Abstract
Introduction
The correlation between total and free polymyxin B (PMB including PMB1 and PMB2) exposure in vivo and acute kidney injury (AKI) remains obscure. This study explores the relationships between plasma exposure of PMB1 and PMB2 and nephrotoxicity, and investigates the risk factors for PMB-induced acute kidney injury (AKI) in critically ill patients.
Methods
Critically ill patients who used PMB and met the criteria were enrolled. The total plasma concentration and plasma binding of PMB1 and PMB2 were analysed by liquid chromatography–tandem mass spectrometry and equilibrium dialysis.
Results
A total of 89 patients were finally included, and AKI developed in 28.1% of them. The peak concentration of PMB1 (Cmax (B1)) (adjusted odds ratio (AOR) = 1.68, 95% CI 1.08–2.62, p = 0.023), baseline BUN level (AOR = 1.08, 95% CI 1.01–1.16, p = 0.039) and hypertension (AOR = 3.73, 95% CI 1.21–11.54, p = 0.022) were independent risk factors for PMB-induced AKI. The area under the ROC curve of the model was 0.799. When Cmax (B1) was 5.23 μg/ml or more, the probability of AKI was higher than 50%. The ratio of PMB1/PMB2 decreased after PMB preparation entered into the body. The protein binding rate in critically ill patients indicated significant individual differences. Free Cmax (B) and free Cmax (B1) levels in the AKI group were significantly (p < 0.05) higher than those in the non-AKI group. Total and free concentrations of PMB in patients showed a positive correlation.
Conclusions
Both the ROC curve and logistic regression model showed that Cmax (B1) was a good predictor for the probability of PMB-induced AKI. Early therapeutic drug monitoring (TDM) of PMB should be considered in critically ill patients. Compared with Cmin (B), Cmax (B) and Cmax (B1) may be helpful for the early prediction of PMB-induced AKI in critically ill patients.
Keywords: Drug exposure, Free concentration, Independent risk factors, Nephrotoxicity, Polymyxin B1, Polymyxin B
Key Summary Points
| Data for the relationship between total and free PMB (PMB1 and PMB2) exposure in vivo and nephrotoxicity is still insufficient. |
| Total and free concentrations of PMB, PMB1 or PMB2 may be used to predict PMB-induced nephrotoxicity. |
| A predicted probability of developing AKI of 50% corresponds to a Cmax (B1) of 5.23 μg/ml. |
| Early therapeutic drug monitoring of PMB should be considered in critically ill patients. Compared with Cmin (B), Cmax (B) and Cmax (B1) may be helpful for the early prediction of PMB-induced AKI in critically ill patients. |
Introduction
The incidence of multidrug-resistant (MDR) gram-negative bacterial (GNB) infections has increased dramatically over the last decade, and such infections have emerged as a major challenge in global public health. The mortality rate of MDR-GNB infections is 40% or higher [1–3]. Novel antibiotic agents for the treatment of MDR-GNB infections are limited. Owing to its potential activity against MDR-GNB infection, the “old” drug polymyxin has been repurposed and recommended as a last-resort therapy for MDR-GNB infections [4, 5].
Polymyxin B (PMB) and colistin, which have similar pharmacologically active moieties, are two different agents in the polymyxin class. PMB is a fermented mixture of more than 30 components from Paenibacillus polymyxa [6]. Nephrotoxicity is the major dose-limiting factor impacting the clinical use of PMB. Previous studies have indicated that the incidence of nephrotoxicity is 20–60% after intravenous administration of PMB [6–11]. Colistin is administered intravenously as a prodrug colistimethate sodium, affected by the ratio of prodrug lost to renal elimination prior to activation. Some studies showed the incidence of AKI of colistin is 30–76.1% [12–14]. Azad et al. found that PMB can noticeably accumulate in the renal cortex, especially in proximal tubular cells, and induce renal tubular epithelial cell apoptosis or necrosis [15], which indicates that the nephrotoxicity of PMB might be related to drug exposure in vivo. For critically ill patients, there are noticeable individual differences in the pharmacokinetics of polymyxin B [7, 8, 16], and the dose of PMB might not represent the actual amount of the drug to which the patient is exposed. Polymyxin B1 (PMB1) and polymyxin B2 (PMB2) are the two major components of polymyxin B (PMB), accounting for more than 60% of the total weight, and have been used to characterize the effects of PMB exposure in vivo for therapeutic drug monitoring (TDM) [16, 17].
Previous studies have reported some risk factors for PMB-induced nephrotoxicity, such as age, baseline SCr (serum creatinine) level, body mass index (BMI), concomitant use of vasoactive drugs and vancomycin, infection site, duration of therapy, and a daily dose of 200 mg or more [7, 8, 18–22]; however, PMB exposure in vivo was not assessed in these previous studies. According to the international consensus [23], the magnitude of polymyxin (i.e. PMB and colistin) exposure is the most important risk factor for polymyxin-associated acute kidney injury. Previously, for PMB, only Han et al. [22] and Wang et al. [24] found that the plasma trough concentration (Cmin) of PMB and an AUCss,24h of at least 100 mg·h/L of PMB were independent risk factors for PMB-induced nephrotoxicity, respectively. Notably, the comorbidity variables included in their studies were limited. Evidence for the relationship between PMB exposure and nephrotoxicity is still insufficient with a limited sample size. Furthermore, whether PMB1 and PMB2, the primary constituents of PMB, are associated with nephrotoxicity has not yet been reported.
On the other hand, only the unbound fraction (fu) of the drug is pharmacologically active, as the protein-bound drug cannot reach the site of infection. The reported protein binding rate of PMB ranges from 58% to 92.4% [25, 26]. The variation in protein binding can affect the volume of distribution and clearance of PMB and, thus, its efficacy and toxicity [27]. Given that pharmacological activity depends on the unbound protein concentration rather than the total plasma concentration, determination of the free protein concentration is essential. However, currently available reports on TDM of PMB did not directly assess the free drug concentration. The relationship between the free drug concentration and renal toxicity is still unknown.
Therefore, the present study explores the relationship between exposure to total and free PMB (including PMB1 and PMB2) in the plasma and nephrotoxicity, and investigates the risk factors for PMB-induced nephrotoxicity in critically ill patients, providing possible reference data for clinical TDM.
Methods
Patients and Data
A retrospective, observational cohort study was conducted at three general tertiary hospitals in China. As the central laboratory of the rational use of anti-infection agent’s technology demonstration base, the TDM laboratory of the Third Hospital of Changsha also undertook the TDM of polymyxin B for the other two hospitals. The Third Hospital of Changsha was the lead institution. The study was approved by the Ethics Research Committee of the Third Hospital of Changsha (No. CS3-KY-2021EC-008), and the requirement for written informed consent was waived. Ethics approval and the informed consent waiver were also accepted by the other two hospitals.
Critically ill patients (aged 18 years or more) with suspected or confirmed MDR-GNB infections who received intravenous injection of PMB sulfate (SPH No. 1, Biochemical and Pharmaceutical Co., Ltd. (Shanghai, China); PMB1/PMB2 ratio of approximately 5.18 ± 0.13) from May 1, 2019 to December 31, 2020 were enrolled. All patients received TDM of PMB. Patients were excluded if (a) they were undergoing haemodialysis and haemodiafiltration or extracorporeal membrane oxygenation; (b) they received PMB for a period of 48 h or less; or (c) the necessary medical data for the patient were lacking. The following medical information was collected: demographics, dosage regimen of PMB (including daily dosage, total dosage and duration days), comorbidities, type of infection, isolated microorganisms, concomitant drugs, duration of hospitalization, and laboratory test results. Creatinine clearance (CrCL) was calculated using the Cockcroft–Gault equation [28] and the CrCL before PMB treatment was used as an observed variable for PMB-induced AKI. The age-adjusted Charlson comorbidity index was used to evaluate the prognostic value of comorbidities [29, 30]. APACHE II scores were calculated on the basis of acute physiology measurements, age, and chronic health evaluation [31]. The definition of severe pneumonia was according to the 2019 ATS/IDSA Guidelines for the Diagnosis and Treatment of Adults with Community-Acquired Pneumonia [32].
PMB Administration and Sample Collection
Patient blood samples were collected at the sixth or later injection of PMB sulfate. The time for Cmax (Tmax) of PMB1 and PMB2 was achieved immediately after the end of PMB infusion [17]. Considering the practical operability and the half-life of PMB, blood samples (2–3 ml) were collected 10 min after completion of PMB infusion to determine the Cmax. In addition, the time of blood sample collection of trough PMB plasma concentration (Cmin) was collected immediately before PMB injection [22, 33].
