Abstract
Cyanobacteria are prominent constituents of the marine biosphere that account for a significant percentage of oceanic primary productivity. In an effort to resolve how open-ocean cyanobacteria persist in regions where the Fe concentration is thought to be limiting their productivity, we performed a number of Fe stress experiments on axenic cultures of marine Synechococcus spp., Crocosphaera sp., and Trichodesmium sp. Through this work, we determined that all of these marine cyanobacteria mount adaptive responses to Fe stress, which resulted in the induction and/or repression of several proteins. We have identified one of the Fe stress-induced proteins as an IdiA homologue. Genomic observations and laboratory data presented herein from open-ocean Synechococcus spp. are consistent with IdiA having a role in cellular Fe scavenging. Our data indicate that IdiA may make an excellent marker for Fe stress in open-ocean cyanobacterial field populations. By determining how these microorganisms respond to Fe stress, we will gain insight into how and when this important trace element can limit their growth in situ. This knowledge will greatly increase our understanding of how marine Fe cycling impacts oceanic processes, such as carbon and nitrogen fixation.
Primary producers play key roles in both oceanic food chain dynamics and marine biogeochemistry. The factors that control their growth directly impact oceanic processes, such as the marine carbon and nitrogen cycles. Cyanobacteria, including species of Synechococcus, Prochlorococcus, Trichodesmium, and Crocosphaera, are prominent constituents of the marine biosphere that account for a significant percentage of oceanic primary productivity (10, 35, 36, 42, 48, 57). Additionally, in warm waters nonheterocystous, diazotrophic cyanobacteria (i.e., Trichodesmium and Crocosphaera) are vital components of the global nitrogen cycle through the production of “new” nitrogen (10, 57).
Recent work has indicated that primary production in the equatorial Pacific Ocean is limited by Fe bioavailability (12, 28), thereby implying that Fe may be an important limiting factor for open-ocean cyanobacterial growth in other regions of the ocean. Determining how these microorganisms respond to Fe stress and how they acquire Fe from the environment will provide insights into the factors that limit their growth in situ and greatly increase our understanding of how marine Fe cycling impacts oceanic processes.
With the exception of some strains of Lactobacillus (4) and Borrelia burgdorferi (37), iron is an essential trace element for all bacteria. The exceedingly low solubility (10−18 M) of ferric iron (Fe3+), the aerobically dominant form of iron at neutral pH, has led to the evolution of molecular systems specifically designed to acquire Fe from the environment in many bacteria (9). Under conditions of Fe stress, these bacteria secrete high-affinity binding ligands (L) or siderophores that specifically bind and solubilize Fe.
Because these Fe-L complexes are frequently quite large (>600 Da), passive diffusion through the outer membrane in gram-negative bacteria is very slow or nonexistent. To circumvent this diffusional barrier, specific outer membrane receptor proteins have evolved to aid in the transport of Fe-L complexes across the outer membrane. However, since the transport across the outer membrane is an energy-dependent step, three proteins (TonB, ExbB, and ExbD) are required to transfer the energy from the inner membrane to allow transport of the Fe-L complex into the periplasm (9, 26). Once in the periplasm, the Fe is transferred to a periplasmic iron-binding protein that delivers it to an inner membrane channel from where, via an ATP-dependent process, the Fe in shuttled into the cytoplasm.
Once inside the cell, Fe homeostasis is regulated at the transcriptional level by the ferric uptake regulator (Fur) in both gram-negative and some gram-positive bacteria (14). Fur is a DNA-binding protein that acts mostly as a repressor (14) and sometimes as an activator (13) of Fe homeostasis genes in response to available Fe2+ in the cell. The Fur regulon from Escherichia coli is complex, including genes encoding Fe transport machinery, siderophore biosynthesis, central metabolism regulators, and oxidative/acid stress response proteins (14).
Several researchers have examined the effects of Fe deprivation on freshwater and coastal cyanobacteria, including several Synechococcus spp. and Synechocystis sp. strain PCC6803 (17, 22, 23, 27, 30–32, 34, 38, 45, 51–56). This work has defined many Fe stress-induced phenotypic changes, including reduced growth rate and pigmentation, induction of Fe stress proteins, and in some cases production of Fe binding ligands (siderophores). In freshwater Synechococcus sp. strains PCC6301 and PCC7942, a number of Fe-regulated genes have been identified, including isiA, isiB (flavodoxin), idiA, fur, irpA, and mapA (17, 27, 32, 38, 51). Four of the above proteins have proposed biochemical functions in iron homeostasis inside the cell (IsiA, IsiB, Fur, and IdiA), while the roles of IrpA and MapA are less clearly defined (38, 51). Recently, 15 genes encoding the putative components of the Fe scavenging system in Synechocystis sp. strain PCC6803 were systematically inactivated (23). This work determined that four genes, futA1, futA2, futB, and futC, predicted to encode a periplasmic binding protein-dependent ABC transporter, were required for efficient Fe3+ transport into the cell. The FutA1/A2 proteins are homologous to the iron deficiency-induced protein A (IdiA) from freshwater Synechococcus sp. strain PCC6301.
The IdiA protein has been studied in detail in freshwater Synechococcus sp. strain PCC6301, where it is thought to be involved in acquiring Mn atoms for photosystem II (PSII) during Fe or Mn stress (30–32). This model is based on the observations that IdiA was: induced in response to Fe or Mn stress, associated with the thylakoid membranes, and required for optimal PSII activity under Fe or Mn stress. Furthermore, the expression of IdiA in PCC6301 has been shown to be dependent on two proteins, DpsA and IdiB, in addition to the predicted requirement for Fur (31).
Several IdiA homologues have been characterized, including SfuA from Serratia marcescens, FbpA from Neisseria spp., HitA from Haemophilus influenzae, and FutA1/FutA2 from Synechocystis sp. strain PCC6803 (1–3, 16, 22, 44). These proteins are the Fe binding protein components of an unusual type of Fe transport system that does not appear to have a dedicated outer membrane Fe-L receptor protein. Detailed analyses of the Fe donor for the periplasmic SfuA protein have determined that it can accept Fe solubilized by citrate, sodium pyrophosphate, and oxaloacetate (58). Similar experiments with HitA have shown that it can acquire Fe from the chemical iron chelator 2,2′-dipyridyl when the Fe:L complex diffuses into the periplasmic space, which is independent of its role in acquiring Fe from transferrin (1).
FbpA has similar characteristics to HitA and SfuA, except it appears to be expressed on the cell surface in addition to the periplasmic space (16). Although the exact cellular locations of FutA1 and FutA2 in Synechocystis sp. strain PCC6803 are unknown, they are predicted to reside in the periplasmic space due to their requirement for efficient Fe3+ transport into the cell (23).
A complete high-affinity Fe scavenging system (i.e., outer membrane ferric siderophore receptor protein, periplasmic iron binding protein, inner membrane channel, and ATPase) has yet to be characterized or demonstrated in any marine cyanobacterium. However, as discussed previously, siderophore production has been investigated extensively in freshwater, coastal, and some marine cyanobacteria (18, 24, 25, 52). One siderophore, schizokinen, has been purified from Fe-stressed cultures of Anabaena sp. strain PCC7102 (18, 25).
Interestingly, many freshwater cyanobacteria make detectable siderophores, while there are no reports of siderophores in open-ocean Synechococcus spp. or Trichodesmium, even though nitrogen-fixing Trichodesmium cells assayed in the field have very high cellular Fe requirements (47). Additionally, it is well documented that Synechococcus spp. and Prochlorococcus spp. are ubiquitous in the ocean even in regions where the Fe concentrations are very low and suspected to be limiting (6). These observations would argue for the existence of some type of high-affinity Fe scavenging system in these oligotrophic open-ocean cyanobacteria.
