Abstract
Background
Interleukin 6 (IL-6) signaling is being investigated as a therapeutic target for atherosclerotic cardiovascular disease (CVD). While changes in circulating high-sensitivity C-reactive protein (hsCRP) are used as a marker of IL-6 signaling, it is not known whether there is effect heterogeneity in relation to baseline hsCRP levels or other cardiovascular risk factors. The aim of this study was to explore the association of genetically predicted IL-6 signaling with CVD risk across populations stratified by baseline hsCRP levels and cardiovascular risk factors.
Methods
Among 397,060 White British UK Biobank participants without known CVD at baseline, we calculated a genetic risk score for IL-6 receptor (IL-6R)-mediated signaling, composed of 26 variants at the IL6R gene locus. We then applied linear and non-linear Mendelian randomization analyses exploring associations with a combined endpoint of incident coronary artery disease, ischemic stroke, peripheral artery disease, aortic aneurysm, and cardiovascular death stratifying by baseline hsCRP levels and cardiovascular risk factors.
Results
The study participants (median age 59 years, 53.9% females) were followed-up for a median of 8.8 years, over which time a total of 46,033 incident cardiovascular events occurred. Genetically predicted IL-6R-mediated signaling activity was associated with higher CVD risk (hazard ratio per 1-mg/dL increment in absolute hsCRP levels: 1.11, 95% CI: 1.06–1.17). The increase in CVD risk was linearly related to baseline absolute hsCRP levels. There was no evidence of heterogeneity in the association of genetically predicted IL-6R-mediated signaling with CVD risk when stratifying the population by sex, age, body mass index, estimated glomerular filtration rate, or systolic blood pressure, but there was evidence of greater associations in individuals with low-density lipoprotein cholesterol ≥ 160 mg/dL.
Conclusions
Any benefit of inhibiting IL-6 signaling for CVD risk reduction is likely to be proportional to absolute reductions in hsCRP levels. Therapeutic inhibition of IL-6 signaling for CVD risk reduction should therefore prioritize those individuals with the highest baseline levels of hsCRP.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12916-022-02446-6.
Keywords: Inflammation, Interleukin-6, Atherosclerosis, Cardiovascular disease, C-reactive protein, Cytokines, Human genetics, Mendelian randomization
Background
Chronic inflammation is an emerging therapeutic target for cardiovascular disease (CVD) [1]. Among pharmacological candidates, agents impacting interleukin (IL)-6 signaling have attracted attention due to converging evidence supporting the relevance of IL-6 in atherosclerosis [2]. Data from the Canakinumab Anti-inflammatory Thrombosis Outcome Study (CANTOS) trial showed that the cardiovascular benefit of IL-1β inhibition with canakinumab was proportional to the reductions in IL-6 and high-sensitivity C-reactive protein (hsCRP) levels [3]. A recent phase 2 trial found that ziltivekimab, a monoclonal antibody directly inhibiting IL-6, effectively and safely reduces biomarkers of inflammation and thrombosis among patients with chronic kidney disease [4]. While indirectly targeting the IL-6 pathway with canakinumab led to hsCRP reductions of 35–40% [3], monthly subcutaneous administration of ziltivekimab resulted in a decrease of hsCRP by 77–92% [4]. However, it remains unknown whether larger hsCRP reductions will translate to greater reductions in CVD risk [4], and the ongoing phase 3 cardiovascular outcomes trial testing ziltivekimab will not be completed before 2025 [5].
Mendelian randomization (MR) leverages genetic variants to investigate the effect of exposures on outcomes and can be applied to explore the therapeutic potential of specific drug targets [6]. The random allocation of genetic variants at conception makes this approach less vulnerable to confounding and reverse causation that can impede causal inference in traditional epidemiological investigations [6, 7]. In this study, we performed MR analyses in 397,060 White British UK Biobank participants to investigate (i) the effect of IL-6 receptor (IL-6R)-mediated signaling on CVD risk in relation to baseline hsCRP levels and (ii) whether the effect of IL-6R-mediated signaling on CVD risk varies across population subgroups stratified by cardiovascular risk factors. These results may inform on patient subgroups for inclusion into interventional trials targeting IL-6 signaling for reducing CVD risk.
Methods
Study population
This study follows the reporting recommendations by the STROBE-MR Guidelines (Research Checklist) [8]. Analyses were performed in UK Biobank (application #2532), a prospective cohort study of 502,460 individuals aged 37–73 years recruited between 2006 and 2010. The UK Biobank obtained approval from the Northwest Multi-Center Research Ethics Committee. All participants provided written informed consent. The current analysis was based on White British individuals of European genetic ancestry without known CVD with available genetic, biomarker, and outcome data (Table 1).
