Abstract
Measurement of the flux through the citrate fermentation pathway in resting cells of Lactococcus lactis CRL264 grown in a pH-controlled fermentor at different pH values showed that the pathway was constitutively expressed, but its activity was significantly enhanced at low pH. The flux through the citrate-degrading pathway correlated with the magnitude of the membrane potential and pH gradient that were generated when citrate was added to the cells. The citrate degradation rate and proton motive force were significantly higher when glucose was metabolized at the same time, a phenomenon that could be mimicked by the addition of lactate, the end product of glucose metabolism. The results clearly demonstrate that citrate metabolism in L. lactis is a secondary proton motive force-generating pathway. Although the proton motive force generated by citrate in cells grown at low pH was of the same magnitude as that generated by glucose fermentation, citrate metabolism did not affect the growth rate of L. lactis in rich media. However, inhibition of growth by lactate was relieved when citrate also was present in the growth medium. Citrate did not relieve the inhibition by other weak acids, suggesting a specific role of the citrate transporter CitP in the relief of inhibition. The mechanism of citrate metabolism presented here provides an explanation for the resistance to lactate toxicity. It is suggested that the citrate metabolic pathway is induced under the acidic conditions of the late exponential growth phase to make the cells (more) resistant to the inhibitory effects of the fermentation product, lactate, that accumulates under these conditions.
The main activity of lactic acid bacteria (LAB) is the conversion of carbohydrates to lactate. At the end of the fermentation process, lactate accumulates in the medium to high concentrations and inhibits growth. The toxicity of lactate is both an advantage and a disadvantage in the application of LAB. The toxicity of lactate results in reduced product formation and affects the viability of cultures. For similar reasons, lactate can be used as a food preservative.
A limited number of LAB are also capable of fermenting carboxylic acids like citrate and malate. The transporter responsible for the uptake of citrate, CitP, has been cloned, sequenced, and functionally characterized. In vitro experiments using membrane vesicles have demonstrated that CitP catalyzes efficient exchange of citrate and lactate, suggesting that the physiological role of CitP is the concomitant uptake of citrate and excretion of lactate produced by glycolysis (2, 11). This could provide citrate-fermenting LAB with a mechanism for resistance to the inhibitory effects of lactate accumulated in the medium.
The mechanism of citrate fermentation has been studied in detail in Leuconostoc mesenteroides (11, 12). The first step in the breakdown of citrate inside the cell involves its conversion to acetate and oxaloacetate by citrate lyase, a three-subunit enzyme that recently was cloned and sequenced (3). In the next step, oxaloacetate is decarboxylated by oxaloacetate decarboxylase yielding pyruvate and carbon dioxide. The pathway generates a proton motive force (PMF) by a secondary mechanism (9). Electrogenic exchange of divalent citrate and monovalent lactate, catalyzed by CitP, efficiently generates a membrane potential, inside negative. Moreover, a pH gradient (inside alkaline) is formed through the consumption of scalar protons in the decarboxylation of oxaloacetate. Together, the membrane potential and pH gradient constitute the PMF, which contributes significantly to the growth advantage observed during cometabolism of citrate and glucose by L. mesenteroides. A similar mechanism of secondary PMF generation by citrate metabolism has been reported in Leuconostoc oenos (16).
Although the initial metabolic steps of citrate metabolism are the same in Lactococcus lactis, the energetics of the pathway have been a matter of debate (see reference 2 and references therein). In the study described in reference 2, it was demonstrated that the amino acid sequences of the citrate transporters of L. mesenteroides and L. lactis are 99% identical and that the functional properties of the two proteins are indistinguishable. Since the energy-coupling mechanism of the transporter plays a crucial role in a secondary metabolic energy-generating pathway, these results strongly suggest that citrate metabolism in L. lactis also generates a PMF. Part of the problems encountered in the energetic studies of citrate metabolism in L. lactis may relate to the recent finding that the citrate transporter is induced by acid stress (5). In this study, we analyzed the energetics of citrate metabolism in L. lactis grown at different pH values and showed unequivocally that the pathway generates metabolic energy by a mechanism similar to that described for L. mesenteroides. Growth experiments suggest an important physiological function of the pathway related to the induction of the pathway at low pH: resistance to lactate toxicity. The proposed mechanism of citrate metabolism provides an explanation for the observed resistance.
MATERIALS AND METHODS
Materials.
The 3,3′-dipropylthiocarbocyanine iodide [DiSC3(5)] and 2′,7′-bis-(2-carboxyethyl)-5(and -6)-carboxyfluorescein (BCECF) probes were obtained from Molecular Probes, Eugene, Oreg. Enzyme kits for citrate and glucose were obtained from Boehringer, Mannheim, Germany. M17 broth was obtained from Difco Laboratories, Detroit, Mich.
Growth conditions.
