Abstract
Radioimmunotherapy, an approach using radiolabeled antibodies, has had minimal success in the clinic with several β-emitting radionuclides for the treatment of ovarian cancer. Alternatively, radioimmunotherapy with α-emitters offers the advantage of depositing much higher energy over shorter distances but was thought to be inappropriate for the treatment of solid tumors, for which antibody penetration is limited to a few cell diameters around the vascular system. However, the deposition of high-energy α-emitters to tumor markers adjacent to a typical leaky tumor vascular system may have large antitumor effects at the tumor vascular level, and their reduced penetration in normal tissue would be expected to lower off-target toxicity. Methods: To evaluate this concept, DOTAylated-huCC49 was labeled with the α-emitter 225Ac to target tumor-associated glycoprotein 72–positive xenografts in a murine model of ovarian cancer. Results: 225Ac-labeled DOTAylated-huCC49 radioimmunotherapy significantly reduced tumor growth in a dose-dependent manner (1.85, 3.7, and 7.4 kBq), with the 7.4-kBq dose extending survival by more than 3-fold compared with the untreated control. Additionally, a multitreatment regime (1.85 kBq followed by 5 weekly doses of 0.70 kBq for a total of 5.4 kBq) extended survival almost 3-fold compared with the untreated control group, without significant off-target toxicity. Conclusion: These results establish the potential for antibody-targeted α-radionuclide therapy for ovarian cancer, which may be generalized to α-radioimmunotherapy in other solid tumors.
Keywords: TAG-72, 225Ac, radioimmunotherapy, ovarian cancer
Ovarian cancer, the fifth cause of cancer death among women, has an overall survival rate of about 50% in patients with all stages of ovarian cancer but only about 17% in patients diagnosed with later-stage disease. Since only about 15% of patients are identified in the early stages, there is an urgent need for improved therapies (1). Across multiple clinical trials, the increase in progression-free survival is measured in months (2–4). Antibody-targeted therapies, including radioimmunotherapy and antibody–drug conjugates, offer promise in that they target a cytotoxic agent directly to the tumor. Among the tumor-associated antigens identified in ovarian cancer, tumor-associated glycoprotein 72 (TAG-72), expressed in 88% of all stages of ovarian cancer, stands out as an excellent candidate for targeted therapy (5). TAG-72 is a mucinlike molecule that arises from aberrantly glycosylated mucins in cancer (6). Although TAG-72 is expressed in normal endometrial tissues during the secretory phase, it is overexpressed in a multitude of carcinomas, including ovarian cancer (7,8). The monoclonal antibody CC49 was developed for its advantages over the first-generation B72.3 antibody (9). Although anti-TAG-72 antibodies exhibit excellent tumor targeting both in preclinical models and in the clinic (10), they have no therapeutic effects on their own, requiring conjugation to therapeutic agents.
β-emitting radionuclides have been investigated for radioimmunotherapy in solid tumors because of their high tissue penetration (11). In the case of TAG-72–positive solid tumors, 3 β-emitter radionuclides, 131I, 90Y, and 177Lu, on anti-TAG-72 antibodies have been tested for their antitumor effects in clinical studies (12–14). Unfortunately, radioimmunotherapy clinical trials based on the anti-TAG-72 antibody have never made it past phase 2 for the treatment of colon cancer and ovarian cancer, because of the lack of a significant clinical response rate or the presence of bone marrow toxicity. In general, radioimmunotherapy trials using β-emitters have been shown to exhibit a dose-limiting toxicity to the bone marrow due to the high penetrative power of their emissions while the antibody is in circulation (15). Recently, radioimmunotherapy using α-emitting radionuclides has been explored because of their higher linear energy transfer and lower penetrative distance (47–85 μm) in tissue (16). On the basis of their low penetrative power, α-emitting radionuclides in radioimmunotherapy were investigated mainly in the treatment of nonsolid cancers such as leukemia and multiple myeloma (17,18). Recently, the use of α-emitters for the treatment of solid tumors has begun to show promise (19,20). Among the newly available α-emitters, 225Ac stands out because of its long half-life (10 d) and the release of 4 α-particles in its decay scheme (21). Examples of preclinical studies with 225Ac-antibody conjugates include treatment of ovarian cancer xenografts (22), colon tumor xenografts (23), neuroblastoma xenografts (24), and breast cancer metastases (25). The expression of tumor antigens near the tumor vascular system, along with the deposition of higher energies, may make α-radionuclide–based radioimmunotherapy more generally effective than other targeted therapies. To test this concept in an OVCAR3 murine model of ovarian cancer, DOTAylated-huCC49 antibody was radiolabeled with the α-emitter 225Ac, and single versus multitreatment strategies were compared.
MATERIALS AND METHODS
Humanization of anti-TAG-72 murine CC49 mAb antibody and absorbed dose calculations are described in the supplemental materials (available at http://jnm.snmjournals.org).
Antibodies, Reagents, and Cell Lines
The murine CC49 mAb clone was from ATCC and purified by protein A chromatography. Daratumumab (anti-CD38; Janssen Biotech Inc.) was used as an untargeted antibody control. OVCAR3 was from ATCC; DOTA-N-hydroxysuccinimide ester was from Macrocyclics, Inc.; 64Cu was from Washington University; 124I was from 3D Imaging; 111In was from Curium US LLC; and 225Ac was from the Department of Energy, Oak Ridge National Laboratory.
Animal Studies
Animal studies were performed on female NOD SCID mice (Jackson Laboratory) in accordance with protocols 14043 and 91037 approved by the City of Hope Institutional Animal Care and Use Committee, in accordance with the National Institute of Health Office of Laboratory Animal Welfare guidelines. OVCAR3 cells were injected subcutaneously (1.5 × 106 cells per mouse), and tumor size was measured by calipers [0.5(length × width2)]. The mice were euthanized once tumors reached 1,500 mm3. Whole-body toxicity was measured by weight loss, with more than 20% weight loss considered an endpoint.
