Abstract
Background
Although many transplant programs have been forced to suspend living donor transplants due to the emergence of coronavirus disease (COVID‐19), there are relatively few real‐time databases to assess center‐level transplant activities. We aimed to delineate the actual impact of COVID‐19 on living donor transplant programs and the resumption process in Japan.
Methods
In a nationwide survey, questionnaires were sent to 32 liver transplant programs that had performed at least more than one case of living donor liver transplantation in 2019 and 132 kidney transplant programs that had performed more than one living donor kidney transplantation in 2018.
Results
Thirty‐one (96.9%) and 125 (94.7%) liver and kidney transplant programs responded, respectively. In the early pandemic period, 67.7% (21/31) of liver programs and 29.8% (37/125) of kidney programs were able to maintain transplant activities similar to those during the pre‐pandemic period. After temporal suspension, 58.1% of kidney programs resumed their transplant activity after the number of local COVID‐19 cases peaked. Establishing institutional COVID‐19 screening, triage, and therapeutic management protocols was mandatory to resume transplant activity for 64.5% and 67.7% of liver and kidney programs, respectively. In the future wave of COVID‐19, 67.7% of liver programs would be affected by institutional COVID‐19 intensive care unit‐bound patient numbers, and 55.7% of kidney programs would stop if hospital‐acquired severe acute respiratory syndrome coronavirus‐2 (SARS‐CoV‐2) infection spreads.
Conclusions: This nationwide survey revealed for the first time how living donor liver and kidney
transplant programs changed in response to the COVID‐19 pandemic in a country where living donor transplantations are predominant.
Keywords: COVID‐19, living donor kidney transplantation, living donor liver transplantation
- COVID‐19
coronavirus disease
- DDKT
deceased donor kidney transplantation
- DDLT
deceased donor liver transplantation
- JST
Japan Society for Transplantation
- LDKT
living donor kidney transplantation
- LDLT
living donor liver transplantation
- SARS‐CoV‐2
severe acute respiratory syndrome coronavirus‐2
1. INTRODUCTION
Severe acute respiratory syndrome coronavirus‐2 (SARS‐CoV‐2) was identified in December 2019 and was subsequently determined to be the cause of coronavirus disease (COVID‐19). 1 , 2 The risk of death from COVID‐19 is higher among immunosuppressed patients than in the general population. 3 Accordingly, elective surgeries, including living donor transplant procedures, were paused in many countries. 4 , 5 It is important to note that organ transplantation in Japan depends heavily on living donors due to a shortage of deceased donors. Responding to the inquiries from multiple transplant programs, the Japan Society for Transplantation (JST) published a set of guidelines on March 6, 2020 (version 1). In the guidelines, the JST recommended continuing transplants for immediate life‐saving organs such as the heart, lung, and liver (for fulminant liver failure) after performing a risk–benefit assessment for each case. Nonurgent transplants of the kidney, pancreas, and bowel, as well as all nonlife‐threatening living donor transplantations, were recommended to be postponed. For urgent living donor transplants, including living donor liver transplantation (LDLT), preventative measures such as 14 days of home or inpatient quarantine and a COVID‐19 screening test for both donors and recipients were recommended. Many transplant programs have suspended their living donor transplants in response to the guidelines in Japan.
As the first wave of COVID‐19 peaked, the JST announced the fourth edition of the guidelines on May 29, 2020 (version 4) and proposed a checklist to help determine the appropriate time to resume living donor kidney transplantation (LDKT) programs. These guidelines also recommended resuming living donor transplant activities based on the local community and institutional transmission risk. For living donor liver and kidney transplant donors and recipients, preventive measures, such as 14 days of home or inpatient quarantine and COVID‐19 polymerase chain reaction (PCR) tests, were recommended. Most recently, the JST announced the fifth edition of the guidelines on November 8, 2021 (version 5) and proposed a checklist to help determine de novo transplantation for both living donors and recipients who recovered from COVID‐19.
Each transplant facility faced a difficult situation in which they had to make decisions individually to resume or postpone transplant surgeries. Since there are relatively few real‐time databases to assess center‐level transplant activities and no evidence‐based guidelines for the management of transplant patients (especially from living donors in Japan), communication of knowledge is vital. 6
This study aimed to understand the impact of COVID‐19 on transplant activities across Japan and to explore center‐level variations in activity, clinical practice, testing, and policies. Through this study, we examined how living donor transplant programs in Japan were previously suspended or continued; this information is critical for the maintenance of programs and for the future if COVID‐19 re‐spikes.
2. MATERIALS AND METHODS
The survey was conducted from October 20, 2020, to November 30, 2020. Questionnaires were constituted only for this survey. The surveys were reviewed by the project members of the grant and adapted based on the feedback. Questionnaires were sent via e‐mail to 32 liver transplant programs that had performed at least more than one case of LDLT in 2019 and 132 kidney transplant programs that had performed more than one LDKT in 2018. Data were collected from 96.9% (31/32) of the LDLT programs and 94.7% (125/132) of the LDKT programs. One answer from the two LDKT programs (Hirosaki University and Oyokyo Kidney Research Institute Hirosaki Hospital, run by the same team) was counted as one program for the analysis. The questionnaire consisted of four sections: (1) institutional transplant activities, (2) influence of COVID‐19 on the institutional transplantation program, (3) institutional transplant outpatient practices, and (4) institutional COVID‐19 treatment practices (Table S1).
Section 1 focused on the number of LDLT, LDKT, deceased donor liver transplantation (DDLT), and deceased donor kidney transplantations (DDKT) performed in 2017, 2018, and 2019, as well as the number of LDLT, LDKT, DDLT, and DDKT performed per month in 2020.
Section 2 focused on the present status of the institutional transplant program at the time of the survey (October 2020). The survey asked who approved the institutional transplant activities (institution, department, or government) during the pandemic, if there were any adverse effects on the recipients by resuming the transplant program, how to manage future transplant programs and the preoperative COVID‐19 preventative measures for both donors and recipients.
Section 3 focused on outpatient management. Specifically, items about the recipient routine check‐up list, number of donor/recipient outpatients, number of patients who delayed their routine visits, and institutional measures taken to reduce the risk of COVID‐19 in outpatient clinics were included.
Section 4 focused on the hospital's experience in dealing with COVID‐19. Items about center‐level COVID‐19 treatment systems were included.
