Abstract
Background
Severe acute respiratory syndrome coronavirus 2 vaccination reduces morbidity and mortality associated with coronavirus disease 2019 (COVID-19); unfortunately, it is associated with serious adverse events, including sudden unexplained death (SUD).
Objective
We aimed to study the genetic basis of SUD after COVID-19 vaccination in Thailand.
Methods
From April to December 2021, cases with natural but unexplained death within 7 days of COVID-19 vaccination were enrolled for whole exome sequencing.
Results
Thirteen were recruited, aged between 23 and 72 years; 10 (77%) were men, 12 were Thai; and 1 was Australian. Eight (61%) died after receiving the first dose of vaccine, and 7 (54%) died after receiving ChAdOx1 nCoV-19; however, there were no significant correlations between SUD and either the number or the type of vaccine. Fever was self-reported in 3 cases. Ten (77%) and 11 (85%) died within 24 hours and 3 days of vaccination, respectively. Whole exome sequencing analysis revealed that 5 cases harbored SCN5A variants that had previously been identified in patients with Brugada syndrome, giving an SCN5A variant frequency of 38% (5 of 13). This is a significantly higher rate than that observed in Thai SUD cases occurring 8–30 days after COVID-19 vaccination during the same period (10% [1 of 10]), in a Thai SUD cohort studied before the COVID-19 pandemic (12% [3 of 25]), and in our in-house exome database (12% [386 of 3231]).
Conclusion
These findings suggest that SCN5A variants may be associated with SUD within 7 days of COVID-19 vaccination, regardless of vaccine type, number of vaccine dose, and presence of underlying diseases or postvaccine fever.
Keywords: COVID-19, Vaccination, Genetics, Sudden unexplained death (SUD), SCN5A
Introduction
Coronavirus disease 2019 (COVID-19) is a devastating infectious disease that has had resulted in health, social, and economic disasters worldwide. Vaccines are 1 intervention that is known to reduce the risk of severe disease and death. Unfortunately, serious adverse events after immunization with any type of COVID-19 vaccine have been demonstrated.1 These include fulminant myocarditis,2 cardiac arrhythmia,3 and sudden unexplained death (SUD).4 Although fatal cases with postmortem investigations have been reported,4 few studies have investigated the genetic basis of SUD after COVID-19 vaccination.
Methods
From April to December 2021, all cases of unexplained death occurring within 7 days of COVID-19 vaccination were sent for medicolegal investigation by forensic pathologists of the National Forensic Network comprising 9 medical centers throughout Thailand. All cases tested negative for severe acute respiratory syndrome coronavirus 2 by reverse transcription polymerase chain reaction. No consent from relatives was required because the autopsy was legally mandated. Those in whom the cause of death was found to be natural were sent for genetic studies. The research reported in this article adhered to Helsinki Declaration guidelines.
Genetic testing was approved by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University (institutional review board no. 264/62). Postmortem blood was obtained for genomic DNA extraction; then, whole exome sequencing (WES) was performed as described previously.5 Briefly, the sequencing libraries were enriched using a SureSelect Human All Exon V7 kit (Agilent Technologies [Santa Clara, CA])” here for SureSelect Human All Exon V7 kit and were sequenced using Illumina HiSeq 4000 (Illumina, San Diego, CA])” for Illumina HiSeq 4000 device. The only unequivocal, major genetic locus responsible for Brugada syndrome (BrS) is SCN5A-SCN10A,6 , 7 and its common variants could have susceptible effects on the phenotype.8 We therefore used minor allele frequencies of <5% as a filtering criterion for this SCN5A-SCN10A locus. Minor allele frequencies of <1% were used for all other genes. In addition, to be considered pathogenic, variants had to be reported in the literature in patients with BrS, cardiac arrhythmias, cardiomyopathies, or SUD.
