Abstract
AIM
To assess the differences in average and sectoral peripapillary retinal nerve fiber layer (pRNFL) thickness using spectral domain optical coherence tomography (SD-OCT) in patients with non-arteritic anterior ischemic neuropathy (NAION) compared with those with primary open angle glaucoma (POAG).
METHODS
A comprehensive literature search of the PubMed, Cochrane Library, and Embase databases were performed prior to October, 2021. Studies that compared the pRNFL thickness in NAION eyes with that in POAG eyes with matched mean deviation of the visual fields were included. The weighted mean difference (WMD) with 95% confidence interval (CI) was used to pool continuous outcomes.
RESULTS
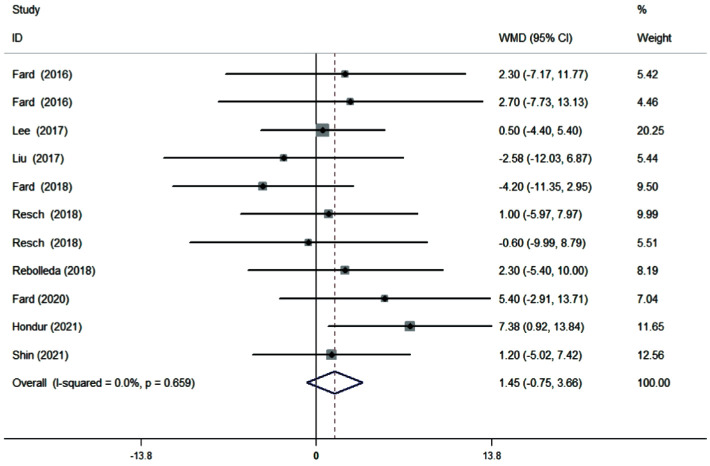
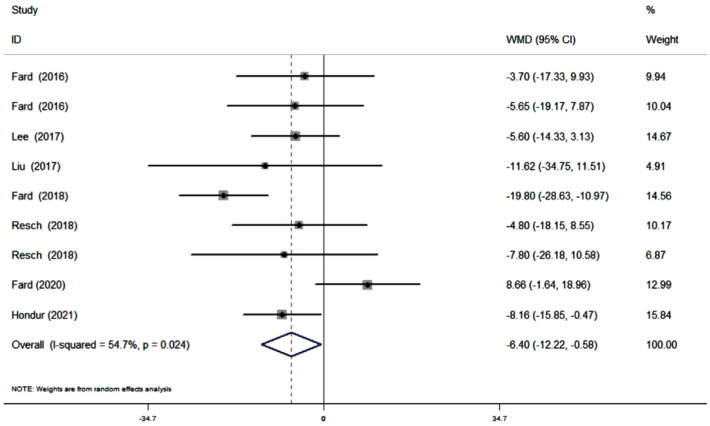
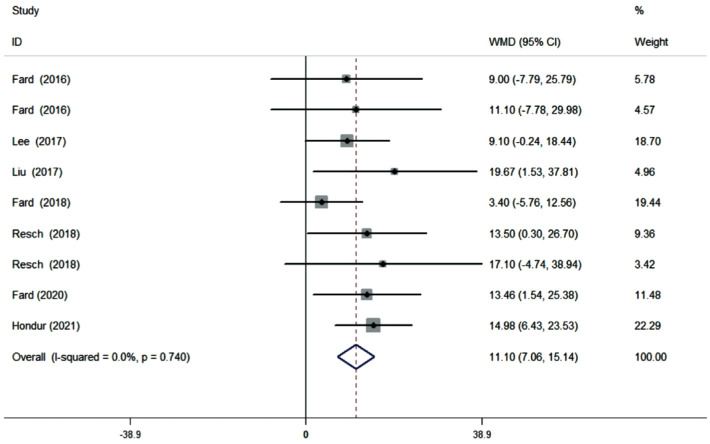
Ten cross-sectional studies (11 datasets) comprising a total of 625 eyes (278 NAION eyes, 347 POAG eyes) were included in the qualitative and quantitative analyses. The pooled results demonstrated that the superior pRNFL was significantly thinner in NAION eyes than in POAG eyes (WMD=-6.40, 95%CI: -12.22 to -0.58, P=0.031), whereas the inferior pRNFL was significant thinner in POAG eyes than in NAION eyes (WMD=11.10, 95%CI: 7.06 to 15.14, P≤0.001). No difference was noted concerning the average, nasal, and temporal pRNFL thickness (average: WMD=1.45, 95%CI: -0.75 to 3.66, P=0.196; nasal: WMD=-2.12, 95%CI: -4.43 to 0.19, P=0.072; temporal: WMD=-1.24, 95%CI: -3.96 to 1.47, P=0.370).
CONCLUSION
SD-OCT based evaluation of inferior and superior pRNFL thickness can be potentially utilized to differentiate NAION from POAG, and help to understand the different pathophysiological mechanisms between these two diseases. Further longitudinal studies and studies using eight-quadrant or clock-hour classification method are required to validate the obtained findings.
Keywords: non-arteritic anterior ischemic optic neuropathy, primary open angle glaucoma, optical coherence tomography, peripapillary retinal nerve fiber layer thickness
INTRODUCTION
Primary open angle glaucoma (POAG) is an age-related neurodegenerative optic neuropathy characterized by the progressive deterioration of retinal ganglion cells (RGCs) and their axons[1]–[3], followed by the excavation of the optic nerve head (ONH) and impaired visual field (VF)[4]–[5]. Non-arteritic anterior ischemic optic neuropathy (NAION) is a non-glaucomatous optic neuropathy that presents with the sudden painless loss of vision, optic disc edema with resolution after several weeks and optic disc pallor at the atrophic stage[6]–[7]. Similar to POAG, NAION also results in the loss of RGCs and their axons. However, the pathophysiological mechanisms underlying the different ONH configuration changes in these two diseases are not completely understood[3],[7].
Evaluation of the peripapillary retinal nerve fiber layer (pRNFL) thickness enables clinicians to assess the degree and the pattern of the damage to the RGC axons coursing toward the ONH. Multiple quantitative retinal imaging techniques comprising scanning laser polarimetry, Heidelberg retinal tomography, as well as optical coherence tomography (OCT)[8]–[13], have been utilized to measure RNFL thickness. Among them, spectral-domain optical coherence tomography (SD-OCT), which is the latest generation of OCT, provides a high-resolution and enhance-depth visualization of the retina and the ONH[14]–[16].
With the utilization of SD-OCT in daily routine diagnosis, several studies have demonstrated that the attenuation of average pRNFL thickness is common in NAION and glaucoma[5],[13],[17]–[33], where NAION may mimic with POAG[34]. However, the pattern and severity of sectoral pRNFL thickness thinning were inconsistent[18]–[20],[22],[24]–[26],[28]–[29], which may help reveal the different underlying mechanisms that induce optic damage in these two ophthalmic neuropathies and help differentiate NAION from POAG in a non-invasive manner.
Therefore, we performed this systematic review and Meta-analysis to compare the average and sectoral pRNFL thickness in patients with NAION and those with POAG with similar VF mean deviation (MD), facilitating a better understanding of the biomechanisms that lead to the different patterns of neurodegeneration.
MATERIALS AND METHODS
The present systematic review and Meta-analysis were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) Statement and the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines[35]–[36]. Three individual investigators (Tong YX, Zhang XY, and He Y) independently conducted the literature search, qualification, data extraction, quality evaluation by Agency for Healthcare Research and Quality (AHQR) checklist, and risk of bias assessment. The present study was registered in PROSPERO (registration number: CRD42020220934).
