Apremilast is an anti-psoriatic medicine that works intra-cellularly by inhibiting phosphodiesterase 4 (PDE4), thereby blocking the degradation of cyclic adenosine monophosphate (cAMP), leading to its elevation (1, 2). The inhibition of PDE4 results in modification of the immune system, i.e. decreased expression of proinflammatory mediators, and increased anti-inflammatory mediators, such as IL-10 (1–3).
A recent double-blind, placebo-controlled, randomized study demonstrated that apremilast may have a beneficial role in the treatment of palmoplantar psoriasis (4). Given that psoriasis has recently been defined as an autoimmune disease, apremilast may play a role in attenuating not only immune skewing, but also epidermal abnormalities. Therefore, in the current study, apremilast was administered to 6 patients with intractable palmoplantar keratoderma (PPKs) that resulted from psoriasis or other dermatodes, including pityriasis rubra pilaris (PRP), congenital ichthyosiform erythroderma (CIE) and acquired PPK. Surprisingly, all 6 patients showed favourable responses to apremilast within 4 months. Thus, the current study highlights the beneficial clinical effect of apremilast on PPKs in a variety of skin diseases. It also suggests the possibility that an inhibitory effect of apremilast on keratinocyte proliferation may ameliorat e hyperkeratosis lesions in PPK.
CASE REPORTS
Case 1. A woman, aged in her 60s, presented with a 1-year history of yellowish hyperkeratosis on her palms and fingers (Fig. S1a1). Histological findings of her palm revealed hyperkeratosis, loss of granular layers, acanthosis, inflammatory cell infiltrates around small vessels in the upper dermis, and the accumulation of neutrophils in the stratum corneum. In addition, scaly erythema on her extremities was shown. She was then diagnosed with psoriasis vulgaris, and was treated with apremilast. Two months later, her hyperkeratotic erythema in the palmoplantar regions were markedly ameliorated (Fig. S1b1).
Case 2. A woman, aged in her 50s, had a 1-year history of hyperkeratosis and scaly erythema on her palms, fingers, and soles (Fig. S1c1). The patient also showed scaly erythema on her trunk. She was diagnosed with psoriasis vulgaris. Apremilast treatment was then initiated. Three months later, the patient’s hyperkeratotic skin lesions had attenuated (Fig. S1d1).
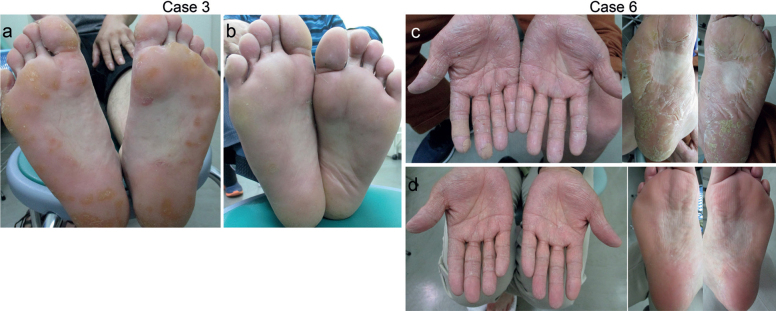
Case 3. A man, aged in his 40s, had hyperkeratosis and scaly erythema on his palms, fingers, and soles (Fig. 1a). Scaly erythema was observed on his face, extremities and trunk. The patient was diagnosed with psoriasis vulgaris. He was started on apremilast treatment. Two months later, the skin lesions were markedly ameliorated (Fig. 1b).
Fig. 1.
(a) Hyperkeratotic skin lesions on the soles in Case 3. (b) Apremilast resulted in a striking improvement of skin lesions within 3 months. (c) The yellowish hyperkeratosis and scaly erythema in Case 6. (d) Apremilast resulted in a striking improvement of palmoplantar keratoderma (PPK) within 2 months.
Case 4. A woman, aged in her 30s, with Down syndrome presented with a 3-year history of diffuse hyperkeratosis on her palms and soles with fissures (Fig. S1e1). The presence of follicular hyperkeratosis in her elbows and knees enabled us to diagnose her with juvenile-onset PRP. The patient had hypothyroidism. She was treated with apremilast, which resulted in the attenuation of hyperkeratosis and fissures in her palms and soles within 3 months (Fig. S1f1).
