Abstract
Metabolic demands of skeletal muscle are substantial and are characterized normally as highly flexible and with a large dynamic range. Skeletal muscle composition (e.g., fiber type and mitochondrial content) and metabolism (e.g., capacity to switch between fatty acid and glucose substrates) are altered in obesity, with some changes proceeding and some following the development of the disease. Nonetheless, there are marked interindividual differences in skeletal muscle composition and metabolism in obesity, some of which have been associated with obesity risk and weight loss capacity. In this review, we discuss related molecular mechanisms and how current and novel treatment strategies may enhance weight loss capacity, particularly in diet-resistant obesity.
Keywords: metabolic disorders, metabolic regulation, mitochondria, oxidative phosphorylation, skeletal muscle
Introduction
According to the World Health Organization, more than 1 billion adults are overweight and an additional 650 million adults are living with obesity. The global prevalence of obesity has tripled since the 1970s, with dramatic increases in the rates of childhood obesity [1–3]. Obesity is a major global public health concern as it is a risk factor for many cardiometabolic diseases including coronary heart disease, hypertension, Type 2 diabetes mellitus (T2D), and non-alcoholic fatty liver disease (NAFLD) [1,4–6]. Obesity is also associated with increased risk for many types of cancer, musculoskeletal disorders (e.g., osteoarthritis), and all-cause mortality [1,4,5,7–11]. The high healthcare costs associated with treating comorbidities associated with obesity demonstrate the critical need for weight loss strategies to manage this disease [12–14]. Achieving a modest weight loss of ≥5% has been shown to significantly modify risk factors for cardiometabolic disease [15]. Specifically, adjusted regression models in a retrospective analysis of electronic health records from patients with a history of obesity who had either maintained their current weight (i.e. not lost weight), lost >5% of body weight and subsequently regained weight, or lost and maintained >5% of their body weight, revealed that achieving a weight loss of 5–10% reduces T2D risk and lowers HbA1c, and that weight loss of 10–15% improves blood pressure and reduces plasma LDL and increases HDL [16]. Weight loss of >20% is generally observed with bariatric surgery and is further associated with decreased risk of cancer and increased life expectancy by up to 3 years [17].
Obesity is a complex, multifactorial disease involving the excess deposition of body fat, predominantly stored not only in adipose tissues but also ectopically in tissues such as liver and skeletal muscle. The expansion of adipose tissue drives increases in body mass and body mass index (BMI). Obesity is most commonly identified as a BMI exceeding 30 kg/m², while overweight is identified as a BMI greater than 25 kg/m² but less than 30 kg/m². The development of obesity is ultimately driven by an imbalance between dietary energy intake and energy expenditure. Thus, weight loss interventions require a negative energy balance, through reduced dietary energy intake, and/or increased energy expenditure (e.g., through exercise).
Individuals with obesity often attempt multiple treatment approaches before seeking help from medical professionals; they frequently report using self-guided caloric restriction or popularized diet strategies, exercise, commercial weight-loss programs, and over-the-counter dietary supplements [18–20]. Once in the care of medical and allied health professionals, the focus is on behavioral strategies and pharmacotherapies. Behavioral programs frequently employ very low-calorie total meal replacements in combination with behavioral counseling. The statistics on sustained weight loss success with these approaches have been discouraging [21], and the hope is that more personalized approaches and novel pharmacotherapies will significantly improve weight loss outcomes. While greater degrees of weight loss and sustained weight loss are associated with bariatric surgeries, these approaches are invasive, not universally available, and typically are only for those with severe obesity, or obesity-associated comorbidities [22]. Long-term mortality significantly improves with surgical intervention [23]; however, there is still a significant risk of complications associated with bariatric surgery, including a 0.7% risk of mortality [24].
The objective of this review is to discuss the role of mitochondrial health and energy expenditure in the context of variability in weight loss success, and more specifically, factors associated with skeletal muscle metabolism that are associated with enhanced or perturbed weight loss success. While exercise is a major factor, and will be addressed here, other factors that augment energy expenditure of skeletal muscle will also be discussed.
Variability in weight loss success
Many studies have shown that there is substantial interindividual variability in weight loss response to traditional interventions [25,26], even after controlling for factors such as diet adherence, age, biological sex, exercise, and medications [25,27]. Max Wishnofsky first proposed the highly propagated idea that weight loss could be linearly quantified in caloric equivalents of a 3500-calorie deficit for one pound of body weight [28], suggesting that decreasing energy intake should lead to similar weight loss outcomes in patients following the same dietary intervention. The idea that low response to diet-induced weight loss is simply attributable to adherence was forwarded in a study by Lichtman et al., which described that individuals with a history of diet-resistant weight loss had large discrepancies in self-reported and actual energy intake and energy expenditure [29]. Remarkably complex mechanisms govern food intake control, and impairments in the perception of food intake can contribute to the development of obesity and make successful treatment more challenging [30,31]. However, a growing body of literature has since refuted the concept that poor response is simply due to poor adherence by demonstrating that weight loss after controlling for program adherence remains highly variable, in-part associated with complex metabolic adaptations in energy expenditure. In addition to the existence of adaptations in energy expenditure during weight loss, the recognition that obesity is a chronic disease has helped denounce the common bias suggesting that obese individuals resistant to weight loss simply lack discipline and/or have an inadequate perception of their food and/or activity habits [32]. While predictive models of weight loss are improving [33,34], there remains a great need for an improved understanding of the causes of variable weight loss success in order to identify more effective personalized treatment strategies.
Extensive research has examined genetic factors related to the propensity for obesity and weight loss. In the early 1990s, studies on monozygotic twins elucidated the heritability of body weight. Male twins overfed by 1000 kcal/day for 84 out of 100 days had similar intrapair variance in body weight and fat mass increases, but the variance between pairs of twins was significantly greater [35]. Similarly, monozygotic male twins who completed an exercise intervention in negative energy balance had similar anthropometric responses in body weight and regional fat distribution within a genotype but responses varied significantly between twin pairs [36]. The contribution of genetics to obesity susceptibility is also supported by adoptee studies, in which the BMI of adoptive children is more closely related to their biological parents than their adoptive parents [37]. In the 30 years following these classical studies, large-scale genome-wide association studies (GWAS) have identified ∼100 loci associated with obesity [38]. The heritability of obesity is high and is estimated to be between 40 and 70% [38–40], with over 500 genetic loci associated with adiposity traits [41]. Recent genome-wide polygenic risk score computational models have been able to robustly predict BMI and obesity [42], providing new opportunities for obesity prevention and mechanistic insights into the pathophysiology of obesity. However, genetic variants associated with increased obesity susceptibility only explain >20% of BMI variation. There are a number of valid explanations for ‘missing heritability’. Despite the information provided by genome-wide association studies, many genetic variants with weak but real effects on a trait, such as BMI, do not reach GWAS significance (P<10−8). Array technologies also do not capture rare variants that are not in linkage disequilibrium with genotyped variants (the non-random association of alleles at different loci) [43]. Newer approaches that make use of whole genome sequencing (WGS) data and a methodology termed GREML (genomic-relatedness-based restricted maximum likelihood) have markedly increased the proportion of predicted heritability explained to date for traits such coronary heart disease, height, and BMI [44,45]. Additional and important layers exist including the epigenome and its interactions with the environment [44]. Finally, even in the context of high genetic risk, clearly lifestyle factors can markedly influence the phenotypic outcome, highlighting the influence of other biological, environmental, and lifestyle factors in the development of obesity [42].
Our group has been investigating the factors that contribute to variability in weight loss in cohorts sampled from over 5000 patients who have completed the same intensively supervised 900 kcal/day meal replacement (OptiFast 900, Nestle) and behavioral program. We have shown that patients in the top quintile for weight loss (diet-sensitive, DS) have many physiological characteristics that distinguish them from patients in the lower quintile for weight loss (diet-resistant, DR). Many of these characteristics relate to skeletal muscle composition and metabolism including greater expression of genes involved in glucose and fatty acid metabolism, higher mitochondrial proton leak, and a greater antioxidant capacity in skeletal muscle from DS patients compared with DR patients [25,27,46,47]. Patients with diet-resistant obesity also exhibit lower weight loss capacity for up to 26-weeks following Roux-en-Y gastric bypass surgery [48], further supporting a biological basis for weight loss capacity.
The role of energy expenditure in weight loss success
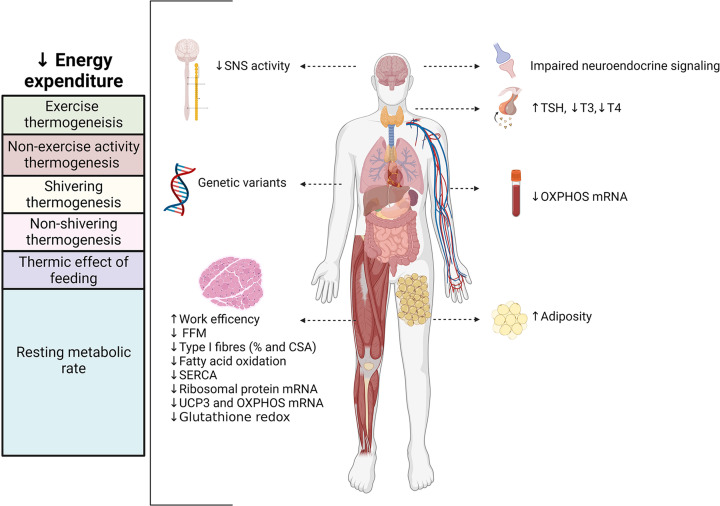
Differences in energy expenditure and resting metabolic rate (RMR) can also contribute to the risk for obesity development [49]. Energy expenditure is classically defined as the sum of the basal metabolic rate, thermic effect of feeding, and non-resting energy expenditure. Fat-free mass (FFM) is the most metabolically active component of body composition, and thus, the best predictor of resting metabolic rate [50,51]. Non-resting energy expenditure is the most variable component to total energy expenditure, and the metabolic variable most affected by weight change [52]. Non-resting energy expenditure can further be categorized into energy expenditure from thermoregulatory energy expenditure, exercise/voluntary physical activity and non-exercise activity thermogenesis (NEAT) that is related to non-volitional movements, such as fidgeting [53]. Thermoregulatory energy expenditure mechanisms include shivering and non-shivering thermogenesis. Energy expenditure fluctuates across the lifespan and, also, demonstrates high interindividual variation. Highlighted in a recent study with over 6000 participants in 29 different countries, analysis of doubly labelled water data revealed that total daily energy expenditure can vary ±20% even after accounting for age, fat-free mass, and sex [54]. The large interindividual variation in energy expenditure has a strong genetic component, with highly similar RMRs observed between monozygotic versus dizygotic twins [55], and siblings within the same family [49]. Starting in late childhood, low RMR is associated with a greater weight gain in adolescence [56]; these observations persist in adulthood, with a continued association between low 24-h energy expenditure and weight gain over 2 years [49]. When obesity develops, there can be increases in both total energy expenditure and RMR [57–60], which are largely due to increases in FFM that accompany the increase in body weight [58].
Reducing energy intake can elicit an evolutionarily conserved adaptive decline in RMR to preserve energy during times of dietary energy deficits and starvation [61,62]. This adaptive thermogenesis was elucidated as part of the Minnesota semistarvation study in which 24 weeks of semistarvation was found to reduce resting energy expenditure by 15.5%, independent of changes in body mass and FFM [63]. While interventions that have used caloric restriction as a tool to induce weight loss in individuals with obesity generally use moderate reductions in caloric intake, more recent studies have also demonstrated that weight loss can induce a decline in energy expenditure that persists for several months to years in the weight-reduced state [64–67]. However, some studies report no changes in energy expenditure in subjects formerly with obesity [68,69].
Weight-loss-induced adaptive declines in thermogenesis can oppose successful weight loss maintenance, with larger decreases in energy expenditure conferring increased susceptibility to weight regain [65,70,71]. Even in healthy lean males, those who exhibited greater decreases in energy expenditure while fasting gained more weight during a 6-week low-protein overfeeding intervention, indicating that larger reductions in fasting energy expenditure increase obesity susceptibility [70,71]. Similarly, individuals with a limited capacity to increase energy expenditure following overfeeding, originally referred to as ‘luxuskonsumption’, have increased susceptibility to weight gain compared with those who have greater increases in energy expenditure with overfeeding [49,71,72]. Consistent with the inverse association between energy expenditure and weight gain susceptibility, individuals with diet-resistant obesity have a greater decrease in energy expenditure while on a low-calorie diet compared to individuals who lose weight quickly [73–75]. Diet-induced weight loss can prompt the undesirable loss of FFM [76], exacerbating weight-loss-induced adaptive thermogenesis. Interindividual differences in energy metabolism likely underlie the considerable variability in adaptive thermogenesis that may enhance or perturb weight loss capacity.
The molecular mechanisms for adaptive thermogenesis and resistance to diet-induced weight loss are vast and involve metabolic and neuroendocrine factors. Upon achieving a greater than 10% weight loss, there are substantial changes to sympathetic nervous system (SNS) activity, notable decreases in thyroid hormones and leptin, and increases in skeletal muscle work efficiency (energy cost per muscle contraction) [61,62]. Total mass, contractile activity and metabolism of skeletal muscle greatly influence weight loss capacity. Skeletal muscle accounts for 40–45% of total body mass, and when the body is at rest, skeletal muscle is responsible for approximately 20% of resting energy expenditure (this proportion increases with exercise) [77,78]. Beyond its overall energy demands, skeletal muscle is the primary site of postprandial glucose uptake [79] and plays a major role in maintaining whole-body glucose homeostasis [80]. Skeletal muscle also serves as a reservoir for amino acids permitting synthesis of new proteins during fasting periods or supporting energy transduction via proteolysis when other macronutrient sources are depleted [81].
Skeletal muscle and mitochondrial bioenergetics
Skeletal muscle metabolism can rapidly adapt to meet energy demands and relies heavily on mitochondrial oxidative metabolism to transduce macronutrient energy into adenosine triphosphate (ATP) [82]. Oxidation of energy substrates provides reducing equivalents (i.e., electrons) that then drive the activity of the mitochondrial electron transport chain (ETC). The electrons can enter at complexes I–III (CI–CIII) of the ETC, and the flow of electrons coincides with the pumping of protons by CI, CIII and CIV from the matrix to the intermembrane space, thereby contributing to the generation of protonmotive force (PMF) across the inner membrane. PMF is comprised of two components: a charge gradient (mitochondrial membrane potential, Δψm) and a chemical gradient (ΔpH). Positively charged ions generate the Δψm charge gradient, which has a much larger contribution to PMF than the chemical separation that determines the ΔpH [83,84]. The return of protons to the matrix, down the electrochemical gradient via the F1F0-ATP synthase produces ATP and decreases PMF [85].
In most cells of the body, OXPHOS yields 90% of the ATP needed to support cellular work; however, the efficiency of mitochondrial ATP production can be highly variable due to the uncoupling of energy substrate oxidation from ATP synthesis through a process termed proton leak. When ATP demand is low, protons can migrate across the inner membrane into the matrix, independently from the ATP synthase, leading to increased activity of the ETC, consuming oxygen in the process. Proton leak is thought to be a protective redox regulation mechanism, preventing excess formation of reactive oxygen species (ROS) by allowing electron flow through the ETC, rather than a build-up and escape of electrons to form superoxide anion.
The mechanisms of proton leak are complex and include both basal and inducible forms of proton conductance. In skeletal muscle proton leak, uncoupling is thought to be responsible for up to 50% of resting muscle energy expenditure, and this proportion is thought to decrease as ATP demands increase [86]. At the protein level, the only UCP expressed in skeletal muscle is UCP3, but it is expressed at approximately two orders of magnitude lower than the levels of UCP1 in brown adipose tissue mitochondria. UCP1 is well-recognized for its role in mediating proton leak uncoupling in brown and beige adipose tissues in many mammals [87–89], and its activity in adult humans is associated with reduced risk for many chronic metabolic diseases [90,91]. Adenine nucleotide translocase (ANT), an abundant mitochondrial inner membrane protein in skeletal muscle that supplies ADP from the cytosol to the mitochondrial matrix in exchange for ATP (i.e. ADP/ATP exchange). ANT also mediates proton leak (basal and fatty acid-activated) independently of ADP/ATP exchange and may facilitate the permeability transition pore opening through mitochondrial depolarization [86,92–95]. Fatty acids act as co-factors for H+ transport by ANT and may bind to the reputed ADP/ATP binding site as H+ transport by ANT is negatively regulated by ANT-mediated ADP/ATP exchange [93,96,97]. Taken together, these data are consistent with the conclusion that ANT has two distinct and partially competing functions: fatty-acid activated proton leak and ADP/ATP exchange. Post-translational modifications of ANT through acetylation, and glutathionylation have been reported and require further investigation in light of the recently identified reciprocal activities of ADP/ATP transport versus H+ leak [98,99].
If proton leak is responsible in humans (as in rats) for upwards of 50% of energy expenditure in resting muscle, and as muscle energy expenditure is responsible for roughly 20% of RMR, then proton leak in muscle alone could be responsible for approximately 10% of RMR in humans [100–102]. A role for low proton leak in muscle in the context of diet-resistant obesity is supported by results from our group, and is discussed below.
Declines in skeletal muscle mitochondrial content have repeatedly been associated with obesity as evidenced by 20–60% reductions in mitochondrial surface area (using electron microscopy), and significantly lower expression of various mitochondrial genes and proteins [103–105]. It is well established that impaired skeletal muscle mitochondrial function is characteristic of obesity, and is reflected by impaired fatty acid metabolism, lower activity of mitochondrial enzymes, and increased H2O2 emission [106–113].
Skeletal muscle mitochondrial bioenergetics and weight loss
Skeletal muscle fiber composition and the efficiency of skeletal muscle mitochondrial energy transduction have been linked to weight loss characteristics in individuals with obesity (Figure 1) [102]. Adults with obesity have lower proportions of mitochondria-dense type I fibers compared with proportions in muscle of lean controls [114–117], and the proportion of type I fibers contributes to obesity susceptibility [118,119]. Moreover, a recent systemic review revealed a negative relationship between the proportion of type I fibers and BMI, and a positive relationship between the proportion of type IIX fibers and BMI, with no clear relationship between type IIa fiber proportion and obesity [120]. The proportion of type I fibers also contributes to weight loss capacity, with a high degree of weight loss following bariatric surgery strongly associated with type I fiber content in rectus abdominis muscle [114]. Similarly, vastus lateralis muscle from DS patients with obesity has a greater proportion of type I fibers and fiber hypertrophy compared with both DR patients and lean controls [27]. Altogether, findings support the conclusion that variability in skeletal muscle fiber composition and size are associated with obesity risk and weight loss.
Figure 1. Potential factors contributing to adaptive thermogenesis during weight loss and resistance to diet-induced weight loss.
Restricting energy intake can elicit metabolic adaptations in energy expenditure that oppose successful weight loss. The mechanisms that underlie adaptations in energy-expenditure are associated with genetic factors, decreased activity of the sympathetic nervous system (SNS), impaired neuroendocrine signaling, changes in thyroid hormones, and variations in body composition (loss of fat free mass [FFM]). Moreover, resistance to diet-induced weight loss is associated with lower expression of OXPHOS and ribosomal genes, diminished glutathione redox, less type I muscle fibers, and decreased oxidative capacity. Figure created with BioRender.com.
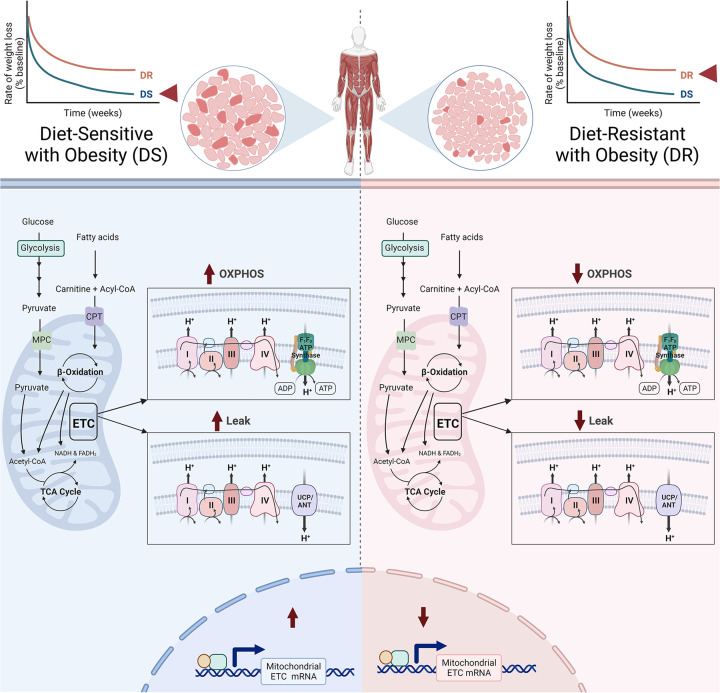
While it is clear that skeletal muscle mitochondrial content is decreased with obesity [103,104], a direct link between skeletal muscle mitochondrial content and propensity for weight loss has yet to be observed. However, substantial evidence supports the conclusion that mitochondrial oxidative capacity may drive weight loss success. Gene set enrichment analysis of repeated skeletal muscle biopsies obtained from patients undergoing a hypocaloric diet revealed that DS patients who lost an average of 49% more weight than DR patients had significantly higher enrichment of gene transcripts encoding mitochondrial ETC proteins [27]. Higher enrichment of mitochondrial ETC transcripts was even observed in circulation prior to weight loss, suggesting that oxidative capacity could predict weight loss success [121]. The differences in ETC transcripts between DR and DS individuals translate into functional differences in skeletal muscle, where maximal mitochondrial respiration and complex I+II OXPHOS are lower in muscle biopsies of DR versus DS participants (Figure 2) [46]. However, lower enrichment of mitochondrial ETC transcripts in DR muscle did not translate into decreased expression of key ETC subunits in primary myotubes isolated from DR individuals [47].
Figure 2. Mitochondria-specific differences observed in skeletal muscle from diet-resistant versus diet-sensitive patients with obesity.
Skeletal muscle from diet-resistant individuals has less mitochondrial proton leak and lower expression of genes involved in the electron transport chain and fatty acid metabolism compared to diet-sensitive individuals. Moreover, when challenged with a high-fat meal, skeletal muscle fatty acid oxidation and maximal oxidative phosphorylation was lower in DR individuals (see text for details). Figure created with BioRender.com.
