Significance
Both nitrogen (N) and phosphorus (P) drive water quality and are heavily altered by human activities that amplify their supplies to lakes, rivers, and oceans. Considerable attention is given to management of absolute nutrient levels but less to their relative abundances, their N:P stoichiometry. This study documents high N:P ratios in low-nutrient Flathead Lake that persist despite considerably lower N:P ratios in river nutrient inputs. The lake’s elevated N:P ratios are associated with phytoplankton P limitation, impaired food quality for zooplankton, and potential production of the greenhouse gas methane by P-limited microbes. These findings highlight the need to consider not only absolute levels of N and P in aquatic ecosystems, but also their stoichiometric balance.
Keywords: phosphorus, nitrogen, stoichiometry, limnology, ecosystem
Abstract
Considerable attention is given to absolute nutrient levels in lakes, rivers, and oceans, but less is paid to their relative concentrations, their nitrogen:phosphorus (N:P) stoichiometry, and the consequences of imbalanced stoichiometry. Here, we report 38 y of nutrient dynamics in Flathead Lake, a large oligotrophic lake in Montana, and its inflows. While nutrient levels were low, the lake had sustained high total N: total P ratios (TN:TP: 60 to 90:1 molar) throughout the observation period. N and P loading to the lake as well as loading N:P ratios varied considerably among years but showed no systematic long-term trend. Surprisingly, TN:TP ratios in river inflows were consistently lower than in the lake, suggesting that forms of P in riverine loading are removed preferentially to N. In-lake processes, such as differential sedimentation of P relative to N or accumulation of fixed N in excess of denitrification, likely also operate to maintain the lake’s high TN:TP ratios. Regardless of causes, the lake’s stoichiometric imbalance is manifested in P limitation of phytoplankton growth during early and midsummer, resulting in high C:P and N:P ratios in suspended particulate matter that propagate P limitation to zooplankton. Finally, the lake’s imbalanced N:P stoichiometry appears to raise the potential for aerobic methane production via metabolism of phosphonate compounds by P-limited microbes. These data highlight the importance of not only absolute N and P levels in aquatic ecosystems, but also their stoichiometric balance, and they call attention to potential management implications of high N:P ratios.
The emergence of the Anthropocene era has been marked by major changes in all of Earth’s major biogeochemical cycles (1). For example, fluxes of carbon (C) (as CO2) to the atmosphere have increased by ∼14% during the last 120 y largely due to fossil fuel combustion. Fluxes of nitrogen (N) into the biosphere have increased by at least 100% due to application of the Haber-Bosch reaction for fertilizer production, land use change favoring N-fixing legumes, and conversion of atmospheric N2 to available forms (NOx) by high temperature combustion of petroleum and fossil gas (2). Finally, large-scale mining of phosphorus (P)-rich geological deposits for production of fertilizers has amplified rates of P cycling in the biosphere by ∼400% (1). Each of these perturbations has biophysical and ecological impacts at differing time and space scales. For C, its accumulation in the atmosphere has altered Earth’s radiative balance, warming the planet and perturbing precipitation patterns globally. Amplified inputs of reactive N to the Earth system enter the hydrosphere and, thus, potentially lead to overenrichment of lakes, rivers, and coastal oceans across broad regions. Amplifications of P inputs often impair water quality at watershed and local scales (3), stimulating phytoplankton production and contributing, along with N, to harmful algal blooms, fish kills, and “dead zones” (4, 5). These differential amplifications and their contrasting spatial scales indicate that ecosystems are experiencing not only absolute changes in biogeochemical cycling, but also perturbations in the relative inputs and outputs of biologically important elements (6). Studies of elemental coupling and uncoupling in ecosystems are not yet widespread, but emerging work has shown how C, N, and P are differentially processed as they pass through watersheds (7).
The potential for differential alteration in supplies of N and P to aquatic ecosystems suggests that understanding the nutrient status of a water body requires knowledge of not only absolute supplies of limiting nutrients, but also their relative proportions (i.e., their N:P stoichiometry). This work has been facilitated in recent years by the emergence of the theory of ecological stoichiometry (8). For example, seminal work by Redfield (9) found that N:P ratios in marine organic matter were tightly constrained around 16:1 (molar, here and throughout), a value that may represent the central tendency for the N:P ratio of phytoplankton undergoing balanced growth in which major pools of N (protein) and P (RNA) are produced at the same rate (10). In lakes, N:P ratios show much wider variation—around a value of ∼30—perhaps reflecting the biogeochemical connections of lakes to terrestrial systems where N:P ratios have a similar value and range of variation (11). Nevertheless, this classic “Redfield ratio” of 16:1 can be thought of as representing a balanced nutrient supply for primary producers in pelagic ecosystems. When the ratios of N and P supplied deviate from this balanced ratio, primary limitation of growth by N (when N:P is low) or by P (when N:P is high) can occur. For example, phytoplankton growth in lakes with imbalanced total N: total P (TN:TP) ratios that exceed 30:1 is generally P limited (12). Disproportionate inputs of N relative to P from atmospheric deposition can increase lake TN:TP ratios and shift lake phytoplankton from N to P limitation (13), inducing P limitation in zooplankton (14). Imbalanced N:P ratios in nutrient supplies can also shift the competitive advantage among phytoplankton and enhance production of potentially toxic compounds during harmful algal blooms. For example, skewed supplies of N relative to P can increase production of N-rich secondary compounds by phytoplankton, while disproportionate inputs of P relative to N can induce production of C-based toxins (15). High N:P ratios can also enhance proliferation of fungal parasites of phytoplankton (16).
Imbalanced N:P ratios can impact aquatic ecosystems in other ways. For example, they can alter the functioning of food webs. In particular, shifts in nutrient supply regimes that enhance P limitation can impede energy flow in trophic interactions because biomass of P-limited primary producers is of low quality for animals due to its low P content (8). Ecosystem shifts to high N:P ratios and more prevalent P limitation can also impact the cycling of the greenhouse-active gas methane (CH4) because phosphate limitation can result in production of methane under aerobic conditions in both marine and freshwater phytoplankton and bacteria (17, 18). Both chemoheterotrophic and photoautotrophic bacteria (e.g., Pseudomonas, SAR11, Trichodesmium, Synechococcus) can metabolize organic P compounds, called phosphonates, to acquire P. Microbial cleavage of one type of phosphonate, methylphosphonic acid (MPn), to acquire P results in formation of methane (17). While it is likely that anaerobic methane production due to oxygen depletion in response to P-driven eutrophication is the dominant process connecting P to methane dynamics, the significance of aerobic phosphonate metabolism to global methane cycles remains to be assessed. However, contributions are potentially large, given the prevalence of P limitation in both freshwater and marine ecosystems. In light of emerging trends that suggest overall increases in ecosystem N:P ratios due to human impacts (6), these trophic and biogeochemical impacts of stoichiometric imbalance show that it is critical to consider not only absolute levels of nutrients, but also their stoichiometry. In particular, high N:P ratios can accentuate P limitation, causing a suite of ecological impacts that, currently, are poorly described.
In this paper, we illustrate the utility of stoichiometric approaches by combining analyses of long-term records of nutrient supply and dynamics, together with contemporary experiments, to examine how imbalances in N:P stoichiometry (e.g., strong divergence from classic Redfield proportions) influence plankton ecology and biogeochemistry across multiple scales in Flathead Lake, a large lake in western Montana. The lake is itself relatively unperturbed by human impacts and, thus, maintains low overall nutrient levels. However, the strong stoichiometric imbalance that we describe makes Flathead Lake appropriate for assessing ecosystem consequences of what appear to be general trends of increasing N:P ratios in global ecosystems (6). Numerous limnological properties of the lake and its inflow rivers have been monitored continuously for several decades, including concentrations of various forms of N and P. Thus, these time-series data allow us not only to assess long-term variability or stability in the stoichiometry of N and P in the lake and its river inflows over decadal time scales, but also to connect its stoichiometry with potential consequences for nutrient limitation, food web dynamics, and biogeochemical cycling under low-nutrient conditions.