Results
Thirteen cases comprising 10 men (77%) and 3 women (23%), aged between 23 and 72 years, were recruited (Table 1 ); 12 were Thai and 1 was Australian. Underlying diseases were reported in 5 patients, but none were arrhythmia. No cases had a history of SUD in family members. Eight (61%), 4 (31%), and 1 (8%) died after receiving the first, second, and third doses of vaccine, respectively. The total number of individuals receiving 1, 2, and 3 doses of COVID-19 vaccines in Thailand during the same period were 51,295,617 (49%), 46,145,566 (44%), and 7,002,986 (7%) doses, respectively. There was no significant correlation between the number of vaccine and SUD (Online Supplemental Table S1). SUD occurred after all types of vaccines used in Thailand, with 7 (54%), 2 (15%), 2 (15%), 1 (8%), and 1 (8%) deaths after receiving ChAdOx1 nCoV-19 (AstraZeneca), BBIBP-CorV (Vero Cells) (Sinopharm [Beijing]), CoronaVac (Sinovac), BNT162b2 (Pfizer/BioNTech), and mRNA-1273 (Moderna), respectively (Table 1). During the same period in Thailand, the number of administered doses of vaccines were 41,839,326 (41%), 14,091,514 (14%), 27,607,314 (27%), and 17,587,080 (18%) for AstraZeneca, Sinopharm, Sinovac, and Pfizer/BioNTech, respectively (no formal data for Moderna). There was no significant correlation between the type of vaccine and SUD (Online Supplemental Table S2). Fever after vaccination was noted in the self-reported web-based registry in 3 cases (23%). Ten (77%) and 11 (85%) deaths occurred within 24 hours and 3 days of vaccination, respectively. In 6 cases (46%), the autopsy identified no explainable causes of death. Another 6 were found to have cardiac causes including coronary atherosclerosis in 4 (31%), dilated cardiomyopathy (DCM) in 1 (8%), and arrhythmogenic right ventricular dysplasia in 1 (8%). The remaining case had thalassemia, leading to hemochromatosis and liver cirrhosis.
Table 1.
Clinical and genetic characteristics of Thai cases with SUD within 7 d of COVID-19 vaccinations
| No | Gender | Age (y) | Ethnic | Underlying disease | Type of vaccine∗ | Self-reported side effects | Days of death after vaccination | Forensic impression and autopsy findings | Gene† | DNA changes | Amino acid changes | Type of mutation | Zygosity | gnomAD |
Thai exomes (6462 alleles) | Reported in patients in PubMed or ClinVar | PubMed or ClinVar no. | ACMG (A) | SIFT (S) | Polyphen (P) | mCAP (m) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (alleles) | |||||||||||||||||||||
| I. Cases with variants in SCN5A (n = 5)‡ | |||||||||||||||||||||
| 1 | M | 23 | Thai | None | SV-AZ | Fever, headache, fatigue | 1 | Unexplained | SCN5A | c.3269C>T | p.Pro1090Leu | Missense | Hetero- | 411/245980 | 84 | BrS | 16344400, 15996170 | Y | |||
| SCN10A | c.1157T>G | p.Phe386Cys | Missense | Hetero- | 129/251388 | 16 | BrS, LQTS | 28407228, 24998131 | Y | Y | Y | ||||||||||
| 2 | M | 33 | Thai | Schizophrenia with clonazepam, diazepam, fluoxetine | SV-AZ | None | 1 | Unexplained | SCN5A | c.3578G>A | p.Arg1193Gln | Missense | Hetero- | 1414/272838 | 323 | BrS, atrioventricular block, and DCM | 16344400, 19808398 | ||||