Blood samples were centrifuged immediately at 3500g and 2–8 °C for 10 min to obtain the plasma, and the plasma was collected in two tubes (one for determination of total PMB1 and PMB2 levels and the other for determination of the plasma protein binding rates of PMB1 and PMB2) and then stored at − 80 °C until analysis.
Quantification of Total and Unbound PMB1 and PMB2 Concentrations
A validated liquid chromatography–tandem mass spectrometry (HPLC–MS/MS) method was used to quantify PMB1 and PMB2 levels at the TDM laboratory of the Third Hospital of Changsha. Reference standards of PMB1 sulfate (purity 94.078%) and PMB2 sulfate (purity 99.358%) used for the preparation of calibration standards and quality control samples were obtained from TOKU-E (Bellingham, WA, USA). A Shim-pack GIST C18 column (2.1 × 100 mm, 3 μm, Shimadzu, Japan) was used for liquid chromatographic separation. The mobile phase included solvent A (0.1% formic acid in water) and solvent B (acetonitrile). The following gradient elution was performed at a total flow rate of 0.4 ml/min for analyte elution: 5% B for 0.5 min, 5–50% B for 3.0 min, 50% to 60% for 2.0 min and 5% B for 4.5 min. The temperature of the autosampler was maintained at 6 °C. The HPLC system was combined with a Shimadzu LC MS-8050 mass spectrometer and performed with multiple reaction monitoring (MRM) in positive ionization mode with an m/z of 402.1 → 101.15 (PMB1) and 397.4 → 101.15 (PMB2). The PMB concentration was calculated using the following equation:
| 1 |
where C represents the concentration and M is the molar mass.
For total PMB1 and PMB2 concentration analysis, 20 μl polymyxin E solution (2.66 μg/ml) was added to 100 μl of human plasma sample and vortexed for 30 s. Human plasma was extracted with 20 μl formic acid solution and 280 μl acetonitrile. After the extract was mixed for 2 min and centrifuged at 15,000 rpm at 4 °C for 10 min, 150 μl supernatant was collected carefully and resuspended in 150 μl purified water. A 2-μl aliquot of this solution was injected into the LC–MS/MS system for analysis.
The free PMB1 and PMB2 concentrations were analysed by equilibrium dialysis. The 48-well plate equilibrium dialysis plate was assembled with a semipermeable membrane (Thermo Fisher Scientific, Waltham, MA, USA) separating two reservoirs of the cell unit. The semipermeable membrane is selective for high molecular polymers, i.e. only small molecular substances can pass through the membrane while proteins and cells are excluded. Considering the adequacy of dialysis and stability of PMB in whole blood for 6 h at 37 °C determined in our previous study [34], the plasma samples were dialyzed against isotonic phosphate buffer (pH 7.4) at 37 °C for 5 h. For dialysis samples, 50 μl of blank plasma or phosphate buffer was added to the corresponding sample and processed as described above. The fraction of the plasma fu value was calculated using the following equation:
| 2 |
where Cd represents the concentration in the dialysate after completion of dialysis and Cp is the corresponding concentration in the plasma (μg/ml).
The calibration curves showed acceptable linearity over 0.033–18.816 μg/ml for PMB1 and 0.034–19.872 μg/ml for PMB2. The interday and intraday precision were less than 12% and less than 9%, respectively. The accuracy was 99.8–110.4%.
Nephrotoxicity Definition
Acute kidney injury (AKI) was studied to assess PMB-induced nephrotoxicity. RIFLE criteria [35] were used to define AKI in this study according to SCr. Increased SCr × 1.5 was defined as risk stage of renal function; increased SCr × 2 was defined as injury; increased SCr × 3 was defined as failure. Persistent acute renal failure or loss of function for more than 4 weeks was defined as loss. Patients who met this criterion were defined as the AKI group. Assessment of SCr levels was performed at baseline, during PMB treatment, and at the end of PMB treatment.
Data Analysis
Statistical analysis was performed using R version 4.1.1 (R Foundation for Statistical Computing; Vienna, Austria; ISBN 3-000051-07-0, http://www.R-project.org) and SPSS for Windows (version 22.0). Continuous variables are presented as the mean ± standard deviation (SD) if the data were normally distributed and were compared using Student’s t tests. The median and interquartile range (IQR) are presented for non-normally distributed data, and the Mann–Whitney U test was used. Categorical variables are expressed as counts and percentages, and the chi-square test or Fisher’s exact test was used. Spearman’s rank correlation coefficient (r) was used to analyse the correlation between the total concentrations and free concentrations of PMB, PMB1 and PMB2. Univariate analysis was performed for all variables to identify possible risk factors for AKI. Variables found to be statistically significant (p < 0.05) were entered into the multivariate logistic regression models. A forward stepwise (likelihood ratio) method was used to determine the risk factors. Adjusted odds ratios (AOR), corresponding two-sided 95% confidence intervals (CIs) and p values are presented for the final logistic model. The Hosmer–Lemeshow test was used to determine the fitness of the model. The variance inflation factor (VIF) was used to test for multicollinearity among the risk factors in the final logistic models to ensure the independence of each variable. VIF values greater than 4.0 were considered to indicate an interaction among predictors [36]. A receiver operating characteristic (ROC) curve was drawn to evaluate the discriminatory power of the factors in the final multivariate logistic regression model. Discriminatory power was used as a measure of the model’s ability to distinguish between patients with AKI and without AKI. A p value less than 0.05 was considered statistically significant.
Results
Characteristics
A total of 117 patients were included in the study. Twenty-eight patients were excluded; 11 patients had a PMB therapy duration less than 48 h, and necessary medical data was missing for 17 patients. Thus, 89 patients met the inclusion criteria (Fig. 1). The demographic characteristics of all patients are summarized in Table 1. The average APACHE II score of patients was 19.1 ± 7.4. The most common pathogenic bacteria were Acinetobacter baumannii (N = 59; 66.3%), followed by Klebsiella pneumoniae (N = 31; 34.8%) and Pseudomonas aeruginosa (N = 22; 24.7%). Sepsis (N = 26; 29.2%) and lung infection (N = 54; 60.7%) were the main types of infection. Seventeen patients had chronic renal dysfunction.
Fig. 1.
Patient selection flow chart
Table 1.
