichment analysis and KEGG pathway enrichment analysis screening conditions were set as P < 0.05, q < 0.05.
2.4. Molecular Docking Verification
The core target genes screened by topological analysis were molecularly docked to curcumin. The protein 3D maps of the core target genes were downloaded from the PDB database (https://www.rcsb.org/). Also, download the 3D structure of the curcumin molecule from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) and convert the format using Open Babel software. With the help of AutoDockTools, the molecular structure of curcumin was optimized, the structure of core gene protein was dehydrated and hydrogenated, and the molecular and protein preservation format was changed to PDBQT format. Molecular docking verification was performed using Auto Dock Vina software, and finally the obtained results were visualized using PyMOL.
3. Results
3.1. Targets of Curcumin in the Treatment of Retinoblastoma
504 curcumin targets were retrieved from the HERB, PharmMapper, and SwissTargetPrediction databases. The 966 differentially expressed genes of retinoblastoma were screened by GEO2R from three gene chips, GSE24673, GSE97508, and GSE110811. After mapping the curcumin targets to the retinoblastoma targets, 44 intersecting targets were obtained (Figure 2), which were predicted as potential targets for curcumin treatment of retinoblastoma.
Figure 2.
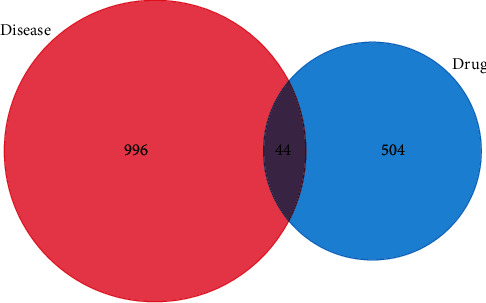
Venny analysis diagram of the mapping targets of curcumin and retinoblastoma.
3.2. GO Functional Enrichment Analysis and KEGG Pathway Enrichment Analysis
The R 4.1.0 software was used to analyze the biological processes and molecular functions of the intersecting targets involved in retinoblastoma and to investigate the possible molecular mechanisms of curcumin in the treatment of retinoblastoma. The GO functional enrichment analysis yielded 517 entries, including 462 for Biological Process, 21 for Cellular Component, and 34 for Molecular function. The results were ranked in order of significance, and the top 10 entries for each type of analysis were selected for visualization in R software (Figure 3). The biological process mainly involved the regulation of DNA-binding transcription factor activity, peptidyl-tyrosine phosphorylation, peptidyl-tyrosine modification, histone phosphorylation, etc. Cellular Component mainly involved RNA polymerase II transcription regulator complex, chromosome, centromeric region, microtubule-associated complex, spindle microtubule, etc. Molecular function mainly involved protein serine/threonine kinase activity, RNA polymerase II-specific DNA-binding transcription factor binding, DNA-binding transcription factor binding, etc. KEGG pathway enrichment analysis screened 94 signaling pathways (P < 0.05) and visualized the top 30 pathways (Figure 4). The main pathways involved were chemical carcinogenesis-receptor activation, chemical carcinogenesis-reactive oxygen species, viral carcinogenesis, Th17 cell differentiation, etc.
Figure 3.