| 3 | M | 43 | Thai | Asthma, gout | SV-SV-PZ | Fever, myalgia | 1 | DCM | SCN5A | c.833A>G | p.His278Arg | Missense | Hetero- | 1/152256 | 0 | BrS | 29202755 | Y | Y | Y | |
| 4 | M | 46 | Thai | Hyperthyroidism | SV | None | 3 | Unexplained | SCN5A | c.3578G>A | p.Arg1193Gln | Missense | Hetero- | 1414/272838 | 323 | BrS, atrioventricular block, and DCM | 16344400, 19808398 | ||||
| 5 | F | 28 | Thai | None | SV | None | 7 | Arrhythmogenic right ventricular dysplasia | SCN5A | c.3068G>A | p.Arg1023His | Missense | Hetero- | 72/279064 | 2 | BrS, LQTS | 16344400, 25935074 | Y | |||
| II. Cases with variants in non-SCN5A genes (n = 6) | |||||||||||||||||||||
| 6 | M | 35 | Thai | None | SP | Fever, pain in both knees | 1 | Unexplained | SCN3B | c.328G>A | p.Val110Ile | Missense | Hetero- | 77/251476 | 13 | BrS, sudden death of the young | 23257389, 27435932, 30662450 | Y | Y | ||
| 7 | M | 36 | Thai | Alcoholic hepatitis | SV-AZ | None | 1 | Unexplained | MYBPC3 | c.2807C>T | p.Thr936Met | Missense | Hetero- | 10/247610 | 0 | LVHT | 28798025 | Y | Y | Y | |
| Thai | Hetero- | ||||||||||||||||||||
| 8 | M | 38 | Thai | None | SP-SP | None | 1 | Coronary atherosclerosis | SLC22A5 | c.865C>T | p.Arg289Ter | Null | Hetero- | 4/251492 | 0 | PCD and SCD | 30863740, 26828774 | Y | |||
| SLC22A5 | c.1022G>A | p.Arg341Gln | Missense | Hetero- | 6/251448 | 2 | PCD and HCM | ClinVar:VCV000650541.1 | Y | Y | Y | ||||||||||
| 9 | M | 72 | Thai | None | AZ | Chest pain | 1 | Coronary atherosclerosis | TTN | c.20165-1G>A | N/A | Null | Hetero- | 6/244112 | 0 | DCM | ClinVar:VCV001053732.1 | ||||
| DNMT3A | c.939G>A | p.Trp313Ter | Null | Hetero- | 0 | 2 | DCM in Tatton-Brown-Rahman syndrome | 32435502 | |||||||||||||
| 10 | F | 53 | Thai | B-thalassemia§ | AZ | None | 1 | Thalassemia with liver cirrhosis | TTN | c.6668A>T | p.His2223Leu | Missense | Hetero- | 17/251140 | 48 | CM | 23861362 | Y | |||
| 11 | F | 59 | Thai | None | AZ | None | 1 | Coronary atherosclerosis | TTN | c.18530T>G | p.Leu6177Arg | Missense | Homo- | 26/273246 | 12 | SCD | 28704380 | ||||
| III. Cases with no candidate variants (n = 2) | |||||||||||||||||||||
| 12 | M | 34 | Thai | None | AZ | None | 1 | Unexplained | |||||||||||||
| 13 | M | 56 | Australian | None | MD | None | 4 | Coronary atherosclerosis | |||||||||||||
ACMG = The American College of Medical Genetics and Genomics; BrS = Brugada syndrome; CM = cardiomyopathy; COVID-19 = coronavirus disease 2019; DCM = dilated cardiomyopathy; F = female; HCM = hypertrophic cardiomyopathy; LQTS = long QT syndrome; LVHT = left ventricular hypertrabeculation; M = male; PCD = primary carnitine deficiency; SCD = sudden cardiac death; SUD = sudden unexplained death; Y = yes.
Vaccines: AZ: ChAdOx1 nCoV-19 (AstraZeneca); MD: mRNA-1273 (Moderna); PZ: BNT162b2 (Pfizer/BioNTech); SP: BBIBP-CorV (Vero Cells) (Sinopharm [Beijing]); SV: CoronaVac (Sinovac).
Transcripts used: SCN5A (NM_001099404.2), SCN10A (NM_006514.3), SCN3B (NM_018400.4), MYBPC3 (NM_000256.3), SLC22A5 (NM_003060.4), TTN (NM_001256850.1), DNMT3A (NM_022552.5).
In the last 4 columns, (A): pathogenic; (S) deleterious; (P): damaging; (m): pathogenic.
Compound heterozygous for p.Glu27Lys and p.Phe42Leufs∗19 in the HBB gene.
