Abstract
Background
The burden of Type 2 Diabetes Mellitus (T2DM) in South Asian countries is increasing rapidly. Self-care behaviour plays a vital role in managing T2DM and preventing complications. Research on self-care behaviours among people with T2DM has been widely conducted in South Asian countries, but there are no systematic reviews that assess self-care behaviour among people with T2DM in South Asia. This study systematically assessed the studies reporting self-care behaviours among people with T2DM in South-Asia.
Methods
Adhering to the PRISMA guidelines, we searched six bibliographic databases (Scopus, PubMed, CINAHL, Embase, Web of Science, and PsychInfo) to identify the relevant articles published between January 2000 through March 2022. Eligibility criteria included all observational and cross-sectional studies reporting on the prevalence of self-care behaviours (ie, diet, physical activity, medication adherence, blood glucose monitoring, and foot care) conducted in South Asian countries among people with T2DM.
Results
The database search returned 1567 articles. After deduplication (n = 758) and review of full-text articles (n = 192), 92 studies met inclusion criteria and were included. Forward and backward reference checks were performed on included studies, which resulted in an additional 18 articles. The pooled prevalence of adherence to blood glucose monitoring was 65% (95% CI = 49-80); 64% for medication adherence (95% CI = 53-74); 53% for physical activity (95% CI = 39-66); 48% for diet (95% CI = 38-58); 42% for foot care (95% CI = 30-54). About a quarter of people with T2DM consumed alcohol (25.2%, IQR = 13.8%-38.1%) and were using tobacco products (18.6%, IQR = 10.6%-23.8%).
Conclusions
Our findings suggest that the prevalence of self-care behaviours among people with T2DM in South Asia is low. This shows an urgent need to thoroughly investigate the barriers to the practising of self-care and design and implement interventions to improve diabetes self-care behaviour among people with T2DM in South Asia.
Diabetes mellitus is a major public health concern worldwide. The number of people with diabetes mellitus has increased from 108 million in 1980 to 422 million in 2014 [1] and 537 million in 2021 [2]. According to the International Diabetes Federation (IDF), this number is likely to reach 643 million by 2030 [2]. Type 2 Diabetes Mellitus (T2DM) constitutes more than 90% of all diabetes cases around the globe [2]. In recent years, the T2DM prevalence has significantly increased in low- and middle-income countries compared to higher-income countries in recent years [3]. The prevalence of T2DM in the South-Asian region specifically has doubled from 4.1% in 1980 to 8.6% in 2014 [3] and is estimated to reach 11.3% by 2045 [2].
South Asia is the southern region of Asia that comprises eight countries: Nepal, India, Bangladesh, Maldives, Sri Lanka, Pakistan, Bhutan, and Afghanistan [4]. South Asians are at higher risk of developing Non-Communicable Disease (NCDs), including T2DM, compared to other ethnic groups [5]. They tend to have more abdominal fat, more insulin resistance, low levels of adiponectin, low high-density lipoproteins, high low-density lipoproteins, and high triglycerides – characteristics which are responsible for the development of T2DM and cardiovascular diseases [5]. The prevalence of T2DM is the highest in Pakistan (26.7%) followed by India (8.3%), Bhutan (8.8%), Sri Lanka (9.8%), Bangladesh (12.5%), Maldives (6.7%), Afghanistan (8.7%) and Nepal (6.3%) [2]. The increased prevalence of T2DM negatively affects the socioeconomic circumstances for South Asian people by increasing diabetes-related health expenditure [6]. Poor knowledge about the disease, delayed diagnosis, poor adherence to self-care behaviours, and administration of harmful alternative medicines are the challenges for the treatment of T2DM among South-Asians [7,8].
The IDF has identified indicators for data collection (at least once in 12-24 months) to monitor the effectiveness of diabetes management, including self-care. The components of self-care are smoking status, alcohol consumption, self-monitoring (glucose, blood pressure, body weight), diet, physical activity, driving risk, medication adherence, insulin techniques, and dental care [9]. Similarly, the American Association of Diabetes Educators (AADE) has identified seven self-care behaviours: healthy eating, being active, monitoring, taking medication, problem-solving, healthy coping, and reducing risks as a framework for delivering patient-oriented diabetes care and education [10]. Adherence to self-care behaviours is essential for controlling adequate metabolism and preventing long-term complications [9,11-13]. Adherence to healthier behaviours significantly reduces the T2DM related complications and the mortality rate [14,15]. Despite this evidence, South-Asians adhere poorly to T2DM self-care behaviours [7,8,16]. To date, the components of self-care for management of T2DM in the South Asian region have not been defined by South Asian or the regional federations on diabetes.
As many South Asian countries have adopted the WHO’s Global Action Plan for the Prevention and Control of Non-Communicable Diseases [17], there is a need for research examining the planning, implementation, and evaluation of NCD prevention, control, and management strategies. Similarly, the higher risks of developing T2DM among South Asians have drawn the attention of policy makers in management and control of T2DM in this region. There are many studies examining the prevalence of self-care behaviours among those with T2DM in South-Asian countries [18-21]. However, a comprehensive systematic review and meta-analysis of this collective body of literature has not yet been conducted; posing challenges for policymakers to act on this. The literature on the prevalence of self-care behaviours among people with T2DM has been systematically analysed in regions other than South Asia [11,22-25] and reported widely varying rates of self-care behaviours. The findings from such reviews improve our understanding on practice of self-care among the people with T2DM across the regions. Because of the increasing burden of T2DM in South Asia [2] and South Asians being at higher risk for developing T2DM [5], there is an urgent need to design and implement effective prevention and management programs for T2DM. Improving our understanding on the practice of self-care behaviours among people with T2DM will help forward this agenda. This systematic review and meta-analysis aim to assess and summarize the findings on self-care behaviours among people with T2DM in South-Asia.
METHODS
This systematic review is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [26]. The South Asian countries included in this study were Bangladesh, Bhutan, India, Nepal, Pakistan, Maldives, Sri Lanka, and Afghanistan [4]. The review was registered with PROSPERO, an international prospective register of systematic reviews (Registration number: CRD42021242930).
Search strategy
We systematically searched six bibliographic databases: Scopus, PubMed, CINAHL, Embase, Web of Science, and PsychInfo for articles published between January 2000 through March 2022. This time frame aligns with the launch of the Millennium Development Goals (MDGs) in 2000 [27]. A search strategy for each database was developed with all the possible combinations of three keywords, “Type 2 Diabetes Mellitus”, “Self-care behaviour”, and “South-Asia” (See Appendix S1 in the Online Supplementary Document). Search tools such as PICO, PICOS, or SPIDER were not used because this study only reviewed observational and cross-sectional studies. Medical Subject Headings (MeSH), boolean operators, wildcards, truncation, and field tags were used where appropriate. Both the reference lists (ie, backward search) and articles citing (ie, forwards search) of included studies were checked by two authors (GP, PD) for additional relevant studies.
Inclusion and exclusion criteria
Observational, and cross-sectional studies that quantitatively reported on the practice of self-care behaviours (ie, diet, physical activity, medication adherence, blood glucose monitoring, and foot care) among adults with T2DM from South-Asian countries that were published in the English language were included. These five self-care behaviours were based on the key indicators for self-care behaviour as suggested by the IDF and AADE [9,10]. Furthermore, these domains of self-care were also assessed by several review studies on self-care among people with T2DM in other settings [11,22-25]. Studies not mentioning the type of diabetes examined, studies based on the same data set, and studies without a full-text publication available were excluded.
Screening
One author (GP) performed the online database search in first week of April 2021 (updated on 25 March 2022). Articles identified through the search were exported into the EndNote referencing software and deduplicated. Titles and abstracts were screened independently by two reviewers (GP, PD). The potentially eligible studies underwent full-text screening using the selection criteria. Disagreements between the two reviewers (GP and PD) was discussed in consultation with a third reviewer (LR). Remaining disagreements were discussed within the study team (GP, PD, TB, LR, UNY, TS, and CV) until a consensus was reached. A detailed study selection process is presented in the PRISMA flowchart (Figure 1) [26].
Figure 1.
PRISMA flow diagram (2009) for reporting systematic review and meta-analysis.
Data extraction and quality assessment
A data extraction template similar to the one used in the systematic review of Stephani et al. [24] was developed in Microsoft Excel to collect information from the selected studies for the analysis. Information on the primary author, publication year, country, study design, sample size, demographic characteristics of the population (eg, age, gender, and other contextual information), and reported self-care behaviours were extracted.
The Jonna Briggs Institute (JBI) critical appraisal checklist was used to assess the methodological quality of the selected studies following each study design [28]. Two independent reviewers (GP, PD) critically appraised the selected articles using the JBI critical appraisal checklist. This tool involves assessing the study’s methodological quality in dealing with bias at different study stages. A checklist for analytical cross-sectional studies (with 8 appraising items) and another for prevalence studies (with 9 appraising items) were used. Each response was scored with 1 (if the response to the question was “yes”) and 0 (if the response to the question was “no” or “unclear” or “not applicable”). Based on the score, studies were categorized into high (80% and above), moderate (60%-80%) and low quality (<60%) [29]. Discrepancies between reviewers (GP, PD) on study quality assessment were resolved through discussion and consultation with a third reviewer (LR).
Meta-analysis
A systematic narrative synthesis was performed to describe the characteristics and results of all included studies. The narrative synthesis followed the Guidance of the Conduct of Narrative Synthesis in Systematic Reviews [30]. Data synthesis and analysis were performed by one reviewer (GP) and the findings were discussed with all other team members.
In the meta-analysis, the overall pooled prevalence for each domain of self-care behaviours was conducted. A subgroup analysis of self-care behaviours including diet, physical activity, and foot care was conducted based on studies that either used a standardised tool to assess self-care domains or those that clearly defined the self-care domain and studies that did not clarify either the measure used to assess self-care domain or that did not define self-care domain. Medication adherence was not considered for subgroup analysis as most included studies used standard tools, while some reported regular intake of medication as recommended by their health care providers. Similarly, blood glucose monitoring differs from person to person based on their blood glucose level, so subgroup analysis was based on studies that assessed blood glucose monitoring on monthly basis and studies that assessed blood glucose monitoring at least once in three months. We used the quality effects model (QE) for bias adjustment [31]. The advantage of the QE model is that the between-study variability is adjusted based on the relative quality rank of the studies instead of on random variables assigned by the random effect (RE) model. The heterogeneity of the studies was reported by the I-squared value (I2) which measures the proportion of total variance between studies beyond random error [32]. As significant heterogeneity was detected among the studies (I2>50%) in the meta-analysis, a random-effects model was used. All the analyses were conducted using the MetaXL software version 5.3 [33]. Publication bias was assessed using both a graphical (Doi plot) and quantitative (Luis Furuya-Kanamori (LFK) index) examination for potential small-study effects [32]. LFK indices are defined as no (±1), minor (between ±1 and ±2), and major (>±2) asymmetry, respectively. Sensitivity and subgroup analysis were performed for extreme levels of heterogeneity between studies (I2≥90%) [32].
RESULTS
Study selection
A total of 1585 studies were identified through the database search (n = 1567) and forward and backward reference checking (n = 18). The duplicates (n = 809) were removed, and 758 studies underwent the title and abstract screening. Of these, 260 studies were eligible for full-text retrieval and 192 studies were retrieved with full-text articles. It was not possible to access the full-text of 68 studies, which were conference abstracts or articles published in local paper-based journals. After full text review, 92 studies met the inclusion criteria and were included in the qualitative synthesis, and 70 were eligible to be included in the meta-analysis (22 were excluded due to insufficiently disaggregated data). A detailed process of study screening and selection is presented in the PRISMA flow diagram (Figure 1).
Quality assessment of the included studies
Quality assessment of 92 studies included in this review was done using Jonna Briggs Institute (JBI) Critical Appraisal Tool based on study design. Thirty-five studies (38%) were assessed as high quality, 33 studies (36%) were assessed as moderate quality, and 24 studies (26%) were assessed as poor quality (Appendix S2 in the Online Supplementary Document). The detailed information on quality assessment of individual study is presented in Table 1.
Table 1.
