Abstract
Spontaneous preterm birth (sPTB) remains a worldwide healthcare challenge. Pre-term labor (PTL) is thought to be the largest reversible cause of sPTB, but current tocolytic therapies are ineffective and associated with systemic side effects from chronic use. Therefore, identifying novel mechanisms that promote human uterine smooth muscle (hUSM) relaxation is essential to improving clinical management of PTL. Here, we aimed to determine if an extraocular opsin receptor (OPN 3,4,5) system is expressed in pregnant hUSM and to characterize how photo-mediated relaxation of pre-contracting hUSM may be facilitated by external application of light. Translational studies were performed with hUSM from healthy late gestation patients (n=8) and non-pregnant, similarly-aged patients undergoing hysterectomy (n=4). First, RT-PCR screened for mRNA coding for components of the classical extraocular light receptors (OPN 3,4,5). We found a restricted repertoire of opsin receptors (OPN3) expressed in pregnant hUSM tissue. Immunohistochemistry was performed to confirm protein expression. Pre-contracting late gestation hUSM strips were studied in functional organ bath studies to determine if photo-mediated relaxation is intensity or wavelength dependent. Functional organ bath studies revealed acute photo-mediated relaxation occurring in an intensity and wavelength-dependent manner. Finally, coimmunoprecipitation of OPN3 with Gs following light activation suggests that a component of photo-relaxation occurs via GPCR machinery. This is the first report of light-mediated relaxation of pre-contracted human myometrium. Activation of endogenous light receptors on human myometrium may become a novel, non-invasive tocolytic strategy.
Keywords: Myometrium, opsin 3 receptor, photo-relaxation
Introduction:
Given that approximately 10% of babies in the US are born before 37 weeks’ gestation, spontaneous preterm birth (sPTB) remains a public health priority in the United States [1]. While the etiologies leading to sPTB are numerous, the largest potentially reversible cause involves the development of pre-term labor (PTL) [2]. Unfortunately, long-term suppression of premature uterine contractions once they have become established remains largely ineffective [3]. While a variety of reasons have been proposed for this shortcoming, the prevailing evidence suggests the pharmacodynamic profiles of conventional tocolytics are poorly suited for laboring myometrium. Indeed, emerging studies show that the milieu of PTL may contribute to altered normal uterine smooth muscle (USM) relaxation pathways [4]. An additional concern includes the divergent physiological effects these drugs exhibit when comparing acute versus long term systemic use. In particular, while conventional tocolytics (e.g. Nifedipine, Terbutaline, etc.) may be successful at acutely suppressing uterine contractions, issues related to reductions in drug efficacy and the development of off-target side effects severely limit their chronic use [5-8]. The clinical evidence for chronic administration for PTL is marred by excessive maternal morbidity risk [9]. These collective dilemmas have appropriately led to a quest to find other novel pathways and targets more selective for uterine smooth muscle (USM) to treat PTL. Interestingly, while several candidate targets have emerged in the last 5 years, none have shown clinical utility to date. Most would agree the ideal treatment would be non-invasive, avoid systemic administration, and allow for focused treatment directly on the uterus to facilitate reversible and titrate-able relaxation. Yet, no approaches to date have sought to incorporate all of these principles.
Recent reports now describe endogenous light-receptor expression systems that exist in peripheral human tissues outside of the CNS/retina, and that they may be therapeutically targeted [10-16]. Indeed, our group and others have shown that it is possible to target human smooth muscle opsin-light receptor complexes with external light to induce potent relaxation from pre-contracted states in airway tissues [10, 17]. While it is unknown if pregnant human uterine smooth muscle (USM) functionally express these systems, the possibility of light-mediated photo-activation of these specialized receptor systems as a novel tocolytic approach may allow for a non-systemic, non-invasive, and completely revolutionary means to address PTL. Therefore, the first goal of this study sought to determine the repertoire of any peripheral opsin receptor system expressed in late gestation human uterine smooth muscle. We also questioned whether photo-mediated activation can induce human USM relaxation from a pre-contracted state in a dose and wavelength-dependent manner, as has been previously shown in other smooth muscle studies. Lastly, given that opsin receptors classically signal via GPCR machinery, we questioned if mechanistic coupling of endogenous human USM opsins to Gs, a well-established pro-relaxation signaling molecule, could be demonstrated.
Methods
Ethical Approval:
Human uterus was obtained from healthy, pregnant patients (n=8 patients) undergoing elective C-section in accordance with the institutional review board (IRB)-approved protocol (#AAAL4005). De-identified human uterine tissue from the superior margin of the uterine incision was collected from patients at 38-40 weeks of gestation. For comparative purposes, non-pregnant human uterus samples were obtained after hysterectomies of similarly aged women (n=4 patients), as is described in the IRB-approved protocol (#AAAI0337).
Reagents/Chemicals:
G-protein receptor kinase 2 inhibitor (Santa Cruz, Dallas, Texas), 9-cis retinal (Sigma Aldrich St. Louis, MO).
Human USM Specimens:
In all cases, excised tissue was immediately placed in cold sterile M199 media, and transported to the lab on ice. Tissues were then washed copiously in cold Hank's Balanced Salt Solution and processed for organ bath studies, establishing primary cell cultures of human USM cells, and/or embedded in OCT compound for histology as previously described [18-19].
Cell Culture:
Primary uterine smooth muscle cell cultures were established from fresh myometrium tissue samples (n=8 patients) utilizing a papain enzymatic dissociation in accordance with manufacturer recommendation (Worthington-Papain Kit; Lakewood, NJ, USA). As previously described, myometrial tissue was cut into small pieces using dissecting scissors and enzymatically dissociated using papain and collagenase under sterile conditions at 37°C. Samples were oxygenated intermittently by bubbling media with 95% oxygen, separated using ovamucoid/albumin (density separation), and plated onto a 75 cm2 culture flasks. Experimental protocols restricted cultured cells to less than 7 passages, and all cells were maintained in Smooth Muscle Growth Medium-2, fortified with manufacturer recommended additives (Lonza, Walkersville, Maryland).