Characteristics of patients
| Characteristics | Value (N = 89) |
|---|---|
| Demographics | |
| Age (years) (mean ± SD) | 60.7 ± 15.1 |
| Gender (male) (n (%)) | 63 (70.8%) |
| Weight (kg) | 55.0 (50.0–61.0) |
| APACHE II scores | 19.1 ± 7.4 |
| Hospitalization days | 27 (21–35) |
| Comorbiditiesa | |
| ACCI (median (IQR)) | 3 (1–4) |
| Sepsis (n (%)) | 26 (29.2%) |
| Severe pneumonia (n (%)) | 26 (29.2%) |
| Sepsis + severe pneumonia (n (%)) | 12 (13.5%) |
| Lung infection (n (%)) | 54 (60.7%) |
| Respiratory failure (n (%)) | 18 (20.2%) |
| Anaemia (n (%)) | 12 (13.5%) |
| Coronary heart disease (n (%)) | 22 (24.7%) |
| Hypertension (n (%)) | 43 (48.3%) |
| Type 2 diabetes (n (%)) | 13 (14.6%) |
| Chronic liver disease (n (%)) | 15 (16.9%) |
| Chronic renal dysfunction (n (%)) | 17 (19.1%) |
| Cerebral infarction (n (%)) | 20 (22.5%) |
| Pathogenic bacteria | |
| Acinetobacter baumannii (n (%)) | 59 (66.3%) |
| Klebsiella pneumoniae (n (%)) | 31 (34.8%) |
| Pseudomonas aeruginosa (n (%)) | 22 (24.7%) |
| Escherichia coli (n (%)) | 5 (5.6%) |
| Stenotrophomonas maltophilia (n (%)) | 7 (7.9%) |
| Candida (n (%)) | 23 (25.8%) |
| PMB treatment | |
| PMB daily dosage (mg) (median (IQR)) | 100 (100–100) |
| PMB total dosage (mg) (median (IQR)) | 1000 (700–1300) |
| PMB duration (days) (median (IQR)) | 10 (7–12)) |
| PMB monotherapy (n (%)) | 14 (15.7%) |
| Concomitant with nephrotoxic drugsb (n (%)) | 35 (39.3%) |
| Concomitant with β-lactam (n (%)) | 45 (50.6%) |
| Carbapenem (n (%)) | 62 (69.7%) |
| Tigecycline (n (%)) | 23 (25.8%) |
| Laboratory examination (before PMB treatment) | |
| WBC (× 109/L) (median (IQR)) | 10.9 (6.8–14.0) |
| Percentage of neutrophils (%) | 80.4 (71.2–86.1) |
| Albumin (g/L) (median (IQR)) | 32.4 (29.6–35.6) |
| Serum creatinine (µmol/L) (median (IQR)) | 82.6 (57.7–124.0) |
| BUN (mmol/L) (median (IQR)) | 11.5 (7.3–18.4) |
| Procalcitonin (ng/ml) (median (IQR)) | 1.2 (0.2–4.4) |
| HCRP (mg/L) (median (IQR)) | 96.0 (54.0–123.0) |
| CrCL (ml/min) | 58.1 (36.4–86.0) |
APACHE Acute Physiology and Chronic Health Evaluation, ACCI age-adjusted Charlson comorbidity index, PMB polymyxin B, WBC white blood cell count, BUN blood urea nitrogen, HCRP high-sensitivity C-reactive protein, CrCL creatinine clearance, IQR interquartile range
aFrom the clinic diagnosis of electronic medical records
bIncludes vancomycin, sulfamethoxazole, gentamicin, amikacin, ciprofloxacin and levofloxacin in this study
Total and Free Concentrations
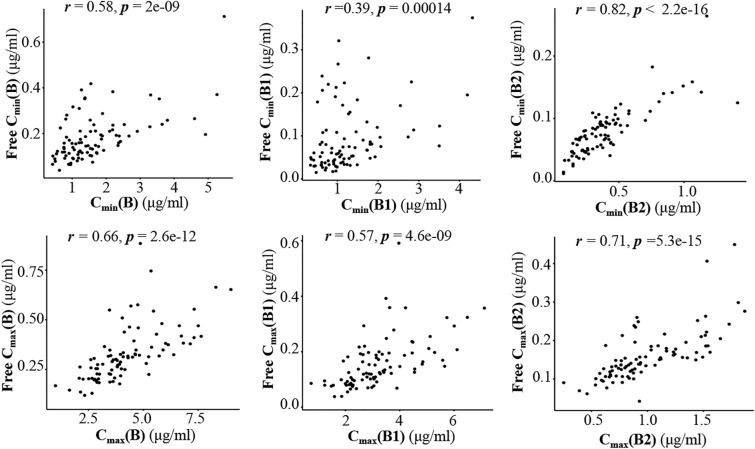
The total and free concentrations of PMB1, PMB2 and PMB are shown in Table 3. The median protein binding rates of PMB, PMB1 and PMB2 in patients were 91.8% (range 65.7–96.0%), 95.1% (range 61.7–98.2%) and 82.8% (range 59.2–95.5%), respectively. Spearman’s rank correlation analysis showed that the total concentrations of PMB, PMB1 and PMB2 were positively correlated with their free concentrations (Fig. 2).
Table 3.
Total concentration and free concentration of PMB in patients’ plasma
| All | Non-AKI group | AKI group | p value | |
|---|---|---|---|---|
| (N = 89) | (N = 64) | (N = 25) | ||
| Total concentration (median (IQR)) | ||||
| Cmin (B) (μg/ml) | 1.39 (0.97–1.91) | 1.25 (0.92–1.87) | 1.70(1.39–1.94) | 0.02* |
| Cmin (B1) (μg/ml) | 1.04 (0.72–1.50) | 0.98 (0.64–1.46) | 1.30 (1.03–1.50) | 0.02* |
| Cmin (B2) (μg/ml) | 0.36 (0.23–0.46) | 0.30 (0.22–0.42) | 0.43 (0.34–0.48) | 0.03* |
| Cmax (B) (μg/ml) | 3.99 (3.01–5.18) | 3.82 (2.92–4.84) | 4.55 (3.73–5.96) | 0.01* |
| Cmax (B1) (μg/ml) | 3.07 (2.30–3.82) | 2.89 (2.22–3.68) | 3.45 (3.00–4.50) | 0.01* |
| Cmax (B2) (μg/ml) | 0.91 (0.75–1.24) | 0.89 (0.69–1.17) | 0.99 (0.85–1.44) | 0.04* |
| Cmin (B1)/Cmin (B2) | 3.28 (2.90–3.66) | 3.27 (2.91–3.65) | 3.37 (2.85–3.68) | 0.98 |
| Cmax (B1)/Cmax (B2) | 3.27 (3.02–3.70) | 3.23 (2.97–3.69) | 3.46 (3.13–3.78) | 0.08 |
| Protein binding rate (median (IQR)) | ||||
| Cmin (B) (%) | 89.7 (83.4–92.7) | 88.6 (83.4–92.1) | 90.1 (84.5–93.2) | 0.21 |
| Cmin (B1) (%) | 94.5 (88.9–96.1) | 94.3 (88.7–96.1) | 94.7 (89.6–96.1) | 0.58 |
| Cmin (B2) (%) | 78.5 (73.3–83.0) | 77.9 (71.6–81.9) | 80.6 (76.1–85.1) | 0.10 |
| Cmax (B) (%) | 92.7 (91.1–93.7) | 92.4 (91.0–93.7) | 92.9 (91.1–93.8) | 0.43 |
| Cmax (B1) (%) | 95.6 (94.3–96.5) | 95.8 (94.3–96.5) | 95.5 (94.3–96.3) | 0.80 |
| Cmax (B2) (%) | 84.4 (82.2–86.4) | 83.9 (82.1–86.0) | 85.9 (83.2–87.1) | 0.10 |
| Free concentration (median (IQR)) | ||||
| Free Cmin (B) (μg/ml) | 0.16 (0.12–0.23) | 0.16 (0.12–0.21) | 0.14 (0.11–0.35) | 0.54 |
| Free Cmin (B1) (μg/ml) | 0.07 (0.04–0.11) | 0.07 (0.04–0.10) | 0.05 (0.04–0.19) | 0.51 |
| Free Cmin (B2) (μg/ml) | 0.07 (0.05–0.10) | 0.07 (0.05–0.10) | 0.07 (0.06–0.09) | 0.69 |
| Free Cmax (B) (μg/ml) | 0.31 (0.23–0.38) | 0.29 (0.22–0.37) | 0.33 (0.29–0.47) | 0.02* |
| Free Cmax (B1) (μg/ml) | 0.14 (0.10–0.20) | 0.14 (0.11–0.18) | 0.19 (0.12–0.26) | 0.02* |
| Free Cmax (B2) (μg/ml) | 0.15 (0.12–0.19) | 0.14 (0.11–0.18) | 0.16 (0.14–0.19) | 0.13 |
Cmin trough concentration, Cmax peak concentration, B polymyxin B, B1 polymyxin B1, B2 polymyxin B2
*Denotes p < 0.05, differences between the AKI group and the non-AKI group were tested for statistical significance
Fig. 2.
Spearman’s rank correlation between total concentrations and free concentrations (r represents the Spearman’s rank correlation coefficient). Cmax (B), Cmax (B1) and Cmax (B2) represent the peak plasma concentrations of PMB, PMB1 and PMB2, respectively; Cmin (B), Cmin (B1) and Cmin (B2) represent the trough plasma concentrations of PMB, PMB1 and PMB2, respectively
The ratio of the Cmin of PMB1 (Cmin (B1)) to the Cmin of PMB2 (Cmin (B2)) in patients ranged from 1.53 to 6.76, and the ratio of the Cmax of PMB1 (Cmax (B1)) to the Cmax of PMB2 (Cmax (B2)) ranged from 1.48 to 4.45. Only two patients had a PMB1/PMB2 ratio within the range of 5.18 ± 20% (the ratio of PMB1/PMB2 in the preparation was ± 20%). The concentrations of PMB1 and PMB2 in patient plasma are shown in Fig. 3.
Fig. 3.