An additional survey was performed to reveal the COVID‐19 test status of the donors and the recipients who underwent transplantation during the survey period. Twenty‐eight (85.7%) liver transplant programs and 111 (89.5%) kidney transplant programs responded to the additional survey.
On April 7, 2020, to prevent the spread of COVID‐19, a state of emergency was declared in seven prefectures (Saitama, Chiba, Tokyo, Kanagawa, Osaka, Hyogo, and Fukuoka), which had an increasing number of new cases. On April 16, six prefectures (Hokkaido, Ibaraki, Ishikawa, Gifu, Aichi, and Kyoto) were added. These 13 prefectures either had a high population density and high levels of traffic or had clusters of COVID‐19. In general, for living donor transplantation programs in Japan, recipients and donors are from the same or neighboring prefectures, except in pediatric cases. We categorized the transplantation programs into two groups based on their location: red zone—transplant programs located in the 13 prefectures where the state of emergency was declared (Figure 1A), and blue zone—the remaining 34 prefectures where a state of emergency was not issued. Based on this definition, 58.1% of the transplantation programs were categorized as the red zone group, and the remaining 41.9% were categorized as the blue zone group (Figure 1B).
FIGURE 1.
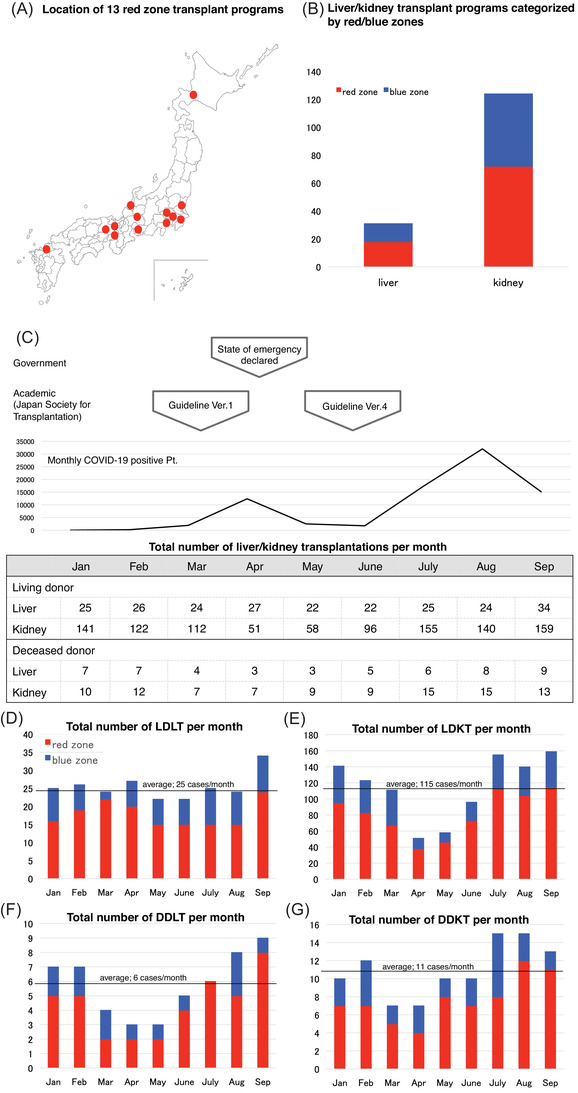
(A) The 13 prefectures where the state of emergency was declared in Japan on April 7 and 16, 2020 to prevent the spread of coronavirus disease (COVID‐19) (Saitama, Chiba, Tokyo, Kanagawa, Osaka, Hyogo, Fukuoka, Hokkaido, Ibaraki, Ishikawa, Gifu, Aichi, and Kyoto). (B) Red zone: transplant programs located in the 13 prefectures where the state of emergency was issued. Blue zone: the remaining 34 prefectures where the state of emergency was not issued. (C) The monthly case numbers of liver and kidney transplantations in 2020 along with the timing of when the state of emergency was declared; the first and fourth versions of the guidelines were published by the Japan Society for Transplantation (JST). In version 1, the JST recommended continuing transplants for life‐saving organs such as the heart, lungs, and status 1 livers, requiring a risk–benefit assessment for each case. The JST recommended postponing any nonurgent transplants of the kidney, pancreas, and bowel, as well as all nonlife‐threatening living donor transplantations. In version 4, the JST recommended resuming living donor transplant programs based on the local community and institutional transmission risk. They also proposed a checklist to help determine the restart of the living donor kidney transplantation (LDKT) programs. The JST guidelines are summarized in Table S2. (D)–(G) The monthly case numbers of liver and kidney transplantations in 2020 categorized by blue and red zone groups. The straight line indicates the average transplantation case number during the respective period. (D) Living donor liver transplantation (LDLT); (E) deceased donor liver transplantation (DDLT); (F) LDKT; (G) deceased donor kidney transplantation (DDKT). Blue zones indicate prefectures where emergency measures were not implemented; red zones indicate prefectures where emergency measures were implemented
3. RESULTS
Answers from 31 LDLT and 124 LDKT programs were analyzed in the study.
3.1. Institutional transplantation activities
3.1.1. Transplantation numbers in the pre‐pandemic era
The yearly number of cases of living donor liver and kidney transplantations was 334/1402 in 2017, 343/1555 in 2018, and 312/1701 in 2019. From deceased donors, the number of yearly liver/kidney transplant cases was 63/127 in 2017, 59/121 in 2018, and 88/176 in 2019. Categorized by zone, 219 (70.2%) LDLTs, 66 (75.0%) DDLTs, 1192 (70.1%) LDKTs, and 135 (76.7%) DDKTs were performed in 2019 in the red zone.