Search Strategy
Literature search was conducted from inception to October, 2021 in PubMed, Cochrane Library and Embase databases. The following search terms were used: “non-arteritic anterior ischemic optic neuropathy”, “NAION”, “glaucoma”, “retinal nerve fiber layer”, “RNFL”, “optical coherence tomography”, “OCT” with no restriction. The search strategies were modified by the requirements of the different databases. Potential eligible articles were included by detailed screening of full-text versions of the literature.
Inclusion and Exclusion Criteria
We included cross-sectional studies if they met the following requirements: 1) original article; 2) inclusion of NAION at the atrophic stage where optic disc swelling had to occur at least 3mo prior to the documentation and has resolved at the time of the study; 3) inclusion of both NAION and POAG with the same diagnostic standards; 4) inclusion of NAION and POAG with similar severities in terms of the MD of the VF; 5) inclusion of pRNFL thickness assessed by SD-OCT.
Exclusion criteria were: 1) conference abstracts, reviews, case reports and animal experiments; 2) enrollment of NAION at the acute stage; 3) different diagnostic standards; 4) non-inclusion of SD-OCT based evaluation of pRNFL thickness; 5) studies without extractable data.
Data Extraction
The following information was extracted and summarized: title, first author, publication year, region, study type, number of patients and eyes, source of patients, time periods for identifying patients, mean age of patients, female/male ratios, types of OCT devices, episode of NAION, types of glaucoma, diagnostic criteria, average and quadrant pRNFL thickness, scan protocol and area of ONH, MD of the VF, severity of glaucoma, onset time of NAION. Disagreements were resolved by discussing among all the authors.
Quality Assessment
Ten included cross-sectional studies were evaluated based on the AHRQ methodology checklist.
Statistical Analysis
Stata version 12.0 (StataCorp, Texas, USA) and Review Manager version 5.4.1 (Cochrane Collaboration, London, UK) were used for the statistical analyses. We used weighted mean difference (WMD) with a 95% confidence interval (CI) to pool the mean differences in average and sectoral pRNFL thickness between the NAION and POAG groups. A P value <0.05 was regarded to be statistically significant. Statistical heterogeneities among different groups were measured using Cochrane's Q test and quantified by I2. We used a fixed-effects model when I2<50%[37], indicating the heterogeneity was acceptable; otherwise, we employed a random-effects model when I2>50%. The stratified analyses were performed by the onset time of NAION and OCT device types. Egger et al's[38] and Begg et al's[39] tests were used to evaluate the potential publication bias. The “leave-one-out” sensitivity analysis concerning the average and quadrant pRNFL thickness was performed to explore the sources of heterogeneity.
In some studies, RNFL thickness was displayed by six-quadrant classification method, and in others the four-quadrants classification method was used. To transform the six-quadrant data to four-quadrant data (since the majority of the articles used four-quadrant classification method), we used a modified method as previously described[40].
RESULTS
Literature Search
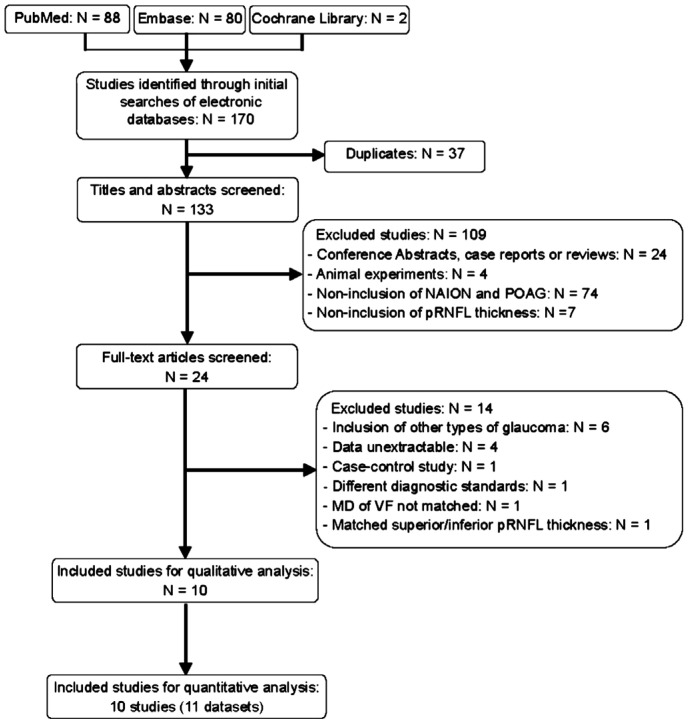
A total of 170 studies were initially identified, of which 37 duplicates were removed. Of the remaining 133 articles, 109 were excluded after screening the titles and abstract: 74 did not include NAION and POAG patients; 24 were conference abstracts, case reports, or reviews; seven lacked SD-OCT-based pRNFL thickness data; and four performed animal experiments. With full-text screening, another 14 studies were removed: six included other types of glaucoma rather than POAG, four reported unextractable data, one was a case-control study, one used different diagnostic standards, one included NAION and POAG with un-matched MD of VF, and one included NAION and POAG with matched superior or inferior pRNFL thickness. Thus, 10 eligible articles were included in the qualitative analysis, and 10 studies (11 datasets) were integrated in the quantitative analysis. A flow diagram of the literature search is shown in Figure 1.
Figure 1. Flow diagram of studies identified, included and excluded.
Characteristics and Qualities of Included Studies
Ten included cross-sectional studies (11 datasets) comprising 625 eyes (278 NAION eyes, 347 POAG eyes) were published between 2016 and 2021 across different regions (Turkey, Iran, Spain, Austria, Korea, and Taiwan, China). The mean age varied from 54.1 to 68.6y in NAION patients and 53.75 to 72.3y in POAG patients. The pRNFL thickness was detected by using three types of SD-OCT devices: Spectralis, Cirrus, and Optovue. Most of the scan protocols were centered at the 3.4-3.5 mm circle around the ONH. Moreover, the MD of the VF was comparable between NAION and POAG eyes, and the majority of the included studies enrolled patients with moderate to severe POAG patients. The included studies recruited NAION patients with time from the onset of more than 3 or 6mo. The detailed characteristics are summarized in Table 1.
Table 1. Characteristics of included studies.