In an effort to resolve how open-ocean cyanobacteria persist in regions where the Fe concentration is thought to be limiting, we have performed Fe stress experiments on axenic cultures of marine Synechococcus, Crocosphaera, and Trichodesmium. We determined that these marine cyanobacteria mount adaptive responses to Fe stress, which ultimately result in the expression of significant amounts of the IdiA protein. Genomic observations and data presented here from experiments on open-ocean Synechococcus are consistent with the IdiA protein's being required for cellular Fe scavenging.
MATERIALS AND METHODS
Bacterial strains.
The three strains of open-ocean Synechococcus used in this study were WH7803 (nonmotile, phycoerythrins with low phycourobilin [PUB] content), WH8102, and WH8103 (both motile, phycoerythrins with high PUB content). The two diazotrophic strains studied were Crocosphaera sp. strain WH8501 and Trichodesmium erythraeum IMS101. All cultures except WH8102 were axenic as verified by purity medium and direct microscopic observation, as described (49). All strains were maintained by the lab of John B. Waterbury at the Woods Hole Oceanographic Institution and isolated as described (33, 48, 49). The Crocosphaera strain was previously designated Synechocystis sp. strain WH8501 (42, 48, 57).
Culture conditions.
Cultures were maintained using described methods (49) unless otherwise noted.
Synechococcus and Crocosphaera spp.
Stock cultures of Synechococcus spp., strains WH7803, WH8102, and WH8103, were maintained in SN medium with nitrate as the source of combined nitrogen in constant light (10 μEin/m2/s) at 23°C. The Crocosphaera sp. strain was maintained in SO (SN medium without fixed nitrogen source) on a 14-h light (30 μEin/m2/s)–10-h dark cycle at 27.5°C. The seawater base of the SN medium was derived from filtered Vineyard Sound water (Woods Hole, Mass.).
Trichodesmium sp.
Trichodesmium medium was made of 3/4 Sargasso seawater filtered successively through 1.0- and 0.2-μm Millipore membrane filters then Tyndalized by heating in a microwave oven to boiling in Teflon containers. Sargasso seawater is diluted with steam-sterilized Millipore Q-water (Millipore, Bedford, Mass.). All chemical additions to the medium are tissue culture tested and purchased from Sigma. Stocks (1,000×) are sterilized by either filtration or steam and added to 3/4 strength seawater base. The additions include 1.5 × 10−6 M EDTA, 8 × 10−6 M phosphoric acid, 5 × 10−8 M Fe (ferric citrate), 10−7 M MnSO4, 10−8 M ZnCl2, 10−8 M NaMoO4, 10−10 M CoCl2, 10−10 M NiCl2, 10−10 M NaSeO3, and 1.5 μg of vitamin B12/liter. All Trichodesmium cultures are grown in Nalgene polycarbonate flasks or Nalgene polycarbonate 1.5-liter culture chambers equipped with internally suspended magnetic stir bars, washed successively with Micro (International Products Corporation Burlington, N.J.) and 0.5 N HCl. Growth conditions vary but typically include a 14 h–10 h light-dark cycle using cool white or warm, white, deluxe fluorescent lamps at 30 to 300 μEin m−2 s−1 and temperatures ranging from 24 to 28°C. Small-volume cultures are not stirred but are shaken gently daily.
Nutrient stress experiments. (i) Synechococcus and Crocosphaera spp.
Induction of nutrient stress in cultures of Synechococcus and Crocosphaera used a variation of the protocol developed earlier (32). All nutrient stress experiments were done with at least two or more replicates. Cells from a late-log-phase culture were used as an inoculum (>1/50 dilution) for parallel cultures where the Fe was either supplemented to 20 μM or omitted from the trace metal mix. To restrict carryover Fe from the inoculum, the EDTA concentration was held at 15 μM in both conditions.
For Synechococcus experiments, the cells were inoculated in either SN or ASW medium (49) and incubated in constant light (≈30 μEin/m2/s) at 23°C for 6 to 10 days. Crocosphaera was inoculated in SO medium and incubated in the same conditions as the stock culture for 6 to 10 days. Fe stress was evident through reduced biomass and pigmentation in the Fe-omitted culture relative to the Fe-replete control. In some experiments, aliquots of cells were counted directly by light microscopy using epifluorescence (49). Nitrogen-and phosphorus-stressed cultures were obtained using the same techniques described above for Fe.
(ii) Trichodesmium sp.
Trichodesmium sp. strain IMS101 cultures (30 to 100 ml) grown as described above were gently filtered down onto 5-μm polycarbonate filters (Osmonics, Kent, Wash.). The cells were then washed twice with 50 to 100 ml of medium without Fe and resuspended in medium with or without Fe. Fe stress was visually assessed by reduced biomass and pigmentation relative to an Fe-replete control.
Genomic analyses.
Detailed analyses of the cyanobacterial genomes were performed using the ERGO interface on the Integrated Genomics (Chicago, Ill.) website (http://wit.integratedgenomics.com/IGwit/). Preliminary sequence data from Synechococcus sp. strain WH8102, Prochlorococcus sp. strains MED4 and MIT9313, and Nostoc punctiforme were obtained from the Department of Energy (DOE) Joint Genome Institute (JGI) at http://www.jgi.doe.gov/tempweb/JGI_microbial/html/index.html. If homologues to IdiA, DpsA, IdiB, TonB, ExbB, and ExbD were not identified using BLASTP when analyzing data from unclosed genomes, DNA sequences of the gene in question from closely related organisms were used to verify that the gene was absent. The BLOSUM62 scoring matrix was used during BLASTP analysis to generate the percentage of identical and positive residues in the IdiA homologues (20). All homologues discussed had significant similarity over the whole gene (>82%).
Generation of bulk protein extracts. (i) Synechococcus spp.
To generate crude protein extracts, the cells were isolated by centrifugation at 15,000 × g for 5 min, the supernatant was completely removed, and the pellet was resuspended in protein sample buffer (5) and heated at 95°C for 5 min. Protein extracts were separated via sodium dodecyl sulfate–12% polyacrylamide gel electrophoresis (SDS-PAGE) in Tris-glycine buffer using the Miniprotean3 and the manufacturer's instructions (Bio-Rad, Hercules, Calif.). Once visualized via Coomassie staining, protein concentrations in extracts were normalized using a Chem Imager 4000 (Alpha Innotech Corp. San Leandro, Calif.) densitometry scanning of invariant protein bands or direct quantitation with the DC protein quantitation kit (Bio-Rad, Hercules, Calif.).
(ii) Crocosphaera sp.
Cells were isolated as described for Synechococcus spp., washed in 1 ml of 50 mM Tris-HCl, pH 8.0, and resuspended in the same buffer, and crude protein extracts were generated by bead beating in a mini-bead beater (Biospec Products, Bartlesville, Okla.) with 0.5-μm glass beads for 250 s. The extracts were mixed with sample buffer, proteins were visualized, and their concentrations were normalized as described above.
(iii) Trichodesmium sp
Crude protein extracts from the Trichodesmium sp. strain were generated by filtering the cells to dryness on 5-μm polycarbonate filters, resuspending the cells in sample buffer, and heating the solution at 95°C for 5 min. Proteins in the extracts were visualized and concentrations were normalized as described above.
IdiA detection.
IdiA was detected using polyclonal antiserum raised against the IdiA protein from freshwater Synechococcus sp. strain PCC6301 (generously provided by E. Pistorius). Briefly, equivalent amounts of protein were separated as discussed and electrophoretically transferred to nitrocellulose, and antibody binding was visualized with the amplified alkaline phosphatase goat anti-rabbit immunoblot assay kit (Bio-Rad, Hercules, Calif.).
