Abstract
Background
Adipose-derived mesenchymal stem cells (ADMSCs) are a promising source of material source for medical regeneration of cartilage. Growth factors, including transforming growth factor-β (TGFβ) subfamily members and bone morphogenetic proteins (BMPs), play important roles in inducing and promoting chondrogenic differentiation of MSCs. However, these exogenous growth factors have some drawbacks related to their cost, biological half-life, and safety for clinical application. Several studies have reported that statins, the competitive inhibitors of 3-hydroxy-2-methylglutaryl coenzyme A (HMG-CoA) reductase, induce the expression of BMP2 in multiple cell types as the pleotropic effects. The objective of this study was to investigate the effects of fluvastatin during chondrogenic differentiation of human ADMSCs (hADMSCs).
Methods
The effects of fluvastatin were analyzed during chondrogenic differentiation of hADMSCs in the pellet culture without exogenous growth factors by qRT-PCR and histology. For functional studies, Noggin, an antagonist of BMPs, mevalonic acid (MVA) and geranylgeranyl pyrophosphate (GGPP), metabolites of the mevalonate pathway, ROCK inhibitor (Y27632), or RAC1 inhibitor (NSC23766) were applied to cells during chondrogenic differentiation. Furthermore, RhoA activity was measured by RhoA pulldown assay during chondrogenic differentiation with or without fluvastatin. Statistically significant differences between groups were determined by Student’s t-test or the Tukey–Kramer test.
Results
Fluvastatin-treated cells expressed higher levels of BMP2, SOX9, ACAN, and COL2A1 than control cells, and accumulated higher levels of glycosaminoglycans (GAGs). Noggin significantly inhibited the fluvastatin-mediated upregulation of ACAN and COL2A1. Both MVA and GGPP suppressed the effects of fluvastatin on the expressions of BMP2, SOX9, ACAN, and COL2A1. Furthermore, fluvastatin suppressed the RhoA activity, and inhibition of RhoA–ROCK signaling by Y27632 increased the expressions of BMP2, SOX9, ACAN, and COL2A1, as well as fluvastatin.
Conclusions
Our results suggest that fluvastatin promotes chondrogenic differentiation of hADMSCs by inducing endogenous BMP2, and that one of the mechanisms underlying the effects is inhibition of RhoA–ROCK signaling via suppression of GGPP. Fluvastatin is a safe and low-cost compound that holds promise for use in transplantation of hADMSCs for cartilage regeneration.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40360-022-00600-7.
Keywords: Fluvastatin, ADMSCs, Chondrogenic differentiation, BMP2, Mevalonate pathway, GGPP, RhoA–ROCK signaling
Background
Due to their high self-renewal capacity and chondrogenic differentiation potential, mesenchymal stem cells (MSCs) are a promising source of material source for medical regeneration of cartilage [1, 2]. MSCs can be isolated from various tissues such as bone marrow, adipose tissue, and synovium. Adipose-derived mesenchymal stem cells (ADMSCs) have advantages over other cells in terms of harvest volume and the safety of the procedure [3, 4]. Johnstone et al. first reported in vitro chondrogenic differentiation of MSCs in pellet culture [5], which requires growth factors and other compounds [6]. In particular, growth factors play important roles in inducing and promoting chondrogenic differentiation of MSCs, including transforming growth factor-β (TGFβ) subfamily members and bone morphogenetic proteins (BMPs) [7]. However, these exogenous growth factors have some drawbacks related to their cost, biological half-life, and safety for clinical application [8–11].
Statins are competitive inhibitors of 3-hydroxy-2-methylglutaryl coenzyme A (HMG-CoA) reductase and are used clinically throughout the world to decrease serum cholesterol levels by inhibiting the mevalonate pathway [12]. Importantly, several studies have suggested that statins have pleotropic effects, including alterations of cell differentiation, by inhibiting synthesis of isoprenoid, which is required for post-translational modification of small GTP-binding proteins such as the members of the Rho family [13].
Mundy et al. first reported that statins induced the expression of BMP2 in osteoblasts and stimulated new bone formation in vitro and in vivo [14]. Subsequently, statins were reported to induce the expression of BMP2 in multiple cell types, including MSCs during osteogenic differentiation [15], chondrocytes [16, 17], intervertebral disc cells [18], vascular smooth muscle cells [19], and colorectal cancer cell lines [20]. In mesenchymal cells to the chondrogenic lineage and in growth plate chondrocytes, BMP signaling stimulates chondrogenic differentiation and maturation. Among the members of this family, BMP2 plays a crucial role in the condensation of MSCs and promotes the synthesis of extracellular matrix [7, 21, 22]. Furthermore, exogenous BMP2, alone or combined with other growth factors, has been used to differentiate MSCs into the chondrogenic lineage [23–27]. Therefore, we hypothesized that statins would promote chondrogenic differentiation of MSCs by inducing endogenous BMP2.
In this study, we showed that fluvastatin could promote chondrogenic differentiation of human ADMSCs (hADMSCs) in pellet culture without exogenous growth factors, and investigated the underlying mechanism.
Materials and methods
Cell culture
hADMSCs were purchased from Promo Cell (Heidelberg, Germany). The cells were cultured in MSC Growth Medium 2 (Promo Cell) at 37 °C in an atmosphere containing 5% CO2 and maintained at sub-confluence to prevent spontaneous differentiation, according to instructions recommended by the manufacturer. Cells from passages 3–6 were used in this study.
