Abstract
Escherichia coli MC4100 was grown in anaerobic glucose-limited chemostat cultures, either in the presence of an electron acceptor (fumarate, nitrate, or oxygen) or fully fermentatively. The steady-state NADH/NAD ratio depended on the nature of the electron acceptor. Anaerobically, the ratio was highest, and it decreased progressively with increasing midpoint potential of the electron acceptor. Similarly, decreasing the dissolved oxygen tension resulted in an increased NADH/NAD ratio. As pyruvate catabolism is a major switch point between fermentative and respiratory behavior, the fluxes through the different pyruvate-consuming enzymes were calculated. Although pyruvate formate lyase (PFL) is inactivated by oxygen, it was inferred that the in vivo activity of the enzyme occurred at low dissolved oxygen tensions (DOT ≤ 1%). A simultaneous flux from pyruvate through both PFL and the pyruvate dehydrogenase complex (PDHc) was observed. In anaerobic cultures with fumarate or nitrate as an electron acceptor, a significant flux through the PDHc was calculated on the basis of the redox balance, the measured products, and the known biochemistry. This result calls into question the common assumption that the complex cannot be active under these conditions. In vitro activity measurements of PDHc showed that the cellular content of the enzyme varied with the internal redox state and revealed an activity for dissolved oxygen tension of below 1%. Whereas Western blots showed that the E3 subunit of PDHc (dihydrolipoamide dehydrogenase) did not vary to a large extent under the conditions tested, the E2 subunit (dihydrolipoamide acetyltransferase) amount followed the trend that was found for the in vitro PDHc activity. From this it is concluded that regulation of the PDHc is exerted at the E1/E2 operon (aceEF). We propose that the external redox state (measured as the midpoint potentials of those terminal acceptors with which the cell has sufficient capacity to react) is reflected by the internal redox state. The latter may subsequently govern both the expression and the activity of the two pyruvate-catabolizing enzymes.
In the family Enterobacteriaceae, as in many prokaryotes and all eukaryotes, the pyridine nucleotides NAD and NADH play a central role in catabolism. These coenzymes (as well as their phosphorylated forms) function as the most important redox carriers involved in metabolism. These nucleotides not only serve as electron acceptors in the breakdown of catabolic substrates but also provide the cell with the reducing power needed in energy-conserving redox reactions such as occur in anaerobic and aerobic respiration. A balance in the rates of oxidation and reduction of these nucleotides is a prerequisite for the continuation of both catabolism and anabolism, since the turnover of the nucleotides is very high compared to their concentrations. Whereas for a given carbon and energy source, catabolic NADH formation occurs under all conditions by a rather limited set of redox reactions (e.g., glycolysis), a wide variety of mechanisms has evolved to fulfill the requirement of NADH reoxidation. Thus, in many bacterial species, e.g., Escherichia coli, a variety of compounds can serve as acceptors of the electrons from NADH. These acceptors may either be present in the environment (external acceptors) or they may be generated intracellularly. Electron transfer may occur either in a cytoplasmatic, nonvectorial process or in a membrane-bound vectorial process. The former reactions (fermentation) result in the reoxidation of NADH and the formation of reduced compounds only, whereas with the latter NADH oxidation may be coupled to the conservation of free energy (respiration).
The regulatory mechanisms that underlie the expression of genes coding the enzymes specific to respiration and fermentation have been studied extensively (15, 20). In E. coli, these genes are under control of at least three global regulators which exert their effects dependent on the redox environment of the cell. The first of these is FNR, which is involved in the regulation of expression of some fermentation-related enzymes (30), and the others are the two-component regulatory systems Nar (31) and Arc (14). FNR can function as both an activator and a repressor of many anaerobically controlled genes. Its regulatory mechanism is thought to reside in the binding of dimeric FNR to the promoter regions of the relevant genes with affinities that depend on the redox state of the cysteine-rich N terminus (6). Molecular oxygen can oxidize the iron-sulfur cluster contained in this region, resulting in monomerization of the protein and the subsequent loss of its ability to bind DNA (16, 18). Nar, which serves primarily as a nitrate-sensing system (31), belongs to the two-component redox regulation systems. It comprises a membrane spanning sensor (NarX) that may act as a kinase under the proper environmental conditions, causing phosphorylation of the regulator (NarL). In addition, a second sensor (NarQ) as well as a second regulator (NarP) have been identified and the NarP/NarQ system supposedly has a somewhat different role in gene control. The Nar system as a whole activates transcription of the genes involved in nitrate reduction (e.g., nitrate and nitrite reductase) and represses the fumarate reductase gene (for a review, see reference 31). The Arc system also belongs to the two-component regulation systems. In its active form it mainly represses enzymes of aerobic catabolism (e.g., the tricarboxylic acid [TCA] cycle and the respiratory chain) (14). The mechanism of this system involves a transphosphorylation from the sensor ArcB to the regulator ArcA, although no precise mechanism has been put forward unequivocally (11, 12). Although in vitro studies have shown that both lactate and NADH stimulate the activation (phosphorylation) of Arc (11, 12), it is not known what the biochemical signal is that results in activation of the system.
Pyruvate is a key intermediate in the catabolism in E. coli and is a common product for most if not all free-energy sources irrespective of the environmental conditions. Its subsequent conversion by either pyruvate formate lyase (PFL) or the pyruvate dehydrogenase complex (PDHc) can be considered as a major switch point between fermentative routes (mixed acid fermentation) and oxidative routes (the TCA cycle and subsequent respiration). Both PFL and PDHc have been subjected to extensive molecular studies (7, 19, 23). In E. coli expression of the PFL is regulated by the Fnr and Arc systems, whereas PDHc synthesis is regulated by the Arc system (13, 24).