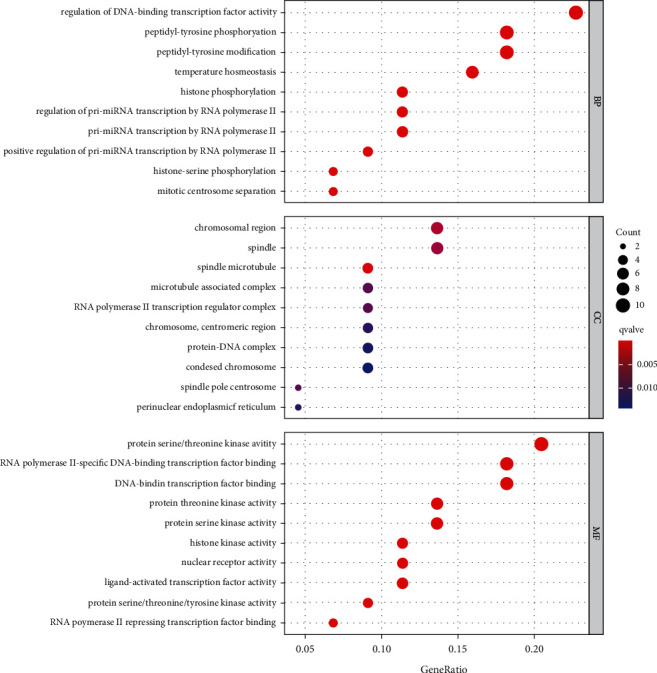
GO functional enrichment results of potential targets of curcumin against retinoblastoma.
Figure 4.
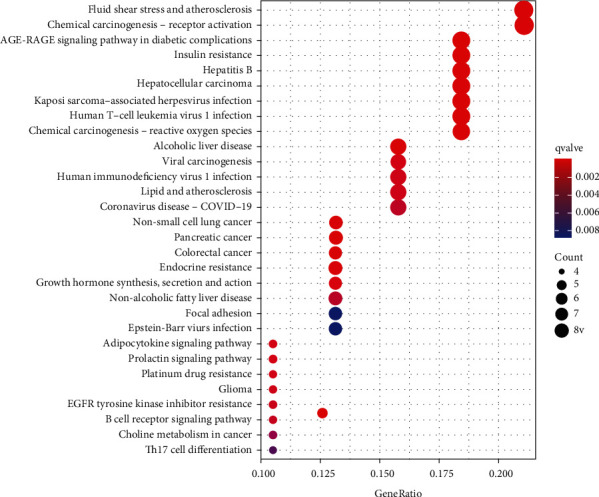
KEGG enrichment results of potential targets of curcumin against retinoblastoma.
3.3. PPI Network Construction, Analysis, and Core Gene Screening
The intersection targets were imported into the STRING database to obtain a total of 44 protein nodes for the PPI network, save the PPI network map (Figure 5) with TSV format files, and import the files into Cytoscape 3.7.0 software. Cytoscape 3.7.0 software plug-in CytoNCA calculated the Betweenness Centrality (BC), Closeness Centrality (CC), Degree Centrality (DC), and Eigenvector Centrality (EC) of 44 intersecting target proteins. The calculated median values of BC, CC, DC, and EC were 16.59, 0.46, 7, and 0.15, respectively. A total of 9 target proteins with parameters greater than the median value were filtered to create a sub-network (Figure 6). The second time, BC, CC, DC, and EC of 9 target proteins in the sub-network were calculated. The median values of BC, CC, DC, and EC were 15.7, 0.73, 5, and 0.34, respectively. A total of 3 target proteins with parameters greater than the median value were obtained and the network was created (Figure 6). The three target proteins were the key target proteins of curcumin for the treatment of retinoblastoma.
Figure 5.
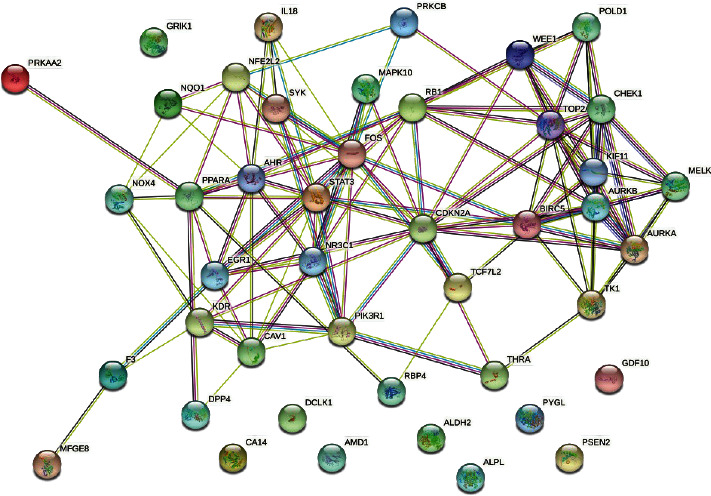
The protein-protein interaction network among potential targets of curcumin in the treatment of retinoblastoma.