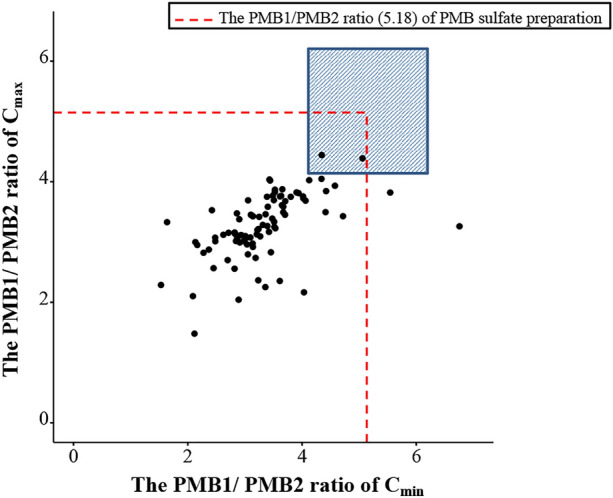
Distribution of the ratio (PMB1/PMB2) in the clinical sample. The blue area represents the range (5.18 ± 20%) of the ratio (PMB1/PMB2) of PMB preparation. Cmax represents the peak plasma concentration. Cmin represents the trough plasma concentration
AKI Analysis
According to the RIFLE criteria, 25 (28.1%) patients experienced AKI. The characteristics of the AKI group (N = 25) and the non-AKI group (N = 64) were analysed (Table 2). Patients in the AKI group were more likely to suffer from hypertension (p = 0.020), type 2 diabetes (p = 0.025) and chronic renal dysfunction (p = 0.011) group and had higher baseline BUN levels (p = 0.009) than patients in the non-AKI. The plasma concentrations (Cmin and Cmax) of PMB, PMB1 and PMB2 (p < 0.05) and the free Cmax (B) and free Cmax (B1) were significantly higher (p < 0.05) in the AKI group than in the non-AKI group (Table 3). On the basis of these results, a total of 12 possible risk factors were identified by the univariate analysis (Tables 2 and 3).
Table 2.
Characteristics of patients in the AKI group and non-AKI group
| Characteristics | Non-AKI group | AKI group | p value |
|---|---|---|---|
| (N = 64) | (N = 25) | ||
| Demographics | |||
| Age (years) (mean ± SD) | 60.6 ± 14.8 | 61 ± 16.2 | 0.91 |
| Gender (male) (n (%)) | 46 (71.9%) | 17 (68.0%) | 0.53 |
| Weight (kg) (median (IQR)) | 55.0 (49.5–60.3) | 60.0 (52.0–61.0) | 0.40 |
| APACHE II scores | 18.7 ± 7.1 | 19.9 ± 8.1 | 0.54 |
| Hospitalization days (median (IQR)) | 28 (19–36) | 27 (24–33) | 0.71 |
| Comorbiditiesa | |||
| ACCI (median (IQR)) | 3 (1–4) | 3 (2–5) | 0.32 |
| Sepsis (n (%)) | 16 (25.0%) | 10 (40.0%) | 0.16 |
| Severe pneumonia (n (%)) | 19 (29.7%) | 7 (28.0%) | 0.88 |
| Sepsis + severe pneumonia (n (%)) | 8 (12.5%) | 4 (16.0%) | 0.93 |
| Lung infection (n (%)) | 37 (57.8%) | 17 (68.0%) | 0.43 |
| Respiratory failure (n (%)) | 15 (23.4%) | 3 (12.0%) | 0.36 |
| Anaemia (n (%)) | 7 (10.9%) | 5 (20.0%) | 0.44 |
| Coronary heart disease (n (%)) | 15 (23.4%) | 7 (28.0%) | 0.65 |
| Hypertension (n (%)) | 26 (40.6%) | 17(68.0%) | 0.02* |
| Type 2 diabetes (n (%)) | 6 (9.4%) | 7 (28.0%) | 0.03* |
| Chronic liver disease (n (%)) | 9 (14.1%) | 6 (24.0%) | 0.26 |
| Chronic renal dysfunction (n (%)) | 8 (12.5%) | 9 (36.0%) | 0.01* |
| Cerebral infarction (n (%)) | 14 (21.9%) | 6 (24.0%) | 0.83 |
| Pathogenic bacteria | |||
| Acinetobacter baumannii (n (%)) | 41 (64.1%) | 18 (72.0%) | 0.48 |
| Klebsiella pneumoniae (n (%)) | 23 (35.9%) | 8 (32.0%) | 0.73 |
| Pseudomonas aeruginosa (n (%)) | 17 (26.6%) | 5 (20.0%) | 0.52 |
| Escherichia coli (n (%)) | 4 (4.7%) | 1 (4.0%) | 1.00 |
| Stenotrophomonas maltophilia (n (%)) | 6 (9.4%) | 1 (4.0%) | 0.68 |
| Candida (n (%)) | 20 (31.3%) | 3 (12.0%) | 0.11 |
| PMB treatment | |||
| PMB daily dosage (mg) (median (IQR)) | 100 (100–100) | 100 (100–100) | 0.90 |
| PMB total dosage (mg) (median (IQR)) | 1050 (780–1400) | 900 (650–1100) | 0.23 |
| PMB duration (days) (median (IQR)) | 11 (7–13) | 9 (8–11) | 0.32 |
| PMB monotherapy (n (%)) | 9 (14.1%) | 5 (20.0%) | 0.71 |
| Concomitant with nephrotoxic drugsb (n (%)) | 24 (37.5%) | 11 (44.0%) | 0.57 |
| Concomitant with β-lactam (n (%)) | 33 (51.6%) | 12 (48.0%) | 0.76 |
| Carbapenem (n (%)) | 47 (73.4%) | 15 (60.0%) | 0.22 |
| Tigecycline (n (%)) | 16 (25.0%) | 7 (28.0%) | 0.77 |
| Laboratory examination (before PMB treatment) | |||
| WBC (× 109/L) (median (IQR)) | 10.2 (6.7–13.9) | 12.8 (7.6–14.2) | 0.29 |
| Percentage of neutrophils (%) | 80.0 (70.8–87.1) | 81.0 (75.4–85.0) | 0.06 |
| Albumin (g/L) (median (IQR)) | 32.7 (30.2–36.3) | 32.1 (28.5–35.0) | 0.32 |
| Serum creatinine (µmol/L) (median (IQR)) | 92.9 (60.9–127.1) | 73.9 (51.9–102.5) | 0.13 |
| BUN (mmol/L) (median (IQR)) | 10.8 (7.3–14.5) | 18.7 (12.6–23.8) | 0.01* |
| Procalcitonin (ng/ml) (median (IQR)) | 1.3 (0.2–5.2) | 1.3 (0.7–3.7) | 0.89 |
| HCRP (mg/L) (median (IQR)) | 92.0 (42.5–119.0) | 101 (81.7–133) | 0.25 |
| CrCL (ml/min) | 53.6 (36.7–81.7) | 77.4 (34.1–104.3) | 0.15 |
APACHE Acute Physiology and Chronic Health Evaluation, ACCI age-adjusted Charlson comorbidity index, PMB polymyxin B, WBC white blood cell count, BUN blood urea nitrogen, HCRP high-sensitivity C-reactive protein, CrCL creatinine clearance, IQR interquartile range
aFrom the clinic diagnosis of electronic medical records
bIncludes vancomycin, sulfamethoxazole, gentamicin, amikacin, ciprofloxacin and levofloxacin in this study
*Denotes p < 0.05, differences between the AKI group and the non-AKI group were tested for statistical significance
These 12 variables (p < 0.05) were used to develop a multivariate logistic regression model. Finally, Cmax (B1) (AOR = 1.68, 95% CI 1.08–2.62, p = 0.023), the baseline BUN level (AOR = 1.08, 95% CI 1.01–1.16, p = 0.039) and hypertension (AOR = 3.73, 95% CI 1.21–11.54, p = 0.022) were included in the final model (Table 4). The predicted probability of AKI was calculated using the logistic function probability = 1 / (1 + e−β), where β represents the summation of the model constant and the covariates. Therefore, the equation of the final obtained prediction model was
| 3 |
Table 4.