The following protocol was used to detect IdiA with a 1:4,000 dilution of IdiA antiserum and preimmune serum. The blots were blocked in 5% nonfat milk for 90 min, probed with primary antibody for 120 min, probed with secondary antibody for 90 min, incubated with the streptavidin-biotinylated alkaline phosphatase complex for 60 min, and color developed for 30 min. All antisera were prepared in 5% nonfat milk. Specificity of the IdiA protein detected was confirmed by probing the same extracts with preimmune serum.
Membrane preparations. (i) Soluble and insoluble protein separation.
Fe-stressed and replete cultures (300 ml) of Synechococcus sp. strain WH7803 were grown as described above. The cells were harvested by centrifugation at 10,000 × g for 10 min and washed in 50 mM Tris-HCl, pH 8.0. Cell pellets were resuspended in the same buffer (1 ml and 3 ml for Fe-stressed and replete, respectively), and protein extracts were generated by two French press treatments (≈20,000 lb/in2). Whole cells were removed from 0.5 ml of the extract by centrifugation at 10,000 × g for 10 min, and soluble proteins were separated from insoluble material by ultracentrifugation in an L8-M ultracentrifuge (Beckman, Fullerton, Calif.) at 110,000 × g for 90 min. The orange membrane-containing pellet was resuspended in 100 and 50 μl of TE (10 mM Tris-HCl, 1 mM EDTA, pH 8.0) for the Fe-replete and stressed samples, respectively. The resuspended membrane extracts and reddish supernatant fractions were mixed with sample buffer and subjected to 12% SDS–12% PAGE–Tris–glycine analysis. Proteins were visualized by Coomassie staining.
(ii) Outer membrane isolation.
Cultures of Synechococcus sp. strain WH7803 were subjected to the outer membrane stripping protocol (8, 40), with the following modifications. One liter of culture was grown in SN as described above to late log phase, two 500-ml aliquots of culture were harvested by centrifugation (10 min at 10,000 × g), the pellets were resuspended in 10 ml of SN-Fe and used as inoculum for two 1-liter cultures (SN+Fe and SN−Fe). Fe stress was apparent in the SN−Fe culture, due to reduced biomass and pigmentation after 7 days of growth at 30 μEin/m2/s at 23°C. The cells were isolated by centrifugation at 7,500 × g for 15 min at 20°C, washed in 15 ml of sterile SN, and resuspended in stripping buffer (50 mM Tris-HCl, pH 8.0, 50 mM disodium EDTA, 15% sucrose), and incubated on ice for 30 to 40 min The outer membrane-containing fraction was isolated via ultracentrifugation (110,000 × g for 90 min at 4°C), and the high-speed supernatant fraction was concentrated 14-fold using Micron YM-10 filtration columns (Millipore, Bedford, Mass.). The high-speed pellet was resuspended in 100 μl of TE. The absorbance spectra of both fractions were obtained using a Shimadzu UV160 spectrophotometer (Shimadzu Scientific Instruments, Columbia, Md.).
RESULTS
Genomic survey for putative Fe scavenging genes in open-ocean cyanobacteria.
Three proteins predicted to be involved in Fe scavenging have been identified, based on similarity, in the genome of Synechococcus sp. strain WH8102: an Fe binding protein (ERGO RSN04338; 45% identity to IdiA from freshwater Synechococcus sp. strain PCC6301), inner membrane channel (ERGO RSN04339; 35% identity to HitB from Haemophilus influenzae), and an ATPase (ERGO RSN03833; 42% identity with SfuC from S. marcescens). Similar homologues were identified in the genomes of Prochlorococcus sp. strains MED4 and MIT9313. Together these three proteins could compose a complete periplasmic binding protein-dependent ABC transporter for Fe.
As discussed, the Fe binding protein has significant similarity with both IdiA, a thylakoid membrane-associated Fe and Mn binding protein from freshwater Synechococcus sp. strain PCC6301 (30–32) and a family of periplasmic Fe binding proteins including SfuA of the SfuABC operon from S. marcescens (2, 3). In addition, both Prochlorococcus spp. and Synechococcus sp. strain WH8102 appear to be lacking any defined outer membrane receptor proteins for Fe-siderophore complexes, siderophore biosynthetic genes, and the proteins TonB, ExbB, and ExbD (F. Larimer, unpublished data). In contrast, searches of the freshwater cyanobacterial genomes (Anabaena PCC7120, Synechocystis sp. strain PCC6803, and Nostoc punctiforme) identified multiple copies of the global Fe homeostasis transcriptional regulator Fur, homologues to TonB, ExbB, and ExbD, and putative outer membrane Fe-siderophore receptor proteins.
IdiA homologues.
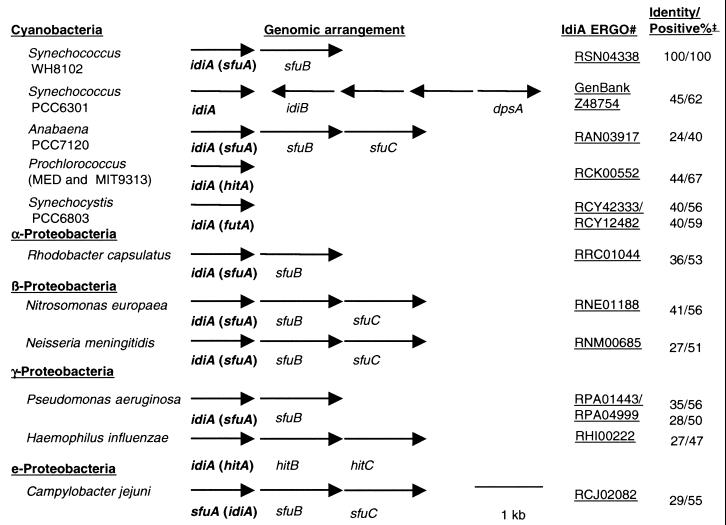
Although IdiA is not universally present, many bacteria from several divisions, including the cyanobacteria and α-, β-, γ-, and Ε-proteobacteria contain an IdiA homologue. Figure 1 compares both the genetic arrangement of the DNA immediately surrounding the idiA gene and the similarity of the IdiA homologues from several bacteria to IdiA from open-ocean Synechococcus sp. strain WH8102. Included are IdiA homologues from freshwater Synechococcus sp. strain PCC6301 and several organisms whose genomes are present in the ERGO interface on the Integrated Genomics website.
FIG. 1.
idiA gene from open-ocean Synechococcus sp. strain WH8102 compared to idiA homologues and surrounding genes in other bacteria and cyanobacteria whose genomes are present in the ERGO interface on the Integrated Genomics website. The GenBank accession number is shown for the region surrounding the idiA in Synechococcus sp. strain PCC6301; in all other cases an ERGO number is presented. Percent positive and identical bases was determined using the BLOSUM62 matrix.
Of the IdiA homologues shown, all were similar over >85% of the protein, with similarity ranging from 24 to 45% identical for Anabaena sp. strain PCC7120 and Synechococcus sp. strain PCC6301, respectively. The region of the protein with the most heterogeneity was the N-terminal 10 to 30 amino acid residues, which are predicted to encode a leader sequence for transport from the cytoplasm (15). The predicted IdiA transcript varies from tricistronic to monocistronic, with the ATPase encoded by the SfuC homologue most frequently encoded elsewhere in the genome.