Figure 6.
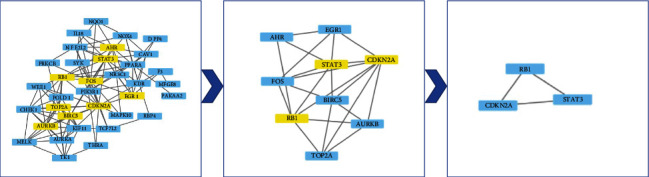
Hub genes acquired from the protein-protein interaction network.
3.4. Docking Results Analysis
We selected the core targets, including RB1, STAT3, and CDKN2A, for molecular docking with curcumin. The results showed that curcumin has a good affinity with RB1, STAT3, and CDKN2A, and the docking results were visualized by PyMOL software (Figure 7 and Table 2).
Figure 7.
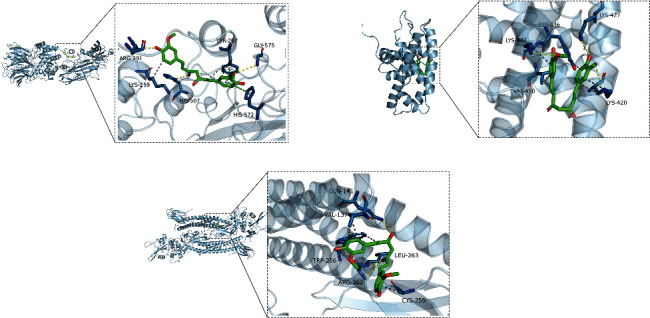
Modes of docking between curcumin and core targets protein molecules.
Table 2.
Chemical information and binding energy of curcumin.
| Mol ID | Chemical abstracts service number | Chemical formula | Molecular weight | Docking protein | Binding energy/(kcal·mol−1) |
|---|---|---|---|---|---|
| MOL000090 | 485-37-7 | C21H20O6 | 368.37 | RB1 | −6.9 |
| STAT3 | −4.8 | ||||
| CDKN2A | −7.4 |
4. Discussion
Retinoblastoma is the second most common pediatric malignancy and is a serious threat to children's lives [8]. Retinoblastoma develops mostly under the age of 5, and about 2/3 of children develop the disease before the age of 3 [9, 10]. Timely diagnosis and intervention in the early stages of retinoblastoma results in a survival rate of over 95%, while once extraocular metastases occur, the survival rate is less than 50% [11]. Therefore, in order to achieve early diagnosis and treatment of retinoblastoma, it is increasingly important to find its key genes and biomarkers. Curcumin has antitumor effects on a variety of tumors [12–14]. Curcumin exerts its effects through the downregulation of multiple cellular signaling pathways, including NF-κB, STAT3, activated protein-1, and epidermal growth response-1, all of which are essential for cell development and progression [15]. Yang et al. [16] showed that inhibition of STAT3 phosphorylation contributed to the antiproliferative effect of curcumin on lung cancer cells and inhibited the migration and invasion of cancer cells. Zhang et al. [17] found that curcumin induced endoplasmic reticulum stress in hepatocellular carcinoma BEL-7404 cells by downregulating STAT3 expression, which ultimately promoted apoptosis in BEL-7404 cells. However, the underlying mechanism of curcumin in the treatment of retinoblastoma remains unclear.
