Abstract
In the present study we focused on the anti-asthmatic and antioxidant effects of Zingiber officinalis roscoe L. (ZO) aqueous extract. This study includes 20 adult male rats, which were grouped into four; Group I: control group; Group II: asthmatic group (Ovalbumin sensitized/challenge model, Oval group); Group III: received ovalbumin sensitized/challenge associated a dose of 207 mg/kg body weight (BW) of ZO (Oval + D1 group); Group IV: received ovalbumin sensitized/challenge associated a dose of 414 mg/k BW of ZO (Oval + D2 group). After 21 days, blood and lung samples were collected for biochemical, hematological, and histopathological analyses. The ameliorative effect of ZO phytochemical compounds was also assessed by in silico approach on transducer and activator of transcription 6 (STAT6) and tumor necrosis factor-α (TNF-α) receptors. The oxidative/antioxidative status was evaluated in the lung tissues. Our results show that ZO extract alleviated the ovalbumin-induced hematological and biochemical disruptions associated oxidative injury. In fact, white and red blood cells (WBC and RBC, respectively), aspartate aminotransaminase (ASAT), malondialdehyde (MDA), glutathione (GSH), and glutathione peroxidase (GPx) were significantly disrupted (p < 0.05) in Oval group and alleviated following ZO treatment. Besides, several histopathological features were outlined in lung tissues of Oval group. Interestingly, ZO was found to exert ameliorative effects on tissue level. In silico analyses, particularly the binding affinities, the number of H-bonds, the embedding distance and the molecular interactions of ZO phytochemical compounds with either STAT6 or TNF-α supported the in vivo results. These findings confirm the potential ethno-pharmacological effects of ZO against asthma and its associated complications.
Keywords: Asthma, Inflammation, Molecular interactions, Ovalbumin, Oxidative damage, Zingiber officinalis roscoe L.
Introduction
Asthma is an inflammatory chronic disease that affects respiratory airways. It associates with a harsh and occasional airflow restriction, hyper-responsiveness, and diverse respiratory symptoms. The disease’s epidemiology is still grown throughout the World (Mims, 2015). According to the World Health Organization (WHO), 300 million people suffer from asthma, which constitute the third leading cause of death in the World (Delmas and Fuhrman, 2010). In Tunisia, a 15-years epidemiological survey estimated its incidence to 15.4%, with regional disparities across the country (Joobeur et al. 2015). It is a multifactorial disease for which the pathophysiologic process is still debated in term of management and prevention. In most cases, asthma is linked to a state of inflammation as a primary cause (Khan et al. 2015; Badraoui et al. 2021a). The disease management and related work absenteeism lead to important socioeconomic burden.
Current treatment of asthma is mainly based on use of bronchodilator, particularly corticosteroids, beta agonist and several other inhaled anti-inflammatory medications. Besides their costs, the continual use of such medicines sounds to be associated with several drawbacks and toxicological disorders as assessed by absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties or pharmacokinetics (Badraoui et al. 2021b; Ade et al. 2017). Recently, a great interest was highlighted regarding the efficiency of natural products and their beneficial effects against several pathologies including asthma and several other respiratory diseases. This might constitute an adequate alternative medication to prevent and manage such health disorders. In particular, plant-derived substances were shown to alleviate inflammation and its associated diseases (Saoudi et al. 2021; Akacha et al. 2022; Mzid et al. 2017; Zammel et al. 2021a). They provide promising content for drug design and development (Rouhi et al. 2006; Badraoui et al. 2020). In this context, Zingiber officinalis roscoe L. (ZO) has a long history of nutritional and medical usage. In Ayurvedic medicine, it is considered as a great medicament because of its huge beneficial virtue (Afzal et al. 2001; Zammel et al. 2018, 2021a). It is also ranked among the most used spices in Chinese medicine (Jittiwat and Wattanathorn, 2012). ZO was proven to possess anti-inflammatory, antioxidant, antipyretic, antibacterial, antiviral, and antidiabetic effects (Zammel et al. 2021a; Gupta and Sharma, 2014). Ginger was mentioned in Ayurvedic and Tibb medicine as a helpful treatment against migraine, neurological disorders, nausea, stomachache, diarrhea, and toothache (Borrelli et al. 2005; Haksar et al. 2006; Podlogar and Verspohl 2012). Accordingly, it was reported to suppress hypersensitivity to allergens and might reduce asthma incidence. The cumulus of these biological activities might certainly be linked to its richness in several phytochemical components with significant biological activities (Zammel et al. 2021b, 2018; Gupta and Sharma, 2014). Nevertheless, the effect of ZO on asthmatic diseases is not well documented and need to be further studied to justify the ethnobotanical use of the plant and to detail its mechanistic possible ameliorative effects.
Hence, this work aimed to investigate the potential effects of ZO aqueous extract on a very reproducible murine experimental model of asthma due to ovalbumin sensitization. Furthermore, an in silico simulation was carried out to check the intermolecular interactions of the plant phytochemical compounds with signal transducer and activator of transcription 6 (STAT6) and tumor necrosis factor-alpha (TNF-α), which are commonly involved in asthma, specifically the chronic inflammation of the lower respiratory tract (Li et al. 2018).
Materials and methods
Chemicals and reagents
All used chemicals were of analytical grade and purchased from Sigma–Aldrich Company.
Plant material
Ginger rhizomes (Zingiber officinalis roscoe L.) were obtained from a local market. The plant material was dried and grinded, using Moulinex the original grinder (AR110O27), to obtain a fine powder. The plant material was water-macerated for 24 h before preparing concentrations of 50 g/L. After cooling in room temperature, the obtained solutions were filtrated using Whatman No. 1 paper and orally administrated to rats to give final doses of 207 mg/kg BW (D1) or 414 mg/kg BW (D2). The selection of dose referred to previous published reports and common use in ethno-pharmacological trials (Zammel et al. 2021a, b).
Animal breeding
Our study was performed on 20 mature male Albino Wistar rats weighting about of 250 g. They were housed, 5%, at a temperature of 23 ± 2 °C, 40–50% of air humidity and 12/12 light–dark cycle. They were fed standard calibrated food-pellet and drinking water was given ad libitum. Animals were allowed to acclimate to the animal house condition for at least 1 week, before the commencement of the experiment.
