Abstract
In insulin-dependent diabetes, the islet β cells do not produce enough insulin and the patients must receive exogenous insulin to control blood sugar. However, there are still many deficiencies in exogenous insulin supplementation. Therefore, the replacement of destroyed functional β cells with insulin-secreting cells derived from functional stem cells is a good idea as a new therapeutic idea. This review introduces the development schedule of mouse and human embryonic islets. The differences between mouse and human pancreas embryo development were also listed. Accordingly to the different sources of stem cells, the important research achievements on the differentiation of insulin-secreting β cells of stem cells and the current research status of stem cell therapy for diabetes were reviewed. Stem cell replacement therapy is a promising treatment for diabetes, caused by defective insulin secretion, but there are still many problems to be solved, such as the biosafety and reliability of treatment, the emergence of tumors during treatment, untargeted differentiation and autoimmunity, etc. Therefore, further understanding of stem cell therapy for insulin is needed.
Keywords: Diabetes mellitus, Stem cell therapy, Transplantation, β cell, Differentiation
Core Tip: Diabetes mellitus is one of the major health problems. Although traditional treatments such as exogenous insulin injection can relieve diabetes to a certain extent, they have failed to achieve a radical cure. Stem cell replacement therapy is a promising treatment for diabetes. So in this review, we introduce the development schedule of mouse and human embryonic islets and summarize the important research progressions in stem cell therapy for diabetes.
INTRODUCTION
Diabetes mellitus is one of the major health problems now. This disease currently affects more than 425 million individuals and the prevalence of it is a year by year (http://www.idf.org/diabetesatlas). About 537 million adults (20-79 years) worldwide are living with diabetes. This number is predicted to rise to 643 million by 2030 and 783 million by 2045. Among all patients with diabetes, about 10% individuals suffer from type 1 diabetes mellitus (T1DM), a kind of diabetes that is caused by the autoimmune destruction of insulin-secreting β cells in the pancreas, and about 90% of those are affected by type 2 diabetes mellitus (T2DM) due to the insulin resistance in key metabolic tissues and the dysfunction of pancreatic insulin-secreting β cells[1,2]. Because both types of patients require exogenous insulin supplementation to regulate blood sugar, they are collectively call insulin-dependent diabetes[3].
Although treatment can be effective, it often induces hypoglycemia and complex complications[4,5]. Therefore, β cells replacement therapy had transplant in 1966[6] and the development of schemes for islet isolation[7], and the first clinical islet transplantation was carried out in 1977[8]. However, both pancreas and islet transplantation are severely limited affected by donor tissue sources and immun osuppressive demand[9,10].
Compared with the limitations of human donor-derived β cells, the differentiation of insulin β cells from pluripotent stem cells (PSCs) may be a more suitable method. Embryonic stem cells were first isolated by Evans and Kaufman in 1981[11], and human embryonic stem cells were first cultured in 1998 by Thomson et al[12]. In 2007, Voltarelli et al[13] conducted a phase I/II trial of T1DM patients with hematopoietic stem cells (one kind of adult stem cells) transplantation, which showed that autologous stem cells transplantation could produce insulin. In this review, we will summarize the specific process of differentiation of definitive endoderm (DE) into insulin-secreting β cells in the existing literature, and discuss the advances of promising stem cell therapy for insulin-dependent diabetes in recent years, and finally, explore its future development direction.
PANCREAS DEVELOPMENT: FROM MULTIPOTENT PANCREAS PROGENITORS TO ENDOCRINE CELLS
The pancreas is an important organ with both endocrine and digestive functions. Because human materials are difficult to obtain, most researchers currently use mouse models to explore pancreatic embryonic development. In this review, mouse embryonic age is converted into human embryonic age, and the possible process of human pancreatic embryonic development is described from front to back on the basis of mouse model according to the timeline.
The timeline of human embryonic development is based on age estimates until 60 d post conception when identifiable human characteristics become apparent and then the name changes from embryo to fetus[14].
Under the activation of epidermal growth factor, fibroblast growth factor 1, 7 and 10 (FGF1, FGF7and FGF10), multipotent pancreas progenitors (MPPs) proliferate in a large amount[15]. The levels of FGF7 and FGF10 parallel MPPs expansion[16], and they activate the proliferation of human pancreatic epithelial cells in vitro[17]. During 6-7 wpc, pancreatic trunk and tip domains are formed from the foregut[18], and the former will continue to differentiate into ductal cells and endocrine cells, while the latter will differentiate into acinar cells. Progenitors in the tip domain have the ability to develop into all pancreatic epithelial cells initially, including acinar, ductal, and endocrine cells. The cells remaining at the distal end of the epithelial structure then undergo acinar differentiation, while those near the apical end become bipotent progenitors (endocrine/duct), also known as sex determining region Y-box 9 (SOX9+) bipotent progenitors. Acinar apical cells are isolated from the trunk by the antagonistic relationship between Nirenberg and Kim homeobox factor 6.1 (NKX6.1) and pancreas transcription factor 1A (PTF1A). PTF1A is gradually localized only in tip cells, while NKX6.1, SOX9 and hepatocyte nuclear factor 1 homeobox B (HNF1B) are located in the trunk domain, whilst SOX9, NKX6.1 are limited to ductal lineages ultimately. Under the mediation of FGF7, FGF 10, lamimin-1, and WNT-activating ligands, the ducts and acini form the exocrine part of the pancreas[19]. Acini secrete digestive enzymes such as trypsin, chymotrypsin, lipase and amylase, and their differentiation is regulated by a series of transcription factors, including PTF1A and MIST1[20], While the molecular mechanisms by which ducts are regulated is not completely clear, it is thought that HNF1B and HNF6 are active in ductal cells[19]. Ductal cells are polar and ciliated, forming tubular networks and secreting bicarbonates and mucins[21].