In this study, we used network pharmacology and molecular docking to explore the molecular mechanisms of curcumin for retinoblastoma treatment in a more comprehensive manner. We identified three potential targets for curcumin for retinoblastoma treatment through network data mining. GO functional enrichment analysis revealed 517 pathways. KEGG pathway enrichment analysis revealed 94 signaling pathways related to chemical carcinogenesis-receptor activation, chemical carcinogenesis-reactive oxygen species, viral carcinogenesis, Th17 cell differentiation, and other signaling pathways. Molecular docking results showed that curcumin was stably bound to RB1, STAT3, and CDKN2A, among which CDKN2A had the strongest binding to curcumin.
RB1, STAT3, and CDKN2A are the most likely core targets of curcumin for retinoblastoma treatment. Molecular docking analysis has shown that curcumin has a good affinity for these three targets, with CDKN2A showing the highest binding. STAT3 is a well-studied transcription factor in recent years. Several studies have shown that STAT3 has been aberrantly expressed and activated in a variety of tumor tissues and cell lines, can participate in tumor development by inducing cell overproliferation and inhibiting apoptosis, and is aberrantly expressed in retinoblastoma [18, 19]. The RB1 gene is the first family of oncogenes discovered in humans and is an oncogene that regulates the cell cycle, which is closely linked to the development of retinoblastoma [20, 21]. CDKN2A is directly involved in cell cycle regulation and is a novel anticancer gene [22]. Its gene inactivation is closely related to the development of malignant tumors [23]. The main mechanisms include effective inhibition of RNA polymerase activity, alteration of cellular chromatin structure, and prevention of transcription factors from binding to DNA [24]. CNKN2A was found to be differentially expressed in a variety of tumor tissues and showed upregulated expression, which correlated with the pathological characteristics and prognosis of patients [25].
The KEGG enrichment results suggested that the chemical carcinogenesis-reactive oxygen species signaling pathway plays a crucial role, indicating that this signaling pathway is a key link in the treatment of retinoblastoma with curcumin. Reactive oxygen species are produced in the process of oxidative phosphorylation and play an important role in cell and tissue proliferation, differentiation, and apoptosis. Numerous studies have shown that reactive oxygen species are closely associated with the development of retinoblastoma, uveal melanoma, age-related macular degeneration, age-related cataract, dry eye, pterygium, and other ocular-related diseases [26]. Compared with normal cells, the content of reactive oxygen species is higher in tumor cells, and different concentrations of reactive oxygen species have different effects on the development of tumors [27]. Reactive oxygen species not only influence tumorigenesis and development but also play a key role in tumor treatment [27]. Hypoxia promotes the production of reactive oxygen species and activates STAT3 transcription factors, while treatment with reactive oxygen species inhibitors and the antioxidant NAC inhibits STAT3 transcription factor activation, thereby blocking its mediated angiogenic pathway [28]. In the study of Cho et al. [29] on prostate cancer, it was found that that epidermal growth factor promotes prostate cancer cell invasion and metastasis by promoting reactive oxygen species production, which in turn drives STAT3 phosphorylation and regulates the HIF-1α/TWIST1/N-cadherin (N-cadherin) signaling pathway. Therefore, STAT3 may be a key target of reactive oxygen species in regulating tumor invasion and metastasis.
5. Conclusion
In conclusion, this comprehensive network-based pharmacological analysis suggests a number of testable speculations on the potential molecular mechanisms of curcumin in the treatment of retinoblastoma and predicts RB1, STAT3, and CDKN2A as potential therapeutic targets. This study provides a basis for further experimental studies and ideas to investigate the mechanism of curcumin in the treatment of retinoblastoma, and further studies and validation are needed.