Cell viability assay
hADMSCs were seeded in 96-well plates at a density of 5 × 103 cells/well, incubated in high-glucose GlutaMAX DMEM medium (Gibco, Langley, OK, USA) with 1% FBS supplemented with fluvastatin (Toronto Research Chemicals, North York, Canada) at 0, 0.01, 0.1 or 1 μM for 2 weeks. Culture media were replaced every 3–4 days. The cell viability assay was performed using a CellTiter-Glo Luminescent Cell Viability Assay kit (Promega, Fitchburg, WI) in accordance with the manufacturer’s protocol. Luminescence was measured using a MicroLumatPlus LB96V Microplate Luminometer (EG&G Berthold, Bad Wildbad, Germany).
In vitro chondrogenic differentiation
hADMSCs were seeded in 96-well U-bottom plates (Sumitomo Bakelite, Tokyo, Japan) at a density of 2 × 105 cells/well in high-glucose GlutaMAX DMEM medium and incubated for 48 h. Under these conditions, the cells spontaneously formed 3D pellets. Then, to induce chondrogenic differentiation, the pellets were incubated in chondrogenic medium for 2 weeks at 37 °C in an atmosphere containing 5% CO2. High-glucose GlutaMAX DMEM medium supplemented with L-proline (40 μg/ml) (Sigma-Aldrich, St. Louis, MO, USA), dexamethasone (0.1 μM) (Sigma-Aldrich), 1% ITS (Sigma-Aldrich), L-ascorbic acid (50 μg/ml) (Sigma-Aldrich), and sodium pyruvate (110 μg/ml) (Gibco) was used as control chondrogenic medium. Chondrogenic differentiation of hADMSCs was performed under the following three conditions: control medium, control medium with human TGFβ1 (10 ng/ml) (R&D Systems, Minneapolis, MN, USA) or fluvastatin (0.1 μM). Chondrogenic media were replaced every 3–4 days.
Total RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was extracted from three hADMSC pellets for each condition. Briefly, pellets were crushed in a mini homogenizer and lysed in TRIzol reagent (Invitrogen, Carlsbad, CA, USA). Complementary DNA was synthesized using the PrimeScript RT Reagent (Takara Bio, Kusatsu, Japan). qRT-PCR was performed on a LightCycler 2.0 Instrument (F. Hoffmann–La Roche AG, Basel, Switzerland) using TB Green Premix EX TaqII (Takara Bio). GAPDH was used as an internal control and used to normalize the levels of other target genes across samples. The sequences of the forward and reverse primers were as Table 1.
Table 1.
Primer sequences used for qRT-PCR
| Genes | Forward (5’-3’) | Reverse (5’-3’) |
|---|---|---|
| Human SOX9 | AGACCTTTGGGCTGCCTTAT | TAGCCTCCCTCACTCCAAGA |
| Human ACAN | AGGCAGCGTGATCCTTACC | GGCCTCTCCAGTCTCATTCTC |
| Human COL2A1 | CAACACTGCCAACGTCCAGAT | CTGCTTCGTCCAGATAGGCAAT |
| Human COL10A1 | ACGCTGAACGATACCAAATG | TGCTATACCTTTACTCTTTATGGTGTA |
| Human BMP2 | CGAAACACAAACAGCGGAAAC | GCCACAATCCAGTCGTTCCA |
| Human GAPDH | GGTGAAGGTCGGAGTCAACGGA | GAGGGATCTCGCTCCTGGAAGA |
Alcian blue staining
Pellets of hADMSCs were differentiated under each condition for 2 weeks, fixed with 4% paraformaldehyde for 24 h, and embedded in paraffin. Sections were deparaffinized, rehydrated, and stained with 0.3% Alcian blue 8GX for 10 min. The percentages of positively stained areas were measured using the BZ-II Analyzer software (Keyence, Osaka, Japan).
BMP inhibition experiment
Recombinant human Noggin 500 ng/mL (R&D Systems) was applied to cells in control chondrogenic medium with or without 0.1 μM fluvastatin, as mentioned above. On days 14, total RNA extraction and qRT-PCR were performed, and the expressions of ACAN and COL2A1 was confirmed.
Mevalonate pathway experiment
Mevalonic acid (100 μM) (MVA; Sigma-Aldrich) or geranylgeranyl pyrophosphate (20 μM) (GGPP; Sigma-Aldrich) was applied to cells in chondrogenic medium with 0.1 μM fluvastatin, as described above. On days 4 and 7, total RNA extraction and qRT-PCR were performed, and the expressions of BMP2, SOX9, ACAN, and COL2A1 was confirmed.
RhoA–ROCK or RAC1 signaling inhibition
Y27632 (20 μM) (R&D Systems), a ROCK inhibitor, or NSC23766 (50 μM) (Sigma-Aldrich), a RAC1 inhibitor, was applied to cells in control chondrogenic medium, as described above. On days 4 and 7, total RNA extraction and qRT-PCR were performed, and the expressions of BMP2, SOX9, ACAN, and COL2A1 was confirmed.
