Abstract
Increased vasoactive-inotropic score (VIS) is a reliable predictor of mortality and morbidity after cardiac surgery. Here, we retrospectively evaluated the association between VIS and adverse outcomes in adult patients after off-pump coronary artery bypass grafting (OPCAB). We included 2149 patients who underwent OPCAB. The maximal VIS was calculated for the initial 48 postoperative hours using standard formulae. The primary outcome was 1-year death. The composite adverse outcome was death, resuscitation or mechanical support, myocardial infarction, revascularization, new-onset atrial fibrillation, infection requiring antibacterial therapy, acute kidney injury, and stroke. Path-analysis was conducted using lactate and prognostic nutritional index (PNI). VIS was associated with 1-year death (odds ratio [OR] 1.07 [1.04–1.10], p < 0.001) and 1-year composite outcome (OR 1.02 [1.0–1.03], p = 0.008). In path-analysis, high VIS showed a direct effect on the increased risk of 1-year death and composite outcome. In the pathway using lactate as a mediating variable, VIS showed an indirect effect on the composite outcome but no significant effect on death. Low PNI directly affected the increased risk of 1-year death and composite outcome, and had an indirect effect on both outcomes, even when VIS was used as a mediating variable. In patients undergoing OPCAB, high VIS independently predicted morbidity and 1-year death. Patients with increased lactate levels following high VIS had an increased risk of postoperative complications, although not necessarily resulting in death. However, patients with poor preoperative nutritional status had an increased risk of unfavourable outcomes, including death, implying the importance of preoperative nutritional support.
Subject terms: Cardiology, Risk factors
Introduction
The vasoactive-inotropic score (VIS) is a reliable predictor of mortality and morbidity after cardiac surgery1,2. Although several studies have evaluated the predictive value of VIS in pediatric cardiac surgery3,4, there is limited evidence for adult patients undergoing cardiac surgeries, especially those undergoing off-pump coronary artery bypass grafting (OPCAB). Previous studies have shown that a higher VIS predicts unfavourable outcomes in a dose-dependent manner1–4.
Although timely initiation of optimal vasopressor and inotrope therapy is essential for patients to maintain adequate tissue perfusion5, the use of high-dose vasoactive-inotropic agents can induce impaired tissue perfusion. Blood lactate concentrations have been widely used as markers of altered tissue perfusion, and high blood lactate is associated with major complications after cardiac surgery6,7. To date, various independent factors have been proposed, such as VIS and lactate, to predict unfavourable outcomes in coronary artery bypass grafting (CABG), but the causal relationship of variables has not yet been evaluated8,9.
Structural equation modelling (SEM) was used to evaluate the causal relationships of significant independent variables. SEM is a multivariate statistical analysis technique that can solve the problem of multicollinearity and identify relationships between explanatory variables, thus developing a more accurate prediction model for the outcome10. In the current study, we investigated whether VIS predicted unfavourable outcomes in adult patients after OPCAB. Moreover, we hypothesised that impaired tissue perfusion is the mechanism leading to unfavourable outcomes following high VIS, and consequently, we investigated whether lactate is a mediator between VIS and unfavourable outcomes using SEM. However, although VIS and blood lactate are independent predictors of unfavourable outcomes, they are not modifiable factors; thus, we conducted further analysis to identify any modifiable factors that impact outcomes, such as patients’ prognostic nutritional index (PNI) or any comorbidities.
The aim of this study was to determine independent variables predicting long-term outcomes after OPCAB within 1-year and to investigate causal relationships among independent variables.
Methods
Study population
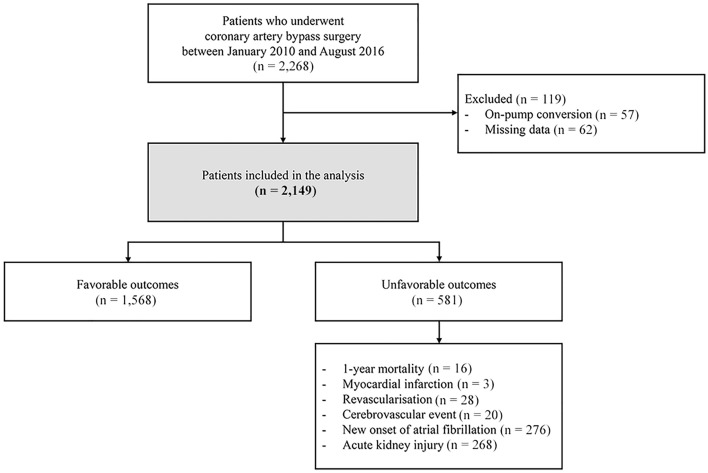
We performed a single-centre retrospective cohort study including OPCAB patients admitted to the intensive care unit (ICU) at Samsung Medical Center, Seoul, Korea. We screened the records of 2268 patients who underwent OPCAB between January 2010 and August 2016; among whom, 57 patients who underwent on-pump conversion were excluded. Of the remaining 2211 patients, 62 patients were further excluded because of missing data and readmission to the ICU (Fig. 1). This study was approved by the Institutional Review Board of Samsung Medical Center (IRB No. 2020-02-166) and the study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice Guidelines. Considering the retrospective nature and minimal risk to participants, the IRB waived the need for individual consent.
Figure 1.
Flowchart of patients.
Data collection
From January 2010 to August 2016, 2149 adult patients who underwent OPCAB were enrolled in the analysis. Our institution operates as a paperless hospital with an electronic medical record (EMR) system that archives all patient medication information and laboratory findings. Data in this study were extracted from EMRs and curated using “Clinical Data Warehouse Darwin-C”, an electronic system designed to search and retrieve de-identified medical records. After finalising the patients for inclusion in the study, independent researchers who were blinded to the perioperative medical data organised de-identified data, including baseline characteristics, clinical data, and postoperative outcomes, into a standardised form.
Maximal VIS was calculated (VIS = dopamine dose [mcg/kg/min] + dobutamine [mcg/kg/min] + 100*epinephrine dose [mcg/kg/min] + 10*milrinone dose [mcg/kg/min] + 10,000*vasopressin [units/kg/min] + 100*norepinephrine dose [mcg/kg/min]) using the maximum dosing rates of vasoactive and inotropic medications (mcg/kg/min or IU/kg/min) during the initial 48 postoperative hours after ICU admission. All the data were reviewed and validated by two independent researchers.