In open-ocean Synechococcus sp. strain WH8102, the idiA gene is predicted to be transcribed with a gene encoding an inner membrane channel protein, an SfuB homologue, while in Prochlorococcus sp. strains MED4 and MIT9313, the idiA appears to be monocistronic (although the other components [SfuB and -C] are encoded elsewhere on the genome). Although the idiB and dpsA genes are encoded 3′ of the idiA gene in Synechococcus sp. strain PCC6301, this arrangement is an exception in the genomes currently available. The IdiB protein is currently only found in the genomes of freshwater cyanobacteria (Synechocystis spp., Anabaena sp. strain PCC7120, Synechococcus sp. strain PCC6301, and N. punctiforme). DpsA, since it contains a helix-turn-helix catabolite activator protein motif (29), is found by itself in several genomes with three notable exceptions, Prochlorococcus sp. strains MED4 and MIT9313 and Synechococcus sp. strain WH8102.
Demonstration of Fe stress in Synechococcus spp.
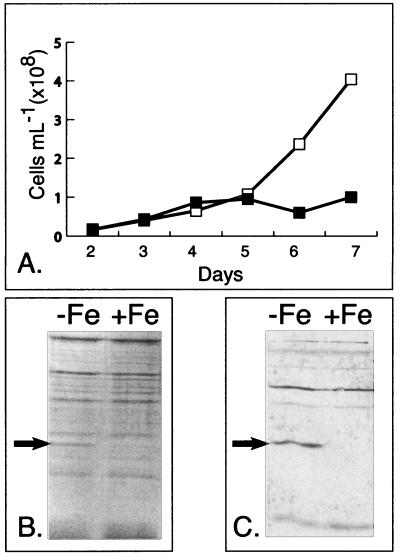
Since it had been shown previously that open-ocean Synechococcus sp. strain WH7803 mounted an adaptive response to Fe stress that results in the expression of an approximately 36-kDa protein (11), we sought to verify this result and determine if the induced protein was the IdiA protein described above. Axenic cultures of Synechococcus sp. strain WH7803 were grown under Fe-replete and Fe-omitted conditions, and growth was monitored by epifluorescence microscopy (Fig. 2A). Bulk protein extracts were generated immediately following cessation of growth in the Fe-omitted culture (day 7), separated by SDS-PAGE, and visualized by Coomassie staining. Figure 2B shows that an approximately 39-kDa protein was overexpressed in the Fe-omitted culture. As shown in the Western blot in Fig. 2C using the anti-IdiA polyclonal antiserum, the 39-kDa protein was identified as an IdiA homologue (representative preimmune serum for Synechococcus sp. strain WH7803 shown in blot in Fig. 2B and 4B). Although the amounts of IdiA induced in open-ocean Synechococcus sp. strain WH8102 and 8103 were barely detectable with Coomassie staining (data not shown), Western blots with WH8103 and WH8102 (not shown) confirmed its expression in similar experiments (see Fig. 4).
FIG. 2.
Fe stress response of Synechococcus sp. strain WH7803. (A) Representative growth curve of Synechococcus sp. strain WH7803 grown in Fe-replete (□) and Fe-omitted (▪) artificial seawater medium. (B) Coomassie staining of SDS-PAGE-separated protein samples, Fe omitted (−Fe) or replete conditions (+Fe), from day 7 of the growth curve. An arrow denotes the location of an abundant 39-kDa Fe stress-induced protein. (C) Representative Western blot detecting IdiA in marine Synechococcus sp. strain WH7803. An arrow denotes the IdiA-specific band.
FIG. 4.
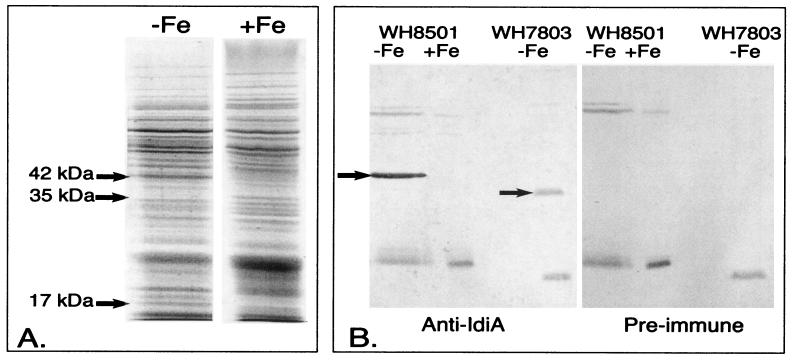
Proteins induced in response to Fe stress in Crocosphaera sp. strain WH8501. (A) Coomassie-stained SDS-PAGE from axenic Crocosphaera sp. strain WH8501grown in Fe-replete (+Fe) or Fe-omitted (−Fe) SN medium. Arrows denote three differentially expressed proteins. (B) Western blots from Fe-stressed and -replete Crocosphaera sp. strain WH8501 and Synechococcus WH7803 probed with polyclonal Synechococcus sp. strain PCC6301 anti-IdiA antiserum and preimmune serum. Arrows identify the IdiA proteins. Open-ocean Synechococcus sp. strain WH7803 is presented as a positive control for the blot.
Demonstration of Fe stress in the diazotrophs Trichodesmium sp. strain IMS101 and Crocosphaera sp. strain WH8501.
Since IdiA expression was conserved in the Synechococcus isolates tested, we sought to determine if the open-ocean, nonheterocystous, diazotrophic cyanobacteria Trichodesmium and Crocosphaera also expressed the IdiA protein during Fe stress. Parallel cultures (with and without Fe) were grown as described in Materials and Methods. Cultures lacking Fe visually displayed reduced biomass and pigmentation relative to the Fe-replete control. Immediately upon the cessation of growth in the Fe-omitted culture (which varied from 4 to 6 days depending on the inoculum source between the replicates), the cells were harvested, bulk protein extracts were separated by SDS-PAGE, and proteins were visualized by Coomassie staining.
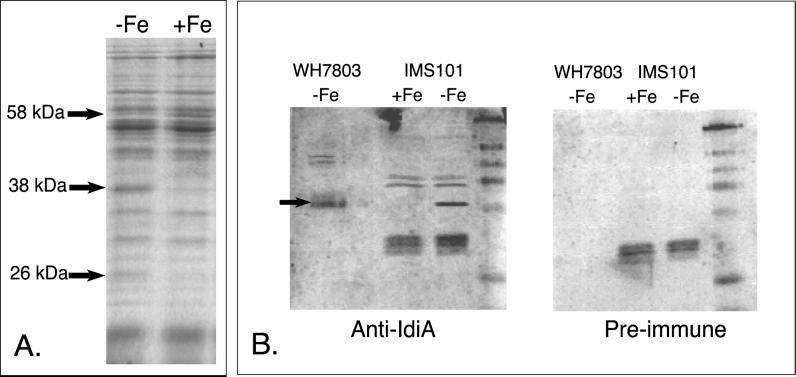
Comparison of the protein banding patterns from the Fe-deficient and -replete cultures shows that Trichodesmium sp. strain IMS101 differentially expresses at least three proteins (Fig. 3A). Specifically, a 58-kDa protein is repressed while 26- and 38-kDa proteins are induced. Using the polyclonal anti-IdiA antiserum, the 38-kDa protein was identified as an IdiA homologue (Fig. 3B). As for Trichodesmium extracts, Coomassie-stained one-dimensional SDS-PAGE gels from Crocosphaera extracts also showed three differentially expressed proteins. As shown in Fig. 4A, two proteins, 42 and 17 kDa, were induced in Fe-deficient conditions, while a 35-kDa protein was repressed. Western blotting with IdiA antiserum verified that the 42-kDa protein was an IdiA homologue (Fig. 4B).
FIG. 3.