General characteristics of included studies
| Sample characteristics |
Reported self-care behaviours |
Quality score | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Author
|
Year
|
Country
|
Sample size
|
Male
|
Female
|
Mean age (SD)
|
Diet
|
Physical Activity
|
Medication intake
|
Foot care
|
SMBG
|
|
| Shah, Kamdar and Shah [34] |
2009 |
India |
238 |
120 |
118 |
55.8 (±10.2) |
X |
|
|
X |
X |
Low |
| Sultana et al. [35] |
2010 |
India |
218 |
104 |
114 |
51.5 (±12.3) |
|
|
X |
|
|
Moderate |
| Malathy et al. [36] |
2011 |
India |
207 |
85 |
122 |
52.1 |
X |
X |
X |
|
|
Low |
| Gopichandran et al. [37] |
2012 |
India |
200 |
82 |
118 |
NR |
X |
X |
X |
|
X |
High |
| Patel et al. [38] |
2012 |
India |
399 |
259 |
140 |
53.1 (±7.9) |
X |
X |
|
|
X |
High |
| Sasi et al. [39] |
2013 |
India |
546 |
303 |
243 |
55.4 |
X |
X |
X |
X |
|
Moderate |
| Arulmozhi and Mahalakshmy [40] |
2014 |
India |
150 |
75 |
75 |
54.0 (±12.0) |
X |
X |
X |
X |
|
Moderate |
| Khan et al. [41] |
2014 |
India |
184 |
81 |
103 |
51.4 (±12.2) |
|
|
X |
|
|
Moderate |
| Santhanakrishnan, Lakshminarayanan and Kar [42] |
2014 |
India |
135 |
27 |
108 |
59.0 (±12.0) |
X |
X |
X |
X |
|
Low |
| Saurabh et al. [43] |
2014 |
India |
103 |
48 |
55 |
54.8 (±11.8) |
X |
X |
|
X |
X |
Low |
| Sajith et al. [44] |
2014 |
India |
105 |
60 |
45 |
NR |
X |
X |
X |
|
|
Low |
| Abraham et al. [45] |
2015 |
India |
60 |
25 |
35 |
50.7 (±7.0) |
X |
X |
|
X |
X |
High |
| Divya and Nadig [46] |
2015 |
India |
150 |
104 |
46 |
49.1 |
|
|
X |
|
|
Low |
| Basu et al. [47] |
2015 |
India |
385 |
159 |
226 |
53.1 (±10.2) |
X |
X |
X |
|
|
High |
| Rajasekharan et al. [48] |
2015 |
India |
290 |
174 |
116 |
47.9 (±8.9) |
X |
X |
X |
X |
X |
High |
| Das et al. [49] |
2016 |
India |
232 |
199 |
33 |
57.0 (±8.9) |
X |
X |
X |
X |
X |
High |
| Karthikeyan, Madhusudhan and Selvamuthukumaran [50] |
2016 |
India |
345 |
185 |
160 |
NR |
|
|
X |
|
|
Low |
| Pathania et al. [51] |
2016 |
India |
48 |
25 |
23 |
57.4 (±10.6) |
|
|
X |
|
|
Moderate |
| Dinesh, Kulkarni and Gangadhar [52] |
2016 |
India |
400 |
245 |
155 |
NR |
X |
X |
X |
X |
X |
High |
| Debnath et al. [53] |
2017 |
India |
450 |
253 |
197 |
64.8 (±4.6) |
|
X |
X |
X |
X |
Moderate |
| Kumar et al. [54] |
2017 |
India |
124 |
68 |
56 |
Median = 60 (IQR = 50-68) years |
|
|
X |
|
X |
High |
| Samu, Amirthalingam and Mohammed [55] |
2017 |
India |
86 |
38 |
48 |
NR |
|
|
X |
|
|
High |
| Sheeba, Ak and Biju [56] |
2017 |
India |
100 |
60 |
40 |
NR |
X |
X |
X |
X |
X |
Low |
| Srinath, Basavegowda and Tharuni [57] |
2017 |
India |
400 |
172 |
228 |
NR |
X |
X |
X |
X |
X |
Moderate |
| Britto et al. [58] |
2018 |
India |
25 |
NR |
NR |
58.8 (±8.9) |
|
X |
|
|
|
Moderate |
| Pati et al. [59] |
2018 |
India |
321 |
204 |
117 |
51.0 (±12.8) |
X |
X |
|
|
|
Low |
| Ravi, Kumar and Gopichandran [60] |
2018 |
India |
200 |
96 |
104 |
NR |
X |
X |
|
X |
X |
High |
| Venkatesan, Dongre and Ganapathy [61] |
2018 |
India |
328 |
149 |
179 |
57.3 (±12.1) |
|
|
X |
|
|
High |
| Jasmine and Iyer [62] |
2019 |
India |
77 |
33 |
44 |
NR |
X |
X |
X |
X |
X |
Low |
| Acharya et al. [63] |
2019 |
India |
200 |
74 |
126 |
49.8 (±10.5) |
|
|
X |
|
|
Low |
| Aravind, Joy and Rakesh [64] |
2019 |
India |
68 |
39 |
29 |
62.5 (±11.2) |
X |
X |
|
|
X |
Moderate |
| Banerjee et al. [65] |
2019 |
India |
347 |
210 |
137 |
NR |
|
X |
|
|
|
Moderate |
| Raj, Selvaraj and Thomas [66] |
2019 |
India |
205 |
110 |
95 |
62.3 (±9.3) |
|
X |
|
|
|
High |
| Sirari et al. [67] |
2019 |
India |
60 |
30 |
30 |
54.9 (±9.2) |
X |
X |
|
X |
X |
High |
| Bashir et al. [68] |
2020 |
India |
203 |
99 |
104 |
53.9 (±10.5) |
X |
X |
|
|
|
Moderate |
| Chandrika et al. [69] |
2020 |
India |
208 |
95 |
113 |
51.3 (±9.4) |
X |
X |
X |
|
X |
High |
| Kowsalya et al. [70] |
2020 |
India |
60 |
32 |
28 |
NR |
|
|
X |
|
|
Low |
| Palathingal et al. [71] |
2020 |
India |
200 |
123 |
77 |
NR |
|
|
X |
|
X |
Low |
| Patnaik et al. [72] |
2020 |
India |
100 |
58 |
42 |
54.2 (±12.0) |
X |
|
|
|
|
Moderate |
| Shrivastva et al. [73] |
2020 |
India |
166 |
109 |
57 |
NR |
X |
X |
|
|
X |
Moderate |
| Achappa [74] |
2020 |
India |
70 |
28 |
42 |
58.9 (±14.5) |
|
|
X |
|
|
Low |
| Karthik et al. [75] |
2020 |
India |
250 |
137 |
113 |
NR |
X |
X |
X |
X |
X |
Moderate |
| Kumar et al. [76] |
2021 |
India |
105 |
43 |
62 |
54.8 (±8.9) |
X |
X |
|
|
X |
High |
| Rana et al. [77] |
2021 |
India |
200 |
100 |
100 |
56.2 (±8.3) |
X |
X |
X |
|
X |
Low |
| Verma, et al. [18] |
2021 |
India |
416 |
243 |
173 |
NR |
|
X |
|
X |
|
High |
| Burman et al. [78] |
2021 |
India |
367 |
172 |
195 |
51.4 (±9.3) |
X |
X |
X |
X |
X |
Moderate |
| Durai et al. [79] |
2021 |
India |
390 |
104 |
286 |
56.2 (±10.4) |
X |
X |
X |
X |
X |
Moderate |
| Mishra et al. [80] |
2021 |
India |
277 |
158 |
119 |
50.8 (±10.6) |
|
|
X |
|
|
Moderate |
| Singh et al. [81] |
2021 |
India |
350 |
179 |
171 |
NR |
|
|
X |
|
|
Moderate |
| Aravindakshan et al. [82] |
2021 |
India |
218 |
87 |
131 |
62.1 (±12.2) |
|
|
X |
|
|
Moderate |
| Zuberi, Syed and Bhatti [83] |
2011 |
Pakistan |
286 |
128 |
158 |
NR |
X |
X |
X |
X |
|
High |
| Ahmed et al. [84] |
2015 |
Pakistan |
139 |
60 |
79 |
43.0 (±16.0) |
X |
X |
X |
X |
X |
Moderate |
| Javaid et al. [85] |
2016 |
Pakistan |
120 |
38 |
62 |
50.7 (±10.6) |
X |
X |
|
|
|
Low |
| Bukhsh et al. [86] |
2017 |
Pakistan |
130 |
55 |
75 |
51.3 (±10.4) |
X |
X |
|
|
X |
Moderate |
| Iqbal et al. [87] |
2017 |
Pakistan |
300 |
180 |
120 |
51.2 (±9.5) |
|
|
X |
|
|
High |
| Nazirl et al. [88] |
2017 |
Pakistan |
392 |
222 |
170 |
50.7 (±9.6) |
|
|
X |
|
|
High |
| Rana et al. [89] |
2017 |
Pakistan |
145 |
54 |
91 |
50.2 (±8.5) |
|
|
X |
|
|
Low |
| Bukhsh et al. [90] |
2018 |
Pakistan |
218 |
112 |
106 |
50.7 (±13.3) |
X |
X |
|
|
X |
High |
| Farooq et al. [91] |
2018 |
Pakistan |
180 |
82 |
98 |
50.3 (±11.2) |
X |
|
|
|
|
Low |
| Zafar et al. [92] |
2018 |
Pakistan |
220 |
93 |
127 |
52.9 (±12.5) |
|
|
|
X |
|
Moderate |
| Hussain, Said and Khan [93] |
2020 |
Pakistan |
524 |
0 |
524 |
64.0 |
|
|
X |
|
|
Moderate |
| Siddique et al. [94] |
2020 |
Pakistan |
154 |
68 |
86 |
NR |
X |
X |
X |
|
X |
Moderate |
| Malik et al. [19] |
2020 |
Pakistan |
363 |
241 |
122 |
45.7 |
X |
X |
X |
X |
X |
Moderate |
| Sayeed et al. [95] |
2020 |
Pakistan |
317 |
174 |
143 |
NR |
X |
X |
|
|
X |
Moderate |
| Ishaq et al. [96] |
2021 |
Pakistan |
300 |
180 |
120 |
51.2 (±9.6) |
|
|
X |
|
|
Moderate |
| Shrestha et al. [97] |
2013 |
Nepal |
100 |
48 |
52 |
58.1 (±11.6) |
|
|
X |
|
|
Low |
| Parajuli et al. [98] |
2014 |
Nepal |
385 |
187 |
198 |
54.4 (±11.5) |
X |
X |
|
|
|
High |
| Sharma and Bhandari [99] |
2014 |
Nepal |
100 |
56 |
44 |
NR |
X |
X |
|
|
X |
Low |
| Bhandari and Kim [20] |
2016 |
Nepal |
230 |
91 |
139 |
56.9 (±10.8) |
X |
X |
X |
X |
X |
High |
| Ghimire [100] |
2017 |
Nepal |
197 |
111 |
86 |
54.7 (±11.3) |
X |
X |
|
|
|
High |
| Shrestha et al. [101] |
2017 |
Nepal |
183 |
116 |
67 |
58.7 (±12.9) |
X |
X |
X |
|
|
Low |
| Ghimire and Devi [102] |
2018 |
Nepal |
115 |
62 |
53 |
60.0 (±10.3) |
X |
X |
|
|
X |
Moderate |
| Kadariya and Aro [103] |
2018 |
Nepal |
270 |
167 |
103 |
53 (ranging from 30-70 y) |
|
X |
|
|
|
High |
| Sapkota et al. [104] |
2018 |
Nepal |
200 |
116 |
84 |
51.9 (±11.5) |
|
X |
X |
|
X |
Moderate |
| Thapa [105] |
2018 |
Nepal |
141 |
71 |
70 |
NR |
X |
X |
X |
X |
X |
Low |
| Pokhrel et al. [106] |
2019 |
Nepal |
480 |
236 |
244 |
58.3 (±12.5) |
X |
X |
X |
|
X |
High |
| Bhattarai et al. [107] |
2019 |
Nepal |
214 |
104 |
110 |
NR |
X |
X |
X |
|
X |
Low |
| Sharma et al. [108] |
2021 |
Nepal |
296 |
120 |
176 |
59.5 (±11.7) |
|
|
X |
|
|
High |
| Shrestha et al. [109] |
2021 |
Nepal |
354 |
156 |
198 |
51.7 (±12.6) |
X |
X |
X |
X |
X |
High |
| Kandel et al. [110] |
2022 |
Nepal |
411 |
177 |
234 |
NR |
X |
X |
X |
X |
X |
High |
| Saleh et al. [111] |
2012 |
Bangladesh |
160 |
72 |
88 |
45.1 (±5.6) |
X |
X |
|
|
|
Low |
| Mumu et al. [112] |
2014 |
Bangladesh |
374 |
157 |
217 |
51.0 (±11.3) |
X |
X |
|
X |
X |
Moderate |
| Saleh et al. [113] |
2014 |
Bangladesh |
500 |
249 |
251 |
54.2 (±11.2) |
X |
X |
X |
X |
X |
High |
| Ahmed et al. [114] |
2017 |
Bangladesh |
122 |
67 |
55 |
57.5 (±8.7) |
|
|
X |
|
X |
High |
| Chowdhury et al. [115] |
2018 |
Bangladesh |
11917 |
4418 |
7499 |
50.0 (±12.0) |
X |
X |
|
|
X |
Moderate |
| Bukht et al. [116] |
2019 |
Bangladesh |
977 |
468 |
509 |
56.0
(±8.0) |
|
X |
|
|
|
High |
| Majid et al. [117] |
2019 |
Bangladesh |
420 |
248 |
172 |
47.2 (±6.4) |
X |
|
|
|
|
High |
| Islam et al. [118] |
2020 |
Bangladesh |
265 |
133 |
132 |
50.3 (±9.9) |
|
X |
|
X |
X |
High |
| MahmudulHasan et al. [119] |
2021 |
Bangladesh |
379 |
175 |
204 |
NR |
X |
X |
|
|
|
High |
| Mannan et al. [21] |
2021 |
Bangladesh |
2061 |
1233 |
828 |
50.6 (±12.1) |
X |
|
X |
|
|
High |
| Medagama and Galgomuwa [120] |
2018 |
Sri-Lanka |
400 |
113 |
287 |
55.4 (±8.9) |
|
X |
|
|
|
Moderate |
| Rathish et al. [121] | 2019 | Sri-Lanka | 200 | 100 | 100 | NR | X | Moderate | ||||
SMBG – self-monitoring of blood glucose, NR – not reported, IQR – interquartile range, SD – standard deviation
Characteristics of the included studies
Of the total included studies (n = 92), 50 were conducted in India [18,34-82], 15 in Pakistan [19,83-96], 15 in Nepal [20,97-110], 10 in Bangladesh [21,111-119] and two in Sri-Lanka [120,121]. No studies were conducted in the Maldives, Bhutan, and Afghanistan. 78 studies were based on data recorded in health facilities (hospitals, primary health care centres, diabetic clinics, and pharmacies) while 14 studies were based on data collected in community settings. The general characteristics of the studies are summarized in Table 1. The total number of participants in the included studies was 36 180 (16 601 male and 19 559 female) and ranged from 48 participants in the smallest study [51] to 11 917 in the largest study [115]. The mean age of the participants, as reported by 66 studies, ranged from 43 to 64 years. 24 studies reported the length of time participants were living with T2DM, which ranged from 1.5 to 9.7 years. Thirty studies reported on smoking habits (n = 30) while fifteen studies reported on alcohol use (n = 15). The median score for tobacco use (smoke and/or smokeless form) was 18.6% (IQR = 10.6%-23.8%) and 25.2% (IQR = 13.8%-38.1%) for alcohol consumption among the participants.
Domains of T2DM self-care behaviours
Among all the self-care behaviours, physical activity (n = 61) was the most reported self-care behaviour followed by medication intake (n = 57), dietary habits (n = 56), self-monitoring of blood glucose (n = 42), and foot care (n = 30). Studies adopted a wide range of scales (n = 58) to assess the different domains of self-care behaviours. Many studies did not provide information about the tool used (n = 27) and some studies (n = 7) reported using author-developed tools. The Summary of Diabetes Self-Care Activities measure (SDSCA) [122] was used by 16 studies and the Diabetes Self-Management Questionnaire (DSMQ) [123] by seven studies to assess the different domains of self-care behaviours. Similarly, the Morisky Medication Adherence Scale (MMAS) scale was used by 23 studies in assessing the status of medication adherence among the study participants [124,125]. Four studies used the Global Physical Activity Questionnaire (GPAQ) [126] and the International Physical Activity Questionnaire (IPAQ) [127] was used by three studies to measure the physical activity level of the study participants. The findings on self-care behaviours were reported in the form of a percentage, mean and median.
Physical activity
Physical activity was assessed by 61 studies (Table 2). The Summary of Diabetes Self-Care Activities measure (n = 15), the Diabetes Self-Management Questionnaire (n = 7), the Global Physical Activity Questionnaire (n = 4), and the International Physical Activity Questionnaire (n = 3) were the most used tools in assessing the study participants’ physical activity. Two studies reported the mean number of days in a week participants were physically active, ranging from 4.08 to 4.23 days [20,45]. Additionally, four studies reported the mean score for physical activity ranging from 3.74 to 5.1 (scale range of 0-10, where 10 represents the optimal practice of self-care) [73,76,86,95].
Table 2.