Postoperative lactate values and PNI were also collected as mediating variables in SEM. Maximal postoperative lactate was defined as the maximal value of lactate during the initial 48 h after ICU admission. PNI was calculated as 10 × serum albumin (g/dL) + 0.005 × total lymphocyte count (per mm3).
All patients were transferred to the specific critical care unit for cardiac surgery patients after OPCAB, and the arterial blood pressure and hemodynamic data measured by the pulmonary artery catheter were continuously monitored. Postoperative patient care was provided at the discretion of the attending ICU physician based on the institutional protocol.
Study outcomes and definitions
The primary outcome was 1-year death. The secondary outcome was a composite of postoperative complications in the year following surgery, including death, cerebral infarction, cerebral haemorrhage, myocardial infarction (MI), new-onset atrial fibrillation, and postoperative acute kidney injury. Cerebral infarction and cerebral haemorrhage were defined as new neurological deficits with new radiological evidence after cardiac surgery. MI was defined as evidence of coronary thrombus with symptoms or electrocardiographic changes compatible with ischemic aetiology according to the fourth universal definition of MI11. Postoperative acute kidney injury (AKI) was defined based on the Kidney Disease Improving Global Outcomes criteria using creatinine level. An absolute increase > 0.3 mg/dl or a relative increase of > 50% from the preoperative baseline level was defined as AKI12. Postoperative AKI in our study outcome included both the new onset AKI and aggravating AKI based on the perioperative changes of serum creatinine level. Additionally, subgroup analysis was performed to observe the effects of our study variables on long-term mortality and the composite adverse outcome in separate patient groups with different operation risks. For risk stratification, we used European System for Cardiac Operative Risk Evaluation-II (EuroSCORE-II) system, which is a widely used system for perioperative risk scoring in the cardiac surgery patients13. Based on the EuroSCORE-II distribution in our study population, low-risk group was defined as EuroSCORE-II less than 2% and high-risk group was defined as EuroSCORE-II over 2%.
Statistical analysis
Continuous variables are described as median (interquartile range [IQR]), and categorical variables are expressed as numbers (%). Baseline characteristics were compared between the favourable and unfavourable groups using the chi-square test or Fisher’s exact test for categorical variables and the Wilcoxon rank sum test for continuous variables.
A logistic regression model was used to assess the associations between associated factors and outcomes. A stepwise selection process was performed for multivariable analysis with p < 0.05 for inclusion of variables, and p > 0.10 for removal of variables. The results are described using odds ratios (ORs) with 95% confidence intervals (CIs). The predictability of increased VIS with respect to mortality up to 1 year was assessed by the area under the curve (AUC) of the receiver operating characteristics (ROC) curve.
We used VIS as a continuous variable in the most of our study analysis. However, in the additional analysis to reveal the relationship between VIS and postoperative maximal lactate or the long-term mortality, we used VIS as a categorical variable. We selected VIS cut-off values to define five groups according to the values of an adult population undergoing cardiac surgeries demonstrated in a previous study (five VIS categories: 0–5, > 5–15, > 15–30, > 30–45, and > 45 points)1.
In addition to the multivariable logistic regression analysis, we applied path analysis among various SEM models to examine the relationships between patient and perioperative variables and postoperative outcomes. The path analysis model includes independent, dependent (outcome variable), and mediating variables (e.g. lactate or PNI). We chose path analysis models that consisted of three explanatory variables. Because VIS was the most important variable in the risk factor of death, VIS was set as an independent variable, and a composite outcome, or 1-year death was set as the dependent variable. Since postoperative lactate levels indicate tissue perfusion after surgery, lactate was used as the mediating variable in the model. The same analysis was conducted for composite outcome and death. Because there were different results in the indirect effect through lactate between the 1-year death and a composite outcome, we assumed that a patient may have a different prognosis depending on the patient's own underlying nutritional status even if the patient was treated with a similar dosage of vasoactive-inotropic medication or showed a comparable increase in serum lactate. Moreover, in the original logistic regression, PNI was the only variable included in the final model for both 1-year death and composite outcomes; therefore, we added the PNI in SEM and generated an additional path analysis, including the PNI.
All reported p values were two-sided, and statistical significance was set at p < 0.05. Statistical analyses were performed using SAS (version 9.4; SAS Institute, Cary, NC), AMOS 26.0, SPSS 20.0 (IBM Corp., Chicago, IL), or R 3.5.2 (R Development Core Team, Vienna, Austria; http://www.R-project.org/).
Results
Patient characteristics
A flowchart of the patients is shown in Fig. 1. Of the 2149 patients who were included in the final analysis, 1565 received vasoactive or inotropic support within 48 h of ICU admission. In these patients who received cardiovascular drug support, VIS ranged from 0.3 to 108. The median VIS was 5 (IQR 0–10) overall, and 7 (IQR 4–12) among patients who received vasoactive or inotropic medication. Distribution of patients (%) and postoperative mortality rate (%) according to the five VIS groups are demonstrated in Supplementary Fig. S1A. Long-term mortality rate after OPCAB increased with higher VIS group (Supplementary Fig. S1A). Overall, 581 (27.1%) patients had one or more postoperative complications within 1 year, 16 patients died, 3 patients experienced myocardial infarction, 28 patients underwent coronary revascularization, 20 patients had cerebrovascular events, 276 patients showed new-onset atrial fibrillation, and 268 patients experienced acute kidney injury (Fig. 1). Compared to patients who had no postoperative complications, patients who had any of the composite outcomes were older and more likely to have a history of stroke and dialysis (Table 1).
Table 1.