| Study Year | Region | Design | No. eye |
Mean±SD age (y) |
Gender (F/M) |
Device (SD-OCT) | Scan area of ONH (mm2) | MD of the VF (dB) |
Severity of glaucoma | Onset time of NAION | ||||
| NAION | POAG | NAION | POAG | NAION | POAG | NAION | POAG | |||||||
| Shin 2021[17] | Korea | CS, R | 27 | 27 | 65.2±7.8 | 61.7±11.1 | 11/16 | 17/10 | Cirrus | 3.45 mm circle | -15.13±8.34 | -15.11±7.57 | Not indicated | >6mo |
| Hondur 2021[18] | Turkey | CS | 21 | 26 | 61±7 | 62±11 | 9/12 | 13/13 | Spectralis | 4.5×4.5 | -18.79±6.13 | -20.99±6.36 | Severe | >6mo |
| Fard 2020[19] | Iran | CS, P | 19 | 37 | 57.4±12.2 | 63.6±0.4 | N/A | N/A | Optovue | 3.4 mm circle | -14.0±5.3 | -12.7±5.5 | Moderate to severe | >6mo |
| Robolleda 2019[20] | Spain | CS | 23 | 25 | 68.6±10.3 | 72.3±9.8 | 12/11 | 11/14 | Spectralis | 3.5 mm circle | -14.7±8.6 | -12.9±8.32 | Not indicated | >6mo |
| Resch 2018[22] | Austria | CS | 20 | 20 | 66.8±8.3 | 71.2±6.0 | 8/12 | 11/9 | Spectralis & Cirrus | 3.4 mm circle | -9.85±4.79 | -10.24±4.71 | Moderate to severe | >6mo |
| Fard 2018[24] | Iran | CS | 31 | 42 | 54.1±11 | 60.2±8 | 15/16 | 16/26 | Spectralis | 3.45 mm circle | -18.4±8.6 | -17.2±8.6 | Moderate to severe | >6mo |
| Liu 2017[25] | Taiwan, China | CS, R | 10 | 16 | 59.9±10.7 | 53.75±10.21 | 6/4 | 7/9 | Optovue | 3.45 mm circle | -12.78±6.34 | -9.69±3.56 | Moderate | >3mo |
| Lee 2017[26] | Korea | CS, R | 35 | 70 | 63.6±8.6 | 64.4±9.0 | 20/15 | 40/30 | Cirrus | Optic disc cube 200×200 | -17.7±8.7 | -13.5±7.5 | Moderate to severe | >3mo |
| Fard 2016[29] | Iran | CS | 30 | 32 | 58.4±10.5 | 65.6±13.1 | 17/13 | 10/22 | Spectralis | 3.4 mm circle | -16.47±9.05 | -16.28±9.98 | Moderate to severe | >3mo |
| Fard 2016[28] | Iran | CS | 42 | 32 | 58.02±8.83 | 64.59±9.79 | 22/20 | 10/22 | Spectralis | 3.4 mm circle | -16.47±9.05 | -16.28±9.98 | Moderate to severe | >3mo |
F/M: Female/male; SD-OCT: Spectral-domain optical coherence tomography; ONH: Optic nerve head; MD: Mean deviation; VF: Visual field; NAION: Non-arteritic anterior ischemic optic neuropathy; POAG: Primary open angle glaucoma; CS: Cross-sectional study; P: Prospective study; R: Retrospective study.
In terms of evaluating methodological quality, the AHRQ scores of all included studies were more than 3, indicating adequate quality (Table 2).
Table 2. Methodological quality of included studies.
| Study | 11-item check list recommended by AHRQ checklist |
||||||||||||
| i | ii | iii | iv | v | vi | vii | viii | ix | x | xi | Score | Quality | |
| Shin 2021[17] | ★ | ★ | ★ | ★ | ★ | ★ | 6 | M | |||||
| Hondur 2021[18] | ★ | ★ | ★ | ★ | 4 | M | |||||||
| Fard 2020[19] | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 7 | M | ||||
| Robolleda 2019[20] | ★ | ★ | ★ | ★ | ★ | 5 | M | ||||||
| Resch 2018[22] | ★ | ★ | ★ | ★ | ★ | 5 | M | ||||||
| Fard 2018[24] | ★ | ★ | ★ | ★ | ★ | ★ | 6 | M | |||||
| Liu 2017[25] | ★ | ★ | ★ | ★ | 4 | M | |||||||
| Lee 2017[26] | ★ | ★ | ★ | ★ | ★ | 5 | M | ||||||
| Fard 2016[29] | ★ | ★ | ★ | ★ | ★ | ★ | ★ | 7 | M | ||||
| Fard 2016[28] | ★ | ★ | ★ | ★ | ★ | ★ | 6 | M | |||||
AHRQ: Agency for Healthcare Research and Quality; H: High quality; M: Moderate quality; L: Low quality; high quality (score: 8-11); moderate quality (score: 4-7); low quality (score: 0-3). i: Define the source of information; ii: List inclusion and exclusion criteria for exposed and unexposed subjects (cases and controls) or refer to previous publications; iii: Indicate time period used for identifying patients; iv: Indicate whether or not subjects were consecutive if not population-based; v: Indicate if evaluators of subjective components of study were masked to other aspects of the status of the participants; vi: Describe any assessments undertaken for quality assurance purposes; vii: Explain any patient exclusions from analysis; viii: Describe how confounding was assessed and/or controlled; ix: If applicable, explain how missing data were handled in the analysis; x: Summarize patient response rates and completeness of data collection; xi: Clarify what follow-up, if any, was expected and the percentage of patients for which incomplete data or follow-up was obtained.
RNFL Thickness in NAION and POAG Patients
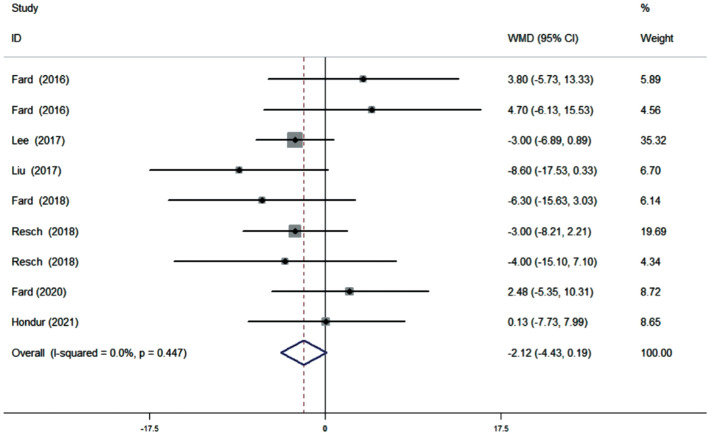
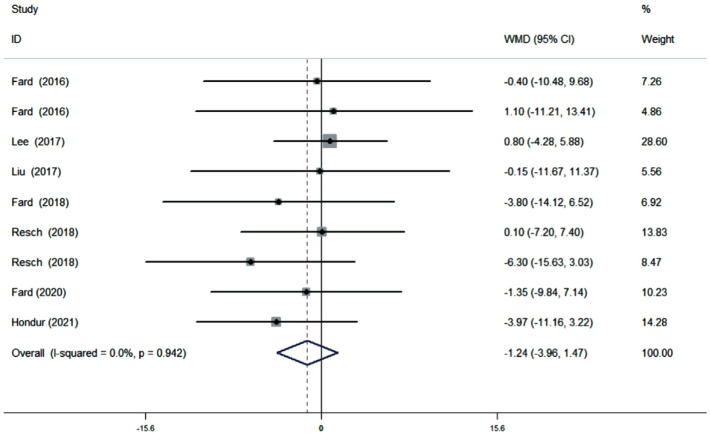
Ten studies (11 datasets) assessing the average pRNFL thickness showed no heterogeneity (I2=0). The pooled results demonstrated that no difference in average pRNFL thickness between NAION and POAG eyes (WMD=1.45, 95%CI: -0.75 to 3.66, P=0.196; Figure 2). However, eight studies (nine datasets) evaluating the sectoral pRNFL thickness demonstrated that the superior pRNFL thickness was significantly lower in NAION patients than in POAG patients (WMD=-6.40, 95%CI: -12.22 to -0.58, P=0.031; Figure 3), whereas the inferior pRNFL was significantly thinner in POAG eyes (WMD=11.10, 95%CI: 7.06 to 15.14, P≤0.001; Figure 4). No difference in the nasal and temporal quadrants was found between NAION and POAG patients (nasal: WMD=-2.12, 95%CI: -4.43 to 0.19, P=0.072; Figure 5; temporal: WMD=-1.24, 95%CI: -3.96 to 1.47, P=0.370; Figure 6).
Figure 2. Forrest plot of average pRNFL thickness in patients with NAION and POAG.
Figure 3. Forrest plot of superior pRNFL thickness in patients with NAION and POAG.