Isolation of RNA and reverse transcription polymerase chain reaction (RT-PCR):
A PALM Microbeam IV laser capture micro-dissection (LCMD) system (Zeiss, Oberkochen, Germany) was used to isolate smooth muscle and to extract pure smooth muscle RNA from human uterine samples. First, human uterine samples were embedded in OCT compound followed by isopentane/dry ice freezing. Sections of 6 μm thickness were created from frozen OCT blocks under RNase-free conditions and placed on a single 1-mm PEN-membrane-coated slide (PALM Microlaser Technologies, Westchester, NY, USA). Cresyl Violet staining of the uterine sample (Ambion AM1935 (Thermofisher Waltham, MA, USA) was performed to improve the histologic and morphologic targeting of USM cells. Only central portions of smooth muscle were acquired, avoiding contamination from other adjacent cell types. Then, RNA was extracted using the RNAqueous-micro kit (Ambion, Austin, TX) according to manufacturer recommendation. Recovered RNA was reversed transcribed into cDNA using a commercially available kit, Superscript VILO (Invitrogen, Carlsbad, CA).
Using the Advantage 2 Polymerase Kit (Clontech, Mountain View, CA, USA), 5 μl of cDNA was amplified. Sense and antisense primers (0.4 μM), each designed to target distinct exons separated by a large intron (Table 1), were used to screen respective messages for extra-ocular opsin receptors. cDNA samples were denatured at 94°C for 10s with annealing and extension temperatures maintained at 68°C for 1 min. Each sample underwent 35 cycles of amplification in a PTC-200 Peltier thermal cycler (Bio-Rad, Hercules, CA, USA). PCR products were analyzed on a 5% nondenaturing polyacrylamide gel in tris-acetate, EDTA buffer. The gel was then stained with ethidium bromide (Molecular Probes, Eugene, OR, USA) and visualized with a gel imager (Biospectra UVP, Upland, CA, USA) and the Visionworks software (Biospectra UVP, Jena, Germany). Results were repeated with samples obtained from 3 different patients.
Table 1.
Primer sequence and predicted product size used in RT-PCR OPN survey
Immunohistochemistry for Opsin 3 in human pregnant uterus:
Samples of pregnant human myometrial smooth muscle taken from the superior margin of the uterine incision were processed for qualitative presence of OPN3. Strips of myometrial smooth muscle were grossly dissected from surrounding connective tissue. Samples were fixed in 4% paraformaldehyde (4°C overnight) and incubated in 30% sucrose for 24 hours. Samples were then frozen and sectioned into 6μm slices. After a PBS wash, samples were permeabilized with 0.1% Triton X-100 for 10 minutes, blocked with 15% goat serum and incubated overnight at 4°C in primary antibodies. The primary antibodies were Opsin 3 (1:200; Anti-OPN3, LS-C151367; LS Biosciences, Seattle, WA), and rhodamine phalloidin (1:200; Life Technologies). The secondary antibody for Opsin 3 consisted of goat anti-rabbit conjugated with AlexaFluor 488 (1:400 dilution; ThermoFisher, Waltham, MA). Mounting media contained 4′,6-diamidino-2-phenylindole (DAPI) stain for cell nuclei (#H-1500; Vector laboratories, Burlingame, California). Negative controls consisted of smaples prepared without primary antibody incubation. Experiments were repeated 3 times. Immunofluorescence images were acquired using confocal microscopy with NIS software version 4.10 (Nikon, Minato, Tokyo, Japan).
Immunocytochemistry for Opsin 3 in human pregnant uterus:
Primary cells derived from pregnant human myometrial smooth muscle were used, as described previously. Cells were washed twice with PBS prior to fixation with 4% paraformaldehyde for 10 minutes at room temperature. Cells were then washed three times with ice cold PBS. Then, cells were permeabilized with 0.1% Triton X-100 for 10 minutes at room temperature. Cells were washed three times for 5 minutes. Afterwards, cells were then blocked with 22.52 mg/mL Glycine and 10% goat serum in PBST (PBS + 0.1% Tween 20) for 1 hour. Opsin 3 (1:100; Anti-OPN3, LC-C151367; LS Biosciences, Seattle, WA) was incubated for 2 hours in a humidified chamber. Cells were washed thrice in PBS for 5 minutes each. The secondary antibody for Opsin 3 consisted of goat anti-rabbit conjugated with AlexaFluor 488 (1:2000 dilution; ThermoFisher, Waltham, MA). Mounting media contained 4′,6-diamidino-2-phenylindole (DAPI) stain for cell nuclei (#H-1500; Vector laboratories, Burlingame, California). No primary antibody control for nonspecific secondary binding was performed. Images were acquired under 20x with a confocal microscope with NIS Software version 5.02 (Ti Microscope, Minato, Tokyo, Japan). DAPI excitation at 405 and emission at 450 with a laser power of 2.7. AlexaFluor excitation at 488 and emission at 525 with a laser power of 6.8. Experiments were repeated with three primary cell lines from three different patients.
