ABSTRACT
Background:
Amenorrhoea is considered a kind of menstrual disorder in a woman of reproductive age. It is a symptom with many potential causes such as an abnormality in the hypothalamic–pituitary–ovarian axis, anatomical abnormalities of the genital tract or functional causes.
Aims:
In this study, we aimed to investigate chromosomal abnormalities in patients presenting with primary amenorrhoea.
Study Setting and Design:
This study was conducted in the medical genetic laboratory.
Materials and Methods:
Chromosomal analysis was carried out in 134 cases that were referred to the human genetic laboratory from 2010 to 2017, employing (GTG) Giemsa banding.
Statistical Analysis:
Statistical analyses were carried out by Microsoft Office Excel (2019).
Results:
The karyotype results revealed 77.6% (n = 104) with normal chromosome composition while 22.38% (n = 30) showed chromosomal abnormalities. Among the patients with abnormal chromosome constituents, 53.54% exhibited numerical aberration and 46.66% showed structural abnormalities.
Conclusion:
The present study has emphasised that karyotyping is one of the fundamental investigations in the evaluation of primary amenorrhoea.
KEYWORDS: Chromosomal abnormality, cytogenetic study, karyotyping, primary amenorrhoea
INTRODUCTION
Most girls begin menstruating between the ages of 9 and 18, with an average of around 12 years. If other pubertal changes occur naturally, the absence of menstruation until the age of 15 is not considered primary amenorrhoea. Primary amenorrhoea may occur with or without other signs of puberty.[1,2]
Although there are many reasons for primary amenorrhoea including constitutional delay, chronic systemic disease, hypothalamic-pituitary dysfunction and absent ovarian function, genetic or chromosomal causes are the most important as their presence affects subsequent management.[3,4]
Primary amenorrhoea can be caused by chromosomal disorders such as Turner syndrome, gonadal disorders, structural problems with the uterus and structural abnormalities of the Fallopian tubes which could prevent or inhibit the normal development or the release of eggs into the uterus.[5,6] According to previous studies, the percentage of chromosomal abnormalities varies from 15.9% to 63.3% in patients with primary amenorrhoea.[3,6] This study was undertaken to determine the frequency and type of chromosomal abnormalities that result in primary amenorrhoea in the local population.
MATERIALS AND METHODS
In this study, we used the data obtained from patients (n = 134) referred with primary amenorrhoea for chromosomal analysis. The cases were referred from different parts of Iran between 2010 and 2017. The age group of the participants ranged from 17 to 30 years. For chromosomal abnormality investigation, routine lymphocyte cell culture and the G-banding methods were used.
About 1 ml of blood was added to 6 ml of Roswell Park Memorial Institute medium supplemented with 10% of foetal bovine serum and 0.1 μg/ml of phytohaemagglutinin and was incubated at 37°C. Cells were treated with 0.1 μg/ml of colcemid after a 72-h incubation period, after which metaphase chromosomes of each patient were spread on a slide and stained using the standard G-banding technique.[7,8] A minimum of 25 metaphase cells per slide were analysed to find any numerical or structural chromosomal abnormalities using a microscope. If an abnormal karyotype was found in this first analysis, further cells (up to 60) could be examined.
This research and methods were performed under the ethical principles, national norms, relevant guidelines and regulations for conducting medical research in Iran. Written informed consent was obtained from all eligible subjects. The project was approved by the science and research branch of Tehran's Islamic azad university's ethical committee (Tehran, Iran, IR.IAU.SRB.REC.1398.001).
For this cross-sectional study, the sample size was calculated based on Cochran's formula using an online sample size calculator website (https://www.calculator.net/sample-size-calculator.html). Obtained desired sample size for this study was 130 individuals.
RESULTS
Among the 134 patients, 30 had abnormal karyotypes. The chromosomal analysis and the obtained karyotype for the patients are summarised in Table 1. Chromosomal abnormalities can be classified into five main types with or without mosaicism [Figure 1]:
Table 1.
