Abstract
Background
SARS-CoV-2 has been identified as the cause of the COVID-19, which caused a global pandemic. It is a pathogen that causes respiratory disease and can easily navigate the interspecies barrier. A significant number of COVID-19 cases in animals have been reported worldwide, including but not limited to animals in farms, captivity, and household pets. Thus, assessing the affected population and anticipating ‘at risk’ population becomes essential.
Objectives
This article aims to emphasize the zoonotic potential of SARS- CoV-2 and discuss the One Health aspects of the disease.
Content
This is a narrative review of recently published studies on animals infected with SARS-CoV-2, both experimental and natural. The elucidation of the mechanism of infection by binding SARS-CoV-2 spike protein to the ACE-2 receptor cells in humans has led to bioinformatic analysis that has identified a few other susceptible species in silico. While infections in animals have been extensively reported, no intermediary host has yet been identified for this disease. The articles collected in this review have been grouped into four categories; experimental inoculations, infection in wild animals, infection in farm animals and infection in pet animals, along with a review of literature in each category. The risk of infection transmission between humans and animals and vice versa and the importance of the One Health approach has been discussed at length in this article.
Keywords: Covid 19, SARS-CoV-2, One Health, Zoonosis
COVID-19 is a viral disease caused by Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2). The virus has a positive sense linear single-strand linear RNA genome that can cause respiratory, hepatic, enteric and neurological disorders in mammalian and avian species. Epidemiological links trace the first few reports back to the Huanan seafood market in Wuhan, known for housing several live animal species for sale, facilitating domestic-wild animal-human interactions. Sequence analyses revealed that there is a stark similarity between the coronavirus affecting horseshoe bats and SARS-CoV-2 that affects humans, raising questions whether the former virus was the ancestral one that traversed the species barrier, underwent adaptive genetic recombination and caused a global pandemic, causing several socio-economic-political problems across the world alike [1].
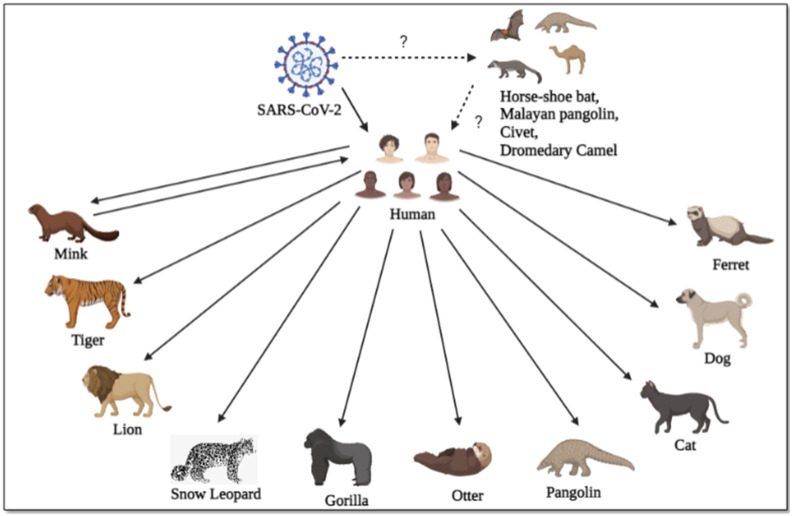
While the geographic epicenter was identified, the exact biological source of the virus remains to be discovered. Several intermediary hosts have been considered (Malayan pangolin, civet cats, and dromedary camels) for the lack of credible evidence that supports direct transmission from bats to humans [2]. Phylogenetic analysis affirms that the viruses isolated from environmental samples from the Huanan market and the early cases of COVID 19 are closely related. Arguments prevail as to whether COVID-19 should be called a ‘zoonotic disease’ for this reason, but several cases of COVID-19 in animals have been reported worldwide (Fig. 1 ).
Fig. 1.
Overview of COVID-19 spread to animals from humans and from animals to humans. Solid arrows represent confirmation of disease transmission and dotted arrows represent transmission that we are unsure of.
The genetic makeup of SARS-CoV-2 reveals it is a chimera of bat CoVRaTG13 and the receptor-binding domain (RBD) of pangolin CoV, optimized to bind the Angiotensin-Converting- Enzyme-2 (ACE-2) receptor in human cells. Further, the unique furin cleavage site activated by the host cell enzyme at the spike protein raises concerns due to low probability of presence of GCC codon that encodes the PRRA amino acid sequence at the cleavage site. Such natural gain of function recombination is rare because the coronavirus affecting bats and pangolins need to affect the same cell simultaneously at a location with a sparse density of pangolins with lower affinity of the viral spike protein. This supports the claim regarding manmade origins of the virus [3].
The ability of the viral spike protein to bind to the ACE-2 receptor across an extensive range of hosts raises questions about the host specificity of the virus and spill-back infections from humans to animals or vice versa. Such infections risk the emergence of mutant strains with increased virulence, transmissibility, lower sensitivity to neutralizing antibodies and/or reducing vaccine ability. This review captures the available literature on cases reported in animals worldwide and experimental inoculations, presenting a list of susceptible species identified so far and the need for One Health approach to combat the disease.