Results and Discussion
Lake-Scale Stoichiometry.
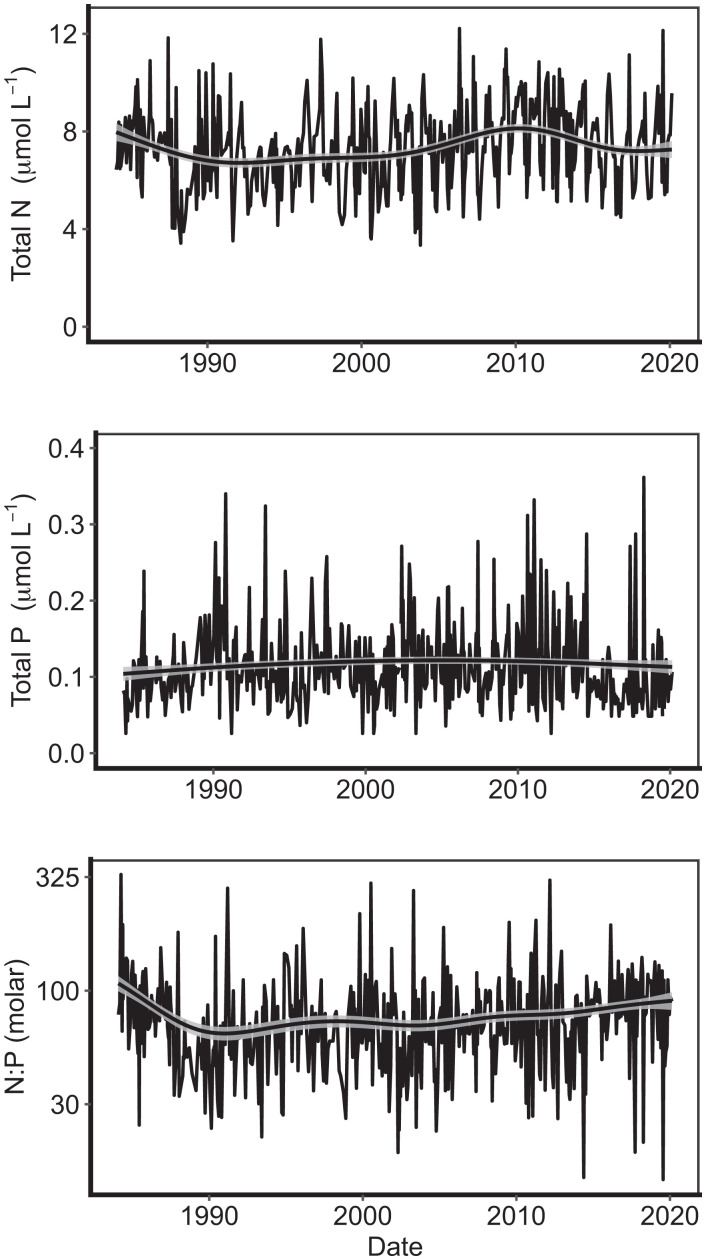
To assess the long-term status of N and P in Flathead Lake, we summarized data for average concentrations of TN and TP and their stoichiometric ratio in the upper 30 m of Flathead Lake since 1983 (Fig. 1). While exhibiting considerable seasonal variability, TN and TP concentrations, as well as the TN:TP ratios, showed no long-term trends over the 38 y of observation (Fig. 1). Overall, concentrations of TN and TP were low, falling in the lower 2.5% and 1%, respectively, of observations in the US Environmental Protection Agency's 2017 National Lakes Assessment (19). TN concentrations did decline somewhat during a 5- to 6-y period during the late 1980s and had a temporary increase around 2010 (Fig. 1A), while TP concentrations were constant throughout (Fig. 1B). Following a modest decline at the start of the record (Fig. 1C), TN:TP ratios, while quite variable at short time scales, have held steady at high values (80 to 90:1) for the 30 y since 1990. Current TN:TP ratios of ∼90:1 place Flathead Lake in the upper 87th percentile for TN:TP ratio in lakes of the United States (19). Thus, Flathead Lake has sustained a strong and steady stoichiometric imbalance for decades. This constancy of nutrient levels and ratios is notable, given that during the period of our observations, human population in the watershed—while still at relatively low density—doubled, and wildfires have burned sizable portions of the lake’s watershed.
Fig. 1.
(A–C) Biweekly or monthly concentrations of (A) TN, (B) TP, and (C) the TN:TP stoichiometric ratio (molar) in integrated samples of the upper 30 m of Flathead Lake from 1983 to 2019 (note natural log scale). GAMs were used to assess trends in the time series data, with solid black lines indicating the fitted model and gray shading indicating model error.
To connect the stoichiometric status of the lake to external inputs of N and P, we analyzed long-term data on river nutrient concentrations and discharge to estimate rates of nutrient loading and the N:P stoichiometry of that loading for the past 38 y. While highly variable, annual TN loading (Fig. 2A) has declined slightly during the study period. Annual loading of TP also displayed considerable year-to-year variability, declining modestly (∼10%) over the study period (Fig. 2B). Reflecting this long-term stability in TN and TP loading, the TN:TP ratio of annual nutrient loading, while variable (range: 5 to 60 around an overall geometric mean of 21.5), showed no long-term trend (Fig. 2C). We note that the average loading N:P ratio from Flathead Lake’s rivers is somewhat lower than national values of river and stream TN:TP ratios (median value of 31:1; ref. 20). This relatively low N:P of loading likely reflects the status of the lake’s watershed. Manning et al. (20) report that rivers and streams in forested watersheds generally have lower TN:TP ratios than those draining developed or agricultural watersheds. This is consistent with the relatively low TN:TP of loading to Flathead Lake, as the watershed is heavily forested with limited developed or agricultural land areas. Furthermore, much of this landscape lies within federally protected areas, including Glacier National Park, and federally designated wilderness areas. While there was no long-term trend in loading N:P, interannual variability in loading N:P ratios (Fig. 2C) was notable and appeared to be driven by changes in P loading (Fig. 2B). We hypothesized that this variation was driven by interannual changes in discharge. Indeed, loading N:P ratios (log-transformed) negatively correlated with annual discharge (P < 0.001, r = −0.65). That is, years when the loading N:P ratios were particularly low correspond to years with high absolute loading rates of N and, especially, P (see Fig. 2; 1990 to 1991, 1996 to 1997, 2002). This hydrologic variation itself may be due to decadal-scale climate variation (e.g., El Niño/La Niña cycles), as annual discharge into Flathead Lake is negatively correlated (P = 0.03, r = −0.34) with the Oceanic Niño Index. (Note that in western Montana, strong El Niño produces dry conditions, while La Niña is associated with wet conditions; ref. 21.) Following previous work (20, 22), we hypothesize that years of high discharge in the Flathead watershed include flood events in spring and early summer that mobilize and transport low N:P sediments from in-river or near-channel deposits in the floodplain.
Fig. 2.
(A–C) Annual riverine loading for (A) TN, (B) TP, and (C) TN:TP loading ratios (molar) since 1983. Inputs from the Flathead River dominate both the riverine hydrologic and nutrient loading to the lake. The dynamics of lake TN:TP ratio and associated GAM are shown in the upper dotted line to ease comparison.