Chromosomal abnormalities in primary amenorrhea
| Karyotypes | n |
|---|---|
| 45, X/46, XX | 7 |
| 45, X | 6 |
| 48, XXXX | 1 |
| 45, X/46, i (Xq)/47, X, i (Xq), i (Xq) | 1 |
| 45, X/46, XY | 1 |
| 46, XY | 4 |
| 46, X, i (Xq) | 4 |
| 46, XX, del (X)(q13) | 1 |
| 46, XX, del (X)(q23) | 1 |
| 46, XX, del (X)(q26) | 1 |
| 45, X/46, XX, del (X)(q23) | 1 |
| 46, XX, del (Xp) | 1 |
| 46, i (Xp) | 1 |
| Total | 30 |
Figure 1.
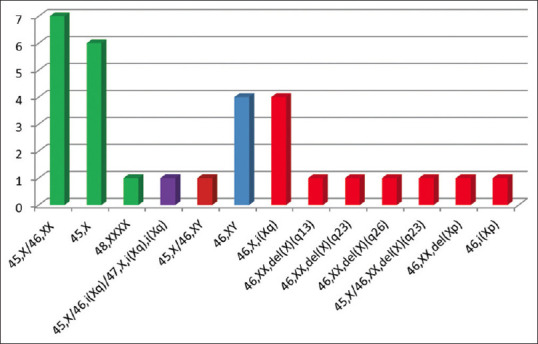
Chromosome abnormalities in primary amenorrhoea
The most frequent karyotype was X chromosome aneuploidies (n = 14) that include Turner syndrome 45, X (n = 6), mosaic Turner 45, X/46, XX (n = 7) and 48, XXXX (n = 1).
A male karyotype of 46, XY was present in four patients.
Structural anomalies of the X chromosome were detected in 10 cases. Four patients were found to have an isochromosome of the long arm of the X chromosome (×46, i[Xq]), one patient had an isochromosome of the short arm of the X chromosome (×46, i[Xp]) and five patients had a partial deletion of the X chromosome.
Mosaicism of male chromosome constitution and X chromosome aneuploidy was present in one case (45, X [39]/46, XY [21]).
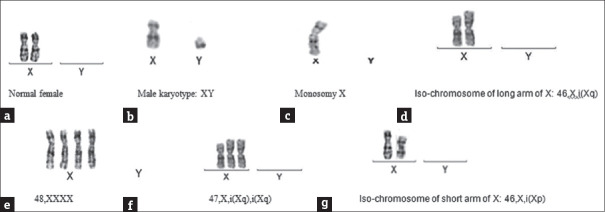
Mosaicism of X chromosome aneuploidy and structural anomalies of the X chromosome were found in one case 45, X [12]/46, i (Xq) [27]/47, X, i (Xq), i (Xq).[17] Partial karyotype images of the chromosome abnormalities are shown in Figure 2.
Figure 2.
Partial karyotype images of the chromosome abnormalities (only sex chromosome images of the karyotypes are shown). (a) Normal female. (b) Male karyotype: XY. (c) Monosomy X. (d) Iso-chromosome of long arm of X: 46,X,i(Xq). (e) 48,XXXX. (f) 47,X,i(Xq),i(Xq). (g) Iso-chromosome of short arm of X: 46,X,i(Xp)
DISCUSSION
Primary amenorrhoea is the complete absence of menstruation. Genetic factors have a causal effect in addition to endocrine disturbances and constitutional and environmental factors in primary amenorrhoea.[9] Several cytogenetic studies have been performed aimed at understanding the frequency and nature of chromosomal abnormalities in primary amenorrhoea.[10,11] In the present study, the incidence of chromosomal abnormalities in amenorrhoea was found to be 22.38%. The reported incidence in different kinds of literature ranges from 13% to 32% and numerical X chromosomal abnormality is frequent.[12]
