Abstract
Glucocorticoids have short- and long-term effects on adrenal gland function and development. RNA sequencing (RNA-seq) was performed to identify early transcriptomic responses to the synthetic glucocorticoid, dexamethasone (Dex), in vitro and in vivo. In total, 1711 genes were differentially expressed in the adrenal glands of the 1-h Dex-treated mice. Among them, only 113 were also considered differentially expressed genes (DEGs) in murine adrenocortical Y-1 cells treated with Dex for 1 h. Gene ontology analysis showed that the upregulated DEGs in the adrenal gland of the 1-h Dex-treated mice were highly associated with the development of neuronal cells, suggesting the adrenal medulla had a rapid response to Dex. Interestingly, only 4.3% of Dex-responsive genes in the Y-1 cell line under Dex treatment for 1 h were differentially expressed under Dex treatment for 24 h. The heatmaps revealed that most early responsive DEGs in Y-1 cells during 1 h of treatment exhibited a transient response. The expression of these genes under treatment for 24 h returned to basal levels similar to that during control treatment. In summary, this research compared the rapid transcriptomic effects of Dex stimulation in vivo and in vitro. Notably, adrenocortical Y-1 cells had a transient early response to Dex treatment. Furthermore, the DEGs had a minimal overlap in the 1-h Dex-treated group in vivo and in vitro.
Keywords: glucocorticoids, adrenal gland, transcriptome, dexamethasone, Y-1 cells
Introduction
Glucocorticoids (GCs) are primary stress hormones and critical regulators of several physiological mechanisms. GCs commonly elicit their function through glucocorticoid receptor (GR)-mediated genomic effects. However, these hormones can also stimulate several non-genomic mechanisms via the membrane-bound GR (1). Non-transcriptional effects have short-term outcomes that control several critical physiological processes, including inflammation, behavioral changes, and even negative feedback from the hypothalamic–pituitary–adrenal (HPA) axis (2). Although GCs have non-genomic actions, they may still rapidly affect gene expression via different mechanisms such as mRNA destabilization and coactivator competition (3, 4). Transcriptional responses can occur within 1 h after the initial stimulus (5, 6). Therefore, the rapid response of gene expression is observed immediately after GC administration via both genomic and non-genomic actions.
Synthetic GC dexamethasone (Dex) is widely used for therapeutic and diagnostic purposes in clinical settings due to its potent GR activity and predominant effects. Treatment with Dex suppresses the HPA axis activity leading to decreased steroidogenesis in the adrenal cortex, eventually reducing endogenous GC secretion. The mechanisms of the abovementioned Dex-mediated negative feedback have been widely assessed (7). However, the immediate transcriptomic response (i.e. within approximately 1 h) of the adrenal gland to Dex administration is not completely elucidated at the genome-wide level. In this study, RNA sequencing (RNA-seq) and downstream bioinformatics analysis were performed to explore the early transcriptional response to Dex exposure in vivo and in vitro. This study provided data about the transcriptomic response of Y-1 cell, a widely used murine adrenocortical cell line, to Dex treatment for 1 and 24 h. The rapid response (within 1 h) in vitro vs in vivo was also compared to highlight the difference between Y-1 adrenocortical cells and the C57BL/6 mice.
Materials and methods
Animals and dexamethasone treatment
Male C57BL/6 mice aged 35–60 days were housed in a 14 h light:10 h darkness cycle, were given free access to water and rodent chow, and were treated with Dex as previously described (8). In short, mice were familiarized with general handling, weighing procedures, and intraperitoneal injections for 4 days (once a day with 0.25 mL of sterile 1× PBS). On the fifth day, in the morning, animals were weighed, injected with 0.25 mL of either vehicle (sterile 1× PBS) or 10 μg dexamethasone (1DEX027, Covetrus, Portland, ME, USA), and returned to their cages. Mice were euthanized by CO2 inhalation 1 h after injection, and all samples were collected immediately after sacrification. Adrenal glands were quickly removed from mice, and the surrounding adipose tissue was trimmed away. The right adrenal gland from each mouse was snap-frozen in liquid nitrogen and stored at −80°C until use. All procedures followed the protocols approved by the Institutional Animal Care and Use Committees at Auburn University.
Cell culture
Y-1 mouse adrenocortical cells (ATCC #CCL-79) were purchased from American Type Culture Collection. Y-1 cells were cultured in DMEM/F12 media supplemented with 10% fetal bovine serum and 1% streptomycin/penicillin (E490-20ML, VWR, Radnor, PA, USA). Cells were subcultured separately into four groups (control, 1-h Dex-treated, 24-h Dex-treated, and 48-h Dex-treated) using six-well plates with 400,000 cells per well. After reaching 70% confluency, the growth media was aspirated and replaced by either vehicle- (growth media with 0.05% ethanol) or Dex- (growth media with 0.05% ethanol and 100 nM Dex) containing media. The 100 nM Dex is commonly used in the cell culture system for studying the Dex effect and is considered a high-dose treatment (9, 10, 11, 12). At the end of the treatment, the culture medium was aspirated, immediately followed by RNA extraction. Vehicle-treated control groups were harvested along with the 1-h Dex-treated group and the 24-h Dex-treated group. For the RNA-seq study, the 24-h vehicle-treated samples were used as the control group. Three experimental replicates were performed independently on different days starting from different cell passages. For the qPCR study, the control group was the 1-h vehicle-treated samples.
RNA isolation and sequencing
Total RNA from cell cultures was extracted using 400 µL TRIzol (Invitrogen). Total RNA from tissues was extracted from frozen adrenal glands using Monarch Total RNA Miniprep Kit (New England Biolabs, Ipswich, MA, USA) according to the manufacturer’s instructions. Adrenal glands from two to three animals were pooled as one biological replicate, with two biological replicates per condition (a total of five mice were included in each condition). Total RNA was sent to Novogene Life Sciences Co., Ltd., for the PE150 RNA-seq service. All samples that passed the quality control (concentration > 25 ng/µL, RIN ≥ 7.5) were used for library preparation and subsequent RNA-seq.
Bioinformatics data analysis
Unstranded paired-end read files were first analyzed using the FASTQ error check feature of Chipster (13). Quality control was then run on the raw read files using FastQC. The trimmed reads were then aligned to the mouse genome (mm10, GRCm38.95) using STAR (v. 2.5) (14). Reads were mapped to each gene and counted using HTSeq (v.0.6.1) (15). The maximal mappable prefix (MMP) method ensures precise mapping results for exon–exon junction reads. Differential expression analysis between control and Dex-treated groups was performed using the DESeq2 (16) in R version 3.6.2 with FDR cutoff α = 0.05. Differential expression analysis between the 1-h and 24-h Dex treatment groups was also performed using the same parameter (Supplementary Fig. 1, see section on supplementary materials given at the end of this article). Three replicates per group were included in the in vitro study and two replicates per group were used for the in vivo study. Statistical significance of differential expression was assigned to genes with adjusted P-values < 0.05, irrespective of fold change. Genes must have at least one fragment per kilobase per million mapped reads (FPKM) in at least one sample to be considered differentially expressed genes (DEGs). Heatmaps for DEGs were generated using heatmap.2 in R, with Ward’s minimum variance method for clustering (ward.D2) and Euclidean distance measurement method. The Panther Gene Ontology website (geneontology.org) was used for GO analysis. GO-Figure! was used to generate Fig. 2D. GO enrichment analysis in Fig. 3 was analyzed using g:Profiler and the enrichGO function in the clusterProfiler package (3.16.0) in R with P < 0.05 as a cutoff value (17, 18).
Figure 2.