During 7-8 wpc, insulin+ cells are first detected in human pancreas[18]. At around 8 wpc, the left ventral bud degenerate and the right ventral bud fuse with the dorsal bud due to the gut rotation movement[22]. The expression of transcription factor neurogenin 3 (NGN3) increases with the appearance of human fetal β cells around 8 wpc, but the expression of NGN3 in human fetus is transient and reaches its peak around 10-14 wpc, and cannot be detected after 35 wpc[23]. Around e9.5, a small group of cells in the thickened DE epithelium begin to express the basic helix-loop-helix transcription factor NGN3[24]. Studies have shown that these NGN3+ cells are islet progenitors because SOX9+ bipotent progenitors depend on the regulation of NGN3 to differentiate into endocrine or ductal cells, and cells expressing NGN3 produce all islet lineage cells[18]. The evidence to this effect are as follows: islet cells are not observed in NGN3 knockout mice[24]. Mice with NGN3 gene defects failed to develop all endocrine cells and died of diabetes 1-3 d after birth[24]. Genealogical tracing experiments showed that NGN3+ cells could produce all pancreatic endocrine cells[25].
In adult pancreas, purified NGN3+ cells activated by partial ductal ligation could differentiate into all islet cell types after being injected into embryonic pancreas in vitro[26]. In human, biallelic mutations of NGN3 caused permanent neonatal diabetes mellitus[27]. The efficiency of endocrine cells induced by NGN3 was low. Only 3%-4.5% of SOX9+ progenitors express NGN3 at the peak, which means that in order to produce a sufficient number of islet cells in the human body, it takes a long time for endocrine cells to be induced, and it is still unknown why only some SOX9+ cells activate the expression of NGN3[23].
After the expression of NGN3, pro-endocrine cells trigger the expression of downstream endocrine transcription factor genes. These include pancreatic and duodenal homeobox 1 (PDX1), NKX6.1, paired box protein 4 and 6 (PAX4 and PAX6), neurogenic differentiation 1, aristaless related homeobox, regulatory factor X6, NKX2.2 etc., expressed around 8 wpc[28]. These progenitors migrate into mesenchyme and form islets composed of α, β, δ, pancreatic polypeptide (PP) and ε cells. Glucagon, insulin, somatostatin (SST), PPY and ghrelin are produced respectively. Insulin acts on peripheral tissues such as liver, muscle, and adipose tissue to increase glucose utilization and lower blood glucose, while glucagon increases blood glucose concentration by acting on liver, brain, adipose tissue, and heart[29]. These two hormones are the key to maintaining blood glucose homeostasis. There is a close paracrine regulatory loop between α and β cells. For example, β cells secrete urocortin 3 to stimulate the release of SST, and SST inhibits the secretion of glucagon from α cells[30]; α cells also produce ghrelin to inhibit insulin secretion and stimulate their own glucagon secretion[31]. Around 8.5 wpc, the expression of glucagon or SST can be probed; at 9 wpc, cells with polypeptide+ or ghrelin+ appear[18,32]. Not all cells with the positive expression of NGN3 are identical in their developmental potential, and we do not fully understand how NGN3+ cells decide to differentiate into specific endocrine subtypes yet.
Although most researchers currently use mouse models to explore pancreatic embryonic development due to the lack of human materials, we cannot ignore the differences between mouse and human pancreatic embryogenesis. The following outlines the differences between human and mouse pancreatic embryogenesis that are currently known (Table 1).
Table 1.
The differences between human and mouse pancreatic embryogenesis
|
Differences
|
Mice
|
Human
|
Ref.
|
| Morphological change | Early separation of foregut from notochord: e8.75 | Delayed separation of foregut from notochord: 4-5 wpc | [58,59] |
| Early formation of tip and trunk of pancreass: e14-e15 | The tip and trunk pancreas form late: 6-8 wpc | [18,60,61] | |
| Late islet formation: Endocrine cells do not aggregate until birth to form islets | Islet formation is early: formation begins at 12 wpc | [62,63] | |
| Expression of transcription factors | PDX1: Early expression, the current intestinal and notochord is still in contact with the expression | PDX1: Late expression, delayed until the foregut and notochord separated from each other | [58,59] |
| NKX2.2: When it was confined to NGN3+ progenitor cells, it was widely expressed in mouse MPPs up to e13 | NKX2.2: This expression does not appear until the cells have differentiated into endocrine lineage in human | [18,64] | |
| SOX17: It was not present in mouse pancreatic epithelial cells | SOX17: Markers specific to the endoderm of the human islet | [65] | |
| Endocrine cell formation | α cells: e8.5 | α cells: 8-9 wpc | [25,62,66] |
| β cells: e10.5-e.12.5 | β cells: 6 wpc | ||
| δ cells: e14.5 | δ cells: 10 wpc | ||
| PP cells: e10.5-e.12.5 | PP cells: 17 wpc |
IMPORTANT ACHIEVEMENTS IN DIFFERENTIATION OF PLURIPOTENT STEM CELLS INTO INSULIN-SECRETING Β CELLS
After fertilization, mammalian embryos undergo a series of cellular divisions to form morulae and are transformed into blastocyst by further cell division. The cells at the outer layer of the blastocyst differentiate into the trophoblast, forming the majority of the placenta that sustains nutrient supply to the embryo, and the inner cell mass (ICM) cells located inside the blastocyst maintain pluripotency and produce all cell types of the extraembryonic tissues and the embryos. In 1981, Evans and Kaufman[11] demonstrated that mouse ICM cells could be isolated and cultured in vitro without losing their pluripotency. Because they could mimic the differentiation ability of ICM cells, the cultured cells were called embryonic stem cells. Embryonic stem cells are a class of cells isolated from early embryos that could proliferate and self-renew indefinitely. Whether in vivo or in vitro, embryonic stem cells can be induced to differentiate into almost all types of cells in the inner, middle and outer germ layers. If embryonic stem cells are transplanted into patients, it is possible to replace a variety of damaged cells, and restore the corresponding function.