Fe stress response of axenic Trichodesmium sp. strain IMS101. (A) Coomassie-stained SDS-PAGE gel from cultures grown under Fe-replete (+Fe) or Fe-omitted (−Fe) conditions. Arrows denote differentially expressed proteins and their approximate molecular masses. (B) Western blots detecting IdiA expression in Fe-stressed axenic Trichodesmium sp. strain IMS101 with polyclonal Synechococcus sp. strain PCC6301 anti-IdiA antiserum and preimmune serum. An arrow identifies the IdiA protein. Open-ocean Synechococcus sp. strain WH7803 is presented as a positive control for the blot.
Specificity of IdiA induction.
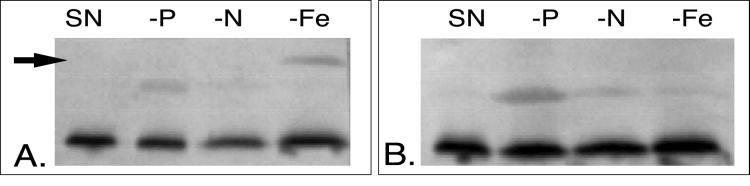
To determine if IdiA was induced specifically in response to Fe depletion, other nutrient omission experiments were performed. As shown in the Western blot in Fig. 5, although a growth lag was apparent in the nonreplete Synechococcus sp. strain WH8103 cultures (−N, −P, and −Fe), IdiA was only detected in extracts from the Fe-omitted culture. Although cross-reactivity was detected when probing with the preimmune serum, no cross-reactive bands were detected in the IdiA size range. Similar results were obtained with open-ocean Synechococcus sp. strain WH7803 grown in the four culture conditions described above (data not shown). Furthermore, IdiA was not detectable in Trichodesmium and Crocosphaera cultures when P was omitted from the medium (data not shown).
FIG. 5.
Western blot detecting IdiA expression in Synechococcus sp. strain WH8103 grown in SN medium formulated to cause conditions of phosphate (−P), nitrogen (−N), and iron (−Fe) stress. (A) A clear 36-kDa IdiA band, denoted by an arrow, is detected only in the Fe-omitted sample when probed with the IdiA antiserum. (B) Preimmune serum blot showing that no cross-reactive bands were detected in the 36-kDa range.
To assess the permanence of IdiA expression, Synechococcus sp. strain WH7803 was grown in changing Fe regimens from Fe depleted to replete while cellular expression of IdiA was monitored (Fig. 6). Throughout the experiment, aliquots of the culture were removed each day for epifluorescent cell enumeration (49). Once the growth in Fe-omitted culture was significantly lagging behind the replete culture (day 4), the total Fe concentration was supplemented up to 20 μM (standard SN Fe concentration). On select days (3, 4, and 7), protein extracts were generated and IdiA expression was probed as described in Materials and Methods. Since equal amounts of protein (11 μg) were loaded in each lane, relative expression of IdiA detected in the Western blot can be compared.
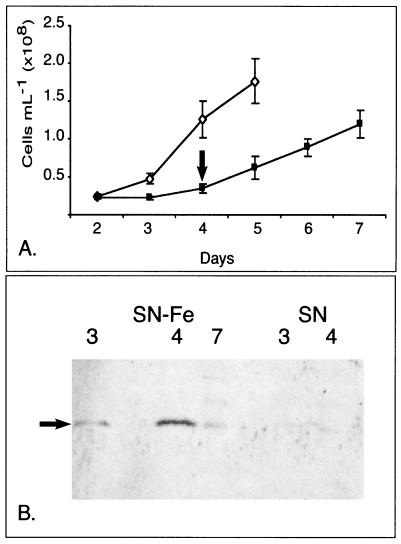
FIG. 6.
Assessment of the permanence of IdiA expression in Synechococcus sp. strain WH7803 upon switch from Fe-stressed to Fe-replete conditions in SN-based medium. (A) Growth curve comparing Fe-replete (⋄) and Fe-omitted (▪) growth. Error bars show standard deviation between the cell counts obtained from three culture replicates. Cells were counted directly by epifluorescence. On day 4 (arrow), the total Fe in the Fe-omitted culture was supplemented by the addition of Fe (20 μM). (B) Western blot detecting IdiA (denoted by the arrow) on select days throughout the growth curve. The initial conditions of the cultures were Fe replete (SN) and Fe omitted (SN−Fe).
Very little IdiA was detected in the logarithmically growing control culture (Fig. 6B, SN days 3 and 4). In contrast, by days 3 and 4 significant amounts of IdiA were detected in the Fe-stressed culture (Fig. 6B, SN−Fe days 3 and 4). Once the medium was supplemented with Fe and allowed to grow for 2 additional days (SN−Fe day 7), densitometry scanning determined that the amount of IdiA detected was reduced by 66% relative to the level on day 4.
Cellular location of IdiA.
To gain further insight into the role of IdiA in open-ocean cyanobacteria, membrane separations of Synechococcus sp. strain WH7803 were performed. Western blotting with high-speed-separated soluble and insoluble protein extracts determined that IdiA was detected exclusively in the membrane-associated fraction (Fig. 7A). Similar results were obtained with Synechococcus sp. strain WH8103 (data not shown).
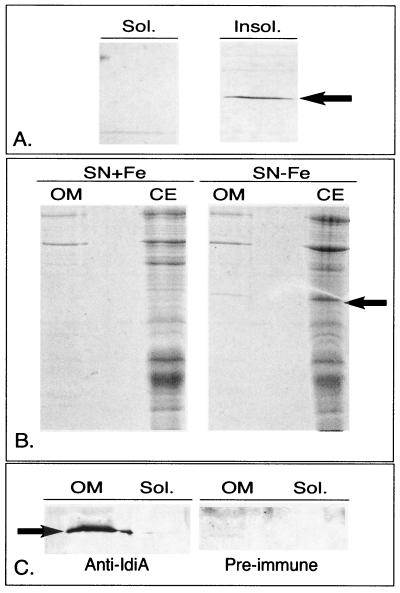
FIG. 7.
Localization of the IdiA protein from Synechococcus sp. strain WH7803. (A) Western blot probing high-speed soluble (sol.) and insoluble (insol.) proteins in Fe-stressed cultures of Synechococcus sp. strain WH7803. (B) Outer membrane (OM) fraction and cellular extracts (CE) from Fe-replete (SN+Fe) and -stressed (SN−Fe) cultures of Synechococcus sp. strain WH7803 stained with Coomassie. (C) Western blots from Fe-stressed Synechococcus sp. strain WH7803 outer membrane fraction probed with polyclonal Synechococcus sp. strain PCC6301 anti-IdiA antiserum and preimmune serum. In all panels, an arrow denotes the IdiA protein.
To refine the location of IdiA, the outer membrane of Synechococcus sp. strain WH7803 grown with and without Fe was isolated as described in Materials and Methods. Coomassie staining and Western blotting identified significant amounts of IdiA in the outer membrane fractions from Synechococcus sp. strain WH7803, while none could be detected in the soluble fraction (Fig. 7B and 7C). The absorbance spectrum obtained from the outer membrane extract was very similar to the spectrum published by Resch (40), detecting carotenoid peaks at approximately 435, 460, and 490 nm. Contamination from cellular lysis was minimal in the preparations, as evidenced by the inability to detect chlorophyll a and phycobiliproteins in the preparations.