Patient and clinical characteristics according to death and unfavourable outcome.
| All patients (n = 2149) | Unfavourable outcome | p value | ||
|---|---|---|---|---|
| No (n = 1568) | Yes (n = 581) | |||
| Male sex | 1672 (77.8) | 1219 (77.7) | 453 (78.0) | 0.911 |
| Age (years) | 64.0 (57.0–71.0) | 63.0 (56.0–70.0) | 66.0 (59.0–72.0) | < 0.001 |
| Smoking | 569 (26.5) | 408 (25.7) | 166 (28.6) | 0.181 |
| BMI | 24.5 (22.8–26.6) | 24.5 (22.8–26.5) | 24.6 (22.9–26.7) | 0.302 |
| Comorbidities | ||||
| Hypertension | 1705 (79.3) | 1237 (78.9) | 468 (80.55) | 0.398 |
| Diabetes | 962 (44.8) | 689 (43.9) | 273 (47.0) | 0.207 |
| History of previous MI | 187 (8.7) | 140 (8.9) | 47 (8.1) | 0.540 |
| Acute MI | 249 (11.6) | 173 (11.03) | 76 (13.1) | 0.188 |
| Previous PCI | 378 (17.6) | 287 (18.3) | 91 (15.7) | 0.153 |
| Previous CABG | 9 (0.42) | 7 (0.45) | 2 (0.34) | > 0.999 |
| PAOD | 114 (5.3) | 75 (4.8) | 39 (6.7) | 0.076 |
| COPD | 28 (1.3) | 18 (1.2) | 10 (1.7) | 0.298 |
| History of stroke | 272 (12.7) | 178 (11.4) | 94 (16.2) | 0.003 |
| Chronic kidney disease | 122 (5.7) | 82 (5.2) | 40 (6.9) | 0.141 |
| Dialysis | 61 (2.8) | 37 (2.4) | 24 (4.1) | 0.028 |
| Cancer | 70 (3.3) | 46 (2.9) | 24 (4.1) | 0.165 |
| Heart failure | 29 (1.4) | 20 (1.3) | 9 (1.6) | 0.626 |
| Valve disease | 10 (0.5) | 6 (0.4) | 4 (0.7) | 0.474 |
| Aortic disease | 15 (0.7) | 11 (0.7) | 4 (0.7) | > 0.999 |
| PTE DVT | 2 (0.1) | 2 (0.1) | 0 (0) | > 0.999 |
| Medication | ||||
| ACEi | 141 (6.6) | 97 (6.2) | 44 (7.6) | 0.249 |
| ARB | 549 (25.6) | 405 (25.8) | 144 (24.8) | 0.622 |
| Aspirin | 1981 (92.2) | 1455 (92.8) | 526 (90.5) | 0.083 |
| BB | 710 (33.0) | 499 (31.8) | 211 (36.3) | 0.049 |
| CCB | 668 (31.1) | 475 (30.3) | 193 (33.2) | 0.193 |
| Clopidogrel | 1242 (57.8) | 885 (56.4) | 357 (61.5) | 0.037 |
| Statin | 1106 (51.5) | 793 (50.6) | 313 (53.9) | 0.174 |
| Intraoperative parameter | ||||
| Anastomosis number | 4 (1–8) | 4 (1–8) | 4 (1–7) | 0.314 |
| OP duration | 260 (222–307) | 260 (221–305) | 262.0 (225.0–313.0) | 0.255 |
| RBC transfusion | 2.0 (1.0–3.0) | 2.0 (1.0–3.0) | 2.0 (1.0–3.0) | 0.712 |
Values are presented as n (%) or median (interquartile range).
BMI body mass index, MI myocardial infarction, PCI percutaneous coronary intervention, CABG coronary artery bypass grafting, PAOD peripheral artery occlusion disease, COPD chronic obstructive pulmonary disease, PTE pulmonary thromboembolism, DVT deep vein thrombosis, ACEi angiotensin-converting enzyme inhibitor, ARB angiotensin 2 receptor blocker, BB beta-blocker, CCB calcium channel blocker, OP operation, RBC red blood cell. Chi-square test or Fisher’s exact test was used for categorical variables and Wilcoxon rank sum test was used for continuous variables.
Variables associated with the composite outcome
Increased VIS during the immediate postoperative 48 h was independently associated with the occurrence of 1-year composite outcome (adjusted OR 1.02; 95% CI 1.0–1.03; p value = 0.008; Table 2). The maximum value of blood lactate within 48 h postoperatively was also independently related to the 1-year composite outcome (adjusted OR 1.21; 95% CI 1.14–1.29; p < 0.001). In addition, a higher VIS was significantly associated with increased lactate levels in a dose-dependent manner (p < 0.001; Supplementary Fig. S1B). Preoperative poor PNI was independently associated with the risk of the composite outcome (adjusted OR 0.98; 95% CI 0.96–0.99; p = 0.011). Multivariable analysis also confirmed a significant association between composite outcome and history of stroke, preoperative infectious state, and preoperative intake of aspirin and beta-blockers (Table 2). The results of subgroup analysis were consistent with the results of entire population (Supplementary Table S1A).
Table 2.
Logistic regression model for a composite outcome.