It has been assumed for many years that the PDHc could not be active under anaerobic conditions (see also reference 7). The absence of PDHc activity under anaerobic conditions in E. coli has been explained by the high sensitivity towards NADH inhibition (8). However, in Enterococcus faecalis it was found that the distribution over PDHc and PFL of the catabolic flux correlated with the in vivo steady-state redox potential of the NADH/NAD couple (28). Thus, in this organism high in vivo activities of the PDHc were found, even under anaerobic conditions, provided that growth conditions were such that the steady-state NADH/NAD ratio was sufficiently low (29). This, for example, could be achieved when pyruvate was used as the sole energy source.
In contrast to E. faecalis, E. coli is capable of both anaerobic (nitrate and fumarate) and aerobic respiration. It is to be expected that these respiration types affect the redox state of the cell to various extents and hence possibly the level and/or activity of the PDHc. Indeed, Kaiser and Sawers (17) have provided circumstantial evidence that the PDHc of E. coli can be active anaerobically when the cells are provided with nitrate as an external electron acceptor.
We report here on the effect of different electron acceptors on the steady-state metabolic fluxes in E. coli grown in glucose-limited chemostat cultures. For all conditions, the steady-state flux distribution through the PFL and PDHc rates were determined for both wild-type cells and mutant strains lacking PFL or PDHc. In addition, the steady-state cellular redox state (as reflected by the NADH/NAD ratio) and the in vitro activity of PDHc were determined. The steady-state NADH/NAD ratio appeared to be dependent on the nature of external electron acceptors. It was demonstrated that the synthesis and activity of the PDHc does occur under anaerobic respiratory conditions and that both correlate with the cellular steady-state redox state.
MATERIALS AND METHODS
Strains and growth conditions.
The following strains were used: E. coli MC4100 [F− araD139 (argF-lac)U169 rpsL150 relA1 deoC1 flb-5301 ptsF1 (1) (wild type)], E. coli RM201 [see MC4100 Δpfl-25 Ω(pfl::cat pACYC184) (27)], and E. coli RM319 [see MC4100 Δ(aroP-aceEF) (17)]. The strains were maintained on beads in Luria-Bertani medium with 50% (wt/vol) glycerol at −20°C. Organisms were cultured in a 700-ml fermentor (Modular Fermentor Series III (L. H. Engineering Co., Ltd., Stoke Poges, England), and microaerobic cultures were performed in a 3-liter Bioflow III fermentor (New Brunswick) equipped with an Ingold polarographic oxygen electrode coupled to a feedback regulation to maintain the set point dissolved oxygen tension (DOT) value by agitation speed. A 100% DOT value was set by sparging the fermentor with air, and a 0% DOT value was set by sparging with nitrogen gas. Growth media were simple salts media as specified by Evans et al. (4) but, instead of citrate, nitrilotriacetic acid (2 mM) was used as the chelator. Selenite (30 μg/liter) and thiamine (15 mg/liter) were added to the medium. For growth of RM201 without an external electron acceptor, the medium was supplemented with 5 mM acetate. The dilution rate was set at 0.10 or 0.3 h−1. The pH of the culture was maintained at 6.5 ± 0.1 by using sterile 4 M NaOH. For cultures of RM201 without an external electron acceptor, 1 M Na2CO3 was added to the NaOH in order to supply the culture with CO2. The temperature was set to 35°C. Anaerobic cultures were stirred at 1,000 rpm. To prevent excessive foaming, BDH silicone antifoaming agent (1% [wt/vol]) was added at a rate of approximately 0.5 ml · h−1. Anaerobiosis was maintained by the method described previously (33).
Analyses.
Steady-state bacterial dry weight was measured by the procedure of Herbert et al. (9). Glucose, pyruvate, lactate, formate, acetate, succinate, and ethanol were determined by high-pressure liquid chromatography (LKB) with an Aminex HPX 87H organic acid analysis column (Bio-Rad) at a temperature of 65°C with 5 mM H2SO4 as the eluent by using a 2142 refractive index detector (LKB) and an SP 4270 integrator (Spectra Physics). CO2 production or O2 consumption was measured by passing the effluent gas from the fermentor through a Servomex CO2 analyzer and a Servomex O2 analyzer.
Enzyme activities in vitro.
To obtain cell extracts, cells were taken from a steady-state culture, centrifuged (3,020 × g, 10 min), washed twice with 50 mM sodium phosphate buffer (pH 7.0), and lysed with a Branson Sonifier 250 (4 min; duty cycle, 50%; output control, 35%). The cell debris was removed by centrifugation (12,100 × g, 15 min).
The overall activity of the PDHc was measured in a standard reaction mixture containing 50 mM sodium phosphate (pH 7.0), 12.5 mM MgCl2, 0.18 mM thiamine pyrophosphate (TPP), 0.175 mM coenzyme A (CoA) 2 mM NAD, 5 mM pyruvate, and 1 mM potassium ferricyanide. The reaction was started by the addition of cell extract, and the initial rate of reaction was monitored spectrophotometrically by monitoring the reduction of ferricyanide at 430 nm (ɛ = 1,030 M−1 · cm−1).
The activity of the E2 component (dihydrolipoamide acetyltransferase) of the PDHc was measured in a standard reaction mixture containing 10 mM acetylphosphate, 4 mM lipoamide-(SH)2NH2, 0.13 mM CoA, and 2 U of phosphotransacetylase acid in 50 mM Tris (pH 7.0). The reaction was started by the addition of cell extract, and the initial rate was monitored by following the production of acetyl lipoate at 240 nm (ɛ = 5,000 M−1 · cm−1).
Western blotting.