Physical activity
| Authors | Year | Country | Sample size | Measure | Practice rates |
|---|---|---|---|---|---|
| Malathy et al.[36] |
2011 |
India |
207 |
Performing exercise regularly (Self-reported) |
41% |
| Gopichandran et al. [37] |
2012 |
India |
200 |
Good exercise behaviour (at least 20 min a day exercise for 5 d in last week) |
19.5% |
| Patel et al. [38] |
2012 |
India |
399 |
Following recommended Physical Activity |
54% |
| Sasi et al. [39] |
2013 |
India |
546 |
Performing physical exercise for at least 30 min a day and 5 d a week |
37% |
| Arulmozhi and Mahalakshmy [40] |
2014 |
India |
150 |
Physical exercise for at least 30 min for at least 4 d/week |
22.7% |
| Santhanakrishnan, Lakshminarayanan and Kar [42] |
2014 |
India |
135 |
Practicing Physical Activity |
37.0% |
| Saurabh et al. [43] |
2014 |
India |
103 |
Performing Physical Activity in addition to their routine work |
45.6%, |
| Sajith et al. [44] |
2014 |
India |
105 |
Exercise adherence |
32.3%, |
| Abraham et al. [45] |
2015 |
India |
60 |
Mean (SD) number of days in a week performing at least 30 min of physical activity or exercise |
4.1 (±2.8) |
| Basu et al. [47] | 2015 | India | 385 | Specific exercise session averaging 30 min/d |
3.6 (±2.3) |
| a. <5 d (non-adherent) in the previous 7 d |
52% |
||||
|
|
|
|
|
b. ≥5 d (adherent) in the previous 7 d |
48% |
| Rajasekharan et al. [48] |
2015 |
India |
290 |
Practicing Physical Activity of at least 30 min on all days of the week |
43.4% |
| Das et al. [49] |
2016 |
India |
232 |
Exercise being done regularly |
53.9% |
| Dinesh, Kulkarni and Gangadhar [52] |
2016 |
India |
400 |
Exercising at least 5 d a week for 20-30 min |
20.5% |
| Debnath et al. [53] |
2017 |
India |
450 |
Performing good physical activity (regular walking) |
38% |
| Sheeba, Ak and Biju [56] |
2017 |
India |
100 |
Performing regular exercise |
46% |
| Srinath, Basavegowda and Tharuni [57] |
2017 |
India |
400 |
Participated in walking in the last week |
27.7% |
| Britto et al. [58] | 2018 | India | 25 | Inactive |
20% |
| Moderately active |
52% |
||||
|
|
|
|
|
Highly active |
28% |
| Pati et al. [59] |
2018 |
India |
321 |
Performing Physical Activity frequently |
59% |
| Ravi, Kumar and Gopichandran [60] |
2018 |
India |
200 |
Median number of days in the past week participating in at least 30 min of physical activity |
0 (IQR:0-7) |
| Jasmine and Iyer [62] |
2019 |
India |
77 |
Following regular physical exercise |
15.6% |
| Aravind, Joy and Rakesh [64] |
2019 |
India |
68 |
Good physical activity |
39.7% |
| Banerjee et al. [65] | 2019 | India | 347 | High level of physical activity |
34.9% |
| Moderate level of physical activity |
31.1% |
||||
|
|
|
|
|
Low level of physical activity |
34% |
| Raj, Selvaraj and Thomas [66] | 2019 | India | 205 | Low Physical Activity |
61.5% |
| Moderate Physical Activity |
19.5% |
||||
|
|
|
|
|
High Physical Activity |
19.0% |
| Sirari et al. [67] | 2019 | India | 60 | Performing at least 30 min of Physical Activity |
61.3% |
|
|
|
|
|
Performing specific exercise session |
48.4% |
| Bashir et al. [68] |
2020 |
India |
203 |
Mean (SD) score for daily exercising |
2.6 (±0.9) |
| Performing daily exercise as recommended |
38.9% |
||||
| Chandrika et al. [69] |
2020 |
India |
208 |
Performed physical activity for at least 30 min for minimum 5 d in the last week |
30.3% |
| Chandrika et al. [69] |
2020 |
India |
208 |
Performed physical activity for at least 30 min for minimum 5 d in the last week |
30.3% |
| Shrivastva et al. [73] |
2020 |
India |
166 |
Mean (SD) score for physical activity |
4.9 (±2.8), |
| Karthik et al. [75] |
2020 |
India |
250 |
Performing satisfactory level of exercise |
19.2% |
| Kumar et al. [76] |
2021 |
India |
105 |
Mean (SD) score for physical activity |
5.1 (±1.6) |
| Rana et al. [77] |
2021 |
India |
200 |
Mean (SD) score adhering the exercise |
1.2 (±1.3) |
| Verma et al. [18] |
2021 |
India |
416 |
Performing physical activity |
72% |
| Burman et al. [78] |
2021 |
India |
367 |
Performing satisfactory level of exercise for at least 30 min in a week |
76.5% |
| Durai et al. [79] |
2021 |
India |
390 |
Performing physical activity (at least 30 min for 3 or more days a week) |
46% |
| Zuberi, Syed and Bhatti [83] |
2011 |
Pakistan |
286 |
Compliant with exercise |
28.0% |
| Ahmed et al. [84] |
2015 |
Pakistan |
139 |
Following regular physical activity |
8.6% |
| Javaid et al. [85] |
2016 |
Pakistan |
120 |
Low physical activity |
67.0% |
| Moderate physical activity |
33.0% |
||||
| Bukhsh et al. [86] |
2017 |
Pakistan |
130 |
Mean (SD) score for physical activity |
4.0 (±3.1) |
| Bukhsh et al. [90] |
2018 |
Pakistan |
218 |
Median (IQR) score for physical activity |
3.3 (1.11–6.67) |
| Siddique et al. [94] |
2020 |
Pakistan |
154 |
Performing exercise daily for 30 min |
27.9% |
| Malik et al. [19] |
2020 |
Pakistan |
363 |
Exercising at least 20-30 min per day for at least five days a week |
65.3% |
| Sayeed et al. [95] |
2020 |
Pakistan |
317 |
Mean (SD) score for physical activity |
3.7 (±1.03) |
| Parajuli et al. [98] | 2014 | Nepal | 385 | Mean (SD) score for adherence to Physical Activity |
67 (±23.9) |
| a. Non-adherence |
42.1% |
||||
| b. Poor adherence |
36.6% |
||||
|
|
|
|
|
c. Good adherence |
21.0% |
| Sharma and Bhandari [99] | 2014 | Nepal | 100 | Exercise frequency |
|
| a. Daily |
72.0% |
||||
| b. 2-3 d a week |
18.0% |
||||
| c. 4-5 d a week |
10.0% |
||||
| Exercise duration |
|||||
| a. 20 min |
22.0% |
||||
| b. 30 min |
30.0% |
||||
|
|
|
|
|
c. 60 min |
48.0% |
| Bhandari and Kim [20] |
2016 |
Nepal |
230 |
Mean (SD) number of days in a week performing exercise |
4.2(±2.8) |
| Ghimire [100] |
2017 |
Nepal |
197 |
Non-compliant to exercise recommendation |
46.0% |
| Shrestha et al. [101] |
2017 |
Nepal |
183 |
Performing physical exercise |
67.7% |
| Kadariya and Aro [103] | 2018 | Nepal | 270 | Low level of physical activity |
20.4% |
| Medium level of physical activity |
51.8% |
||||
|
|
|
|
|
High level of physical activity |
27.8% |
| Ghimire and Devi [102] |
2018 |
Nepal |
115 |
Performing good physical activity |
56.5% |
| Sapkota et al. [104] |
2018 |
Nepal |
200 |
Performing exercise regularly |
27% |
| Thapa [105] |
2018 |
Nepal |
141 |
Performing exercise regularly |
56.7% |
| Pokhrel et al. [106] |
2019 |
Nepal |
480 |
High adherence to exercise |
38.3% |
| Bhattarai et al. [107] |
2019 |
Nepal |
214 |
Not performing exercise regularly |
63.6% |
| Shrestha et al. [109] |
2021 |
Nepal |
354 |
Performing physical activity |
44% |
| Kandel et al. [110] |
2022 |
Nepal |
411 |
Recreational physical activity 7 d a week |
48.2% |
| Saleh et al. [111] |
2012 |
Bangladesh |
160 |
Performing exercise |
23.0% |
| Mumu et al. [112] |
2014 |
Bangladesh |
374 |
Non-adherence to exercise (<30 min a day): |
25.0% |
| Saleh et al. [113] |
2014 |
Bangladesh |
500 |
Non-adherence to exercise (exercise <45 min/d) |
33.2% |
| Chowdhury et al. [115] |
2018 |
Bangladesh |
11917 |
Performing regular exercise (more than 30 min/ at least 5 d per week) |
69.0% |
| Bukht et al. [116] |
2019 |
Bangladesh |
977 |
Inactive/low (<150 min/week) |
74.0% |
| Moderate-to-vigorous (≥150minutes/week) |
26.0% |
||||
| Islam et al. [118] |
2020 |
Bangladesh |
265 |
Walk (30 min/d) for at least 5 d (last week) |
70.9% |
| MahmudulHasan et al. [119] |
2021 |
Bangladesh |
379 |
Adherence to recommended Physical Activity (≥150 min in 7 d) |
38.5% |
| Medagama and Galgomuwa [120] | 2018 | Sri Lanka | 400 | Physically inactive |
21.5% |
| Minimally active |
33.8% |
||||
| Physically active | 44.8% | ||||
SD – standard deviation, IQR – interquartile range
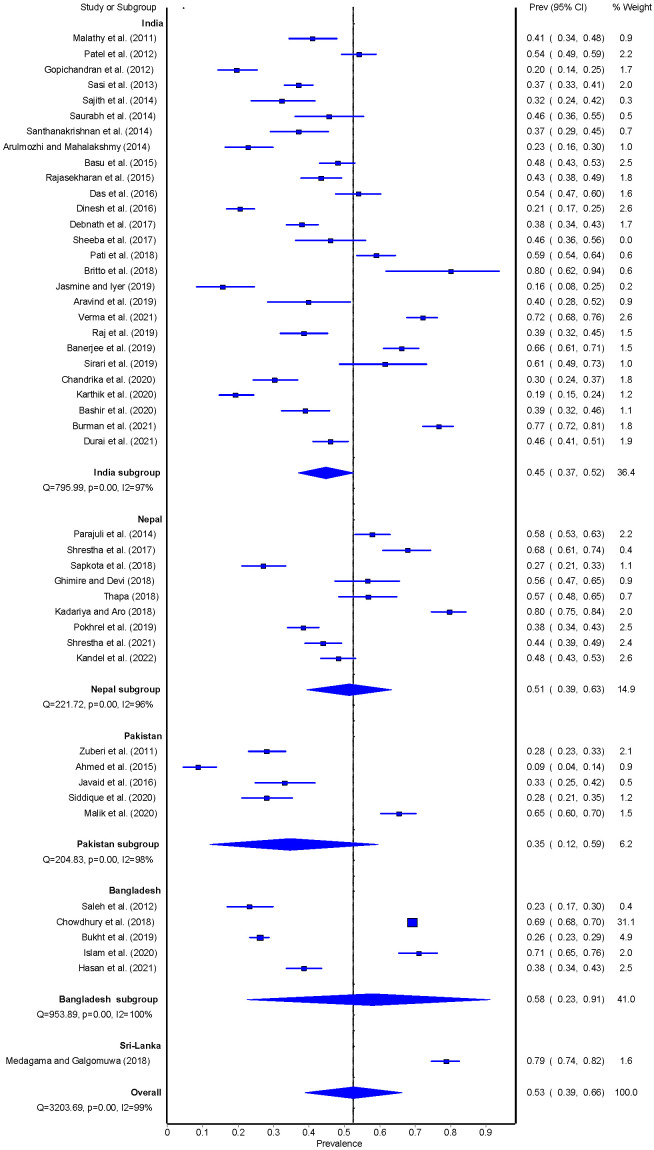
The overall pooled prevalence of adherence to sufficient physical activity was 53% (95% CI = 39-66) and ranged from 9% to 80%. In terms of country-specific pooled prevalence, studies conducted in Sri Lanka (n = 1) reported an adherence of 79% (95% CI = 74-82), followed by Bangladesh (n = 5; 58%, 95% CI = 23-91), Nepal (n = 9; 51%, 95% CI = 39-63), India (n = 27; 45%, 95% CI = 37-52) and Pakistan (n = 5; 35%, 95% CI = 12-59) (Figure 2). Adherence to sufficient physical activity was 54% (95% CI = 38%-69%) for studies that either used a standardised tool to assess physical activity or studies that clearly defined what sufficient physical activity constitutes. Adherence to sufficient physical activity was 47% (95% CI = 34%-59%) for studies that did not clarify either the measure used to assess physical activity or studies that did not define what sufficient physical activity constitutes (Figure S1-S2 in the Online Supplementary Document).
Figure 2.
Pooled estimate of physical activity among people with T2DM.
Medication use
57 studies measured adherence to the medication use (Table 3). The Morisky Medication Adherence Scale (n = 23) and the Summary of Diabetes Self-Care Activities measure (n = 10) were the most used tools in measuring adherence to medication use. A study from Nepal reported a mean of 6.77 number of days per week participants’ adhering to medication [20]. Non-adherence to Oral Hypoglycaemic Agents and Insulin was assessed by a study from Bangladesh [113], where 20% and 6.6% were non-adherence to Oral Hypoglycaemic Agents and Insulin respectively.
Table 3.
Medication adherence
| Authors | Year | Country | Sample size |
Measures | Practice rates | |
|---|---|---|---|---|---|---|
| Sultana et al. [35] |
2010 |
India |
218 |
Good adherence to medication |
47.7% |
|
| Malathy et al.[36] |
2011 |
India |
207 |
Regularly taking the doses of diabetes medication |
58.4% |
|
| Gopichandran et al. [37] |
2012 |
India |
200 |
Drug adherence |
79.8% |
|
| Sasi et al. [39] | 2013 | India | 546 | Good adherence to medication |
61% |
|
|
|
|
|
|
Poor adherence to medication |
39% |
|
| Arulmozhi and Mahalakshmy [40] | 2014 | India | 150 | Low adherence |
26% |
|
| Moderate adherence |
24.7% |
|||||
|
|
|
|
|
High adherence |
49.3% |
|
| Khan et al. [41] |
2014 |
India |
184 |
Good adherence with the prescribed therapy |
48.4% |
|
| Santhanakrishnan, Lakshminarayanan and Kar [42] |
2014 |
India |
135 |
Compliance to pharmacological treatment |
76.3% |
|
| Sajith et al. [44] | 2014 | India | 105 | Low adherence |
21.9% |
|
| Moderate adherence |
37.1% |
|||||
|
|
|
|
|
High adherence |
40.9% |
|
| Basu et al. [47] | 2015 | India | 385 | Good medication adherence |
74.5% |
|
|
|
|
|
|
Poor medication adherence |
25.5% |
|
| Divya and Nadig [46] | 2015 | India | 150 | Non-adherence (low) |
54.7% |
|
|
|
|
|
|
Adherence (Moderate-high) |
45.3% |
|
| Rajasekharan et al. [48] | 2015 | India | 290 | Adherence to OHA's on all days of the week |
60.5% |
|
|
|
|
|
|
Adherence to insulin injections on all days of the week |
66.9% |
|
| Das et al. [49] |
2016 |
India |
232 |
Medicines taken regularly |
90.5% |
|
| Karthikeyan, Madhusudhan and Selvamuthukumaran [50] | 2016 | India | 345 | Low adherence |
95.6% |
|
| Moderate adherence |
4.3% |
|||||
|
|
|
|
|
High adherence |
0 |
|
| Pathania et al. [51] | 2016 | India | 48 | Low adherence |
56.2% |
|
| Moderate adherence |
29.2% |
|||||
|
|
|
|
|
High adherence |
14.6% |
|
| Dinesh, Kulkarni and Gangadhar [52] |
2016 |
India |
400 |
Taking drugs every day and regularly |
48% |
|
| Debnath et al. [53] | 2017 | India | 450 | Good medication adherence |
38% |
|
|
|
|
|
|
Poor medication adherence |
62% |
|
| Kumar et al. [54] | 2017 | India | 124 | Low adherence |
43.5% |
|
| Moderate adherence |
29% |
|||||
|
|
|
|
|
High adherence |
27.4% |
|
| Samu, Amirthalingam and Mohammed [55] |
2017 |
India |
86 |
Low medication adherence |
4.3 (±2.3) |
|
| Sheeba, Ak and Biju [56] |
2017 |
India |
100 |
Taking regular medication |
88% |
|
| Srinath, Basavegowda and Tharuni [57] |
2017 |
India |
400 |
Good compliance for medication |
92.5% |
|
| Venkatesan, Dongre and Ganapathy [61] |
2018 |
India |
328 |
Low adherent for medication |
45.4% |
|
| Acharya et al. [63] | 2019 | India | 200 | Low adherence |
33% |
|
| Moderate adherence |
34.5% |
|||||
|
|
|
|
|
High adherence |
32.5% |
|
| Jasmine and Iyer [62] | 2019 | India | 77 | Good compliance to treatment |
64.9% |
|
|
|
|
|
|
Poor compliance to treatment |
35.1% |
|
| Chandrika et al. [69] |
2020 |
India |
208 |
Good drug adherence |
56.3% |
|
| Kowsalya et al. [70] | 2020 | India | 60 | Low adherence |
2% |
|
| Moderate adherence |
20% |
|||||
|
|
|
|
|
High adherence |
78% |
|
| Palathingal et al. [71] | 2020 | India | 200 | Low adherence |
71.5% |
|
| Moderate adherence |
24% |
|||||
|
|
|
|
|
High adherence |
4.5% |
|
| Achappa [74] | 2020 | India | 70 | Good adherence to medication |
80% |
|
|
|
|
|
|
Poor adherence to medication |
20% |
|
| Karthik et al. [75] | 2020 | India | 250 | Low adherence |
29.6% |
|
|
|
|
|
|
High adherence |
70.4% |
|
| Rana et al. [77] |
2021 |
India |
200 |
Mean (SD) score adhering the medication |
0.3 (±0.7) |
|
| Burman et al. [78] |
2021 |
India |
367 |
Taking medication daily |
93% |
|
| Durai et al. [79] |
2021 |
India |
390 |
Adherence to medication |
57.2% |
|
| Mishra et al. [80] | 2021 | India | 277 | Good adherence |
44% |
|
|
|
|
|
|
Poor adherence |
56% |
|
| Singh et al. [81] | 2021 | India | 350 | Low adherence |
26% |
|
| Moderate adherence |
42% |
|||||
|
|
|
|
|
High adherence |
32% |
|
| Aravindakshan et al. [82] | 2021 | India | 218 | Low adherence |
10.5% |
|
| Moderate adherence |
29.4% |
|||||
|
|
|
|
|
High adherence |
60.1% |
|
| Zuberi, Syed and Bhatti [83] | 2011 | Pakistan | 286 | Taking dose on time |
84% |
|
|
|
|
|
|
Taking recommended dose of medication |
83% |
|
| Ahmed et al. [84] |
2015 |
Pakistan |
139 |
Taking medication on time |
7.9% |
|
| Iqbal et al. [87] | 2017 | Pakistan | 300 | Low adherence |
7.3% |
|
| Moderate adherence |
37% |
|||||
|
|
|
|
|
High adherence |
55.6% |
|
| Nazirl et al. [88] | 2017 | Pakistan | 392 | Low adherence |
71.9% |
|
| Moderate adherence |
24.7% |
|||||
|
|
|
|
|
High adherence |
3.32% |
|
| Rana et al. [89] | 2017 | Pakistan | 145 | Low adherence |
19.3%, |
|
| Moderate adherence |
43.4% |
|||||
|
|
|
|
|
High adherence |
37.2% |
|
| Hussain, Said and Khan [93] |
2020 |
Pakistan |
524 |
Mean (SD) score adhering the medication |
3.1 (±0.5) |
|
| Siddique et al. [94] |
2020 |
Pakistan |
154 |
Taking medication daily |
74% |
|
| Malik et al. [19] |
2020 |
Pakistan |
363 |
Daily medication use |
66.4% |
|
| Ishaq et al. [96] |
2021 |
Pakistan |
300 |
Low adherence |
7.3% |
|
| Moderate adherence |
37% |
|||||
| High adherence |
55.6% |
|||||
| Shrestha et al. [97] |
2013 |
Nepal |
100 |
Non-adherence to medication |
38% |
|
| Bhandari and Kim [20] |
2016 |
Nepal |
230 |
Mean (SD) number of days in a week adhering the medication |
6.8(±1.1) |
|
| Shrestha et al. [101] |
2017 |
Nepal |
183 |
Adherence to medication |
77% |
|
| Sapkota et al. [104] | 2018 | Nepal | 200 | Forgot to take diabetes tablet/insulin in the last year |
||
| a. <5 times |
76% |
|||||
|
|
|
|
|
b. ≥5 times |
24% |
|
| Thapa [105] | 2018 | Nepal | 141 | Adherence to OHA on 7 d of a week |
86.5% |
|
|
|
|
|
|
Adherence to insulin on 7 d of the week |
78% |
|
| Pokhrel et al. [106] |
2019 |
Nepal |
480 |
Low adherence |
36.6% |
|
| High adherence |
63.4% |
|||||
| Bhattarai et al. [107] |
2019 |
Nepal |
214 |
Adherence to medication |
44.9% |
|
| Non-adherence to medication |
55.1% |
|||||
| Sharma et al. [108] |
2021 |
Nepal |
296 |
Adherence to medication |
86.8% |
|
| Shrestha et al. [109] |
2021 |
Nepal |
354 |
Adherence to medication |
92% |
|
| Kandel et al. [110] | 2022 | Nepal | 411 | Adherence to OHA |
98.2% |
|
|
|
|
|
|
Adherence to insulin |
100% |
|
| Saleh et al. [113] |
2014 |
Bangladesh |
500 |
Non-adherence to OHA |
20% |
|
| Non-adherence to insulin |
6.6% |
|||||
| Ahmed et al. [114] |
2017 |
Bangladesh |
122 |
Taking medication regularly as prescribed |
43% |
|
| Taking medication irregularly |
57% |
|||||
| Mannan et al. [21] |
2021 |
Bangladesh |
2061 |
Low adherence |
46.3% |
|
| Medium- to-high adherence |
53.7% |
|||||
| Rathish et al. [121] | 2019 | Sri-Lanka | 200 |
Low adherence |
7% |
|
| Moderate adherence |
70% |
|||||
| High adherence | 23% | |||||
SD – standard deviation, OHA – oral hypoglycaemic agent, d – day
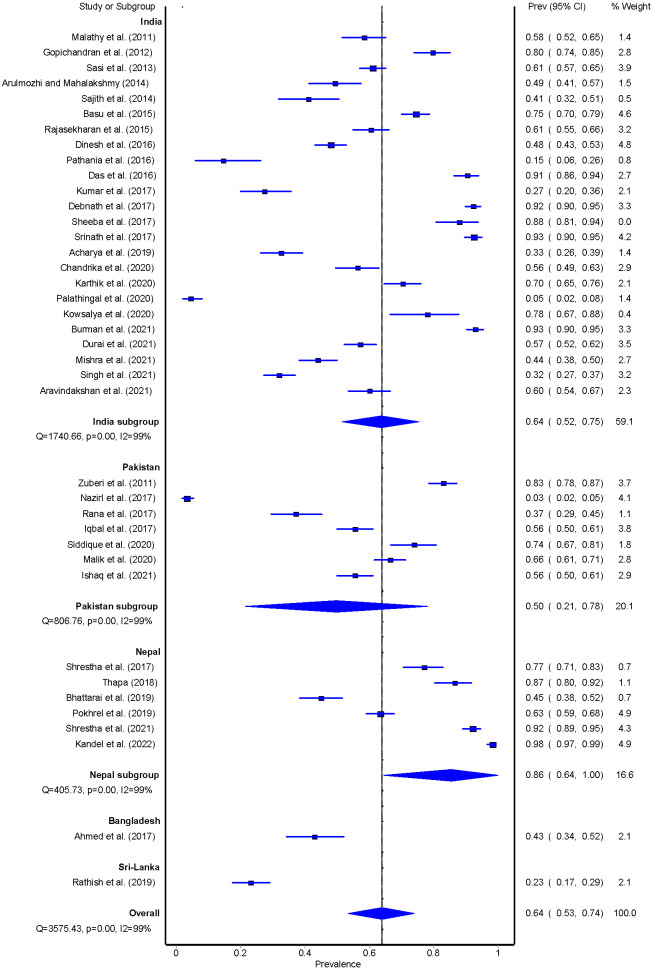
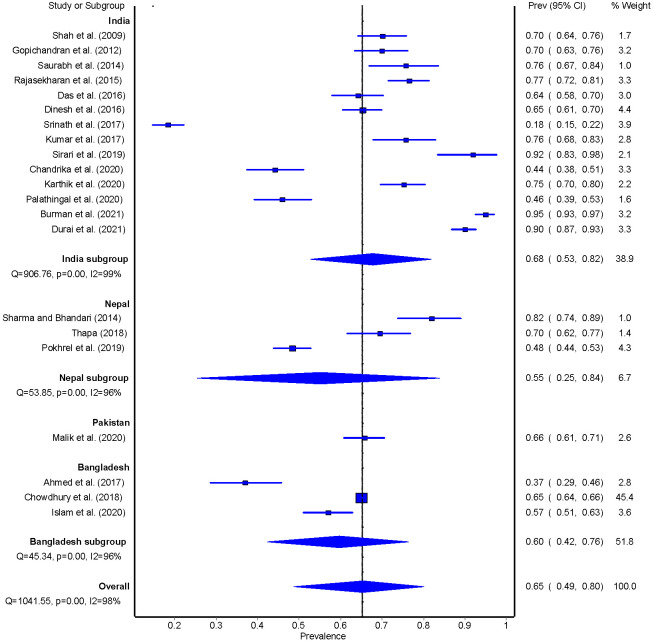
The pooled prevalence of adherence to medication use was 64% (95% CI = 53-74) and ranged between 3% and 98%. Studies conducted in Nepal reported a higher prevalence of adherence to medication use (n = 6; 86%, 95% CI = 64-100), followed by India (n = 19; 64%, 95% CI = 52-75), Pakistan (n = 6; 50%, 95% CI = 21-78), Bangladesh (n = 1; 43%, 95% CI = 34-52) and Sri-Lanka (n = 1; 23%, 95% CI = 17-29) (Figure 3).