L. lactis subsp. lactis biovar diacetylactis CRL264 was obtained from the collection of the Centro de Referencia para Lactobacilos, Tucuman, Argentina (18), and grown in a pH-controlled fermentor in M17 broth containing 0.5% (wt/vol) glucose and 20 mM citrate at pH 4.5 or 6.5. The fermentor had a volume of 150 ml and was stirred slowly with a magnetic bar. The pH was monitored continuously, and the pH of the growth medium was kept constant by two pumps controlled by the pH meter and connected to flasks containing 1 M NaOH and 1 M HCl. The fermentor was inoculated with an overnight batch culture in M17 medium containing 0.5% glucose. Alternatively, cells were grown in a batch culture in the same medium at pH 5 in 100-ml serum bottles. Cells were harvested in the exponential growth phase at an optical density at 660 nm (OD660) of 0.6 to 0.8 by centrifugation, washed once with 50 mM potassium phosphate buffer (pH 5.5), and stored on ice in the same buffer until used. Growth rates were determined in sterile low-protein-binding microplates (Greiner). Exponentially growing cultures were diluted in M17 broth (pH 5) containing the substrates and inhibitors indicated to a final OD660 of 0.05 in a total volume of 250 μl. Growth was monitored by measuring the OD660 every 10 min in a SPECTRAmax 340 (Molecular Devices) microplate reader.
Measurement of internal pH and membrane potential.
The internal pH was measured by loading the cells with the pH-sensitive fluorescent probe BCECF as previously described (14). Briefly, 1 μl of a 10 mM BCECF solution was added to 20 μl of a cell suspension typically containing 50-mg/ml protein, followed by 2.5 μl of 0.5 N HCl to shock the probe into the cells. The suspension was left for 5 min at room temperature, after which 1 ml of 50 mM potassium phosphate buffer (pH 5.5) was added. The cells were spun down, resuspended in 200 μl of buffer, and kept on ice until use. For each experiment, 10 μl of the BCECF-loaded cells was added to 2 ml of buffer in a 3-ml cuvette equilibrated at 30°C. The cuvette was stirred with a magnetic stirring bar. The fluorescent signal was sampled every second. Distortions in the traces caused by the opening of the measurement compartment necessary to add substrates were removed from the traces. This resulted in loss of data during the first 5 to 6 s after an addition. The internal pH was determined from the fluorescence signal as previously described (14).
The membrane potential was measured qualitatively with the fluorescent probe DiSC3(5) (20). An increase in electrical potential across the membrane correlates with a decrease in fluorescence intensity. Freshly harvested cells were resuspended to an OD660 of 6 in 50 mM potassium phosphate buffer (pH 5.5) and kept on ice until use. For each experiment, 10 μl of the cell suspension was added to 2 ml of buffer in a cuvette equilibrated at 30°C, and 4 μl of a 1 mM solution of DiSC3(5) was subsequently added. In the membrane potential and pH gradient measurements, glucose and citrate were added at concentrations of 0.5 and 2 mM, respectively, when indicated.
Measurement of citrate and glucose consumption rates.
Cells were harvested and resuspended to an OD660 of 6 in 50 mM potassium phosphate buffer (pH 5.5). Citrate utilization was initiated by the addition of 500 μl of cells to 1,500 μl of buffer containing 2 mM citrate. When indicated, glucose and lactate were included at concentrations of 0.5 and 2 mM, respectively. Glucose utilization was initiated by addition of 50 μl of cells to 1,950 μl of buffer containing 0.5 mM glucose and 2 mM citrate when indicated. At the indicated times, 250 μl of the cell suspension was centrifuged for 15 s in a Eppendorf tabletop centrifuge operating at maximal speed, and a sample of the supernatant was stored in liquid nitrogen until analysis. The concentrations of citrate, pyruvate, and glucose in the supernatants were measured by commercially available enzyme kits for citrate and d-glucose (Boehringer). The protocol for the measurement of citrate was modified slightly to allow the measurement of pyruvate (and oxaloacetate) at the same time, as described before (12). Both protocols were modified for use in 96-well plates as follows. For the citrate assay, 100 μl of the buffer provided by the manufacturer was mixed with 50 μl of water, after which the A340 was measured. Subsequently, 50 μl of the supernatant was added and the OD was read again. The difference between the two readings is a measure of the pyruvate (or oxaloacetate) concentration in the sample. After addition of 1 μl of the citrate lyase solution, the absorbance was read again, which provides the data for the calculation of the citrate concentration as indicated in the protocol. For glucose determination, the volumes in the manufacturer’s protocol were scaled down to give a total volume of 200 μl. The ODs were read by using a SPECTRAmax 340 microplate spectrophotometer.
RESULTS
Citrate metabolism in resting cells of L. lactis CRL264 grown at pHs 6.5 and 4.5.