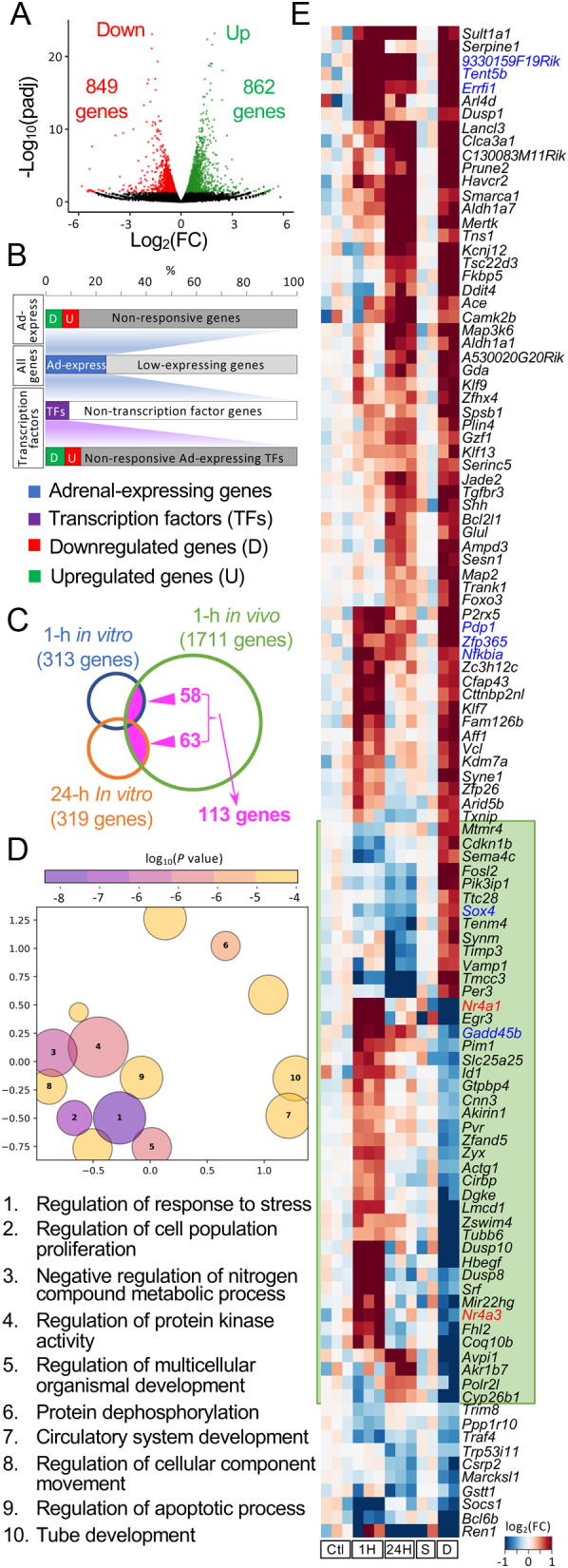
Transcriptomic response of adult mouse adrenal gland to 1-h dexamethasone treatment. (A) The volcano plot showed the DEGs in the whole adrenal gland transcriptome in male mice under a 1-h Dex treatment. (B) Around 26.2% of genes were identified as adrenal-expressed genes. 12.0% of adrenal-expressed genes were differentially expressed under a 1-h Dex treatment. Transcription factors (TFs) had a similar response rate compared to non-TF genes. (C) Minimal overlap of DEGs among three groups. (D) GO analysis of the 58 overlapping DEGs in the 1-h treated in vivo and in vitro studies. GO terms that are most similar in semantic spaces (indicated on the X and Y axis) are placed nearest to each other. The ten most significant terms are labeled with numbers and their descriptions are provided below the plot. (E) The clustered heatmap of the 113 genes that were differentially expressed both in vivo and in vitro. Genes in the green box had opposing responses to Dex treatment in vivo vs in vitro. DEGs identified in all three conditions are highlighted in blue. The fold change used for heatmaps was calculated based on FPKM +0.001. Ctl, untreated control (in vitro); 1H, 1-h Dex-treated group (in vitro); 24H, 24-h Dex-treated group (in vitro); S, saline-treated group (in vivo); D, Dex-treated group (in vivo).
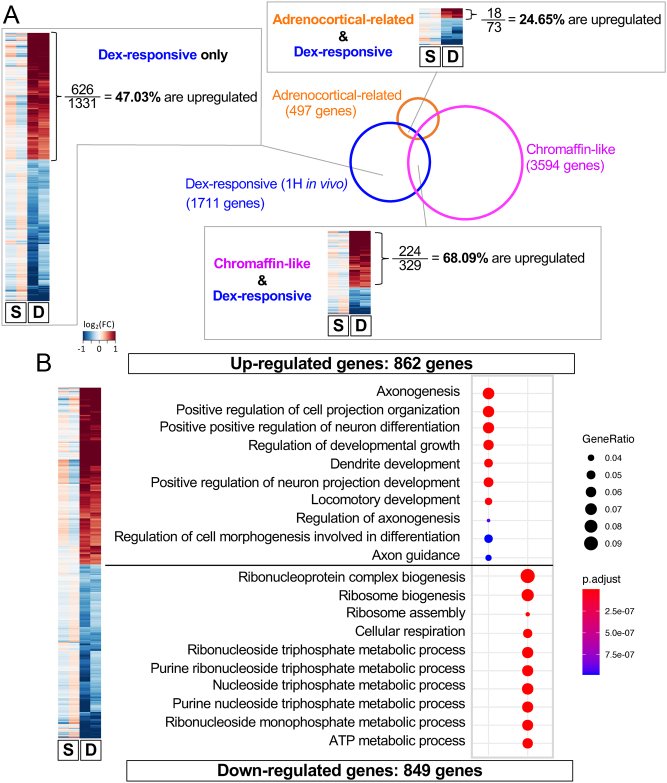
Figure 3.
In the in vivo 1-h dexamethasone treatment, the upregulated genes and the downregulated genes were linked to different functions and different groups of cells. (A) Venn diagram comparing Dex-responsive genes with ‘adrenocortical-related genes’ and ‘chromaffin-like’ genes. Clustered heatmaps showed how genes were differentially expressed in each overlapping gene set. In Dex-responsive genes that overlap with the ‘chromaffin-like’ gene set, 68.09% were upregulated (significantly higher than the ‘Dex-response only’ group, z = 6.84, P < 0.0001). In genes that were both ‘Dex-responsive’ and ‘adrenocortical-related’, only 24.65% were upregulated (significantly lower than the ‘Dex-response only’ group, z = −3.74, P < 0.0002). (B) GO analysis showed that all upregulated genes in the in vivo 1-h Dex treatment group were highly associated with neural cell development and function. The fold change used for heatmaps was calculated based on FPKM +0.001. S, saline-treated group; D, Dex-treated group.
Quantitative RT-PCR
The relative expression of some key DEGs was verified using quantitative RT-PCR (qPCR). Total RNA was used to generate cDNA using SuperScript IV transcriptase and dT primers. qPCR was performed using the off-the-shelf TaqMan Gene Expression Assays (Supplementary Table 1). Gapdh and Rplp0 were used as the internal control. The relative gene expressions were calculated using the relative standard curve method. Each data point was an average of three independent cell culture experiments with three technical replicates in each experiment.
Statistical analysis
Student’s t-test was used to compare the expressions of DEGs between the control and Dex-treated groups in Y-1 cells (Fig. 5).
Results
Differentially expressed genes had a minimal overlap in the 1-h and 24-h Dex-treated Y-1 cells
RNA-seq analysis identified 313 and 319 DEGs in Y-1 cells in the 1-h and 24-h Dex-treated groups, respectively (Fig. 1). There were 203 upregulated genes and 110 downregulated genes in the 1-h Dex-treated group. Meanwhile, 200 and 119 genes were upregulated and downregulated, respectively, in the 24-h Dex-treated group (Supplementary Table 2). The clustered heatmap showed that these DEGs belong to four major groups, which were as follows: (a) upregulated mainly in the 1-h Dex-treated group, (b) upregulated mainly in the 24-h Dex-treated group, (c) downregulated mainly in the 24-h Dex-treated group, and (d) downregulated mainly in the 1-h Dex-treated group (Fig. 1A). Interestingly, most DEGs in the 1-h Dex-treated group were not considered DEGs in the 24-h Dex-treated group. The expressions of these genes returned to basal levels similar to those of the non-treated control group. Among 606 Dex-responsive genes found in Y-1 cells, only 26 (4.3%) were differentially expressed in the 1-h and 24-h Dex-treated groups, which is equivalent to 8.3 and 8.2% of DEGs in each group, respectively (Fig. 1B). Interestingly, the heatmap showed that almost all of the 26 genes had a similar response to Dex treatment for 1 and 24 h. Sdf2l1 and Plk2 might have an opposite direction of response on the heatmap. However, their expression did not significantly differ, as revealed on qPCR. Sdf2l1 was differentially expressed on qPCR only in the 24-h Dex-treated group but not in the 1-h Dex-treated group (P = 0.003 vs P = 0.94). In contrast, Plk2 remained stable in the 1-h and 24-h Dex-treated groups (P = 0.57 vs P = 0.42). Notably, Y-1 cells had low Plk2 expression levels (FPKM of <20 in all samples, Supplementary Table 2).