Figure 4. Forrest plot of inferior pRNFL thickness in patients with NAION and POAG.
Figure 5. Forrest plot of nasal pRNFL thickness in patients with NAION and POAG.
Figure 6. Forrest plot of temporal pRNFL thickness in patients with NAION and POAG.
Subgroup Analysis
The subgroup analysis regarding the onset time of NAION (Table 3) also demonstrated that the inferior pRNFL thickness was significantly lower in POAG eyes than in NAION eyes (onset time >3mo: WMD=10.89, 95%CI: 3.97 to 17.82, P=0.002; onset time >6mo: WMD=11.20, 95%CI: 6.23 to 16.18, P≤0.001). However, in contrast to the combined pooled data, significant difference in the superior pRNFL thickness was not found between the NAION eyes and POAG eyes regardless of the onset time of NAION (onset time >3mo: WMD=-5.65, 95%CI: -11.87 to 0.57, P=0.075; onset time >6mo: WMD=-6.55, 95%CI: -16.58 to 3.48, P=0.201). Similarly, no difference in the average, nasal, and temporal pRNFL thicknesses was noted.
Table 3. Subgroup analysis of pRNFL thickness according to the onset time of NAION.
| Subgroup | No. | Heterogeneity |
WMD (95%CI) | Overall effect |
||
| I 2 | P | Z | P | |||
| Onset time >3mo | ||||||
| Average | 4 | 0 | 0.869 | 0.58 (-3.12, 4.28) | 0.31 | 0.758 |
| Superior | 4 | 0 | 0.953 | -5.65 (-11.87, 0.57) | 1.78 | 0.075 |
| Inferior | 4 | 0 | 0.779 | 10.89 (3.97, 17.82) | 3.08 | 0.002 |
| Nasal | 4 | 42.5% | 0.157 | -2.28 (-5.47, 0.91) | 1.40 | 0.161 |
| Temporal | 4 | 0 | 0.996 | 0.53 (-3.46, 4.52) | 0.26 | 0.795 |
| Onset time >6mo | ||||||
| Average | 7 | 9.6% | 0.355 | 1.93 (-0.81, 4.68) | 1.38 | 0.167 |
| Superior | 5 | 76.6% | 0.002 | -6.55 (-16.58, 3.48) | 1.28 | 0.201 |
| Inferior | 5 | 1.7% | 0.397 | 11.20 (6.23, 16.18) | 4.42 | ≤0.001 |
| Nasal | 5 | 0 | 0.623 | -1.94 (-5.30, 1.41) | 1.14 | 0.256 |
| Temporal | 5 | 0 | 0.845 | -2.77 (-6.47, 0.94) | 1.46 | 0.143 |
pRNFL: Peripapillary retinal nerve fiber layer; NAION: Non-arteritic anterior ischemic optic neuropathy; SD-OCT: Spectral-domain optical coherence tomography; WMD: Weighted mean difference; CI: Confidence interval; I2: I-square heterogeneity statistic; Z: Z-statistic.
Stratified analysis according to the different types of SD-OCT (Table 4) also revealed that the inferior pRNFL was significantly thinner in POAG eyes when different SD-OCT devices were utilized (Spectralis: WMD=10.11, 95%CI: 4.69 to 15.53, P≤0.001; Cirrus: WMD=10.57, 95%CI: 2.94 to 18.19, P=0.007; Optovue: WMD=15.33, 95%CI: 5.37 to 25.29, P=0.003). The pooled results showed that the superior pRNFL was significantly thinner in NAION eyes than in POAG eyes when the Spectralis SD-OCT was used (WMD=-10.20, 95%CI: -16.51 to -3.90, P=0.002), whereas no difference was demonstrated when Cirrus and Optovue SD-OCT were used (Cirrus: WMD=-5.36, 95%CI: -12.67 to 1.94, P=0.150; Optovue: WMD=1.27, 95%CI: -17.85 to 20.40, P=0.896). Similarly, no difference in the average, nasal, and temporal pRNFL thickness was indicated between NAION and POAG eyes regardless of SD-OCT type.
Table 4. Subgroup analysis of pRNFL thickness according to the type of SD-OCT.
| Subgroup | No. | Heterogeneity |
WMD (95%CI) | Overall effect |
||
| I2 | P | Z | P | |||
| Spectralis | ||||||
| Average | 6 | 14.9% | 0.319 | 1.92 (-1.37, 5.22) | 1.14 | 0.252 |
| Superior | 5 | 34.9% | 0.189 | -10.20 (-16.51, -3.90) | 3.17 | 0.002 |
| Inferior | 5 | 0 | 0.444 | 10.11 (4.69, 15.53) | 3.66 | ≤0.001 |
| Nasal | 5 | 0 | 0.469 | -0.38 (-4.63, 3.87) | 0.17 | 0.862 |
| Temporal | 5 | 0 | 0.870 | -3.20 (-7.40, 1.00) | 1.50 | 0.135 |
| Cirrus | ||||||
| Average | 3 | 0 | 0.983 | 0.82 (-2.55, 4.19) | 0.48 | 0.632 |
| Superior | 2 | 0 | 0.922 | -5.36 (-12.67, 1.94) | 1.44 | 0.150 |
| Inferior | 2 | 0 | 0.594 | 10.57 (2.94, 18.19) | 2.72 | 0.007 |
| Nasal | 2 | 0 | 1.000 | -3.00 (-6.12, 0.12) | 1.89 | 0.059 |
| Temporal | 2 | 0 | 0.877 | 0.57 (-3.60, 4.74) | 0.27 | 0.788 |
| Optovue | ||||||
| Average | 2 | 35.3% | 0.214 | 1.92 (-4.32, 8.16) | 0.60 | 0.54 6 |
| Superior | 2 | 59.4% | 0.116 | 1.27 (-17.85, 20.40) | 0.13 | 0.896 |
| Inferior | 2 | 0 | 0.575 | 15.33 (5.37, 25.29) | 3.02 | 0.003 |
| Nasal | 2 | 70.1% | 0.067 | -2.84 (-13.69, 8.01) | 0.51 | 0.607 |
| Temporal | 2 | 0 | 0.869 | -0.93 (-7.76, 5.91) | 0.27 | 0.790 |
WMD: Weighted mean difference.
Publication Bias
Results of Begg's test and Egger's test demonstrated no significant risk of publication bias in the average and sectoral pRNFL thickness (P>0.05; Table 5).
Table 5. Begg's test and Egger's test results of average and sectoral pRNFL thickness.
| pRNFL thickness | Begg's test |
Egger's test |
||
| z | P>|z| | t | P>|t| | |
| Average | 0.16 | 0.876 | -0.07 | 0.947 |
| Superior | 0.10 | 0.917 | 0.34 | 0.743 |
| Inferior | 0.52 | 0.602 | 1.06 | 0.323 |
| Nasal | 0.94 | 0.348 | 0.69 | 0.514 |
| Temporal | -0.10 | 1.000 | -0.89 | 0.404 |
pRNFL: Peripapillary retinal nerve fiber layer.
