Abstract
BACKGROUND
The gut microbiota (GM) has been proven to play a role in the regulation of host lipid metabolism, which provides a new theory about the pathogenesis of dyslipidemia. However, the associations of GM with dyslipidemia based on sex differences remain unclear and warrant elucidation.
AIM
To investigate the associations of GM features with serum lipid profiles based on sex differences in a Chinese population.
METHODS
This study ultimately recruited 142 participants (73 females and 69 males) at Honghui Hospital, Xi’an Jiaotong University. The anthropometric and blood metabolic parameters of all participants were measured. According to their serum lipid levels, female and male participants were classified into a high triglyceride (H_TG) group, a high total cholesterol (H_CHO) group, a low high-density lipoprotein cholesterol (L_HDL-C) group, and a control (CON) group with normal serum lipid levels. Fresh fecal samples were collected for 16S rRNA gene sequencing. UPARSE software, QIIME software, the RDP classifier and the FAPROTAX database were used for sequencing analyses.
RESULTS
The GM composition at the phylum level included Firmicutes and Bacteroidetes as the core GM. Different GM features were identified between females and males, and the associations between GM and serum lipid profiles were different in females and males. The GM features in different dyslipidemia subgroups changed in both female patients and male patients. Proteobacteria, Lactobacillaceae, Lactobacillus and Lactobacillus_salivarius were enriched in H_CHO females compared with CON females, while Coriobacteriia were enriched in L_HDL-C females. In the comparison among the three dyslipidemia subgroups in females, Lactobacillus_salivarius were enriched in H_CHO females, and Prevotellaceae were enriched in L_HDL-C females. Compared with CON or H_TG males, Prevotellaceae, unidentified_Ruminococcaceae, Roseburia and Roseburia_inulinivorans were decreased in L_HDL-C males (P value < 0.05), and linear discriminant analysis effect size analysis indicated an enrichment of the above GM taxa in H_TG males compared with other male subgroups. Additionally, Roseburia_inulinivorans abundance was positively correlated with serum TG and total cholesterol levels, and Roseburia were positively correlated with serum TG level. Furthermore, Proteobacteria (0.724, 95%CI: 0.567-0.849), Lactobacillaceae (0.703, 95%CI: 0.544-0.832), Lactobacillus (0.705, 95%CI: 0.547-0.834) and Lactobacillus_salivarius (0.706, 95%CI: 0.548-0.835) could distinguish H_CHO females from CON females, while Coriobacteriia (0.710, 95%CI: 0.547-0.841), Coriobacteriales (0.710, 95%CI: 0.547-0.841), Prevotellaceae (0.697, 95%CI: 0.534-0.830), Roseburia (0.697, 95%CI: 0.534-0.830) and Roseburia_inulinivorans (0.684, 95%CI: 0.520-0.820) could discriminate H_TG males from CON males. Based on the predictions of GM metabolic capabilities with the FAPROTAX database, a total of 51 functional assignments were obtained in females, while 38 were obtained in males. This functional prediction suggested that cellulolysis increased in L_HDL-C females compared with CON females, but decreased in L_HDL-C males compared with CON males.
CONCLUSION
This study indicates associations of GM with serum lipid profiles, supporting the notion that GM dysbiosis may participate in the pathogenesis of dyslipidemia, and sex differences should be considered.
Keywords: Dyslipidemia, Gut microbiota, 16S rRNA, Sequencing, Sex differences, Northwestern China
Core Tip: Dyslipidemia is the circulating lipid expression of metabolic syndrome, and alterations of gut microbiota (GM) are indicated to participate in the pathogenesis of dyslipidemia; however, little evidence was found in the literature on sex differences for these associations. Our results demonstrated the GM features in different dyslipidemia subgroups in females and males, suggesting a complex interaction between GM and lipid metabolism. Our observations may provide new evidence that different GM taxa may be associated with distinct lipids, and that GM may affect specific aspects of lipid metabolism. More studies are required to propose specific taxa that have the potential to ameliorate dyslipidemia.
INTRODUCTION
Obesity has become a worldwide public health challenge, with its prevalence nearly tripled since 1975[1]. Obesity is defined as a chronic accumulation of excessive lipids in tissues[2], which is related to the disruption of lipid metabolism[3]. As a defect in lipid metabolism, dyslipidemia is defined as any abnormality in blood lipid levels, and is characterized by an elevation of circulating triglyceride (TG), total cholesterol (TC), and low-density lipoprotein cholesterol (LDL-C), or a decrease in high-density lipoprotein cholesterol (HDL-C). Since observational studies have shown that 60%-70% of adults have lipid levels outside the recommended range[4,5], it is essential to reveal the underlying mechanism of dyslipidemia. Circulating lipid levels are known to have an important genetic contribution from over 500 single-nucleotide polymorphisms in more than 150 Loci, explaining approximately 40% of the total individual variation[6]. However, the unexplained 60% variation has been attributed to undiscovered elements and unquantified environmental factors, such as dietary intake and physical activity[7,8]. In recent years, considerable progress has been made in elucidating the mechanism responsible for dyslipidemia, and accumulated evidence has shown that the gut microbiota (GM) may play a potential role in obesity and related metabolic diseases, such as dyslipidemia[9-12].
The human gastrointestinal tract harbors over 100 trillion microorganisms[13], and the gut bacteria having effects on human health are the most prevalent and well-studied. In humans, GM profiles vary among different ethnicities (host genetics) and between different sexes[14,15], and are mainly shaped by early life events and stabilize in adolescence[16]. However, its composition and activity can be dynamic, and may be altered dramatically by multiple factors, such as medications, chronic dietary patterns and other environmental exposures[15,17,18]. This “microbial organ” has been recognized to perform various physiological functions[19-21], and is often called “a new virtual metabolic organ”[22,23]. The first indication of associations between GM and disorder statuses were for inflammation[24], and altered GM community (dysbiosis) has now been established in the development of cardiometabolic phenotypes[25]. These lines of evidence have raised an interest in GM as an important candidate in accounting for the unexplained variation in serum lipid levels in humans, and as a target for the therapeutic benefit of dyslipidemia[26]. Recent studies have convincingly linked GM to dyslipidemia, and GM was reported to explain substantial variation in TG and HDL-C levels independent of genetic factors in a Dutch study[26]. In addition, accumulating data from animal studies demonstrate that GM can affect host lipid metabolism through multiple direct and indirect biological mechanisms[27,28]. Nevertheless, determining associations between GM and host lipid metabolism remains a challenge in humans. Furthermore, sex is an important factor that may influence GM profiles, and sex differences can be observed in serum lipid profiles[29-31]. However, the relationship between GM and dyslipidemia based on sex differences remains unclear.
Collectively, ethnicity, geography and sex are potent factors that could influence the GM community[14,15,32]. Thus, this study focused on the associations of GM features with dyslipidemia based on sex differences in a northwestern Chinese population. We first reveal sex differences regarding GM features and then introduce dyslipidemia, highlighting its intricate relationships with GM, and discuss possible altered GM functions.
MATERIALS AND METHODS
Study design
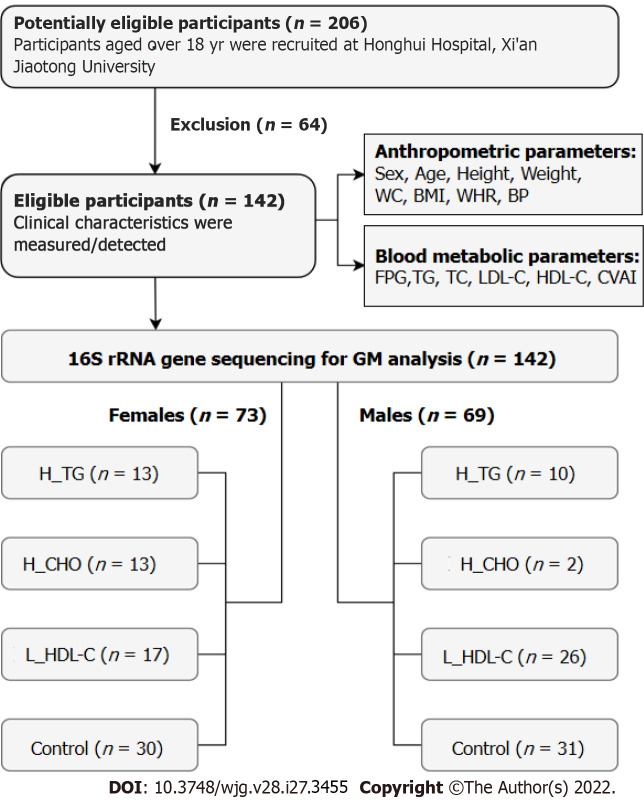
From July 2018 to January 2020, this study recruited 206 adult individuals (107 females and 99 males) from the outpatient clinics at Honghui Hospital, Xi’an Jiaotong University, China. Individuals were excluded if one of the following conditions existed: (1) With any gastrointestinal diseases, infectious or chronic diseases, serious systematic dysfunctions, or surgery histories of the gastrointestinal tract; (2) Taking any medications that could disrupt the original GM community, such as probiotics or prebiotics, antimicrobial therapies, anti-inflammatory drugs, acid-suppressing drugs, immunosuppressants, or anti-dyslipidemia/anti-dysglycemia/anti-hypertension drugs within the past month prior to sampling[33]; and (3) Females who were pregnant or lactating. The individuals who met the above conditions were included as participants in this study, and informed consent was obtained. Finally, 142 participants (73 females and 69 males) were recruited, including 81 dyslipidemia patients and 61 controls (CON) with normal serum lipid levels. Data privacy was ensured by using anonymized identifiers, and the study flow is shown in Figure 1. This study was approved by the Ethical Committee of Honghui Hospital, Xi’an Jiaotong University (Protocol Number: 201801022, approved January 8, 2018).
Figure 1.
Flow chart of this study. WC: Waist circumference; BMI: Body mass index; WHR: Waist circumference/height ratio; BP: Blood pressure; FPG: Fasting plasma glucose; TG: Triglyceride; TC: Total cholesterol; LDL-C: Low-density lipoprotein cholesterol; HDL-C: High-density lipoprotein cholesterol; CVAI: Chinese visceral adiposity index; GM: Gut microbiota; H_TG: High triglyceride group; H_CHO: High cholesterol group; L_HDL-C: Low high-density lipoprotein cholesterol group.
Measurement of anthropometric parameters
On their first visit, related medical information was documented for all participants. Body weight (W) and height (H) were measured without shoes and heavy clothing to the nearest 0.1 cm and 0.1 kg, respectively. Waist circumference (WC) was measured in the middle of the lower rib margin and the iliac crest with a nonexpandable tape to the nearest 0.1 cm in the standing position. Body mass index (BMI) and WC/height ratio (WHR) were calculated accordingly. Blood pressure (BP) was assessed using a medical electronic sphygmomanometer (HEM-7130 professional, OMRON, Dalian, China) on the left arm positioned at the heart level with palm face up in a sitting position. The participants were required to rest in a seated position for at least 5 min before BP assessment, with triplicate measurements at 1-min intervals. All equipment was calibrated at the beginning of the study.
