Highlights
-
•
HWWS is a congenital anomaly characterized by uterine didelphys, unilateral blind hemivagina, and ipsilateral renal agenesis.
-
•
We presented a case of HWWS-associated cervical cancer in which vaginal bleeding was the first presentation of HWWS.
-
•
HWWS may be present in the elderly women, even without a previous diagnosis of urogenital anomaly.
-
•
A literature review revealed an increased incidence of clear cell carcinoma histology in women with HWWS.
Keywords: Herlyn-Werner-Wunderlich syndrome, Cervical cancer, Clear cell carcinoma
Abstract
Herlyn-Werner-Wunderlich syndrome (HWWS) is a rare congenital urogenital anomaly characterized by uterine didelphys, unilateral blind hemivagina, and ipsilateral renal agenesis. We present a very rare case of HWWS-associated cervical cancer in which the presence of a genital anomaly was not noticed until the patient experienced postmenopausal vaginal bleeding. A 74-year-old nulliparous Japanese woman presented with vaginal bleeding. Pre-treatment workup revealed uterine didelphys, obstructed hemivagina/hemicervix, renal agenesis, and cancer development from the remnant-obstructed hemivagina/hemicervix. The patient was diagnosed with HWWS and HWWS-associated vaginal or cervical cancer, treated with radical surgery, and a diagnosis of clear cell carcinoma (CCC) of the uterine cervix was histopathologically confirmed. A literature review revealed an increased incidence of CCC in women with HWWS.
1. Introduction
Herlyn-Werner-Wunderlich syndrome (HWWS), also known as OHVIRA (Obstructed Hemivagina and Ipsilateral Renal Anomaly) syndrome, is a rare congenital abnormality caused by abnormal development of Müllerian and Wolffian ducts (Burgis, 2001). HWWS was originally characterized by three key anomalies: uterus didelphys, unilateral blind hemivagina, and ipsilateral renal agenesis, and its incidence is estimated to be 0.1–3.8% (Burgis, 2001).
HWWS is commonly diagnosed during puberty and presents with pelvic pain or dysmenorrhea shortly after menarche, all of which are associated with hematocolpos, hematometra, or hematosalpinx (Zhu et al., 2015). However, in asymptomatic cases during puberty and the subsequent period, the diagnosis of HWWS is delayed, and unusual presentations of HWWS, including renal or vaginal hemorrhage, endometriosis, infertility, and benign and malignant tumors of the genitourinary system, have been observed (Jindal et al., 2009). With regard to HWWS-associated malignant gynecological cancer, only 11 cases, including ours, have been reported in the English literature (Watanabe et al., 2012, Kaba et al., 2013, Cordoba et al., 2017, Kusunoki et al., 2018, Oka et al., 2020, Mei, 2020, Tanase et al., 2021, AlMulhim and AlRasheed, 2021, Kobayashi et al., 2021). Given the rarity of this condition, we believe that reporting individual cases is important for establishing an optimal diagnostic method and treatment.
We present a very rare case of HWWS-associated cervical cancer in which the presence of a congenital genital anomaly was not noticed until the patient experienced postmenopausal vaginal bleeding and was successfully treated with radical hysterectomy plus total vaginectomy. We also summarized the current knowledge regarding HWWS-associated gynecological cancers.
1.1. Informed consent statement
A written informed consent was obtained from the patient for publication of this case report and accompanying images.
1.2. Case report
A 74-year-old nulliparous Japanese woman who had been evaluated at a general hospital for postmenopausal vaginal bleeding was referred to our hospital. Her surgical and medical history was unremarkable, except for hypertension. After her menarche at age 15, she had regular cycles without dysmenorrhea and experienced menopause at 50. She was informed about her left kidney agenesis during childhood.
On speculum evaluation, a macroscopic tumor sized 1-cm was observed at the left vaginal wall, 2-cm above the vaginal opening. A biopsy obtained from this lesion revealed adenocarcinoma. She had a grossly normal but slightly right-shifted uterine cervix with a normal pap smear result. Transvaginal ultrasound revealed a 4-cm cystic mass adjacent to her normal cervix, which was later proven to be a hydrometra without accompanying abnormal endometrial thickness on pelvic MRI (Fig. 1A).