Skeletal muscle energy transduction efficiency is also important. Differences in mitochondrial proton leak uncoupling have been associated with weight loss capacity. Proton leak was found to be 50% higher in mitochondria isolated from rectus femoris muscle of DS individuals who achieved high rates of diet-induced weight loss compared to DR individuals with obesity [25]. Enhanced proton leak in DS muscle appears to be cell autonomous as proton leak is also higher in primary myotubes isolated from DS individuals [47]. The difference in skeletal muscle mitochondrial proton leak between DS and DR has been attributed to altered expression of UCP3, as UCP3 mRNA expression is greater in isolated mitochondria from DS rectus femoris muscle [25]. When the human form of UCP3 is overexpressed in mouse skeletal muscle by ∼2.5-fold above normal, mice are protected from diet-induced obesity and display metabolic characteristics of enhanced fatty acid oxidation [122–125]. Genetic polymorphisms may play a key role in determining UCP3 function, as a recent meta-analysis concluded that -55C/T polymorphism in UCP3 protects from obesity and T2D [126,127]. Moreover, evidence from murine models indicates that ADP sensitivity is impaired in skeletal muscle from mice with diet-induced obesity [128], suggesting that ANT-mediated ADP/ATP exchange is decreased in obesity. However, ANT-mediated proton leak may remain intact in obesity due to the accumulation of intramuscular fatty acids which may promote H+ transport [107,129,130]. These data support the conclusion that differences in mitochondrial uncoupling in muscle impact propensity for obesity and weight loss.
There is evidence to support the possibility that blunted capacity for fat oxidation contributes to obesity and the capacity for weight loss. In response to a defined high-fat meal (∼35% of daily kcal requirements based on indirect calorimetry; >60% calories from fat), diet-resistant women exhibit lower fatty acid clearance, suggesting that lower fatty acid oxidation may contribute to the diet-resistant phenotype [46]. Low resting respiratory exchange ratio (RER, VCO2/VO2 i.e., carbohydrate versus fat oxidation) has previously been identified in women with a high rate of weight loss [131]; whereas high resting RER has been linked to weight gain [132]. However, meta-analyses of studies associating 24-h RQ and body weight have not confirmed this hypothesis [133]. Ex vivo studies at the level of skeletal muscle have shown that fatty acid oxidation is lower in DR muscle [46], which may be attributed to decreased fatty acid availability and reduced fatty acid mobilization from adipose tissue [134,135].
With the increasing availability of omics platforms, researchers have a greater capacity to investigate differences in protein and metabolite patterns that could explain weight loss variability. In a large cohort of adults with obesity, predictive models of baseline parameters identified plasma metabolites that could explain up to 57% of variation in weight loss success prior to diet-induced weight loss. Circulating concentrations of branched-chain amino acids (BCAAs), tyrosine, specific lipid species, and citrate have consistently been linked to weight loss success in principal component analyses of plasma and serum metabolites [73,131,136]. There is a clear pattern for a metabolic basis of variations in body weight; however, much research is still required to translate this to novel and personalized approaches for the treatment of obesity.
Interventions to boost skeletal muscle mitochondrial bioenergetics for diet-resistant obesity
While comprehensive weight loss programs that focus on decreasing caloric intake can result in a 5–8% weight reduction in many individuals [21], it is clear that diet-centric approaches do not work for all patients, and that maintenance of the reduced body weight is the exception rather than the norm. Bariatric surgery is the most effective treatment strategy and typically results in 20–45% weight loss 12 months following Roux-en-Y gastric bypass (RYGB). Moreover, the weight loss is maintained in 70% of patients for over 7 years [137]. However, as discussed, the invasive and irreversible nature of this treatment, as well as the lifelong changes to food intake and diet supplements, mean that this approach is not available or desirable for many with obesity. Table 1 summarizes possible interventions to boost skeletal muscle bioenergetics in diet-resistant obesity.
Table 1. Interventions to boost skeletal muscle mitochondrial bioenergetics.
| Intervention | Mode of action and adaptions to skeletal muscle metabolism | Clinically relevant weight loss | Status and remarks | Safety and adverse effects | References |
|---|---|---|---|---|---|
| Lifestyle | |||||
| Aerobic exercise training | ↑ Energy expenditure ↑ Type I fibers ↓ Fat mass ↑ Mitochondrial content and function |
Aerobic exercise alone, low weight loss; Exercise + diet, high weight loss | Recommended in most weight loss programs | Non-serious adverse events related to muscle pain and fatigue | [146,147,151,369,370] |
| Resistance exercise training | ↑ Protein translation ↑ FFM ↑ Hypertrophy |
Resistance Exercise alone, no weight loss; Resistance exercise + diet high | Recommended in most weight loss programs | Non-serious adverse events related to muscle pain and fatigue | [212,217,370,371] |
| Cold exposure | ↑ Energy expenditure ↑ Heat production ↑ SNS activity ↑ Skeletal muscle contractions ↑ SERCA Ca2+ cycling ↑ UCP content and proton leak |
Insufficient evidence | Hypothermia | [372] | |
| Mitochondrial uncouplers | |||||
| (2,4)-Dinitrophenol (DNP) (3–5 mg/kg/day; 75–300 mg/day) |
↑ Energy expenditure ↑ Heat production ↑ Proton leak |
High | Not approved for weight loss treatment | Major safety concerns; hyperthermia, mortality | [266] |
| Salsalate (3–4 g/day) | Animal studies: ↑ AMPK activation ↑ Energy expenditure ↑ Heat production ↑ Proton leak |
No | Not approved for weight loss. Human trials show increased or no change in body weight | Side effects include tinnitus, headache, rash, gastrointestinal disturbances | [273,275,373] |
| Niclosamide, niclosamide ethanolamine and nicloasmide piperazine | Animal studies: ↑ Proton leak ↑ Fatty acid oxidation |
Insufficient evidence in humans | Not approved for weight loss treatment; Approved as anthelmintic | Generally well tolerated; nausea, gastrointestinal disturbances, dizziness, pruritus | [277,278,374] |
| Appetite suppressants | |||||
| Amphetamine Methamphetamine |
↓ Energy intake ↑ Energy expenditure ↑ SNS activity ↑ Thyroid hormones ↑ Catecholamine and monoamines ↑ (α and β)-Adrenergic receptor stimulation Skeletal muscle (animal studies): ↑ Motor stimulation ↑ UCP3 ↑ SERCA activity |
High | Withdrawn | High risk for addiction and abuse; adverse psychiatric effects, neurotoxicity | [287,288,375,376] |
| Sympathomimetic agents/Amphetamine Congeners e.g. Amfepramone, Diethylpropion, Mazindol Phentermine (15–30 mg), Phentermine/topiramate, Phenmetrazine (3 × 25 mg/day), Phendimetrazine (6 × 35 mg) |
↓ Energy intake ↑ SNS activity ↑ Release of monoamines ↑ (α and β)-Adrenergic receptor stimulation ↑ Motor stimulation |
High | Approved for short-term use | Side effects include paresthesia, cardiovascular abnormalities, nausea | [263,377,378] |
| Sibutramine (10–20 mg) | ↓ Energy intake ↑ SNS activity ↑ Energy expenditure ↑ (α and β)-Adrenergic receptor stimulation |
High | Withdrawn | Major safety concerns; Hypertension, increased risk of heart attack and stroke, mortality | [377,379,380] |
| Serotonergic agents Fenfluramine, Dexfenfluramine |
↓ Energy intake ↑ Serotonin release ↓ Serotonin reuptake ↑ SNS activity ↑ (α and β)-Adrenergic receptor stimulation Muscle: ↑ Glucose uptake, lactate production (Fenfluramine) |
High | Withdrawn | Major safety concerns; Increased risk of cardiovascular events, valvulopathy, pulmonary hypertension |
[377,381] |
| Other | |||||
| Thyroid Hormones T3 (18–117 µg/70 kg/day) |
↑ Energy expenditure Muscle: ↑ ATP turnover ↑ Proton leak ↑ UCP3 |
Insufficient evidence in euthyroid patients | Not approved for weight loss treatment; Approved for hypothyroidism | Safety concerns for euthyroid patients. Adverse effects include thyrotoxicosis, cardiovascular abnormalities, decreased bone density, muscle catabolism | [382,383] |
| Metformin (500–3000 mg/day) | ↓ Energy intake ↑ AMPK activation ↑ Fatty acid oxidation |
Medium | Not approved for weight loss treatment; Approved for hyperglycemia | [335,384] | |
| β-Adrenoceptor agonists e.g Isoproterenol, Isoprenaline (non-selective β-adrenoceptor agonists) Mirabegron (β3-adrenoceptor agonist) Formoterol (β2-adrenoceptor agonist) |
↑ β-Adrenergic receptor stimulation ↑ Energy expenditure ↑ Lipolysis ↑ Fatty acid oxidation ↑ Lean mass |
Insufficient evidence from long-term trials | Not approved for weight loss treatment | [302,385] | |
| GLP-1 agonists Liraglutide (3 mg/day) Semaglutide (2.4 mg subcutaneous injection/week) |
↓ Energy intake ↑ Energy expenditure ↑ Fatty acid oxidation ↓ Muscle loss |
High | Approved for weight loss treatment | Nausea, diarrhea, constipation | [386,387] |
| Supplements | |||||
| Caffeine (60–600 mg/day) | ↑ Energy expenditure ↑ SNS activity ↑ Catecholamines ↑ Fatty acid oxidation |
Low | Approved for sale as a dietary supplement | Generally well tolerated in moderate doses. Mild side effects include sleep disturbances, increased blood pressure, diuresis, nausea, and gastrointestinal discomfort. Toxic at doses of 15 mg/kg | [360–362,388] |
| (L)-Carnitine (1.8–4 g/day) | ↑ Fatty acid oxidation | Low | Approved for sale as a dietary supplement | Generally well tolerated. Mild side effects include nausea and gastrointestinal disturbances | [367,368] |
| Ephedrine/Ephedra (20–150 mg/day) | ↑ Energy expenditure ↑ SNS activity ↑ Fatty acid oxidation |
Low | Not approved for sale as a dietary supplement | Major safety concerns. Adverse effects include psychiatric symptoms, gastrointestinal disturbances, cardiovascular abnormalities and events, mortality | [352,353] |
| Conjugated Linoleic acid 2.4–6 g/day | ↑ Lipolysis | Low | Approved for sale as a dietary supplement | Generally well tolerated. Mild side effects include gastrointestinal discomfort | [354] |
| Green tea Green tea catechins (141–1207 mg/day) |
↑ Energy expenditure ↑ Fatty acid oxidation |
Low | Approved for sale as a dietary supplement | Generally well tolerated. Mild side effects include nausea, gastrointestinal discomfort, increased blood pressure | [353,356] |
Interventions that target energy expenditure and skeletal muscle metabolism and their associated weight loss efficacy in humans. Clinically relevant weight loss scale: low = 0–2 kg weight loss, medium = 2–5 kg weight loss, high ≥ 5 kg weight loss.
SERCA, sarcoplasmic/endoplasmic reticulum Ca2+ ATPase; SNS, sympathetic nervous system; UCP3, uncoupling protein 3.
Exercise
The use of exercise in the absence of dietary approaches as a means to induce weight loss has a controversial history in obesity treatment. Despite abundant research demonstrating the beneficial effects of exercise in mitigating the risk of cardiometabolic disease and all-cause mortality [138–141], exercise is often overlooked as a primary treatment for obesity. Exercise is well recognized for its beneficial effects on skeletal muscle glucose homeostasis and has repeatedly been shown to enhance insulin sensitivity in individuals with obesity [142–144]. However, caloric restriction is ultimately more effective at inducing weight loss than exercise is alone [145–152]. Given the above-described deficits in mitochondrial energetics in DR obesity, exercise interventions may be particularly beneficial in DR obesity; on the other hand, if the deficits are ‘hard-wired’ (e.g., genetically), then exercise interventions may not be of particular benefit to those with DR obesity.
Exercise evokes ATP demand to support the energy needs during muscle contractile activity. The three most ATP demanding cellular processes include the sarcolemmal Na+/K+ ATPase, Ca2+ reuptake into the sarcoplasmic reticulum by the Ca2+ ATPase, and actin-myosin cross-bridge cycling by myosin ATPase. The substrate source for ATP supply during exercise is largely determined by exercise intensity and duration. Intense, short-term exercise is fuelled by anaerobic (non-mitochondrial) pathways that facilitate the breakdown of phosphocreatine and glycogen to produce ATP. As the intensity decreases and the duration of exercise increases, oxidative phosphorylation becomes the primary source of ATP for contracting muscle. The metabolic demand for ATP during exercise is accompanied by the activation of intracellular stress signaling pathways in skeletal muscle, including production of ROS [153], release of proinflammatory myokines [154], calcium (Ca2+) [155], and the unfolded protein response [156,157]. These molecular stressors elicit physiological adaptations that subsequently enhance mitochondrial oxidative capacity [158–161]. Resistance and aerobic exercise training promote different, complementary but potentially interfering adaptations in skeletal muscle that improve skeletal muscle metabolism [162].
Aerobic exercise
Aerobic exercise training is traditionally associated with improved skeletal muscle oxidative capacity and bioenergetic metabolism. A single aerobic exercise bout initiates mitochondrial biogenesis through transient increases in mRNA and protein expression of peroxisome proliferator-activated receptor (PPAR)-γ coactivator-1α (PGC1α), along with PGC1α translocation to the nucleus [163,164]. PGC1α orchestrates mitochondrial biogenesis by interacting with and activating additional transcription factors that promote expression of nuclear- and mitochondrial-encoded genes involved in β-oxidation, OXPHOS, and antioxidant enzymes [161,165–168]. With prolonged endurance training, the induction of mitochondrial biogenesis and synthesis of new mitochondrial proteins result in increases in skeletal muscle mitochondrial content [163,169–171] that are stoichiometrically associated with exercise-induced improvements in mitochondrial respiratory capacity [172]. There is some evidence from muscle-specific loss of PGC1α rodent models suggesting that PGC1α can be dispensable for exercise-induced increases in mitochondrial content and function [173], suggesting that redundant pathways confer exercise-induced improvements in mitochondrial health. However, human studies generally agree that PGC1α plays a central role in facilitating the exercise-induced improvements in oxidative capacity. In addition to initiating transcription for mitochondrial proteins, aerobic exercise training has profound affects on mitochondrial ribosomal protein translation [172], mitochondrial supercomplex formation [174], and intrinsic mitochondrial function. Together, enhanced mitochondrial density and function contribute to increases in skeletal muscle oxidative capacity that correlate with improvements in cardiorespiratory fitness [163,169–171].
AMP-activated protein kinase (AMPK) and p38 MAP kinase are also activated in skeletal muscle with aerobic exercise and are intimately linked to PCG1α to support increases in mitochondrial content and oxidative capacity [175–180]. In response to aerobic exercise, AMPK and p38 MAPK phosphorylate and directly activate PGC1α, and also indirectly activate other transcription factors to support mitochondrial biogenesis, including myocyte enhancer factor 2 (MEF2), ATF-2, and p53 [179,180]. In addition to contributing to exercise-induced mitochondrial biogenesis, AMPK also improves skeletal muscle glucose and fatty acid metabolism by enhancing substrate uptake and oxidation. Moreover, in rodent models and untrained humans, aerobic exercise has been shown to increase UCP3 mRNA expression in skeletal muscle [181,182], which seems counterintuitive, but may be associated with an enhanced capacity for fatty acid oxidation and the associated need to minimize fatty acid oxidation associated ROS [122,183,184].
Beyond induction of mitochondrial biogenesis, aerobic exercise is associated with enhanced mitochondrial networking which also confers improvements to oxidative capacity. Mitochondria form as an interconnected network and can adapt and reorganize to meet the bioenergetic demands of a cell. Mitochondria appear smaller in skeletal muscle from individuals with obesity, and the mitochondrial reticulum length is shorter [185,186]. A single aerobic exercise bout increases the number of electron-dense contact sites between adjacent mitochondrial membranes [187] and augments mRNA expression of mitofusins 1 and 2 (MFN1 and MFN2) [188]. Aerobic exercise training promotes elongation of the mitochondrial reticulum by enhancing the ratio of fusion to fission proteins [189]. Moreover, endurance training enhances mitophagy, a protective mechanism that clears damaged mitochondria to maintain the health of the mitochondrial network and prevent cellular apoptosis [190,191]. The energetic imbalance imposed by exercise activates AMPK while suppressing mTOR leading to the induction of mitophagy through the PTEN-induced putative kinase protein 1 (PINK1)/Parkin pathway [192–195]. Overall, the exercise-induced adaptations in mitochondrial networking improve mitochondrial turnover, quality, and function.
Resistance exercise
Resistance training robustly increases anabolic signaling and promotes skeletal muscle hypertrophy as evidenced by increases in cross-sectional area of myofibers [196–198]. While long-term aerobic exercise training can also promote hypertrophy particularly in untrained individuals [199], a recent meta-analysis validated the superiority of resistance training at promoting skeletal muscle hypertrophy [200]. Resistance exercise-induced skeletal muscle hypertrophy is primarily driven by enhanced myofibrillar protein synthesis [201] and increases in translational capacity via ribosomal biogenesis [202].
There is some evidence suggesting that resistance exercise can enhance mitochondrial function, but not content [203]. However, while increases in skeletal muscle mitochondrial respiration have been observed following 12 weeks of resistance training in the absence of changes in markers of mitochondrial content [203], other studies have failed to report this relationship [204].
Aerobic and/or resistance exercise and weight loss
Weight-loss interventions that combine exercise training with caloric restriction demonstrate that exercise can enhance weight loss capacity compared to caloric restriction alone [147,150–152,205]. Even after RYGB bariatric surgery, higher levels of exercise/physical activity are associated with greater reductions in body weight and fat mass [206]. When comparing the effects on mitochondrial oxidative capacity in patients with obesity, diet-induced weight loss fails to elicit improvements in mitochondrial capacity, whereas diet combined with exercise enhances muscle mitochondrial content and ETC activity [207–209].
While most weight loss studies have focused on aerobic exercise training [210], the combination of aerobic and resistance exercise training may prove to be particularly useful during caloric restriction in DR patients with obesity. Aerobic exercise plus caloric restriction promotes greater decreases in adipose tissue than dieting alone [211] and is likely more effective at promoting decreases in body weight and fat mass than resistance exercise [212,213]. At the molecular level, aerobic exercise training may improve the previously observed deficits in skeletal muscle oxidative capacity in DR individuals by promoting mitochondrial biogenesis. Aerobic exercise can also increase the proportion and cross sectional area of type I fibers [214,215], which may be particularly useful in individuals with diet-resistant obesity who have been shown to have lower proportions and cross sectional area of type I fibers in vastus lateralis muscle [27]. In contrast, resistance exercise would maintain lean body mass and preserve myocellular quality during weight loss [211,216]. Within skeletal muscle, resistance training may enhance protein synthesis and ribosomal biogenesis in DR individuals, who have been shown to have lower ribosomal protein transcripts in whole blood and in skeletal muscle [27]. In line with the idea that a combined aerobic and resistance exercise program may preferentially benefit DR individuals with obesity, when examining the effects of diet plus aerobic and/or resistance exercise on weight loss outcomes, interventions using diet plus combined aerobic/resistance training generally are the most effective at inducing weight loss, improving physical function, and preserving lean body mass [216,217].
Cold exposure
The discovery of functional brown adipose tissue (BAT) in adult humans has renewed interest in exploiting cold-induced thermogenesis to treat obesity. The use of positron-emission tomographic and computed tomographic (PET-CT) imaging using radiolabeled tracers demonstrated that BAT is generally expressed in fairly low amounts of 0–200 g in adults, and that amounts of BAT decrease with both age and BMI [218,219]. BAT is activated through the release of catecholamines by the sympathetic nervous system. In mature brown adipocytes, catecholamines stimulate β-adrenergic receptors that are coupled to Gs proteins, which then activate adenylyl cyclase to increase intracellular cAMP levels. Increases in intracellular cAMP activate protein kinase A (PKA) and hormone sensitive lipase to enhance lipolysis of triglycerides and the expression of UCP1 and ancillary thermogenic proteins [220,221]. As well, the released FFAs acutely activate UCP1, prompting the rapid oxidation of fatty acids, causing thermogenesis and increasing whole-body energy expenditure [221]. β3-adrenergic receptors mediate thermogenesis in rodents, whereas Blondin et al. [2020] recently demonstrated that β2-adrenergic receptors mediate BAT thermogenesis in humans [222].
Importantly, while much of the thermogenic response is attributable to β-adrenergic receptor activation, brown adipocytes also contain α-adrenergic receptors that regulate thermogenic responses [223]. α1-adrenergic receptors coupled to G proteins (Gq) potentiate the thermogenic response by activating phospholipase C to cleave phosphatidylinositol 4,5-bisphosphate (PIP2) into diacylglycerol (DAG) and inositol 1,4,5-triphosphate (IP3) [224–226]. DAG stimulates PKC to induce the expression of thermogenic proteins, while IP3 enhances cytosolic Ca2+ concentrations by activating calcium channels in the endoplasmic reticulum [225,227]. In contrast, activation of α2-adrenergic receptors inhibits thermogenesis through the coupled actions of Gi proteins that inhibit β-adrenergic receptor stimulation of adenylyl cyclase [221,228].
In addition to UCP1-mediated thermogenesis in brown adipocytes, substrate cycling also contributes to enhanced energy expenditure in BAT and is thought to function independent from UCP1. For example, creatine kinase B liberates excess ADP derived from ATP in a 1 ATP:1 creatine stoichiometric relationship, creating a futile creatine-dependent ADP/ATP substrate cycle that stimulates thermogenic respiration in brown adipocytes [229,230]. Similarly, lipid cycling from the resynthesis of TAGs and calcium cycling are also thought to contribute to thermogenesis in BAT independently from UCP1 [231,232].
Low levels of UCP1 were originally hypothesized to be important in obesity susceptibility [233], and the benefits of BAT were recently highlighted by Becher et al. who demonstrated that individuals with BAT have a lower prevalence of cardiometabolic diseases and improved blood glucose and lipid profiles [90]. However, individuals with obesity demonstrate blunted responses to cold exposure, including reduced sympathetic responsiveness, and lower BAT glucose uptake during cold exposure [234]. Moreover, adipocytes from individuals with obesity display lipolytic catecholamine resistance [235]. Overfeeding and cold exposure evoke similar interindividual variation patterns in energy expenditure and metabolism, through diet-induced, and cold-induced thermogenesis, respectively [236,237].
