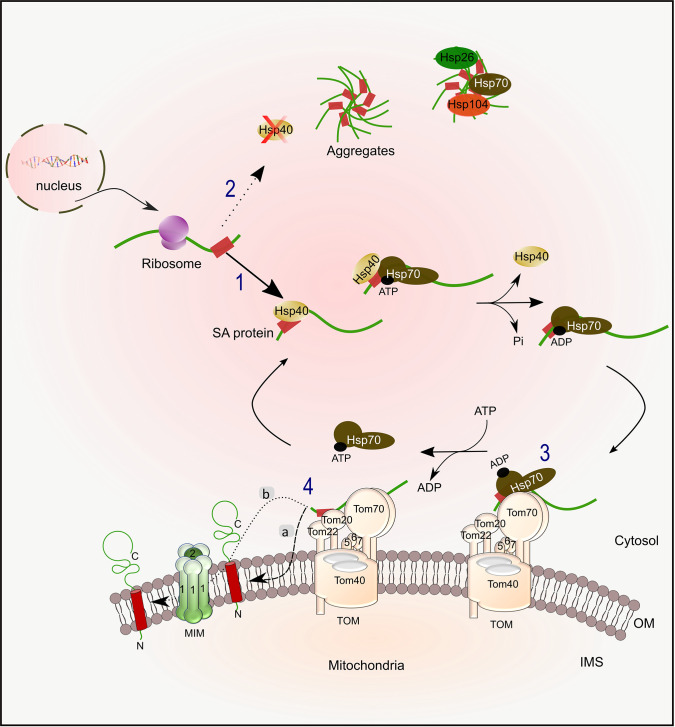
Figure 10. Working model for the biogenesis of SA proteins.
After SA proteins get synthesized on cytosolic ribosomes, they can associate with Hsp40 chaperones (like Ydj1 and Sis1)(1). Upon depletion of Hsp40 chaperones, newly synthesized SA proteins might tend to form aggregates, which can then associate with disaggregases chaperones such as Hsp104 and Hsp26 (2). Hsp40 chaperones drive the transfer of the newly synthesized protein to Hsp70 chaperone (Ssa1/2) by facilitating the conversion of Hsp70 from its ATP form to the ADP one that has a higher affinity for polypeptides. Next, the protein-chaperone complex is recognized by Tom70 receptor (3), followed by disassociation of the chaperone. Subsequently to the recognition by Tom70, which may involve also Tom20, the substrate is then inserted into the OM, either through an unassisted route (4 a), or via a pathway which is facilitated by the MIM complex (4b).