Thus, a notable feature that emerges from this analysis of the long-term status of N:P stoichiometry in this coupled lake-watershed ecosystem is that the lake maintains unusually high N:P ratios (90:1) despite loading N:P ratios that are relatively low (∼21:1). Various mechanisms might produce this discrepancy between N:P of the lake and its river inflows. One possible contributor is that river loading N:P ratios might not reflect what nutrients are available for biological use and subsequent processing in the lake water column. For example, based on measurements of nutrient pools in river inflows since 2016, at least ∼50% of TP concentrations in river water are in particulate form versus only ∼23% for N. This observation is consistent with earlier studies in the Flathead River showing that much P carried to Flathead Lake is in nonlabile forms associated with inorganic sediments (23). Thus, much inflowing P appears particle associated, entering Flathead Lake during periods of elevated spring and early summer runoff that is high in suspended particles. Much of this particulate load is likely lost to sedimentation soon after it enters the lake. Alternatively, low bioavailability of dissolved organic N (DON) may also increase TN:TP ratios. In a large study (348 watersheds) of organic N in runoff, 50% of exported N from forested watersheds was in the form of DON, and the bioavailability of this DON was only ∼20% (24). Given that the Flathead Lake watershed is mostly forested, we expect DON to represent a large fraction of TN loading as well. While data needed to estimate DON contributions are lacking in the long-term record, assessments from spring 2021 indicate that DON contributed 49% to TN concentrations in river water, higher than nitrate (37%), a highly bioavailable form of N. Furthermore, dissolved organic P (DOP) carried by Flathead’s inflows may be more bioavailable than its DON, as for rivers entering the Baltic Sea (75% of DOP bioavailable vs. 30% for DON) (25). Thus, riverine inputs of relatively refractory DON may allow N to accumulate in the lake. Unfortunately, long-term data on the relative bioavailability of DON and DOP in river loading to Flathead Lake are not available. High lake TN:TP ratios, despite comparatively low N:P ratios in river discharge, may also be explained if low river N:P ratios are offset by contributions from high N:P nutrient sources, such as atmospheric deposition to the lake surface. Atmospheric deposition in this region does have a high N:P ratio (∼50:1) and has been an increasing contributor to the lake’s nutrient budget (26). Furthermore, most of the deposited N is in the form of highly available NH4+, and direct aerosol deposition to lake surface waters may make it an important nutrient source of N during summer periods of low river discharge (26). Nevertheless, this direct deposition constitutes <10 and 4% to the lake’s overall N and P inputs, respectively (26), and thus is unlikely to have a major influence on the overall nutrient regime. Thus, we hypothesize that in-lake processes are primarily responsible for the maintenance of Flathead Lake’s imbalanced N:P stoichiometry.
These in-lake processes might include N2 fixation. While taxa of cyanobacteria capable of N fixation are present in both the phytoplankton and periphyton of Flathead Lake (27), direct measurements of N fixation for the lake are lacking. Nevertheless, even relatively low rates of N fixation would accumulate in the lake’s N inventory if rates of denitrification are also low, as would be expected, given that the entire water column and even upper sediment layers are well oxygenated year-round. Sedimentation processes might also contribute to high TN:TP in the water column of Flathead Lake, as particulate P contributes a greater fraction (∼39 to 49%) of water column TP than particulate N contributes to TN (18%). Since particle-bound nutrients sink but dissolved forms do not, sedimentation might differentially remove P relative to N from the water column, raising overall TN:TP ratios. Another internal process that might amplify or mitigate stoichiometric imbalance is eddy diffusion of nutrients from the hypolimnion into surface waters during stratified periods. To assess this, we examined N:P ratios in hypolimnetic (90 m) pools during summer for 2018 to 2020. These ratios were also high (TN:TP of 137:1 and dissolved inorganic nitrogen: soluble reactive phosphorus of at least 190:1), implying that internal resupply of nutrients to surface waters would further reinforce stoichiometric imbalance and potential P limitation in the lake. These hypothesized internal processes contributing to the high TN:TP ratios observed in Flathead Lake require investigation but are consistent with a recent global analysis of 573 lakes (28) indicating that deep lakes (in which mean depth exceeds summer mixing depth) such as Flathead Lake have high TN:TP ratios and, thus, more potential for P limitation of phytoplankton growth because of increased efficiency of P sedimentation and diminished influence of benthic denitrification.
Consequences at the Ecological Level: Phytoplankton Nutrient Limitation.
Regardless of its underlying causes, what are the ecological consequences of Flathead Lake’s sustained highly imbalanced TN:TP ratios? We do not have continuous long-term data on summer phytoplankton nutrient limitation or C:N:P stoichiometry of seston (suspended particulate matter) in Flathead Lake. However, high in-lake TN:TP ratios could cause phytoplankton P limitation (12), at least in summer when light intensities are high, potentially leading to high C:P and N:P ratios in lake seston. Consistent with this possibility, nutrient enrichment bioassays performed in early to midsummer 2018 and 2019 show evidence of phytoplankton growth response to addition of P alone, with larger responses to combined N and P enrichment but no significant response to the addition of N alone (Fig. 3). According to a taxonomy of N × P enrichment experiment outcomes (29), these results would be consistent with serial N&P limitation, with P as the primary limiting nutrient during these summer months. Unfortunately, experiments testing N versus P limitation of phytoplankton in Flathead Lake are quite limited for earlier periods relevant to our study. Nevertheless, a set of N × P enrichment experiments similar to ours were performed in 1987 and 1988 (30, 31). While these found no response of chlorophyll-a concentrations to enrichment with P alone (in contrast to our results for three of six experiments; Fig. 3A), they did demonstrate significant stimulation by combined N&P enrichment, consistent with the strong response to combined N&P enrichment that we observed in all six of our experiments. These midsummer bioassays support a primary P limitation of phytoplankton during this period; however, we note that the N:P ratio of dissolved organic nutrients in lake must be quite high since particulate N:P is generally lower than TN:TP, even after inorganic N (nitrate) is depleted as summer progresses (SI Appendix, Fig. S1). This observation suggests differential accumulation of organic N pools (relative to P) that are not bioavailable. Thus, N&P colimitation or even N limitation of phytoplankton growth seems possible once pools of available N (NO3−) are depleted by late summer (see Fig. S1 in SI Appendix). That N can colimit phytoplankton growth is supported by our experimental results that show a consistent secondary stimulation when N is added along with P (Fig. 3), suggesting that, concordant with our inferences about river-borne DON, much of the lake’s DON is also not readily bioavailable. In fact, during summer (June to September 2016 to 2021), DON contributed an average of 68% of TN in lake surface waters (DOP contributed 56% of TP). A contributing role of N in colimiting phytoplankton growth in oligotrophic Flathead Lake, despite its high TN:TP ratios, is consistent with earlier comparative analysis indicating that the N:P ratio at which phytoplankton growth shifts to primary P limitation is higher at low nutrient levels (32). Consistent with the lake’s overall high TN:TP ratios and our bioassay data indicating that early to midsummer (June to July) phytoplankton growth is P limited, seston C:P and N:P ratios (Fig. 4) were also consistently high (>300:1 and >30:1, respectively) in the lake’s surface layers during summer. Indeed, epilimnetic seston C:P ratios reached as high as 490, consistently surpassing the threshold elemental ratio for P limitation of the important herbivore Daphnia (∼200 to 300; ref. 33).
Fig. 3.
P primarily limited Flathead Lake phytoplankton growth during summer, as estimated from factorial enrichment of N and P in six 5-d experiments testing response of chlorophyll-a concentration. (A) Raw data for three experiments in 2018 (Expt 1: 18 June; Expt 2: 2 July; Expt 3: 9 July) and 2019 (Expt 4: 24 June; Expt 5: 1 July; Expt 6: 8 July) (note log transformation of y axis). Lines connect means of the +P treatments. (B) Predicted values based on a mixed-effects linear model, with intercept as a random effect and N and P effects as fixed for all data combined. Model structure is where is ln (chlorophyll-a concentration) in replicate i in experiment j. and are dummy variables for N and P addition, and is independent and identically distributed error. Parameter estimates (±SE) are P effect, = 0.49 ± 0.067; N effect, = 0.02 ± 0.067; and NP interaction, = 0.39 ± 0.096. Overall, P addition increased chlorophyll-a when added alone or with N, but N addition only had an effect when added with P (B).