Radiolabeling Studies
DOTAylated-mAbs (30-molar excess of DOTA-N-hydroxysuccinimide ester) were radiolabeled with 64Cu at a ratio of 0.37:1 MBq/μg,124I at a ratio of 0.37:1 MBq/μg, 111In at a ratio of 0.37:1 MBq/μg, and 225Ac at a ratio of 1.85:1 kBq/μg and purified by size-exclusion chromatography on a 10/30 GL column (GE Biosciences). Radiolabel efficiencies were 89%–100% by instant thin-layer chromatography. The average number of chelates per antibody was determined by quadrupole time-of-flight mass spectrometry. Immunoreactivity was determined by incubation with a 20-fold excess of bovine submaxillary mucin to the radiolabeled antibody, followed by size-exclusion chromatography.
PET Imaging, Biodistribution Studies, and Pharmacokinetics
Tumor-bearing mice were injected intravenously with radiolabeled mAbs (3.7 kBq/mouse for 64Cu; 0.37 kBq/mouse for 124I or 111In) after an intraperitoneal injection of immunoglobulin injection of 1 mg/mL per mouse 2 h beforehand, and imaged at various times on a Siemens Inveon PET/CT scanner. Images were reconstructed as previously described (26). For 124I studies, the mice were given potassium iodide 24–48 h before injections (27).
Therapy Studies
The mice (tumor size, 50–100 mm3) were treated with saline, untargeted control mAb (225Ac-DOTAylated-Dara), or targeted mAb (225Ac-DOTAylated-huCC49), given intravenously, after intravenous immunoglobulin. For the multitreatment study, the mice were injected weekly, for a total of 6 doses. All therapy doses were made up to 30 μg of mAb by the addition of unlabeled mAb.
Statistical Analysis
One-way ANOVA (Tukey multiple-comparison test) was used to compare the tumor growth curves for each treatment group with that of the untreated saline control group. A log-rank Mantel–Cox test was used for the survival curves. Survival was defined as the time required for the tumor to reach a volume of 1,500 mm3. All statistical analyses were performed in Prism, version 7.02 (GraphPad Software).
RESULTS
TAG-72 Expression in OVCAR3 Tumors
The OVCAR3 tumors had strong staining for TAG-72 expression with huCC49 and were negative for CD38 with anti-CD38 antibody (Dara) (Supplemental Fig. 1).
Radiolabeling and Pharmacokinetics of DOTAylated-huCC49
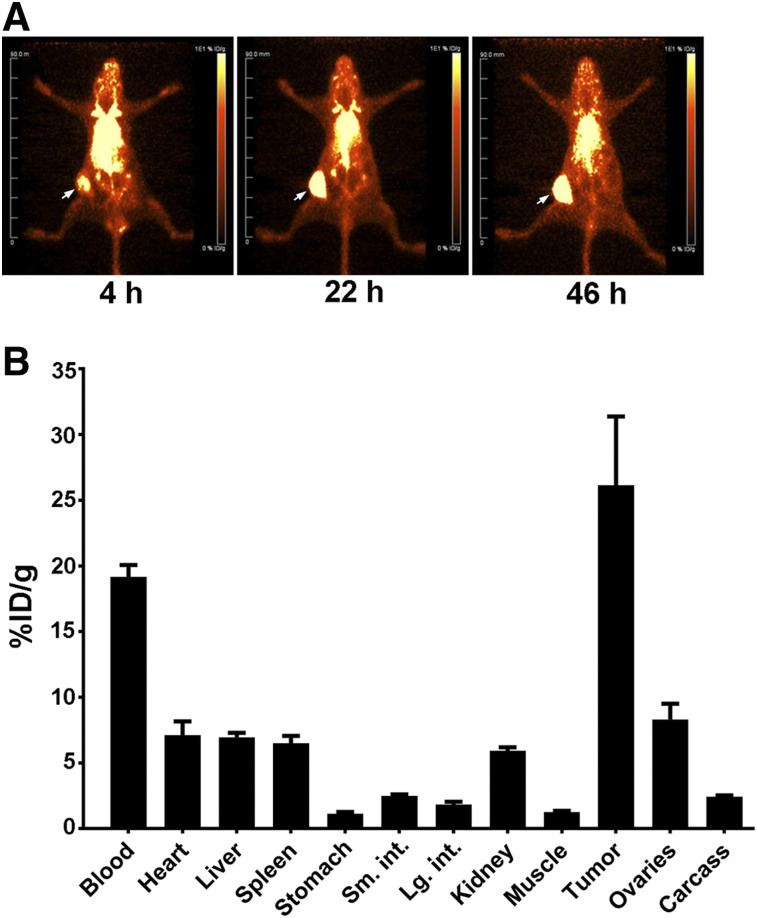
Conjugation of huCC49 with DOTA-N-hydroxysuccinimide gave a product with an average of 6 chelates per antibody, more than 90% labeling with 225Ac, and more than 85% immunoreactivity (Supplemental Fig. 2). Blood clearance of murine CC49 mAb and huCC49 showed similar half-lives of 17.7 and 16.7 h, respectively (Supplemental Fig. 3). PET images of 64Cu-DOTAylated-huCC49 show that huCC49 antibody targeted OVCAR3 tumors (Fig. 1A), reaching 26% injected dose/g at 46 h after injection (Fig. 1B).
FIGURE 1.
PET imaging of 64Cu-DOTAylated-huCC49 mice bearing OVCAR3 xenografts. (A) PET images of 64Cu-DOTAylated-huCC49 (arrow indicates tumor site). (B) Biodistribution at terminal time point (n = 4). Total dose to tumor was 7.8% (26% injected dose (ID)/g × 0.30 g = 7.8% injected dose).