DISCUSSION
Cyanobacteria are ubiquitous constituents of the ocean, where they significantly contribute to primary productivity. Despite their environmental importance and the belief that Fe limits their growth in many regimens (6, 12, 28), we know very little about how open-ocean cyanobacteria acquire Fe. Although siderophores have yet to be identified in axenic Fe-stressed cultures of Synechococcus sp. strain WH7803, it is clear that this cyanobacterium can use Fe bound by a number of structurally different siderophores (21), leading to the hypothesis that it is using a ligand exchange mechanism (46). Clear demonstration of the molecular mechanism of Fe acquisition employed by open-ocean cyanobacteria has yet to be established.
Genomic insights into Fe scavenging in open-ocean cyanobacteria.
Analyses of the cyanobacterial genomes currently available have identified some intriguing absences in predicted Fe-scavenging capabilities of open-ocean cyanobacteria. No known siderophore biosynthesis genes could be identified in the genomes of Synechococcus sp. strain WH8102 and Prochlorococcus sp. strains MED4 and MIT9313. Additionally, neither putative outer membrane receptor proteins for Fe siderophore complexes nor the proteins TonB, ExbB, and ExbD could be identified in these marine cyanobacterial genomes, even though these protein homologues are identifiable in the freshwater cyanobacterial genomes.
The absence of outer membrane receptor proteins in Synechococcus sp. strain WH8102 and Prochlorococcus spp. was not too surprising, since it has been demonstrated that these proteins can diverge significantly between organisms and still perform the same function (19). However, the absence of the proteins TonB, ExbB, and ExbD was more unexpected, since they are all required for Fe-siderophore transport across the outer membrane (reference 46 and references therein). It is possible that the genes encoding TonB, ExbB, and ExbD are present in the genomes but reside in the gaps not represented by the contigs in the unfinished genomes, or that the DNA sequences of tonB and exbBD have diverged significantly. However, it is also possible that open-ocean Synechococcus and Prochlorococcus spp. use a different mechanism of Fe-siderophore translocation through the outer membrane from the developed paradigms for gram-negative bacteria (9, 46).
IdiA homologues.
Many bacteria contain IdiA homologues. Based on the diverse niches of the organisms that have IdiA homologues (Fig. 1), there does not appear to be a clear correlation between their physiologies (i.e., autotroph or heterotroph) and the presence of IdiA. As demonstrated by our analyses of the IdiA protein in oceanic cyanobacteria, there is considerable variation in the size of the protein (≈5 kDa). This heterogeneity is not reserved to cyanobacteria, since it is also seen in the IdiA homologues listed in Fig. 1. Additionally, since several organisms that are known to make siderophores (i.e., Anabaena sp. strain PCC7120 and Pseudomonas aeruginosa) have an IdiA homologue (18, 25, 39), it is plausible that IdiA might be able to receive Fe solubilized by a number of siderophores.
Role of IdiA in open-ocean cyanobacteria.
The exact cellular location and role of the IdiA homologues vary from organism to organism. However, the majority are Fe stress-induced proteins, localized in the periplasmic space, where they make up the periplasmic Fe binding component of a binding protein-dependent ABC transporter (1–3, 7, 22). In other organisms, the Fe stress-induced IdiA homologue is localized in the outer membrane or on the cell surface (16). In contrast, IdiA from freshwater Synechococcus sp. strain PCC6301 is associated with the thylakoids Fe and Mn, stress induced, and thought to be involved in scavenging Mn atoms for PSII (30–32).
We have currently been unable to Mn stress Synechococcus or Crocosphaera spp. in Sargasso and artificial seawater-based media and therefore have not yet tested the Mn induction of IdiA in these organisms. However, we have recently been able to Mn limit Trichodesmium. In these experiments, IdiA was only significantly detected in cellular extracts from the Fe-omitted culture (unpublished data). Additionally, unlike what has been shown in freshwater Synechococcus sp. strain PCC6301, we have determined that significant amounts of IdiA are associated with outer membrane preparations from marine Synechococcus sp. strain WH7803.
Although the absorbance spectra from our outer membrane preparations did not have detectable chlorophyll a, we cannot preclude the possibility of cytoplasmic or thylakoid contamination, but we do not believe that contamination could explain the large amounts of IdiA detected in the outer membrane, as demonstrated in the Coomassie-stained gel in Fig. 7B. Additionally, genomic observations from Prochlorococcus sp. strains MED4 and MIT9313 and Synechococcus sp. strain WH8102 suggest that the IdiA protein is the only potential Fe binding protein identifiable to scavenge cellular Fe.
If the sole role of the IdiA protein in marine Synechococcus sp. strains is to acquire Mn atoms for PSII, it would further obscure our understanding of how the cell acquires Fe. Fitting with the requirement of IdiA for Fe transport, the genes encoding the two IdiA homologues found in Synechocystis sp. strain PCC6803 were recently inactivated and shown to be required for Fe3+ transport into the cell (23). Moreover, it has been shown that IdiA expression in freshwater Synechococcus sp. strain PCC6301 is dependent on two proteins, IdiB and DpsA (31), which were not identifiable in the genomes of Synechococcus sp. strain WH8102 and Prochlorococcus sp. strains MED4 and MIT9313. These findings are not inconsistent with IdiA's functioning in Synechococcus sp. strain WH7803 (and possibly all oligotrophic marine cyanobacteria) as a periplasmic Fe binding protein, in the same manner as SfuA (an IdiA homologue) in Serratia marcescens (3). We are currently in the process of inactivating the idiA gene in Synechococcus sp. strain WH8102 to test this proposal.
IdiA as an Fe stress indicator.
The present study has revealed insights into the molecular mechanism of Fe scavenging employed by species of the oligotrophic marine cyanobacteria Synechococcus, Trichodesmium, and Crocosphaera. In these organisms, in addition to the well-defined growth rate and pigmentation changes (11, 41), Fe stress results in the induction or repression of several proteins. We have identified one of the major proteins induced during Fe stress in all three genera as an IdiA homologue.
Three lines of evidence indicate that IdiA will make an excellent biomarker for Fe stress in oceanic cyanobacteria: it is expressed at high levels in Fe-stressed cells of all the oceanic cyanobacteria tested, it is dynamically expressed in response to changing Fe regimens, and it appears to be extracytoplasmically located. The possession of a diagnostic for Fe stress in these important oligotrophic cyanobacteria will make it possible to directly test the hypothesis that Fe is a significant limiting growth factor in a variety of oceanic environments.
ACKNOWLEDGMENTS
This work was partially funded by the Woods Hole Oceanographic Institution postdoctoral scholarship and a subcontract from the Center for Bioinorganic Chemistry at Princeton University (grant no. CHE-9810248) to E.A.W. Additional funds were supplied by the Seaver Institute to J.W.M.
We thank E. Pistorius for generously supplying the IdiA and preimmune sera. Additionally we thank Katrina Edwards, Sonya Dyhrman, and Frederica Valois for reviewing drafts of the manuscript and Bruce Woodin and Michael Thomas for helpful discussion. Preliminary sequence data was obtained from the DOE Joint Genome Institute (JGI) at http://www.jgi.doe.gov/tempweb/JGI_microbial/html/index.html.
Footnotes
Woods Hole Oceanographic Institution contribution no. 10466.