Figure 1.
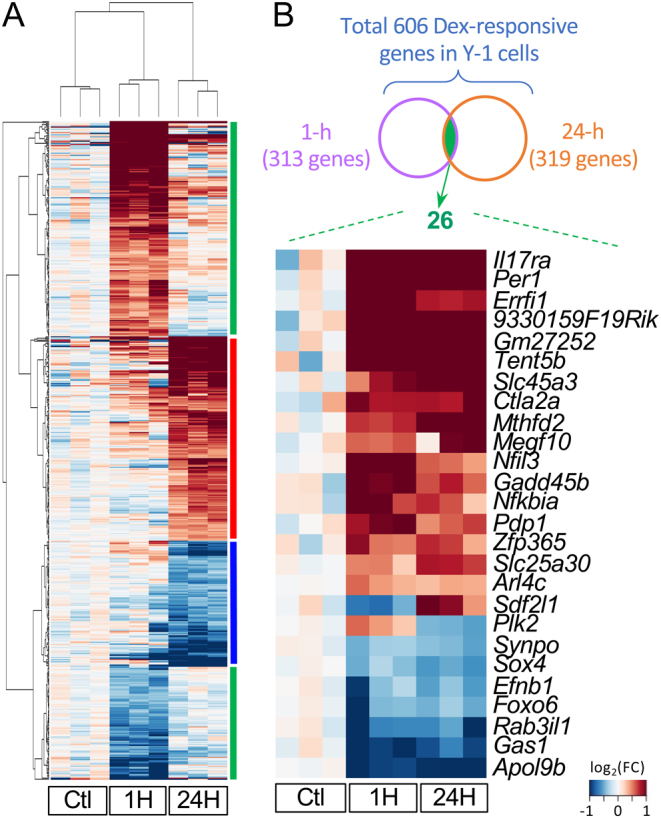
Y-1 cells responded differently to the 1-h and the 24-h dexamethasone treatment. (A) The clustered heatmap showed the fold change of all DEGs identified either in the 1-h or the 24-hour Dex treatment in Y-1 cells. Genes were clustered into four major groups. Genes in two clusters (marked by the red bar and the blue bar on the right) were either upregulated or downregulated mainly in the 24-h group with no or minor changes in the 1-h group. Genes in another two clusters (marked by green bars) had a transient response to Dex. In these two clusters, expressions of genes in the 24-h group moved back toward the non-treated control group. (B) There were 313 and 319 genes differentially expressed in the 1-h treatment group and the 24-h treatment group, respectively. A total of 26 genes were differentially expressed in both groups. Sdf2l1 and Plk2 are the only two genes that showed an opposite change between the 1-h and 24-h treatment groups. Ctl, vehicle control. Genes were excluded if the average FPKM of each group was less than 1 across all three groups. The fold change used for heatmaps was calculated based on FPKM +0.001.
Genes responsive to Dex treatment in vivo and in vitro
To compare the early response to Dex treatment in vitro vs in vivo, adult C57BL/6 mice were also treated with high-dose Dex, which can have a sufficient effect on the HPA axis at the pituitary level within 1 h (8). Approximately 26.2% of 54,533 genes in the mouse genome had at least 1 FPKM in 1 sample. These genes were classified as adrenal-expressed genes (14,300 genes). The number of adrenal-expressed genes in the saline-treated group was similar to that in another genome-wide study of adult C57BL/6 mice (19). The in vivo 1-h Dex-treated group had 862 upregulated genes and 849 downregulated genes (Fig. 2A, Supplementary Table 3). These 1711 Dex early responsive genes comprise 12.0% of all adrenal-expressed genes (Fig. 2B). Approximately 13.3% of adrenal-expressed transcription factors (TFs) were differentially expressed in the 1-h Dex-treated group. The percentage of Dex early responsive adrenal-expressed genes and Dex-responsive adrenal-expressed TFs was similar. Hence, the response rate to Dex treatment was similar between the TF and non-TF genes (Fig. 2B). Interestingly, only a small percentage of DEGs in vitro was also identified (Fig. 2C): 58 DEGs in the 1-h Dex-treated Y-1 cells were differentially expressed in the in vivo study; meanwhile, 63 genes in the 24-h in vitro Dex-treated group were also identified in the in vivo treatment group. The Venn diagram showed that only a small portion of DEGs overlapped among three groups (58 + 63 − 113 = 8 genes). Gene ontology (GO) analysis showed that 58 overlapping DEGs between the 1-h in vitro and in vivo groups were highly associated with the ‘regulation of response to stress’ (GO: 0080135, fold enrichment of >5.00, FDR P = 0.0003) (Fig. 2D).
Dynamic expression of differentially expressed genes in vivo and in vitro
A clustered heatmap was used to better visualize the response of 113 genes to Dex in vivo and in vitro (Fig. 2E). Interestingly, genes that exhibited an opposite direction of response during in vivo and in vitro treatments were clustered together (highlighted in the green box in Fig. 2E, containing 44 genes). For example, the expressions of nuclear receptor genes Nr4a1 and Nr4a3 and the zona fasciculata marker gene Akr1b7 were upregulated either in the 1- or 24-h Dex-treated Y-1 cells. However, the expressions were downregulated in vivo. ‘cellular response to corticotropin-releasing hormone stimulus’ (GO: 0071376, fold enrichment of >100, FDR P = 0.047) was the leading GO term obtained from the 44 clustered genes. The other GO terms included ‘cell migration involved in sprouting angiogenesis’ (GO:0002042, fold enrichment of 84.7, FDR P = 0.020), ‘blood vessel endothelial cell migration’ (GO:0043534, fold enrichment of 53.6, FDR P = 0.035), and ‘tissue development’ (GO:0009888, fold enrichment of 4.47, FDR P = 0.009).
Opposite response of genes associated with steroidogenesis and catecholamine synthesis
We then compared 1711 Dex-early responsive genes identified in the in vivo treatment, chromaffin-like genes, (20) and adrenocortical-related genes (21). In the Dex-treated adrenal glands of mice, 24.65% of the adrenocortical-related Dex-responsive genes and 68.09% of the chromaffin-like Dex-responsive genes were upregulated (Fig. 3A, Supplementary Table 2). This difference was in accordance with the current understanding that the effect of GCs in the adrenal cortex could be opposite to that in the adrenal medulla. GCs can both suppress adrenocortical steroidogenesis directly or indirectly (22) and can induce medullary function by upregulating the expression of phenylethanolamine N-methyltransferase (PNMT) (23, 24), an epinephrine synthesis enzyme. To further understand the functional meaning of this dynamic response at the transcriptome level, GO analysis was performed to evaluate the potential function of upregulated and downregulated genes. GO analysis revealed that several GO terms associated with the 862 Dex-upregulated genes were associated with the development or function of neuronal cells (Fig. 3B). To provide better visualization of the response of adrenal glands to Dex, the genes were classified based on their function, and clustered heatmaps were used to show the relative expression of genes both in the in vivo and in vitro treatment groups. Interestingly, results revealed that genes associated with catecholamine synthesis may be upregulated in general in the in vivo group (Fig. 4), which was in accordance with the result of GO analysis. Although none of these catecholamine synthesis-associated genes were considered DEG, several genes associated with the regulation of neurotransmitter secretion (GO:0046928, fold enrichment of 4.0, FDR P = 0.000407) were included in the Dex-upregulated genes. Regarding steroid hormone production, several genes associated with steroidogenesis were downregulated primarily in the in vivo study (Fig. 4). No specific trend was observed in genes associated with nuclear receptors or cholesterol metabolism. Most genes in these two categories did not change or were minimally affected in the in vitro treatment groups.
Figure 4.