Coimmunoprecipitation and western blot:
Primary uterine smooth muscle cells derived from pregnant human uteruses were cultured on two 150mm dishes and grown to at least 80% confluence. Two dishes were used at a time. The cells were first incubated for 30 minutes in darkness (5% CO2, 37°C) followed by a 15 minute treatment with G protein receptor kinase 2 (GRK2) inhibitor (95 μM) and 9-cis-retinal (28 μM) under ambient red light (previously shown to not have a pro-relaxant effect), and finally a 2 minute treatment with 435nm blue light (CoolLED; Andover, England) [10,17]. Then, the cells were lysed with 3 mL PD buffer (40 mM Tris-HCl, 500 mM NaCl, 0.1% Nonidet P-40, 6 mM EGTA, 10 mM NaF, 1 tablet of cOmplete Mini Protease Inhibitor Cocktail [Sigma Aldrich; St. Louis, MO], 1% Phosphatase Inhibitor Cocktail 3 [Sigma Aldrich; St. Louis, MO] ). 3mL lysis buffer was transferred and scrapped between the two cell culture dishes. Resulting lysate was centrifuged (5 minutes, 300 rcf). The supernatant was pre-cleared of non-specific protein binding using protein A-based Dynabeads (1:10 Bead:Lysate volume). New beads were linked to Gs antibody (100 μL for 200 μg/mL antibody for 3mL of lysate, Santa Cruz sc-55545; 1:10 Bead:Lysate by volume) according to manufacture protocol. This bead-antibody complex was then added to the supernatant of pre-cleared protein and magnetically pulled down. Wash steps and volumes were determined according to the manufacturer. The bead-antibody-antigen complex was eluted and denatured in 70°C with Laemmli’s buffer (2% SDS, 5% 2-mercaptoethanol, 10% glycerol, 0.002% bromophenol blue, 0.0625M Tris-HCl, pH 6.8) for 10 minutes. The resulting protein isolate was then probed for OPN3 receptor via western blot. Plain beads and antibody linked beads alone were eluted as controls for nonspecific binding between the western blot primary and secondary antibodies and coimmunoprecipitation reagents (data not shown).
Western blot was performed with the Gs-targeted co-immunoprecipitated protein isolate. Briefly, 45 μL of denatured isolated was loaded into a Mini Protean TGX 4-15% acrylamide gradient gel. DynaMarker Protein MultiColor Stable was used as the protein ladder (Biodynamics Laboratory, Japan). Following gel electrophoresis (100V for 1 hour), protein was transferred from the gel to a methanol-activated PVDF membrane in 4°C overnight at 5V using transfer buffer (25 mM Tris, pH 8.3, 192 mM glycine, 20% methanol). Membranes were blocked (5% milk protein in TBS (50 mM Tris, 150 mM NaCl)) for 1 hour at room temperature. The membranes were incubated overnight at 4°C with antibodies against Opsin 3 (1:1000 LS Biosciences, Seattle, WA). Membrane images were developed using darkroom techniques for chemiluminescence (Biospectra UVP, Cambridge, UK) using Supersignal West Femto reagents (ThermoFisher, Waltham, MA) and 1:3000 goat anti-rabbit secondary antibody (BioRad, Hercules, CA).
Human USM Organ Bath Study:
With IRB approval (#AAAL4005), ex vivo organ bath experiments were performed utilizing strips of freshly obtained late gestation human USM (33 strips from 8 different patients). Pregnant (late gestation) human uterine tissue was dissected into strips approximately 4x6x12 mm. Strips were equilibrated under 2.5 gram of isometric tension for 1 hour in a modified Krebs-Henseleit buffer (concentration in mM: NaCl 115.0, KCl 2.5, CaCl2 1.9, MgSO4 2.5, NaHCO3 25.0, NaH2PO4 1.4, D-glucose 5.6). Isometric contractions were achieved via fixation of both ends of the uterine strip with noncompliant suture material. All USM strips were pre-contracted with 0.5 μM oxytocin (EC85) and allowed to equilibrate for 1 hour in the dark. Pretreatment with 9-cis retinal (5.7 μM) and GRK2 inhibitor (Methyl 5-[(E)-2-(5-nitrofuran-2-yl)ethenyl]furan-2-carboxylate, 20 μM) was used to prevent ambient light desensitization. USM strips were then exposed to increasing intensities of white light (11.6-17.8 Log2 [lux]) or specific wavelengths (no light, 630 nm (red), 532 nm (green) or 405 nm (blue)). Changes in muscle tension were measured by a Grass FT03 force transducer and digital recordings were acquired using Biopac MP100 hardware and AcqKnowledge software (Biopac Systems, Goleta, CA). Analysis was performed by comparing intervals of Integral Tension (g*sec), normalized to an intra-experimental control strip contracted with 0.5 μM oxytocin.
Statistical Analysis:
Data are presented as mean ± SEM with α=0.05. One-way ANOVA with Bonferroni's Multiple Comparison Test was used to analyze differences between groups.
Results
Opsin mRNA expression:
Representative gel images demonstrating the results of RT-PCR analysis screening for mRNA encoding extraocular opsin receptors are presented in Fig. 1c. Analysis was performed on total RNA isolated from tissues of late gestation human uterine smooth or non-pregnant uterine muscle using laser capture microdissection (LCMD). Although the screen included all extraocular opsin family members (OPN3, OPN4, OPN5) only messenger RNA encoding opsin 3 was detected in both non-pregnant and pregnant smooth muscle. Neither Opsin 4 nor 5 was detected in both non-pregnant and pregnant smooth muscle. Water blanks serving as negative controls (devoid of cDNA) did not demonstrate PCR product. Human brain (cDNA), which served as a positive control, demonstrated PCR products of expected sizes. All RT-PCR images are representative of three independent experiments.
Fig. 1c. OPN3 protein localization in hUSM cells.
Primary uterine smooth muscle cell stained for OPN3 protein (green) with fluorescein isothiocyanate-conjugated goat anti-rabbit immunoglobulin G as the secondary antibody. DAPI was used for counterstaining of cell nuclei (blue). Cells were viewed at 20x and membrane localization of the OPN3 receptor was observed.
OPN3 protein expression:
After screening extraocular opsin mRNA candidates, the OPN3 receptor was targeted for immunological detection of protein expression on confocal microscopy (Fig. 1b). Antibodies directed against OPN3 receptor demonstrate positive expression of OPN3. Positive control experiments demonstrate ocular expression of OPN3, while omitting primary antibody generated a negative result demonstrating the absence of non-specific binding of the secondary fluorescent antibody. Counterstaining of the nucleus with DAPI (blue) and actin staining (red) was performed to accentuate the smooth muscle cell phenotype. Immunocytochemistry was performed (Fig. 1c) to demonstrate cellular expression in primary cultured uterine smooth muscle cells.