Single-Treatment Radioimmunotherapy Study
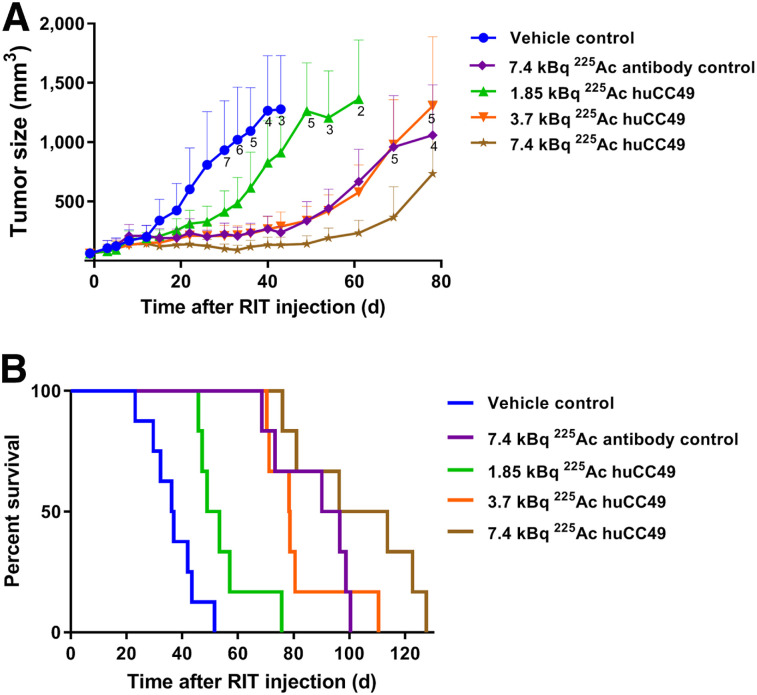
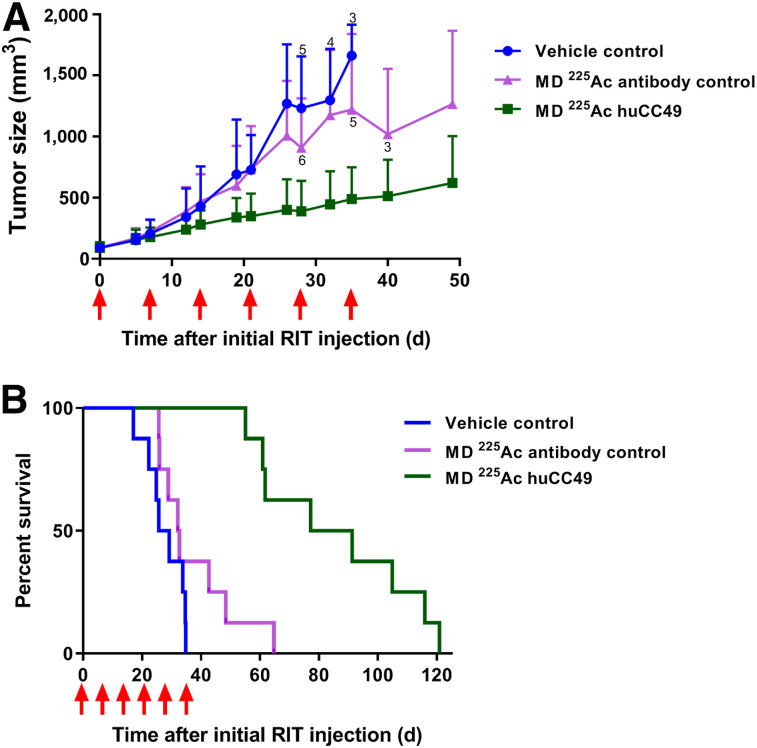
Single treatments of OVCAR3 xenografts with 1.85, 3.7, or 7.4 kBq of 225Ac-DOTAylated-huCC49 led to a dose-dependent reduction in tumor growth (Fig. 2A). Although the 1.85-kBq dose of 225Ac-DOTAylated-huCC49 did not show a significant difference, there was a significant difference in the 3.7- and 7.4-kBq doses compared with the control. The untargeted 225Ac-DOTAylated-Dara control dose of 7.4 kBq was equivalent to the targeted dose of 3.7 kBq. The tumor response seen with the 7.4-kBq untargeted control dose was likely due to enhanced permeability and retention of antibodies. The enhanced permeability and retention effect is the favored accumulation of greater than 40-kDa molecules in tumors over normal tissues (28).
FIGURE 2.
Dose escalation of 225Ac-DOTAylated-huCC49 mice bearing OVCAR3 xenografts. (A) Tumor volume vs. time after radioimmunotherapy (RIT). (B) Kaplan–Meier survival plot (n = 8 for control group; n = 6 for all other groups). Tumor growth curves for 7.4-kBq untargeted (P = 0.0001), 3.7-kBq targeted (P = 0.0001), and 7.4-kBq targeted (P < 0.0001) groups were statistically significant compared with untreated controls. Survival curves for 7.4-kBq untargeted (P = 0.0011), 1.85-kBq targeted (P = 0.0054), 3.7-kBq targeted (P = 0.0003), and 7.4-kBq targeted (P = 0.0003) groups were statistically significant compared with controls.
There was a significant dose-dependent increase in survival (Fig. 2B). In this analysis, mean survival was extended from 32.3 d in the vehicle control to 54.6 d in the 1.85-kBq targeted dose, whereas the 3.7-kBq targeted dose extended survival to 81.6 d and the 7.4-kBq targeted dose extended survival to 103 d (Table 1). Thus, the mean survival of the 7.4-kBq targeted treatment group was more than 3 times the mean survival of the control group, with 3 of 6 mice maintaining reduced tumor growth past day 100 (Supplemental Fig. 4). The 7.4-kBq untargeted antibody control group showed a survival advantage similar to that of the 3.7-kBq 225Ac-DOTAylated-huCC49 group.
TABLE 1.