Figure 3.
Pooled estimate of medication intake among people with T2DM.
Dietary habits
56 studies explored the study participants’ dietary intake (Table 4) using a range of dietary measurement tools. Summary of the Diabetes Self-Care Activities measure (n = 15) and the Diabetes Self-Management Questionnaire (n = 7) were the most used self-care tools in assessing the dietary practice of the study participants. Some studies reported the mean number of days in a week participant’s adhering to a healthy diet ranging from 4.32 to 5.42 days [20,45,47]. In addition, the reported mean score for dietary control (limiting sweets and carbohydrate-rich foods, consuming recommended diet) varied from 3.9 to 6.6 (scale range of 0-10, where 10 represents the optimal practice of self-care) [73,76,86,95]. Non-adherence to healthy dietary habits was reported by four studies whose values range from 41% to 88% [100,107,112,113].
Table 4.
Dietary habits
| Authors | Year | Country | Sample size | Measure | Practice rates |
|---|---|---|---|---|---|
| Shah, Kamdar and Shah [34] |
2009 |
India |
238 |
Including fruits in diet regularly |
54.2% |
| Taking green leafy vegetables in diet |
31.9% |
||||
| Malathy et al.[36] |
2011 |
India |
207 |
Following a controlled and planned diet (self-reported) |
50% |
| Gopichandran et al. [37] |
2012 |
India |
200 |
Having good dietary behaviour |
29% |
| Patel et al. [38] |
2012 |
India |
399 |
Following the recommended diabetic diet |
73% |
| Sasi et al. [39] |
2013 |
India |
546 |
Following the diabetic meal plans |
41% |
| Arulmozhi and Mahalakshmy [40] |
2014 |
India |
150 |
Consumed recommended diet for at least 4 d/week |
67.3% |
| Santhanakrishnan, Lakshminarayanan and Kar [42] |
2014 | India | 135 | Reduced the quantity of food intake |
77% |
|
|
|
|
Increased frequency of food intake |
50.3% |
|
| Saurabh et al. [43] |
2014 |
India |
103 |
Following the diet-control |
58.3% |
| Sajith et al. [44] |
2014 |
India |
105 |
Dietary adherence |
3.8% |
| Abraham et al. [45] | 2015 | India | 60 | Mean number of days in a week following general diet* |
5.3 |
|
|
|
|
|
Mean number of days in a week following specific diet† |
5.4 |
| Basu et al. [47] |
2015 |
India |
385 |
Mean (SD) number of days in a week following a healthy eating plan |
4.8 (±1.4) |
| Rajasekharan et al. [48] | 2015 | India | 290 | Following healthy eating plan on all days of the week |
45.9% |
|
|
|
|
|
Incorporating fruits/vegetables in the diets on all days of the week |
26.2% |
| Das et al. [49] |
2016 |
India |
232 |
Following the planned and the controlled diet |
76.3% |
| Dinesh, Kulkarni and Gangadhar [52] |
2016 |
India |
400 |
Having a good dietary behaviour |
24% |
| Sheeba, Ak and Biju [56] |
2017 |
India |
100 |
Following the proper diet |
72% |
| Srinath, Basavegowda and Tharuni [57] | 2017 | India | 400 | Compliant to diabetic diet as advised by the doctor |
72.0% |
| Had vegetables on all seven days in the last week |
96.2% |
||||
|
|
|
|
|
Consuming fruits on all seven days in the last week |
5.5% |
| Pati et al. [59] |
2018 |
India |
321 |
Following the strict diabetic diet control |
45% |
| Ravi, Kumar and Gopichandran [60] | 2018 | India | 200 | Median (IQR) number of days following healthy eating plan in the past week |
6 (2-6) |
|
|
|
|
|
Median (IQR) number of days in the past week taking five or more servings of fruits/vegetables |
0 (0) |
| Aravind, Joy and Rakesh [64] |
2019 |
India |
68 |
Following good diet |
45.6% |
| Jasmine and Iyer [62] |
2019 |
India |
77 |
Good diabetic diet practice |
44.9% |
| Sirari et al. [67] | 2019 | India | 60 | Compliant for not eating high-fat foods |
93.5% |
| Compliant with prescribed eating plan |
51.6% |
||||
|
|
|
|
|
Compliant with eating 5 or more servings of fruits and vegetables |
59.7% |
| Bashir et al. [68] |
2020 |
India |
203 |
Mean (SD) score of consumption of healthiest diet |
1.0 (±0.2) |
| Mean (SD) score of consumption of least healthy diet |
2.6 (±0.7) |
||||
| Chandrika et al. [69] |
2020 |
India |
208 |
Good dietary behaviour |
29.8% |
| Patnaik et al. [72] | 2020 | India | 100 | Follow instructions provided to avoid certain foods |
77% |
| Follow the recommended amount of diet |
67% |
||||
|
|
|
|
|
Taking sweets |
38% |
| Shrivastva et al. [73] |
2020 |
India |
166 |
Mean (SD) score for dietary control: |
6.6 (±1.9) |
| Karthik et al. [75] |
2020 |
India |
250 |
Following satisfactory level of diet: |
35.2% |
| Kumar et al. [76] |
2021 |
India |
105 |
Mean (SD) score of dietary control‡ |
5.7 (±1.5) |
| Rana et al. [77] |
2021 |
India |
200 |
Mean (SD) score adhering the diet: |
1.1 (±0.8) |
| Burman et al. [78] |
2021 |
India |
367 |
Consumption of satisfactory level of fruits and vegetables in last 7 d: |
61.5% |
| Durai et al. [79] |
2021 |
India |
390 |
Adherent to dietary modifications: |
25.4% |
| Zuberi, Syed and Bhatti [83] |
2011 |
Pakistan |
286 |
Complying with the dietary restrictions: |
61.2% |
| Ahmed et al. [84] |
2015 |
Pakistan |
139 |
Following a proper diet plan: |
4.3% |
| Javaid et al. [85] |
2016 |
Pakistan |
120 |
Good dietary practice: |
71.7% |
| Bukhsh et al. [86] |
2017 |
Pakistan |
130 |
Mean (SD) value of dietary control: |
4.8 (±2.8) |
| Bukhsh et al. [90] |
2018 |
Pakistan |
218 |
Median (IQR) score for dietary control: |
4.17 (2.5– 6.9) |
| Farooq et al. [91] | 2018 | Pakistan | 180 | Strictly following a recommended dietary plan: |
36.1% |
| Changing diet following diabetes diagnosis: |
82.2% |
||||
| Taking three meals a day: |
55.6% |
||||
|
|
|
|
|
Eating same meal as their family: |
79.4% |
| Siddique et al. [94] |
2020 |
Pakistan |
154 |
Following the dietary plan daily: |
50% |
| Malik et al. [19] |
2020 |
Pakistan |
363 |
Following well-balanced and planned diet: |
68.9% |
| Sayeed et al. [95] |
2020 |
Pakistan |
317 |
Mean (SD) score for dietary control: |
3.87 (±1.04) |
| Parajuli et al. [98] |
2014 | Nepal |
385 |
Dietary advice: |
30.0 (±16.3) |
| a. Non-adherence |
87.5% |
||||
| b. Poor adherence |
12.5% |
||||
|
|
c. Good adherence |
0% |
|||
| Sharma and Bhandari [99] | 2014 | Nepal | 100 | Food intake per day: |
|
| a. Two times |
20% |
||||
| b. Three times |
42% |
||||
|
|
|
|
|
c. Four times |
38% |
| Bhandari and Kim [20] |
2016 |
Nepal |
230 |
Mean (SD) number of days in a week adhering the diet: |
4.3(±1.4) |
| Ghimire [100] |
2017 |
Nepal |
197 |
Non-compliant with the dietary recommendation |
41% |
| Shrestha et al. [101] | 2017 | Nepal | 183 | Dietary habits: |
|
| a. Vegetarian |
12% |
||||
|
|
|
|
|
b. non-vegetarian |
88% |
| Ghimire and Devi [102] |
2018 |
Nepal |
115 |
Having good dietary management |
47% |
| Thapa [105] | 2018 | Nepal | 141 | Following recommended dietary plans |
95.7% |
| Eating fruits and vegetables for at least 5 d/week |
73.8%, |
||||
|
|
|
|
|
Consuming high fat food |
39% |
| Pokhrel et al. [106] |
2019 |
Nepal |
480 |
Adhering the recommended meal plan: |
64.6% |
| Bhattarai et al. [107] |
2019 |
Nepal |
214 |
Not following the diabetic diet: |
85.7% |
| Shrestha et al. [109] |
2021 |
Nepal |
354 |
Dietary adherence: |
38% |
| Kandel et al. [110] | 2022 | Nepal | 411 | Ate ≥5 small meals every day in last 7 d |
15.3% |
| Ate >2 bowls of vegetables every day in last 7 d |
78.3% |
||||
| Ate >1 bowl of fruits every day in last 7 d |
45.3% |
||||
| Ate fatty food or red meat at most once in last 7 d |
55.5% |
||||
|
|
|
|
|
Refused offered sweets within the past 1 mo |
70% |
| Saleh et al. [111] |
2012 |
Bangladesh |
160 |
Following dietary control: |
18% |
| Mumu et al. [112] |
2014 |
Bangladesh |
374 |
Non-adherence to recommended diet plan: |
88% |
| Saleh et al. [113] |
2014 |
Bangladesh |
500 |
Non-adherence to diet: |
44.8% |
| Chowdhury et al. [115] |
2018 |
Bangladesh |
11 917 |
Taking food timely: |
69% |
| Have habit of extra salt intake: |
69% |
||||
| Majid et al. [117] | 2019 | Bangladesh | 420 |
A. Carbohydrate intake:
|
259.2 (±57.2)
|
| a. low |
5.7% |
||||
| b. ideal |
36.2% |
||||
| c. high |
58.1% |
||||
|
B. Protein intake:
|
87.2 (±19.1)
|
||||
| a. low |
14.3% |
||||
| b. ideal |
55.2% |
||||
| c. high |
30.5% |
||||
|
C. Fat intake:
|
65.1 (±12.2)
|
||||
| a. low |
1.9% |
||||
| b. ideal |
42.9% |
||||
|
|
|
|
|
c. high |
55.2% |
| MahmudulHasan et al. [119] |
2021 |
Bangladesh |
379 |
Adherence to recommended diet |
24.3% |
| Mannan et al. [21] | 2021 | Bangladesh | 2061 | Consumption of fruit and vegetables: |
|
| a. ≥3 times/d |
4.9% |
||||
| b. <3 times/d | 95.1% | ||||
SD – standard deviation, IQR – interquartile range, d – days
*General diet: Consumption of generally helpful or prescribed diet.
†Specific diet: Consumption of five or more servings of “fruits and vegetables” and avoiding fat foods.
‡Dietary control: Limiting sweets and carbohydrate rich foods, consuming recommended diet.
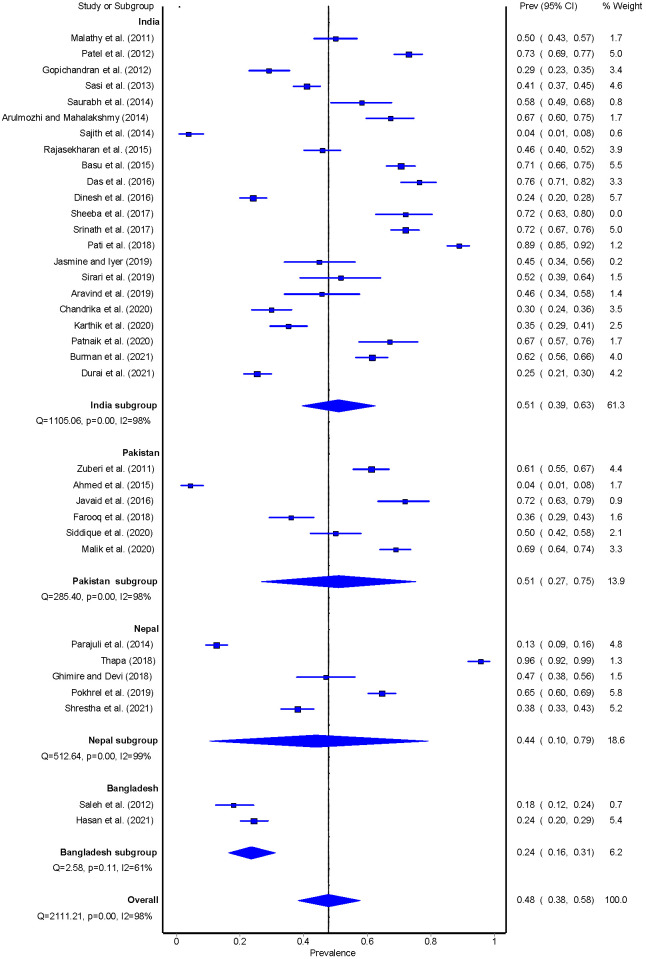
The prevalence of adherence to a healthy diet varied widely across studies, from 0% to 95.7%. The overall pooled prevalence of adherence to a healthy diet was 48% (95% CI = 38-58). In terms of country-specific analysis, the studies conducted in India (n = 22) had an adherence to a healthy diet of 51% (95% CI = 39-63), followed by Pakistan (n = 6; 51%, 95%CI: 27-75), Nepal (n = 4; 44%, 95%CI: 10-79) and Bangladesh (n = 2; 24%, 95%CI: 16-31) (Figure 4). Adherence to a healthy diet was 40% (95%CI = 29%-53%) for studies that either used a standardised tool to assess diet or studies that clearly defined what a healthy diet constitutes. Adherence to a healthy diet was 57% (95%CI = 42%-72%) for studies that did not clarify either the measure used to assess diet or studies that did not define what a healthy diet constitutes (Figure S3-S4 in the Online Supplementary Document).