Fig. 4.
(A–C) Average (A) seston C concentrations and (B) C:P and (C) N:P ratios (molar) at 5-, 50-, and 90-m depth during summer (June to mid-September) 2016 to 2019. Error bars indicate one SEM of five sampling dates within each year. Note that x axes in B and C are natural log-transformed. The gray bar in B indicates the range of values (200 to 300) for the threshold elemental ratio, above which Daphnia P limitation is predicted to occur. The dotted lines in B and C indicate the Redfield ratios for C:P (106) and N:P (16), respectively.
Consequences for Trophic Interactions: Food Quality for Zooplankton.
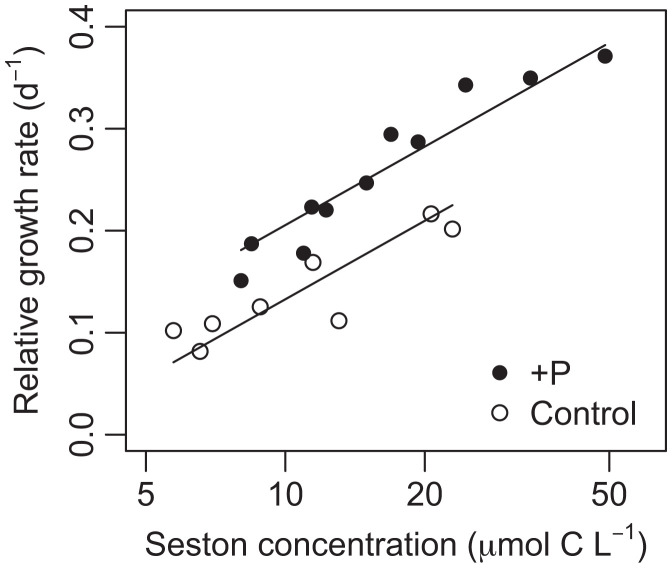
High C:P and N:P ratios such as those in Flathead Lake seston indicate potential P limitation of animal growth (34), but it remains unclear if such stoichiometric food quality limitations operate at low food concentrations (35) such as those in Flathead Lake (Fig. 4A). To test to what extent these high seston C:P ratios impose P limitation on Daphnia despite low overall food concentrations, we performed growth experiments involving manipulations of both lake seston concentration and C:P ratio (see SI Appendix for details) in summer 2019. In short, we incubated seston from the epilimnion of Flathead Lake (after screening out zooplankton as well as phytoplankton larger than 45 µm that are less edible for Daphnia) with or without PO43− enrichment for several days, allowing P uptake to lower seston C:P ratios and to increase seston biomass concentrations above low ambient levels. On a daily basis, we then diluted (using filtered lake water) or concentrated subsamples of that seston to achieve a wide gradient of food concentrations for both control (high C:P ratio) and +P treatments (low C:P ratio). Juvenile Daphnia were then grown in these various food concentration and P enrichment treatments using daily transfers for 5 d, after which body growth rate of each animal was recorded. P enrichment of lake seston lowered seston C:P ratios (from ∼280 to 591 in control seston to ∼83 to 104 in enriched seston) and improved growth of Daphnia across all food concentrations (Fig. 5). While vertical migration into deeper water to feed on seston with lower C:P ratios (Fig. 4B) might ameliorate this food quality effect, the current dominant daphnid in the lake, Daphnia thorata, prefers warmer waters and tends to remain in the upper 5 to 10 m (36). Together with phytoplankton bioassay results, these findings support the idea that imbalanced N and P ratios in Flathead Lake are associated with P limitation in the food web at both the first and second trophic levels, affecting filter-feeding Daphnia with potential impacts on its roles in modulating water quality (37) and transferring primary production to higher trophic levels. One implication of our data is that a more stoichiometrically balanced nutrient regime in the lake, achieved perhaps by adding N removal to wastewater treatment in the basin or by mitigating atmospheric N deposition, might alleviate P limitation; lower seston C:P ratios; and, thus, increase Daphnia abundances, imposing stronger water column filtering and further increasing the lake’s already considerable water transparency.
Fig. 5.
Daphnia relative growth rate (g) response to seston concentration depended on both seston concentration and seston C:P ratio in Flathead Lake. Regression model was g = −0.123 + 0.256ln(C) + 0.073P (R2 = 0.92), where C is the seston concentration, and P is a categorical value contrasting the control and P-enriched treatments. That is, this model showed that P enrichment of seston increased growth rate by 0.073 d−1 (CI 0.40 to 0.98 d−1) compared to unenriched seston, independent of food quantity. Note log-scaled x axis.
Biogeochemical Consequences: Aerobic Methane Production.
We also assessed whether the strong stoichiometric imbalance in Flathead Lake was associated with potential for aerobic production of methane via metabolic processing of methylphosphonates by P-limited lake microbes. Consistent with these mechanisms, experimental addition of MPn (together with nitrate and glucose) to epilimnetic water from Flathead Lake strongly stimulated methane production (Fig. 6), but this stimulation was suppressed when PO43− was added to satisfy microbial P demand. These findings support the view that the high N:P stoichiometric ratios in Flathead Lake and associated P limitation could induce aerobic methane production, providing a feedback between lake nutrient stoichiometry and dynamics of this greenhouse gas. While P-sensitive aerobic methane production via phosphonate metabolism has been observed in oligotrophic Lake Saiko in Japan (18) and in mesotrophic Yellowstone Lake in the United States (38), data are needed from eutrophic lakes to establish the broader significance of our findings from oligotrophic Flathead Lake. However, it is likely that conventional anaerobic pathways of methane production dominate in eutrophic lakes due to oxygen depletion driven by high rates of organic matter production and burial.
Fig. 6.
Time-course experiment showing phosphate suppression of methane production by planktonic methylphosphonic acid use in Flathead Lake (August 2018). Experimental treatments were unamended (circles) or amended with 106 µmol C L−1 as glucose, 16 µmol L−1 nitrate (as a source of N), and either 1 µmol L−1 PO43− (triangles), 1 µmol L−1 MPn (squares), or 0.5 µmol L−1 PO43− + 0.5 µmol L−1 MPn (diamonds).
Implications.
Taken together, our observations indicate that strong imbalances in lake N:P stoichiometry can be sustained over long periods and that this imbalance can have a variety of ecological consequences. While high N:P ratios in Flathead Lake primarily reflect natural processes occurring in the lake and in its relatively undisturbed watershed, our findings are timely given that various human activities can also accentuate or attenuate stoichiometric imbalances in aquatic ecosystems. For example, rapid population expansion, sewage inputs, and fertilizer use in China during recent decades lowered N:P ratios in its freshwater ecosystems (39). However, subsequent implementation of advanced wastewater treatment in China has counteracted those tendencies, leading to increasing TN:TP ratios (40). Increases in TN concurrent with decreases in TP have occurred in the Laurentian Great Lakes in recent decades and are attributed to implementation of P controls within the 1978 Great Lakes Water Quality Agreement (41). Similar processes also affect nutrient regimes in coastal oceans. Burson and colleagues (42) demonstrated strong declines in inorganic N:P ratios as well as seston N:P ratios moving offshore as phytoplankton nutrient limitation shifted from P limitation inshore to N limitation offshore in the North Sea. They attributed this pattern of inshore P limitation to high N:P ratios in river inflows due to anti-eutrophication efforts in northern Europe, which together have differentially reduced P relative to N supply to this ecosystem. In particular, PO43− detergent bans and advanced wastewater treatment have effectively removed P, while N inputs from agricultural fertilizers have increased, and those from atmospheric deposition have declined only modestly. These shifts have likely affected phytoplankton community composition, toxin production during harmful algal blooms, and trophic interactions in coastal areas of the North Sea (42). Finlay et al. (43) have also linked P reduction to accumulation of N in lakes, resulting in stoichiometric imbalance in lake N:P ratios. While the mechanisms of this outcome are not completely established, Finlay et al. hypothesized that decreases in P availability (possibly due to mitigation of P pollution) weaken the ability of lakes to assimilate N and convey it to microhabitats (such as benthic sediments) for denitrification.