Detection and computation of metabolic indicators
Venous blood samples were drawn from an antecubital vein in the morning following an overnight (at least 8 h) fast. Levels of fasting plasma glucose, TG, TC, LDL-C and HDL-C were detected by an automatic biochemical analyzer (Cobas c701, Roche, Mannheim, Germany). This instrument system was calibrated regularly. Moreover, the Chinese visceral adiposity index (CVAI) was calculated using the formula[34]: CVAI for females = -187.32 + 1.71 × age + 4.23 × BMI + 1.12 × WC (cm) + 39.76 × Log10TG (mmol/L) - 11.66 × HDL-C (mmol/L), and CVAI for males = -267.93 + 0.68 × age + 0.03 × BMI + 4.00 × WC + 22.00 × Log10TG - 16.32 × HDL-C.
Diagnostic criteria and grouping
In accordance with the “Guidelines for prevention and treatment of dyslipidemia in Chinese adults” (revised in 2016)[35] and the stratification standard of dyslipidemia in the primary prevention population of arteriosclerotic cardiovascular disease in China[36], dyslipidemia was defined as the presence of one or more abnormal serum lipid levels without any lipid-lowering medication: TG ≥ 1.7 mmol/L, TC ≥ 5.2 mmol/L, LDL-C ≥ 3.4 mmol/L, and/or HDL-C < 1.0 mmol/L. Subsequently, participants were divided into subgroups according to their serum lipid profiles. Specifically, patients were classified into the high TG (H_TG) group if only high TG level existed. Patients with increased serum TC and/or LDL-C levels and without HDL-C or TG abnormalities were classified into the high cholesterol (H_CHO) group. Patients were classified into the low HDL-C (L_HDL-C) group if only low serum HDL-C level existed. Participants with normal serum lipid profiles served as CON.
Fecal sample collection, DNA extraction, and 16S rRNA gene sequencing
Fresh fecal samples were collected from each participant at home, and stored in foam boxes with frozen cold packs[37]. Within 6 h after defecation, all fecal samples were transported to the Clinical Laboratory of Honghui Hospital, Xi’an Jiaotong University, and immediately stored at -80 °C until further processing. Genomic DNA was extracted from all samples using the QIAamp Fast DNA Stool Mini Kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions. After quality evaluation and concentration determination, DNA samples, greater than 1 μg and with an OD value between 1.8-2.0, were considered to be qualified for subsequent sequencing. Then, the V3-V4 regions of the 16S rRNA gene were amplified by universal primers (338F: 5’-ACT CCT ACG GGA GGC AGC AG-3’; 806R: 5’-GGA CTA CHV GGG TWT CTA AT-3’) with barcodes, and all PCRs were performed using Phusion® High Fidelity PCR Master Mix (New England Biolabs, Ipswich, MA, United States). Next, the PCR products were mixed and purified using a GeneJETTM Gel Extraction Kit (Thermo Scientific, Waltham, MA, United States). The sequencing library for each sample was constructed with the NEB Next® UltraTM DNA Library Prep Kit for Illumina (New England Biolabs, Ipswich, MA, United States), and library quality was assessed on a Qubit® 2.0 Fluorometer (Thermo Scientific, Waltham, MA, United States). Finally, the generated libraries were pair-end (2 × 250 bp) sequenced on the Illumina NovaSeq 6000 platform (Illumina, San Diego, CA, United States).
16S rRNA gene sequencing analyses
UPARSE (v7.0.1001) and QIIME software (v1.7.0) were introduced for sequencing analyses. According to a similarity threshold of 97%, acquired clean reads with high quality were de novo clustered into the same operational taxonomic unit (OTU), and the representative sequence of each OTU was screened and used to annotate taxonomic information based on the RDP classifier (v2.2). The GM diversity and composition were assessed based on the annotated OTUs. The alpha diversity (α-diversity) of GM was estimated by four indices, including Chao1, abundance coverage-based estimator (Ace), Shannon and Simpson. The comparisons of these indices between groups were conducted by the Wilcoxon rank-sum test, using function wilcox.test from the R package stats. To investigate the significance of differences in the GM community, beta diversity (β-diversity) was estimated using the unweighted UniFrac method to calculate the distances between samples, and visualized by the principal coordinates analysis (PCoA) model. The “WGCNA”, “stats” and “ggplot2” packages in R were utilized. The top ten GM taxa sorted by higher relative abundances at the six taxonomic levels, including phylum, class, order, family, genus and species, were identified and visualized in each group. The linear discriminant analysis (LDA) effect size (LEfSe) algorithm was applied to identify the enriched significant taxa in each group. The LDA score threshold was set to 2 on a log10 scale. The FAPROTAX (v1.2.2) database, containing 90 different types of metabolic assignments, was introduced to obtain the functional information of the GM community[38], and the differences between groups were assessed by the Wilcoxon rank-sum test, with a P value < 0.05 considered significant.
Statistical analysis
SPSS (v23.0.0.0, IBM SPSS Inc., Chicago, IL), R platform (v4.0.2, R Foundation, Vienna, Austria), GraphPad Prism (v8.4.3, GraphPad Software Inc., San Diego, CA, United States) and MedCalc (v19.0.4, MedCalc Software Bvba, Ostend, Belgium) were employed for statistical analysis and figure construction. The normal distribution of quantitative variables was assessed by the Shapiro-Wilk test. Clinical parameters are presented as the mean ± SD, and were analyzed among groups by the variance analysis (ANOVA) or Welch’s ANOVA depending on the homogeneity of variance. In addition, the Games-Howell test was used for multiple comparisons if a P value < 0.05 existed. The “ggcorrplot” package in R was utilized for Spearman correlation analysis between clinical parameters. Moreover, Spearman correlation analysis was also applied to evaluate the potential associations between GM features and clinical parameters, using the function cor.test (method = spearman) in R. Receiver operating characteristic (ROC) curve analysis, with areas under the curve, was applied to evaluate the diagnostic performance of specific GM taxa. All statistical tests with a P value < 0.05 were considered significant.
The bioinformatics analyses and statistical methods/techniques mentioned in this study were conducted, verified and reviewed by our expert Ji-Han Wang, PhD, from Institute of Medical Research, Northwestern Polytechnical University, and Guo-Dong Wang, Master Degree, from Department of Quality Control, Xi’an Mental Health Center.
RESULTS
Basic information of the study population
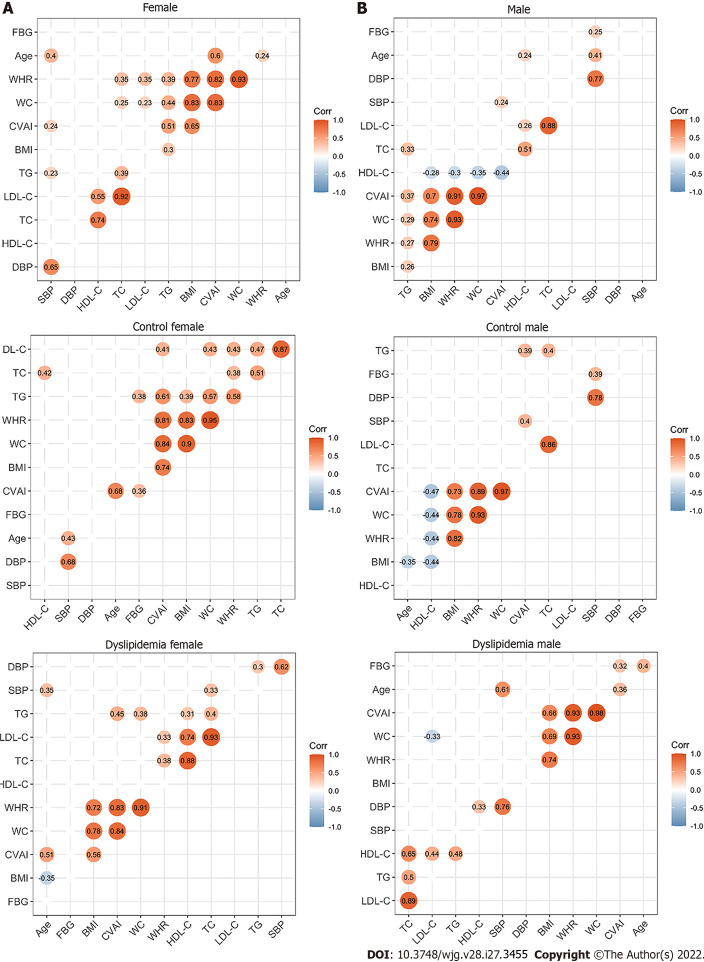
The present study finally included 142 participants (73 females and 69 males), as shown in Figure 1. Table 1 shows the clinical characteristics of all the participants grouped by sex and serum lipid profiles. Serum lipid levels and CVAI showed differences among female subgroups as did height, WC and WHR (P value < 0.05). Serum lipid levels and CVAI showed differences among male subgroups as well (P value < 0.05). Please refer to Supplementary Table 1 for detailed information and differences between females and males. The correlation analysis between the clinical characteristics of females and males is shown in Figure 2, and indicates positive correlations between the serum lipid indicators.
Table 1.
Clinical characteristics of the study population
|
|
Females
|
|
Males
|
|
||||||
|
High TG
|
High CHO
|
Low HDL-C
|
Control
|
P
value
|
High TG
|
High CHO
|
Low HDL-C
|
Control
|
P
value
|
|
| Number | 13 | 13 | 17 | 30 | 10 | 2 | 26 | 31 | ||
| Age (yr) | 55 ± 8 | 56 ± 5 | 54 ± 16 | 52 ± 12 | 0.420 | 54 ± 12 | 58 ± 11 | 52 ± 12 | 56 ± 13 | 0.770 |
| Height (cm) | 155.0 ± 7.2 | 147.0 ± 5.8 | 153.6 ± 5.4 | 154.3 ± 4.8 | 0.001a | 164.7 ± 6.2 | 160.0 ± 1.2 | 166.8 ± 5.4 | 164.8 ± 6.1 | 0.300 |
| Weight (kg) | 61.6 ± 9.3 | 53.6 ± 8.6 | 55.0 ± 9.2 | 55.3 ± 8.8 | 0.102 | 65.8 ± 9.0 | 56.2 ± 4.9 | 65.0 ± 8.3 | 61.0 ± 10.5 | 0.231 |
| WC (cm) | 90.2 ± 9.6 | 85.4 ± 7.2 | 82.3 ± 7.8 | 82.4 ± 8.6 | 0.035a | 88.3 ± 6.1 | 75.2 ± 9.5 | 87.0 ± 8.2 | 83.1 ± 8.8 | 0.066 |
| BMI (kg/m2) | 25.7 ± 3.7 | 24.7 ± 3.3 | 23.3 ± 3.2 | 23.2 ± 3.2 | 0.103 | 24.2 ± 3.0 | 22.0 ± 2.2 | 23.3 ± 2.8 | 22.4 ± 3.6 | 0.403 |
| WHR | 0.58 ± 0.07 | 0.58 ± 0.05 | 0.54 ± 0.06 | 0.53 ± 0.06 | 0.017a | 0.54 ± 0.03 | 0.47 ± 0.06 | 0.52 ± 0.05 | 0.50 ± 0.50 | 0.153 |
| SBP (mmHg) | 141 ± 17 | 139 ± 17 | 125 ± 19 | 129 ± 25 | 0.127 | 137 ± 23 | 136 ± 12 | 128 ± 21 | 134 ± 18 | 0.569 |
| DBP (mmHg) | 82 ± 10 | 78 ± 8 | 73 ± 9 | 76 ± 11 | 0.110 | 84 ± 12 | 80 ± 5 | 78 ± 11 | 81 ± 10 | 0.509 |
| FPG (mmol/L) | 5.00 ± 1.33 | 4.96 ± 0.57 | 5.48 ± 1.17 | 4.90 ± 0.59 | 0.346 | 5.05 ± 0.73 | 4.85 ± 2.23 | 4.81 ± 0.80 | 4.97 ± 1.10 | 0.895 |
| TG (mmol/L) | 2.15 ± 0.48 | 1.40 ± 0.22 | 1.01 ± 0.31 | 1.21 ± 0.33 | < 0.001a | 4.04 ± 0.86 | 5.75 ± 0.65 | 3.07 ± 0.76 | 3.97 ± 0.55 | 0.012a |
| TC (mmol/L) | 4.31 ± 0.50 | 5.58 ± 0.43 | 2.57 ± 0.63 | 0.41 ± 0.67 | < 0.001a | 2.01 ± 0.68 | 4.26 ± 0.95 | 1.77 ± 0.54 | 2.19 ± 0.45 | < 0.001a |
| LDL-C (mmol/L) | 2.16 ± 0.43 | 3.44±0.49 | 1.51 ± 0.56 | 2.25 ± 0.56 | < 0.001a | 1.13 ± 0.09 | 1.20 ± 0.27 | 0.79 ± 0.14 | 1.48 ± 0.32 | 0.001a |
| HDL-C (mmol/L) | 1.29 ± 0.23 | 1.75±0.31 | 0.77 ± 0.15 | 1.50 ± 0.36 | < 0.001a | 2.98 ± 0.78 | 4.55 ± 0.92 | 2.28 ± 0.69 | 2.49 ± 0.54 | < 0.001a |
| CVAI | 113.63 ± 25.96 | 94.16 ± 19.92 | 86.84 ± 32.86 | 76.30 ± 37.05 | 0.008a | 112.13 ± 26.62 | 54.08 ± 53.32 | 103.91 ± 36.87 | 78.76 ± 38.08 | 0.011a |
P < 0.05.
