Abstract
Three decades have passed from the initial discovery of a microRNA (miRNA) in Caenorhabditis elegans to our current understanding that miRNAs play essential roles in regulating fundamental physiological processes and that their dysregulation can lead to many human pathologies, including cancer. In effect, restoration of miRNA expression or downregulation of aberrantly expressed miRNAs using miRNA mimics or anti-miRNA inhibitors (anti-miRs/antimiRs), respectively, continues to show therapeutic potential for the treatment of cancer. Although the manipulation of miRNA expression presents a promising therapeutic strategy for cancer treatment, it is predominantly reliant on nucleic acid-based molecules for their application, which introduces an array of hurdles, with respect to in vivo delivery. Because naked nucleic acids are quickly degraded and/or removed from the body, they require delivery vectors that can help overcome the many barriers presented upon their administration into the bloodstream. As such, in this review, we discuss the strengths and weaknesses of the current state-of-the-art delivery systems, encompassing viral- and nonviral-based systems, with a specific focus on nonviral nanotechnology-based miRNA delivery platforms, including lipid-, polymer-, inorganic-, and extracellular vesicle-based delivery strategies. Moreover, we also shed light on peptide carriers as an emerging technology that shows great promise in being a highly efficacious delivery platform for miRNA-based cancer therapeutics.
Keywords: microRNA, miRNA therapeutics, miRNA delivery, RNA silencing, cancer, cell-penetrating peptides, CPPs
1. Introduction
Since the reported finding of the first human miRNA in 2000 [1] and, subsequently, the discovery of cancer-associated miRNAs several years later [2], miRNAs have come into focus as feasible therapeutic drug candidates and/or therapeutic targets for cancer intervention. With the global incidence of cancer burden and cancer-related deaths rising to 19.3 million and 10 million in 2020, respectively, and due to a predicted ~50% increase in cancer burden worldwide by 2040, according to the most recent GLOBOCAN estimates from the International Agency for Research on Cancer (Global Cancer Observatory; Lyon, France) [3,4], it is evident that continued therapeutic efforts are needed to help combat this devastating disease. Thus, with miRNAs being found to be aberrantly expressed in many cancer types [5,6,7,8,9], it is not surprising that miRNA therapeutics has evolved and developed into a promising approach (via its manipulation of miRNA expression levels) for cancer treatment.
Despite the progress made in the preclinical development of miRNA therapeutics over the past several decades [10,11,12,13], its transition to the clinic still faces significant hurdles. Presently, of the 11 developed miRNA therapeutics that have reached clinical trials (Table 1) [10,12,14,15], four studies have been terminated/discontinued/withdrawn, five have completed phase I or II trials, and two are currently undergoing active phase I or II trials. Notably, though, none have yet reached phase III trials, nor are being apparently further developed and/or advanced for cancer-related treatments. This slow transition to the clinic could be in-part due to the numerous pharmacological challenges confronted by miRNA-based cancer therapies, which can include rapid degradation by ribonucleases in the bloodstream, renal and reticuloendothelial system (RES) clearance, poor penetration into tumor tissues due to mechanical and biological barriers, immunotoxicity, neurotoxicity, cell/tissue-type specific delivery, poor intracellular delivery, and endosomal entrapment, with subsequent degradation in lysosomes [13]. Notwithstanding, continued advancements in RNA chemistry, and in particular, delivery technologies, are enabling miRNA therapies to overcome these limitations, making the application of this form of therapy in the clinic for cancer treatment that much more a possible reality. In this context, the main focus of this review is on the advancements of varied miRNA delivery platforms for cancer intervention, along with a discussion of the emergence of peptides as potential carriers for miRNA-based cancer therapeutics.
Table 1.
Status of miRNA therapeutics-based clinical trials in USA.
| Drug | miRNA Therapeutic | Delivery System | Disease | Status * | Manufacturer |
|---|---|---|---|---|---|
| Lademirsen (RG-012/SAR339375) |
anti-miR-21 | AMO a inhibitor | Alport Syndrome | Phase II (active) NCT02855268 |
Regulus Therapeutics; Genzyme |
| AZD4076/RG-125 | anti-miR-103/107 | GalNAc-conjugated antimiR |
Type 2 diabetes; nonalcoholic fatty liver diseases |
Phase I (active) NCT02612662; Phase I/IIa (completed) NCT02826525 |
AstraZeneca; Regulus Therapeutics |
| Remlarsen (MRG-201) |
miR-29 mimic | Cholesterol-conjugated miRNA duplex |
Keloid | Phase II (completed) NCT03601052 |
miRagen Therapeutics |
| Cobomarsen (MRG-106) |
anti-miR-155 | LNA-modified antimiR |
Lymphomas; Leukemias |
Phase I (completed) NCT02580552; Phase II (terminated) NCT03837457 NCT03713320 |
miRagen Therapeutics |
| MRG-110 | anti-miR-92a | LNA-modified antimiR |
Skin wound | Phase I (completed) NCT03603431 |
miRagen Therapeutics |
| MesomiR-1/ TargomiR |
miR-16 mimic | EDVs-nonliving bacterial minicells |
Mesothelioma; non-small cell lung cancer | Phase I (completed) NCT02369198 |
EnGeneIC; Asbestos Diseases Research Foundation |
| Miravirsen | anti-miR-122 | LNA-modified antimiR |
Hepatitis C | Phase I and II (completed) NCT01646489 NCT01200420 NCT01872936 NCT02031133 NCT02508090 |
Santaris Pharma A/S; Hoffmann-La Roche |
| RGLS4326 | anti-miR-17 | AMO inhibitor | Polycystic kidney disease |
Phase I (completed) NCT04536688 |
Regulus Therapeutics |
| RG-101 | anti-miR-122 | GalNAc-conjugated antimiR |
Hepatitis C | Phase I and II (discontinued) |
Regulus Therapeutics |
| MRX34 | miR-34 mimic | Lipid-based nanoparticle (liposome) |
Cancer | Phase I (terminated) NCT01829971 NCT02862145 |
Mirna Therapeutics |
| pSil-miR200c and PMIS miR200a | Plasmid DNAs encoding miR-200c and a miRNA inhibitor targeting miR-200a |
Biodegradable sponge |
Tooth extraction | Phase I (withdrawn) NCT02579187 |
University of Iowa |
* ClinicalTrials.gov government identifier numbers are listed for each miRNA therapeutic, if applicable. a Abbreviations: AMO, antisense miRNA oligonucleotide; GalNAc, N-acteyl-D-galactosamine; LNA, locked nucleic acid; EDVs, EnGeneIC delivery vehicles.
2. RNA Silencing and miRNAs
RNA silencing is an evolutionary conserved eukaryotic post-transcriptional gene regulatory mechanism by which noncoding RNAs (ncRNAs) induce the sequence-specific inhibition of target gene expression and/or protein synthesis. One particular class of ncRNAs that can induce RNA silencing and affects many cellular processes and developmental pathways are the small (20–25 nucleotides (nt) in length) ncRNAs known as miRNAs [16,17,18,19]. Specifically, miRNAs regulate gene expression by either repressing translation or inducing mRNA degradation, depending on the degree of sequence complementarity with the target mRNAs [16,20]. To date, >60% of human protein-coding genes are predicted to contain miRNA binding sites and the total number of human miRNAs are estimated to be ~2300 [21,22,23,24].
The biogenesis of mature miRNAs initiates from long primary transcripts, termed primary-miRNAs (pri-miRNAs), which harbor hairpin structures comprising the miRNA sequences that are transcribed predominantly by RNA polymerase II, but also by RNA polymerase III [17,20,25]. Many canonical miRNAs are derived from the introns/exons of long ncRNA transcripts, as well as the introns of protein-coding precursor-mRNAs, where these pri-miRNAs can consist of either a single miRNA gene unit or a cluster of often related miRNA genes [17,19,20]. Following transcription, the hairpin structures within the pri-miRNAs are recognized and processed by the nuclear microprocessor complex, comprising an RNase III enzyme, Drosha, in complex with DGCR8, resulting in the liberation of ~70 nt stem-loop precursor miRNAs known as pre-miRNAs [26]. Subsequently, the pre-miRNAs are exported to the cytoplasm by the action of Exportin-5 [27,28,29], where they undergo final processing by another RNase III enzyme, Dicer, to generate ~20 base-pair miRNA duplexes [17,30,31,32]. Afterward, the miRNA duplex is then incorporated into an Argonaute (AGO) protein, where one strand (i.e., the “guide” strand) is selected to become the mature miRNA and its complementary strand (i.e., the “passenger” strand) is discarded, thus forming the mature RNA-induced silencing complex (RISC) [17,19,20,33,34,35]. Strand selection preference is given based on the strand possessing the least stable paired 5′-end [36,37], as well as a 5′-terminal A or U residue [38,39]. Once loaded in the RISC, the mature miRNA subsequently pairs to target sites generally located in the 3′-UTR of mRNAs to direct RNA silencing [16,23,40].
The interactions of miRNAs with their mRNA targets are largely based on their seed sequence, which comprises nucleotides 2–8 (from the 5′-end), and whose sequence similarities are used as a basis for grouping miRNAs into families [16,17]. In addition, other atypical sites on the miRNA, such as the 3′-supplementary site, can further supplement the seed interactions through additional base pairings [16,17,41,42,43]. Thus, the extent of the pairing between the miRNA and its mRNA target will ultimately dictate the mechanism of gene silencing, where very extensive pairings will result in cleavage/slicing of the mRNA target and less extensive pairings will induce translational repression or mRNA decay via deadenylation, decapping, and 5′-3′ exonuclease activity [17,20,44,45]. Although both the translational inhibition and mRNA decay modes of miRNA-mediated repression are thought to be interconnected [20,45], the mechanism of mRNA decay appears to be generally responsible for 66–90% of silencing in cells [17,20,46,47].
3. miRNAs, Cancer, and Therapeutic Approaches to miRNA Replacement/Inhibition
miRNAs can form complex, intertwined networks of interactions in their abilities to regulate gene expression [48], as a single miRNA can silence many different mRNAs, and one mRNA can be regulated by multiple miRNAs [20]. Moreover, individual miRNAs and miRNA clusters can regulate entire cellular pathways, with multiple miRNAs also being able to control intermeshed regulatory networks [20,48]. Hence, through this network-regulatory role of miRNAs, combined with miRNAs also having been implicated in regulating practically every cellular process and being essential for animal development, cell differentiation, and homeostasis [17,20,48,49], it is not surprising that alterations in miRNA expression have been associated with different human diseases, including cancer [48,50].