Fig. 1.
Pretreatment imaging results. (A), T2-weighted pelvic MRI (Coronal view) of the right uterus. A 4-cm cystic mass (blue arrow) adjacent to her normal right cervix (white arrow), showing a high signal on T2-weighted imaging, was observed in her left pelvis. As this cyst showed iso/high signal on T1-weighted imaging (C) and being communicating with the right normal cervix, this was proven to be a hematometra of her right uterus that was deviated to the left side. (B), T2-weighted pelvic MRI (Left; Coronal view, Right; Sagittal view) of the left uterus and vagina. Her remnant obstructed left hemivagina/hemicervix was observed in front of her deviated right uterus. In addition to a hematometra and a hematocolpos (blue arrow), a solid tumor exhibiting heterogeneous high signal intensity on T2-weighted image was observed in her left hemivagina/hemicervix (white arrow). (C), Axial views of T1-weighted pelvic MRI (left) and T2-weighted pelvic MRI (Right). In addition to the hematometra of the left (blue arrow) and right uteruses (white arrow), a tubular structure (yellow arrow) showing high signal on T1-weighted image suggesting methemoglobin was observed adjacent to her left hematometra, corresponding to a left ectopic ureter.(D), Abdominal CT imaging demonstrating right renal agenesis. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
Bimanual pelvic examination revealed a 5–6 cm immovable mass on the left anterior side of the abovementioned 4-cm cystic mass that extended to the left vaginal lesion. Transvaginal ultrasonography revealed that the mass consisted of two parts: a 2-cm round solid mass adjacent to the left vaginal tumor (lower solid portion), which communicated with a 3–4 cm cystic mass with internal echoes resembling an endometrioma (superior cystic portion). Based on pelvic MRI (Fig. 1B), the solid and cystic structures were thought to correspond to a malignant tumor that developed from a left obstructed hemivagina/hemicervix and a resulting hematometra, respectively. In addition, on the left side of the obstructed hemivagina/hemicervix, another fluid-filled tubular structure exhibiting high signal intensity on T1-weighted image was observed (Fig. 1C), which corresponded to a blind ectopic ureter containing the reflux of hematic fluid from the obstructed hemivagina/hemicervix. Both the ovaries were normal.
Given the presence of left renal agenesis (Fig. 1D), uterine didelphys (two separate hematometras), and an obstructed hemivagina/hemicervix, a diagnosis of Herlyn-Werner-Wunderlich syndrome (HWWS) and a provisional diagnosis of adenocarcinoma extending from the obstructed hemivagina/hemicervix were made. Computed tomography (CT) scans of the abdomen and chest showed no evidence of metastatic disease, and laboratory findings were unremarkable, except for an elevated serum CA-125 of 69.3 U/mL (normal range < 35 U/mL).
After discussing the treatment options, including surgical treatment and definitive radiotherapy, with the patients and their families, the patient received surgical treatment. During surgery, no intraperitoneal adhesions, uterine didelphys and normal adnexa on either side were noted. Radical hysterectomy, total vaginectomy, bilateral salpingo-oophorectomy, and pelvic lymphadenectomy were performed to resect the tumor with adequate surgical margins. During the procedure, a fluid-filled tubular structure was identified on the left side of her obstructed hemivagina/hemicervix, which was proven to be her left ectopic ureter leading to the obstructed hemivagina/hemicervix and was resected along with her obstructed hemivagina/hemicervix.