Embryonic stem cells were the first type of stem cell used to induce β cell in vitro. In 2001, Lumelsky et al[33] induced mouse embryonic stem cells to differentiate into insulin+ cells by “five-stage differentiation protocol” for the first time. In 2005, D'Amour et al[34] also designed a "five-stage differentiation protocol" to induce embryonic stem cells to differentiate into insulin producing cells (IPCs). This method mainly simulated the process of pancreatic development in vivo. In 2008, Kroon et al[35] designed a "four-stage differentiation protocol" based on the "five-stage differentiation protocol" of D'Amour’s team. The result showed that after pancreatic endoderm derived from human ESCs was transplanted into mice in vitro, the team obtained IPCs that were matured, functional and responsive to the changes of glucose level in the environment, and its function was similar to that of human mature islets. In 2014, Pagliuca et al[36] induced human ESCs to differentiate into insulin-secreting β cells for the first time.
Induced pluripotent stem cells are autologous pluripotent stem cells with multipotent differentiation potential generated by reprogramming somatic cells. In 2006, Takahashi et al[37] reprogrammed mouse somatic cells into pluripotent stem cells by adding four transcription factors (Oct3/4, Sox2, c-Myc, and Klf4) for the first time. They showed that pluripotent stem cells can be directly induced by the addition of only a few defined factors. Until now, the classical induction pattern of four transcription factors has been broken, and the number of added transcription factors has been reduced from four to two or even one[38]. Since ectopic expression of c-Myc can cause tumorigenicity of progeny, generation of pluripotent stem cells with minimal factors may reduce the risk of treatment. The study found that neural stem cells expressed endogenous levels of Sox2 and c-Myc higher than embryonic stem cells in adult mice, and that exogenous Oct4 together with either Klf4 or c-Myc is sufficient to induce plu ripotent stem cells. In 2016, Zhu et al[39] reprogrammed skin cells to transform into endodermal progenitors using drugs and genetic molecules for the first time. After adding four other molecules, the endodermal progenitors were transformed into pancreatic precursors and they developed into fully functional pancreatic β cells. These cells protect mice from diabetes and are capable of producing different doses of insulin in response to the changes in blood glucose levels.
Adult stem cells are a kind of undifferentiated cells that exist in differentiated tissues. They have the characteristics of self-renewal and multi-differentiation potential, and they exist in a variety of tissues and organs. Bone marrow mesenchymal stem cells[40], adipose mesenchymal stem cells[41,42] and pancreatic mesenchymal stem cells[43,44] are widely studied. In 2013, Lima et al[44] induced pancreatic mesenchymal stem cells to differentiate into induced pluripotent stem cells (iPSCs) by adenovirus transfection of PDX1, NGN3, macrophage-activating factor A and PAX4.
THE PROGRESSIONS IN STEM CELL THERAPY FOR INSULIN-INDEPENDENT DIABETES
At present, among many methods for treating diabetes, islet replacement therapy can be regarded as an effective treatment method to relieve diabetes, especially for insulin dependent type 1 diabetes. However, there are two problems in islet donors[45,46]: (1) islet donors are in short supply, and recipients may face huge costs for surgery and postoperative follow-up treatment; and (2) the most critical is the existence of organ immune rejection. The emergence of stem cell therapy provides a new way to solve these problems.
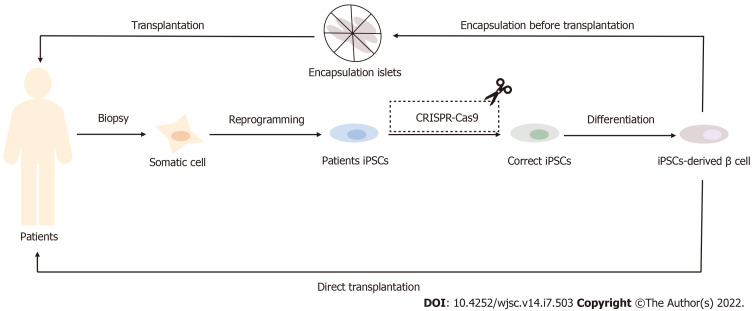
As mentioned above, stem cells are produced in various ways and from various sources. Different types of stem cells have different applications and clinical limitations (Table 2). Embryonic stem cells (ESCs, derived from the ICM of pre-implanted embryo) have limitations such as high tumorigenic risk, obvious host immune rejection and ethical controversy[36,38,39]. Therefore, the clinical application of ESCs is not clear. The biological characteristics of IPSCs (derived from embryonic gonadal ridge or postnatal testes) are highly similar to ESCs, and their biggest advantage lie in: by obtaining specific IPSCs from diabetic patients themselves, immune rejection and ethical controversies in ESCs transplantation can be effectively avoided[41]. IPSCs-derived β cells have been considered as a potential alternative source of β cells for T1DM[47] (Figure 1). However, IPSCs technology still faces the following problems[48-50]: (1) high genetic variability between individual cell lines can result in immature function of derived β cells. This mutation has been found to be repairable using genome editing tools such as CRISPR-Cas9, etc[51]. To target diabetes caused by single-gene disease in 2020 the study demonstrated that CRISPR-Cas9 correction of diabetes-induced gene variants enhances differentiation of autologous SC-cells, corrects glucose in diabetic mice, and alleviates endoplasmic reticulum and mitochondrial stress in β cells, thereby protecting pancreatic β cells[52]; (2) IPSCs have very low differentiation efficiency in vitro and may become cancerous. In 2022, Chinese scholar Deng Hongkui's research group[53] adjusted the in vitro differentiation scheme and realized efficient induction of differentiation, thus making it possible to prepare functional mature islet cells on a large scale in vitro. The research team transplanted islet cells differentiated from IPSCs into diabetic money model, which effectively reversed diabetes in mice, and no tumor-causing phenomenon was observed in all tran splanted mice during the observation period of up to 48 wk. And this year, Chinese researchers have found[54] that the bromine-containing domain and the additional terminal domain family protein inhibitor