Fig. 1b. OPN3 protein expression in hUSM.
Representative image of hUSM stained for OPN3 protein (green) with primary antibody against OPN3 and fluorescein isothiocyanate-conjugated goat anti-rabbit immunoglobulin G as the secondary antibody. Positive OPN3 protein expression was found in hUSM. DAPI was used for counterstaining of cell nuclei (blue). Mouse eye was used as a positive control for OPN3 protein expression. Counterstaining for F-actin (red) with rhodamine phalloidin was also performed to illustrate cellular localization of OPN3.
Human uterine smooth muscle light mediated relaxation in intensity dependent manner:
Precontracted human uterine smooth muscle was exposed to light at varying intensities following pretreatment with the GRK2 inhibitor and 9-cis retinal to demonstrate the relationship between light intensity and uterine smooth muscle relaxation. Human uterine smooth muscle strip relaxation increased as the white light intensity increased over the range of 0-228,000 lux units or 0-17.8 log2 (lux) units (Fig. 2a). Compiled reductions in percent tension as a function of light intensity dose response was determined. The half-maximal inhibitory intensity (IC50) values for oxytocin-induced (EC85) contractility of human uterine smooth muscle was 14.69 log2 lux units (Fig. 2b) (n=3).
Fig. 2. Intensity dependent relaxation of hUSM.


A. Representative tension tracing of hUSM strips in an organ bath pre-contracted with 0.5 μM oxytocin (EC85). Human USM strips were pretreated with a G protein receptor kinase 2 (GRK2) inhibitor (20 μM) and 9-cis retinal (5.7 μM) and exposed to white light at increasing intensities (11.6-17.8 Log2 [lux]). The top tracing demonstrates increased relaxation when human USM was exposed to increasing intensity of white light. Bottom tracing demonstrates no light (dark) treatment and time control.
B. Light induced relaxation is intensity (Log2 [lux]) dependent. Compiled reductions in percent Integral Tension (g*sec) as a function of light dose response in human USM pre-contracted with 0.5 μM oxytocin (EC85). Samples were pretreated with GRK2 inhibitor / 9-cis retinal to minimize light desensitization and then exposed to increasing intensities of white light (11.6-17.8 Log2 [lux]). Precontracted human USM photo-mediated inhibition to white light is dose (intensity) dependent, with significant relaxation seen at our lowest light threshold (11.7 Log2 [lux]) and an IC50 of 14.69 Log2 [lux] (***p<0.001, n=5 patients).
Light mediated USM relaxation is wavelength and chromophore dependent:
Oxytocin precontracted human uterine smooth muscle strips were pretreated with the GRK2 inhibitor and 9-cis retinal and then exposed to specific wavelengths of light [no light treatment, red (630 nm), green (532 nm) or blue (405 nm). The representative tracing (Fig. 3a) demonstrates spectral dependent relaxation at 405nm. Fig. 3b are compiled results of percent change in Integral Tension (g*sec) following treatment with specific wavelengths of light. There was significant relaxation in the treatment group receiving blue light compared to changes in tension measured in no light control. There was no significant change in tension measured in human USM strips receiving red or green light treatments, respectively. Fig. 3c illustrates maximal OPN3 mediated USM photo-relaxation induced at 405 nm is chromophore (9-cis retinal) dependent.
Fig. 3. Wavelength and chromophore dependent relaxation of human USM.
A. Representative tension tracings of human USM strips following contractile challenge with 0.5 μM oxytocin (EC85). All USM strips were pretreated with 20 mM GRK2 inhibitor and 5.7 μM 9-cis retinal and allowed to equilibrate (~1 hour). Top (black) tracing: no light treatments time control. Bottom (blue) tracing: sequential exposure to three specific wavelengths of light (red = 630 nm, green = 532 nm, and blue = 405 nm).
B. Compiled results of percent change in Integral Tension (g*sec) following treatment with specific wavelengths of light revealed significant relaxation in the treatment group receiving GRK inhibitor and 9-cis retinal pretreatment followed by 405 nm blue light in comparison to changes in tension measured in the no light control (***p<0.001). There was no significant change in tension (n.s.) measured in human USM strips receiving GRK2 inhibitor and 9-cis retinal pretreatments followed by red (630 nm) or green (532 nm) light treatments, respectively (n=8 patients).
C. Compiled results of percent change in Integral Tension (g*sec) following treatment with blue light (405 nm) alone, blue light (405 nm), GRK inhibitor + blue light, or 9-cis retinal + GRK inhibitor + blue light. USM strips exposed to blue light alone exhibited no significant change in tension compared to no-light control. Strips pretreated with both 9-cis retinal and GRK inhibitor and exposed to blue light (9-cis + GRK inh. + light) demonstrated significant relaxation compared to both the light-only group (Light) (***p<0.001) and GRK inhibitor + light group (GRK inh.+light) (**p<0.01, n=7 patients).
Light mediated USM relaxation is reversible subsequent to prolonged light exposure:
Oxytocin precontracted uterine smooth muscle strips were pretreated with GRK2 inhibitor and 9-cis retinal and then exposed to 1 hour of 405nm wavelength light. The representative tracing (Fig. 4a) demonstrates that the pro-relaxant effect of blue light was sustained for as long as the light was directed at the muscle strip, and the USM tension recovered significantly after cessation of blue light exposure (Fig. 4b). After spontaneously recovering contractility for 30 minutes, strips were challenged with 60 mM KCl to induce tetanic contraction and confirm tissue viability.
Fig. 4. Reversible relaxation of USM to 405 nm blue light.
