Abstract
Including health outcomes for carers as well as patients in economic evaluations can change the results and conclusions of the analysis. Whilst in many disease areas there can be clear justification for including carers’ health-related quality of life (HRQL) in health technology assessments (HTAs), we believe that, in general, the perspective of carers is under-represented in HTA. We were interested in the extent, and methods by which, HTA bodies include carers’ HRQL in economic evaluation. We reviewed guidance from 13 HTA bodies across the world regarding carers’ HRQL. We examined five interventions, as case studies, assessed by different HTA bodies, and extracted information on whether carers’ HRQL was included by the manufacturers or assessors in their dossiers of evidence, the data and methods used, and the impact on the results. We developed recommendations to guide analysts on including carers’ HRQL in economic evaluations. When reviewing the methods guides: two bodies recommend including carers’ HRQL in the base case, two referred to outcomes for all individuals, two preferred to exclude carers, three said it depended on other conditions, and it was unclear for four. Across the five case studies: five source studies for carers’ HRQL and two different modelling approaches were used. Including carers’ HRQL increased incremental quality-adjusted life-years (QALYs) in 19/23 analyses (decreased it in two); there was substantial variation in the magnitude of change. We recommend: (1) the inclusion of carers is clearly justified, (2) the use of HRQL data from the population under comparison where possible, (3) the use of data from another disease area or country is clearly justified (and transferability/applicability issues are discussed), (4) the use of external data to derive comparisons for cross-sectional data is justified, (5) assumptions and implications of the modelling approach are explicit, and (6) disaggregated results for patients and carers are presented.
Key Points
| Health technology assessment (HTA) bodies globally take different views on whether carers’ health-related quality of life (HRQL) should be included in economic evaluation. |
| A variety of methods and data have been considered by HTA bodies across appraisals for including carers’ HRQL. |
| We provide six recommendations for including carers’ HRQL in economic evaluation. |
Introduction
The relationship between the health/wellbeing of carers, the health of the patient they care for, and the volume of care provided has long been established [1, 2]. It is therefore reasonable to expect that healthcare interventions that change patients’ health or the volume of informal (unpaid) care required will also affect health outcomes for carers [3]. This impact is particularly notable where informal carers provide a high volume of care, such as parents providing round-the-clock care for a child with a long-term health condition, or a spouse living with a person with a neurological condition. The inclusion of these changes in carers’ health-related quality of life (HRQL) (sometimes termed a ‘spillover’ effect) in addition to patients’ HRQL in economic evaluations may, as a result, change the incremental quality-adjusted life-years (QALYs) associated with an intervention. This in turn would impact the resulting cost-effectiveness, and may change the eventual decision to adopt (or not adopt) the technology. Indeed, a recent review of paediatric economic evaluations found that in four cases, including carers’ HRQL reduced the incremental cost-effectiveness ratio (ICER) [4], and a review of National Institute for Health and Care Excellence (NICE) appraisals found that including carers’ HRQL increased incremental QALYs or decreased ICERs in 14 cases [5].
While some economists argue that the inclusion of carers’ HRQL in economic evaluations is essential to ensure efficiency and equity [6], others argue that including it may lead to unintended effects on the distribution of health and access to healthcare. The ‘unintended effects’ argument states that spillover effects are likely to be affected by socioeconomic status for both the interventions being assessed and those being displaced, which may lead to further inequity [7]. In practice, local health technology assessment (HTA) guidance dictates whether carers’ HRQL can be included in economic evaluations. However, even where HTA bodies do recommend the inclusion of carers’ HRQL, it is also noted that not all assessments will include carers. For example, the previously mentioned review of NICE appraisals found that only 12 of 414 technology appraisals included carers’ HRQL [5].
Based on these factors, the objective of this work was to understand whether guidance provided by various international HTA bodies recommended the inclusion of carers’ HRQL. In addition to whether carer HRQL was included, we aimed to understand the methodological approach taken, and resulting impact on the economic evaluation across HTA bodies.
Methods
Guidance
We reviewed guidance on economic evaluations from 13 international HTA bodies whose guidance on methods was available in English online. These 13 bodies reflected the authors’ combined experiences of HTA assessment globally and represent a range of healthcare systems and reimbursement processes:
Agenzia Italiana Del Farmaco (AIFA), Italy.
All Wales Medicine Strategy Group (AWMSG), Wales.
Canadian Agency for Drugs and Technologies in Health (CADTH), Canada.
Haute Autorité de Santé (HAS), France.
Institute for Clinical and Economic Reviews (ICER), the USA.
Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen (IQWIG), Germany.
National Institute for Health and Care Excellence (NICE), England.
Tandvårds- och läkemedelsförmånsverket (TLV), Sweden.
Pharmac, New Zealand.
Pharmaceutical Benefits Advisory Committee (PBAC), Australia.
Scottish Medicines Consortium (SMC), Scotland.
The National Centre for PharmacoEconomics (NCPE), using guidance from the Health Information and Quality Authority (HIQA), Ireland.
Zorginstituut Nederlands (ZiN), the Netherlands.
We extracted information relating to the inclusion of benefits or outcomes for carers/caregivers.
Case Studies
We aimed to identify five case studies where multiple HTA bodies had assessed the same intervention and at least one body had included carers’ HRQL. We believed that five individual interventions would give a good overview of some of the issues and differences between HTA bodies (allowing for trends to emerge), whilst being achievable given the small number of assessments that have historically included carers’ HRQL. We began by identifying NICE assessments in the last 5 years where carers’ HRQL was included. We started with NICE as we were aware, from previous research, that the inclusion of carers’ HRQL is relatively uncommon; however, NICE recommends the inclusion of carers’ HRQL ‘where relevant’ [5].
We searched the NICE website for assessments in disease areas where the literature suggested carer HRQL had been included in economic evaluations, or where there was anticipated to be a particularly notable carer burden. These disease areas were:
Meningitis [11].
Multiple sclerosis [12].
Paediatric diseases [4] including asthma, juvenile arthritis, cystic fibrosis.
Parkinson’s disease [9].
Rare/orphan diseases [5] using NICE’s Highly Specialised Technology (HST) programme.
Spinal muscular atrophy [13].
Stroke [14].
Where we identified NICE assessments with final guidance published, we searched for assessments of the same intervention by the other 11 HTA bodies.
Different HTA bodies follow different processes for evaluating the cost-effectiveness of interventions. For example, companies submit evidence to NICE, which is then reviewed by an independent assessment group (the Evidence Review Group (ERG)) before it is discussed by the committee—there is consequently information on the company analysis, the ERG analysis, and the committee’s preferred assumptions. Similarly, TLV and CADTH base their assessments on company submissions (making changes, where deemed appropriate). Other bodies may only review evidence submitted by the company (e.g., SMC and PBAC) or undertake their own assessment (e.g., ZiN and ICER). As a result, there are different analyses and levels of detail available from the different bodies.
We reviewed the available documentation and extracted information on:
Whether carers’ HRQL was included by either the company or the HTA body.
The methods used to include carers’ HRQL in any analysis.
The data used to include carers’ HRQL in any analysis.
The effect of including carers’ HRQL on the incremental QALYs in any analysis.