Data Availability
All data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Rodriguez-Galindo C., Orbach D. B., VanderVeen D. Retinoblastoma. Pediatric clinics of north America . 2015;62(1):201–223. doi: 10.1016/j.pcl.2014.09.014. [DOI] [PubMed] [Google Scholar]
- 2.Ojo O. A., Adeyemo T. R., Rotimi D., et al. Anticancer properties of curcumin against colorectal cancer: a review. Frontiers in Oncology . 2022;12 doi: 10.3389/fonc.2022.881641.881641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sravani A. B., Mathew E. M., Ghate V., Lewis S. A. A sensitive seectroflurimetric method for curcumin analysis. Journal of Fluorescence . 2022;8:1–11. doi: 10.1007/s10895-022-02947-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vadukoot A. K., Mottemmal S., Vekaria P. H. Curcumin as a potential therapeutic agent in certain cancer types. Cureus . 2022;14(3) doi: 10.7759/cureus.22825.e22825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sreenivasan S., Thirumalai K., Danda R., Krishnakumar S. Effect of curcumin on miRNA expression in human Y79 retinoblastoma cells. Current Eye Research . 2012;37(5):421–428. doi: 10.3109/02713683.2011.647224. [DOI] [PubMed] [Google Scholar]
- 6.Zoi V., Galani V., Tsekeris P., Kyritsis A. P., Alexiou G. A. Radiosensitization and radioprotection by curcumin in glioblastoma and other cancers. Biomedicines . 2022;10(2):p. 312. doi: 10.3390/biomedicines10020312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li Y., Sun W., Han N., Zou Y., Yin D. Curcumin inhibits proliferation, migration, invasion and promotes apoptosis of retinoblastoma cell lines through modulation of miR-99a and JAK/STAT pathway. BMC Cancer . 2018;18(1):p. 1230. doi: 10.1186/s12885-018-5130-y. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 8.Dimaras H., Kimani K., Dimba E. A., et al. Retinoblastoma. The Lancet . 2012;379(9824):1436–1446. doi: 10.1016/s0140-6736(11)61137-9. [DOI] [PubMed] [Google Scholar]
- 9.Pichi F., Lembo A., De Luca M., Hadjistilianou T., Nucci P. Bilateral retinoblastoma: clinical presentation, management and treatment. International Ophthalmology . 2013;33(5):589–593. doi: 10.1007/s10792-012-9703-5. [DOI] [PubMed] [Google Scholar]
- 10.Dimaras H., Corson T. W. Retinoblastoma, the visible CNS tumor: a review. Journal of Neuroscience Research . 2019;97(1):29–44. doi: 10.1002/jnr.24213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Soliman S. E., Racher H., Zhang C., MacDonald H., Gallie B. L. Genetics and molecular diagnostics in retinoblastoma--an update. Asia-Pacific journal of ophthalmology (Philadelphia, Pa) . 2017;6(2):197–207. doi: 10.22608/APO.201711. [DOI] [PubMed] [Google Scholar]
- 12.Xie L., Ji X., Zhang Q., Wei Y. Curcumin combined with photodynamic therapy, promising therapies for the treatment of cancer. Biomedicine & Pharmacotherapy . 2022;146 doi: 10.1016/j.biopha.2021.112567.112567 [DOI] [PubMed] [Google Scholar]
- 13.Sudhesh Dev S., Zainal Abidin S. A., Farghadani R., Othman I., Naidu R. Receptor tyrosine kinases and their signaling pathways as therapeutic targets of curcumin in cancer. Frontiers in Pharmacology . 2021;12 doi: 10.3389/fphar.2021.772510.772510 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Abadi A. J., Mirzaei S., Mahabady M. K., et al. Curcumin and its derivatives in cancer therapy: potentiating antitumor activity of cisplatin and reducing side effects. Phytotherapy Research . 2022;36(1):189–213. doi: 10.1002/ptr.7305. [DOI] [PubMed] [Google Scholar]