| Any unfavourable outcome (n = 581) | No unfavourable outcome (n = 1568) | Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|---|---|
| Unadjusted OR (95% CI) | p value | Adjusted OR (95% CI) | p value | |||
| VIS | 5.0 (2.0–11.0) | 5.0 (0.0–9.0) | 1.03 (1.02–1.04) | < 0.001 | 1.02 (1.00–1.03) | 0.008 |
| Lactate | 2.0 (1.1–2.8) | 1.5 (0.8–2.5) | 1.24 (1.16–1.31) | < 0.001 | 1.21 (1.14–1.29) | < 0.001 |
| PNI | 51.4 (47.7–55.0) | 52.5 (48.9–55.8) | 0.96 (0.95–0.98) | < 0.001 | 0.98 (0.96–0.99) | 0.011 |
| Demographics | ||||||
| Male sex | 453 (78.0) | 1219 (77.7) | 0.99 (0.79–1.24) | 0.911 | ||
| Age (years) | 66.0 (59.0–72.0) | 63.0 (56.0–70.0) | 1.02 (1.01–1.03) | < 0.001 | 1.01 (1.00–1.02) | 0.013 |
| Smoking | 166 (28.6) | 408 (25.7) | 1.16 (0.94–1.43) | 0.181 | ||
| BMI | 24.6 (22.9–26.7) | 24.5 (22.8–26.5) | 1.01 (0.99–1.05) | 0.303 | ||
| Comorbidities | ||||||
| Hypertension | 468 (80.55) | 1237 (78.9) | 1.11 (0.87–1.41) | 0.397 | ||
| Diabetes | 273 (47.0) | 689 (43.9) | 1.13 (0.93–1.37) | 0.207 | ||
| History of previous MI | 47 (8.1) | 140 (8.9) | 0.90 (0.64–1.27) | 0.54 | ||
| Acute MI | 76 (13.1) | 173 (11.03) | 1.21 (0.91–1.62) | 0.188 | ||
| Previous PCI | 91 (15.7) | 287 (18.3) | 0.83 (0.64–1.07) | 0.154 | ||
| Previous CABG | 2 (0.34) | 7 (0.45) | 0.77 (0.16–3.72) | 0.745 | ||
| PAOD | 39 (6.7) | 75 (4.8) | 1.43 (0.96–2.14) | 0.078 | ||
| COPD | 10 (1.7) | 18 (1.2) | 1.51 (0.69–3.29) | 0.301 | ||
| History of stroke | 94 (16.2) | 178 (11.4) | 1.51 (1.15–1.98) | 0.003 | 1.33 (1.01–1.76) | 0.045 |
| Chronic kidney disease | 40 (6.9) | 82 (5.2) | 1.34 (0.91–1.98) | 0.142 | ||
| Dialysis | 24 (4.1) | 37 (2.4) | 1.78 (1.06–3.01) | 0.030 | ||
| Cancer | 24 (4.1) | 46 (2.9) | 1.43 (0.86–2.36) | 0.167 | ||
| Heart failure | 9 (1.6) | 20 (1.3) | 1.22 (0.55–2.69) | 0.626 | ||
| Valve disease | 4 (0.7) | 6 (0.4) | 1.81 (0.51–6.42) | 0.361 | ||
| Aortic disease | 4 (0.7) | 11 (0.7) | 0.98 (0.31–3.09) | 0.974 | ||
| PTE DVT | 0 (0) | 2 (0.1) | 0.54 (0.00–6.63) | 0.744 | ||
| Infection (CRP pre) | 0.1 (0.1–0.5) | 0.2 (0.1–0.6) | 1.09 (1.04–1.14) | < 0.001 | 1.06 (1.01–1.1) | 0.022 |
| Haemoglobin | 0.93 (0.88–0.98) | 0.004 | ||||
| Creatinine | 1.01 (0.94–1.08) | 0.865 | ||||
| Medication | ||||||
| ACEi | 44 (7.6) | 97 (6.2) | 1.24 (0.86–1.80) | 0.25 | ||
| ARB | 144 (24.8) | 405 (25.8) | 0.95 (0.76–1.18) | 0.622 | ||
| Aspirin | 526 (90.5) | 1455 (92.8) | 0.74 (0.53–1.04) | 0.084 | 0.68 (0.48–0.96) | 0.029 |
| BB | 211 (36.3) | 499 (31.8) | 1.22 (1.01–1.49) | 0.049 | 1.25 (1.01–1.53) | 0.037 |
| CCB | 193 (33.2) | 475 (30.3) | 1.15 (0.93–1.40) | 0.193 | ||
| Clopidogrel | 357 (61.5) | 885 (56.4) | 1.23 (1.01–1.49) | 0.037 | ||
| Statin | 313 (53.9) | 793 (50.6) | 1.14 (0.94–1.38) | 0.174 | ||
| Operation | ||||||
| Anastomosis number | 4 (1–7) | 4 (1–8) | 0.92 (0.75–1.13) | 0.444 | ||
| OP duration | 262.0 (225.0–313.0) | 260 (221–305) | 1.00 (1.00–1.00) | 0.191 | ||
| RBC transfusion | 2.0 (1.0–3.0) | 2.0 (1.0–3.0) | 1.01 (0.95–1.08) | 0.653 | ||
VIS vasoactive inotropic score, PNI prognostic nutritional index, BMI body mass index, MI myocardial infarction, PCI percutaneous cardiac intervention, CABG coronary artery bypass graft, PAOD peripheral arterial occlusive disease, COPD chronic obstructive pulmonary disease, PTE pulmonary thromboembolism, DVT deep vein thrombosis, ACEi angiotensin-converting enzyme inhibitors, ARB angiotensin receptor blocker, BB beta-blocker, CCB calcium channel blocker, OP operation, RBC red blood cell.
Predictors of 1-year death
Increased postoperative VIS was independently associated with the risk of postoperative death within 1-year (adjusted OR 1.06; 95% CI 1.03–1.10; p value < 0.001; Table 3). Increased postoperative lactate showed a significant association with the risk of postoperative 1-year death in univariable analysis (OR 1.37; 95% CI 1.14–1.66; p = 0.001) but failed to be included in the final multivariable model. PNI was significantly associated with postoperative 1-year death in both the univariate and multivariate analyses (OR 0.86; 95% CI 0.80–0.93; p value < 0.001, and adjusted OR 0.92; 95% CI 0.84–0.99; p value = 0.041, respectively). The preoperative need for dialysis was also associated with the risk of 1-year death (adjusted OR 4.95; 95% CI 1.22–20.16; p = 0.026; Table 3). In the further analysis using ROC analysis to observe the predictability of VIS on death, the cut-off value of VIS for increased risk of 1-year death was 10.5, with an AUC of 0.82 [95% CI 0.72–0.92; p < 0.001; Supplementary Fig. S2). The results of subgroup analysis were consistent with the results of entire population except for the insignificance of the association between PNI and 1-year death in low-risk patient group, but it might be explained by the increase of type II error induced by decreased number of deaths following subgroup stratification (Supplementary Table S1B).
Table 3.
Logistic regression model for 1-year death.
