Abstract
Background
The role of consolidative chemotherapy (CCT) for locally advanced esophageal squamous cell carcinoma (LA-ESCC) patients treated with definitive concurrent chemoradiotherapy (dCCRT) is unclear. We aimed to compare the overall survival (OS) of those treated with vs without CCT via a population based approach.
Methods
Eligible LA-ESCC patients diagnosed between 2011 and 2017 were identified via the Taiwan Cancer Registry. We used propensity score (PS) weighting to balance observable potential confounders between groups. The hazard ratio (HR) of death and incidence of esophageal cancer mortality (IECM) were compared between those with vs without CCT. We also evaluated the OS in supplementary analyses via alternative approaches.
Results
Our primary analysis consisted of 368 patients in whom covariates were well balanced after PS weighting. The HR of death when CCT was compared to without was 0.67 (95% confidence interval 0.52–0.86, P = 0.002). The HR of IECM was 0.66 (P = 0.04). The HR of OS remained similarly in favor of CCT in supplementary analyses.
Conclusions
We found that CCT was associated with significantly improved OS for LA-ESCC patients treated with dCCRT. Randomized controlled trials were needed to confirm this finding.
Keywords: Consolidative chemotherapy, Definitive concurrent chemoradiotherapy, Esophageal squamous cell carcinoma
Background
Esophageal cancer was one of the major causes of cancer mortality around the world including Taiwan [1, 2]. Squamous cell carcinoma (SqCC) was the common histology in the East whereas adenocarcinoma was more prevalent in the West [1, 2]. Most esophageal cancer patients were presented with locally advanced stage disease for whom definitive concurrent chemoradiotherapy (dCCRT) was commonly employed [3–6]. However, the long term survival outcomes of locally advanced esophageal cancer patients treated with dCCRT was still not satisfactory [7–10].
Treatment intensification via the use of consolidative (or called adjuvant) chemo therapy (CCT) after dCCRT for these patients may theoretically improve the outcome. However, it was not universally adopted as reflected in its mandatory use in some landmark randomized controlled trials (RCT) [8, 9] but excluded in the other RCTs [7, 10]. The role of CCT was also not clearly addressed in the current treatment guidelines [3–6]. A systematic review published in 2021 reported overall survival (OS) was significantly improved in the short term (1 year hazard ratio (HR) 0.542, P < 0.001) but not in the long term (5 year HR 0.923 P = 0.555) when CCT was compared to without CCT [11]. However, all the six studies regarding CCT in this systematic review were retrospective reviews from limited institutes [12–17]. Due to the lack of population based study, we aimed to compare the OS of locally advanced esophageal squamous cell carcinoma (LA-ESCC) patients treated with dCCRT with/without CCT via a population based approach.
Material and methods
Data source
Our study was a retrospective cohort study based on cancer registry. The analyzed data with personal identifiers removed was obtained from Health and Welfare Data Science Center (HWDC) database. The database included the Taiwan cancer registry (TCR), death registration, and reimbursement data for the whole Taiwan population provided by the Bureau of National Health Insurance (NHI). The TCR with comprehensive information (such as patient demographics, patient/disease/treatment characteristics) had been reported to be a good quality cancer registry [18]. This study had been approved by the Central Regional Research Ethics Committee at China Medical University Taichung Taiwan (CRREC-108-080 (CR2)).
Study design, study population, and intervention
The inclusion criteria of our study populations were (1) LA-ESCC adult (≥ 18 years old) patients diagnosed within 2011–2017 with locally-advanced stage defined as clinical stage cT2-4N0M0 or cT1-4N+M0 for the 7th American Joint Committee on Cancer staging; (2) treated with dCCRT without surgery according to the recording in TCR, with external beam radiotherapy 50–70 Gy in conventional fractionation. We excluded patients with multiple treatment records or prior other cancer(s) to ensure data quality. The study flowchart in concordant with STROBE statement [19] was depicted in Fig. 1.
Fig. 1.
STROBE study flowchart and the number of individuals at each stage of the study. 1We only included those treated (class 1–2) to ensure data consistency. 2Clinical stage cT2-4N0M0 or cT1-4N+M0 for the 7th American Joint Committee on Cancer staging. 350–70 Gy in 1.8–2 Gy/fraction. 4Without missing information in the TCR and death registry regarding survival status, and cause of death
The intervention (i.e., explanatory variable, with vs without CCT), the primary outcome (overall survival, OS) and the supplementary outcome (incidence of esophageal cancer mortality, IECM) were determined via the recordings of TCR or death registry. We defined the diagnostic date in TCR as the index date, and calculated OS/IECM from the index date to the death date (or Dec 31, 2019, i.e. the censoring date in death registry).
Covariates
We collected covariates according to our clinical knowledge [20] via modification from recent relevant studies [21] and our clinical research experiences [22–24]. We used these covariates to adjust for potential nonrandomized treatment selection as defined as follows.
Patient demographics (age, gender, residency): age was classified as ≤ 58 or > 58 years old according a relevant study [21]. Patient residency region was classified as non-north or northern in Taiwan based on the variation in disease and care pattern we observed from clinical care and research experiences [24]. Patient characteristics (comorbidity, body mass index (BMI), drinking, smoking): comorbidity was determined by the modified Charlson comorbidity index score [25] and classified as with or without. BMI was classified as ≤ 18.5 or > 18.5 kg/m2 according to a relevant recent study [21]. The drinking and smoking were classified as no or yes.
Disease characteristics (grade, tumor location, tumor size, clinical T- & N-stage, clinical stage): Grade was classified as poorly or well/moderately differentiated. Tumor location was classified as upper, middle or lower. Tumor size was classified by a diameter ≤ 5 or > 5 cm. The clinical T-stage was classified as T1–T2 or T3–T4. The clinical N-stage was classified as N0 or N1–N2. The clinical stage was classified as II or III.