Sensitivity Analysis
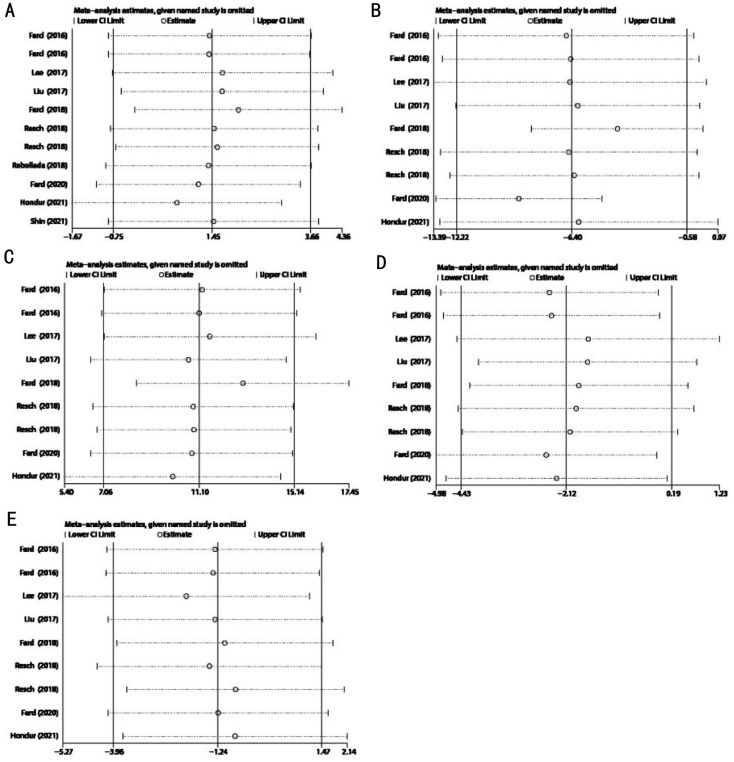
No obvious change in the results was noted after excluding each study (Figure 7), demonstrating the stability and reliability of our results. Two studies by Fard et al[19],[24] were found to contribute most to the heterogeneity of the superior pRNFL thickness (Table 6).
Figure 7. Sensitivity analyses of pRNFL thickness in patients with NAION and POAG.
A: Average pRNFL thickness; B: Superior pRNFL thickness; C: Inferior pRNFL thickness; D: Nasal pRNFL thickness; E: Temporal pRNFL thickness.
Table 6. Sensitivity analysis of superior pRNFL thickness.
| Study excluded | Fixed-effects model |
Random-effects model |
HeterogeneityI2 | ||
| WMD (95%CI) | P | WMD (95%CI) | P | ||
| Hondur 2021[18] | -6.50 (-10.70, -2.30) | 0.002 | -6.05 (-13.08, 0.97) | 0.09 | 60% |
| Fard 2020[19] | -9.16 (-13.11, -5.22) | <0.00001 | -9.09 (-13.29, -4.89) | <0.0001 | 8% |
| Resch 2018[22] | -6.84 (-10.60, -3.08) | 0.0004 | -6.28 (-12.58, 0.03) | 0.05 | 60% |
| Resch 2018[22] | -7.05 (-10.89, -3.22) | 0.0003 | -6.56 (-13.06, -0.07) | 0.05 | 60% |
| Fard 2018[24] | -4.15 (-8.21, -0.10) | 0.004 | -4.09 (-8.44, 0.25) | 0.06 | 9% |
| Liu 2017[25] | -6.76 (-10.49, -3.02) | 0.0004 | -6.11 (-12.28, 0.07) | 0.05 | 60% |
| Lee 2017[26] | -7.16 (-11.22, -3.09) | 0.0006 | -6.50 (-13.39, 0.39) | 0.06 | 60% |
| Fard 2016[29] | -6.98 (-10.81, -3.15) | 0.0004 | -6.46 (-12.96, 0.03) | 0.05 | 60% |
| Fard 2016[28] | -7.13 (-10.96, -3.30) | 0.0003 | -6.69 (-13.14, -0.23) | 0.04 | 60% |
pRNFL: Peripapillary retinal nerve fiber layer.
DISCUSSION
In the present study, we pooled the average and quadrant pRNFL thickness in NAION and POAG patients. Our data demonstrated no significant difference in the average pRNFL thickness between the NAION and POAG eyes (P=0.196). This finding is consistent with those of previous studies[5],[17]–[26],[29]–[30],[32]–[33],[41]. Currently, POAG has been reported to be associated with multiple risk factors including race, age, elevated intraocular pressure, family history, myopia, and diastolic perfusion pressure, etc[3],[42]–[47]. In glaucomatous neurodegeneration, the increase in intraocular pressure or imbalance of trans-laminar cribrosa pressure continuously stresses the RGCs and their supporting glia, leading to the progressive loss of RGCs and axons[48]. In contrast to glaucoma, NAION is presumed to be correlated with factors that can disrupt vascular autoregulation, such as nocturnal hypotension and other predisposed conditions such as small-to-disc ratio, etc[49]–[50]. Transient infarction of the anterior segment of the optic nerve nourished by the posterior ciliary artery circulation also results in the deterioration of RGCs and axons[51], where NAION may mimic POAG with regard to the average pRNFL thickness.
Despite no difference in average pRNFL thickness, the superior pRNFL was significantly thinner in NAION eyes (P=0.031), whereas the inferior sector was significantly thicker (P≤0.001) when the MD of the VF was similar between NAION and POAG. These findings could be explained in several aspects. First, studies have reported that the inferior altitudinal VF defect was more common in NAION patients, although the VF defects tended to be less diffuse compared to POAG, while the superior hemifield loss was more commonly seen in glaucoma patients[32],[51]–[54]. Moreover, the superior altitudinal VF defects can result from the loss of inferior RNFL thickness since structural deterioration can precede VF defects in glaucoma[55]–[56]. These structure-function relationship findings are in line with our findings that the superior pRNFL is significantly thinner in NAION eyes, whereas the inferior pRNFL thickness is significantly thinner in POAG eyes. Second, a longitudinal study revealed that RNFL thickness was lowest superiorly at 6mo from the onset of NAION, indicating that peripapillary structure attenuation was most severe in the superior quadrant in NAION during this period[57]. Third, the loss of the neuro-retinal rim of glaucoma is shown to start from the inferior temporal to superior nasal sector[58], which may account for the different progression patterns between these two diseases. In addition, the diagnostic ability of inferior pRNFL thickness is highest in glaucoma[59]–[60]. Recently, with the use of OCT angiography, a study reported a stronger correlation between inferior peripapillary capillary density and inferior pRNFL thickness in POAG patients compared with NAION patients, suggesting a higher susceptibility of inferior peripapillary capillary density that may contribute to the attenuation of inferior pRNFL in POAG[24]. Another study revealed that the inferior parafoveal deep vasculature in POAG eyes was lower than that in NAION eyes, indicating that the vulnerable regions of POAG were more susceptible to optic damage[19].
To clarify the sources of heterogeneity, we performed a “leave-one-out” sensitivity analysis. No obvious change in the average and sectoral pRNFL thickness was observed after excluding each study, demonstrating that our data were stable and reliable. However, the results indicated that the two studies by Fard et al[19],[24] (more deviated from the estimate line; Figure 7B) contributed to the heterogeneity mostly of the superior pRNFL thickness (I2=54.7%). Nevertheless, heterogeneity was largely reduced after excluding these two studies separately (the heterogeneity dropped from 54.1% to 9% when we excluded Fard et al[24]; the heterogeneity dropped from 54.1% to 8% when we excluded Fard et al[19]).