Table 1.
Baseline characteristics of the UK Biobank participants included in our analyses stratified by the median IL-6R-mediated signaling genetic score
| Variable | IL-6R-mediated signaling GRS < median | IL-6R-mediated signaling GRS > median | p-value | ||
|---|---|---|---|---|---|
| Age, years | 59 | 51–64 | 59 | 51–64 | 0.983 |
| Sex, females | 114,383 | 52.8 | 114,563 | 52.9 | 0.585 |
| CRP, mg/dL | 1.28 | 0.63–2.66 | 1.45 | 0.71–2.98 | < 2.2 × 10−16 |
| SBP, mmHg | 137 | 126–151 | 137 | 126–151 | 0.218 |
| DBP, mmHg | 77 | 84–92 | 77 | 84–92 | 0.755 |
| BMI, kg/m2 | 26.8 | 24.2–30.0 | 26.8 | 24.2–30.0 | 0.847 |
| eGFR, mL/min/1.73 m2 | 88.3 | 76.5–100.1 | 88.2 | 76.3–100.0 | 0.062 |
| HbA1c, % | 5.36 | 5.15–5.61 | 5.38 | 5.15–5.62 | 4.1 × 10−07 |
| LDL-cholesterol, mg/dL | 136.5 | 114.1–159.9 | 136.3 | 113.8–159.8 | 0.038 |
| HDL-cholesterol, mg/dL | 53.9 | 45.1–64.6 | 53.8 | 45.1–64.5 | 0.131 |
| Lipid-lowering drug use | 37,296 | 17.2 | 37,742 | 17.4 | 0.044 |
| Antidiabetic drug use | 6,632 | 3.0 | 6,913 | 3.2 | 0.014 |
| Antihypertensive drug use | 47,587 | 22.0 | 47,615 | 22.0 | 0.920 |
The results represent median (interquartile range) or N (%). The p-values are derived from the Mann–Whitney U test for quantitative variables and the chi-square test for binary variables and test the null hypothesis that there is no difference in the listed phenotype by median IL6R signaling genetic risk score (GRS)
Genetic instruments
The genetic risk score (GRS) for IL-6 receptor (IL-6R)-mediated signaling was created as previously described [9–11] and included 26 variants 300 kB within the IL6R gene (clumped at pairwise r2 < 0.1) that were associated with hsCRP, a downstream biomarker of IL-6 signaling (Additional file 1: Table S1, Additional file 2: Fig. S1). As previously described [12], we meta-analyzed a genome-wide association study (GWAS) for hsCRP levels in 204,402 European ancestry individuals (Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) Consortium) [13] with data from 318,279 White British individuals in the UK Biobank [14]. This meta-analysis was performed to maximize the number of genetic variants to be leveraged as instruments for IL-6 signaling, in turn optimizing the power of the MR analysis. We selected variants associated with hsCRP levels (p < 5 × 10−8) after clumping for linkage disequilibrium at r2 < 0.1 (1000G European reference panel). We then created a genetic risk score (GRS) for IL-6R-mediated signaling, using association estimates from the CHARGE GWAS as weights for the 26 identified variants (Additional file 1: Table S1). As weights for the GRS were taken from a population that did not overlap with UK Biobank, risk of weak instrument bias related to participant overlap was minimized [15]. This GRS is associated with other biomarkers of upregulated IL-6 signaling as well (lower circulating IL-6 and soluble IL-6R levels), as has been previously described (Additional file 2: Fig. S1) [12].
Study outcomes
Genetic data were linked to inpatient hospital episode records, primary care data and death registry. The outcome considered was a combined CVD endpoint of incident coronary artery disease, ischemic stroke, peripheral artery disease, aortic aneurysm, and cardiovascular death (codes used to define these outcomes in Additional file 1: Table S2). In sensitivity analyses, we examined an alternative outcome excluding aortic aneurysm, because the disease mechanisms might diverge from those of the other outcomes that are primarily related with atherosclerosis [16].
Statistical analysis
We used the ratio of coefficients method to perform MR analyses [17]. This represents the association of the GRS with the outcome divided by the association of the GRS with hsCRP [18]. Cox regression was used to estimate association of the score with outcomes, incorporating age, sex, principal components 1 to 10 of genetic ancestry, genotyping chip, kinship, and assessment center as covariates. Linear regression was used to estimate the association of the GRS with hsCRP, incorporating the same covariates. In sensitivity analyses, we excluded individuals with evidence of relatedness within the UK Biobank cohort (kinship coefficient < 0.0884).