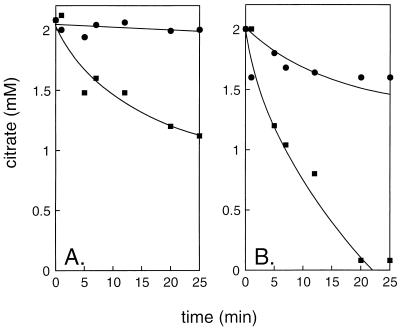
Growth of LAB results in acidification of the growth medium due to the production of lactic acid. To measure the effect of pH on the induction of the citrate metabolic pathway, L. lactis CRL264 was grown in a pH-controlled fermentor. Resting cells cultured at different pH values on rich medium containing glucose and citrate were assayed for the ability to metabolize citrate in the same buffer at pH 5.5 (Fig. 1). Cells grown at a constant pH of 6.5 metabolized citrate very slowly, while cells preincubated with glucose decreased the citrate concentration significantly, revealing the presence of the enzymes of the citrate metabolic pathway. The rate of citrate metabolism by cells cultured at pH 4.5 was significantly higher. Also, in these cells, the rate of metabolism increased in the presence of glucose. The results were not significantly different when citrate was omitted from the growth medium (data not shown). The first intermediates in citrate breakdown in lactic acid bacteria, the keto acids oxaloacetate and pyruvate, could not be detected in the medium (data not shown). This contrasts with the results of similar experiments with L. mesenteroides 19D in which citrate metabolism resulted in a burst of pyruvate in the medium (12). This suggests that in L. lactis, pyruvate-converting routes are much more active. Under the conditions of the experiments, the products are the neutral compounds acetoin and diacetyl, which leave the cell by passive diffusion (7).
FIG. 1.
Citrate metabolism by resting cells of L. lactis CRL264. Cells grown in a pH-controlled fermentor at pH 6.5 (A) and pH 4.5 (B) were preincubated for 10 min at 30°C in 50 mM potassium phosphate (pH 5.5), after which citrate (●) or citrate plus glucose (■) was added. At the indicated time points, residual citrate in the medium was analyzed as described in Materials and Methods.
In conclusion, the citrate metabolic pathway is constitutively expressed in L. lactis CRL264, but a higher activity is observed when the cells are grown under acidic conditions, suggesting higher levels of expression of the rate-controlling enzyme(s). The higher rate of citrate metabolism in the presence of glucose suggests that metabolic-energy-dependent steps play a role in the pathway, for instance, uptake of citrate from the medium, but see below.
Energetics of citrate metabolism in L. lactis CRL264.
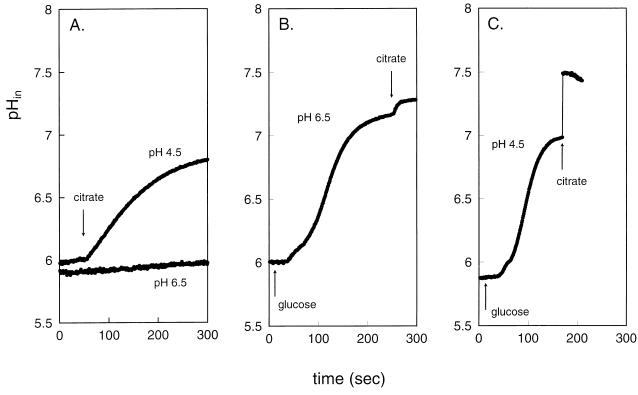
The energetic consequences of citrate metabolism in resting cells were investigated by monitoring, under the same conditions of the experiments presented in Fig. 1, the two components of the PMF, i.e., the pH gradient across the cytoplasmic membrane and the membrane potential. Changes in the pH gradient were monitored by measuring the internal pH with the pH-sensitive fluorescent probe BCECF that was trapped inside the cells. The resting cells maintained a pH gradient across the membrane of about 0.5 pH unit, inside alkaline, when suspended in pH 5.5 buffer (Fig. 2). The citrate consumption rates in the absence of glucose by cells grown at constant pH values of 6.5 and 4.5 correlated with the changes observed in the internal pH. Cells grown at pH 6.5 showed hardly any citrate degradation, and no change in internal pH was observed, while citrate consumption by cells grown at acidic pH correlated with an increase in cytoplasmic pH, resulting in an increase of the transmembrane pH gradient of almost 1.5 pH units, (Fig. 2A). The rise in internal pH is diagnostic for the generation of metabolic energy during citrate metabolism (8). Addition of glucose to resting cells raised the internal pH to about 7 in cells grown at both pHs 6.5 and 4.5 (Fig. 2B and C). Alkalinization of the cytoplasm is caused by proton pumping by F1F0-ATPase upon hydrolysis of ATP produced in glycolysis. The results presented in Fig. 2 show that addition of citrate under these “energized” conditions resulted in further increases in the internal pH and in the transmembrane pH gradient. The observation that cells grown at acidic pH have a greater potential to raise the pH gradient in the presence of citrate correlates with the higher flux through the citrate metabolic pathway (Fig. 1B).
FIG. 2.
Transmembrane pH gradient during citrate metabolism by resting cells of L. lactis CRL264. Cells grown in a pH-controlled fermentor at pH 6.5 (A, B) and pH 4.5 (A, C) were loaded with the fluorescent pH indicator probe BCECF in 50 mM potassium phosphate (pH 5.5), after which citrate (A) or glucose followed by citrate (B, C) was added at the time points indicated by the arrows.