A. Representative tension tracing of human USM strips in organ bath following contractile challenge with 0.5 μM oxytocin (EC85). All USM strips were pretreated with 5.7 μM 9-cis retinal and 20 μM GRK2 inhibitor and exposed to blue light (405 nm) for 1 hour. Tracing demonstrates sustained attenuation of tension and contractile frequency for the duration of blue light exposure, followed by spontaneous recovery of muscle tension when blue light was turned off. Strips were recontracted with 60 mM KCl. End point of recontraction resulted in tetany, confirming recovery of contractility.
B. Compiled results of percent change in Integral Tension (g*sec) of EC85 oxytocin-contracted USM strips pretreated with 9-cis retinal and GRK2 inhibitor and exposed to blue light (405 nm) for 1 hour, followed by 30 minutes of no light. There was a significant reduction in tension on sustained exposure to 405 nm light as well as significant recovery of tension upon cessation of light exposure (***p<0.001, n=8 patients).
Coimmunoprecipitation:
Primary human uterine smooth muscle cells were pretreated with darkness for at least 30 minutes followed by GRK2 inhibitor and 9-cis retinal treatment (15 minutes, concurrently), added to minimize desensitization effects of ambient light exposure on the blue light-mediated photorelaxation mechanism. Then, the cells were subjected to 435 nm blue light and lysed for coimmunoprecipitation with primary antibody for Gs. The isolated protein from the coimmunoprecipitation was used in a western blot for OPN3. We observed appropriate banding at the expected molecular weight for OPN3 (44 kDa) (Fig. 5). A no primary antibody membrane was used as a negative control. No nonspecific binding between secondary antibody and the coimmunoprecipitation isolate was seen (Fig. 5). Images are representative of three experiments (n=3 different patients).
Fig. 5. OPN3 associates with Gs in primary hUSM cells.

Representative image of an immunoblot for protein isolated from coimmunoprecipitation of Gs for two patients (labeled as 1, and 2; n=3 different patients). Antibodies against OPN3 revealed banding at the anticipated molecular weight (44 kDa). Cells were pretreated with darkness for 30 minutes as well as 28 μM 9-cis retinal and 95 μM GRK2 inhibitor for 15 minutes. Subsequently, cells were treated to 435 nm blue light (2 minutes) prior to coimmunoprecipitation. From the negative western blot primary control (right), there was no observed nonspecific binding between the secondary and the coimmunoprecipitation isolate.
Discussion:
1. Principle findings:
Late gestation human uterine smooth muscle expresses the opsin 3 (OPN3), which can be photo-activated ex vivo to elicit relaxation. Consistent with opsin 3 G-protein mediated signaling, we also observed dose (intensity) and spectral dependency that required cis-retinal and GRK2 inhibition. Under these conditions, we observed the greatest sensitivity to blue light (405 nm). In addition, we show OPN3 receptor coimmunoprecipitates with Gs, thus providing a plausible mechanism for the photo-mediated smooth muscle relaxation we observed.
2. Results:
Our findings represent the first description of a functional response of human late gestation uterine smooth muscle to light and the initial characterization of this response ex vivo. While the presence of a light-sensing receptor does raise some interesting questions as to its possible role in myometrial biology, many studies have now reported non-visual responses to light in other smooth muscle tissues. Indeed, the observation that light can mediate relaxation of smooth muscle dates back over 50 years [20]. Subsequent reports of “photo-relaxation” in smooth muscle followed in trachea, vascular smooth muscle, urethra and corpus cavernosum [21-23]. Interestingly, in smooth muscle this phenomenon has been shown to be wavelength dependent with two spectral regions, wavelengths at UV and blue spectra, eliciting relaxation via distinct mechanisms. At UV wavelengths photo-relaxation has been shown to occur in vascular smooth muscle that is mediated by a nitric oxide (NO) synthase-independent release of NO from local stores [24-26]. Interestingly, early studies demonstrated that UV light could induce a photo-relaxation effect in non-pregnant rabbit USM if the preparations were pre-treated with an exogenous NO donor (sodium nitrite 10−4 g/ml, an equivalent of 1.45 mmol) [27]. However, given the negative association with UV and its theoretical propensity to induce DNA damage, experimental translation of this phenomenon in pregnant models was never undertaken. Given this conundrum, we did not investigate UV-mediated photo-relaxation in human USM in this study. However, more recent work in vascular smooth muscle has revealed that at blue wavelengths, photo-relaxation is mediated by G-protein coupled receptor activation via the opsin family of receptors [11,16]. This group found OPN4 (melanopsin) and OPN3 (panopsin) can act as G-protein coupled receptors to mediate blue-light specific photo-relaxation responses. This group has also demonstrated a unique role between GRK2 and OPN3/OPN4 signaling by which excessive light activation mediates receptor desensitization, similar to β-arrestin mediated desensitization seen during β-adrenergic receptor over-stimulation. Our group has since extended these findings to airway smooth muscle (ASM), where we have demonstrated similar photo-mediated relaxation occurs in airway smooth muscle in response to blue light and OPN receptor activation [17]. Interestingly, human ASM expresses only OPN3 while in human vascular pulmonary smooth muscle there appears to be a co-expression of OPN3 and OPN4 [10, 17]. We observed the same conservation of OPN3 expression in human myometrium and no significant expression of OPN4 at both the mRNA and protein levels. Our findings are in agreement with published human transcriptome data from late gestation human myometrium which also shows expression of OPN3 at the mRNA level [27]. While this transcriptome analysis did not show a significant difference in expression of OPN3 as a function of normal labor, the comparison of OPN3 between pregnant and non-pregnant USM or in samples in which sPTL was present remains to be determined.