To explore the shape of the association between genetically predicted IL-6R-mediated and CVD risk across individuals with varying baseline hsCRP levels, we stratified the population into strata based on residual hsCRP levels, defined as a participant’s hsCRP minus the genetic contribution to hsCRP from the GRS. Stratifying on hsCRP directly would introduce collider bias to distort estimates [19]. For each stratum, we calculated the MR estimate for the association of genetically proxied IL-6R-mediated signaling with the outcome using the ratio of coefficients method [18]. Using a flexible semiparametric framework [20], we then performed a meta-regression of the linear MR estimates obtained for each decile against the median hsCRP value per decile. A fractional polynomial test was used to investigate whether a non-linear model fit this meta-regression better than a linear model. This analysis was performed for both absolute and ln-transformed hsCRP levels. In alternative analyses, we stratified the analyses in centiles of hsCRP rather than deciles.
To investigate if the associations between genetically proxied IL-6R-mediated signaling and CVD vary depending on levels of other cardiovascular risk factors, we performed MR analyses stratified by sex and age, and residual values of body mass index (BMI), cystatin C-based estimated glomerular filtration (eGFR), glycated hemoglobin (HbA1c), low-density lipoprotein cholesterol (LDL-C), and systolic blood pressure (SBP) [19].
Statistical significance for all analyses was set at a two-sided p-value < 0.05. Statistical analyses were performed in R (v4.1.1).
Results
From 502,460 individuals enrolled to the UK Biobank, a total of 397,060 individuals were included in analyses (Additional file 2: Fig. S2, Table 1). Median age at recruitment was 59 years (interquartile range 51–64) and 53.9% of the participants were female. The median hsCRP levels among UK Biobank participants were 1.35 mg/dL (interquartile range 0.67–2.80, Table 1). Levels of hsCRP followed a right-skewed distribution (Additional file 2: Fig. S3) were higher among females and older individuals, correlated positively with BMI, LDL-C, SBP, and HbA1c, and correlated negatively with eGFR (Additional file 2: Fig. S4). The GRS for IL-6R-mediated signaling was associated with hsCRP levels among both female and male participants (Additional file 2: Fig. S5).
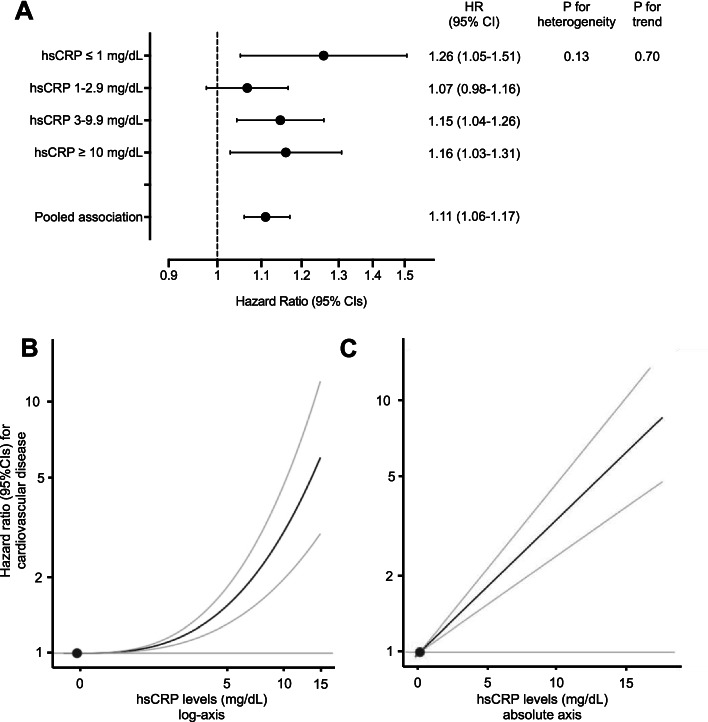
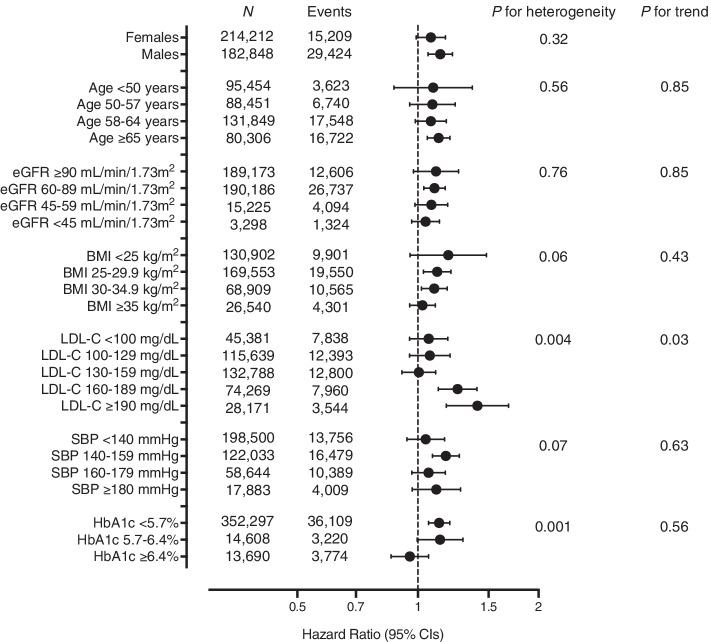
Over a median follow-up of 8.8 years (interquartile range 8.1–9.5 years), there were a total of 46,033 incident CVD events. MR analyses identified significant associations between genetically predicted IL-6R-mediated signaling and risk of the composite CVD outcome (hazard ratio per 1-mg/dL increment in absolute hsCRP levels: 1.11, 95% CI: 1.06–1.17, p = 6.7 × 10−5). This association was similar across individuals with varying baseline hsCRP levels (Fig. 1A) and followed a linear dose–response