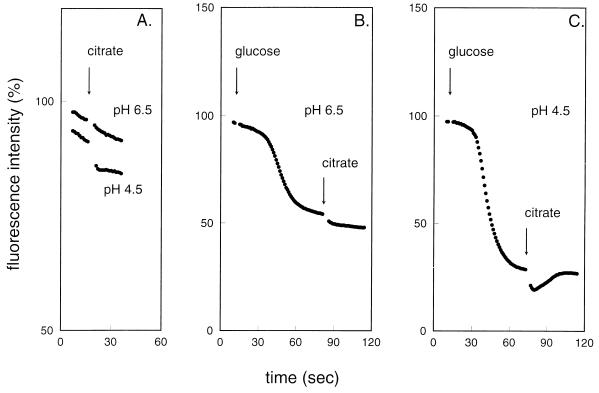
The membrane potential was measured qualitatively by the fluorescent probe DiSC3(5). The probe partitions in the membrane, and its fluorescence is quenched by an electrical potential difference across the membrane (20). The results for the membrane potential parallel those obtained for the transmembrane pH gradient (Fig. 3). When citrate was added to cells grown at pH 6.5, no change in the membrane potential was observed, while addition to cells grown at pH 4.5 resulted in a small but significant increase in membrane potential. When glucose was first added to the cells, a high membrane potential was generated, which was further increased by the addition of citrate. Again, the increase in membrane potential was the highest in cells grown at pH 4.5.
FIG. 3.
Electrical membrane potential during citrate metabolism by resting cells of L. lactis CRL264. Cells grown in a pH-controlled fermentor at pH 6.5 (A, B) and pH 4.5 (A, C) were incubated in 50 mM potassium phosphate (pH 5.5). At the zero time point, the fluorescent membrane potential indicator probe DiSC3(5) was added, after which citrate (A) or glucose followed by citrate (B, C) was added at the time points indicated by the arrows.
The difference in the kinetics of the generation of both the pH gradient and membrane potential following the addition of glucose and citrate is of importance. Both gradients increase upon the addition of glucose after a lag time which reflects the time necessary to build up the ATP pool inside the cells. Only when sufficient ATP is available does F1F0-ATPase efficiently pump out protons from the cell. In contrast, upon addition of citrate, the increases in the pH gradient and membrane potential are immediate, indicating that the generation of the gradients is intimately associated with the citrate metabolic pathway.
The experiments show that citrate fermentation in L. lactis CRL264 results in the generation of a PMF consisting of both a pH gradient and a membrane potential. The ability to generate the electrochemical proton gradient correlates with the flux through the metabolic pathway that depends on the growth conditions and the presence or absence of glucose. Importantly, the increased rate of citrate metabolism in the presence of glucose reflects not an energy-dependent step in the pathway but rather a kinetic restriction in the pathway in the absence of glucose.
The role of lactate in citrate metabolism of L. lactis CRL264.
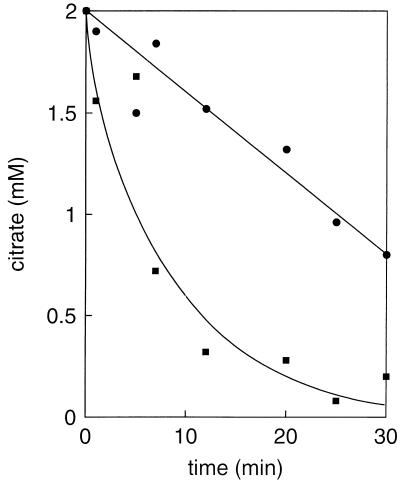
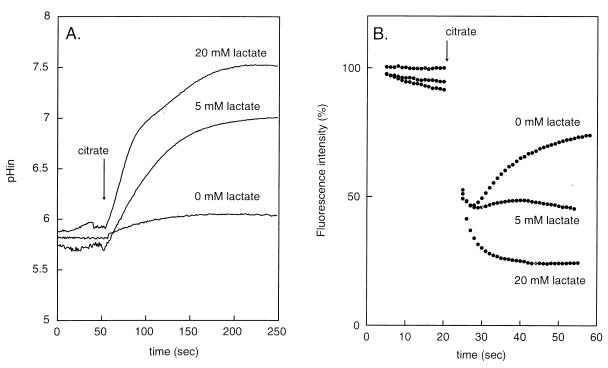
Experiments with membrane vesicles prepared from L. lactis cells capable of citrate fermentation have demonstrated that the citrate transporter CitP catalyzes heterologous citrate-lactate exchange much more efficiently than it catalyzes citrate-proton symport (2). This suggests that lactate produced from glucose played a role in the enhancement of citrate metabolism in the above-described experiments. Indeed, addition of lactate resulted in a stimulation of citrate metabolism by resting cells similar to that observed when glucose was added (Fig. 4). The increased rate of metabolism resulted in the generation of the transmembrane pH gradient and membrane potential: the higher the lactate concentration, the higher the gradients (Fig. 5). At 20 mM lactate, a pH gradient of 2 pH units was generated. The membrane potential induced by citrate in the absence of lactate was transient, but the transient character was absent in the presence of lactate. The pH gradient and membrane potential generated by citrate metabolism in the presence of saturating concentrations of lactate (∼20 mM) were of the same magnitude as the PMF generated by glucose metabolism (data not shown). The observed gradients were strictly dependent on citrate metabolism, since the addition of lactate in the absence of citrate showed no effect.