After confirming OPN3 expression in human USM, we then sought to determine whether this receptor could mediate a photo-relaxation effect. The first evidence for OPN3’s role in mediating a photo-relaxation effect was provided by illustrating that ex vivo relaxation is GRK2 dependent. Indeed, this was a significant issue for our initial ex vivo organ bath studies since we obtain our uterine tissue during elective c-section, when the tissue is exposed to very bright lights in the operating room. As a result, our initial studies using human myometrium demonstrated very small relaxation responses to either full spectrum (cold white light) or to 405 LED (cold blue light). Negative data is not shown. Interestingly, the same relationship is described in vascular and airway smooth muscle [16, 17]. Specifically, overstimulation of the OPN receptor ex vivo leads to a GRK2 mediated desensitization to subsequent light exposure. Therefore, in ex vivo experiments where tissue is exposed to ambient light an attenuated response to blue light alone is consistent with an OPN3/GRK2 coupling. We demonstrate that under reversal of this coupling by GRK2 inhibition, we were able to demonstrate photo-relaxation. This finding is in agreement with the other studies that illustrate OPN-mediated photo-relaxation in airway and vascular smooth muscle preparations exposed to ambient light require GRK2 inhibition to achieve maximal relaxation effects. Given that ambient light exposure represents several orders of light intensity beyond what these tissues are exposed to in vivo, it is logical that GRK2 inhibition is required to elicit photo-relaxation effects ex vivo. What remains to be answered is whether blue light delivered to myometrium in its native, dark (in vivo) setting may achieve photo-relaxation without requiring GRK inhibition. This enhancement of relaxation due to dark treatment was demonstrated in airway [17].
OPN3 receptor activation classically also displays dose dependency to light intensity and we demonstrate that human USM smooth muscle relaxation increases with an increase in white light intensity. Additionally, OPN3 activation is reportedly most sensitive to 415-470 nm light, but wavelength specificity is also determined by binding of specific chromophores. The importance of the chromophore cannot be understated. Photosensitivity requires the OPN3 receptor to complex with a chromophore and form a light sensing complex. In the retina, the prototypical chromophore is 11-cis retinal (a metabolite of vitamin A), which when bound to the opsin receptor forms a retinylidene protein, also known as the retinal OPN complex. This complex will change conformation in the presence of light and transmit this activation intracellularly through G-protein coupled signaling pathways. Different variations in OPN subtype and chromophore type can lead to differential activation sensitivity (e.g. wavelength) and differential downstream G protein couplings (i.e. Gs vs. Gq) [28-33]. In the case of smooth muscle, other studies have shown the chromophore 9-cis retinal induces sensitivity to blue-light wavelength as low as 405 nm. With this paradigm, we used a cold blue LED light (405 nm) to demonstrate a wavelength specific photo-relaxation effect in the presence of exogenous 9-cis retinal. Furthermore, we demonstrate diminished cold blue (405 nm)-mediated relaxation in the absence of 9-cis retinal. Using the chromophore 9-cis in this way implicates endogenous OPN3 activation. Given the role G protein receptor kinase (GRK) plays in desensitizing both OPN3 and β-adrenoceptors, we hypothesized myometrial OPN3 mediated photo-relaxation may share mechanistic overlap with β-adrenoceptor mediated relaxation by also signaling through Gs pathways. To implicate this mechanism, we performed a coimmunoprecipitation of OPN3 and Gs to explore whether in human myometrium these two molecules indeed couple to one another. Given our results that show Gs and OPN3 combine following activation with 435 nm light, photo-relaxation at this wavelength likely involves Gs-signaling pathways. However, this does not rule out other mechanisms that may also contribute; therefore, more detailed investigations are warranted [34].
3. Clinical implications:
In this translational study, we provide the first report of light-mediating relaxation of pre-contracted human uterine smooth muscle ex vivo. Additionally, we have provided several layers of evidence implicating the OPN3 receptor is not only endogenously expressed but pharmacologic targeting of the opsin 3 receptor indeed allows uterine tissue to become photosensitive. This holds several important potential clinical implications with respect to tocolysis of premature uterine contractions. The possibility that regionally applied light therapy may be used to promote uterine smooth muscle relaxation is a novel idea that may eventually be harnessed to offer therapeutic effects without many of the same side effects that plague current tocolytic drugs in use.
4. Strengths/Limitations:
The strengths of our findings are that we have strong functional data that complements and reinforces the mechanistic characterization underlying the photo-relaxation effect we observed in human pregnant myometrial tissue. This initial characterization provides a target receptor system to be explored by future studies. A major limitation of our study is that we harvest tissue following elective cesarean delivery, and the tissue is exposed to intense light that is several orders more intense than what is typically observed in vivo/in situ. To combat this desensitization, we employed inhibitors of this desensitization pathway in order to study this system ex vivo. Confirmatory and feasibility studies in vivo remain to be conducted. Additionally, future experiments will need to explore how chromophore modification may enhance light receptivity/spectral sensitivity. Furthermore, we have not replicated our findings on myometrium obtained from women with a history of PTL, so it is unknown what effects the pathogenesis of PTL may have on opsin-receptor activation and signaling. Lastly, we acknowledge that our co-immunoprecipitation with Gs does not establish a functional signaling relationship between these two molecules. However, it does provide qualitative evidence of a relationship between Opsin3 and a known pro-relaxant signaling pathway. Further signaling studies need to be done in order to validate the possibility of relaxation being solely attributable to Gs signaling.
5. Future Directions:
The endogenous role of nonvisual opsins have been described in the processes of embryonic and neonatal development as well as circadian rhythm [35-37]. As the issue photon tissue penetrance remains controversial, other evidence has demonstrated that other biologically relevant ligands do exist [38,39]. Future studies will be needed to be performed to fully uncover the endogenous activation and the role of nonvisual opsins in human peripheral tissue biology.
6. Conclusions:
This is the first report of light mediated relaxation of pre-contracted human myometrium. Activation of endogenous light receptors on human myometrium may become a non-invasive tocolytic strategy with minimal side effects.
Fig. 1a. OPSIN 3, 4, and 5 mRNA expression in hUSM.