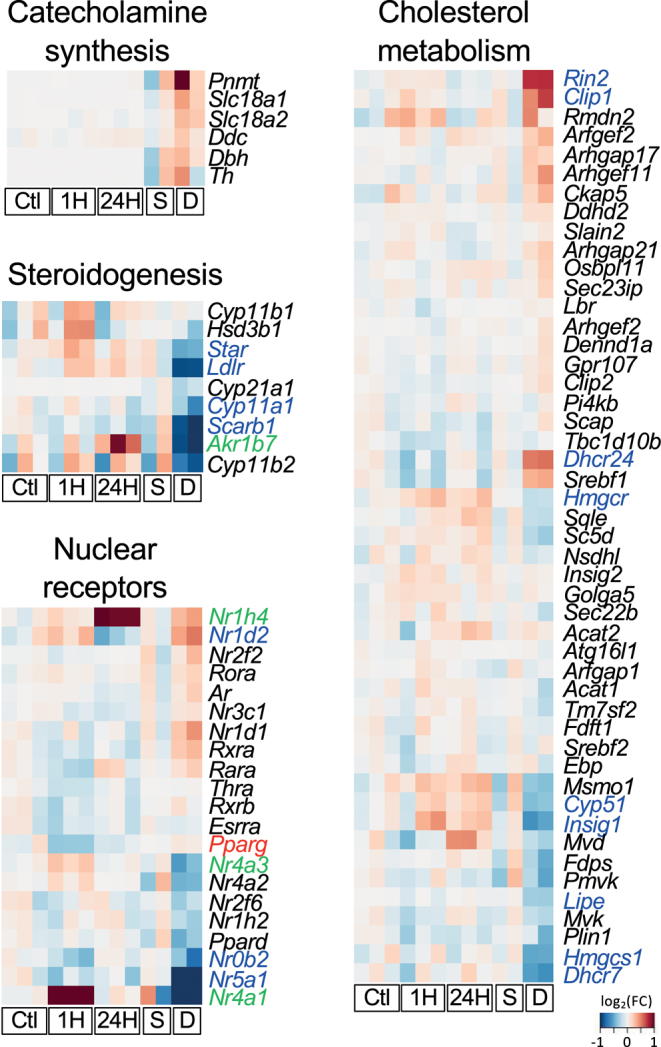
Heatmaps of the genes related to adrenal gland functions, cholesterol metabolism, and nuclear receptors in the adrenal gland. Heatmaps showed the fold change of Dex-treated groups vs control groups. The fold change was calculated based on FPKM +10 to reduce the influence of low expressing genes. Genes must have an original FPKM > 10 in at least one sample to be included in these heatmaps. Gene names in blue, red, or green were identified as DEGs in vivo, in vitro, or both groups, respectively. Ctl, vehicle control (in vitro); 1H, 1-h Dex-treated group (in vitro); 24H, 24-h Dex-treated group (in vitro); S, saline-treated group (in vivo); D, Dex-treated group (in vivo).
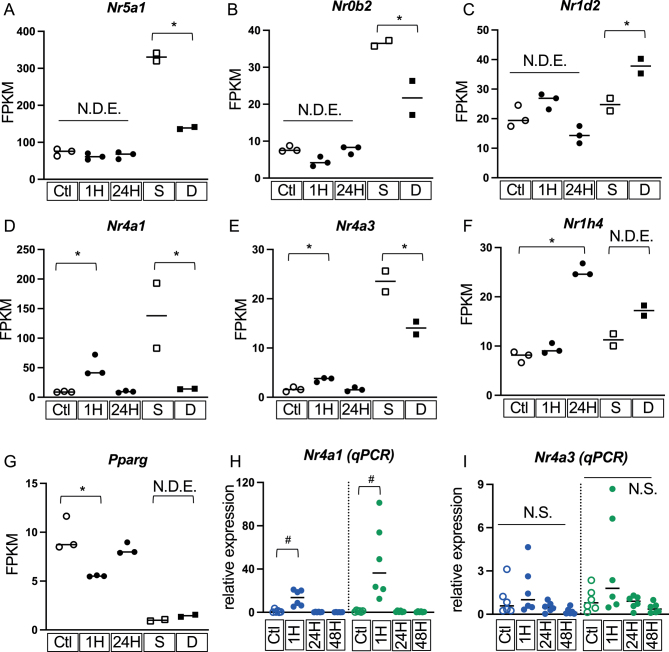
Dynamic response of nuclear receptors to Dex treatment in vivo and in vitro
It is interesting that the downregulation of genes was associated with steroidogenesis particularly in the in vivo treatment groups. This notion was in accordance with the long-term understanding that Dex can suppress the endogenous secretion of GCs via the negative feedback loop. In the in vivo treatment groups, Nr5a1, which is a nuclear receptor known as the master regulator of steroidogenesis, is downregulated (Fig. 5A). The expression of several genes known as the Nr5a1-induced genes (e.g. Fdx1, Alas1, and Akr1b7) was also downregulated (Table 1) (25, 26, 27). Moreover, the downregulation of Nr5a1 may explain the overall suppression of steroidogenesis-related genes (Fig. 4). Nr0b2, Nr1d2, Nr4a1, Nr4a3, Nr1h4, and Pparg, in addition to Nr5a1, were the nuclear receptors considered DEGs (Fig. 5B, C, D, E, F and G). Notably, Nr4a1 and Nr4a3 were the two TFs that exhibited a transient upregulation in Y-1 cells during Dex treatment for 1 h (Fig. 5D and E). qPCR confirmed the transient differential expression of Nr4a1 (Fig. 5H). Although some qPCR data points showed a transient upregulation of Nr4a3 during Dex treatment for 1 h, the results did not significantly differ (Fig. 5I). The difference in Nr4a3 expression might be attributed to the extremely low Nr4a3 expression in Y-1 cells (FPKM of <4).
Figure 5.
Expression of differentially expressed nuclear receptors. Dot plots showed (A, B, C, D, E, F, G) FPKM levels with means. (H and I) qPCR was used to verify the transient differential expression of Nr4a1 and Nr4a3. Data in blue used Gapdh as the internal control gene. Data in green used Rplp0 as the internal control gene. The relative expression of the untreated control was set as one. *Identified as DEGs using DESeq2 in the RNA-seq; #Identified as genes with a significant difference with the control group (t-test, P < 0.05); N.S., no significant difference (t-test); N.D.E., not differently expressed using DESeq2 in the RNA-seq. Ctl, vehicle control (in vitro); 1H, 1-h Dex-treated group (in vitro); 24H, 24-h Dex-treated group (in vitro); 48H, 48-h Dex-treated group (in vitro); S, saline-treated group (in vivo); D, Dex-treated group (in vivo).
Table 1.
Top differentially expressed genes in adrenal glands in Dex-treated male mice.