Data are presented as mean ± SD. P values are from the variance analysis (ANOVA) or Welch’s ANOVA depending on the homogeneity of variance. CHO: Cholesterol; WC: Waist circumference; BMI: Body mass index; WHR: Waist circumference/height ratio; SBP: Systolic blood pressure; DBP: Diastolic blood pressure; FPG: Fasting plasma glucose; TG: Triglyceride; TC: Total cholesterol; LDL-C: Low-density lipoprotein cholesterol; HDL-C: High-density lipoprotein cholesterol; CVAI: Chinese visceral adiposity index.
Figure 2.
Correlations between the clinical characteristics in the study population. A: Correlations between the clinical characteristics of the enrolled females; B: Correlations between the clinical characteristics of the enrolled males. The circle represents the correlation coefficient of each two parameters (P value < 0.05). The number presented in the circle is the correlation coefficient. The larger the absolute value is, the stronger the correlation is. Blue indicates a negative correlation and orange indicates a positive correlation. The depth of the color represents the strength of the correlation. The deeper the color is, the stronger the correlation is. The “ggcorrplot” package in R was utilized for Spearman’s correlation analysis. WC: Waist circumference; BMI: Body mass index; WHR: Waist circumference/height ratio; SBP: Systolic blood pressure; DBP: Diastolic blood pressure; TG: Triglyceride; TC: Total cholesterol; LDL-C: Low-density lipoprotein cholesterol; HDL-C: High-density lipoprotein cholesterol; CVAI: Chinese visceral adiposity index.
Diversity analysis of GM in the study population
To identify the associations of GM features with serum lipid profiles, we performed 16S rRNA gene sequencing analyses of GM from fecal samples. After quality CON, sequencing reads from 142 fecal samples were processed to determine the OTUs. Our data indicated that females had more unique OTUs, and the number of common OTUs shared by the female subgroups was larger than that shared by the male subgroups (data not shown).
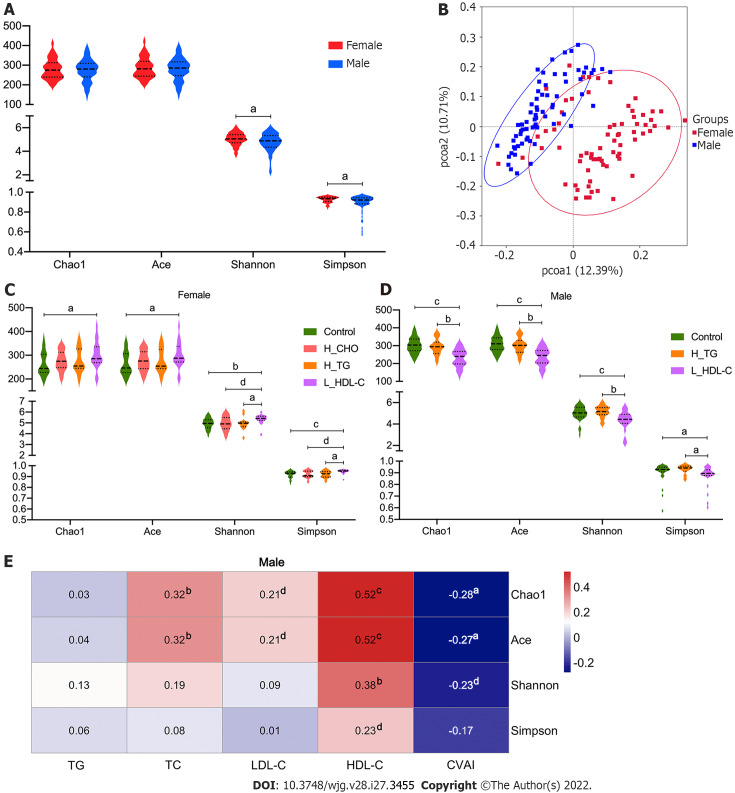
We evaluated the diversity of the GM community to assess the richness and evenness for females and males, and Shannon and Simpson indices suggested a higher α-diversity in females (P value < 0.05, Figure 3A). In addition, a dissimilarity between females and males was observed according to the visualized PCoA model for β-diversity analysis (Figure 3B). Since sex is an important determinant of GM and serum lipids[30,32,39], the study population was divided into a female group and a male group for the subsequent analyses.
Figure 3.
Diversity analysis of gut microbiota in the study population. A: Violin plots of α-diversity analysis of gut microbiota (GM) in females and males of the study population. Each plot represents one index of the α-diversity distribution of GM, including Chao1, Ace, Shannon and Simpson indices, for each group. The Wilcoxon rank-sum test was used to evaluate the differences between groups; B: Plots of principal coordinate analysis (PCoA) based on the operational taxonomic unit level in females and males of the study population. Each square represents the GM community in one sample, and the axis title represents the percentage change of interpretation. The distance between squares represents the similarity or dissimilarity of the GM community in the study population, and PCoA analysis was conducted by unweighted UniFrac method; C: Violin plots of α-diversity analysis of GM in females of the study population; D: Violin plots of α-diversity analysis of GM in males of the study population; E: Correlations between GM diversity and serum lipid profiles in males of the study population. Spearman’s correlation analysis was conducted. The number presented in each cell is the correlation coefficient. The larger the absolute value is, the stronger the correlation is. Blue indicates a negative correlation and red indicates a positive correlation. The depth of the color represents the strength of the correlation. The deeper the color is, the stronger the correlation is. aP < 0.05, bP < 0.01, cP < 0.001, dP < 0.1. TG: Triglyceride; TC: Total cholesterol; LDL-C: Low-density lipoprotein cholesterol; HDL-C: High-density lipoprotein cholesterol; CVAI: Chinese visceral adiposity index; H_CHO: High total cholesterol; H_TG: high triglyceride; L_HDL-C: Low high-density lipoprotein cholesterol.
To reveal the associations of GM diversity with serum lipid profiles based on sex differences, we conducted analogous analyses in the enrolled females and males. Our results showed that the α-diversity of GM was different in different dyslipidemia subgroups in females and males (Figure 3C and D). Of note, the α-diversity in L_HDL-C females was higher than that in CON females (P value < 0.05), while it was lower in L_HDL-C males than that in CON males (P value < 0.05). Additionally, the α-diversity of GM in H_TG males was higher than that in L_HDL-C males (P value < 0.05). The PCoA results suggested that the GM community in different dyslipidemia subgroups varied from that in the CON group in females and males, respectively, but could not be separated accurately and clearly (data not shown). Furthermore, the α-diversity of GM was found to be correlated with the serum lipid profiles and CVAI in males (Figure 3E). Specifically, the Chao1, Ace and Shannon indices were positively correlated with the serum HDL-C levels, while the Chao1 and Ace indices were negatively correlated with CVAI. However, similar results could not be observed in females. The bioinformatics analyses above revealed the associations between the general state of GM and the human serum lipid profiles.