An overwhelming amount of evidence indicates that miRNAs exhibit abnormal expression levels in malignant cells and tissues and that they are deeply involved in tumor onset and progression through their tumor-promoting and/or tumor suppressive behaviors [10,50]. More specifically, the dysregulation of miRNA expression levels in cancer has been associated with a number of biological hallmarks of human cancer development, including sustained proliferative signaling, evasion of anti-growth signals and immune destruction, resistance of apoptosis, activation of invasion/metastasis, induction of angiogenesis, and promotion of genomic instability and inflammation [5,10]. The underlying mechanisms that contribute to the dysregulation of miRNA expression levels in human malignancies are often attributed to the amplification, deletion, or translocation of miRNA genes (i.e., the alteration of genomic miRNA copy numbers or gene locations) [5]. However, the perturbation of other cellular mechanisms can also contribute to their abnormal expression, which can involve changes to their transcriptional or epigenetic control, as well as defects in the miRNA biogenesis machinery [5].
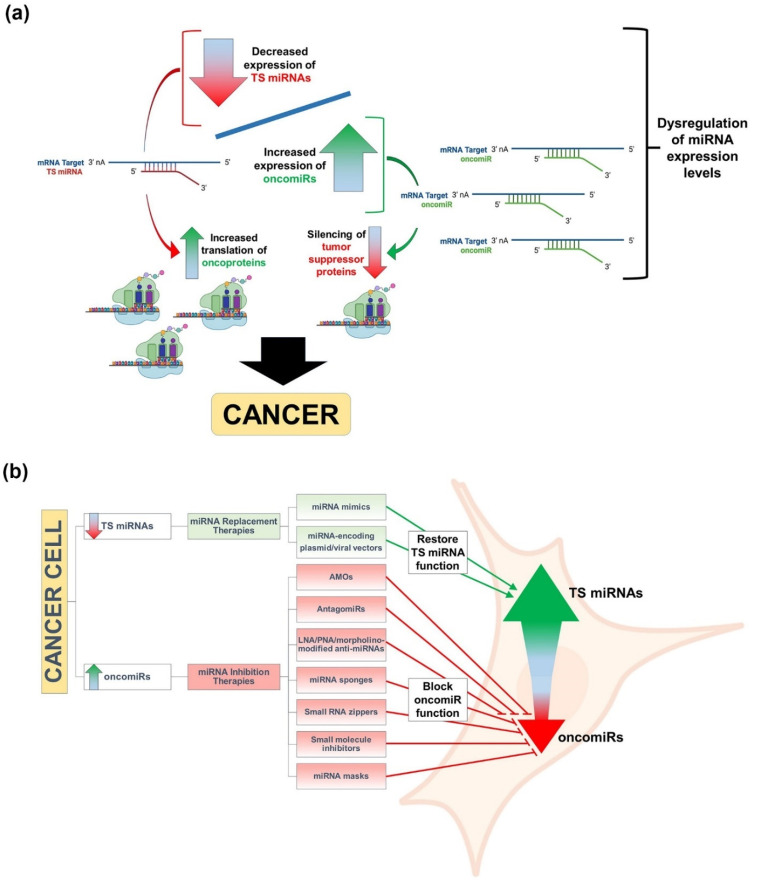
Based on their modulating effect on the expression of their target genes, tumor-associated miRNAs can be classified into two categories: (1) oncogenic miRNAs (oncomiRs) and (2) tumor suppressor miRNAs (TS miRNAs) [8,10]. In general, oncomiRs are typically found to be overexpressed in malignant cells/tissues and promote cancer by silencing mRNAs encoding tumor suppressor proteins, whereas TS miRNAs are usually downregulated and result in increases in the translation of their target oncogenic mRNAs (Figure 1a) [8,10,50]. Deep sequencing and miRNA profiling studies have provided direct evidence that miRNA expression is dysregulated in cancer and that its varied and unique signatures can be used for tumor classification, diagnosis, and prognosis [5,6,7,9]. More importantly, the dysregulation of miRNAs in cancer has also been harnessed as a potential therapeutic modality, by either miRNA replacement therapy using miRNA mimics or inhibition of miRNA function by antimiRs to restore TS miRNA or suppress oncomiR activities, respectively (Figure 1b) [10,50,51,52,53]. Approaches to enhance TS miRNA activity via miRNA replacement therapy have been achieved through the use of chemically synthesized/modified, double-stranded miRNA mimics, as well as through the use of plasmid or viral vectors engineered to encode specific TS miRNAs that can replenish the lost miRNAs within the cancer cells, thus inducing silencing of the targeted oncogenic mRNAs and impairing tumor progression [54,55,56,57,58]. Alternatively, approaches to antagonize oncomiR activity via the use of antimiRs capable of restoring tumor suppressor protein expression and halting cancer progression have been achieved through the use of antisense miRNA oligonucleotides (AMOs), antagomiRs, locked nucleic acid (LNA)/peptide nucleic acid/morpholino-modified anti-miRNAs, miRNA sponges, small RNA zippers, small-molecule inhibitors, or miRNA masks [59,60,61,62,63,64,65,66,67,68,69]. Nonetheless, although advances have been made in the abovementioned strategies in circumventing the abnormal expression of miRNAs in cancer cells, the intracellular delivery of these therapeutic miRNA mimics and antimiRs still presents a challenge to the development of effective miRNA-based cancer therapeutics, especially in vivo, that must be overcome in order to make them clinically relevant. Fortunately, there has been and continues to be much progress made in the development of highly efficacious delivery systems that are making miRNA therapeutics a clinical reality for cancer intervention.
Figure 1.
Graphical depictions highlighting the different types of cancer-associated miRNAs and the molecular therapeutic approaches to alleviating their abnormal expression in cancer. (a) Classes of tumor-associated miRNAs and their mechanisms of promoting cancer. Tumor-associated miRNAs are classified as either oncogenic miRNAs (oncomiRs) or tumor suppressor miRNAs (TS miRNAs), and the dysregulation of the expression levels of these tumor-associated miRNAs leads to cancer onset and progression. In particular, oncomiRs are typically overexpressed in malignant cells/tissues and promote cancer by silencing mRNAs encoding tumor suppressor proteins (green curved arrow), whereas TS miRNAs are downregulated and result in increases in the translation of their target oncoprotein-encoding mRNAs (red curved arrow). (b) miRNA inhibition and replacement therapies for cancer treatment. miRNA inhibition and replacement therapies are molecular interventions designed to either suppress oncomiR or restore TS miRNA functions, respectively. Approaches to enhance TS miRNA function via miRNA replacement therapy have been achieved through the use of chemically synthesized/modified, double-stranded miRNA mimics, as well as through the use of plasmid or viral vectors engineered to encode specific TS miRNAs that can replenish the lost miRNAs within cancer cells. Alternatively, approaches to antagonize oncomiR function via the use of antimiRs, resulting in the impairment of cancer progression, have been achieved through the use of antisense miRNA oligonucleotides (AMOs), antagomiRs, locked nucleic acid (LNA)/peptide nucleic acid (PNA)/morpholino-modified anti-miRNAs, miRNA sponges, small RNA zippers, small-molecule inhibitors, or miRNA masks. Select images within the figure were acquired from BioRender.com.
4. Delivery Platforms for miRNA-Based Cancer Therapeutics
Some of the most significant barriers to the widespread use of miRNA replacement/inhibition therapies for the treatment of cancer are consequences of systemic delivery and limitations in tumor-tissue specificity. Thus, because the therapeutic manipulation of miRNA expression is predominantly reliant on nucleic acid-based molecules, delivery vectors are needed to help them overcome not only the systemic barriers, such as rapid renal clearance, but also clinical barriers, such as biosafety [13]. The following section highlights the current state-of-the-art viral and nonviral delivery systems that have been demonstrated to deliver miRNA-based cancer therapies in vitro and in vivo, with a focus on nonviral nanotechnology-based miRNA delivery systems. The graphical depictions of these varied delivery systems, including their generalized modes of cell entry, as well as their strengths and weaknesses, can be found in Figure 2 and Table 2 below.
Figure 2.
Graphical depictions highlighting the varied miRNA delivery platforms developed for cancer therapeutics, as well as their mechanisms of cellular internalization. Schematic diagram illustrating the generalized modes of cell entry for both viral and nonviral miRNA delivery systems. To cross the plasma membrane of the targeted cell, many of the delivery systems utilize multiple different cellular entry routes, but in general, utilize either direct cellular entry mechanisms or endocytosis-based uptake pathways [70,71]. For example, viral vectors, such as adenovirus (AV), adeno-associated virus (AAV), and lentivirus (LV), can utilize either direct entry mechanisms or endocytosis-based uptake pathways in their delivery of miRNA mimic-encoded RNA/DNA cargo into cells. Other examples, in terms of nonviral delivery systems, can include direct cytoplasmic entry via lipid fusion of a lipid-based vector with the plasma membrane or direct cellular entry of a polymer (e.g., polyamidoamine (PAMAM))-based vector in the delivery of antagomiR cargo. Additionally depicted are examples of cellular internalizations via endocytosis of inorganic (e.g. gold (Au)), extracellular vesicle (EV), and peptide-based vectors in complex with miRNA mimic cargo. Select images within the figure were acquired from BioRender.com.
Table 2.
The highlighted advantages and disadvantages of both viral and nonviral delivery systems in their transport of miRNA-based therapeutics for the treatment of cancer.