3.2. Transplantation numbers in the pandemic era
Figure 1C shows the total number of transplantations per month from January to September 2020. Although LDLTs were routinely performed at 25 cases per month on average, the number of LDKTs dropped to 55% (an average of 115 cases per month) from March to June; the rate increased gradually thereafter. In total, 229 LDLTs and 1034 LDKTs were performed. The number of deceased donor transplantations decreased slightly at an earlier stage. On average, six cases of DDLTs and 11 cases of DDKTs were performed per month from January to September 2020. In total, 52 DDLTs and 97 DDKTs were performed. To examine the regional impact of COVID‐19, the total number of transplantations was sorted according to the red and blue zones. Figure 1D shows the total number of LDLT cases per month, which were routinely performed during the study period. Figure 1E shows the total number of DDLT cases per month. The total number decreased in March, April, and May, and a decrease was observed in both the red and blue zones. The number of DDLT cases increased in June. In total, 22 liver transplantations (18 LDLTs and four DDLTs) were performed per month in the red zone, whereas only nine (eight LDLTs and one DDLT) were performed in the blue zone. Throughout the pandemic era, 161 (70.3%) LDLTs and 39 (75.0%) DDLTs were performed in the red zone. Figure 1F,G shows the total number of kidney transplantation cases per month. The number of LDKT cases decreased drastically in April and May and that of DDKT cases decreased in March and April. A decrease was observed in both the red and blue zones. The number of LDKT started to increase in June and DDKT in May. In total, 89 kidney transplantations (81 LDKTs and eight DDKTs) were performed per month in the red zone, and 36 (33 LDKTs and three DDKTs) were performed per month in the blue zone. Throughout the pandemic era, 759 (73.4%) LDKTs and 71 (73.2%) DDKTs were performed in the red zone. In short, although the number of cases of LDLT did not change, DDLT and kidney transplantation decreased in the pandemic era.
3.3. Influence of COVID‐19 on institutional transplantation programs
3.3.1. Institutional transplantation activities
Twenty‐seven (87.1%) LDLT and 117 (94.4%) LDKT programs followed the guidelines proposed by the JST on the management of COVID‐19. In addition, 110 (88.7%) LDKT programs referred to the checklist in the JST guidelines. Table 1 shows the institutional transplantation activities at the end of October 2020. Although almost 70% of the LDLT programs could continue transplant activities without any regulations, almost 60% of the LDKT programs resumed after the number of local COVID‐19 patients peaked. Most transplant programs were approved by either an institutional or departmental board. As an indicator of urgent LDLT, the Model for End‐Stage Liver Disease score was used, ranging from 15 to 30 (average, 22). Acute liver failure (n = 29) and pediatric transplantation (n = 14) were also evaluated to determine the urgency of LDLT. For LDKT, difficulty in creating vascular access (n = 48), preemptive cases (n = 53), pediatric cases (n = 36), and marginal donors (n = 26) were considered factors for immediate transplantation. Although most of the transplant programs indicated that interruption of transplant programs did not impact the awaiting donors and recipients, death of the recipient during the waiting time (n = 1), and deterioration of patient condition beyond the indication (n = 2) were reported from LDLT programs, and patient death due to acute myocardial infarction (n = 1), deterioration of patient condition (n = 3), and unexpected temporal introduction of hemodialysis (n = 16) were reported from LDKT programs.
TABLE 1.
Institutional transplantation activities
| Liver | Kidney | |
|---|---|---|
| Institutional activity on October 2020 | ||
| Without any regulation | 67.7% | 29.8% |
| Only urgent/selected transplantation | 19.4% | 6.5% |
| Resume after local COVID‐19 peak out | 9.7% | 58.1% |
| Pause | 3.2% | 5.6% |
| Institutional approval for transplantation activities | ||
| Institutional board | 61.3% | 52.4% |
| Department board | 67.7% | 68.6% |
| Government guidelines | 0% | 1.6% |
Abbreviation: COVID‐19, coronavirus disease.
Table 2 indicates the reasons that led to institutional transplant resumption. Most of the transplant programs were resumed because institutional COVID‐19‐related treatment and the screening system were established. Additional analyses were performed to examine the regional impacts of COVID‐19. Tables 3 and 5 show institutional liver/kidney transplantation activities, and Tables 4 and 6 indicate the reasons for resuming liver/kidney transplant programs categorized by red and blue zones. Although liver transplant programs could continue without any regulations regardless of the zones, kidney transplant programs in the red zone resumed once there was no institutional COVID‐19 spread, and the blue zone groups resumed once the regional SARS‐CoV‐2 infection status improved.
TABLE 2.
Reasons to resume the institutional transplant program
| Liver | Kidney | |
|---|---|---|
| Institutional | ||
| Operation restriction was released | 22.6% | 26.6% |
| COVID‐19 treatment system was constituted | 64.5% | 67.7% |
| COVID‐19 infection status was improved | 25.8% | 25.0% |
| No COVID‐19 spread | 51.6% | 42.7% |
| Regional | ||
| COVID‐19 infection status was improved | 41.9% | 46.0% |
Abbreviation: COVID‐19, coronavirus disease.
TABLE 3.
Institutional liver transplantation activities categorized by zones
| Red zone | Blue zone | |
|---|---|---|
| Institutional activity on October 2020 | ||
| Without any regulation | 72.2% | 61.5% |
| Only urgent/selected transplantation | 16.7% | 23.1% |
| Resume after local COVID‐19 peak out | 11.1% | 7.7% |
| Pause | 0% | 7.7% |
Abbreviation: COVID‐19, coronavirus disease.
TABLE 5.
Institutional kidney transplantation activities categorized by zones
| Red zone | Blue zone | |
|---|---|---|
| Institutional activity on October 2020 | ||
| Without any regulation | 29.2% | 30.1% |
| Only urgent/selected transplantation | 8.3% | 3.8% |
| Resume after local COVID‐19 peak out | 58.3% | 57.7% |
| Pause | 4.2% | 7.7% |
Abbreviation: COVID‐19, coronavirus disease.
TABLE 4.
Reasons to resume the institutional liver transplant program categorized by zones
| Red zone | Blue zone | |
|---|---|---|
| Institutional | ||
| Operation restriction was released | 11.1% | 23.1% |
| COVID‐19 treatment system was constituted | 66.7% | 61.5% |
| COVID‐19 infection status was improved | 33.3% | 30.8% |
| No COVID‐19 spread | 44.4% | 46.2% |
| Regional | ||
| COVID‐19 infection status was improved | 22.2% | 69.2% |
Abbreviation: COVID‐19, coronavirus disease.
TABLE 6.
Reasons to resume the institutional kidney transplant program
| Red zone | Blue zone | |
|---|---|---|
| Institutional | ||
| Operation restriction was released | 30.6% | 21.2% |
| COVID‐19 treatment system was constituted | 70.8% | 63.5% |
| COVID‐19 infection status was improved | 31.9% | 26.9% |
| No COVID‐19 spread | 48.6% | 25.0% |
| Regional | ||
| COVID‐19 infection status was improved | 34.7% | 59.6% |
Abbreviation: COVID‐19, coronavirus disease.