Studies have attempted to target β-adrenergic receptors with sympathomimetics to directly stimulate BAT without the need for cold exposure but have yielded mixed results. Both the systemic infusion of isoprenaline and the intramuscular injection with ephedrine fail to elicit glucose uptake by BAT in humans [238,239]. Ephedrine administered orally has been shown to enhance BAT glucose uptake in lean adults, but not individuals with obesity [240]. Direct activation of β3-adrenergic receptors through oral admiration of mirabegron (50 and 200 mg), a medication approved for treating overactive bladders, increases energy expenditure in humans [222,241]. Despite increases in energy expenditure, mirabegron failed to stimulate BAT oxidative metabolism in humans and induced undesirable cardiovascular responses owing to contaminant non-selective β-adrenergic receptor activation, including increased heart rate and blood pressure [222]. To avoid the unwanted effects on the cardiovascular system, most studies have employed cold exposure to investigate BAT thermogenesis in humans. However, as a method to study the contributions of BAT, cold exposure is somewhat limited by the inevitable shivering activity that evokes contractions of skeletal muscles, even when measures are taken to minimize shivering activity [242,243]. Cold-induced increases in metabolic rate are closely associated with shivering intensity [243]. During mild cold exposure, shivering preferentially recruits type I muscle fibers in proximal muscle groups for contraction [243] and evokes the mild uncoupling of skeletal muscle mitochondria [244]. These contractions are accompanied by large increases in glucose uptake with mild cold exposure, with skeletal muscle accounting for 50% more glucose turnover than BAT during acute cold exposure [243]. Thus, while the cold-induced rate of glucose uptake in BAT is high relative to its small volume, skeletal muscle is the predominant site of glucose disposal during cold exposure [243]. Results from animal studies have supported the idea that skeletal muscle plays a central role in cold-induced thermogenesis even in the absence of shivering (i.e., non-shivering thermogenesis). Skeletal muscle mitochondrial uncoupling has been linked to increases in metabolic rate in several species such as dogs [245], pigeons [246], fur seals [247], rodents [248], and penguin chicks [249]. Furthermore, animal studies have demonstrated that alterations in mitochondrial coupling are accompanied by the stimulation of skeletal muscle angiogenesis [250], and high rates of calcium (Ca2+) cycling, an ATP-dependent process in the sarcoplasmic reticulum [251,252].
Beyond proton leak and mitochondrial oxidative capacity, there is emerging evidence to support a role for the sarcoplasmic/endoplasmic reticulum Ca2+ ATPases (SERCAs) in cold-induced thermogenesis. SERCAs are a family of membrane-bound P-type ATPases that utilize energy derived from ATP hydrolysis to translocate Ca2+ against the chemical gradient from cytosol into the sarcoplasmic reticulum [253,254]. Calcium is released from the sarcoplasmic reticulum into the cytoplasm via the ryanodine receptors to initiate a muscle contraction. Following contraction, SERCA ion pumps rapidly facilitate reuptake of calcium into the sarcoplasmic reticulum [255]. However, SERCA-mediated ATP hydrolysis can be uncoupled from Ca2+ transport, resulting in a futile cycle controlled by two membrane phosphoproteins: phospholamban and sarcolipin [256]. Data from rodent studies have confirmed a role for sarcolipin-mediated thermogenesis, as mice null for sarcolipin are unable to maintain their body temperature during an acute cold challenge [257]. Furthermore, overexpression of sarcolipin confers resistance against diet-induced obesity and induces mitochondrial biogenesis [258,259], suggesting that SERCA uncoupling could be exploited to enhance skeletal muscle energy expenditure in obesity.
Importantly, many of the molecular adaptations to cold exposure in skeletal muscle appear to diverge in response to either acute or prolonged cold exposure. UCP3 mRNA and protein expression are rapidly up-regulated in rat skeletal muscle within one day of cold exposure; however, prolonged cold exposure is associated with UCP3 downregulation [260]. In humans, UCP3 mRNA expression decreases after 60 h of mild cold exposure, without affecting UCP3 protein content [261]; however, authors found that interindividual differences in UCP3 protein were closely associated with individual differences in 24-h energy expenditure [261]. When comparing the effects of acute cold exposure versus four weeks of cold acclimation on mitochondrial bioenergetics, proton leak dependent respiration was 4.4-fold higher than baseline following acute cold exposure, but this effect was eliminated with prolonged cold acclimation despite similar levels of whole-body substrate utilization, consistent with the idea of inter-tissue adapations (e.g., muscle and BAT) [262]. Cold exposure, or mimetics of cold exposure may prove to be useful in boosting both skeletal muscle and BAT thermogenesis.
Pharmacotherapy
Research over the past two decades has markedly improved our understanding of the neuroendocrine basis of obesity, and this has led to the development of various anti-obesity medications. Before the development of newer age anti-obesity medications, there were repeated cycles of launching and withdrawing medications due to unforeseen adverse effects. Anti-obesity medications can generally be classified into five groups: (1) mitochondrial uncouplers, (2) appetite suppressants, (3) medications that promote nutrient malabsorption, (4) hormone replacement therapies, and (5) diabetes medications that also promote weight loss. Not all of these classes exert known effects on skeletal muscle metabolism and therefore only some will be addressed here [see detailed anti-obesity medication reviewed by Muller et al. [263]].
Mitochondrial uncouplers
As this review focuses to a large extent on variable efficiency of mitochondrial energetics, it is relevant to discuss past and current pharmacologic approaches to induce mitochondrial uncoupling. In the late 1880s, a weak lipophilic acid commonly used to manufacture explosives and dyes, [2,4]-dinitrophenol (DNP), was shown to markedly enhance energy expenditure in dogs. Tainter and Cutting then demonstrated that 300–400 mg/day of DNP could induce rapid weight loss by enhancing resting metabolism in humans [264,265]. DNP is a weak uncoupler that induces diffusion of protons across the inner membrane (i.e., bypassing ATP synthase) and thus increasing oxidative metabolism. Recently, Bertholet et al. challenged the idea that protonophores induce proton leak through protein-independent mechanisms by demonstrating that protonophores, including DNP, activate ANT and UCP1 to facilitate protonophoric activity [96]. DNP (300 mg) was then shown to induce weight loss averaging 6.3 kg in 113 patients with obesity with minimal side effects [266]. The success of DNP popularized its use as an over-the-counter weight loss agent until multiple adverse side effects, including cardiac arrests, leading to its withdrawal from the market in 1938 [267].
Since then, there has been interest in the development of novel mitochondrial uncouplers with improved tolerability and a wider dynamic range. Several promising compounds have been investigated, but poor absorption and adverse side effects have presented challenges [268]. Recent studies, for example, have focused on BAM15 ((2-fluorophenyl){6-[(2-fluorophenyl)amino](1,2,5-oxadiazolo[3,4-e]pyrazin-5-yl)}amine), a mitochondrial-targeted uncoupler that prevents and reverses diet-induced obesity in rodents [269,270]. High-fat diet-fed mice that were treated with 0.1% or 0.15% BAM15 (w/w) exhibited higher whole-body oxygen consumption, had lower RERs, and gained less fat mass compared with untreated mice [269,270], and effects were partly attributable to elevated hepatic lipid oxidation [270]. Moreover, BAM15 enhanced skeletal muscle mitophagy and mitochondrial content, in old mice fed a HF diet that also exhibited attenuated age-related declines in muscle mass and strength [271]. Recently, SHC517 was identified as a more potent derivative of BAM15 that was able to prevent diet-induced obesity and improve glucose tolerance in mice [272]. While these compounds have demonstrated promise in rodent models for the prevention of diet-induced obesity, caution is warranted for concerns related to potential toxicities (i.e., narrow safety windows) and abuse.
A handful of approved medications can also act as protonophores and induce mild mitochondrial uncoupling. The glucose-lowering prodrug salsalate can directly activate the β1 subunit of AMPK, but also enhances mitochondrial uncoupling at clinical concentrations [273]. Rodent studies have demonstrated that salsalate can enhance skeletal muscle thermogenesis and attenuate diet-induced weight gain [274]; however, clinical trials examining the glucose lowering effects of salasate have reported no change in body weight [275,276]. The anthelmintic medication niclosamide and its salt derivatives, niclosamide ethanolamine and nicloasmide piperazine, have shown promising effects at preventing diet-induced obesity by promoting mitochondrial uncoupling and enhancing lipid oxidation in rodents [277,278]. While niclosamide and its derivatives are tolerable for short-term use [279,280], future studies examining the long-term safety are needed. Altogether, mitochondrial-targeted uncouplers that have less safety concerns may be beneficial at enhancing skeletal muscle metabolism by increasing proton leak in individuals with obesity.
Appetite suppressants
Many weight loss medications act to increase satiety and lower caloric intake through the modulation of monoamine neurotransmitters [281]. Centrally acting agents increase satiety by modulating serotonin, noradrenaline, or dopamine in the hypothalamus to block catecholamine reuptake. There are two main classes of centrally acting appetite suppressants that are approved for short-term use: sympathomimetics and serotonergic agents.
Soon after DNP was withdrawn, amphetamine (benzedrine sulphate) was widely used to induce weight loss mainly by suppressing hunger [282–284]. Amphetamine was successful at inducing weight loss, but the highly addictive drug led to adverse psychiatric effects, physical dependence and abuse. Sympathomimetic agents were developed as phenylethylamine derivatives, which are structurally similar to amphetamine, but without the α-methylated side chain. These phenylethylamine derivatives are synthesized from tyrosine and act on hypothalamic and limbic regions of the brain to stimulate the release of norepinephrine and dopamine and increase satiety. In addition to increased satiety, certain sympathomimetic agents may also increase energy expenditure by stimulating β-adrenergic receptors and delaying gastric emptying, such as phentermine and mazindol [285]. The peripheral effects of sympathomimetic medications on skeletal muscle are relatively unexplored; however, there is evidence to suggest that amphetamines may enhance skeletal muscle thermogenesis. For example, rodents administered methamphetamine or 3,4-methylenedioxymethamphetamine (MDMA) exhibit increases in skeletal muscle temperature [286], and increases body temperature correlate with UCP3 protein expression [287]. Moreover, UCP3 knockout mice display have a blunted hyperthermic response to MDMA, and pharmacological blockade of SERCA-mediated Ca2+ release attenuates methamphetamine-induced hyperthermia [288]. Since diet-resistant obesity exists after controlling for compliance to a 900 kcal/day meal replacement diet [25], it is likely that appetite suppressants would not benefit DR obesity to a greater degree than DS obesity. However, the stimulation of β-adrenergic receptors by certain sympathomimetic agents could enhance skeletal muscle metabolism. In both rodent models and humans, individuals with increased susceptibility to weight gain often display decreased adrenergic-dependent thermogenic capacity [289].
Skeletal muscle also contains β1-, β2-, and β3-adrenergic receptors, with β2-adrenergic receptors as the predominate muscle isoform [290]. Density of β-adrenergic receptors is higher in type I fibers, and correlates with succinate dehydrogenase activity, a marker of oxidative capacity [291]. Similar to the mechanism in BAT, stimulation of β-adrenergic receptors in skeletal muscle activates PKA via cAMP, which in turn phosphorylates the RyR1, leading to SERCA-mediated Ca2+ release and generation of contractile force [292,293]. Pharmacological blockade of β-adrenergic receptors in humans is associated with decreases in isokinetic endurance and lower thermogenesis in vastus lateralis biopsies [294]. Β-adrenergic receptor agonism in humans is associated with increases in glucose uptake, lipolysis, and skeletal muscle hypertrophy, resulting from both increases in protein synthesis and suppression of catabolic pathways [295–297]. Evidence from in vitro studies suggests that β2-adrenergic receptor activation enhances mitochondrial function in myotubes [298]. However, chronic administration of β2-agonists is associated with increases in the proportions type II fibers in rodents, and decreases in oxidative enzyme activity [299,300], although there is some evidence that deleterious shifts in fiber type can be mitigated with low-intensity exercise [301]. In contrast, mirabegron, the β3-adrenergic receptor agonist, has recently been shown to promote fiber-type switching to type I fibers and reduces skeletal muscle content [302]. Altogether, activation of β-adrenergic receptors by sympathomimetic agents could theoretically enhance thermogenesis in individuals with diet-resistant obesity, particularly when combined with exercise to mitigate shifts in fiber type.
Thyroid hormones
Thyroid hormones have repeatedly been associated with body weight, BMI, and mitochondrial uncoupling thermogenesis [303–307]. Thyroid-stimulating hormone (TSH) is produced by the pituitary and acts as the primary signal for thyroxine (T4) and triiodothyronine (T3) production and release by follicular cells in the thyroid gland. T4 is an iodine-containing tyrosine-based precursor hormone that is converted into T3 [308]. T3 binds to nuclear thyroid hormone receptors (TR) to up-regulate the transcription of T3-responsive genes. T3 and T4 increase basal energy expenditure by modulating major metabolic pathways, augmenting ATP turnover, and uncoupling mitochondrial oxidative phosphorylation. Hypothyroidism has long been associated with weight gain; whereas patients with hyperthyroidism frequently present with weight loss, and thus, desiccated thyroid, thyroxine, and triiodothyronine have historically been used to treat obesity [309,310]. Similarly, weight loss is associated with small decreases in T3 [311–317]. Supplementation with triiodothyronine during a hypocaloric liquid diet has been shown to enhance diet-induced weight loss [318]. Similarly, patients with high baseline levels of free T3 achieve significantly greater weight loss following bariatric surgery [319]. The actions of thyroid hormones appear to be partially mediated by leptin [320].
In skeletal muscle, T3 also augments the expression and activity of Na+/K+ ATPase in the plasma membrane and the Ca2+ ATPase of the sarcoplasmic reticulum via SERCA1 [321,322]. In skeletal muscle, T3 also increases TCA cycle flux and promotes mitochondrial uncoupling [323]. Supplementation with 75 μg of T3 upregulates mRNA expression of UCP3 and adenine nucleotide translocases 1 and 2 in vastus lateralis muscle of healthy volunteers [324]. Moreover, thyroid hormones enhance lipolysis and mobilization of triglycerides from adipose tissue, and lipid utilization [325]. In rodents, hypothyroidism and hyperthyroidism cause decreased and increased levels, respectively, of ‘energy wasting’ mitochondrial proton leak in hepatocytes [326]. Induction of mitochondrial proton leak uncoupling in skeletal muscle by T3 occurs in vivo [323] and in ex vivo preparations [327–330]. Because DR obesity is associated with lower OXPHOS, decreased fatty acid utilization, and lower mitochondrial uncoupling, a greater understanding of the effects of thyroid hormones on muscle physiology in obesity, particularly DR obesity, is needed.
Diabetes medications: metformin, DPP4 inhibitors, and GLP-1
Metformin is well recognized as the first line of pharmacotherapy for patients with Type 2 diabetes and pre-diabetes. Despite its introduction in 1950, the exact mechanism of metformin is not fully understood, but many of its actions are associated with the activation of AMPK and inhibition of complex I [331]. Metformin does not directly activate AMPK [332] but rather is thought to modulate the ADP:ATP ratio leading to AMPK activation [331]. The AMPK-dependent actions of metformin have been linked to increases in skeletal muscle mitochondrial fatty acid oxidation and decreased expression of lipogenic genes [333,334]. Recent large-scale meta-analyses have revealed that metformin can reduce body weight and adiposity, which has been attributed to decreased energy intake [335,336].
Newer aged diabetes medications, Liraglutide and Semaglutide, are agonists of gut-derived peptide hormones (incretins) that have recently been approved in many countries for the treatment of obesity. Glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are incretins secreted by intestinal L and K cells into the hepatic portal system to facilitate glucose-stimulated insulin secretion [337,338]. Both GLP-1 and GIP are rapidly inactivated by the enzymatic removal of the N-terminal dipeptide by dipeptidyl peptidase 4 (DPP4) [339]. The discovery that GIP activity was impaired in patients with T2D [340] lead to the pharmacological development of incretin analogues, DPP4 inhibitors, and receptor agonists for the treatment of T2D. DPP4 inhibitors are generally considered to be weight neutral, as they are associated with variable results on body weight [341,342]. However, GLP-1 receptor agonists can elicit weight loss. GLP-1 receptors are involved in appetite regulation and are highly expressed in the brain [343]. As such, GLP-1 receptor agonism induces weight loss by enhancing satiety signals. Indeed, early investigative studies demonstrated that intravenous infusion with GLP-1 suppressed appetite, decreased energy intake and delayed gastric emptying in individuals with obesity and in healthy adults [344–347]. Clinical studies then demonstrated that GLP-1 receptor agonists, such as Liraglutide, can aid in weight loss when combined with a hypocaloric diet [348]. Specifically, when combined with behavioral therapy, GLP-1 agonists are associated with a 5–7% weight loss after 12 months, with greater success at higher doses. GLP-1 agonists exert anabolic effects within muscle and can ameliorate muscle atrophy to preserve lean body mass in mice [349], and have recently been shown to overcome anabolic resistance to feeding in older humans [350]. The appetite suppressing effects of GLP-1 agonists would likely confer equal benefits to DS and DR patients, while the anabolic effects of GLP-1 on skeletal may prove to be helpful in those with DR obesity.
Supplements
Dietary supplements hold appeal because they can be easier to implement than large lifestyle changes such as diet and exercise. Supplements are also often readily available without a prescription and many are associated with prodigious claims regarding weight loss. Approximately 15% of adults have reported using non-prescription dietary supplements to promote weight loss [351]. In contrast with the regulation of prescription medications, weight loss supplements often contain multiple ingredients and many clinical trials involving dietary supplements are often of poor methodological quality. Of the widely available supplements, there is some evidence to support weight-loss claims for caffeine, (L-)carnitine, conjugated linoleic acid, green tea, and ephedrine. However, meta-analyses have concluded that ephedrine [352,353] and conjugated linoleic acid [354] can elicit weight loss of less than 1 kg, for which the clinical relevance is low.
The effects of green tea on weight loss are attributed to caffeine and catechins in green tea. When examining the effects of green tea catechins on weight loss many studies are confounded by the presence of caffeine in green tea. While green tea extract has been shown to increase thermogenesis and fat oxidation in humans [355], meta-analyses have determined that green tea does not elicit significant weight loss [353,356]. However, caffeine has been associated with modest weight loss in both short-term clinical interventions and large-scale epidemiological studies. Caffeine is a methylxanthine that increases activity of the sympathetic nervous system by acting as a competitive inhibitor of phosphodiesterase and adenosine, leading to increases in intracellular cyclic adenosine monophosphate (cAMP) and suppressed catecholamine release. The resulting caffeine-induced increase in SNS activity has been shown to promote lipolysis, enhance fatty acid oxidation, and cause short-term increases in energy expenditure in a dose-dependent manner [357]. Caffeine is also an agonist of ryanodine receptors and can stimulate calcium ion flux in the sarcoplasmic reticulum [358], which may enhance skeletal muscle thermogenesis through increased SERCA activity [359]. Most clinical studies that have examined the efficacy of caffeine on weight loss have been of short duration and many have used caffeine in combination with other ingredients. However, a recent meta-analysis of RCTs determined that caffeine consumption is associated with an average weight loss of <2 kg after 4 weeks [360]. Moreover, data from cross sectional and observational studies have suggested that increased caffeine intake is associated with less weight gain in the long-term [361], and may also be beneficial for the maintenance of weight loss [362].
(L-)Carnitine (L-b-hydroxy-c-N-trimethylaminobutyric acid) is an endogenous compound that plays an essential role in lipid metabolism by acting as a cofactor to facilitate long-chain fatty acid transport across the mitochondrial membrane via formation of acylcarnitines [363]. L-Carnitine also participates in glucose metabolism by maintaining the acetyl-CoA/CoA ratio. Intramitochondrial free CoA availability increases as fatty acids are transported into mitochondria via carnitine, and the rise in free CoA stimulates the pyruvate dehydrogenase complex [364]. More than 95% of total carnitine is stored in skeletal muscle, and chronic oral L-carnitine with simultaneous carbohydrate ingestion can increase intramuscular carnitine concentrations and alter fuel utilization during exercise [365,366]. Meta-analyses of randomized controlled trials have concluded that L-carnitine is associated with modest weight loss between 1.2 and 1.3 kg and an average of 2.08 kg of fat mass [367,368]. We have previously demonstrated that patients with DR obesity have a lower capacity for fatty acid oxidation, reflected by less fatty acid-supported respiration in permeabilized skeletal muscle and greater circulating long- and medium-chained acylcarnitines following a high fat meal [46]. Thus, (L-)carnitine supplementation may enhance the capacity for fat oxidation.
Conclusion
The use of caloric restriction and very low-calorie diet programs often yield considerable variability in weight loss success. Understanding the metabolic processes that contribute to overall energy expenditure and adaptive thermogenesis will aid in the development of novel treatment strategies for individuals who fail to respond adequately to diets despite documented adherence. With new technologies and integrative -omics platforms, future research avenues may lead to more personalized lifestyle and nutrition treatment strategies for more effective weight loss outcomes. Interventions that enhance energy expenditure, mitochondrial biogenesis, and mitochondrial uncoupling may benefit individuals with diet-resistant obesity. Exercise is often given low priority in weight loss programs but is beneficial in offsetting adaptive decreases in metabolic rate. Cold exposure and sympathomimetic drugs (appetite suppressants) and supplements (caffeine and ephedrine) may stimulate β-adrenergic receptors to enhance energy expenditure during weight loss, but effects are generally modest. Mitochondrial uncouplers can be effective at enhancing energy expenditure through futile processes but most have deleterious side effects that currently limit their use. The development of new anti-obesity drugs and weight loss agents could exploit processes such as uncoupling in skeletal muscle to enhance energy expenditure.
Abbreviations
- ADP
adenosine diphosphate
- AMPK
AMP-activated protein kinase
- ANT
adenine nucleotide translocase
- ATP
adenosine triphosphate
- BAT
brown adipose tissue
- BMI
body mass index
- cAMP
cyclic adenosine monophosphate
- DNP
(2,4)-dinitrophenol
- DPP4
dipeptidyl peptidase 4
- DR
diet resistant
- DS
diet sensitive
- ETC
electron transport chain
- FFM
fat-free mass
- GIP
glucose-dependent insulinotropic polypeptide
- GLP-1
glucagon-like peptide-1
- MFN1/2
mitofusin 1 and 2
- NEAT
non-exercise activity thermogenesis
- OXPHOS
oxidative phosphorylation
- PGC1α
peroxisome proliferator-activated receptor (PPAR)-γ coactivator-1α
- PMF
protonmotive force
- RER
respiratory exchange ratio
- RMR
resting metabolic rate
- ROS
reactive oxygen species
- RQ
respiratory quotient
- RYGB
Roux-en-Y gastric bypass
- SERCA
sarcoplasmic/endoplasmic reticulum Ca2+ ATPase
- SNS
sympathetic nervous system
- T2D
type 2 diabetes
- TSH
thyroid-stimulating hormone
- UCP
uncoupling protein
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
Funding
CIHR FDN-143278 (M.E.H.).
CRediT Author Contribution
Chantal A. Pileggi: Visualization, Writing—original draft, Writing—review & editing. Breana G. Hooks: Visualization, Writing—original draft, Writing—review & editing. Ruth McPherson: Writing—original draft, Writing—review & editing. Robert Dent: Writing—original draft, Writing—review & editing. Mary-Ellen Harper: Writing—original draft, Writing—review & editing.