- 15.Wong K. E., Ngai S. C., Chan K. G., Lee L. H., Goh B. H., Chuah L. H. Curcumin nanoformulations for colorectal cancer: a review. Frontiers in Pharmacology . 2019;10:p. 152. doi: 10.3389/fphar.2019.00152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang C. L., Liu Y. Y., Ma Y. G., et al. Curcumin blocks small cell lung cancer cells migration, invasion, angiogenesis, cell cycle and neoplasia through Janus kinase-STAT3 signalling pathway. PLoS One . 2012;7(5) doi: 10.1371/journal.pone.0037960.e37960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang M., Zhang Y., Shen Z., Jin R. Curcumin regulates the mechanism of endoplasmic reticulum stress in STAT3-induced apoptosis in human hepatocellular carcinoma BEL-7404 cells. Lishizhen Medicine and Materia Medica Research . 2020;31(3):594–597. [Google Scholar]
- 18.Jo D. H., Lee S., Bak E., et al. Antitumor activity of novel signal transducer and activator of transcription 3 inhibitors on retinoblastoma. Molecular Pharmacology . 2021;100(1):63–72. doi: 10.1124/molpharm.120.000231. [DOI] [PubMed] [Google Scholar]
- 19.Yang L., Shen J., Cheng Y., Hong S. Expressions of p-STAT3‚MMP-2and VEGF in retinoblastoma. Journal of Shandong University (HEALTH SCIENCES) . 2009;47(5):113–116. [Google Scholar]
- 20.Yang X., Sun H., Tang T., Zhang W., Li Y. Netrin-1 promotes retinoblastoma-associated angiogenesis. Annals of Translational Medicine . 2021;9(22):p. 1683. doi: 10.21037/atm-21-5560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wong K. M., King D. A., Schwartz E. K., Herrera R. E., Morrison A. J. Retinoblastoma protein regulates carcinogen susceptibility at heterochromatic cancer driver loci. Life science alliance . 2022;5(4) doi: 10.26508/lsa.202101134.e202101134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Abou-Zeid A. A., Azzam A. Z., Kamel N. A. Methylation status of the gene promoter of cyclin-dependent kinase inhibitor 2A (CDKN2A) in ovarian cancer. Scandinavian Journal of Clinical and Laboratory Investigation . 2011;71(7):542–547. doi: 10.3109/00365513.2011.590224. [DOI] [PubMed] [Google Scholar]
- 23.Panani A. D., Maliaga K., Babanaraki A., Bellenis I. Numerical abnormalities of chromosome 9 and p16CDKN2A gene deletion detected by FISH in non-small cell lung cancer. Anticancer Research . 2009;29(11):4483–4487. [PubMed] [Google Scholar]
- 24.Zhou C., Li J., Li Q. CDKN2A methylation in esophageal cancer: a meta-analysis. Oncotarget . 2017;8(30):50071–50083. doi: 10.18632/oncotarget.18412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xia C., Ding Y., Xu X., Liu P. Screening and prognostic analysis of differential genes in non-small cell lung cancer. Journal of Clinical Pulmonary Medicine . 2020;25(4):578–583. [Google Scholar]
- 26.Wang W., Chen j., Wang Y. Research progress on relationship between oxidative stress and ocular surface diseases. Recent Advances in Ophthalmology . 2014;34(6):585–588. [Google Scholar]
- 27.Zheng K., Ga L., Ma Y., Yang H., Zhao Y., Luo H. Research progress of reactive oxygen species (ROS)-dependent anticancer drugs. Journal of Guangdong Pharmaceutical University . 2022;38(1):130–136. [Google Scholar]
- 28.Yu M. O., Park K. J., Park D. H., Chung Y. G., Chi S. G., Kang S. H. Reactive oxygen species production has a critical role in hypoxia-induced Stat3 activation and angiogenesis in human glioblastoma. Journal of neuro-oncology . 2015;125(1):55–63. doi: 10.1007/s11060-015-1889-8. [DOI] [PubMed] [Google Scholar]
- 29.Cho K. H., Choi M. J., Jeong K. J., et al. A ROS/STAT3/HIF-1α signaling cascade mediates EGF-induced TWIST1 expression and prostate cancer cell invasion. The Prostate . 2014;74(5):528–536. doi: 10.1002/pros.22776. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data used to support the findings of this study are included within the article.