| miRNA Delivery Platforms for Cancer Therapeutics | ||||||
|---|---|---|---|---|---|---|
| Advantages | Viral | Nonviral | ||||
| Virus | Lipid | Polymer | Inorganic | EV | Peptide | |
| • Highly efficacious gene delivery |
• Non- immunogenic • Control of size, lipid composition and functional groups, and drug loading • Co-delivery of multiple drugs |
• Non- immunogenic • Control of size, polymer composition and functional groups, and drug loading • Co-delivery of multiple drugs |
• Non- immunogenic • Ease of production • Control of size, composition and functional groups, and drug loading |
• Non- immunogenic • Control of functional groups and drug loading • Co-delivery of multiple drugs • Tissue/organ- specific delivery |
• Ease/cost of production • Control of physiochemical properties and functions • Tissue/organ- specific delivery |
|
| Disadvantages | • Immunogenic • Biosafety concerns |
• Non-specific delivery • Low in vivo efficacy • Cytotoxicity |
• Non-specific delivery • Low in vivo efficacy • Cytotoxicity |
• Low in vivo efficacy |
• Lack of experimental data/studies • Inherent diverse composition of EV cargos • Cost of production |
• Lack of experimental data/studies |
4.1. Viral Delivery
Virus-mediated delivery of miRNAs has been shown to be highly efficacious, where viral vectors can be designed to deliver miRNAs at different stages of biogenesis (i.e., pri-miRNAs and pre-miRNAs). Driven by a viral promotor, pri/pre-miRNA cloned within a plasmid can be transcribed and further processed to the mature miRNA form, enabling it to subsequently act on the target mRNA [57]. Adenoviruses (AdVs), particularly oncolytic AdVs (OAdVs), have been successful in the delivery of antimiRs and miRNA mimics. In fact, OAdV-mediated delivery of antimiRs in the form of long ncRNAs (lncRNAs), which has the therapeutic design advantage of targeting multiple copies of the same miRNA or different miRNAs within a single lncRNA molecule, has been shown to inhibit tumor growth in xenograft murine models of triple-negative breast cancer (TNBC) by simultaneously suppressing oncomiR levels of miR-9-5p, miR-10b-5p, miR-21-5p, miR-23a-3p, miR-29a-3p, miR-155-5p, miR-222-3p, miR-301a-3p, and miR-373-3p [72] and to decrease sorafenib resistance in sorafenib-resistant hepatocellular carcinoma (HCC) by concomitantly targeting miR-21, miR-153, miR-216a, miR-217, miR-494, and miR-10a-5p [73]. Virus-mediated overexpression of miRNAs can also have its advantages by suppressing particular oncogenes, as it has been shown that overexpression of miR-143 can inhibit cell growth and induce apoptosis by targeting KRAS in human colorectal cancer, in vitro [74]. Moreover, virus-mediated overexpression of miR-199 and miR-34a has been found to lead to control of tumor growth by targeting mTOR, c-MET, HIF-1, and CD44, as well as complete tumor regression by targeting Bcl-2 and SIRT1 in xenograft murine models of HCC [75,76], respectively. Similarly to AdV, adeno-associated virus (AAV)-mediated delivery systems have also had success in the treatment of HCC, as overexpression of miR-342-3p and miR-26a has each demonstrated anti-tumor effects in murine models [77,78] by targeting MCT1 and cyclins D2 and E2, respectively. Additionally, Bhere and colleagues recently showed that AAV-mediated delivery of anti-miR-21 and miR-7 resulted in decreased cell proliferation, migration, and invasion of human prostate and colon cancer cells, in vitro, and a significant reduction in tumor burden in glioblastoma murine models [79] through targeting of the PI3K/AKT and JAK/STAT3 signaling pathways (via anti-miR-21) and down-regulation of EGFR and p-AKT (via miR-7). Lentivirus-mediated delivery of miR-15a and miR-16 in murine models of chronic lymphoid leukemia [80] and a miRNA sponge targeting miR-494 in murine models of breast cancer [81] have also resulted in beneficial therapeutic effects; however, concerns of lentiviral integration into the host genome have limited their clinical application as a delivery vector.
4.2. Nonviral Delivery
Despite being highly efficacious, virus-mediated miRNA-based therapeutic delivery platforms lack clinical desirability due to a number of biosafety concerns, including viral immunogenicity. Although classically thought of as inefficient compared to viral vectors, recent advancements in nonviral delivery platforms, however, are paving the way for nucleic acid-based therapies that make their application that much more feasible in the clinic [13]. As such, the following sections highlight and discuss the pros/cons of the varied nonviral delivery technologies that have been developed for miRNA-based cancer therapeutics, including polymer nanoparticles, lipid-based nanoparticles, inorganic nanoparticles, extracellular vesicles, and an emerging technology, which is of particular relevance to our own studies—peptide carriers.
4.2.1. Polymer Nanoparticles
Polymeric delivery systems have found success as suitable vectors for delivery of nucleic acids due to their high stability and flexibility, and the facile ability to make substitutions and/or additions of functional groups [82]. In fact, one particular polymer, poly lactic-co-gycolic acid, has gained FDA approval as a delivery vector and is in phase II clinical trials for the delivery of therapeutic small interfering RNA (siRNA) molecules (NCT01676259) [83]. The ability to control the molecular weight, polymer composition, and architecture of polymers allows for the manipulation of size, morphology, charge, pKa, membrane interactions, and biodegradability [84]. Moreover, polymer-based vectors are composed of a variety of materials, including natural polymers, such as collagen, gelatin, and chitosan, synthetic polymers, and combinations of natural and synthetic polymers. The following subsections pertaining to polymer nanoparticles detail polymer-based vectors that have found success in the delivery of specific miRNA-based cancer therapies.
Polyethylenimines
Polyethylenimines (PEI) are second-generation cationic polymers that are frequently utilized for therapeutic gene delivery [84]. PEI is composed of many positively charged amino groups, allowing for complexation with anionic RNA molecules and shielding from degradation, as well as enabling the proton sponge effect, which promotes escape from endosomes after endocytosis [85]. Indeed, PEI has been shown to be an effective delivery vector of miRNA for treatment of various cancers, including miR-33a and miR-145 for the treatment of colon cancer [86] and miR-708-5p mimics for the treatment of metastatic non-small cell lung cancer [87]. Although successful, PEI alone is not as desirable as other delivery vectors, due to the excess positive charge and low degradability due to the binding of serum proteins [57]. As such, PEI in combination/conjugation with other lipids and polymers has been investigated to mitigate these undesirable effects and has found some success. For example, PEI-polyethylene glycol (PEG)-mediated delivery of miR-34a and miR-150 has been shown to be effective for treatment of HCC [88] and to effectively reduce the cell viability of chronic myeloid leukemia cells [89], respectively. Additionally, PEI-hyaluronic acid (HA)-PEG-mediated delivery of a plasmid encoding miR-125b has been demonstrated to inhibit tumor growth and induce apoptosis in a murine lung cancer model [90]. Likewise, polyurethane-PEI-mediated delivery of a plasmid encoding miR-145 has shown success for the treatment of glioblastoma [91], and PEI-antagomiR-126 complexes, loaded into liposomes, have been demonstrated to effectively target leukemic stem cells in vivo for the treatment of acute myeloid leukemia [92].
Polyamidoamine
Polyamidoamine (PAMAM) is a hyperbranched synthetic polymer that is positively charged and biodegradable. While the overall positive charge of the polymer allows for complexation with nucleic acids, it promotes hepatic accumulation and toxicity [93]. Consequently, PAMAM is frequently found in combination/conjugation with other lipids and polymers. For instance, PAMAM-PEG-mediated delivery of a miR-34a-expressing plasmid has been shown to have anti-tumor effects in non-small cell lung cancer [94], and nanographene oxide (NGO)-PEG-PAMAM-mediated delivery of anti-miR-21 has proven effective in reducing migration and invasion of non-small-cell lung cancer A549 cells, in vitro [95].
Chitosan
Chitosan is a biocompatible, natural polysaccharide. It is a deacetylated derivative of chitin, which is found in the exoskeleton of insects, crustaceans, and fungi, making it the second most abundant natural polymer [96]. Chitosan consists of repeating units of β-1,4 linked N-acetyl-D-glucosamine and D-glucosamine [97], and has been described as having a profound binding affinity for miRNAs [10]. Chitosan has been shown to be an effective delivery vector in the treatment of breast cancer, through the complexation of miR-200c [98,99], and in the treatment of prostate bone metastasis, through the complexation of miR-34a mimics [100].
Poly Lactic-Co-Gycolic Acid
As previously mentioned, poly lactic-co-gycolic acid (PLGA) is an FDA-approved polymeric delivery vector. These polymers are polyesters and are negatively charged, biodegradable, and biocompatible. PLGA is also hydrophobic, which is thought to impair its miRNA delivery efficacy [101]. As a result, PLGA in combination/conjugation with various lipids and polymers, both synthetic and natural, have been investigated, with some combinations proving effective in mediating miRNA delivery for the treatment of various cancers. In particular, PLGA-chitosan complexes containing miR-34a mimics have been shown to inhibit tumor growth of multiple myeloma xenografts and resulted in the greater survival of treated NOD-SCID tumor-bearing mice [102]. Additionally, PLGA-HA-PEI in complex with a miR-145-encoding plasmid was shown to reduce tumor growth in a murine xenograft model of colon cancer [58], and PLGA-PEG-anti-miR-21, PLGA-PEG-anti-miR-10b/21, and PLGA-PEG-miR-122 complexes were demonstrated to be effective in treating HCC [103], breast cancer [104,105], and colon cancer [106], respectively.
4.2.2. Lipid-Based Nanoparticles
The ease of use and versatility of lipid-based nanoparticles in the form of liposomes have made them the most widely used nanoparticle for the delivery of nucleic acid-based therapies, which includes miRNAs. Liposomes are spherical structures with a hydrophilic core that is separated from the external environment by a phospholipid bilayer. Liposomes can accommodate hydrophobic molecules within the bilayer, hydrophilic molecules within the liposome core, and amphiphilic molecules at the interphase between the bilayer and core [107]. Due to their phospholipid composition, liposomes can interact with cell membranes, which leads to efficient delivery of cargo.
Cationic Liposomes
The first generation of liposomes were cationic in nature, which allowed for electrostatic interactions with nucleic acid-based cargos, as well as with the negatively charged surfaces of cells [108,109]. While advantageous for drug loading and delivery, this positive-charge property, however, was found to limit the cell specificity of cationic liposomes, allowed interactions with serum proteins, and increased the susceptibility of uptake by RES [110,111]. Despite these challenges within the circulatory system, a significant tumor reduction in a xenograft tumor mouse model of colorectal cancer was still observed through cationic liposome-based nanoparticles loaded with miR-139-5p mimics, albeit with the liposomes also possessing other functionalized moieties [112]. Decreased levels of miR-143 and miR-145, which are associated with colorectal carcinoma, and the delivery of miR-143 and miR-145 mimics using cationic liposomes to restore their levels have also been shown to reduce cell proliferation in a number of colorectal cancer cell lines [113]. In addition to colorectal cancer, cationic liposome-mediated delivery of a miR-7-encoding plasmid and miR-29 mimics have been demonstrated to significantly reduce tumor sizes in xenograft tumor mouse models of lung cancer [114,115].
Neutral Liposomes
To reduce the charge-associated shortcomings of cationic liposomes, neutral liposomes were developed by the inclusion of helper lipids, such as 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE) [116], PEG [117], phosphatidylcholines (PCs) [118], and cholesterol [119]. These modifications have led to reduced RES uptake, which allows for increased half-life of the neutral liposomes within the bloodstream [110,111]. Taking advantage of the helper lipid phosphocholine, an intermediate of PC, Trang and colleagues found that neutral liposome-mediated delivery of miR-34a and let-7b mimics resulted in the significant reduction in tumor burden in a K-Ras-activated autochthonous mouse model of non-small cell lung cancer (NSCLC) [120].