RhoA pulldown assay and western blotting
RhoA activity was analyzed using the Rho Activation Assay Kit (Cytoskelton, Denver, CO, USA) according to the instructions of the manufacturer. hADMSCs were seeded in 96-well U-bottom plates at a density of 2 × 105 cells/well. After the cells spontaneously formed 3D pellets, the cells were induced chondrogenic differentiation with or without 0.1 μM fluvastatin. On day 1, the culture medium was replaced with fresh medium of each condition, and after 1 h, whole-cell lysates were extracted from five pellets. Briefly, pellets were crushed in a mini homogenizer and lysed using lysis buffer. Then, the cell lysates were clarified by centrifugation and incubated with rhotekin-Rho binding domain (RBD) beads at 4 °C for 1 h to selectively precipitate GTP-RhoA, the active form RhoA. Whole-cell lysates and the precipitates were determined the amount of RhoA by western blotting. The cell lysates were electrophoresed in 4–12% gradient polyacrylamide gels (Invitrogen), and the resolved proteins were transferred to nitrocellulose membranes (Amersham Biosciences, Arlington Heights, IL, USA). Membranes were blocked with blocking buffer (Takara Bio), washed in TBS with Tween (TBST), and incubated with primary antibodies against RhoA (number ARH05; Cytoskelton) and GAPDH (number 5174; Cell Signaling Technology, Danvers, MA, USA) diluted in Can Get Signal Immunoreaction Enhancer Solution 1 (TOYOBO, Osaka, Japan) (RhoA, 1:500; GAPDH, 1:1000) at 4 °C overnight. After the membranes were washed in TBST, secondary anti-mouse IgG antibodies (sc-516102; Santa Cruz Biotechnology, Dallas, TX, USA) or anti-rabbit IgG antibodies (number 7074; Cell Signaling Technology) were added. Immunoreactivity was detected with ECL Prime (Amersham Biosciences) and imaged on an Ez Capture MG (ATTO, Tokyo, Japan). Band densities were calculated using CS Analyzer 3.0 (ATTO).
Statistical analysis
All experiments were repeated at least three times. Data are presented as means ± standard deviation. Statistically significant differences between groups were determined by Student’s t-test or the Tukey–Kramer test. All data analyses were performed using the JMP 15 statistical software (SAS Institute, Cary, NC, USA). p < 0.05 was considered statistically significant.
Results
Fluvastatin promotes SOX9, ACAN and COL2A1 expressions during chondrogenic differentiation of hADMSCs (Fig. 1)
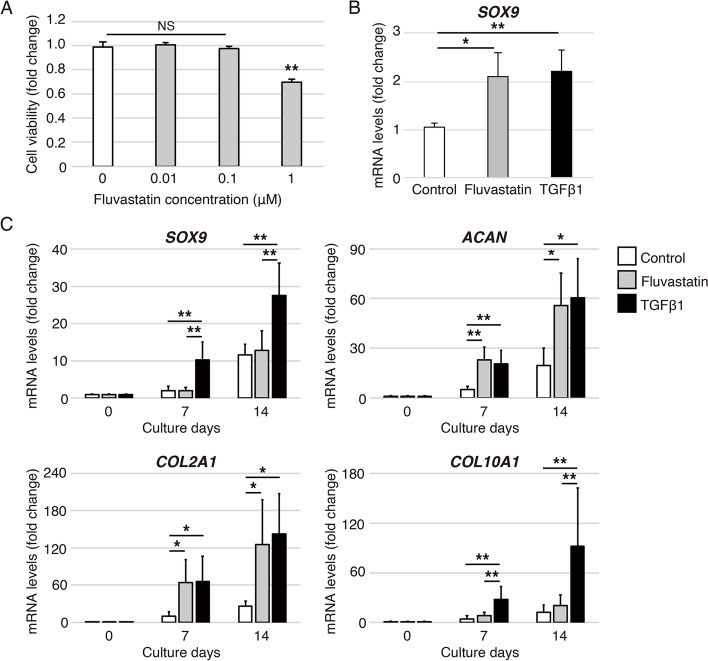
Fig. 1.
Effects of fluvastatin during chondrogenic differentiation of hADMSCs. A Effects of fluvastatin on cell viability of hADMSCs. hADMSCs were treated with fluvastatin at 0, 0.01, 0.1 or 1 μM for 2 weeks. Cell viability is shown relative to the level on treatment with 0 μM fluvastatin. Error bars denote the means ± standard deviation (n = 5). **p < 0.01, NS: not significant by Tukey–Kramer test. B Relative SOX9 mRNA levels on day 4 during chondrogenic differentiation of hADMSCs assessed by qRT-PCR; control, fluvastatin, and TGFβ1 groups are shown. Gene expression is given relative to the level in the control group. Error bars denote means ± standard deviation (n = 4). *p < 0.05, **p < 0.01 by Tukey–Kramer test. C Effects of fluvastatin on gene expression during chondrogenic differentiation of hADMSCs, as determined by qRT-PCR. Relative mRNA levels of SOX9, ACAN, COL2A1, and COL10A1 in chondrogenic differentiation of hADMSCs on days 0, 7, and 14 among control, fluvastatin, and TGFβ1 groups. Gene expression at each stage is shown relative to the level on day 0. Error bars denote the means ± standard deviation (n = 5). *p < 0.05, **p < 0.01 by Tukey–Kramer test
First, we investigated the effect of fluvastatin on cell viability of hADMSCs. Incubation with fluvastatin at concentrations below 0.1 μM for 2 weeks did not affect cell viability of hADMSCs (Fig. 1A).
Next, to examine whether fluvastatin promotes chondrogenic differentiation of hADMSCs, the following three conditions were prepared: the control, fluvastatin, and TGFβ1. TGFβ1, a major inducer of chondrogenic differentiation, was used as a positive control. The expressions of SOX9, ACAN, and COL2A1 as chondrogenic differentiation markers, and COL10A1 as a hypertrophic differentiation marker, were measured. The expression of SOX9 was significantly higher in the TGFβ1 and fluvastatin groups than in the control group on day 4 (Fig. 1B). SOX9 expression in the TGFβ1 group on days 7 and 14 was significantly higher than in the other groups (Fig. 1C). The expressions of ACAN and COL2A1 were significantly higher in the TGFβ1 and fluvastatin groups than in the control group on days 7 and 14. There were no significant differences in ACAN and COL2A1 expression between the TGFβ1 and fluvastatin groups (Fig. 1C). The expression of COL10A1 was significantly higher in the TGFβ1 group than in the other groups on days 7 and 14 (Fig. 1C).