Diagnostic and treatment characteristics (use of positron emission tomography (PET), reason for no surgery, radiotherapy (RT) modality, RT break, RT dose, induction chemotherapy): The reason for “no surgery” was classified as either with contraindication or without contraindication (but patient refused or surgery was not planned). RT modality were classified as three-dimensional radiotherapy (3DCRT) or intensity-modulated radiotherapy (IMRT). The use of PET was classified as no or yes. For RT break, patients with radiotherapy prolongation was classified as ≤ 1 or > 1 week. RT dose was classified as low (50–50.4 Gy) or high (50.4–70 Gy) dose. The induction chemotherapy (ICT) was classified as with ICT (according to the recording in TCR plus systemic therapy at least 3 weeks before radiotherapy [11, 21]) or without ICT (patients started systemic therapy no earlier than 1 week before radiotherapy was started [11, 21]).
Statistical analyses
In the primary analysis (PA), we adopted propensity score (PS) weighting (PSW) approach using overlap weight as the framework for analysis [26, 27]. To balance the measured potential confounders [28–30], we evaluated the probability of receiving CCT (vs. without CCT) as PS via a logistic regression model based on the above covariates, and then assessed the balance in covariates between groups via standardized difference [20, 30, 31]. In the weighted sample, we compared the hazard ratio (HR) of death between groups via Cox proportional hazards model for point estimation, and used the bootstrap method to estimate the 95% confidence interval (95% CI) [32–34]. We evaluated the impact of potential unmeasured confounder(s) via E-value as suggested in the literature [35]. We also estimated IECM via the competing risk approach [36] between groups in the weighted sample.
In the first supplementary analysis (SA-1), we used alternative analytic framework (PS matching, PSM) among the study population of primary analysis, and then constructed 1:1 PS matched cohorts to compare the HR of death between groups via a robust variance estimator [32]. In the second supplementary analysis (SA-2), we limited our study population to those with clinical response recorded in TCR and performed the PSW analysis in this subgroup to compare the HR of death as well as the response rate between groups.
All statistical analyses in this study were performed with the software SAS 9.4 (SAS Institute, Cary, NC) and R version 4.1.0 (R Development Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Results
Study population in the primary analysis
Our study population consisted of 368 eligible locally advanced esophageal squamous cell carcinoma patients treated with dCCRT plus CCT (n = 103) or no CCT (n = 265) within 2011–2017 (Fig. 1). The patient characteristics were described in Table 1. Two covariates (tumor location, use of PET) were imbalanced before PS weighting, but all covariates achieved balance [20, 31] after PS weighting via overlap weights.
Table 1.
Patient characteristics of the study population in the primary analysis
| Patient characteristics before PSW | Patient characteristics (%) after PSWa | |||||
|---|---|---|---|---|---|---|
| CCT (n = 103) | Without CCT (n = 265) | Standardized differenceb | CCT | Without CCT | Standardized differenceb | |
| Number (%)b or mean (SD)b | Number (%)b or mean (SD)b | |||||
| Age (years) | ||||||
| ≤ 58 | 53 (51) | 139 (52) | 0.020 | 50 | 50 | ≈ 0 |
| > 58 | 50 (49) | 126 (48) | 50 | 50 | ||
| Gender | ||||||
| Female | 5 (5) | 12 (5) | 0.015 | 5 | 5 | ≈ 0 |
| Male | 98 (95) | 253 (95) | 95 | 95 | ||