Cell extracts of chemostat cultures of E. coli were loaded and run on a sodium dodecyl sulfate-polyacrylamide electrophoresis gel (12.5%). Equal amounts of protein were loaded on each lane. The gel was then incubated in a transfer buffer (20 mM TrisCl, pH 8.7; 150 mM glycine; 20% methanol) for 30 min. The proteins were blotted on a nitrocellulose filter by electrophoretic transfer. The membrane was then floated in Tris-buffered saline (TBS; 10 mM Tris-Cl, pH 7.6; 150 mM NaCl). Excess protein binding sites were blocked by TBST (TBS plus 0.05% Tween 20) and incubated in TBST with 1% bovine serum albumin. Next, the primary antibody was applied to the filter for 30 min. The membrane was washed in TBST for 5 min to remove the unbound antibody. After this the secondary antibody was applied (anti-immunoglobulin G alkaline phosphatase conjugate) for 30 min. The membrane was washed again in TBST for 5 min to remove unbound antibody and subsequently dried on filter paper. The color reaction was started by transferring the membrane to the color development solution (NBT substrate in AP buffer [100 mM Tris-HCl, pH 9.5; 100 mM NaCl, 5 mM MgCl2 mixed with BCIP [5-bromo-4-chloro-3-indolylphosphate toluidinium] substrate). Within 1 to 15 min, the color developed and the reaction was stopped by rinsing the filter in deionized water.
NADH and NAD+.
Levels of these two pyridine nucleotides were measured by first extracting the nucleotides from a culture sample and then assaying for the nucleotides in the neutralized, filtered extract, as described previously (28).
RESULTS
Catabolic fluxes. (i) Anaerobic conditions without external electron acceptors.
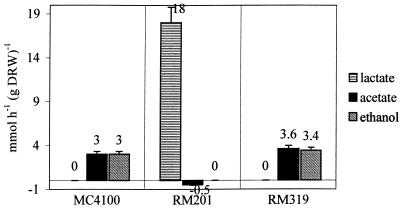
All E. coli strains were grown in glucose-limited chemostat cultures at a dilution rate of 0.1 h−1 at pH 6.5. Anaerobic growth of strain RM 201 (PFL mutant) was only observed when the culture medium was supplied with CO2 and acetate. Presumably this is due to the need for C2 compounds for biosynthetic purposes (36). Therefore, with this strain carbonate was used as a titrant, and acetate (5 mM) was added to the medium. For all cultures, carbon balances of 100 ± 10% could be calculated on the basis of glucose consumption rates and product formation rates (Fig. 1). The main fermentation product of strain RM201 was lactate, confirming the absence of PFL and indicating that under these conditions PDHc was not active. It should be noted here that the specific rate of succinate production was invariably found to be the same for all strains (0.8 mmol/g [dry weight]/h). Lactate formation from glucose is redox neutral. Consequently, all NAD(H)-dependent reactions leading to succinate and biomass should add up to a closed redox balance. This is confirmed by the observation that strain RM319 produced ethanol and acetate in equimolar amounts. This must have occurred via PFL (the strain lacks PDHc) which, again, is an overall redox-neutral process. Similarly, it can be concluded that in wild-type E. coli no in vivo activity of PDHc occurs (see Fig. 4a).
FIG. 1.
Production rates of lactate, acetate, and ethanol in anaerobic glucose-limited chemostat cultures of E. coli MC4100, RM201, and RM319. Strains were grown at a dilution rate of 0.1 h−1 at pH 6.5 and at 35°C.
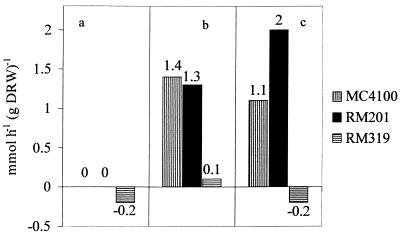
FIG. 4.
Calculated fluxes through the PDHc in E. coli MC4100, RM201, and RM319 grown under the conditions described in Fig. 1 (a), Fig. 2 (b), and Fig. 3 (c). The fluxes are calculated according to the electron flow to the external acceptor minus the difference between the acetate and ethanol production rates.
(ii) Anaerobic conditions with fumarate or nitrate as the electron acceptor.
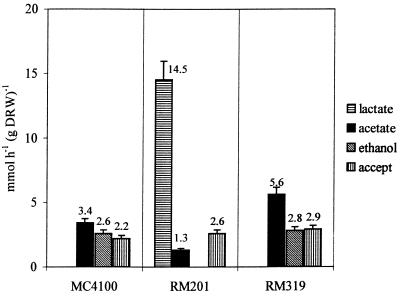
In the presence of fumarate (25 mM medium concentration) a significant shift in the relative production rates was observed (Fig. 2) compared to the findings in fermentative conditions. Succinate production rates (from glucose) similar to those observed under fermentative conditions were measured (data not shown). With all strains, it was found that the fumarate added to the medium was fully reduced to succinate, thus providing the cells with an additional NADH sink. When this NADH oxidizing flux is included in the calculation of the redox balance, it follows that for the wild-type cells breakdown of pyruvate solely by PFL would not provide sufficient reducing equivalents to allow for the observed fumarate reduction rate since in that case the excess of acetate production relative to ethanol should equal the fumarate reduction rate. Indeed, such a stoichiometry was observed for RM319, which can produce acetate solely via PFL. Since in the wild type the difference between acetate and ethanol production rates is less than the fumarate reduction rate, an additional NADH-generating pathway must have been operational to feed fumarate reduction. We propose that here PDHc activity provides the reducing equivalents to fumarate respiration. Support for this possibility is provided by the observation that now strain RM201 produces acetate rather than being dependent for its growth on the availability of acetate in the medium. In the absence of PFL, acetate production can only be ascribed to PDHc activity. On the basis of a closed redox balance, the in vivo fluxes via the PDHc have been calculated, and the results are presented (see Fig. 4b).
FIG. 2.
Production rates of lactate, acetate, and ethanol and the rate of NADH oxidation via fumarate respiration (accept) in anaerobic glucose-limited chemostat cultures of E. coli MC4100, RM201, and RM319 with 25 mM fumarate added to the medium. Strains were grown at a dilution rate of 0.1 h−1 at pH 6.5 at 35°C.