| Death (n = 16) | No death (n = 1875) | Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|---|---|
| Unadjusted OR (95% CI) | p value | Adjusted OR (95% CI) | p value | |||
| VIS | 19.0 (8.0–35.0) | 5.0 (0–9.8) | 1.08 (1.05–1.11) | < 0.001 | 1.07 (1.04–1.10) | < 0.001 |
| Lactate | 2.60 (1.00–4.15) | 1.60 (0.90–2.50) | 1.37 (1.14–1.66) | 0.001 | ||
| PNI | 46.9 (40.75–52.85) | 52.24 (48.66–55.59) | 0.86 (0.80–0.93) | < 0.001 | 0.92 (0.84–0.99) | 0.041 |
| Demographics | ||||||
| Male sex | 13 (81.3) | 1659 (77.8) | 0.81 (0.23–2.85) | 0.74 | ||
| Age (years) | 72.50 (62.0–77.5) | 64.0 (56.0–71.0) | 1.09 (1.02–1.16) | 0.007 | ||
| Smoking | 3 (18.8) | 566 (26.5) | 0.64 (0.18–2.25) | 0.486 | ||
| BMI | 24.4 (22.5–25.6) | 24.6 (22.8–26.6) | 0.90 (0.76–1.07) | 0.223 | ||
| Comorbidities | ||||||
| Hypertension | 13 (81.3) | 1692 (79.3) | 1.13 (0.32–3.98) | 0.85 | ||
| Diabetes | 6 (37.5) | 956(44.8) | 0.74 (0.27–2.04) | 0.559 | ||
| History of previous MI | 1 (6.3) | 186 (8.7) | 0.70 (0.09–5.31) | 0.728 | ||
| Acute MI | 5 (31.3) | 244 (11.4) | 3.52 (1.21–10.2) | 0.021 | ||
| Previous PCI | 0 (0) | 378 (17.7) | 0.14 (0.00–1.04) | 0.173 | ||
| Previous CABG | 0 (0) | 9 (0.4) | 6.78 (0.05–57.5) | 0.217 | ||
| PAOD | 2 (12.5) | 112 (5.3) | 2.58 (0.58–11.5) | 0.214 | ||
| COPD | 1 (6.3) | 27 (1.3) | 5.20 (0.66–40.8) | 0.117 | ||
| History of stroke | 4 (25) | 268 (12.6) | 2.32 (0.74–7.24) | 0.148 | ||
| Chronic kidney disease | 5 (31.3) | 117 (5.5) | 7.83 (2.68–22.9) | < 0.001 | ||
| Dialysis | 4 (25) | 57 (2.7) | 12.1 (3.80–38.8) | < 0.001 | 4.95 (1.22–20.16) | 0.026 |
| Cancer | 1 (6.3) | 69 (3.2) | 2.00 (0.26–15.31) | 0.507 | ||
| Heart failure EF | 0 (0) | 29 (1.4) | 2.16 (0.02–16.67) | 0.601 | ||
| Valve disease | 0 (0) | 10 (0.5) | 6.13 (0.05–51.30) | 0.239 | ||
| Aortic disease | 0 (0) | 15 (0.7) | 4.14 (0.03–33.26) | 0.345 | ||
| PTE DVT | 0 (0) | 2 (0.1) | 25.8 (0.19–334.16) | 0.089 | ||
| Infection (CRP pre) | 1.02 (0.33–3.26) | 0.14 (0.05–0.49) | 1.15 (1.05–1.25) | 0.002 | ||
| Haemoglobin | 13.1 (11.7–14.3) | 13.4 (12.1–14.5) | 0.68 (0.53–0.87) | 0.002 | ||
| Creatinine | 0.90 (0.75–1.13) | 0.92 (0.79–1.08) | 1.15 (1.05–1.25) | 0.002 | ||
| Medication | ||||||
| ACEi | 2 (12.5) | 139 (6.5) | 2.05 (0.46–9.11) | 0.345 | ||
| ARB | 4 (25) | 545 (25.6) | 0.97 (0.31–3.02) | 0.96 | ||
| Aspirin | 14 (87.5) | 1967 (92.2) | 0.59 (0.13–2.62) | 0.489 | ||
| BB | 4 (25) | 706 (33.1) | 0.67 (0.22–2.10) | 0.495 | ||
| CCB | 5 (31.3) | 663 (31.1) | 1.01 (0.35–2.91) | 0.989 | ||
| clopidogrel | 10 (62.5) | 1232 (57.8) | 1.22 (0.44–3.37) | 0.703 | ||
| Statin | 7 (43.8) | 1099 (51.5) | 0.73 (0.27–1.97) | 0.537 | ||
| Operation | ||||||
| Anastomosis number | 4 (2–8) | 4 (1–8) | 1.13 (0.58–2.18) | 0.725 | ||
| OP duration | 239.5 (208.5–302.0) | 260.0 (222.0–307.0) | 0.99 (0.99–1.01) | 0.633 | ||
| RBC transfusion | 4.0 (3.0–4.0) | 2.0 (1.0–3.0) | 1.49 (1.19–1.87) | 0.001 | ||
VIS vasoactive inotropic score, PNI prognostic nutritional index, BMI body mass index, MI myocardial infarction, PCI percutaneous cardiac intervention, CABG coronary artery bypass graft, PAOD peripheral arterial occlusive disease, COPD chronic obstructive pulmonary disease, PTE pulmonary thromboembolism, DVT deep vein thrombosis, ACEi angiotensin-converting enzyme inhibitors, ARB angiotensin receptor blocker, BB beta-blocker, CCB calcium channel blocker, OP operation, RBC red blood cell.
Causal relationship of VIS, lactate, and PNI on outcomes
Figure 2 shows the path analysis using postoperative lactate as a mediating variable. High VIS score showed significant effects on the occurrence of the composite outcome directly (p < 0.001) and indirectly though lactate (p < 0.001 from VIS to lactate and p < 0.001 from lactate to composite outcome; Fig. 2a). High VIS also showed a direct significant effect on 1-year death (p = 0.006) but did not show an indirect effect of lactate on 1-year death (Fig. 2b). In detail, higher VIS had a direct significant effect on the higher postoperative lactate levels (p < 0.001), but the effect of transiently elevated lactate on the occurrence of 1-year death was insignificant (p = 0.2; Fig. 2b).
Figure 2.