Among the different types of chromosomal abnormalities, either complete monosomy of X (n = 6) or mosaicism (n = 7) was observed in 43.3% of cases. The obtained results were in line with the previous reports that indicate Turner's syndrome as the leading cause of primary amenorrhoea. The obtained results further strengthened the role of gene composition on the X chromosome in normal female physiology and reproduction. The presence of extra X chromosomes causes ovarian failure due to hormonal imbalance.[14] Complete or partial deletion of chromosome X and consequent gene insufficiency in deleted regions also leads to gonadal dysgenesis.[13] It is known that deletion of the long arm of the X chromosome leads to ovarian failure.[14] The ZFX gene is another critical gene for the normal development of female gonads that is located on the short arm of the X chromosome.[15] The lack of Short Stature Homeobox (SHOX), a vital growth development gene located on the short arm of the X chromosome, has also been reported to be a major cause of Turner syndrome phenotype and primary amenorrhoea.[14]
Sex-reversed female, individuals with female phenotype but 46, XY karyotype, occurs in the embryos with a mutation or deletion on testis determining factor, the sex determination region of the chromosome Y (SRY gene).[14,16] These subjects often represent normally developed Müllerian ducts, gonadal dysgenesis, poorly developed breast, absence of the uterus or ovaries and primary amenorrhoea.[14,16] The chromosomal abnormalities in primary amenorrhoea could be grouped as the X numerical; X structural, structural and numerical abnormalities together, 46, XY female and sex reversal condition [Figure 3]. Their frequencies from different reported articles are: 45, X (40%–50%), X mosaicism (25%–36%), X structural (8%), 46, XY female (16%) and mosaicism of male chromosome constitution (45, X/46, XY) (1.36%).[5,6,15,17] The frequency of chromosomal anomalies among different populations is summarised in Table 2. In the present study, the respective frequency is 20% (45, X), 23.3% (X mosaicism), 32% (X structural), 13.3% (46, XY) and 3.3% (45, X/46, XY). A rare type of aneuploidy together with structural anomalies of the X chromosome (45, X/46, X, i[Xq]/47, X, i[Xq], i[Xq]) was observed in one of the studied cases. Two reports of this anomaly have previously been published by Melaragno MI, et al., (1993) and Gorukmez O, et al. (2015).[18,19] Although the results of this study emphasise the role of chromosomal abnormalities as a causative factor of primary amenorrhoea, as a limitation of the study, other possible factors such as molecular markers have not been included in this study, which need further investigation.
Figure 3.
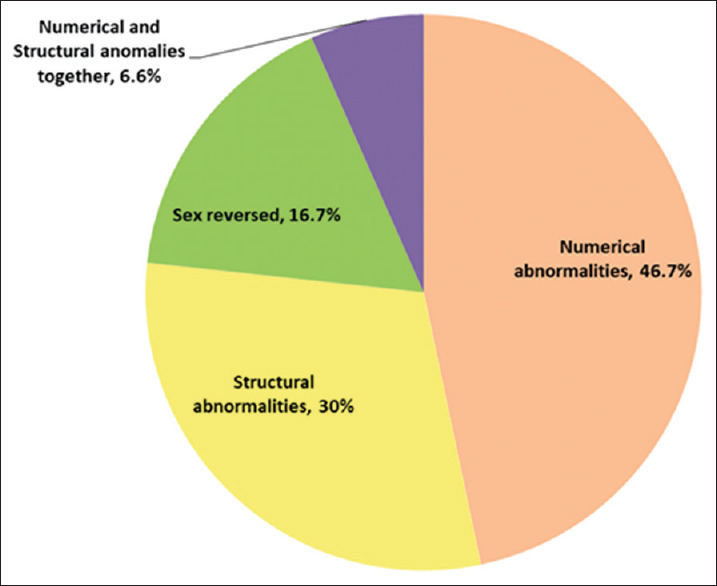
Chromosomal abnormalities in primary amenorrhoea
Table 2.