| Top upregulated genes | Top downregulated genes | ||||||
|---|---|---|---|---|---|---|---|
| Gene namea | Average FPKM | Average FPKM | Fold change | ||||
| Saline | Dex | Fold change | Gene nameb | Saline | Dex | ||
| Abca1 | 10.2 | 42.8 | 4.20 | Nr4a1 | 138.0 | 14.0 | 0.10 |
| Pdk4 | 3.0 | 11.7 | 3.83 | 2010003K11Rik | 16.3 | 3.2 | 0.20 |
| Zbtb16 | 6.6 | 25.4 | 3.83 | Gm13889 | 18.9 | 4.2 | 0.22 |
| Chrna7 | 3.8 | 14.2 | 3.79 | 2010109I03Rik | 14.7 | 4.1 | 0.28 |
| Ddit4 | 28.6 | 107.9 | 3.77 | Tnfrsf12a | 225.4 | 70.5 | 0.31 |
| Fam107a | 5.3 | 19.0 | 3.60 | Mob4 | 78.9 | 25.9 | 0.33 |
| Nt5e | 4.1 | 13.8 | 3.40 | Apoc4 | 11.6 | 3.9 | 0.34 |
| Fzd4 | 3.6 | 11.6 | 3.25 | Coq10b | 63.5 | 21.6 | 0.34 |
| Greb1 | 20.3 | 63.4 | 3.12 | Cryba4 | 24.9 | 9.0 | 0.36 |
| Lrg1 | 9.2 | 28.3 | 3.07 | Yam1 | 6933.1 | 2565.2 | 0.37 |
| Lnpep | 8.2 | 24.2 | 2.94 | Mapkapk3 | 13.9 | 5.3 | 0.38 |
| Maf | 4.6 | 13.3 | 2.87 | Fdx1 | 2978.8 | 1129.6 | 0.38 |
| Tsc22d3 | 28.3 | 80.4 | 2.84 | Avpi1 | 60.7 | 23.1 | 0.38 |
| Tbx20 | 4.8 | 13.6 | 2.83 | Junb | 44.5 | 17.0 | 0.38 |
| Rin2 | 6.6 | 18.2 | 2.74 | Ldlr | 31.9 | 12.3 | 0.38 |
| Vcam1 | 9.3 | 25.4 | 2.72 | Hba-a2 | 17.0 | 6.7 | 0.40 |
| AW551984 | 3.9 | 10.4 | 2.70 | Tubb6 | 121.8 | 49.0 | 0.40 |
| Dock10 | 4.1 | 11.0 | 2.68 | Ptp4a1 | 42.6 | 17.6 | 0.41 |
| Klf15 | 9.5 | 25.5 | 2.67 | Smim3 | 31.0 | 12.8 | 0.41 |
| Rab3c | 8.7 | 23.1 | 2.65 | Atf4 | 146.0 | 60.9 | 0.42 |
| Pde10a | 6.0 | 15.8 | 2.65 | Nr5a1 | 330.6 | 138.7 | 0.42 |
| Arl4d | 5.6 | 14.5 | 2.60 | Scx | 32.3 | 13.6 | 0.42 |
| Errfi1 | 14.2 | 36.6 | 2.58 | Dusp10 | 10.0 | 4.2 | 0.42 |
| Hmgcs2 | 4.8 | 12.3 | 2.54 | Hes6 | 174.1 | 75.7 | 0.43 |
| Atp7a | 5.4 | 13.7 | 2.54 | Srrd | 11.4 | 5.0 | 0.44 |
| Fkbp5 | 47.6 | 121.0 | 2.54 | Etnk2 | 22.9 | 10.1 | 0.44 |
| Bmp4 | 5.4 | 13.7 | 2.53 | Gm10275 | 16.6 | 7.5 | 0.45 |
| Ston1 | 8.1 | 20.5 | 2.52 | Alas1 | 423.9 | 191.0 | 0.45 |
| Ptprn2 | 5.4 | 13.6 | 2.52 | Tmem120a | 29.4 | 13.6 | 0.46 |
| Irs2 | 4.3 | 10.8 | 2.49 | Adamts1 | 12.6 | 5.9 | 0.46 |
| Bdkrb2 | 4.5 | 11.2 | 2.47 | Creb3l1 | 19.1 | 9.1 | 0.47 |
| Klhl24 | 6.0 | 14.9 | 2.47 | Zswim4 | 16.5 | 7.9 | 0.48 |
| Pdp1 | 11.9 | 29.3 | 2.47 | Dusp14 | 12.4 | 5.9 | 0.48 |
| Tgfbr3 | 38.4 | 93.2 | 2.43 | Fam222a | 34.9 | 16.7 | 0.48 |
| Slc9a7 | 5.0 | 12.0 | 2.41 | Akr1b7 | 1279.3 | 611.1 | 0.48 |
| Rims3 | 5.1 | 12.3 | 2.40 | Cldn5 | 11.8 | 5.7 | 0.48 |
| Osbpl3 | 19.7 | 47.1 | 2.39 | Naa38 | 20.3 | 9.8 | 0.48 |
| Cebpd | 16.7 | 39.6 | 2.36 | 2310009B15Rik | 16.1 | 7.8 | 0.48 |
| Arrdc2 | 4.4 | 10.1 | 2.32 | Agtr1b | 13.5 | 6.6 | 0.49 |
| Adcyap1r1 | 12.2 | 28.3 | 2.31 | RP24-139E15.1 | 16.4 | 8.1 | 0.49 |
| Klhl11 | 5.5 | 12.7 | 2.31 | Lsm5 | 14.5 | 7.2 | 0.49 |
| Ncoa7 | 5.8 | 13.4 | 2.30 | Esm1 | 18.9 | 9.3 | 0.50 |
| Mcf2l | 4.8 | 10.9 | 2.30 | Mir22hg | 15.2 | 7.5 | 0.50 |
| Map3k6 | 8.7 | 19.9 | 2.29 | Hbb-bt | 19.5 | 9.7 | 0.50 |
| Lars2 | 270.7 | 134.8 | 0.50 | ||||
aOnly list the genes that have FPKM > 10 in the Dex-treated group; bOnly list the genes that have FPKM > 10 in the saline-treated group.
Discussion
Rapid transcriptional effects of GCs
A typical action of GCs is that they bind with cytosolic steroid receptors, which in turn migrate to the nucleus and regulate the expression of target genes. For example, GCs can activate or suppress gene expression via GR or mineralocorticoid receptor that binds with the glucocorticoid-responsive elements. GCs could also tether/interact with other TFs to regulate gene expression (28). Although the genetic effect of steroid hormones is not commonly short-term, several data have shown that steroid hormone exposure causes transcriptional changes more rapidly than previously believed. Our in vitro experiments revealed that Y-1 cells had a dynamic early response to Dex treatment. The DEGs identified in the 1-h Dex-treated group are extremely different from those identified in the 24-h Dex-treated group.
The rapid cytoplasmic movement of steroid receptors toward the cell nucleus occurs within minutes after steroid hormone administration (29). In vitro studies have shown that Dex-induced transcriptional changes can be a direct effect independent from other confounding external stimuli present in organismal model systems. For example, a 40-min exposure to estrogen in human umbilical vein endothelial cell culture leads to an acute PI3K-mediated upregulation of 250 genes (30). GR ChIP-seq studies have shown a significant enrichment in GR-binding sites after 1 h of Dex treatment in cells originating from different types of tissues (31, 32). RNA-seq analysis using rat primary cardiomyocytes revealed 98 differentially regulated genes after 1 h of Dex stimulation. This indicated the upregulation of several genes involved in the development of hypertrophic cardiomyopathy, which is a common adverse effect of Dex (33). The same study revealed that the treatment with Dex in 10-week-old mice could mimic findings found in primary cardiomyocytes. The abovementioned in vivo and in vitro studies provided important evidence showing that Dex treatment causes short-term expression changes at a genome-wide level.
Rapid and transient responses of the Nr4a subfamily
In Y-1 cells, Dex treatment leads to a rapid and transient upregulation of Nr4a1 and Nr4a3, which are orphan nuclear receptors belonging to the Nr4a subfamily (contains Nr4a1, Nr4a2, and Nr4a3). Although the expression of Nr4a1 and Nr4a3 in Dex-treated Y-1 cells increased in the 1-h Dex-treated group, the expression was low in the 24- and 48-h Dex-treated groups. This rapid and transient upregulation of the Nr4a subfamily has been observed in several different types of cells that are stimulated. The expression of Nr4a1 and Nr4a3 in 3T3-L1 preadipocytes increases only within the first 4 h during the cell differentiation process from preadipocyte to adipocyte (34). The upregulation of Nr4a1 and Nr4a2 in T cells reaches the peak level within the first hour after antigen stimulation (35). In addition, the expression of Nr4a1 and Nr4a3 increases within the first hour and then decreases in macrophages if coincubated with apoptotic thymocytes (36). Interestingly, Nr4a1 and Nr4a3 are enriched in the GO term ‘cellular response to corticotropin-releasing hormone stimulus’ (GO: 0071376), which is the leading biological process GO term identified using 44 genes that were classified because of the opposite response in vitro and in vivo (Fig. 2E). Most of the 44 genes had a transient response in the in vitro treatment groups. Because our study does not specifically focus on the GR-dependent and/or GR-independent pathways, the data cannot confirm the possible crosstalk between GR and the Nr4a subfamily (37). However, a previous GR ChIP-seq study did not detect GR bindings around the promotor region of Nr4a1 in a Dex-treated human cell line (38).
Effects of Dex on steroidogenesis
GC suppresses the HPA axis activity partly by reducing the secretion of steroid hormones from the adrenal cortex. This GC-mediated suppression is commonly associated with an hour-to-day timescale. Although the short-term non-transcriptional and long-term transcriptional effects of GCs on steroid hormone secretion are known, most studies focused on time points several hours after establishing baseline transcription levels at time 0. For example, efforts for classifying GC-associated mechanisms in Leydig MA10 cells showed suppressed cAMP-mediated Star transcription via NR4A1 after 4 h of Dex incubation (10). In bovine adrenocortical primary cells, 48 h of Dex treatment resulted in a 50% suppression of the adrenocorticotropic hormone (ACTH) or forskolin-induced CYP11A and CYP17 expressions (9). Another study revealed that 24-h incubation with a physiological range of Dex inhibits steroidogenic gene expression including STAR, CYP11A1, and HSD3B in the human glial cell line via GR (11). All these in vitro studies have shown that the GC-mediated suppression of steroidogenesis could be direct and independent of the HPA axis. At least, immunostaining results showed that GR is expressed in all cortical zones and the adrenal medulla as well (39, 40). Although GC was found to have an inhibitor effect on steroidogenesis, seemingly contradictory results have also been reported. For example, in the human adrenocortical (H295A) cell line, 24-h Dex treatment did not affect the transcription of CYP11A1, CYP17, and HSD3B (41).