References
- 1.GBD 2015 Obesity Collaborators (2017) Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 377, 13–27 10.1056/NEJMoa1614362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.NCD Risk Factor Collaboration (NCD-RisC) (2016) Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 387, 1377–1396 10.1016/S0140-6736(16)30054-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Abarca-Gómez L., Abdeen Z.A., Hamid Z.A., Abu-Rmeileh N.M., Acosta-Cazares B., Acuin C.et al. (2017) Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet 390, 2627–2642 10.1016/S0140-6736(17)32129-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pi-Sunyer X. (2009) The medical risks of obesity. Postgrad. Med. 121, 21–33 10.3810/pgm.2009.11.2074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Twig G., Yaniv G., Levine H., Leiba A., Goldberger N., Derazne E.et al. (2016) Body-mass index in 2.3 million adolescents and cardiovascular death in adulthood. N. Engl. J. Med. 374, 2430–2440 10.1056/NEJMoa1503840 [DOI] [PubMed] [Google Scholar]
- 6.Bjerregaard L.G., Jensen B.W., Ängquist L., Osler M., Sørensen T.I.A. and Baker J.L. (2018) Change in overweight from childhood to early adulthood and risk of type 2 diabetes. N. Engl. J. Med. 78, 1302–1312 10.1056/NEJMoa1713231 [DOI] [PubMed] [Google Scholar]
- 7.Berrington de Gonzalez A., Hartge P., Cerhan J.R., Flint A.J., Hannan L., MacInnis R.J.et al. (2010) Body-mass index and mortality among 1.46 million white adults. N. Engl. J. Med. 363, 2211–2219 10.1056/NEJMoa1000367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Calle E.E. and Thun M.J. (2004) Obesity and cancer. Oncogene 23, 6365–6378 10.1038/sj.onc.1207751 [DOI] [PubMed] [Google Scholar]
- 9.Arnold M., Pandeya N., Byrnes G., Renehan A.G., Stevens G.A., Ezzati M.et al. (2015) Global burden of cancer attributable to high body-mass index in 2012: a population-based study. Lancet Oncol. 16, 36–46 10.1016/S1470-2045(14)71123-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fontaine K.R., Redden D.T., Wang C., Westfall A.O. and Allison D.B. (2003) Years of life lost due to obesity. JAMA 289, 187–193 10.1001/jama.289.2.187 [DOI] [PubMed] [Google Scholar]
- 11.Prospective Studies Collaboration (2009) Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 373, 1083–1096 10.1016/S0140-6736(09)60318-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dee A., Kearns K., O'Neill C., Sharp L., Staines A., O'Dwyer V.et al. (2014) The direct and indirect costs of both overweight and obesity: a systematic review. BMC Res. Notes 7, 1–9 10.1186/1756-0500-7-242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Specchia M.L., Veneziano M.A., Cadeddu C., Ferriero A.M., Mancuso A., Ianuale C.et al. (2015) Economic impact of adult obesity on health systems: a systematic review. Eur. J. Public Health 25, 255–262 10.1093/eurpub/cku170 [DOI] [PubMed] [Google Scholar]
- 14.Tremmel M., Gerdtham U.-G., Nilsson P.M. and Saha S. (2017) Economic burden of obesity: a systematic literature review. Int. J. Environ. Res. Public Health 14, 435 10.3390/ijerph14040435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wing R.R., Lang W., Wadden T.A., Safford M., Knowler W.C., Bertoni A.G.et al. (2011) Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care 34, 1481–1486 10.2337/dc10-2415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bailey-Davis L., Wood G.C., Benotti P., Cook A., Dove J., Mowery J.et al. (2022) Impact of sustained weight loss on cardiometabolic outcomes. Am. J. Cardiol. 162, 66–72 10.1016/j.amjcard.2021.09.018 [DOI] [PubMed] [Google Scholar]
- 17.Carlsson L.M.S., Sjöholm K., Jacobson P., Andersson-Assarsson J.C., Svensson P.-A., Taube M.et al. (2020) Life expectancy after bariatric surgery in the Swedish obese subjects study. N. Engl. J. Med. 383, 1535–1543 10.1056/NEJMoa2002449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kruger J., Galuska D.A., Serdula M.K. and Jones D.A. (2004) Attempting to lose weight: specific practices among US adults. Am. J. Prev. Med. 26, 402–406 10.1016/j.amepre.2004.02.001 [DOI] [PubMed] [Google Scholar]
- 19.Serdula M.K., Collins M.E., Williamson D.F., Anda R.F., Pamuk E. and Byers T.E. (1993) Weight control practices of US adolescents and adults. Ann. Intern. Med. 119, 667–671 10.7326/0003-4819-119-7_Part_2-199310011-00008 [DOI] [PubMed] [Google Scholar]
- 20.Serdula M.K., Mokdad A.H., Williamson D.F., Galuska D.A., Mendlein J.M. and Heath G.W. (1999) Prevalence of attempting weight loss and strategies for controlling weight. JAMA 282, 1353–1358 10.1001/jama.282.14.1353 [DOI] [PubMed] [Google Scholar]
- 21.American College of Cardiology/American Heart Association Task Force on Practice Guidelines, Obesity Expert Panel (2014) Expert Panel Report: Guidelines (2013) for the management of overweight and obesity in adults. Obesity (Silver Spring) 22, S41–S410 10.1002/oby.20660 [DOI] [PubMed] [Google Scholar]
- 22.Steinbrook R. (2004) Surgery for severe obesity. N. Engl. J. Med. 350, 1075–1079 10.1056/NEJMp048029 [DOI] [PubMed] [Google Scholar]
- 23.Adams T.D., Gress R.E., Smith S.C., Halverson R.C., Simper S.C., Rosamond W.D.et al. (2007) Long-term mortality after gastric bypass surgery. N. Engl. J. Med. 357, 753–761 10.1056/NEJMoa066603 [DOI] [PubMed] [Google Scholar]
- 24.Smith M.D., Patterson E., Wahed A.S., Belle S.H., Berk P.D., Courcoulas A.P.et al. (2011) Thirty-day mortality after bariatric surgery: independently adjudicated causes of death in the longitudinal assessment of bariatric surgery. Obes. Surg. 21, 1687–1692 10.1007/s11695-011-0497-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Harper M.-E., Dent R., Monemdjou S., Bézaire V., Van Wyck L., Wells G.et al. (2002) Decreased mitochondrial proton leak and reduced expression of uncoupling protein 3 in skeletal muscle of obese diet-resistant women. Diabetes 51, 2459–2466 10.2337/diabetes.51.8.2459 [DOI] [PubMed] [Google Scholar]
- 26.King N.A., Hopkins M., Caudwell P., Stubbs R.J. and Blundell J.E. (2008) Individual variability following 12 weeks of supervised exercise: identification and characterization of compensation for exercise-induced weight loss. Int. J. Obes. 32, 177–184 10.1038/sj.ijo.0803712 [DOI] [PubMed] [Google Scholar]
- 27.Gerrits M.F., Ghosh S., Kavaslar N., Hill B., Tour A., Seifert E.L.et al. (2010) Distinct skeletal muscle fiber characteristics and gene expression in diet-sensitive versus diet-resistant obesity. J. Lipid Res. 51, 2394–2404 10.1194/jlr.P005298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wishnofsky M. (1958) Caloric equivalents of gained or lost weight. Am. J. Clin. Nutr. 6, 542–546 10.1093/ajcn/6.5.542 [DOI] [PubMed] [Google Scholar]
- 29.Lichtman S.W., Pisarska K., Berman E.R., Pestone M., Dowling H., Offenbacher E.et al. (1992) Discrepancy between self-reported and actual caloric intake and exercise in obese subjects. N. Engl. J. Med. 327, 1893–1898 10.1056/NEJM199212313272701 [DOI] [PubMed] [Google Scholar]
- 30.Scagliusi F.B., Polacow V.O., Artioli G.G., Benatti F.B. and Lancha A.H. Jr (2003) Selective underreporting of energy intake in women: magnitude, determinants, and effect of training. J. Am. Diet. Assoc. 103, 1306–1313 10.1016/S0002-8223(03)01074-5 [DOI] [PubMed] [Google Scholar]
- 31.Fricker J., Baelde D., Igoin-Apfelbaum L., Huet J.-M. and Apfelbaum M. (1992) Underreporting of food intake in obese “small eaters”. Appetite 19, 273–283 10.1016/0195-6663(92)90167-5 [DOI] [PubMed] [Google Scholar]
- 32.Brewis A.A. (2010) Big-body symbolism meanings, and norms. Obesity: Cultural and Biocultural Perspectives, pp. 99–124, Rutgers University Press [Google Scholar]
- 33.Thomas D.M., Gonzalez M.C., Pereira A.Z., Redman L.M. and Heymsfield S.B. (2014) Time to correctly predict the amount of weight loss with dieting. J. Acad. Nutr. Diet. 114, 857–861 10.1016/j.jand.2014.02.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hall K.D., Sacks G., Chandramohan D., Chow C.C., Wang Y.C., Gortmaker S.L.et al. (2011) Quantification of the effect of energy imbalance on bodyweight. Lancet 378, 826–837 10.1016/S0140-6736(11)60812-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bouchard C., Tremblay A., Després J.-P., Nadeau A., Lupien P.J., Thériault G.et al. (1990) The response to long-term overfeeding in identical twins. N. Engl. J. Med. 322, 1477–1482 10.1056/NEJM199005243222101 [DOI] [PubMed] [Google Scholar]
- 36.Bouchard C., Tremblay A., Després J., Thériault G., Nadeauf A., Lupien P.J.et al. (1994) The response to exercise with constant energy intake in identical twins. Obes. Res. 2, 400–410 10.1002/j.1550-8528.1994.tb00087.x [DOI] [PubMed] [Google Scholar]
- 37.Stunkard A.J., Sørensen T.I.A., Hanis C., Teasdale T.W., Chakraborty R., Schull W.J.et al. (1986) An adoption study of human obesity. N. Engl. J. Med. 314, 193–198 10.1056/NEJM198601233140401 [DOI] [PubMed] [Google Scholar]
- 38.Locke A.E., Kahali B., Berndt S.I., Justice A.E., Pers T.H., Day F.R.et al. (2015) Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206 10.1038/nature14177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bouchard C. (2021) Genetics of obesity: what we have learned over decades of research. Obesity 29, 802–820 10.1002/oby.23116 [DOI] [PubMed] [Google Scholar]
- 40.Maes H.H.M., Neale M.C. and Eaves L.J. (1997) Genetic and environmental factors in relative body weight and human adiposity. Behav. Genet. 27, 325–351 10.1023/A:1025635913927 [DOI] [PubMed] [Google Scholar]
- 41.Loos R.J.F. (2018) The genetics of adiposity. Curr. Opin. Genet. Dev. 50, 86–95 10.1016/j.gde.2018.02.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Khera A.V., Chaffin M., Wade K.H., Zahid S., Brancale J., Xia R.et al. (2019) Polygenic prediction of weight and obesity trajectories from birth to adulthood. Cell 177, 587–596 10.1016/j.cell.2019.03.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Slatkin M. (2008) Linkage disequilibrium—understanding the evolutionary past and mapping the medical future. Nat. Rev. Genet. 9, 477–485 10.1038/nrg2361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nikpay M., Stewart A.F.R. and McPherson R. (2017) Partitioning the heritability of coronary artery disease highlights the importance of immune-mediated processes and epigenetic sites associated with transcriptional activity. Cardiovasc. Res. 113, 973–983 10.1093/cvr/cvx019 [DOI] [PubMed] [Google Scholar]
- 45.Yang J., Bakshi A., Zhu Z., Hemani G., Vinkhuyzen A.A.E., Lee S.H.et al. (2015) Genetic variance estimation with imputed variants finds negligible missing heritability for human height and body mass index. Nat. Genet. 47, 1114–1120 10.1038/ng.3390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Thrush A.B., Antoun G., Nikpay M., Patten D.A., DeVlugt C., Mauger J.F.et al. (2018) Diet-resistant obesity is characterized by a distinct plasma proteomic signature and impaired muscle fiber metabolism. Int. J. Obes. 42, 353–362 10.1038/ijo.2017.286 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Thrush A.B., Zhang R., Chen W., Seifert E.L., Quizi J.K., McPherson R.et al. (2014) Lower mitochondrial proton leak and decreased glutathione redox in primary muscle cells of obese diet-resistant versus diet-sensitive humans. J. Clin. Endocrinol. Metab. 99, 4223–4230 10.1210/jc.2014-1726 [DOI] [PubMed] [Google Scholar]
- 48.Azar M., Nikpay M., Harper M., McPherson R. and Dent R. (2016) Can response to dietary restriction predict weight loss after R oux-en-Y gastroplasty? Obesity (Silver Spring) 24, 805–811 10.1002/oby.21395 [DOI] [PubMed] [Google Scholar]
- 49.Ravussin E., Lillioja S., Knowler W.C., Christin L., Freymond D., Abbott W.G.H.et al. (1988) Reduced rate of energy expenditure as a risk factor for body-weight gain. N. Engl. J. Med. 318, 467–472 10.1056/NEJM198802253180802 [DOI] [PubMed] [Google Scholar]
- 50.Ravussin E., Lillioja S., Anderson T.E., Christin L. and Bogardus C. (1986) Determinants of 24-hour energy expenditure in man. Methods and results using a respiratory chamber. J. Clin. Invest. 78, 1568–1578 10.1172/JCI112749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Webb P. (1981) Energy expenditure and fat-free mass in men and women. Am. J. Clin. Nutr. 34, 1816–1826 10.1093/ajcn/34.9.1816 [DOI] [PubMed] [Google Scholar]
- 52.Leibel R.L., Rosenbaum M. and Hirsch J. (1995) Changes in energy expenditure resulting from altered body weight. N. Engl. J. Med. 332, 621–628 10.1056/NEJM199503093321001 [DOI] [PubMed] [Google Scholar]
- 53.Levine J.A., Schleusner S.J. and Jensen M.D. (2000) Energy expenditure of nonexercise activity. Am. J. Clin. Nutr. 72, 1451–1454 10.1093/ajcn/72.6.1451 [DOI] [PubMed] [Google Scholar]
- 54.Pontzer H., Yamada Y., Sagayama H., Ainslie P.N., Andersen L.F., Anderson L.J.et al. (2021) Daily energy expenditure through the human life course. Science 373, 808–812 10.1126/science.abe5017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Fontaine E., Savard R., Tremblay A., Despres J.P., Poehlman E. and Bouchard C. (1985) Resting metabolic rate in monozygotic and dizygotic twins. Acta Genet. Medicae Gemellol Twin Res. 34, 41–47 10.1017/S0001566000004906 [DOI] [PubMed] [Google Scholar]
- 56.Hohenadel M.G., Hollstein T., Thearle M., Reinhardt M., Piaggi P., Salbe A.D.et al. (2019) A low resting metabolic rate in late childhood is associated with weight gain in adolescence. Metabolism 93, 68–74 10.1016/j.metabol.2018.12.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.James W.P.T., Bailes J., Davies H.L. and Dauncey M.J. (1978) Elevated metabolic rates in obesity. Lancet 311, 1122–1125 10.1016/S0140-6736(78)90300-8 [DOI] [PubMed] [Google Scholar]
- 58.Ravussin E., Burnand B., Schutz Y. and Jequier E. (1982) Twenty-four-hour energy expenditure and resting metabolic rate in obese, moderately obese, and control subjects. Am. J. Clin. Nutr. 35, 566–573 10.1093/ajcn/35.3.566 [DOI] [PubMed] [Google Scholar]
- 59.Prentice A.M., Black A.E., Coward W.A., Davies H.L., Goldberg G.R., Murgatroyd P.R.et al. (1986) High levels of energy expenditure in obese women. Br. Med. J. (Clin. Res. Ed) 292, 983–987 10.1136/bmj.292.6526.983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Prentice A.M., Black A.E., Coward W.A. and Cole T.J. (1996) Energy expenditure in overweight and obese adults in affluent societies: an analysis of 319 doubly-labelled water measurements. Eur. J. Clin. Nutr. 50, 93–97 [PubMed] [Google Scholar]
- 61.Rosenbaum M. and Leibel R.L. (2010) Adaptive thermogenesis in humans. Int. J. Obes. 34, S47–S55 10.1038/ijo.2010.184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Müller M.J. and Bosy-Westphal A. (2013) Adaptive thermogenesis with weight loss in humans. Obesity 21, 218–228 10.1002/oby.20027 [DOI] [PubMed] [Google Scholar]
- 63.Taylor H.L., Keys A., Hylander C.J., Meyerhoff H.A. and Taylor R.L. (1950) Adapting to caloric restriction. Science 112, 215–218 10.1126/science.112.2904.215 [DOI] [PubMed] [Google Scholar]
- 64.Doucet E., St-Pierre S., Alméras N., Després J.-P., Bouchard C. and Tremblay A. (2001) Evidence for the existence of adaptive thermogenesis during weight loss. Br. J. Nutr. 85, 715–723 10.1079/BJN2001348 [DOI] [PubMed] [Google Scholar]
- 65.Rosenbaum M., Hirsch J., Gallagher D.A. and Leibel R.L. (2008) Long-term persistence of adaptive thermogenesis in subjects who have maintained a reduced body weight. Am. J. Clin. Nutr. 88, 906–912 10.1093/ajcn/88.4.906 [DOI] [PubMed] [Google Scholar]
- 66.Fothergill E., Guo J., Howard L., Kerns J.C., Knuth N.D., Brychta R.et al. (2016) Persistent metabolic adaptation 6 years after “The Biggest Loser” competition. Obesity 24, 1612–1619 10.1002/oby.21538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Astrup A., Gøtzsche P.C., van de Werken K., Ranneries C., Toubro S., Raben A.et al. (1999) Meta-analysis of resting metabolic rate in formerly obese subjects. Am. J. Clin. Nutr. 69, 1117–1122 10.1093/ajcn/69.6.1117 [DOI] [PubMed] [Google Scholar]
- 68.De Peuter R., Withers R.T., Brinkman M., Tomas F.M. and Clark D.G. (1992) No differences in rates of energy expenditure between post-obese women and their matched, lean controls. Int. J. Obes. Relat. Metab. Disord. 16, 801–808 [PubMed] [Google Scholar]
- 69.Goldberg G.R., Black A.E., Prentice A.M. and Coward W. (1991) No evidence of lower energy expenditure in post-obese women. Proc. Nutr. Soc. 50, 109A, [Abstract] [Google Scholar]
- 70.Hollstein T., Ando T., Basolo A., Krakoff J., Votruba S.B. and Piaggi P. (2019) Metabolic response to fasting predicts weight gain during low-protein overfeeding in lean men: further evidence for spendthrift and thrifty metabolic phenotypes. Am. J. Clin. Nutr. 110, 593–604 10.1093/ajcn/nqz062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schlögl M., Piaggi P., Pannacciuli N., Bonfiglio S.M., Krakoff J. and Thearle M.S. (2015) Energy expenditure responses to fasting and overfeeding identify phenotypes associated with weight change. Diabetes 64, 3680–3689 10.2337/db15-0382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hollstein T., Basolo A., Ando T., Krakoff J. and Piaggi P. (2021) Reduced adaptive thermogenesis during acute protein-imbalanced overfeeding is a metabolic hallmark of the human thrifty phenotype. Am. J. Clin. Nutr. 114, 1396–1407 10.1093/ajcn/nqab209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Whytock K.L., Corbin K.D., Parsons S.A., Pachori A., Bock C.P., Jones K.P.et al. (2021) Metabolic adaptation characterizes short-term resistance to weight loss induced by a low-calorie diet in overweight/obese individuals. Am. J. Clin. Nutr. 114, 267–280 10.1093/ajcn/nqab027 [DOI] [PubMed] [Google Scholar]
- 74.Tremblay A., Royer M.M., Chaput J.P. and Doucet E. (2013) Adaptive thermogenesis can make a difference in the ability of obese individuals to lose body weight. Int. J. Obes. 37, 759–764 10.1038/ijo.2012.124 [DOI] [PubMed] [Google Scholar]
- 75.Reinhardt M., Thearle M.S., Ibrahim M., Hohenadel M.G., Bogardus C., Krakoff J.et al. (2015) A human thrifty phenotype associated with less weight loss during caloric restriction. Diabetes 64, 2859–2867 10.2337/db14-1881 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Chaston T.B., Dixon J.B. and O'Brien P.E. (2007) Changes in fat-free mass during significant weight loss: a systematic review. Int. J. Obes. 31, 743–750 10.1038/sj.ijo.0803483 [DOI] [PubMed] [Google Scholar]
- 77.Zurlo F., Larson K., Bogardus C. and Ravussin E. (1990) Skeletal muscle metabolism is a major determinant of resting energy expenditure. J. Clin. Invest. 86, 1423–1427 10.1172/JCI114857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Elia M. (1992) Organ and tissue contribution to metabolic rate. Energy Metabolism: Tissue Determinants and Cellular Corollaries, pp. 61–80, Raven Press, New York, pp. [Google Scholar]
- 79.Thiebaud D., Jacot E., DeFronzo R.A., Maeder E., Jequier E. and Felber J.P. (1982) The effect of graded doses of insulin on total glucose uptake, glucose oxidation, and glucose storage in man. Diabetes 31, 957–963 10.2337/diacare.31.11.957 [DOI] [PubMed] [Google Scholar]
- 80.Martin B.C., Warram J.H., Krolewski A.S., Soeldner J.S., Kahn C.R., Martin B.C.et al. (1992) Role of glucose and insulin resistance in development of type 2 diabetes mellitus: results of a 25-year follow-up study. Lancet 340, 925–929 10.1016/0140-6736(92)92814-V [DOI] [PubMed] [Google Scholar]
- 81.Wolfe R.R. (2006) The underappreciated role of muscle in health and disease. Am. J. Clin. Nutr. 84, 475–482 10.1093/ajcn/84.3.475 [DOI] [PubMed] [Google Scholar]
- 82.Hood D.A., Irrcher I., Ljubicic V. and Joseph A.-M. (2006) Coordination of metabolic plasticity in skeletal muscle. J. Exp. Biol. 209, 2265–2275 10.1242/jeb.02182 [DOI] [PubMed] [Google Scholar]
- 83.Nicholls D.G. (1974) The influence of respiration and ATP hydrolysis on the proton-electrochemical gradient across the inner membrane of rat-liver mitochondria as determined by ion distribution. Eur. J. Biochem. 50, 305–315 10.1111/j.1432-1033.1974.tb03899.x [DOI] [PubMed] [Google Scholar]