Multivariable logistic regression model of AKI
| Risk factors | Adjusted OR | 95% CI | p value |
|---|---|---|---|
| Cmax (B1) | 1.68 | 1.08–2.62 | 0.023* |
| Baseline BUN | 1.08 | 1.01–1.16 | 0.039* |
| Hypertension | 3.73 | 1.21–11.54 | 0.022* |
BUN blood Urea nitrogen, Cmax (B1) peak concentration of polymyxin B
*Denotes p < 0.05, differences between the AKI group and the non-AKI group were tested for statistical significance
The Hosmer–Lemeshow test showed that the model had favourable fitness (p = 0.60). The VIF values of the predictor variables in this model were all less than 1.2, indicating no multicollinearity. A ROC curve was used to calculate the discriminatory power of the model (Fig. 4). Figure 5 depicts that the model shows better discriminatory power for PMB-induced AKI than other single factors. The AUCs were as follows: model (0.799) > baseline BUN level (0.683) > Cmax (B1) (0.680) > Cmax (B) (0.674) > Cmin (B) (0.666).
Fig. 4.
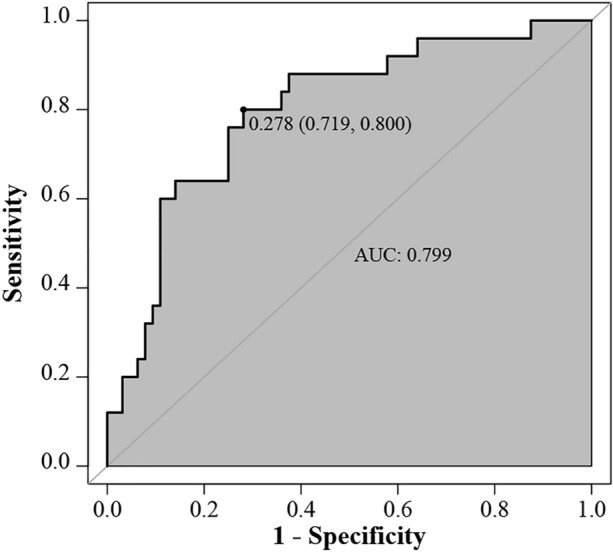
ROC of the final multivariate logistic regression model
Fig. 5.
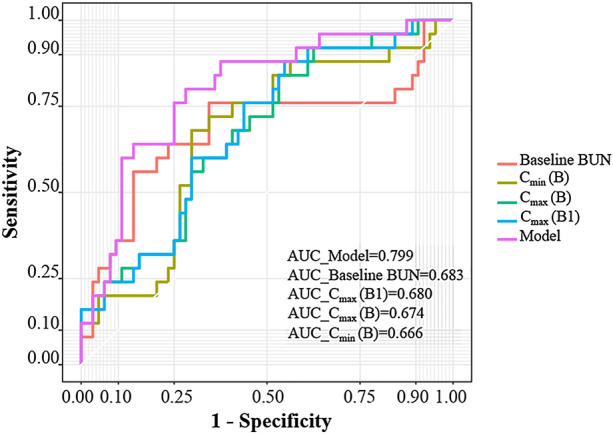
Comparison of the ROCs of the final model and other single factors. BUN represents blood urea nitrogen. Cmax (B) represents the peak plasma concentrations of PMB. Cmin (B) represents the trough plasma concentrations of PMB. Cmax (B1) represents the peak plasma concentrations of PMB1. Model indicates the final multivariate logistic regression model
In addition, a univariate logistic regression was developed to determine the correlation between the Cmax (B1) and the probability of developing AKI (Fig. 6). The results showed that the Cmax (B1) (OR = 1.68, 95% CI 1.15–2.47, p = 0.008) was a significant predictor of AKI and that the predicted probability of AKI was higher than 50% when the Cmax (B1) was 5.23 μg/ml or higher according to Eq. (4).
| 4 |
Fig. 6.
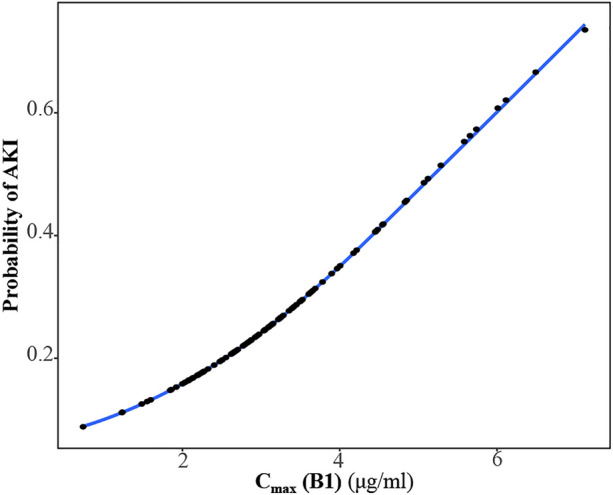
Univariate logistic regression model for the risk probability of acute kidney injury (AKI). Cmax (B1) represents the peak plasma concentrations of PMB1
In addition, the relationship between the Cmax (B1) and the severity of AKI was assessed. Patients with AKI were stratified according to the RIFLE criteria (Table 5). Only one patient was classified as “failure”. To compare the difference of Cmax (B1) among groups of “risk” and “injury”, a Student’s t test was performed. The result showed there was no significant difference between the “risk” group and the “injury” group (p = 0.52), indicating that the Cmax (B1) was not associated with the severity of AKI in this study.
Table 5.
Relationship between patients and severity of AKI
| Category criteria | Number of patients (%) | Cmax (B1) (mean ± SD) |
|---|---|---|
| Risk (R) | 16 (18.0%) | 3.65 ± 1.26 |
| Injury (I) | 8 (9.0%) | 4.04 ± 1.59 |
| Failure (F) | 1 (1.1%) | 6.17 |
| Loss (L) | 0 | NA |
NA not available
Discussion
The present study demonstrated that the Cmax (B1), hypertension and baseline BUN levels were independent risk factors for AKI development during PMB treatment in critically ill patients. To our knowledge, this is the first report showing that Cmax (B1) is related to PMB-induced nephrotoxicity. Furthermore, we were the first to explore the correlation between the free concentration of PMB (PMB1, PMB2) and PMB-induced nephrotoxicity in critically ill patients, and we found that the free Cmax (B) and free Cmax (B1) in the AKI group were significantly higher than those in the non-AKI group.
The ultrafiltration tube method and equilibrium dialysis are the two commonly used methods for determining free concentrations. The results of the ultrafiltration tube method are affected by multiple factors, such as temperature, centrifugal force, pH and the ultrafiltration membrane [37, 38]. Equilibrium dialysis is based on drug diffusion across a semipermeable membrane that separates the sample to be investigated from a buffer solution and is still considered the gold standard for monitoring free drug concentrations [39]. Equilibrium dialysis was adopted in our study, and the results showed that the median protein binding rates of PMB, PMB1 and PMB2 were 91.7% (range 65.7–96.0%), 95.2% (range 61.7–98.2%) and 82.7% (range 59.2–95.5%), respectively. It was reported that the plasma protein binding rate of PMB in eight critically ill patients (range 78.5–92.4%) was higher than that in healthy humans (55.9% ± 4.7%) [26], which is consistent with our study. However, Sandri and colleagues found that the median protein binding rate in 23 critically ill patients was 58% (36–74%) [25], which is quite different from that observed in our study. Special pathophysiological and iatrogenic factors in critically ill patients can affect protein concentrations by altering synthesis and catabolism or promoting protein movement from the plasma to extravascular sites [40]. Alterations in protein levels vary significantly among individuals and might result in variability in the free concentration of PMB in critically ill patients. Total concentrations and published protein binding values are usually used to predict the unbound drug concentration in clinical practice in general. However, the measured total concentration is not an adequate surrogate for the free concentration in some antibiotics studies [41, 42]. In the present study, a positive correlation between the total and free concentrations was found (Fig. 2). We attempted to use the clinical data of patients to develop a multivariate linear regression equation (stepwise regression) to predict the free concentration. However, these variables were not included in the final equation. Furthermore, although the free Cmax (B1) was not independently associated with AKI in this study, the significant difference between the two groups in the univariate analysis (Table 3) suggests that the free Cmax (B) and Cmax (B1) may also be associated with AKI. Our results indicated that monitoring of free drug concentrations should be considered in the management of critically ill patients administered PMB.