Whether the HTA body felt that carers’ HRQL should be included in the base case, sensitivity analysis, or not at all.
Where possible, we read guidance and assessment information published in English. We searched for the terms ‘carer’ and ‘caregiver’ within the text, and read the utility/HRQL and results sections of documents. Where English language documentation was not available, we used Google Translate to translate text from the utility/HRQL and results sections into English to identify where carers’ HRQL was included. For detailed translations (where required), assistance was requested from bilingual colleagues.
Results
Guidance
Table 1 presents a summary of the guidance on the inclusion of carers’ HRQL in economic evaluation from global HTA bodies. NICE, HAS, HIQA and ZiN were all clear in indicating carers’ HRQL should be included in the reference case analysis, and PBAC and PHARMAC made it clear that carers’ HRQL should not be included. In other cases, the HTA body stated conditions under which they would include carers’ HRQL, for example CADTH state carers should be included only if the target population for the intervention is both patient and carers, otherwise carers should be included in the sensitivity analysis only [15]; for IQWIG it depends on the perspective in the commission [16]; and ICER states that carers can be included in the modified societal co-base case [17]. The guidance was unclear in the remainder of cases: TLV state that a societal perspective should be used but this appears focussed on costs rather than outcomes [18]; and SMC state that outcomes should be all direct health effects for patients or other parties (principally carers), but then later states that evidence on carer QALYs should be presented in a separate analysis as it is outside SMC’s perspective [19]. Guidance from AWMSG and AIFA did not mention carers [20, 21].
Table 1.
Summary of global health technology assessment guidance on the inclusion of carers' health-related quality of life (HRQL)
| Guidance on carers’ HRQL | Institute | Country | Section | References | Quotation |
|---|---|---|---|---|---|
| Specifically states carer outcomes should be included in reference case analysis (where relevant) | National Institute for Health and Care Excellence (NICE) | England and Wales* | 4.3.1.7 | [22] | “Evaluations should consider all health effects for patients, and, when relevant, carers. When presenting health effects for carers, evidence should be provided to show that the condition is associated with a substantial effect on carer's health-related quality of life and how the technology affects carers.” |
| Haute Autorité de Santé (HAS) | France | 1.3 | [23] | “The evaluation of health outcomes identifies the relevant health effects from the point of view of the populations concerned (i.e., patients, healthcare system users and informal caregivers under a collective perspective…)” | |
| Refers to including outcomes for all individuals in reference case analysis | Health Information and Quality Authority (HIQA) | Ireland | 2.3 | [24] | “All health benefits accruing to individuals should be included in the assessment of outcomes.” |
| Zorginstituut Nederland (ZiN) | Netherlands | 1.2 | [25] | “All relevant societal costs and benefits, irrespective of who bears the cost or to who the benefits go, should therefore be taken into account in the evaluation and reporting.” | |
| Inclusion of carer outcomes depends on other factors | Canadian Agency for Drugs and Technologies in Health (CADTH) | Canada |
3 5 10 |
[15] | “Based on the target population specified in the decision problem, researchers should consider any potential spillover impact…any associated spillover beyond the target population(s)…should be addressed in a non-reference case analysis.” |
| Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen (IQWIG) | Germany |
3.1.1 4.1.2 |
[16] | “Interventions can also have consequences for those indirectly affected, for example relatives and carers. If appropriate, these consequences can also be considered within the framework of the Institute’s reports.” | |
| Institute for Clinical and Economic Reviews (ICER) | USA |
3.5 3.10 |
[17] | “Specific scenario analyses (including one using a modified societal perspective that incorporates estimates such as … caregiver burden)… are conducted when appropriate.” | |
| Unclear | Scottish Medicines Consortium (SMC) | Scotland |
6.7.3 6.9 |
[19] |
“The perspective on outcomes should be all direct health effects whether for patients, or, where relevant, other individuals (principally carers).” “If appropriate data on utilities/QALYs for carers or other groups….it is outside the perspective adopted by the SMC.” |
| Tandvårds- och läkemedelsförmånsverket (TLV) | Sweden | Health Economics | [18] | “A societal perspective also includes costs and effects outside the heath care sector. In TLV’s health economic evaluations a societal perspective is applied.” | |
| All Wales Medicines Strategy Group (AWMSG) | Wales* | Not mentioned | [20] | ||
| Agenzia Italiana Del Farmaco (AIFA) | Italy | Not mentioned | [21] | ||
| Specifically states carer outcomes should not be included in reference case analysis | Pharmaceutical Benefits Advisory Committee (PBAC) | Australia | 3A 1.4 | [26] |
“The PBAC’s preferred health care system perspective includes health…and health-related outcomes…outcomes are those associated with the patient.” “…where the beneficiaries of health or other relevant outcomes are broader than the treated patient population (e.g., community, carers, dependants), include these are supplementary analyses.” |
| Pharmac | New Zealand | 6.1.2 | [27] | “It is recommended that only the health-related quality of life (HR-QoL) of the patient being treated should be included in the base-case analysis. If the treatment might have a measurable but indirect impact on the HR-QoL of others, such as family and caregivers, this could be estimated and discussed in the report as a scenario.” |
*Medicines are funded by NHS Wales following guidance from NICE and AWMSG: AWMSG considers NICE’s future work programme when determining whether to appraise a medicine and AWMSG guidance is interim to NICE guidance
Case Studies
Recent cases studies with assessments by multiple HTA bodies were focussed predominantly in multiple sclerosis and rare/orphan diseases. We did not identify suitable case studies in either dementia or stroke because there were no recent assessments of interventions. While there was a recent ICER assessment in Alzheimer’s (aducanumab) [28], this intervention was not assessed by other HTA bodies (we note that the NICE appraisal was suspended because the European Medicines Agency’s Committee for Medicinal Products for Human Use adopted a negative opinion, recommending the refusal of the marketing authorisation [29]). We identified five case studies with recent assessments conducted by NICE and at least one other HTA body at the time of review:
Elosulfase alfa for mucopolysaccharidosis type IV (an enzyme replacement therapy to delay progression of a rare disease that causes tissue damage).
Ocrelizumab for (relapsing remitting) multiple sclerosis (an intravenous infusion to treat a chronic, disabling neurological condition).
Nusinersen for spinal muscular atrophy (an intrathecal injection to improve survival and motor function in people with a rare, progressive neuromuscular disorder).
Patisiran for hereditary transthyretin amyloidosis (a disease-modifying intravenous infusion for a rare multi-system condition).
Voretigene neparvovec for retinal disease (an injection in the eye to improve visual function and functional vision in a rare genetic eye disease).
Not every HTA body appraised all the interventions in our case studies. For example, the AWMSG referred to NICE guidance for elosulfase alfa, and had not appraised any of the other case studies. IQWIG had appraised each of the case studies but had not considered health economics evidence for any of them. AIFA had not conducted a health economics assessment for any of the case studies. An overview of the five case studies for the remaining HTA bodies is shown in Table 2.
Table 2.