I-BET151 can effectively promote the amplification of PPs. These expandable islet progenitors (ePPs) maintain the islet progenitor status for a long time and have the ability to efficiently differentiate into functional mature islet β cells (ePP-β). In particular, ePP-β cells can be transplanted in vivo to rapidly improve diabetes in mice, and thus have great potential in cell transplantation therapy; and (3) There is transplantation immune rejection. Studies have shown that using CRISPR gene editing to knock out the β2-microglobulin gene could eliminate all HLA class I molecules, or deleting double alleles of HLA-A and HLAB, leaving only one allele of HLA-C. This allows the IPSCs-induced β cells to avoid T and NK cell attack after transplantation[55]. Mesenchymal stem cells (MSCs) are adult stem cells (derived from postnatal tissues) with self-replicating ability and multidirectional differentiation potential. Among them, human bone marrow MSCs, umbilical cord blood mesenchymal stem cells and adipose tissue MSCs are widely used. In recent years, MSC based clinical trials in patients with T1DM and T2DM diabetes were conducted, and in 2014, the first islet organ transplantation from MSC was performed in T1D patients to evaluate the efficacy and safety of stem cells in the treatment of type 1 diabetes[47]. In 2018, it was found that MSCS (ASC) in adipose tissue can effectively reduce blood glucose, improve insulin sensitivity, improve islet β cell function and reduce fat deposition in liver of type 2 diabetic mice[56].
Table 2.
Advantages and disadvantages of different types of stem cell therapy for diabetes
|
Cell types
|
Advantages
|
Disadvantages
|
Ref.
|
| Embryonic stem cell | High degree of differentiation | ESCs is weak in directional differentiation and difficult to induce | [35,36] |
| There are ethical issues: ESCs are usually allogeneic | |||
| Teratoma, immune rejection and gene mutation may occur after transplantation | |||
| Induced pluripotent stem cell | IPSC technology does not use embryonic or egg cells, so ethical problems are less likely | At present, the differentiation scheme of induced pluripotent stem cells is not mature, and the induction efficiency is low, the stability is poor, and the cost is high | [39] |
| Proprietary stem cells can be made from a patient's own cells, so there is less immune rejection | The use of virus vectors poses security problems | ||
| Adult stem cell | It is easy to achieve targeted differentiation, and some studies have shown that adult stem cells can be used to treat diabetes | The direction of differentiation is limited, not omnipotent | [43,44] |
| After transplantation, the ability of induced differentiated cells to secrete insulin was usually lower than that of normal islet β cells, and the cell survival rate was also lower | |||
| The efficiency of inducing differentiation at different stages is still low based on reprogramming and small molecule screening |
IPSC: Induced pluripotent stem cell.
Figure 1.
Application of induced pluripotent stem cells in the treatment of diabetes mellitus. The In insulin-dependent diabetes patients, induced pluripotent stem cells (IPSCs)-derived β cells can be induced by autologous IPSCs and then directly or indirectly transplanted back into the body after encapsulation to achieve the effect of diabetes treatment. For patients with monogenic diabetes, such as Wolfram syndrome patients, IPSCs-derived β cells with correct coding can also be obtained after CRISPR-Cas9 gene modification technology and then transplanted. IPSCs: Induced pluripotent stem cells.
ViaCyte's research and development focuses on the targeted differentiation of pluripotent stem cells into PPs, which are encapsulated and trialed to treat type 1 diabetes. These pancreatic progenitor cells encased in biomaterial "envelopes" (cysts) require further differentiation in vivo to develop into islet cells for optimal implant size for therapeutic effect and long-term viability of transplanted cells[54]. In July 2021, the company released the latest clinical data of its VC-02 islet cell replacement therapy for type 1 diabetes: implanted islet progenitor cells produced endogenous insulin in patients with clinical manifestation of increased glucose reactive C-peptide levels, with time delay, and decreased HbA1C. This led to the further development of stem cell therapy for diabetes. ViaCyte, in partnership with CRISPR Therapeutics, is currently developing immune avoidant stem cell lines that combine the two strategies. Methods designed to induce immune protection may produce cells that cannot be recognized and thus cleared by the immune system[57], which will further improve the safety of stem cell-derived islets transplanted into diabetic patients and effectively improve their postoperative quality of life. Unlike ViaCyte, Vertex is designed to grow fully differentiated islet cells in the laboratory and transplant them directly into patients without encapsulation. In February 2021, the FDA approved the application for clinical trial of a new drug (VX-880) for the treatment of type 1 diabetes with stem cells. In October, Vertex's clinical trial showed that the first patient treated with its stem-derived islet cells not only resumed insulin production 90 d after treatment, but also reduced daily insulin use by 91 percent.
CONCLUSION
In the past 20 years, researchers have made great progresses on how to induce stem cells to differentiate into pancreatic cells, and then insulin-secreting β cells. We also have a further understanding of the treatment of diabetes by islet transplantation. However, although some studies have been able to induce stem cells to differentiate into insulin-secreting β cells, these cells are cells with immature phenotypes, which are different from normal human β cells. Moreover, these studies have low differentiation efficiencies, thus stem cells cannot fully develop into insulin-secreting β cells.