In the present study, we preliminarily analysed the in vivo PMB1/PMB2 ratio. The PMB1/PMB2 Cmin ratio and PMB1/PMB2 Cmax ratio in patients ranged from 1.53 to 6.76 and from 1.48 to 4.44, respectively. The ratio of PMB1/PMB2 was less than 80% of that in the preparation in most patients and exhibited noticeable individual differences (Fig. 3). The PMB1/PMB2 ratio in the plasma was related not only to the ratio in the preparation but also to PK parameters such as volume of distribution (Vd) and t1/2. Reports on the PK parameters of PMB1 and PMB2 in patients are limited. Wang et al. reported the PK parameters of PMB1 and PMB2 in 15 patients, and a significant interindividual difference in Vd was observed [16]. Vd is influenced by plasma protein binding [43]. For PMB, there were significant individual differences in the plasma protein binding rate among patients [25, 26]. We speculated that interindividual differences in the PK parameters of PMB1 and PMB2 in different populations could be a reasonable explanation for the discrepancy. Considering that Cmax (B1) was independently related to AKI, TDM of PMB1 and PMB2 should be warranted.
The clinical risk factors for PMB-induced nephrotoxicity reported in previous studies include age, baseline SCr levels, body mass index (BMI), concomitant use of medications such as vasoactive drugs and vancomycin, and the infection site [18–22]. Notably, the identification of risk factors had yielded mixed results from different studies. For example, Mendes et al. [19] and Han et al. [22] found that the baseline SCr level is a risk factor for PMB-induced nephrotoxicity. However, three other reports [21, 44, 45] involving critically ill patients indicated that the baseline SCr level was not associated with PMB-induced nephrotoxicity. This difference might be related to differences in the severity of illness in the patients included in the different studies. For instance, the in-hospital mortality rate (61.4%) of patients in the study by Mendes et al. [19] was higher than that (23–42%) of patients in the other three studies [21, 44, 45] that reported that the SCr level is not associated with PMB-induced nephrotoxicity. CrCL is calculated according to age, weight and sex and may be a more accurate index for evaluating renal function than the SCr level. However, it was not identified as a risk factor in our research or two other studies [20, 43]. The BUN level is another indicator of renal function and was found to be associated with kidney injury induced by other antibiotics, such as vancomycin-related nephrotoxicity [46]. It should be noted that BUN may be too nonspecific for kidney injury [47], and elevated BUN may be caused by non-renal factors such as protein intake, catabolic state, upper gastrointestinal bleeding, volume status and therapy with high-dose steroids [48–51]. In the present study, although BUN eventually entered a multivariate logistic regression model, the ROC curves (Fig. 5) showed the predictive power of BUN is lower than that of the multivariate logistic regression model, which indicated that the predictive power of BUN may be limited when it was used as a single indicator for predicting drug-related AKI.
The kidney is an essential organ for blood pressure regulation and one of the main target organs damaged by hypertension [52]. A previous study has reported that hypertension may be a potential risk factor for colistin-induced nephrotoxicity [53]. In our study, hypertension was identified as another risk factor for PMB-induced nephrotoxicity. The mechanism of this damage may be related to oxidative stress and haemodynamics [54]. In addition, previous studies have found that kidney injury molecule 1 (KIM-1), a factor associated with diabetic nephropathy, may be associated with colistin-induced nephrotoxicity [55–57]. In our study, we found that the incidence rates of type 2 diabetes and chronic renal dysfunction were different between the two groups in the univariate analysis (Table 2). Although the difference became weak in the multivariate analysis, it did not increase the discriminative ability of the final logistic regression model. The meta-analysis [58] reported that underlying diabetes mellitus was a risk factor for polymyxin-induced nephrotoxicity. Although this meta-analysis focused on polymyxins, and polymyxin B and colistin were not analysed separately, the possible risk of AKI in patients with diabetes should be considered.
In our study, the onset of nephrotoxicity in patients ranged from day 3 to day 12. Four patients (16%) experienced nephrotoxicity on day 3. Considering that early PMB-induced nephrotoxicity on day 3 is a predictive factor for later nephrotoxicity [44], early monitoring of renal function during PMB treatment is necessary.
The area under the ROC curve (AUC) of the final multivariate logistic regression model reached 0.799, which is similar to the AUC (0.813) of Han et al.’s combined predictor (Cmin (B) and baseline SCr level) [22]. Sorlí and colleagues also found that trough plasma level is an independent risk factor for colistin-induced AKI [33]. The optimal cut-off trough concentrations for predicting PMB-related and colistin-related nephrotoxicity in these two studies were 3.55 mg/L and 3.33 mg/L, respectively. However, Cmin (B) was not included in the final logistic model in our study. This may have been in part related to the relatively low Cmin (B) in patients included in our study. Considering that the Cmin (B) showed a significant difference in univariate analysis (Table 3), we attempted to develop a univariate logistic regression model to observe the correlation between Cmin (B) and the probability of AKI development. The results showed that the predicted risk of AKI reached 50% when the Cmin (B) was 3.63 mg/L or higher. However, when the Cmin (B) was used alone to distinguish patients with AKI, the area under the ROC curve was normal (0.666). The predictive value of the Cmin (B) for AKI should also be considered in future research. The multivariate model (Eq. 3) is convenient for calculating the predicted risk probability of AKI according to the Cmax (B1), presence of hypertension and baseline BUN levels. When data such as baseline BUN levels are missing, Eq. 4 can also provide a preliminary method for predicting AKI risk. Because samples of Cmax (B) were collected 10 min after completion of PMB infusion and Cmin (B) was obtained immediately before infusion in our study, we contend that Cmax (B) and Cmax (B1) may be helpful for the early prediction of PMB-induced AKI in critically ill patients. Overall, our model might provide a helpful method for early identification of patients with a high risk of AKI and formulate corresponding intervention strategies.
The study had several limitations. First, this was a retrospective study. We could not analyse other possible risk factors, such as more sensitive indicators reflecting early renal function injury. Second, we were unable to determine the PK parameters in patients to assess the relationship between the AUC and AKI risk of PMB, and the difference in PK parameters between PMB1 and PMB2 was not further assessed. Third, although the enrolled patients came from three medical centres, the sample size was relatively small, limiting the generalizability of our results. Fourth, although the TDM of PMB was recommended by the guideline, the quantification method of PMB1 has not yet been performed in numerous laboratories, which might limit the application of our findings in clinical routine practice.
Conclusions
The present work identified the Cmax (B1), baseline BUN levels, and hypertension as independent risk factors for PMB-induced AKI. The ratio of PMB1/PMB2 may decrease after PMB preparation entered into the body. Both the ROC curve and logistic regression model showed that Cmax (B1) was a good predictor for the probability of PMB-induced AKI. Early therapeutic drug monitoring of PMB should be considered in critically ill patients. Compared with Cmin (B), Cmax (B) and Cmax (B1) may be helpful for the early prediction of PMB-induced AKI in critically ill patients.
Acknowledgements
Funding
This study, and the journal’s Rapid Service Fees, was supported by the Project of Health Commission of Hunan Province, China [No. 20201375] and the Hunan Provincial Science and Technology Department Foundation, China [No. 2020SK52901, No. 2016SK4008].
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed to the study conception and design. The manuscript was written by Yang Deng and Jun-Yuan Gu; the research was designed by Xin Li and Yang Deng; the research was performed by Yang Deng, You Li, Si-Wei Guo and Bing Xu; data analysis was performed by Yang Deng, Bi-Kui Zhang, Ying Li and Hai-Ying Huang; new reagents/analytical tools was contributed by Jun-Yuan Gu, Huan Tong and Gui-Ying Xiao. All authors approved the final manuscript.
Disclosures
Yang Deng, Jun-yuan Gu, Xin Li, Huan Tong, Si-wei Guo, Bing Xu, You Li, Bi-kui Zhang, Ying Li, Hai-ying Huang, Gui-ying Xiao have nothing to disclose.
Compliance with Ethics Guidelines
As the central laboratory of the rational use of anti-infection agent’s technology demonstration base, the TDM laboratory of the Third Hospital of Changsha also undertook the TDM of polymyxin B for the other two medical institutions, and the Third Hospital of Changsha was the lead institution of this retrospective study. According to the “Deepening the Reform of the Review and Approval System and Encouraging the Innovation of Drugs and Medical Devices” issued (on October 8, 2017) by the State Council of China [59], this study was only approved by the Ethics Research Committee of the Third Hospital of Changsha (No. CS3-KY-2021EC-008), and the requirement for written informed consent was waived. Ethics approval and the informed consent waiver were also accepted by the other two hospitals.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yang Deng and Jun-yuan Gu contributed equally to this work.