Study design and experimental procedure
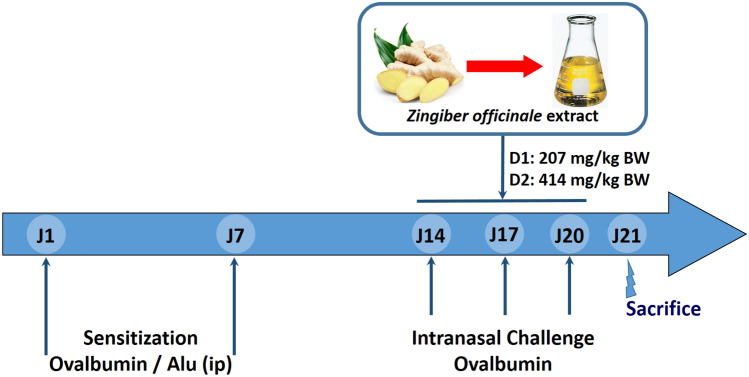
Animals were randomly divided into four groups of five rats each. The first group served as negative control and continue to have standard food pellets and tap water for drinking (Control group). Other groups were sensitized by intraperitoneal injections of 10 µL of ovalbumin-alu solution (20 mg/mL), at the 1st and the 7th days. At 14th, 17th, and 20th days, the rats were stimulated by intranasal installation of 20 μL of ovalbumin solution (0.4 mg/mL). One group was kept untreated (Oval group: the positive control group) and the two others received either 207 mg/kg BW or 414 mg/kg BW of the ginger water–maceration per gavage (Oval + D1 and Oval + D2, respectively). The treatment by the Ginger water–maceration was given from the 14th to the 20th days of the experimental procedure (Fig. 1). Rats were euthanized by cervical dislocation under general anesthesia using 8% chloral hydrate (400 mg/100 g BW) at the 21st day. Blood samples and lungs were collected for hematological, histological, and biochemical analyses. All animal procedures were performed in accordance with the Guidelines for Care and Use of Laboratory Animals of Sfax University and approved by its local (Faculty of Medicine—University of Sfax, 12ES15, 2018–21) Animal Ethics Committee.
Fig. 1.
Scheme of the experimental procedure: sensitization, challenge, and treatment protocols for the different experimental groups: Control, Oval, Oval + D1, and Oval + D2
Some of the blood samples (0.5 mL) have been collected in EDTA containing tubes and used for hematological analysis. For biochemical analysis, the blood samples were centrifuged at 1500× g for 15 min and plasma was separated and stored at –80 °C until analyzed.
The lungs were gently removed and separated from adjacent tissues and weighted. The right lung was used for histological examination. The remaining pulmonary tissues were homogenized into a buffered saline solution (1:2, w/v) and centrifuged at 5000× g for 30 min at 4 °C. The collected supernatants were aliquoted, stored at –80 °C until analyzed and served to determine the oxidative stress parameters.
Hematological and biochemical analyses
Hematological parameters were determined using automated apparatus (KX21 hemogram, CHU Habib Bourguiba, Sfax, Tunisia). The assessed parameters include the numeration of white blood cells (WBC), red blood cells (RBC), hematocrit (HCT), hemoglobin (HGB), mean globular volume (VGM), and platelets (PLT). Aspartate aminotransaminase (ASAT), alanine aminotransaminase (ALAT), C-reactive protein (CRP), urea, and creatinine were analyzed using specific kits from Biolabo (France) and used according to the manufacturer’s instructions. Both hematological and biochemical analyses have been carried in Hédi Chaker University Hospital (Sfax, Tunisia), specifically the Hematology and Biochemistry labs, respectively.
Evaluation of oxidative stress markers
To prove the effect of asthma on oxidative stress status, the prepared lung tissues homogenates were used to quantify MDA, the advanced oxidation of protein products (AOPP), GPx activity and GSH. One gram of lungs was cut into small pieces, immersed in 2 mL ice-cold and Tris-buffered saline (TBS, pH = 7.04) then centrifugated (5000× g, 30 min, 4 °C). Supernatants were collected and used.
The MDA assessment method is based on the use of thiobarbituric acid (TBA) as previously reported by Draper and Hadley (1990). The assay requires 0.5 mL of supernatant that is mixed with 1 mL of trichloroacetic acid (TCA, 30%). After centrifugation (2500× g, 10 min) and addition of TBA (1 mL, 0.67%), 0.5 mL of the supernatant was incubated at 90 °C for 15 min. The reaction with TBA produces a colored pigment that is assayed by measuring the optical density at 532 nm. Values for thiobarbituric acid reactive substances (TBARS) were expressed as nmoles/mg of protein.
The AOPP assay is based on the method described by Kayali et al. (2006). The concentration of each sample in AOPP was calculated using the extinction coefficient (261 cm−1 mM−1). The AOPP was expressed in nmoles/mg of protein.
GPx assay was realized according to the method described by Flohé and Günzler (1984) and Akacha et al. (2022). Briefly, at a pH 7 and a temperature of 25 °C, one unit of GPx was characterized as an oxidation by H2O2 of 1 µmol of reduced glutathione (GSH) per min. The activity was measured spectrophotometrically at 430 nm.
GSH assay principle is based on the reaction of the thiol group of GSH with the DTNB (5,5'-dithio-bis-2 nitrobenzoic acid, Ellman reactive). The product of the reaction is reduced by GSH reductase to recycle GSH and produce more TNB (5-thiol-2-nitrobenzic acid). This production is proportional to the concentration of GSH existed in the homogenate. The absorbance was measured spectrophotometrically at 412 nm as previously reported (Ellman 1959; Jollow et al. 1974).
Lung histological analysis
The morphology of the lung was examined to investigate structural damage associated with ovalbumin-induced asthma and inflammation. The lung tissues prepared for standard histological examination were fixed in 10% formalin for 24 h at 25 °C, dehydrated, embedded in paraffin, and sectioned at 4-μm thick. The tissue sections were then mounted on glass slides, dewaxed and stained with standard hematoxylin and eosin (H&E) as previously published (Amri et al. 2017; Hakim et al. 2008; Akacha et al. 2022).