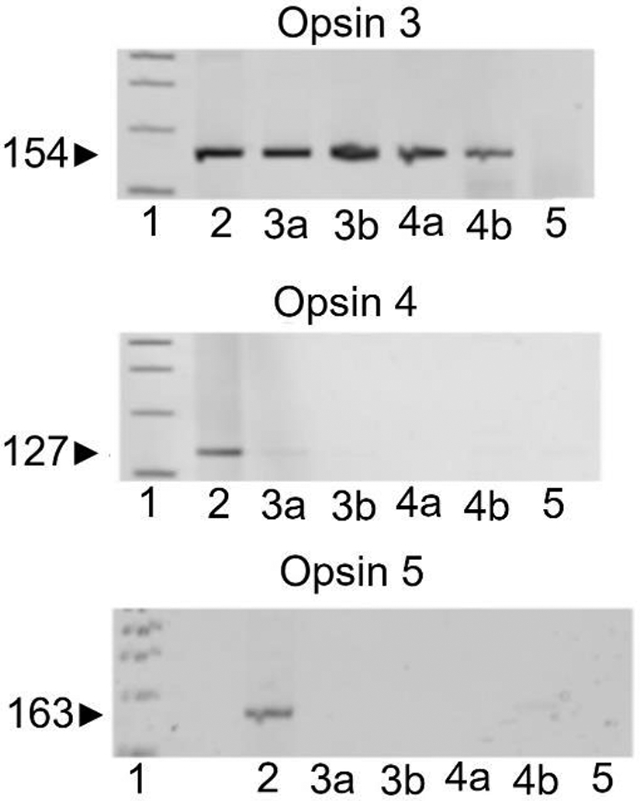
Representative gel image of RT-PCR products from primers targeting mRNA encoding opsin proteins, OPN3, OPN4, OPN5 in both nonpregnant (3a, 3b) and pregnant human (4a, 4b) USM tissue samples (n=3 different patients). Brain tissue was used as a positive control (2) and water blank was used as a negative control (5). Both nonpregnant and pregnant human USM samples demonstrate positive mRNA expression of OPSIN 3 mRNA. The lane labeled 1 represents the ladder.
Acknowledgments
Funding: Funding provided by Yim (Foundation for Anesthesia Education and Research - Mentored Research Training Grant), Vink (K08 HD088758) and Gallos (R01 HD082251)
Footnotes
Conflicts of interest/Competing interests: none for all authors
Ethics approval: Human uterus was obtained from healthy, pregnant patients (n=8 patients) undergoing elective C-section in accordance with the institutional review board (IRB)-approved protocol (#AAAI0337). De-identified human uterine tissue from the superior margin of the uterine incision was collected from patients at 38-40 weeks of gestation. For comparative purposes, non-pregnant human uterus samples were obtained after hysterectomies of similarly aged women (n=4 patients), as is described in the IRB-approved protocol (#AAAI0337).
Consent to participate: All subjects were consented to the acquisition of tissue as well as pertinent clinical information described in IRB approved protocols at Columbia University (#AAAI0337) (#AAAI0337).
Consent for publication: All authors consent to the publication of this paper.
Availability of data and material: All raw data available upon request.
Code availability: not applicable
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of a an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
References:
- 1.Center for Disease Control and Prevention. Premature Birth. Center for Disease Control and Prevention. 2018. https://www.cdc.gov/features/prematurebirth/index.html. Accessed 1 Feb 2019.
- 2.Younger JD, Reitman E, Gallos G. Tocolysis: Present and future treatment options. Semin Perinatol. 2017;41(8):493–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Haas DM, et al. Tocolytic therapy for preterm delivery: systematic review and network meta-analysis. BMJ. 2012;345:e6226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Michel MC, et al. Decreased myometrial β-adrenoceptors in women receiving β2-adrenergic tocolytic therapy: Correlation with lymphocyte β-adrenoceptors. Clin Pharmacol Ther 1989;45(1):1–8. [DOI] [PubMed] [Google Scholar]
- 5.Barnett SD, et al. S-Nitrosoglutathione reductase underlies the dysfunctional relaxation to nitric oxide in preterm labor. Sci Rep. 2018;8(5614). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Robinson C, et al. Oxytocin-induced desensitization of the oxytocin receptor. AJOG. 2003;188(2):497–502. [DOI] [PubMed] [Google Scholar]
- 7.Group TCPLI. Treatment of preterm labor with the beta-adrenergic agonist ritodrine. New Engl J Med. 1992;327(5):308–312. [DOI] [PubMed] [Google Scholar]
- 8.van Veen AJ, et al. Severe hypotension and fetal death due to tocolysis with nifedipine. BJOG. 2005;112(4):509–510. [DOI] [PubMed] [Google Scholar]
- 9.FDA. FDA Drug Safety Communication: New warnings against use of terbutaline to treat preterm labor. FDA. 2011. https://www.fda.gov/Drugs/DrugSafety/ucm243539.htm. Accessed 1 Feb 2019. [Google Scholar]
- 10.Barreto Ortiz S, et al. Opsin 3 and 4 mediate light-induced pulmonary vasorelaxation that is potentiated by G protein-coupled receptor kinase 2 inhibition. Am J Physiol Lung Cell Mol Physiol. 2018;314(1):L93–L106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Buscone S, et al. A new path in defining light parameters for hair growth: Discovery and modulation of photoreceptors in human hair follicle. Laser Surg Med. 2017;49(7):705–718. [DOI] [PubMed] [Google Scholar]
- 12.de Assis LVM, et al. Melanopsin and rhodopsin mediate UVA-induced immediate pigment darkening: Unravelling the photosensitive system of the skin. Eur J Cell Biol. 2018;97(3):150–162. [DOI] [PubMed] [Google Scholar]
- 13.Jiao J, et al. Opsin3 sensitizes hepatocellular carcinoma cells to 5-fluorouracil treatment by regulating the apoptotic pathway. Cancer Lett. 2012;320(1):96–103. [DOI] [PubMed] [Google Scholar]
- 14.Kasper G, et al. Different structural organization of the encephalopsin gene in man and mouse. Gene. 2002;295(1):27–32. [DOI] [PubMed] [Google Scholar]