- 84.Lambert A.J. and Brand M.D. (2004) Superoxide production by NADH: ubiquinone oxidoreductase (complex I) depends on the pH gradient across the mitochondrial inner membrane. Biochem. J. 382, 511–517 10.1042/BJ20040485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chance B.W.G.R. (1955) Respiratory enzymes in oxidative phosphorylation. J. Biol. Chem. 217, 383–395 10.1016/S0021-9258(19)57189-7 [DOI] [PubMed] [Google Scholar]
- 86.Brand M.D., Pakay J.L., Ocloo A., Kokoszka J., Wallace D.C., Brookes P.S.et al. (2005) The basal proton conductance of mitochondria depends on adenine nucleotide translocase content. Biochem. J. 392, 353–362 10.1042/BJ20050890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Fedorenko A., Lishko P.V. and Kirichok Y. (2012) Mechanism of fatty-acid-dependent UCP1 uncoupling in brown fat mitochondria. Cell 151, 400–413 10.1016/j.cell.2012.09.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Carpentier A.C., Blondin D.P., Virtanen K.A., Richard D., Haman F. and Turcotte É.E. (2018) Brown adipose tissue energy metabolism in humans. Front. Endocrinol. (Lausanne) 9, 447 10.3389/fendo.2018.00447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zingaretti M.C., Crosta F., Vitali A., Guerrieri M., Frontini A., Cannon B.et al. (2009) The presence of UCP1 demonstrates that metabolically active adipose tissue in the neck of adult humans truly represents brown adipose tissue. FASEB J. 23, 3113–3120 10.1096/fj.09-133546 [DOI] [PubMed] [Google Scholar]
- 90.Becher T., Palanisamy S., Kramer D.J., Eljalby M., Marx S.J., Wibmer A.G.et al. (2021) Brown adipose tissue is associated with cardiometabolic health. Nat. Med. 27, 58–65 10.1038/s41591-020-1126-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Brand M.D., Chien L.-F., Ainscow E.K., Rolfe D.F.S. and Porter R.K. (1994) The causes and functions of mitochondrial proton leak. Biochim. Biophys. Acta-Bioenerg. 1187, 132–139 10.1016/0005-2728(94)90099-X [DOI] [PubMed] [Google Scholar]
- 92.Bevilacqua L., Seifert E.L., Estey C., Gerrits M.F. and Harper M.-E. (2010) Absence of uncoupling protein-3 leads to greater activation of an adenine nucleotide translocase-mediated proton conductance in skeletal muscle mitochondria from calorie restricted mice. Biochim. Biophys. Acta-Bioenerg. 1797, 1389–1397 10.1016/j.bbabio.2010.02.018 [DOI] [PubMed] [Google Scholar]
- 93.Bertholet A.M., Chouchani E.T., Kazak L., Angelin A., Fedorenko A., Long J.Z.et al. (2019) H+ transport is an integral function of the mitochondrial ADP/ATP carrier. Nature 571, 515–520 10.1038/s41586-019-1400-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bertholet A.M. and Kirichok Y. (2022) Mitochondrial H+ leak and thermogenesis. Annu. Rev. Physiol. 84, 381–407 10.1146/annurev-physiol-021119-034405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bernardi P., Rasola A., Forte M. and Lippe G. (2015) The mitochondrial permeability transition pore: channel formation by F-ATP synthase, integration in signal transduction, and role in pathophysiology. Physiol. Rev. 95, 1111–1155 10.1152/physrev.00001.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Bertholet A.M., Natale A.M., Bisignano P., Suzuki J., Fedorenko A., Hamilton J.et al. (2022) Mitochondrial uncouplers induce proton leak by activating AAC and UCP1. Nature 606, 180–187 10.1038/s41586-022-04747-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Andreyev A.Y., Bondareva T.O., Dedukhova V.I., Mokhova E.N., Skulachev V.P., Tsofina L.M.et al. (1989) The ATP/ADP-antiporter is involved in the uncoupling effect of fatty acids on mitochondria. Eur. J. Biochem. 182, 585–592 10.1111/j.1432-1033.1989.tb14867.x [DOI] [PubMed] [Google Scholar]
- 98.Queiroga C.S.F., Almeida A.S., Martel C., Brenner C., Alves P.M. and Vieira H.L.A.. Glutathionylation of adenine nucleotide translocase induced by carbon monoxide prevents mitochondrial membrane permeabilization and apoptosis. J. Biol. Chem. 285(22), 17077–17088 10.1074/jbc.M109.065052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Mielke C., Lefort N., McLean C.G., Cordova J.M., Langlais P.R., Bordner A.J.et al. (2014) Adenine nucleotide translocase is acetylated in vivo in human muscle: modeling predicts a decreased ADP affinity and altered control of oxidative phosphorylation. Biochemistry 53, 3817–3829 10.1021/bi401651e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Rolfe D.F.S. and Brand M.D. (1996) Contribution of mitochondrial proton leak to skeletal muscle respiration and to standard metabolic rate. Am. J. Physiol. - Cell Physiol. 271, C1380–C1389 10.1152/ajpcell.1996.271.4.C1380 [DOI] [PubMed] [Google Scholar]
- 101.Rolfe D.F.S. and Brand M.D. (1997) The physiological significance of mitochondrial proton leak in animal cells and tissues. Biosci. Rep. 17, 9–16 10.1023/A:1027327015957 [DOI] [PubMed] [Google Scholar]
- 102.Harper M.-E., Green K. and Brand M.D. (2008) The efficiency of cellular energy transduction and its implications for obesity. Annu. Rev. Nutr. 28, 13–33 10.1146/annurev.nutr.28.061807.155357 [DOI] [PubMed] [Google Scholar]
- 103.Ritov V.B., Menshikova E.V., He J., Ferrell R.E., Goodpaster B.H. and Kelley D.E. (2005) Deficiency of subsarcolemmal mitochondria in obesity and type 2 diabetes. Diabetes 54, 8–14 10.2337/diabetes.54.1.8 [DOI] [PubMed] [Google Scholar]
- 104.Kelley D.E., He J., Menshikova E.V. and Ritov V.B. (2002) Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes. Diabetes 51, 2944–2950 10.2337/diabetes.51.10.2944 [DOI] [PubMed] [Google Scholar]
- 105.Simoneau J.A., Veerkamp J.H., Turcotte L.P. and Kelley D.E. (1999) Markers of capacity to utilize fatty acids in human skeletal muscle: relation to insulin resistance and obesity and effects of weight loss. FASEB J. 13, 2051–2060 10.1096/fasebj.13.14.2051 [DOI] [PubMed] [Google Scholar]
- 106.Anderson E.J., Lustig M.E., Boyle K.E., Woodlief T.L., Kane D.A., Lin C.-T.et al. (2009) Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J. Clin. Invest. 119, 573–581 10.1172/JCI37048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kelley D.E., Goodpaster B., Wing R.R. and Simoneau J.A. (1999) Skeletal muscle fatty acid metabolism in association with insulin resistance, obesity, and weight loss. Am. J. Physiol. 277, E1130–E1141 10.1152/ajpendo.1999.277.6.E1130 [DOI] [PubMed] [Google Scholar]
- 108.Kim J.Y., Hickner R.C., Cortright R.L., Dohm G.L. and Houmard J.A. (2000) Lipid oxidation is reduced in obese human skeletal muscle. Am. J. Physiol. Metab. 279, E1039–E1044 10.1152/ajpendo.2000.279.5.E1039 [DOI] [PubMed] [Google Scholar]
- 109.Simoneau J.A., Colberg S.R., Thaete F.L. and Kelley D.E. (1995) Skeletal muscle glycolytic and oxidative enzyme capacities are determinants of insulin sensitivity and muscle composition in obese women. FASEB J. 9, 273–278 10.1096/fasebj.9.2.7781930 [DOI] [PubMed] [Google Scholar]
- 110.Colberg S., Simoneau J.A., Thaete F.L. and Kelley D.E. (1995) Impaired FFA utilization by skeletal muscle in women with visceral obesity. J. Clin. Invest. 95, 1846–1853 10.1172/JCI117864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Goodpaster B.H., He J., Watkins S. and Kelley D.E. (2001) Skeletal muscle lipid content and insulin resistance: evidence for a paradox in endurance-trained athletes. J. Clin. Endocrinol. Metab. 86, 5755–5761 10.1210/jcem.86.12.8075 [DOI] [PubMed] [Google Scholar]
- 112.Koves T.R., Ussher J.R., Noland R.C., Slentz D., Mosedale M., Ilkayeva O.et al. (2008) Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 7, 45–56 10.1016/j.cmet.2007.10.013 [DOI] [PubMed] [Google Scholar]
- 113.Pileggi C.A., Parmar G. and Harper M. (2021) The lifecycle of skeletal muscle mitochondria in obesity. Obes. Rev. 22, e13164 10.1111/obr.13164 [DOI] [PubMed] [Google Scholar]
- 114.Tanner C.J., Barakat H.A., Dohm G.L., Pories W.J., MacDonald K.G., Cunningham P.R.G.et al. (2002) Muscle fiber type is associated with obesity and weight loss. Am. J. Physiol. Metab. 282, E1191–E1196 10.1152/ajpendo.00416.2001 [DOI] [PubMed] [Google Scholar]
- 115.Hickey M.S., Carey J.O., Azevedo J.L., Houmard J.A., Pories W.J., Israel R.G.et al. (1995) Skeletal muscle fiber composition is related to adiposity and in vitro glucose transport rate in humans. Am. J. Physiol. Metab. 268, E453–E457 10.1152/ajpendo.1995.268.3.E453 [DOI] [PubMed] [Google Scholar]
- 116.Wade A.J., Marbut M.M. and Round J.M. (1990) Muscle fibre type and aetiology of obesity. Lancet 335, 805–808 10.1016/0140-6736(90)90933-V [DOI] [PubMed] [Google Scholar]
- 117.Helge J.W., Fraser A.M., Kriketos A.D., Jenkins A.B., Calvert G.D., Ayre K.J.et al. (1999) Interrelationships between muscle fibre type, substrate oxidation and body fat. Int. J. Obes. Relat. Metab. Disord. 23, 986–991 10.1038/sj.ijo.0801030 [DOI] [PubMed] [Google Scholar]
- 118.Sun G., Ukkola O., Rankinen T., Joanisse D.R. and Bouchard C. (2002) Skeletal muscle characteristics predict body fat gain in response to overfeeding in never-obese young men. Metab. Exp. 51, 451–456 10.1053/meta.2002.31324 [DOI] [PubMed] [Google Scholar]
- 119.Karjalainen J., Tikkanen H., Hernelahti M. and Kujala U.M. (2006) Muscle fiber-type distribution predicts weight gain and unfavorable left ventricular geometry: A 19 year follow-up study. BMC Cardiovasc. Disord. 6, 1–8 10.1186/1471-2261-6-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Damer A., Meniaway S., McPherson R., Wells G. and Harper M.-E.D.R. (2022) Association of Muscle Fibre Type with Measures of Obesity: A Systematic Review. Obes. Rev. 23, e13444 10.1111/obr.13444 [DOI] [PubMed] [Google Scholar]
- 121.Ghosh S., Dent R., Harper M.E., Stuart J. and McPherson R. (2011) Blood gene expression reveal pathway differences between diet-sensitive and resistant obese subjects prior to caloric restriction. Obesity 19, 457–463, 10.1038/oby.2010.209 10.1038/oby.2010.209 [DOI] [PubMed] [Google Scholar]
- 122.Aguer C., Fiehn O., Seifert E.L., Bezaire V., Meissen J.K., Daniels A.et al. (2013) Muscle uncoupling protein 3 overexpression mimics endurance training and reduces circulating biomarkers of incomplete beta-oxidation. FASEB J. 27, 4213–4225 10.1096/fj.13-234302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Costford S.R., Chaudhry S.N., Salkhordeh M. and Harper M.-E. (2006) Effects of the presence, absence, and overexpression of uncoupling protein-3 on adiposity and fuel metabolism in congenic mice. Am. J. Physiol. Metab. 290, E1304–E1312 10.1152/ajpendo.00401.2005 [DOI] [PubMed] [Google Scholar]
- 124.Bezaire V., Spriet L.L., Campbell S., Sabet N., Gerrits M., Bonen A.et al. (2005) Constitutive UCP3 overexpression at physiological levels increases mouse skeletal muscle capacity for fatty acid transport and oxidation. FASEB J. 19, 977–979 10.1096/fj.04-2765fje [DOI] [PubMed] [Google Scholar]
- 125.Wang S., Subramaniam A., Cawthorne M.A. and Clapham J.C. (2003) Increased fatty acid oxidation in transgenic mice overexpressing UCP3 in skeletal muscle. Diabetes Obes. Metab. 5, 295–301 10.1046/j.1463-1326.2003.00273.x [DOI] [PubMed] [Google Scholar]
- 126.Argyropoulos G., Brown A.M., Willi S.M., Zhu J., He Y., Reitman M.et al. (1998) Effects of mutations in the human uncoupling protein 3 gene on the respiratory quotient and fat oxidation in severe obesity and type 2 diabetes. J. Clin. Invest. 102, 1345–1351 10.1172/JCI4115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Walder K., Norman R.A., Hanson R.L., Schrauwen P., Neverova M., Jenkinson C.P.et al. (1998) Association between uncoupling protein polymorphisms (UCP2-UCP3) and energy metabolism/obesity in Pima indians. Hum. Mol. Genet. 7, 1431–1435 10.1093/hmg/7.9.1431 [DOI] [PubMed] [Google Scholar]
- 128.Miotto P.M., LeBlanc P.J. and Holloway G.P. (2018) High-fat diet causes mitochondrial dysfunction as a result of impaired ADP sensitivity. Diabetes 67, 2199–2205 10.2337/db18-0417 [DOI] [PubMed] [Google Scholar]
- 129.Bonen A., Parolin M.L., Steinberg G.R., Calles-Escandon J., Tandon N.N., Glatz J.F.et al. (2004) Triacylglycerol accumulation in human obesity and type 2 diabetes is associated with increased rates of skeletal muscle fatty acid transport and increased sarcolemmal FAT/CD36. FASEB J. 18, 1144–1146 10.1096/fj.03-1065fje [DOI] [PubMed] [Google Scholar]
- 130.Bonnard C., Durand A., Peyrol S., Chanseaume E., Chauvin M.A., Morio B.et al. (2008) Mitochondrial dysfunction results from oxidative stress in the skeletal muscle of diet-induced insulin-resistant mice. J. Clin. Invest. 118, 789–800 10.1172/JCI32601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Piccolo B.D., Keim N.L., Fiehn O., Adams S.H., Van Loan M.D. and Newman J.W. (2015) Habitual physical activity and plasma metabolomic patterns distinguish individuals with low vs. high weight loss during controlled energy restriction. J. Nutr. 145, 681–690 10.3945/jn.114.201574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Zurlo F., Lillioja S., Esposito-Del Puente A., Nyomba B.L., Raz I., Saad M.F.et al. (1990) Low ratio of fat to carbohydrate oxidation as predictor of weight gain: study of 24-h RQ. Am. J. Physiol. Metab. 259, E650–E657 10.1152/ajpendo.1990.259.5.E650 [DOI] [PubMed] [Google Scholar]
- 133.Péronnet F. and Haman F. (2019) Low capacity to oxidize fat and body weight. Obes. Rev. 20, 1367–1383 10.1111/obr.12910 [DOI] [PubMed] [Google Scholar]
- 134.Kelley D.E. and Mandarino L.J. (2000) Fuel selection in human skeletal muscle in insulin resistance: a reexamination. Diabetes 49, 677–683 10.2337/diabetes.49.5.677 [DOI] [PubMed] [Google Scholar]
- 135.Randle P.J., Garland P.B., Hales C.N. and Newsholme E.A. (1963) The glucose fatty-acid cycle its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet 281, 785–789 10.1016/S0140-6736(63)91500-9 [DOI] [PubMed] [Google Scholar]
- 136.Stroeve J.H.M., Saccenti E., Bouwman J., Dane A., Strassburg K., Vervoort J.et al. (2016) Weight loss predictability by plasma metabolic signatures in adults with obesity and morbid obesity of the D i OG enes study. Obesity 24, 379–388 10.1002/oby.21361 [DOI] [PubMed] [Google Scholar]
- 137.Courcoulas A.P., King W.C., Belle S.H., Berk P., Flum D.R., Garcia L.et al. (2018) Seven-year weight trajectories and health outcomes in the Longitudinal Assessment of Bariatric Surgery (LABS) study. JAMA Surg. 153, 427–434 10.1001/jamasurg.2017.5025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Lee I.-M. and Skerrett P.J. (2001) Physical activity and all-cause mortality: what is the dose-response relation? Med. Sci. Sport. Exerc. 33, S459–S471 10.1097/00005768-200106001-00016 [DOI] [PubMed] [Google Scholar]
- 139.Arem H., Moore S.C., Patel A., Hartge P., Berrington De Gonzalez A., Visvanathan K.et al. (2015) Leisure Time Physical Activity and Mortality: A Detailed Pooled Analysis of the Dose-Response Relationship. JAMA Intern Med. 175, 959–967 10.1001/jamainternmed.2015.0533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Kodama S., Saito K., Tanaka S., Maki M., Yachi Y., Asumi M.et al. (2009) Cardiorespiratory fitness as a quantitative predictor of all-cause mortality and cardiovascular events in healthy men and women: a meta-analysis. JAMA 301, 2024–2035 10.1001/jama.2009.681 [DOI] [PubMed] [Google Scholar]
- 141.Paffenbarger R.S. Jr, Hyde R., Wing A.L. and Hsieh C. (1986) Physical activity, all-cause mortality, and longevity of college alumni. N. Engl. J. Med. 314, 605–613 10.1056/NEJM198603063141003 [DOI] [PubMed] [Google Scholar]
- 142.Holloszy J.O. (2005) Exercise-induced increase in muscle insulin sensitivity. J. Appl. Physiol. 99, 338–343 10.1152/japplphysiol.00123.2005 [DOI] [PubMed] [Google Scholar]
- 143.Goodyear L.J. and Kahn B.B. (1998) Exercise, glucose transport, and insulin sensitivity. Annu. Rev. Med. 49, 235–261 10.1146/annurev.med.49.1.235 [DOI] [PubMed] [Google Scholar]
- 144.Borghouts L.B. and Keizer H.A. (2000) Exercise and insulin sensitivity: a review. Int. J. Sports Med. 21, 1–12 10.1055/s-2000-8847 [DOI] [PubMed] [Google Scholar]
- 145.Thomas D.M., Bouchard C., Church T., Slentz C., Kraus W.E., Redman L.M.et al. (2012) Why do individuals not lose more weight from an exercise intervention at a defined dose? An energy balance analysis Obes. Rev. 13, 835–847 10.1111/j.1467-789X.2012.01012.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Verheggen R.J.H.M., Maessen M.F.H., Green D.J., Hermus A.R.M.M., Hopman M.T.E. and Thijssen D.H.T. (2016) A systematic review and meta-analysis on the effects of exercise training versus hypocaloric diet: distinct effects on body weight and visceral adipose tissue. Obes. Rev. 17, 664–690 10.1111/obr.12406 [DOI] [PubMed] [Google Scholar]
- 147.Miller W.C., Koceja D.M. and Hamilton E.J. (1997) A meta-analysis of the past 25 years of weight loss research using diet, exercise or diet plus exercise intervention. Int. J. Obes. 21, 941–947 10.1038/sj.ijo.0800499 [DOI] [PubMed] [Google Scholar]
- 148.Foster-Schubert K.E., Alfano C.M., Duggan C.R., Xiao L., Campbell K.L., Kong A.et al. (2012) Effect of diet and exercise, alone or combined, on weight and body composition in overweight-to-obese postmenopausal women. Obesity 20, 1628–1638 10.1038/oby.2011.76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Skender M.L., Goodrick G.K., Del Junco D.J., Reeves R.S., Darnell L., Gotto A.M.et al. (1996) Comparison of 2-year weight loss trends in behavioral treatments of obesity: diet, exercise, and combination interventions. J. Am. Diet. Assoc. 96, 342–346 10.1016/S0002-8223(96)00096-X [DOI] [PubMed] [Google Scholar]
- 150.Ross R., Dagnone D., Jones P.J.H., Smith H., Paddags A., Hudson R.et al. (2000) Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men: a randomized, controlled trial. Ann. Intern. Med. 133, 92–103 10.7326/0003-4819-133-2-200007180-00008 [DOI] [PubMed] [Google Scholar]
- 151.Franz M.J., VanWormer J.J., Crain A.L., Boucher J.L., Histon T., Caplan W.et al. (2007) Weight-loss outcomes: a systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J. Am. Diet. Assoc. 107, 1755–1767 10.1016/j.jada.2007.07.017 [DOI] [PubMed] [Google Scholar]
- 152.Clark J.E. (2015) Diet, exercise or diet with exercise: comparing the effectiveness of treatment options for weight-loss and changes in fitness for adults (18-65 years old) who are overfat, or obese; systematic review and meta-analysis. J. Diab. Metab. Disord. 14, 1–28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Pinho R.A., Andrades M.E., Oliveira M.R., Pirola A.C., Zago M.S., Silveira P.C.L.et al. (2006) Imbalance in SOD/CAT activities in rat skeletal muscles submitted to treadmill training exercise. Cell Biol. Int. 30, 848–853 10.1016/j.cellbi.2006.03.011 [DOI] [PubMed] [Google Scholar]
- 154.Garneau L., Parsons S.A., Smith S.R., Mulvihill E.E., Sparks L.M. and Aguer C. (2020) Plasma myokine concentrations after acute exercise in non-obese and obese sedentary women. Front. Physiol. 11, 1–8 10.3389/fphys.2020.00018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Place N., Ivarsson N., Venckunas T., Neyroud D., Brazaitis M., Cheng A.J.et al. (2015) Ryanodine receptor fragmentation and sarcoplasmic reticulum Ca2+ leak after one session of High-intensity interval exercise. Proc. Natl. Acad. Sci. U.S.A. 112, 15492–15497 10.1073/pnas.1507176112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Wu J., Ruas J.L., Estall J.L., Rasbach K.A., Choi J.H., Ye L.et al. (2011) The unfolded protein response mediates adaptation to exercise in skeletal muscle through a PGC-1α/ATF6α complex. Cell Metab. 13, 160–169 10.1016/j.cmet.2011.01.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Ogborn D.I., McKay B.R., Crane J.D., Parise G. and Tarnopolsky M.A. (2014) The unfolded protein response is triggered following a single, unaccustomed resistance-exercise bout. Am. J. Physiol. - Regul. Integr. Comp. Physiol. 307, R664–R669 10.1152/ajpregu.00511.2013 [DOI] [PubMed] [Google Scholar]