pattern based with absolute, but not ln-transformed hsCRP levels (Fig. 1B, C). A similar dose–response pattern was observed when stratifying the analyzed UK Biobank population into centiles of hsCRP rather than deciles (Additional file 2: Fig. S6). We observed similar results when excluding aortic aneurysm cases from our main outcome (Additional file 2: Fig. S7). The results also remained materially unchanged when excluding individuals with evidence of relatedness within the UK Biobank (Additional file 2: Fig. S8). Associations were similar in both sexes and across age subgroups. There was no evidence of a trend when stratifying by BMI, eGFR, HbA1c, or SBP (Fig. 2). There was evidence of heterogeneity across subgroups stratified by HbA1c (pheterogeneity = 0.001) and LDL-C levels (pheterogeneity = 0.004), with estimates of greater magnitude in individuals with LDL-C levels ≥ 160 mg/dl (ptrend = 0.03).
Fig. 1.
Associations between genetically predicted IL-6R-mediated signaling and risk of incident cardiovascular disease across measured hsCRP levels. A Mendelian randomization analyses stratified by baseline hsCRP levels. The hazard ratios are scaled for 1 mg/dL increment in absolute hsCRP levels. The p-values for heterogeneity and for trend are derived from the Cochran Q statistic and linear meta-regression analyses across deciles of measured hsCRP. B, C Mendelian randomization analyses of genetically predicted IL6R-mediated signaling and CVD risk across B ln-transformed measured hsCRP levels and C absolute measured hsCRP levels. For B, C, results are obtained from fractional polynomial models across associations derived for deciles of measured hsCRP levels. The reference is set to the minimum hsCRP value in the UK Biobank sample (0.08 mg/dL). The p-values for non-linearity are 0.001 for ln-transformed hsCRP levels and 0.99 for absolute hsCRP levels. For all graphs, the residual values of hsCRP are used to stratify, as determined in models regressing the genetic risk score for IL-6 signaling on measured hsCRP levels
Fig. 2.
Association between genetically predicted IL-6R-mediated signaling activity and risk of cardiovascular disease across clinically relevant subgroups. The hazard ratios are scaled on 1 mg/dL increment in absolute hsCRP levels. The p-values for heterogeneity and for trend are derived from the Cochran Q statistic and linear meta-regression analyses across strata of the different measured variables. For all variables except for age and sex, the residual values are used to stratify, as determined in models regressing the genetic score for IL-6 signaling on these variables
Discussion
Our findings are consistent with a linear dose–response relationship between genetically predicted IL-6R signaling and CVD risk in relation to absolute baseline hsCRP levels. For pharmacological purposes, this translates to greater efficacy against CVD of IL-6 signaling inhibition that achieves larger hsCRP reductions.