Mean Survival and Total Absorbed Tumor Dose for 225Ac Radioimmunotherapy of OVCAR3 Xenografts
| Parameter | Control* (n = 16) | 7.4 kBq untargeted antibody (n = 6) | 1.85 kBq huCC49 (n = 6) | 3.7 kBq huCC49 (n = 6) | 7.4 kBq huCC49 (n = 6) | Multitreatment untargeted antibody (n = 8) | Multitreatment huCC49 (n = 8) |
| Mean survival | 32.3 | 87.9† | 54.6† | 81.6† | 103† | 37.6 (NS) | 86.0 |
| Maximum tumor dose (Gy)‡ | NA | 2.93 | 2.50 | 5.01 | 10.0 | 7.74 | 17.7 |
| Minimum tumor dose (Gy)¶ | NA | 0.73 | 0.63 | 1.25 | 2.50 | 1.94 | 4.43 |
Number of radioimmunotherapy controls combined from single and multitreatment groups.
Significant difference from no radioimmunotherapy controls.
Assuming delivery of all 4 α-particles at site of tumor based on biodistribution study.
Assuming delivery of only 1 α-particle at site of tumor based on biodistribution study.
NA = not applicable; NS = not statistically significant.
Whole-body toxicity was measured as percentage weight loss after radioimmunotherapy. Although there was a dose-dependent weight loss with the 225Ac-DOTAylated-huCC49–targeted treatment groups, the weights were maintained within the acceptable less than 20% weight loss (Supplemental Fig. 5). Overall, the 7.4-kBq targeted 225Ac-DOTAylated-huCC49 group showed improved whole-body toxicity compared with the 7.4-kBq untargeted control group, likely because of its longer circulation time.
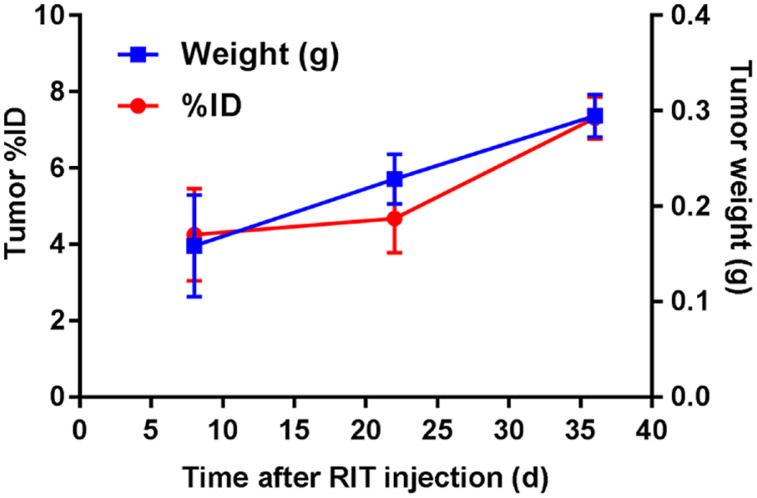
TAG-72 expression was monitored after a single targeted dose of 3.7 kBq of 225Ac-DOTAylated-huCC49 at 1, 3, and 5 wk after the initial 3.4-kBq 225Ac-DOTAylated-huCC49 radioimmunotherapy injection by injection with 64Cu-DOTAylated-huCC49 and terminal biodistribution studies after 3 d. huCC49 uptake correlated with tumor size, confirming that TAG-72 expression was not reduced by a single treatment of 225Ac-DOTAylated-huCC49 (Fig. 3). Since the OVCAR3 xenografts still expressed TAG-72 after a single radioimmunotherapy dose, we explored a multitreatment regimen.
FIGURE 3.
TAG-72 expression measured by 64Cu-DOTAylated-huCC49 tumor uptake after single dose of 3.7 kBq of 225Ac-DOTAylated-huCC49. Two mice per week at 1, 3, and 5 wk after radioimmunotherapy (RIT) (3.7 kBq) were injected with 64Cu-DOTAylated-huCC49, and tumor uptake and weight were measured 72 h after injection. ID = injected dose.
Multitreatment Radioimmunotherapy Study
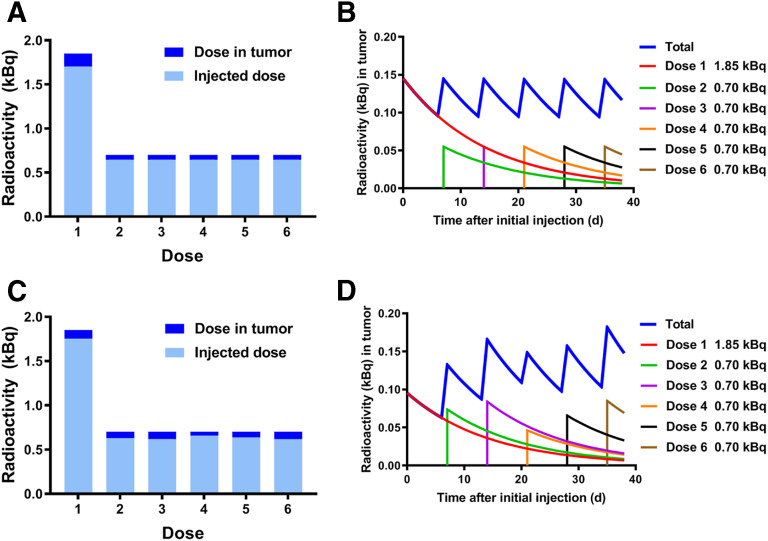
Since the single dose of 1.85 kBq of 225Ac-DOTAylated-huCC49 showed the least whole-body toxicity and only a slight increase in survival, we hypothesized that a multitreatment study with an initial dose of 1.85 kBq followed by subsequent lower doses would have potential benefits over regimens with a higher single dose. The multitreatment experiment was designed to maintain the same amount of radioactivity in the tumor over the course of 6 wk as that achieved by a single treatment of 1.85 kBq. Since the 1.85-kBq dose deposited 7.8% of the dose (0.14 kBq) in the tumor (Fig. 1B), subsequent doses were calculated to restore 0.14 kBq in the tumor 7 d later. Thus, we calculated that mice treated with a single 1.85-kBq dose followed by 5 doses of 0.70 kBq of 225Ac-DOTAylated-huCC49 every 7 d would maintain the tumor dose between 0.09 and 0.14 kBq over each 7-d period (Figs. 4A and 4B). Accounting for radioactive decay, the radioactivity in the tumor (starting at 0.14 kBq) would drop to 0.09 kBq by day 7. A subsequent dose of 0.70 kBq of 225Ac-DOTAylated-huCC49 would boost the amount of radioactivity in the tumor back to 0.14 kBq. This approach allows for the amount of radioactivity in the tumor to be kept within the initial 1.85-kBq single-dose range seen in the first 7 d, but over the course of 6 wk.