Fluvastatin increases staining of glycosaminoglycans in pellets of hADMSCs
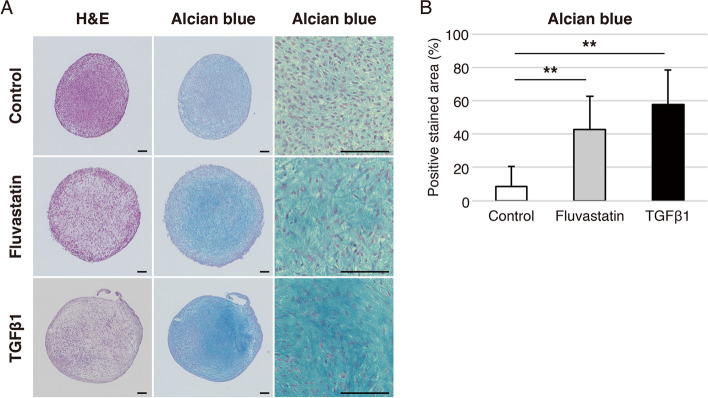
After 14 days of chondrogenic differentiation under the three conditions, we evaluated accumulation of glycosaminoglycans (GAGs) in pellets stained with Alcian blue. Levels of GAGs were significantly higher in the TGFβ1 and fluvastatin groups, but did not differ significantly between the TGFβ1 and fluvastatin groups (Fig. 2A and B).
Fig. 2.
Histological appearance of pellets of hADMSCs cultured in chondrogenic medium for 14 days. A Histological appearance of pellets of hADMSCs stained with hematoxylin and eosin (H&E) and Alcian blue; control, fluvastatin, and TGFβ1 groups are shown. Bars represent 100 μm. B Percentages of Alcian blue–positive areas in the control, fluvastatin, and TGFβ1 groups. Error bars denote means ± standard deviation (n = 10). **p < 0.01 by Tukey–Kramer test
Fluvastatin induces BMP2 expression in chondrogenic differentiation of hADMSCs
To elucidate the molecular mechanism of fluvastatin-promoted chondrogenic differentiation of hADMSCs, we confirmed the expression of BMP2. The results revealed that expression of BMP2 on day 4 during chondrogenic differentiation of hADMSCs was significantly higher in the TGFβ1 and fluvastatin groups than in the control group. Again, there was no significant difference between the TGFβ1 and fluvastatin groups (Fig. 3A).
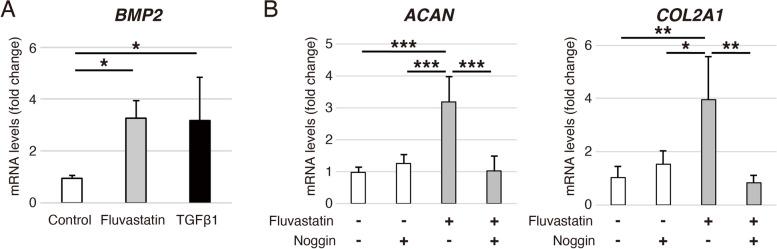
Fig. 3.
Fluvastatin-induced BMP2 expression and its effect on chondrogenic differentiation of hADMSCs. A Relative BMP2 mRNA levels on day 4 during chondrogenic differentiation of hADMSCs assessed by qRT-PCR; control, fluvastatin, and TGFβ1 groups are shown. Gene expression is given relative to the level in the control group. Error bars denote means ± standard deviation (n = 4). *p < 0.05 by Tukey–Kramer test. B Effects of Noggin on gene expression promoted by fluvastatin in chondrogenic differentiation of hADMSCs assessed by qRT-PCR. Relative ACAN and COL2A1 mRNA levels on day 14 in chondrogenic differentiation of hADMSCs cultured with or without fluvastatin or Noggin. Gene expression is expressed relative to the level in cells cultured without fluvastatin or Noggin. Error bars denote means ± standard deviation (n = 4). *p < 0.05, **p < 0.01, ***p < 0.001 by Tukey–Kramer test
Noggin blocks the expressions of ACAN and COL2A1 promoted by fluvastatin in chondrogenic differentiation of hADMSCs (Fig. 3)
To determine whether BMP2 induced by fluvastatin promotes the expressions of ACAN and COL2A1 during chondrogenic differentiation of hADMSCs, we administered 500 ng/mL Noggin to cells. In chondrogenic medium containing fluvastatin, Noggin significantly blocked the elevation of ACAN and COL2A1 expression on day 14 (Fig. 3B). On the other hand, Noggin did not suppress the baseline chondrogenic differentiation in control medium without fluvastatin (Fig. 3B).
Mevalonic acid and geranylgeranyl pyrophosphate suppress the effects of fluvastatin in chondrogenic differentiation of hADMSCs
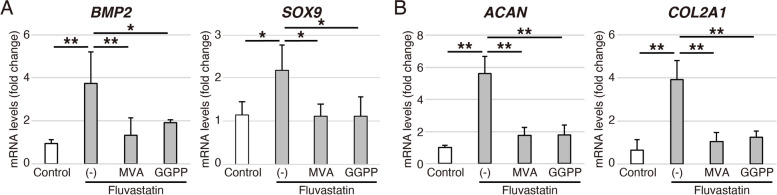
In the mevalonate pathway, MVA is produced from HMG-CoA by HMG-CoA reductase, and subsequently metabolized to GGPP [28]. Hence, for the further mechanistic analyses, we administered 100 μM MVA or 20 μM GGPP to cells in chondrogenic medium with fluvastatin. Both MVA and GGPP significantly suppressed the expressions of BMP2 and SOX9 on day 4 (Fig. 4A), and the expressions of ACAN and COL2A1 on day 7 (Fig. 4B).
Fig. 4.