FIG. 4.
Stimulation of citrate metabolism by lactate in resting cells of L. lactis CRL264. Cells grown in a batch culture at pH 5 were incubated for 10 min at 30°C, after which 2 mM citrate (●) or 2 mM citrate plus 5 mM lactate (■) was added. At the indicated time points, residual citrate was determined.
FIG. 5.
Generation of a pH gradient (A) and membrane potential (B) by citrate metabolism in the presence of lactate. (A) Cells grown in a batch culture at pH 5 were loaded with BCECF and incubated with the indicated concentrations of lactate for 1 min, after which 2 mM citrate was added. (B) Cells were incubated for 1 min with the indicated concentrations of lactate, after which, at the zero time point, the DiSC3(5) probe was added, followed by 2 mM citrate.
The cells used in these experiments were grown in a batch culture at pH 5 without pH control. The distribution of the PMF generated by citrate over the pH gradient and the membrane potential was opposite to what was observed for the cells grown at constant pH (compare Fig. 5A and 2A and Fig. 5B and 3A). Possibly, this reflects different mechanisms of pH homeostasis in the cells imposed by the different growth conditions.
Growth of L. lactis CRL264 at acidic pH in the presence of citrate and lactate.
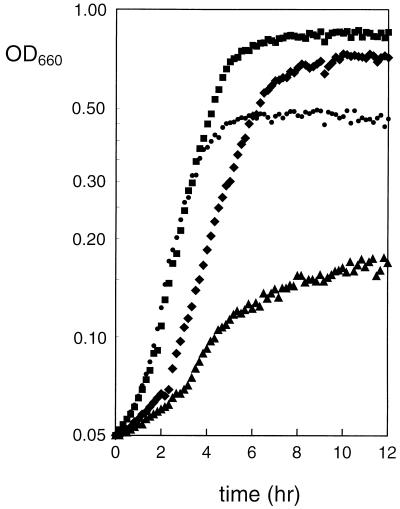
Cometabolism of glucose and citrate by L. lactis species in a batch culture at neutral pH has only a marginal growth advantage over growth on glucose alone. The experiments presented above show that citrate metabolism in cells of L. lactis CRL264 grown at pH 5 can generate a PMF that is at least of the same magnitude as that observed with glucose as the energy source. Therefore, the potential advantage of cometabolism was reinvestigated for growth at acidic pH. In batch cultures in M17 medium, the growth rate of L. lactis CRL264 with glucose at pH 5 was not significantly different in the presence or absence of citrate (Fig. 6, ■ and ●, respectively). However, a prominent difference was observed in the twofold increase in biomass in the presence of citrate. The growth rate and biomass production were severely inhibited when 60 mM lactate was included in the growth medium (Fig. 6, ▴). Remarkably, the inhibition was almost completely reversed when citrate was added in addition to lactate (Fig. 6, ⧫). After a lag time, the rate of growth was almost as fast as that observed in the absence of lactate and the amount of biomass produced at the end of growth was equally high.
FIG. 6.
Growth of L. lactis CRL264 at pH 5 in the presence of citrate and lactate. Cells were grown in M17 medium containing 0.5% glucose and the following: no addition, (●), 10 mM citrate (■), 60 mM lactate (▴), or 10 mM citrate plus 60 mM lactate (⧫). Cells were grown in a total volume of 250 μl in a microtiter plate. The OD660 was plotted on a logarithmic scale.
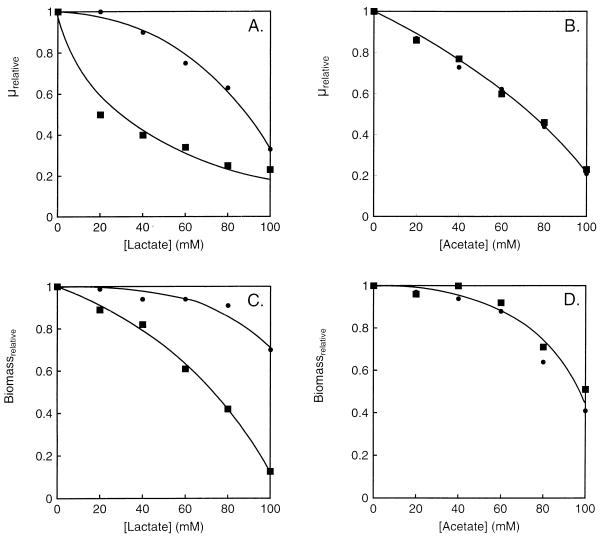
Uncoupling of the transmembrane pH gradient by organic weak acids which in their protonated form are membrane permeable is generally considered to be a mechanism by which end products like lactate, acetate, and formate inhibit bacterial growth. To gain insight into the inhibition of growth by lactate and relief of the inhibition by citrate, growth of L. lactis CRL264 was studied in more detail at acidic pH in the presence of two different weak acids, lactate and acetate. At pH 5, 5.6 and 36% of lactic acid and acetic acid are in the protonated, membrane-permeable form, respectively. In spite of the smaller fraction of the protonated species, titration of the growth rate and biomass production showed that lactate, especially at the lower concentrations, was more inhibitory (Fig. 7). The difference may be explained by different permeabilities of the protonated species of the two molecules or by specific inhibitory effects. At any rate, the presence of citrate in the medium, in addition to the inhibitory weak acid, relieved the inhibition imposed by lactate but not that imposed by acetate. This demonstrates that the inhibition is not caused by some global effect imposed on the cells by any weak acid.