Our results expand previous genetic data supporting a causal role of IL-6 signaling on atherosclerotic CVD [9, 10, 21] and are consistent with the known effects of IL-6 signaling on increasing CRP generation [22]. In primary prevention cohorts, circulating levels of both IL-6 and CRP have been found to be independently associated with risk of incident CVD [23–25]. Furthermore, the magnitude of cardiovascular risk reduction in the CANTOS trial was directly related to the degree of IL-6 reduction achieved [3]. Taken together, our current genetic findings add to the body of epidemiological and trial evidence supporting a dose response-relationship between IL-6 signaling mediated CRP lowering and CVD risk reduction. In terms of mechanisms underlying such a dose–response relationship, IL-6 is involved in upregulating cellular adhesion molecules at the vessel wall [26], increasing vascular permeability and disrupting endothelial barrier function [27], and promoting vascular smooth muscle growth [28]. It follows that greater absolute reductions in IL-6 signaling, as measured by hsCRP reduction, would confer greater benefit in cardiovascular risk reduction.
When stratifying on other cardiovascular risk factors, there was evidence of greater CVD risk reduction through IL-6R signaling inhibition in individuals with higher LDL-C. This aligns with the notion that inflammation is the result of lipid accumulation in atherosclerotic plaques, and as such, greater benefits may be expected among patients with high baseline LDL-C levels. However, previous genetic analyses have suggested no departure from additive effects on CHD risk when considering genetic proxies for inhibition of IL-6R signaling and pharmacological LDL-C-lowering [11].
A limitation of this work is that it considered European ancestry individuals and may not translate across other ethnic groups. This is particularly relevant as the risk factors and pathophysiological mechanisms underlying CVD may vary across populations of different ethnic ancestry. Furthermore, we considered individuals without CVD at baseline, and it is unclear how these findings will apply to individuals with established CVD, who are at greater absolute risk and therefore likely to be prioritized for treatment. We also did not consider potential adverse effects of perturbing IL-6 signaling, including implications for allergic, autoimmune and infectious disease [10]. Of note, observational and genetic evidence has supported an association of low CRP levels with increased risk of Alzheimer’s disease [29], and further work is required to delineate the dose–response relationship between IL-6 signaling mediated changes in CRP levels and potential adverse outcomes. Finally, these genetic analyses explore small lifelong effects, and may not be directly extrapolated to short-term clinical interventions [6].
Conclusions
In summary, we find genetic evidence to support that any benefit of pharmacologically inhibiting IL-6 signaling for CVD risk reduction is likely to be proportional to absolute reductions in hsCRP levels. Our results indicate that therapeutic inhibition of IL-6 signaling for CVD risk reduction should prioritize those individuals with the highest baseline levels of hsCRP.
Supplementary Information
Additional file 1: Table S1. Genetic variants included in the genetic risk score for interleukin-6 receptor signaling downregulation and their associations with hsCRP. Table S2. Definition of outcomes in the current analysis.
Additional file 2: Fig. S1. Associations of genetically predicted IL6 receptor-mediated signaling (measured in 1 unit increment in ln-transformed hsCRP levels) with circulating biomarkers. Fig. S2. Selection of study participants. Fig. S3. Distribution of (A) absolute and (B) ln-transformed hsCRP levels in the analyzed UK Biobank population. Fig. S4. Levels of hsCRP levels by vascular risk factors in the analyzed UK Biobank participants. Fig. S5. Levels of high sensitivity C-reactive protein (hsCRP) across deciles of genetic risk score for IL-6 receptor mediated signaling in (A) males and (B) females. Fig. S6. Associations between genetically predicted IL-6R-mediated signaling across centiles of measured hsCRP levels and risk of incident cardiovascular disease. Fig. S7. Associations between genetically predicted IL-6R-mediated signaling and risk of incident cardiovascular disease across measured hsCRP levels after excluding aortic aneurysm cases from the definition of the outcome. Fig. S8. Associations between genetically predicted IL-6R-mediated signaling and risk of incident cardiovascular disease across measured hsCRP levels after excluding individuals with evidence of relatedness within the cohort (kinship coefficient < 0.0884).
Acknowledgements
Not applicable.
Abbreviations
- CVD
Cardiovascular disease
- GWAS
Genome-wide association study
- hsCRP
High-sensitivity C-reactive protein
- IL-6
Interleukin-6
- IL-6R
Interleukin-6 receptor
- MR
Mendelian randomization
Authors’ contributions
MKG and DG conceived and designed the study. MKG performed the statistical analysis. MKG and DG drafted the first version of the manuscript. All authors interpreted the data and revised the manuscript for intellectual content. All authors read and approved the final manuscript.