Five cases harbored variants in SCN5A (Table 1); 4 were men, aged between 23 and 46 years, who died within 3 days of the last COVID-19 vaccination. Only 2 of our 5 SUD cases with SCN5A variants reported fever. Six other SUD cases in our cohort harbored variants in other genes, which were previously reported to be associated with cardiac arrhythmias, cardiomyopathies, or SUD. Two had no variants, consistent with the filtering criteria.
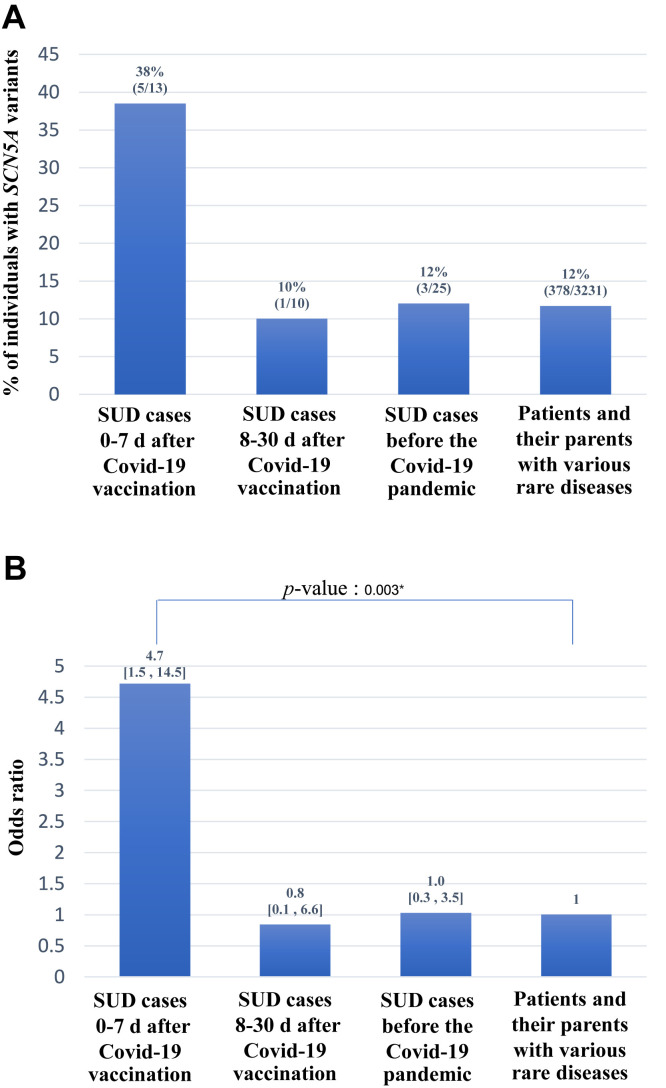
The frequency of SCN5A variants in our cohort was 38% (5 of 13). For comparative purposes, we thereafter determined the frequency of SCN5A variants in Thai SUD cases occurring 8–30 days after COVID-19 vaccination during the same period. The rate in these SUD cases that occurred after 7 days was considerably lower (1 of 10 [10%]; Online Supplementary Table S3), as were the rates in a Thai cohort of SUD before the COVID-19 pandemic (3 of 25 [12%]),9 and in our in-house exome database enrolling patients and their parents with various rare diseases (186 of 3231 [12%]), after applying the same filtering criteria (Figure 1A). The odds ratios for SCN5A variants and SUD in these other groups and that of the present study are shown in Figure 1B. Evidence from our present study supports the hypothesis that SUD within 7 days of COVID-19 vaccination is associated with SCN5A variants.
Figure 1.
A: Frequencies of SCN5A variants in our cohort with sudden unexplained death (SUD) cases within 7 days of coronavirus disease 2019 (COVID-19) vaccination, in Thai SUD cases during 8–30 days after COVID-19 vaccination during the same period, in a Thai cohort of SUD before the COVID-19 pandemic, and in our in-house exome database enrolling patients and their parents with various rare diseases. B: The odds ratios and 95% confidence intervals for SCN5A variants and SUD in study groups, compared with the in-house exome controls as a reference. The asterisk indicates that the P-value reaches statistical significance of less than .05.