Overview of inclusion of carers' health-related quality of life (HRQL)
| Carers: | CADTH | HAS | ICER | NICE | NCPE | PBAC | Pharmac | SMC | TLV | ZiN | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Elosulfase alfa | Includeda | Scenario analysis [30] | No published assessment | No published assessment | Base case[31] | Not mentioned [32] | Not mentioned [33] | No published assessment | Not mentioned [34] | Scenario analysis [35] | No published assessment |
| Methodb | Disutilities | Disutilities | Disutilities | ||||||||
| Numberc | NR | 1 | 1 | ||||||||
| Nusinersen | Includeda | Not mentioned [36] | Not mentioned [37] | Thinking presented, excluded in analysis [38] | Base case[39] [40] [41] | No published assessment | Not mentioned [42] | Not mentioned [43] | Scenario analysis [44] | Scenario analysis [45] | Scenario analysis [46] |
| Methodb | Disutilities | Disutilities | NR | Utilities | Utilities | ||||||
| Numberc | 2 |
Early onset: 3 Late onset: 2 (3 in worst) |
NR | 1 | 1 | ||||||
| Ocrelizumab | Includeda | Scenario analysis [47] | No published assessment | Not mentioned [48] | Base case [49] [50] [51] | Not mentioned [52] | Not mentioned [53] | No published assessment | Not mentioned [54] | Scenario analysis [55] | No published assessment |
| Methodb | Disutilities | Disutilities | NR | ||||||||
| Numberc | NR | 1 | NR | ||||||||
| Patisiran | Includeda | Excluded [56] | No published assessment | Scenario analysis [57] | Base case [58] [59, 60] | Not mentioned [61] | No published assessment | No published assessment | Not mentioned[62] | Scenario analysis [63] | No published assessment |
| Methodb | Disutilities | Disutilities | Disutilities | ||||||||
| Numberc | NR |
Stage 1-2: 1 Stage 3: 2 |
1 | ||||||||
| Voretigene neparvovec | Includeda | Scenario analysis [64] | No published assessment | Not mentioned [65] | Base case [66] [67] [68] | Not mentioned [69] | No published assessment | No published assessment | Base case [70] | Scenario analysis [71] | Scenario analysis [72] |
| Methodb | NR | Disutilities | NR | Disutilities | Disutilities | ||||||
| Numberc | NR |
Company: children: 1, adults: 0.5 ERG: children 1.78, adults: 0 |
NR | NR |
Children: 1 Adults: 0.5 |
CADTH Canadian Agency for Drugs and Technologies in Health, ERG evidence review group, HAS: Haute Autorité de Santé, ICER Institute for Clinical and Economic Reviews, NCPE National Centre for PharmacoEconomics, NICE National Institute for Health and Care Excellence, NR not reported, PBAC Pharmaceutical Benefits Advisory Committee, SMC Scottish Medicines Consortium, TLV Tandvårds- och läkemedelsförmånsverket, ZiN Zorginstituut Nederlands
aRefers to whether carers’ HRQL was included in the model. Base case/scenario analysis/excluded refers to HTA body’s final analysis and not company submission
bRefers to the method used for including carers’ HRQL in the model. Please refer to Sect. 4.1 in the text for further information
cRefers to the number of carers whose HRQL was included in the model
Notably, despite covering a range of technologies and diseases across multiple jurisdictions, carer utilities were derived from only four sources (Table 3). References were not provided for the carer utilities included in the TLV and ZiN appraisals of nusinersen, but based on the values (0.85) we may expect these correspond to population norms (average utilities for the general population of the same age as the carer).
Table 3.
Studies used as data for carers' health-related quality of life (HRQL)
| Study | Population | Utility instrument | Country | Values reported | Used in case studies |
|---|---|---|---|---|---|
| Gani et al. (2008) [12] |
Modelled carers of people with multiple sclerosis Values from Neumann et al (1999) study of 679 carers of Alzheimer’s patients [71] |
Health Utilities Index: 2 | USA |
Gani et al (2008) report carer disutilities ranging from 0.00 to 0.14 Neumann et al (1999) report a minimum carer utility of 0.86 |
Elosulfase alfa: NICE elosulfase alfa: TLV Ocrelizumab: NICE patisiran: NICE Patisiran: TLV |
| Kuhlthau et al. (2010) [72] | 2412 parents of children with an activity limitation, compared to 13,560 parents of children without an activity limitation | EQ-5D (US Value Set) | USA |
Unadjusted utility scores: 0.82 where child has activity limitation, 0.9 otherwise. Regression-adjusted difference: − 0.07 |
Voretigene neparvovec: NICE (company) Voretigene neparvovec: TLV Voretigene neparvovec: ZiN |
| Al-Janabi et al. (2016) [73] | 1053 family members close to a person with after-effects of meningitis, compared to 517 family members close to a person with no after-effects | EQ-5D-5L, scored using interim UK value set | UK | Mean difference in utility between family members of person with after effects and family members of person without: 0.041 | Voretigene neparvovec: NICE (ERG) |
| Lopez-Bastida et al. (2017) [13] | 81 caregivers of people with spinal muscular atrophy | EQ-5D-5L (value set unclear) | Spain | Mean utility: 0.484 (0.472 for 60 caregivers of Type II patients) |
Nusinersen: NICE Nusinersen: ICER |
ERG evidence review group, ICER Institute for Clinical and Economic Reviews, NICE National Institute for Health and Care Excellence, NR not reported, TLV Tandvårds- och läkemedelsförmånsverket, ZiN Zorginstituut Nederlands
Impact of Including Carers’ HRQL on Results
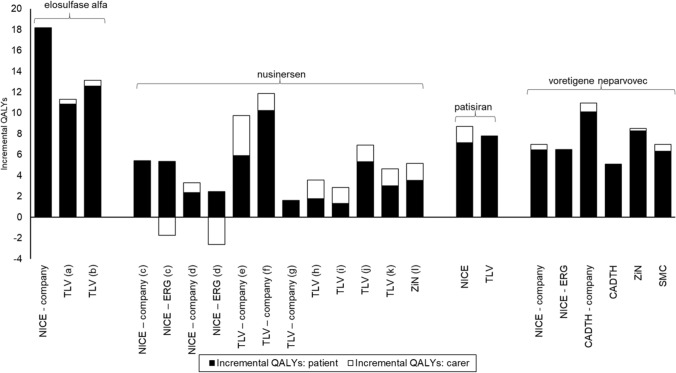
Figure 1 shows the incremental QALYs for patients and carers for each assessment (no results were available for ocrelizumab). The size of the incremental QALYs for carers varies between assessments, ranging from no change and negligible gains to gains almost as large as those for the patient (e.g., TLV company (e) SMA type I, where incremental carer QALYs are 3.83 and incremental patient QALYs are 5.93). In the nusinersen case study the incremental QALY gains for carers are greater for the TLV and ZiN scenarios, which consider carer utilities, than the NICE scenarios which consider carer disutilities. In the patisiran case study, the NICE and TLV analyses generate similar incremental patient QALYs, but the incremental carer QALYs are greater for the NICE analysis, despite using the same source data for carer disutilities.
Fig. 1.