1. ACE-2 receptor binding and predicted hosts
Though the host specificity of SARS-CoV-2 is inexplicit, the mechanism of infection has been elucidated using bioinformatic techniques and X-ray crystallographic methods. The homotrimeric spike glycoprotein present in the envelope of SARS-CoV-2 bind to the ACE-2 receptor cells in the human cells, initiating a series of events that result in the fusion of the cell membrane with the viral membrane facilitating viral entry into the cell. Upon entry into the host, the viral spike protein is cleaved into two subunits, S1 and S2, using host proteases. The S1 domain comprises the RBD, while the S2 subunit has functional elements necessary for the fusion of the viral membrane with the host membrane. The RBD in S1 binds to the peptidase domain (PD) of the ACE-2 receptor, prompting the host proteases to cleave the C terminal of the S1 protease. Once the S1 subunit binds with the ACE-2 receptor, it disassociates from the S2 subunit. The S2 subunit then undergoes a post-fusion transformation into a dumbbell-shaped conformation that inserts into the host cell membrane. The binding of S1 receptor simultaneously prompts cleavage of the ACE 2 receptor using a disintegrin and metallopeptidase domain 17 (ADAM17)/tumor necrosis factor-converting enzyme (TACE) at the ectodomain sites and by TMPRSS2 at the intracellular C terminal domain. Following this, the cell membrane and the viral membrane fuse, releasing the viral RNA into the cytoplasm [4].
Fang et al. reported 173 species with 60% or more similarity to the human ACE-2 receptor and further identified 22 species with a high probability of susceptibility based on homology modeling and binding energies of the spike-receptor complex [4]. An in silico study reported that human ACE-2 protein shares 35%–40% sequence similarity with ectoparasitic arthropods such as water flea, deer tick and common tick. Protein structure analysis and binding energy calculations showed feasibility of the spike-receptor binding, opening up the possibility of ectoparasites being carriers of the disease [5]. The susceptibility of marine animals like whales, dolphins, seals and otters to COVID-19 owing to similar ACE protein structures to be transmitted through untreated wastewater discharge in oceans also raises profound concerns [6,7].
2. Covid 19 in wildlife
Incidence of SARS-CoV-2 in wildlife is very scant, but it is worth anticipating. In silico studies have predicted several wildlife species that are at risk, including non-human primates, lemur, reindeer, anteaters, bison, giraffes, leopards, lions, tigers, hippopotamus, etc. [7]. Natural infections or in vivo experiments have demonstrated that a few species are susceptible to infection. Lions, tigers, snow leopards, gorillas, Malayan pangolins and otters at zoological parks are reported to be infected (Table 3 ). In vivo experiments have successfully established infection in deer, raccoons, ferrets, African green monkey, rhesus monkey, and tree shrews [2,[8], [9], [10], [11], [12]].
Table 3.
SARS-CoV-2 infection in captive animals.
| S.no | Reference | Country | Animal/Category | Method of Transmission | Symptoms | Comments |
|---|---|---|---|---|---|---|
| 1. | Mcaloose et al., 2020 [35] | USA | Lion and Tiger in zoo | NA | Intermittent cough, wheezing | Lions and tigers in the Bronx Zoo were tested for SARS-CoV-2 after developing intermittent cough and wheezing. Imaging techniques like radiography and ultrasonography revealed consolidations in the thoracic cavity. ISH and RT-PCR were used to confirm viral RNA in tracheal, nasal and oropharangeal wash fluid. WGS revealed that the genome sequence of virus infecting one of the tigers is identical to that of the virus isolated from one of the keepers. |
| 2 | Xiao et al., 2020 [36] | Malayan and Chinese Pangolin in Wildlife reserves | Lung samples collected from Malayan pangolins tested positive for Sars-CoV-2 by RT-PCR while those from Chinese pangolins did not show. | |||
| 3. | Nath, 2021 https://www.indiatoday.in/cities/story/after-lioness-dies-in-chennai-zoo-nine-lions-test-positive-for-covid-1810949-2021-06-04 | India | Lion | NA | Loss of appetite, nasal discharge | Following the death of a nine year old lion on June 3, 2021, 11 lions in Aringar Anna Zoological Park were tested for COVID 19, and nine of them were found to be positive by RT PCR. Subsequently, another 12 year old lion succumbed to the disease on June 16, 2021. |
| 4. | Eight Asiatic Lions Test Positive for Coronavirus in Hyderabad Zoo - The Hindu, 2021 https://www.thehindu.com/news/national/telangana/eight-asiatic-lions-test-positive-first-in-india/article34477645.ece#:∼:text=Highly%20placed%20sources%20at%20the,a%20few%20days%20to%20confirm. | India | Lion | NA | Nasal discharge | Eight lions tested positive for the virus by RT-PCR. |
| 5. | After Hyderabad, Lion Tests Positive for Covid-19 in Jaipur Zoo - Coronavirus Outbreak News, 2021 https://www.indiatoday.in/coronavirus-outbreak/story/lion-tests-positive-covid-19-jaipur-zoo-1801929-2021-05-13 | India | Lion | NA | NA | One lion was positive for the virus by RT-PCR. |
| 6. | Lion at Etawah Safari Park Tests Covid-19 Positive - Coronavirus Outbreak News, 2021 https://www.indiatoday.in/coronavirus-outbreak/story/lion-at-etawah-safari-park-tests-covid-19-positive-1800062-2021-05-07 | India | Lion | NA | None | One lion was positive by RT-PCR |
| 7. | Gorillas At San Diego Zoo Safari Park In US Diagnosed With Coronavirus, 2021 https://www.ndtv.com/world-news/gorillas-at-san-diego-zoo-safari-park-in-us-diagnosed-with-coronavirus-2351156 (accessed July 7, 2021). | USA | Gorilla | NA | Cough and congestion | Fecal samples were collected from 8 gorillas and they were tested positive for the virus. They have recovered well. |