Taxonomic composition of GM in the study population
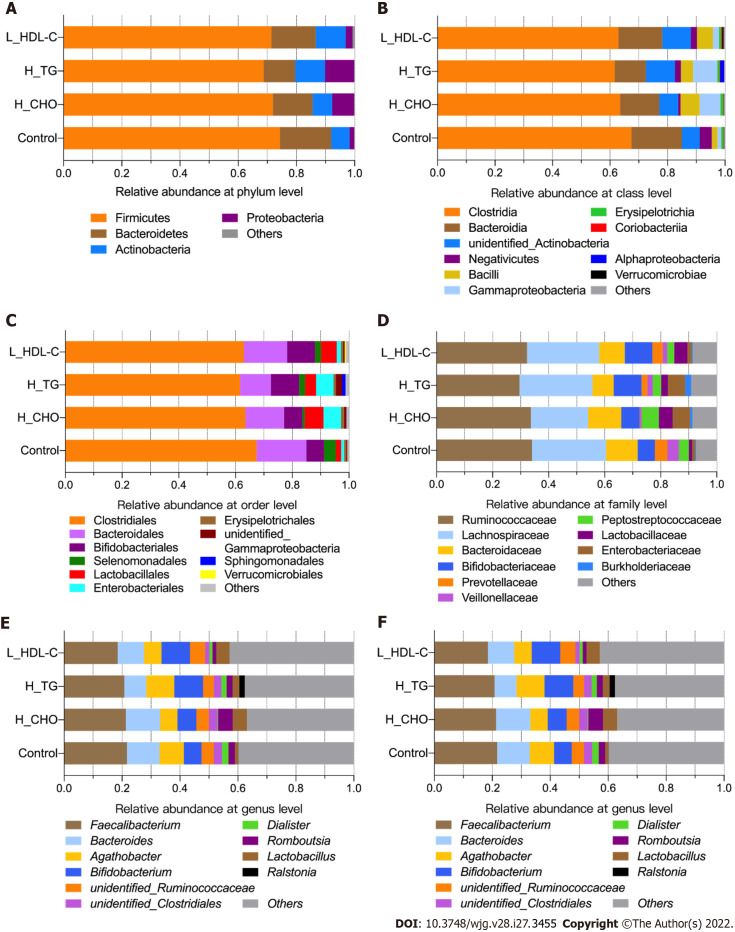
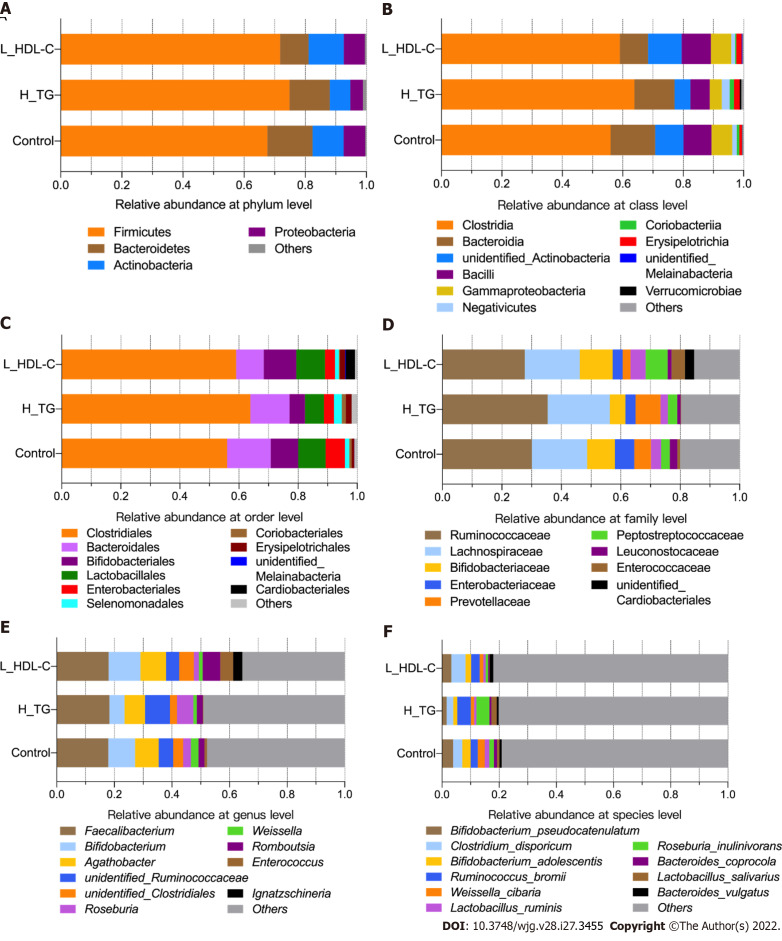
After diversity analysis, we focused on the relative abundances of GM taxa at six different taxonomic levels in different groups, and the top ten taxa with higher relative abundances at each level are shown in Supplementary Figure 1 for females and males. Similarly, the top ten taxa at each level were identified in the female subgroups (Figure 4). Consistent with previous data, the GM composition at the phylum level included Firmicutes and Bacteroidetes as the core GM, with lower relative abundances of Actinobacteria, Proteobacteria and others. The most abundant GM taxa were Clostridia, Bacteroidia, Clostridiales, Bacteroides, Ruminococcaceae, Lachnospiraceae, Bacteroidaceae, Faecalibacterium and Bacteroides. GM taxa with relative abundances of no less than 0.0001 at each level were included in the following analyses. The relative abundances of Proteobacteria, Lactobacillaceae and Lactobacillus were no less than 0.010 in H_CHO females, and were greater than those in CON females (P value < 0.05); while the relative abundance of Coriobacteriia was higher in L_HDL-C females than that in CON females (P value < 0.05). In the comparison of H_CHO and L_HDL-C females, Prevotellaceae abundance was greater in L_HDL-C females (P value < 0.05), and Lactobacillus_salivarius abundance was greater in H_CHO females (P value < 0.05). In the comparison of H_CHO and H_TG females, Agathobacter abundance was higher in H_TG females (P value < 0.05), while the relative abundances of Ruminococcus_bromii and Lactobacillus_salivarius were higher in H_CHO females (P value < 0.05). Additionally, Prevotellaceae abundance was greater in L_HDL-C females than that in H_TG females (P value < 0.05). Subsequently, analogous comparison analysis was conducted in the male subgroups (Figure 5). The most abundant taxa were Clostridia, Bacteroidia, Clostridiales, Bacteroidales, Ruminococcaceae, Lachnospiraceae, Faecalibacterium, Bifidobacterium, Bifidobacterium_pseudocatenulatum and Clostridium_disporicum. In the comparison of L_HDL-C and CON males, the relative abundances of Bacteroidetes, Bacteroidia, Bacteroidales, Prevotellaceae, unidentified_Ruminococcaceae, Roseburia and Roseburia_inulinivorans were lower in L_HDL-C males (P value < 0.05). Meanwhile, Coriobacteriia, Coriobacteriales, Prevotellaceae, unidentified_Ruminococcaceae, Roseburia and Roseburia_inulinivorans were lower in L_HDL-C males than those in H_TG males (P value < 0.05). Interestingly, no differences were observed in the comparison between the H_TG group and CON group (P value > 0.05) in females or males.
Figure 4.
The taxonomic composition of gut microbiota in females of the study population. Bar plots show the relative abundances of the top ten taxa at the six taxonomic levels, including phyla, class, orders, family, genus and species, in females. Each component of the cumulative bar chart indicates a phylum, a class, an order, a family, a genus or a species. A: Relative abundance at phylum level; B: Relative abundance at class level; C: Relative abundance at order level; D: Relative abundance at family level; E: Relative abundance at genus level; F: Relative abundance at species level. H_CHO: High total cholesterol; H_TG: high triglyceride; L_HDL-C: Low high-density lipoprotein cholesterol.
Figure 5.
The taxonomic composition of gut microbiota in males of the study population. Bar plots show the relative abundances of the top ten taxa at the six taxonomic levels, including phyla, class, orders, family, genus and species, in males. Each component of the cumulative bar chart indicates a phylum, a class, an order, a family, a genus or a species. A: Relative abundance at phylum level; B: Relative abundance at class level; C: Relative abundance at order level; D: Relative abundance at family level; E: Relative abundance at genus level; F: Relative abundance at species level. H_TG: high triglyceride; L_HDL-C: Low high-density lipoprotein cholesterol.
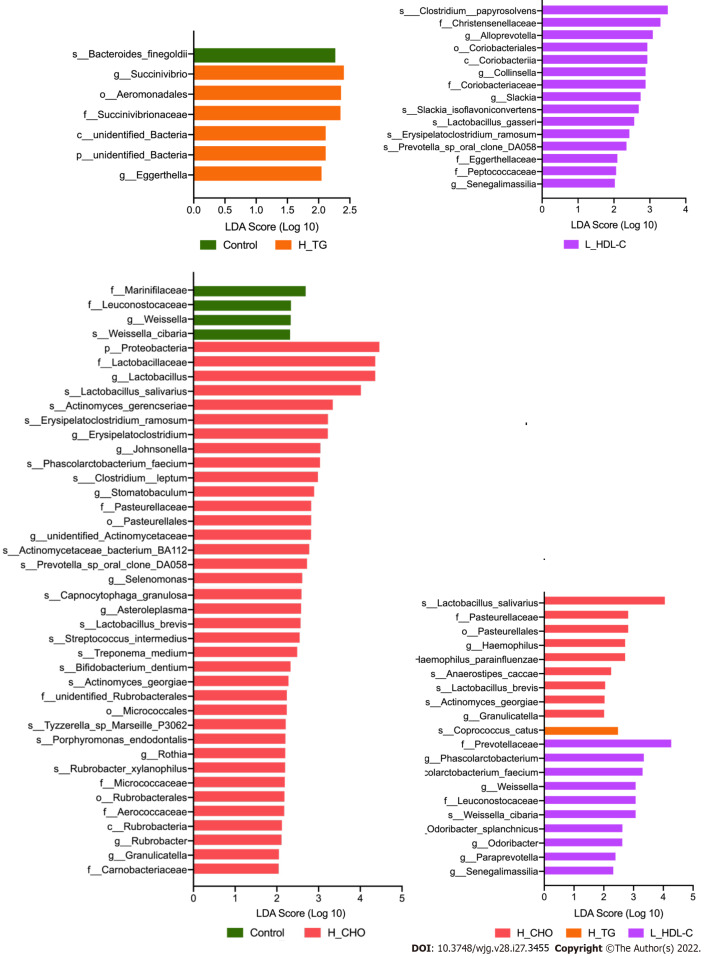
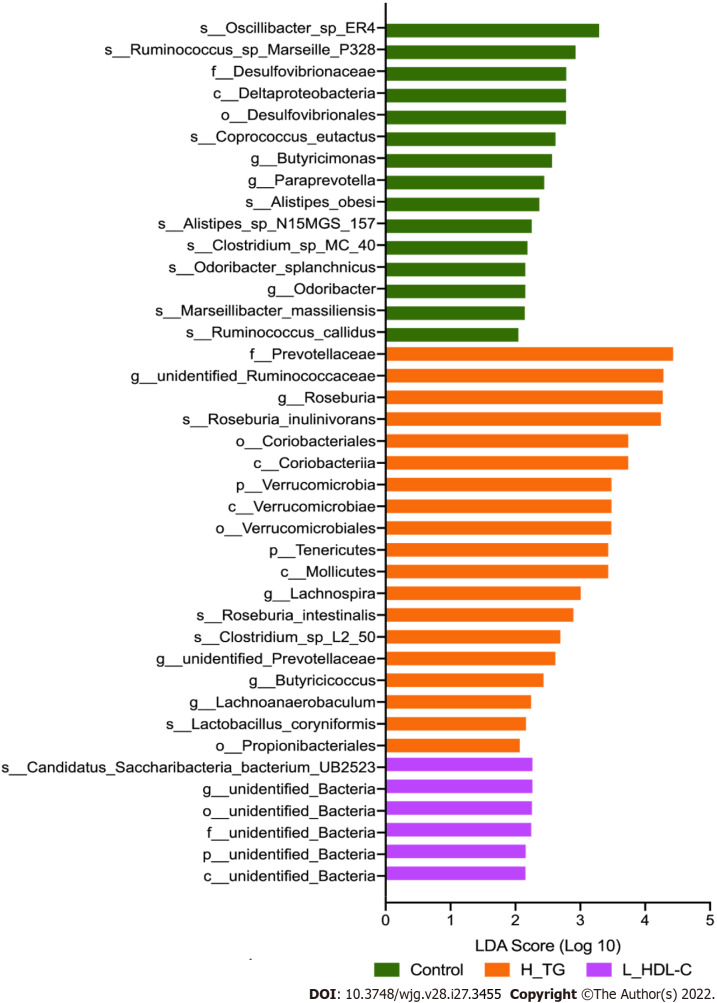
LEfSe analysis was conducted in different dyslipidemia subgroups and the CON group in females and males. Compared with CON females (Figure 6), LEfSe analysis revealed the enrichment of Proteobacteria, Lactobacillaceae, Lactobacillus and Lactobacillus_salivarius in H_CHO females, and the enrichment of Coriobacteriia in L_HDL-C females. In the comparison among the three dyslipidemia subgroups in females, Lactobacillus_salivarius were enriched in H_CHO females, and Prevotellaceae were enriched in L_HDL-C females. Simultaneously, LEfSe analysis revealed that Prevotellaceae, unidentified_Ruminococcaceae, Roseburia, Roseburia_inulinivorans, Coriobacteriia, Coriobacteriales and Verrucomicrobiae were enriched in H_TG males compared with other males (Figure 7).