Our data on nutrient limitation of phytoplankton, seston C:N:P stoichiometry, and food quality for Daphnia provide direct evidence for some of the potential consequences of sustained decoupling of P and N proposed by Finlay et al. (43). While our food quality experiment in Flathead Lake (Fig. 5) highlights the role of stoichiometric constraints in a low-nutrient system, as seen in previous experiments (14, 34), food quality impacts of high seston C:P likely operate also in mesotrophic and eutrophic lakes, as shown in earlier work (44, 45). Furthermore, our finding that imbalanced N:P stoichiometry in Flathead Lake is accompanied by the potential for aerobic methane production is also important, given that inland waters contribute ∼50% of global methane inputs to the atmosphere (46). While most aquatic methane flux derives from anaerobic processes that, themselves, are amplified by P-driven eutrophication (47), our data raise the possibility that aerobic methane production may be accentuated by establishment of high N:P ratios in lakes and other aquatic ecosystems. Overall, we propose that while much attention has been productively focused on processes that sustain or disrupt a lake’s overall nutrient concentrations and thus its trophic status, aquatic scientists should also seek to better document and understand the stoichiometric status of aquatic ecosystems, given the various consequences that sustained imbalance in N:P stoichiometry can impose. Our data on impacts of high N:P ratios pertain to the oligotrophic conditions of Flathead Lake; the relevance of these impacts in more nutrient-rich lakes requires further assessment. Nevertheless, given that human interventions now dominate both the N and P cycles (1), we contend that management of nutrients informed by stoichiometric understanding will be of increasing importance during the Anthropocene (6, 43, 48, 49). Nutrient balance matters.
Materials and Methods
Study Site.
Flathead Lake is located in northwestern Montana (47.902° N, −114.104° W). The lake is fed by the Flathead and Swan Rivers (along with other minor tributaries, <5% total load) and flows out to the Flathead River. The lake has a short residence time (∼2.2 y). Lake water level is held at a stable elevation (881 m above sea level) throughout the summer months by operation of a dam at the outlet; however, lake elevation can vary by as much as 3 m on an annual cycle. At full pool, the mean depth is 39 m, and the maximum depth is 105 m. Much of Flathead Lake’s watershed lies in Glacier National Park and adjacent wilderness areas (33%) and in managed national forest land in the United States and Canada (28%), while the remainder of the watershed (39%) lies in intermountain lowlands (prairie, pasture, farmlands, and expanding urban and suburban land use). The current year-round population in Flathead County (the largest in the watershed) is ∼104,400. In addition, the area experiences considerable and expanding tourism visitation, especially in summer. Midsummer water transparency (Secchi depth) ranges from 10 to 15 m, and epilimnetic chlorophyll-a concentrations are consistently less than 3 µg L−1 during the period of record (37). Nutrient concentrations are summarized in the main text. The lake’s upper food web is dominated by introduced fish species, especially lake trout (Salvelinus namaycush), whose abundance has increased following invasion by Mysis diluviana (opossum shrimp) in the early 1980s (37).
Lake Monitoring and Nutrient Loading.
Lake monitoring.
The Flathead Lake Monitoring program has regularly measured physical, chemical, and biological parameters at the deepest point of the lake (Midlake Deep) and in tributaries since 1977. Dissolved and total nutrient concentrations were measured ∼15 times per year at Midlake Deep and each of the major lake tributaries. Nutrient concentrations were determined using standard colorimetric segmented flow methods with alkaline persulfate digestions to assess organic pools. Samples for dissolved nutrient analyses (total dissolved P, [NO2−+ NO3−]-N, NH4+-N) were filtered to 0.45 µm, while total nutrient concentrations were determined on unfiltered water samples. In addition, a limited set of samples were assessed for total dissolved N. Detailed methodology and metadata regarding the Flathead Lake Monitoring program’s sampling and analysis protocols are available on the Flathead Lake Biological Station (FLBS) Public Data Portal (https://flbs.umt.edu/PublicData) and have been described previously in the literature (26, 37, 50). River discharge data were obtained from the United States Geological Survey's National Water Information System for both the Flathead River (USGS 12355500) and the Swan River (USGS 12370000) gauges.
Close inspection of the time series of nutrient chemistry in the lake called attention to a potentially anomalous period in the data for TP concentrations (beginning 2013); this period coincided with a change in the analytical methods used to measure the time series P pools. Beginning in July 2013, the colorimetric P determinations shifted to include a correction for potential silica interference (SI Appendix). The lack of silica correction in the pre-2013 analysis led to an overestimation of TP concentrations by 1.6 µg L−1 (51.6 nmol L−1) in lake samples and an Si-proportional overestimate of P in river samples. Hence, for this study, we transformed data prior to 2013 accordingly (see SI Appendix for details). Because TP levels in river samples were relatively high, the Si correction did not have a large effect on estimates of riverine TP loading, but the 51.6 nmol L−1 correction of lake TP concentrations for the early periods represented a ∼25 to 30% reduction in values reported for this observation period. Corrected data are displayed in Figs. 1 and 2. See SI Appendix for additional information.
Nutrient concentration time series.
Generalized additive models (GAMs) and mixed-effect models were used to identify trends for unaggregated Midlake Deep nutrient concentrations through time. Additive models are used because the Flathead Lake Monitoring program dataset time series contains missing data, has intermittent changes in the frequency of and intervals between sampling events, and likely has autoregressive tendencies that preclude standard linear models (51). Models included a random effect parameter accounting for the month sampled, thus preserving inherent variation due to seasonality or variable sampling frequencies throughout the dataset. To examine trends in the long-term time series data, GAMs were constructed with the R package mgcv structured with concentration as a function of date with a splining smoother term. Model optimization (i.e., the number of nodes used in the smoothing function) was based on a restricted maximum likelihood estimate approach. Models were assessed (52) using the package mgcv in R (53).
Nutrient loading time series.
Annual loads of TP and TN were estimated using the rloadest and loadflex packages (54) in R (55). rloadest is based on the USGS LOADEST program (56), and loadflex allows for composite models accounting for variation in concentration to hydrograph relationships through time. These packages were used to estimate loads based on time series data of hydrologic regimes and nutrient concentrations. Total mass daily loadings were estimated and summed to calculate the annual load for the entirety of the Flathead Lake Monitoring program dataset. Models were automatically selected based on maximum likelihood estimation or least absolute deviation when residuals were not normally distributed. The TN:TP ratio was calculated daily and averaged annually. GAMs were then used to assess patterns of change in nutrient concentrations, loading, and stoichiometric balance through time as indicated above.
Nutrient Enrichment Experiments.
Six in situ incubation experiments were performed to test for nutrient (N, P) limitation of phytoplankton growth via factorial enrichment of N and P in June and July of 2018 and 2019. All water samples were collected from Yellow Bay, a deep embayment on the eastern side of the lake. Water was collected along a 5-m column using an integrated tube sampler (2018) or a Kemmerer sampler (2019) at four random sites within Yellow Bay. All samples were filtered through a 125-µm Nitex mesh to remove macrozooplankton prior to enrichment. Initial water samples were reserved to determine the initial chlorophyll-a concentrations.