| 8. | Four Lions at Spanish Zoo Test Positive for COVID-19 - The Hindu, 2021 https://www.thehindu.com/news/international/four-lions-at-spanish-zoo-test-positive-for-covid-19/article33281698.ece (accessed July 7, 2021). | Spain | Lion | Contact with infected zoo keeper | Cough | Four lions were found to be positive for COVID-19 by RT-PCR test |
| 9. | Louisville Zoo Female Snow Leopard Tests Positive for SARS-CoV-2, 2021 https://louisvillezoo.org/louisville-zoo-female-snow-leopard-tests-positive-for-sars-cov-2-media-release/(accessed July 7, 2021). | USA | Snow leopard | NA | Dry cough, sneeze, wheezing | One snow leopard tested positive for SARS-CoV-2 and recovered fully. |
| 10 | Confirmation of COVID-19 in Otters at an Aquarium in Georgia, 2021 https://www.aphis.usda.gov/aphis/newsroom/stakeholder-info/sa_by_date/sa-2021/sa-04/covid-georgia-otters (accessed July 7, 2021). | USA | Otters | NA | Sneezing, runny nose, lethargy, coughing | Otters at an aquarium in Georgia tested positive for COVID-19. They have recovered fully. |
Abbreviations: RT-PCR -- Real Time Polymerase Chain Reaction, WGS -- Whole Genome Sequencing, ISH – In-situ Hybridisation, ELISA -- Enzyme Linked Immunosorbent Assay, SARS- CoV-2 – Severe Acute Respiratory Syndrome Coronavirus 2, COVID-19 – Coronavirus Disease-19, NA – Not Available.
The possible routes of SARS-CoV-2 entry in the wild could be activities of wildlife researchers, feral population control staff, forestry workers, conservationists, ecological consultancy workers and wildlife tourists. The spread of COVID-19 in wildlife cannot be contained since surveillance, quarantine facilities and appropriate health care for the infected animals cannot be easily provided. Animals living in packs or herds are at a higher risk. Infections in the prey-predator-scavenger triad is also possible if any of them are carriers.
Birds are host to many gamma and delta coronaviruses. They are potential hosts with high biodiversity, the ability to travel long distances and a unique adaptive immune system equipped for moving viruses past topographical and political boundaries. Wild birds have been implicated in spreading profoundly pathogenic H5Nx avian influenza viruses, tick-borne encephalitis virus, West Nile virus, Newcastle disease virus (NDV), and influenza A virus (IAV). These factors may make wild birds alarming sources of zoonotic CoV-spreading vectors, though SARS-CoV-2 has not been reported in birds so far [13].
The MERS epidemic witnessed the dromedary camels become reservoir hosts for the disease and humans were affected when they were in contact with them. Establishing a novel animal reservoir could become a bottleneck in human disease control measures. Moreover, the virus could replicate and mutate in the reservoir host, thus causing new epidemics and hindering vaccine or drug discoveries [1].
3. Covid 19 in farm animals
Farm animals regularly in contact with humans have always been at risk of being affected by human infections and vice versa. Experimental inoculation in calves has proven that they are not very susceptible. Out of six calves inoculated with SARS-CoV-2, nasal swabs from two were positive for COVID-19 by RT-PCR. Viral isolation was not possible in this study meaning absence of productive viral replication. Similarly, six calves were intranasally inoculated with SARS-CoV-2. Viral RNA was detected only for a short period after inoculation in two of them, which elicited a lower but identifiable serological reaction. Three naïve cattle introduced to the infected cattle developed no disease or symptoms. However, ex-vivo tissues of both bovine and ovine origins showed significant viral replication upon infection [2]. Infection was not observed during experimental inoculation in pigs and chickens [14,15]. The body temperature of pigs (39.3 °C–39.8 °C) and chickens (41 °C–42 °C) is considered to be a significant factor in disease inhibition [1].
Several cases of COVID-19 have been reported in the American mink that is farmed for its fur across Europe and USA [[16], [17], [18], [19], [20]]. Denmark has culled all of its existing minks and banned mink farming after the identification of ‘Cluster 5′ SARS-CoV-2, which caused infection in twelve humans [21]. Similarly, in Netherlands, whole genome sequencing (WGS) analysis has revealed that in sixteen farms, 68% of farm workers/people associated with the farm had COVID-19 with the virus showing an animal signature, meaning the infection traversed from mink to human [16]. Free-roaming mink, suspected to have escaped from mink farms in Utah, tested positive for SARS-CoV-2, adumbrating the possibilities of inter-species transmission in the free-ranging wildlife [22]. In 2021, Hoffman et al., reported a mutation Y453F in viruses isolated from minks. This mutation helps the virus evade antibody response in humans, should they acquire it from minks [23]. Large-scale, sustained infection in minks suggests that they could be a potential intermediate host for the disease.
4. COVID-19 in pets
Despite claims by WHO that pets are safe from COVID-19 and cannot effectively transmit the disease even if infected, several cases of infection in pets have been reported worldwide. Most of these pets were tested from households where humans were ill. WGS has also proved that the sequences obtained from humans and the pets were identical, proving spill-back infection from pets to humans [24,25]. Cats are more susceptible to COVID-19 infection than dogs and can transmit the disease to naive cats as well [15]. Multiple protein sequence analysis of human ACE-2 proteins with some common pets revealed that ACE-2 proteins of cats, golden hamsters and rabbits differ in only 3 of the 22 amino acid residues responsible for binding with SARS-CoV-2. Dogs differ in 5 of the 22 amino acid residues [26]. The possibility of reverse zoonosis is yet to be ascertained in the case of pets.