- 15.Kato M, et al. Two Opsin 3-Related Proteins in the Chicken Retina and Brain: A TMT-Type Opsin 3 Is a Blue-Light Sensor in Retinal Horizontal Cells, Hypothalamus, and Cerebellum. PLoS One. 2016;11(11):e0163925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sikka G, Hussmann GP, Pandey D, et al. Melanopsin mediates light-dependent relaxation in blood vessels. PNAS. 2014; 111(50):17977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yim PD, et al. Airway smooth muscle photorelaxation via opsin receptor activation. Am J Physiol Lung Cell Mol Physiol. 2018;316(1):L82–L93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bernstein K, et al. Calcium-activated chloride channels anoctamin 1 and 2 promote murine uterine smooth muscle contractility. AJOG. 2014;211(6):688.e681–688.e610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Danielsson J, et al. Antagonists of the TMEM16A calcium-activated chloride channel modulate airway smooth muscle tone and intracellular calcium. Anesthesiology. 2015;123(3):569–581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Furchgott RF, et al. Relaxation of Arterial Strips by Light, and the Influence of Drugs on This Photodynamic Effect. J Pharmacol Exp Ther. 1955;113(113):1. [Google Scholar]
- 21.Chen X, Gillis CN. Enhanced photorelaxation in aorta, pulmonary artery and corpus cavernosum produced by BAY K 8644 or N-nitro-L-arginine. Biochem Biophys Res Commun. 1992;186(3):1522–1527. [DOI] [PubMed] [Google Scholar]
- 22.Chang KC, et al. NO- and N02-Carrying Molecules Potentiate Photorelaxation in Rat Trachea and Aorta. Biochem Biophys Res Commun. 1993;191(2):509–514. [DOI] [PubMed] [Google Scholar]
- 23.Triguero D, et al. Spontaneous photo-relaxation of urethral smooth muscle from sheep, pig and rat and its relationship with nitrergic neurotransmission. J Physiol (Lond). 2000,522 Pt 3(Pt 3):443–456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Flitney FW, Megson IL. Nitric oxide and the mechanism of rat vascular smooth muscle photorelaxation. J Physiol (Lond). 2003;550(Pt 3):819–828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kim JH, Hong Y, Shim CS. Mechanism of UV light-induced photorelaxation in isolated rat aorta. J Vet Sci. 2000;1(2):5. [PubMed] [Google Scholar]
- 26.Batenburg WWK, et al. Light-induced vs. bradykinin-induced relaxation of coronary arteries: do S-nitrosothiols act as endothelium-derived hyperpolarizing factors? J Hypertens. 2009;27(8):9. [DOI] [PubMed] [Google Scholar]
- 27.Ehrreich SJ, Furchgott RF. Relaxation of Mammalian Smooth Muscles by Visible and Ultraviolet Radiation. Nature. 1968;218(5142):682–684. [DOI] [PubMed] [Google Scholar]
- 28.Dartnall HJA, Mollon JD, Barlow HB. Human visual pigments: microspectrophotometric results from the eyes of seven persons. Proc R Soc Lond. 1983;220(1218):15. [DOI] [PubMed] [Google Scholar]
- 29.Kanaho Y, et al. Rhodopsin-enhanced GTPase activity of the inhibitory GTP-binding protein of adenylate cyclase. J Biol Chem 1984;259(12):7378–7381. [PubMed] [Google Scholar]
- 30.Karunarathne WKA, et al. Optical control demonstrates switch-like PIP3 dynamics underlying the initiation of immune cell migration. PNAS. 2013;110(17):E1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Koyanagi M, et al. Jellyfish vision starts with cAMP signaling mediated by opsin-Gs cascade. PNAS. 2008;105:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ockenfels A, Schapiro I, Gärtner W. Rhodopsins carrying modified chromophores – the ‘making of’, structural modelling and their light-induced reactivity. Photochem Photobiol Sci. 2016;15(2):297–308. [DOI] [PubMed] [Google Scholar]
- 33.Enright JM, et al. Cyp27c1 Red-Shifts the Spectral Sensitivity of Photoreceptors by Converting Vitamin A1 into A2. Curr Bio 2015;25(23):3048–3057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yau K-W, Hardie RC. Phototransduction motifs and variations. Cell. 2009;139(2):246–264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Buhr ED, Yue WWS, Ren X, Jiang Z, Liao H-WR, Mei X, et al. Neuropsin (OPN5)-mediated photoentrainment of local circadian oscillators in mammalian retina and cornea. Proceedings of the National Academy of Sciences. 2015;112(42):13093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vemaraju S, Nayak G, Buhr ED, Odaka Y, Zhang KX, Mocko JA, et al. An adipocyte light-Opsin 3 pathway regulates the circadian clock and energy balance. bioRxiv. 2018:368373. [Google Scholar]
- 37.Nguyen M-TT, Vemaraju S, Nayak G, Odaka Y, Buhr ED, Alonzo N, et al. An opsin 5–dopamine pathway mediates light-dependent vascular development in the eye. Nature Cell Biology. 2019;21(4):420–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.AzimiHashemi N, Erbguth K, Vogt A, Riemensperger T, Rauch E, Woodmansee D, et al. Synthetic retinal analogues modify the spectral and kinetic characteristics of microbial rhodopsin optogenetic tools. Nature Communications. 2014;5:5810. [DOI] [PubMed] [Google Scholar]
- 39.Shen YC, Sasaki T, Matsuyama T, Yamashita T, Shichida Y, Okitsu T, et al. Red-Tuning of the Channelrhodopsin Spectrum Using Long Conjugated Retinal Analogues. Biochemistry. 2018;57(38):5544–56. [DOI] [PubMed] [Google Scholar]