Table 7 shows the items that would affect transplant activities if there were to be a future wave of COVID‐19. Although most of the LDLT programs said that they would depend on the COVID‐19 intensive care unit (ICU)‐bound patient number, LDKT programs said that they would stop if institutional transmission of COVID‐19 occurred. Tables 8 and 9 list the items that would affect transplant activities stratified by red and blue zones; these were not significantly different between the groups. In short, after the constitution of the COVID‐19‐related screening system, most of the LDLT programs could continue transplantation activities, and the number of transplantations that can be performed in the future would be affected by institutional COVID‐19 ICU‐bound patient numbers regardless of the facility location. Although LDKT programs in the red zone resumed as there was no institutional COVID‐19 spread and blue zone groups resumed as the regional SARS‐CoV‐2 infection status improved, future transplant activity would be stopped once institutional COVID‐19 infection appears.
TABLE 7.
Items that affect future transplant activities
| Liver | Kidney | |
|---|---|---|
| Stop once institutional COVID‐19 infection appears | 29.0% | 55.7% |
| Institutional COVID‐19 patient number | 35.5% | 42.0% |
| Institutional COVID‐19 ICU‐bound patient number | 67.7% | 40.3% |
| Will not stop transplant program | 22.6% | 8.9% |
Abbreviations: COVID‐19, coronavirus disease; ICU, intensive care unit.
TABLE 8.
Items that affect future liver transplant activities categorized by zones
| Red zone | Blue zone | |
|---|---|---|
| Stop once institutional COVID‐19 infection appears | 22.2% | 38.5% |
| Institutional COVID‐19 patient number | 33.3% | 38.5% |
| Institutional COVID‐19 ICU‐bound patient number | 55.6% | 92.3% |
| Will not stop transplant program | 27.8% | 7.7% |
Abbreviations: COVID‐19, coronavirus disease; ICU, intensive care unit.
TABLE 9.
Items that affect future kidney transplant activities categorized by zones
| Red zone | Blue zone | |
|---|---|---|
| Stop once institutional COVID‐19 infection appears | 54.2% | 51.9% |
| Institutional COVID‐19 patient number | 43.1% | 44.2% |
| Institutional COVID‐19 ICU‐bound patient number | 41.7% | 38.5% |
| Will not stop transplant program | 9.7% | 7.7% |
Abbreviations: COVID‐19, coronavirus disease; ICU, intensive care unit.
3.3.2. Preoperative check‐up system for donors and recipients
Table 10 shows the preoperative check‐up systems for the donors and recipients. A preoperative COVID‐19 screening test was performed in almost 90% of transplant programs for both recipients and donors. A nasopharyngeal swab was mainly used for test samples, and as a COVID‐19 screening test, more than 90% of the transplant programs used reverse transcription polymerase chain reaction (RT‐PCR. Computed tomography (CT) chest scans for the recipients were routinely performed in 83.9% of the LDLT and 65.3% of the LDKT programs, and for donors, the proportion decreased to 54.8% and 58.9%, respectively. More than 60% of the transplant programs set the preoperative self‐quarantine period for the recipients as 14 days. In terms of the preoperative isolation period for donors, almost 50% of the transplant programs set the period as 14 days.
TABLE 10.
Preoperative check‐up system for donors and recipients
| Liver | Kidney | |
|---|---|---|
| COVID‐19 preoperative screening test | ||
| Recipient | 93.6% | 87.9% |
| Donor | 90.3% | 86.3% |
| COVID‐19 test samples | ||
| Nasopharyngeal swab | 71.0% | 56.5% |
| Nasal swab | 25.8% | 21.8% |
| Sputum | 3.2% | 3.2% |
| Saliva | 19.4% | 32.3% |
| COVID‐19 screening test | ||
| RT‐PCR | 96.8% | 93.6% |
| Qualitive antigen test | 3.2% | 12.1% |
| Quantitative antigen test | 3.2% | 6.5% |
| Antibody test | 0% | 3.2% |
| Chest CT scan for the recipient | ||
| Routinely performed | 83.9% | 65.3% |
| Performed initially but stopped | 0% | 12.1% |
| Not performed | 16.1% | 22.6% |
| Chest CT scan for the donor | ||
| Routinely performed | 54.8% | 58.9% |
| Performed initially but stopped | 0% | 12.1% |
| Not performed | 45.2% | 29.0% |
| Preoperative self‐quarantine period for the recipient | ||
| 28 days | 3.2% | 3.2% |
| 14 days | 67.7% | 60.5% |
| 7 days | 0% | 12.9% |
| Not fixed | 29.0% | 23.4% |
| Preoperative self‐quarantine period for the donor | ||
| 28 days | 0% | 1.6% |
| 14 days | 48.4% | 53.2% |
| 7 days | 19.4% | 9.7% |
| Not fixed | 32.3% | 35.5% |
Abbreviations: COVID‐19, coronavirus disease; CT, computed tomography; RT‐PCR, reverse transcription polymerase chain reaction.
3.4. Institutional transplant outpatient practices
The usual check‐up list for the recipients included patients’ body weight, blood pressure, heart rate, body temperature, water intake, urinary volume (kidney transplant), adherence to medication, number of steps (liver transplant), and glucose level at outpatient clinics with modifications at each facility. In total, 5842 (average 195, 5–1000) liver transplant recipients and 3280 (average 106, 0–700) donors visited the outpatient clinic, whereas 20 451 (average 165, 10–2500) kidney transplant recipients and 10 365 (average 86, 0–700) donors visited the outpatient clinic. From January 1, 2020, to September 30, 2020, a total of 991 (average 32, 0–95) LDLT recipients and 5079 (average 41, 0–100) LDKT recipients postponed their consultation at the outpatient clinic. Most of the programs implemented several preventative measures to reduce COVID‐19 transmission risk, such as reducing the frequency of outpatient clinic visits, extending the interval for refilling prescriptions, reducing the frequency of tests, shortening the length of hospital stay, and minimizing contact between other patients. To reduce the frequency of outpatient visits, 38.7% of LDLT and 21.8% of LDKT programs conducted telemedicine. Moreover, 87% of LDLT and 66.9% of LDKT facilities utilized an application system to support remote consultations.