5. One health approach
The vast host range of SARS-CoV-2 is evident from the above discussion. Despite undue efforts by multifarious disciplines of infectious diseases, the reservoir host of SARS-CoV-2 is still undetermined.
Currently, two accepted models of viral disease emergence prevail; linear spillover model and circulatory model. The linear spillover model posits that the origin of zoonosis must be an animal reservoir that is highly capable of producing the virus. While the animal reservoir can consistently sustain the virus, it spills over to deluge humans (or other species). This leads to a high-frequency infection in humans. The circulation model gives a non-linear path of viral emergence, where there are susceptible or resistant hosts, sans a reservoir, where the ability of the virus to infect the host is based on its compatibility with the host cell receptor that facilitates the viral entry [1]. Provided SARS-CoV-2 is the product of a cross-species spillover, it has undergone genetic refinement in an intermediary host that has a very similar ACE-2 receptor as that of humans. If the refinement had occurred after the spillover to humans, the continuous transmission of the virus from human to human would result in natural selection opportunities that would favor the virus [22]. Howevr, there is no conclusive evidence of either of these hypotheses.
Many viral diseases have been zoonotically transmitted, like ebola, Marburg virus, SARS, MERS, chikunguniya, rabies etc. Human-to-animal transmission of SARS-CoV-2 has been speculated in many cases of COVID-19. In some cases, concrete proof of human-to-animal transmission is available; as in the case of infection in minks in Denmark, Netherlands and USA and in pet and captive animals across the globe, where the animals have been in contact with infected humans and the source of infection in these animals have been conclusively traced back to be of human origin (Table 1, Table 2, Table 3, Table 4 ). Another reason to worry is the traceback of human infection outbreaks to mink sources in the Netherlands [16].
Table 1.
SARS -CoV-2 infection in experimental animal models.
| S.no | Reference | Country | Animal/Category | Method of infection | Symptoms | Comments |
|---|---|---|---|---|---|---|
| 1. | Falkenberg et al., 2021 | USA | Deer | Experimental inoculation in fawns via nasal route. | Increase in body temperature in the fawns inoculated with SARS- CoV2 virus | RT PCR was used to detect infection in inoculated fawns in nasal secretions and fecal samples. It was further confirmed by ISH of palatine tonsils and medial retropharyngeal lymph nodes. |
| 2. | Fagre et al., 2020 [28] | USA | Deer Mice | Experimental inoculation in deer mice via nasal route | NA | RT-PCR was used to confirm infection in oral swabs (until 21st day post infection) and in lungs. There was an increase in activity of several innate immune response genes in the lungs, namely IFNα, TBK1, Pycard, Oas2, Cxcl10. Deer mice that were exposed to the infected mice were positive for viral RNA within two days after contact. Immunohistochemistry showed the presence of viral antigens in the brain tissue of infected animals, which suggests the successful break down of blood brain barrier by the virus. |
| 3. | Freuling et al., 2020 | Germany | Racoons | Nasal route in one group while the second group was exposed to infected animals to test for direct transmission | Lethargy | Viral RNA was identified in nasal and oroparangeal swabs of inoculated animals between 2nd and 4th day post infection and after the 10th day in animals that were exposed. Immunohistochemistry confirmed the presence of viral antigens on the 4th and 8th day post infection in the nasal respiratory and the olfactory epithelium. ELISA confirmed the presence of viral antibodies in 4 out of 7 inoculated animals on the eighth day post infection. |
| 4. | Selvan et al., 2021 | USA | Cats | Intratracheal inoculation | Fever, lethargy, respiratory distress, ocular and nasal discharge, wheezhing. | Necropsy of inoculated cats after euthanisation on the 4th and 8th day post inoculation showed dark red regions of consolidation exudating edema upon cut surface. Further examination revealed that the histologic changes in the lungs and vascular tissues were very identical to that of COVID-19 in humans. |
| 5. | Falkenberg et al., 2021 | USA | Calves | Intratracheal and intravenous inoculation | NA | Nasal swabs, whole blood and voided urine collected at regular intervals post infection were positive for virus by RT-PC as well as ISH and were also positive for viral antibodies by ELISA. |
| 6. | Schlottau et al., 2020 | Germany | Pigs, Chicken, fruit bats and ferrets | Intranasally | None | Pigs, chicken, fruit bats and ferrets were inoculated intranasally. Pigs and chickens did not test positive for the virus by RT-PCR nor showed antibodies for the virus. Seven (78%) of nine fruit bats had infection and was confirmed by RT-PCR, ISH and immunohistochemistry from trachea, lung and lung-associated lymphatic tissue. One out of three contact bats were infected with the virus. Efficient virus replication was observed in ferrets although clinical signs were absent. No transmission was observed with contact animals. |