FIGURE 4.
Theoretic and actual doses administered in multitreatment strategy. (A) Total calculated dose in kilobecquerels given compared with amount found in tumor with initial dose of 1.85 kBq followed by 5 subsequent doses of 0.70 kBq (Fig. 1). (B) Total calculated amount of radioactivity in tumor, accounting for radioactive decay. (C) Actual injected dose in kilobecquerels determined by 111In-DOTAylated-huCC49 biodistribution performed after each dose of 225Ac-DOTAylated-huCC49 compared with amount in tumor, where initial dose is 1.85 kBq followed by 5 subsequent doses of 0.70 kBq. (D) Total actual amount of radioactivity in tumor, accounting for radioactive decay.
The results of the actual amount of radioactivity delivered to the tumor versus the amount given at the time of injection for each of the 6 doses of 225Ac-DOTAylated-huCC49 were determined by an 111In-DOTAylated-huCC49 tumor uptake study (Fig. 4C). Accounting for radioactivity decay between doses, the actual amount of radioactivity in the tumor over time is shown in Figure 4D. The theoretic doses over time (Fig. 4B) versus the actual doses (Fig. 4D) closely agree.
The results of the multitreatment 225Ac-DOTAylated-huCC49 group showed a significant antitumor effect compared with the control group, whereas the untargeted mAb group showed no significant difference for either tumor growth (Fig. 5A) or the survival curves (Fig. 5B). The multidose 225Ac-DOTAylated-huCC49 group (mean survival, 86.0 d) almost tripled the mean survival compared with both the control group (mean survival, 32.3 d) and the untargeted mAb group (mean survival, 37.6 d; Table 1). In the multitreatment targeted group, 4 of 8 mice maintained reduced tumor growth past day 80, and 3 of 8 mice maintained reduced tumor growth past day 100 (Supplemental Fig. 6).
FIGURE 5.
Multitreatment radioimmunotherapy (RIT) mice bearing OVCAR3 xenografts. Doses were given at days 0, 7, 14, 21, 28, and 35. (A) Tumor volume vs. time after radioimmunotherapy. (B) Kaplan–Meier survival plots (n = 8 for all groups). Tumor growth curve for radioimmunotherapy-targeted group was statistically significant (P = 0.043) compared with controls. Survival curve for targeted radioimmunotherapy was statistically significant (P < 0.0001) compared with controls.
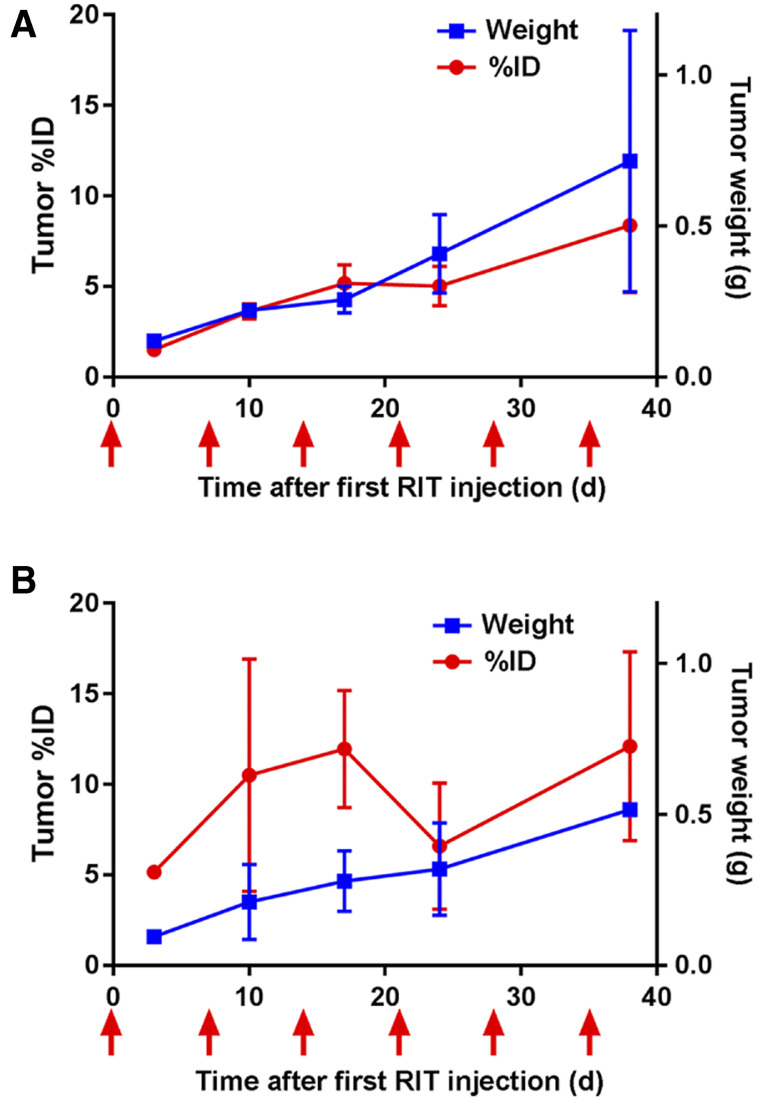
To measure the uptake of targeted versus untargeted mAbs in tumors, the 2 multitreatment radioimmunotherapy groups were probed with 111In-labeled mAbs. In the mice that received 111In-labeled untargeted mAb instead of the untargeted radioimmunotherapy, the tumor uptake was 1.51% (Fig. 6A). The 2 mice that received 111In-labeled targeted mAb instead of targeted radioimmunotherapy had a tumor uptake of 5.16% (Fig. 6B). Thus, the lack of an antitumor response for the multitreatment untargeted controls can be explained by the uptake of only 1.51% compared with 5.16% for the multitreatment targeted radioimmunotherapy group. Untargeted mAb uptake correlates with increasing tumor weight (Fig. 6A), likely because of the enhanced permeability and retention effect. The targeted mAb uptake also correlates with tumor weight (with the exception of a dip after 3 doses of targeted radioimmunotherapy [Fig. 6B], likely caused by continued expression of TAG-72). The dip after the third targeted treatment may be due to the selective killing of cells that express the targeted antigen. The resurgence of TAG-72 expression after the fifth dose suggests that additional targeted radioimmunotherapy could have been performed.