FIG. 7.
Inhibition of growth of L. lactis CRL264 by lactate and acetate. Cells were grown in M17 plus 0.5% glucose (■) or 0.5% glucose plus citrate (●) and in the presence of increasing concentrations of lactate (A, C) or acetate (B, D). The growth rate (μ) and absorbance of the cultures in the stationary phase (Biomass) in the absence of the weak acids were set to 1.
DISCUSSION
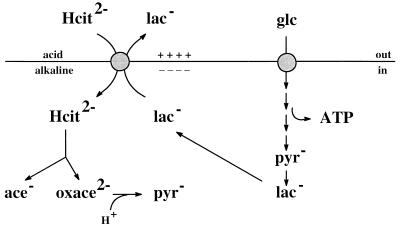
Citrate metabolism by the LAB L. mesenteroides is a secondary PMF-generating pathway (9, 12). The transporter that is responsible for the uptake of citrate from the medium is termed CitP. In a previous study, we showed that the nucleotide sequences of the genes coding for the CitPs of L. mesenteroides and L. lactis are almost identical, and in vitro functional studies of the two transporters revealed no differences (2). Since the properties of the transporter are crucial to the mechanism of PMF generation, this strongly suggested that citrate metabolism in L. lactis is likely to generate metabolic energy in a way similar to that observed in L. mesenteroides. The present data confirm this suggestion. Thus, citrate metabolism resulted in the generation of both a pH gradient and a membrane potential that together form a PMF of physiological polarity. The rate of metabolism of citrate and the efficiency of PMF generation increased dramatically during cometabolism with glucose, a phenomenon that could be mimicked by the addition of lactate, the end product of glucose metabolism. The role of lactate is evident from the metabolic scheme depicted in Fig. 8, which shows the mechanism of PMF generation by glucose and citrate cometabolism in L. lactis under physiological conditions. CitP catalyzes heterologous exchange between divalent citrate and monovalent lactate, which results in a membrane potential, inside negative. Lactate is generated inside the cell by glucose metabolism and is exported by CitP. The transmembrane pH gradient is generated by scalar proton consumption in the oxaloacetate decarboxylation step yielding pyruvate. In the absence of glucose, i.e., when no lactate is produced, CitP functions as a proton-citrate symporter, transporting divalent citrate and a single proton which also is membrane potential generating (11). This mode of transport is much less efficient than citrate-lactate exchange and results in lower rates of citrate metabolism and, consequently, less effective PMF generation (Fig. 1, 2A, and 3A). Addition of lactate to the outside of the cells induces a lactate shuttle by which lactic acid enters the cells passively and the lactate anion is exported by CitP, allowing the latter to operate in the faster exchange mode (12). The result is increased flux through the citrate metabolic pathway and a higher pH gradient and membrane potential. Under physiological conditions, lactate is generated inside the cell and its outward-directed gradient contributes to the force that drives the transport reaction catalyzed by CitP.
FIG. 8.
Schematic representation of the mechanism of citrate fermentation and lactate extrusion in L. lactis CRL264. Citrate metabolism and glucose metabolism are shown on the left and right, respectively. Electrogenic exchange of citrate and lactate results in the generation of a membrane potential, while proton consumption in the oxaloacetate decarboxylation step results in alkalinization of the cytoplasm and the generation of a pH gradient across the membrane. Lactate produced from glucose is pumped out of the cell by the citrate transporter.