In the 1-h in vivo treatment groups, the expression of several genes involved in steroidogenesis was downregulated, thereby indicating the suppression of steroidogenesis in mouse adrenocortical cells at the transcriptional level 1 h after Dex treatment. Because the expression of most downregulated genes identified in our in vivo treatment groups did not change in the in vitro treatment groups (except Akr1b7, which was upregulated after 24 h of treatment), a systemic effect from the HPA axis may influence the adrenal gland at the transcriptional level. Our data did show that 1 h of Dex treatment significantly increased the expression of pituitary Rasd1, a gene associated with mechanisms mediating the early response of GCs to negative feedback (8). Although a systemic effect from HPA axis cannot be ruled out, a direct effect is more probable in our 1-h in vivo treatment study. Notably, carbon dioxide inhalation was used for sacrification in our study. Sacrification via CO2 inhalation is significant stress and can lead to high plasma ACTH and corticosterone levels in rodents (42). A possible influence from acute stress may still exist even though high-dose Dex treatment should significantly suppress the HPA axis activity via the negative feedback.
Effects of Dex on neuronal cells
GCs are critical regulators of neuronal cell development and function (43). They upregulate the expression of PNMT in the adrenal medulla, which converts norepinephrine to epinephrine. The GC-induced PNMT expression has been evaluated using several models including in hypophysectomized rats. Results revealed that a low PNMT expression can be restored by ACTH or supraphysiological doses of external GC (23, 44, 45). The rich vasculature within the adrenal gland allows the rapid delivery of GCs to cortical layers and the medulla (46). Endogenous GCs are supplied to the adrenal medulla via the corticomedullary portal system. This phenomenon leads to extremely high GC concentrations in the adrenal medulla and induction of PNMT expression. The lack of PNMT in Cyp11a1 knockout embryos further confirms that a high concentration of intra-adrenal GC is required to stimulate adrenal PNMT (47). In the current study, although the heatmaps showed that the expression of several genes associated with catecholamine synthesis might be elevated in the Dex-treated groups, none of them were DEGs. One possibility is that the Dex treatment suppresses endogenous GC secretion. Thus, the effect of Dex on the medulla, at least at the acute regulation of catecholamine synthesis, is attenuated due to the reduced quantity of endogenous GC entering the adrenal medulla. This also explains why the adrenal PNMT expression increases as early as 20 min after Dex treatment in hypophysectomized rats. However, there was no change in non-hypophysectomized rats (48). Although PNMT did not significantly increase based on our data, GO analysis did show that the upregulated genes were associated with the function and development of neuronal cells. An RNA-seq study showed that the proliferation of cells is the leading functional network in hypothalamic neural-progenitor/stem cells treated with Dex (49), thereby indicating the direct effect of Dex on neuronal cell development.
Difference between adrenal cortex and adrenocortical Y-1 cells
Notably, the Y-1 cells are adrenocortical tumor cells; therefore, the characteristics (i.e. pathways and mechanisms) of these cells significantly differ from those of normal adrenocortical cells. For example, Y-1 cells do not express 21-hydroxylase (Cyp21a1) and, thus, do not secrete corticosterone (50). Moreover, Y-1 cells had a significantly low expression of the lipid transport protein apolipoprotein E (encoded by Apoe), a key modulator of cholesterol homeostasis and corticosterone secretion in the adrenal gland (FPKM of approximately 5 in Y-1 cells vs FPKM of >500 in the whole adrenal gland RNA-seq) (51). In the current study, only 58 genes were differentially expressed during 1 h of Dex treatment both in vivo and in vitro. Notably, most Dex-responsive genes identified in the in vitro treatment were not observed in the in vivo treatment, and vice versa. It is understandable that data on Y-1 cell line, which is an adrenocortical tumor cell line of a LAF1 (C57L × A/HeJ) male mouse, differ in an in vivo study using the whole adrenal glands of B6 mice. The difference could also be attributed to complex physiological responses, including the HPA axis negative feedback, in living animals. Although we did not use a cell type-specific approach for our in vivo study, the whole adrenal gland RNA-seq still provides valuable resource data emphasizing the difference in the Dex-mediated effect between Y-1 cells and the in vivo mouse model at the transcriptome level.
Sexually dimorphic adrenal gland and effects of Dex
The histology and transcriptome of the adrenal gland are sexually dimorphic in mice (19, 21, 52, 53). Moreover, Dex elicits its effect in a sex-specific manner in several aspects via complex mechanisms that are still not completely understood (54, 55, 56, 57). Several immortalized adrenocortical cell lines from transgenic mice, which include a cell line generated using female mice (58), are developed. However, the Y-1 cell line is most widely used. Because the Y-1 cell line was collected from a male mouse, only male mice were included in our study. With consideration of GR-mediated sexual dimorphism in several tissues (59, 60, 61), the Dex-mediated short-term effect in the adrenal gland and adrenocortical cells in female mice could differ from that in male mice. Further studies need to be conducted to assess the short-term effects mediated by Dex in the female adrenal gland and female-derived adrenocortical cells.
In conclusion, we assessed the early/rapid murine adrenal transcriptome response to Dex treatment in vivo and in vitro. Results showed that 1 h of Dex treatment had acute transcriptional effects in the adrenal gland and Y-1 cells. However, less than 10% of Dex-responsive genes were differentially expressed both in vivo and in vitro. Interestingly, a group of Dex-responsive genes had opposite directions of response in vivo and in vitro, indicating a dynamic early response of the mouse adrenal gland and adrenocortical Y-1 cells to Dex at the transcriptomic level.
Supplementary Material
The supplementary data is also available on figshare:
Supplementary Figure 1: https://doi.org/10.6084/m9.figshare.19799401.v1
Supplementary Table 1: https://doi.org/10.6084/m9.figshare.16828537.v1
Supplementary Table 2: https://doi.org/10.6084/m9.figshare.16828534.v1
Supplementary Table 3: https://doi.org/10.6084/m9.figshare.16828531.v1
Declaration of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Funding
NIH R00HD082686 (C J H), Animal Health and Disease Research Program (C D F), Animal Health and Disease Research Program (S R P).
Data availability
The RNA-seq data have been deposited in NCBI’s Gene Expression Omnibus and are accessible through GEO Series accession numbers GSE189182 and GSE189305 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE189182; https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE189305)
Acknowledgments
This work was supported by NIH grant R00 HD082686 (to C J H), Animal Health and Disease Research Program at Auburn University (to C D F), and Animal Health and Disease Research Program at Auburn University (to S R P). H S Z was supported by the Endocrine Society Summer Research Fellowship. Some equipment used in the study was supported by the Auburn University new faculty start-up funds to C J H.