Case 5. A man, aged in his 40s, exhibited erythroderma with scaling on his whole body since childhood. He presented with erythroderma with scaly lesions over his trunk and face, and marked palmoplantar hyperkeratosis and fissures (Fig. S1g1). His older sister had similar skin lesions. He had hyperuricemia, dyslipidaemia and diabetes mellitus. His parents were consanguineous and they had a homozygous missense mutation (c.4601C>T, p.Thr1534Met) in the ABCA12 gene. Therefore, the patient was diagnosed with CIE. He was treated with apremilast despite the fact that was an off-label use. Strikingly, apremilast attenuated his PPK within 4 months (Fig. S1h1).
Case 6. A man, aged in his 60s, presented with PPK, which had developed 6 months earlier (Fig. 1c). No other skin lesions were noted. He had no prior skin disease, infection or systemic comorbidities before the development of PKK and he had not been exposed to any medicine or chemicals. No malignancy was found on laboratory or imaging examination. He was diagnosed with acquired PPK. We decided to initiate apremilast treatment despite the off-label use. Apremilast treatment resulted in a striking improvement in PPK within 2 months (Fig. 1d).
DISCUSSION
We recently demonstrated the striking impact of apremilast on psoriasis verrucosa, characterized by verruca-like papillomatous epidermal hyperplasia and hyperkeratosis, which is refractory to all other treatments (5). Collectively, apremilast is beneficial for subtypes of psoriasis with marked epidermal hyperplasia and/or hyperkeratosis. Table I shows the characteristics of all case reports described in the current study.
Table I.
Details of patients with palmoplantar keratoderma treated with apremilast
| Patient | Age/Sex | Systemic disease | Diagnosis | Prior treatment | Concomitant therapy | Effect of apremilast | Period needed, months |
|---|---|---|---|---|---|---|---|
| Case 1 | 60s/F | None | Psoriasis vulgaris | Topical corticosteroids and vitamin D3 analogues | Topical corticosteroids and vitamin D3 analogues | Excellent | 2 |
| Case 2 | 50s/F | None | Psoriasis vulgaris | Topical corticosteroids, NB-UVB, etretinate | Topical corticosteroids | Good | 3 |
| Case 3 | 40s/F | None | Psoriasis vulgaris | Topical corticosteroids, NB-UVB, cyclosporine | Topical corticosteroids and vitamin D3 analogues | Excellent | 2 |
| Case 4 | 30s/F | Hypothyroidism | Pityriasis rubra pilaris | Topical vitamin D3 analogues | Topical corticosteroids and moisturizer | Mild | 3 |
| Case 5 | 40s/M | Hyperuricaemia, dyslipidaemia and diabetes mellitus | Congenital ichthyosiform erythroderma | Topical corticosteroids and vitamin D3 analogues, etretinate | Topical corticosteroids and vitamin D3 analogues, etretinate | Mild | 4 |
| Case 6 | 60s/M | None | Acquired PPK | Topical corticosteroids, urea- and salicylic acid-containing ointments, etretinate | Topical corticosteroids and vitamin D3 analogues | Excellent | 2 |
NB-UVB: narrow-band ultraviolet B; PPK: palmoplantar keratoderma.
Case 4, reported here, is the third case of PRP in Down syndrome (6, 7). Similar to our case with PRP, a few studies demonstrated that PPK in patients with PRP improved in response to apremilast (8, 9). Moreover, the recent study reported that anti-IL-12/23 p40 antibody ameliorated the skin lesions in the patients with PRP (10, 11). These results suggest that IL-12/23p40 pathway may be associated with the pathogenesis of PRP.
We report here a case of CIE with an ABCA12 mutation whose PPK was improved by treatment with apremilast (Case 5). Several studies have showed that the pathogenesis of ichthyosis-linked Th17 activation and proliferation (12, 13). In addition, the paper described by Haiges demonstrated that anti-IL-17 antibody ameliorated a case of CIE cornification disorder (14). These findings indicate that immunological IL-17 signals may be involved in the pathogenesis of ichthyosis.
Recently, we investigated the effect of apremilast on human keratinocytes, and found that it downregulated the expression of proinflammatory mediator genes, such as IL-1 (15). We also revealed that inhibitory action of apremilast was due to attenuation of MEK/ERK signalling in keratinocytes, independent of cAMP levels (15). In addition to immune cells, apremilast directly affects keratinocytes through attenuation of the ERK signalling, which is important for cell proliferation and inflammation (15). Therefore, it is likely that apremilast treatment is beneficial not only for psoriasis, but also for PKK, which is characterized by keratinocyte proliferation and inflammation.
Further studies with larger cohorts of patients will be required to elucidate the efficacy of apremilast for the treatment of PPK.