Discussion
This study aimed to identify associations between genetic variants and SUD that occurred within 7 days of COVID-19 vaccination. A total of 13 variants in 7 genes were identified by WES in 11 of 13 cases (85%), compared with approximately one-third to one-half of the diagnostic yield of WES for patients with previously undiagnosed diseases.10 , 11 This suggests that SUD after COVID-19 vaccination may have a genetic susceptibility more commonly identified by WES, compared with other rare and undiagnosed diseases.
Four SCN5A variants were found in 5 cases. All have previously been identified in patients with BrS. Two variants—c.833A>G in case 3 and c.3068G>A in case 5—had low allele frequencies: 0 and 2 alleles, respectively, out of 6462 alleles in our in-house exome database. Case 3 also had DCM, which could possibly have resulted from SCN5A variants,12 suggesting that SCN5A c.833A>G (p.His278Arg) is a severe loss-of-function variant. The frequency of SCN5A c.3269C>T (p.Pro1090Leu) identified in case 1 is 0.013 (84 of 6462) in our database. In addition to carrying the SCN5A variant, case 1 harbored a c.1157T>G (p.Phe386Cys) variant in SCN10A, previously found in patients with BrS (Table 1). Variants in SCN5A and SCN10A are a major SUD risk locus.6 Although the SCN5A c.3578G>A (p.Arg1193Gln) variants found in cases 2 and 4 have an allele frequency of 0.049 (323 of 6462) in our database, these have previously been demonstrated to encode a protein with aberrant function contributing to various cardiac arrhythmias, including long QT syndrome and BrS.13 It was also previously identified in 2 of 25 (8%) Thai SUD cases.9 Moreover, case 2 had schizophrenia and was prescribed with clonazepam, diazepam, and fluoxetine. Psychotropic drugs are associated with sudden death of cardiac origin,14 especially diazepam, which causes a faster block and unblock of Na+ channels,15 and may therefore aggravate abnormalities caused by the aberrant SCN5A variant. Taken together, these lines of evidence suggest that all 4 SCN5A variants had a contributory role in the SUD of these 5 cases.
Six other SUD cases in our cohort harbored variants in 5 other genes. Three cases carried likely pathogenic variants in TTN, a huge gene encoding a 34,350 -amino acid protein in which mutations are known to cause cardiomyopathies.16 The heterozygous splice site c.20165-1G>A variant identified in case 9 was previously reported in the ClinVar database in an individual with DCM.17 Case 9 was also heterozygous for a nonsense variant, c.939G>A (p.Trp313Ter) in DNMT3A, mutations in which cause Tatton-Brown-Rahman syndrome, a syndrome with possible cardiac abnormalities, inherited in an autosomal dominant manner.18 Case 10 with underlying beta thalassemia was heterozygous for the c.6668A>T (p.His2223Leu) variant in TTN previously reported in a patient with cardiomyopathy.19 Case 11 was homozygous for the TTN c.18530T>G (p.Leu6177Arg) variant, which was recently identified heterozygously in a 25-year-old Thai male with SUD.9 Cases 6, 7, and 8 harbored variants in SCN3B, MYBPC3, and SLC22A5, which were previously associated with BrS,20 left ventricular hypertrabeculation,21 and autosomal recessive primary carnitine deficiency with hypertrophic cardiomyopathy and sudden death,22 respectively.
The association between SCN5A variants and SUD within 7 days of COVID-19 vaccination (Figure 1) might be due to COVID-19 vaccination unmasking fatal arrhythmia in individuals with SCN5A. Fever is a well-known trigger for arrhythmia in BrS.23 A possible mechanism might therefore include an indirect effect of the vaccine in which an individual with a susceptible pathogenic SCN5A variant had vaccine-induced fever and malaise serving as a precipitating factor leading to fatal arrhythmias.24 Nonetheless, since fever did not seem to play a major role in our study, the other possibility is a direct effect. The vaccine may cause myocarditis in the right ventricular outflow tract area, which in turn unmasks the preexisting aberrant conduction in individuals with SCN5A 25 and leads to death. It is important to note that the possible association of genetic variants and SUD after COVID-19 vaccination in our study while plausible is primarily hypothesis generating and must be independently confirmed by other studies.