- 158.Memme J.M., Erlich A.T., Phukan G. and Hood D.A. (2021) Exercise and mitochondrial health. J. Physiol. 599, 803–817 10.1113/JP278853 [DOI] [PubMed] [Google Scholar]
- 159.Drake J.C., Wilson R.J. and Yan Z. (2016) Molecular mechanisms for mitochondrial adaptation to exercise training in skeletal muscle. FASEB J. 30, 13–22 10.1096/fj.15-276337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Lira V.A., Benton C.R., Yan Z. and Bonen A. (2010) PGC-1α regulation by exercise training and its influences on muscle function and insulin sensitivity. Am. J. Physiol. - Endocrinol. Metab. 299, 145–161 10.1152/ajpendo.00755.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Finck B.N. and Kelly D.P. (2006) PGC-1 coactivators: inducible regulators of energy metabolism in health and disease. J. Clin. Invest. 116, 615–622 10.1172/JCI27794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Wilson J.M., Marin P.J., Rhea M.R., Wilson S.M.C., Loenneke J.P. and Anderson J.C. (2012) Concurrent training: a meta-analysis examining interference of aerobic and resistance exercises. J. Strength Cond. Res. 26, 2293–2307 10.1519/JSC.0b013e31823a3e2d [DOI] [PubMed] [Google Scholar]
- 163.Perry C.G.R., Lally J., Holloway G.P., Heigenhauser G.J.F., Bonen A. and Spriet L.L. (2010) Repeated transient mRNA bursts precede increases in transcriptional and mitochondrial proteins during training in human skeletal muscle. J. Physiol. 588, 4795–4810 10.1113/jphysiol.2010.199448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Gidlund E., Ydfors M., Appel S., Rundqvist H., Sundberg C.J. and Norrbom J. (2015) Rapidly elevated levels of PGC-1α-b protein in human skeletal muscle after exercise: exploring regulatory factors in a randomized controlled trial. J. Appl. Physiol. 119, 374–384 10.1152/japplphysiol.01000.2014 [DOI] [PubMed] [Google Scholar]
- 165.Holloszy J.O. (1967) Biochemical adaptations in muscle. Effects of exercise on mitochondrial oxygen uptake and respiratory enzyme activity in skeletal muscle. J. Biol. Chem. 242, 2278–2282 10.1016/S0021-9258(18)96046-1 [DOI] [PubMed] [Google Scholar]
- 166.Lin J., Handschin C. and Spiegelman B.M. (2005) Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab. 1, 361–370 10.1016/j.cmet.2005.05.004 [DOI] [PubMed] [Google Scholar]
- 167.Gomez-Cabrera M.-C., Domenech E. and Viña J. (2008) Moderate exercise is an antioxidant: upregulation of antioxidant genes by training. Free Radic. Biol. Med. 44, 126–131 10.1016/j.freeradbiomed.2007.02.001 [DOI] [PubMed] [Google Scholar]
- 168.Lee W.J., Kim M., Park H.S., Kim H.S., Jeon M.J., Oh K.S.et al. (2006) AMPK activation increases fatty acid oxidation in skeletal muscle by activating PPARα and PGC-1. Biochem. Biophys. Res. Commun. 340, 291–295 10.1016/j.bbrc.2005.12.011 [DOI] [PubMed] [Google Scholar]
- 169.Holloszy J.O. and Booth F.W. (1976) Biochemical adaptations to endurance exercise in muscle. Annu. Rev. Physiol. 38, 273–291 10.1146/annurev.ph.38.030176.001421 [DOI] [PubMed] [Google Scholar]
- 170.Hoppeler H., Lüthi P., Claassen H., Weibel E.R. and Howald H. (1973) The ultrastructure of the normal human skeletal muscle. Pflugers Arch. 344, 217–232 10.1007/BF00588462 [DOI] [PubMed] [Google Scholar]
- 171.Jacobs R.A., Flück D., Bonne T.C., Bürgi S., Christensen P.M., Toigo M.et al. (2013) Improvements in exercise performance with high-intensity interval training coincide with an increase in skeletal muscle mitochondrial content and function. J. Appl. Physiol. 115, 785–793 10.1152/japplphysiol.00445.2013 [DOI] [PubMed] [Google Scholar]
- 172.Granata C., Caruana N.J., Botella J., Jamnick N.A., Huynh K., Kuang J.et al. (2021) High-intensity training induces non-stoichiometric changes in the mitochondrial proteome of human skeletal muscle without reorganisation of respiratory chain content. Nat. Commun. 12, 7056 10.1038/s41467-021-27153-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Rowe G.C., El-Khoury R., Patten I.S., Rustin P. and Arany Z. (2012) PGC-1α is dispensable for exercise-induced mitochondrial biogenesis in skeletal muscle. PLoS ONE 7, e41817 10.1371/journal.pone.0041817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Greggio C., Jha P., Kulkarni S.S., Lagarrigue S., Broskey N.T., Boutant M.et al. (2017) Enhanced respiratory chain supercomplex formation in response to exercise in human skeletal muscle. Cell Metab. 25, 301–311 10.1016/j.cmet.2016.11.004 [DOI] [PubMed] [Google Scholar]
- 175.Chen Z.P., Stephens T.J., Murthy S., Canny B.J., Hargreaves M., Witters L.A.et al. (2003) Effect of exercise intensity on skeletal muscle AMPK signaling in humans. Diabetes 52, 2205–2212 10.2337/diabetes.52.9.2205 [DOI] [PubMed] [Google Scholar]
- 176.Fujii N., Hayashi T., Hirshman M.F., Smith J.T., Habinowski S.A., Kaijser L.et al. (2000) Exercise induces isoform-specific increase in 5′ AMP-activated protein kinase activity in human skeletal muscle. Biochem. Biophys. Res. Commun. 273, 1150–1155 10.1006/bbrc.2000.3073 [DOI] [PubMed] [Google Scholar]
- 177.Wojtaszewski J.F.P., Nielsen P., Hansen B.F., Richter E.A. and Kiens B. (2000) Isoform-specific and exercise intensity-dependent activation of 5′-AMP-activated protein kinase in human skeletal muscle. J. Physiol. 528, 221–226 10.1111/j.1469-7793.2000.t01-1-00221.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Puigserver P., Rhee J., Lin J., Wu Z., Yoon J.C., Zhang C.-Y.et al. (2001) Cytokine stimulation of energy expenditure through p38 MAP kinase activation of PPARγ coactivator-1. Mol. Cell. 8, 971–982 10.1016/S1097-2765(01)00390-2 [DOI] [PubMed] [Google Scholar]
- 179.Jørgensen S.B., Richter E.A. and Wojtaszewski J.F.P. (2006) Role of AMPK in skeletal muscle metabolic regulation and adaptation in relation to exercise. J. Physiol. 574, 17–31 10.1113/jphysiol.2006.109942 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Richter E.A. and Ruderman N.B. (2009) AMPK and the biochemistry of exercise: implications for human health and disease. Biochem. J. 418, 261–275 10.1042/BJ20082055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Zhou M., Lin B.Z., Coughlin S., Vallega G. and Pilch P.F. (2000) UCP-3 expression in skeletal muscle: Effects of exercise, hypoxia, and AMP-activated protein kinase. Am. J. Physiol. - Endocrinol. Metab. 279, E622–E629 10.1152/ajpendo.2000.279.3.E622 [DOI] [PubMed] [Google Scholar]
- 182.Noland R.C., Hickner R.C., Jimenez-Linan M., Vidal-Puig A., Zheng D., Dohm G.L.et al. (2003) Acute endurance exercise increases skeletal muscle uncoupling protein-3 gene expression in untrained but not trained humans. Metabolism 52, 152–158 10.1053/meta.2003.50021 [DOI] [PubMed] [Google Scholar]
- 183.Bézaire V., Seifert E.L. and Harper M.-E. (2007) Uncoupling protein-3: clues in an ongoing mitochondrial mystery. FASEB J. 21, 312–324 10.1096/fj.06-6966rev [DOI] [PubMed] [Google Scholar]
- 184.Seifert E.L., Bézaire V., Estey C. and Harper M.E. (2008) Essential role for uncoupling protein-3 in mitochondrial adaptation to fasting but not in fatty acid oxidation or fatty acid anion export. J. Biol. Chem. 283, 25124–25131 10.1074/jbc.M803871200 [DOI] [PubMed] [Google Scholar]
- 185.Kristensen M.D., Petersen S.M., Møller K.E., Lund M.T., Hansen M., Hansen C.N.et al. (2018) Obesity leads to impairments in the morphology and organization of human skeletal muscle lipid droplets and mitochondrial networks, which are resolved with gastric bypass surgery-induced improvements in insulin sensitivity. Acta Physiol. 224, e13100 10.1111/apha.13100 [DOI] [PubMed] [Google Scholar]
- 186.Toledo F.G.S., Watkins S. and Kelley D.E. (2006) Changes induced by physical activity and weight loss in the morphology of intermyofibrillar mitochondria in obese men and women. J. Clin. Endocrinol. Metab. 91, 3224–3227 10.1210/jc.2006-0002 [DOI] [PubMed] [Google Scholar]
- 187.Picard M., Gentil B.J., McManus M.J. and White K. (2013) St Louis K, Gartside SE, et al. Acute exercise remodels mitochondrial membrane interactions in mouse skeletal muscle. J. Appl. Physiol. 115, 1562–1571 10.1152/japplphysiol.00819.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Cartoni R., Léger B., Hock M.B., Praz M., Crettenand A., Pich S.et al. (2005) Mitofusins 1/2 and ERRα expression are increased in human skeletal muscle after physical exercise. J. Physiol. 567, 349–358 10.1113/jphysiol.2005.092031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Axelrod C.L., Fealy C.E., Mulya A. and Kirwan J.P. (2019) Exercise training remodels human skeletal muscle mitochondrial fission and fusion machinery towards a pro-elongation phenotype. Acta Physiol. 225, e13216 10.1111/apha.13216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Ashrafi G. and Schwarz T.L. (2013) The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ. 20, 31–42 10.1038/cdd.2012.81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Tarpey M.D., Davy K.P., McMillan R.P., Bowser S.M., Halliday T.M., Boutagy N.E.et al. (2017) Skeletal muscle autophagy and mitophagy in endurance-trained runners before and after a high-fat meal. Mol. Metab. 6, 1597–1609 10.1016/j.molmet.2017.10.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Balan E., Schwalm C., Naslain D., Nielens H., Francaux M. and Deldicque L. (2019) Regular endurance exercise promotes fission, mitophagy, and oxidative phosphorylation in human skeletal muscle independently of age. Front. Physiol. 10, 1088 10.3389/fphys.2019.01088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Greene A.W., Grenier K., Aguileta M.A., Muise S., Farazifard R., Haque M.E.et al. (2012) Mitochondrial processing peptidase regulates PINK1 processing, import and Parkin recruitment. EMBO Rep. 13, 378–385 10.1038/embor.2012.14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Scheele C., Petrovic N., Faghihi M.A., Lassmann T., Fredriksson K., Rooyackers O.et al. (2007) The human PINK1 locus is regulated in vivo by a non-coding natural antisense RNA during modulation of mitochondrial function. BMC Genomics 8, 74 10.1186/1471-2164-8-74 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Kim J., Kundu M., Viollet B. and Guan K.-L. (2011) AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat. Cell Biol. 13, 132–141 10.1038/ncb2152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Konopka A.R. and Harber M.P. (2014) Skeletal muscle hypertrophy after aerobic exercise training. Exerc. Sport Sci. Rev. 42, 53–61 10.1249/JES.0000000000000007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Phillips S.M. (2014) A brief review of critical processes in exercise-induced muscular hypertrophy. Sport Med. 44, 71–77 10.1007/s40279-014-0152-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Charette S.L., McEvoy L., Pyka G., Snow-Harter C., Guido D., Wiswell R.A.et al. (1991) Muscle hypertrophy response to resistance training in older women. J. Appl. Physiol. 70, 1912–1916 10.1152/jappl.1991.70.5.1912 [DOI] [PubMed] [Google Scholar]
- 199.Short K.R., Vittone J.L., Bigelow M.L., Proctor D.N. and Nair K.S. (2004) Age and aerobic exercise training effects on whole body and muscle protein metabolism. Am. J. Physiol. Metab. 286, E92–E101 10.1152/ajpendo.00366.2003 [DOI] [PubMed] [Google Scholar]
- 200.Grgic J., Mcllvenna L.C., Fyfe J.J., Sabol F., Bishop D.J., Schoenfeld B.J.et al. (2019) Does aerobic training promote the same skeletal muscle hypertrophy as resistance training? A systematic review and meta-analysis Sport Med. 49, 233–254 10.1007/s40279-018-1008-z [DOI] [PubMed] [Google Scholar]
- 201.West D.W.D., Burd N.A., Staples A.W. and Phillips S.M. (2010) Human exercise-mediated skeletal muscle hypertrophy is an intrinsic process. Int. J. Biochem. Cell Biol. 42, 1371–1375 10.1016/j.biocel.2010.05.012 [DOI] [PubMed] [Google Scholar]
- 202.Figueiredo V.C. and McCarthy J.J. (2019) Regulation of ribosome biogenesis in skeletal muscle hypertrophy. Physiology 34, 30–42 10.1152/physiol.00034.2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Porter C., Reidy P.T., Bhattarai N., Sidossis L.S. and Rasmussen B.B. (2014) Resistance exercise training alters mitochondrial function in human skeletal muscle. Med. Sci. Sport Exerc. 47, 1922–1931 10.1249/MSS.0000000000000605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Robinson M.M., Dasari S., Konopka A.R., Johnson M.L., Manjunatha S., Esponda R.R.et al. (2017) Enhanced protein translation underlies improved metabolic and physical adaptations to different exercise training modes in young and old humans. Cell Metab. 25, 581–592 10.1016/j.cmet.2017.02.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Brennan A.M., Standley R.A., Yi F., Carnero E.A., Sparks L.M. and Goodpaster B.H. (2020) Individual response variation in the effects of weight loss and exercise on insulin sensitivity and cardiometabolic risk in older adults. Front. Endocrinol. 11, 632 10.3389/fendo.2020.00632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Carnero E.A., Dubis G.S., Hames K.C., Jakicic J.M., Houmard J.A., Coen P.M.et al. (2017) Randomized trial reveals that physical activity and energy expenditure are associated with weight and body composition after RYGB. Obesity (Silver Spring) 25, 1206–1216 10.1002/oby.21864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Toledo F.G.S., Menshikova E.V., Azuma K., Radiková Z., Kelley C.A., Ritov V.B.et al. (2008) Mitochondrial capacity in skeletal muscle is not stimulated by weight loss despite increases in insulin action and decreases in intramyocellular lipid content. Diabetes 57, 987–994 10.2337/db07-1429 [DOI] [PubMed] [Google Scholar]
- 208.Menshikova E.V., Ritov V.B., Dube J.J., Amati F., Stefanovic-Racic M., Toledo F.G.S.et al. (2018) Calorie restriction-induced weight loss and exercise have differential effects on skeletal muscle mitochondria despite similar effects on insulin sensitivity. J. Gerontol. Ser. A. 73, 81–87 10.1093/gerona/glw328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Menshikova E.V., Ritov V.B., Ferrell R.E., Azuma K., Goodpaster B.H. and Kelley D.E. (2007) Characteristics of skeletal muscle mitochondrial biogenesis induced by moderate-intensity exercise and weight loss in obesity. J. Appl. Physiol. 103, 21–27 10.1152/japplphysiol.01228.2006 [DOI] [PubMed] [Google Scholar]
- 210.Jakicic J.M., Clark K., Coleman E., Donnelly J.E., Foreyt J., Melanson E.et al. (2001) Appropriate intervention strategies for weight loss and prevention of weight regain for adults. Med. Sci. Sport Exerc. 33, 2145–2156 10.1097/00005768-200112000-00026 [DOI] [PubMed] [Google Scholar]
- 211.Ross R., Pedwell H. and Rissanen J. (1995) Effects of energy restriction and exercise on skeletal muscle and adipose tissue in women as measured by magnetic resonance imaging. Am. J. Clin. Nutr. 61, 1179–1185 10.1093/ajcn/61.6.1179 [DOI] [PubMed] [Google Scholar]
- 212.Ismail I., Keating S.E., Baker M.K. and Johnson N.A. (2012) A systematic review and meta-analysis of the effect of aerobic vs. resistance exercise training on visceral fat. Obes. Rev. 13, 68–91 10.1111/j.1467-789X.2011.00931.x [DOI] [PubMed] [Google Scholar]
- 213.Willis L.H., Slentz C.A., Bateman L.A., Shields A.T., Piner L.W., Bales C.W.et al. (2012) Effects of aerobic and/or resistance training on body mass and fat mass in overweight or obese adults. J. Appl. Physiol. 113, 1831–1837 10.1152/japplphysiol.01370.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Dubé J.J., Amati F., Stefanovic-Racic M., Toledo F.G.S., Sauers S.E. and Goodpaster B.H. (2008) Exercise-induced alterations in intramyocellular lipids and insulin resistance: the athlete's paradox revisited. Am. J. Physiol. Metab. 294, E882–E888 10.1152/ajpendo.00769.2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Fry C.S., Noehren B., Mula J., Ubele M.F., Westgate P.M., Kern P.A.et al. (2014) Fibre type-specific satellite cell response to aerobic training in sedentary adults. J. Physiol. 592, 2625–2635 10.1113/jphysiol.2014.271288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Colleluori G., Aguirre L., Phadnis U., Fowler K., Armamento-Villareal R., Sun Z.et al. (2019) Aerobic plus resistance exercise in obese older adults improves muscle protein synthesis and preserves myocellular quality despite weight loss. Cell Metab. 30, 261–273 10.1016/j.cmet.2019.06.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Villareal D.T., Aguirre L., Gurney A.B., Waters D.L., Sinacore D.R., Colombo E.et al. (2017) Aerobic or resistance exercise, or both, in dieting obese older adults. N. Engl. J. Med. 376, 1943–1955 10.1056/NEJMoa1616338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Cypess A.M., Lehman S., Williams G., Tal I., Rodman D., Goldfine A.B.et al. (2009) Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 360, 1509–1517 10.1056/NEJMoa0810780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Nedergaard J., Bengtsson T. and Cannon B. (2007) Unexpected evidence for active brown adipose tissue in adult humans. Am. J. Physiol. Metab. 293, E444–E452 10.1152/ajpendo.00691.2006 [DOI] [PubMed] [Google Scholar]
- 220.Arch J.R.S., Ainsworth A.T., Cawthorne M.A., Piercy V., Sennitt M.V., Thody V.E.et al. (1984) Atypical β-adrenoceptor on brown adipocytes as target for anti-obesity drugs. Nature 309, 163–165 10.1038/309163a0 [DOI] [PubMed] [Google Scholar]
- 221.Cannon B. and Nedergaard J. (2004) Brown adipose tissue: function and physiological significance. Physiol. Rev. 84, 277–359 10.1152/physrev.00015.2003 [DOI] [PubMed] [Google Scholar]
- 222.Blondin D.P., Nielsen S., Kuipers E.N., Severinsen M.C., Jensen V.H., Miard S.et al. (2020) Human brown adipocyte thermogenesis is driven by β2-AR stimulation. Cell Metab. 32, 287–300 10.1016/j.cmet.2020.07.005 [DOI] [PubMed] [Google Scholar]
- 223.Mohell N., Nedergaard J. and Cannon B. (1983) Quantitative differentiation of α-and β-adrenergic respiratory responses in isolated hamster brown fat cells: evidence for the presence of an α1-adrenergic component. Eur. J. Pharmacol. 93, 183–193 10.1016/0014-2999(83)90136-X [DOI] [PubMed] [Google Scholar]
- 224.Thonberg H., Fredriksson J.M., Nedergaard J. and Cannon B. (2002) A novel pathway for adrenergic stimulation of cAMP-response-element-binding protein (CREB) phosphorylation: mediation via α1-adrenoceptors and protein kinase C activation. Biochem. J. 364, 73–79 10.1042/bj3640073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Zhao J., Cannon B. and Nedergaard J. (1997) α1-Adrenergic stimulation potentiates the thermogenic action of β3-adrenoreceptor-generated cAMP in brown fat cells. J. Biol. Chem. 272, 32847–32856 10.1074/jbc.272.52.32847 [DOI] [PubMed] [Google Scholar]
- 226.Granneman J.G., Zhai Y. and Lahners K.N. (1997) Selective up-regulation of α1a-adrenergic receptor protein and mRNA in brown adipose tissue by neural and β3-adrenergic stimulation. Mol. Pharmacol. 51, 644–650 10.1124/mol.51.4.644 [DOI] [PubMed] [Google Scholar]
- 227.Wilcke M. and Nedergaard J. (1989) α1-and β-adrenergic regulation of intracellular Ca2+ levels in brown adipocytes. Biochem. Biophys. Res. Commun. 163, 292–300 10.1016/0006-291X(89)92134-7 [DOI] [PubMed] [Google Scholar]
- 228.García-Sáinz J.A., Hoffman B.B., Li S.-Y., Lefkowitz R.J. and Fain J.N. (1980) Role of alpha1 adrenoceptors in the turnover of phosphatidylinositol and of alpha2 adrenoceptors in the regulation of cyclic AMP accumulation in hamster adipocytes. Life Sci. 27, 953–961 10.1016/0024-3205(80)90105-8 [DOI] [PubMed] [Google Scholar]
- 229.Kazak L., Chouchani E.T., Jedrychowski M.P., Erickson B.K., Shinoda K., Cohen P.et al. (2015) A creatine-driven substrate cycle enhances energy expenditure and thermogenesis in beige fat. Cell 163, 643–655 10.1016/j.cell.2015.09.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Rahbani J.F., Roesler A., Hussain M.F., Samborska B., Dykstra C.B., Tsai L.et al. (2021) Creatine kinase B controls futile creatine cycling in thermogenic fat. Nature 590, 480–485 10.1038/s41586-021-03221-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Ukropec J., Anunciado R.P., Ravussin Y., Hulver M.W. and Kozak L.P. (2006) UCP1-independent thermogenesis in white adipose tissue of cold-acclimated Ucp1-/-mice. J. Biol. Chem. 281, 31894–31908 10.1016/S0021-9258(19)84104-2 [DOI] [PubMed] [Google Scholar]