Ionizable Liposomes
Further optimization of lipid-based nanoparticles has resulted in the generation of ionizable liposomes, which are cationic at low pH and neutral/anionic at neutral or higher pH levels. The ability to change charge states with respect to extracellular pH gives ionizable liposomes enhanced cell selectively characteristics that makes them more clinically translatable. In fact, an ionizable liposome-miRNA complex (comprising miR-34 mimics; MRX34) made it as far as phase I clinical trials for treatment of liver cancer and metastasis (NCT01829971; Table 1) [121,122]. However, this trial was stopped due to severe immune-related adverse events, which resulted in the death of four patients [122]. Despite this unfortunate setback, another group has shown that ionizable liposomes could still hold promise as delivery vehicles for miRNA therapeutics, as they have found, at least at the preclinical level, that ionizable liposome-mediated delivery of miR-200c mimics could result in enhanced radiosensitivity in a xenograft mouse model of lung cancer [123]. Additionally, another study found that delivery of a miR-199b-5p mimic using an ionizable cationic liposome [15] could significantly impair Hes-1 (a downstream effector of the canonical Notch and noncanonical SHH pathways) and cancer stem cell markers in a number of different tumorigenic cell lines, including colon (HT-29, CaCo-2, and SW480), breast (MDA-MB231T and MCF-7), prostate (PC-3), glioblastoma (U-87), and medulloblastoma (Daoy, ONS-76, and UW-228) cancer cell lines [124].
4.2.3. Inorganic Nanoparticles
Inorganic nanoparticles are desirable as delivery vectors because they can be designed to be biocompatible, nonimmunogenic, and nontoxic, and the size, shape, and porosity of particles can be controlled [10,125]. Nevertheless, the use of inorganic materials for delivery of miRNAs still faces challenges, such as protection from degradation in vivo, as well as endosomal escape [13]. The following section describes examples of common inorganic vectors utilized for delivery of miRNAs, however, the examples discussed herein are not a complete representation of all developed technologies.
Calcium Phosphate
Calcium phosphate (CaP) nanoparticles, composed of hydroxyapatite [Ca5(PO4)3OH], the inorganic component of bone and teeth, are described as being the most successful inorganic vectors for miRNA therapeutics [10], particularly for the treatment of colon/colorectal cancers. CaP owes this success to its unique in vivo characteristics, including its biocompatibility and biodegradability properties. Moreover, CaP’s susceptibility to acidic conditions allows for endocytic escape, where once these CaP nanoparticles are endocytosed, the acidic environment of the endosome dissolves them, resulting in subsequent increases in ionic strength that lead to osmotic swelling and the release of cargo [126]. As previously mentioned, CaP nanoparticle-mediated delivery of miRNAs has been particularly successful in the treatment of colon/colorectal cancers, as delivery of miR-4711-5p [127], miR-4689 [128], and miR-29b [129] mimics were found to effectively inhibit tumor growth in xenograft colon/colorectal cancer mouse models.
Silica
Silica-based nanoparticle technologies are desirable due to their biocompatibility, large surface area, well-defined chemical properties, and ability to control characteristics, such as pore structure [130]. One type of silica-based nanoparticle that has found success with miRNA delivery is the mesoporous silica nanoparticle (MSNP). MSNPs have a/an: (i) tunable particle size, which is important for endocytosis; (ii) stable and rigid framework, making them more resistant to heat, pH, mechanical stress, and hydrolysis-mediated degradation; (iii) uniform and tunable pore size, allowing for controlled drug loading; (iv) high surface area (>900 m2/g) and large pore volume (>0.9 cm3/g), which allows for increased drug loading; (v) interior and exterior surface, permitting selective functionalization of either surface; (vi) unique “honeycomb-like” porous structure, which aids in decreased premature drug release or leaking [131]. Taking advantage of these MSNP properties, Bertucci and colleagues successfully induced apoptosis in temozolomide (TMZ)-resistant T98G glioblastoma cells, in vitro, by loading the MSNPs with the anti-cancer drug TMZ and decorating them on the surface with a polyarginine-peptide nucleic acid (R8-PNA) antimiR conjugate designed to target miR-221, a miRNA, whose downregulation was previously reported to sensitize glioma cells to TMZ [132,133].
Gold
Gold (Au)-based nanoparticles (AuNPs) are well suited for delivery of nucleic acids, particularly after the addition of various functional groups. AuNPs have multifunctional monolayers, allowing for the addition of multiple functional moieties, which can control cytotoxicity, biodistribution, and excretion [134,135,136,137,138,139]. AuNPs can also be easily scaled with low size dispersity [140]. Due to these characteristics, AuNPs have found success in delivery of miRNAs for the treatment of various cancers. In particular, miR-375 mimic-coated AuNPs were observed to reduce tumor volume in primary and xenograft tumor mouse models of HCC [141]. Additionally, AuNPs formulated with PEG were found to mediate the highly efficient cell uptake of miRNAs and could decrease cell proliferation upon delivery of a miR-31 mimic into neuroblastoma (NGP and SH-SY5Y) and ovarian (OVCAR8 and HEYA8) cancer cell lines [142]. Moreover, Gilam and colleagues showed that in combination with the chemotherapy drug, cisplatin, AuNPs functionalized with PEG and a tumor-homing peptide, embedded within a hydrogel, could mediate the efficient local, selective, and sustained release of co-complexed miR-96 or miR-182 mimics, leading to the reduction in primary tumor size and metastasis in a breast cancer mouse model [143].
4.2.4. Extracellular Vesicles
Extracellular vesicles (EVs) are cell-derived nanovesicles that transport DNA, RNA, proteins, and lipids for cellular communication and activation of signaling pathways [144]. While EVs transport mRNA and other RNA species, such as lncRNAs and circular RNAs, miRNAs are perhaps the most abundant cargo molecule in EVs, particularly in exosomes [145]. In fact, these exosome-associated miRNAs have significant roles in the post-transcriptional regulation of gene expression and participate in the mediation of inflammatory reactions, cell migration, proliferation, apoptosis, autophagy, and epithelial-mesenchymal transition [145]. It, therefore, stands to reason that exploiting EVs for therapeutic miRNA delivery has its advantages over other delivery vectors. As a natural biomolecular carrier possessing specific ligands, EVs can be selectively delivered to cell types bearing specific surface receptors [146]. Additionally, EV ligands, such as CD47, can aid in their protection from phagocytes [147]. Moreover, lipid bilayer-encapsulated miRNA cargo is protected from RNase-mediated degradation, as well as from other circulatory system obstacles. This lipid bilayer also allows for direct fusion of the EV with the target cell membrane, with the subsequent release of cargo directly into the cytoplasm of the target cell, thus evading potential endosomal entrapment of the cargo; however, it should be noted that EVs can also undergo receptor-mediated endocytosis [148,149,150]. Though a relatively new delivery platform for therapeutic nucleic acid-based drugs, EV-mediated delivery of miRNAs has already shown promising therapeutic responses in various cancers. For example, EV-mediated delivery of a chemically modified miR-143 mimic, a plasmid expressing miR-146b, and a miR-145 mimic has each been observed to have therapeutic effects in colon cancer [151], glioma [152], and lung cancer [153], respectively. Other therapeutic uses of EVs have also been reported in the delivery of a plasmid expressing miR-122 in HCC [154] and a chemically synthesized miR-199a-3p mimic in ovarian cancer [155]. Similar to nonviral vectors discussed thus far, exosomes also have the advantage of being modified to contain different functional surface moieties. One interesting example of this type of modification was reported by Ohno and colleagues, where they engineered exosomes to express an epidermal growth factor receptor (EGFR)-specific targeting peptide, GE11, on their surfaces, which were then subsequently used to target a TS miRNA to EGFR-expressing breast cancer cells [156]. In particular, when GE11-positive exosomes containing the TS miRNA, let-7a, were administered to EGFR-expressing breast cancer xenograft tumor-bearing mice, these GE11-positive let-7a-loaded exosomes were observed to not only target the tumors, but also impair their development [156].