Therefore, the selection of stem cells, the identification of maturation inducing factors in vivo, and the improvement methods of culture efficiency are all problems that need to be solved. In addition, there are other difficulties to overcome as follows: (1) the evaluation method of stem cells derived endocrine cells function; (2) the selection of surface antigens of progenitors in specific differentiation stage and the formulation of cell purification methods; and (3) the appearances of tumor, untargeted differentiation and autoimmunity during the treatment with stem cells, as well as the biosafety and reliability of the treatment.
On the whole, the application of genomics, epigenomics, proteomics and other methods to characterize the differentiation products and the cooperation with biotechnology and pharmaceutical departments are conducive to promoting the development of progenitors in specific stages to mature insulin-secreting β cells. In the near future, stem cell replacement therapy may be clinically applicable to diabetes.
Footnotes
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: March 17, 2022
First decision: April 19, 2022
Article in press: June 26, 2022
Specialty type: Endocrinology and metabolism
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): A, A
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Aydin S, Turkey; Mrozikiewicz-Rakowska B, Poland; Salvadori M, Italy; Song BW, South Korea S-Editor: Gong ZM L-Editor: A P-Editor: Gong ZM
Contributor Information
Lu Yang, Department of Endocrinology, Xiang’an Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen 361100, Fujian Province, China.
Zhu-Meng Hu, Department of Endocrinology, Xiang’an Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen 361100, Fujian Province, China.
Fang-Xu Jiang, Department of Endocrinology, Xiang’an Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen 361100, Fujian Province, China; School of Biomedical Science, University of Western Australia, Nedlands 6009, Australia; School of Health and Medical Sciences, Edith Cowan University, Perth 6000, Australia.
Wei Wang, Department of Endocrinology, Xiang’an Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen 361100, Fujian Province, China. wwei19742007@hotmail.com.
References
- 1.Halban PA, Polonsky KS, Bowden DW, Hawkins MA, Ling C, Mather KJ, Powers AC, Rhodes CJ, Sussel L, Weir GC. β-cell failure in type 2 diabetes: postulated mechanisms and prospects for prevention and treatment. Diabetes Care. 2014;37:1751–1758. doi: 10.2337/dc14-0396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jiang FX, Morahan G. Multipotent pancreas progenitors: Inconclusive but pivotal topic. World J Stem Cells. 2015;7:1251–1261. doi: 10.4252/wjsc.v7.i11.1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yalow RS, Berson SA. Immunoassay of endogenous plasma insulin in man. J Clin Invest. 1960;39:1157–1175. doi: 10.1172/JCI104130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Magliano DJ, Harding JL, Shaw JE. Glycemic control and excess mortality in type 1 diabetes. N Engl J Med. 2015;372:880. doi: 10.1056/NEJMc1415677. [DOI] [PubMed] [Google Scholar]
- 5.Khunti K, Davies M, Majeed A, Thorsted BL, Wolden ML, Paul SK. Hypoglycemia and risk of cardiovascular disease and all-cause mortality in insulin-treated people with type 1 and type 2 diabetes: a cohort study. Diabetes Care. 2015;38:316–322. doi: 10.2337/dc14-0920. [DOI] [PubMed] [Google Scholar]
- 6.Kelly WD, Lillehei RC, Merkel FK, Idezuki Y, Goetz FC. Allotransplantation of the pancreas and duodenum along with the kidney in diabetic nephropathy. Surgery. 1967;61:827–837. [PubMed] [Google Scholar]
- 7.Ricordi C, Lacy PE, Scharp DW. Automated islet isolation from human pancreas. Diabetes. 1989;38 Suppl 1:140–142. doi: 10.2337/diab.38.1.s140. [DOI] [PubMed] [Google Scholar]
- 8.Najarian JS, Sutherland DE, Matas AJ, Steffes MW, Simmons RL, Goetz FC. Human islet transplantation: a preliminary report. Transplant Proc. 1977;9:233–236. [PubMed] [Google Scholar]
- 9.Kandaswamy R, Stock PG, Gustafson SK, Skeans MA, Urban R, Fox A, Israni AK, Snyder JJ, Kasiske BL. OPTN/SRTR 2018 Annual Data Report: Pancreas. Am J Transplant. 2020;20 Suppl s1:131–192. doi: 10.1111/ajt.15673. [DOI] [PubMed] [Google Scholar]
- 10.Balamurugan AN, Naziruddin B, Lockridge A, Tiwari M, Loganathan G, Takita M, Matsumoto S, Papas K, Trieger M, Rainis H, Kin T, Kay TW, Wease S, Messinger S, Ricordi C, Alejandro R, Markmann J, Kerr-Conti J, Rickels MR, Liu C, Zhang X, Witkowski P, Posselt A, Maffi P, Secchi A, Berney T, O'Connell PJ, Hering BJ, Barton FB. Islet product characteristics and factors related to successful human islet transplantation from the Collaborative Islet Transplant Registry (CITR) 1999-2010. Am J Transplant. 2014;14:2595–2606. doi: 10.1111/ajt.12872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- 12.Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 13.Voltarelli JC, Couri CE, Stracieri AB, Oliveira MC, Moraes DA, Pieroni F, Coutinho M, Malmegrim KC, Foss-Freitas MC, Simões BP, Foss MC, Squiers E, Burt RK. Autologous nonmyeloablative hematopoietic stem cell transplantation in newly diagnosed type 1 diabetes mellitus. JAMA . 2007;297:1568–1576. doi: 10.1001/jama.297.14.1568. [DOI] [PubMed] [Google Scholar]