The loss of pRNFL thickness is reported to reach a plateau at 6mo from the onset of NAION, and to be more correlated with VF at the atrophic stage[57]. Similarly, the pRNFL decreases rapidly in early-to-moderate glaucoma, showing a “floor effect” where the pRNFL decreases relatively slower in the advanced stage[30]. For this reason, we included most of the studies that enrolled POAG patients at moderate or more severe stages with comparable MD of the VF to ensure the similar severities in these diseases. However, pRNFL thickness is also shown to decrease slowly 3mo after NAION onset[27]. Based on these findings, we performed a subgroup analysis attributed by the time from the onset of NAION. Our pooled results indicated a significant decrease of inferior pRNFL thickness in POAG eyes compared with that in NAION eyes, regardless of the onset time (onset time >3mo: P=0.002; onset time >6mo: P≤0.001). Nevertheless, unlike the combined data of the superior pRNFL thickness (n=9), there was no significant difference between NAION and POAG eyes (onset time >3mo, P=0.075, n=4; onset time >6mo, P=0.201, n=5). This may be due to the sources of heterogeneity introduced by Fard et al[19],[24] when patients were enrolled at 6mo from the onset, and the relatively small sample size of this subgroup (n=5).
In the subgroup analysis according to the SD-OCT type, the pooled results demonstrated that the inferior pRNFL thickness was significantly lower in the POAG eyes regardless of the SD-OCT type (Spectralis: P≤0.001; Cirrus: P=0.007; Optovue: P=0.003). Interestingly, in the subgroup analysis regarding the superior pRNFL thickness, a significant decrease was found in patients compared to that in POAG patients when the Spectralis SD-OCT was used (P=0.002). However, the superior pRNFL thickness was lower in NAION than in POAG, but no significant difference was observed when Cirrus and Optovue SD-OCT were used (Cirrus: P=0.150; Optovue: P=0.896). One reason may be the differences in the parameters, algorithms and software in different types of SD-OCT, although all types of SD-OCT devices can provide similar diagnostic abilities to detect the typical pattern of glaucomatous pRNFL deterioration[61]. In addition, since the relatively small sample size in Cirrus (n=2) and Optovue (n=2) groups, more studies are needed to fully assess the influence of SD-OCT types on measuring the sectoral pRNFL thickness.
This study has several limitations despite its strengths. First, the 10 included studies were cross-sectional studies. Further longitudinal studies and studies using eight-quadrant or clock-hour classification method are needed to verify our findings. Second, the sample size in the subgroup analyses was relatively small when Cirrus and Optovue SD-OCT were used. This may have introduced difficulties in completely assessing heterogeneity due to inadequate data. Therefore, interpreting the results should be cautious. In addition, we did not perform subgroup analysis according to the severity of glaucoma because most of the studies recruited patients with moderate to severe glaucoma. Nevertheless, we only included studies that enrolled NAION and POAG patients with comparable MD of the VF, to ensure similar severities in these diseases.
In conclusion, SD-OCT-based evaluation of pRNFL thickness reveals that the superior pRNFL was significantly lower in NAION eyes, whereas the inferior pRNFL thickness was significantly lower in POAG eyes. In the future, the application of SD-OCT in evaluating pRNFL thickness may help us better understand the different pathophysiological mechanisms between NAION and POAG, and help in differentiating these two diseases in a non-invasive manner.
Acknowledgments
Authors' contributions: Tong YX, Zhang XY and Jiang B: Conceptualization and design. Tong YX, Zhang XY and He Y: Literature search, data extraction, quality assessment and data analysis. Tong YX, Zhang XY, He Y, Chen ZL and Jiang B: Manuscript writing and editing. Jiang B: Supervision.
Foundations: Supported by National Natural Science Foundation of China (No.82070967; No.81770930); National Natural Science Foundation of Hunan Province Grant (No.2020jj4788); China Hunan Provincial Science and Technology Department (No.2020SK2086).
Conflicts of Interest: Tong YX, None; Zhang XY, None; He Y, None; Chen ZL, None; Jiang B, None.
REFERENCES
- 1.Stein JD, Khawaja AP, Weizer JS. Glaucoma in adults-screening, diagnosis, and management: a review. JAMA. 2021;325(2):164–174. doi: 10.1001/jama.2020.21899. [DOI] [PubMed] [Google Scholar]
- 2.Lee EJ, Han JC, Park DY, Kee C. A neuroglia-based interpretation of glaucomatous neuroretinal rim thinning in the optic nerve head. Prog Retin Eye Res. 2020;77:100840. doi: 10.1016/j.preteyeres.2020.100840. [DOI] [PubMed] [Google Scholar]
- 3.Weinreb RN, Khaw PT. Primary open-angle glaucoma. Lancet. 2004;363(9422):1711–1720. doi: 10.1016/S0140-6736(04)16257-0. [DOI] [PubMed] [Google Scholar]
- 4.Jonas JB, Aung T, Bourne RR, Bron AM, Ritch R, Panda-Jonas S. Glaucoma. Lancet. 2017;390(10108):2183–2193. doi: 10.1016/S0140-6736(17)31469-1. [DOI] [PubMed] [Google Scholar]
- 5.Danesh-Meyer HV, Boland MV, Savino PJ, Miller NR, Subramanian PS, Girkin CA, Quigley HA. Optic disc morphology in open-angle glaucoma compared with anterior ischemic optic neuropathies. Invest Ophthalmol Vis Sci. 2010;51(4):2003–2010. doi: 10.1167/iovs.09-3492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bajpai V, Madan S, Beri S. Arteritic anterior ischaemic optic neuropathy: an update. Eur J Ophthalmol. 2021;31(6):2818–2827. doi: 10.1177/11206721211009447. [DOI] [PubMed] [Google Scholar]
- 7.Hayreh SS. Ischemic optic neuropathy. Prog Retin Eye Res. 2009;28(1):34–62. doi: 10.1016/j.preteyeres.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 8.Kalyani VK, Bharucha KM, Goyal N, Deshpande MM. Comparison of diagnostic ability of standard automated perimetry, short wavelength automated perimetry, retinal nerve fiber layer thickness analysis and ganglion cell layer thickness analysis in early detection of glaucoma. Indian J Ophthalmol. 2021;69(5):1108–1112. doi: 10.4103/ijo.IJO_2409_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brusini P. OCT Glaucoma Staging System: a new method for retinal nerve fiber layer damage classification using spectral-domain OCT. Eye (Lond) 2018;32(1):113–119. doi: 10.1038/eye.2017.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Toprak I, Yaylalı V, Yildirim C. Diagnostic consistency and relation between optical coherence tomography and standard automated perimetry in primary open-angle glaucoma. Semin Ophthalmol. 2017;32(2):185–190. doi: 10.3109/08820538.2015.1046557. [DOI] [PubMed] [Google Scholar]