| Residency | ||||||
| Non-north | 77 (75) | 185 (70) | 0.111 | 73 | 73 | ≈ 0 |
| North | 26 (25) | 80 (30) | 27 | 27 | ||
| Comorbidity | ||||||
| Without | 91 (88) | 233 (88) | 0.013 | 89 | 89 | ≈ 0 |
| Withc | 12 (12) | 32 (12) | 11 | 11 | ||
| BMI (kg/m2) | ||||||
| ≤ 18.5 | 22 (21) | 62 (23) | 0.049 | 21 | 21 | ≈ 0 |
| > 18.5 | 81 (79) | 203 (77) | 79 | 79 | ||
| Drinking | ||||||
| No | 14 (14) | 46 (17) | 0.104 | 14 | 14 | ≈ 0 |
| Yes | 89 (86) | 219 (83) | 86 | 86 | ||
| Smoking | ||||||
| No | 10 (10) | 43 (16) | 0.195 | 11 | 11 | ≈ 0 |
| Yes | 93 (90) | 222 (84) | 89 | 89 | ||
| Grade | ||||||
| Poorly | 34 (33) | 59 (22) | 0.242 | 30 | 30 | ≈ 0 |
| Well/moderately differentiated | 69 (67) | 206 (78) | 70 | 70 | ||
| Tumor location | ||||||
| Upper | 56 (54) | 97 (37) | 50 | 50 | ||
| Middle | 34 (33) | 122 (46) | 0.269 | 36 | 36 | ≈ 0 |
| Lower | 13 (13) | 46 (17) | 0.133 | 14 | 14 | ≈ 0 |
| Tumor size (cm) | ||||||
| ≤ 5 cm | 43 (42) | 99 (37) | 0.090 | 41 | 41 | ≈ 0 |
| > 5 cm | 60 (58) | 166 (63) | 59 | 59 | ||
| Clinical T-stage | ||||||
| T1–T2 | 10 (10) | 32 (12) | 0.076 | 10 | 10 | ≈ 0 |
| T3–T4 | 93 (90) | 233 (88) | 90 | 90 | ||
| Clinical N-stage | ||||||
| N0 | 9 (9) | 23 (9) | 0.002 | 9 | 9 | ≈ 0 |
| N1-N2 | 94 (91) | 242 (91) | 91 | 91 | ||
| Clinical stage | ||||||
| II | 11 (11) | 30 (11) | 0.020 | 12 | 12 | ≈ 0 |
| III | 92 (89) | 235 (89) | 88 | 88 | ||
| Reason for no surgery | ||||||
| Without contraindication | 99 (96) | 248 (94) | 0.115 | 95 | 95 | ≈ 0 |
| With contraindication | 4 (4) | 17 (6) | 5 | 5 | ||
| RT modality | ||||||
| 3DCRT | 7 (7) | 7 (3) | 0.197 | 5 | 5 | ≈ 0 |
| IMRT | 96 (93) | 258 (97) | 95 | 95 | ||
| Use of PET | ||||||
| No | 45 (44) | 77 (29) | 0.308 | 37 | 37 | ≈ 0 |
| Yes | 58 (56) | 188 (71) | 63 | 63 | ||
| RT break | ||||||
| ≤ 1 week | 79 (77) | 197 (74) | 0.055 | 76 | 76 | ≈ 0 |
| > 1 week | 24 (23) | 68 (26) | 24 | 24 | ||
| RT dose | ||||||
| Low | 25 (24) | 88 (33) | 0.198 | 26 | 26 | ≈ 0 |
| High | 78 (76) | 177 (67) | 74 | 74 | ||
| Induction chemotherapy | ||||||
| Without | 98 (95) | 258 (97) | 0.117 | 96 | 96 | ≈ 0 |
| With | 5 (5) | 7 (3) | 4 | 4 | ||
3DCRT, three-dimensional radiotherapy; BMI, Body Mass Index; CCT, consolidative chemotherapy; IGRT, image-guided radiotherapy; IMRT, intensity-modulated radiotherapy; PET, positron emission tomography; PSW, Propensity Score (PS) Weighting; RT, radiotherapy; SD, standard deviation
aWeighted proportion for each group
bRounded
cModified Carlson comorbidity score ≥ 1
Primary analysis
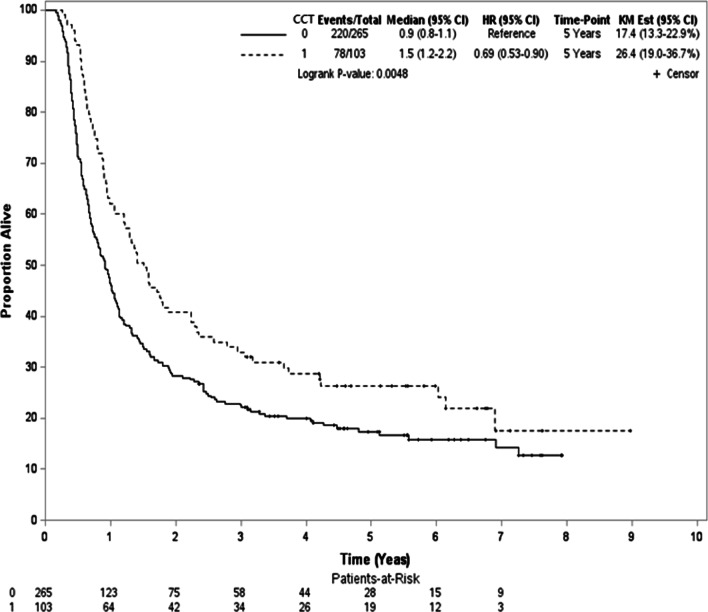
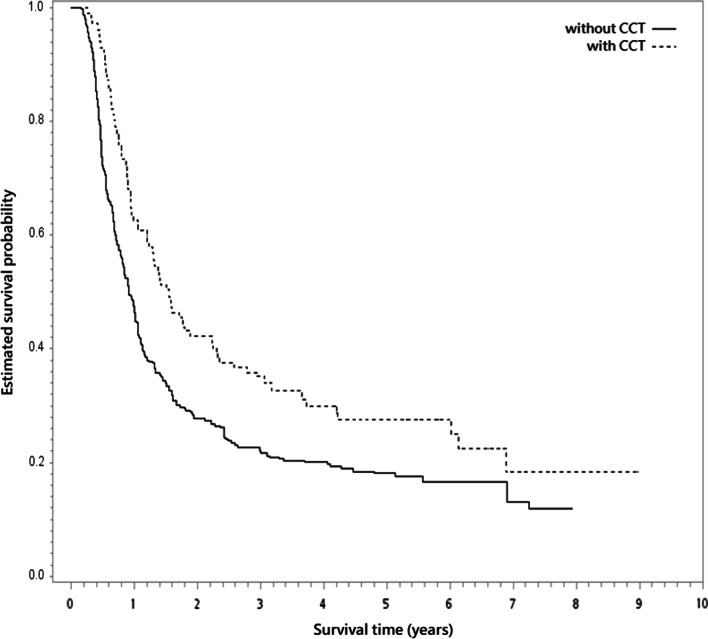
During the follow-up period with median follow-up 12 months (range 2–107 months), 298 deaths were observed (78 and 220 for patients with CCT or without CCT respectively). For survivors, the median follow-up was 63 months (range 28–107). In the unadjusted analysis, the 5-year OS rate was 26% and 17% for those with CCT and without CCT respectively (log-rank test, P = 0.005; Fig. 2). In the PSW analysis, the 5-year PSW-adjusted OS rate between groups were 28% (with CCT) and 18% (without CCT) respectively. The overlap weights adjusted OS curve was shown in Fig. 3. When CCT was compared to without CCT, the PSW adjusted HR of death was 0.67 (95% confidence interval (95% CI) 0.52–0.86, P = 0.002). The observed HR 0.67 for OS could be explained by an unmeasured confounder associated with both selection of treatment and survival by a risk ratio of 1.97 (E-value) fold each, but weaker confounding factors could not. The result was also in favor of CCT for IECM (HR = 0.66, 95% CI 0.44–0.99, P = 0.04).