4.2.5. Peptides
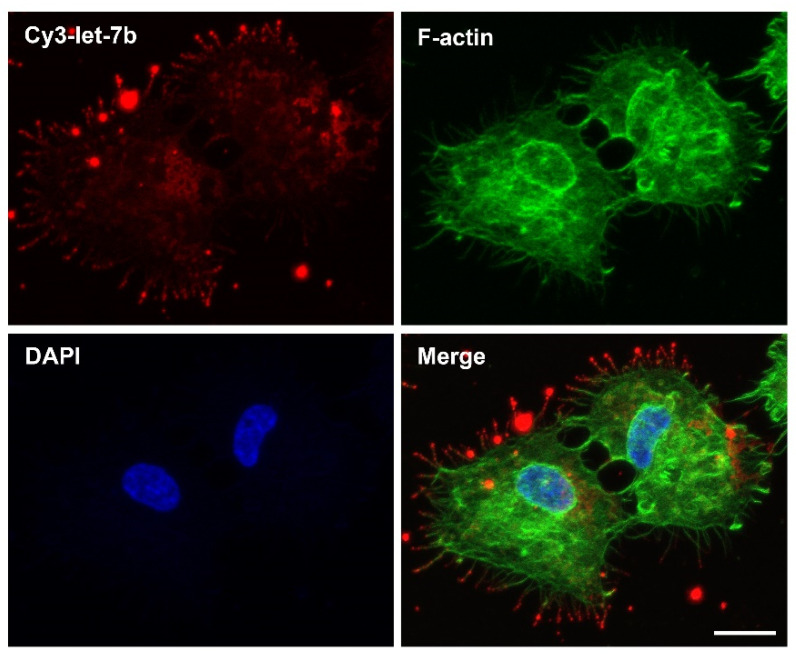
The use of peptides as a delivery vector of nucleic acid-based therapeutics was initially described over two decades ago [157,158,159], but has only recently begun to gain popularity. Peptides are favorable delivery vectors because of the diversity of their physiochemical properties and functions [160]. The controlled ability to modify their amino acid sequences and ease of synthesis allows for the production of peptides that can overcome many of the systemic circulation-associated barriers faced by nucleic acid-based therapies. As such, many different classes of peptide carriers exist, one of which, the cell-penetrating peptides (CPPs), are proving to be highly efficacious and clinically translatable for the treatment of various cancers, as suggested by their presence in phase I and II clinical trials [161,162,163,164,165], as either a therapeutic agent alone or as a delivery agent for macromolecular therapeutics [166]. CPPs are typically 4–40 amino acid residues in length [167], can penetrate the plasma membrane of a cell and facilitate the delivery of different cargos [168], and are considered by some to be the most promising nonviral delivery platform for improvement of intracellular trafficking of nucleic acid-based cargos [169], which have included DNA, RNA, siRNA [170], and more recently, even those associated with miRNA-based therapeutics. For instance, one particular peptide carrier, named FA-R9-FPcas3, comprising a folate receptor-targeting ligand, folic acid (FA), a nona-arginine CPP (R9), and a Caspase-3-sensitive imaging probe (FPcas3), was used to form a multi-functional peptide-miRNA nanocomplex consisting of the miR-34a mimic that was capable of suppressing tumor growth upon tail vein injection into living mice bearing subcutaneous HeLa tumors [171]. Moreover, because molecular imaging is such a powerful tool for visualization and quantification of pathological processes, such as cancer, Yang and colleagues recently demonstrated that a CPP, PepFect6, could also be used in complex with a radioactively-labeled AMO designed to target the oncomiR, miR-21, to successfully image miR-21 expression in A549 lung adenocarcinoma xenografts, thus demonstrating a promising method for the noninvasive imaging of miRNA expression levels in vivo [172]. Additionally, although not examples of cancer-related applications, peptides such as LMWP and PepFect6 have also been shown to successfully deliver miRNA mimics, including miR-29b, to stem cells to promote osteoblastic differentiation [173] and miR-146a (a known anti-inflammatory miRNA) to inhibit inflammatory responses in a murine model of irritant contact dermatitis [174], respectively. Lastly, regarding our own studies, we have also reported that CPPs can be effective carriers of therapeutic small ncRNAs, particularly siRNAs, which are similar to miRNAs in function in that they both can induce post-transcriptional gene silencing, but differ in that siRNAs typically inhibit the expression of a single mRNA target, whereas miRNAs normally regulate the expression of multiple mRNA targets [43]. More specifically, we have demonstrated that a CPP, named 599, could enhance the intracellular delivery and bioavailability of siRNAs in oral cancer cells in vitro, as well as induce oncogene silencing upon intratumoral administration, resulting in significant inhibition of tumor growth in an orthotopic oral cancer mouse model [54,175]. In subsequent work, we also demonstrated that co-complexation of 599 with a cancer cell-targeting peptide could synergistically mediate the effective targeting/delivery of siRNAs to xenograft oral cancer tumors upon systemic administration and significantly enhance silencing of the targeted oncogene [176]. More recently, in an effort to improve upon the efficacy of siRNA uptake and gene silencing mediated by 599, we found that a 599 peptide variant, RD3AD, which exhibited enhanced siRNA uptake and gene silencing in comparison to 599, also directed siRNAs to specific cell-surface protrusions, identified as filopodia [177]. Intriguingly, filopodia are highly dynamic, elongated, and thin cellular processes that have been reported to facilitate the highly efficient cell entry of viruses, bacteria, activated receptors, lipo/polyplexes, and exosomes by mediating their retrograde transport and/or “surfing” along the structures toward the cell body [178,179,180,181,182], where, at the filopodial base, endocytic hotspots potentially allow for easier cell entry [179]. Of particular relevance regarding exosomes, which are known transporters of miRNAs [183], is that they can utilize filopodia to “surf” toward endocytic hotspots at the filopodial base, internalize, and then traffic within endosomes to the ER [178], which coincidentally is the central nucleation site of siRNA and miRNA-mediated RNA silencing [184,185]. Hence, one can envision how the targeting of filopodia and the subsequent directed transport of RD3AD-siRNA/miRNA complexes from the filopodia to the ER would potentially allow for a very efficient trafficking route of exogenous siRNAs/miRNAs into the cellular RNA silencing machinery. In fact, recent preliminary data from our lab have found that complexation of RD3AD with a synthetic fluorescently-labeled let-7b miRNA duplex could similarly localize the miRNA mimic to filopodia and direct its entry into cancer cells (Figure 3). The significance is that one can, thus, potentially exploit filopodia-directed cell-entry machineries and subcellular-trafficking routes via CPPs for the development of more effective miRNA therapeutics.
Figure 3.
RD3AD peptide-mediated localization of complexed let-7b miRNAs to filopodia and delivery into cancer cells. Confocal fluorescence microscopy analysis of CAL 27 oral cancer cells 2 h post-treatment with a synthetic Cy3-labeled let-7b miRNA duplex (Cy3-let-7b; red) in complex with the RD3AD peptide. Filopodia (green) were stained with the F-actin label Alex Fluor 488 phalloidin, and nuclei (blue) were counterstained with DAPI. The merged images are also presented. Scale bar: 15 µm. For details on materials and methods used, please see Supplementary Materials.
5. Conclusions
Despite the pre-clinical promises of miRNA-based cancer therapeutics, the clinical advances of this form of therapy for human cancer intervention have been unfortunately very limited. Of the varied nonviral delivery platforms discussed earlier, only the lipid-based miRNA delivery system, MRX34, had reached Phase I clinical trials (NCT01829971; Table 1) [121,122], but had to be prematurely terminated, as a result of it inducing severe immune-related adverse events that led to a number of patient deaths [122]. Even unconventional miRNA delivery technologies, such as the bacterially-derived nanocell-delivery system (i.e., the EnGeneIC Delivery Vehicle) that was adapted to deliver miR-16 mimics for the treatment of mesothelioma and non-small cell lung cancer and had shown promise after the completion of a Phase I clinical trial (NCT02369198; Table 1) [186,187,188], has had no new updates regarding its progression to Phase II clinical trials since 2017. Nevertheless, although challenges persist in the development of effective delivery vehicles for miRNA-based cancer therapies, the hope remains that with the advances being made in delivery platforms for miRNA therapeutics in conferring stability to the miRNA-associated drug candidate, enhancing cancer cell-specific targeting, and promoting more efficient intracellular delivery of the therapeutic cargo, while limiting potential toxicities and adverse immune responses, it is feasible that this form of cancer therapy will become a clinical reality in the near future. With the advent of nonviral delivery technologies, such as peptide carriers, particularly CPPs, which can be tailored to target specific cancer cells and designed to enhance the cell entry of their associated drug cargo, for example, through the potential exploitation of filopodia-directed cell-entry machineries for improved drug efficacy, it is not unreasonable to assume that through further studies focused on the mechanisms of cell entry of the varied drug delivery systems and subcellular trafficking fates of the delivered nucleic acid-based cargos, that miRNA-based cancer therapeutics will soon have “big” clinical impacts.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cells11152332/s1, Materials and Methods.
Author Contributions
All authors reviewed the research findings/information available in the bibliography, designed the figures, and conceptualized and wrote the original draft of the manuscript. A.J. supervised the writing and organization of the manuscript, as well as reviewed and edited the final version. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest. The funders also had no role in the writing of the manuscript or in the decision to publish it.
Funding Statement
This work was supported in-part by NIH/NIDCR, grant numbers R21DE027231 (A.J.) and T32DE017551 (C.H.); NIH/NCATS, grant number UL1TR001450; Cell & Molecular Imaging Shared Resource, Hollings Cancer Center, MUSC (NIH/NCI P30CA138313); MUSC College of Dental Medicine Department of Oral Health Sciences.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Pasquinelli A.E., Reinhart B.J., Slack F., Martindale M.Q., Kuroda M.I., Maller B., Hayward D.C., Ball E.E., Degnan B., Müller P., et al. Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA. Nature. 2000;408:86–89. doi: 10.1038/35040556. [DOI] [PubMed] [Google Scholar]
- 2.Calin G.A., Dumitru C.D., Shimizu M., Bichi R., Zupo S., Noch E., Aldler H., Rattan S., Keating M., Rai K., et al. Frequent deletions and down-regulation of micro- RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA. 2002;99:15524–15529. doi: 10.1073/pnas.242606799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.World Health Organization International Agency for Research on Cancer—GLOBOCAN 2020. [(accessed on 30 March 2021)]. Available online: https://gco.iarc.fr/