- 14.O'Rahilly R, Müller F. Developmental stages in human embryos: revised and new measurements. Cells Tissues Organs. 2010;192:73–84. doi: 10.1159/000289817. [DOI] [PubMed] [Google Scholar]
- 15.Bonfanti P, Nobecourt E, Oshima M, Albagli-Curiel O, Laurysens V, Stangé G, Sojoodi M, Heremans Y, Heimberg H, Scharfmann R. Ex Vivo Expansion and Differentiation of Human and Mouse Fetal Pancreatic Progenitors Are Modulated by Epidermal Growth Factor. Stem Cells Dev. 2015;24:1766–1778. doi: 10.1089/scd.2014.0550. [DOI] [PubMed] [Google Scholar]
- 16.Benazra M, Lecomte MJ, Colace C, Müller A, Machado C, Pechberty S, Bricout-Neveu E, Grenier-Godard M, Solimena M, Scharfmann R, Czernichow P, Ravassard P. A human beta cell line with drug inducible excision of immortalizing transgenes. Mol Metab. 2015;4:916–925. doi: 10.1016/j.molmet.2015.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ye F, Duvillié B, Scharfmann R. Fibroblast growth factors 7 and 10 are expressed in the human embryonic pancreatic mesenchyme and promote the proliferation of embryonic pancreatic epithelial cells. Diabetologia. 2005;48:277–281. doi: 10.1007/s00125-004-1638-6. [DOI] [PubMed] [Google Scholar]
- 18.Jennings RE, Berry AA, Kirkwood-Wilson R, Roberts NA, Hearn T, Salisbury RJ, Blaylock J, Piper Hanley K, Hanley NA. Development of the human pancreas from foregut to endocrine commitment. Diabetes. 2013;62:3514–3522. doi: 10.2337/db12-1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jacobson EF, Tzanakakis ES. Human pluripotent stem cell differentiation to functional pancreatic cells for diabetes therapies: Innovations, challenges and future directions. J Biol Eng. 2017;11:21. doi: 10.1186/s13036-017-0066-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Reichert M, Rustgi AK. Pancreatic ductal cells in development, regeneration, and neoplasia. J Clin Invest. 2011;121:4572–4578. doi: 10.1172/JCI57131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Benitez CM, Goodyer WR, Kim SK. Deconstructing pancreas developmental biology. Cold Spring Harb Perspect Biol. 2012;4 doi: 10.1101/cshperspect.a012401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sarkar A, Hochedlinger K. The sox family of transcription factors: versatile regulators of stem and progenitor cell fate. Cell Stem Cell. 2013;12:15–30. doi: 10.1016/j.stem.2012.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Salisbury RJ, Blaylock J, Berry AA, Jennings RE, De Krijger R, Piper Hanley K, Hanley NA. The window period of NEUROGENIN3 during human gestation. Islets. 2014;6:e954436. doi: 10.4161/19382014.2014.954436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gradwohl G, Dierich A, LeMeur M, Guillemot F. neurogenin3 is required for the development of the four endocrine cell lineages of the pancreas. Proc Natl Acad Sci U S A. 2000;97:1607–1611. doi: 10.1073/pnas.97.4.1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Johansson KA, Dursun U, Jordan N, Gu G, Beermann F, Gradwohl G, Grapin-Botton A. Temporal control of neurogenin3 activity in pancreas progenitors reveals competence windows for the generation of different endocrine cell types. Dev Cell. 2007;12:457–465. doi: 10.1016/j.devcel.2007.02.010. [DOI] [PubMed] [Google Scholar]
- 26.Xu X, D'Hoker J, Stangé G, Bonné S, De Leu N, Xiao X, Van de Casteele M, Mellitzer G, Ling Z, Pipeleers D, Bouwens L, Scharfmann R, Gradwohl G, Heimberg H. Beta cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell. 2008;132:197–207. doi: 10.1016/j.cell.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 27.Rubio-Cabezas O, Jensen JN, Hodgson MI, Codner E, Ellard S, Serup P, Hattersley AT. Permanent Neonatal Diabetes and Enteric Anendocrinosis Associated With Biallelic Mutations in NEUROG3. Diabetes. 2011;60:1349–1353. doi: 10.2337/db10-1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jennings RE, Berry AA, Strutt JP, Gerrard DT, Hanley NA. Human pancreas development. Development. 2015;142:3126–3137. doi: 10.1242/dev.120063. [DOI] [PubMed] [Google Scholar]
- 29.Campbell JE, Drucker DJ. Islet α cells and glucagon--critical regulators of energy homeostasis. Nat Rev Endocrinol. 2015;11:329–338. doi: 10.1038/nrendo.2015.51. [DOI] [PubMed] [Google Scholar]
- 30.van der Meulen T, Donaldson CJ, Cáceres E, Hunter AE, Cowing-Zitron C, Pound LD, Adams MW, Zembrzycki A, Grove KL, Huising MO. Urocortin3 mediates somatostatin-dependent negative feedback control of insulin secretion. Nat Med. 2015;21:769–776. doi: 10.1038/nm.3872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chuang JC, Sakata I, Kohno D, Perello M, Osborne-Lawrence S, Repa JJ, Zigman JM. Ghrelin directly stimulates glucagon secretion from pancreatic alpha-cells. Mol Endocrinol. 2011;25:1600–1611. doi: 10.1210/me.2011-1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Riedel MJ, Asadi A, Wang R, Ao Z, Warnock GL, Kieffer TJ. Immunohistochemical characterisation of cells co-producing insulin and glucagon in the developing human pancreas. Diabetologia. 2012;55:372–381. doi: 10.1007/s00125-011-2344-9. [DOI] [PubMed] [Google Scholar]