Fig. 2.
Kaplan–Meier unadjusted overall survival curve (in years) in the primary analysis. CCT, consolidative chemotherapy
Fig. 3.
The overlap weights adjusted overall survival curve (in years) in the primary analysis. CCT, consolidative chemotherapy
Supplementary analyses (SA-1, SA-2)
In the SA-1, we achieved all covariates balance (standardized difference ≤ 0.25 [31]) after PSM in the PS-matched subgroup (n = 182; Table 2). The 5-year OS rate was 26% (with CCT) and 19% (without CCT) respectively. The Kaplan Meier OS curve was shown in Fig. 4. There was also statistically significant difference for OS (HR = 0.69, 95% CI 0.50–0.94, P = 0.02).
Table 2.
SA-1: patient characteristics of the PS-matched subgroup
| CCT (n = 91) | Without CCT (n = 91) | Standardized differencea | |||
|---|---|---|---|---|---|
| Number or mean (SD)a | (%)a | Number or mean (SD)a | (%)a | ||
| Age (years) | |||||
| ≤ 58 | 47 | (52) | 44 | (48) | 0.066 |
| > 58 | 44 | (48) | 47 | (52) | |
| Gender | |||||
| Female | 5 | (5) | 3 | (3) | 0.107 |
| Male | 86 | (95) | 88 | (97) | |
| Residency | |||||
| Non-north | 66 | (73) | 70 | (77) | 0.101 |
| North | 25 | (27) | 21 | (23) | |
| Comorbidity | |||||
| Without | 79 | (87) | 83 | (91) | 0.141 |
| Withb | 12 | (13) | 8 | (9) | |
| BMI (kg/m2) | |||||
| ≤ 18.5 | 20 | (22) | 14 | (15) | 0.170 |
| > 18.5 | 71 | (78) | 77 | (85) | |
| Drinking | |||||
| No | 13 | (14) | 15 | (16) | 0.061 |
| Yes | 78 | (86) | 76 | (84) | |
| Smoking | |||||
| No | 10 | (11) | 10 | (11) | 0 |
| Yes | 81 | (89) | 81 | (89) | |
| Grade | |||||
| Poorly | 28 | (31) | 23 | (25) | 0.123 |
| Well/moderately differentiated | 63 | (69) | 68 | (75) | |
| Tumor location | |||||
| Upper | 46 | (51) | 44 | (48) | |
| Middle | 32 | (35) | 35 | (39) | 0.068 |
| Lower | 13 | (14) | 12 | (13) | 0.032 |
| Tumor size (cm) | |||||
| ≤ 5 cm | 38 | (42) | 43 | (47) | 0.111 |
| > 5 cm | 53 | (58) | 48 | (53) | |
| Clinical T-stage | |||||
| T1–T2 | 10 | (11) | 9 | (10) | 0.036 |
| T3–T4 | 81 | (89) | 82 | (90) | |
| Clinical N-stage | |||||
| N0 | 9 | (10) | 8 | (9) | 0.038 |
| N1–N2 | 82 | (90) | 83 | (91) | |
| Clinical stage | |||||
| II | 11 | (12) | 10 | (11) | 0.034 |
| III | 80 | (88) | 81 | (89) | |
| Reason for no surgery | |||||
| Without contraindication | 87 | (96) | 87 | (96) | 0 |
| With contraindication | 4 | (4) | 4 | (4) | |
| RT modality | |||||
| 3DCRT | 6 | (7) | 3 | (3) | 0.152 |
| IMRT | 85 | (93) | 88 | (97) | |
| Use of PET | |||||
| No | 34 | (37) | 36 | (40) | 0.045 |
| Yes | 57 | (63) | 55 | (60) | |
| RT break | |||||
| ≤ 1 week | 69 | (76) | 70 | (77) | 0.026 |
| > 1 week | 22 | (24) | 21 | (23) | |
| RT dose | |||||
| Low | 23 | (25) | 22 | (24) | 0.025 |
| High | 68 | (75) | 69 | (76) | |
| Induction chemotherapy | |||||
| Without | 87 | (96) | 86 | (95) | 0.051 |
| With | 4 | (4) | 5 | (5) | |
3DCRT, three-dimensional radiotherapy; BMI, Body Mass Index; CCT, consolidative chemotherapy; IGRT, image-guided radiotherapy; IMRT, intensity-modulated radiotherapy; PET, positron emission tomography; RT, radiotherapy; SD, standard deviation
aRounded
bModified Carlson comorbidity score ≥ 1
Fig. 4.
Kaplan–Meier survival curve (in years) for the PS-matched subgroup (SA-1). CCT, consolidative chemotherapy
In the SA-2, covariate balance was also achieved after PSW although some were imbalanced before PSW as shown in Table 3 (n = 246). Comparisons between groups revealed significantly better OS for those with CCT versus without CCT [PSW adjusted HR 0.68 (95% CI 0.49–0.92, P = 0.013). The crude response rate (77% vs. 68%) was higher for those with vs without CCT, but without statistical significance (PSW adjusted odds ratio (OR) 1.61, 95% CI 0.62–2.60, P = 0.23).
Table 3.