Effects of MVA or GGPP on fluvastatin-induced gene expression by during chondrogenic differentiation of hADMSCs. A Relative BMP2 and SOX9 mRNA levels on day 4 and B relative ACAN and COL2A1 mRNA levels on day 7 in chondrogenic differentiation of hADMSCs cultured with or without fluvastatin, MVA, or GGPP, as determined by qRT-PCR. Gene expression is expressed relative to the level in cells cultured without fluvastatin, MVA, or GGPP. Error bars denote means ± standard deviation (n = 4). *p < 0.05, **p < 0.01 by Tukey–Kramer test
Inhibition of RhoA–ROCK signaling increases BMP2 and SOX9 expressions followed by ACAN and COL2A1 during chondrogenic differentiation of hADMSCs
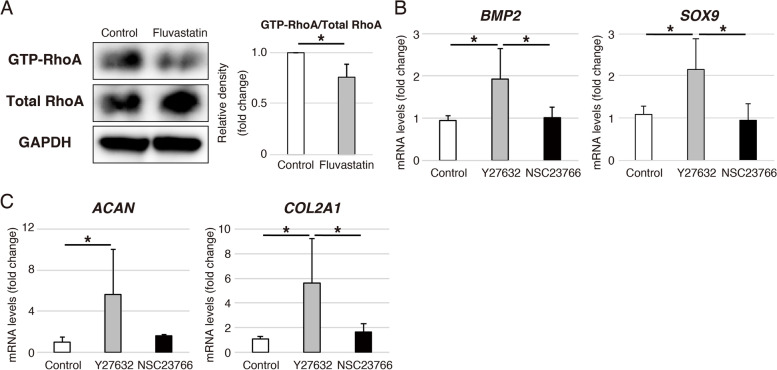
The GTP-RhoA, the active form RhoA, in cells treated with fluvastatin was significantly decreased compared to control on day 1 during chondrogenic differentiation of hADMSCs (Fig. 5A). Next, we applied 20 μM ROCK inhibitor (Y27632) and 50 μM RAC1 inhibitor (NSC23766) to cells in control chondrogenic medium in order to investigate the inhibitory effect of RhoA–ROCK or RAC1 signaling. ROCK inhibitor, but not RAC1 inhibitor, increased the expressions of BMP2 and SOX9 on day 4 (Fig. 5B), and the expression of ACAN and COL2A1 on day 7 (Fig. 5C).
Fig. 5.
Effects of fluvastatin on the RhoA activity in chondrogenic differentiation of hADMSCs. A Levels of GTP-RhoA, total RhoA, and GAPDH on day 1 in chondrogenic differentiation of hADMSCs cultured with or without fluvastatin, as determined by western blotting. GAPDH was used as a loading control. These blot images were cropped. Error bars denote means ± standard deviation (n = 3). *p < 0.05 by Student’s t-test. B Relative BMP2 and SOX9 mRNA levels on day 4 and (C) relative ACAN and COL2A1 mRNA levels on day 7 in chondrogenic differentiation of hADMSCs cultured with Y27632 or NSC23766, as determined by qRT-PCR. Gene expression is expressed relative to the level in cells cultured without Y27632 or NSC23766. Error bars denote means ± standard deviation (BMP2 and SOX9, n = 4; ACAN and COL2A1, n = 5). *p < 0.05 by Tukey–Kramer test
Discussion
In the present study, we revealed the positive effects of fluvastatin on chondrogenic differentiation of hADMSCs in the absence of exogenous growth factors. Fluvastatin significantly induced BMP2, SOX9, ACAN and COL2A1 expressions and accumulated GAGs in the pellet of hADMSCs. In the functional analyses, BMP inhibition by Noggin significantly inhibited the fluvastatin-mediated upregulation of ACAN and COL2A1. Exogenously added MVA and GGPP, HMG-CoA products, reversed the effects of fluvastatin on the gene expressions. Fluvastatin suppressed the RhoA activity, and inhibition of RhoA–ROCK signaling by Y27632 increased BMP2, SOX9, ACAN, and COL2A1 expressions. Taken together, our findings suggest that fluvastatin promotes chondrogenic differentiation of hADMSCs by inducing endogenous BMP2.
Statins were previously reported to promote chondrogenic differentiation of ATDC5 cells, an in vitro model of chondrogenic differentiation, and to rescue the chondrogenic differentiation ability of induced pluripotent stem (iPS) cells from patients with achondroplasia or thanatophoric dysplasia [29, 30]. These studies proposed that the mechanism by which statins influence chondrogenic differentiation is related to the Indian Hedgehog pathway or fibroblast growth factor receptor 3 (FGFR3); however, it is not fully understood. In this study, we focused on BMP2 as a statin-induced growth factor [15–20]. Exogenous BMP2 promotes chondrogenic differentiate of MSCs [23–27], and endogenous BMP2 expression is elevated during the early stage of TGFβ1-induced chondrogenic differentiation of MSCs [26, 31]. As we hypothesized, fluvastatin promoted the chondrogenic differentiation in hADMSCs accompanied by elevated BMP2 expression. This is the first to elucidate the potential of fluvastatin on chondrogenic induction of MSCs as alternative to exogenous growth factors for clinical application.
Statins exert the pleotropic effects by inhibiting isoprenoid synthesis, which is required for post-translational modification of small GTP-binding proteins such as the members of the Rho family [13]. In fact, fluvastatin decreased the RhoA activity, a downstream effector of GGPP, one of the isoprenoids, and the effects of fluvastatin were abolished by exogenous GGPP addition. Furthermore, the chemical inhibition of RhoA–ROCK signaling mimicked the anabolic effects of fluvastatin on chondrogenic differentiation of hADMSC. Several previous studies reported that the inhibition of RhoA–ROCK signaling enhanced chondrogenic gene expressions and cartilaginous extracellular matrix accumulation by reducing cytoskeletal tension in MSCs pellet culture [32–34]. Therefore, the underlying mechanism of the effects of fluvastatin might be involved in the inhibition of RhoA–ROCK signaling and the subsequent cytoskeletal reorganization.