| 7. | Bosco-lauth et al., 2020 [30] | USA | Cats and dogs | Intranasally | NA | RT- PCR confirmed the infection in both cats and dogs. ELISA confirmed the presence of viral antibodies in both cats and dogs. Cats were more susceptible to infection and mounted a stronger immune response than cats. When the cats were reinfected, no infection was observed. |
| 8. | Kim & Webby, 2020 | Republic of Korea | Ferret | Intranasally | Fever | Viral RNA was detected by RT-PCR on the 2nd day post infection. ISH and histopathological analyses also confirmed viral infection. |
| 9. | Gaudreault et al., 2021 | USA | Cat | Intranasally | NA | Cats that were initially exposed to the virus were re-exposed 21st day post infection. The cats developed infection |
| 10. | Woolsey et al., 2020 [32] | USA | African Green Monkey | Intranasally | Fever, loss of appetite | Virus was detected from nasal swabs 2nd day post infection in nasal swabs and bronchoalveolar lavage by RT-PCR and ISH (from pnemonocytes). Histological analyses on the 5th day post infection revealed multifocal pulmonary lesions. |
| 11. | Munster et al., 2020 | USA | Rhesus Monkey | Intranasally | Respiratory distress, coughing, loss of appetite, dehydration, hunched posture | RT-PCR detected the presence of virus after one day post infection in nasal swabs. Antibodies against the virus were detected in the blood samples by ELISA on the tenth day post infection. Necropsy on 21st day post infection showed multiple lesions in the lungs. |
| 12. | Sia et al., 2020 [33] | Hong Kong | Golden Hamsters | Intranasally | None | Viral RNA was detected in nasal swabs on the 2nd day post infection. Immunohistochemistry revealed the presence of viral antigens on nasal mucosa on the second day post infection. Naïve hamsters housed with the diseased hamsters were also infected. |
| 13. | Zhao et al., 2020 | Tree shrew | Intranasally | Fever | Viral RNA was observed by RT-PCR in nasal swabs on the 6th day post infection. Necropsy revealed lesions in the lungs, widened pulmonary septum, and local hemorrhagic necrosis. | |
| 14. | Bertzbach et al., 2020 [34] | Germany | Chinese Hamster | Intranasally | Drop in weight and body temperature | RT-PCR and ISH performed on bucco larangeal swabs, lungs and blood from the second day post infection were positive for virus. Necropsy revealed alveolar damage from the second day until 14th day post infection. |
| 15. | Shi et al., 2020 | Ferrets, cats, dogs, ducks, chicken | Intranasally | NA | Ferrets and cats are highly susceptible to SARS-CoV-2; dogs have low susceptibility; and pigs, chickens, and ducks are not susceptible to the virus |
Abbreviations: RT-PCR -- Real Time Polymerase Chain Reaction, WGS -- Whole Genome Sequencing, ISH – In-situ Hybridisation, ELISA -- Enzyme Linked Immunosorbent Assay, SARS- CoV-2 – Severe Acute Respiratory Syndrome Coronavirus 2, COVID-19 – Coronavirus Disease-19, NA – Not Available.
Table 2.
SARS-CoV-2 infection in farm animals.
| S.no | Reference | Country | Animal/Category | Method of Transmission | Symptoms | Comments |
|---|---|---|---|---|---|---|
| 1. | Munnink et al., 2021 | Netherlands | Mink | From infected animal handlers | Respiratory distress | Minks and animal handlers of 16 farms were tested for infection. WGS showed that the virus infecting 16 farms belonged to 5 different clusters. It also revealed that on 14 of the 16 farms, the sequence of virus infecting human and mink were almost identical. |
| 2. | Doma et al., 2021 | Poland | Mink | NA | None | Oral and rectal swabs from minks in 28 farms were collected. 20 minks belonging to the same farm tested positive for the virus by RT-PCR. 120 blood samples were collected from infected animals prior to culling, out of which 30 were positive for viral antibodies. Throat swabs and blood samples were collected from additional 30 animals, out of which 15 were positive for both viral RNA and antibody. |
| 3. | Boklund et al., 2021 | Denmark | Mink | NA | Respiratory symptoms and reduced feed intake. | Minks in 215 farms in Denmark tested positive by RT-PCR from June to November 2020. Infection was also seen in dogs and cats near certain farms. |
| 4. | Oreshkova et al., 2020 | Netherlands | Mink | NA | Respiratory distress, nasal discharge | Minks were tested in two farms from a period of April to May 2020 in Netherlands. Throat swabs and rectal swabs confirmed the presence of viral RNA in 36 minks on both farms. Twenty four stray cats in the surroundings of the farms were tested and very low amount of viral RNA was detected in one cat. |
| 5. | Hammer et al., 2021 | Denmark | Mink | NA | NA | Minks were tested in three farms in Northern Jutland in Denmark. Throat, nasal and fecal swabs showed they were positive viral DNA by RT-PCR. ELISA confirmed the presence of viral antibodies in blood samples collected from these animals. Humans in contact with the minks also tested positive for the virus. WGS showed that both the human and mink were infected with the European 20B clade of the global SARS-CoV-2 tree. |
Abbreviations: RT-PCR -- Real Time Polymerase Chain Reaction, WGS -- Whole Genome Sequencing, ISH – In-situ Hybridisation, ELISA -- Enzyme Linked Immunosorbent Assay, SARS- CoV-2 – Severe Acute Respiratory Syndrome Coronavirus 2, COVID-19 – Coronavirus Disease-19, NA – Not Available.
Table 4.