Figure 6.
Linear discriminant analysis effect size analysis in females of the study population. Linear discriminant analysis (LDA) scores indicate differentially represented gut microbiota taxa (biomarkers) in each female subgroups. The length of each bar represents the LDA score format with log 10, and the logarithmic threshold for discriminative features was set to 2.0. H_CHO: High total cholesterol; H_TG: high triglyceride; L_HDL-C: Low high-density lipoprotein cholesterol.
Figure 7.
Linear discriminant analysis effect size analysis in males of the study population. Linear discriminant analysis (LDA) scores indicate differentially represented gut microbiota taxa (biomarkers) in each male subgroups. The length of each bar represents the LDA score format with log 10, and the logarithmic threshold for discriminative features was set to 2.0. H_TG: high triglyceride; L_HDL-C: Low high-density lipoprotein cholesterol.
Associations of GM taxa with serum lipid profiles in the study population
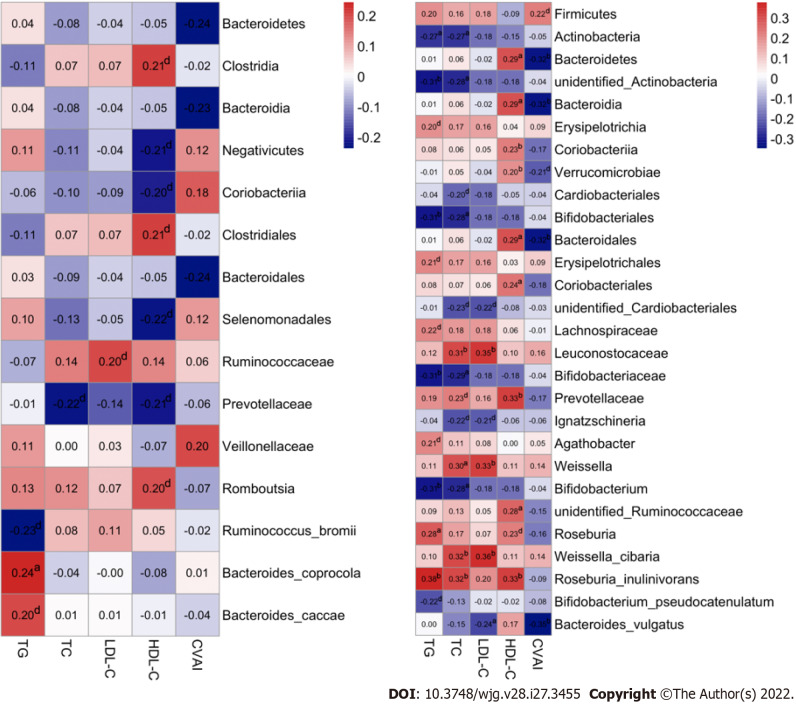
To further explore the clinical implications of GM in dyslipidemia, Spearman correlation analysis was introduced to assess the associations between differential GM taxa and the serum lipid profiles/CVAI, and a number of reliable correlations were revealed. In females, a positive correlation of Bacteroides_coprocola with serum TG level was observed, while negative correlations of Bacteroidetes, Bacteroidia and Bacteroidales with CVAI were noted (P value < 0.05, Figure 8A). Moreover, more correlations were identified in males (P value < 0.05, Figure 8B). Specifically, the relative abundances of Actinobacteria, unidentified_Actinobacteria, Bifidobacteriales, Bifidobacteriaceae and Bifidobacterium were negatively correlated with the serum TG and TC levels, the relative abundance of Roseburia_inulinivorans was positively correlated with the serum TG and TC levels, and the relative abundance of Roseburia was positively correlated with the serum TG level. In addition, the relative abundances of Leuconostocaceae, Weissella and Weissella_cibaria were correlated with increased serum TC and LDL-C levels, and the relative abundance of Bacteroides_vulgatus was correlated with decreased serum LDL-C level. Furthermore, we found that the relative abundances of Bacteroidetes, Bacteroidia, Bacteroidales, Coriobacteriales, Prevotellaceae, unidentified_Ruminococcaceae and Roseburia_inulinivorans were positively correlated with serum HDL-C level, and the relative abundances of Bacteroidetes, Bacteroidia, Bacteroidales and Bacteroides_vulgatus were negatively correlated with CVAI in males.
Figure 8.
Associations of gut microbiota taxa with serum lipid indicators in the study population. A: Correlations of gut microbiota (GM) taxa with serum lipid indicators in females. Spearman’s correlation analysis was conducted. The number presented in each cell is the correlation coefficient. The larger the absolute value is, the stronger the correlation is. Blue indicates a negative correlation and red indicates a positive correlation. The depth of the color represents the strength of the correlation. The deeper the color is, the stronger the correlation is; B: Correlations of GM taxa with serum lipid indicators in males. aP < 0.05, bP < 0.01, dP < 0.1. TG: Triglyceride; TC: Total cholesterol; LDL-C: Low-density lipoprotein cholesterol; HDL-C: High-density lipoprotein cholesterol; CVAI: Chinese visceral adiposity index.
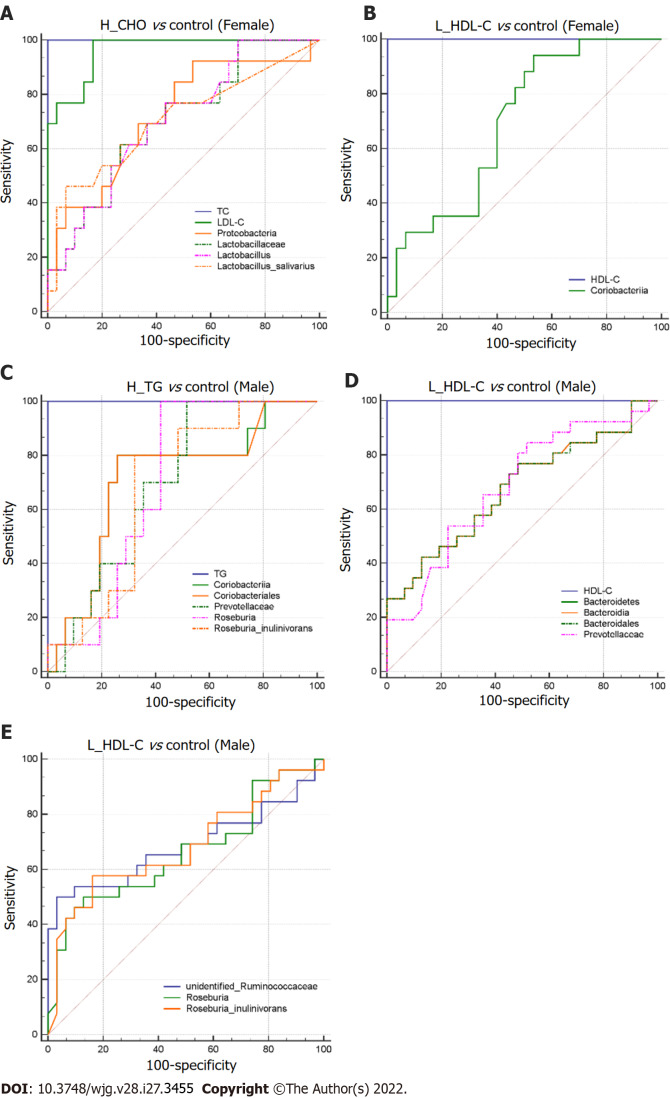
To search for the specific GM taxa that may facilitate the differentiation of lipid profiles in dyslipidemia patients from CON, ROC curve analysis was conducted in the female and male subgroups (P value < 0.05, Figure 9). The GM taxa, that effectively distinguished H_CHO females from CON females, were Proteobacteria (0.724, 95%CI: 0.567-0.849), Lactobacillaceae (0.703, 95%CI: 0.544-0.832), Lactobacillus (0.705, 95%CI: 0.547-0.834) and Lactobacillus_salivarius (0.706, 95%CI: 0.548-0.835). In addition, Coriobacteriia (0.697, 95%CI: 0.546-0.822) may help discriminate L_HDL-C females from CON females. Moreover, the GM taxa in favor of the differentiation of H_TG males from CON males were Coriobacteriia (0.710, 95%CI: 0.547-0.841), Coriobacteriales (0.710, 95%CI: 0.547-0.841), Prevotellaceae (0.697, 95%CI: 0.534-0.830), Roseburia (0.697, 95%CI: 0.534-0.830) and Roseburia_inulinivorans (0.684, 95%CI: 0.520-0.820). Additionally, seven GM taxa may play a role in distinguishing L_HDL-C males from CON males: Bacteroidetes (0.676, 95%CI: 0.539-0.794), Bacteroidia (0.676, 95%CI: 0.539-0.794), Bacteroidales (0.676, 95%CI: 0.538-0.793), Prevotellaceae (0.685, 95%CI: 0.548-0.802), unidentified_Ruminococcaceae (0.687, 95%CI: 0.551-0.804), Roseburia (0.662, 95%CI: 0.524-0.782) and Roseburia_inulinivorans (0.682, 95%CI: 0.545-0.799).
Figure 9.
Differential gut microbiota taxa-based classification of dyslipidemia in the study population. Receiver operating characteristic curve analysis for identifying dyslipidemia subgroup from controls by gut microbiota taxa in females and males. A: High total cholesterol vs control (Female); B: Low high-density lipoprotein cholesterol (L_HDL-C) vs control (Female); C: High triglyceride vs control (Male); D and E: L_HDL-C vs control (Male). H_CHO: High total cholesterol; H_TG: high triglyceride; L_HDL-C: Low high-density lipoprotein cholesterol.