In this study, the expression of SOX9, a master regulator of chondrogenesis, was higher on day 4 in the fluvastatin group than in the control group, after that, there was no significant difference between the two groups. In general, SOX9 is a transcription factor that acts during the early phase of chondrogenic differentiation of MSCs [35, 36]. BMP2 signaling and cytoskeletal reorganization, which we propose to be the mechanism by which fluvastatin promotes chondrogenesis, have been reported to be closely involved in SOX9 expression. BMP2 signaling increases the level of SOX9 expression by the p38 MAPK pathway within half a day after BMP2 administration to mouse embryonic fibroblasts [37]. The cytoskeletal reorganization of MSCs by the continuous low-intensity ultrasound stimulation also induces SOX9 upregulation immediately via the phosphorylation of ERK1/2 [38]. Based on these findings, the impact of fluvastatin on SOX9 expression should be predominant in the early stage of chondrogenic differentiation of hADMSCs.
This study has several limitations. First, we used primary hADMSCs, which may differ by donor. Second, we conducted only in vitro studies of chondrogenic differentiation, therefore we need to confirm the effects of fluvastatin in vivo. Third, this study failed to show that fluvastatin directly inhibits the intracellular synthesis of MVA and GGPP during chondrogenic differentiation of hADMSCs, because reliable microanalysis could not be performed under pellet culture. However, a number of reports used exogenous MVA and GGPP in the rescue experiment to abolish the effect of statin [39–41]. In addition, fluvastatin is a lipophilic statin with capable of transferring into a cell. Hence, fluvastatin exogenously administered could surely function in cytoplasm to inhibit the intracellular synthesis of MVA and GGPP.
Conclusions
In summary, we demonstrated that fluvastatin promotes chondrogenic differentiation of hADMSCs by inducing endogenous BMP2. One of the mechanisms underlying the effects of fluvastatin administration effects was inhibition of RhoA–ROCK signaling via suppression of GGPP. Although more work is needed to elucidate its molecular mechanism, fluvastatin is a safe and low-cost compound that hold promise for use in transplantation of hADMSCs for cartilage regeneration.
Supplementary Information
Additional file 1. Fig. S1 The original uncropped blot in Figure 5A. Black-boxed areas in the original blot were included in the main paper.
Acknowledgements
We thank Hitomi Kimura for supporting histological preparing.
Abbreviations
- MSCs
Mesenchymal stem cells
- hADMSCs
human adipose-derived mesenchymal stem cells
- TGFβ
transforming growth factor β
- BMPs
bone morphogenetic proteins
- HMG-CoA
3-hydroxy-2-methylglutaryl coenzyme A
- DMEM
Dulbecco’s modified Eagle’s medium
- FBS
fetal bovine serum
- 3D
three-dimensional
- ITS
insulin–transferrin–selenium
- RT
reverse transcription
- qRT-PCR
quantitative real-time polymerase chain reaction
- GAPDH
glyceraldehyde-3-phosphate dehydrogenase
- SOX9
SRY-box transcription factor 9
- ACAN
aggrecan
- COL2A1
collagen type II alpha 1 chain
- COL10A1
collagen type X alpha 1 chain
- GAGs
glycosaminoglycans
- MVA
mevalonic acid
- GGPP
geranylgeranyl pyrophosphate
- RhoA
ras homolog family member A
- ROCK
Rho associated coiled-coil containing protein kinase
- RAC1
Rac family small GTPase 1
- iPS cells
induced pluripotent stem cells
- FGFR3
fibroblast growth factor receptor 3
- MAPK
mitogen-activated protein kinase
Authors’ contributions
MK, YA, HT, and YN contributed to the conception and design of the study. MK, NG, IK, TS, MT, TU, TT, and RH contributed to the acquisition of data. MK and YA analyzed and interpreted data, and drafted the manuscript. All authors read and approved the final manuscript.
Funding
This study was supported by a Grant-in-Aid for Young Scientists (A) 17H05097 from the Japan Society for the Promotion of Science and a grant from the Nakatomi Foundation.