Conclusion
Our study suggests that SCN5A variants could be associated with SUD within 7 days of COVID-19 vaccination, regardless of vaccine type, number of vaccine dose, and presence of underlying diseases or postvaccine fever. Given the observational nature of our study, these findings should be further explored and confirmed in surveillance programs in other settings. Until then, it seems prudent to closely monitor individuals who harbor variants in SCN5A, and possibly in other genes that predispose to cardiac arrhythmias or cardiomyopathies, for 7 days after the administration of COVID-19 vaccines, regardless of preexisting underlying diseases and the presence of vaccination-associated fever.
Acknowledgments
We thank Professor Arthur A.M. Wilde, PhD of the Heart Centre, University of Amsterdam, The Netherlands, for critical comments.
Footnotes
Funding Sources: This work was supported by the Health Systems Research Institute (65-040) and National Research Council of Thailand (N35A640274).
Disclosures: The authors have no conflicts of interest to disclose.
Supplementary data associated with this article can be found in the online version at https://doi.org/10.1016/j.hrthm.2022.07.019.
Appendix
References
- 1.Beatty A.L., Peyser N.D., Butcher X.E., et al. Analysis of COVID-19 vaccine type and adverse effects following vaccination. JAMA Netw Open. 2021;4 doi: 10.1001/jamanetworkopen.2021.40364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Verma A.K., Lavine K.J., Lin C.Y. Myocarditis after Covid-19 mRNA vaccination. N Engl J Med. 2021;385:1332–1334. doi: 10.1056/NEJMc2109975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Patone M., Mei X.W., Handunnetthi L., et al. Risks of myocarditis, pericarditis, and cardiac arrhythmias associated with COVID-19 vaccination or SARS-CoV-2 infection. Nat Med. 2022;28:410–422. doi: 10.1038/s41591-021-01630-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sessa F., Salerno M., Esposito M., Di Nunno N., Zamboni P., Pomara C. Autopsy findings and causality relationship between death and COVID-19 vaccination: a systematic review. J Clin Med. 2021;10:5876. doi: 10.3390/jcm10245876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shotelersuk V., Wichadakul D., Ngamphiw C., et al. The Thai reference exome (T-REx) variant database. Clin Genet. 2021;100:703–712. doi: 10.1111/cge.14060. [DOI] [PubMed] [Google Scholar]
- 6.Barc J., Tadros R., Glinge C., et al. Genome-wide association analyses identify new Brugada syndrome risk loci and highlight a new mechanism of sodium channel regulation in disease susceptibility. Nat Genet. 2022;54:232–239. doi: 10.1038/s41588-021-01007-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Makarawate P., Glinge C., Khongphatthanayothin A., et al. Common and rare susceptibility genetic variants predisposing to Brugada syndrome in Thailand. Heart Rhythm. 2020;17:2145–2153. doi: 10.1016/j.hrthm.2020.06.027. [DOI] [PubMed] [Google Scholar]
- 8.Huang Y., Chen X.M., Barajas-Martinez H., Jiang H., Antzelevitch C., Hu D. Common variants in SCN10A gene associated with Brugada syndrome. Hum Mol Genet. 2021;31:157–165. doi: 10.1093/hmg/ddab217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Suktitipat B., Sathirareuangchai S., Roothumnong E., et al. Molecular investigation by whole exome sequencing revealed a high proportion of pathogenic variants among Thai victims of sudden unexpected death syndrome. PLoS One. 2017;12 doi: 10.1371/journal.pone.0180056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Splinter K., Adams D.R., Bacino C.A., et al. Effect of genetic diagnosis on patients with previously undiagnosed disease. N Engl J Med. 2018;379:2131–2139. doi: 10.1056/NEJMoa1714458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kamolvisit W., Phowthongkum P., Boonsimma P., et al. Rapid exome sequencing as the first-tier investigation for diagnosis of acutely and severely ill children and adults in Thailand. Clin Genet. 2021;100:100–105. doi: 10.1111/cge.13963. [DOI] [PubMed] [Google Scholar]