Figure 4.
Pooled estimate of Dietary habit among people with T2DM.
Blood glucose monitoring
42 studies investigated blood glucose monitoring (Table 5). The Summary of Diabetes Self-Care Activities measure (n = 10) and the Diabetes Self-Management Questionnaire (n = 7) were the most used tools in assessing the blood glucose monitoring among the study participants. The mean number of days in a week practicing adequate self-monitoring of blood glucose was reported by two studies and ranged from 0.61 to 1.33 days [20,45]. Similarly, the reported mean scores for glucose management ranged from 3.92 to 6.82 (scale ranging from 0 to 10, where 10 represents the highest practice of self-care behaviour) [73,76,86,95]. However, non-adherence to blood glucose monitoring was reported by a study from Bangladesh [112,113] which ranged from 32% to 37%, while 46.61% of participants did not monitor the glucose level regularly in Nepal [107].
Table 5.
Blood glucose monitoring
| Authors | Year | Country | Sample size | Measure | Practice rates |
|---|---|---|---|---|---|
| Shah, Kamdar and Shah [34] |
2009 |
India |
238 |
Checking blood glucose monthly |
70.2% |
| Gopichandran et al. [37] |
2012 |
India |
200 |
Regular monitoring of blood glucoses (at least once in the previous 3 mo) |
70% |
| Patel et al. [38] |
2012 |
India |
399 |
Self-monitoring blood glucose |
37% |
| Saurabh et al. [43] |
2014 |
India |
103 |
Checking blood glucose at least once in 3 mo |
75.7% |
| Abraham et al. [45] |
2015 |
India |
60 |
Mean (SD) number of days in a week testing the blood glucose* |
1.3 |
| Rajasekharan et al. [48] |
2015 |
India |
290 |
Blood glucose testing at least for once in past 3 mo |
76.6% |
| Das et al. [49] |
2016 |
India |
232 |
Last checked blood glucose as advised |
64.2% |
| Dinesh, Kulkarni and Gangadhar [52] |
2016 |
India |
400 |
Checking of blood glucoses at least once in 3 mo |
65.2% |
| Checking of blood glucoses as advised by doctor |
72.7% |
||||
| Debnath et al. [53] | 2017 | India | 450 | Blood glucose check-up |
|
| Good |
48.7% |
||||
| Average |
39.1% |
||||
|
|
|
|
|
Poor |
12.2% |
| Kumar et al. [54] | 2017 | India | 124 | Blood glucose monitoring: |
|
| a. Regular (once in a month) |
75.8% |
||||
|
|
|
|
|
b. Occasional |
24.2% |
| Sheeba, Ak and Biju [56] |
2017 |
India |
100 |
Regularly monitoring blood glucose level |
63% |
| Srinath, Basavegowda and Tharuni [57] |
2017 |
India |
400 |
Blood glucose check as advised by doctor |
18.2% |
| Ravi, Kumar and Gopichandran [60] |
2018 |
India |
200 |
Median (IQR) score for blood glucose testing at least once in past 3 mo |
1 (0-1) |
| Aravind, Joy and Rakesh [64] |
2019 |
India |
68 |
Good glucose management |
52.9% |
| Jasmine and Iyer [62] | 2019 | India | 77 | Regular blood glucose check-up at Primary Health Center |
|
| a. good practice |
88.3% |
||||
|
|
|
|
|
b. poor practice |
11.7% |
| Sirari et al. [67] |
2019 |
India |
60 |
Blood glucose monitoring at least once in every 3 mo |
91.9% |
| Chandrika et al. [69] |
2020 |
India |
208 |
Blood glucose monitoring at least once within the previous 3 mo |
44.2% |
| Palathingal et al. [71] | 2020 | India | 200 | Blood glucose monitoring: |
|
| a. once in a month |
46% |
||||
| b. once in three months |
46% |
||||
| c. once in six months |
7% |
||||
|
|
|
|
|
d. once a year |
1% |
| Shrivastva et al. [73] |
2020 |
India |
166 |
Mean (SD) score for glucose management |
6.8 (±1.7) |
| Karthik et al. [75] |
2020 |
India |
250 |
Regularly monitoring/checking-up the blood glucose |
75.2% |
| Kumar et al. [76] |
2021 |
India |
105 |
Mean (SD) score for glucose management |
5.7(±1.1) |
| Rana et al. [77] |
2021 |
India |
200 |
Mean (SD) score adhering the self-monitoring of blood glucose |
0.3 (±0.8) |
| Burman et al. [78] |
2021 |
India |
367 |
Checking blood glucose level in the past 3 mo |
95% |
| Durai et al. [79] |
2021 |
India |
390 |
Blood glucose testing once in 3 mo |
90% |
| Ahmed et al. [84] |
2015 |
Pakistan |
139 |
Regularly checking blood glucose level at home |
8.6% |
| Bukhsh et al. [86] |
2017 |
Pakistan |
130 |
Mean (SD) score for glucose management |
5.3 (±2.9) |
| Bukhsh et al. [90] |
2018 |
Pakistan |
218 |
Median (IQR) score for glucose management |
4.7 (3.3–7.3) |
| Siddique et al. [94] |
2020 |
Pakistan |
154 |
Monitoring glucose twice a week |
54.5% |
| Malik et al. [19] | 2020 | Pakistan | 363 | Checking blood glucose at home as per health practitioners |
69.7% |
| Checking HbA1c levels every three months |
28.4% |
||||
|
|
|
|
|
Checking random blood glucose level at least once every three months |
65.8% |
| Sayeed et al. [95] |
2020 |
Pakistan |
317 |
Mean (SD) score for glucose management |
3.9 (±0.6) |
| Sharma and Bhandari [99] | 2014 | Nepal | 100 | Blood glucose test: |
|
| a. once a week |
2% |
||||
| b. once a month |
82% |
||||
|
|
|
|
|
c. half yearly |
16% |
| Bhandari and Kim [20] |
2016 |
Nepal |
230 |
Mean (SD) number of days in a week monitoring the blood glucose |
0.6(±0.9) |
| Ghimire and Devi [102] |
2018 |
Nepal |
115 |
Good glucose management practice |
68.2% |
| Sapkota et al. [104] | 2018 | Nepal | 200 | Checking blood glucose |
|
| a. once within a day to 1 mo |
19% |
||||
|
|
|
|
|
b. once within a month to 1 y |
81% |
| Thapa [105] |
2018 |
Nepal |
141 |
Monitoring blood glucose in every 3 mo |
69.5% |
| Pokhrel et al. [106] |
2019 |
Nepal |
480 |
Blood glucose monitoring: |
|
| a. weekly |
2.1% |
||||
| b. monthly |
48.3% |
||||
| c. triannual |
31.2% |
||||
| d. biannual |
14.6% |
||||
|
|
|
|
|
e. yearly |
3.8% |
| Bhattarai et al. [107] |
2019 |
Nepal |
214 |
Not monitoring the blood glucose level regularly |
46.6% |
| Shrestha et al. [109] |
2021 |
Nepal |
354 |
Optimal blood glucose testing |
77% |
| Kandel et al. [110] |
2022 |
Nepal |
411 |
Blood glucose testing at least 3 times in the last 7 d |
14.4% |
| Mumu et al. [112] |
2014 |
Bangladesh |
374 |
Non-adherence to blood glucose monitoring (missing the scheduled blood testing) |
32% |
| Saleh et al. [113] |
2014 |
Bangladesh |
500 |
Non-adherence to blood glucose monitoring |
37% |
| Ahmed et al. [114] | 2017 | Bangladesh | 122 | Blood glucose monitoring: |
|
| a. Daily |
8% |
||||
| b. Weekly |
15% |
||||
| c. Monthly |
37% |
||||
|
|
|
|
|
d. Never |
40% |
| Chowdhury et al. [115] | 2018 | Bangladesh | 11 917 | Blood glucose monitoring: |
|
| a. Daily |
6% |
||||
| b. Weekly |
1% |
||||
| c. Monthly |
65% |
||||
|
|
|
|
|
d. Never |
28% |
| Islam et al. [118] | 2020 | Bangladesh | 265 | Self-monitoring of blood glucose at home: |
|
| a. Weekly |
12.4% |
||||
| b. Monthly |
30.6% |
||||
| c. Every 2-3 mo or later | 57% | ||||
IQR – interquartile range, SD – standard deviation, HbA1c – glycated haemoglobin, d – days, mo – months
*Testing blood glucose: Testing of blood glucose and as recommended by health care provider
The overall pooled prevalence of blood glucose monitoring was 65% (95%CI: 49-80), ranging between 18% to 95%. A higher prevalence of adequate blood glucose monitoring among people with T2DM was found in India (n = 14; 68%, 95% CI = 53-82), followed by Pakistan (n = 1; 66%, 95% CI = 61-71), Bangladesh (n = 3; 60%, 95% CI = 42-76), and Nepal (n = 3; 55%, 95% CI: 25-84) (Figure 5). The sub-group analysis for blood glucose monitoring was assessed based on monitoring blood glucose levels at least once a month and/or at least once in three months.
Figure 5.
Pooled estimate of blood glucose monitoring among people with T2DM.
In the sub-group analysis, the pooled prevalence of monthly blood glucose monitoring was 63% (95% CI = 48-77), while it was 67% (95% CI = 53-79) for the at least once in three months interval (Figure S5-S6 in the Online Supplementary Document).
Foot care
Thirty studies investigated the practice of foot care (Table 6). The Summary of Diabetes Self-Care Activities measure tool (n = 10) was most used in assessing the study participants’ foot care. The mean number of days in a week participants practicing foot care ranged between 0.55 and 2.16 days [20,45]. A study from India among 200 people with T2DM reported that the median number of days in the past week inspecting shoes or footwear was “0” [60]. Studies from Bangladesh reported on non-adherence to foot care ranging from 43.2 to 70% [112,113].
Table 6.
Foot care
| Authors | Year | Country | Sample size | Measures | Practice rates |
|---|---|---|---|---|---|
| Shah, Kamdar and Shah [34] |
2009 |
India |
238 |
Checking the feet regularly |
56% |
| Sasi et al. [39] |
2013 |
India |
546 |
Adequate foot care |
31% |
| Inadequate foot care |
69% |
||||
| Arulmozhi and Mahalakshmy [40] | 2014 | India | 150 | Inspected foot at least 4 d/week: |
22.7% |
|
|
|
|
|
Foot care at least 4 d/week (drying between toes after wash) |
24% |
| Santhanakrishnan, Lakshminarayanan and Kar [42] |
2014 |
India |
135 |
Practicing foot care |
54% |
| Saurabh et al. [43] | 2014 | India | 103 | Daily inspection of feet or their footwear |
47.6% |
| Daily washing and drying of feet |
80.6% |
||||
| Poor practice of foot care |
44.7% |
||||
| Satisfactory practice of foot care |
35.9% |
||||
|
|
|
|
|
Good practice of foot care |
19.4% |
| Abraham et al. [45] |
2015 |
India |
60 |
Mean (SD) number of days in a week practicing foot care* |
0.6 |
| Rajasekharan et al. [48] | 2015 | India | 290 | Washing feet on all days of the week |
64.8% |
| Drying between the toes on all days of week |
70.7% |
||||
| Examining feet on all days of the week |
28.3% |
||||
|
|
|
|
|
Inspecting the inner surface of shoes on all days of the week |
13.4% |
| Das et al. [49] |
2016 |
India |
232 |
Regularly practicing the foot care |
55.6% |
| Dinesh, Kulkarni and Gangadhar [52] |
2016 |
India |
400 |
Checking the feet daily |
0.5% |
| Inspecting inside of shoes/footwear daily |
0.5% |
||||
| Debnath et al. [53] |
2017 |
India |
450 |
Practicing good foot care |
6.2% |
| Sheeba, Ak and Biju [56] |
2017 |
India |
100 |
Performing proper foot care |
79% |
| Srinath, Basavegowda and Tharuni [57] |
2017 |
India |
400 |
Checking feet daily (last week) |
24.2% |
| Ravi, Kumar and Gopichandran [60] | 2018 | India | 200 | Median number of days in the past week checking feet |
0 (IQR = 0) |
|
|
|
|
|
Median number of days in the past week inspecting inside of shoes |
0 (IQR = 0) |
| Jasmine and Iyer [62] |
2019 |
India |
77 |
Good practice of inspecting feet |
13% |
| Good practice of using footwear |
51.9% |
||||
| Sirari et al. [67] |
2019 |
India |
60 |
Inspecting shoes from inside |
66.1% |
| Performing foot care checking feet |
67.7% |
||||
| Karthik et al. [75] |
2020 |
India |
250 |
Practicing satisfactory foot care |
17.6% |
| Practicing unsatisfactory foot care |
82.4% |
||||
| Verma et al. [18] | 2021 | India | 416 | Poor practice of foot care |
20.6% |
| Satisfactory practice of foot care |
32.7% |
||||
|
|
|
|
|
Good practice of foot care |
46.7% |
| Burman et al. [78] |
2021 |
India |
367 |
Taking care of foot regularly |
54.5% |
| Durai et al. [79] |
2021 |
India |
390 |
Inspecting the foots regularly |
26.7% |
| Zuberi, Syed and Bhatti [83] |
2011 |
Pakistan |
286 |
Compliant with foot care |
82% |
| Ahmed et al. [84] |
2015 |
Pakistan |
139 |
Proper cutting of nails |
5.8% |
| Zafar et al. [92] | 2018 | Pakistan | 220 | Poor practice of foot care |
24.1% |
| Average practice of foot care |
59.1% |
||||
|
|
|
|
|
Good practice of foot care |
16.8% |
| Malik et al. [19] |
2020 |
Pakistan |
363 |
Checking the feet daily |
58.4% |
| Bhandari and Kim [20] |
2016 |
Nepal |
230 |
Mean (SD) number of days in a week practicing foot care |
2.2(±2.4) |
| Thapa [105] | 2018 | Nepal | 141 | Washing feet daily |
100% |
| Habit of inspecting feet |
92.2% |
||||
| Trim nails regularly |
100% |
||||
|
|
|
|
|
Drying the toes on all day of the week |
78% |
| Shrestha et al. [109] |
2021 |
Nepal |
354 |
Optimum foot care |
42% |
| Kandel et al. [110] |
2022 |
Nepal |
411 |
Checked feet every day in the last 7 d: |
51.1% |
| Mumu et al. [112] |
2014 |
Bangladesh |
374 |
Non-adherence to foot care (not following the recommended foot care) |
70% |
| Saleh et al. [113] |
2014 |
Bangladesh |
500 |
Non-adherence to foot care |
43.2% |
| Islam et al. [118] | 2020 | Bangladesh | 265 | Practicing the foot care (last week) | 37.4% |
SD – standard deviation
*Foot care: Checking feet and inside of shoe, and washing, and drying feet.
The overall pooled prevalence of adherence to foot care was 42% (95% CI = 30-54) and ranged between 6% and 92%. Studies conducted in Pakistan reported the highest adherence to foot care (n = 2; 72%, 95%CI = 47-94) followed by Nepal (n = 3; 52%, 95% CI = 19-84), Bangladesh (n = 1; 37%, 95% CI = 32-43) and India (n = 15; 33%, 95% CI = 21-45) (Figure 6). Sufficient adherence to foot care was 37% (95% CI = 18-57) for studies that either used a standardised tool to assess foot care or studies that clearly defined what adequate foot care constitutes. Adherence to sufficient foot care was 29% (95% CI = 8-57) for studies that did not clarify either the measure used to assess foot care or studies that did not define what sufficient foot care constitutes (Figures S7-S8 in the Online Supplementary Document).
Figure 6.
Pooled estimate of foot care among people with T2DM.