Availability of data and materials
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Makris EA, Gomoll AH, Malizos KN, Hu JC, Athanasiou KA. Repair and tissue engineering techniques for articular cartilage. Nat Rev Rheumatol. 2015;11(1):21–34. doi: 10.1038/nrrheum.2014.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hunziker EB, Lippuner K, Keel MJ, Shintani N. An educational review of cartilage repair: precepts & practice–myths & misconceptions–progress & prospects. Osteoarthritis Cartilage. 2015;23(3):334–350. doi: 10.1016/j.joca.2014.12.011. [DOI] [PubMed] [Google Scholar]
- 3.Filardo G, Perdisa F, Roffi A, Marcacci M, Kon E. Stem cells in articular cartilage regeneration. J Orthop Surg Res. 2016;11:42. doi: 10.1186/s13018-016-0378-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Torres-Torrillas M, Rubio M, Damia E, et al. Adipose-Derived Mesenchymal Stem Cells: A Promising Tool in the Treatment of Musculoskeletal Diseases. Int J Mol Sci. 2019;20(12):3105. [DOI] [PMC free article] [PubMed]
- 5.Johnstone B, Hering TM, Caplan AI, Goldberg VM, Yoo JU. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998;238(1):265–272. doi: 10.1006/excr.1997.3858. [DOI] [PubMed] [Google Scholar]
- 6.Vater C, Kasten P, Stiehler M. Culture media for the differentiation of mesenchymal stromal cells. Acta Biomater. 2011;7(2):463–477. doi: 10.1016/j.actbio.2010.07.037. [DOI] [PubMed] [Google Scholar]
- 7.Kwon H, Paschos NK, Hu JC, Athanasiou K. Articular cartilage tissue engineering: the role of signaling molecules. Cell Mol Life Sci. 2016;73(6):1173–1194. doi: 10.1007/s00018-015-2115-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bakker AC, van de Loo FA, van Beuningen HM, et al. Overexpression of active TGF-beta-1 in the murine knee joint: evidence for synovial-layer-dependent chondro-osteophyte formation. Osteoarthritis Cartilage. 2001;9(2):128–136. doi: 10.1053/joca.2000.0368. [DOI] [PubMed] [Google Scholar]
- 9.Blaney Davidson EN, Vitters EL, van Beuningen HM, van de Loo FA, van den Berg WB, van der Kraan PM. Resemblance of osteophytes in experimental osteoarthritis to transforming growth factor beta-induced osteophytes: limited role of bone morphogenetic protein in early osteoarthritic osteophyte formation. Arthritis Rheum. 2007;56(12):4065–4073. doi: 10.1002/art.23034. [DOI] [PubMed] [Google Scholar]
- 10.Yeh TT, Wu SS, Lee CH, et al. The short-term therapeutic effect of recombinant human bone morphogenetic protein-2 on collagenase-induced lumbar facet joint osteoarthritis in rats. Osteoarthritis Cartilage. 2007;15(12):1357–1366. doi: 10.1016/j.joca.2007.04.019. [DOI] [PubMed] [Google Scholar]
- 11.Kwon HJ, Lee GS, Chun H. Electrical stimulation drives chondrogenesis of mesenchymal stem cells in the absence of exogenous growth factors. Sci Rep. 2016;6:39302. doi: 10.1038/srep39302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shepherd J, Cobbe SM, Ford I, et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group. N Engl J Med. 1995;333(20):1301–7. doi: 10.1056/NEJM199511163332001. [DOI] [PubMed] [Google Scholar]
- 13.Bedi O, Dhawan V, Sharma PL, Kumar P. Pleiotropic effects of statins: new therapeutic targets in drug design. Naunyn Schmiedebergs Arch Pharmacol. 2016;389(7):695–712. doi: 10.1007/s00210-016-1252-4. [DOI] [PubMed] [Google Scholar]
- 14.Mundy G, Garrett R, Harris S, et al. Stimulation of bone formation in vitro and in rodents by statins. Science. 1999;286(5446):1946–1949. doi: 10.1126/science.286.5446.1946. [DOI] [PubMed] [Google Scholar]
- 15.Benoit DS, Nuttelman CR, Collins SD, Anseth KS. Synthesis and characterization of a fluvastatin-releasing hydrogel delivery system to modulate hMSC differentiation and function for bone regeneration. Biomaterials. 2006;27(36):6102–6110. doi: 10.1016/j.biomaterials.2006.06.031. [DOI] [PubMed] [Google Scholar]
- 16.Hatano H, Maruo A, Bolander ME, Sarkar G. Statin stimulates bone morphogenetic protein-2, aggrecan, and type 2 collagen gene expression and proteoglycan synthesis in rat chondrocytes. J Orthop Sci. 2003;8(6):842–848. doi: 10.1007/s00776-003-0724-9. [DOI] [PubMed] [Google Scholar]
- 17.Terabe K, Takahashi N, Cobb M, Askew EB, Knudson CB, Knudson W. Simvastatin promotes restoration of chondrocyte morphology and phenotype. Arch Biochem Biophys. 2019;665:1–11. doi: 10.1016/j.abb.2019.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang H, Lin CY. Simvastatin stimulates chondrogenic phenotype of intervertebral disc cells partially through BMP-2 pathway. Spine. 2008;33(16):E525–31. doi: 10.1097/BRS.0b013e31817c561b. [DOI] [PubMed] [Google Scholar]
- 19.Emmanuele L, Ortmann J, Doerflinger T, Traupe T, Barton M. Lovastatin stimulates human vascular smooth muscle cell expression of bone morphogenetic protein-2, a potent inhibitor of low-density lipoprotein-stimulated cell growth. Biochem Biophys Res Commun. 2003;302(1):67–72. doi: 10.1016/S0006-291X(03)00109-8. [DOI] [PubMed] [Google Scholar]
- 20.Kodach LL, Bleuming SA, Peppelenbosch MP, Hommes DW, van den Brink GR, Hardwick JC. The effect of statins in colorectal cancer is mediated through the bone morphogenetic protein pathway. Gastroenterology. 2007;133(4):1272–1281. doi: 10.1053/j.gastro.2007.08.021. [DOI] [PubMed] [Google Scholar]
- 21.Demoor M, Ollitrault D, Gomez-Leduc T, et al. Cartilage tissue engineering: molecular control of chondrocyte differentiation for proper cartilage matrix reconstruction. Biochim Biophys Acta. 2014;1840(8):2414–2440. doi: 10.1016/j.bbagen.2014.02.030. [DOI] [PubMed] [Google Scholar]