SA-2: patient characteristics of the subgroup with clinical response recorded
| Patient characteristics before PSW | Patient characteristics (%) after PSWa | |||||
|---|---|---|---|---|---|---|
| CCT (n = 79) | Without CCT (n = 167) | Standardized differenceb | CCT | Without CCT | Standardized differenceb | |
| Number (%)b or mean (SD)b | Number (%)b or mean (SD)b | |||||
| Age (years) | ||||||
| ≤ 58 | 42 (53) | 90 (54) | 0.015 | 51 | 51 | ≈ 0 |
| > 58 | 37 (47) | 77 (46) | 49 | 49 | ||
| Gender | ||||||
| Female | 4 (5) | 7 (4) | 0.042 | 5 | 5 | ≈ 0 |
| Male | 75 (95) | 160 (96) | 95 | 95 | ||
| Residency | ||||||
| Non-north | 60 (76) | 119 (71) | 0.107 | 74 | 74 | ≈ 0 |
| North | 19 (24) | 48 (29) | 26 | 26 | ||
| Comorbidity | ||||||
| Without | 69 (87) | 146 (87) | 0.003 | 88 | 88 | ≈ 0 |
| Withc | 10 (13) | 21 (13) | 12 | 12 | ||
| BMI (kg/m2) | ||||||
| ≤ 18.5 | 15 (19) | 36 (22) | 0.064 | 19 | 19 | ≈ 0 |
| > 18.5 | 64 (81) | 131 (78) | 81 | 81 | ||
| Drinking | ||||||
| No | 11 (14) | 22 (13) | 0.022 | 14 | 14 | ≈ 0 |
| Yes | 68 (86) | 145 (87) | 86 | 86 | ||
| Smoking | ||||||
| No | 8 (10) | 20 (12) | 0.059 | 11 | 11 | ≈ 0 |
| Yes | 71 (90) | 147 (88) | 89 | 89 | ||
| Grade | ||||||
| Poorly | 25 (32) | 40 (24) | 0.172 | 31 | 31 | ≈ 0 |
| Well/moderately differentiated | 54 (68) | 127 (76) | 69 | 69 | ||
| Tumor location | ||||||
| Upper | 43 (54) | 65 (39) | 50 | 50 | ||
| Middle | 25 (32) | 75 (45) | 0.275 | 35 | 35 | ≈ 0 |
| Lower | 11 (14) | 27 (16) | 0.063 | 15 | 15 | ≈ 0 |
| Tumor size (cm) | ||||||
| ≤ 5 cm | 34 (43) | 61 (37) | 0.133 | 43 | 43 | ≈ 0 |
| > 5 cm | 45 (57) | 106 (63) | 57 | 57 | ||
| Clinical T-stage | ||||||
| T1–T2 | 7 (9) | 17 (10) | 0.045 | 8 | 8 | ≈ 0 |
| T3–T4 | 72 (91) | 150 (90) | 92 | 92 | ||
| Clinical N-stage | ||||||
| N0 | 7 (9) | 8 (5) | 0.162 | 7 | 7 | ≈ 0 |
| N1–N2 | 72 (91) | 159 (95) | 93 | 93 | ||
| Clinical stage | ||||||
| II | 9 (11) | 14 (8) | 0.101 | 10 | 10 | ≈ 0 |
| III | 70 (89) | 153 (92) | 90 | 90 | ||
| Reason for no surgery | ||||||
| Without contraindication | 75 (95) | 154 (92) | 0.111 | 94 | 94 | ≈ 0 |
| With contraindication | 4 (5) | 13 (8) | 6 | 6 | ||
| RT modality | ||||||
| 3DCRT | 7 (9) | 4 (2) | 0.283 | 5 | 5 | ≈ 0 |
| IMRT | 72 (91) | 163 (98) | 95 | 95 | ||
| Use of PET | ||||||
| No | 34 (43) | 42 (25) | 0.384 | 35 | 35 | ≈ 0 |
| Yes | 45 (57) | 125 (75) | 65 | 65 | ||
| RT break | ||||||
| ≤ 1 week | 63 (80) | 125 (75) | 0.117 | 79 | 79 | ≈ 0 |
| > 1 week | 16 (20) | 42 (25) | 21 | 21 | ||
| RT dose | ||||||
| Low | 19 (24) | 54 (32) | 0.185 | 26 | 26 | ≈ 0 |
| High | 60 (76) | 113 (68) | 74 | 74 | ||
| Induction chemotherapy | ||||||
| Without | 74 (94) | 162 (97) | 0.159 | 95 | 95 | ≈ 0 |
| With | 5 (6) | 5 (3) | 5 | 5 | ||
3DCRT, three-dimensional radiotherapy; BMI, Body Mass Index; CCT, consolidative chemotherapy; IGRT, image-guided radiotherapy; IMRT, intensity-modulated radiotherapy; PET, positron emission tomography; PSW, propensity score weighting; RT, radiotherapy; SD, standard deviation
aWeighted proportion for each group
bRounded
cModified Carlson comorbidity score ≥ 1
Discussion
In our population based cohort study, we found that CCT was associated with significantly improved OS for LA-ESCC patients treated with dCCRT. This was the 1st population based study to our knowledge.