3.5. Institutional COVID‐19 treatment practices
The facilities of 27 (87.1%) LDLT and 106 (85.5%) LDKT programs had experience accepting COVID‐19 patients. The outpatient fever clinic was established in 27 (87.1%) LDLT and 99 (79.8%) LDKT programs. The general ward was used for COVID‐19 patients in 27 (87.1%) LDLT and 102 (82.3%) LDKT programs, and COVID‐19 patients were treated in the ICU in 29 (93.6%) LDLT and 84 (67.7%) LDKT programs. The routes for transplant and COVID‐19 patients were separated into 26 (83.9%) LDLT and 108 (87.1%) LDKT programs, and 31 (100%) LDLT and 112 (90.3%) LDKT programs had already established a collaboration system between infectious disease specialists and intensivists to treat transplant recipients who had COVID‐19. Table 11 shows the management of febrile transplant patients. More than half of the patients were referred to fever clinics or managed at transplant outpatient clinics. COVID‐19 testing was mainly performed if the disease was suspected after a medical history interview. Of the LDLT and LDKT programs, 40% and 30.7% performed chest CT if the recipients presented with cough, respectively.
TABLE 11.
Management of fever transplant patients
| Liver | Kidney | |
|---|---|---|
| Management of the patient | ||
| At fever clinic | 61.3% | 66.9% |
| At transplant clinic | 58.1% | 48.4% |
| COVID‐19 screening test | ||
| Routinely performed | 19.4% | 20.2% |
| Only suspicious cases | 83.9% | 66.1% |
| Chest CT | ||
| Routinely performed | 19.4% | 30.7% |
| Only with cough cases | 41.9% | 30.7% |
Abbreviations: COVID‐19, coronavirus disease; CT, computed tomography.
3.6. Impact of COVID‐19 on transplantation
Twenty‐eight (87.5%) liver transplant programs responded to the additional survey. Among the living donors and the recipients who underwent LDLT from January to September 2020, no patient was diagnosed with COVID‐19 before transplantation. After transplantation, one donor (3.57%) and four recipients (10.71%) were diagnosed with COVID‐19. Based on the national registry of the Japanese Liver Transplant Society, the 1‐year patient survival after LDLT was 85.4% and after DDLT was 89.2% during the pre‐COVID period (accessed at http://jlts.umin.ac.jp/images/annual/JLTSRegistry2019.pdf [in Japanese]). For the patients who underwent liver transplantation during the COVID‐19 era from January to September 2020, the 1‐year survival for LDLT was 90.2% and 88.1% for DDLT.
One hundred and eleven (89.5%) kidney transplant programs responded to the additional survey. There were no donors or recipients who were diagnosed with COVID‐19 before transplantation. After transplantation, one donor (0.92%) and 15 recipients (11.82%) were diagnosed with COVID‐19. Based on the national registry, the 1‐year patient survival was 99.5%, and graft survival was 98.8% in the pre‐COVID period. For the patients who underwent kidney transplantation during the COVID‐19 era from January to September 2020, the 1‐year patient survival was 99.8%, and the graft survival was 99.2%.
4. DISCUSSION
To the best of our knowledge, this is the first national survey that has been conducted to examine the impact of COVID‐19 on institutional living donor transplantation programs. We found that although most of the LDLT programs continued liver transplantation without any regulations, most of the LDKT programs were forced to pause; accordingly, the number of cases substantially decreased during this period. Most transplant programs resumed living donor transplant activities after constituting an institutional COVID‐19 treatment system. Almost 70% of transplantations were performed before and during the era of the COVID‐19 pandemic in areas where the state of emergency was declared. In areas where the state of emergency was not issued, the regional SARS‐CoV‐2 infection status had to be considered. In the case of a future COVID‐19 wave, liver transplant activity would continue if the number of ICU‐bound COVID‐19 patients was not too high, while kidney transplant activity would stop if institutional SARS‐CoV‐2 spread appeared.
Several systematic reviews and meta‐analyses have shown that transplant recipients have a higher risk of developing critical COVID‐19 illness due to chronic immunosuppression than the general population. 7 , 8 International and national registries showed a mortality rate between 19% and 32% among solid organ transplant recipients with COVID‐19.9–11 Regarding the clinical outcomes of liver transplant recipients with COVID‐19, international registries reported a mortality rate of 18%–19%. 12 , 13 In Japan, the Japanese registry by JST, accessed on February 8, 2021, reported that the case‐fatality rate for organ transplant recipients was 7.1% (accessed at http://square.umin.ac.jp/jst‐covid‐19 [in Japanese]). Based on the Johns Hopkins Coronavirus Resource Center accessed on February 11, 2021 (accessed at https://coronavirus.jhu.edu/data/mortality), the case‐fatality rate for the general population was 2.9% in the United Kingdom, 1.7% in the United States, 2.1% in Spain, 2.3% in France, and 1.6% in Japan. To date, some studies have concluded that there is no difference in overall mortality between the general population and solid organ transplant recipients, 14 , 15 and there is also an opinion that this conclusion needs further analysis because the study group was limited only to inpatients or ICU‐bound patients. 16 As LDLT and LDKT programs are performed in the presence of living donors, concrete policies to reduce the risk of COVID‐19 infections should be thoroughly discussed at each transplant center.