The simplest secondary PMF-generating pathways consist of only two enzymes, a transporter and a decarboxylase. Well-known examples are the malolactic fermentation pathway in LAB (15) and oxalate decarboxylation in Oxalobacter formigenes (1). The transporters catalyze exchange of the substrate and the decarboxylation product, i.e., malate-lactate exchange and oxalate-formate exchange, respectively, and are therefore termed precursor-product exchangers. One case has been reported in which the transporter functions as an electrogenic uniporter and the decarboxylation product leaves the cell passively (17). These simple pathways operate autonomously in the cell without the need for intermediates of other pathways. The citrate metabolic pathways of LAB are the only secondary PMF-generating pathways described to date that are more complicated, not only because of the involvement of more cytoplasmic enzymes but, in particular, because of their interaction with glycolysis. This interaction is needed because citrate uptake via the citrate transporter occurs in exchange with lactate, which is not a direct product of citrate metabolism. The interaction between the two pathways differs slightly in L. lactis and L. mesenteroides because of differences in the carbohydrate fermentation pathways in these two organisms. In L. mesenteroides, cometabolism of citrate and glucose results in a metabolic shift in the heterofermentative pathway for glucose breakdown in which redox equivalents are shuttled to pyruvate produced from citrate, yielding lactate (4, 7). In other words, lactate is a product of citrate metabolism when glycolysis provides the necessary redox equivalents. Therefore, in L. mesenteroides, the citrate transporter CitP is a precursor-product exchanger and the process is termed citrolactic fermentation (12). In the homofermentative bacterium L. lactis, lactate is the product of glycolysis and not of citrate metabolism, even though the product of the latter feeds into the common pyruvate pool. In L. lactis, the terms precursor-product exchanger and citrolactic fermentation seem less appropriate for CitP and citrate metabolism, respectively. Summarizing, the coupling between the citrate metabolic pathway and glycolysis provides the necessary lactate to allow CitP to function in the citrate-lactate exchange mode. In L. mesenteroides, the coupling is at the level of the redox state of the cell, and in L. lactis, it is at the level of the end product of glycolysis.
An important physiological difference between the citrate fermentation pathways of L. lactis and L. mesenteroides is the regulation of expression of the pathways. In this paper, we demonstrate that the pathway in L. lactis is constitutively expressed at neutral pH values of the growth medium. At lower pH values, an increased activity is observed, which is in agreement with the increased expression of the citrate transporter at low pH reported before (5). The presence of citrate in the growth medium did not seem to induce the pathway. Likewise, in L. mesenteroides, constitutive expression of the citrate transporter has been reported (13). However, in this bacterium, increased expression of the transporter was observed when citrate was added to the growth medium. The effect of acidity on the expression in L. mesenteroides has not been investigated. Therefore, in both organisms, a low level of constitutive expression is observed, but in L. lactis, enhanced expression is induced by low pH, and in L. mesenteroides, it is induced by citrate. The different regulation of expression is likely to reflect different physiological functions of citrate metabolism in the two bacteria.
The mechanism of citrate metabolism in LAB has a number of physiological consequences that may be of importance, depending on the organisms and their habitat. Citrate metabolism contributes to the energy status of the cell by generating a PMF, it contributes to pH homeostasis because of the alkalinization of the cytoplasm, and eventually, it will alkalinize the external medium, which may allow the bacterium to recover from acid stress. Furthermore, intermediates of the pathway have been shown to be precursors of amino acid synthesis (13). In our search for the effects of citrate metabolism on the growth parameters of L. lactis, we discovered another benefit of citrate metabolism that may be of specific importance to L. lactis strains, the relief of lactate toxicity. During growth of L. lactis in a batch culture, the fermentation of sugars results in accumulation of lactic acid and acidification of the medium. Both conditions inhibit growth, and the inhibitory effects are synergistic when present at the same time. Because of the synergism, i.e., the inhibition by lactate, and in general by weak organic acids, increases with decreasing pH (e.g., see reference 6), the inhibitory effect is usually ascribed to the undissociated acid that is membrane permeable and may compromise pH homeostasis of the cytoplasm. Other, specific inhibitory effects of the dissociated acids have also been suggested (19). The growth studies presented in Fig. 7 demonstrate that citrate metabolism specifically relieves the inhibition imposed by lactate but not the inhibition by acetic acid. This suggests that the inhibitory effect of both weak acids is not caused by a negative effect on pH homeostasis, since in that case, the alkalinizing effect of citrate metabolism should relieve the inhibition caused by any weak acid. Mechanistically, the beneficial mode of action of citrate is readily evident from the scheme in Fig. 8, which shows that citrate metabolism actively and specifically removes lactate from the cytoplasm. Since lactate is especially inhibitory at low pH, resistance to lactate toxicity provides a rationale for the induction of the citrate fermentation pathway at low pH.
ACKNOWLEDGMENTS
This work was supported by grants from Fundación Antorchas, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), and Agencia Nacional de Promoción Científica y Tecnológica (FONCYT). C. Magni and D. de Mendoza are Career Investigators from CONICET.