In silico simulation and intermolecular interactions
The tridimensional structure of the tumor necrosis factor-alpha (TNF-α; ID: 1KD7) and signal transducer and activator of transcription 6 (STAT6; ID: 5D39) were obtained from RCSB data bank. The main previously identified components of ZO (6-gingerol, 8-gingerol, 10-gingerol, 6-shogaol, caffeic acid, rosmarinic acid, syringic acid, amentoflavone, and ferulic acid) were collected from PubChem websites or drawed using ChemDraw 4.1 and saved in pdb format. A blind binding based on the CHARMm force field was used in the complex formations (Mhadhbi et al. 2022; Hchicha et al. 2021). The ligands were assessed for their ability to the targeted macromolecules (TNF-α and STAT6) after removing water molecules and adding polar hydrogens and Kollman charges as previously reported (Akacha et al. 2022; Zammel et al. 2021a; Badraoui et al. 2021b). Both receptors are involved in classical immune responses linked to asthma and several associated diseases including pathogenesis and signaling pathways of asthma itself (Li et al. 2018).
Statistical analysis
All the results collected were analyzed with the Statistical Package for Social Sciences (SPSS) for windows version 20.0 software package. All data were expressed as the mean ± standard deviation (SD). Data were subjected to ANOVA followed by Tukey post hoc test, to elucidate if there were significant differences between the studied groups. The statistical probability of p < 0.05 was considered significant.
Results
Blood hematology and biochemistry analysis
Table 1 exhibits blood cells numeration and biochemical changes due to the various treatment. The WBC count was significantly increased by the asthma induction (12.84 ± 1.17 × 103 µL−1 in comparison to control negative group (7.34 ± 4.63 × 103 µL−1). This change was fully reestablished by the daily oral intake of ginger solution at a dose of 414 mg/kg BW (7.19 ± 4.47 × 103 µL−1). The distribution of immune cells suggests a huge mobilization of lymphocytes as indicated by their percent coming over 85% in ovalbumin-sensitized animals (Oval, Oval + D1, and Oval + D2). Alleviative effect was noted with ginger treatment and the effect was dose–dependent. The ameliorative effect was more prominent with D2 once compared with D1. Furthermore, rates of granulocytes and monocytes were disrupted following asthma induction. RBC count falls down in response to ovalbumin (3.80 ± 3.80 × 106 µL−1, in comparison to control and Oval + D2 groups (7.09 ± 2.38 × 106 µL−1 and 6.84 ± 2.29 × 106 µL−1, respectively).
Table 1.
Hematological and biochemical parameters for the different experimental groups
| Groups | ||||
|---|---|---|---|---|
| Control | Oval | Oval + D1 | Oval + D2 | |
| Hematological parameters | ||||
| WBC (103/µL) | 7.34 ± 4.63b | 12.84 ± 1.17a,d | 9.42 ± 1.25 | 7.19 ± 4.47b |
| RBC (106/µL) | 7.09 ± 2.38b | 3.80 ± 3.80a,d | 5.41 ± 1.70 | 6.84 ± 2.29b |
| HCT (%) | 30.94 ± 15.20 | 18.92 ± 7.92 | 27.64 ± 5.79 | 31.36 ± 9.95 |
| HGB (g/dL) | 11.02 ± 4.18 | 6.92 ± 2.76 | 9.98 ± 1.80 | 9.88 ± 4.59 |
| MGV (fL) | 49.5 ± 2.81 | 46.3 ± 3.82 | 44.02 ± 1.45 | 51.02 ± 10.94 |
| PLT (103/µL) | 521 ± 335 | 281 ± 182 | 536 ± 297 | 716 ± 373 |
| Biochemical parameters | ||||
| Urea (mmol/L) | 7.30 ± 0.75b,c | 12.64 ± 1.09a,c,d | 9.4 ± 1.22a,b | 8.15 ± 1.43b |
| Creatinine (µmol/L) | 23.60 ± 4.16 | 26.60 ± 3.65 | 26.8 ± 4.09 | 23.40 ± 7.27 |
| ALAT (U/L) | 98 ± 39 | 108 ± 42 | 133 ± 62 | 164 ± 104 |
| ASAT (IU/L) | 345 ± 104b,c | 782 ± 150a,c,d | 517 ± 139a,b | 411 ± 74b |
| CRP (mg/L) | 37.6 ± 7.44b | 49.00 ± 10.17a,b,c | 36.00 ± 6.96b | 36.20 ± 7.98b |
Data represent mean ± SD.
p <0.05 by ANOVA and Tukey post hoc test
ap < 0.05 versus Control group
bp < 0.05 versus Oval group
cp < 0.05 versus Oval + D1 group
dp < 0.05 versus Oval + D2 group
In this study, the biochemical analysis showed further changes in hemostasis resulting from asthma induction due to ovalbumin treatment. In fact, there were significant variations in urea, CRP and ASAT concentrations in Oval group once compared with control rats. These changes were rectified by the oral intake of ZO extract, particularly with D2.
Oxidative stress analysis
As shown in Fig. 2, both MDA and AOPP levels showed statistically significant increase in Oval group in comparison to control. The oral intake of the plant extract significantly (p < 0.05) reduced the lipid peroxidation induced by ovalbumin sensitization, in a dose dependent manner.
Fig. 2.
Effect of the different experimental treatments on MDA, AOPP, GPx, and GSH parameters for the different groups: Control, ovalbumin-sensitized rats (Oval), ovalbumin-sensitized rats receiving oral ginger aqueous solution at 207 mg/kg BW (D1) or 414 mg/kg BW (D2) (Oval + D1 and Oval + D2, respectively). *p < 0.05 vs Control; #p < 0.05 vs Oval and §p < 0.05 between Oval + D1 and Oval + D2
Lower panels of Fig. 2 exhibit a significant (p < 0.05) levelling-down of both lungs’ glutathione content and glutathione peroxidase activity in animals with induced asthma. However, the administration of ZO tended to reestablish the GSH level, which was significantly augmented in a dose dependent manner following D1 and D2 intake in comparison to the positive control group (11.63 ± 0.62 nmoL/mg of protein for Oval group).
Histological findings
The microscopic observation showed slightly collapsed alveoli with abundant infiltration of immune cells in asthma-like rats; the ovalbumin group (Fig. 3). Furthermore, Oval-treated rats exhibited a disorganized columnar epithelium with mostly desquamating epithelial cells, paler cytoplasm, and cell membrane denaturation. Once compared with control rats, the histological sections showed a thickening of bronchiole lining tissues in ovalbumin-sensitized rats associated bronchial spasm aspects. The oral intake of ginger water–maceration reduced the outlined histopathological features (alveoli collapse and the inflammatory aspects) seemingly in a dose dependent manner. In fact, the beneficial effect was more prominent in Oval + D2 in comparison with Oval + D1.