- 232.Chondronikola M., Volpi E., Børsheim E., Porter C., Saraf M.K., Annamalai P.et al. (2016) Brown adipose tissue activation is linked to distinct systemic effects on lipid metabolism in humans. Cell Metab. 23, 1200–1206 10.1016/j.cmet.2016.04.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Himms-Hagen J. (1984) Thermogenesis in brown adipose tissue as an energy buffer: implications for obesity. N. Engl. J. Med. 311, 1549–1558 10.1056/NEJM198412133112407 [DOI] [PubMed] [Google Scholar]
- 234.Orava J., Nuutila P., Noponen T., Parkkola R., Viljanen T., Enerbäck S.et al. (2013) Blunted metabolic responses to cold and insulin stimulation in brown adipose tissue of obese humans. Obesity (Silver Spring) 21, 2279–2287 10.1002/oby.20456 [DOI] [PubMed] [Google Scholar]
- 235.Reynisdottir S., Wahrenberg H., Carlström K., Rössner S. and Arner P. (1994) Catecholamine resistance in fat cells of women with upper-body obesity due to decreased expression of beta 2-adrenoceptors. Diabetologia 37, 428–435 10.1007/BF00408482 [DOI] [PubMed] [Google Scholar]
- 236.Wijers S.L.J., Saris W.H.M. and van Marken Lichtenbelt W.D. (2007) Individual thermogenic responses to mild cold and overfeeding are closely related. J. Clin. Endocrinol. Metab. 92, 4299–4305 10.1210/jc.2007-1065 [DOI] [PubMed] [Google Scholar]
- 237.Wijers S.L.J., Saris W.H.M. and van Marken Lichtenbelt W.D. (2010) Cold-induced adaptive thermogenesis in lean and obese. Obesity (Silver Spring) 18, 1092–1099 10.1038/oby.2010.74 [DOI] [PubMed] [Google Scholar]
- 238.Vosselman M.J., Van Der Lans A.A.J.J., Brans B., Wierts R., Van Baak M.A., Schrauwen P.et al. (2012) Systemic β-adrenergic stimulation of thermogenesis is not accompanied by brown adipose tissue activity in humans. Diabetes 61, 3106–3113 10.2337/db12-0288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Cypess A.M., Chen Y.-C., Sze C., Wang K., English J., Chan O.et al. (2012) Cold but not sympathomimetics activates human brown adipose tissue in vivo. Proc. Natl. Acad. Sci. USA 109, 10001–10005 10.1073/pnas.1207911109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Carey A.L., Formosa M.F., Van Every B., Bertovic D., Eikelis N., Lambert G.W.et al. (2013) Ephedrine activates brown adipose tissue in lean but not obese humans. Diabetologia 56, 147–155 10.1007/s00125-012-2748-1 [DOI] [PubMed] [Google Scholar]
- 241.Cypess A.M., Weiner L.S., Roberts-Toler C., Elía E.F., Kessler S.H., Kahn P.A.et al. (2015) Activation of human brown adipose tissue by a β3-adrenergic receptor agonist. Cell Metab. 21, 33–38 10.1016/j.cmet.2014.12.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Ouellet V., Labbé S.M., Blondin D.P., Phoenix S., Guérin B., Haman F.et al. (2012) Brown adipose tissue oxidative metabolism contributes to energy expenditure during acute cold exposure in humans. J. Clin. Invest. 122, 545–552 10.1172/JCI60433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Blondin D.P., Labbé S.M., Phoenix S., Guérin B., Turcotte É.E., Richard D.et al. (2015) Contributions of white and brown adipose tissues and skeletal muscles to acute cold-induced metabolic responses in healthy men. J. Physiol. 593, 701–714 10.1113/jphysiol.2014.283598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Wijers S.L.J., Schrauwen P., Saris W.H.M. and van Marken Lichtenbelt W.D. (2008) Human skeletal muscle mitochondrial uncoupling is associated with cold induced adaptive thermogenesis. PLoS ONE 3, e1777 10.1371/journal.pone.0001777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Davis T.R. (1967) Contribution of skeletal muscle to nonshivering thermogenesis in the dog. Am. J. Physiol. Content 213, 1423–1426 10.1152/ajplegacy.1967.213.6.1423 [DOI] [PubMed] [Google Scholar]
- 246.Skulachev V.P. and Maslov S.P. (1960) The role of nonphosphorylating oxidation in temperature regulation. Biochemistry-Moscow 25, 826–832, [In Russian] [Google Scholar]
- 247.Grav H.I. and Blix A.S. (1979) A source of nonshivering thermogenesis in fur seal skeletal muscle. Science 204, 87–89 10.1126/science.219477 [DOI] [PubMed] [Google Scholar]
- 248.Mollica M.P., Lionetti L., Crescenzo R., Tasso R., Barletta A., Liverini G.et al. (2005) Cold exposure differently influences mitochondrial energy efficiency in rat liver and skeletal muscle. FEBS Lett. 579, 1978–1982 10.1016/j.febslet.2005.02.044 [DOI] [PubMed] [Google Scholar]
- 249.Duchamp C., Barre H., Delage D., Rouanet J.-L., Cohen-Adad F. and Minaire Y. (1989) Nonshivering thermogenesis and adaptation to fasting in king penguin chicks. Am. J. Physiol. Integr. Comp. Physiol. 257, R744–R751 10.1152/ajpregu.1989.257.4.R744 [DOI] [PubMed] [Google Scholar]
- 250.Deveci D. and Egginton S. (2003) Cold exposure differentially stimulates angiogenesis in glycolytic and oxidative muscles of rats and hamsters. Exp. Physiol. 88, 741–746 10.1113/eph8802630 [DOI] [PubMed] [Google Scholar]
- 251.Dumonteil E., Barré H. and Meissner G. (1994) Effects of palmitoyl carnitine and related metabolites on the avian Ca (2+)-ATPase and Ca2+ release channel. J. Physiol. 479, 29–39 10.1113/jphysiol.1994.sp020275 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Dumonteil E., Barre H. and Meissner G. (1995) Expression of sarcoplasmic reticulum Ca2+ transport proteins in cold-acclimating ducklings. Am. J. Physiol. Physiol. 269, C955–C960 10.1152/ajpcell.1995.269.4.C955 [DOI] [PubMed] [Google Scholar]
- 253.Toyoshima C., Nakasako M., Nomura H. and Ogawa H. (2000) Crystal structure of the calcium pump of sarcoplasmic reticulum at 2.6 Å resolution. Nature 405, 647–655 10.1038/35015017 [DOI] [PubMed] [Google Scholar]
- 254.MacLennan D.H., Rice W.J. and Green N.M. (1997) The mechanism of Ca2+ transport by sarco (endo) plasmic reticulum Ca2+-ATPases. J. Biol. Chem. 272, 28815–28818 10.1074/jbc.272.46.28815 [DOI] [PubMed] [Google Scholar]
- 255.Periasamy M. and Kalyanasundaram A. (2007) SERCA pump isoforms: their role in calcium transport and disease. Muscle Nerve Off. J. Am. Assoc. Electrodiagn Med. 35, 430–442 10.1002/mus.20745 [DOI] [PubMed] [Google Scholar]
- 256.Vangheluwe P., Schuermans M., Zádor E., Waelkens E., Raeymaekers L. and Wuytack F. (2005) Sarcolipin and phospholamban mRNA and protein expression in cardiac and skeletal muscle of different species. Biochem. J. 389, 151–159 10.1042/BJ20050068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Bal N.C., Maurya S.K., Sopariwala D.H., Sahoo S.K., Gupta S.C., Shaikh S.A.et al. (2012) Sarcolipin is a newly identified regulator of muscle-based thermogenesis in mammals. Nat. Med. 18, 1575–1579 10.1038/nm.2897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Maurya S.K., Herrera J.L., Sahoo S.K., Reis F.C.G., Vega R.B., Kelly D.P.et al. (2018) Sarcolipin signaling promotes mitochondrial biogenesis and oxidative metabolism in skeletal muscle. Cell Rep. 24, 2919–2931 10.1016/j.celrep.2018.08.036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Maurya S.K., Bal N.C., Sopariwala D.H., Pant M., Rowland L.A., Shaikh S.A.et al. (2015) Sarcolipin is a key determinant of the basal metabolic rate, and its overexpression enhances energy expenditure and resistance against diet-induced obesity. J. Biol. Chem. 290, 10840–10849 10.1074/jbc.M115.636878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Lin B., Coughlin S. and Pilch P.F. (1998) Bidirectional regulation of uncoupling protein-3 and GLUT-4 mRNA in skeletal muscle by cold. Am. J. Physiol. Metab. 275, E386–E391 10.1152/ajpendo.1998.275.3.E386 [DOI] [PubMed] [Google Scholar]
- 261.Schrauwen P., Westerterp-Plantenga M.S., Kornips E., Schaart G. and van Marken Lichtenbelt W.D. (2002) The effect of mild cold exposure on UCP3 mRNA expression and UCP3 protein content in humans. Int. J. Obes. 26, 450–457 10.1038/sj.ijo.0801943 [DOI] [PubMed] [Google Scholar]
- 262.Blondin D.P., Daoud A., Taylor T., Tingelstad H.C., Bézaire V., Richard D.et al. (2017) Four-week cold acclimation in adult humans shifts uncoupling thermogenesis from skeletal muscles to brown adipose tissue. J. Physiol. 595, 2099–2113 10.1113/JP273395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Müller T.D., Blüher M., Tschöp M.H. and DiMarchi R.D. (2022) Anti-obesity drug discovery: advances and challenges. Nat. Rev. Drug Discov. 21, 201–223 10.1038/s41573-021-00337-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Tainter M.L. and Cutting W.C. (1933) Febrile, respiratory and some other actions of dinitrophenol. J. Pharmacol. Exp. Ther. 48, 410–429 [Google Scholar]
- 265.Cutting W.C., Mehrtens H.G. and Tainter M.L. (1933) Actions and uses of dinitrophenol: promising metabolic applications. J. Am. Med. Assoc. 101, 193–195 10.1001/jama.1933.02740280013006 [DOI] [Google Scholar]
- 266.Tainter M.L., Stockton A.B. and Cutting W.C. (1933) Use of dinitrophenol in obesity and related conditions: a progress report. J. Am. Med. Assoc. 101, 1472–1475 10.1001/jama.1933.02740440032009 [DOI] [Google Scholar]
- 267.Colman E. (2007) Dinitrophenol and obesity: an early twentieth-century regulatory dilemma. Regul. Toxicol. Pharmacol. 48, 115–117 10.1016/j.yrtph.2007.03.006 [DOI] [PubMed] [Google Scholar]
- 268.Childress E.S., Alexopoulos S.J., Hoehn K.L. and Santos W.L. (2017) Small molecule mitochondrial uncouplers and their therapeutic potential: miniperspective. J. Med. Chem. 61, 4641–4655 10.1021/acs.jmedchem.7b01182 [DOI] [PubMed] [Google Scholar]
- 269.Axelrod C.L., King W.T., Davuluri G., Noland R.C., Hall J., Hull M.et al. (2020) BAM15-mediated mitochondrial uncoupling protects against obesity and improves glycemic control. EMBO Mol. Med. 12, e12088 10.15252/emmm.202012088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Alexopoulos S.J., Chen S.-Y., Brandon A.E., Salamoun J.M., Byrne F.L., Garcia C.J.et al. (2020) Mitochondrial uncoupler BAM15 reverses diet-induced obesity and insulin resistance in mice. Nat. Commun. 11, 2397 10.1038/s41467-020-16298-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Dantas W.S., Zunica E.R.M., Heintz E.C., Vandanmagsar B., Floyd Z.E., Yu Y.et al. (2022) Mitochondrial uncoupling attenuates sarcopenic obesity by enhancing skeletal muscle mitophagy and quality control. J. Cachexia Sarcopenia Muscle 13, 1821–1836 10.1002/jcsm.12982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Chen S.-Y., Beretta M., Alexopoulos S.J., Shah D.P., Olzomer E.M., Hargett S.R.et al. (2021) Mitochondrial uncoupler SHC517 reverses obesity in mice without affecting food intake. Metabolism 117, 154724 10.1016/j.metabol.2021.154724 [DOI] [PubMed] [Google Scholar]
- 273.Smith B.K., Ford R.J., Desjardins E.M., Green A.E., Hughes M.C., Houde V.P.et al. (2016) Salsalate (salicylate) uncouples mitochondria, improves glucose homeostasis, and reduces liver lipids independent of AMPK-β1. Diabetes 65, 3352–3361 10.2337/db16-0564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Nie L., Yuan X.-L., Jiang K.-T., Jiang Y.-H., Yuan J., Luo L.et al. (2017) Salsalate activates skeletal muscle thermogenesis and protects mice from high-fat diet induced metabolic dysfunction. EBioMedicine 23, 136–145 10.1016/j.ebiom.2017.08.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Goldfine A.B., Fonseca V., Jablonski K.A., Chen Y.-D.I., Tipton L., Staten M.A.et al. (2013) Salicylate (salsalate) in patients with type 2 diabetes: a randomized trial. Ann. Intern. Med. 159, 1–12 10.7326/0003-4819-159-1-201307020-00003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Goldfine A.B., Fonseca V., Jablonski K.A., Pyle L., Staten M.A., Shoelson S.E.et al. (2010) The effects of salsalate on glycemic control in patients with type 2 diabetes: a randomized trial. Ann. Intern. Med. 152, 346–357 10.7326/0003-4819-152-6-201003160-00004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Tao H., Zhang Y., Zeng X., Shulman G.I. and Jin S. (2014) Niclosamide ethanolamine-induced mild mitochondrial uncoupling improves diabetic symptoms in mice. Nat. Med. 20, 1263–1269 10.1038/nm.3699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Guo J., Tao H., Alasadi A., Huang Q. and Jin S. (2019) Niclosamide piperazine prevents high-fat diet-induced obesity and diabetic symptoms in mice. Eat Weight Disord. Anorexia, Bulim Obes. 24, 91–96 10.1007/s40519-017-0424-7 [DOI] [PubMed] [Google Scholar]
- 279.Andrews P., Thyssen J. and Lorke D. (1982) The biology and toxicology of molluscicides, Bayluscide. Pharmacol. Ther. 19, 245–295 10.1016/0163-7258(82)90064-X [DOI] [PubMed] [Google Scholar]
- 280.Frayha G.J., Smyth J.D., Gobert J.G. and Savel J. (1997) The mechanisms of action of antiprotozoal and anthelmintic drugs in man. Gen. Pharmacol. Vasc. Syst. 28, 273–299 10.1016/S0306-3623(96)00149-8 [DOI] [PubMed] [Google Scholar]
- 281.Bray G.A. (2005) Drug insight: appetite suppressants. Nat. Clin. Pract. Gastroenterol. Hepatol. 2, 89–95 10.1038/ncpgasthep0092 [DOI] [PubMed] [Google Scholar]
- 282.Lesses M.F. and Myerson A. (1938) Human autonomic pharmacology: XVI. Benzedrine sulfate as an aid in the treatment of obesity. N. Engl. J. Med. 218, 119–124 10.1056/NEJM193801202180307 [DOI] [PubMed] [Google Scholar]
- 283.Kunstadter R., Necheles H. and Weiner M. (1943) Studies on the effect and mechanism of amphetamine sulfate on weight reduction. Am. J. Med. Sci. 205, 820–825 10.1097/00000441-194306000-00011 [DOI] [Google Scholar]
- 284.Harris S.C., Ivy A.C. and Searle L.M. (1947) The mechanism of amphetamine-induced loss of weight: a consideration of the theory of hunger and appetite. J. Am. Med. Assoc. 134, 1468–1475 10.1001/jama.1947.02880340022005 [DOI] [PubMed] [Google Scholar]
- 285.Bray G.A. and Greenway F.L. (1999) Current and potential drugs for treatment of obesity. Endocr. Rev. 20, 805–875 10.1210/edrv.20.6.0383 [DOI] [PubMed] [Google Scholar]
- 286.Mills E.M., Banks M.L., Sprague J.E. and Finkel T. (2003) Uncoupling the agony from ecstasy. Nature 426, 403–404 10.1038/426403a [DOI] [PubMed] [Google Scholar]
- 287.Sprague J.E., Yang X., Sommers J., Gilman T.L. and Mills E.M. (2007) Roles of norepinephrine, free fatty acids, thyroid status, and skeletal muscle uncoupling protein 3 expression in sympathomimetic-induced thermogenesis. J. Pharmacol. Exp. Ther. 320, 274–280 10.1124/jpet.106.107755 [DOI] [PubMed] [Google Scholar]
- 288.Makisumi T., Yoshida K., Watanabe T., Tan N., Murakami N. and Morimoto A. (1998) Sympatho-adrenal involvement in methamphetamine-induced hyperthermia through skeletal muscle hypermetabolism. Eur. J. Pharmacol. 363, 107–112 10.1016/S0014-2999(98)00758-4 [DOI] [PubMed] [Google Scholar]
- 289.Jung R.T., Shetty P.S., James W.P.T., Barrand M.A. and Callingham B.A. (1979) Reduced thermogenesis in obesity. Nature 279, 322–323 10.1038/279322a0 [DOI] [PubMed] [Google Scholar]
- 290.Elfellah M.S., Dalling R., Kantola I.M. and Reid J.L. (1989) Beta-adrenoceptors and human skeletal muscle characterisation of receptor subtype and effect of age. Br. J. Clin. Pharmacol. 27, 31–38 10.1111/j.1365-2125.1989.tb05332.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Williams R.S., Caron M.G. and Daniel K. (1984) Skeletal muscle beta-adrenergic receptors: variations due to fiber type and training. Am. J. Physiol. Metab. 246, E160–E167 10.1152/ajpendo.1984.246.2.E160 [DOI] [PubMed] [Google Scholar]
- 292.Andersson D.C., Betzenhauser M.J., Reiken S., Umanskaya A., Shiomi T. and Marks A.R. (2012) Stress-induced increase in skeletal muscle force requires protein kinase A phosphorylation of the ryanodine receptor. J. Physiol. 590, 6381–6387 10.1113/jphysiol.2012.237925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Shan J., Kushnir A., Betzenhauser M.J., Reiken S., Li J., Lehnart S.E.et al. (2010) Phosphorylation of the ryanodine receptor mediates the cardiac fight or flight response in mice. J. Clin. Invest. 120, 4388–4398 10.1172/JCI32726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Fagher B., Liedholm H., Monti M. and Moritz U. (1986) Thermogenesis in human skeletal muscle as measured by direct microcalorimetry and muscle contractile performance during β-adrenoceptor blockade. Clin. Sci. 70, 435–441 10.1042/cs0700435 [DOI] [PubMed] [Google Scholar]
- 295.Lynch G.S. and Ryall J.G. (2008) Role of β-adrenoceptor signaling in skeletal muscle: implications for muscle wasting and disease. Physiol. Rev. 88, 729–767 10.1152/physrev.00028.2007 [DOI] [PubMed] [Google Scholar]
- 296.Simonsen L., Bulow J., Madsen J. and Christensen N.J. (1992) Thermogenic response to epinephrine in the forearm and abdominal subcutaneous adipose tissue. Am. J. Physiol. Metab. 263, E850–E855 10.1152/ajpendo.1992.263.5.E850 [DOI] [PubMed] [Google Scholar]
- 297.Hagström-Toft E., Enoksson S., Moberg E., Bolinder J. and Arner P. (1998) β-Adrenergic regulation of lipolysis and blood flow in human skeletal muscle in vivo. Am. J. Physiol. Metab. 275, E909–E916 10.1152/ajpendo.1998.275.6.E909 [DOI] [PubMed] [Google Scholar]
- 298.Azevedo Voltarelli V., Coronado M., Gonçalves Fernandes L., Cruz Campos J., Jannig P.R., Batista Ferreira J.C.et al. (2021) β2-adrenergic signaling modulates mitochondrial function and morphology in skeletal muscle in response to aerobic exercise. Cells 10, 146 10.3390/cells10010146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Pellegrino M.A., D'Antona G., Bortolotto S., Boschi F., Pastoris O., Bottinelli R.et al. (2004) Clenbuterol antagonizes glucocorticoid-induced atrophy and fibre type transformation in mice. Exp. Physiol. 89, 89–100 10.1113/expphysiol.2003.002609 [DOI] [PubMed] [Google Scholar]
- 300.Zeman R.J., Ludemann R., Easton T.G. and Etlinger J.D. (1988) Slow to fast alterations in skeletal muscle fibers caused by clenbuterol, a beta 2-receptor agonist. Am. J. Physiol. Metab. 254, E726–E732 10.1152/ajpendo.1988.254.6.E726 [DOI] [PubMed] [Google Scholar]
- 301.Lynch G.S., Hayes A., Campbell S.P. and Williams D.A. (1996) Effects of β2-agonist administration and exercise on contractile activation of skeletal muscle fibers. J. Appl. Physiol. 81, 1610–1618 10.1152/jappl.1996.81.4.1610 [DOI] [PubMed] [Google Scholar]
- 302.Finlin B.S., Memetimin H., Zhu B., Confides A.L., Vekaria H.J., El Khouli R.H.et al. (2020) The β3-adrenergic receptor agonist mirabegron improves glucose homeostasis in obese humans. J. Clin. Invest. 130, 2319–2331 10.1172/JCI134892 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Knudsen N., Laurberg P., Rasmussen L.B., Bülow I., Perrild H., Ovesen L.et al. (2005) Small differences in thyroid function may be important for body mass index and the occurrence of obesity in the population. J. Clin. Endocrinol. Metab. 90, 4019–4024 10.1210/jc.2004-2225 [DOI] [PubMed] [Google Scholar]
- 304.Lowell B.B. and Spiegelman B.M. (2000) Towards a molecular understanding of adaptive thermogenesis. Nat 404, 652–660 10.1038/35007527 [DOI] [PubMed] [Google Scholar]
- 305.Silva J.E. (1995) Thyroid hormone control of thermogenesis and energy balance. Thyroid 5, 481–492 10.1089/thy.1995.5.481 [DOI] [PubMed] [Google Scholar]
- 306.Ritz P., Dumas J.F., Salle A., Simard G., Malthiery Y. and Rohmer V. (2002) [Thyroid hormones and obesity]. Ann. Endocrinol. (Paris) 63, 135–139, [In French] [PubMed] [Google Scholar]
- 307.Nyrnes A., Jorde R. and Sundsfjord J. (2006) Serum TSH is positively associated with BMI. Int. J. Obes. 30, 100–105 10.1038/sj.ijo.0803112 [DOI] [PubMed] [Google Scholar]
- 308.Moreno M., De Lange P., Lombardi A., Silvestri E., Lanni A. and Goglia F. (2008) Metabolic effects of thyroid hormone derivatives. Thyroid 18, 239–253 10.1089/thy.2007.0248 [DOI] [PubMed] [Google Scholar]
- 309.Bray G.A., Melvin K.E.W. and Chopra I.J. (1973) Effect of triiodothyronine on some metabolic responses of obese patients. Am. J. Clin. Nutr. 26, 715–721 10.1093/ajcn/26.6.715 [DOI] [PubMed] [Google Scholar]