The response rate for this study was as high as ≥90%, implying a strong national need to better understand the impact of COVID‐19. In short, the results of the study can be summarized as follows: (1) although the LDKT program decreased drastically, the LDLT program was not affected by the emergence of COVID‐19; (2) transplant programs stopped once and restarted based on either institutional or department policies; (3) almost 20% of the transplanted recipients postponed their routine check‐up at the outpatient clinic; (4) more than 85% of transplant programs accepted COVID‐19 patients, and (5) short‐term patient/graft survival was not affected by the emergence of COVID‐19. In the raw data, there were some differences between liver and kidney transplant programs. In general, LDKT programs implemented more rigorous restrictions than LDLT programs. While most liver patients require urgent transplantation, kidney transplantation is an alternative option for renal replacement therapy. As resources outlining safety measures for COVID‐19 in the early phase of the pandemic were scarce, most kidney transplant programs were more cautious in avoiding potential harm to both recipients and donors. Moreover, the JST recommended postponing nonurgent kidney transplants. All these factors decreased the total number of kidney transplantations that were performed during the COVID‐19 era. However, a recent report from Belgium found that the cumulative incidence of COVID‐19 was 5.31% in hemodialysis patients, 1.82% in peritoneal dialysis patients, and 1.40% in kidney transplant patients, 17 suggesting that transplant patients have a decreased chance of contracting COVID‐19, possibly due to less frequent visits to the hospital compared to dialysis patients. Furthermore, delaying kidney transplantation by 1 year would decrease patient survival. 18 A risk–benefit analysis should be carefully performed for each patient considering the availability of resources, the intensity of the pandemic in the patient's region, and their comorbidities. 19 COVID‐19 preoperative screening was performed in more than 90% of transplant programs, as recommended in the JST guidelines (accessed at https://square.umin.ac.jp/jst‐covid‐19/images/guidance4.1.pdf [in Japanese]). The RT‐PCR assay was used in most cases; however, since RT‐PCR was not available in Japan during the early phases of the pandemic, less sensitive antigen assays were performed in two (6.4%) LDLT and 23 (18.6%) LDKT programs. Although RT‐PCR is highly sensitive, a single negative result is insufficient to exclude the diagnosis of COVID‐19 if the suspicion of COVID‐19 remains high. 20 Thus, the JST guidelines emphasize the importance of practices that minimize the risk of COVID‐19 exposure, including social distancing and self‐quarantine for 14 days before donation and transplantation for both donors and recipients. Our survey found that approximately 20% of transplant programs did not encourage self‐quarantine before donation or transplantation. Although the reasons why these measures were not implemented were unknown, pre‐transplant self‐quarantine may not be feasible for some donors and recipients, especially when they can still work before transplantation. In such cases, simple measures such as social distancing, universal masking, and frequent hand washing should be advised to minimize the risk of COVID‐19 during the pre‐transplant period. Importantly, there were no liver/kidney donors or recipients who were diagnosed with COVID‐19 before transplantation, thus supporting the JST guidelines of preoperative self‐quarantine.
A limitation of the study is that because this survey collected responses from transplant surgeons, the results may have been different if the survey answers were collected from referring physicians, as they evaluate transplant indication. Accordingly, the number of transplants is extensively influenced by the number of referred patients from physicians. Another limitation is that because the survey was conducted to clarify general practices and policies at each transplant program, patient‐level data on COVID‐19 treatments or changes in immunosuppressant regimens were not collected. In the study, more transplants occurred in the red zone than in the blue zone. Based on the Japan Statistical Yearbook 2022, the total population of 13 red zone prefectures was 77 million, which was almost 61.6% of the total Japanese population as of December 1, 2020 (accessed at https://www.stat.go.jp/English/data/nenkan/71nenkan/index.html). Transplant centers and universities were historically established in the area, which had a large population and convenient location. Judging from the fact that almost 70% of transplantations were already performed in the red zone before the era of the COVID‐19 pandemic, the case number bias was caused by the definition of the red zone itself that these areas had a high population density and high levels of traffic or had clusters of COVID‐19.
Based on the Japanese Ministry of Health, Labor, and Welfare data, the actual ICU utilization rate ranged from approximately 70% to 75% during the survey period (https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/0000121431_00180.html). As the ICU utilization rate itself does not reflect the availability of ICUs for transplant patients, we focused on the monthly number of newly introduced respirators and extracorporeal membrane oxygenation (ECMO). Based on the cross‐ICU searchable information system data, the monthly number of newly introduced respirators ranged from 20 (in June) to 393 (in April), and the monthly number of newly introduced ECMO ranged from 6 (in June) to 110 (in April) (https://www.ecmonet.jp/crisis). These data suggest that although the actual ICU utilization rate did not drastically change, ICU‐bound patient disease distribution shifted to COVID‐19. Despite the negative surroundings, the total number of LDLTs was preserved, which was the result of an excellent team effort of all concerned with transplantation.
In conclusion, this nationwide survey in Japan revealed that transplant programs for LDKT decreased drastically at one point and recovered after institutional resumption policies were implemented. Most of the transplant programs complied with the JST guidelines, including performing preoperative COVID‐19 tests and promoting self‐quarantine for both donors and recipients, while also considering the regional COVID‐19 infection status. Although vaccination and therapeutics were introduced after the survey era, how the living donor transplant program was modified in Japan at the early phase of the pandemic was elucidated through the study. There were no donors/recipients who were diagnosed with COVID‐19 before transplantation, thus affirming self‐quarantine. The maintained LDLT case numbers with shifted COVID‐19 ICU surroundings suggested efforts by the transplant team with the ICU team to protect end‐stage liver disease patients. Supported by preserved transplant patient survival during the era of COVID‐19, this study provides valuable information to transplant facilities on how to prepare for future pandemics so that we can ensure that such living donor transplant programs continue unhindered.
CONFLICT OF INTEREST
The authors declare no conflicts of interest.
AUTHOR CONTRIBUTIONS
All authors contributed to the manuscript and fulfilled criteria as per the uniform requirements set forth by the International Committee of Medical Journal Editors (ICJME) guidelines. All authors have reviewed and approved the final version of the manuscript.
Supporting information
Supplemental TABLE S1. Questionnaires sent to transplant programs
Supplemental TABLE S2. Guidelines for transplant medicine from the Japan Society for Transplantation
ACKNOWLEDGMENTS
This study was supported by grants from the Ministry of Health, Labor, and Welfare in Japan. We are grateful to our colleagues at the transplant institutions who responded to the questionnaires and made this study possible. For the revised analysis, Dr. Koji Umeshita (Osaka University, Osaka) and Dr. Yuki Nakagawa (Juntendo University, Tokyo) performed the national data analysis. We are also grateful to Mrs. Mine Yamanaga, who helped edit the entire manuscript. This study was supported by grants from the Ministry of Health, Labor, and Welfare in Japan.
LDLT facilities: Okinawa Chubu Hospital (Dr. Murakami), Fujita Health University (Dr. Suzuki), Kanagawa Children's Medical Center (Dr. Shinkai), Kyoto University (Dr. Ito), Nagasaki University (Dr. Soyama), Kumamoto University (Dr. Shimata), Chiba University (Dr. Otsuka), Hiroshima University (Dr. Ohira), Tokyo Medical University (Dr. Kawachi), Okayama University (Dr. Yagi), Saitama Medical Center (Dr. Maki), Iwate Medical University (Dr. Takahara), Tokyo Women's Medical University (Dr. Egawa), Mie University (Dr. Tanemura), Hokkaido University (Dr. Goto), Tokyo University (Dr. Akamatsu), Jichi Medical University (Dr. Sakuma), Nagoya University (Dr. Ogura), Osaka University (Dr. Goto), Asahikawa Medical University (Dr. Furukawa), Yamaguchi University (Dr. Nagano), Tohoku University (Dr. Miyagi), Kyushu University (Dr. Yoshizumi), National Center for Child Health and Development (Dr. Kasahara), Tokyo Jikei University School of Medicine (Dr. Ikegami), Shinshu University (Dr. Soejima), Saitama Children's Medical Center (Dr. Mizuta), Ehime University (Dr. Takada), Fukushima Medical University (Dr. Maruhashi), Keio University (Dr. Hasegawa), and Kobe University.