In our mind, our results were compatible with the results in the above-mentioned systematic review in that the point estimate of HR for OS was in favor of CCT [11]. In another systematic review published in 2021 (not limited to SqCC but consisted of mainly SqCC patients) [37], favorable OS (HR 0.72; 95% CI 0.59–0.86, P < 0.001) and response rate (OR 1.44; 95% CI 0.62–3.35, P = 0.393) were reported. Our results were relatively close to these results. When we looked at the relevant individual studies [12–17] included in the above systematic review [11], the details were summarized below. Wu et al. compared 67 patients in the CCT group vs 142 patients in control group treated at a single institute and found CCT improved the overall survival with HR 0.67 [12]. In 524 PS matched patients treated from two institutes, Liu et al. reported OS HR 0.92 [13]. Chen et al. investigated 187 patients (89 with CCT whereas 98 without CCT) treated at two institutes and reported OS HR 0.971 in the univariate analyses [14]. Among 124 patients (65 with CCT and 59 without CCT) treated with dCCRT from a single institute, Chen et al. reported the median OS to be 19 months (without CCT) vs. 25 months (with CCT) [15]. From 73 patients treated with dCCRT at three institutes, Koh et al. reported CCT improved OS (3-year, 24.2% vs. 11.8%, P = 0.004) [16]. Among 222 patients (113 with CCT and 109 without CCT) treated with dCCRT from a single institute, Zhang et al. reported the median OS to be 18 months (without CCT) vs. 33 months (with CCT) (P = 0.003) [17]. Therefore, our results were compatible with most of these studies [12, 15–17] in favor of CCT. Furthermore, our study utilized papulation-based cancer registry so were more representative than these studies relied on patients from one ~ three institutes.
The interpretation of our results seems strait forward because the outcomes were improved after treatment intensification. However, RCT were needed to confirm our finding because negative results of CCT had been reported in other disease sites such as lung cancer [38]. The generalizability of our finding to current practice was also not clear in the era of immunotherapy [39, 40].
There were several limitations in our study. First of all, there were always concerns regarding potential unmeasured confounder(s) in non-randomized studies although we had used propensity score to adjust for measured covariates and used E value to address the impact of the potential unmeasured confounders. For example, radiotherapy volume or chemotherapy regimens or cycles may be imbalance between groups but were not considered in our study due to data limitation. Therefore, we reported the E value (1.97) as suggested in the literature to evaluate the potential impact of possible unmeasured confounder(s) [35]. Secondly, other endpoints such as progression free survival or quality of life may also be important but were not investigated due to data limitation as well.
Conclusions
We found that CCT was associated with significantly improved OS for LA-ESCC patients treated with dCCRT. RCT was needed to confirm this finding.
Acknowledgements
The data analyzed in this study were provided by the Health and Welfare Data Science Center, Ministry of Health and Welfare, Executive Yuan, Taiwan. We are grateful to Health Data Science Center, China Medical University Hospital for providing administrative, technical and funding support.
Abbreviations
- 95% CI
95% Confidence interval
- 3DCRT
Three-dimensional radiotherapy
- BMI
Body Mass Index
- CCT
Chemo therapy
- dCCRT
Definitive concurrent chemoradiotherapy
- HR
Hazard ratio
- ICT
Induction chemotherapy
- HWDC
Health and Welfare Data Science Center
- IECM
Incidence of esophageal cancer mortality
- IMRT
Intensity-modulated radiotherapy
- LA-ESCC
Locally advanced esophageal squamous cell carcinoma
- NHI
National Health Insurance
- OS
Overall survival
- PA
Primary analysis
- PET
Positron emission tomography
- PS
Propensity Score
- PSW
Propensity Score Weighting
- RCT
Randomized controlled trial
- RT
Radiotherapy
- SA-1
The first supplementary analysis
- SA-2
The second supplementary analysis
- SqCC
Squamous cell carcinoma
- TCR
Taiwan cancer registry
Author contributions
CRC participated in the concept and design, analysis and interpretation of data, and drafting of the manuscript. CYL, MYL, CCC, HYF, YSL, CKC, JXC, TYL, TMH, TCH, and SSS participated in the concept and design, interpretation of data, and drafting of the manuscript. CCL participated in the concept and design, analysis of data and drafting of the manuscript. All authors read and approved the final manuscript.
Funding
Not applicable.
Availability of data and materials
The data that support the findings of this study are available from [Health and Welfare Data Science Center, Ministry of Health and Welfare, Executive Yuan, Taiwan] but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of [Health and Welfare Data Science Center, Ministry of Health and Welfare, Executive Yuan, Taiwan].
Declarations
Ethics approval and consent to participate
All methods were carried out in accordance with relevant guidelines and regulations. All experimental protocols were approved by Central Regional Research Ethics Committee China Medical University. All study participants in the Taiwan Cancer Registry were deidentified so that the Central Regional Research Ethics Committee China Medical University had waived our study from the need of participants’ informed consent [CRREC-108-080 (CR2)].
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Chen-Yuan Lin, Ming-Yu Lien, Chi-Ching Chen and Hsin-Yuan Fang contributed equally to this work
References
- 1.Lagergren J, Smyth E, Cunningham D, Lagergren P. Oesophageal cancer. Lancet. 2017;390(10110):2383–2396. doi: 10.1016/S0140-6736(17)31462-9. [DOI] [PubMed] [Google Scholar]
- 2.Chien CR, Lin CY, Chen CY. Re: incidence of adenocarcinoma of the esophagus among white Americans by sex, stage, and age. J Natl Cancer Inst. 2009;101(20):1428; author reply 1429. [DOI] [PubMed]
- 3.Lordick F, Mariette C, Haustermans K, Obermannová R, Arnold D; ESMO Guidelines Committee. Oesophageal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27(suppl 5):v50–7. [DOI] [PubMed]
- 4.Kitagawa Y, Uno T, Oyama T, Kato K, Kato H, Kawakubo H, et al. Esophageal cancer practice guidelines 2017 edited by the Japan esophageal society: part 2. Esophagus. 2019;16(1):25–43. doi: 10.1007/s10388-018-0642-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shah MA, Kennedy EB, Catenacci DV, Deighton DC, Goodman KA, Malhotra NK, et al. Treatment of locally advanced esophageal carcinoma: ASCO guideline. J Clin Oncol. 2020;38(23):2677–2694. doi: 10.1200/JCO.20.00866. [DOI] [PubMed] [Google Scholar]
- 6.National Comprehensive Cancer Network Guidelines for Esophageal and Esophagogastric Junction Cancers, version 4.2021 [free registration required] https://www.nccn.org/professionals/physician_gls/pdf/esophageal.pdf. Accessed 14 Oct 2021.