DISCUSSION
Adherence to self-care behaviour prevents T2DM-associated morbidities and mortalities [14,15]. The systematic reviews that included studies from Ethiopia [22,23], Sub-Saharan Africa [24], and LMICs [25] reported the poor practice of self-care behaviours among the people with T2DM and stressed the need for developing and implementing interventions to improve self-care behaviour. South Asians are at higher risk of developing NCDs, including type 2 diabetes [5], and the health care resources in this region are limited [128,129]. While previous systematic reviews on self-care behaviours have been conducted in various regions [11,22-25], no reviews have focused on studies conducted in South Asia. To the best of our knowledge, this is the first systematic review and meta-analysis to systematically assess and report the evidence on self-care behaviours among people with T2DM in South Asia countries. We also conducted a meta-analysis to calculate the pooled prevalence of different domains of self-care behaviour reported in 70 studies. The prevalence of T2DM self-care behaviour was highest for blood glucose monitoring, followed by medication adherence, physical activity, diet, and foot care.
Physical activity was the most frequently measured self-care behaviour among included studies. The pooled prevalence of adherence to physical activity was 53%. This shows insufficient participation in physical activity among South Asians with T2DM. Similar findings were observed in reviews from Ethiopia conducted by Dagnew et al. [23] (pooled prevalence = 48.29%) and Katema et al. [22] (pooled prevalence = 49%). In contrast, lower adherence to physical activity was reported in Canadian studies (21%) by Thiel et al. [130] and LMICs (41.2%) by Morge et al. [25]. This discrepancy might be because of the higher presence of manual labour in low-income countries compared to sedentary occupation and personal motorized transportation in high-income countries [131]. However, studies identified that time constraint, unwillingness, poor awareness level, comorbid conditions, social issues, lack of infrastructure and insufficient emphasis by physicians were the barriers to physical activity among South Asians with T2DM [59,100,120,132]. Engaging in sufficient physical activity reduces the risk of T2DM and plays a significant role in reducing the glycaemic level among people with T2DM [133,134]. This illustrates that physical activity is a key self-care behaviour for diabetes management and needs to be improved in South Asians with T2DM by designing culturally acceptable and person-centred interventions to facilitate and encourage people to adopt healthy behaviours.
The pooled prevalence of adherence to medication use in this review was 64%. This finding is consistent with the review from Sub-Saharan Africa conducted by Stephani et al. [24] that reported a mean adherence to medication use of 64% (range = 39%-88%). However, a higher prevalence of adherence to medication use (71%, range = 59%-83%) is reported by Morge et al. [25]. The lower adherence to medication use in South Asia might be the result of a poor understanding of the role of medication use in controlling blood glucose levels and the preference for traditional home remedies [135]. The practice of fasting, a cultural practice observed by people from various faiths, may impede compliance with medication intake [136,137]. Unaffordability, lack of information about prescribed medicines and their importance, ignorance and unwillingness, forgetfulness, poor drug supply from health facilities and poor doctor-patient relationship are significant factors for the lower adherence to medication use among the people with T2DM in South Asian countries [19,46,61,104,107]. Although adherence to medication use has a positive impact in controlling the glycaemic level [138], almost half of the South Asians did not adhere to their prescribed medication use and are at risk of developing acute and long-term complications, consequently leading to an increased hospitalization rate and higher medical costs [138-140]. Interventions aiming to raise awareness of the role of regular use of medication in controlling blood glucose levels are to be designed and implemented to adhere to the treatment regimen, and primary health care facilities need to be equipped with proper infrastructure required for screening of diabetes, provision of regular drug supplies and counselling services.
Less than half (48%) of the study participants adhered to healthy dietary behaviour. This finding is consistent with the reviews from Ethiopia conducted by Dagnew et al. [23] and Katema et al. [22], both reporting a pooled estimate of 50% for good dietary practice. A study by Stephani et al. [24] from Sub-Saharan Africa reported adherence to a healthy diet ranging between 33%-87%, while a study by Morge et al. [25] examining adherence in low- and middle-income countries reported 58%. A study by Coyle et al. [11] examining adherence in high-income countries reported healthy diet adherence ranging between 50% and 80.9%. As such, these studies reported a relatively higher prevalence of adherence to dietary habits than those reported in our study. The differences in study outcomes might be due to differences in foods consumed in other countries and the difference in cultural and traditional values. For example, South Asians often eat certain foods because of their cultural and traditional importance, even if they are known to be unhealthy [135]. In addition, South Asians consume high amounts of white rice, other refined grains, saturated fats, and low amounts of fibre and vegetables; these food patterns increase the risk of T2DM [141,142]. Furthermore, poor quality information on dietary modification and misconceptions on what a healthy diet constitutes might have also restricted adherence to healthy dietary behaviours among South Asians [135]. Cost constraints are also a barrier to consuming the recommended amount of fruits and vegetables among South Asians [143]. Evidence shows that the practice of fasting, a religious belief observed with Hindus (Navratri, Mahashivratri, Janmashtami, Ashtami, Ekadashi), Muslim (Ramadan), and Jain (Ekasana, Digambarupvas), has also impacted the practicing of healthy dietary habits [136]. As diet plays an important role in controlling glycaemic levels and preventing T2DM complications [144,145], there is an need for addressing dietary behaviours among people with T2DM in South Asia. This can be achieved by implementing culturally tailored and contextual interventions on healthy diets, given the importance of diet in maintaining the recommended level of blood glucose.
Regular blood glucose monitoring improves blood glucose level among people with T2DM [146] and its frequency varies from person to person depending on the patient’s needs and health care provider’s advice. We found that the pooled prevalence of blood glucose monitoring was 65%. However, studies by Morge et al. [25] (range = 13%-79%), Ketema et al. [22] (pooled prevalence = 28%), and Dagnew et al. [23] (pooled prevalence = 31.89%) found a lower prevalence of blood glucose monitoring than our study. This might be because we only considered those monitoring the blood glucose level at monthly or at least once in three months intervals as adhering to the blood glucose monitoring, while the above-mentioned studies considered daily, weekly, monthly, three-monthly, bi-annually and other intervals. Unlike other self-care behaviours, it was difficult to compare the practice of blood glucose monitoring among people with T2DM due to varied treatment goals. Also, the access to glucometers at the household level in South-Asia is minimal because of cost constraints, thus contributing to suboptimal blood glucose monitoring. The engagement of community health workers in primary health care centres can ensure comprehensive health care services are delivered and self-management of NCDs is promoted [147]. As such, community health workers can be trained to conduct regular visits to patients with T2DM and provide social prescriptions required for adopting healthy self-care behaviour.
The overall pooled prevalence of adherence to foot care was 42%. A lower prevalence of foot care than in this study was observed in the review by Morge et al. [25] which reported a median adherence to foot care of 36.5% (IQR = 13.6%-59.2%). However, the studies conducted by Dagnew et al. [23] (pooled prevalence = 63.61%) and Ketema et al. [22] (pooled prevalence = 58%) reported higher adherence to diabetic foot-care. The lower prevalence of foot care among people with T2DM in the South-Asian region might be the result of the practice of barefoot walking, use of inappropriate footwear, poor awareness of foot care and its complications, and poor counselling on foot care from service providers [148,149]. Foot problems due to T2DM can have a large economic impact which deteriorates the quality of life and ultimately results in physical impairment [150]. There is a high need for health literacy programs on foot care and its complication among people with T2DM in this region.
Strengths and limitations:
This study has several strengths. This review adhered to the PRISMA guidelines [26] and is registered in the PROSPERO website [151] (Registration number: CRD42021242930). We included cross-sectional and observational studies searching multiple databases and performed forward and backward reference checking to ensure no relevant articles were missed. In addition, this study considered and categorized the studies as those defining the study tool and/or providing a clear definition of self-care domain and those not reporting the study tool and/or not providing a clear definition of self-care domain (reporting only good or poor practice). This allowed for sub-group analyses and pooled prevalence calculation to be done separately. Moreover, this study is the first of its kind to provide comprehensive findings on the practice of self-care behaviours among people with T2DM in South Asian countries.
This study also had some limitations. The findings of the sub-group analysis, specifically among studies not defining study tools and/or not providing a clear definition of self-care domains assessed, should be used with caution. Another limitation was that only “high adherence” to medication intake was categorized as adequate. This is because moderate adherence was unevenly calculated (scoring for moderate adherence differed) among the included studies. In addition, the pooled prevalence of adequate blood glucose monitoring might be over-reported, as we included only those monitoring blood glucose every month or at least once in three months in the meta-analysis. Moreover, the country-specific findings on each domain of self-care should be interpreted with caution, as the number of studies varied between countries, with some countries reporting very low numbers. Finally, outcomes might be biased as most of the included studies only assessed self-reported self-care behaviours.
CONCLUSION
The findings of this meta-analysis suggest that the overall self-care behaviour among people with T2DM in South Asia was low. Of five self-care domains, blood glucose monitoring and medication adherence were relatively common compared to physical activity, diet, and foot care. There is a need for designing and implementing high-quality, community-based, cost-effective, and culturally-tailored interventions to improve self-care among people with T2DM in South Asia.
Additional material
Acknowledgements
We would like to thank and acknowledge Dr Rashidul Alam Mahumud at Faculty of Medicine and Health, The University of Sydney for his valuable suggestions during the process of conducting the review and drafting the manuscript.
Footnotes
Funding: This research was supported by an Australian Government Research Training Program Scholarship.
Authorship contributions: GP conceptualised the study and designed the protocol under the supervision of LR and CV. GP, LR, and CV identified and finalised the search strategies. GP independently performed the search; GP and PD contributed to screening, data extraction, and quality assessment of studies. TB conducted the meta-analysis and GP drafted the manuscript. LR, CV, UNY, TS, TB, and PD reviewed the draft manuscript, provided critical comments, and approved the final manuscript.
Disclosure of interest: The authors completed the ICMJE Unified Competing Interest Form (available upon request from the corresponding author) and declare no relevant interests.
REFERENCES
- 1.World Health Organization Diabetes 2020. Available: https://www.who.int/news-room/fact-sheets/detail/diabetes. Accessed: 7 March 2021.
- 2.International Diabetes Federation. IDF Diabetes Atlas (10th Ed.): International Diabetes Federation, Brussels, Belgium; 2021. Available: www.diabetesatlas.org. Accessed: 21 March 2021.
- 3.World Health Organization. GLOBAL REPORT ON DIABETES 2016. Available: https://apps.who.int/iris/bitstream/handle/10665/204871/9789241565257_eng.pdf?sequence=1. Accessed: 11 February 2021.
- 4.The World Bank. World Bank Country and Lending Groups 2021. Available: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups. Accessed: 21 March 2021.
- 5.Unnikrishnan R, Anjana RM, Mohan V.Diabetes in South Asians: is the phenotype different? Diabetes. 2014;63:53-5. 10.2337/db13-1592 [DOI] [PubMed] [Google Scholar]
- 6.Waisundara VY, Naofumi S. Diabetes Mellitus in South Asia In: G. AR, editor. Diabetes and Its Complications. London: IntechOpen; 2017. [Google Scholar]
- 7.Misra A, Sattar N, Tandon N, Shrivastava U, Vikram NK, Khunti K, et al. Clinical management of type 2 diabetes in south Asia. Lancet Diabetes Endocrinol. 2018;6:979-91. 10.1016/S2213-8587(18)30199-2 [DOI] [PubMed] [Google Scholar]
- 8.Sohal PS.Prevention and Management of Diabetes in South Asians. Can J Diabetes. 2008;32:206-10. 10.1016/S1499-2671(08)23011-X [DOI] [Google Scholar]
- 9.International Diabetes Federation. IDF Diabetes Atlas (9th Ed.). International Diabetes Federation, Brussels, Belgium (2019), ISBN: 978-2-930229-87-4. [Google Scholar]
- 10.AADE7 Self-Care Behaviors. Diabetes Educ. 2008;34:445-9. 10.1177/0145721708316625 [DOI] [PubMed] [Google Scholar]
- 11.Coyle ME, Francis K, Chapman Y.Self-management activities in diabetes care: a systematic review. Aust Health Rev. 2013;37:513-22. 10.1071/AH13060 [DOI] [PubMed] [Google Scholar]
- 12.Modarresi M, Gholami S, Habibi P, Ghadiri-Anari A.Relationship between Self Care Management with Glycemic Control in Type 2 Diabetic Patients. Int J Prev Med. 2020;11:127. 10.4103/ijpvm.IJPVM_207_19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.World Health Organization. Adherence to Long-term Therapies: Evidence for action 2003. Available: https://www.who.int/chp/knowledge/publications/adherence_full_report.pdf?ua=1. Accessed: 12 February 2021.
- 14.Gaede P, Vedel P, Parving HH, Pedersen O.Intensified multifactorial intervention in patients with type 2 diabetes mellitus and microalbuminuria: the Steno type 2 randomised study. Lancet. 1999;353:617-22. 10.1016/S0140-6736(98)07368-1 [DOI] [PubMed] [Google Scholar]
- 15.Macisaac RJ, Jerums G.Intensive glucose control and cardiovascular outcomes in type 2 diabetes. Heart Lung Circ. 2011;20:647-54. 10.1016/j.hlc.2010.07.013 [DOI] [PubMed] [Google Scholar]
- 16.Gray J, Millett C, Saxena S, Netuveli G, Khunti K, Majeed A.Ethnicity and quality of diabetes care in a health system with universal coverage: population-based cross-sectional survey in primary care. J Gen Intern Med. 2007;22:1317-20. 10.1007/s11606-007-0267-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.World Health Organization. Global action plan for the prevention and control of noncommunicable diseases 2013-2020 Available: https://apps.who.int/iris/bitstream/handle/10665/94384/9789241506236_eng.pdf?sequence=1. Accessed: 21 March 2022.
- 18.Verma M, Sharma N, Rashi, Arora V, Bashar MA, Nath B, et al. Diabetic Foot Care Knowledge and Practices in Rural North India: Insights for Preventive Podiatry. J Assoc Physicians India. 2021;69:30-4. [PubMed] [Google Scholar]
- 19.Malik S, Khaliq IH, Mahmood HZ, Khaqan S, Abdullah Y, Rasheed N, et al. Self-management practices among type 2 diabetes patients attending physicians and pharmacists: An exploratory study from Lahore, Pakistan. Ethiop J Health Dev. 2020;34:165-70. [Google Scholar]
- 20.Bhandari P, Kim M.Self-Care Behaviors of Nepalese Adults With Type 2 Diabetes A Mixed Methods Analysis. Nurs Res. 2016;65:202-14. 10.1097/NNR.0000000000000153 [DOI] [PubMed] [Google Scholar]
- 21.Mannan A, Hasan MM, Akter F, Rana MM, Chowdhury NA, Rawal LB, et al. Factors associated with low adherence to medication among patients with type 2 diabetes at different healthcare facilities in southern Bangladesh. Glob Health Action. 2021;14:1872895. 10.1080/16549716.2021.1872895 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ketema DB, Leshargie CT, Kibret GD, Assemie MA, Alamneh AA, Kassa GM, et al. Level of self-care practice among diabetic patients in Ethiopia: a systematic review and meta-analysis. BMC Public Health. 2020;20:309. 10.1186/s12889-020-8425-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dagnew B, Debalkie Demissie G, Abebaw Angaw D.Systematic Review and Meta-Analysis of Good Self-Care Practice among People Living with Type 2 Diabetes Mellitus in Ethiopia: A National Call to Bolster Lifestyle Changes. Evid Based Complement Alternat Med. 2021;2021:8896896. 10.1155/2021/8896896 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stephani V, Opoku D, Beran D.Self-management of diabetes in Sub-Saharan Africa: a systematic review. BMC Public Health. 2018;18:1148. 10.1186/s12889-018-6050-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mogre V, Johnson NA, Tzelepis F, Shaw JE, Paul C.A systematic review of adherence to diabetes self-care behaviours: Evidence from low- and middle-income countries. J Adv Nurs. 2019;75:3374-89. 10.1111/jan.14190 [DOI] [PubMed] [Google Scholar]
- 26.Moher D, Liberati A, Tetzlaff J, Altman DG.Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. 10.1371/journal.pmed.1000097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.United Nations Millennium Development Goals. Millennium Summit, 6-8 September 2000, New York Available: https://www.un.org/en/conferences/environment/newyork2000. Accessed: 25 December 2021.
- 28.Joanna Briggs Institute. Critical Appraisal Tools 2017. Available: https://jbi.global/critical-appraisal-tools. Accessed: 21 March 2021.