LDKT facilities: Nagano Red Cross Hospital (Dr. Imao), Yokkaichi Municipal Hospital (Dr. Okumura), University Hospital Kyoto Prefectural University of Medicine (Dr. Ushigome), Tokyo Dental College Ichikawa General Hospital (Dr. Kohno), Nagoya University (Dr. Kato), Okayama Medical Center (Dr. Fujiwara), Kinki University (Dr. Nose), Kawashima Hospital (Dr. Minakuchi), Toyama Prefectural Central Hospital (Dr. Seto), Toyooka Public Hospital (Dr. Machida), Kanazawa Medical University Hospital (Dr. Tanaka), Kure Medical Center (Dr. Tashiro), Hidaka Hospital (Dr. Soeno), Juntendo University Urayasu Hospital (Dr. Nosaki), Kitano Hospital (Dr. Ikeuchi), Okinawa Chubu Hospital (Dr. Shimabukuro), Kagawa University (Dr. Taoka), Yamaguchi University (Dr. Isoyama), Edogawa Hospital (Dr. Koga), Miyazaki University (Dr. Uemura), Ehime Prefectural Central Hospital (Dr. Okamoto), Saga‐ken Medical Center Koseikan (Dr. Tokuda), Iwate Medical University (Dr. Sugimura), Showa University (Dr. Yoshitake), Ota Memorial Hospital (Dr. Mizutani), Tokyo Women's Medical University (Dr. Unagami), Jikei University School of Medicine (Dr. Yamamoto), National Defense Medical College Hospital (Dr. Tasaki), Niigata University (Dr. Saito), Masuko Memorial Hospital (Dr. Uchida), Joban Hospital (Dr. Shinmura), Sumitomo Hospital (Dr. Ichimaru), Tokyo University (Dr. Yamada), Yuuai Medical Center (Dr. Ota), Akita University (Dr. Sato), Toranomon Hospital (Dr. Nakamura), Tokyo Medical University (Dr. Iwamoto), Kochi Health Sciences Center (Dr. Shibuya), Osaka Medical College Hospital (Dr. Hirano), Japan Community Health care Organization Sendai Hospital (Dr. Haga), Sapporo Hokuyu Hospital (Dr. Miura), Okayama University (Dr. Araki), Komaki City Hospital (Dr. Uehira), Osaka City General Hospital (Dr. Asai), Shizuoka Children's Hospital (Dr. Kitayama), Kansai Medical University Hospital (Dr. Yanishi), Gifu University (Dr. Tsuchiya), Japanese Red Cross Medical Center (Dr. Ishibashi), Toyohashi Municipal Hospital (Dr. Nagasaka), Osaka Women's and Children's Hospital (Dr. Yazawa), International University of Health and Welfare Mita Hospital (Dr. Tonsho), Osaka General Medical Center (Dr. Tsutahara), Nara Medical University Hospital (Dr. Yoneda), Ehime University (Dr. Miyauchi), Mito Medical Center (Dr. Yuzawa), Saku Central Hospital (Dr. Murakami), Dokkyo Medical University Saitama Medical Center (Dr. Tokumoto), Jichi Medical University Hospital (Dr. Iwami), Hokkaido University (Dr. Hotta), Kagoshima University (Dr. Yamada), Tokyo Women's Medical University (Dr. Hattori), Tokyo Metropolitan Children's Medical Center (Dr. Sato), Itabashi Chuo Medical Center (Dr. Koyama), Fujita Health University (Dr. Itoh), Yamanashi University (Dr. Kamiyama), Shonan Kamakura General Hospital (Dr. Igarashi), Fukui University (Dr. Fukiage), Keio University (Dr. Morita), Japanese Red Cross Fukuoka Hospital (Dr. Motoyama), University of Tsukuba Hospital (Dr. Takahashi), Japanese Red Cross Wakayama Medical Center (Dr. Itoh), Yonago Medical Center (Dr. Sugitani), Japanese Red Cross Nagoya Daini Hospital (Dr. Watarai), Nagasaki Medical Center (Dr. Onita), Chiba University (Dr. Maruyama), Miyazaki Prefectural Miyazaki Hospital (Dr. Terasaka), Tokai University (Dr. Nakamura), Sapporo City General Hospital (Dr. Sasaki), Saitama University (Dr. Okada), Toranomon Hospital (Dr. Ishii), Kyushu University (Dr. Okabe), International University of Health and Welfare Atami Hospital (Dr. Tojinbara), Wakayama Medical University Hospital (Dr. Yoshikawa), Shizuoka General Hospital (Dr. Shiraishi), Yokohama City University Medical Center (Dr. Teranishi), Hospital of Hyogo College of Medicine (Dr. Yamada), Mie University (Dr. Nishikawa), Toda Chuo General Hospital (Dr. Shimizu), Tokushima University (Dr. Yamaguchi), Kobe University (Dr. Ishimura), Hyogo Prefectural Nishinomiya Hospital (Dr. Kishikawa), Osaka University (Dr. Imamura), Saitama Medical Center (Dr. Maki), Kameda Medical Center (Dr. Ochi), Shinshu University (Dr. Minagawa), St. Luke's International Hospital (Dr. Nagahama), Hamamatsu University (Dr. Isobe), Hiroshima Prefectural Hospital (Dr. Ishimoto), National Center for Child Health and Development (Dr. Sato), Ohkubo Hospital (Dr. Shirakawa), Hiroshima University (Dr. Ide), Japan Community Health Care Organization Osaka Hospital (Dr. Fujimoto), Kyoto University (Dr. Kobayashi), Osaka City University Hospital (Dr. Uchida), Kushiro City General Hospital (Dr. Morita), St. Marianna University (Dr. Marui), Toho University Omori Medical Center (Dr. Shinoda), Aichi Medical University (Dr. Kobayashi), Hirosaki University (Dr. Hatakeyama), Oyokyo Kidney Research Institute Hirosaki Hospital (Dr. Hatakeyama), St. Mary Hospital (Dr. Taniguchi), Tokyo Women's Medical University Yachiyo Medical Center (Dr. Inui), Kanazawa University (Dr. Kadono), Nagasaki University (Dr. Mochizuki), Kitasato University (Dr. Yoshida), Ryukyu University (Dr. Kimura), Saiseikai Yahata General Hospital (Dr. Yasunaga), Oita University (Dr. Ando), Yamagata University (Dr. Nishida), Fukuoka University (Dr. Nakamura), Iwate Prefectural Isawa Hospital (Dr. Yoneda), Tohoku University (Dr. Tokodai), Fukushima Medical University (Dr. Hata), Sapporo Medical University (Dr. Tanaka), and the Japanese Red Cross Kumamoto Hospital (Dr. Yamanaga).