Fig. 3.
Microscopic slides of lung tissues (Alveoli: A–D and bronchioles: A’–D’) in control (A and A’), ovalbumin-sensitized non-treated (B and B’) and treated rats by ginger aqueous extract, respectively, at doses of 207 mg/kg BW (C and C’) 414 mg/kg BW (D and D’). Magnification ×400. AL alveolar lumen, BL bronchiole’s lumen, BM basement membrane, BV blood vessels, AC alveolar channel, CE columnar epithelium, E endothelial cell, F fibroblast, M muscle bundles, P (1, 2) pneumocyte type 1 and 2, S inter-alveolar septum, and (*) infiltrated immune cells
Computational findings
For better understanding the mechanism by which ZO extract persuaded its efficacy, an in silico analysis was realized for the plant phytochemical compounds, which have been previously identified. The binding affinities, with some key receptors have been assessed; STAT6 and TNF-α. Our results showed that all the compounds interacted with the targeted receptors with negative free binding affinities. Interestingly, the binding affinity ranged between –6.1 to –10.2 kcal/mol and –5.1 to –8.1 kcal/mol for STAT6 and TNF-α, respectively. Furthermore, all the ZO assessed compounds were deeply embedded in the targeted proteins. The distance between ligand and receptor ranged between 1.80 and 2.83 Å (Table 2). Amentoflavone was predicted to possess the best binding score and interacted with seven residues in both STAT6 and TNF-α (Fig. 4). Rosmarinic acid established the highest number of conventional H-bonds (n = 7) with TNF-α macromolecule (Fig. 5). It was also well embedded in it, only 1.80 Å. The molecular interactions study revealed that several key residues were involved during the formation of protein–ligand complexes.
Table 2.
Binding affinity (kcal/mol), closest interacting residues, and conventional hydrogen bonds of ZO phytochemical compounds and the selected receptors: STAT6 and TNF-α
| Compound (Ligand) | Target receptors | Molecular interactions | |||
|---|---|---|---|---|---|
| Binding affinity (kcal/mol) | No. closest interacting residue | Closest interacting residues (Distance, Å) |
No H-bonds | ||
| 6-Gingerol | STAT6 | –6.1 | 5 | Pro396 (2.21), Gly397 (2.06), Phe301 (3.61), Leu394 (5.39), Ile401 (5.40), Phe301 (5.08) | 2 |
| TNF-α | –5.7 | 8 | Lys184 (2.05), Lys215 (2.23), Gln260 (2.58), Gln260 (2.17), Tyr163 (4.92), Lys160 (5.35), Ile158 (4.41), Ile158 (3.97), Leu226 (5.27), Phe165 (4.34) | 4 | |
| 8-Gingerol | STAT6 | –6.3 | 6 | Lys480 (2.90), Lys480 (2.83), Asp565 (3.37), Lys480 (3.77), Leu487 (5.17), Leu475 (4.69), Leu475 (4.84), Phe476 (4.71), Tyr546 (4.22), Tyr546 (4.17) | 2 |
| TNF-α | –5.1 | 6 | Gln148 (2.63), Gln148 (2.55), Gln148 (2.43), Ser171 (2.39), Phe194 (5.38), Tyr196 (4.40), Tyr196 (3.89), Phe278 (4.96), Ile150 (4.71) | 4 | |
| 10-Gingerol | STAT6 | –6.2 | 5 | Lys480 (2.45), Asp565 (1.99), Phe476 (5.28), Lys480 (5.20), Leu507 (5.40), Leu507 (4.28), Leu508 (5.45), Leu507 (5.17) | 2 |
| TNF-α | –5.2 | 6 | Tyr206 (2.15), Tyr206 (2.86), Ala207 (2.55), Tyr163 (2.67), Pro264 (4.08), Pro264 (4.52), Ile233 (5.32), Leu211 (5.32), Ile233 (5.42), Tyr163 (4.75), Pro264 (5.08) | 4 | |
| 6-Shogaol | STAT6 | –6.4 | 9 | Lys205 (2.57), Lys205 (2.54), Gln209 (2.84), Arg437 (2.60), Met435 (2.41), Ser433 (3.68), Gln202 (3.67), Arg437 (5.05), Pro439 (5.33), Pro439 (4.70), Leu406 (4.37), Lys205 (4.65), Arg206 (4.83), Met435 (4.58), Lys205 (5.23) | 5 |
| TNF-α | –5.6 | 3 | Lys184 (1.94), Gln260 (2.36), Gly161 (2.53), Lys160 (5.05), Ile158 (4.38), Ile158 (4.64), Lys160 (4.36), Phe165 (4.37), Lys160 (5.02) | 6 | |
| Amentoflavone | STAT6 | –10.2 | 7 | Arg322 (2.76), Arg322 (2.10), Glu318 (2.77), Glu318 (3.71), Glu318 (3.53), Glu377 (3.43), Glu444 (3.56), Glu444 (3.89), Trp499 (5.09), Trp499 (4.61), Trp499 (5.37), Cys371 (5.35) | 3 |
| TNF-α | –8.1 | 7 | Lys160 (2.74), Lys160 (2.50), Lys184 (2.89), Gln260 (2.91), Glu182 (2.45), Trp168 (2.47), Ile158 (3.55), Phe165 (5.19), Ile158 (5.46), Lys184 (4.78) | 6 | |
| Caffeic Acid | STAT6 | –6.9 | 8 | Val275 (2.14), Arg437 (2.70), Arg437 (2.81), Ser407 (2.28), Leu406 (2.29), Gln208 (2.98), Val275 (1.94), Pro409 (3.50), Lys205 (4.84), Lys 205 (3.82), Val275 (5.01), Leu408 (5.49) | 6 |
| TNF-α | –5.9 | 4 | Thr157 (2.96), Gln269 (2.42), Ser269 (2.90), Ser153 (2.12), Ser153 (2.54), Leu272 (3.89) | 5 | |
| Ferulic acid | STAT6 | –7.0 | 6 | Gln209 (2.53), Val275 (2.18), Arg437 (2.14), Arg437 (2.73), Gln208 (2.39), Val275 (2.19), Leu408 (3.52), Phe440 (4.89), Lys205 (5.09) | 7 |
| TNF-α | –5.6 | 5 | Tyr206 (2.15), Arg231 (2.14), Cys232 (1.94), Arg231 (3.57), Pro264 (4.56), Arg265 (5.27) | 3 | |
| Rosmarinic acid | STAT6 | –6.9 | 6 | Leu399 (2.05), Ser236 (2.04), Leu394 (2.31), Pro396 (2.30), Leu394 (2.23), Tyr235 (5.01), Pro396 (4.63), Leu399 (5.42), Ile401 (4.76) | 5 |
| TNF-α | –6.4 | 5 | Thr157 (2.64), Asn267 (2.25), Gln269 (2.75), Gln269 (2.89), Ile270 (1.80), Ser153 (2.31), Gln269 (2.21), Leu272 (4.48) | 7 | |
| Syringic acid | STAT6 | –6.3 | 8 | Gln278 (2.21), Arg437 (2.32), Arg437 (2.65), Leu406 (2.15), Gln209 (2.55), Leu408 (3.45), Lys205 (4.29), Arg206 (4.75), Phe440 (5.03), Lys205 (4.96) | 5 |
| TNF-α | –5.3 | 8 | Gln144 (2.55), Asp145 (2.38), Asp145 (2.40), Thr143 (2.09), Asp145 (2.40), Asp145 (3.75), Lys283 (3.64), Leu284 (3.74), Val142 (4.52), Asp145 (3.91), Glu189 (4.30), Lys283 (3.55), Leu285 (4.48) | 5 | |
Bold residues: interaction via conventional H-bond
Underlined residues: closest interacting residue
Fig. 4.