Impact of including carers' HRQL on incremental QALYs. a Starting treatment aged 18 years; b starting treatment aged 3 years; c early onset; d late onset; e spinal muscular atrophy (SMA) type I; f SMA type II, g SMA type III; h SMA type Ia; i SMA type Ib; j SMA type IIa; k SMA type IIb; l SMA type II/I
In most cases, the incremental carer QALYs are positive, so there is a benefit to carers from the intervention. The exceptions to this are the NICE ERG analyses for nusinersen, where improving patient survival had a negative impact for carers. This finding is due to the use of carer disutilities linked to patient health states and may occur where the gain in carer QALYs from improving patient’s health (better patient health states are associated with smaller carer disutilities) is less than the loss in carer QALYs from the patient living longer (carer disutilities accrue over a longer period). Whether this finding occurs depends on the variation in carer disutilities associated with patient health states; the relationship between improvement in quality of life/delay in disease progression, any extension of life of the patient; and whether a carer disutility for bereavement is included (and the magnitude of this disutility). Unfortunately, the values used by the NICE ERG are redacted, preventing further analysis. By contrast, when carer utilities are included (as in the TLV and ZiN scenarios), improving patient survival will always result in a QALY gain for carers.
Discussion
HTA bodies globally take different views on whether carers’ HRQL should be included in economic evaluation. They also have differing preferences for how carers’ HRQL should be included in terms of the methods and data to do this. Although this is not unexpected given the general variation in HTA methods globally, the variety in preferences for (and means of assessing) carer HRQL could further increase inequality of access to treatments globally.
Perhaps more surprising is the inconsistency seen in appraisals by the same HTA bodies when appraising different interventions. Examples include the inclusion of carers’ HRQL in the base case for voretigene neparvovec at the SMC but not for other interventions, or the TLV using carer utilities for nusinersen and carer disutilities for voretigene neparvovec, patisiran and elosulfase alfa. Even in the case of NICE, who allowed the inclusion of carer disutilities for all five case studies, the number of carers differed between appraisals, and the level of critique applied to the same data source [12], varied substantially between appraisals.
Carer Utilities or Carer Disutilities
The case studies used two fundamentally different approaches for including carers’ HRQL: carer utilities or carer disutilities. The more common disutility approach considers the detrimental impact of caring on HRQL, and the extent to which this is relieved or exacerbated when the patient’s health changes. The interpretation of this approach appears problematic when considering the HRQL of the carer when the patient dies: removing the disutility implies that the carers’ HRQL improves when the patient dies. Further to this there is limited data to provide empirical evidence for changes in carers’ HRQL over time when patients die, although a bereavement effect has been observed in at least one study [73]. Furthermore, this assumption seems at odds with patients’ and carers’ preferences (they would prefer patients did not die), and raises equity issues of how patients and carers should be valued where an intervention can lead to gains for one and a loss for the other. The utility approach used in the TLV and ZiN nusinersen appraisals seemingly solves this issue by including carers as a separate entity, whose HRQL is included as an additional utility that can change as patients’ health changes (although it is unclear whether the utility does change in the nusinersen examples as no details were provided). However, in this scenario it is unclear whether, and how, carers should still be included in the model once the patient dies, and what the normative interpretation of this is—to maintain consistency across arms, it would seem most logical that the time frame should be extended out to include the carers’ lifetime.
There does not appear to be an obvious answer as to which approach is superior, and there is a surprising lack of transparency around the assumptions within the approaches. While the NICE committee discussion for nusinersen notes that the patient experts found it ‘perverse’ that including carers’ HRQL could make a life-extending treatment less cost-effective [41], the carer disutility approach has been used historically in multiple sclerosis appraisals, since at least 2008 [12], seemingly without considering its limitations. ICER note that the disutility approach can lead to ‘counter-intuitive’ findings but do not propose a better method, instead preferring to use a qualitative approach to consider carers [17].
Sources of Evidence
The case studies highlight the paucity of evidence of carers’ HRQL, with many case studies utilising data from other disease areas. In some cases, the HTA bodies appear to accept this with the justification that precedence has been set in previous appraisals, but in other cases the same data were critiqued as not being robust. For example, the study by Gani et al. [12] was used in NICE appraisals for elosulfase alfa and patisiran, but in the ocrelizumab appraisal the ERG referred to a previous assessment in multiple sclerosis where this was criticised as it assumes that the HRQL of carers would increase from 0.86 to 1.00 (perfect health) if they did not have to care for people with dementia, whereas carers of people with dementia will often be quite old, and age-adjusted population norms may be similar to 0.86 for non-carers [74]. Similarly, TLV used this study in their appraisal of patisiran but critiqued it in their appraisal of elosulfase alfa for being from another disease area and not having a control arm [35].
A further inconsistency is in the way the same study is used to derive carer HRQL values: TLV included a disutility of 0.01 for the most severe state in their appraisal of patisiran [63], and a maximum disutility of 0.14 in their appraisal of elosulfase alfa [35], both referenced to the same study [12]. In the nusinersen appraisals, the carer utility values reported in the source study [13] were applied differently by the company (carer disutilities for different health states were estimated based on differences in patient utility between health states) [39] and the ERG (carer disutilities used directly from the source study) [40] in their NICE submission and by ICER (carer disutilities for different health states were estimated using patient utilities for some health states and interpolating between values) [38].
Lastly, in some appraisals the issue of transferability of evidence from other countries was raised in some appraisals, such as the NICE assessment of voretigene neparvovec, where the company used data from a US study [66], but the ERG [67] and NICE committee [68] preferred data from a study of UK carers, with utilities derived using a UK value set. However, the US study was used in the appraisals of voretigene by TLV [71] and ZiN [72], without discussion of its applicability.
Definition of Carers and How They are Affected
Although we specifically searched for the terms ‘carer’ and ‘caregiver’ within assessments, we read whole sections of documents to ensure any relevant information was extracted. In the TLV assessments, we included analyses where three different terms were used: ‘vårdgivare’, ‘vårdnadshavaren’, ‘anhöriga’, which translate as ‘caregiver’, ‘custodian’, and ‘relatives’, respectively (according to Google Translate). We included all of these within our analyses but note that they may have different interpretations and implications for whom is affected by the interventions and how. Bobinac et al. differentiated between the ‘caregiver effect’ and the ‘family effect’, noting that HRQL for people close to a patient may be affected by both the extent of caregiving duties as well as caring about a loved one [1, 2]. Furthermore, Al-Janabi et al. identified six mechanisms behind carer wellbeing effects: information, management of care, patient outcomes, alienation, compliance, and timing or location [76]. It is entirely feasible that different groups of people beyond the patient are affected differently by the same intervention, and so the comparisons between these assessments may not be consistent.
Limitations
Our review of guidelines was designed to be pragmatic and illustrative in identifying key issues, rather than to be exhaustive. As such, there are HTA bodies who we have not included here (particularly those without English language methods documentation) whose guidance may differ. Our selection of case studies was limited by the small number of assessments which have included carer HRQL. Some of the key disease areas where carer burden is observed are excluded here as there were no interventions assessed in these diseases. While a recent systematic review identified economic evaluations which included carers’ HRQL in a broader range of disease areas, the evaluations often compared different programmes of delivery of care [75], rather than drugs as typically assessed by HTA bodies.
Recommendations
Whilst recommending that analysts adhere to guidance from each individual body, we provide six recommendations for including carers’ HRQL in economic evaluation. These recommendations are informed by the issues we identified and may become less salient as the evidence base improves.