FIGURE 6.
Antibody uptake and tumor weights in targeted and untargeted radioimmunotherapy (RIT). Two mice at indicated time points after radioimmunotherapy were injected with either 111In-labeled untargeted (A) or 111In-labeled targeted (B) antibodies in place of each radioimmunotherapy dose. Timing of 111In-labeled antibody injections is indicated with arrows. ID = injected dose.
The whole-body toxicity of the multitreatment study indicated no significant difference in weight for the multitreatment targeted radioimmunotherapy group versus the control group. However, the multitreatment untargeted radioimmunotherapy group exhibited a significant weight loss compared with the control group (Supplemental Fig. 7). In summary, the multitreatment study demonstrated a significant antitumor effect, with no significant whole-body toxicity from the multitreatment targeted radioimmunotherapy compared with the multitreatment untargeted radioimmunotherapy. Since the multitreatment strategy led to a total dose of 5.4 kBq, a dose falling between two of the single doses administered, we extrapolated this dose as described in Supplemental Figs. 8–11.
Absorbed Radiation Dose in Tumors of Single Treatment Versus Multitreatment Radioimmunotherapy
The absorbed radiation dose in tumors of each of the radioimmunotherapy groups is summarized in Table 1. For the maximum dose, we assumed all of the daughter α-radionuclides remained in the tumor, a likely overestimate. For the minimum dose, we assumed that 75% of the daughter α-radionuclides diffused away into the bloodstream, a likely underestimate (29). The absorbed tumor dose of 10.0 Gy of targeted therapy with a single treatment of 7.4 kBq resulted in the longest survival of 103 d, compared with 87.9 d when 2.03 Gy of untargeted therapy was delivered to the tumor with a single treatment of 7.4 kBq. Survival was improved only 1.17-fold with targeted over untargeted therapy, even with a 5-fold increase in radiation dose to the tumor. Thus, at the highest targeted dose of 7.4 kBq, whole-body toxicity has reduced the potential for even better survival than would be predicted by a 5-fold increase in radiation dose. In the case of targeted multitreatment therapy, a survival of 86 d was achieved with a tumor radiation dose of 17.7 Gy, compared with 37.6-d survival with an untargeted multitreatment tumor radiation dose of 7.74 Gy. There was a 2.29-fold increase in survival with a 2.28-fold increase in radiation dose delivered to the tumor. Thus, a multitreatment regimen is a promising strategy, in that delivery of the radiation dose over an extended time not only reduces whole-body toxicity but also leads to a more linear increase in survival.
DISCUSSION
Murine CC49 radiolabeled with 225Ac in a colon cancer model was previously compared with a CH2-domain deleted CC49 antibody (∆CH2 CC49), an antibody fragment predicted to have better tissue penetration and less toxicity than intact IgG (23). However, it was found that radioimmunotherapy with ∆CH2 CC49 had no benefit over intact IgG. In that study, a single dose of 18.5 kBq of 225Ac-murine CC49 mAb was found to reduce tumor growth by 18 d but had unacceptable levels of toxicity. A lower dose of 9.25 kBq was investigated, with no evidence of tumor regression. Compared with our results in the ovarian cancer model, these findings suggest that 225Ac-CC49 radioimmunotherapy effects may be cancer model–specific.
In an α-emitter–labeled Muc-1 antibody study on an ovarian cancer model, Song et al. (30) showed that a single injection of 355 kBq/kg of 213Bi-C595 prolonged survival by 25 d. Although encouraging, the short half-life of 213Bi (45 min) has limited its use in the treatment of solid tumors.
A study with 225Ac-DOTA–labeled trastuzumab showed high tumor uptake (56%–60% injected dose/g) in a xenograft model of ovarian cancer, with a median survival of 52–126 d compared with controls (33–44 d) when treated with 8.14–16.65 kBq of targeted antibody (22). This study gave results comparable to our 7.4-kBq targeted dose but had high toxicity at the higher doses administered, comparable to our 7.4-kBq untargeted dose.
In our study, the treatment of TAG-72–positive OVCAR3 tumors with 225Ac-DOTAylated-huCC49 led to a potent and prolonged antitumor effect, suggesting that α-emitter–based radioimmunotherapy for solid tumors is feasible. Although we did not determine the physical localization of tumor-deposited antibody, it is unlikely that antibody was directed to every tumor cell, since the immunostaining of the tumor xenografts revealed the typical architecture of a solid tumor. Thus, it is likely that the antitumor effect was due to other effects.
Although α-emitter–based radioimmunotherapy has been shown to effectively treat leukemia and multiple myeloma malignancies (17,18), the rationale for α-emitter–based radioimmunotherapy in solid tumors is debatable since it is well known that systemically delivered antibodies remain localized to the interface between the tumor vasculature and the tumor itself (31). Since α-particles penetrate no deeper than a few cell diameters, one would predict little effect on tumor cells more than a few cell diameters away from the tumor vasculature. However, if the tumor depends on access to vasculature rich in tumor antigens, a likely situation (32), then the deposition of large amounts of energy across the tumor vasculature could be lethal to the entire tumor. Future studies are required to determine whether that mechanism is responsible for the antitumor effect we observed.