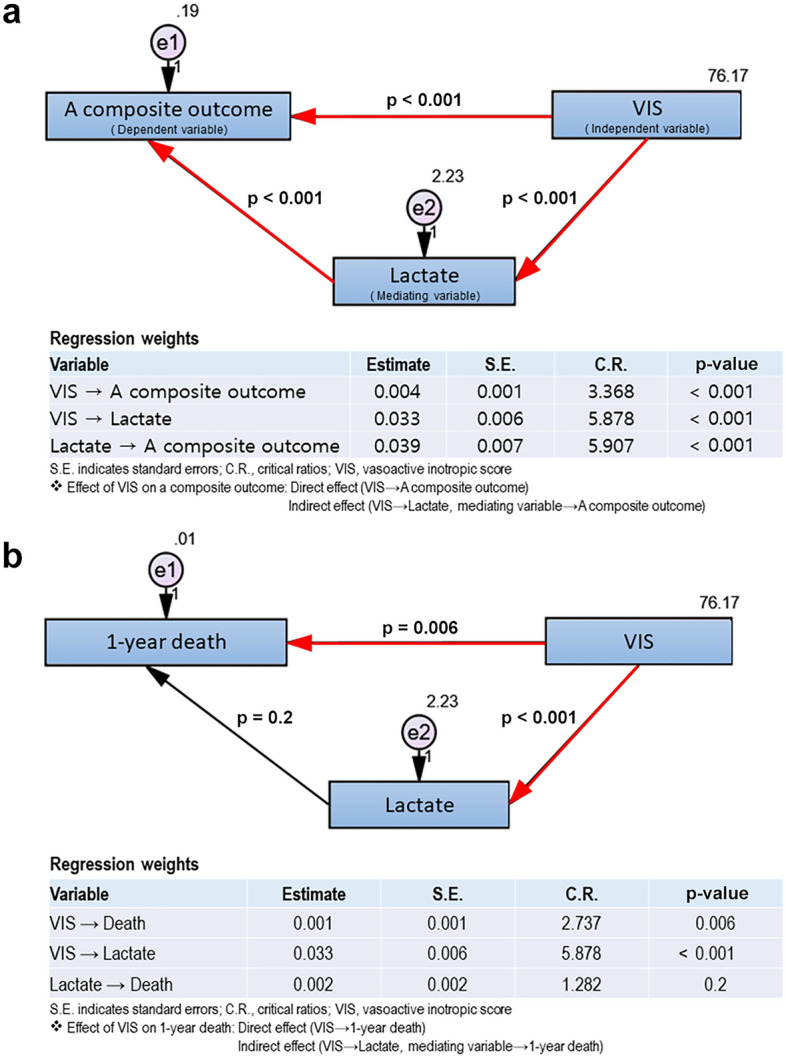
Path analysis using lactate as a mediating variable.
The PNI was added to the path analysis to determine other factors leading from complications to death (Fig. 3). The PNI showed a significant direct effect on a 1-year composite outcome (p < 0.001), and also had an indirect effect on the composite outcome when VIS was used as a mediating variable (p < 0.001 from PNI to VIS and p = 0.004 from VIS to a composite outcome; Fig. 3a). The PNI also had a significant direct effect on 1-year death (p = 0.025; Fig. 3b). In the pathway using VIS as a mediating variable, PNI also showed an indirect effect through VIS on 1-year death (p < 0.001 from PNI to VIS and p = 0.006 from VIS to death; Fig. 3b). There was a consistent trend toward lower PNI values in the death group, although statistical significance was achieved only in patients with a VIS value of ≥ 30 (44 vs. 50, p = 0.037).
Figure 3.
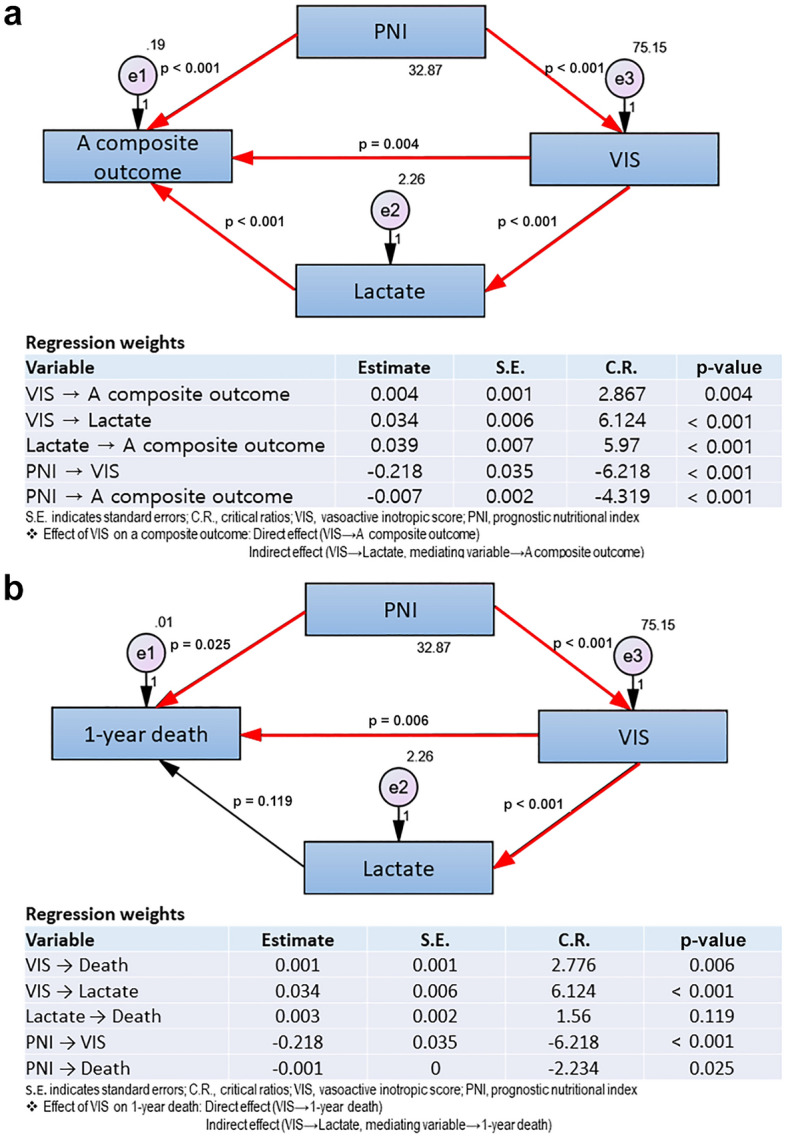
Path analysis using PNI and lactate as a mediating variable.
Discussion
In this retrospective analysis, we found that increased VIS during the immediate postoperative 48 h following OPCAB was significantly associated with long-term morbidity and mortality up to 1-year. In the pathway analysis, increased blood lactate during the immediate postoperative period reflecting temporary impaired tissue perfusion following high VIS may explain the occurrence of various postoperative complications, but not the occurrence of death. However, low PNI was a consistent independent risk factor for both postoperative 1-year death and composite outcome. The pathway using PNI showed that a poor preoperative nutritional state might increase perioperative inotropic requirements, which is consequently related to an increased risk of death and a composite of various complications.