Recommendation 1: Clearly justify whether, and why, carers’ HRQL is included in the economic evaluation We believe evaluations should specify explicitly when carers’ HRQL is included, whether in the base case or sensitivity analysis, and the rationale for doing so. Furthermore, we encourage the provision of evidence that carers’ HRQL is of relevance to the intervention under consideration, such that all can understand and accept why it has been included. Consideration of the mechanisms by which carers’ HRQL is affected by the intervention could provide further rationale for its inclusion [76].
This issue emerged during our review of the assessments, particularly when comparing guidance from ICER and CADTH, whose sensitivity analysis using a ‘societal’ perspective includes carers’ HRQL, with TLV’s base case including the ‘societal’ perspective, which does not include carers’ HRQL. We note that the term ‘societal perspective’ can have different interpretations for whether this includes outcomes beyond the patient, and so may not be an effective description—similar to the term ‘cost-effective’ without reference to a threshold.
Analysts should also consider the number of carers’ whose HRQL is affected, noting that this ranged between one and three in the case studies, with the literature suggesting as many as eight people could be affected by spillover effects depending on the population and severity [77].
Recommendation 2: Where possible, provide evidence of carers’ HRQL for the population under consideration Ideally, carers’ HRQL data should be collected alongside patients’ HRQL data during the company’s evidence generation activities and meet the same standards of evidence. This means planning for collecting carers’ HRQL data in early-stage development of the interventions. Generating evidence specific to the disease area, intervention and country provides a more compelling case for including carers’ HRQL in economic evaluation.
Recommendation 3: If carers’ HRQL is informed by data from a different disease area and/or country, justify its use and discuss its limitations Analysts should state explicitly if carers’ HRQL data has been derived by data from another disease area and explain its relevance/applicability. Consideration should be given to differences and similarities in the carer burden between the source data and the intervention under consideration, in terms of both the volume and the type of care provided. For example, the heavy reliance on data from Alzheimer’s to model carers of paediatric patients would be questionable without a detailed explanation of the relevance of this data source.
In addition to considering differences between disease areas, we recommend consideration also be given to differences between carers’ HRQL data from different countries. Of note, formal care provision differs across healthcare systems, therefore informal care burden may not necessarily be the same across jurisdictions. Similarly, societal expectations may lead to differences in the norms (and thus deviations from these) in patient-reported outcomes. These factors should be considered in addition to the value set used to derive utilities.
Recommendation 4: Where cross-sectional data informs carers’ HRQL, justify the choice of external data used to derive comparisons Preferably, carers’ HRQL data would be collected at multiple time points to demonstrate how it changes (as patient health changes). However, where only cross-sectional data are available; for example, a survey of carers collected at one time point, analysts should carefully consider how to translate this into values for use in the economic evaluations. Assuming that carers’ HRQL would be equivalent to full health in the absence of any caring responsibilities is unlikely to be appropriate given the effects of ageing and comorbidities [78]. Comparison to age-adjusted population norms may present a more appropriate approach, reflecting that no group has a mean utility of 1.
Recommendation 5: Carefully consider the implications of the modelling approach and be explicit about the assumptions made Evaluations should explain how the modelling approach incorporates carers’ HRQL, and what this assumes about changes in carers’ HRQL as patients’ health changes. This should include an explanation of assumptions about whether carers’ HRQL is included in the evaluation throughout and beyond the patient’s lifetime. Justification should be provided for the chosen approach, and the limitations discussed. Analysts should not be constrained by previous methods to combine patients’ and carers’ HRQL but consider how this can be improved.
Recommendation 6: Present disaggregated results for patients and carers, using sensitivity analysis to include/exclude carers Presenting the results for the two groups separately allows for clear identification of where the costs and benefits accrue. Including sensitivity analysis excluding carers, or including them if they are excluded in the base case, allows the cost-effectiveness to be considered in circumstances where carers’ HRQL is deemed not to be relevant, or relevant, if excluded in the base case.
We found that the magnitude and direction of incremental QALYs for carers varied dramatically between assessments (Fig. 1), demonstrating that the impact of including carers’ HRQL cannot be estimated or understood without seeing the patient and carer QALYs separately. Equally, when only ‘healthcare’ versus ‘societal’ perspectives were considered, it was not possible to know whether the intervention was leading to improvements in carer quality of life, or reductions in costs, which are relevant both for decision makers, and prospective recipients of the technology.
Conclusions
Carers’ HRQL is inconsistently analysed and included globally in HTA reviews. The data used are often of limited quality, and the methods used to include carers in economic evaluations are not well developed. Given the potential impact of carers’ HRQL on cost-effectiveness results, it is imperative that data and methods are improved and consistently applied. Failure to do so results in economic evaluations that do not reflect the experience of patients and their carers, and the overall importance of carers in some of these disease areas, resulting in decision making that is inconsistent and likely to be sub-optimal. Whilst our recommendations are not wholly prescriptive regarding modelling approaches to include carers, they would improve the quality and transparency of how carers’ HRQL is included in HTA.
Acknowledgements
We are grateful to Jeanni van Loon and Paolo Cortesi for reviewing materials published in Dutch and Italian, respectively, and the team at Delta Hat for supporting in shaping the final manuscript.
Declarations
Funding
This study was funded by Takeda UK Ltd. Helen Taylor and Jack Eaton are employees of Takeda UK Ltd. Becky Pennington and Anthony J. Hatswell received fees from Takeda UK Ltd for undertaking the research. Article Processing Charges were paid for by Takeda UK Ltd. Becky Pennington is supported by the National Institute for Health Research (NIHR) Fellowship (NIHR300160). The views expressed in this publication are those of the authors and not necessarily those of the National Institute for Health Research or Takeda UK Ltd.
Conflict of interest
Becky Pennington is supported by an NIHR fellowship researching carers’ health-related quality of life.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
All material analysed during this review are referenced in this article and are available in the public domain from the websites of the Health Technology Assessment bodies.
Code availability
Not applicable.
Author’s contributions
All authors contributed to the study conception and design. Data extraction and analysis were performed by BP in close discussions with AJH and reviewed by JE and HT. The first draft of the manuscript was written by BP and all authors contributed to subsequent versions. All authors read and approved the final manuscript.