Frequency of chromosomal abnormalities among different populations
| Population (references) | Number of cases with chromosomal anomalies | Numerical abnormalities (%) | Structural abnormalities (%) | Sex reversed (%) | Numerical and structural anomalies together (%) |
|---|---|---|---|---|---|
| India[3] | 162 out of 620 cases | 43.83 | 21.6 | 34.57 | - |
| India[6] | 39 out of 140 cases | 53.84 | 2.56 | 20.5 | 23.1 |
| India[6] | 36 out of 150 cases | 50 | 30.5 | 8.3 | 11.2 |
| India[12] | 121 out of 490 cases | 43.8 | 26.4 | 29.8 | - |
| India[15] | 1177 out of 3776 cases | 31.2 | 21.7 | 33.9 | 13.2 |
| India[16] | 23 out of 174 cases | 34.78 | 8.7 | 43.47 | 13.05 |
| Indonesia[18] | 24 out of 79 cases | 45.8 | 4.2 | 33.3 | 16.7 |
| Turkey[20] | 44 out of 175 cases | 34 | 32 | 29.5 | 4.5 |
| China[21] | 58 out of 237 cases | 50 | 12 | 34.5 | 3.5 |
| Malaysia[22] | 34 out of 117 cases | 26.5 | 11.8 | 50 | 11.7 |
| Tunis[23] | 16 out of 40 cases | 43.8 | 18.8 | 31.2 | 6.2 |
| Romania[24] | 269 out of 493 cases | 79.9 | 5.6 | 5.3 | 9.2 |
| Our study | 30 out of 134 cases | 46.7 | 30 | 16.7 | 6.6 |
CONCLUSION
Our study confirms that chromosomal abnormalities are significant factors in gonadal dysgenesis resulting in primary amenorrhoea. Furthermore, an appreciable proportion of these individuals with sex chromosome abnormalities is mosaics. Genetic counselling should include the risk of premature menopause for patients with Turner syndrome, use of hormonal replacement therapy, risk of gonadal malignancy in patients with XY gonadal dysgenesis and possibility of infertility in the future children of patients with mosaic Turner syndrome.
Availability of data and materials
All data obtained and analysed during this study are included in this article. Details are available from the corresponding author, on reasonable request.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Marsh CA, Grimstad FW. Primary amenorrhea: Diagnosis and management. Obstet Gynecol Surv. 2014;69:603–12. doi: 10.1097/OGX.0000000000000111. [DOI] [PubMed] [Google Scholar]
- 2.Klein DA, Poth MA. Amenorrhea: An approach to diagnosis and management. Am Fam Physician. 2013;87:781–8. [PubMed] [Google Scholar]
- 3.Rajangam S, Nanjappa L. Cytogenetic studies in amenorrhea. Saudi Med J. 2007;28:187–92. [PubMed] [Google Scholar]
- 4.Rosa RF, Dibi RP, Picetti Jdos S, Rosa RC, Zen PR, Graziadio C, et al. Amenorrhea and X chromosome abnormalities. Rev Bras Ginecol Obstet. 2008;30:511–7. doi: 10.1590/s0100-72032008001000006. [DOI] [PubMed] [Google Scholar]
- 5.Vijayalakshmi J, Koshy T, Kaur H, Mary FA, Selvi R, Parvathi VD, et al. Cytogenetic analysis of patients with primary amenorrhea. Int J Hum Genet. 2010;10:71–6. [Google Scholar]
- 6.Ghosh S, Roy S, Pal P, Dutta A, Halder A. Cytogenetic analysis of patients with primary amenorrhea in Eastern India. J Obstet Gynaecol. 2018;38:270–5. doi: 10.1080/01443615.2017.1353595. [DOI] [PubMed] [Google Scholar]
- 7.Yunis JJ, Sanchez O. G-banding and chromosome structure. Chromosoma. 1973;44:15–23. doi: 10.1007/BF00372570. [DOI] [PubMed] [Google Scholar]
- 8.Harada N. G-banding: Fetal chromosome analysis by using chromosome banding techniques. In: Fetal Morph Functional Diagnosis. Part of the Comprehensive Gynecology and Obstetrics book series: Springer. 2021. pp. 309–18. [Google Scholar]
- 9.Yoon JY, Cheon CK. Evaluation and management of amenorrhea related to congenital sex hormonal disorders. Ann Pediatr Endocrinol Metab. 2019;24:149–57. doi: 10.6065/apem.2019.24.3.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dutta UR, Ponnala R, Pidugu VK, Dalal AB. Chromosomal abnormalities in amenorrhea: A retrospective study and review of 637 patients in South India. Arch Iran Med. 2013;16:267–70. [PubMed] [Google Scholar]