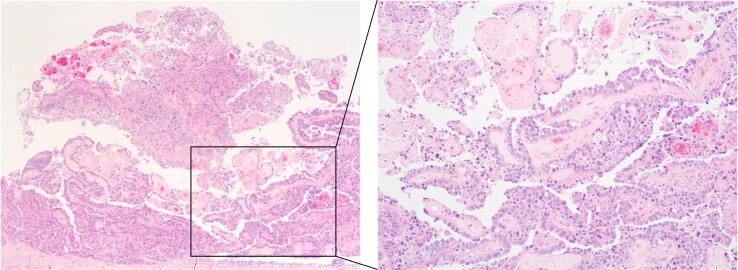
Pathological examination of the surgical specimen revealed a clear cell carcinoma that developed from the obstructed hemivagina or hemicervix extending superficially to the uterine corpus without lymph node metastasis (Fig. 2). Pathological examinations failed to distinguish the origin of the adenocarcinoma (vagina vs. cervix). The surgical margin was negative, and parametrial or lymphovascular space invasion was not observed. Although a vaginal tumor was predominant, as the uterine cervix was involved, we concluded that this was a cervical cancer of the remnant uterus (T2A1N0M0). Due to the lack of clinical evidence demonstrating the benefit of adjuvant radiotherapy for such unusual cervical cancer that developed from the remnant uterus, the patient did not receive postoperative adjuvant treatment. She is currently free of disease three months after the radical surgery.
Fig. 2.
Histopathological findings from surgically-resected specimens. Pathological examination of the surgical specimen revealed a clear cell carcinoma that developed from the obstructed hemivagina or hemicervix (hematoxylin-eosin staining: left, x1; right, × 40).
2. Discussion
We present a very rare case of HWWS-associated cervical cancer, in which the presence of a congenital genital anomaly was not noticed until the patient experienced postmenopausal vaginal bleeding. After the diagnosis of HWWS and vaginal or cervical cancer extending from the non-visible obstructed hemivagina/hemicervix, the patient was successfully treated with radical hysterectomy, total vaginectomy, and pelvic lymphadenectomy. Postoperative histopathology revealed a clear cell carcinoma that developed from the obstructed hemivagina/hemicervix extending to the entire uterus, indicating the difficulty in precisely diagnosing the origin of gynecological cancer in the remnant uterus in women with HWWS.
HWWS is a complex congenital malformation of the female urogenital tract caused by Müllerian (paramesonephric) and Wolffian (mesonephric) duct anomalies. This syndrome was first reported in 1922 by Purslow (Purslow, 1922), but its current denomination derives from two subsequent reports. In 1971, Herlyn and Werner described the simultaneous occurrence of an open Gartner duct cyst, a homolateral aplasia of the kidney, and a double uterus (Herlyn–Werner syndrome) (Herlyn and Werner, 1971). Five years later, in 1976, Wunderlich described an association between right renal aplasia and a bicornuate uterus and simple vagina in the presence of an isolated hematocervix (Wunderlich, 1976). HWWS was originally described as a triad of symptoms: didelphytic uterus, low genital obstruction (hemivagina and hemicervix), and unilateral renal anomaly (renal agenesis, dysplasia, polycystic kidney, duplication, or crossed fused ectopia). However, the significant number of cases with a non-didelphytic uterus, the acronym OHVIRA, which refers to two features, obstructed hemivagina/hemicervix with ipsilateral renal anomaly, gained wide acceptance, and HWWS has been referred to as OHVIRA syndrome since 2007 (Smith and Laufer, 2007).
A previous investigation conducted by Zhu et al. suggested that the syndrome can be classified into two types according to the complete or partial obstruction of the hemivagina/hemicervix: classification 1, a completely obstructed hemivagina/hemicervix, and classification 2, an incompletely obstructed hemivagina/hemicervix (Zhu et al., 2015). In Classification 2 HWWS, as the menses from the obstructed side can be drained through the contralateral vagina, the clinical presentation is generally delayed when compared with Classification 1. However, as communication is generally limited, drainage is still impeded and may cause symptoms similar to those observed in patients with Classification 1 HWWS. In classification 1 HWWS, patients usually experience pelvic pain shortly after menarche, which is associated with hematocolpos, hematometra, or hematosalpinx. However, our case, Classification 1 HWWS, was asymptomatic and unaware of urogenital malformation until she had been evaluated for postmenopausal vaginal bleeding. Thus, we must recognize that HWWS may be present in the elderly, even without a previous diagnosis of urogenital anomaly or history of dysmenorrhea or abdominal/pelvic pain.