References
- 1.Bobinac A, van Exel NJ, Rutten FF, Brouwer WB. Caring for and caring about: disentangling the caregiver effect and the family effect. J Health Econ. 2010;29(4):549–556. doi: 10.1016/j.jhealeco.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 2.Bobinac A, van Exel NJ, Rutten FF, Brouwer WB. Health effects in significant others: separating family and care-giving effects. Med Decis Making. 2011 Mar-Apr;31(2):292-8. [DOI] [PubMed]
- 3.Al-Janabi H, Nicholls J, Oyebode J. The need to ‘carer proof’ healthcare decisions. BMJ. 2016;59:352. doi: 10.1136/bmj.i1651. [DOI] [PubMed] [Google Scholar]
- 4.Lavelle TA, D'Cruz BN, Mohit B, Ungar WJ, Prosser LA, Tsiplova K, et al. Family spillover effects in pediatric cost-utility analyses. Appl Health Econ Health Policy. 2019;17(2):163–174. doi: 10.1007/s40258-018-0436-0. [DOI] [PubMed] [Google Scholar]
- 5.Pennington BM. Inclusion of carer health-related quality of life in national institute for health and care excellence appraisals. Value in Health. 2020;23(10):1349–1357. doi: 10.1016/j.jval.2020.05.017. [DOI] [PubMed] [Google Scholar]
- 6.Brouwer WBF. The Inclusion of Spillover Effects in Economic Evaluations: Not an Optional Extra. Pharmacoeconomics. 2019;37(4):451–456. doi: 10.1007/s40273-018-0730-6. [DOI] [PubMed] [Google Scholar]
- 7.McCabe C. Expanding the scope of costs and benefits for economic evaluations in health: some words of caution. Pharmacoeconomics. 2019;37(4):457–460. doi: 10.1007/s40273-018-0729-z. [DOI] [PubMed] [Google Scholar]
- 8.Lin PJ, D'Cruz B, Leech AA, Neumann PJ, Sanon Aigbogun M, Oberdhan D, et al. Family and caregiver spillover effects in cost-utility analyses of alzheimer's disease interventions. Pharmacoeconomics. 2019;37(4):597–608. doi: 10.1007/s40273-019-00788-3. [DOI] [PubMed] [Google Scholar]
- 9.Krol M, Papenburg J, van Exel J. Does including informal care in economic evaluations matter? A systematic review of inclusion and impact of informal care in cost-effectiveness studies. Pharmacoeconomics. 2015;33(2):123–135. doi: 10.1007/s40273-014-0218-y. [DOI] [PubMed] [Google Scholar]
- 10.Wittenberg E, James LP, Prosser LA. Spillover effects on Caregivers' and family members' utility: a systematic review of the literature. Pharmacoeconomics. 2019;37(4):475–499. doi: 10.1007/s40273-019-00768-7. [DOI] [PubMed] [Google Scholar]
- 11.Bhadhuri A, Jowett S, Jolly K, Al-Janabi H. A comparison of the validity and responsiveness of the EQ-5D-5L and SF-6D for measuring health spillovers: a study of the family impact of meningitis. Med Decis Making. 2017;37(8):882–893. doi: 10.1177/0272989X17706355. [DOI] [PubMed] [Google Scholar]
- 12.Gani R, Giovannoni G, Bates D, Kemball B, Hughes S, Kerrigan J. Cost-effectiveness analyses of natalizumab (Tysabri) compared with other disease-modifying therapies for people with highly active relapsing-remitting multiple sclerosis in the UK. Pharmacoeconomics. 2008;26(7):617–627. doi: 10.2165/00019053-200826070-00008. [DOI] [PubMed] [Google Scholar]
- 13.Lopez-Bastida J, Pena-Longobardo LM, Aranda-Reneo I, Tizzano E, Sefton M, Oliva-Moreno J. Social/economic costs and health-related quality of life in patients with spinal muscular atrophy (SMA) in Spain. Orphanet J Rare Dis. 2017;12(1):141. doi: 10.1186/s13023-017-0695-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McLoughlin C, Goranitis I, Al-Janabi H. Validity and responsiveness of preference-based quality-of-life measures in informal carers: a comparison of 5 measures across 4 conditions. Value Health. 2020;23(6):782–790. doi: 10.1016/j.jval.2020.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Canadian Agency for Drugs and Technologies in Health. Guidelines for the Economic Evaluation of Health Technologies: Canada (4th Edition). 2017 [cited 2021 16 January]; https://www.cadth.ca/dv/guidelines-economic-evaluation-health-technologies-canada-4th-edition.
- 16.Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen. General Methods (Version 5.0). 2017 [cited 2021 16 January]; https://www.iqwig.de/en/about-us/methods/methods-paper/.
- 17.Institute for Clinical and Economic Review. Value assessment framework. 2020 [cited 2021 10 May]; https://icer.org/wp-content/uploads/2020/10/ICER_2020_2023_VAF_102220.pdf.
- 18.Tandvårds- och läkemedelsförmånsverket. Health Economics. 2020 [cited 2021 16 January]; https://www.tlv.se/in-english/medicines/health-economics.html.
- 19.Scottish Medicines Consortium. Guidance to submitting companies for completion of New Product Assessment Form. 2020 [cited 2021 16 January]; https://www.scottishmedicines.org.uk/media/5599/20200611-guidance-on-npaf.pdf.
- 20.All Wales Medicines Strategy Group. Guidance on Appraisal Structure and Evidence Considered. 2019 [cited 2021 16 January]; https://awmsg.nhs.wales/files/appraisal-process/guidance-on-appraisal-structure-and-evidence-considered-pdf-780kb/.
- 21.Agenzia Italiana Del Farmaco. Linee Guida per la compilazione del dossier a support di rimborsabilita e prezzo di un medicinale. 2020 [cited 2021 03 February]; https://www.aifa.gov.it/en/-/l-aifa-approva-le-nuove-linee-guida-per-la-contrattazione-dei-prezzi-e-rimborsi-dei-farmaci.
- 22.National Institute for Health and Care Excellence. NICE health technology evaluations: the manual 2022 [cited 2022 04 May]; https://www.nice.org.uk/process/pmg36/chapter/economic-evaluation.
- 23.Haute Autorité de Santé. Choices in Methods for Economic Evaluation. 2020 [cited 2021 10 May]; https://www.has-sante.fr/jcms/r_1499251/en/choices-in-methods-for-economic-evaluation.
- 24.Health Information and Quality Authority. Guidelines for the Economic Evaluation of Health Technologies in Ireland. 2020 [cited 2021 10 May]; https://www.hiqa.ie/reports-and-publications/health-techNlogy-assessment/guidelines-economic-evaluation-healt.
- 25.Zorginstituut Nederlands. Guidelines for economic evaluations in Healthcare. 2016 [cited 2021 10 May]; https://english.zorginstituutnederland.nl/about-us/publications/reports/2016/06/16/guideline-for-economic-evaluations-in-healthcare.
- 26.Pharmaceutical Benefits Advisory Committee. Guidelines for preparing a submission to the Pharmaceutical Benefits Advisory Committee version 5.0. 2016 [cited 2021 16 January]; https://pbac.pbs.gov.au/information/about-the-guidelines.html.
- 27.PHARMAC. Prescription for Pharmacoeconomic Analysis. 2020 [cited 2021 31 August]; https://pharmac.govt.nz/medicine-funding-and-supply/the-funding-process/policies-manuals-and-processes/economic-analysis/prescription-for-pharmacoeconomic-analysis-methods-for-cost-utility-analysis/6-estimating-health-benefits/.
- 28.Institute for Clinical and Economic Review. Aducanumab for Alzheimer's disease: effectiveness and Value. 2021 [cited 2022 04 May]; https://icer.org/assessment/alzheimers-disease-2021/.
- 29.National Institute for Health and Care Excellence. Aducanumab for treating mild cognitive impairment and mild dementia caused by Alzheimer's disease. 2021 [cited 2022 04 May]; https://www.nice.org.uk/guidance/indevelopment/gid-ta10739.