Illustration of the interactions within the Amentoflavone and STAT6 complex, which showed the best free binding energy (–10.2 kcal/mol). A The 3D structure of amentoflavone. B Amentoflavone docked to the 3D ribbon structure of STAT6. C and D 3D and 2D diagrams of interactions, respectively. E and F Representation of hydrophobicity and H-bond interactions, respectively
Fig. 5.
Illustration of the interactions within the rosmarinic acid and TNF-α complex, which showed the highest number of conventional H-bonds (n = 7). A The 3D structure of rosmarinic acid. B Rosmarinic acid docked to the 3D ribbon structure of TNF-α. C and D 3D and 2D diagrams of interactions, respectively. E and F Representation of hydrophobicity and H-bond interactions, respectively
Discussion
Asthma represents a variable level of chronic airway obstruction, which is frequently associated with bronchoconstriction and inflammation within the airway that causes a wide range of decrease in lung performance (Mims, 2015). Asthma affects many individuals worldwide. It causes severe morbidity and even mortality if exacerbated as several pulmonary complications (Badraoui et al. 2021a). Currently, a wide variety of drugs have been developed to treat asthma; these include leukotriene inhibitors, mast cell stabilizer steroids, and beta2 adrenergic agonists. To date, many herbal medicines were commonly used as ethno-pharmaceutical way to treat or alleviate asthma, despite that the therapeutic efficacies and action modes of several used herbal medicines are currently unclear. In the present study, we investigated the anti-asthmatic effect of ZO extract in OVAL sensitized/challenge rat asthma model. Our results demonstrated disrupted hematological parameters (WBC, RBC, and PLT), oxidative injury (MDA, AOPP, and SOD) within the pulmonary tissues associated severe pathological features in the lung parenchyma as assessed by both serum biomarkers (creatinine, urea, uric acid) and histopathological examination. These findings supported those previously reported by Bayrami and Boskabady (2012) and others. It also confirms that OVAL sensitized/challenge rat is a suitable and reproducible asthma model due to inflammatory development within the lung tissues. Furthermore, our results showed that ZO was efficient against OVAL induced lung inflammation by moderating, oxidative damage, biochemical, and histological injuries.
ZO have been associated with several biological functions including antiemetic, antidiabetic antibacterial, antifungal, and antiviral effects. Recently, there have been various reports about ZO that show potential anti-inflammatory and antioxidant effects (Zammel et al. 2021a, b).
Asthma inflammatory disease of the airways is commonly associated with increased production of reactive oxygen species (ROS) creating an unbalance of the pro-oxidants and antioxidants (Badraoui et al. 2007; Hussain et al. 2016). Indeed, recent research suggests that oxidative stress is involved in the pathophysiology of several diseases including asthma and could be partially associated with a deficiency of antioxidant elements (Saoudi et al. 2021; Misso et al. 2005; Zammel et al. 2018; Hakim et al. 2008). In this context, GPx is the key antioxidant enzyme that regulates the level of ROS by its ability to reduce not only hydrogen peroxide in water, but also hydroperoxides resulting from the oxidation of unsaturated fatty acids. In addition, this enzyme would manage to protect cells against damage generated by foreign agents such as ovalbumin (Bano et al. 2012). Its significant decrease, in the present study, would be mainly due to over production of hydrogen peroxide and the exhaustion of GSH. This decrease can be explained by the elimination and detoxification of the allergen by GSH (Dasari et al. 2018). Indeed, several studies reported a decrease in the GPx activity in serum and erythrocytes of patients with asthma (Al-Afaleg et al. 2011).
Our findings recorded a significant decrease in the GSH level in the OVAL group, which is in agreement with previous studies (Cho et al. 2004; Mukherjee et al. 2017).
Also, the MDA parameter is an early indicator of foreign aggression and is often used as a biomarker of oxidative stress (Amri et al. 2017; Akacha et al. 2022; Badraoui et al. 2007). In our study, Ovalbumin constituted a foreign aggression, thus following its administration, the level of MDA increased in the ovalbumin-sensitized rats, which may confirm the occurrence of damage on the cell membranes of lung tissue. This finding is in agreement with the results of other studies, which have highlighted oxidative injury, specifically increased lipid peroxidation after treatment with various allergens (ovalbumin, mites, etc.) (Mukherjee et al. 2017). In this same context, it has been proved that the impact of ovalbumin on antioxidant enzymes activities resulted on increased amounts of free radicals as a result of increased number of activated inflammatory cells in the lung airways, which can initiate a lipo-peroxidation (Al-Harbi et al. 2016). AOPP findings in our study was supported by another work (Tokac et al. 2017) that showed a significant increase of AOPP level in the sensitized group.