As shown (Table 1), so far, 11 cases of HWWS-associated gynecological malignancies have been reported. Most malignancies developed from the remnant uterus or vagina (nine cases), and vaginal bleeding was the most frequent presenting symptom. Of these, six patients had been previously diagnosed with HWWS. Of the five patients who had received marsupialization for HWWS, four patients had a correct pretreatment diagnosis of gynecological cancer, which was completely consistent with a postoperative diagnosis. In contrast, among the 5 patients who had not been previously diagnosed with HWWS, a preoperative diagnosis of gynecological cancer was not consistent with a postoperative diagnosis in 3 cases, indicating the difficulty in precisely diagnosing cancer developed from the remnant uterus or vagina in women with HWWS.
Table 1.
Summary of a reports of HWWS-associated gynecological malignancies.
| Author Reference Year |
Age Gravida/Para |
HPV status | DES status | Affected side | Pretreatment diagnosis | Treatment | Posttreatment diagnosis Stage |
Histology | Previous diagnosis of HWWS | Symptom |
|---|---|---|---|---|---|---|---|---|---|---|
| Watanabe, et al.4 2012 |
33 y.o. 2/2 |
Negative | No | Remnant | Cervical or vaginal cancer | Chemotherapy, EBRT, followed by PE | Cervical or vaginal cancer T4ANxM0 |
EM | Yes | VB |
| 53 y.o. 2/2 |
Negative | No | Remnant | Cervical or vaginal cancer | RH | Vaginal cancer T1NxM0 |
CCC | Yes Marspialization was performed |
VB Vaginal pain |
|
| Kaba, et al.5 2013 |
49 y.o. 2/2 |
NA | NA | Remnant | Corpus cancer | Radical surgery | Cervical cancer T1B1N0M0 |
EM | No | Dysmenorrhea VB |
| Cordoba, et al.6 2017 |
37 y.o. 2/2 |
NA | NA | Remnant | Cervical cancer | Pelvic LND followed by CCRT | Cervical cancer T3AN1M0 |
A | Yes Marspialization was performed |
Dysmenorrhea VB |
| Kusunoki, et al.7 2018 |
65 y.o. 3/2 |
NA | No | Remnant | Paracervical mass in the remnant uterus | Radical surgery followed by CCRT | Cervical cancer T1B2N0M0 |
CCC | No | VB |
| Oka, et al.8 2020 |
38 0/0 |
Negative | No | Remnant | Cervical cancer | Radical surgery followed by RT | Cervical cancer T2A1N0M0 |
A | Yes Marspialization was performed |
VB |
| Mei, et al.9 2020 |
40 1/1 |
NA | No | Remnant | Vaginal cancer | Radical surgery | Vaginal cancer T1N0M0 |
CCC | No | VB |
| Tanase, et al.10 2021 |
52 y.o. 0/0 |
NA | No | Remnant | Cervical cancer | Radical surgery | Cervical cancer T1B1N0M0 |
CCC | Yes Marspialization was performed |
VB |
| Almulhim, et al.11 2021 |
29 y.o. 0/0 |
NA | NA | Right ovary | Ovarian tumor | Cystectomy | Ovarian cancer T1ANxM0 |
Serous borderline tumor | No | Infertility Ovarian tumor |
| Kobayashi, et al.12 2021 |
58 y.o. 2/1 |
NA | NA | Visible side uterus | Uterine corpus | Radical surgery | Corpus cancer T3AN0M0 |
EM | Yes Marspialization was performed |
VB |
| Current case | 74 y.o. 0/0 |
NA | No | Remnant | Cervical or vaginal cancer | Radical surgery | Cervical cancer T2A1N0M0 |
CCC | No | VB |
NA; not available, HPV; Human papillomavirus, DES; diethylstilbestol, HWWS; Herlyn-Werner-Wunderlich syndrome, RH; radical hysterectomy, CCRT; concurrent chemoradiotherapy, LND; lymphadenectomy, EBRT; external-beam radiotherapy, PE; pelvic exenteration, CCC; clear cell carcinoma, EM; endometrioid carcinoma, A; adenocarcinoma, VB; vaginal bleeding.