Functional analysis of GM in the study population
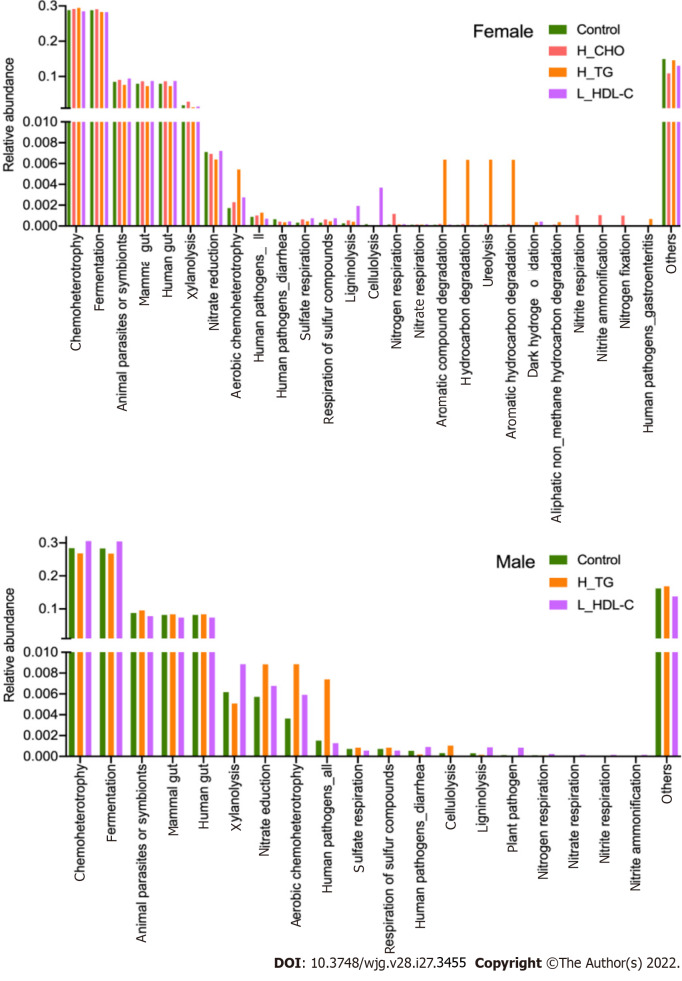
Finally, we evaluated the functions of the GM community using the FAPROTAX database, and obtained the main metabolic processes of microorganisms associated with different biogeochemical cycles. A total of 51 functional assignments, with relative abundances larger than 0.0001 of the average level, were obtained in females, and 38 assignments were obtained in males. Apparently, GM functions showed different patterns between females and males, further supporting our strategy of separation and independent analysis in females and males. The top twenty annotated functions with higher relative abundances in females and males are shown in Figure 10. Furthermore, comparison analysis between the different subgroups was conducted. Compared with CON females, the relative abundance of aerobic chemoheterotrophy was increased in H_CHO females (P value < 0.05), while cellulolysis abundance was increased in L_HDL-C females (P value < 0.05). Additionally, cellulolysis abundance was increased in L_HDL-C females when compared with that in H_CHO females (P value < 0.05). Compared with CON males, the relative abundances of sulfate respiration, respiration of sulfur compounds and cellulolysis were decreased in L_HDL-C males (P value < 0.05), while the relative abundance of nitrate respiration was increased in L_HDL-C males (P value < 0.05).
Figure 10.
The main functional annotations of gut microbiota in the study population. Bar plots show the relative abundances of top twenty annotated functions with higher relative abundances in female subgroups and male subgroups, respectively, using the FAPROTAX database. H_CHO: High total cholesterol; H_TG: high triglyceride; L_HDL-C: Low high-density lipoprotein cholesterol.
DISCUSSION
Dyslipidemia is considered a defect of lipid metabolism in circulation, characterized by increased or decreased levels of serum lipids. Notably, alterations in the GM community may participate in the pathogenesis of dyslipidemia, and the GM composition could be influenced by sex, host genotype and geographic location. To our knowledge, little evidence is present in the literature on the correlations between GM and serum lipids based on sex differences[39]; hence, the initial objective of this study was to identify the associations of GM features with serum lipid profiles based on sex differences in a Chinese population.
Considered as a whole, there were significant differences in serum lipid profiles and GM features between females and males. Specifically, females have higher GM diversities, and the GM composition was quite different from that in males, which is consistent with previous data[40]. In addition, the functional analysis of GM also showed an obvious dissimilarity between females and males, further supporting our strategy of respective GM analysis in females and males[14]. Of note, the associations between GM and dyslipidemia in humans have been investigated and demonstrated by various studies, suggesting the alteration of GM in patients with impaired lipid metabolism[28,41]. For instance, Cotillard et al[42] noted that reduced GM richness, commonly observed in obese patients, was linked to increased serum TG and TC levels. Following that study, more studies have demonstrated negative correlations of circulating TG and LDL-C levels with GM diversity, and a positive correlation of HDL-C with GM richness[9,26,43,44]. Moreover, certain GM taxa were found to be correlated with specific lipid profiles, suggesting that different GM taxa may affect distinct classes of lipids[26,42,43]. Although the present data have not clearly defined the GM pattern of patients with dyslipidemia, these observations provide new avenues for validation and follow-up studies. Therefore, dyslipidemia female and male patients in this study were divided into a H_TG group, a H_CHO group and a L_HDL-C group, for a more targeted investigation and interpretation.
As an important indicator of the GM community and a general measure of gut health, higher GM diversities have been proposed to be associated with healthy lipid levels, such as increased HDL-C and decreased TG levels[26]. Our results showed a positive correlation between GM diversities and the serum HDL-C level in males, consistent with previous data[26]. Additionally, GM diversities were negatively correlated with CVAI in males. As a valuable indicator of “adipose distribution and function”, CVAI has been suggested to be a reliable and applicable indicator for the evaluation of visceral fat dysfunction in Chinese individuals, which is based on simple and obtainable clinical parameters and the serum lipid levels[34,45]. The CVAI is essentially correlated with lipid metabolism, and its negative correlation with GM diversities could further support the reliability of our data.
Butyrate has been suggested to facilitate the prevention and treatment of diet-induced obesity by reducing fat accumulation and insulin resistance[46,47], and the ability to produce butyrate is widely distributed among gram-positive anaerobic bacteria[48]. As members of the butyrate-producing bacteria[48], Roseburia and Roseburia_inulinivorans have been investigated in certain diseases. By analyzing the GM composition of patients with symptomatic atherosclerosis, Karlsson et al[49] noted an enrichment of Roseburia in CVD patients, while a lower abundance of Roseburia was be observed in patients with diabetes[44]. In this study, we found that Roseburia_inulinivorans were positively correlated with the serum TG, TC and HDL-C levels, and Roseburia were positively correlated with the serum TG level in males. In addition, decreased Roseburia and Roseburia_inulinivorans abundances distinguished L_HDL-C males from CON males, while increased Roseburia and Roseburia_inulinivorans abundances distinguished H_TG males from CON males. Furthermore, strains of Bifidobacterium and Lactobacillus have potential therapeutic purposes[50]. An et al[51] once described a comparable, positive anti-obesity and lipid-lowering effect of Bifidobacterium spp. in obese rats fed a high-fat diet. Consistent with the evidence, our results demonstrated that the relative abundances of Bifidobacteriales, Bifidobacteriaceae and Bifidobacterium were negatively correlated with the serum TG and TC levels in males. However, certain favorable GM taxa, such as Lactobacillaceae, Lactobacillus and Lactobacillus_salivarius, were enriched in H_CHO females. As a probiotic supplement, Lactobacillus were noted to have a negative correlation with serum lipid profiles[52]. An animal study, focused on mice fed a high-fat high-cholesterol diet and supplemented with Lactobacillus curvatus and/or Lactobacillus plantarum, revealed that these probiotic bacteria play important roles in normalizing lipid metabolism, such as decreasing TC levels in plasma and liver, and reducing the accumulation of hepatic TG[53]. Obviously, certain conflicting findings exist, which should be further investigated. Nevertheless, these observations indicate that different GM taxa associate with certain lipids, and may affect specific aspects of lipid metabolism[26,42,43].
A recent study on rats[54] demonstrated that a high-fat diet decreased the proportions of Bacteroidetes and its genera Bacteroides and Prevotella, and a study on swine indicated that decreased Bacteroidetes proportions were accompanied by decreases in the circulating TG level[55]; however, whether Bacteroidetes can alter cholesterol or TG levels in humans remains disputed. In humans, decreased Bacteroidetes proportions, including Bacteroides_vulgatus and Bacteroides_dorei, were observed in patients with coronary artery disease[56,57]. In addition, some species of Bacteroides were shown to be decreased in patients with CVDs[49], and may be used as biomarkers for evaluating the alleviation of obesity[54]. Our results showed that Bacteroides_vulgatus were negatively correlated with the serum LDL-C level but positively correlated with the serum TG level in females, while Bacteroidetes, Bacteroidia and Bacteroidales were negatively correlated with CVAI in females. Moreover, Bacteroidetes, Bacteroidia and Bacteroidales were positively correlated with the serum HDL-C level, differentiated L_HDL-C males from CON males, and were negatively correlated with CVAI in males. These observed associations may further support their roles in the favorable regulation of lipid metabolism. Interestingly, Prevotellaceae were enriched in L_HDL-C group in the comparison among the three dyslipidemia subgroups in females. However, the relative abundance of Prevotellaceae was decreased significantly in L_HDL-C males, and could distinguish L_HDL-C males from CON males. Additionally, increased Prevotellaceae also helped discriminate H_TG males from CON males in our study. Of note, Kelly et al[58] reported that genera within the family Prevotellaceae had different effects; some were associated with an increased and others with a decreased CVD risk profile. Meanwhile, Coriobacteriia were indicated to be enriched in L_HDL-C females, and could differentiate L_HDL-C females from CON females. Moreover, we identified that Coriobacteriales were correlated with the increased serum HDL-C level in males, and increased Coriobacteriia and Coriobacteriales could distinguish H_TG males from CON males. However, these taxa have not been studied thoroughly, and it is difficult to assess their metabolic functions in human lipid metabolism.
Different GM taxa may have distinct activities and modes of action[50,59], and certain taxa may exert synergistic and cooperative interactions[60], indicating the importance of balance in the GM community. Our results may suggest a complex interaction between GM and distinct lipid metabolisms based on sex differences, and provide new evidence of the involvement of GM in dyslipidemia. Nevertheless, more studies are required to determine which specific taxa have the potential to ameliorate dyslipidemia.
There were several limitations in this study, the most marked of which was the small sample size in each subgroup. This was a single-center study, recruiting Chinese participants near Xi’an, a central city in northwestern China. In addition, certain confounders, such as dietary habit, that could have influenced the GM composition[61], must be taken into consideration. However, elucidation of certain associations of GM taxa with host lipid metabolism in this study is still valuable. We hope that our observations will facilitate prospective studies investigating diverse aspects of GM influences on human dyslipidemia based on sex differences. In future studies, it is recommended that the sample size be increased, more confounders be considered, and various research methods be integrated to ascertain potential associations.
CONCLUSION
Based on the thorough analyses of GM features with dyslipidemia in females and males, potent associations of GM-host relations based on sex differences were revealed, and the potential of GM for dyslipidemia diagnosis was demonstrated. Although this study could not provide a conclusive association between GM and dyslipidemia, it may provide new insights into the pathogenesis of dyslipidemia.
ARTICLE HIGHLIGHTS
Research background
Dyslipidemia is a common chronic disorder, and is defined as any abnormality in blood lipid levels. In recent years, considerable progress has been made in elucidating the mechanisms of dyslipidemia, and the gut microbiota (GM) has been indicated to play a pivotal role in its pathophysiology. However, the associations between GM and dyslipidemia remain to be elucidated.
Research motivation
Although recent studies have convincingly linked GM to dyslipidemia, proving the associations between GM and host lipid metabolism remains a challenge in humans. In addition, ethnicity, geography and sex are potent factors that could influence the GM community. Therefore, it is important to clarify the relationship between GM and dyslipidemia, and to explore the importance of sex differences. In this study, we first revealed sex differences regarding the GM features, and then introduced dyslipidemia, highlighting its intricate relationships with GM and possible altered GM functions.
Research objectives
This study focused on the associations of GM features with dyslipidemia based on sex differences in a northwestern Chinese population.