- 12.McNair W.P., Sinagra G., Taylor M.R., et al. Familial Cardiomyopathy Registry Research Group SCN5A mutations associate with arrhythmic dilated cardiomyopathy and commonly localize to the voltage-sensing mechanism. J Am Coll Cardiol. 2011;57:2160–2168. doi: 10.1016/j.jacc.2010.09.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang Q., Chen S., Chen Q., et al. The common SCN5A mutation R1193Q causes LQTS-type electrophysiological alterations of the cardiac sodium channel. J Med Genet. 2004;41:e66. doi: 10.1136/jmg.2003.013300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Timour Q., Frassati D., Descotes J., Chevalier P., Christe G., Chahine M. Sudden death of cardiac origin and psychotropic drugs. Front Pharmacol. 2012;3:76. doi: 10.3389/fphar.2012.00076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Backus K.H., Pflimlin P., Trube G. Action of diazepam on the voltage-dependent Na+ current: comparison with the effects of phenytoin, carbamazepine, lidocaine and flumazenil. Brain Res. 1991;548:41–49. doi: 10.1016/0006-8993(91)91104-9. [DOI] [PubMed] [Google Scholar]
- 16.Hinson J.T., Chopra A., Nafissi N., et al. Heart disease: titin mutations in iPS cells define sarcomere insufficiency as a cause of dilated cardiomyopathy. Science. 2015;349:982–986. doi: 10.1126/science.aaa5458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.ClinVar. NM_001267550.2(TTN):c.21116-1G>A. https://www.ncbi.nlm.nih.gov/clinvar/variation/VCV001053732.1 Accession VCV001053732.1.
- 18.Cecchi A.C., Haidar A., Marin I., Kwartler C.S., Prakash S.K., Milewicz D.M. Aortic root dilatation and dilated cardiomyopathy in an adult with Tatton-Brown-Rahman syndrome. Am J Med Genet A. 2022;188:628–634. doi: 10.1002/ajmg.a.62541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ng D., Johnston J.J., Teer J.K., et al. NIH Intramural Sequencing Center (NISC) Comparative Sequencing Program. Interpreting secondary cardiac disease variants in an exome cohort. Circ Cardiovasc Genet. 2013;6:337–346. doi: 10.1161/CIRCGENETICS.113.000039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ishikawa T., Takahashi N., Ohno S., et al. Novel SCN3B mutation associated with Brugada syndrome affects intracellular trafficking and function of Nav1.5. Circ J. 2013;77:959–967. doi: 10.1253/circj.cj-12-0995. [DOI] [PubMed] [Google Scholar]
- 21.Miszalski-Jamka K., Jefferies J.L., Mazur W., et al. Novel genetic triggers and genotype-phenotype correlations in patients with left ventricular noncompaction. Circ Cardiovasc Genet. 2017;10 doi: 10.1161/CIRCGENETICS.117.001763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mazzini M., Tadros T., Siwik D., et al. Primary carnitine deficiency and sudden death: in vivo evidence of myocardial lipid peroxidation and sulfonylation of sarcoendoplasmic reticulum calcium ATPase 2. Cardiology. 2011;120:52–58. doi: 10.1159/000333127. [DOI] [PubMed] [Google Scholar]
- 23.Keller D.I., Rougier J.S., Kucera J.P., et al. Brugada syndrome and fever: genetic and molecular characterization of patients carrying SCN5A mutations. Cardiovasc Res. 2005;67:510–519. doi: 10.1016/j.cardiores.2005.03.024. [DOI] [PubMed] [Google Scholar]
- 24.Caturano A., Pafundi P.C., Sasso F.C., Dendramis G., Brugada P., Russo V. Brugada syndrome and COVID-19 vaccines. Europace. 2021;23:1871–1872. doi: 10.1093/europace/euab211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Blok M., Boukens B.J. Mechanisms of arrhythmias in the Brugada syndrome. Int J Mol Sci. 2020;21:7051. doi: 10.3390/ijms21197051. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.