Funding
MKG is supported by a Walter-Benjamin fellowship from the German Research Foundation (Deutsche Forschungsgemeinschaft [DFG], GZ: GE 3461/1-1) and the FöFoLe program of Ludwig-Maximilians-University Munich (FöFoLe-Forschungsprojekt Reg.-Nr. 1120). This work was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany’s Excellence Strategy within the framework of the Munich Cluster for Systems Neurology (EXC 2145 SyNergy – ID 390857198 to MKG). CDA is supported by the US National Institutes of Health (R01NS103924, U01NS069763) and the American Stroke Association-Bugher Foundation. SB is supported by a Sir Henry Dale Fellowship jointly funded by the Wellcome Trust and the Royal Society (204623/Z/16/Z) and supported by the National Institute for Health Research Cambridge Biomedical Research Centre (BRC-1215-20014). MD received funding from the German Research Foundation (DFG) as part of the Collaborative Research Centre 1123 (B3). DG is supported by the British Heart Foundation Centre of Research Excellence (RE/18/4/34215) at Imperial College London and a National Institute for Health Research Clinical Lectureship at St. George’s, University of London (CL-2020-16-001). The views expressed are those of the authors and not necessarily those of the National Institute for Health Research or the Department of Health and Social Care. This research was funded by United Kingdom Research and Innovation Medical Research Council (MC_UU_00002/7 to DG). For the purpose of open access, SB has applied a Creative Commons Attribution (CC BY) license to any Author Accepted Manuscript version arising from this submission.
Availability of data and materials
Data from the UK Biobank are available for research purposes following submission of a research proposal. The summary statistics used to generate the IL-6 signaling genetic score are provided in Additional file 1: Table S1.
Declarations
Ethics approval and consent to participate
The UK Biobank obtained approval from the Northwest Multi-Center Research Ethics Committee (UK Biobank is 11/NW/0382). All participants provided written informed consent according to the Declaration of Helsinki. Data access for this analysis was granted for application #2532.
Consent for publication
Not applicable.
Competing interests
CDA has received sponsored research support from the American Heart Association, Massachusetts General Hospital, and Bayer AG, and has consulted for ApoPharma. JMMH is employed by Novo Nordisk. TGR, GKH, and DG are employed part-time by Novo Nordisk. DG is an Editorial Board Member of BMC Medicine; he had no role in the handling or peer review of the manuscript. The remaining authors have no conflicts of interest to declare.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Marios K. Georgakis, Email: mgeorgakis@mgh.harvard.edu, Email: marios.georgakis@med.uni-muenchen.de
Dipender Gill, Email: dipender.gill@imperial.ac.uk.
References
- 1.Soehnlein O, Libby P. Targeting inflammation in atherosclerosis - from experimental insights to the clinic. Nat Rev Drug Discov. 2021;20(8):589–610. doi: 10.1038/s41573-021-00198-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ridker PM, Rane M. Interleukin-6 signaling and anti-interleukin-6 therapeutics in cardiovascular disease. Circ Res. 2021;128(11):1728–1746. doi: 10.1161/CIRCRESAHA.121.319077. [DOI] [PubMed] [Google Scholar]
- 3.Ridker PM, MacFadyen JG, Everett BM, Libby P, Thuren T, Glynn RJ, Group CT Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: a secondary analysis from the CANTOS randomised controlled trial. Lancet. 2018;391(10118):319–328. doi: 10.1016/S0140-6736(17)32814-3. [DOI] [PubMed] [Google Scholar]
- 4.Ridker PM, Devalaraja M, Baeres FMM, Engelmann MDM, Hovingh GK, Ivkovic M, Lo L, Kling D, Pergola P, Raj D, et al. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet. 2021;397(10289):2060–2069. doi: 10.1016/S0140-6736(21)00520-1. [DOI] [PubMed] [Google Scholar]
- 5.ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). 2000 Feb 29 - . Identifier NCT05021835, ZEUS - a research study to look at how ziltivekimab works compared to placebo in people with cardiovascular disease, chronic kidney disease and inflammation (ZEUS); August 26, 2021 [cited June 25, 2022]. Available from: https://clinicaltrials.gov/ct2/show/results/NCT05021835.