- 4.Sung H., Ferlay J., Siegel R.L., Laversanne M., Soerjomataram I., Jemal A., Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 5.Peng Y., Croce C.M. The role of MicroRNAs in human cancer. Signal Transduct. Target. Ther. 2016;1:15004. doi: 10.1038/sigtrans.2015.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li M.-H., Fu S.-B., Xiao H.-S. Genome-wide analysis of microRNA and mRNA expression signatures in cancer. Acta Pharmacol. Sin. 2015;36:1200–1211. doi: 10.1038/aps.2015.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lu J., Getz G., Miska E.A., Alvarez-Saavedra E., Lamb J., Peck D., Sweet-Cordero A., Ebert B.L., Mak R.H., Ferrando A.A., et al. MicroRNA expression profiles classify human cancers. Nature. 2005;435:834–838. doi: 10.1038/nature03702. [DOI] [PubMed] [Google Scholar]
- 8.Otmani K., Lewalle P. Tumor Suppressor miRNA in Cancer Cells and the Tumor Microenvironment: Mechanism of Deregulation and Clinical Implications. Front. Oncol. 2021;11 doi: 10.3389/fonc.2021.708765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Volinia S., Calin G.A., Liu C.G., Ambs S., Cimmino A., Petrocca F., Visone R., Iorio M., Roldo C., Ferracin M., et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc. Natl. Acad. Sci. USA. 2006;103:2257–2261. doi: 10.1073/pnas.0510565103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Forterre A., Komuro H., Aminova S., Harada M. A Comprehensive Review of Cancer MicroRNA Therapeutic Delivery Strategies. Cancers. 2020;12:1852. doi: 10.3390/cancers12071852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hanna J., Hossain G.S., Kocerha J. The Potential for microRNA Therapeutics and Clinical Research. Front. Genet. 2019;10:478. doi: 10.3389/fgene.2019.00478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rupaimoole R., Slack F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017;16:203–222. doi: 10.1038/nrd.2016.246. [DOI] [PubMed] [Google Scholar]
- 13.Chen Y., Gao D.-Y., Huang L. In vivo delivery of miRNAs for cancer therapy: Challenges and strategies. Adv. Drug Deliv. Rev. 2014;81:128–141. doi: 10.1016/j.addr.2014.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang S., Cheng Z., Wang Y., Han T. The Risks of miRNA Therapeutics: In a Drug Target Perspective. Drug Des. Dev. Ther. 2021;ume 15:721–733. doi: 10.2147/DDDT.S288859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dasgupta I., Chatterjee A. Recent Advances in miRNA Delivery Systems. Methods Protoc. 2021;4:10. doi: 10.3390/mps4010010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bartel D.P. MicroRNAs: Target Recognition and Regulatory Functions. Cell. 2009;136:215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bartel D.P. Metazoan MicroRNAs. Cell. 2018;173:20–51. doi: 10.1016/j.cell.2018.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ebert M.S., Sharp P.A. Roles for MicroRNAs in Conferring Robustness to Biological Processes. Cell. 2012;149:515–524. doi: 10.1016/j.cell.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Michlewski G., Cáceres J.F. Post-transcriptional control of miRNA biogenesis. RNA. 2018;25:1–16. doi: 10.1261/rna.068692.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gebert L.F.R., Macrae I.J. Regulation of microRNA function in animals. Nat. Rev. Mol. Cell Biol. 2019;20:21–37. doi: 10.1038/s41580-018-0045-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Di Leva G., Garofalo M., Croce C.M. MicroRNAs in Cancer. Annu. Rev. Pathol. Mech. Dis. 2014;9:287–314. doi: 10.1146/annurev-pathol-012513-104715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alles J., Fehlmann T., Fischer U., Backes C., Galata V., Minet M., Hart M., Abu-Halima M., Grässer F.A., Lenhof H.-P., et al. An estimate of the total number of true human miRNAs. Nucleic Acids Res. 2019;47:3353–3364. doi: 10.1093/nar/gkz097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Friedman R.C., Farh K.K.-H., Burge C.B., Bartel D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009;19:92–105. doi: 10.1101/gr.082701.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hücker S.M., Fehlmann T., Werno C., Weidele K., Lüke F., Schlenska-Lange A., Klein C.A., Keller A., Kirsch S. Single-cell microRNA sequencing method comparison and application to cell lines and circulating lung tumor cells. Nat. Commun. 2021;12:1–13. doi: 10.1038/s41467-021-24611-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Macfarlane L.-A., Murphy P.R. MicroRNA: Biogenesis, Function and Role in Cancer. Curr. Genom. 2010;11:537–561. doi: 10.2174/138920210793175895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee Y., Ahn C., Han J., Choi H., Kim J., Yim J., Lee J., Provost P., Rådmark O., Kim S., et al. The nuclear RNase III Drosha initiates microRNA processing. Nature. 2003;425:415–419. doi: 10.1038/nature01957. [DOI] [PubMed] [Google Scholar]
- 27.Yi R., Qin Y., Macara I.G., Cullen B.R. Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev. 2003;17:3011–3016. doi: 10.1101/gad.1158803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lund E., Güttinger S., Calado A., Dahlberg J.E., Kutay U. Nuclear Export of MicroRNA Precursors. Science. 2004;303:95–98. doi: 10.1126/science.1090599. [DOI] [PubMed] [Google Scholar]
- 29.Bohnsack M.T., Czaplinski K., Görlich D. Exportin 5 is a RanGTP-dependent dsRNA-binding protein that mediates nuclear export of pre-miRNAs. RNA. 2004;10:185–191. doi: 10.1261/rna.5167604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hutvágner G., Mclachlan J., Pasquinelli A.E., Bálint É., Tuschl T., Zamore P.D. A cellular function for the RNA-interference enzyme Dicer in the maturation of the let-7 small temporal RNA. Science. 2001;293:834–838. doi: 10.1126/science.1062961. [DOI] [PubMed] [Google Scholar]
- 31.Grishok A., Pasquinelli A.E., Conte D., Li N., Parrish S., Ha I., Baillie D.L., Fire A., Ruvkun G., Mello C.C. Genes and Mechanisms Related to RNA Interference Regulate Expression of the Small Temporal RNAs that Control C. elegans Developmental Timing. Cell. 2001;106:23–34. doi: 10.1016/S0092-8674(01)00431-7. [DOI] [PubMed] [Google Scholar]
- 32.Ketting R.F., Fischer S.E., Bernstein E., Sijen T., Hannon G.J., Plasterk R.H. Dicer functions in RNA interference and in synthesis of small RNA involved in developmental timing in C. elegans. Genes Dev. 2001;15:2654–2659. doi: 10.1101/gad.927801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kobayashi H., Tomari Y. RISC assembly: Coordination between small RNAs and Argonaute proteins. Biochim. Et Biophys. Acta (BBA)-Gene Regul. Mech. 2016;1859:71–81. doi: 10.1016/j.bbagrm.2015.08.007. [DOI] [PubMed] [Google Scholar]
- 34.Iwasaki S., Kobayashi M., Yoda M., Sakaguchi Y., Katsuma S., Suzuki T., Tomari Y. Hsc70/Hsp90 Chaperone Machinery Mediates ATP-Dependent RISC Loading of Small RNA Duplexes. Mol. Cell. 2010;39:292–299. doi: 10.1016/j.molcel.2010.05.015. [DOI] [PubMed] [Google Scholar]
- 35.Kawamata T., Tomari Y. Making RISC. Trends Biochem. Sci. 2010;35:368–376. doi: 10.1016/j.tibs.2010.03.009. [DOI] [PubMed] [Google Scholar]
- 36.Khvorova A., Reynolds A., Jayasena S.D. Functional siRNAs and miRNAs Exhibit Strand Bias. Cell. 2003;115:209–216. doi: 10.1016/S0092-8674(03)00801-8. [DOI] [PubMed] [Google Scholar]
- 37.Schwarz D.S., Hutvágner G., Du T., Xu Z., Aronin N., Zamore P.D. Asymmetry in the Assembly of the RNAi Enzyme Complex. Cell. 2003;115:199–208. doi: 10.1016/S0092-8674(03)00759-1. [DOI] [PubMed] [Google Scholar]
- 38.Suzuki H., Katsura A., Yasuda T., Ueno T., Mano H., Sugimoto K., Miyazono K. Small-RNA asymmetry is directly driven by mammalian Argonautes. Nat. Struct. Mol. Biol. 2015;22:512–521. doi: 10.1038/nsmb.3050. [DOI] [PubMed] [Google Scholar]
- 39.Frank F., Sonenberg N., Nagar B. Structural basis for 5′-nucleotide base-specific recognition of guide RNA by human AGO2. Nature. 2010;465:818–822. doi: 10.1038/nature09039. [DOI] [PubMed] [Google Scholar]
- 40.Lewis B.P., Burge C.B., Bartel D.P. Conserved Seed Pairing, Often Flanked by Adenosines, Indicates that Thousands of Human Genes are MicroRNA Targets. Cell. 2005;120:15–20. doi: 10.1016/j.cell.2004.12.035. [DOI] [PubMed] [Google Scholar]
- 41.Chi S.W., Hannon G.J., Darnell R.B. An alternative mode of microRNA target recognition. Nat. Struct. Mol. Biol. 2012;19:321–327. doi: 10.1038/nsmb.2230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Shin C., Nam J.-W., Farh K.K.-H., Chiang H.R., Shkumatava A., Bartel D.P. Expanding the MicroRNA Targeting Code: Functional Sites with Centered Pairing. Mol. Cell. 2010;38:789–802. doi: 10.1016/j.molcel.2010.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lam J.K.W., Chow M.Y.T., Zhang Y., Leung S.W.S. siRNA Versus miRNA as Therapeutics for Gene Silencing. Mol. Ther.-Nucleic Acids. 2015;4:e252. doi: 10.1038/mtna.2015.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Huntzinger E., Izaurralde E. Gene silencing by microRNAs: Contributions of translational repression and mRNA decay. Nat. Rev. Genet. 2011;12:99–110. doi: 10.1038/nrg2936. [DOI] [PubMed] [Google Scholar]
- 45.Jonas S., Izaurralde E. Towards a molecular understanding of microRNA-mediated gene silencing. Nat. Rev. Genet. 2015;16:421–433. doi: 10.1038/nrg3965. [DOI] [PubMed] [Google Scholar]
- 46.Eichhorn S.W., Guo H., McGeary S.E., Rodriguez-Mias R.A., Shin C., Baek D., Hsu S.-H., Ghoshal K., Villén J., Bartel D.P. mRNA Destabilization Is the Dominant Effect of Mammalian MicroRNAs by the Time Substantial Repression Ensues. Mol. Cell. 2014;56:104–115. doi: 10.1016/j.molcel.2014.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Guo H., Ingolia N.T., Weissman J.S., Bartel D.P. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature. 2010;466:835–840. doi: 10.1038/nature09267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bracken C.P., Scott H.S., Goodall G.J. A network-biology perspective of microRNA function and dysfunction in cancer. Nat. Rev. Genet. 2016;17:719–732. doi: 10.1038/nrg.2016.134. [DOI] [PubMed] [Google Scholar]