- 29.Ifeagwu SC, Yang JC, Parkes-Ratanshi R, Brayne C.Health financing for universal health coverage in Sub-Saharan Africa: a systematic review. Glob Health Res Policy. 2021;6:8. 10.1186/s41256-021-00190-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Popay J, Roberts H, Sowden A, Petticrew M, Arai L, Rodgers M, et al, editors. Guidance on the conduct of narrative synthesis in systematic Reviews. A Product from the ESRC Methods Programme. Version 12006. [Google Scholar]
- 31.Borenstein M, Higgins JP, Hedges LV, Rothstein HR.Basics of meta-analysis: I(2) is not an absolute measure of heterogeneity. Res Synth Methods. 2017;8:5-18. 10.1002/jrsm.1230 [DOI] [PubMed] [Google Scholar]
- 32.Furuya-Kanamori L, Barendregt JJ, Doi SAR.A new improved graphical and quantitative method for detecting bias in meta-analysis. Int J Evid-Based Healthc. 2018;16:195-203. 10.1097/XEB.0000000000000141 [DOI] [PubMed] [Google Scholar]
- 33.Barendregt J, Doi S. MetaXL user guide version 5.3. EpiGear International Pty Ltd. 2016. [Google Scholar]
- 34.Shah VN, Kamdar PK, Shah N.Assessing the knowledge, attitudes and practice of type 2 diabetes among patients of Saurashtra region, Gujarat. Int J Diabetes Dev Ctries. 2009;29:118-22. 10.4103/0973-3930.54288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sultana G, Kapur P, Aqil M, Alam M, Pillai K.Drug utilization of oral hypoglycemic agents in a university teaching hospital in India. J Clin Pharm Ther. 2010;35:267-77. 10.1111/j.1365-2710.2009.01080.x [DOI] [PubMed] [Google Scholar]
- 36.Malathy R, Narmadha MP, Ramesh S, Alvin JM, Dinesh BN.Effect of a diabetes counseling programme on knowledge, attitude and practice among diabetic patients in Erode district of South India. J Young Pharm. 2011;3:65-72. 10.4103/0975-1483.76422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gopichandran V, Lyndon S, Angel MK, Manayalil BP, Blessy KR, Alex RG, et al. Diabetes self-care activities: A community-based survey in urban southern India. Natl Med J India. 2012;25:14-7. [PubMed] [Google Scholar]
- 38.Patel M, Patel IM, Patel YM, Rathi SK.Factors associated with consumption of diabetic diet among type 2 diabetic subjects from Ahmedabad, Western India. J Health Popul Nutr. 2012;30:447-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sasi ST, Kodali M, Burra KC, Muppala BS, Gutta P, Bethanbhatla MK.Self Care Activities, Diabetic Distress and other Factors which Affected the Glycaemic Control in a Tertiary Care Teaching Hospital in South India. J Clin Diagn Res. 2013;7:857-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Arulmozhi S, Mahalakshmy T.Self care and medication adherence among type 2 diabetics in Puducherry, Southern India: A hospital based study. J Clin Diagn Res. 2014;8:UC01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Khan GH, Aqil M, Pillai KK, Ahmad MA, Kapur P, Ain MR, et al. Therapeutic adherence: A prospective drug utilization study of oral hypoglycemic in patients with type 2 diabetes mellitus. Asian Pac J Trop Dis. 2014;4:S347-52. 10.1016/S2222-1808(14)60469-2 [DOI] [Google Scholar]
- 42.Santhanakrishnan I, Lakshminarayanan S, Kar SS.Factors affecting compliance to management of diabetes in Urban Health Center of a tertiary care teaching hospital of south India. J Nat Sci Biol Med. 2014;5:365-8. 10.4103/0976-9668.136186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Saurabh S, Sarkar S, Selvaraj K, Kar S, Kumar S, Roy G.Effectiveness of foot care education among people with type 2 diabetes in rural Puducherry, India. Indian J Endocrinol Metab. 2014;18:106-10. 10.4103/2230-8210.126587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sajith M, Pankaj M, Pawar A, Modi A, Sumariya R, editors. Medication Adherence To Antidiabetic Therapy In Patients With Type 2 Diabetes Mellitus.2014. [Google Scholar]
- 45.Abraham AM, Sudhir PM, Philip M, Bantwal G.Illness perceptions and perceived barriers to self-care in patients with type 2 diabetes mellitus: an exploratory study from India. Int J Diabetes Dev Ctries. 2015;35:137-44. 10.1007/s13410-014-0266-z [DOI] [Google Scholar]
- 46.Divya S, Nadig P.Factors contributing to non-adherence to medication among type 2 diabetes mellitus in patients attending tertiary care hospital in South India. Asian J Pharm Clin Res. 2015;8:274-6. [Google Scholar]
- 47.Basu S, Khobragade M, Kumar A, Raut DK.Medical adherence and its predictors in Diabetes Mellitus patients attending government hospitals in the Indian Capital, Delhi, 2013: a cross sectional study. Int J Diabetes Dev Ctries. 2015;35:S95-101. 10.1007/s13410-014-0232-9 [DOI] [Google Scholar]
- 48.Rajasekharan D, Kulkarni V, Unnikrishnan B, Kumar N, Holla R, Thapar R.Self-care activities among patients with diabetes attending a tertiary care hospital in Mangalore Karnataka, India. Ann Med Health Sci Res. 2015;5:59-64. 10.4103/2141-9248.149791 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Das T, Wallang B, Semwal P, Basu S, Padhi TR, Ali MH.Changing Clinical Presentation, Current Knowledge-Attitude-Practice, and Current Vision Related Quality of Life in Self-Reported Type 2 Diabetes Patients with Retinopathy in Eastern India: The LVPEI Eye and Diabetes Study. J Ophthalmol. 2016;2016:3423814. 10.1155/2016/3423814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Karthikeyan V, Madhusudhan S, Selvamuthukumaran S.Intervention to improve adherence to type 2 diabetes mellitus subjects in rural teaching hospital. Pharm Lett. 2016;8:26-32. [Google Scholar]
- 51.Pathania M, Dutt HK, Gogoi JB, Rathaur V, Singh G, Singh P.Study the impact of diabetes camps on adherence to medication and glycaemic control in Uttarakhand. J Clin Diagn Res. 2016;10:OC22-6. 10.7860/JCDR/2016/16053.7271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dinesh PV, Kulkarni AG, Gangadhar NK.Knowledge and self-care practices regarding diabetes among patients with Type 2 diabetes in Rural Sullia, Karnataka: A community-based, cross-sectional study. J Family Med Prim Care. 2016;5:847-52. 10.4103/2249-4863.201176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Debnath P, Natasha K, Ali L, Bhaduri T, Roy TK, Bera S, et al. Ayurpharmacoepidemiology Perspective: Health Literacy (Knowledge and Practice) Among Older Diabetes Patients Visiting Ayurveda Teaching Hospitals in India. J Evid Based Complementary Altern Med. 2017;22:242-50. 10.1177/2156587216643641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kumar N, Unnikrishnan B, Thapar R, Mithra P, Kulkarni V, Holla R, et al. Distress and its effect on adherence to antidiabetic medications among Type 2 diabetes patients in coastal South India. J Nat Sci Biol Med. 2017;8:216-20. 10.4103/0976-9668.210008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Samu AM, Amirthalingam PS, Mohammed OS.Assessment of patient medication adherence among the type 2 diabetes mellitus population with peripheral diabetic neuropathy in South India. J Taibah Univ Med Sci. 2017;12:164-8. 10.1016/j.jtumed.2016.12.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sheeba S, Ak S, Biju V.Knowledge and self-care practices among diabetics. Asian J Pharm Clin Res. 2017;10:234-7. 10.22159/ajpcr.2017.v10i12.21072 [DOI] [Google Scholar]
- 57.Srinath KM, Basavegowda M, Tharuni NS.Diabetic self care practices in rural Mysuru, Southern Karnataka, India - A need for Diabetes Self Management Educational (DSME) program. Diabetes Metab Syndr. 2017;11 Suppl 1:S181-S186. 10.1016/j.dsx.2016.12.029 [DOI] [PubMed] [Google Scholar]
- 58.Britto A, Kumar S, Hazari A, Jadhav R, Babu AS, Bhat V, et al. Physical activity measurement using an accelerometer among indians with type 2 diabetes mellitus: A cross-sectional study. Crit Rev Phys Rehabil Med. 2018;30:141-50. 10.1615/CritRevPhysRehabilMed.2018025969 [DOI] [Google Scholar]
- 59.Pati S, Lobo E, Pati S, Desaraju S, Mahapatra P.Type 2 diabetes and physical activity: Barriers and enablers to diabetes control in Eastern India. Indian J Endocrinol Metab. 2018;22:S43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ravi S, Kumar S, Gopichandran V.Do supportive family behaviors promote diabetes self-management in resource limited urban settings? A cross sectional study. BMC Public Health. 2018;18:826. 10.1186/s12889-018-5766-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Venkatesan M, Dongre AR, Ganapathy K.A Community-Based Study on Diabetes Medication Nonadherence and its Risk Factors in Rural Tamil Nadu. Indian J Community Med. 2018;43:72-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jasmine A, Iyer RH.Knowledge, attitude, practice and barriers for self-care among type 2 diabetes mellitus patients in rural tamil nadu. Indian J Public Health Res Dev. 2019;10:419-24. 10.5958/0976-5506.2019.02463.X [DOI] [Google Scholar]
- 63.Acharya AS, Gupta E, Prakash A, Singhal N.Self-reported Adherence to Medication among Patients with Type II Diabetes Mellitus attending a Tertiary Care Hospital of Delhi. J Assoc Physicians India. 2019;67:26-9. [PubMed] [Google Scholar]
- 64.Aravind MS, Joy TM, Rakesh PS.Self care practices among people with type II diabetes mellitus in an urban area of Ernakulam, Kerala. Indian J Public Health Res Dev. 2019;10:1-5. 10.5958/0976-5506.2019.01843.6 [DOI] [Google Scholar]
- 65.Banerjee G, Kumar R, Basu D, Chakraborty SN, Hansda K, Munshi BD.Physical activity pattern and its impact on glycemic control among patients with type-2 diabetes mellitus attending an integrated diabetes and gestational diabetes clinic of eastern India. Natl J Physiol Pharm Pharmacol. 2019;10:177-83. 10.5455/njppp.2020.10.1238622122019 [DOI] [Google Scholar]
- 66.Raj JP, Selvaraj DR, Thomas JC.Dietary Calcium Intake and Physical Activity Levels among Urban South Indian Type 2 Diabetic Patients. Topics Clin Nutr. 2019;34:39-46. 10.1097/TIN.0000000000000166 [DOI] [Google Scholar]
- 67.Sirari T, Patro B, Datta P, Lakshmi PVM.Levels of compliance of self-care practices of diabetes mellitus type 2 patients: a study from a tertiary care hospital of North India. Int J Diabetes Dev Ctries. 2019;39:193-200. 10.1007/s13410-018-0623-4 [DOI] [Google Scholar]
- 68.Bashir A, Pandey K, Azharuddin M, Kumari A, Rashid I, Siddiqui NA, et al. Association between diet quality scores, adiposity, glycemic status and nutritional biomarkers among Indian population with type 2 diabetes mellitus: A cross-sectional study. Clin Epidemiol Glob Health. 2020;8:7. 10.1016/j.cegh.2019.04.007 [DOI] [Google Scholar]
- 69.Chandrika K, Das B, Syed S, Challa S.Diabetes self-care activities: A community-based survey in an urban slum in Hyderabad, India. Indian J Community Med. 2020;45:307-10. 10.4103/ijcm.IJCM_236_19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kowsalya P.Karthika, Krishna Kumar R, Joyce S, Thivya N. A study to assess the compliance to medication among type 2 diabetic patients at selected rural community in kanchipuram district, tamil nadu. Med-Leg Update. 2020;20:128-31. [Google Scholar]
- 71.Palathingal JT, Tom R, Naresh Babu V, Chacko SE, Kumar AS, Saravanan M, et al. Assessment of medication adherence in rural population with type 2 diabetes: Study in a tertiary care hospital In South India. Int J Biomed Sci. 2020;16:21-9. [Google Scholar]
- 72.Patnaik L, Muduli AK, Sahoo AK, Beura S, Panigrahi SK.Dietary Habits of Patients with Type 2 Diabetes Mellitus-An Exploratory Study in a Tertiary Care Hospital of Eastern India. J Clin Diagn Res. 2020;14:4. 10.7860/JCDR/2020/37234.13511 [DOI] [Google Scholar]
- 73.Shrivastva A, Phadnis S, Rao NK, Gore M.A study on knowledge and self-care practices about Diabetes Mellitus among patients with type 2 Diabetes Mellitus attending selected tertiary healthcare facilities in coastal Karnataka. Clin Epidemiol Glob Health. 2020;8:689-92. 10.1016/j.cegh.2020.01.003 [DOI] [Google Scholar]
- 74.Achappa S.Adherence to Medication among the Patients with Type 2 Diabetes Mellitus in a Rural Area: A Community based Study. Indian J Public Health Res Dev. 2020;11. [Google Scholar]
- 75.Karthik RC, Radhakrishnan A, Vikram A, Arumugam B, Jagadeesh S.Self-care practices among type II diabetics in rural area of Kancheepuram district, Tamil Nadu. J Family Med Prim Care. 2020;9:2912-8. 10.4103/jfmpc.jfmpc_356_20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kumar M, Paul B, Dasgupta A, Bandyopadhyay L, Roy S, Bandyopadhyay S.Diabetic peripheral neuropathy and its association with diabetes self-care: A clinic-based study in an urban health centre, kolkata. J Clin Diagn Res. 2021;15:LC13-6. 10.7860/JCDR/2021/45807.14557 [DOI] [Google Scholar]
- 77.Rana S, Kumar GL, Seema N, Nandinee D.Profile of diabetes patients’ chronic illness care in India and its role in their adherence. Diabetes Metab Syndr. 2021;15:303-8. 10.1016/j.dsx.2021.01.001 [DOI] [PubMed] [Google Scholar]
- 78.Burman J, Bhattacharya A, Chattopdhyay A, Dey I, Sembiah S, Negi R.Self-care practice and its predictors amongst Type-2 Diabetes Mellitus patients in the outpatient department of a tertiary hospital of Kolkata, Eastern India - A cross-sectional study. J Family Med Prim Care. 2021;10:1377-82. 10.4103/jfmpc.jfmpc_2070_20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Durai V, Samya V, Akila G, Shriraam V, Jasmine A, Muthuthandavan A, et al. Self-care practices and factors influencing self-care among type 2 diabetes mellitus patients in a rural health center in South India. J Edu Health Promotion. 2021;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mishra R, Sharma SK, Verma R, Kangra P, Dahiya P, Kumari P, et al. Medication adherence and quality of life among type-2 diabetes mellitus patients in India. World J Diabetes. 2021;12:1740-9. 10.4239/wjd.v12.i10.1740 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Singh R, Kumari P, Prashar A, Sardana O, Singh V.Assessment of medication adherence among type-2 diabetes mellitus in a tertiary care hospital of North India. Environ Sci Pollut Res Int. 2022;29:24951-5. 10.1007/s11356-021-17434-1 [DOI] [PubMed] [Google Scholar]
- 82.Aravindakshan R, Abraham SB, Aiyappan R.Medication Adherence to Oral Hypoglycemic Drugs among Individuals with Type 2 Diabetes Mellitus - A Community Study. Indian J Community Med. 2021;46:503-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zuberi SI, Syed EU, Bhatti JA.Association of depression with treatment outcomes in Type 2 Diabetes Mellitus: a cross-sectional study from Karachi, Pakistan. BMC Psychiatry. 2011;11:27. 10.1186/1471-244X-11-27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ahmed MU, Seriwala HM, Danish SH, Khan AM, Hussain M, Husain M, et al. Knowledge, Attitude, and Self Care Practices Amongsts Patients WithType 2 Diabetes in Pakistan. Glob J Health Sci. 2015;8:1-8. 10.5539/gjhs.v8n7p1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Javaid RA, Akhtar MS, Ali K, Khan MM, Bucha S, Kamboyo SH, et al. Physical activity, dietary practices and body mass indices relationship in type II diabetics patients. Pak J Med Health Sci. 2016;10:92-7. [Google Scholar]
- 86.Bukhsh A, Lee SWH, Pusparajah P, Schmitt A, Khan TM.Psychometric properties of the Diabetes Self-Management Questionnaire (DSMQ) [in Urdu]. Health Qual Life Outcomes. 2017;15:200. 10.1186/s12955-017-0776-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Iqbal Q, Bashir S, Iqbal J, Iftikhar S, Godman B.Assessment of medication adherence among type 2 diabetic patients in Quetta city, Pakistan. Postgrad Med. 2017;129:637-43. 10.1080/00325481.2017.1328251 [DOI] [PubMed] [Google Scholar]
- 88.Nazirl SR, Hassali MA, Saleem F, Bashir S, Aljadhey H.Does adherence to the therapeutic regimen associate with health related quality of life: Findings from an observational study of type 2 diabetes mellitus patients in Pakistan. Pak J Pharm Sci. 2017;30:2159-65. [PubMed] [Google Scholar]