SARS- CoV-2 infection in pets.
| S.no | Reference | Country | Animal/Category | Method of Transmission | Symptoms | Comments |
|---|---|---|---|---|---|---|
| 1 | Sit et al., 2020 [37] | Hong Kong | Dog | From infected owners | In Hong Kong, nasal swabs and blood samples were collected from seven cats and fifteen dogs whose owners have been diagnosed with COVID-19. Two of the dogs tested positive for the virus and viral antibodies by RT-PCR and ELISA respectively. | |
| 2. | Gaudreault et al., 2021 [31] | Cat | NA | NA | The study tested the serum samples of stray cats collected for different purposes before the pandemic and during the pandemic and reported that no cats had antibodies for SARS-CoV-2 in the former case and only 1% of the total cats tested positive in the latter. | |
| 3 | Klaus et al., 2021 | Italy | Cat | From infected owner | Sneezing, cough, wheezing | The 12-year-old cat that had lymphoma tested positive for SARS- CoV-2 by RT-PCR when the owners were infected. Blood samples collected from the cat six months after the infection still showed the presence of antibodies. |
| 4 | Klaus et al., 2021 [38] | Switzerland | Cat | From infected owner | Sneezing, cough, wheezing in one of the cats. The other one was asymptomatic | Nasal swab, fecal sample and fur tested positive for virus by RT PCR and ELISA confirmed the presence of viral antibodies. |
| 5 | Verde et al., 2021 [39] | Ferret | None | Blood samples were collected from 127 ferrets and ELISA confirmed the presence of viral antibodies in two of them | ||
| 6 | Hamer et al., 2020 | USA | Cat and dog | From infected owners | None | Nasal and rectal swabs collected from cats snd/or dogs in the households with confirmed COVID 19 patients were positive for the virus by RT-PCR. WGS of the virus isolated from the infected animals. |
| 7. | Neira et al., 2021 | Chile | Cats | From infected owners | Nasal swabs and fecal samples collected from cats from households where the owners were infected with COVID 19 were positive for virus by RT-PCR. Serology showed that few cats and dogs had antibodies for the virus as well. WGS of the virus isolated from the cats and dogs were identical to those that were obtained from humans. | |
| 8. | Pagani et al., 2021 [40] | Italy | Cat | Infected pet owner | Sneezing | Nasal swab was collected from the four year old short hared cat following sneezing was positive for RT-PCR. WGS analysis performed on the strains retrieved from both the cat and the owner showed 99.9% nucleotide similarity. |
Abbreviations: RT-PCR -- Real Time Polymerase Chain Reaction, WGS -- Whole Genome Sequencing, ISH – In-situ Hybridisation, ELISA -- Enzyme Linked Immunosorbent Assay, SARS- CoV-2 – Severe Acute Respiratory Syndrome Coronavirus 2, COVID-19 – Coronavirus Disease-19, NA – Not Available.
Following infection in minks, stray cats that were not in contact with infected humans in and around mink farms were found to be positive for SARS-CoV-2 antibodies, suggesting possible transmission [19]. Another concern is the possibility of disease transmission to other wild species, like ferrets. Experimental inoculation in ferrets has been very successful and the progression of infection in ferrets is deemed very close to humans recommending that they be used as model organisms in SARS-CoV-2-related studies [10,14,15]. The incidence of natural infection in such a susceptible population leads to establishment of reservoir hosts risking the transmission of the animal-adapted virus back to humans, thus beginning a vicious cycle that would lead to continued viral disease outbreaks even after the initial outbreaks have been contained [22].
The World Health Organisation (WHO) defines One Health as “an approach to designing and implementing programs, policies, legislation and research in which multiple sectors communicate and work together to achieve better public health outcomes”. This requires coordination from various fields, including physicians, epidemiologists, veterinarians, and researchers in natural sciences, social sciences and environmental sciences. While the presence of an intermediary host is still an enigma, it becomes necessary to focus on One Health to curb transmission in the human-animal interface and stop the persecution of animals alleged to be reservoirs.
The World Organization For Animal Health (OIE) recognizes the threat of the emergence of new animal-adapted strains and the ability of such strains to hinder treatment of vaccine efficacy in humans. It urges collaborations between human and animal health authorities to understand the nuances of the situation. It advocates countries to protect animal welfare which in turn could prevent disease in humans. The plan of action proposed by OIE for this purpose is preventing viral transmission from humans to susceptible animals; monitoring susceptible animals; reporting the cases of animal COVID-19; Sharing genetic sequences of virus isolated from the infected animals to the global health community (https://wahis.oie.int/#/report-info?reportId=16330).
Stringent systematic surveillance methods are needed to monitor disease spread in farm animals, pet animals and captive animals housed in zoological parks. This approach might seem impractical for wild animals, but appropriate safety measures are required. While there is no information regarding the immunization efficiency of SARS-CoV-2 in animal front, pharmaceutical companies have concocted different kinds of vaccines for the human front. A similar approach might prove helpful on the animal front.
While it is difficult to identify vaccine leads for several different animal species and bring a candidate to the pipelines, Karni-Kov, the first vaccine for carnivorous animals, has been launched in Russia. The vaccine is designed to protect carnivores against SARS-CoV-2 and trials have been successful in arctic foxes, cats, rats and mink [27]. The LinearDNATM is also an animal vaccine candidate for cats. The United States Department of Agriculture (USDA) has given regulatory approval for this candidate (http://pharmabiz.com/NewsDetails.aspx?aid=133853&sid=2). Eight great apes in the San Diego zoo have been vaccinated against SARS-CoV-2 with an experimental vaccine candidate developed by Zoetis (https://www.nationalgeographic.com/animals/article/first-great-apes-at-us-zoo-receive-coronavirus-vaccine-made-for-animals). Vaccinations are necessary for pets and farm animals with a notable human-animal interface. Even though vaccines do not produce sterilizing immunity in animals, they remain a precaution to reduce the shared risk and strengthen the One Health approach.