- 49.Bartel D.P. MicroRNAs: Genomics, Biogenesis, Mechanism, and Function. Cell. 2004;116:281–297. doi: 10.1016/S0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 50.Gambari R., Brognara E., Spandidos D.A., Fabbri E. Targeting oncomiRNAs and mimicking tumor suppressor miRNAs: New trends in the development of miRNA therapeutic strategies in oncology (Review) Int. J. Oncol. 2016;49:5–32. doi: 10.3892/ijo.2016.3503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kong Y.W., Ferland-McCollough D., Jackson T.J., Bushell M. microRNAs in cancer management. Lancet Oncol. 2012;13:e249–e258. doi: 10.1016/S1470-2045(12)70073-6. [DOI] [PubMed] [Google Scholar]
- 52.A Broderick J., Zamore P.D. MicroRNA therapeutics. Gene Ther. 2011;18:1104–1110. doi: 10.1038/gt.2011.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bader A.G., Brown D., Winkler M. The Promise of MicroRNA Replacement Therapy. Cancer Res. 2010;70:7027–7030. doi: 10.1158/0008-5472.CAN-10-2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Alexander-Bryant A.A., Dumitriu A., Attaway C.C., Yu H., Jakymiw A. Fusogenic-oligoarginine peptide-mediated silencing of the CIP2A oncogene suppresses oral cancer tumor growth in vivo. J. Control. Release. 2015;218:72–81. doi: 10.1016/j.jconrel.2015.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Daige C.L., Wiggins J.F., Priddy L., Nelligan-Davis T., Zhao J., Brown D. Systemic Delivery of a miR34a Mimic as a Potential Therapeutic for Liver Cancer. Mol. Cancer Ther. 2014;13:2352–2360. doi: 10.1158/1535-7163.MCT-14-0209. [DOI] [PubMed] [Google Scholar]
- 56.Fan J., Feng Y., Zhang R., Zhang W., Shu Y., Zeng Z., Huang S., Zhang L., Huang B., Wu D., et al. A simplified system for the effective expression and delivery of functional mature microRNAs in mammalian cells. Cancer Gene Ther. 2019;27:424–437. doi: 10.1038/s41417-019-0113-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hosseinahli N., Aghapour M., Duijf P.H.G., Baradaran B. Treating cancer with microRNA replacement therapy: A literature review. J. Cell. Physiol. 2018;233:5574–5588. doi: 10.1002/jcp.26514. [DOI] [PubMed] [Google Scholar]
- 58.Liang G., Zhu Y., Jing A., Wang J., Hu F., Feng W., Xiao Z., Chen B. Cationic microRNA-delivering nanocarriers for efficient treatment of colon carcinoma in xenograft model. Gene Ther. 2016;23:829–838. doi: 10.1038/gt.2016.60. [DOI] [PubMed] [Google Scholar]
- 59.Fei J., Lan F., Guo M., Li Y., Liu Y. Inhibitory effects of anti-miRNA oligonucleotides (AMOs) on A549 cell growth. J. Drug Target. 2008;16:688–693. doi: 10.1080/10611860802295946. [DOI] [PubMed] [Google Scholar]
- 60.Ma L., Reinhardt F., Pan E., Soutschek J., Bhat B., Marcusson E.G., Teruya-Feldstein J., Bell G.W., A Weinberg R. Therapeutic silencing of miR-10b inhibits metastasis in a mouse mammary tumor model. Nat. Biotechnol. 2010;28:341–347. doi: 10.1038/nbt.1618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Corsten M.F., Miranda R., Kasmieh R., Krichevsky A.M., Weissleder R., Shah K. MicroRNA-21 Knockdown Disrupts Glioma Growth In vivo and Displays Synergistic Cytotoxicity with Neural Precursor Cell–Delivered S-TRAIL in Human Gliomas. Cancer Res. 2007;67:8994–9000. doi: 10.1158/0008-5472.CAN-07-1045. [DOI] [PubMed] [Google Scholar]
- 62.Cheng C.J., Bahal R., Babar I.A., Pincus Z., Barrera F.N., Liu C., Svoronos A., Braddock D.T., Glazer P., Engelman D.M., et al. MicroRNA silencing for cancer therapy targeted to the tumour microenvironment. Nature. 2014;518:107–110. doi: 10.1038/nature13905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chang R.-M., Xiao S., Lei X., Yang H., Fang F., Yang L.-Y. miRNA-487a Promotes Proliferation and Metastasis in Hepatocellular Carcinoma. Clin. Cancer Res. 2017;23:2593–2604. doi: 10.1158/1078-0432.CCR-16-0851. [DOI] [PubMed] [Google Scholar]
- 64.Chen L., Zhang K., Shi Z., Zhang A., Jia Z., Wang G., Pu P., Kang C., Han L. A lentivirus-mediated miR-23b sponge diminishes the malignant phenotype of glioma cells in vitro and in vivo. Oncol. Rep. 2014;31:1573–1580. doi: 10.3892/or.2014.3012. [DOI] [PubMed] [Google Scholar]
- 65.Monroig P.D.C., Chen L., Zhang S., Calin G.A. Small molecule compounds targeting miRNAs for cancer therapy. Adv. Drug Deliv. Rev. 2014;81:104–116. doi: 10.1016/j.addr.2014.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ors-Kumoglu G., Gulce-Iz S., Biray-Avci C. Therapeutic microRNAs in human cancer. Cytotechnology. 2019;71:411–425. doi: 10.1007/s10616-018-0291-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhang T., Hu Y., Ju J., Hou L., Li Z., Xiao D., Li Y., Yao J., Wang C., Zhang Y., et al. Downregulation of miR-522 suppresses proliferation and metastasis of non-small cell lung cancer cells by directly targeting DENN/MADD domain containing 2D. Sci. Rep. 2016;6:srep19346. doi: 10.1038/srep19346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Miroshnichenko S., Patutina O. Enhanced Inhibition of Tumorigenesis Using Combinations of miRNA-Targeted Therapeutics. Front. Pharmacol. 2019;10 doi: 10.3389/fphar.2019.00488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Meng L., Liu C., Lü J., Zhao Q., Deng S., Wang G., Qiao J., Zhang C., Zhen L., Lu Y., et al. Small RNA zippers lock miRNA molecules and block miRNA function in mammalian cells. Nat. Commun. 2017;8:13964. doi: 10.1038/ncomms13964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kootstra N.A., Verma I.M. Gene Therapy with Viral Vectors. Annu. Rev. Pharmacol. Toxicol. 2003;43:413–439. doi: 10.1146/annurev.pharmtox.43.100901.140257. [DOI] [PubMed] [Google Scholar]
- 71.Donahue N.D., Acar H., Wilhelm S. Concepts of nanoparticle cellular uptake, intracellular trafficking, and kinetics in nanomedicine. Adv. Drug Deliv. Rev. 2019;143:68–96. doi: 10.1016/j.addr.2019.04.008. [DOI] [PubMed] [Google Scholar]
- 72.Ang L., Guo L., Wang J., Huang J., Lou X., Zhao M. Oncolytic virotherapy armed with an engineered interfering lncRNA exhibits antitumor activity by blocking the epithelial mesenchymal transition in triple-negative breast cancer. Cancer Lett. 2020;479:42–53. doi: 10.1016/j.canlet.2020.03.012. [DOI] [PubMed] [Google Scholar]
- 73.Tang S., Tan G., Jiang X., Han P., Zhai B., Dong X., Qiao H., Jiang H., Sun X. An artificial lncRNA targeting multiple miRNAs overcomes sorafenib resistance in hepatocellular carcinoma cells. Oncotarget. 2016;7:73257–73269. doi: 10.18632/oncotarget.12304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Luo Q., Basnet S., Dai Z., Li S., Zhang Z., Ge H. A novel E1B55kDa-deleted oncolytic adenovirus carrying microRNA-143 exerts specific antitumor efficacy on colorectal cancer cells. Am. J. Transl. Res. 2016;8:3822–3830. [PMC free article] [PubMed] [Google Scholar]
- 75.Callegari E., Elamin B.K., D’Abundo L., Falzoni S., Donvito G., Moshiri F., Milazzo M., Altavilla G., Giacomelli L., Fornari F., et al. Anti-Tumor Activity of a miR-199-dependent Oncolytic Adenovirus. PLoS ONE. 2013;8:e73964. doi: 10.1371/journal.pone.0073964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lou W., Chen Q., Ma L., Liu J., Yang Z., Shen J., Cui Y., Bian X.-W., Qian C. Oncolytic adenovirus co-expressing miRNA-34a and IL-24 induces superior antitumor activity in experimental tumor model. Klin. Wochenschr. 2013;91:715–725. doi: 10.1007/s00109-012-0985-x. [DOI] [PubMed] [Google Scholar]
- 77.Komoll R.-M., Hu Q., Olarewaju O., von Döhlen L., Yuan Q., Xie Y., Tsay H.-C., Daon J., Qin R., Manns M.P., et al. MicroRNA-342-3p is a potent tumour suppressor in hepatocellular carcinoma. J. Hepatol. 2020;74:122–134. doi: 10.1016/j.jhep.2020.07.039. [DOI] [PubMed] [Google Scholar]
- 78.Kota J., Chivukula R.R., O’Donnell K.A., Wentzel E.A., Montgomery C.L., Hwang H.-W., Chang T.-C., Vivekanandan P., Torbenson M., Clark K.R., et al. Therapeutic microRNA Delivery Suppresses Tumorigenesis in a Murine Liver Cancer Model. Cell. 2009;137:1005–1017. doi: 10.1016/j.cell.2009.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bhere D., Arghiani N., Lechtich E.R., Yao Y., Alsaab S., Bei F., Matin M.M., Shah K. Simultaneous downregulation of miR-21 and upregulation of miR-7 has anti-tumor efficacy. Sci. Rep. 2020;10:1–10. doi: 10.1038/s41598-020-58072-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kasar S., Salerno E., Yuan Y., Underbayev C., Vollenweider D., Laurindo M.F., Fernandes H., Bonci D., Addario A., Mazzella F., et al. Systemic in vivo lentiviral delivery of miR-15a/16 reduces malignancy in the NZB de novo mouse model of chronic lymphocytic leukemia. Genes Immun. 2011;13:109–119. doi: 10.1038/gene.2011.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Liu Y., Lai L., Chen Q., Song Y., Xu S., Ma F., Wang X., Wang J., Yu H., Cao X., et al. MicroRNA-494 Is Required for the Accumulation and Functions of Tumor-Expanded Myeloid-Derived Suppressor Cells via Targeting of PTEN. J. Immunol. 2012;188:5500–5510. doi: 10.4049/jimmunol.1103505. [DOI] [PubMed] [Google Scholar]
- 82.Fernandez C.A., Rice K.G. Engineered Nanoscaled Polyplex Gene Delivery Systems. Mol. Pharm. 2009;6:1277–1289. doi: 10.1021/mp900033j. [DOI] [PubMed] [Google Scholar]
- 83.Yin H., Kanasty R.L., Eltoukhy A.A., Vegas A.J., Dorkin J.R., Anderson D.G. Non-viral vectors for gene-based therapy. Nat. Rev. Genet. 2014;15:541–555. doi: 10.1038/nrg3763. [DOI] [PubMed] [Google Scholar]
- 84.Berg A.I.v.D., Yun C.-O., Schiffelers R.M., Hennink W.E. Polymeric delivery systems for nucleic acid therapeutics: Approaching the clinic. J. Control. Release. 2021;331:121–141. doi: 10.1016/j.jconrel.2021.01.014. [DOI] [PubMed] [Google Scholar]
- 85.Höbel S., Aigner A. Polyethylenimines for siRNA and miRNA delivery in vivo. WIREs Nanomed. Nanobiotechnology. 2013;5:484–501. doi: 10.1002/wnan.1228. [DOI] [PubMed] [Google Scholar]
- 86.Ibrahim A.F., Weirauch U., Thomas M., Grünweller A., Hartmann R.K., Aigner A. MicroRNA Replacement Therapy for miR-145 and miR-33a Is Efficacious in a Model of Colon Carcinoma. Cancer Res. 2011;71:5214–5224. doi: 10.1158/0008-5472.CAN-10-4645. [DOI] [PubMed] [Google Scholar]