REFERENCES
- 1.Anantharam V, Allison M J, Maloney P C. Oxalate:formate exchange: the basis for energy coupling in Oxalobacter. J Biol Chem. 1989;264:7244–7250. [PubMed] [Google Scholar]
- 2.Bandell M, Lhotte M E, Marty-Teysset C, Veyrat A, Prévost H, Dartois V, Diviès C, Konings W N, Lolkema J S. Mechanism of the citrate transporters in carbohydrate and citrate cometabolism in Lactococcus and Leuconostoc species. Appl Environ Microbiol. 1998;64:1594–1600. doi: 10.1128/aem.64.5.1594-1600.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bekal S, Van Beeumen J, Samyn B, Garmyn D, Henini S, Diviès C, Prévost H. Purification of Leuconostoc mesenteroides citrate lyase and cloning and characterization of the citCDEFG gene cluster. J Bacteriol. 1998;180:647–654. doi: 10.1128/jb.180.3.647-654.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cogan T M. Co-metabolism of citrate and glucose by Leuconostoc ssp.: effects on growth, substrates and products. J Appl Bacteriol. 1987;63:551–558. [Google Scholar]
- 5.Garcí-Quintáns N, Magni C, de Mendoza D, López P. The citrate transport system of Lactococcus lactis subsp. lactis biovar diacetylactis is induced by acid stress. Appl Environ Microbiol. 1998;64:850–857. doi: 10.1128/aem.64.3.850-857.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Houtsma P C, Kusters B J M, de Wit J C, Rombouts F M, Zwietering M H. Modelling growth rates of Listeria innocua as a function of lactate concentration. J Food Microbiol. 1994;24:113–123. doi: 10.1016/0168-1605(94)90111-2. [DOI] [PubMed] [Google Scholar]
- 7.Hugenholtz J. Citrate metabolism in lactic acid bacteria. FEMS Microbiol Rev. 1993;12:165–178. [Google Scholar]
- 8.Lolkema J S, Poolman B, Konings W N. Role of scalar protons in metabolic energy generation in lactic acid bacteria. J Bioenerg Biomembr. 1995;27:467–473. doi: 10.1007/BF02110009. [DOI] [PubMed] [Google Scholar]
- 9.Lolkema J S, Poolman B, Konings W N. Secondary transporters and metabolic energy generation. In: Konings W N, Kaback H R, Lolkema J S, editors. Handbook of biological physics. Amsterdam, The Netherlands: Elsevier; 1996. pp. 229–260. [Google Scholar]
- 10.Magni C, Lopez-de Felipe F, Sesma F, Lopez P, de Mendoza D. Citrate transport in Lactococcus lactis biovar diacetylactis: expression of the plasmid borne citrate permease P. FEMS Microbiol Lett. 1994;118:75–82. [Google Scholar]
- 11.Marty-Teysset C, Lolkema J S, Schmitt P, Divies C, Konings W N. Membrane-potential-generating transport of citrate and malate by CitP of Leuconostoc mesenteroides. J Biol Chem. 1995;270:25370–25376. doi: 10.1074/jbc.270.43.25370. [DOI] [PubMed] [Google Scholar]
- 12.Marty-Teysset C, Posthuma C, Lolkema J S, Schmitt P, Divies C, Konings W N. Proton motive force generation by citrolactic fermentation in Leuconostoc mesenteroides. J Bacteriol. 1996;178:2175–2185. doi: 10.1128/jb.178.8.2178-2185.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marty-Teysset C, Lolkema J S, Schmitt P, Diviès C, Konings W N. The citrate metabolic pathway in Leuconostoc mesenteroides: expression, amino acid synthesis, and α-ketocarboxylate transport. J Bacteriol. 1996;178:6209–6215. doi: 10.1128/jb.178.21.6209-6215.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Molenaar D, Abee T, Konings W N. Continuous measurement of the cytoplasmic pH in Lactococcus lactis with a fluorescence pH indicator. Biochim Biophys Acta. 1991;1115:75–83. doi: 10.1016/0304-4165(91)90014-8. [DOI] [PubMed] [Google Scholar]
- 15.Poolman B, Molenaar D, Smid E J, Ubbink T, Abee T, Renault P P, Konings W N. Malolactic fermentation: electrogenic malate uptake and malate/lactate antiport generate metabolic energy. J Bacteriol. 1991;173:6030–6037. doi: 10.1128/jb.173.19.6030-6037.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ramos A, Poolman B, Santos H, Lolkema J S, Konings W N. Uniport of anionic citrate and proton consumption in citrate metabolism generates a proton motive force in Leuconostoc oenos. J Bacteriol. 1994;176:4899–4905. doi: 10.1128/jb.176.16.4899-4905.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Salema M, Poolman B, Lolkema J S, Loureiro Dias M C, Konings W N. Uniport of monoanionic l-malate in membrane vesicles from Leuconostoc oenos. Eur J Biochem. 1994;225:289–295. doi: 10.1111/j.1432-1033.1994.00289.x. [DOI] [PubMed] [Google Scholar]
- 18.Sesma F, Gardiol D, de Ruiz Holgado A P, de Mendoza D. Cloning and expression of the citrate permease gene of Lactococcus lactis subsp. lactis biovar diacetylactis in Escherichia coli. Appl Environ Microbiol. 1990;56:2099–2103. doi: 10.1128/aem.56.7.2099-2103.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shelef L A. Antimicrobial effect of lactates: a review. J Food Microbiol. 1994;57:445–450. doi: 10.4315/0362-028X-57.5.445. [DOI] [PubMed] [Google Scholar]
- 20.Sims P J, Waggoner A S, Wang C-H, Hoffman J F. Studies on the mechanism by which cyanine dyes measure membrane potential in red blood cells and phosphatidylcholine vesicles. Biochemistry. 1974;13:3315–3330. doi: 10.1021/bi00713a022. [DOI] [PubMed] [Google Scholar]