- 30.Canadian Agency for Drugs and Technologies in Health. Pharmacoeconomic Report: elosulfase alfa (Vimizim). 2015 [cited 2021 09 February]; https://www.cadth.ca/elosulfase-alfa-vimizim. [PubMed]
- 31.BioMarin. Elosulfase alfa for the treatment of mucopolysaccharidosis type IVA. 2015 [cited 2021 09 February]; https://www.nice.org.uk/guidance/hst2/history.
- 32.National Centre for Pharmacoeconomics. Cost-effectiveness of elosulfase alfa (Vimizim®) for the treatment of Morquio A Syndrome in patients of all ages. 2016 [cited 2021 09 February]; https://www.scottishmedicines.org.uk/medicines-advice/elosulfase-alfa-vimizim-fullsubmission-107215/.
- 33.Pharmaceutical Benefits Scheme. Public Summary Document—March 2016 PBAC Meeting. 2016 [cited 2021 09 February]; https://www.pbs.gov.au/pbs/industry/listing/elements/pbac-meetings/psd/2016-03/elosulfase-alfa-vimizim-psd-03-2016.
- 34.Scottish Medicines Consortium. elosulfase alfa, 1mg/mL concentrate for solution for infusion (Vimizim®). 2015 [cited 2021 09 February]; https://www.scottishmedicines.org.uk/medicines-advice/elosulfase-alfa-vimizim-fullsubmission-107215/.
- 35.Tandvårds- och läkemedelsförmånsverket. Underlag för beslut i landstingen Vimizim (elosulfas alfa) 2020 [cited 2021 09 February]; https://www.tlv.se/lakemedel/kliniklakemedelsuppdraget/avslutade-halsoekonomiska-bedomningar/arkiv/2020-05-29-halsoekonomisk-bedomning-av-vimizim-vid-mps-iva-morquio-a-syndrom.html
- 36.Canadian Agency for Drugs and Technologies in Health. Pharmacoeconomic Review Report (Resubmission) NUSINERSEN (SPINRAZA). 2019 [cited 2021 20 January]; https://www.cadth.ca/nusinersen-0 [PubMed]
- 37.Haute Autorite de Sante. Avis D’Efficience Spinraza®(nusinersen). 2017 [cited 2021 20 January]; https://www.has-sante.fr/upload/docs/application/pdf/2018-03/spinraza_12122017_avis_efficience.pdf.
- 38.Institute for Clinical and Economic Review. Spinraza® and Zolgensma® for Spinal Muscular Atrophy: Effectiveness and Value. 2019 [cited 2021 20 January]; https://icer.org/assessment/spinal-muscular-atrophy-2019/.
- 39.Biogen International GmBH. Nusinersen for treating spinal muscular atrophy. 2018 [cited 2021 20 January]; https://www.nice.org.uk/guidance/ta588/evidence.
- 40.Tappenden PH, JKaltenthaler J, Hock E, Rawdin E, Mukuria A, Clowes C, Simonds M, Childs AA. Nusinersen for treating spinal muscular atrophy: A Single Technology Appraisal. 2018 [cited 2021 20 January]; https://www.nice.org.uk/guidance/ta588/evidence
- 41.National Institute for Health and Care Excellence Nusinersen for treating spinal muscular atrophy. 2019 [cited 2021 20 January]; https://www.nice.org.uk/guidance/ta588.
- 42.Pharmaceutical Benefits Scheme. Public Summary Document – July 2020 PBAC meeting. 2020 [cited 2021 20 January]; https://www.pbs.gov.au/pbs/industry/listing/elements/pbac-meetings/psd/2020-07/nusinersen-solution-for-injection-12-mg-in-5-ml-spinraza.
- 43.PHARMAC. TAR 398 Nusinersen for Spinal Muscular Atrophy. 2020 [cited 2021 01 September]; https://pharmac.govt.nz/assets/176-3-2020-03-TAR-398-Nusinersen-for-Spinal-Muscular-Atrophy-FOR-RELEASE-Optimized.pdf.
- 44.Scottish Medicines Consortium. nusinersen 12mg solution for injection (Spinraza® ) 2018 [cited 2021 20 January]; https://www.scottishmedicines.org.uk/medicines-advice/nusinersen-spinraza-fullsubmission-131818/.
- 45.Tandvårds- och läkemedelsförmånsverket. Underlag för beslut i landstingen Spinraza (nusinersen). 2017 [cited 2021 20 January]; https://www.tlv.se/om-oss/press/nyheter/arkiv/2017-11-10-halsoekonomisk-bedomning-av-spinraza-vid-spinal-muskelatrofi-av-typ-5q.html.
- 46.Zorginstituut Nederland. Pakketadvies sluisgeneesmiddel nusinersen (Spinraza®) voor de behandeling van spinale musculaire atrofie (SMA). 2018 [cited 2021 20 January]; https://www.zorginstituutnederland.nl/publicaties/adviezen/2018/02/07/pakketadvies-nusinersen-spinraza-voor-de-behandeling-van-spinale-musculaire-atrofie-sma.
- 47.Canadian Agency for Drugs and Technologies in Health. Pharmacoeconomic Review Report OCRELIZUMAB (OCREVUS) 2017 [cited 2021 08 February]; https://www.cadth.ca/ocrelizumab. [PubMed]
- 48.Institute for Clinical and Economic Review. Disease-modifying therapies for relapsing remitting and primary-progressive multiple sclerosis: effectiveness and value. 2017 [cited 2021 08 February]; : https://icer.org/assessment/multiple-sclerosis-2017/.
- 49.Roche Products Ltd. Ocrelizumab in relapsing forms of multiple sclerosis. 2017 [cited 2021 08 February]; https://www.nice.org.uk/guidance/ta533/history.
- 50.Lord J, Onyimadu O, Harris P, Shepherd J, Frampton G. Ocrelizumab for treating relapsing multiple sclerosis: A Single Technology Appraisal. 2018 [cited 2021 08 February]; https://www.nice.org.uk/guidance/ta533/documents/committee-papers.
- 51.National Institute for Health and Care Excellence. Ocrelizumab for treating relapsing–remitting multiple sclerosis. 2018 [cited 2021 08 February]; https://www.nice.org.uk/guidance/ta533.
- 52.National Centre for Pharmacoeconomics. Cost-effectiveness of ocrelizumab (Ocrevus®) for the treatment of adult patients with relapsing-remitting multiple sclerosis (RRMS). 2018 [cited 2021 08 February]; http://www.ncpe.ie/drugs/ocrelizumab-ocrevus-for-rms/.
- 53.Pharmaceutical Benefits Scheme. Public Summary Document—July 2017 PBAC meeting. 2017 [cited 2021 08 February]; https://www.pbs.gov.au/pbs/industry/listing/elements/pbac-meetings/psd/2017-07/ocrelizumab-psd-july-2017.
- 54.Scottish Medicines Consortium. ocrelizumab 300mg concentrate for solution for infusion (Ocrevus®) 2018 [cited 2021 08 February]; https://www.scottishmedicines.org.uk/medicines-advice/ocrelizumab-ocrevus-resubmission-smc2121/.