- 7.Hulshof MCCM, Geijsen ED, Rozema T, Oppedijk V, Buijsen J, Neelis KJ, et al. Randomized study on dose escalation in definitive chemoradiation for patients with locally advanced esophageal cancer (ARTDECO study) J Clin Oncol. 2021;39(25):2816–2824. doi: 10.1200/JCO.20.03697. [DOI] [PubMed] [Google Scholar]
- 8.Minsky BD, Pajak TF, Ginsberg RJ, Pisansky TM, Martenson J, Komaki R, et al. INT 0123 (radiation therapy oncology group 94–05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol. 2002;20(5):1167–1174. doi: 10.1200/JCO.2002.20.5.1167. [DOI] [PubMed] [Google Scholar]
- 9.Bedenne L, Michel P, Bouché O, Milan C, Mariette C, Conroy T, et al. Chemoradiation followed by surgery compared with chemoradiation alone in squamous cancer of the esophagus: FFCD 9102. J Clin Oncol. 2007;25(10):1160–1168. doi: 10.1200/JCO.2005.04.7118. [DOI] [PubMed] [Google Scholar]
- 10.Stahl M, Stuschke M, Lehmann N, Meyer HJ, Walz MK, Seeber S, et al. Chemoradiation with and without surgery in patients with locally advanced squamous cell carcinoma of the esophagus. J Clin Oncol. 2005;23(10):2310–2317. doi: 10.1200/JCO.2005.00.034. [DOI] [PubMed] [Google Scholar]
- 11.Wang J, Xiao L, Wang S, Pang Q, Wang J. Addition of induction or consolidation chemotherapy in definitive concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone for patients with unresectable esophageal cancer: a systematic review and meta-analysis. Front Oncol. 2021;11:665231. doi: 10.3389/fonc.2021.665231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu SX, Li XY, Xu HY, Xu QN, Luo HS, Du ZS, et al. Effect of consolidation chemotherapy following definitive chemoradiotherapy in patients with esophageal squamous cell cancer. Sci Rep. 2017;7(1):16870. doi: 10.1038/s41598-017-17254-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen Y, Guo L, Cheng X, Wang J, Zhang Y, Wang Y, et al. With or without consolidation chemotherapy using cisplatin/5-FU after concurrent chemoradiotherapy in stage II-III squamous cell carcinoma of the esophagus: a propensity score-matched analysis. Radiother Oncol. 2018;129(1):154–160. doi: 10.1016/j.radonc.2017.10.031. [DOI] [PubMed] [Google Scholar]
- 14.Chen M, Shen M, Lin Y, Liu P, Liu X, Li X, et al. Adjuvant chemotherapy does not benefit patients with esophageal squamous cell carcinoma treated with definitive chemoradiotherapy. Radiat Oncol. 2018;13(1):150. doi: 10.1186/s13014-018-1086-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen H, Zhou L, Yang Y, Yang L, Chen L. Clinical effect of radiotherapy combined with chemotherapy for non-surgical treatment of the esophageal squamous cell carcinoma. Med Sci Monit. 2018;24:4183–4191. doi: 10.12659/MSM.910326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Koh HK, Park Y, Koo T, Park HJ, Lee MY, Chang AR, et al. Adjuvant chemotherapy and dose escalation in definitive concurrent chemoradiotherapy for esophageal squamous cell carcinoma. Anticancer Res. 2020;40(3):1771–1778. doi: 10.21873/anticanres.14131. [DOI] [PubMed] [Google Scholar]
- 17.Zhang AD, Su XH, Shi GF, Han C, Wang L, Liu H, et al. Survival comparison of three-dimensional radiotherapy alone vs chemoradiotherapy for esophageal squamous cell carcinoma. Arch Med Res. 2020;51(5):419–28. doi: 10.1016/j.arcmed.2020.04.013. [DOI] [PubMed] [Google Scholar]
- 18.Chiang CJ, Wang YW, Lee WC. Taiwan's nationwide cancer registry system of 40 years: past, present, and future. J Formos Med Assoc. 2019;118(5):856–858. doi: 10.1016/j.jfma.2019.01.012. [DOI] [PubMed] [Google Scholar]
- 19.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370(9596):1453–1457. doi: 10.1016/S0140-6736(07)61602-X. [DOI] [PubMed] [Google Scholar]
- 20.Ali MS, Groenwold RH, Belitser SV, Pestman WR, Hoes AW, Roes KC, et al. Reporting of covariate selection and balance assessment in propensity score analysis is suboptimal: a systematic review. J Clin Epidemiol. 2015;68(2):112–121. doi: 10.1016/j.jclinepi.2014.08.011. [DOI] [PubMed] [Google Scholar]
- 21.Liu S, Luo L, Zhao L, Zhu Y, Liu H, Li Q, et al. Induction chemotherapy followed by definitive chemoradiotherapy versus chemoradiotherapy alone in esophageal squamous cell carcinoma: a randomized phase II trial. Nat Commun. 2021;12(1):4014. doi: 10.1038/s41467-021-24288-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li CC, Liang JA, Chen WTL, Chien CR. Effectiveness of image-guided radiotherapy for rectal cancer patients treated with neoadjuvant concurrent chemoradiotherapy: a population-based propensity score-matched analysis. Asia Pac J Clin Oncol. 2019;15(5):e197–203. doi: 10.1111/ajco.13196. [DOI] [PubMed] [Google Scholar]