ACKNOWLEDGEMENTS
The authors thank Dr Takuya Takeichi for genetic analysis related to the diagnosis of congenital ichthyosiform erythroderma. The patients reported here provided written informed consent for publication of their case details.
The authors have no conflicts of interest to declare.
REFERENCES
- 1.Schafer PH, Parton A, Gandhi AK, Capone L, Adams M, Wu L, et al. Apremilast, a cAMP phosphodiesterase-4 inhibitor, demonstrates anti-inflammatory activity in vitro and in a model of psoriasis. Br J Pharmacol 2010; 159: 842–855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pincelli C, Schafer PH, French LE, Augustin M, Krueger JG. Mechanisms underlying the clinical effects of apremilast for psoriasis. J Drugs Dermatol 2018; 17: 835–840. [PubMed] [Google Scholar]
- 3.Papadavid E, Rompoti N, Theodoropoulos K, Kokkalis G, Rigopoulos D. Real-world data on the efficacy and safety of apremilast in patients with moderate-to-severe plaque psoriasis. J Eur Acad Dermatol Venereol 2018; 32: 1173–1179. [DOI] [PubMed] [Google Scholar]
- 4.Bissonnette R, Haydey R, Rosoph LA, Lynde CW, Burhalo M, Fowler JF, et al. Apremilast for the treatment of moderate-to-severe palmoplantar psoriasis: results from a double-blind, placebo-controlled, randomized study. J Eur Acad Dermatol Venereol 2018; 32: 403–410. [DOI] [PubMed] [Google Scholar]
- 5.Okazaki S, Osawa R, Nakajima H, Nakajima K, Sano S. Favorable response to apremilast in a patient with refractory psoriasis verrucosa. J Dermatol 2019;46: 544–547. [DOI] [PubMed] [Google Scholar]
- 6.Hazini AR, Rongioletti F, Rebona A. Pityriasis rubra pilaris and vitiligo in Down’s syndrome. Clin Exp Dermatol 1988; 13: 334–335. [DOI] [PubMed] [Google Scholar]
- 7.Terasaki K, Kanekura T, Saruwatari H, Kanzaki T. Classical juvenile pityriasis rubra pilaris with Down synsrome. Clin Dermatol 2004; 29: 49–51. [DOI] [PubMed] [Google Scholar]
- 8.Krase IZ, Cavanaugh K, Curiel-Lewandrowski C. Treatment of refractory pityriasis rubra pilaris with novel phosphodiesterase 4 (PDE4) inhibitor apremilast. JAMA Dermatol 2016; 152: 348–350. [DOI] [PubMed] [Google Scholar]
- 9.Cho M, Honda T, Ueshima C, Kataoka T, Otsuka A, Kabashima K. A case of pityriasis rubra pilaris treated successfully with the phosphodiesterase-4 inhibitor apremilast. Acta Derm Venereol 2018; 98: 975–976. [DOI] [PubMed] [Google Scholar]
- 10.Feldmeyer L, Mylonas A, Demaria O, Mennella A, Yawalkar N, Laffitte E, et al. Interleukin 23-helper T cell 17 axis as a treatment target for pityriasis rubra pilaris. JAMA Dermatol 2017; 153: 304–308. [DOI] [PubMed] [Google Scholar]
- 11.Matsuda T, Yamazaki F, Ueda-Hayakawa I, Kambe N, Okamoto H. Case of pityriasis rubra pilaris progressed to generalized erythroderma following blocade of interleukin-17A, but improved after blockade of interleukin 23. J Dermatol 2019; 46: 70–72. [DOI] [PubMed] [Google Scholar]
- 12.Paller AS. Profiling immune expression to consider repurposing therapeutics for the ichthyoses. J Invest Dermatol 2019; 139: 535–540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Malik K, He H, Huynh TN, Tran G, Mueller K, Doytcheva K, et al. Ichthyosis molecular fingerprinting shows profound T H 17 skewing and a unique barrier genomic signature. J Allergy Clin Immunol 2019; 143: 604–618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Haiges D, Fisher J, Horer S, Has C, Schempp CM. Biologic therapy targeting IL-17 ameliorates a case of congenital ichthyosiform cornification disorder. J Dtsh Dermatol Ges 2019; 17: 70–72. [DOI] [PubMed] [Google Scholar]
- 15.Kataoka S, Takaishi M, Nakajima K, Sato S. Phosphodiesterase-4 inhibitors reduce the expression of proinflammatory mediators by human epidermal keratinocytes independent of intracellular cAMP elevation J Dermatol Sci 2020; 100: 230–233. [DOI] [PubMed] [Google Scholar]