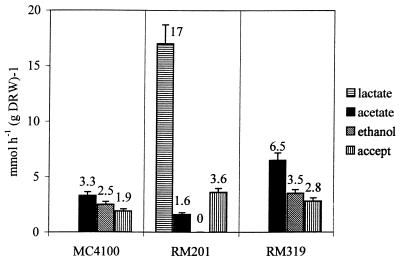
A similar approach was followed by analyzing the relative fluxes to end products when the three strains where grown anaerobically in glucose-limited chemostat cultures with 5 mM nitrate added to the medium. Specific product formation and nitrate utilization rates are given in Fig. 3. The results show that the presence of nitrate as an electron acceptor resulted in a shift in flux distribution over PFL and PDHc that was qualitatively similar to the shift found with fumarate. Again, the observed acetate production by strain RM201 can only be ascribed to the activity of PDHc, whereas in wild-type cells in vivo PDHc activity can be inferred from the difference between acetate and ethanol production being lower than the rate of NADH oxidation by nitrate reduction (Fig. 4). In contrast, with the control strain RM319 this difference was found to be in accordance with the NADH oxidation rate via nitrate respiration, assuming that here no flux through PDHc occurred. The calculated fluxes are presented in Fig. 4c.
FIG. 3.
Production rates of lactate, acetate, and ethanol and the rate of NADH oxidation via nitrate respiration (accept) in anaerobic glucose-limited chemostat cultures of E. coli MC4100, RM201, and RM319 with 5 mM nitrate added to the medium. Strains were grown at a dilution rate of 0.1 h−1 at pH 6.5 at 35°C.
(iii) Growth in the presence of oxygen.
Under fully aerobic conditions, the PFL is inactivated (19), and the conversion of pyruvate is solely catalyzed by the PDHc. In Table 1, the specific consumption and production rates in aerobic chemostat cultures are given. As expected with glucose-limited cultures (10), with the wild-type strain all glucose is catabolized completely to CO2 and the same was found for E. coli RM201. Strain RM319 behaved quite differently. Lacking the ability to feed the TCA cycle with acetyl-CoA, part of the glycolytically formed pyruvate was reduced to lactate, but also high concentrations of pyruvate were found in the effluent (up to 50 mM). The data on this strain in Table 1 do not present a redox-balanced product profile. We have no explanation for this. In this context it should be mentioned that the organisms were difficult to grow under these conditions, and often a long lag phase was observed. Similar observations have been made (2, 5) with another mutant lacking PDHc. Increased expression of pyruvate oxidase or the addition of acetate to the culture medium (5) resulted in better growth. This may answer the question as to how CO2 is formed by RM319. Pyruvate oxidase is not active when cells express PDHc but can partially replace PDHc (2). In addition, its activity would yield also the necessary C2 compounds needed for biosynthesis.
TABLE 1.
Specific rates of substrate utilization and product formation in glucose-limited chemostat cultures of various E. coli strainsa
| Strain | Substrate utilization-product formation rate (mmol g [dry wt]−1 h−1)
|
||||
|---|---|---|---|---|---|
| qglc | qpyr | qlac | qCO2 | qO2 | |
| MC4100 | −1.4 | 0.0 | 0.0 | 4.0 | 3.8 |
| RM201 | −1.7 | 0.0 | 0.0 | 4.8 | 4.6 |
| RM319 | −8.1 | 8.0 | 3.8 | 5.7 | 5.3 |
Strains were grown aerobically at D = 0.1 h−1 at pH 6.5. Subscripts: glc, glucose; pyr, pyruvate; lac, lactate. The values are the means of at least four independent measurements. The standard deviation is ±10%.
E. coli MC4100 was also cultured under conditions with various steady-state DOT values. From the observed steady-state fluxes (Table 2) it can be concluded that at 1 and 0.5% DOT values, fermentation and respiration occur simultaneously, as can be deduced by the production of formate and consumption of oxygen. In these microaerobic cultures, significant amounts of acetate and ethanol are being produced. In conclusion, under these conditions NADH is reoxidized by fermentative ethanol production as well as by respiratory activity. Again, from the rate of oxygen reduction it can be concluded that additional NADH generation occurred during glucose catabolism, presumably partially by invoking PDHc activity. However, as the TCA cycle is expected to be active as well, the data do not allow for a quantification of the contribution of PDHc to the formation of acetyl-CoA.
TABLE 2.
Steady-state fluxes in glucose-limited chemostat cultures of E. coli MC4100a
| DOT (%) | Steady-state flux (mmol h−1 g [dry wt]−1)
|
|||||||
|---|---|---|---|---|---|---|---|---|
| qglc | qsucc | qlac | qform | qac | qEtOH | qCO2 | qO2 | |
| 10.0 | −4.1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 8.9 | 9.0 |
| 5.0 | −3.8 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 8.6 | 8.4 |
| 2.0 | −3.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.1 | 8.6 | 8.8 |
| 1.0 | −15.4 | 2.4 | 0.2 | 11.3 | 12.8 | 8.1 | 7.6 | 5.9 |
| 0.5 | −12.5 | 2.3 | 0.2 | 4.8 | 11.3 | 6.9 | 11.2 | 5.0 |
Strain MC4100 was grown at D = 0.3 h−1 at pH 6.5. The DOT is given as the percentage of air saturation. The values are the means of at least four independent measurements. The standard deviation is ±10%. Subscripts: glc, glucose; succ, succinate; lac, lactate; form, formate; EtOH, ethanol.
Internal redox state.