- 6.Gill D, Georgakis MK, Walker VM, Schmidt AF, Gkatzionis A, Freitag DF, Finan C, Hingorani AD, Howson JMM, Burgess S, et al. Mendelian randomization for studying the effects of perturbing drug targets. Wellcome Open Res. 2021;6:16. doi: 10.12688/wellcomeopenres.16544.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Georgakis MK, Gill D. Mendelian randomization studies in stroke: exploration of risk factors and drug targets with human genetic data. Stroke. 2021;52(9):2992–3003. doi: 10.1161/STROKEAHA.120.032617. [DOI] [PubMed] [Google Scholar]
- 8.Skrivankova VW, Richmond RC, Woolf BAR, Yarmolinsky J, Davies NM, Swanson SA, VanderWeele TJ, Higgins JPT, Timpson NJ, Dimou N, et al. Strengthening the reporting of observational studies in epidemiology using mendelian randomization: the STROBE-MR statement. JAMA. 2021;326(16):1614–1621. doi: 10.1001/jama.2021.18236. [DOI] [PubMed] [Google Scholar]
- 9.Georgakis MK, Malik R, Gill D, Franceschini N, Sudlow CLM, Dichgans M. Interleukin-6 signaling effects on ischemic stroke and other cardiovascular outcomes: a Mendelian randomization study. Circ Genom Precis Med. 2020;13(3):e002872. doi: 10.1161/CIRCGEN.119.002872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Georgakis MK, Malik R, Li X, Gill D, Levin MG, Vy HMT, Judy R, Ritchie M, Verma SS, Regeneron Genetics C et al. Genetically downregulated interleukin-6 signaling is associated with a favorable cardiometabolic profile: a phenome-wide association study. Circulation. 2021;143(11):1177–1180. doi: 10.1161/CIRCULATIONAHA.120.052604. [DOI] [PubMed] [Google Scholar]
- 11.Georgakis MK, Malik R, Burgess S, Dichgans M. Additive effects of genetic IL-6 signaling downregulation and LDL-cholesterol lowering on cardiovascular disease: a 2x2 factorial Mendelian randomization analysis. J Am Heart Assoc. 2021; in press. [DOI] [PMC free article] [PubMed]
- 12.Georgakis MK, Malik R, Burgess S, Dichgans M. Additive effects of genetic interleukin-6 signaling downregulation and low-density lipoprotein cholesterol lowering on cardiovascular disease: a 2x2 factorial Mendelian randomization analysis. J Am Heart Assoc. 2022;11(1):e023277. doi: 10.1161/JAHA.121.023277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ligthart S, Vaez A, Vosa U, Stathopoulou MG, de Vries PS, Prins BP, Van der Most PJ, Tanaka T, Naderi E, Rose LM, et al. Genome analyses of >200,000 individuals identify 58 loci for chronic inflammation and highlight pathways that link inflammation and complex disorders. Am J Hum Genet. 2018;103(5):691–706. doi: 10.1016/j.ajhg.2018.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sinnott-Armstrong N, Tanigawa Y, Amar D, Mars NJ, Aguirre M, Venkataraman GR, Wainberg M, Ollila HM, Pirruccello JP, Qian J et al: Genetics of 38 blood and urine biomarkers in the UK Biobank. bioRxiv 2019:660506. 10.1101/660506.
- 15.Burgess S, Davies NM, Thompson SG. Bias due to participant overlap in two-sample Mendelian randomization. Genet Epidemiol. 2016;40(7):597–608. doi: 10.1002/gepi.21998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Quintana RA, Taylor WR. Cellular mechanisms of aortic aneurysm formation. Circ Res. 2019;124(4):607–618. doi: 10.1161/CIRCRESAHA.118.313187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Burgess S, Small DS, Thompson SG. A review of instrumental variable estimators for Mendelian randomization. Stat Methods Med Res. 2017;26(5):2333–2355. doi: 10.1177/0962280215597579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Burgess S, Thompson SG. Use of allele scores as instrumental variables for Mendelian randomization. Int J Epidemiol. 2013;42(4):1134–1144. doi: 10.1093/ije/dyt093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Coscia C, Gill D, Benitez R, Perez T, Malats N, Burgess S. Avoiding collider bias in Mendelian randomization when performing stratified analyses. Eur J Epidemiol. 2022. 10.1007/s10654-022-00879-0. [DOI] [PMC free article] [PubMed]
- 20.Staley JR, Burgess S. Semiparametric methods for estimation of a nonlinear exposure-outcome relationship using instrumental variables with application to Mendelian randomization. Genet Epidemiol. 2017;41(4):341–352. doi: 10.1002/gepi.22041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Levin MG, Klarin D, Georgakis MK, Lynch J, Liao KP, Voight BF, O’Donnell CJ, Chang KM, Assimes TL, Tsao PS, et al. A missense variant in the IL-6 receptor and protection from peripheral artery disease. Circ Res. 2021;129(10):968–70. [DOI] [PMC free article] [PubMed]