For each factorial N × P experiment, 16 clear 500-mL bottles were rinsed and then filled with water from Yellow Bay, four each for a control (C), N, P, and a combined NP treatment. The N and P treatments were 8 µmol L−1 N (equimolar as NH4Cl + NaNO3) and 0.5 µmol L−1 P (as Na2HPO4), respectively, while the N&P treatment received both 8 µmol L−1 N and 0.5 µmol L−1 P enrichment (16:1 atomic ratio). In 2019, bottles used in experiments 2 and 3 all received the same level of potassium (K) as part of a separate study. All bottles were suspended at a depth of 1.5 m (equivalent to ∼50% of incident light) in Yellow Bay. After 4 d, 250 mL from each bottle were filtered onto a glass fiber filter (Whatman GF/F) that was frozen until extraction. Chlorophyll-a was extracted by holding filters in the dark at 4 °C for 12 to 24 h in either methanol or 90% acetone. Extracts were analyzed following EPA method 445.0 (57) using a Turner Designs fluorometer and a hydrochloric acid correction for phaeophytin.
We estimated the effect of N, P, and N&P additions in multiple experiments by fitting a random intercept model predicting chlorophyll-a concentration for replicate i in experiment j with slopes for N, P, and N&P as fixed effects. Values for N and P were 0 if not added and 1 if added. This model was
where is normally distributed error. We fit this model using lmer() in package lme4 in R.
Daphnia Food Quality Experiments.
Seston manipulation.
We used a 5-L Van Dorn horizontal water sampler to collect lake water from depths of 0.5, 3, 5, and 6 m in Yellow Bay. Water was combined into a single sample, passed through a 45-µm nylon net to remove zooplankton and phytoplankton inedible to Daphnia, and transferred to 20-L polycarbonate carboys. We conducted two experiments (21 August and 16 September 2019) to assess Daphnia growth at different food concentrations for ambient seston (high C:P ratio) and for P-enriched seston with low C:P ratio. In both experiments, food (seston) quantity and quality were manipulated by enriching N to 0.45 µmol L−1 (as NaNO3) to control carboys and P to +0.16 µmol L−1 and +0.5 µmol L−1 P (as K2HPO4) in the first experiment and +0.48 µmol L−1 P in the second experiment to P-enriched carboys. Carboys were then placed in an outdoor incubator circulated with surface lake water to maintain temperature. The incubator reduced light intensity to 30% of ambient levels using blue Plexiglas shading. The carboys were held for 3 to 5 d to allow seston concentration to increase before the start of the Daphnia growth experiment, allowing us to examine animal growth with high and low seston C:P and covering a range of food concentrations after dilution (see below).
Seston C:P ratio and concentration.
Seston C concentrations and C:P ratios in both the lake and the carboys were measured daily. Seston was filtered onto precombusted and acid-washed Whatman glass fiber filters (GF/F grade), dried for at least 24 h at 60 °C, and then digested using modified persulfate oxidation followed by infrared detection (for C measurement) and the ascorbic acid method (for P measurement) (58). In the first experiment, seston concentration for each treatment carboy was measured daily by filtering seston onto a GF/F filter and measuring C concentration prior to dilution with filtered lake water (see below). In the second experiment, seston concentration in each individual jar used for animal growth was estimated daily by measuring turbidity (using a Hach 2100N turbidimeter) and then converting the turbidity measurement to C concentration based on a previous calibration.
Food concentration gradient preparation.
In the first experiment, gradients of three food concentrations were established for seston from the control, +0.16 µmol L−1 P, and +0.5 µmol L−1 P treatments. For all the treatments, food concentrations were set at 25, 50, and 100% of the actual in situ concentration and prepared by diluting ambient seston (100%) with Whatman GF/F-filtered (nominal pore size: 0.7 µm) lake water. In the second experiment, gradients of five food concentrations were established for both control and P-enriched (+0.5 µmol L−1 P) seston. For the P-enriched treatments, food concentrations were set at 6.25, 12.5, 25, 50, and 100% of the initial concentration. For the control treatment, food concentrations were lower, and, thus, levels of seston were set at 12.5, 25, 50, 100, and 200% of the initial concentration in the carboy. To prepare these treatments, a 5-µm Nitex net was used to concentrate seston from the control carboy to 200% of the ambient level, and then the resulting concentrated seston preparation was diluted to different target concentrations with Whatman GF/F-filtered water. For the enriched treatment, food concentration gradients were prepared by diluting ambient seston (100%) with GF/F-filtered water.
Daphnia culture.
A population of Daphnia pulicaria isolated from Flathead Lake was maintained in the laboratory in modified COMBO media (59) on a diet of nutrient-sufficient Scenedesmus acutus. Adult Daphnia were separated from the main culture to produce offspring used in the experiments. Neonates were collected between 24 and 48 h in age, imaged for initial body size using a digital camera on a dissecting microscope, and placed individually into 60-mL glass vials with 60 mL prepared seston (see above). Each food treatment had eight replicate animals for the first experiment and seven replicates for the second experiment. Vials were kept on a rotating plankton wheel to prevent food particles from setting. The wheel was kept inside a growth chamber at 19 to 20 °C with continuous illumination. Every day, animals were transferred individually into freshly prepared seston. The experiments ran for 5 d, after which animals were individually reimaged. Relative growth rate (g) of each Daphnia was calculated from the pre- and postexperimental body sizes as g (d−1) = ln (S2/S1)/dt, where S is individual body area at beginning (S1) and end (S2) of the experiment, and dt is the duration of the experiment (5 d).
Data analysis.
In the first experiment, daily measurements of seston concentration in each jar were averaged across all replicates for that food concentration treatment, and the resulting mean 5-d seston C concentration was used for analysis of Daphnia relative growth rate. In the second experiment, daily measurements of seston C concentrations of each treatment were averaged and used in analysis. Examination of data indicated that P enrichment resulted in similar C:P ratios in the two experiments and that growth rate data for the two experiments also collapsed onto a similar response function with seston C concentration. So, data for the two experiments were combined for comparison of growth rate of animals fed P-enriched seston with growth data for animals receiving unenriched seston, while controlling for seston concentration.
To do so, we fit the following regression model to the data:
| [1] |
where g is relative growth rate, C is seston concentration (log transformed to facilitate linearity), P is a dummy variable designating P-enriched or control treatments, and is normally distributed error. This model assumes a variable intercept and a common slope and is equivalent to an Analysis of Covariance without an interaction term. Models with an interaction term (i.e., ) had P = 0.07 for and Akaike information criterion values three units higher than obtained for Eq. 1, indicating an overfitted model.
Methane Experiment.
A time-course experiment to examine potential plankton production of CH4 based on the use of MPn was conducted in August 2018. Near-surface Flathead Lake water was collected into four 20-L polycarbonate carboys and transported to the laboratory. In the laboratory, carboys were amended with treatment-specific nutrient substrates (glucose, N, P, and MPn). Experimental treatments were compared to unamended lake water controls. Nutrients (final concentrations) were added as C + N + PO43− = 106 µmol L−1 C (as glucose) + 16 µmol L−1 NO3− (as NaNO3) + 1 µmol L−1 PO43− (as Na2PO4); C + N + MPn = 106 µmol L−1 C (as glucose) + 16 µmol L−1 NO3− + 1 µmol L−1 MPn; C + N + MPn + PO43− = 106 µmol L−1 C (as glucose) + 16 µmol L−1 NO3− + 0.5 µmol L−1 MPn + 0.5 µmol L−1 PO43−. Water was subsampled from each carboy into 160-mL glass serum bottles, and bottles were crimp-sealed with Teflon-lined silicone stoppers. Incubations were conducted at in situ temperatures in the dark. Triplicate serum bottles from each treatment (and control) were harvested at each sampling time point for quantification of CH4 concentrations as described above. Samples were killed by injecting 200 µL of an 8 M NaOH solution and analyzed by gas chromatography via headspace (20 mL) gas introduction.