- 23.Li CC, Chen CY, Chou YH, Huang CJ, Ku HY, Chien CR. Chemotherapy alone versus definitive concurrent chemoradiotherapy for cT4b esophageal squamous cell carcinoma: a population-based study. BMC Gastroenterol. 2021;21(1):153. doi: 10.1186/s12876-021-01742-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kuo YH, Chien YW, Chen PR, Feng CL, Li CC, Chien CR. Impact of the interval between neoadjuvant concurrent chemoradiotherapy and esophagectomy in the modern era: a population-based propensity-score-matched retrospective cohort study in Asia. World J Surg Oncol. 2019;17(1):222. doi: 10.1186/s12957-019-1712-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sun JW, Rogers JR, Her Q, Welch EC, Panozzo CA, Toh S, et al. Adaptation and validation of the combined comorbidity Score for ICD-10-CM. Med Care. 2017;55(12):1046–1051. doi: 10.1097/MLR.0000000000000824. [DOI] [PubMed] [Google Scholar]
- 26.Thomas LE, Li F, Pencina MJ. Overlap weighting: a propensity score method that mimics attributes of a randomized clinical trial. JAMA. 2020;323(23):2417–2418. doi: 10.1001/jama.2020.7819. [DOI] [PubMed] [Google Scholar]
- 27.Mao H, Li L, Greene T. Propensity score weighting analysis and treatment effect discovery. Stat Methods Med Res. 2019;28(8):2439–2454. doi: 10.1177/0962280218781171. [DOI] [PubMed] [Google Scholar]
- 28.Lalani N, Jimenez RB, Yeap B. Understanding propensity score analyses. Int J Radiat Oncol Biol Phys. 2020;107(3):404–407. doi: 10.1016/j.ijrobp.2020.02.638. [DOI] [PubMed] [Google Scholar]
- 29.Rosenbaum PR. Part II. Observational studies-5. Between observational studies and experiments. In: Rosenbaum PR, editor. Observation and experiment: an introduction to causal inference. Cambridge: Harvard University Press; 2017. p. 65–99.
- 30.Webster-Clark M, Stürmer T, Wang T, Man K, Marinac-Dabic D, Rothman KJ, et al. Using propensity scores to estimate effects of treatment initiation decisions: State of the science. Stat Med. 2021;40(7):1718–1735. doi: 10.1002/sim.8866. [DOI] [PubMed] [Google Scholar]
- 31.Garrido MM, Kelley AS, Paris J, Roza K, Meier DE, Morrison RS, et al. Methods for constructing and assessing propensity scores. Health Serv Res. 2014;49(5):1701–1720. doi: 10.1111/1475-6773.12182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Austin PC. The use of propensity score methods with survival or time-to-event outcomes: reporting measures of effect similar to those used in randomized experiments. Stat Med. 2014;33(7):1242–1258. doi: 10.1002/sim.5984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cole SR, Hernán MA. Adjusted survival curves with inverse probability weights. Comput Methods Programs Biomed. 2004;75(1):45–49. doi: 10.1016/j.cmpb.2003.10.004. [DOI] [PubMed] [Google Scholar]
- 34.Austin PC. Variance estimation when using inverse probability of treatment weighting (IPTW) with survival analysis. Stat Med. 2016;35(30):5642–5655. doi: 10.1002/sim.7084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Haneuse S, VanderWeele TJ, Arterburn D. Using the E-value to assess the potential effect of unmeasured confounding in observational studies. JAMA. 2019;321(6):602–603. doi: 10.1001/jama.2018.21554. [DOI] [PubMed] [Google Scholar]
- 36.Bolch CA, Chu H, Jarosek S, Cole SR, Elliott S, Virnig B. Inverse probability of treatment-weighted competing risks analysis: an application on long-term risk of urinary adverse events after prostate cancer treatments. BMC Med Res Methodol. 2017;17(1):93. doi: 10.1186/s12874-017-0367-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xia X, Liu Z, Qin Q, Di X, Zhang Z, Sun X, et al. Long-term survival in nonsurgical esophageal cancer patients who received consolidation chemotherapy compared with patients who received concurrent chemoradiotherapy alone: a systematic review and meta-analysis. Front Oncol. 2021;10:604657. doi: 10.3389/fonc.2020.604657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Senan S, Brade A, Wang LH, Vansteenkiste J, Dakhil S, Biesma B, et al. PROCLAIM: randomized phase iii trial of pemetrexed-cisplatin or etoposide-cisplatin plus thoracic radiation therapy followed by consolidation chemotherapy in locally advanced nonsquamous non-small-cell lung cancer. J Clin Oncol. 2016;34(9):953–962. doi: 10.1200/JCO.2015.64.8824. [DOI] [PubMed] [Google Scholar]
- 39.Faivre-Finn C, Vicente D, Kurata T, Planchard D, Paz-Ares L, Vansteenkiste JF, et al. Four-year survival with durvalumab after chemoradiotherapy in stage III NSCLC-an update from the PACIFIC trial. J Thorac Oncol. 2021;16(5):860–867. doi: 10.1016/j.jtho.2020.12.015. [DOI] [PubMed] [Google Scholar]
- 40.Kelly RJ, Ajani JA, Kuzdzal J, Zander T, Van Cutsem E, Piessen G, et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl J Med. 2021;384(13):1191–1203. doi: 10.1056/NEJMoa2032125. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from [Health and Welfare Data Science Center, Ministry of Health and Welfare, Executive Yuan, Taiwan] but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of [Health and Welfare Data Science Center, Ministry of Health and Welfare, Executive Yuan, Taiwan].