References
- 1.Stahn C, Buttgereit F. Genomic and nongenomic effects of glucocorticoids. Nature Clinical Practice: Rheumatology 20084525–533. ( 10.1038/ncprheum0898) [DOI] [PubMed] [Google Scholar]
- 2.Panettieri RA, Schaafsma D, Amrani Y, Koziol-White C, Ostrom R, Tliba O. Non-genomic effects of glucocorticoids: an updated view. Trends in Pharmacological Sciences 20194038–49. ( 10.1016/j.tips.2018.11.002) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Treviño LS, Gorelick DA. The interface of nuclear and membrane steroid signaling. Endocrinology 2021162 bqab107. ( 10.1210/endocr/bqab107) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Uchoa ET, Aguilera G, Herman JP, Fiedler JL, Deak T, de Sousa MB. Novel aspects of glucocorticoid actions. Journal of Neuroendocrinology 201426557–572. ( 10.1111/jne.12157) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lopez-Maury L, Marguerat S, Bahler J. Tuning gene expression to changing environments: from rapid responses to evolutionary adaptation. Nature Reviews: Genetics 20089583–593. ( 10.1038/nrg2398) [DOI] [PubMed] [Google Scholar]
- 6.Yosef N, Regev A. Impulse control: temporal dynamics in gene transcription. Cell 2011144886–896. ( 10.1016/j.cell.2011.02.015) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dallman MF, Akana SF, Levin N, Walker CD, Bradbury MJ, Suemaru S, Scribner KS. Corticosteroids and the control of function in the hypothalamo-pituitary-adrenal (HPA) axis. Annals of the New York Academy of Sciences 199474622–31; discussion 31,–22, 64–27. ( 10.1111/j.1749-6632.1994.tb39206.x) [DOI] [PubMed] [Google Scholar]
- 8.Foradori CD, Mackay L, Huang CJ, Kemppainen RJ. Expression of Rasd1 in mouse endocrine pituitary cells and its response to dexamethasone. Stress 202124659–666. ( 10.1080/10253890.2021.1907340) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Trzeciak WH, LeHoux JG, Waterman MR, Simpson ER. Dexamethasone inhibits corticotropin-induced accumulation of CYP11A and CYP17 messenger RNAs in bovine adrenocortical cells. Molecular Endocrinology 19937206–213. ( 10.1210/mend.7.2.8385739) [DOI] [PubMed] [Google Scholar]
- 10.Martin LJ, Tremblay JJ. Glucocorticoids antagonize cAMP-induced star transcription in Leydig cells through the orphan nuclear receptor NR4A1. Journal of Molecular Endocrinology 200841165–175. ( 10.1677/JME-07-0145) [DOI] [PubMed] [Google Scholar]
- 11.Koibuchi F, Ritoh N, Aoyagi R, Funakoshi-Tago M, Tamura H. Dexamethasone suppresses neurosteroid biosynthesis via downregulation of steroidogenic enzyme gene expression in human glioma GI-1 cells. Biological and Pharmaceutical Bulletin 2014371241–1247. ( 10.1248/bpb.b14-00014) [DOI] [PubMed] [Google Scholar]
- 12.Páez-Pereda M, Kovalovsky D, Hopfner U, Theodoropoulou M, Pagotto U, Uhl E, Losa M, Stalla J, Grübler Y, Missale C, et al. Retinoic acid prevents experimental Cushing syndrome. Journal of Clinical Investigation 20011081123–1131. ( 10.1172/JCI11098) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kallio MA, Tuimala JT, Hupponen T, Klemela P, Gentile M, Scheinin I, Koski M, Kaki J, Korpelainen EI. Chipster: user-friendly analysis software for microarray and other high-throughput data. BMC Genomics 201112 507. ( 10.1186/1471-2164-12-507) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dobin A, Davis CA, Schlesinger F, Drenkow J, Zaleski C, Jha S, Batut P, Chaisson M, Gingeras TR. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 20132915–21. ( 10.1093/bioinformatics/bts635) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Anders S, Pyl PT, Huber W. HTSeq--a Python framework to work with high-throughput sequencing data. Bioinformatics 201531166–169. ( 10.1093/bioinformatics/btu638) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biology 201415 550. ( 10.1186/s13059-014-0550-8) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Raudvere U, Kolberg L, Kuzmin I, Arak T, Adler P, Peterson H, Vilo J. g:profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Research 201947 W191–W198. ( 10.1093/nar/gkz369) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics 201216284–287. ( 10.1089/omi.2011.0118) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lyu Q, Wang H, Kang Y, Wu X, Zheng HS, Laprocina K, Junghans K, Ding X, Huang CJ. RNA-seq reveals sub-zones in mouse adrenal zona fasciculata and the sexually dimorphic responses to thyroid hormone. Endocrinology 2020161 bqaa126. ( 10.1210/endocr/bqaa126) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chan WH, Komada M, Fukushima T, Southard-Smith EM, Anderson CR, Wakefield MJ. RNA-seq of isolated chromaffin cells highlights the role of sex-linked and imprinted genes in adrenal medulla development. Scientific Reports 20199 3929. ( 10.1038/s41598-019-40501-0) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dumontet T, Sahut-Barnola I, Septier A, Montanier N, Plotton I, Roucher-Boulez F, Ducros V, Lefrançois-Martinez AM, Pointud JC, Zubair M, et al. PKA signaling drives reticularis differentiation and sexually dimorphic adrenal cortex renewal. JCI Insight 20183. ( 10.1172/jci.insight.98394) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Spiga F, Zavala E, Walker JJ, Zhao Z, Terry JR, Lightman SL. Dynamic responses of the adrenal steroidogenic regulatory network. PNAS 2017114E6466–E6474. ( 10.1073/pnas.1703779114) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wong DL, Lesage A, Siddall B, Funder JW. Glucocorticoid regulation of phenylethanolamine N-methyltransferase in vivo. FASEB Journal 199263310–3315. ( 10.1096/fasebj.6.14.1426768) [DOI] [PubMed] [Google Scholar]
- 24.Wurtman RJ, Axelrod J. Adrenaline synthesis: control by the pituitary gland and adrenal glucocorticoids. Science 19651501464–1465. ( 10.1126/science.150.3702.1464) [DOI] [PubMed] [Google Scholar]
- 25.Ju Y, Mizutani T, Imamichi Y, Yazawa T, Matsumura T, Kawabe S, Kanno M, Umezawa A, Kangawa K, Miyamoto K. Nuclear receptor 5A (NR5A) family regulates 5-aminolevulinic acid synthase 1 (ALAS1) gene expression in steroidogenic cells. Endocrinology 20121535522–5534. ( 10.1210/en.2012-1334) [DOI] [PubMed] [Google Scholar]
- 26.Imamichi Y, Mizutani T, Ju Y, Matsumura T, Kawabe S, Kanno M, Yazawa T, Miyamoto K. Transcriptional regulation of human ferredoxin 1 in ovarian granulosa cells. Molecular and Cellular Endocrinology 20133701–10. ( 10.1016/j.mce.2013.02.012) [DOI] [PubMed] [Google Scholar]
- 27.Aigueperse C, Val P, Pacot C, Darne C, Lalli E, Sassone-Corsi P, Veyssiere G, Jean C, Martinez A. SF-1 (steroidogenic factor-1), C/EBPbeta (CCAAT/enhancer binding protein), and ubiquitous transcription factors NF1 (nuclear factor 1) and Sp1 (selective promoter factor 1) are required for regulation of the mouse aldose reductase-like gene (AKR1B7) expression in adrenocortical cells. Molecular Endocrinology 20011593–111. ( 10.1210/mend.15.1.0577) [DOI] [PubMed] [Google Scholar]
- 28.Oakley RH, Cidlowski JA. The biology of the glucocorticoid receptor: new signaling mechanisms in health and disease. Journal of Allergy and Clinical Immunology 20131321033–1044. ( 10.1016/j.jaci.2013.09.007) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Picard D, Yamamoto KR. Two signals mediate hormone-dependent nuclear localization of the glucocorticoid receptor. EMBO Journal 198763333–3340. ( 10.1002/j.1460-2075.1987.tb02654.x) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pedram A, Razandi M, Aitkenhead M, Hughes CC, Levin ER. Integration of the non-genomic and genomic actions of estrogen. Membrane-initiated signaling by steroid to transcription and cell biology. Journal of Biological Chemistry 200227750768–50775. ( 10.1074/jbc.M210106200) [DOI] [PubMed] [Google Scholar]
- 31.John S, Sabo PJ, Thurman RE, Sung MH, Biddie SC, Johnson TA, Hager GL, Stamatoyannopoulos JA. Chromatin accessibility pre-determines glucocorticoid receptor binding patterns. Nature Genetics 201143264–268. ( 10.1038/ng.759) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vockley CM, D'Ippolito AM, McDowell IC, Majoros WH, Safi A, Song L, Crawford GE, Reddy TE. Direct GR binding sites potentiate clusters of TF binding across the human genome. Cell 20161661269, .e19–1281.e19. ( 10.1016/j.cell.2016.07.049) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Severinova E, Alikunju S, Deng W, Dhawan P, Sayed N, Sayed D. Glucocorticoid receptor‐binding and transcriptome signature in cardiomyocytes. Journal of the American Heart Association 20198 e011484. ( 10.1161/JAHA.118.011484) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fu M, Sun T, Bookout AL, Downes M, Yu RT, Evans RM, Mangelsdorf DJ. A nuclear receptor atlas: 3T3-L1 adipogenesis. Molecular Endocrinology 2005192437–2450. ( 10.1210/me.2004-0539) [DOI] [PubMed] [Google Scholar]