Perioperative VIS is known to be a good predictor of poor clinical outcomes after cardiac surgery in various patient populations1–4. In the present study, consistent with previous studies, increased requirement of vasoactive and inotropic agents during the immediate postoperative 48 h following OPCAB also increased the risk of postoperative complications, including death up to 1 year. Moreover, this increased VIS showed a dose–response relationship with postoperative lactate levels. The increase in postoperative lactate may reflect a temporary decrease in systemic organ perfusion, which may be attributed to the prolonged use of various vasopressors, potent inotropic agent support, or the severity of the patient’s illness.
Blood lactate concentration has been widely used as a marker of altered tissue perfusion in critically ill patients14. Indeed, previous studies have reported that even minor increases in lactate concentrations are associated with morbidity and higher mortality rates15. In the current study, high postoperative blood lactate levels showed a significant association with composite occurrence of various postoperative complications. However, interestingly, the increased blood lactate level was neither a significant risk factor nor could explain the incidence of 1-year death following high VIS. Although blood lactate is a significant predictor of short-term mortality in critically ill patients6,16–18, a more complex process may be involved in the occurrence of longer-term mortality after OPCAB, and other important factors may also play a crucial role in long-term death.
Because poor preoperative PNI was consistently associated with the occurrence of both postoperative complications and death in multivariable models, we performed further pathway analysis, including PNI as a mediating variable. Low PNI, which refers to the decreased response of albumin and lymphocytes to acute disease, indicates a low immune-nutritional status, which consequently induces depression in cellular immunity associated with surgical events19. The increase in postoperative morbidity and mortality following poor PNI can be explained by the functional alterations in the immune system associated with surgical stress and is related to a shift in the physiological homeostatic balance between pro-inflammatory and regulatory cytokines19,20. Because poor nutritional status has many clinical consequences, including decreased quality of life, impaired ambulation, reduced treatment response, and increased treatment-related toxicity, previous studies have reported the significance of PNI on long-term outcomes, including death21–23. Accordingly, PNI has been widely used to assess the prognosis of various surgical groups24–27. Some studies have suggested that PNI should be considered as a clinical element and indicator of disease severity in patients undergoing CABG21. Recently, several studies reported that lower PNI levels were significantly associated with higher mortality and morbidity in patients with cardiovascular disease, including coronary artery disease, undergoing CABG21,28. In the current study, PNI was also an independent risk factor for morbidity and mortality.
The current study has strength in that the study population was more homogenous than that included in previous studies29, given that all patients underwent off-pump CABG and the surgery was performed by three experienced surgeons. Because patients with coronary artery disease who undergo CABG have a distinct nature compared to other patients with structural heart disease, there is a need to test the predictability of VIS in this population. Given that developing a validated evaluation tool to predict an operative outcome that could be applied evenly to patients in the high-risk vs. low-risk groups is demanding, the results of the present study is worthwhile. The result of subgroup analysis showed that VIS could be useful predictor for post-operative outcomes in both high-risk and low risk patients undergoing off-pump CABG. Insignificant result of the association between PNI and 1-year death might be explained by increase of type II error induced by decreased number of deaths following subgroup stratification. Furthermore, we applied SEM, which has an advantage in that it can solve the problem of multicollinearity, referring to a situation in which two or more explanatory variables in a multiple regression model are highly linearly related10. The multivariable regression model showed that VIS, lactate, and PNI were related to the outcome; however, these explanatory variables were correlated. Moreover, the simple regression model used in previous studies cannot explain the cause-and-effect relationship among these variables but can only show a simple relationship.
This study had several limitations. First, as a retrospective study, we could not exclude the possibility of bias from unanalysed (unmeasured or unmeasurable) variables. Although we attempted to include currently known confounders, we cannot rule out the existence of other confounders. Second, this was a single-centre study that evaluated cardiac surgical patients undergoing OPCAB, and, as a result, our results may not be applicable to different surgical populations. Third, the VIS was calculated as the sum of the maximum dosing rate of all administered inotropes or vasopressors during the first 48 h; therefore, VIS cannot be used to evaluate the individual influence of each drug on the outcome.
In conclusion, in patients undergoing OPCAB, high postoperative VIS independently predicted long-term morbidity and mortality up to 1 year after surgery. Patients with increased lactate levels, reflecting temporary tissue hypoperfusion following high postoperative VIS, had an increased risk of postoperative complications, but they did not necessarily lead to longterm death. However, patients with poor preoperative nutritional status had an increased risk of unfavourable outcome occurrences, including death up to 1-year (Supplementary Fig. S3). Our results imply that preoperative nutritional support can change the postoperative outcome including death after OPCAB.
Supplementary Information
Author contributions
J.-H.K., S.Y.Y., J.J.M. designed and conceived the study, performed statistical analysis, drafted the manuscript; S.H., Y.J.B., J.P. conceived the study and participated in its design and coordination; S. K., H.W., W.K., acquired data, coordinated data collection, and helped to draft the manuscript; J.-H.L., H.S.C., participated to design the study and substantially revised the draft; All authors read and approved the final manuscript.
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Ji-Hye Kwon and Seung Yeon Yoo.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-022-16900-1.