Research methods
This study finally recruited 142 participants (73 females and 69 males) at Honghui Hospital, Xi’an Jiaotong University, who fulfilled the criteria for the diagnosis of dyslipidemia according to the “Guidelines for prevention and treatment of dyslipidemia in Chinese adults”. The anthropometric and blood metabolic parameters of all participants were measured. According to their detected serum lipid levels, female and male participants were classified into a high triglyceride (H_TG) group, a high total cholesterol (H_CHO) group, a low high-density lipoprotein cholesterol (L_HDL-C) group, and a control (CON) group with normal serum lipid levels. Fresh fecal samples were collected for 16S rRNA gene sequencing, and UPARSE software, QIIME software, the RDP classifier tool and the FAPROTAX database were used for sequencing analyses.
Research results
Different GM features were identified between females and males, and the associations between GM and serum lipid profiles were different between females and males. In the comparison of the three dyslipidemia subgroups in females, Lactobacillus_salivarius were enriched in H_CHO females, and Prevotellaceae were enriched in L_HDL-C females. Compared with CON or H_TG males, Prevotellaceae, unidentified_Ruminococcaceae, Roseburia and Roseburia_inulinivorans were decreased in L_HDL-C males; while linear discriminant analysis effect size analysis indicated an enrichment of these above GM taxa in H_TG males in comparison to other male subgroups. Additionally, Roseburia_inulinivorans abundance was positively correlated with the serum TG and total cholesterol levels, and Roseburia were positively correlated with the serum TG level. Furthermore, Proteobacteria, Lactobacillaceae, Lactobacillus and Lactobacillus_salivarius distinguished H_CHO females from CON females, while Coriobacteriia, Coriobacteriales, Prevotellaceae, Roseburia and Roseburia_inulinivorans distinguished H_TG males from CON males. Based on the predictions of GM metabolic capabilities from the FAPROTAX database, cellulolysis was increased in L_HDL-C females compared with CON females, but was decreased in L_HDL-C males compared with CON males.
Research conclusions
This study provides evidence of the associations between GM and serum lipid profiles based on sex differences, suggesting a complex interaction between GM and distinct lipid metabolisms, and providing new insights into the pathogenesis of dyslipidemia.
Research perspectives
Future studies are needed to determine which specific taxa have the potential to ameliorate dyslipidemia, to investigate the underlying biological functions of the key GM in dyslipidemia, and to explore the differences in diet and other factors between females and males as possible causes for the observed differences in GM and the lipid profiles.
Footnotes
Institutional review board statement: This study was reviewed and approved by the Ethics Committee of Honghui Hospital, Xi’an Jiaotong University, No. 201801022.
Informed consent statement: All study participants provided informed consent prior to study enrollment.
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: February 18, 2022
First decision: May 9, 2022
Article in press: June, 20, 2022
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Amedei A, Italy; Panduro A, Mexico; Papazafiropoulou A, Greece S-Editor: Fan JR L-Editor: A P-Editor: Qi WW
Contributor Information
Lei Guo, Department of Spine Surgery, Honghui Hospital, Xi'an Jiaotong University, Xi'an 710054, Shaanxi Province, China.
Yang-Yang Wang, School of Electronics and Information, Northwestern Polytechnical University, Xi'an 710129, Shaanxi Province, China.
Ji-Han Wang, Institute of Medical Research, Northwestern Polytechnical University, Xi'an 710072, Shaanxi Province, China.
He-Ping Zhao, Department of Clinical Laboratory, Honghui Hospital, Xi'an Jiaotong University, Xi'an 710054, Shaanxi Province, China.
Yan Yu, Department of Clinical Laboratory, Honghui Hospital, Xi'an Jiaotong University, Xi'an 710054, Shaanxi Province, China.
Guo-Dong Wang, Department of Quality Control, Xi’an Mental Health Center, Xi'an 710100, Shaanxi Province, China.
Kun Dai, Department of Clinical Laboratory, Yanliang Railway Hospital of Xi’an, Xi'an 710089, Shaanxi Province, China.
Yu-Zhu Yan, Department of Clinical Laboratory, Honghui Hospital, Xi'an Jiaotong University, Xi'an 710054, Shaanxi Province, China.
Yan-Jie Yang, Department of Cardiology, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an 710061, Shaanxi Province, China.
Jing Lv, Department of Clinical Laboratory, Honghui Hospital, Xi'an Jiaotong University, Xi'an 710054, Shaanxi Province, China. lvjing-1219@163.com.
Data sharing statement
The raw data supporting the conclusions of this article available from the corresponding author at lvjing-1219@163.com.
References
- 1.World Health Organization. Obesity and overweight Fact Sheet. 2021. [cited 10 January 2022]. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight .
- 2.Sun B, Karin M. Obesity, inflammation, and liver cancer. J Hepatol. 2012;56:704–713. doi: 10.1016/j.jhep.2011.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bloor ID, Symonds ME. Sexual dimorphism in white and brown adipose tissue with obesity and inflammation. Horm Behav. 2014;66:95–103. doi: 10.1016/j.yhbeh.2014.02.007. [DOI] [PubMed] [Google Scholar]
- 4.Gao N, Yu Y, Zhang B, Yuan Z, Zhang H, Song Y, Zhao M, Ji J, Liu L, Xu C, Zhao J. Dyslipidemia in rural areas of North China: prevalence, characteristics, and predictive value. Lipids Health Dis. 2016;15:154. doi: 10.1186/s12944-016-0328-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tóth PP, Potter D, Ming EE. Prevalence of lipid abnormalities in the United States: the National Health and Nutrition Examination Survey 2003-2006. J Clin Lipidol. 2012;6:325–330. doi: 10.1016/j.jacl.2012.05.002. [DOI] [PubMed] [Google Scholar]
- 6.García-Giustiniani D, Stein R. Genetics of Dyslipidemia. Arq Bras Cardiol. 2016;106:434–438. doi: 10.5935/abc.20160074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kokkinos PF, Fernhall B. Physical activity and high density lipoprotein cholesterol levels: what is the relationship? Sports Med. 1999;28:307–314. doi: 10.2165/00007256-199928050-00002. [DOI] [PubMed] [Google Scholar]
- 8.O'Doherty AF, Jones HS, Sathyapalan T, Ingle L, Carroll S. The Effects of Acute Interval Exercise and Strawberry Intake on Postprandial Lipemia. Med Sci Sports Exerc. 2017;49:2315–2323. doi: 10.1249/MSS.0000000000001341. [DOI] [PubMed] [Google Scholar]
- 9.Matey-Hernandez ML, Williams FMK, Potter T, Valdes AM, Spector TD, Menni C. Genetic and microbiome influence on lipid metabolism and dyslipidemia. Physiol Genomics. 2018;50:117–126. doi: 10.1152/physiolgenomics.00053.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tremaroli V, Bäckhed F. Functional interactions between the gut microbiota and host metabolism. Nature. 2012;489:242–249. doi: 10.1038/nature11552. [DOI] [PubMed] [Google Scholar]
- 11.Cani PD. Metabolism in 2013: The gut microbiota manages host metabolism. Nat Rev Endocrinol. 2014;10:74–76. doi: 10.1038/nrendo.2013.240. [DOI] [PubMed] [Google Scholar]
- 12.Plaza-Diaz J. Nutrition, Microbiota and Noncommunicable Diseases. Nutrients. 2020;12 doi: 10.3390/nu12071971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Turnbaugh PJ, Ley RE, Hamady M, Fraser-Liggett CM, Knight R, Gordon JI. The human microbiome project. Nature. 2007;449:804–810. doi: 10.1038/nature06244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, Magris M, Hidalgo G, Baldassano RN, Anokhin AP, Heath AC, Warner B, Reeder J, Kuczynski J, Caporaso JG, Lozupone CA, Lauber C, Clemente JC, Knights D, Knight R, Gordon JI. Human gut microbiome viewed across age and geography. Nature. 2012;486:222–227. doi: 10.1038/nature11053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schloissnig S, Arumugam M, Sunagawa S, Mitreva M, Tap J, Zhu A, Waller A, Mende DR, Kultima JR, Martin J, Kota K, Sunyaev SR, Weinstock GM, Bork P. Genomic variation landscape of the human gut microbiome. Nature. 2013;493:45–50. doi: 10.1038/nature11711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dominguez-Bello MG, Blaser MJ, Ley RE, Knight R. Development of the human gastrointestinal microbiota and insights from high-throughput sequencing. Gastroenterology. 2011;140:1713–1719. doi: 10.1053/j.gastro.2011.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jernberg C, Löfmark S, Edlund C, Jansson JK. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology (Reading) 2010;156:3216–3223. doi: 10.1099/mic.0.040618-0. [DOI] [PubMed] [Google Scholar]
- 18.Wu GD, Chen J, Hoffmann C, Bittinger K, Chen YY, Keilbaugh SA, Bewtra M, Knights D, Walters WA, Knight R, Sinha R, Gilroy E, Gupta K, Baldassano R, Nessel L, Li H, Bushman FD, Lewis JD. Linking long-term dietary patterns with gut microbial enterotypes. Science. 2011;334:105–108. doi: 10.1126/science.1208344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sommer F, Bäckhed F. The gut microbiota--masters of host development and physiology. Nat Rev Microbiol. 2013;11:227–238. doi: 10.1038/nrmicro2974. [DOI] [PubMed] [Google Scholar]
- 20.Rinninella E, Raoul P, Cintoni M, Franceschi F, Miggiano GAD, Gasbarrini A, Mele MC. What is the Healthy Gut Microbiota Composition? Microorganisms. 2019;7 doi: 10.3390/microorganisms7010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oliphant K, Allen-Vercoe E. Macronutrient metabolism by the human gut microbiome: major fermentation by-products and their impact on host health. Microbiome. 2019;7:91. doi: 10.1186/s40168-019-0704-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Evans JM, Morris LS, Marchesi JR. The gut microbiome: the role of a virtual organ in the endocrinology of the host. J Endocrinol. 2013;218:R37–R47. doi: 10.1530/JOE-13-0131. [DOI] [PubMed] [Google Scholar]
- 23.Dieterich W, Schink M, Zopf Y. Microbiota in the Gastrointestinal Tract. Med Sci (Basel) 2018;6 doi: 10.3390/medsci6040116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Blander JM, Longman RS, Iliev ID, Sonnenberg GF, Artis D. Regulation of inflammation by microbiota interactions with the host. Nat Immunol. 2017;18:851–860. doi: 10.1038/ni.3780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nicholson JK, Holmes E, Kinross J, Burcelin R, Gibson G, Jia W, Pettersson S. Host-gut microbiota metabolic interactions. Science. 2012;336:1262–1267. doi: 10.1126/science.1223813. [DOI] [PubMed] [Google Scholar]