- 11.Hood DC. Improving our understanding, and detection, of glaucomatous damage: an approach based upon optical coherence tomography (OCT) Prog Retin Eye Res. 2017;57:46–75. doi: 10.1016/j.preteyeres.2016.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alnawaiseh M, Hömberg L, Eter N, Prokosch V. Comparison between the correlations of retinal nerve fiber layer thickness measured by spectral domain optical coherence tomography and visual field defects in standard automated white-on-white perimetry versus pulsar perimetry. J Ophthalmol. 2017;2017:8014294. doi: 10.1155/2017/8014294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Seymenoğlu G, Başer E, Oztürk B. Comparison of spectral-domain optical coherence tomography and Heidelberg retina tomograph III optic nerve head parameters in glaucoma. Ophthalmologica. 2013;229(2):101–105. doi: 10.1159/000341574. [DOI] [PubMed] [Google Scholar]
- 14.Vazquez LE, Bye A, Aref AA. Recent developments in the use of optical coherence tomography for glaucoma. Curr Opin Ophthalmol. 2021;32(2):98–104. doi: 10.1097/ICU.0000000000000733. [DOI] [PubMed] [Google Scholar]
- 15.Pazos M, Biarnés M, Blasco-Alberto A, Dyrda A, Luque-Fernández MÁ, Gómez A, Mora C, Milla E, Muniesa M, Antón A, Díaz-Alemán VT. SD-OCT peripapillary nerve fibre layer and ganglion cell complex parameters in glaucoma: principal component analysis. Br J Ophthalmol. 2021;105(4):496–501. doi: 10.1136/bjophthalmol-2020-316296. [DOI] [PubMed] [Google Scholar]
- 16.Hou HY, Moghimi S, Zangwill LM, Shoji T, Ghahari E, Penteado RC, Akagi T, Manalastas PIC, Weinreb RN. Macula vessel density and thickness in early primary open-angle glaucoma. Am J Ophthalmol. 2019;199:120–132. doi: 10.1016/j.ajo.2018.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shin JW, Lee JY, Lee BJ, Lim HT, Kook MS. Clinical characteristics of choroidal microvasculature dropout in normal-tension glaucoma versus nonarteritic anterior ischemic optic neuropathy: an optical coherence tomography angiography study. Sci Rep. 2021;11:21391. doi: 10.1038/s41598-021-00868-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hondur G, Sen E, Budakoglu O. Microvascular and structural alterations in the optic nerve head of advanced primary open-angle glaucoma compared with atrophic non-arteritic anterior ischemic optic neuropathy. Graefes Arch Clin Exp Ophthalmol. 2021;259(7):1945–1953. doi: 10.1007/s00417-021-05122-2. [DOI] [PubMed] [Google Scholar]
- 19.Fard MA, Fakhraee G, Ghahvechian H, Sahraian A, Moghimi S, Ritch R. Macular vascularity in ischemic optic neuropathy compared to glaucoma by projection-resolved optical coherence tomography angiography. Am J Ophthalmol. 2020;209:27–34. doi: 10.1016/j.ajo.2019.09.015. [DOI] [PubMed] [Google Scholar]
- 20.Rebolleda G, Pérez-Sarriegui A, Díez-Álvarez L, De Juan V, Muñoz-Negrete FJ. Lamina cribrosa position and Bruch's membrane opening differences between anterior ischemic optic neuropathy and open-angle glaucoma. Eur J Ophthalmol. 2019;29(2):202–209. doi: 10.1177/1120672118782101. [DOI] [PubMed] [Google Scholar]
- 21.Lee EJ, Han JC, Park DY, Kee C. Difference in topographic pattern of prelaminar and neuroretinal rim thinning between nonarteritic anterior ischemic optic neuropathy and glaucoma. Invest Ophthalmol Vis Sci. 2019;60(7):2461–2467. doi: 10.1167/iovs.19-26891. [DOI] [PubMed] [Google Scholar]
- 22.Resch H, Mitsch C, Pereira I, Schwarzhans F, Wasserman L, Hommer A, Reitner A, Vass C. Optic nerve head morphology in primary open-angle glaucoma and nonarteritic anterior ischaemic optic neuropathy measured with spectral domain optical coherence tomography. Acta Ophthalmol. 2018;96(8):e1018–e1024. doi: 10.1111/aos.13804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mastropasqua R, Agnifili L, Borrelli E, Fasanella V, Brescia L, Di Antonio L, Mastropasqua L. Optical coherence tomography angiography of the peripapillary retina in normal-tension glaucoma and chronic nonarteritic anterior ischemic optic neuropathy. Curr Eye Res. 2018;43(6):778–784. doi: 10.1080/02713683.2018.1438630. [DOI] [PubMed] [Google Scholar]
- 24.Fard MA, Suwan, Moghimi S, Geyman LS, Chui TY, Rosen RB, Ritch R. Pattern of peripapillary capillary density loss in ischemic optic neuropathy compared to that in primary open-angle glaucoma. PLoS One. 2018;13(1):e0189237. doi: 10.1371/journal.pone.0189237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liu CH, Wu WC, Sun MH, Kao LY, Lee YS, Chen HSL. Comparison of the retinal microvascular density between open angle glaucoma and nonarteritic anterior ischemic optic neuropathy. Invest Ophthalmol Vis Sci. 2017;58(9):3350–3356. doi: 10.1167/iovs.17-22021. [DOI] [PubMed] [Google Scholar]
- 26.Lee YH, Kim KN, Heo DW, Kang TS, Lee SB, Kim CS. Difference in patterns of retinal ganglion cell damage between primary open-angle glaucoma and non-arteritic anterior ischaemic optic neuropathy. PLoS One. 2017;12(10):e0187093. doi: 10.1371/journal.pone.0187093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Han M, Zhao C, Han QH, Xie SY, Li Y. Change of retinal nerve layer thickness in non-arteritic anterior ischemic optic neuropathy revealed by Fourier domain optical coherence tomography. Curr Eye Res. 2016;41(8):1076–1081. doi: 10.3109/02713683.2015.1084640. [DOI] [PubMed] [Google Scholar]
- 28.Fard MA, Afzali M, Abdi P, Yasseri M, Ebrahimi KB, Moghimi S. Comparison of the pattern of macular ganglion cell-inner plexiform layer defect between ischemic optic neuropathy and open-angle glaucoma. Invest Ophthalmol Vis Sci. 2016;57(3):1011–1016. doi: 10.1167/iovs.15-18618. [DOI] [PubMed] [Google Scholar]
- 29.Fard MA, Afzali M, Abdi P, Chen R, Yaseri M, Azaripour E, Moghimi S. Optic nerve head morphology in nonarteritic anterior ischemic optic neuropathy compared to open-angle glaucoma. Invest Ophthalmol Vis Sci. 2016;57(11):4632–4640. doi: 10.1167/iovs.16-19442. [DOI] [PubMed] [Google Scholar]
- 30.Han S, Jung JJ, Kim US. Differences between non-arteritic anterior ischemic optic neuropathy and open angle glaucoma with altitudinal visual field defect. Korean J Ophthalmol. 2015;29(6):418–423. doi: 10.3341/kjo.2015.29.6.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Savini G, Carbonelli M, Barboni P. Spectral-domain optical coherence tomography for the diagnosis and follow-up of glaucoma. Curr Opin Ophthalmol. 2011;22(2):115–123. doi: 10.1097/ICU.0b013e3283437222. [DOI] [PubMed] [Google Scholar]
- 32.Horowitz J, Fishelzon-Arev T, Rath EZ, Segev E, Geyer O. Comparison of optic nerve head topography findings in eyes with non-arteritic anterior ischemic optic neuropathy and eyes with glaucoma. Graefes Arch Clin Exp Ophthalmol. 2010;248(6):845–851. doi: 10.1007/s00417-010-1323-z. [DOI] [PubMed] [Google Scholar]