References
- 1.Koponen T, et al. Vasoactive-inotropic score and the prediction of morbidity and mortality after cardiac surgery. Br. J. Anaesth. 2019;122:428–436. doi: 10.1016/j.bja.2018.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barge-Caballero E, et al. Evaluation of the preoperative vasoactive-inotropic score as a predictor of postoperative outcomes in patients undergoing heart transplantation. Int. J. Cardiol. 2015;185:192–194. doi: 10.1016/j.ijcard.2015.03.098. [DOI] [PubMed] [Google Scholar]
- 3.Gaies MG, et al. Vasoactive-inotropic score as a predictor of morbidity and mortality in infants after cardiopulmonary bypass. Pediatr. Crit. Care Med. 2010;11:234–238. doi: 10.1097/PCC.0b013e3181b806fc. [DOI] [PubMed] [Google Scholar]
- 4.Gaies MG, et al. Vasoactive-inotropic score is associated with outcome after infant cardiac surgery: An analysis from the Pediatric Cardiac Critical Care Consortium and Virtual PICU System Registries. Pediatr. Crit. Care Med. 2014;15:529–537. doi: 10.1097/PCC.0000000000000153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hollenberg SM. Vasoactive drugs in circulatory shock. Am. J. Respir. Crit Care Med. 2011;183:847–855. doi: 10.1164/rccm.201006-0972CI. [DOI] [PubMed] [Google Scholar]
- 6.Andersen LW. Lactate elevation during and after major cardiac surgery in adults: A review of etiology, prognostic value, and management. Anesth. Analg. 2017;125:743–752. doi: 10.1213/ane.0000000000001928. [DOI] [PubMed] [Google Scholar]
- 7.Hajjar LA, et al. High lactate levels are predictors of major complications after cardiac surgery. J. Thorac. Cardiovasc. Surg. 2013;146:455–460. doi: 10.1016/j.jtcvs.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 8.Kusu-Orkar TE, Kermali M, Oguamanam N, Bithas C, Harky A. Coronary artery bypass grafting: Factors affecting outcomes. J. Card. Surg. 2020;35:3503–3511. doi: 10.1111/jocs.15013. [DOI] [PubMed] [Google Scholar]
- 9.Wu C, et al. Risk score for predicting long-term mortality after coronary artery bypass graft surgery. Circulation. 2012;125:2423–2430. doi: 10.1161/CIRCULATIONAHA.111.055939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Beran TN, Violato C. Structural equation modeling in medical research: A primer. BMC Res. Notes. 2010;3:267. doi: 10.1186/1756-0500-3-267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thygesen K, et al. Fourth universal definition of myocardial infarction. J. Am. Coll. Cardiol. 2018;72:2231–2264. doi: 10.1016/j.jacc.2018.08.1038. [DOI] [PubMed] [Google Scholar]
- 12.Machado MN, Nakazone MA, Maia LN. Acute kidney injury based on KDIGO (Kidney Disease Improving Global Outcomes) criteria in patients with elevated baseline serum creatinine undergoing cardiac surgery. Rev. Bras. Cir. Cardiovasc. 2014;29:299–307. doi: 10.5935/1678-9741.20140049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bozkurt B, et al. Universal definition and classification of heart failure: A report of the Heart Failure Society of America, Heart Failure Association of the European Society of Cardiology, Japanese Heart Failure Society and Writing Committee of the Universal Definition of Heart Failure. J. Card. Fail. 2021 doi: 10.1016/j.cardfail.2021.01.022. [DOI] [PubMed] [Google Scholar]
- 14.Weil MH, Afifi AA. Experimental and clinical studies on lactate and pyruvate as indicators of the severity of acute circulatory failure (shock) Circulation. 1970;41:989–1001. doi: 10.1161/01.cir.41.6.989. [DOI] [PubMed] [Google Scholar]
- 15.Singer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;315:801–810. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Demers P, Elkouri S, Martineau R, Couturier A, Cartier R. Outcome with high blood lactate levels during cardiopulmonary bypass in adult cardiac operation. Ann. Thorac. Surg. 2000;70:2082–2086. doi: 10.1016/s0003-4975(00)02160-3. [DOI] [PubMed] [Google Scholar]
- 17.Maillet JM, et al. Frequency, risk factors, and outcome of hyperlactatemia after cardiac surgery. Chest. 2003;123:1361–1366. doi: 10.1378/chest.123.5.1361. [DOI] [PubMed] [Google Scholar]
- 18.Badreldin AM, et al. Mortality prediction after cardiac surgery: Blood lactate is indispensible. Thorac. Cardiovasc. Surg. 2013;61:708–717. doi: 10.1055/s-0032-1324796. [DOI] [PubMed] [Google Scholar]
- 19.Ni Choileain N, Redmond HP. Cell response to surgery. Arch. Surg. 2006;141:1132–1140. doi: 10.1001/archsurg.141.11.1132. [DOI] [PubMed] [Google Scholar]
- 20.Strohmeyer JC, et al. Standardized immune monitoring for the prediction of infections after cardiopulmonary bypass surgery in risk patients. Cytom. Part B Clin Cytom. 2003;53:54–62. doi: 10.1002/cyto.b.10031. [DOI] [PubMed] [Google Scholar]
- 21.Keskin M, et al. Effect of nutritional status on mortality in patients undergoing coronary artery bypass grafting. Nutrition. 2018;48:82–86. doi: 10.1016/j.nut.2017.10.024. [DOI] [PubMed] [Google Scholar]
- 22.Cheng YL, et al. Prognostic nutritional index and the risk of mortality in patients with acute heart failure. J. Am. Heart Assoc. 2017;6:e004876. doi: 10.1161/JAHA.116.004876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen QJ, et al. Prognostic nutritional index predicts clinical outcome in patients with acute ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Sci. Rep. 2017;7:3285. doi: 10.1038/s41598-017-03364-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dolapoglu A, Avci E, Kiris T, Bugra O. The predictive value of the prognostic nutritional index for postoperative acute kidney injury in patients undergoing on-pump coronary bypass surgery. J. Cardiothorac. Surg. 2019;14:74. doi: 10.1186/s13019-019-0898-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee EH, et al. Preoperative hypoalbuminemia is a major risk factor for acute kidney injury following off-pump coronary artery bypass surgery. Intensive Care Med. 2012;38:1478–1486. doi: 10.1007/s00134-012-2599-8. [DOI] [PubMed] [Google Scholar]
- 26.Gibbs J, et al. Preoperative serum albumin level as a predictor of operative mortality and morbidity: Results from the National VA Surgical Risk Study. Arch. Surg. 1999;134:36–42. doi: 10.1001/archsurg.134.1.36. [DOI] [PubMed] [Google Scholar]
- 27.Park S, et al. The prognostic nutritional index and postoperative complications after curative lung cancer resection: A retrospective cohort study. J. Thorac. Cardiovasc. Surg. 2020;160:276–285 e271. doi: 10.1016/j.jtcvs.2019.10.105. [DOI] [PubMed] [Google Scholar]
- 28.Basta G, et al. The prognostic impact of objective nutritional indices in elderly patients with ST-elevation myocardial infarction undergoing primary coronary intervention. Int. J. Cardiol. 2016;221:987–992. doi: 10.1016/j.ijcard.2016.07.039. [DOI] [PubMed] [Google Scholar]
- 29.Matkovic M, et al. Long term outcomes of the off-pump and on-pump coronary artery bypass grafting in a high-volume center. Sci. Rep. 2019;9:8567. doi: 10.1038/s41598-019-45093-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.