REFERENCES
- 1.Adhikari P, Kirby S D, Nowalk A J, Veraldi K L, Schryvers A B, Mietzner T A. Biochemical characterization of a Haemophilus influenzae periplasmic iron transport operon. J Biol Chem. 1995;270:25142–25149. doi: 10.1074/jbc.270.42.25142. [DOI] [PubMed] [Google Scholar]
- 2.Angerer A, Gaisser S, Braun V. Nucleotide sequences of the sfuA, sfuB, and sfuC genes of Serratia marcescens suggest a periplasmic-binding-protein-dependent iron transport mechanism. J Bacteriol. 1990;172:572–578. doi: 10.1128/jb.172.2.572-578.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Angerer A, Klupp B, Braun V. Iron transport systems of Serratia marcescens. J Bacteriol. 1992;174:1378–1387. doi: 10.1128/jb.174.4.1378-1387.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Archibald F. Lactobacillus plantarum, an organism not requiring iron. FEMS Microbiol Lett. 1983;19:29. [Google Scholar]
- 5.Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. Short protocols in molecular biology. 4th ed. New York, N.Y: Wiley; 1999. [Google Scholar]
- 6.Behrenfeld M J, Kolber Z S. Widespread iron limitation of phytoplankton in the south pacific ocean. Science. 1999;283:840–843. doi: 10.1126/science.283.5403.840. [DOI] [PubMed] [Google Scholar]
- 7.Boos W, Lucht J M. Periplasmic Binding Protein-Dependent ABC Transporters. In: Neidhardt F C, editor. Escherichia coli and Salmonella: cellular and molecular biology. Washington, D.C.: ASM Press; 1999. [Google Scholar]
- 8.Brahamsha B. An abundant cell-surface polypeptide is required for swimming by the nonflagellated marine cyanobacterium Synechococcus. Proc Natl Acad Sci USA. 1996;93:6504–6509. doi: 10.1073/pnas.93.13.6504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Braun V, Killmann H. Bacterial solutions to the iron-supply problem. Trends Biochem Sci. 1999;24:104–109. doi: 10.1016/s0968-0004(99)01359-6. [DOI] [PubMed] [Google Scholar]
- 10.Capone D G, Zehr J P, Paerl H W, Bergman B, Carpenter E J. Trichodesmium, a globally significant marine cyanobacterium. Science. 1997;276:1221–1229. [Google Scholar]
- 11.Chadd H E, Joint I R, Mann N H, Carr N G. The marine picoplankter Synechococcus sp. WH 7803 exhibits an adaptive response to restricted iron availability. FEMS Microbiol Ecol. 1996;21:69–76. [Google Scholar]
- 12.Coale K H, Johnson K S, Fitzwater S E, Gordon R M, Tanner S, Chavez F P, Ferioli L, Sakamoto C, Rogers P, Millero F, Steinberg P, Nightingale P, Cooper D, Cochlan W P, Kudela R. A massive phytoplankton bloom induced by an ecosystem-scale iron fertilization experiment in the Equatorial Pacific Ocean. Nature. 1996;383:495–501. doi: 10.1038/383495a0. [DOI] [PubMed] [Google Scholar]
- 13.Dubrac S, Touati D. Fur positive regulation of iron superoxide dismutase in Escherichia coli: functional analysis of the sodB promoter. J Bacteriol. 2000;182:3802–3808. doi: 10.1128/jb.182.13.3802-3808.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Escolar L, Perez-Martin J, de Lorenzo V. Opening the iron box: transcriptional metalloregulation by the Fur protein. J Bacteriol. 1999;181:6223–6229. doi: 10.1128/jb.181.20.6223-6229.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fekkes P, Driessen A J. Protein targeting to the bacterial cytoplasmic membrane. Microbiol Mol Biol Rev. 1999;63:161–173. doi: 10.1128/mmbr.63.1.161-173.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ferreiros C, Criado M T, Gomez J A. The Neisserial 37 kDa ferric binding protein (FbpA) Comp Biochem Physiol B. 1999;123:1–7. doi: 10.1016/s0305-0491(99)00044-9. [DOI] [PubMed] [Google Scholar]
- 17.Ghassemian M, Straus N A. Fur regulates the expression of iron-stress genes in the cyanobacterium Synechococcus sp. strain PCC 7942. Microbiology. 1996;142:1469–1476. doi: 10.1099/13500872-142-6-1469. [DOI] [PubMed] [Google Scholar]
- 18.Goldman S J, Lammers P J, Berman M S, Sanders-Loehr J. Siderophore-mediated iron uptake in different strains of Anabaena sp. J Bacteriol. 1983;156:1144–1150. doi: 10.1128/jb.156.3.1144-1150.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guerinot M L. Microbial iron transport. Annu Rev Microbiol. 1994;48:743–772. doi: 10.1146/annurev.mi.48.100194.003523. [DOI] [PubMed] [Google Scholar]
- 20.Henikoff S, Henikoff J G. Amino acid substitution matrices from protein blocks. Proc Natl Acad Sci USA. 1992;89:10915–10919. doi: 10.1073/pnas.89.22.10915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hutchins D A, Witter A E, Butler A, Luther G W., III Competition among marine phytoplankton for different chelated iron species. Nature. 1999;400:858–861. [Google Scholar]
- 22.Katoh H, Grossman A R, Hagino N, Ogawa T. A gene of Synechocystis sp. strain PCC 6803 encoding a novel iron transporter. J Bacteriol. 2000;182:6523–6524. doi: 10.1128/jb.182.22.6523-6524.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Katoh H, Hagino N, Grossman A R, Ogawa T. Genes essential to iron transport in the cyanobacterium Synechocystis sp. strain PCC 6803. J Bacteriol. 2001;189:2779–2784. doi: 10.1128/JB.183.9.2779-2784.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kerry A, Laudenbach D E, Trick C G. Influence of iron limitation and nitrogen source on growth and siderophore production by cyanobacteria. J Phycol. 1988;24:566–571. [Google Scholar]
- 25.Lammers P J, Sanders-Loehr J. Active transport of ferric schizokinen in Anabaena sp. J Bacteriol. 1982;151:288–294. doi: 10.1128/jb.151.1.288-294.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Larsen R A, Thomas M G, Postle K. Proton motive force, ExbB and ligand-bound FepA drive conformational changes in TonB. Mol Microbiol. 1999;31:1809–1824. doi: 10.1046/j.1365-2958.1999.01317.x. [DOI] [PubMed] [Google Scholar]
- 27.Leonhardt K, Straus N A. An iron stress operon involved in photosynthetic electron transport in the marine cyanobacterium Synechococcus sp. PCC 7002. J Gen Microbiol. 1992;138:1613–1621. doi: 10.1099/00221287-138-8-1613. [DOI] [PubMed] [Google Scholar]
- 28.Martin J H, Coale K H, Johnson K S, Fitzwater S E, Gordon R M, Tanner S J, Hunter C N, Elrod V A, Nowicki J L, Coley T L, Barber R T, Lindley S, Watson A J, Van Scoy K, Law C S, Liddicoat M I, Ling R, Stanton T, Stocjel J, Collins C, Anderson A, Bidigare R, Ondrusek M, Latasa M, Millero F J, Lee K, Yao W, Zhang J Z, Friederich D, Sakamoto C, Chavez F, Buck K, Kolber Z, Greene R, Falkowski P, Chisholm S W, Hoge F, Swift R, Yungel J, Turner S, Nightingale P, Hatton A, Liss P, Tindale N W. Testing the iron hypothesis in ecosystems of the equatorial Pacific Ocean. Nature. 1994;371:123–129. [Google Scholar]
- 29.McKay D B, Steitz T A. Structure of catabolite gene activator protein at 2.9 A resolution suggests binding to left-handed B-DNA. Nature. 1981;290:744–749. doi: 10.1038/290744a0. [DOI] [PubMed] [Google Scholar]