Kuramitsu K, Yamanaga S, Osawa R, et al. Impact of COVID‐19 on the living donor liver and kidney transplantation programs in Japan in 2020. Transpl Infect Dis. 2022;24:e13845. 10.1111/tid.13845
REFERENCES
- 1. Morens DM, Daszak P, Taubenberger JK. Escaping Pandora's box—another novel coronavirus. N Engl J Med. 2020;382(14):1293‐1295. 10.1056/NEJMp2002106. [DOI] [PubMed] [Google Scholar]
- 2. Morens DM, Fauci AS. Emer ging pandemic diseases: how we got to COVID‐19. Cell. 2020;182(5):1077‐1092. 10.1016/j.cell.2020.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Caillard S, Chavarot N, Francois H, et al. COVID‐19 infection more severe in kidney transplant recipients? Am J Transplant. 2021;21(3):1295‐1303. 10.1111/ajt.16424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ahn C, Amer H, Anglicheau D, et al. Global transplantation COVID report March 2020. Transplantation. 2020;104(10):1974‐1983. 10.1097/TP.0000000000003258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Kumar D, Manuel O, Natori Y, et al. COVID‐19: a global transplant perspective on successfully navigating a pandemic. Am J Transplant. 2020;20(7):1773‐1779. 10.1111/ajt.15876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Strauss AT, Boyarsky BJ, Garonzik‐Wang JM, et al. Liver transplantation in the United States during the COVID‐19 pandemic: national and center‐level responses. Am J Transplant. 2021;21(5):1838‐1847. 10.1111/ajt.16373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Azzi Y, Bartash R, Scalea J, Loarte‐Campos P, Akalin E. COVID‐19 and solid organ transplantation: a review article. Transplantation. 2021;105(1):37‐55. 10.1097/TP.0000000000003523. [DOI] [PubMed] [Google Scholar]
- 8. Raja MA, Mendoza MA, Villavicencio A, et al. COVID‐19 in solid organ transplant recipients: a systematic review and meta‐analysis of current literature. Transplant Rev (Orlando). 2021;35(1):100588. 10.1016/j.trre.2020.100588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cravedi P, Mothi SS, Azzi Y, et al. COVID‐19 and kidney transplantation: results from the TANGO International Transplant Consortium. Am J Transplant. 2020;20(11):3140‐3148. 10.1111/ajt.16185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Sánchez‐Álvarez JE, Pérez Fontán M, Jiménez Martín C, et al. SARS‐CoV‐2 infection in patients on renal replacement therapy. Report of the COVID‐19 Registry of the Spanish Society of Nephrology (SEN). Nefrologia. 2020;40(3):272‐278. 10.1016/j.nefro.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Caillard S, Anglicheau D, Matignon M, et al. An initial report from the French SOT COVID Registry suggests high mortality due to COVID‐19 in recipients of kidney transplants. Kidney Int. 2020;98(6):1549‐1558. 10.1016/j.kint.2020.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Webb GJ, Marjot T, Cook JA, et al. Outcomes following SARS‐CoV‐2 infection in liver transplant recipients: an international registry study. Lancet Gastroenterol Hepatol. 2020;5(11):1008‐1016. 10.1016/S2468-1253(20)30271-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Colmenero J, Rodríguez‐Perálvarez M, Salcedo M, et al. Epidemiological pattern, incidence, and outcomes of COVID‐19 in liver transplant patients. J Hepatol. 2021;74(1):148‐155. 10.1016/j.jhep.2020.07.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Molnar MZ, Bhalla A, Azhar A, et al. Outcomes of critically ill solid organ transplant patients with COVID‐19 in the United States. Am J Transplant. 2020;20(11):3061‐3071. 10.1111/ajt.16280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Chaudhry ZS, Williams JD, Vahia A, et al. Clinical characteristics and outcomes of COVID‐19 in solid organ transplant recipients: a cohort study. Am J Transplant. 2020;20(11):3051‐3060. 10.1111/ajt.16188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Mendoza MA, Raja M, Villavicencio A, Anjan S, Natori Y. Is t he outcome of SARS‐CoV‐2 infection in solid organ transplant recipients really similar to that of the general population? Am J Transplant. 2021;21(4):1670‐1671. 10.1111/ajt.16370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. De Meester J, De Bacquer D, Naesens M, et al. Incidence, characteristics, and outcome of COVID‐19 in adults on kidney replacement therapy: a regionwide registry study. J Am Soc Nephrol. 2021;32(2):385‐396. 10.1681/ASN.2020060875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Massie AB, Boyarsky BJ, Werbel WA, et al. Identifying scenarios of benefit or harm from kidney transplantation during the COVID‐19 pandemic: a stochastic simulation and machine learning study. Am J Transplant. 2020;20(11):2997‐3007. 10.1111/ajt.16117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Thaunat O, Legeai C, Anglicheau D, et al. IMPact of the COVID‐19 epidemic on the moRTAlity of kidney transplant recipients and candidates in a French Nationwide registry sTudy (IMPORTANT). Kidney Int. 2020;98(6):1568‐1577. 10.1016/j.kint.2020.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Arevalo‐Rodriguez I, Buitrago‐Garcia D, Simancas‐Racines D, et al. False‐negative results of initial RT‐PCR assays for COVID‐19: a systematic review. PLoS One. 2020;15(12):e0242958. 10.1371/journal.pone.0242958. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental TABLE S1. Questionnaires sent to transplant programs
Supplemental TABLE S2. Guidelines for transplant medicine from the Japan Society for Transplantation