- 89.Rana B, Bukhsh A, Khan TM, Sarwar A, Omer MO, Jamshed SQ.Evaluation of therapeutic effectiveness of prescribed medications in patients with type 2 diabetes mellitus: Findings from a tertiary care hospital, Lahore, Pakistan. J Pharm Bioallied Sci. 2017;9:121-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Bukhsh A, Khan TM, Nawaz MS, Ahmed HS, Chan KG, Lee LH, et al. Association of diabetes-related self-care activities with glycemic control of patients with type 2 diabetes in Pakistan. Patient Prefer Adherence. 2018;12:2377-85. 10.2147/PPA.S177314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Farooq MU, Mushtaq F, Naeem Z, Iqbal S, Naseem S, Ishtiaq O.Dietary habits and practices of type-2 diabetic patients in a tertiary care centre of Islamabad, Pakistan. J Pak Med Assoc. 2018;68:825-30. [PubMed] [Google Scholar]
- 92.Zafar S, Rashid A, Aleem M, Bashir A, Tariq R, Akram E.Knowledge and practices of foot care in type diabetic patients visiting OPD of a tertiary care hospital. Pak J Med Health Sci. 2018;12:1143-6. [Google Scholar]
- 93.Hussain N, Said ASA, Khan Z.Influence of Health Literacy on Medication Adherence Among Elderly Females With Type 2 Diabetes in Pakistan. Int Q Community Health Educ. 2020;41:35-44. 10.1177/0272684X19896724 [DOI] [PubMed] [Google Scholar]
- 94.Siddique K, Malik R, Usman A, Ishfaq K, Nadeem MS, Qadir M, et al. Self-care behaviors and glycemic control among older Type 2 diabetes mellitus patients in low-income families in Southern Punjab, Pakistan. J Hum Behav Soc Environ. 2022;1:67-76. [Google Scholar]
- 95.Sayeed KA, Qayyum A, Jamshed F, Gill U, Usama SM, Asghar K, et al. Impact of Diabetes-related Self-management on Glycemic Control in Type II Diabetes Mellitus. Cureus. 2020;12:e7845-e. 10.7759/cureus.7845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Ishaq R, Haider S, Saleem F, Bashir S, Tareen AM, Mengal MA, et al. Diabetes-related Knowledge, Medication Adherence, and Health-related Quality of Life: A Correlation Analysis. Altern Ther Health Med. 2021;27 S1:46-53. [PubMed] [Google Scholar]
- 97.Shrestha SS, Shakya R, Karmacharya BM, Thapa P.Medication adherence to oral hypoglycemic agents among type II diabetic patients and their clinical outcomes with special reference to fasting blood glucose and glycosylated hemoglobin levels. Kathmandu Univ Med J (KUMJ). 2013;11:226-32. KUMJ. 10.3126/kumj.v11i3.12510 [DOI] [PubMed] [Google Scholar]
- 98.Parajuli J, Saleh F, Thapa N, Ali L.Factors associated with nonadherence to diet and physical activity among Nepalese type 2 diabetes patients; a cross sectional study. BMC Res Notes. 2014;7:758. 10.1186/1756-0500-7-758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Sharma S, Bhadari SD.Knowledge and Practice regarding Self-Care among the patients with type II Diabetes of Kapan, Kathmandu. Journal of Advanced Academic Research. 2014;1:85-91. 10.3126/jaar.v1i2.16592 [DOI] [Google Scholar]
- 100.Ghimire S.Barriers to Diet and Exercise among Nepalese Type 2 Diabetic Patients. Int Sch Res Notices. 2017;2017:1273084. 10.1155/2017/1273084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Shrestha JTM, Shrestha H, Prajapati M, Karkee A, Maharjan A.Adverse effects of oral hypoglycemic agents and adherence to them among patients with type 2 diabetes mellitus in Nepal. Journal of Lumbini Medical College. 2017;5:34-40. 10.22502/jlmc.v5i1.126 [DOI] [Google Scholar]
- 102.Ghimire GD, Devi WA.Self-Management Behaviors among patients with Type 2 Diabetes at Manipal Teaching Hospital, Nepal. Int J Nurs Educ. 2018;10:49-54. 10.5958/0974-9357.2018.00039.9 [DOI] [Google Scholar]
- 103.Kadariya S, Aro AR.Barriers and facilitators to physical activity among urban residents with diabetes in Nepal. PLoS One. 2018;13:e0199329. 10.1371/journal.pone.0199329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Sapkota RP, Upadhyaya T, Gurung G, Parker M, Raman R, Pardhan S.Need to improve awareness and treatment compliance in high-risk patients for diabetic complications in Nepal. BMJ Open Diabetes Res Care. 2018;6:e000525. 10.1136/bmjdrc-2018-000525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Thapa D.Self-care activities among patients with diabetes attending a tertiary care hospital in Biratnagar, Nepal. Journal of Nobel Medical College. 2018;7:11-7. 10.3126/jonmc.v7i1.20841 [DOI] [Google Scholar]
- 106.Pokhrel S, Shrestha S, Timilsina A, Sapkota M, Bhatt MP, Pardhe BD.Self-Care Adherence And Barriers To Good Glycaemic Control In Nepalese Type 2 Diabetes Mellitus Patients: A Hospital-Based Cross-Sectional Study. J Multidiscip Healthc. 2019;12:817-26. 10.2147/JMDH.S216842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Bhattarai B, Bista B, Shrestha S, Budhathoki B, Dhamala B.Contributing Factors of Non-Adherence to Treatment among the Patients with Type II Diabetes Mellitus. Journal of Manmohan Memorial Institute of Health Sciences. 2019;5:68-78. 10.3126/jmmihs.v5i1.24075 [DOI] [Google Scholar]
- 108.Sharma K, Dhungana G, Adhikari S, Bista Pandey A, Sharma M.Depression and Anxiety among Patients with Type II Diabetes Mellitus in Chitwan Medical College Teaching Hospital, Nepal. Nurs Res Pract. 2021;2021:8846915. 10.1155/2021/8846915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Shrestha M, Ng A, Paudel R, Gray R.Association between subthreshold depression and self-care behaviours in adults with type 2 diabetes: A cross-sectional study. J Clin Nurs. 2021;30:2462-8. 10.1111/jocn.15741 [DOI] [PubMed] [Google Scholar]
- 110.Kandel S, Assanangkornchai S, Wichaidit W.Association between family behaviors and self-care activities among type-II diabetes mellitus patients at a teaching hospital in Kathmandu, Nepal. J Educ Health Promot. 2022;11:25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Saleh F, Mumu SJ, Ara F, Ali L, Hossain S, Ahmed KR.Knowledge, attitude and practice of type 2 diabetic patients regarding obesity: Study in a tertiary care hospital in Bangladesh. J Public Health Afr. 2012;3:e8. 10.4081/jphia.2012.e8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Mumu SJ, Saleh F, Ara F, Afnan F, Ali L.Non-adherence to life-style modification and its factors among type 2 diabetic patients. Indian J Public Health. 2014;58:40-4. 10.4103/0019-557X.128165 [DOI] [PubMed] [Google Scholar]
- 113.Saleh F, Mumu SJ, Ara F, Hafez MA, Ali L.Non-adherence to self-care practices & medication and health related quality of life among patients with type 2 diabetes: a cross-sectional study. BMC Public Health. 2014;14:431. 10.1186/1471-2458-14-431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Ahmed KR, Jebunessa F, Hossain S, Chowdhury HA.Ocular knowledge and practice among type 2 diabetic patients in a tertiary care hospital in Bangladesh. BMC Ophthalmol. 2017;17:171. 10.1186/s12886-017-0560-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chowdhury HA, Fatema K, Hossain S, Natasha K, Khan T, Akter J, et al. Knowledge and self-care practice regarding diabetes among type 2diabetics: experience from a non-profit hospital chain in Bangladesh. Int J Diabetes Dev Ctries. 2018;38:478-85. 10.1007/s13410-017-0597-7 [DOI] [Google Scholar]
- 116.Bukht MS, Ahmed KR, Hossain S, Masud P, Sultana S, Khanam R.Association between physical activity and diabetic complications among Bangladeshi type 2 diabetic patients. Diabetes Metab Syndr. 2019;13:806-9. 10.1016/j.dsx.2018.11.069 [DOI] [PubMed] [Google Scholar]
- 117.Majid MA, Bashet MA, Siddique MRF, Rahman ME.Association between dietary habit and diabetic risk profiles among diagnosed Type-2 diabetic patients in a selected area of Bangladesh. Diabetes Metab Syndr. 2019;13:1633-7. 10.1016/j.dsx.2019.03.030 [DOI] [PubMed] [Google Scholar]
- 118.Islam SMS, Uddin R, Zaman SB, Biswas T, Tansi T, Chegini Z, et al. Healthcare seeking behavior and glycemic control in patients with type 2 diabetes attending a tertiary hospital. Int J Diabetes Dev Ctries. 2020;41:280-7. [Google Scholar]
- 119.Hasan SM, Rahman M, Nakamura K, Tashiro Y, Miyashita A, Seino K.Relationship between diabetes self-care practices and control of periodontal disease among type2 diabetes patients in Bangladesh. PLoS One. 2021;16:e0249011. 10.1371/journal.pone.0249011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Medagama A, Galgomuwa M.Comorbidities and Ethnocultural Factors Limit the Physical Activity of Rural Sri Lankan Patients with Diabetes Mellitus. J Diabetes Res. 2018;2018:4319604. 10.1155/2018/4319604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Rathish D, Hemachandra R, Premadasa T, Ramanayake S, Rasangika C, Roshiban R, et al. Comparison of medication adherence between type 2 diabetes mellitus patients who pay for their medications and those who receive it free: a rural Asian experience. J Health Popul Nutr. 2019;38:4. 10.1186/s41043-019-0161-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Toobert DJ, Hampson SE, Glasgow RE.The summary of diabetes self-care activities measure: results from 7 studies and a revised scale. Diabetes Care. 2000;23:943-50. 10.2337/diacare.23.7.943 [DOI] [PubMed] [Google Scholar]
- 123.Schmitt A, Gahr A, Hermanns N, Kulzer B, Huber J, Haak T.The Diabetes Self-Management Questionnaire (DSMQ): development and evaluation of an instrument to assess diabetes self-care activities associated with glycaemic control. Health Qual Life Outcomes. 2013;11:138. 10.1186/1477-7525-11-138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Morisky DE, Ang A, Krousel-Wood M, Ward HJ.Predictive validity of a medication adherence measure in an outpatient setting. J Clin Hypertens (Greenwich). 2008;10:348-54. 10.1111/j.1751-7176.2008.07572.x [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 125.Morisky DE, Green LW, Levine DM.Concurrent and predictive validity of a self-reported measure of medication adherence. Med Care. 1986;24:67-74. 10.1097/00005650-198601000-00007 [DOI] [PubMed] [Google Scholar]
- 126.World Health Organization. Global physical activity questionnaire (GPAQ) analysis guide. Geneva: World Health Organization. 2012:1-22. [Google Scholar]
- 127.Craig CL, Marshall AL, Sjöström M, Bauman AE, Booth ML, Ainsworth BE, et al. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc. 2003;35:1381-95. 10.1249/01.MSS.0000078924.61453.FB [DOI] [PubMed] [Google Scholar]
- 128.van Weel C, Kassai R, Qidwai W, Kumar R, Bala K, Prasad Gupta P, et al. Primary healthcare policy implementation in South Asia. BMJ Glob Health. 2016;1:e000057. 10.1136/bmjgh-2016-000057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zaidi S, Saligram P, Ahmed S, Sonderp E, Sheikh K.Expanding access to healthcare in South Asia. BMJ. 2017;357:j1645. 10.1136/bmj.j1645 [DOI] [PubMed] [Google Scholar]
- 130.Thiel DM, Al Sayah F, Vallance JK, Johnson ST, Johnson JA.Association between Physical Activity and Health-Related Quality of Life in Adults with Type 2 Diabetes. Can J Diabetes. 2017;41:58-63. 10.1016/j.jcjd.2016.07.004 [DOI] [PubMed] [Google Scholar]
- 131.Guthold R, Stevens GA, Riley LM, Bull FC.Worldwide trends in insufficient physical activity from 2001 to 2016: a pooled analysis of 358 population-based surveys with 1.9 million participants. Lancet Glob Health. 2018;6:e1077-86. 10.1016/S2214-109X(18)30357-7 [DOI] [PubMed] [Google Scholar]
- 132.Advika TS, Idiculla J, Kumari SJ.Exercise in patients with Type 2 diabetes: Facilitators and barriers - A qualitative study. J Family Med Prim Care. 2017;6:288-92. 10.4103/2249-4863.219998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Boniol M, Dragomir M, Autier P, Boyle P.Physical activity and change in fasting glucose and HbA1c: a quantitative meta-analysis of randomized trials. Acta Diabetol. 2017;54:983-91. 10.1007/s00592-017-1037-3 [DOI] [PubMed] [Google Scholar]
- 134.Umpierre D, Ribeiro PA, Kramer CK, Leitão CB, Zucatti AT, Azevedo MJ, et al. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2011;305:1790-9. 10.1001/jama.2011.576 [DOI] [PubMed] [Google Scholar]
- 135.Sohal T, Sohal P, King-Shier KM, Khan NA.Barriers and Facilitators for Type-2 Diabetes Management in South Asians: A Systematic Review. PLoS One. 2015;10:e0136202. 10.1371/journal.pone.0136202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Gupta L, Arora B, Khandelwal D, Lal PR, Kalra S, Bhattacharya S, et al. Dietary Practices During Common Religious Fasts in People with Type 2 Diabetes Mellitus Among Different Religions in North India—A Questionnaire-based Study. US Endocrinol. 2020;16:36. 10.17925/USE.2020.16.1.36 [DOI] [Google Scholar]
- 137.Yadav UN, Lloyd J, Hosseinzadeh H, Baral KP, Dahal S, Bhatta N, et al. Facilitators and barriers to the self-management of COPD: a qualitative study from rural Nepal. BMJ Open. 2020;10:e035700. 10.1136/bmjopen-2019-035700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Capoccia K, Odegard PS, Letassy N.Medication Adherence With Diabetes Medication: A Systematic Review of the Literature. Diabetes Educ. 2016;42:34-71. 10.1177/0145721715619038 [DOI] [PubMed] [Google Scholar]
- 139.Juarez DT, Tan C, Davis J, Mau M.Factors Affecting Sustained Medication Adherence and Its Impact on Health Care Utilization in Patients with Diabetes. J Pharm Health Serv Res. 2013;4:89-94. 10.1111/jphs.12016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Wabe NT, Angamo MT, Hussein S.Medication adherence in diabetes mellitus and self management practices among type-2 diabetics in Ethiopia. N Am J Med Sci. 2011;3:418-23. 10.4297/najms.2011.3418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Malik VS, Willett WC, Hu FB.Global obesity: trends, risk factors and policy implications. Nat Rev Endocrinol. 2013;9:13-27. 10.1038/nrendo.2012.199 [DOI] [PubMed] [Google Scholar]
- 142.Misra A, Khurana L, Isharwal S, Bhardwaj S.South Asian diets and insulin resistance. Br J Nutr. 2009;101:465-73. 10.1017/S0007114508073649 [DOI] [PubMed] [Google Scholar]
- 143.Jayawardena R, Ranasinghe P, Byrne NM, Soares MJ, Katulanda P, Hills AP.Prevalence and trends of the diabetes epidemic in South Asia: a systematic review and meta-analysis. BMC Public Health. 2012;12:380. 10.1186/1471-2458-12-380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Papamichou D, Panagiotakos DB, Itsiopoulos C.Dietary patterns and management of type 2 diabetes: A systematic review of randomised clinical trials. Nutr Metab Cardiovasc Dis. 2019;29:531-43. 10.1016/j.numecd.2019.02.004 [DOI] [PubMed] [Google Scholar]
- 145.Ajala O, English P, Pinkney J.Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am J Clin Nutr. 2013;97:505-16. 10.3945/ajcn.112.042457 [DOI] [PubMed] [Google Scholar]
- 146.Allemann S, Houriet C, Diem P, Stettler C.Self-monitoring of blood glucose in non-insulin treated patients with type 2 diabetes: a systematic review and meta-analysis. Curr Med Res Opin. 2009;25:2903-13. 10.1185/03007990903364665 [DOI] [PubMed] [Google Scholar]
- 147.Yadav UN, Mistry SK, Ghimire S, Schneider CH, Rawal LB, Acharya SP, et al. Recognizing the roles of primary health care in addressing non-communicable diseases in low- and middle-income countries: Lesson from COVID-19, implications for the future. J Glob Health. 2021;11:03120. 10.7189/jogh.11.03120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Garduño-Diaz SD, Khokhar S.Prevalence, risk factors and complications associated with type 2 diabetes in migrant South Asians. Diabetes Metab Res Rev. 2012;28:6-24. 10.1002/dmrr.1219 [DOI] [PubMed] [Google Scholar]
- 149.Begum S, Venkatesan M, Ganapathy K.Foot care practices, its barriers and risk for peripheral neuropathy among diabetic patients attending medical college in rural Puducherry. Int J Community Med Public Health. 2019. Jan;6(1):203-7. [Google Scholar]
- 150.van Acker K, Léger P, Hartemann A, Chawla A, Siddiqui MK.Burden of diabetic foot disorders, guidelines for management and disparities in implementation in Europe: a systematic literature review. Diabetes Metab Res Rev. 2014;30:635-45. 10.1002/dmrr.2523 [DOI] [PubMed] [Google Scholar]
- 151.Booth A, Clarke M, Dooley G, Ghersi D, Moher D, Petticrew M, et al. The nuts and bolts of PROSPERO: an international prospective register of systematic reviews. Syst Rev. 2012;1:2. 10.1186/2046-4053-1-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.