A question that was raised in review concerns the fact that as 225Ac decays, the very short-lived daughter radionuclides of 221Fr and 217At produce 213Bi (with a half-life of 45.62 min), which would accumulate over time, necessitating removal of accumulated 213Bi before its reuse in a multitreatment strategy (we used the same batch of 225Ac twice, once on delivery and again after 7 d). However, this concern is minor since the secular amount of 213Bi is the same in both a freshly delivered sample and a 7-d-old sample of 225Ac (33). The amount of 213Bi in the 7-d 225Ac was verified by spectral analysis, showing that the intensity of the 440-keV photon peak of 213Bi was unchanged compared with the 0-d sample.
Whole-body toxicity remains one of the main drawbacks of radioimmunotherapy. In this respect, the whole-body toxicity effect was dramatically reduced by performing a multitreatment regimen. For example, Vallabhajosula et al. (34) showed that a fractioned dose regimen of 90Y-DOTA-anti-PSMA antibody was more effective than a single dose and exhibited less toxicity. In our study, we chose a minimally effective single dose of 1.85 kBq that resulted in 0.14 kBq deposited in the tumor, decaying over 7 d, and asked if maintaining this tumor dose over 6 wk would lead to effective therapy with less whole-body toxicity. This was achieved by following the 1.85-kBq initial dose with 5 weekly doses of 0.70 kBq for a total of 5.4 kBq. In fact, the multitreatment regimen was as effective as a single 7.4-kBq dose, but with less whole-body toxicity.
CONCLUSION
α-Emitter–based radioimmunotherapy for solid tumors not only is feasible but also can be made less toxic if administered as a multidose regimen.
DISCLOSURE
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under award P30CA033572. No other potential conflict of interest relevant to this article was reported.
KEY POINTS
QUESTION: Is α-radionuclide radioimmunotherapy feasible for the treatment of solid tumors such as ovarian cancer?
PERTINENT FINDINGS: A dose of 7.4 kBq of 225Ac-DOTAylated-huCC49 resulted in a mean survival of 103 d, compared with 32 d for untreated controls, with manageable whole-body toxicity. A multitreatment regimen of 1.85 kBq followed by 5 subsequent weekly treatment of 0.70 kBq resulted in a mean survival of 86 d with no whole-body toxicity.
IMPLICATIONS FOR PATIENT CARE: α-radionuclide radioimmunotherapy is feasible for ovarian cancer, and a multitreatment regimen is effective, with negligible whole-body toxicity.
REFERENCES
- 1.Noone AM, Howlader N, Krapcho M, et al., eds. SEER Cancer Statistics Review, 1975–2015. National Cancer Institute website. https://seer.cancer.gov/archive/csr/1975_2015/. Updated September 10, 2018. Accessed October 1, 2020. [Google Scholar]
- 2.González-Martin AJ, Calvo E, Bover I, et al. Randomized phase II trial of carboplatin versus paclitaxel and carboplatin in platinum-sensitive recurrent advanced ovarian carcinoma: a GEICO (Grupo Espanol de Investigacion en Cancer de Ovario) study. Ann Oncol. 2005;16:749–755. [DOI] [PubMed] [Google Scholar]
- 3.Pfisterer J, Plante M, Vergote I, et al. Gemcitabine plus carboplatin compared with carboplatin in patients with platinum-sensitive recurrent ovarian cancer: an intergroup trial of the AGO-OVAR, the NCIC CTG, and the EORTC GCG. J Clin Oncol. 2006;24:4699–4707. [DOI] [PubMed] [Google Scholar]
- 4.Markman M, Webster K, Zanotti K, Kulp B, Peterson G, Belinson J. Phase 2 trial of single-agent gemcitabine in platinum-paclitaxel refractory ovarian cancer. Gynecol Oncol. 2003;90:593–596. [DOI] [PubMed] [Google Scholar]
- 5.Ponnusamy MP, Venkatraman G, Singh AP, et al. Expression of TAG-72 in ovarian cancer and its correlation with tumor stage and patient prognosis. Cancer Lett. 2007;251:247–257. [DOI] [PubMed] [Google Scholar]
- 6.Johnson VG, Schlom J, Paterson AJ, Bennett J, Magnani JL, Colcher D. Analysis of a human tumor-associated glycoprotein (TAG-72) identified by monoclonal antibody B72.3. Cancer Res. 1986;46:850–857. [PubMed] [Google Scholar]
- 7.Thor A, Szpak CA, Gorstein F, Schlom J, Ohuchi N, Johnston WW. Tumor-associated glycoprotein (TAG-72) in ovarian carcinomas defined by monoclonal antibody 872.3. J Natl Cancer Inst. 1986;76:995–1006. [PubMed] [Google Scholar]
- 8.Osteen KG, Anderson TL, Schwartz K, Hargrove JT, Gorstein F. Distribution of tumor-associated glycoprotein-72 (TAG-72) expression throughout the normal female reproductive tract. Int J Gynecol Pathol. 1992;11:216–220. [DOI] [PubMed] [Google Scholar]
- 9.Muraro R, Kuroki M, Wunderlich D, et al. Generation and characterization of B72.3 second generation monoclonal antibodies reactive with the tumor-associated glycoprotein 72 antigen. Cancer Res. 1988;48:4588–4596. [PubMed] [Google Scholar]
- 10.Bohdiewicz PJ, Scott GC, Juni JE, et al. Indium-111 OncoScint CR/OV and F-18 FDG in colorectal and ovarian carcinoma recurrences: early observations. Clin Nucl Med. 1995;20:230–236. [DOI] [PubMed] [Google Scholar]
- 11.DeNardo SJ, DeNardo GL. Targeted radionuclide therapy for solid tumors: an overview. Int J Radiat Oncol Biol Phys. 2006;66(suppl):S89–S95. [DOI] [PubMed] [Google Scholar]