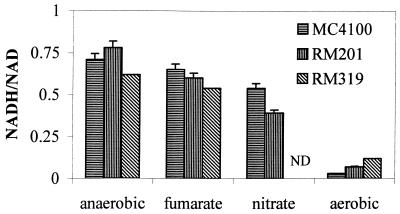
For all growth conditions tested, the steady-state internal redox state, as reflected by the NADH/NAD ratio, was determined. The results are shown in Fig. 5 (for all strains grown under fermentative or respiratory conditions) and Fig. 6 (for the wild-type strain grown at various DOT values). With both strains MC4100 and RM201 it was found that the total dinucleotide pool did not vary significantly (3.1 μmol · g [dry weight]−1 ± 10%) for the conditions tested, whereas for RM319 a total pool was determined that ranged from 3.4 μmol · g (dry weight)−1 under fully aerobic conditions to 5.9 μmol · g (dry weight)−1 under anaerobic conditions. For all strains, the highest ratios were seen during fermentative growth, irrespective of the route of pyruvate conversion. Fumarate respiration resulted in a slightly lower NADH/NAD ratio, and nitrate respiration resulted in a further lowering of the ratio. It was observed that a further increase of the nitrate concentration in the growth medium (7.5 mM) resulted in an even lower NADH/NAD ratio (0.27), but it should be stressed that no steady state could be obtained. When the cells were grown under fully aerobic conditions, the NADH/NAD ratio dropped to values roughly 10-fold lower than those found for fermenting cells. For strain RM319 a higher ratio was observed.
FIG. 5.
NADH/NAD ratios in E. coli MC4100, RM201, and RM319 grown in glucose-limited chemostat cultures (D = 0.1, pH = 6.5) with no acceptor (anaerobic), with 25 mM fumarate, with 5 mM nitrate, or under fully aerobic conditions. The values are the means of at least four independent measurements. ND, not determined.
FIG. 6.
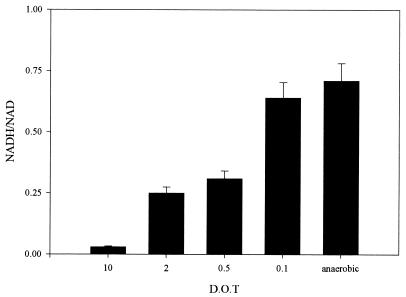
NADH/NAD ratio versus DOT in glucose-limited chemostat cultures of E. coli MC4100 (D = 0.3, pH = 6.5). The values are the means of at least four independent measurements.
In Fig. 6 the NADH/NAD ratios of wild-type cells grown under semiaerobic conditions are presented. At DOT values below 0.5% the NADH/NAD ratio increased sharply. This increase coincides with the fermentative catabolism becoming the major route of glucose breakdown.
PDHc subunit synthesis and in vitro activity.
The overall in vitro activity of PDHc was determined for various growth conditions. It can be seen (Table 3) that the activity, i.e., the cellular content of PDHc, increased when growth occurred in the presence of fumarate; it increased even more so with nitrate and was highest under fully aerobic conditions. No significant differences in activity were found when the availability of oxygen was varied (data not shown).
TABLE 3.
In vitro measurements of PDHc in E. coli RM201 and MC4100 cell extracta
| Strain | PDHc (nmol of ferricyanide reduced/min/mg of protein)
|
|||
|---|---|---|---|---|
| Anaerobic | Fumarate | Nitrate | Aerobic | |
| RM201 | 26 ± 2.1 | 42 ± 3.1 | 83 ± 4.2 | 131 ± 8.5 |
| MC4100 | 22 ± 1.4 | 30 ± 5.3 | 67 ± 4.6 | 128 ± 6.7 |
Cells were obtained from anaerobic glucose-limited chemostat cultures with nitrate (5 mM), with fumarate (25 mM), with no electron acceptor (anaerobic), or aerobically (pH = 6.5, D = 0.1 h−1) as indicated.
Furthermore, a similar trend was found with regard to the expression of dihydrolipoamide transacetylase (the E2 subunit of PDHc) as determined for a limited number of growth conditions by specifically assaying the activity of this subunit (Table 4). The control strain RM319 showed no detectable activity, whereas for both other strains it was found that aerobiosis resulted in a four- to fivefold increase in E2 expression compared to results seen under fermentative conditions.
TABLE 4.
In vitro measurements of the E2 component of the PDHc in E. coli MC4100, RM201, or RM319 cell extracta
| Strain | E2 component of PDHc (nmol of acetyl lipoate formed/min/mg of protein)
|
|||
|---|---|---|---|---|
| Anaerobic | Fumarate | Nitrate | Aerobic | |
| MC4100 | 24 ± 5.4 | ND | ND | 98 ± 5.8 |
| RM201 | 25 ± 2.1 | 51 ± 4.1 | 85 ± 3.2 | 116 ± 6.2 |
| RM319 | <1 | <1 | <1 | <1 |
Cells were obtained from anaerobic chemostat cultures with nitrate (5 mM), with fumarate (12 mM), with no electron acceptor (anaerobic), or in an aerobic chemostat culture as indicated (pH = 6.5, D = 0.1 h−1). ND, not determined.
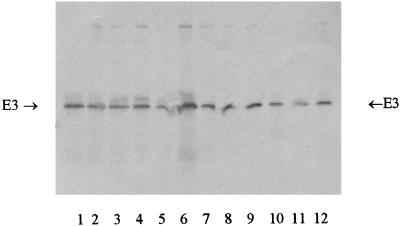
Finally, the synthesis of the E3 (dihydrolipoamide dehydrogenase) subunit of the PDH complex was specifically assayed by Western blotting of cell extracts of cells grown under the conditions as outlined above. As can be seen in Fig. 7, no dramatic changes in the amount of the E3 component were observed for any condition or in the various strains. It should be noted that E. coli RM319 is mutated in the E1 and E2 components (18) and that the E3 component is under the control of a specific promoter. Thus, even in anaerobic cells the E3 subunit was expressed, although there was no in vivo PDHc activity in these cultures.