- 26.Fu J, Bonder MJ, Cenit MC, Tigchelaar EF, Maatman A, Dekens JA, Brandsma E, Marczynska J, Imhann F, Weersma RK, Franke L, Poon TW, Xavier RJ, Gevers D, Hofker MH, Wijmenga C, Zhernakova A. The Gut Microbiome Contributes to a Substantial Proportion of the Variation in Blood Lipids. Circ Res. 2015;117:817–824. doi: 10.1161/CIRCRESAHA.115.306807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ghazalpour A, Cespedes I, Bennett BJ, Allayee H. Expanding role of gut microbiota in lipid metabolism. Curr Opin Lipidol. 2016;27:141–147. doi: 10.1097/MOL.0000000000000278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schoeler M, Caesar R. Dietary lipids, gut microbiota and lipid metabolism. Rev Endocr Metab Disord. 2019;20:461–472. doi: 10.1007/s11154-019-09512-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Magkos F, Mittendorfer B. Gender differences in lipid metabolism and the effect of obesity. Obstet Gynecol Clin North Am. 2009;36:245–265, vii. doi: 10.1016/j.ogc.2009.03.001. [DOI] [PubMed] [Google Scholar]
- 30.Palmisano BT, Zhu L, Eckel RH, Stafford JM. Sex differences in lipid and lipoprotein metabolism. Mol Metab. 2018;15:45–55. doi: 10.1016/j.molmet.2018.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Imamura F, Micha R, Khatibzadeh S, Fahimi S, Shi P, Powles J, Mozaffarian D Global Burden of Diseases Nutrition and Chronic Diseases Expert Group (NutriCoDE) Dietary quality among men and women in 187 countries in 1990 and 2010: a systematic assessment. Lancet Glob Health. 2015;3:e132–e142. doi: 10.1016/S2214-109X(14)70381-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kim YS, Unno T, Kim BY, Park MS. Sex Differences in Gut Microbiota. World J Mens Health. 2020;38:48–60. doi: 10.5534/wjmh.190009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zheng W, Ma Y, Zhao A, He T, Lyu N, Pan Z, Mao G, Liu Y, Li J, Wang P, Wang J, Zhu B, Zhang Y. Compositional and functional differences in human gut microbiome with respect to equol production and its association with blood lipid level: a cross-sectional study. Gut Pathog. 2019;11:20. doi: 10.1186/s13099-019-0297-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xia MF, Chen Y, Lin HD, Ma H, Li XM, Aleteng Q, Li Q, Wang D, Hu Y, Pan BS, Li XJ, Li XY, Gao X. A indicator of visceral adipose dysfunction to evaluate metabolic health in adult Chinese. Sci Rep. 2016;6:38214. doi: 10.1038/srep38214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Joint Committee on Revision of guidelines for prevention and treatment of dyslipidemia in Chinese adults. Guidelines for Prevention and Treatment of Dyslipidemia in Chinese Adults (Revised Edition 2016) Zhongguo Xunhuan Zazhi. 2016;31:937–953. [Google Scholar]
- 36.Zhu X, Li Y, Jiang Y, Zhang J, Duan R, Liu L, Liu C, Xu X, Yu L, Wang Q, Xiong F, Ni C, Xu L, He Q. Prediction of Gut Microbial Community Structure and Function in Polycystic Ovary Syndrome With High Low-Density Lipoprotein Cholesterol. Front Cell Infect Microbiol. 2021;11:665406. doi: 10.3389/fcimb.2021.665406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Franzosa EA, Morgan XC, Segata N, Waldron L, Reyes J, Earl AM, Giannoukos G, Boylan MR, Ciulla D, Gevers D, Izard J, Garrett WS, Chan AT, Huttenhower C. Relating the metatranscriptome and metagenome of the human gut. Proc Natl Acad Sci U S A. 2014;111:E2329–E2338. doi: 10.1073/pnas.1319284111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Louca S, Parfrey LW, Doebeli M. Decoupling function and taxonomy in the global ocean microbiome. Science. 2016;353:1272–1277. doi: 10.1126/science.aaf4507. [DOI] [PubMed] [Google Scholar]
- 39.Razavi AC, Potts KS, Kelly TN, Bazzano LA. Sex, gut microbiome, and cardiovascular disease risk. Biol Sex Differ. 2019;10:29. doi: 10.1186/s13293-019-0240-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Markle JG, Frank DN, Mortin-Toth S, Robertson CE, Feazel LM, Rolle-Kampczyk U, von Bergen M, McCoy KD, Macpherson AJ, Danska JS. Sex differences in the gut microbiome drive hormone-dependent regulation of autoimmunity. Science. 2013;339:1084–1088. doi: 10.1126/science.1233521. [DOI] [PubMed] [Google Scholar]
- 41.Castaner O, Goday A, Park YM, Lee SH, Magkos F, Shiow STE, Schröder H. The Gut Microbiome Profile in Obesity: A Systematic Review. Int J Endocrinol. 2018;2018:4095789. doi: 10.1155/2018/4095789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cotillard A, Kennedy SP, Kong LC, Prifti E, Pons N, Le Chatelier E, Almeida M, Quinquis B, Levenez F, Galleron N, Gougis S, Rizkalla S, Batto JM, Renault P ANR MicroObes consortium, Doré J, Zucker JD, Clément K, Ehrlich SD. Dietary intervention impact on gut microbial gene richness. Nature. 2013;500:585–588. doi: 10.1038/nature12480. [DOI] [PubMed] [Google Scholar]
- 43.Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto JM, Kennedy S, Leonard P, Li J, Burgdorf K, Grarup N, Jørgensen T, Brandslund I, Nielsen HB, Juncker AS, Bertalan M, Levenez F, Pons N, Rasmussen S, Sunagawa S, Tap J, Tims S, Zoetendal EG, Brunak S, Clément K, Doré J, Kleerebezem M, Kristiansen K, Renault P, Sicheritz-Ponten T, de Vos WM, Zucker JD, Raes J, Hansen T MetaHIT consortium, Bork P, Wang J, Ehrlich SD, Pedersen O. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541–546. doi: 10.1038/nature12506. [DOI] [PubMed] [Google Scholar]
- 44.Karlsson FH, Tremaroli V, Nookaew I, Bergström G, Behre CJ, Fagerberg B, Nielsen J, Bäckhed F. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature. 2013;498:99–103. doi: 10.1038/nature12198. [DOI] [PubMed] [Google Scholar]
- 45.Amato MC, Giordano C, Galia M, Criscimanna A, Vitabile S, Midiri M, Galluzzo A AlkaMeSy Study Group. Visceral Adiposity Index: a reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care. 2010;33:920–922. doi: 10.2337/dc09-1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gao Z, Yin J, Zhang J, Ward RE, Martin RJ, Lefevre M, Cefalu WT, Ye J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes. 2009;58:1509–1517. doi: 10.2337/db08-1637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hatayama H, Iwashita J, Kuwajima A, Abe T. The short chain fatty acid, butyrate, stimulates MUC2 mucin production in the human colon cancer cell line, LS174T. Biochem Biophys Res Commun. 2007;356:599–603. doi: 10.1016/j.bbrc.2007.03.025. [DOI] [PubMed] [Google Scholar]
- 48.Louis P, Flint HJ. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol Lett. 2009;294:1–8. doi: 10.1111/j.1574-6968.2009.01514.x. [DOI] [PubMed] [Google Scholar]
- 49.Karlsson FH, Fåk F, Nookaew I, Tremaroli V, Fagerberg B, Petranovic D, Bäckhed F, Nielsen J. Symptomatic atherosclerosis is associated with an altered gut metagenome. Nat Commun. 2012;3:1245. doi: 10.1038/ncomms2266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Suharja AAS, Henriksson A, Liu SQ. Impact of Saccharomyces cerevisiae on viability of probiotic Lactobacillus rhamnosus in fermented milk under ambient conditions. J Food Process Preserv. 2014;38:326–337. [Google Scholar]
- 51.An HM, Park SY, Lee DK, Kim JR, Cha MK, Lee SW, Lim HT, Kim KJ, Ha NJ. Antiobesity and lipid-lowering effects of Bifidobacterium spp. in high fat diet-induced obese rats. Lipids Health Dis. 2011;10:116. doi: 10.1186/1476-511X-10-116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Huang ZR, Chen M, Guo WL, Li TT, Liu B, Bai WD, Ai LZ, Rao PF, Ni L, Lv XC. Monascus purpureus-fermented common buckwheat protects against dyslipidemia and non-alcoholic fatty liver disease through the regulation of liver metabolome and intestinal microbiome. Food Res Int. 2020;136:109511. doi: 10.1016/j.foodres.2020.109511. [DOI] [PubMed] [Google Scholar]
- 53.Yoo SR, Kim YJ, Park DY, Jung UJ, Jeon SM, Ahn YT, Huh CS, McGregor R, Choi MS. Probiotics L. plantarum and L. curvatus in combination alter hepatic lipid metabolism and suppress diet-induced obesity. Obesity (Silver Spring) 2013;21:2571–2578. doi: 10.1002/oby.20428. [DOI] [PubMed] [Google Scholar]
- 54.Zhu Y, Zhang JY, Wei YL, Hao JY, Lei YQ, Zhao WB, Xiao YH, Sun AD. The polyphenol-rich extract from chokeberry (Aronia melanocarpa L.) modulates gut microbiota and improves lipid metabolism in diet-induced obese rats. Nutr Metab (Lond) 2020;17:54. doi: 10.1186/s12986-020-00473-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cui C, Shen CJ, Jia G, Wang KN. Effect of dietary Bacillus subtilis on proportion of Bacteroidetes and Firmicutes in swine intestine and lipid metabolism. Genet Mol Res. 2013;12:1766–1776. doi: 10.4238/2013.May.23.1. [DOI] [PubMed] [Google Scholar]
- 56.Emoto T, Yamashita T, Sasaki N, Hirota Y, Hayashi T, So A, Kasahara K, Yodoi K, Matsumoto T, Mizoguchi T, Ogawa W, Hirata K. Analysis of Gut Microbiota in Coronary Artery Disease Patients: a Possible Link between Gut Microbiota and Coronary Artery Disease. J Atheroscler Thromb. 2016;23:908–921. doi: 10.5551/jat.32672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yoshida N, Emoto T, Yamashita T, Watanabe H, Hayashi T, Tabata T, Hoshi N, Hatano N, Ozawa G, Sasaki N, Mizoguchi T, Amin HZ, Hirota Y, Ogawa W, Yamada T, Hirata KI. Bacteroides vulgatus and Bacteroides dorei Reduce Gut Microbial Lipopolysaccharide Production and Inhibit Atherosclerosis. Circulation. 2018;138:2486–2498. doi: 10.1161/CIRCULATIONAHA.118.033714. [DOI] [PubMed] [Google Scholar]
- 58.Kelly TN, Bazzano LA, Ajami NJ, He H, Zhao J, Petrosino JF, Correa A, He J. Gut Microbiome Associates With Lifetime Cardiovascular Disease Risk Profile Among Bogalusa Heart Study Participants. Circ Res. 2016;119:956–964. doi: 10.1161/CIRCRESAHA.116.309219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hendijani F, Akbari V. Probiotic supplementation for management of cardiovascular risk factors in adults with type II diabetes: A systematic review and meta-analysis. Clin Nutr. 2018;37:532–541. doi: 10.1016/j.clnu.2017.02.015. [DOI] [PubMed] [Google Scholar]
- 60.Chapman CM, Gibson GR, Rowland I. Health benefits of probiotics: are mixtures more effective than single strains? Eur J Nutr. 2011;50:1–17. doi: 10.1007/s00394-010-0166-z. [DOI] [PubMed] [Google Scholar]
- 61.Makki K, Deehan EC, Walter J, Bäckhed F. The Impact of Dietary Fiber on Gut Microbiota in Host Health and Disease. Cell Host Microbe. 2018;23:705–715. doi: 10.1016/j.chom.2018.05.012. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The raw data supporting the conclusions of this article available from the corresponding author at lvjing-1219@163.com.