- 33.Lumelsky N, Blondel O, Laeng P, Velasco I, Ravin R, McKay R. Differentiation of embryonic stem cells to insulin-secreting structures similar to pancreatic islets. Science. 2001;292:1389–1394. doi: 10.1126/science.1058866. [DOI] [PubMed] [Google Scholar]
- 34.D'Amour KA, Bang AG, Eliazer S, Kelly OG, Agulnick AD, Smart NG, Moorman MA, Kroon E, Carpenter MK, Baetge EE. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat Biotechnol. 2006;24:1392–1401. doi: 10.1038/nbt1259. [DOI] [PubMed] [Google Scholar]
- 35.Kroon E, Martinson LA, Kadoya K, Bang AG, Kelly OG, Eliazer S, Young H, Richardson M, Smart NG, Cunningham J, Agulnick AD, D'Amour KA, Carpenter MK, Baetge EE. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat Biotechnol. 2008;26:443–452. doi: 10.1038/nbt1393. [DOI] [PubMed] [Google Scholar]
- 36.Pagliuca FW, Millman JR, Gürtler M, Segel M, Van Dervort A, Ryu JH, Peterson QP, Greiner D, Melton DA. Generation of functional human pancreatic β cells in vitro. Cell. 2014;159:428–439. doi: 10.1016/j.cell.2014.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 38.Kim JB, Zaehres H, Wu G, Gentile L, Ko K, Sebastiano V, Araúzo-Bravo MJ, Ruau D, Han DW, Zenke M, Schöler HR. Pluripotent stem cells induced from adult neural stem cells by reprogramming with two factors. Nature. 2008;454:646–650. doi: 10.1038/nature07061. [DOI] [PubMed] [Google Scholar]
- 39.Zhu S, Russ HA, Wang X, Zhang M, Ma T, Xu T, Tang S, Hebrok M, Ding S. Human pancreatic beta-like cells converted from fibroblasts. Nat Commun. 2016;7:10080. doi: 10.1038/ncomms10080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Karnieli O, Izhar-Prato Y, Bulvik S, Efrat S. Generation of insulin-producing cells from human bone marrow mesenchymal stem cells by genetic manipulation. Stem Cells. 2007;25:2837–2844. doi: 10.1634/stemcells.2007-0164. [DOI] [PubMed] [Google Scholar]
- 41.Chandra V, G S, Phadnis S, Nair PD, Bhonde RR. Generation of pancreatic hormone-expressing islet-like cell aggregates from murine adipose tissue-derived stem cells. Stem Cells. 2009;27:1941–1953. doi: 10.1002/stem.117. [DOI] [PubMed] [Google Scholar]
- 42.Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7:211–228. doi: 10.1089/107632701300062859. [DOI] [PubMed] [Google Scholar]
- 43.Guo XR, Wang XL, Li MC, Yuan YH, Chen Y, Zou DD, Bian LJ, Li DS. PDX-1 mRNA-induced reprogramming of mouse pancreas-derived mesenchymal stem cells into insulin-producing cells in vitro. Clin Exp Med. 2015;15:501–509. doi: 10.1007/s10238-014-0319-0. [DOI] [PubMed] [Google Scholar]
- 44.Lima MJ, Muir KR, Docherty HM, Drummond R, McGowan NW, Forbes S, Heremans Y, Houbracken I, Ross JA, Forbes SJ, Ravassard P, Heimberg H, Casey J, Docherty K. Suppression of epithelial-to-mesenchymal transitioning enhances ex vivo reprogramming of human exocrine pancreatic tissue toward functional insulin-producing β-like cells. Diabetes. 2013;62:2821–2833. doi: 10.2337/db12-1256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nakamura T, Fujikura J, Anazawa T, Ito R, Ogura M, Okajima H, Uemoto S, Inagaki N. Long-term outcome of islet transplantation on insulin-dependent diabetes mellitus: An observational cohort study. J Diabetes Investig. 2020;11:363–372. doi: 10.1111/jdi.13128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bottino R, Knoll MF, Knoll CA, Bertera S, Trucco MM. The Future of Islet Transplantation Is Now. Front Med (Lausanne) 2018;5:202. doi: 10.3389/fmed.2018.00202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen S, Du K, Zou C. Current progress in stem cell therapy for type 1 diabetes mellitus. Stem Cell Res Ther. 2020;11:275. doi: 10.1186/s13287-020-01793-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Araki R, Uda M, Hoki Y, Sunayama M, Nakamura M, Ando S, Sugiura M, Ideno H, Shimada A, Nifuji A, Abe M. Negligible immunogenicity of terminally differentiated cells derived from induced pluripotent or embryonic stem cells. Nature. 2013;494:100–104. doi: 10.1038/nature11807. [DOI] [PubMed] [Google Scholar]