- 12.Murray JL, Macey DJ, Kasi LP, et al. Phase II radioimmunotherapy trial with 131I-CC49 in colorectal cancer. Cancer. 1994;73:1057–1066. [DOI] [PubMed] [Google Scholar]
- 13.Alvarez RD, Partridge EE, Khazaeli MB, et al. Intraperitoneal radioimmunotherapy of ovarian cancer with177Lu-CC49: a phase I/II study. Gynecol Oncol. 1997;65:94–101. [DOI] [PubMed] [Google Scholar]
- 14.Alvarez RD, Huh WK, Khazaeli MB, et al. A phase I study of combined modality 90yttrium-CC49 intraperitoneal radioimmunotherapy for ovarian cancer. Clin Cancer Res. 2002;8:2806–2811. [PubMed] [Google Scholar]
- 15.Larson SM, Carrasquillo JA, Cheung N-KV, Press OW. Radioimmunotherapy of human tumours. Nat Rev Cancer. 2015;15:347–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sgouros G, Roeske JC, McDevitt MR, et al. MIRD pamphlet no. 22 (abridged): radiobiology and dosimetry of α-particle emitters for targeted radionuclide therapy. J Nucl Med. 2010;51:311–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chérel M, Gouard S, Gaschet J, et al. 213Bi radioimmunotherapy with an anti-mCD138 monoclonal antibody in a murine model of multiple myeloma. J Nucl Med. 2013;54:1597–1604. [DOI] [PubMed] [Google Scholar]
- 18.Orozco JJ, Back T, Kenoyer A, et al. Anti-CD45 radioimmunotherapy using 211At with bone marrow transplantation prolongs survival in a disseminated murine leukemia model. Blood. 2013;121:3759–3767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Behling K, Maguire WF, Lopez Puebla JC, et al. Vascular targeted radioimmunotherapy for the treatment of glioblastoma. J Nucl Med. 2016;57:1576–1582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Juergens RA, Zukotynski KA, Juneau D, et al. A phase I study of [225Ac]-FPI-1434 radioimmunotherapy in patients with IGF-1R expressing solid tumors [abstract]. J Clin Oncol. 2019;37(suppl):TPS3152. [Google Scholar]
- 21.Scheinberg DA, McDevitt MR. Actinium-225 in targeted alpha-particle therapeutic applications. Curr Radiopharm. 2011;4:306–320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Borchardt PE, Yuan RR, Miederer M, McDevitt MR, Scheinberg DA. Targeted actinium-225 in vivo generators for therapy of ovarian cancer. Cancer Res. 2003;63:5084–5090. [PubMed] [Google Scholar]
- 23.Kennel SJ, Brechbiel MW, Milenic DE, Schlom J, Mirzadeh S. Actinium-225 conjugates of MAb CC49 and humanized delta CH2CC49. Cancer Biother Radiopharm. 2002;17:219–231. [DOI] [PubMed] [Google Scholar]
- 24.Miederer M, McDevitt MR, Borchardt P, et al. Treatment of neuroblastoma meningeal carcinomatosis with intrathecal application of alpha-emitting atomic nanogenerators targeting disialo-ganglioside GD2. Clin Cancer Res. 2004;10:6985–6992. [DOI] [PubMed] [Google Scholar]
- 25.Song H, Hobbs RF, Vajravelu R, et al. Radioimmunotherapy of breast cancer metastases with alpha-particle emitter 225Ac: comparing efficacy with 213Bi and 90Y. Cancer Res. 2009;69:8941–8948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li L, Bading J, Yazaki PJ, et al. A versatile bifunctional chelate for radiolabeling humanized anti-CEA antibody with In-111 and Cu-64 at either thiol or amino groups: PET imaging of CEA-positive tumors with whole antibodies. Bioconjug Chem. 2008;19:89–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bombardieri E, Giammarile F, Aktolun C, et al. 131I/123I-metaiodobenzylguanidine (mIBG) scintigraphy: procedure guidelines for tumour imaging. Eur J Nucl Med Mol Imaging. 2010;37:2436–2446. [DOI] [PubMed] [Google Scholar]
- 28.Seki T, Fang J, Maeda H. Enhanced delivery of macromolecular antitumor drugs to tumors by nitroglycerin application. Cancer Sci. 2009;100:2426–2430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schwartz J, Jaggi JS, O’Donoghue JA, et al. Renal uptake of bismuth-213 and its contribution to kidney radiation dose following administration of actinium-225-labeled antibody. Phys Med Biol. 2011;56:721–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Song EY, Qu CF, Rizvi SM, et al. Bismuth-213 radioimmunotherapy with C595 anti-MUC1 monoclonal antibody in an ovarian cancer ascites model. Cancer Biol Ther. 2008;7:76–80. [DOI] [PubMed] [Google Scholar]
- 31.Fujimori K, Covell DG, Fletcher JE, Weinstein JN. A modeling analysis of monoclonal antibody percolation through tumors: a binding-site barrier. J Nucl Med. 1990;31:1191–1198. [PubMed] [Google Scholar]
- 32.Azzi S, Hebda JK, Gavard J. Vascular permeability and drug delivery in cancers. Front Oncol. 2013;3:211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Suliman G, Pomme S, Marouli M, et al. Half-lives of 221Fr, 217At, 213Bi, 213Po and 209Pb from the 225Ac decay series. Appl Radiat Isot. 2013;77:32–37. [DOI] [PubMed] [Google Scholar]
- 34.Vallabhajosula S, Smith-Jones PM, Navarro V, Goldsmith SJ, Bander NH. Radioimmunotherapy of prostate cancer in human xenografts using monoclonal antibodies specific to prostate specific membrane antigen (PSMA): studies in nude mice. Prostate. 2004;58:145–155. [DOI] [PubMed] [Google Scholar]