To the best of our knowledge, this is the third case of HWWS-associated CCC of the uterine cervix. Among the reported HWWS-associated gynecological cancers, clear CCC was predominant, followed by endometrioid carcinoma (Table 1). It has been generally accepted that CCC of the vagina or cervix is extremely rare, except for women with a history of prenatal diethylstilbestrol (DES) exposure. No such DES-associated cases were observed in the HWWS-associated gynecological cancers (Table 1). Interestingly, the histological trend in which CCC and endometrioid carcinoma are predominant is consistent with that observed in endometriosis-associated ovarian cancer (Murakami et al., 2020 Jun 24). The precise reason for this trend is unknown, but human papillomavirus (HPV) cannot, as cervical or vaginal CCC is known to be an HPV-independent tumor (Stolnicu et al., 2022). We believe this can be explained by a common feature between obstructed hemivagina/hemicervix in HWWS and ovarian endometriosis: accumulation of menstrual blood containing excess iron and iron-induced reactive oxygen species, which theoretically can accelerate malignant transformation of exposed cells by inducing gene mutations or DNA methylation (Stolnicu et al., 2022). However, as there is no evidence to support this hypothesis, future mechanistic investigations using clinical samples obtained from the affected patients are warranted.
In summary, we presented a case of HWWS-associated cervical cancer in which postmenopausal vaginal bleeding was the first presentation of HWWS and cancer development from the remnant vagina. Given the rarity of the condition, sharing this knowledge may not impact daily patient care. However, we believe that it is worth remembering that HWWS may be present in the elderly, even without a previous diagnosis of urogenital anomaly or a history of dysmenorrhea or abdominal/pelvic pain. Moreover, although cancer development in the remnant gynecological organs is extremely rare and a diagnosis is challenging, to avoid a delay in early diagnosis, this condition should be considered when women suspected of having HWWS exhibit postmenopausal vaginal bleeding.
Ethical approval status: The Ethics Committee of Osaka International Cancer Institute decided that this case report does not require IRB approval due to the nature of this study: a report describing the diagnosis and the treatment of a single patient and thus does not meet the definition of human subjects research.
CRediT authorship contribution statement
Seiji Mabuchi: Conceptualization, Writing – original draft. Harue Hayashida: Data curation. Chiaki Kubo: Supervision, Visualization. Masahiko Takemura: Writing – review & editing. Shoji Kamiura Writing – review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- AlMulhim J., AlRasheed M.R. Herlyn-Werner-Wunderlich syndrome with borderline serous cystadenoma of the ovary: case report and literature review. Radiol. Case Rep. 2021;16(3):744–747. doi: 10.1016/j.radcr.2020.09.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgis J. Obstructive Müllerian anomalies: Case report, diagnosis, and management. Am. J. Obstet. Gynecol. 2001;185(2):338–344. doi: 10.1067/mob.2001.116738. [DOI] [PubMed] [Google Scholar]
- Cordoba A., Escande A., Comte P., Fumagalli I., Bresson L., Mubiayi N., Lartigau E. Locally advanced adenocarcinoma of the cervix on uterus didelphys: a case report. J Contemp Brachytherapy. 2017;1:71–76. doi: 10.5114/jcb.2017.65640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herlyn U., Werner H. Simultaneous occurrence of an open Gartner-duct cyst, a homolateral aplasia of the kidney and a double uterus as a typical syndrome of abnormalities. Geburtshilfe Frauenheilkd. 1971;31:340–347. [PubMed] [Google Scholar]