- 49.Tanosaki S, Tohyama S, Fujita J, Someya S, Hishiki T, Matsuura T, Nakanishi H, Ohto-Nakanishi T, Akiyama T, Morita Y, Kishino Y, Okada M, Tani H, Soma Y, Nakajima K, Kanazawa H, Sugimoto M, Ko MSH, Suematsu M, Fukuda K. Fatty Acid Synthesis Is Indispensable for Survival of Human Pluripotent Stem Cells. iScience. 2020;23:101535. doi: 10.1016/j.isci.2020.101535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yabe SG, Fukuda S, Nishida J, Takeda F, Nashiro K, Okochi H. Induction of functional islet-like cells from human iPS cells by suspension culture. Regen Ther. 2019;10:69–76. doi: 10.1016/j.reth.2018.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kyttälä A, Moraghebi R, Valensisi C, Kettunen J, Andrus C, Pasumarthy KK, Nakanishi M, Nishimura K, Ohtaka M, Weltner J, Van Handel B, Parkkonen O, Sinisalo J, Jalanko A, Hawkins RD, Woods NB, Otonkoski T, Trokovic R. Genetic Variability Overrides the Impact of Parental Cell Type and Determines iPSC Differentiation Potential. Stem Cell Reports. 2016;6:200–212. doi: 10.1016/j.stemcr.2015.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Maxwell KG, Augsornworawat P, Velazco-Cruz L, Kim MH, Asada R, Hogrebe NJ, Morikawa S, Urano F, Millman JR. Gene-edited human stem cell-derived β cells from a patient with monogenic diabetes reverse preexisting diabetes in mice. Sci Transl Med. 2020;12 doi: 10.1126/scitranslmed.aax9106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Du Y, Liang Z, Wang S, Sun D, Wang X, Liew SY, Lu S, Wu S, Jiang Y, Wang Y, Zhang B, Yu W, Lu Z, Pu Y, Zhang Y, Long H, Xiao S, Liang R, Zhang Z, Guan J, Wang J, Ren H, Wei Y, Zhao J, Sun S, Liu T, Meng G, Wang L, Gu J, Wang T, Liu Y, Li C, Tang C, Shen Z, Peng X, Deng H. Human pluripotent stem-cell-derived islets ameliorate diabetes in non-human primates. Nat Med. 2022;28:272–282. doi: 10.1038/s41591-021-01645-7. [DOI] [PubMed] [Google Scholar]
- 54.Ma X, Lu Y, Zhou Z, Li Q, Chen X, Wang W, Jin Y, Hu Z, Chen G, Deng Q, Shang W, Wang H, Fu H, He X, Feng XH, Zhu S. Human expandable pancreatic progenitor-derived β cells ameliorate diabetes. Sci Adv. 2022;8:eabk1826. doi: 10.1126/sciadv.abk1826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Xu H, Wang B, Ono M, Kagita A, Fujii K, Sasakawa N, Ueda T, Gee P, Nishikawa M, Nomura M, Kitaoka F, Takahashi T, Okita K, Yoshida Y, Kaneko S, Hotta A. Targeted Disruption of HLA Genes via CRISPR-Cas9 Generates iPSCs with Enhanced Immune Compatibility. Cell Stem Cell. 2019;24:566–578.e7. doi: 10.1016/j.stem.2019.02.005. [DOI] [PubMed] [Google Scholar]
- 56.Wang M, Song L, Strange C, Dong X, Wang H. Therapeutic effects of adipose stem cells from diabetic mice for the treatment of type 2 diabetes. Mol Ther. 2018;26:1921–1930. doi: 10.1016/j.ymthe.2018.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.de Klerk E, Hebrok M. Stem Cell-Based Clinical Trials for Diabetes Mellitus. Front Endocrinol (Lausanne) 2021;12:631463. doi: 10.3389/fendo.2021.631463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lorberbaum DS, Docherty FM, Sussel L. Animal Models of Pancreas Development, Developmental Disorders, and Disease. Adv Exp Med Biol. 2020;1236:65–85. doi: 10.1007/978-981-15-2389-2_3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Alvarez Fallas ME, Pedraza-Arevalo S, Cujba AM, Manea T, Lambert C, Morrugares R, Sancho R. Stem/progenitor cells in normal physiology and disease of the pancreas. Mol Cell Endocrinol. 2021;538:111459. doi: 10.1016/j.mce.2021.111459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nair G, Hebrok M. Islet formation in mice and men: lessons for the generation of functional insulin-producing β-cells from human pluripotent stem cells. Curr Opin Genet Dev. 2015;32:171–180. doi: 10.1016/j.gde.2015.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Napolitano T, Avolio F, Silvano S, Forcisi S, Pfeifer A, Vieira A, Navarro-Sanz S, Friano ME, Ayachi C, Garrido-Utrilla A, Atlija J, Hadzic B, Becam J, Sousa-De-Veiga A, Plaisant MD, Balaji S, Pisani DF, Mondin M, Schmitt-Kopplin P, Amri EZ, Collombat P. Gfi1 Loss Protects against Two Models of Induced Diabetes. Cells. 2021;10 doi: 10.3390/cells10112805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Piper K, Brickwood S, Turnpenny LW, Cameron IT, Ball SG, Wilson DI, Hanley NA. Beta cell differentiation during early human pancreas development. J Endocrinol. 2004;181:11–23. doi: 10.1677/joe.0.1810011. [DOI] [PubMed] [Google Scholar]
- 63.Perez-Frances M, Abate MV, Baronnier D, Scherer PE, Fujitani Y, Thorel F, Herrera PL. Adult pancreatic islet endocrine cells emerge as fetal hormone-expressing cells. Cell Rep. 2022;38:110377. doi: 10.1016/j.celrep.2022.110377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ali EA, Sardar S, Yassin MA. Priapism in lymphoproliferative disorders: A systematic review. Hematol Oncol Stem Cell Ther. 2021 doi: 10.1016/j.hemonc.2021.05.003. [DOI] [PubMed] [Google Scholar]
- 65.Shaw-Smith C, De Franco E, Lango Allen H, Batlle M, Flanagan SE, Borowiec M, Taplin CE, van Alfen-van der Velden J, Cruz-Rojo J, Perez de Nanclares G, Miedzybrodzka Z, Deja G, Wlodarska I, Mlynarski W, Ferrer J, Hattersley AT, Ellard S. GATA4 mutations are a cause of neonatal and childhood-onset diabetes. Diabetes. 2014;63:2888–2894. doi: 10.2337/db14-0061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Panzer JK, Caicedo A. Targeting the Pancreatic α-Cell to Prevent Hypoglycemia in Type 1 Diabetes. Diabetes. 2021;70:2721–2732. doi: 10.2337/dbi20-0048. [DOI] [PMC free article] [PubMed] [Google Scholar]