- 35.Richard AC, Lun ATL, Lau WWY, Göttgens B, Marioni JC, Griffiths GM. T cell cytolytic capacity is independent of initial stimulation strength. Nature Immunology 201819849–858. ( 10.1038/s41590-018-0160-9) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ipseiz N, Uderhardt S, Scholtysek C, Steffen M, Schabbauer G, Bozec A, Schett G, Krönke G. The nuclear receptor Nr4a1 mediates anti-inflammatory effects of apoptotic cells. Journal of Immunology 20141924852–4858. ( 10.4049/jimmunol.1303377) [DOI] [PubMed] [Google Scholar]
- 37.Kassel O, Herrlich P. Crosstalk between the glucocorticoid receptor and other transcription factors: molecular aspects. Molecular and Cellular Endocrinology 200727513–29. ( 10.1016/j.mce.2007.07.003) [DOI] [PubMed] [Google Scholar]
- 38.Reddy TE, Pauli F, Sprouse RO, Neff NF, Newberry KM, Garabedian MJ, Myers RM. Genomic determination of the glucocorticoid response reveals unexpected mechanisms of gene regulation. Genome Research 2009192163–2171. ( 10.1101/gr.097022.109) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Paust HJ, Loeper S, Else T, Bamberger AM, Papadopoulos G, Pankoke D, Saeger W, Bamberger CM. Expression of the glucocorticoid receptor in the human adrenal cortex. Experimental and Clinical Endocrinology and Diabetes 20061146–10. ( 10.1055/s-2005-873007) [DOI] [PubMed] [Google Scholar]
- 40.Tacon LJ, Soon PS, Gill AJ, Chou AS, Clarkson A, Botling J, Stalberg PL, Skogseid BM, Robinson BG, Sidhu SB, et al. The glucocorticoid receptor is overexpressed in malignant adrenocortical tumors. Journal of Clinical Endocrinology and Metabolism 2009944591–4599. ( 10.1210/jc.2009-0546) [DOI] [PubMed] [Google Scholar]
- 41.Dardis A, Miller WL. Dexamethasone does not exert direct intracellular feedback on steroidogenesis in human adrenal NCI-H295A cells. Journal of Endocrinology 2003179131–142. ( 10.1677/joe.0.1790131) [DOI] [PubMed] [Google Scholar]
- 42.Vahl TP, Ulrich-Lai YM, Ostrander MM, Dolgas CM, Elfers EE, Seeley RJ, D'Alessio DA, Herman JP. Comparative analysis of ACTH and corticosterone sampling methods in rats. American Journal of Physiology: Endocrinology and Metabolism 2005289E823–E828. ( 10.1152/ajpendo.00122.2005) [DOI] [PubMed] [Google Scholar]
- 43.Liston C, Gan WB. Glucocorticoids are critical regulators of dendritic spine development and plasticity in vivo. PNAS 201110816074–16079. ( 10.1073/pnas.1110444108) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wurtman RJ.Control of epinephrine synthesis in the adrenal medulla by the adrenal cortex: hormonal specificity and dose-response characteristics. Endocrinology 196679608–614. ( 10.1210/endo-79-3-608) [DOI] [PubMed] [Google Scholar]
- 45.Wurtman RJ, Axelrod J. Control of enzymatic synthesis of adrenaline in the adrenal medulla by adrenal cortical steroids. Journal of Biological Chemistry 19662412301–2305. ( 10.1016/S0021-9258(1896620-2) [DOI] [PubMed] [Google Scholar]
- 46.Einer-Jensen N, Carter AM. Local transfer of hormones between blood vessels within the adrenal gland may explain the functional interaction between the adrenal cortex and medulla. Medical Hypotheses 199544471–474. ( 10.1016/0306-9877(9590508-1) [DOI] [PubMed] [Google Scholar]
- 47.Huang CC, Shih MC, Hsu NC, Chien Y, Chung BC. Fetal glucocorticoid synthesis is required for development of fetal adrenal medulla and hypothalamus feedback suppression. Endocrinology 20121534749–4756. ( 10.1210/en.2012-1258) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Evinger MJ, Towle AC, Park DH, Lee P, Joh TH. Glucocorticoids stimulate transcription of the rat phenylethanolamine N-methyltransferase (PNMT) gene in vivo and in vitro. Cellular and Molecular Neurobiology 199212193–215. ( 10.1007/BF00712926) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Frahm KA, Peffer ME, Zhang JY, Luthra S, Chakka AB, Couger MB, Chandran UR, Monaghan AP, DeFranco DB. Research resource: the dexamethasone transcriptome in hypothalamic embryonic neural stem cells. Molecular Endocrinology 201630144–154. ( 10.1210/me.2015-1258) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Szyf M, Milstone DS, Schimmer BP, Parker KL, Seidman JG. cis modification of the steroid 21-hydroxylase gene prevents its expression in the Y1 mouse adrenocortical tumor cell line. Molecular Endocrinology 199041144–1152. ( 10.1210/mend-4-8-1144) [DOI] [PubMed] [Google Scholar]
- 51.Thorngate FE, Strockbine PA, Erickson SK, Williams DL. Altered adrenal gland cholesterol metabolism in the apoE-deficient mouse. Journal of Lipid Research 2002431920–1926. ( 10.1194/jlr.m200205-jlr200) [DOI] [PubMed] [Google Scholar]
- 52.Huang CC, Kang Y. The transient cortical zone in the adrenal gland: the mystery of the adrenal X-zone. Journal of Endocrinology 2019241R51–R63. ( 10.1530/JOE-18-0632) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.El Wakil A, Mari B, Barhanin J, Lalli E. Genomic analysis of sexual dimorphism of gene expression in the mouse adrenal gland. Hormone and Metabolic Research 201345870–873. ( 10.1055/s-0033-1349881) [DOI] [PubMed] [Google Scholar]
- 54.Frahm KA, Waldman JK, Luthra S, Rudine AC, Monaghan-Nichols AP, Chandran UR, DeFranco DB. A comparison of the sexually dimorphic dexamethasone transcriptome in mouse cerebral cortical and hypothalamic embryonic neural stem cells. Molecular and Cellular Endocrinology 201847142–50. ( 10.1016/j.mce.2017.05.026) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wallensteen L, Zimmermann M, Thomsen Sandberg M, Gezelius A, Nordenström A, Hirvikoski T, Lajic S. Sex-dimorphic effects of prenatal treatment With dexamethasone. Journal of Clinical Endocrinology and Metabolism 20161013838–3846. ( 10.1210/jc.2016-1543) [DOI] [PubMed] [Google Scholar]
- 56.Kreider ML, Levin ED, Seidler FJ, Slotkin TA. Gestational dexamethasone treatment elicits sex-dependent alterations in locomotor activity, reward-based memory and hippocampal cholinergic function in adolescent and adult rats. Neuropsychopharmacology 2005301617–1623. ( 10.1038/sj.npp.1300716) [DOI] [PubMed] [Google Scholar]
- 57.Duma D, Collins JB, Chou JW, Cidlowski JA. Sexually dimorphic actions of glucocorticoids provide a link to inflammatory diseases with gender differences in prevalence. Science Signaling 20103 ra74. ( 10.1126/scisignal.2001077) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kananen K, Markkula M, Mikola M, Rainio EM, McNeilly A, Huhtaniemi I. Gonadectomy permits adrenocortical tumorigenesis in mice transgenic for the mouse inhibin alpha-subunit promoter/Simian virus 40 T-antigen fusion gene: evidence for negative autoregulation of the inhibin alpha-subunit gene. Molecular Endocrinology 1996101667–1677. ( 10.1210/mend.10.12.8961275) [DOI] [PubMed] [Google Scholar]
- 59.Quinn MA, Cidlowski JA. Endogenous hepatic glucocorticoid receptor signaling coordinates sex-biased inflammatory gene expression. FASEB Journal 201630971–982. ( 10.1096/fj.15-278309) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tejos-Bravo M, Oakley RH, Whirledge SD, Corrales WA, Silva JP, García-Rojo G, Toledo J, Sanchez W, Román-Albasini L, Aliaga E, et al. Deletion of hippocampal glucocorticoid receptors unveils sex-biased microRNA expression and neuronal morphology alterations in mice. Neurobiology of Stress 202114 100306. ( 10.1016/j.ynstr.2021.100306) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cruz-Topete D, Oakley RH, Carroll NG, He B, Myers PH, Xu X, Watts MN, Trosclair K, Glasscock E, Dominic P, et al. Deletion of the cardiomyocyte glucocorticoid receptor leads to sexually dimorphic changes in cardiac gene expression and progression to heart failure. Journal of the American Heart Association 20198 e011012. ( 10.1161/JAHA.118.011012) [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The RNA-seq data have been deposited in NCBI’s Gene Expression Omnibus and are accessible through GEO Series accession numbers GSE189182 and GSE189305 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE189182; https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE189305)



 This work is licensed under a
This work is licensed under a