- 11.Korgaonkar S, Dhangar S, Kulkarni V, Kerketta L, Vundinti BR. Clinical and cytogenetic profile of 490 cases of primary amenorrhea. J Med Sci Clin Res. 2018;6:487–94. [Google Scholar]
- 12.Kalavathi V, Chandra N, Nambiar GR, Shanker J, Sugunashankari P, Meena J, et al. Chromosomal abnormalities in 979 cases of amenorrhea: A review. Int J Hum Genet. 2010;10:65–9. [Google Scholar]
- 13.King TF, Conway GS. Swyer syndrome. Curr Opin Endocrinol Diabetes Obes. 2014;21:504–10. doi: 10.1097/MED.0000000000000113. [DOI] [PubMed] [Google Scholar]
- 14.Koppaka NT, Virulkar SK, Chavan DS, Dalvi RC, Gupta N, Mandava S. Specific chromosomal aberrations in primary amenorrhoea: Study on 3776 cases from Indian population. J Obstet Gynaecol India. 2019;69:457–61. doi: 10.1007/s13224-019-01209-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schaffer L, Tommerup N. An International System for Human Cytogenetic Nomenclature 2005. Recommendations of the International Standing Committee on Human Cytogenetic Nomenclature. Karger Medical and Scientific Publishers, Seoul. 2005 [Google Scholar]
- 16.Pal AK, Ambulkar PS, Sontakke BR, Talhar SS, Bokariya P, Gujar VK. A study on chromosomal analysis of patients with primary amenorrhea. J Hum Reprod Sci. 2019;12:29–34. doi: 10.4103/jhrs.JHRS_125_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ali AB, Indriyati R, Winarni TI, Faradz SM. Cytogenetic analysis and clinical phenotype of primary amenorrhea in Indonesian patients. J Biomed Transl Res. 2018;4:22–7. [Google Scholar]
- 18.Melaragno MI, Fakih LM, Cernach MC, Maccagnan P. Isodicentric X chromosome and mosaicism: report on two cases of 45, X/46, X, idic (Xq)/47, X, idic (Xq), idic (Xq) and review of the literature. American journal of medical genetics. 1993;47(3):357–9. doi: 10.1002/ajmg.1320470312. [DOI] [PubMed] [Google Scholar]
- 19.Gorukmez O, Sag SO, Gulten T, Ture M, Yakut T. TURNER SYNDROME WITH 45, X/46, X, I (Xq)/47, X, I (Xq), I (Xq) KARYOTYPE. Genetic Counseling. 2015;26(2):267. [PubMed] [Google Scholar]
- 20.Geckinli BB, Toksoy G, Sayar C, Soylemez MA, Yesil G, Aydın H, et al. Prevalence of X-aneuploidies, X-structural abnormalities and 46, XY sex reversal in Turkish women with primary amenorrhea or premature ovarian insufficiency. Eur J Obstet Gynecol Reprod Biol. 2014;182:211–5. doi: 10.1016/j.ejogrb.2014.09.033. [DOI] [PubMed] [Google Scholar]
- 21.Wong MS, Lam ST. Cytogenetic analysis of patients with primary and secondary amenorrhoea in Hong Kong: Retrospective study. Hong Kong Med J. 2005;11:267–72. [PubMed] [Google Scholar]
- 22.Ten SK, Chin YM, Noor PJ, Hassan K. Cytogenetic studies in women with primary amenorrhea. Singapore Med J. 1990;31:355–9. [PubMed] [Google Scholar]
- 23.Ayed W, Amouri A, Hammami W, Kilani O, Turki Z, Harzallah F, et al. Cytogenetic abnormalities in Tunisian women with premature ovarian failure. C R Biol. 2014;337:691–4. doi: 10.1016/j.crvi.2014.09.003. [DOI] [PubMed] [Google Scholar]
- 24.Butnariu L, Covic M, Ivanov I, Bujoran C, Gramescu M, Gorduza E. Clinical and cytogenetic correlation in primary and secondary amenorrhea: Retrospective study on 531 patients. Rev Rom Med Lab. 2011;19:51–60. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data obtained and analysed during this study are included in this article. Details are available from the corresponding author, on reasonable request.