- 30.Michel K P, Exss-Sonne P, Scholten-Beck G, Kahmann U, Ruppel H G, Pistorius E K. Immunocytochemical localization of IdiA, a protein expressed under iron or manganese limitation in the mesophilic cyanobacterium Synechococcus PCC 6301 and the thermophilic cyanobacterium Synechococcus elongatus. Planta. 1998;205:73–81. doi: 10.1007/s004250050298. [DOI] [PubMed] [Google Scholar]
- 31.Michel K P, Kruger F, Puhler A, Pistorius E K. Molecular characterization of idiA and adjacent genes in the cyanobacteria Synechococcus sp. strains PCC 6301 and PCC 7942. Microbiology. 1999;144:1473–1484. doi: 10.1099/13500872-145-6-1473. [DOI] [PubMed] [Google Scholar]
- 32.Michel K P, Thole H H, Pistorius E K. IdiA, a 34 kDa protein in the cyanobacteria Synechococcus sp. strains PCC 6301 and PCC 7942, is required for growth under iron and manganese limitations. Microbiology. 1996;142:2635–2645. doi: 10.1099/00221287-142-9-2635. [DOI] [PubMed] [Google Scholar]
- 33.Paerl H W, Prufert-Bebout L E, Guo C. Iron-stimulated N2 fixation and growth in natural and cultured populations of the planktonic marine cyanobacterium Trichodesmium spp. Appl Environ Microbiol. 1994;60:1044–1047. doi: 10.1128/aem.60.3.1044-1047.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Park Y I, Sandstrom S, Gustafsson P, Oquist G. Expression of the isiA gene is essential for the survival of the cyanobacterium Synechococcus sp. PCC 7942 by protecting photosystem II from excess light under iron limitation. Mol Microbiol. 1999;32:123–129. doi: 10.1046/j.1365-2958.1999.01332.x. [DOI] [PubMed] [Google Scholar]
- 35.Partensky F, Blanchot J, Vaulot D. Differential distribution and ecology of Prochlorococcus and Synechococcus in oceanic waters: a review. In: Charpy L, Larkum A W D, editors. Marine cyanobacteria. Vol. 19. Monte Carlo, Monaco: Institut Océanographique; 1999. pp. 457–475. [Google Scholar]
- 36.Partensky F, Hess W R, Vaulot D. Prochlorococcus, a marine photosynthetic prokaryote of global significance. Microbiol Mol Biol Rev. 1999;63:106–127. doi: 10.1128/mmbr.63.1.106-127.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Posey J E, Gherardini F C. Lack of a role for iron in the Lyme disease pathogen. Science. 2000;288:1651–1653. doi: 10.1126/science.288.5471.1651. [DOI] [PubMed] [Google Scholar]
- 38.Reddy K J, Bullerjahn G S, Sherman D M, Sherman L A. Cloning, nucleotide sequence, and mutagenesis of a gene (irpA) involved in iron-deficient growth of the cyanobacterium Synechococcus sp. strain PCC7942. J Bacteriol. 1988;170:4466–4476. doi: 10.1128/jb.170.10.4466-4476.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Reimmann C, Patel H M, Serino L, Barone M, Walsh C T, Haas D. Essential PchG-dependent reduction in pyochelin biosynthesis of Pseudomonas aeruginosa. J Bacteriol. 2001;183:813–820. doi: 10.1128/JB.183.3.813-820.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Resch C M, Gibson J. Isolation of the carotenoid-containing cell wall of three unicellular cyanobacteria. J Bacteriol. 1983;155:345–350. doi: 10.1128/jb.155.1.345-350.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Reuter J G, Hutchins D A, Smith R W, Unsworth N L. Iron nutrition of Trichodesmium. In: Carpenter E J, Capone D G, Reuter J G, editors. Marine pelagic cyanobacteria: Trichodesmium and other diazotrophs. Boston, Mass: Kluwer Academic Publishers; 1992. pp. 289–306. [Google Scholar]
- 42.Rippka R, Castenholz R W, Waterbury J B, Herdman M. Form-genus V. Cyanothece. In: Garrity G M, editor. Bergey's manual of systematic bacteriology. Vol. 2. New York, N.Y: Springer; 2001. p. 501. [Google Scholar]
- 43.Rueter J G, Fujita O K. Response of marine Synechococcus (Cyanophyceae) cultures to iron limitation. J Phycol. 1991;27:173–178. [Google Scholar]
- 44.Sanders J D, Cope L D, Hansen E J. Identification of a locus involved in the utilization of iron by Haemophilus influenzae. Infect Immun. 1994;62:4515–4525. doi: 10.1128/iai.62.10.4515-4525.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Scanlan D J, Mann N H, Carr N G. Effect of iron and other nutrient limitations on the pattern of outer membrane proteins in the cyanobacterium Synechococcus PCC7942. Arch Microbiol. 1989;152:224–228. [Google Scholar]
- 46.Stintzi A, Barnes C, Xu J, Raymond K N. Microbial iron transport via a siderophore shuttle: a membrane ion transport paradigm. Proc Natl Acad Sci USA. 2000;97:10691–10696. doi: 10.1073/pnas.200318797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Tortell P D, Maldonado M T, Granger J, Price N M. Marine bacteria and biogeochemical cycling of iron in the oceans. FEMS Microbiol Ecol. 1999;29:1–11. [Google Scholar]
- 48.Waterbury J B, Rippka R. Subsection I. Order Chroococcaloes. In: Staley J T, editor. Bergey's manual of systematic bacteriology. Vol. 3. Baltimore, Md: Williams & Wilkins; 1989. pp. 1728–1746. [Google Scholar]
- 49.Waterbury J B, Watson S W, Valois F W, Franks D G. Biological and ecological characterization of the marine unicellular cyanobacterium Synechococcus. In: Platt T, Li W K W, editors. Photosynthetic picoplankton. Vol. 214. Ottawa, Canada: Dept. of Fisheries and Oceans; 1986. pp. 71–120. [Google Scholar]
- 50.Waterbury J B, Willey J M. Isolation and growth of marine planktonic cyanobacteria. Methods Enzymol. 1988;167:100–105. [Google Scholar]
- 51.Webb R, Troyan T, Sherman D, Sherman L A. MapA, an iron-regulated, cytoplasmic membrane protein in the cyanobacterium Synechococcus sp. strain PCC7942. J Bacteriol. 1994;176:4906–4913. doi: 10.1128/jb.176.16.4906-4913.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wilhelm S W. Ecology of iron-limited cyanobacteria: a review of physiological responses and implications for aquatic systems. Aquat Microb Ecol. 1995;9:295–303. [Google Scholar]
- 53.Wilhelm S W, MacAuley K, Trick C G. Evidence for the importance of catechol-type siderophores in the iron-limited growth of a cyanobacterium. Limnol Oceanogr. 1998;43:992–997. [Google Scholar]
- 54.Wilhelm S W, Maxwell D P, Trick C G. Growth, iron requirements, and siderophore production in iron-limited Synechococcus PCC 7002. Limnol Oceanogr. 1996;41:89–97. [Google Scholar]
- 55.Wilhelm S W, Trick C G. Iron-limited growth of cyanobacteria: Multiple siderophore production is a common response. Limnol Oceanogr. 1994;39(8):1979–1984. [Google Scholar]
- 56.Wilhelm S W, Trick C G. Physiological profiles of Synechococcus (Cyanophyceae) in iron-limiting continuous cultures. J Phycol. 1995;31:79–85. [Google Scholar]
- 57.Zehr J P, Waterbury J B, Turner P J, Montoya J P, Omoregie E, Steward G F, Hansen A, Karl D M. Unicellular cyanobacteria fix N2 in the subtropical North Pacific Ocean. Nature. 2001;412:635–638. doi: 10.1038/35088063. [DOI] [PubMed] [Google Scholar]
- 58.Zimmermann L, Angerer A, Braun V. Mechanistically novel iron(III) transport system in Serratia marcescens. J Bacteriol. 1989;171:238–243. doi: 10.1128/jb.171.1.238-243.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]