References
- 1.Evans ME, Feola DJ, Rapp RP. Polymyxin B sulfate and colistin: old antibiotics for emerging multiresistant gram-negative bacteria. Ann Pharmacother. 1999;33(9):960–967. doi: 10.1345/aph.18426. [DOI] [PubMed] [Google Scholar]
- 2.Lieberson AD, Winter LW, Behnke RH, Martin RR. Extensive pseudomonal pneumonia ultimately responding to polymyxin therapy. Am Rev Respir Dis. 1969;100(4):558–564. doi: 10.1164/arrd.1969.100.4.558. [DOI] [PubMed] [Google Scholar]
- 3.Landman D, Georgescu C, Martin DA, Quale J. Polymyxins revisited. Clin Microbiol Rev. 2008;21(3):449–465. doi: 10.1128/cmr.00006-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Falagas ME, Kasiakou SK. Colistin: the revival of polymyxins for the management of multidrug-resistant gram-negative bacterial infections. Clin Infect Dis. 2005;40(9):1333–1341. doi: 10.1086/429323. [DOI] [PubMed] [Google Scholar]
- 5.Cai Y, Lee W, Kwa AL. Polymyxin B versus colistin: an update. Expert Rev Anti Infect Ther. 2015;13(12):1481–1497. doi: 10.1586/14787210.2015.1093933. [DOI] [PubMed] [Google Scholar]
- 6.Gales AC, Castanheira M, Jones RN, Sader HS. Antimicrobial resistance among Gram-negative bacilli isolated from Latin America: results from SENTRY Antimicrobial Surveillance Program (Latin America, 2008–2010) Diagn Microbiol Infect Dis. 2012;73(4):354–360. doi: 10.1016/j.diagmicrobio.2012.04.007. [DOI] [PubMed] [Google Scholar]
- 7.Elias LS, Konzen D, Krebs JM, Zavascki AP. The impact of polymyxin B dosage on in-hospital mortality of patients treated with this antibiotic. J Antimicrob Chemother. 2010;65(10):2231–2237. doi: 10.1093/jac/dkq285. [DOI] [PubMed] [Google Scholar]
- 8.Mostardeiro MM, Pereira CAP, Marra AR, Pestana JOM, Camargo LFA. Nephrotoxicity and efficacy assessment of polymyxin use in 92 transplant patients. Antimicrob Agents Chemother. 2013;57(3):1442–1446. doi: 10.1128/aac.01329-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Oliveira MS, Prado GVB, Costa SF, Grinbaum RS, Levin AS. Polymyxin B and colistimethate are comparable as to efficacy and renal toxicity. Diagn Microbiol Infect Dis. 2009;65(4):431–434. doi: 10.1016/j.diagmicrobio.2009.07.018. [DOI] [PubMed] [Google Scholar]
- 10.Akajagbor DS, Wilson SL, Shere-Wolfe KD, Dakum P, Charurat ME, Gilliam BL. Higher incidence of acute kidney injury with intravenous colistimethate sodium compared with polymyxin B in critically ill patients at a tertiary care medical center. Clin Infect Dis. 2013;57(9):1300–1303. doi: 10.1093/cid/cit453. [DOI] [PubMed] [Google Scholar]
- 11.Esaian D, Dubrovskaya Y, Phillips M, Papadopoulos J. Effectiveness and tolerability of a polymyxin B dosing protocol. Ann Pharmacother. 2012;46(3):455–456. doi: 10.1345/aph.1Q294. [DOI] [PubMed] [Google Scholar]
- 12.Wagenlehner F, Lucenteforte E, Pea F, et al. Systematic review on estimated rates of nephrotoxicity and neurotoxicity in patients treated with polymyxins. Clin Microbiol Infect. 2021;27(5):671–686. doi: 10.1016/j.cmi.2020.12.009. [DOI] [PubMed] [Google Scholar]
- 13.Vazin A, Karimzadeh I, Zand A, Hatami-Mazinani N, Firouzabadi D. Evaluating adherence of health-care team to standard guideline of colistin use at intensive care units of a referral hospital in Shiraz, southwest of Iran. Adv Pharm Bull. 2017;7(3):391–397. doi: 10.15171/apb.2017.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Omrani AS, Alfahad WA, Shoukri MM, et al. High dose intravenous colistin methanesulfonate therapy is associated with high rates of nephrotoxicity; a prospective cohort study from Saudi Arabia. Ann Clin Microbiol Antimicrob. 2015 doi: 10.1186/s12941-015-0062-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Azad MAK, Finnin BA, Poudyal A, et al. Polymyxin B induces apoptosis in kidney proximal tubular cells. Antimicrob Agents Chemother. 2013;57(9):4329–4335. doi: 10.1128/aac.02587-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang P, Zhang Q, Qin Z, et al. A simple and robust liquid chromatography with tandem mass spectrometry analytical method for therapeutic drug monitoring of plasma and cerebrospinal fluid polymyxin B1 and B2. Ther Drug Monit. 2020;42(5):716–723. doi: 10.1097/ftd.0000000000000754. [DOI] [PubMed] [Google Scholar]
- 17.Liu X, Chen Y, Yang H, et al. Acute toxicity is a dose-limiting factor for intravenous polymyxin B: a safety and pharmacokinetic study in healthy Chinese subjects. J Infect. 2021;82(2):207–215. doi: 10.1016/j.jinf.2021.01.006. [DOI] [PubMed] [Google Scholar]
- 18.Rigatto MH, Behle TF, Falci DR, et al. Risk factors for acute kidney injury (AKI) in patients treated with polymyxin B and influence of AKI on mortality: a multicentre prospective cohort study. J Antimicrob Chemother. 2015;70(5):1552–1557. doi: 10.1093/jac/dku561. [DOI] [PubMed] [Google Scholar]
- 19.Mendes CAC, Cordeiro JA, Burdmann EA. Prevalence and risk factors for acute kidney injury associated with parenteral polymyxin B use. Ann Pharmacother. 2009;43(12):1948–1955. doi: 10.1345/aph.1M277. [DOI] [PubMed] [Google Scholar]
- 20.Dubrovskaya Y, Prasad N, Lee Y, Esaian D, Figueroa DA, Tam VH. Risk factors for nephrotoxicity onset associated with polymyxin B therapy. J Antimicrob Chemother. 2015;70(6):1903–1907. doi: 10.1093/jac/dkv014. [DOI] [PubMed] [Google Scholar]
- 21.Kubin CJ, Ellman TM, Phadke V, Haynes LJ, Calfee DP, Yin MT. Incidence and predictors of acute kidney injury associated with intravenous polymyxin B therapy. J Infect. 2012;65(1):80–87. doi: 10.1016/j.jinf.2012.01.015. [DOI] [PubMed] [Google Scholar]
- 22.Han L, Xu F-M, Zhang X-S, et al. Trough polymyxin B plasma concentration is an independent risk factor for its nephrotoxicity. Br J Clin Pharmacol. 2021 doi: 10.1111/bcp.15061. [DOI] [PubMed] [Google Scholar]
- 23.Tsuji BT, Pogue JM, Zavascki AP, et al. International Consensus Guidelines for the Optimal Use of the Polymyxins Endorsed by the American College of Clinical Pharmacy (ACCP), European Society of Clinical Microbiology and Infectious Diseases (ESCMID), Infectious Diseases Society of America (IDSA), International Society for Anti-infective Pharmacology (ISAP), Society of Critical Care Medicine (SCCM), and Society of Infectious Diseases Pharmacists (SIDP) Pharmacotherapy. 2019;39(1):10–39. doi: 10.1002/phar.2209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang P, Zhang Q, Zhu Z, et al. Comparing the population pharmacokinetics of and acute kidney injury due to polymyxin B in chinese patients with or without renal insufficiency. Antimicrob Agents Chemother. 2021 doi: 10.1128/aac.01900-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sandri AM, Landersdorfer CB, Jacob J, et al. Population pharmacokinetics of intravenous polymyxin B in critically ill patients: implications for selection of dosage regimens. Clin Infect Dis. 2013;57(4):524–531. doi: 10.1093/cid/cit334. [DOI] [PubMed] [Google Scholar]
- 26.Zavascki AP, Goldani LZ, Cao G, et al. Pharmacokinetics of intravenous polymyxin B in critically ill patients. Clin Infect Dis. 2008;47(10):1298–1304. doi: 10.1086/592577. [DOI] [PubMed] [Google Scholar]
- 27.Dasgupta A. Usefulness of monitoring free (unbound) concentrations of therapeutic drugs in patient management. Clin Chim Acta. 2007;377(1–2):1–13. doi: 10.1016/j.cca.2006.08.026. [DOI] [PubMed] [Google Scholar]