Supplementary Material
Acknowledgments
We thank all of those who contributed to the Flathead Lake long-term monitoring program during the past four decades, including T. Bansak, E. Bilbrey, J. Craft, B. Ellis, J. Nigon, J. Rainieri, J. Stanford, T. Tappenbeck, and many others. M. Trentman provided useful advice on nutrient loading models. D. Whited and B. Bannerman provided information on watershed characteristics. Members of the 2018 and 2019 Field Ecology classes at the FLBS contributed to the bioassay studies. Sampling of Flathead Lake was performed under sampling permits issued by the Confederated Salish and Kootenai Tribe; we are grateful for their partnership. Routine monitoring of Flathead Lake is supported by funds from private philanthropy and from the state of Montana. Early funding for Flathead Lake monitoring came from the McKnight Foundation. Funding for data analysis and manuscript preparation for J.J.E. was provided by the Jessie M. Bierman Professorship, FLBS. We are grateful to R.W. Sterner for early comments on the manuscript. J.Y. was funded by the National Natural Science Foundation of China (41877415) and supported by the Chinese Academy of Sciences scholarship for a 1-y visit at the FLBS. Funding from the US National Science Foundation (DEB-1951002 to M.J.C. and S.P.D. and DEB-1950963 to J.E.D.) supported the methane analyses. Members of the FLBS acknowledge that we live and work within the aboriginal territories of the Salish and Kootenai people, whose descendants among the Bitterroot Salish, Upper Pend d’Oreille, and Kootenai tribes continue to act as careful stewards of these watersheds.
Footnotes
Reviewers: R.M., Universite de Montreal; and J.F., University of Minnesota
The authors declare no competing interest.
This article contains supporting information online at https://www.pnas.org/lookup/suppl/doi:10.1073/pnas.2202268119/-/DCSupplemental.
Data Availability
All observational and experimental data (water chemistry, loading, nutrient limitation, Daphnia growth, methane production) have been deposited via Dryad (https://datadryad.org/) at doi:10.5061/dryad.hdr7sqvkw (60).
References
- 1.Falkowski P., et al. , The global carbon cycle: A test of our knowledge of earth as a system. Science 290, 291–296 (2000). [DOI] [PubMed] [Google Scholar]
- 2.Erisman J. W., et al. , Consequences of human modification of the global nitrogen cycle. Philos. Trans. R. Soc. Lond. B Biol. Sci. 368, 20130116 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schindler D. W., Evolution of phosphorus limitation in lakes. Science 195, 260–262 (1977). [DOI] [PubMed] [Google Scholar]
- 4.Diaz R. J., Rosenberg R., Spreading dead zones and consequences for marine ecosystems. Science 321, 926–929 (2008). [DOI] [PubMed] [Google Scholar]
- 5.Heisler J., et al. , Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae 8, 3–13 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Peñuelas J., Sardans J., The global nitrogen-phosphorus imbalance. Science 375, 266–267 (2022). [DOI] [PubMed] [Google Scholar]
- 7.Maranger R., Jones S. E., Cotner J. B., Stoichiometry of carbon, nitrogen, and phosphorus through the freshwater pipe. Limnol. Oceanogr. Lett. 3, 89–101 (2018). [Google Scholar]
- 8.Sterner R. W., Elser J. J., Ecological Stoichiometry: The Biology of Elements from Molecules to the Biosphere (Princeton University Press, 2002). [Google Scholar]
- 9.Redfield A. C., The biological control of chemical factors in the environment. Am. Sci. 46, 205–221 (1958). [PubMed] [Google Scholar]
- 10.Loladze I., Elser J. J., The origins of the Redfield nitrogen-to-phosphorus ratio are in a homoeostatic protein-to-rRNA ratio. Ecol. Lett. 14, 244–250 (2011). [DOI] [PubMed] [Google Scholar]
- 11.Elser J. J., et al. , Nutritional constraints in terrestrial and freshwater food webs. Nature 408, 578–580 (2000). [DOI] [PubMed] [Google Scholar]
- 12.Downing J. A., McCauley E., The nitrogen: phosphorus relationship in lakes. Limnol. Oceanogr. 37, 936–945 (1992). [Google Scholar]
- 13.Elser J. J., et al. , Shifts in lake N:P stoichiometry and nutrient limitation driven by atmospheric nitrogen deposition. Science 326, 835–837 (2009). [DOI] [PubMed] [Google Scholar]
- 14.Elser J. J., et al. , Atmospheric nitrogen deposition is associated with elevated phosphorus limitation of lake zooplankton. Ecol. Lett. 13, 1256–1261 (2010). [DOI] [PubMed] [Google Scholar]
- 15.Van de Waal D. B., Smith V. H., Declerck S. A. J., Stam E. C. M., Elser J. J., Stoichiometric regulation of phytoplankton toxins. Ecol. Lett. 17, 736–742 (2014). [DOI] [PubMed] [Google Scholar]
- 16.Frenken T., et al. , Changes in N:P supply ratios affect the ecological stoichiometry of a toxic cyanobacterium and its fungal parasite. Front. Microbiol. 8, 1015 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Karl D. M., et al. , Aerobic production of methane in the sea. Nat. Geosci. 1, 473–478 (2008). [Google Scholar]
- 18.Khatun S., et al. , Aerobic methane production by planktonic microbes in lakes. Sci. Total Environ. 696, 133916 (2019). [Google Scholar]
- 19.U.S. Environmental Protection Agency, National Lakes Assessment 2017 (data and metadata files). National Aquatic Resource Surveys (2021). https://www.epa.gov/national-aquatic-resource-surveys/data-national-aquatic-resource-surveys. Accessed 25 May 2021.
- 20.Manning D. W. P., Rosemond A. D., Benstead J. P., Bumpers P. M., Kominoski J. S., Transport of N and P in U.S. streams and rivers differs with land use and between dissolved and particulate forms. Ecol. Appl. 30, e02130 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dracup J. A., Kahya E., The relationships between U.S. streamflow and La Niña Events. Water Resour. Res. 30, 2133–2141 (1994). [Google Scholar]
- 22.Whited D. C., et al. , Climate, hydrologic disturbance, and succession: Drivers of floodplain pattern. Ecology 88, 940–953 (2007). [DOI] [PubMed] [Google Scholar]
- 23.Ellis B. K., Stanford J. A., "Bioavailability of phosphorus fractions in Flathead Lake and its tributary waters." (Project Completion Report. Open File Report, 091–086, 1986).