- 22.Deng ZH, Li YS, Gao X, Lei GH, Huard J. Bone morphogenetic proteins for articular cartilage regeneration. Osteoarthritis Cartilage. 2018;26(9):1153–1161. doi: 10.1016/j.joca.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 23.Schmitt B, Ringe J, Haupl T, et al. BMP2 initiates chondrogenic lineage development of adult human mesenchymal stem cells in high-density culture. Differentiation. 2003;71(9–10):567–577. doi: 10.1111/j.1432-0436.2003.07109003.x. [DOI] [PubMed] [Google Scholar]
- 24.Park Y, Sugimoto M, Watrin A, Chiquet M, Hunziker EB. BMP-2 induces the expression of chondrocyte-specific genes in bovine synovium-derived progenitor cells cultured in three-dimensional alginate hydrogel. Osteoarthritis Cartilage. 2005;13(6):527–536. doi: 10.1016/j.joca.2005.02.006. [DOI] [PubMed] [Google Scholar]
- 25.Sekiya I, Larson BL, Vuoristo JT, Reger RL, Prockop DJ. Comparison of effect of BMP-2, -4, and -6 on in vitro cartilage formation of human adult stem cells from bone marrow stroma. Cell Tissue Res. 2005;320(2):269–276. doi: 10.1007/s00441-004-1075-3. [DOI] [PubMed] [Google Scholar]
- 26.Shirasawa S, Sekiya I, Sakaguchi Y, Yagishita K, Ichinose S, Muneta T. In vitro chondrogenesis of human synovium-derived mesenchymal stem cells: optimal condition and comparison with bone marrow-derived cells. J Cell Biochem. 2006;97(1):84–97. doi: 10.1002/jcb.20546. [DOI] [PubMed] [Google Scholar]
- 27.Mehlhorn AT, Niemeyer P, Kaschte K, et al. Differential effects of BMP-2 and TGF-beta1 on chondrogenic differentiation of adipose derived stem cells. Cell Prolif. 2007;40(6):809–823. doi: 10.1111/j.1365-2184.2007.00473.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McTaggart SJ. Isoprenylated proteins. Cell Mol Life Sci. 2006;63(3):255–267. doi: 10.1007/s00018-005-5298-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ishikawa M, Ishii T, Morikawa T, Iijima Y, Sueishi K. The Effects of Fluvastatin on Indian Hedgehog Pathway in Endochondral Ossification. Cartilage. 2021;13(2_suppl):304S–314S. [DOI] [PMC free article] [PubMed]
- 30.Yamashita A, Morioka M, Kishi H, et al. Statin treatment rescues FGFR3 skeletal dysplasia phenotypes. Nature. 2014;513(7519):507–511. doi: 10.1038/nature13775. [DOI] [PubMed] [Google Scholar]
- 31.Kenyon JD, Sergeeva O, Somoza RA, et al. Analysis of -5p and -3p Strands of miR-145 and miR-140 During Mesenchymal Stem Cell Chondrogenic Differentiation. Tissue Eng Part A. 2019;25(1–2):80–90. doi: 10.1089/ten.tea.2017.0440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhao YH, Lv X, Liu YL, et al. Hydrostatic pressure promotes the proliferation and osteogenic/chondrogenic differentiation of mesenchymal stem cells: The roles of RhoA and Rac1. Stem Cell Res. 2015;14(3):283–296. doi: 10.1016/j.scr.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 33.Wang KC, Egelhoff TT, Caplan AI, Welter JF, Baskaran H. ROCK Inhibition Promotes the Development of Chondrogenic Tissue by Improved Mass Transport. Tissue Eng Part A. 2018;24(15–16):1218–1227. doi: 10.1089/ten.tea.2017.0438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gegg C, Yang F. The Effects of ROCK Inhibition on Mesenchymal Stem Cell Chondrogenesis Are Culture Model Dependent. Tissue Eng Part A. 2020;26(3–4):130–139. doi: 10.1089/ten.tea.2019.0068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Almalki SG, Agrawal DK. Key transcription factors in the differentiation of mesenchymal stem cells. Differentiation. 2016;92(1–2):41–51. doi: 10.1016/j.diff.2016.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shen H, Lin H, Sun AX, et al. Chondroinductive factor-free chondrogenic differentiation of human mesenchymal stem cells in graphene oxide-incorporated hydrogels. J Mater Chem B. 2018;6(6):908–917. doi: 10.1039/C7TB02172K. [DOI] [PubMed] [Google Scholar]
- 37.Pan Q, Yu Y, Chen Q, et al. Sox9, a key transcription factor of bone morphogenetic protein-2-induced chondrogenesis, is activated through BMP pathway and a CCAAT box in the proximal promoter. J Cell Physiol. 2008;217(1):228–241. doi: 10.1002/jcp.21496. [DOI] [PubMed] [Google Scholar]
- 38.Sahu N, Budhiraja G, Subramanian A. Preconditioning of mesenchymal stromal cells with low-intensity ultrasound: influence on chondrogenesis and directed SOX9 signaling pathways. Stem Cell Res Ther. 2020;11(1):6. doi: 10.1186/s13287-019-1532-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Louneva N, Huaman G, Fertala J, Jimenez SA. Inhibition of systemic sclerosis dermal fibroblast type I collagen production and gene expression by simvastatin. Arthritis Rheum. 2006;54(4):1298–1308. doi: 10.1002/art.21723. [DOI] [PubMed] [Google Scholar]
- 40.Yanae M, Tsubaki M, Satou T, et al. Statin-induced apoptosis via the suppression of ERK1/2 and Akt activation by inhibition of the geranylgeranyl-pyrophosphate biosynthesis in glioblastoma. J Exp Clin Cancer Res. 2011;30:74. doi: 10.1186/1756-9966-30-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sorrentino G, Ruggeri N, Specchia V, et al. Metabolic control of YAP and TAZ by the mevalonate pathway. Nat Cell Biol. 2014;16(4):357–366. doi: 10.1038/ncb2936. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Fig. S1 The original uncropped blot in Figure 5A. Black-boxed areas in the original blot were included in the main paper.
Data Availability Statement
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.