- 33.Saito H, Tomidokoro A, Sugimoto E, Aihara M, Tomita G, Fujie K, Wakakura M, Araie M. Optic disc topography and peripapillary retinal nerve fiber layer thickness in nonarteritic ischemic optic neuropathy and open-angle glaucoma. Ophthalmology. 2006;113(8):1340–1344. doi: 10.1016/j.ophtha.2006.01.072. [DOI] [PubMed] [Google Scholar]
- 34.Pasol J. Neuro-ophthalmic disease and optical coherence tomography: glaucoma look-alikes. Curr Opin Ophthalmol. 2011;22(2):124–132. doi: 10.1097/ICU.0b013e328343c1a3. [DOI] [PubMed] [Google Scholar]
- 35.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1–e34. doi: 10.1016/j.jclinepi.2009.06.006. [DOI] [PubMed] [Google Scholar]
- 36.Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–2012. doi: 10.1001/jama.283.15.2008. [DOI] [PubMed] [Google Scholar]
- 37.Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50(4):1088–1101. [PubMed] [Google Scholar]
- 40.Tong YX, Wang TT, Zhang XY, He Y, Jiang B. Optical coherence tomography evaluation of peripapillary and macular structure changes in pre-perimetric glaucoma, early perimetric glaucoma, and ocular hypertension: a systematic review and meta-analysis. Front Med (Lausanne) 2021;8:696004. doi: 10.3389/fmed.2021.696004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Akbari M, Abdi P, Fard MA, Afzali M, Ameri A, Yazdani-Abyaneh A, Mohammadi M, Moghimi S. Retinal ganglion cell loss precedes retinal nerve fiber thinning in nonarteritic anterior ischemic optic neuropathy. J Neuroophthalmol. 2016;36(2):141–146. doi: 10.1097/WNO.0000000000000345. [DOI] [PubMed] [Google Scholar]
- 42.Wu J, Hao J, Du Y, Cao K, Lin C, Sun R, Xie Y, Wang N. The association between myopia and primary open-angle glaucoma: a systematic review and meta-analysis. Ophthalmic Res. 2021 doi: 10.1159/000520468. [DOI] [PubMed] [Google Scholar]
- 43.Torabi R, Harris A, Siesky B, Zukerman R, Oddone F, Mathew S, Januleviciene I, Vercellin A. Prevalence rates and risk factors for primary open angle glaucoma in the middle east. J Ophthalmic Vis Res. 2021;16(4):644–656. doi: 10.18502/jovr.v16i4.9755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Siggs OM, Han XK, Qassim A, Souzeau E, Kuruvilla S, Marshall HN, Mullany S, MacKey DA, Hewitt AW, Gharahkhani P, MacGregor S, Craig JE. Association of monogenic and polygenic risk with the prevalence of open-angle glaucoma. JAMA Ophthalmol. 2021;139(9):1023–1028. doi: 10.1001/jamaophthalmol.2021.2440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schuster AK, Wagner FM, Pfeiffer N, Hoffmann EM. Risk factors for open-angle glaucoma and recommendations for glaucoma screening. Ophthalmologe. 2021;118(2):145–152. doi: 10.1007/s00347-021-01378-5. [DOI] [PubMed] [Google Scholar]
- 46.Liuska PJ, Harju M, Kivelä TT, Turunen JA. Prevalence of MYOC risk variants for glaucoma in different populations. Acta Ophthalmol. 2021;99(7):e1090–e1097. doi: 10.1111/aos.14738. [DOI] [PubMed] [Google Scholar]
- 47.Wurster P, Harris A, Gonzalez AC, Adjei S, Verticchio Vercellin A, Mathew S, Lang M, Eikenberry J, Siesky B. Risk factors for open-angle glaucoma in persons of Latin American descent. J Glaucoma. 2020;29(3):217–225. doi: 10.1097/IJG.0000000000001429. [DOI] [PubMed] [Google Scholar]
- 48.Ren RJ, Wang NL, Zhang XJ, Cui TT, Jonas JB. Trans-lamina cribrosa pressure difference correlated with neuroretinal rim area in glaucoma. Graefes Arch Clin Exp Ophthalmol. 2011;249(7):1057–1063. doi: 10.1007/s00417-011-1657-1. [DOI] [PubMed] [Google Scholar]
- 49.Berry S, Lin WV, Sadaka A, Lee AG. Nonarteritic anterior ischemic optic neuropathy: cause, effect, and management. Eye Brain. 2017;9:23–28. doi: 10.2147/EB.S125311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hayreh SS, Podhajsky P, Zimmerman MB. Role of nocturnal arterial hypotension in optic nerve head ischemic disorders. Ophthalmologica. 1999;213(2):76–96. doi: 10.1159/000027399. [DOI] [PubMed] [Google Scholar]
- 51.Hayreh SS, Zimmerman B. Visual field abnormalities in nonarteritic anterior ischemic optic neuropathy: their pattern and prevalence at initial examination. Arch Ophthalmol. 2005;123(11):1554–1562. doi: 10.1001/archopht.123.11.1554. [DOI] [PubMed] [Google Scholar]
- 52.Choi J, Cho HS, Lee CH, Kook MS. Scanning laser polarimetry with variable corneal compensation in the area of apparently normal hemifield in eyes with normal-tension glaucoma. Ophthalmology. 2006;113(11):1954–1960. doi: 10.1016/j.ophtha.2006.05.040. [DOI] [PubMed] [Google Scholar]
- 53.Gerling J, Meyer JH, Kommerell G. Visual field defects in optic neuritis and anterior ischemic optic neuropathy: distinctive features. Graefes Arch Clin Exp Ophthalmol. 1998;236(3):188–192. doi: 10.1007/s004170050062. [DOI] [PubMed] [Google Scholar]
- 54.Traustason OI, Feldon SE, Leemaster JE, Weiner JM. Anterior ischemic optic neuropathy: classification of field defects by Octopus™ automated static perimetry. Graefes Arch Clin Exp Ophthalmol. 1988;226(3):206–212. doi: 10.1007/BF02181182. [DOI] [PubMed] [Google Scholar]
- 55.Sommer A. Clinically detectable nerve fiber atrophy precedes the onset of glaucomatous field loss. Arch Ophthalmol. 1991;109(1):77. doi: 10.1001/archopht.1991.01080010079037. [DOI] [PubMed] [Google Scholar]
- 56.Quigley HA, Miller NR, George T. Clinical evaluation of nerve fiber layer atrophy as an indicator of glaucomatous optic nerve damage. Arch Ophthalmol. 1980;98(9):1564–1571. doi: 10.1001/archopht.1980.01020040416003. [DOI] [PubMed] [Google Scholar]
- 57.Contreras I, Noval S, Rebolleda G, Muñoz-Negrete FJ. Follow-up of nonarteritic anterior ischemic optic neuropathy with optical coherence tomography. Ophthalmology. 2007;114(12):2338–2344.e1. doi: 10.1016/j.ophtha.2007.05.042. [DOI] [PubMed] [Google Scholar]
- 58.Jonas JB, Fernández MC, Stürmer J. Pattern of glaucomatous neuroretinal rim loss. Ophthalmology. 1993;100(1):63–68. doi: 10.1016/s0161-6420(13)31694-7. [DOI] [PubMed] [Google Scholar]
- 59.Park SB, Sung KR, Kang SY, Kim KR, Kook MS. Comparison of glaucoma diagnostic Capabilities of Cirrus HD and Stratus optical coherence tomography. Arch Ophthalmol. 2009;127(12):1603–1609. doi: 10.1001/archophthalmol.2009.296. [DOI] [PubMed] [Google Scholar]
- 60.Leung CKS, Cheung CYL, Weinreb RN, Qiu QL, Liu S, Li HT, Xu GH, Fan N, Huang LN, Pang CP, Lam DSC. Retinal nerve fiber layer imaging with spectral-domain optical coherence tomography. Ophthalmology. 2009;116(7):1257–1263.e2. doi: 10.1016/j.ophtha.2009.04.013. [DOI] [PubMed] [Google Scholar]
- 61.Chen TC, Hoguet A, Junk AK, Nouri-Mahdavi K, Radhakrishnan S, Takusagawa HL, Chen PP. Spectral-domain OCT: helping the clinician diagnose glaucoma. Ophthalmology. 2018;125(11):1817–1827. doi: 10.1016/j.ophtha.2018.05.008. [DOI] [PubMed] [Google Scholar]