This study outlined an improvement effect of ZO extract. In fact, the treatment with this extract excreted a strong protective effect on OVAL induced oxidative damage. These oxidative stress assessed parameters play a key role in maintaining the redox homeostasis under normal physiological conditions (Dadkhah et al. 2014). ZO extract alleviated the change of these parameters. The ameliorative effect may be explained by enhancing the antioxidant activity. Furthermore, ZO was reported to inhibit the xanthine oxidase system, which mediates the production ROS like superoxide anion (Aeschbach et al. 1994). The histopathological changes confirm both the biochemical and hematological findings. These results paralleled and confirmed the in silico findings.
The interactions of ZO phytochemical compounds with STAT6 and TNF-α showed good free binding energies, which ranged between −5.1 and −10.2 kcal/mol. The variations in binding energies values are mainly related to the chemical 3D structure of the ligands (Badraoui et al. 2021b; Zammel et al. 2021a, b). The studied ZO compounds were sufficiently embedded in the targeted proteins, with distance varying between 1.80 and 2.83 Å, which further support that the biological effect is thermodynamically possible (Mhadhbi et al. 2022; Hchicha et al. 2021). While amentoflavone was predicted to possess the best binding affinity (–10.2 kcal/mol) and interacted with the highest number of residues (n = 7) of either STAT6 or TNF-α, rosmarinic acid established the highest number of conventional H-bonds (n = 7) with TNF-α and showed the deepest embedding (1.80 Å only). Deep embedding was reported to be involved in significant biological effects and also strengthen the stability of ligand–receptor complex (Mhadhbi et al. 2022; Badraoui et al. 2021b; Zammel et al. 2021a). Overall, the molecular interactions results revealed that several key residues were involved during the formation of protein–ligand complexes. Taking into account the importance of the key residues (Zammel et al. 2021a, b; Hchicha et al. 2021; Mhadhbi et al. 2022), the free binding energies (Akacha et al. 2022; Hchicha et al. 2021), and the number of conventional H-bonds (Akacha et al. 2022; Badraoui et al. 2021b) in the pharmacological potential, it could be deduced that ZO phytochemical compounds satisfactorily explain their biological activities and their potential anti-asthmatic and antioxidant effects. The results confirmed previous reports on the antioxidative and anti-inflammatory effects of ZO extract. ZO extract was shown to exert comparative effect to indomethacin as a nonsteroidal anti-inflammatory drug (NSAIDs) (Zammel et al. 2021a). It also supports the findings of a recent study, in which ZO, particularly 6-shogaol mitigated lung inflammation and kidney injury in rats (Yocum et al. 2020; Zammel et al. 2022).
Conclusions
Taken together, the finding of our presents study outline the potential therapeutic effect of ZO administration in a rat model of ovalbumin-induced asthma. The plant material was reported to modulate the oxidative injury, hematological, biochemical and histological parameters. These promising effects suggest that ZO phytochemical compounds are expected to be promising compounds for further analyses for dual pneumo-pathological drugs, particularly anti-asthmatic medication. The ameliorative effects may include antioxidant potential and both STAT6 and TNF-α pathways. Further analyses such as isothermal titration calorimetry (ITC), surface plasmon resonance (SPR) or microscale thermophoresis (MST) experiments may provide additional values and support the current findings for better understood of molecular interactions in real time, thermodynamic parameters of the binding interactions (affinity, enthalpy, and stoichiometry), and detect the ZO phytochemical movement.
Acknowledgements
Authors would like to express their gratitude to Dr. Rim Chaabane for her biochemical support. This research received grants from the deanship of scientific research, University of Ha’il. Project number: RG-21 100.
Author contribution
Conceptualization, O.J., T.R., and R.B.; methodology, O.J., H.B.N., and R.B.; Experimental analysis: O.J., N.Z., T.R., and R.B; validation, W.S.H., S.E., A.J., and R.B.; formal analysis, N.Z., A.E.S, and O.J.; investigation, M.S., H.N., and R.B.; resources, T.R., and R.B.; data curation, N.Z., A.J.S., H.N., and R.B.; software, O.J., H.B.N., and R.B.; writing—original draft preparation, O.J., H.B.N., and R.B.; writing—review and editing, O.J. and R.B.; visualization, M.M.A., A.J.S., A.E.S., and R.B.; supervision, H.B.N., and R.B.; project administration, R.B. All authors have read and agreed to the published version of the manuscript.
Funding
None molecular data accession numbers for16S rRNA gene, other rRNA genes, ITS, WGS, SRA etc., or culture collection numbers for new taxa have been used in this work.
Declarations
Conflict of interest
The authors declare that they have no conflicts of interest in the publication.
Informed consent
Not applicable for this study.
Contributor Information
Olfa Jedli, Email: olfa.jedli92@gmail.com.
Hmed Ben-Nasr, Email: hmedbnasr@gmail.com.
Nourhène Zammel, Email: nourhene.zammel@gmail.com.
Tarek Rebai, Email: tarek.rebai@fmsf.rnu.tn.
Mongi Saoudi, Email: saoudimm@yahoo.fr.
Salem Elkahoui, Email: s.elkahoui@uoh.edu.sa.
Arshad Jamal, Email: arshadjamalus@yahoo.com.
Arif J. Siddiqui, Email: arifjamal13@gmail.com
Abdelmoneim E. Sulieman, Email: abuelhadi@hotmail.com
Mousa M. Alreshidi, Email: mousa.algladi@gmail.com
Houcine Naïli, Email: houcine_naili@yahoo.com.
Riadh Badraoui, Email: riadh.badraoui@fmt.utm.tn.