- 24.Pellerin B. A., Kaushal S. S., McDowell W. H., Does anthropogenic nitrogen enrichment increase organic nitrogen concentrations in runoff from forested and human-dominated watersheds? Ecosystems (N. Y.) 9, 852–864 (2006). [Google Scholar]
- 25.Stepanauskas R., et al. , Summer inputs of riverine nutrients to the Baltic sea: Bioavailability and eutrophication relevance. Ecol. Monogr. 72, 579–597 (2002). [Google Scholar]
- 26.Ellis B. K., Craft J. A., Stanford J. A., Long-term atmospheric deposition of nitrogen, phosphorus and sulfate in a large oligotrophic lake. PeerJ 3, e841 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.DeYoe H. R., Lowe R. L., Marks J. C., Effects of nitrogen and phosphorus on the endosymbiont load of Rhopalodia gibba and Epithemia turgida (Bacillariophyceae). J. Phycol. 28, 773–777 (1992). [Google Scholar]
- 28.Qin B., et al. , Water depth underpins the relative roles and fates of nitrogen and phosphorus in lakes. Environ. Sci. Technol. 54, 3191–3198 (2020). [DOI] [PubMed] [Google Scholar]
- 29.Harpole W. S., et al. , Nutrient co-limitation of primary producer communities. Ecol. Lett. 14, 852–862 (2011). [DOI] [PubMed] [Google Scholar]
- 30.Spencer C. N., Ellis A. B. K., Role of nutrients and zooplankton in regulation of phytoplankton in Flathead Lake (Montana, USA), a large oligotrophic lake. Freshw. Biol. 39, 755–763 (1998). [Google Scholar]
- 31.Dodds W. K., Priscu J. C., A comparison of methods for assessment of nutrient deficiency of phytoplankton in a large oligotrophic lake. Can. J. Fish. Aquat. Sci. 47, 2328–2338 (1990). [Google Scholar]
- 32.Forsberg C., Ryding S. O., Eutrophication parameters and trophic state indices in 30 Swedish waste-receiving lakes. Arch. Hydrobiol. 89, 189–207 (1980). [Google Scholar]
- 33.Urabe J., Watanabe Y., Possibility of N or P limitation for planktonic cladocerans: An experimental test. Limnol. Oceanogr. 37, 244–251 (1992). [Google Scholar]
- 34.Elser J. J., Hayakawa K., Urabe J., Nutrient limitation reduces food quality for zooplankton: Daphnia response to seston phosphorus enrichment. Ecology 82, 898–903 (2001). [Google Scholar]
- 35.Bukovinszky T., et al. , The good, the bad and the plenty: Interactive effects of food quality and quantity on the growth of different Daphnia species. PLoS One 7, e42966 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Spencer C., Impact of predation by Mysis relicta on zooplankton in Flathead Lake, Montana, USA. J. Plankton Res. 21, 51–64 (1999). [Google Scholar]
- 37.Ellis B. K., et al. , Long-term effects of a trophic cascade in a large lake ecosystem. Proc. Natl. Acad. Sci. U.S.A. 108, 1070–1075 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang Q., Dore J. E., McDermott T. R., Methylphosphonate metabolism by Pseudomonas sp. populations contributes to the methane oversaturation paradox in an oxic freshwater lake. Environ. Microbiol. 19, 2366–2378 (2017). [DOI] [PubMed] [Google Scholar]
- 39.Yan Z., et al. , Phosphorus accumulates faster than nitrogen globally in freshwater ecosystems under anthropogenic impacts. Ecol. Lett. 19, 1237–1246 (2016). [DOI] [PubMed] [Google Scholar]
- 40.Tong Y., et al. , Improvement in municipal wastewater treatment alters lake nitrogen to phosphorus ratios in populated regions. Proc. Natl. Acad. Sci. U.S.A. 117, 11566–11572 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Dove A., Chapra S. C., Long-term trends of nutrients and trophic response variables for the Great Lakes. Limnol. Oceanogr. 60, 696–721 (2015). [Google Scholar]
- 42.Burson A., Stomp M., Akil L., Brussaard C. P. D., Huisman J., Unbalanced reduction of nutrient loads has created an offshore gradient from phosphorus to nitrogen limitation in the North Sea. Limnol. Oceanogr. 61, 869–888 (2016). [Google Scholar]
- 43.Finlay J. C., Small G. E., Sterner R. W., Human influences on nitrogen removal in lakes. Science 342, 247–250 (2013). [DOI] [PubMed] [Google Scholar]
- 44.DeMott W. R., Gulati R. D., Phosphorus limitation in Daphnia: Evidence from a long term study of three hypereutrophic Dutch lakes. Limnol. Oceanogr. 44, 1557–1564 (1999). [Google Scholar]
- 45.DeMott W. R., Gulati R. D., Van Donk E., Effects of dietary phosphorus deficiency on the abundance, phosphorus balance, and growth of Daphnia cucullata in three hypereutrophic Dutch lakes. Limnol. Oceanogr. 46, 1871–1880 (2001). [Google Scholar]
- 46.Rosentreter J. A., et al. , Half of global methane emissions come from highly variable aquatic ecosystem sources. Nat. Geosci. 14, 225–230 (2021). [Google Scholar]
- 47.Beaulieu J. J., DelSontro T., Downing J. A., Eutrophication will increase methane emissions from lakes and impoundments during the 21st century. Nat. Commun. 10, 1375 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Paerl H. W., Valdes L. M., Joyner A. R., Piehler M. F., Lebo M. E., Solving problems resulting from solutions: Evolution of a dual nutrient management strategy for the eutrophying Neuse River Estuary, North Carolina. Environ. Sci. Technol. 38, 3068–3073 (2004). [DOI] [PubMed] [Google Scholar]
- 49.Hendriks A. T. W. M., Langeveld J. G., Rethinking wastewater treatment plant effluent standards: Nutrient reduction or nutrient control? Environ. Sci. Technol. 51, 4735–4737 (2017). [DOI] [PubMed] [Google Scholar]
- 50.Devlin S. P., et al. , Spatial and temporal dynamics of invasive freshwater shrimp (Mysis diluviana): Long-term effects on ecosystem properties in a large oligotrophic lake. Ecosystems (N. Y.) 20, 183–197 (2017). [Google Scholar]
- 51.Simpson G. L., Modelling palaeoecological time series using generalised additive models. Front. Ecol. Evol. 6, 149 (2018). [Google Scholar]
- 52.Wood S. N., Generalized Additive Models: An Introduction with R (CRC Press, ed. 2, 2017). [Google Scholar]
- 53.Wood S. N., Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J. R. Stat. Soc. Series B Stat. Methodol. 73, 3–36 (2011). [Google Scholar]
- 54.Lorenz D. L., Rloadest: USGS Water Science R Functions for LOAD Estimation of Constituents in River and Streams. US Geological Survey R Archive Network (US Geological Survey, Washington, DC, 2014). [Google Scholar]
- 55.Computing R., Others R., A Language and Environment for Statistical Computing (R Core Team; ) 2013). [Google Scholar]
- 56.Runkel R. L., Crawford C. G., Cohn T. A., Load estimator (LOADEST): A FORTRAN program for estimating constituent loads in streams and rivers. (Techniques and Methods Rep. 4-A5, USGS, 2004). 10.3133/tm4A5. [DOI]
- 57.Arar E. J., Collins G. B., Method 445.0: In Vitro Determination of Chlorophyll A and Pheophytin A in Marine and Freshwater Algae by Fluorescence (U.S. Environmental Protection Agency, Washington, DC, 1997). [Google Scholar]
- 58.Lampman G. G., Caraco N. F., Cole J. J., A method for the measurement of particulate C and P on the same filtered sample. Mar. Ecol. Prog. Ser. 217, 59–65 (2001). [Google Scholar]
- 59.Kilham S. S., Kreeger D. A., Lynn S. G., Goulden C. E., Herrera L., COMBO: A defined freshwater culture medium for algae and zooplankton. Hydrobiologia 377, 147–159 (1998). [Google Scholar]
- 60.J. Elser, Nutrient concentrations, loading, and N:P stoichiometry (1983 - 2020) and impacts in Flathead Lake (Montana, USA). Dryad. https://datadryad.org/stash/dataset/doi:10.5061%2Fdryad.hdr7sqvkw. Deposited 17 June 2022. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All observational and experimental data (water chemistry, loading, nutrient limitation, Daphnia growth, methane production) have been deposited via Dryad (https://datadryad.org/) at doi:10.5061/dryad.hdr7sqvkw (60).