6. Conclusion
SARS-CoV-2 infection has been reported in various animal species thus far. There is a compelling need to pursue detailed investigations regarding the virus. Implementing the One Health approach is of paramount importance and dynamic risk assessment combined with targeted surveillance programs can help curb the disease.
Declaration of competing interest
The authors have no conflict of interest.
Ethical approval
The authors have declared that ethical statement is not applicable for this manuscript.
Funding
The project was funded by ICMR (Grant ID: ZON/41/2019/ECD-II). The funding agency has no role in planning this study.
References
- 1.Maurin M., Fenollar F., Mediannikov O., Davoust B., Devaux C. Current status of putative animal sources of SARS-CoV-2 infection in humans: wildlife, domestic animals and pets. Microorganisms. 2021;9 doi: 10.3390/microorganisms9040868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Falkenberg S., Buckley A., Laverack M., Martins M., Palmer M.V., Lager K., et al. Experimental inoculation of young calves with SARS-CoV-2. Viruses. 2021;13:1–15. doi: 10.3390/v13030441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Segreto R., Deigin Y. The genetic structure of SARS-CoV-2 does not rule out a laboratory origin. Bioessays. 2020;43 doi: 10.1002/bies.202000240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fang S., Zheng R., Lei C., Wang J., Zheng R., Li M. Key residues influencing binding affinities of 2019-nCoV with ACE2 in different species. Briefings Bioinf. 2020:1–13. doi: 10.1093/bib/bbaa329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lam S.D., Ashford P., Díaz-Sánchez S., Villar M., Gortázar C., Fuente J de la, et al. Arthropod ectoparasites have potential to bind SARS-CoV-2 via ACE. Viruses. 2021;13 doi: 10.3390/v13040708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mathavarajah S., Stoddart A.K., Gagnon G.A., Dellaire G. Pandemic danger to the deep: the risk of marine mammals contracting SARS-CoV-2 from wastewater. Sci Total Environ. 2020:760. doi: 10.1016/j.scitotenv.2020.143346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fischhoff I.R., Catellanos A.A., Rodriguez J.P., Varsani A., Han B.A. Predicting the zoonotic capacity of mammals to transmit SARS-CoV-2.pdf. Prepr BioRxiv. 2021 doi: 10.1101/2021.02.18.431844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhao Y., Wang J., Kuang D., Xu J., Yang M., Ma C., et al. Susceptibility of tree shrew to SARS - CoV - 2 infection. Sci Rep. 2020:1–9. doi: 10.1038/s41598-020-72563-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Freuling C.M., Breithaupt A., Müller T., Sehl J., Balkema-Buschmann A., Rissmann M., et al. Susceptibility of raccoon dogs for experimental SARS-CoV-2 infection. Emerg Infect Dis. 2020;26:2982–2985. doi: 10.3201/eid2612.203733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim S., Webby R.J. Infection and rapid transmission of SARS-CoV-2 in ferrets. Cell Host Microbe. 2020;27:704–709. doi: 10.1016/j.chom.2020.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Munster V.J., Feldmann F., Williamson B.N., Doremalen N Van, Pérez-pérez L., Schulz J., et al. Respiratory disease in rhesus macaques inoculated with SARS-CoV-2. Nature. 2020;585 doi: 10.1038/s41586-020-2324-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Woolsey C, Borisevich V, Prasad AN, Agans KN, Deer DJ, Dobias NS, et al. Establishment of an African green monkey model for COVID-19 and protection against re-infection. Nat Immunol n.d. 10.1038/s41590-020-00835-8. [DOI] [PMC free article] [PubMed]
- 13.Rahman A. Coronaviruses in wild birds – a potential and suitable vector for global distribution. Vet Med Sci. 2021;7 doi: 10.1002/vms3.360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schlottau K., Rissmann M., Graaf A., Schön J., Sehl J., Wylezich C., et al. SSARS-CoV-2 in fruit bats, ferrets, pigs, and chickens: an experimental transmission study. Lancet Microbe. 2020;1:18–25. doi: 10.1016/S2666-5247(20)30089-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shi J., Wen Z., Zhong G., Yang H., Wang C., Huang B., et al. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS-coronavirus 2. Science. 2020;368:1016–1020. doi: 10.1126/science.abb7015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Munnink B.B.O., Sikkema R.S., Nieuwenhuijse D.F., Molenaar R.J., Munger E., Molenkamp R., et al. Transmission of SARS-CoV-2 on mink farms between humans and mink and back to humans. Science. 2021;371:172–177. doi: 10.1126/science.abe5901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Doma K., Orłowska A., Smreczak M., Niemczuk K., Iwan E., Bomba A., et al. Mink SARS-CoV-2 infection in Poland. Short Communication. 2021:1–5. doi: 10.2478/jvetres-2021-0017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Boklund A., Hammer A.S., Quaade M.L., Rasmussen T.B., Lohse L., Strandbygaard B., et al. 2021. SARS-CoV-2 in Danish mink farms : course of the epidemic and a descriptive analysis of the outbreaks in 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Oreshkova N., Molenaar R.J., Vreman S., Harders F., Munnik B.B.O., Hakse-van der Honing R.W., et al. SARS-CoV-2 infection in farmed minks, The Netherlands, April and May 2020. Euro Surveill. 