References
- Ade S, Adjibode O, Awanou B, et al. Résultats de la prise en charge de l’asthme persistant sévère dans des conditions de ressources limitées : expérience du Bénin. Rev FrAllerg. 2017;57(3):271. doi: 10.1016/j.reval.2017.02.171. [DOI] [Google Scholar]
- Aeschbach R, Löliger J, Scott BC, et al. Antioxidant actions of thymol, carvacrol, 6-Gingerol Zingerone and Hydroxytyrosol. Food Chem Toxicol. 1994;32(1):31–36. doi: 10.1016/0278-6915(84)90033-4. [DOI] [PubMed] [Google Scholar]
- Afzal M, Al-Hadidi D, Menon M, et al. Ginger: an ethnomedical, chemical and pharmacological review. Drug Metab Drug Interact. 2001;18(3–4):159–190. doi: 10.1515/DMDI.2001.18.3-4.159. [DOI] [PubMed] [Google Scholar]
- Akacha A, Badraoui R, Rebai T, et al. Effect of Opuntia ficus indica extract on methotrexate-induced testicular injury: a biochemical, docking and histological study. J Biomol Struct Dyn. 2022;40(10):4341–4351. doi: 10.1080/07391102.2020.1856187. [DOI] [PubMed] [Google Scholar]
- Al-Afaleg NO, Al-Senaidy A, El-Ansary A. Oxidative stress and antioxidant status in Saudi asthmatic patients. Clin Biochem. 2011;44(8–9):612–617. doi: 10.1016/j.clinbiochem.2011.01.016. [DOI] [PubMed] [Google Scholar]
- Al-Harbi NO, Nadeem A, Al-Harbi MM, et al. Airway oxidative stress causes vascular and hepatic inflammation via upregulation of IL-17A in a murine model of allergic asthma. Internat Immunopharmacol. 2016;34:173–182. doi: 10.1016/j.intimp.2016.03.003. [DOI] [PubMed] [Google Scholar]
- Amri N, Rahmouni F, Chokri MA, et al. Histological and biochemical biomarkers analysis reveal strong toxicological impacts of pollution in hybrid sparrow (Passer domesticus × Passer hispaniolensis) in Southern Tunisia. Environ Sci Poll Res. 2017;24(21):17845–17852. doi: 10.1007/s11356-017-9352-3. [DOI] [PubMed] [Google Scholar]
- Badraoui R, Sahnoun Z, Abdelmoula NB, et al. May antioxidants status depletion by tetradifon induce secondary genotoxicity in female wistar rats via oxidative stress? Pestic Biochem Physiol. 2007;88(2):149–155. doi: 10.1016/j.pestbp.2006.10.007. [DOI] [Google Scholar]
- Badraoui R, Rebai T, Elkahoui S, et al. Allium Subhirsutum L. as a potential source of antioxidant and anticancer bioactive molecules: HR-LCMS phytochemical profiling, in vitro and in vivo pharmacological study. Antioxidants. 2020;9(10):1003. doi: 10.3390/antiox9101003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badraoui R, Adnan M, Bardakci F, et al. Chloroquine and hydroxychloroquine interact differently with ACE2 domains reported to bind with the coronavirus spike protein: mediation by ACE2 polymorphism. Molecules. 2021;26(3):673. doi: 10.3390/molecules26030673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badraoui R, Alrashedi MM, El-May MV, et al. Acute respiratory distress syndrome: a life threatening associated complication of SARS-CoV-2 infection inducing COVID-19. J Biomol Struct Dyn. 2021;39(17):6842–6851. doi: 10.1080/07391102.2020.1803139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bano T, Kumar N, Dudhe R. Free radical scavenging properties of pyrimidine derivatives. Org Med Chem Lett. 2012;2:34. doi: 10.1186/2191-2858-2-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayrami G, Boskabady MH. The potential effect of the extract of Crocus sativus and Safranal on the total and differential white blood cells of ovalbumin-sensitized guinea pigs. Res Pharm Sci. 2012;7(4):249–255. [PMC free article] [PubMed] [Google Scholar]
- Borrelli F, Capasso R, Aviello G, et al. Effectiveness and safety of ginger in the treatment of pregnancy-induced nausea and vomiting. Obstet Gynecol. 2005;105(4):849–856. doi: 10.1097/01.AOG.0000154890.47642.23. [DOI] [PubMed] [Google Scholar]
- Cho YS, Lee J, Lee TH, et al. Alpha-lipoic acid inhibits airway inflammation and hyperresponsiveness in a mouse model of asthma. J Allergy Clin Immunol. 2004;114(2):429–435. doi: 10.1016/j.jaci.2004.04.004. [DOI] [PubMed] [Google Scholar]
- Dadkhah A, Fatemi F, Farsani ME, et al. Hepatoprotective effects of iranian hypericum scabrum essential oils against oxidative stress induced by acetaminophen in rats. Braz Arch Biol Technol. 2014;57:340–348. doi: 10.1590/S1516-89132014005000012. [DOI] [Google Scholar]
- Dasari S, Ganjayi MS, Yellanurkonda P, et al. Role of glutathione s-transferases in detoxification of a polycyclic aromatic hydrocarbon, methylcholanthrene. Chem Biol Interact. 2018;294:81–90. doi: 10.1016/j.cbi.2018.08.023. [DOI] [PubMed] [Google Scholar]
- Delmas MC, Fuhrman C, pour le groupe épidémiologie et recherche clinique de la SPLF. Asthma in France: a review of descriptive epidemiological data. Rev Mal Respir. 2010;27(2):151–159. doi: 10.1016/j.rmr.2009.09.001. [DOI] [PubMed] [Google Scholar]
- Draper HH, Hadley M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990;186:421–431. doi: 10.1016/0076-6879(90)86135-i. [DOI] [PubMed] [Google Scholar]
- Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959;82(1):70–77. doi: 10.1016/0003-9861(59)90090-6. [DOI] [PubMed] [Google Scholar]
- Flohé L, Günzler WA. Assays of glutathione peroxidase. Methods Enzymol. 1984;105:114–121. doi: 10.1016/s0076-6879(84)05015-1. [DOI] [PubMed] [Google Scholar]
- Gupta S, Sharma A. Medicinal properties of Zingiber officinale roscoe—a review. IOSR J Pharm Biol Sci. 2014;9(5):124–129. doi: 10.9790/3008-0955124129. [DOI] [Google Scholar]
- Hakim A, Kallel H, Sahnoun Z, et al (2008) Lack of nephrotoxicity following 15-day therapy with high doses of colistin in rats. Med Sci Monit 14(4):BR74–BR77. https://www.medscimonit.com/abstract/index/idArt/850282 [PubMed]