- 22.Ridker PM. From C-reactive protein to interleukin-6 to interleukin-1: moving upstream to identify novel targets for atheroprotection. Circ Res. 2016;118(1):145–156. doi: 10.1161/CIRCRESAHA.115.306656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Papadopoulos A, Palaiopanos K, Bjorkbacka H, Peters A, de Lemos JA, Seshadri S, Dichgans M, Georgakis MK. Circulating interleukin-6 levels and incident ischemic stroke: a systematic review and meta-analysis of prospective studies. Neurology. 2022;98(10):e1002–e1012. doi: 10.1212/WNL.0000000000013274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kaptoge S, Seshasai SR, Gao P, Freitag DF, Butterworth AS, Borglykke A, Di Angelantonio E, Gudnason V, Rumley A, Lowe GD, et al. Inflammatory cytokines and risk of coronary heart disease: new prospective study and updated meta-analysis. Eur Heart J. 2014;35(9):578–589. doi: 10.1093/eurheartj/eht367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Emerging Risk Factors C. Kaptoge S, Di Angelantonio E, Lowe G, Pepys MB, Thompson SG, Collins R, Danesh J. C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: an individual participant meta-analysis. Lancet. 2010;375(9709):132–140. doi: 10.1016/S0140-6736(09)61717-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Romano M, Sironi M, Toniatti C, Polentarutti N, Fruscella P, Ghezzi P, Faggioni R, Luini W, van Hinsbergh V, Sozzani S, et al. Role of IL-6 and its soluble receptor in induction of chemokines and leukocyte recruitment. Immunity. 1997;6(3):315–325. doi: 10.1016/S1074-7613(00)80334-9. [DOI] [PubMed] [Google Scholar]
- 27.Alsaffar H, Martino N, Garrett JP, Adam AP. Interleukin-6 promotes a sustained loss of endothelial barrier function via Janus kinase-mediated STAT3 phosphorylation and de novo protein synthesis. Am J Physiol Cell Physiol. 2018;314(5):C589–C602. doi: 10.1152/ajpcell.00235.2017. [DOI] [PubMed] [Google Scholar]
- 28.Ikeda U, Ikeda M, Oohara T, Oguchi A, Kamitani T, Tsuruya Y, Kano S. Interleukin 6 stimulates growth of vascular smooth muscle cells in a PDGF-dependent manner. Am J Physiol. 1991;260(5 Pt 2):H1713–1717. doi: 10.1152/ajpheart.1991.260.5.H1713. [DOI] [PubMed] [Google Scholar]
- 29.Hegazy SH, Thomassen JQ, Rasmussen IJ, Nordestgaard BG, Tybjaerg-Hansen A, Frikke-Schmidt R. C-reactive protein levels and risk of dementia-observational and genetic studies of 111,242 individuals from the general population. Alzheimers Dement. 2022. 10.1002/alz.12568. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Genetic variants included in the genetic risk score for interleukin-6 receptor signaling downregulation and their associations with hsCRP. Table S2. Definition of outcomes in the current analysis.
Additional file 2: Fig. S1. Associations of genetically predicted IL6 receptor-mediated signaling (measured in 1 unit increment in ln-transformed hsCRP levels) with circulating biomarkers. Fig. S2. Selection of study participants. Fig. S3. Distribution of (A) absolute and (B) ln-transformed hsCRP levels in the analyzed UK Biobank population. Fig. S4. Levels of hsCRP levels by vascular risk factors in the analyzed UK Biobank participants. Fig. S5. Levels of high sensitivity C-reactive protein (hsCRP) across deciles of genetic risk score for IL-6 receptor mediated signaling in (A) males and (B) females. Fig. S6. Associations between genetically predicted IL-6R-mediated signaling across centiles of measured hsCRP levels and risk of incident cardiovascular disease. Fig. S7. Associations between genetically predicted IL-6R-mediated signaling and risk of incident cardiovascular disease across measured hsCRP levels after excluding aortic aneurysm cases from the definition of the outcome. Fig. S8. Associations between genetically predicted IL-6R-mediated signaling and risk of incident cardiovascular disease across measured hsCRP levels after excluding individuals with evidence of relatedness within the cohort (kinship coefficient < 0.0884).
Data Availability Statement
Data from the UK Biobank are available for research purposes following submission of a research proposal. The summary statistics used to generate the IL-6 signaling genetic score are provided in Additional file 1: Table S1.