FIG. 7.
Western blot analysis of the E3 component of the PDHc in cell extracts of different chemostat cultures of E. coli strains. Lanes: 1, RM319, aerobic; 2, MC4100, anaerobic with fumarate; 3 and 4, MC4100, anaerobic with nitrate; 5, MC4100, anaerobic; 6, RM201, aerobic; 7 and 8, RM201, anaerobic with nitrate; 9, anaerobic with fumarate; 10 and 11, RM201, anaerobic; 12, MC4100, aerobic.
DISCUSSION
In facultatively aerobic organisms, pyruvate conversion constitutes a major branch point between the fermentative and respiratory modes of catabolism. Central to the relative flux distributions at this point are the relative in vivo activities of pyruvate dehydrogenase and PFL. In this study the in vivo flux distribution through these enzymes has been inferred, depending on the availability of terminal electron acceptors. First, it is concluded that the internal steady-state redox state of the cell, as reflected by the NADH/NAD ratio, is strongly influenced by the availability and nature of external electron acceptors, as it is in E. faecalis (28). Second, a positive correlation is established between the redox state and the PDHc flux: under more oxidizing conditions the flux is higher. In this context, the environmental redox state is operationally defined as the midpoint potential of the terminal electron acceptor used by the cell. This simplification holds only for redox couples for which the cells have a high kinetic capacity and for conditions in which the actual potential does not deviate too much from the midpoint potential. This latter constraint is masked by the great differences in midpoint potentials of the different acceptors used in this study.
It has been assumed that in the family Enterobacteriaceae, PDHc is active under aerobic conditions only (7), but from the flux calculations presented here, the conclusion can be drawn that in E. coli this enzyme complex can contribute to catabolism in the absence of oxygen, as has been suggested before by Kaiser and Sawers (17). Hence, the general conclusion is valid that PDHc activity is not dependent on the presence of oxygen per se but rather on the external redox condition. Our studies indicate further that the cellular response to the redox state of the environment is mediated by the internal redox state as reflected in the NADH/NAD ratio. Our conclusion that the in vivo activity of the PDHc is controlled by the cell’s redox state is in agreement with the rate of an NAD-dependent reaction being related to the actual redox potential of the acceptor. More important, however, is the fact that our results indicate that control of PDHc synthesis is exerted by a redox-dependent regulation of E2 synthesis. Whether this regulation is exerted by NADH per se cannot be concluded here but it should be remarked that such a type of regulation resembles strongly the proposition made for the role of the NADH/NAD ratio with regard to the synthesis of alcohol dehydrogenase (21, 22). Considering the steps involved in pyruvate oxidation by PDHc, it may seem surprising that it was observed that the synthesis of the E3 subunit, which catalyzes a redox-dependent reaction, apparently is not strongly affected by the redox state of the cell. However, E3 is also involved in the synthesis of branched amino acid synthesis (25, 26), and hence the anabolic activity would be hampered if synthesis of this subunit were redox dependent.
The question remains by what mechanism PDHc synthesis is repressed and/or activated. In this context, it is interesting to note that Iuchi (11) found that in vitro phosphorylation of the sensor (ArcB) of the ArcA/B two-component regulatory system was enhanced by NADH. In addition, it is known (24) that PDHc expression is repressed by a phosphorylation cascade via the Arc system, whereas derepression occurs when the Arc system is in its unphosphorylated form. This is compatible with the results presented here as well as with Iuchi’s finding (11) that nitrate addition to anaerobic cultures resulted in the induction of two enzymes related to respiratory catabolism, i.e., succinate dehydrogenase and the flavin-linked d-lactate dehydrogenase. It may very well be that under conditions in which reoxidation of NADH is hampered by a lack of electron acceptor (fermentative catabolism) or occurs at a decreased rate in the presence of acceptors with a lower midpoint potential (anaerobic respiration), a buildup of reduced nucleotides serves as a signal for the repression of PDHc via the Arc system. It should not be excluded that other catabolic intermediates may have a signaling role and that the presumed effect of NAD(H) is an indirect one. Considering the kinetic effects of a changed NAD(H) level on the various enzymes involved, one may expect the changes in the availability of acceptors to be accompanied by changes in intracellular concentrations of intermediates such as pyruvate, lactate, and acetyl-CoA. Interestingly, these metabolites have been proposed by Iuchi (11) as having a signaling function for the ArcA-ArcB system. Whether this is indeed the case is currently under investigation. However, in view of the fact that pyridine nucleotides play a central role in both respiration (as electron donors to the respiratory chain) and fermentation (as they govern product formation by the demand of redox neutrality), it seems logical to attribute to them a regulatory role in the synthesis of those enzymes for which they serve as a substrate.
In the microaerobic cultures studied here, an interesting feature of catabolism is seen: simultaneous respiratory and fermentative catabolism. Surprisingly, PFL is active and since it is known that the PFL is irreversibly damaged by oxygen (19), this observation suggests that under microaerobic conditions the respiratory activity results in maintaining a cytoplasmic environment that is sufficiently anaerobic to allow PFL to function. It should be noted that with regard to the synthesis of PFL, Tseng et al. (34) have reported that under microaerobic conditions (<10% air saturation) many anaerobic respiratory enzymes are induced under the control of FNR, as is PFL.
In principle, carbon fluxes from pyruvate can be distributed over three enzymes: PFL, PDHc, and lactate dehydrogenase (LDH) (the flux through pyruvate oxidase being minor [2, 5]). The absence of in vivo flux via the LDH in MC4100 and RM319 under anaerobic conditions is puzzling. In vitro activity of the enzyme is high (a rate of ca. 400 nmol/min/mg protein was measured in cell extracts under saturating conditions), and pyruvate and NADH are available (2 mM and 1.4 μmol/g [dry weight]). Yet, in glucose-limited-grown cells, no flux through the LDH is observed. This suggests some downregulation of the activity of LDH that is controlled by the limiting availability of the carbon and energy source (under glucose excess conditions, likely to occur in batch cultures, invariably lactate is formed).