- 55.Tandvårds- och läkemedelsförmånsverket. Underlag för beslut i landstingen Ocrevus (okrelizumab). 2018 [cited 2021 08 February]; https://www.tlv.se/lakemedel/kliniklakemedelsuppdraget/avslutade-halsoekonomiska-bedomningar/arkiv/2018-09-12-halsoekonomisk-bedomning-av-ocrevus-vid-behandling-av-multipel-skleros.html.
- 56.Canadian Agency for Drugs and Technologies in Health. Pharmacoeconomic Report: Patisiran. 2019 [cited 2021 08 February]; https://www.cadth.ca/patisiran.
- 57.Institute for Clinical and Economic Review. Inotersen and patisiran for hereditary transthyretin amyloidosis: effectiveness and value. 2018 [cited 2021 08 February]; https://icer.org/assessment/amyloidosis-2018/
- 58.Alnylam Pharmaceuticals. Additional evidence submission for patisiran. 2019 [cited 2021 08 February]; https://www.nice.org.uk/guidance/hst10/evidence.
- 59.Tappenden PNB, A. Stevens, JW. Addendum - ERG critique of the company’s updated model 2019 [cited 2021 08 February]; https://www.nice.org.uk/guidance/hst10/evidence.
- 60.National Institute for Health and Care Excellence. Patisiran for treating hereditary transthyretin amyloidosis. 2019 [cited 2021 08 February]; https://www.nice.org.uk/guidance/hst10/chapter/4-Consideration-of-the-evidence.
- 61.National Centre for Pharmacoeconomics. Cost-effectiveness of patisiran (Onpattro®) for the treatment of hereditary transthyretinmediated (hATTR) amyloidosis in adult patients with stage 1 or stage 2 polyneuropathy. 2020 [cited 2021 08 February]; http://www.ncpe.ie/drugs/patisiran-onpattro/.
- 62.Scottish Medicines Consortium. patisiran 2mg/mL concentrate for solution for infusion (Onpattro®). 2019 [cited 2021 08 February]; https://www.scottishmedicines.org.uk/medicines-advice/patisiran-onpattro-fullsubmission-smc2157.
- 63.Tandvårds- och läkemedelsförmånsverket. Underlag för beslut i landstingen Onpattro (patisiran). 2018 [cited 2021 08 February]; Available from: https://www.tlv.se/lakemedel/kliniklakemedelsuppdraget/avslutade-halsoekonomiska-bedomningar/arkiv/2018-12-18-halsoekonomisk-bedomning-av-onpattro-vid-arftlig-transtyretinamyloidos-ocksa-kallad-skelleftesjukan.html.
- 64.Canadian Agency for Drugs and Technologies in Health. Pharmacoeconomic Report: Voretigene neparvovec (Luxturna). 2021 [cited 2021 09 February]; https://www.cadth.ca/voretigene-neparvovec. [PubMed]
- 65.Institute for Clinical and Economic Reviews. voretigene neparvovec for biallelic RPE65- mediated retinal disease: effectiveness and Value 2018 [cited 2021 09 February]; https://icer.org/assessment/inherited-retinal-disease-2018/.
- 66.Novartis Pharmaceuticals (UK) Ltd. Voretigene neparvovec for treating inherited retinal dystrophies caused by RPE65 gene mutations. 2019 [cited 2021 09 February]; Available from: https://www.nice.org.uk/guidance/hst11/evidence.
- 67.Farmer C, Bullement A, Packman D, Long L, Robinson S, Nikram E, Bello S, Dodman S, Rezaei Hemami M, Churchill A, Lotery A, Hatswell AJ, Melendez-Torres GJ, Crathorne L. Voretigene neparvovec for inherited retinal dystrophies (RPE65 mutations) [ID1054]: a Highly Specialised Technology Evaluation. 2019 [cited 2021 09 February]; https://www.nice.org.uk/guidance/hst11/evidence.
- 68.National Institute for Health and Care Excellence Voretigene neparvovec for treating inherited retinal dystrophies caused by RPE65 gene mutations. 2019 [cited 2021 09 February]; https://www.nice.org.uk/guidance/hst11.
- 69.National Centre for Pharmacoeconomics. Cost-effectiveness of voretigene neparvovec (Luxturna®) for the treatment of adult and paediatric patients with vision loss due to inherited retinal dystrophy caused by confirmed biallelic RPE65 mutations and who have sufficient viable retinal cells. 2020 [cited 2021 09 February]; http://www.ncpe.ie/drugs/voretigene-neparvovec-luxturna/.
- 70.Consortium SM. voretigene neparvovec 5 x 1012 vector genomes/mL concentrate and solvent for solution for injection (Luxturna®). 2019 [cited 2021 09 February]; https://www.scottishmedicines.org.uk/medicines-advice/voretigene-neparvovec-luxturna-uoia-smc2228/.
- 71.Tandvårds- och läkemedelsförmånsverket. Underlag för beslut i landstingen Luxturna (voretigen neparvovek). 2018 [cited 2021 09 February]; https://www.tlv.se/lakemedel/kliniklakemedelsuppdraget/avslutade-halsoekonomiska-bedomningar/arkiv/2019-05-29-halsoekonomisk-bedomning-av-luxturna-vid-behandling-av-synnedsattning-orsakad-av-arftlig-ogonsjukdom.html.
- 72.Zorginstituut Nederland. Pakketadvies voretigene neparvovec (Luxturna®). 2020 [cited 2021 09 February]; Available from: https://www.zorginstituutnederland.nl/publicaties/adviezen/2020/02/17/pakketadvies-voretigene-neparvovec-luxturna.
- 73.Song J, Floyd FJ, Seltzer MM, Greenberg JS, Hong J. Long-term effects of child death on parents' health-related quality of life: a dyadic analysis. Fam Relat. 2010;59(3):269–282. doi: 10.1111/j.1741-3729.2010.00601.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Warwick Evidence. Daclizumab for treating relapsing-remitting multiple sclerosis: A single Technology Appraisal. 2016 [cited 2021 01 March]; https://www.nice.org.uk/guidance/ta441/documents/committee-papers.
- 75.Scope A, Bhadhuri A, Pennington B. Systematic review of cost-utility analyses that have included carer and family member health-related quality of life. Value in Health. 10.1016/j.jval.2022.02.008. [DOI] [PubMed]
- 76.Al-Janabi H, McLoughlin C, Oyebode J, Efstathiou N, Calvert M. Six mechanisms behind carer wellbeing effects: a qualitative study of healthcare delivery. Soc Sci Med. 2019;235:112382. doi: 10.1016/j.socscimed.2019.112382. [DOI] [PubMed] [Google Scholar]
- 77.Canaway A, Al-Janabi H, Kinghorn P, Bailey C, Coast J. Close-person spill-overs in end-of-life care: using hierarchical mapping to identify whose outcomes to include in economic evaluations. Pharmacoeconomics. 2019;37(4):573–583. doi: 10.1007/s40273-019-00786-5. [DOI] [PubMed] [Google Scholar]
- 78.Ara R, Brazier JE. Populating an economic model with health state utility values: moving toward better practice. Value Health. 2010;13(5):509–518. doi: 10.1111/j.1524-4733.2010.00700.x. [DOI] [PubMed] [Google Scholar]