- 310.Lyon D.M. and Dunlop D.M. (1932) The treatment of obesity. A comparison of the effects of diet and of thyroid extract. Q. J. Med. 1, 331–352 [Google Scholar]
- 311.Bray G.A., Fisher D.A. and Chopra I.J. (1976) Relation of thyroid hormones to body-weight. Lancet 307, 1206–1208 10.1016/S0140-6736(76)92158-9 [DOI] [PubMed] [Google Scholar]
- 312.Reinehr T., Isa A., De Sousa G., Dieffenbach R. and Andler W. (2008) Thyroid hormones and their relation to weight status. Horm. Res. Paediatr. 70, 51–57 10.1159/000129678 [DOI] [PubMed] [Google Scholar]
- 313.Moulin De Moraes C.M., Mancini M.C., De Melo M.E., Figueiredo D.A., Villares S.M.F., Rascovski A.et al. (2005) Prevalence of subclinical hypothyroidism in a morbidly obese population and improvement after weight loss induced by Roux-en-Y Gastric Bypass. Obes. Surg. 15, 1287–1291 10.1381/096089205774512537 [DOI] [PubMed] [Google Scholar]
- 314.Kozłowska L. and Rosołowska-Huszcz D. (2004) Leptin, thyrotropin, and thyroid hormones in obese/overweight women before and after two levels of energy deficit. Endocrine 24, 147–153 10.1385/ENDO:24:2:147 [DOI] [PubMed] [Google Scholar]
- 315.Sari R., Balci M.K., Altunbas H. and Karayalcin U. (2003) The effect of body weight and weight loss on thyroid volume and function in obese women. Clin. Endocrinol. (Oxf) 59, 258–262 10.1046/j.1365-2265.2003.01836.x [DOI] [PubMed] [Google Scholar]
- 316.Rosenbaum M., Hirsch J., Murphy E. and Leibel R.L. (2000) Effects of changes in body weight on carbohydrate metabolism, catecholamine excretion, and thyroid function. Am. J. Clin. Nutr. 71, 1421–1432 10.1093/ajcn/71.6.1421 [DOI] [PubMed] [Google Scholar]
- 317.Jung R.T., Shetty P.S. and James W.P. (1980) Nutritional effects on thyroid and catecholamine metabolism. Clin. Sci. (Lond.) 58, 183–191 10.1042/cs0580183 [DOI] [PubMed] [Google Scholar]
- 318.Moore R., Howard A.N., Grant A.M. and Mills I. (1980) Treatment of obesity with triiodothyronine and a very-low-calorie liquid formula diet. Lancet 315, 223–226 10.1016/S0140-6736(80)90715-1 [DOI] [PubMed] [Google Scholar]
- 319.Neves J.S., Souteiro P., Oliveira S.C., Pedro J., Magalhães D., Guerreiro V.et al. (2018) Preoperative thyroid function and weight loss after bariatric surgery. Int. J. Obes. 43, 432–436 10.1038/s41366-018-0071-8 [DOI] [PubMed] [Google Scholar]
- 320.Feldt-Rasmussen U. (2007) Thyroid and leptin. Thyroid 17, 413–419 10.1089/thy.2007.0032 [DOI] [PubMed] [Google Scholar]
- 321.Everts M.E. (1996) Effects of thyroid hormones on contractility and cation transport in skeletal muscle. Acta Physiol. Scand. 156, 325–333 10.1046/j.1365-201X.1996.203000.x [DOI] [PubMed] [Google Scholar]
- 322.Simonides W.S., Thelen M.H.M., Van der Linden C.G., Muller A. and Van Hardeveld C. (2001) Mechanism of thyroid-hormone regulated expression of the SERCA genes in skeletal muscle: implications for thermogenesis. Biosci. Rep. 21, 139–154 10.1023/A:1013692023449 [DOI] [PubMed] [Google Scholar]
- 323.Lebon V., Dufour S., Petersen K.F., Ren J., Jucker B.M., Slezak L.A.et al. (2001) Effect of triiodothyronine on mitochondrial energy coupling in human skeletal muscle. J. Clin. Invest. 108, 733–737 10.1172/JCI200111775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Clément K., Viguerie N., Diehn M., Alizadeh A., Barbe P., Thalamas C.et al. (2002) In vivo regulation of human skeletal muscle gene expression by thyroid hormone. Genome Res. 12, 281–291 10.1101/gr.207702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Pucci E., Chiovato L. and Pinchera A. (2000) Thyroid and lipid metabolism. Int. J. Obes. 24, S109–S112 10.1038/sj.ijo.0801292 [DOI] [PubMed] [Google Scholar]
- 326.Harper M.E. and Brand M.D. (1993) The quantitative contributions of mitochondrial proton leak and ATP turnover reactions to the changed respiration rates of hepatocytes from rats of different thyroid status. J. Biol. Chem. 268, 14850–14860 10.1016/S0021-9258(18)82411-5 [DOI] [PubMed] [Google Scholar]
- 327.Lanni A., Beneduce L., Lombardi A., Moreno M., Boss O., Muzzin P.et al. (1999) Expression of uncoupling protein-3 and mitochondrial activity in the transition from hypothyroid to hyperthyroid state in rat skeletal muscle. FEBS Lett. 444, 250–254 10.1016/S0014-5793(99)00061-7 [DOI] [PubMed] [Google Scholar]
- 328.De Lange P., Lanni A., Beneduce L., Moreno M., Lombardi A., Silvestri E.et al. (2001) Uncoupling protein-3 is a molecular determinant for the regulation of resting metabolic rate by thyroid hormone. Endocrinology 142, 3414–3420 10.1210/endo.142.8.8303 [DOI] [PubMed] [Google Scholar]
- 329.Lombardi A., De Matteis R., Moreno M., Napolitano L., Busiello R.A., Senese R.et al. (2012) Responses of skeletal muscle lipid metabolism in rat gastrocnemius to hypothyroidism and iodothyronine administration: A putative role for FAT/CD36. Am. J. Physiol. - Endocrinol. Metab. 303, 1222–1233 10.1152/ajpendo.00037.2012 [DOI] [PubMed] [Google Scholar]
- 330.Lombardi A., Silvestri E., Moreno M., De Lange P., Farina P., Goglia F.et al. (2002) Skeletal muscle mitochondrial free-fatty-acid content and membrane potential sensitivity in different thyroid states: involvement of uncoupling protein-3 and adenine nucleotide translocase. FEBS Lett. 532, 12–16 10.1016/S0014-5793(02)03690-6 [DOI] [PubMed] [Google Scholar]
- 331.Zhou G., Myers R., Li Y., Chen Y., Shen X., Fenyk-Melody J.et al. (2001) Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 10.1172/JCI13505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Hardie D.G. (2006) Neither LKB1 Nor AMPK are the direct targets of metformin. Gastroenterology 131, 973 10.1053/j.gastro.2006.07.032 [DOI] [PubMed] [Google Scholar]
- 333.Wang C., Liu F., Yuan Y., Wu J., Wang H., Zhang L.et al. (2014) Metformin suppresses lipid accumulation in skeletal muscle by promoting fatty acid oxidation. Clin. Lab. 60, 887–896 10.7754/Clin.Lab.2013.130531 [DOI] [PubMed] [Google Scholar]
- 334.Suwa M., Egashira T., Nakano H., Sasaki H. and Kumagai S. (2006) Metformin increases the PGC-1α protein and oxidative enzyme activities possibly via AMPK phosphorylation in skeletal muscle in vivo. J. Appl. Physiol. 101, 1685–1692 10.1152/japplphysiol.00255.2006 [DOI] [PubMed] [Google Scholar]
- 335.Hui F., Zhang Y., Ren T., Li X., Zhao M. and Zhao Q. (2019) Role of metformin in overweight and obese people without diabetes: a systematic review and network meta-analysis. Eur. J. Clin. Pharmacol. 75, 437–450 10.1007/s00228-018-2593-3 [DOI] [PubMed] [Google Scholar]
- 336.Paolisso G., Amato L., Eccellente R., Gambardella A., Tagliamonte M.R., Varricchio G.et al. (1998) Effect of metformin on food intake in obese subjects. Eur. J. Clin. Invest. 28, 441–446 10.1046/j.1365-2362.1998.00304.x [DOI] [PubMed] [Google Scholar]
- 337.Kreymann B., Ghatei M.A., Williams G. and Bloom S.R. (1987) Glucagon-like peptide-1 7-36: a physiological incretin in man. Lancet 330, 1300–1304 10.1016/S0140-6736(87)91194-9 [DOI] [PubMed] [Google Scholar]
- 338.Baggio L.L. and Drucker D.J. (2007) Biology of incretins: GLP-1 and GIP. Gastroenterology 132, 2131–2157 10.1053/j.gastro.2007.03.054 [DOI] [PubMed] [Google Scholar]
- 339.Deacon C.F. (2004) Circulation and degradation of GIP and GLP-1. Horm. Metab. Res. 36, 761–765 10.1055/s-2004-826160 [DOI] [PubMed] [Google Scholar]
- 340.Nauck M.A., Heimesaat M.M., Orskov C., Holst J.J., Ebert R. and Creutzfeldt W. (1993) Preserved incretin activity of glucagon-like peptide 1 [7-36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus. J. Clin. Invest. 91, 301–307 10.1172/JCI116186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Amori R.E., Lau J. and Pittas A.G. (2007) Efficacy and safety of incretin therapy in type 2 diabetes: systematic review and meta-analysis. JAMA 298, 194–206 10.1001/jama.298.2.194 [DOI] [PubMed] [Google Scholar]
- 342.Goldstein B.J., Feinglos M.N., Lunceford J.K., Johnson J. and Williams-Herman D.E. (2007) Effect of initial combination therapy with sitagliptin, a dipeptidyl peptidase-4 inhibitor, and metformin on glycemic control in patients with type 2 diabetes. Diabetes Care 30, 1979–1987 10.2337/dc07-0627 [DOI] [PubMed] [Google Scholar]
- 343.Larsen P.J., Fledelius C., Knudsen L.B. and Tang-Christensen M. (2001) Systemic administration of the long-acting GLP-1 derivative NN2211 induces lasting and reversible weight loss in both normal and obese rats. Diabetes 50, 2530–2539 10.2337/diabetes.50.11.2530 [DOI] [PubMed] [Google Scholar]
- 344.Flint A., Raben A., Astrup A. and Holst J.J. (1998) Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J. Clin. Invest. 101, 515–520 10.1172/JCI990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Näslund E., Barkeling B., King N., Gutniak M., Blundell J.E., Holst J.J.et al. (1999) Energy intake and appetite are suppressed by glucagon-like peptide-1 (GLP-1) in obese men. Int. J. Obes. 23, 304–311 10.1038/sj.ijo.0800818 [DOI] [PubMed] [Google Scholar]
- 346.Flint A., Raben A., Ersbøll A.K., Holst J.J. and Astrup A. (2001) The effect of physiological levels of glucagon-like peptide-1 on appetite, gastric emptying, energy and substrate metabolism in obesity. Int. J. Obes. 25, 781–792 10.1038/sj.ijo.0801627 [DOI] [PubMed] [Google Scholar]
- 347.Willms B., Werner J., Holst J.J., Orskov C., Creutzfeldt W. and Nauck M.A. (1996) Gastric emptying, glucose responses, and insulin secretion after a liquid test meal: effects of exogenous glucagon-like peptide-1 (GLP-1)-(7-36) amide in type 2 (noninsulin-dependent) diabetic patients. J. Clin. Endocrinol. Metab. 81, 327–332 [DOI] [PubMed] [Google Scholar]
- 348.Astrup A., Rössner S., Van Gaal L., Rissanen A., Niskanen L., Al Hakim M.et al. (2009) Effects of liraglutide in the treatment of obesity: a randomised, double-blind, placebo-controlled study. Lancet 374, 1606–1616 10.1016/S0140-6736(09)61375-1 [DOI] [PubMed] [Google Scholar]
- 349.Gurjar A.A., Kushwaha S., Chattopadhyay S., Das N., Pal S., China S.P.et al. (2020) Long acting GLP-1 analog liraglutide ameliorates skeletal muscle atrophy in rodents. Metabolism 103, 154044 10.1016/j.metabol.2019.154044 [DOI] [PubMed] [Google Scholar]
- 350.Abdulla H., Phillips B.E., Wilkinson D.J., Limb M., Jandova T., Bass J.J.et al. (2020) Glucagon-like peptide 1 infusions overcome anabolic resistance to feeding in older human muscle. Aging Cell. 19, e13202 10.1111/acel.13202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 351.Blanck H.M., Serdula M.K., Gillespie C., Galuska D.A., Sharpe P.A., Conway J.M.et al. (2007) Use of nonprescription dietary supplements for weight loss is common among Americans. J. Am. Diet. Assoc. 107, 441–447 10.1016/j.jada.2006.12.009 [DOI] [PubMed] [Google Scholar]
- 352.Shekelle P.G., Hardy M.L., Morton S.C., Maglione M., Mojica W.A., Suttorp M.J.et al. (2003) Efficacy and safety of ephedra and ephedrine for weight loss and athletic performance: a meta-analysis. J. Am. Med. Assoc. 289, 1537–1545 10.1001/jama.289.12.1537 [DOI] [PubMed] [Google Scholar]
- 353.Maunder A., Bessell E., Lauche R., Adams J., Sainsbury A. and Fuller N.R. (2020) Effectiveness of herbal medicines for weight loss: a systematic review and meta-analysis of randomized controlled trials. Diabetes Obes. Metab. 22, 891–903 10.1111/dom.13973 [DOI] [PubMed] [Google Scholar]
- 354.Onakpoya I.J., Posadzki P.P., Watson L.K., Davies L.A. and Ernst E. (2012) The efficacy of long-term conjugated linoleic acid (CLA) supplementation on body composition in overweight and obese individuals: a systematic review and meta-analysis of randomized clinical trials. Eur. J. Nutr. 51, 127–134 10.1007/s00394-011-0253-9 [DOI] [PubMed] [Google Scholar]
- 355.Dulloo A.G., Duret C., Rohrer D., Girardier L., Mensi N., Fathi M.et al. (1999) Efficacy of a green tea extract rich in catechin polyphenols and caffeine in increasing 24-h energy expenditure and fat oxidation in humans. Am. J. Clin. Nutr. 70, 1040–1045 10.1093/ajcn/70.6.1040 [DOI] [PubMed] [Google Scholar]
- 356.Jurgens T.M., Whelan A.M., Killian L., Doucette S., Kirk S. and Foy E. (2012) Green tea for weight loss and weight maintenance in overweight or obese adults. Cochrane Database Syst. Rev. 12, CD008650 10.1002/14651858.CD008650.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.Astrup A., Toubro S., Cannon S., Hein P., Breum L. and Madsen J. (1990) Caffeine: a double-blind, placebo-controlled study of its thermogenic, metabolic, and cardiovascular effects in healthy volunteers. Am. J. Clin. Nutr. 51, 759–767 10.1093/ajcn/51.5.759 [DOI] [PubMed] [Google Scholar]
- 358.Rousseau E., Ladine J., Liu Q.Y. and Meissner G. (1988) Activation of the Ca2+ release channel of skeletal muscle sarcoplasmic reticulum by caffeine and related compounds. Arch. Biochem. Biophys. 267, 75–86 10.1016/0003-9861(88)90010-0 [DOI] [PubMed] [Google Scholar]
- 359.Pant M., Bal N.C. and Periasamy M. (2016) Sarcolipin: a key thermogenic and metabolic regulator in skeletal muscle. Trends Endocrinol. Metab. 27, 881–892 10.1016/j.tem.2016.08.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Tabrizi R., Saneei P., Lankarani K.B., Akbari M., Kolahdooz F., Esmaillzadeh A.et al. (2019) The effects of caffeine intake on weight loss: a systematic review and dos-response meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 59, 2688–2696 10.1080/10408398.2018.1507996 [DOI] [PubMed] [Google Scholar]
- 361.Lopez-Garcia E., van Dam R.M., Rajpathak S., Willett W.C., Manson J.E. and Hu F.B. (2006) Changes in caffeine intake and long-term weight change in men and women. Am. J. Clin. Nutr. 83, 674–680 10.1093/ajcn.83.3.674 [DOI] [PubMed] [Google Scholar]
- 362.Icken D., Feller S., Engeli S., Mayr A., Müller A., Hilbert A.et al. (2016) Caffeine intake is related to successful weight loss maintenance. Eur. J. Clin. Nutr. 70, 532–534 10.1038/ejcn.2015.183 [DOI] [PubMed] [Google Scholar]
- 363.Bremer J. (1983) Carnitine–metabolism and functions. Physiol. Rev. 63, 1420–1480 10.1152/physrev.1983.63.4.1420 [DOI] [PubMed] [Google Scholar]
- 364.Uziel G., Garavaglia B. and Di Donato S. (1988) Carnitine stimulation of pyruvate dehydrogenase complex (PDHC) in isolated human skeletal muscle mitochondria. Muscle Nerve 11, 720–724 10.1002/mus.880110708 [DOI] [PubMed] [Google Scholar]
- 365.Wall B.T., Stephens F.B., Constantin-Teodosiu D., Marimuthu K., Macdonald I.A. and Greenhaff P.L. (2011) Chronic oral ingestion of l-carnitine and carbohydrate increases muscle carnitine content and alters muscle fuel metabolism during exercise in humans. J. Physiol. 589, 963–973 10.1113/jphysiol.2010.201343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Stephens F.B., Constantin-Teodosiu D., Laithwaite D., Simpson E.J. and Greenhaff P.L. (2006) Insulin stimulates L-carnitine accumulation in human skeletal muscle. FASEB J. 20, 377–379 10.1096/fj.05-4985fje [DOI] [PubMed] [Google Scholar]
- 367.Talenezhad N., Mohammadi M., Ramezani-Jolfaie N., Mozaffari-Khosravi H. and Salehi-Abargouei A. (2020) Effects of l-carnitine supplementation on weight loss and body composition: A systematic review and meta-analysis of 37 randomized controlled clinical trials with dose-response analysis. Clin. Nutr. ESPEN 37, 9–23 10.1016/j.clnesp.2020.03.008 [DOI] [PubMed] [Google Scholar]
- 368.Pooyandjoo M., Nouhi M., Shab-Bidar S., Djafarian K. and Olyaeemanesh A. (2016) The effect of (L-) carnitine on weight loss in adults: a systematic review and meta-analysis of randomized controlled trials. Obes. Rev. 17, 970–976 10.1111/obr.12436 [DOI] [PubMed] [Google Scholar]
- 369.Thorogood A., Mottillo S., Shimony A., Filion K.B., Joseph L., Genest J.et al. (2011) Isolated aerobic exercise and weight loss: a systematic review and meta-analysis of randomized controlled trials. Am. J. Med. 124, 747–755 10.1016/j.amjmed.2011.02.037 [DOI] [PubMed] [Google Scholar]
- 370.Niemeijer A., Lund H., Stafne S.N., Ipsen T., Goldschmidt C.L., Jørgensen C.T.et al. (2020) Adverse events of exercise therapy in randomised controlled trials: a systematic review and meta-analysis. Br. J. Sports Med. 54, 1073–1080 10.1136/bjsports-2018-100461 [DOI] [PubMed] [Google Scholar]
- 371.Walberg J.L. (1989) Aerobic exercise and resistance weight-training during weight reduction. Sport Med. 7, 343–356 10.2165/00007256-198907060-00001 [DOI] [PubMed] [Google Scholar]
- 372.Ivanova Y.M. and Blondin D.P. (2021) Examining the benefits of cold exposure as a therapeutic strategy for obesity and type 2 diabetes. J. Appl. Physiol. 130, 1448–1459 10.1152/japplphysiol.00934.2020 [DOI] [PubMed] [Google Scholar]
- 373.Fleischman A., Shoelson S.E., Bernier R. and Goldfine A.B. (2008) Salsalate improves glycemia and inflammatory parameters in obese young adults. Diabetes Care 31, 289–294 10.2337/dc07-1338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 374.Ofori-Adjei D., Dodoo A.N.O., Appiah-Danquah A. and Couper M. (2008) A review of the safety of niclosamide, pyrantel, triclabendazole and oxamniquine. Int. J. Risk Saf Med. 20, 113–122 10.3233/JRS-2008-0440 [DOI] [Google Scholar]
- 375.Aronne L.J., Wadden T.A., Peterson C., Winslow D., Odeh S. and Gadde K.M. (2013) Evaluation of phentermine and topiramate versus phentermine/topiramate extended-release in obese adults. Obesity (Silver Spring) 21, 2163–2171 10.1002/oby.20584 [DOI] [PubMed] [Google Scholar]
- 376.Samanin R. and Garattini S. (1993) Neurochemical mechanism of action of anorectic drugs. Pharmacol. Toxicol. 73, 63–68 10.1111/j.1600-0773.1993.tb01537.x [DOI] [PubMed] [Google Scholar]
- 377.Müller T.D., Clemmensen C., Finan B., DiMarchi R.D. and Tschöp M.H. (2018) Anti-obesity therapy: from rainbow pills to polyagonists. Pharmacol. Rev. 70, 712–746 10.1124/pr.117.014803 [DOI] [PubMed] [Google Scholar]
- 378.Bowman W.C. and Nott M.W. (1969) Actions of sympathomimetic amines and their antagonists on skeletal muscle. Pharmacol. Rev. 21, 27–72 [PubMed] [Google Scholar]
- 379.Hansen D.L., Toubro S., Stock M.J., Macdonald I.A. and Astrup A. (1999) The effect of sibutramine on energy expenditure and appetite during chronic treatment without dietary restriction. Int. J. Obes. 23, 1016–1024 10.1038/sj.ijo.0801059 [DOI] [PubMed] [Google Scholar]
- 380.Arterburn D.E., Crane P.K. and Veenstra D.L. (2004) The efficacy and safety of sibutramine for weight loss: a systematic review. Arch. Intern. Med. 164, 994–1003 10.1001/archinte.164.9.994 [DOI] [PubMed] [Google Scholar]
- 381.Finder R.M., Brogden R.N., Sawyer P.R., Speight T.M. and Avery G.S. (1975) Fenfluramine. Drugs 10, 241–323 10.2165/00003495-197510040-00001 [DOI] [PubMed] [Google Scholar]
- 382.Kaptein E.M., Beale E. and Chan L.S. (2009) Thyroid hormone therapy for obesity and nonthyroidal illnesses: a systematic review. J. Clin. Endocrinol. Metab. 94, 3663–3675 10.1210/jc.2009-0899 [DOI] [PubMed] [Google Scholar]
- 383.Bartalena L., Bogazzi F. and Martino E. (1996) Adverse effects of thyroid hormone preparations and antithyroid drugs. Drug Saf. 15, 53–63 10.2165/00002018-199615010-00004 [DOI] [PubMed] [Google Scholar]
- 384.Group DPPR (2012) Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care 35, 731–737 10.2337/dc11-1299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Arch J.R.S. (2002) β3-Adrenoceptor agonists: potential, pitfalls and progress. Eur. J. Pharmacol. 440, 99–107 10.1016/S0014-2999(02)01421-8 [DOI] [PubMed] [Google Scholar]
- 386.Moon S., Lee J., Chung H.S., Kim Y.J., Yu J.M., Yu S.H.et al. (2021) Efficacy and safety of the new appetite suppressant, liraglutide: a meta-analysis of randomized controlled trials. Endocrinol. Metab. 36, 647 10.3803/EnM.2020.934 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Wadden T.A., Bailey T.S., Billings L.K., Davies M., Frias J.P., Koroleva A.et al. (2021) Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: the STEP 3 randomized clinical trial. JAMA 325, 1403–1413 10.1001/jama.2021.1831 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA) (2015) Scientific opinion on the safety of caffeine. EFSA J. 13, 4102 10.2903/j.efsa.2015.4102 [DOI] [Google Scholar]