We and others (17) observed that under all conditions tested, PDHc is synthesized and the level of expression varies only about four- to fivefold between fully aerobic and fermentative conditions. We consider the availability of this complex to be yet another example of maximization of metabolic flexibility so often encountered with microorganisms (32). Although regulation of metabolic activity at the level of gene expression may optimize the cell’s enzymatic make-up for a particular steady-state condition, the constitutive presence of apparently redundant enzymes guarantees a rapid response to sudden changes in the environment. Thus, in the case of pyruvate metabolism, the cell can cope with sudden transitions to aerobic conditions since the immediate and irreversible inactivation of PFL can be compensated for by the PDHc, provided that the kinetic constraint caused by the high NADH/NAD ratio is relieved. In this context, it is not surprising that it has been observed that cells grown under fully fermentative conditions retain significant respiratory capacity (3). The time scales at which these responses occur under aerobic-anaerobic (and vice versa) transitions and the accompanying intracellular changes are currently under investigation.
ACKNOWLEDGMENTS
We thank Bas de Bruijn, Heleen Goorissen, Morrow Zalmijn (University of Amsterdam), and Adrie Westphal (Agricultural University, Wageningen, The Netherlands) for helping with the experiments and Gary Sawers (John Innes Centre, Norwich, England) for providing the strains.
REFERENCES
- 1.Casabadan M J, Cohen S N. Lactose genes fused to exogenous promoters in one step using a Mu-lac bacteriophage: in vivo probe for transcriptional control sequences. Proc Natl Acad Sci USA. 1979;76:4530–4533. doi: 10.1073/pnas.76.9.4530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chang Y-Y, Cronan J E., Jr Genetic and biochemical analyses of Escherichia coli strains having a mutation in the structural gene (poxB) for pyruvate oxidase. J Bacteriol. 1983;154:756–762. doi: 10.1128/jb.154.2.756-762.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.De Graef M R. Ph.D. thesis. Amsterdam, The Netherlands: University of Amsterdam; 1999. [Google Scholar]
- 4.Evans C G T, Herbert D, Tempest D W. The continuous culture of microorganisms. 2. Construction of a chemostat. Methods Microbiol. 1970;2:277–327. [Google Scholar]
- 5.Grabau C, Cronan J E. Molecular cloning of the gene (poxB) encoding the pyruvate oxidase of Escherichia coli, a lipid-activated enzyme. J Bacteriol. 1984;160:1088–1092. doi: 10.1128/jb.160.3.1088-1092.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Green J, Guest J R. Activation of FNR-dependent transcription by iron: an in vitro switch for FNR. FEMS Microbiol Lett. 1993;113:219–222. doi: 10.1111/j.1574-6968.1993.tb06517.x. [DOI] [PubMed] [Google Scholar]
- 7.Guest J R, Angier S J, Russell G C. Structure, expression and protein engineering in the pyruvate dehydrogenase complex of Escherichia coli. Ann N Y Acad Sci. 1989;573:76–99. doi: 10.1111/j.1749-6632.1989.tb14988.x. [DOI] [PubMed] [Google Scholar]
- 8.Hansen R G, Henning U. Regulation of pyruvate dehydrogenase activity in Escherichia coli K12. Biochim Biophys Acta. 1966;122:355–358. doi: 10.1016/0926-6593(66)90076-2. [DOI] [PubMed] [Google Scholar]
- 9.Herbert D, Phipps P J, Strange R E. Chemical analysis of microbial cells. Methods Microbiol. 1971;56:209–344. [Google Scholar]
- 10.Holms H. Flux analysis and control of the central metabolic pathways in Escherichia coli. FEMS Microbiol Rev. 1996;19:85–116. doi: 10.1111/j.1574-6976.1996.tb00255.x. [DOI] [PubMed] [Google Scholar]
- 11.Iuchi S. Phosphorylation/deposphorylation of the receiver module at the conserved aspartate residue controls transphosphorylation activity of histidine kinase in sensor protein ArcB of Escherichia coli. J Biol Chem. 1993;268:23972–23980. [PubMed] [Google Scholar]
- 12.Iuchi S, Aristarkhov A, Dong J M, Taylor J S, Lin E C C. Effects of nitrate respiration on expression of the Arc-controlled operons encoding succinate dehydrogenase and flavin-linked l-lactate dehydrogenase. J Bacteriol. 1994;176:1695–1701. doi: 10.1128/jb.176.6.1695-1701.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Iuchi S, Lin E C C. arcA (dye), a global regulatory gene in Escherichia coli mediating repression of enzymes in aerobic pathways. Proc Natl Acad Sci USA. 1988;85:1888–1894. doi: 10.1073/pnas.85.6.1888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Iuchi S, Lin E C C. Adaptation of Escherichia coli to redox environments by gene expression. Mol Microbiol. 1993;9:9–15. doi: 10.1111/j.1365-2958.1993.tb01664.x. [DOI] [PubMed] [Google Scholar]
- 15.Iuchi S, Weiner L. Cellular and molecular physiology of E. coli in the adaptation to aerobic environments. J Biochem. 1996;120:1055–1063. doi: 10.1093/oxfordjournals.jbchem.a021519. [DOI] [PubMed] [Google Scholar]