- Haksar A, Sharma A, Chawla R, et al. Zingiber officinale exhibits behavioral radioprotection against radiation-induced CTA in a gender-specific manner. Pharmacol Biochem Behav. 2006;84(2):179–188. doi: 10.1016/j.pbb.2006.04.008. [DOI] [PubMed] [Google Scholar]
- Hchicha K, Korb M, Badraoui R, et al. A novel sulfate-bridged binuclear copper (II) complex: structure, optical, ADMET and in vivo approach in a murine model of bone metastasis. New J Chem. 2021;45(31):13775–13784. doi: 10.1039/D1NJ02388H. [DOI] [Google Scholar]
- Hussain T, Tan B, Yin Y, et al. Oxidative stress and inflammation: what polyphenols can do for us? Oxid Med Cell Longev. 2016;2016:7432797. doi: 10.1155/2016/7432797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jittiwat J, Wattanathorn J. Ginger pharmacopuncture improves cognitive impairment and oxidative stress following cerebral ischemia. J Acupunct Meridian Stud. 2012;5(6):295–300. doi: 10.1016/j.jams.2012.09.003. [DOI] [PubMed] [Google Scholar]
- Jollow DJ, Mitchell JR, Zampaglione N, et al. Bromobenzene-induced liver necrosis. Protective role of glutathione and evidence for 3,4-Bromobenzene oxide as the hepatotoxic metabolite. PHA. 1974;11(3):151–169. doi: 10.1159/000136485. [DOI] [PubMed] [Google Scholar]
- Joobeur S, Mhamed SC, Saad AB, et al (2015) L’asthme allergique au centre tunisien. Pan Afr Med J 20 (133). 10.11604/pamj.2015.20.133.5642 [DOI] [PMC free article] [PubMed]
- Kayali R, Cakatay U, Akçay T, et al. Effect of alpha-lipoic acid supplementation on markers of protein oxidation in post-mitotic tissues of ageing rat. Cell Biochem Funct. 2006;24(1):79–85. doi: 10.1002/cbf.1190. [DOI] [PubMed] [Google Scholar]
- Khan AM, Shahzad M, Raza Asim MB, et al. Zingiber officinale ameliorates allergic asthma via suppression of Th2-mediated immune response. Pharm Biol. 2015;53(3):359–367. doi: 10.3109/13880209.2014.920396. [DOI] [PubMed] [Google Scholar]
- Li E, Landers CT, Tung HY, et al. Fungi in mucoobstructive airway diseases. Annals ATS. 2018;15(Suppl 3):S198–S204. doi: 10.1513/AnnalsATS.201803-154AW. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mhadhbi N, Issaoui N, Hamadou WS, Alam JM, Elhadi AS, Naïli H, Badraoui R. Physico-chemical properties, pharmacokinetics, molecular docking and in-vitro pharmacological study of a cobalt (ii) complex based on 2-aminopyridine. ChemistrySelect. 2022;7(3):e202103592. doi: 10.1002/slct.202103592. [DOI] [Google Scholar]
- Mims JW. Asthma: definitions and pathophysiology. Int Forum Allergy Rhinol. 2015;5(Suppl 1):S2–6. doi: 10.1002/alr.21609. [DOI] [PubMed] [Google Scholar]
- Misso NLA, Brooks-Wildhaber J, Ray S, et al. Plasma concentrations of dietary and nondietary antioxidants are low in severe asthma. Eur Respir J. 2005;26(2):257–264. doi: 10.1183/09031936.05.00006705. [DOI] [PubMed] [Google Scholar]
- Mukherjee AA, Kandhare AD, Rojatkar SR, et al. Ameliorative effects of artemisia pallens in a murine model of ovalbumin-induced allergic asthma via modulation of biochemical perturbations. Biomed Pharmacother. 2017;94:880–889. doi: 10.1016/j.biopha.2017.08.017. [DOI] [PubMed] [Google Scholar]
- Mzid M, Badraoui R, Khedir SB, et al. Protective effect of ethanolic extract of Urtica Urens L. against the toxicity of imidacloprid on bone remodeling in rats and antioxidant activities. Biomed Pharmacother. 2017;91:1022–1041. doi: 10.1016/j.biopha.2017.05.023. [DOI] [PubMed] [Google Scholar]
- Podlogar JA, Verspohl EJ. Antiinflammatory effects of ginger and some of its components in human bronchial epithelial (BEAS-2B) cells. Phytother Res. 2012;26(3):333–336. doi: 10.1002/ptr.3558. [DOI] [PubMed] [Google Scholar]
- Rouhi H, Ganji F, Nasri H. Effects of ginger on the improvement of asthma [the evaluation of its treatmental effects] Pak J Nutr. 2006;5(4):373–376. doi: 10.3923/pjn.2006.373.376. [DOI] [Google Scholar]
- Saoudi M, Badraoui R, Chira A, et al. The role of Allium Subhirsutum L. in the attenuation of dermal wounds by modulating oxidative stress and inflammation in wistar albino rats. Molecules. 2021;26(16):4875. doi: 10.3390/molecules26164875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokac M, Bacanli M, Dumlu EG, et al. The ameliorative effects of Pycnogenol® on liver ischemia-reperfusion injury in rats. Turk J Pharmaceutic Sci. 2017;14(3):257. doi: 10.4274/tjps.49369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yocum GT, Hwang JJ, Mikami M, et al. Ginger and its bioactive component 6-shogaol mitigate lung inflammation in a murine asthma model. Am J Physiol-Lung Cell Mol Physiol. 2020;318(2):L296–L303. doi: 10.1152/ajplung.00249.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zammel N, Amri N, Chaabane R, et al. Proficiencies of Zingiber officinale against spine curve and vertebral damage induced by corticosteroid therapy associated with gonadal hormone deficiency in a rat model of osteoporosis. Biomed Pharmacother. 2018;103:1429–1435. doi: 10.1016/j.biopha.2018.04.159. [DOI] [PubMed] [Google Scholar]
- Zammel N, Oudadesse H, Allagui I, et al. Evaluation of lumbar vertebrae mineral composition in rat model of severe osteopenia: a Fourier Transform Infrared Spectroscopy (FTIR) analysis. Vibrat Spectrosc. 2021;115:103279. doi: 10.1016/j.vibspec.2021.103279. [DOI] [Google Scholar]
- Zammel N, Saeed M, Bouali N, et al. Antioxidant and anti-inflammatory effects of Zingiber officinale roscoe and Allium Subhirsutum: in silico biochemical and histological study. Foods. 2021;10(6):1383. doi: 10.3390/foods10061383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zammel N, Jedli O, Rebai T, et al (2022) Kidney injury and oxidative damage alleviation by Zingiber officinale: pharmacokinetics and protective approach in a combined murine model of osteoporosis. 3 Biotech 12(5),1–16. 10.1007/s13205-022-03170-x [DOI] [PMC free article] [PubMed]