2020;25(23):1–7. doi: 10.2807/1560-7917.ES.2020.25.23.2001005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hammer A.S., Quaade M.L., Rasmussen T.B., Fonager J., Rasmussen M., Mundbjerg K., et al. SARS-CoV-2 transmission between mink (neovison vison) and humans, Denmark. Emerg Infect Dis. 2021;27:547–551. doi: 10.3201/eid2702.203794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Larsen C.S., Paludan S.R. Corona's new coat: SARS-CoV-2 in Danish minks and implications for travel medicine. Trav Med Infect Dis. 2020;38 doi: 10.1016/j.tmaid.2020.101922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Khan S., Ruchi T., Senthilkumar N., Kuldeep D. SARS-CoV-2 infection in farmed minks , associated zoonotic concerns , and importance of the One Health approach during the ongoing COVID- 19 pandemic. Vet Q. 2021;41:50–60. doi: 10.1080/01652176.2020.1867776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hoffmann M., Zhang Lu, Kr€uger N., Schulz S., Ck H.-M.J., Hlmann S.P. SARS-CoV-2 mutations acquired in mink reduce antibody-mediated neutralization. Cell Rep. 2021;35 doi: 10.1016/j.celrep.2021.109017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hamer S.A., Jenkins-moore M., Mozingo K., Akpalu Y., Ghai R.R., Spengler J.R. Natural SARS-CoV-2 infections, including virus isolation, among serially tested cats and dogs in households with confirmed human COVID-19 cases in Texas, USA. bioRxiv. 2020:1–16. [Google Scholar]
- 25.Neira V., Brito B., Agüero B., Berrios F., Valdés V., Gutierrez A. A household case evidences shorter shedding of SARS-CoV-2 in naturally infected cats compared to their human owners. Emerg Microb Infect. 2021;10 doi: 10.1080/22221751.2020.1863132. https://doi.org/10.1080/22221751.2020.1863132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.De Morais H.A., Pires A., Cannes N., Kmetiuk L.B., Barbosa D.S., Brandão P.E., et al. Natural infection by SARS-CoV-2 in companion animals : a review of case reports and current evidence of their role in the. Epidemiology. 2020;7:1–10. doi: 10.3389/fvets.2020.591216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chavda V.P., Feehan J., Apostolopoulos V. A veterinary vaccine for SARS-CoV-2: the first COVID-19 vaccine for animals. Vaccines. 2021;9:631. doi: 10.3390/vaccines9060631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fagre A., Lewis J., Eckley M., Zhan S., Rocha S.M., Sexton N.R., et al. SARS-CoV-2 infection, neuropathogenesis and transmission among deer mice: implications for reverse zoonosis to New World rodents. bioRxiv. 2020 doi: 10.1101/2020.08.07.241810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bosco-lauth A.M., Hartwig A.E., Porter S.M., Gordy P.W., Nehring M., Byas A.D., et al. Vol. 117. 2020. (Experimental infection of domestic dogs and cats with SARS-CoV-2 : pathogenesis , transmission , and response to reexposure in cats). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gaudreault N.N., Carossino M., Morozov I., Trujillo J.D., Meekins D.A. Experimental re-infected cats do not transmit SARS-CoV-2. Emerg Microb Infect. 2021;10 doi: 10.1080/22221751.2021.1902753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Woolsey C., Borisevich V., Prasad A.N., Agans K.N., Deer D.J., Dobias N.S., et al. Establishment of an African green monkey model for COVID-19. bioRxiv. 2020:1–20. doi: 10.1038/s41590-020-00835-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sia S.F., Yan L., Chin A.W.H., Fung K., Choy K., Wong A.Y.L., et al. Pathogenesis and transmission of SARS-CoV-2 in golden hamsters. Nature. 2020;583 doi: 10.1038/s41586-020-2342-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bertzbach L.D., Vladimirova D., Dietert K., Abdelgawad A., Gruber A.D., Osterrieder N., et al. SARS-CoV-2 infection of Chinese hamsters (Cricetulus griseus) reproduces COVID-19 pneumonia in a well-established small animal model.pdf. Transbound Emerg Dis. 2020:1–5. doi: 10.1111/tbed.13837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mcaloose D., Laverack M., Wang L., Killian L., Caserta L.C., Yuan F., et al. From people to Panthera : natural SARS-CoV-2 infection in tigers and lions at the bronx zoo. Am Soc Microbiol. 2020;11:1–13. doi: 10.1128/mBio.02220-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xiao K., Zhai J., Feng Y., Zhou N., Zhang X., Zou J., et al. Isolation of SARS-CoV-2-related coronavirus from Malayan pangolins. Nature. 2020;583 doi: 10.1038/s41586-020-2313-x. [DOI] [PubMed] [Google Scholar]
- 37.Sit T.H.C., Brackman C.J., Ip S.M., Tam K.W.S., Law P.Y.T., To E.M.W., et al. Canine SARS-CoV-2 infection. Nature. 2020;586:776–778. doi: 10.1038/s41586-020-2334-5.Canine. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Klaus J., Meli M.L., Willi B., Nadeau S., Beisel C., Stadler T., et al. Detection and genome sequencing of SARS-CoV-2 in a domestic cat with respiratory signs in Switzerland. Viruses. 2021;13 doi: 10.3390/v13030496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Verde M., Garc A., Taleb V., Lira-navarrete E. SARS-CoV-2 seroprevalence in household domestic ferrets. Mustela putorius furo) 2021;1–11 doi: 10.3390/ani11030667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pagani G., Lai A., Bergna A., Rizzo A., Stranieri A., Giordano A., et al. Human-to-cat sars-cov-2 transmission: case report and full-genome sequencing from an infected pet and its owner in Northern Italy. Pathogens. 2021;10:1–6. doi: 10.3390/pathogens10020252. [DOI] [PMC free article] [PubMed] [Google Scholar]