- 16.Jordan P A, Thomson A J, Ralph E T, Guest J R, Green J. FNR is a direct oxygen sensor having a biphasic response curve. FEBS Lett. 1997;416:349–352. doi: 10.1016/s0014-5793(97)01219-2. [DOI] [PubMed] [Google Scholar]
- 17.Kaiser M, Sawers G. Pyruvate formate-lyase is not essential for nitrate respiration by Escherichia coli. FEMS Microbiol Lett. 1994;117:163–168. doi: 10.1111/j.1574-6968.1994.tb06759.x. [DOI] [PubMed] [Google Scholar]
- 18.Khoroshilova N, Beinert H, Kiley P J. Association of a polynuclear iron-sulfur center with a mutant FNR protein enhances DNA binding. Proc Natl Acad Sci USA. 1995;92:2499–2503. doi: 10.1073/pnas.92.7.2499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Knappe J, Sawers G. A radical-chemical route to acetyl-CoA: the anaerobically induced pyruvate formate-lyase system of Escherichia coli. FEMS Microbiol Rev. 1990;75:383–398. doi: 10.1111/j.1574-6968.1990.tb04108.x. [DOI] [PubMed] [Google Scholar]
- 20.Lynch A S, Lin E C C. Responses to molecular oxygen. In: Neidhardt F C, Curtiss III R, Ingraham J L, Lin E C C, Low K B Jr, Magasanik B, Reznikoff W S, Riley M, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella: cellular and molecular biology. 2nd ed. Washington, D.C: ASM Press; 1996. pp. 1526–1538. [Google Scholar]
- 21.Leonardo M R, Cunningham P R, Clark D P. Anaerobic regulation of the adhE gene, encoding the fermentative alcohol dehydrogenase of Escherichia coli. J Bacteriol. 1993;175:870–878. doi: 10.1128/jb.175.3.870-878.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Leonardo M R, Dailly Y, Clark D P. Role of NAD regulating the adhE gene of Escherichia coli. J Bacteriol. 1996;178:6013–6018. doi: 10.1128/jb.178.20.6013-6018.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mattevi A, de Kok A, Perham R N. The pyruvate dehydrogenase multienzyme complex. Curr Opin Struct Biol. 1992;2:877–887. [Google Scholar]
- 24.Quail M A, Haydon D J, Guest J R. The pdhR-aceEF-lpd operon of Escherichia coli expresses the pyruvate dehydrogenase complex. Mol Microbiol. 1994;2:95–104. doi: 10.1111/j.1365-2958.1994.tb00998.x. [DOI] [PubMed] [Google Scholar]
- 25.Randle P J, Patson P A, Espinal J. Branched-chain ketoacid dehydrogenase. In: Boyer P D, Krebs E G, editors. The enzymes. Vol. 18. Orlando, Fla: Academic Press, Inc.; 1987. pp. 97–121. [Google Scholar]
- 26.Reed L J, Yeaman S J. Pyruvate dehydrogenase. In: Boyer P D, Krebs E G, editors. The enzymes. Vol. 18. Orlando, Fla: Academic Press, Inc.; 1987. pp. 77–96. [Google Scholar]
- 27.Sawers G, Böck A. Anaerobic regulation of pyruvate formate-lyase from Escherichia coli K-12. J Bacteriol. 1988;170:5330–5336. doi: 10.1128/jb.170.11.5330-5336.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Snoep J L, Teixeira de Mattos M J, Postma P W, Neijssel O M. Involvement of pyruvate dehydrogenase in product formation in pyruvate limited anaerobic chemostat cultures of Enterococcus faecalis NCTC 775. Arch Microbiol. 1990;154:50–55. doi: 10.1007/BF00249177. [DOI] [PubMed] [Google Scholar]
- 29.Snoep J L, Teixeira de Mattos M J, Neijssel O M. Effect of the energy source on the NADH/NAD ratio and pyruvate catabolism in anaerobic chemostate cultures of Enterococcus faecalis NCTC 775. FEMS Microbiol Lett. 1991;81:63–66. [Google Scholar]
- 30.Spiro S, Guest J R. FNR and its role in oxygen-related gene expression in Escherichia coli. FEMS Microbiol Rev. 1990;75:399–428. doi: 10.1111/j.1574-6968.1990.tb04109.x. [DOI] [PubMed] [Google Scholar]
- 31.Stewart V. Nitrate regulation of anaerobic respiratory gene expression in Escherichia coli. Mol Microbiol. 1993;9:425–434. doi: 10.1111/j.1365-2958.1993.tb01704.x. [DOI] [PubMed] [Google Scholar]
- 32.Teixeira de Mattos M J, Neijssel O M. Bioenergetic consequences of microbial adaptation to low nutrient environments. J Biotechnol. 1997;59:117–126. doi: 10.1016/s0168-1656(97)00174-0. [DOI] [PubMed] [Google Scholar]
- 33.Teixeira de Mattos M J, Tempest D W. Metabolic and energetic aspects of the growth of Klebsiella aerogenes NCTC 418 on glucose in anaerobic chemostat cultures. Arch Microbiol. 1983;134:80–85. doi: 10.1007/BF00429412. [DOI] [PubMed] [Google Scholar]
- 34.Tseng C-P, Albrecht J, Gunsalus R. Effect of microaerophilic cell growth conditions on expression of the aerobic (cyoABCDE and cydAB) and anaerobic (narGHJI, frdABCD, and dmsABC) respiratory pathway genes in Escherichia coli. J Bacteriol. 1996;178:1094–1098. doi: 10.1128/jb.178.4.1094-1098.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tsuzuki M, Ishige K, Mizuno T. Phosphotransfer circuitry of the putative multi-signal transducer, ArcB, of Escherichia coli: in vitro studies with mutants. Mol Microbiol. 1995;18:953–962. doi: 10.1111/j.1365-2958.1995.18050953.x. [DOI] [PubMed] [Google Scholar]
- 36.Varenne S, Casse F, Chippaux M, Pascal M C. A mutant of Escherichia coli deficient in pyruvate formate lyase. Mol Gen Genet. 1975;141:181–184. doi: 10.1007/BF00267683. [DOI] [PubMed] [Google Scholar]