- 28.Janmahasatian S, Duffull SB, Ash S, Ward LC, Byrne NM, Green B. Quantification of lean bodyweight. Clin Pharmacokinet. 2005;44(10):1051–1065. doi: 10.2165/00003088-200544100-00004. [DOI] [PubMed] [Google Scholar]
- 29.Koppie TM, Serio AM, Vora K, et al. Age adjusted Charlson comorbidity score is associated with treatment decisions and clinical outcomes for patients undergoing radical cystectomy for bladder cancer. J Urol. 2007;177(4):499. doi: 10.1016/s0022-5347(18)31713-0. [DOI] [PubMed] [Google Scholar]
- 30.Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–1251. doi: 10.1016/0895-4356(94)90129-5. [DOI] [PubMed] [Google Scholar]
- 31.Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med. 1985;13(10):818–829. doi: 10.1097/00003246-198510000-00009. [DOI] [PubMed] [Google Scholar]
- 32.Olson G, Davis AM. Diagnosis and treatment of adults with community-acquired pneumonia. JAMA. 2020;323(9):885–886. doi: 10.1001/jama.2019.21118. [DOI] [PubMed] [Google Scholar]
- 33.Sorli L, Luque S, Grau S, et al. Trough colistin plasma level is an independent risk factor for nephrotoxicity: a prospective observational cohort study. Bmc Infect Dis. 2013 doi: 10.1186/1471-2334-13-380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Deng YXB, Li X, et al. Exploration of LC-MS/MS method for therapeutic drug monitoring and clinical application of polymyxin B. Chin Pharm J. 2021;56(15):1249–1254. [Google Scholar]
- 35.Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Acute Dialysis Quality Initiative Workgroup Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):204–212. doi: 10.1186/cc2872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hayashi T, Boyko EJ, Leonetti DL, et al. Visceral adiposity is an independent predictor of incident hypertension in Japanese Americans. Ann Intern Med. 2004;140(12):992–1000. doi: 10.7326/0003-4819-140-12-200406150-00008. [DOI] [PubMed] [Google Scholar]
- 37.Li X, Wang F, Xu B, et al. Determination of the free and total concentrations of vancomycin by two-dimensional liquid chromatography and its application in elderly patients. J Chromatogr B. 2014;969:181–189. doi: 10.1016/j.jchromb.2014.08.002. [DOI] [PubMed] [Google Scholar]
- 38.Kees MG, Wicha SG, Seefeld A, Kees F, Kloft C. Unbound fraction of vancomycin in intensive care unit patients. J Clin Pharmacol. 2014;54(3):318–323. doi: 10.1002/jcph.175. [DOI] [PubMed] [Google Scholar]
- 39.Musteata FM. Monitoring free drug concentrations: challenges. Bioanalysis. 2011;3(15):1753–1768. doi: 10.4155/bio.11.187. [DOI] [PubMed] [Google Scholar]
- 40.Gonzalez D, Conrado DJ, Theuretzbacher U, Derendorf H. The effect of critical illness on drug distribution. Curr Pharm Biotechnol. 2011;12(12):2030–2036. doi: 10.2174/138920111798808211. [DOI] [PubMed] [Google Scholar]
- 41.Butterfield JM, Patel N, Pai MP, Rosano TG, Drusano GL, Lodise TP. Refining vancomycin protein binding estimates: identification of clinical factors that influence protein binding. Antimicrob Agents Chemother. 2011;55(9):4277–4282. doi: 10.1128/aac.01674-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wong G, Briscoe S, Adnan S, et al. Protein binding of beta-lactam antibiotics in critically ill patients: can we successfully predict unbound concentrations? Antimicrob Agents Chemother. 2013;57(12):6165–6170. doi: 10.1128/aac.00951-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Toutain PL, Bousquet-Melou A. Volumes of distribution. J Vet Pharmacol Ther. 2004;27(6):441–453. doi: 10.1111/j.1365-2885.2004.00602.x. [DOI] [PubMed] [Google Scholar]
- 44.Zeng H, Zeng Z, Kong X, et al. Effectiveness and nephrotoxicity of intravenous polymyxin B in Chinese patients with MDR and XDR nosocomial pneumonia. Front Pharmacol. 2021 doi: 10.3389/fphar.2020.579069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zhang J, Hu Y, Shen X, Zhu X, Chen J, Dai H. Risk factors for nephrotoxicity associated with polymyxin B therapy in Chinese patients. Int J Clin Pharm. 2021;43(4):1109–1115. doi: 10.1007/s11096-020-01225-8. [DOI] [PubMed] [Google Scholar]
- 46.Wang L, Yuan Q, Tan M, et al. Evaluation of efficacy and nephrotoxicity during vancomycin therapy: a retrospective study in China. Exp Ther Med. 2019;17(3):2389–2396. doi: 10.3892/etm.2019.7188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Waikar SS, Bonventre JV. Can we rely on blood urea nitrogen as a biomarker to determine when to initiate dialysis? Clin J Am Soc Nephrol. 2006;1(5):903–904. doi: 10.2215/cjn.02560706. [DOI] [PubMed] [Google Scholar]
- 48.Walser M. Determinants of ureagenesis, with particular reference to renal failure. Kidney Int. 1980;17(6):709–721. doi: 10.1038/ki.1980.84. [DOI] [PubMed] [Google Scholar]
- 49.Griffin BR, Faubel S, Edelstein CL. Biomarkers of drug-induced kidney toxicity. Ther Drug Monit. 2019;41(2):213–226. doi: 10.1097/ftd.0000000000000589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Luke RG. Uremia and the BUN. N Engl J Med. 1981;305(20):1213–1215. doi: 10.1056/nejm198111123052010. [DOI] [PubMed] [Google Scholar]
- 51.Suzuki G, Ichibayashi R, Yamamoto S, et al. Effect of high-protein nutrition in critically ill patients: a retrospective cohort study. Clin Nutr Espen. 2020;38:111–117. doi: 10.1016/j.clnesp.2020.05.022. [DOI] [PubMed] [Google Scholar]
- 52.Batchu SN, Dugbartey GJ, Wadosky KM, et al. Innate immune cells are regulated by Axl in hypertensive kidney. Am J Pathol. 2018;188(8):1794–1806. doi: 10.1016/j.ajpath.2018.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Doshi NM, Mount KL, Murphy CV. Nephrotoxicity associated with intravenous colistin in critically ill patients. Pharmacotherapy. 2011;31(12):1257–1264. doi: 10.1592/phco.31.12.1257. [DOI] [PubMed] [Google Scholar]
- 54.Middlemiss JE, Wilkinson IB, McEniery CM. Mechanisms underlying elevated SBP differ with adiposity in young adults: the Enigma study reply. J Hypertens. 2016;34(7):1443–1444. doi: 10.1097/hjh.0000000000000938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pogue JM, Lee J, Marchaim D, et al. Incidence of and risk factors for colistin-associated nephrotoxicity in a large academic health system. Clin Infect Dis. 2011;53(9):879–884. doi: 10.1093/cid/cir611. [DOI] [PubMed] [Google Scholar]
- 56.Vazin A, Malek M, Karimzadeh I. Evaluation of colistin nephrotoxicity and urinary level of kidney injury molecule-1 in hospitalized adult ICU patients. J Renal Inj Prev. 2020 doi: 10.34172/jrip.2020.13. [DOI] [Google Scholar]
- 57.Mori Y, Ajay AK, Chang J-H, et al. KIM-1 mediates fatty acid uptake by renal tubular cells to promote progressive diabetic kidney disease. Cell Metab. 2021;33(5):1042. doi: 10.1016/j.cmet.2021.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sisay M, Hagos B, Edessa D, Tadiwos Y, Mekuria AN. Polymyxin-induced nephrotoxicity and its predictors: a systematic review and meta-analysis of studies conducted using RIFLE criteria of acute kidney injury. Pharmacol Res. 2021 doi: 10.1016/j.phrs.2020.105328. [DOI] [PubMed] [Google Scholar]
- 59.Website of the Central Government of the People’s Republic of China. The General Office of the State Council of China issued a policy regarding Deepening the Reform of the Review and Approval System and Encouraging the Innovation of Drugs and Medical Devices. 2017. http://www.gov.cn/zhengce/2017-10/08/content_5230105.htm. Accessed 08 Oct 2017.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.