- Jindal G., Kachhawa S., Meena G.L., Dhakar G. Uterus didelphys with unilateral obstructed hemivagina with hematometrocolpos and hematosalpinx with ipsilateral renal agenesis. J. Hum. Reprod. Sci. 2009;2(2):87. doi: 10.4103/0974-1208.57230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaba M., Gungor T., Baser E., Ozdal B., Sirvan L. Cervical cancer in a patient with uterus didelphys and obstructive hemivagina, ipsilateral renal anomaly (OHVIRA) syndrome. Arch. Gynecol. Obstet. 2013;288(1):229–230. doi: 10.1007/s00404-012-2690-y. [DOI] [PubMed] [Google Scholar]
- Kobayashi, M., et al., 2021 Robot-assisted laparoscopic hysterectomy for endometrial cancer in a patient with Herlyn-Werner-Wunderlich syndrome. BMJ Case Rep.,14:e240001. doi: 10.1136/bcr-2020-240001. [DOI] [PMC free article] [PubMed]
- Kusunoki S., Huang K.-G., Magno A. Laparoscopic en bloc resection of a para-cervical cancer with OHVIRA syndrome. Taiwan J. Obstet. Gynecol. 2018;57(1):141–143. doi: 10.1016/j.tjog.2017.12.024. [DOI] [PubMed] [Google Scholar]
- Mei L., et al. Primary vaginal clear cell adenocarcinoma accompanied by Herlyn-Werner-Wunderlich syndrome without prenatal diethylstilbestrol exposure: a case report. Int. J. Clin. Exp. Pathol. 2020;13:2784–2787. [PMC free article] [PubMed] [Google Scholar]
- Murakami K., Kotani Y., Nakai H., Matsumura N. Endometriosis-Associated Ovarian Cancer: The Origin and Targeted Therapy. Cancers (Basel). 2020 Jun 24;12(6):1676. doi: 10.3390/cancers12061676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka, E., et al., 2020.Uterine cervical cancer associated with obstructed hemivagina and ipsilateral renal agenesis (OHVIRA) syndrome: A case report and review of the literature. Gynecol Oncol Rep., 34:100645. doi: 10.1016/j.gore.2020.100645. [DOI] [PMC free article] [PubMed]
- Purslow, C., 1992. A case of unilateral haematokolpos, haematometra and haematosalpinx. J. Obstet. Gynecol. Br .Emp., 29, 643. doi: 10.1111/j.1471-0528.1922.tb16100.x.
- Smith N.A., Laufer M.R. Obstructed hemivagina and ipsilateral renal anomaly (OHVIRA) syndrome: management and follow-up. Fertil. Steril. 2007;87(4):918–922. doi: 10.1016/j.fertnstert.2006.11.015. [DOI] [PubMed] [Google Scholar]
- Stolnicu S., Karpathiou G., Guerra E., Mateoiu C., Reques A., Garcia A., Bart J., Felix A., Fani D., Gama J., Hardisson D., Bennett J.A., Parra-Herran C., Oliva E., Abu-Rustum N., Soslow R.A., Park K.J. Clear Cell Carcinoma (CCC) of the Cervix Is a Human Papillomavirus (HPV)-independent Tumor Associated With Poor Outcome: A Comprehensive Analysis of 58 Cases. Am. J. Surg. Pathol. 2022;46(6):765–773. doi: 10.1097/PAS.0000000000001863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanase Y., Yoshida H., Naka T., Kitamura S., Natsume T., Kobayashi Kato M., Uno M., Ishikawa M., Ali Mahmoud Ali A., Kato T. Clear Cell Carcinoma of the Cervix With OHVIRA Syndrome: A Rare Case Report. World J. Oncol. 2021;12(1):34–38. doi: 10.14740/wjon1362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe Y., Etoh T., Nakai H. Adenocarcinoma of the lower female genital tract in patients with Herlyn-Werner-Wunderlich syndrome. Am. J. Obstet. Gynecol. 2012;207(6):e5–e6. doi: 10.1016/j.ajog.2012.09.009. [DOI] [PubMed] [Google Scholar]
- Wunderlich M. Unusual form of genital malformation with aplasia of the right kidney. Zentralbl. Gynakol. 1976;98:559–562. [PubMed] [Google Scholar]
- Zhu L., Chen N.a., Tong J.-L., Wang W., Zhang L., Lang J.-H. New classification of Herlyn-Werner-Wunderlich syndrome. Chin Med. J. (Engl.). 2015;128(2):222–225. doi: 10.4103/0366-6999.149208. [DOI] [PMC free article] [PubMed] [Google Scholar]