- 87.Wu X., Liu T., Fang O., Dong W., Zhang F., Leach L., Hu X., Luo Z. MicroRNA-708-5p acts as a therapeutic agent against metastatic lung cancer. Oncotarget. 2015;7:2417–2432. doi: 10.18632/oncotarget.6594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hu Q., Wang K., Sun X., Li Y., Fu Q., Liang T., Tang G. A redox-sensitive, oligopeptide-guided, self-assembling, and efficiency-enhanced (ROSE) system for functional delivery of microRNA therapeutics for treatment of hepatocellular carcinoma. Biomaterials. 2016;104:192–200. doi: 10.1016/j.biomaterials.2016.07.016. [DOI] [PubMed] [Google Scholar]
- 89.Biray Avcı Ç., Özcan İ., Balcı T., Özer Ö., Gündüz C. Design of polyethylene glycol-polyethylenimine nanocomplexes as non-viral carriers: Mir-150 delivery to chronic myeloid leukemia cells. Cell Biol. Int. 2013;37:1205–1214. doi: 10.1002/cbin.10157. [DOI] [PubMed] [Google Scholar]
- 90.Talekar M., Trivedi M., Shah P., Ouyang Q., Oka A., Gandham S., Amiji M.M. Combination wt-p53 and MicroRNA-125b Transfection in a Genetically Engineered Lung Cancer Model Using Dual CD44/EGFR-targeting Nanoparticles. Mol. Ther. 2016;24:759–769. doi: 10.1038/mt.2015.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Yang Y.-P., Chien Y., Chiou G.-Y., Cherng J.-Y., Wang M.-L., Lo W.-L., Chang Y.-L., Huang P.-I., Chen Y.-W., Shih Y.-H., et al. Inhibition of cancer stem cell-like properties and reduced chemoradioresistance of glioblastoma using microRNA145 with cationic polyurethane-short branch PEI. Biomaterials. 2012;33:1462–1476. doi: 10.1016/j.biomaterials.2011.10.071. [DOI] [PubMed] [Google Scholar]
- 92.Dorrance A.M., Neviani P., Ferenchak G.J., Huang X., Nicolet D., Maharry K.S., Ozer H.G., Hoellarbauer P., Khalife J., Hill E.B., et al. Targeting leukemia stem cells in vivo with antagomiR-126 nanoparticles in acute myeloid leukemia. Leukemia. 2015;29:2143–2153. doi: 10.1038/leu.2015.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Duncan R., Izzo L. Dendrimer biocompatibility and toxicity. Adv. Drug Deliv. Rev. 2005;57:2215–2237. doi: 10.1016/j.addr.2005.09.019. [DOI] [PubMed] [Google Scholar]
- 94.Wang H., Zhao X., Guo C., Ren D., Zhao Y., Xiao W., Jiao W. Aptamer-Dendrimer Bioconjugates for Targeted Delivery of miR-34a Expressing Plasmid and Antitumor Effects in Non-Small Cell Lung Cancer Cells. PLoS ONE. 2015;10:e0139136. doi: 10.1371/journal.pone.0139136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wang F., Zhang B., Zhou L., Shi Y., Li Z., Xia Y., Tian J. Imaging Dendrimer-Grafted Graphene Oxide Mediated Anti-miR-21 Delivery With an Activatable Luciferase Reporter. ACS Appl. Mater. Interfaces. 2016;8:9014–9021. doi: 10.1021/acsami.6b02662. [DOI] [PubMed] [Google Scholar]
- 96.Ravi Kumar M.N.V. A review of chitin and chitosan applications. React. Funct. Polym. 2000;46:1–27. doi: 10.1016/S1381-5148(00)00038-9. [DOI] [Google Scholar]
- 97.Martirosyan A., Olesen M.J., Howard K.A. Chitosan-Based Nanoparticles for Mucosal Delivery of RNAi Therapeutics. Adv. Genet. 2014;88:325–352. doi: 10.1016/b978-0-12-800148-6.00011-0. [DOI] [PubMed] [Google Scholar]
- 98.Kaban K., Salva E., Akbuga J. The effects of chitosan/miR-200c nanoplexes on different stages of cancers in breast cancer cell lines. Eur. J. Pharm. Sci. 2016;95:103–110. doi: 10.1016/j.ejps.2016.05.030. [DOI] [PubMed] [Google Scholar]
- 99.Kaban K., Salva E., Akbuga J. In Vitro Dose Studies on Chitosan Nanoplexes for microRNA Delivery in Breast Cancer Cells. Nucleic Acid Ther. 2017;27:45–55. doi: 10.1089/nat.2016.0633. [DOI] [PubMed] [Google Scholar]
- 100.Gaur S., Wen Y., Song J.H., Parikh N.U., Mangala L.S., Blessing A.M., Ivan C., Wu S.Y., Varkaris A., Shi Y., et al. Chitosan nanoparticle-mediated delivery of miRNA-34a decreases prostate tumor growth in the bone and its expression induces non-canonical autophagy. Oncotarget. 2015;6:29161–29177. doi: 10.18632/oncotarget.4971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Cheng C.J., Saltzman W.M. Polymer Nanoparticle-Mediated Delivery of MicroRNA Inhibition and Alternative Splicing. Mol. Pharm. 2012;9:1481–1488. doi: 10.1021/mp300081s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Cosco D., Cilurzo F., Maiuolo J., Federico C., Di Martino M.T., Cristiano M.C., Tassone P., Fresta M., Paolino D. Delivery of miR-34a by chitosan/PLGA nanoplexes for the anticancer treatment of multiple myeloma. Sci. Rep. 2015;5:17579. doi: 10.1038/srep17579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Devulapally R., Foygel K., Sekar T.V., Willmann J.K., Paulmurugan R. Gemcitabine and Antisense-microRNA Co-encapsulated PLGA–PEG Polymer Nanoparticles for Hepatocellular Carcinoma Therapy. ACS Appl. Mater. Interfaces. 2016;8:33412–33422. doi: 10.1021/acsami.6b08153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Devulapally R., Sekar N.M., Sekar T.V., Foygel K., Massoud T.F., Willmann J.K., Paulmurugan R. Polymer Nanoparticles Mediated Codelivery of AntimiR-10b and AntimiR-21 for Achieving Triple Negative Breast Cancer Therapy. ACS Nano. 2015;9:2290–2302. doi: 10.1021/nn507465d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Devulapally R., Sekar T.V., Paulmurugan R. Formulation of Anti-miR-21 and 4-Hydroxytamoxifen Co-loaded Biodegradable Polymer Nanoparticles and Their Antiproliferative Effect on Breast Cancer Cells. Mol. Pharm. 2015;12:2080–2092. doi: 10.1021/mp500852s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wang T.-Y., Choe J.W., Pu K., Devulapally R., Bachawal S., Machtaler S., Chowdhury S.M., Luong R., Tian L., Khuri-Yakub B., et al. Ultrasound-guided delivery of microRNA loaded nanoparticles into cancer. J. Control. Release. 2015;203:99–108. doi: 10.1016/j.jconrel.2015.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Ewert K.K., Scodeller P., Simón-Gracia L., Steffes V.M., Wonder E.A., Teesalu T., Safinya C.R. Cationic Liposomes as Vectors for Nucleic Acid and Hydrophobic Drug Therapeutics. Pharmaceutics. 2021;13:1365. doi: 10.3390/pharmaceutics13091365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Malone R.W., Felgner P.L., Verma I.M. Cationic liposome-mediated RNA transfection. Proc. Natl. Acad. Sci. USA. 1989;86:6077–6081. doi: 10.1073/pnas.86.16.6077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.A Mislick K., Baldeschwieler J.D. Evidence for the role of proteoglycans in cation-mediated gene transfer. Proc. Natl. Acad. Sci. USA. 1996;93:12349–12354. doi: 10.1073/pnas.93.22.12349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Cheng X., Lee R.J. The role of helper lipids in lipid nanoparticles (LNPs) designed for oligonucleotide delivery. Adv. Drug Deliv. Rev. 2016;99:129–137. doi: 10.1016/j.addr.2016.01.022. [DOI] [PubMed] [Google Scholar]
- 111.Sun X., Yan X., Jacobson O., Sun W., Wang Z., Tong X., Xia Y., Ling D., Chen X. Improved Tumor Uptake by Optimizing Liposome Based RES Blockade Strategy. Theranostics. 2017;7:319–328. doi: 10.7150/thno.18078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zhao Y., Xu J., Le V.M., Gong Q., Li S., Gao F., Ni L., Liu J., Liang X. EpCAM Aptamer-Functionalized Cationic Liposome-Based Nanoparticles Loaded with miR-139-5p for Targeted Therapy in Colorectal Cancer. Mol. Pharm. 2019;16:4696–4710. doi: 10.1021/acs.molpharmaceut.9b00867. [DOI] [PubMed] [Google Scholar]
- 113.Su J., Liang H., Yao W., Wang N., Zhang S., Yan X., Feng H., Pang W., Wang Y., Wang X., et al. MiR-143 and MiR-145 Regulate IGF1R to Suppress Cell Proliferation in Colorectal Cancer. PLoS ONE. 2014;9:e114420. doi: 10.1371/journal.pone.0114420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Rai K., Takigawa N., Ito S., Kashihara H., Ichihara E., Yasuda T., Shimizu K., Tanimoto M., Kiura K. Liposomal Delivery of MicroRNA-7–Expressing Plasmid Overcomes Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor-Resistance in Lung Cancer Cells. Mol. Cancer Ther. 2011;10:1720–1727. doi: 10.1158/1535-7163.MCT-11-0220. [DOI] [PubMed] [Google Scholar]
- 115.Wu Y., Crawford M., Mao Y., Lee R.J., Davis I.C., Elton T.S., Lee L.J., Nana-Sinkam S.P. Therapeutic Delivery of MicroRNA-29b by Cationic Lipoplexes for Lung Cancer. Mol. Ther.-Nucleic Acids. 2013;2:e84. doi: 10.1038/mtna.2013.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Hattori Y., Suzuki S., Kawakami S., Yamashita F., Hashida M. The role of dioleoylphosphatidylethanolamine (DOPE) in targeted gene delivery with mannosylated cationic liposomes via intravenous route. J. Control. Release. 2005;108:484–495. doi: 10.1016/j.jconrel.2005.08.012. [DOI] [PubMed] [Google Scholar]
- 117.Jokerst J.V., Lobovkina T., Zare R.N., Gambhir S.S. Nanoparticle PEGylation for imaging and therapy. Nanomedicine. 2011;6:715–728. doi: 10.2217/nnm.11.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Thewalt J.L., Bloom M. Phosphatidylcholine: Cholesterol phase diagrams. Biophys. J. 1992;63:1176–1181. doi: 10.1016/S0006-3495(92)81681-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Semple S.C., Chonn A., Cullis P.R. Influence of Cholesterol on the Association of Plasma Proteins with Liposomes. Biochemistry. 1996;35:2521–2525. doi: 10.1021/bi950414i. [DOI] [PubMed] [Google Scholar]
- 120.Trang P., Wiggins J.F., Daige C.L., Cho C., Omotola M., Brown D., Weidhaas J.B., Bader A.G., Slack F.J. Systemic Delivery of Tumor Suppressor microRNA Mimics Using a Neutral Lipid Emulsion Inhibits Lung Tumors in Mice. Mol. Ther. 2011;19:1116–1122. doi: 10.1038/mt.2011.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Beg M.S., Brenner A.J., Sachdev J., Borad M., Kang Y.-K., Stoudemire J., Smith S., Bader A.G., Kim S., Hong D.S. Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Investig. New Drugs. 2016;35:180–188. doi: 10.1007/s10637-016-0407-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Hong D.S., Kang Y.K., Borad M., Sachdev J., Ejadi S., Lim H.Y., Brenner A.J., Park K., Lee J.L., Kim T.Y., et al. Phase 1 study of MRX34, a liposomal miR-34a mimic, in patients with advanced solid tumours. Br. J. Cancer. 2020;122:1630–1637. doi: 10.1038/s41416-020-0802-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Cortez M.A., Valdecanas D., Zhang X., Zhan Y., Bhardwaj V., Calin G.A., Komaki R., Giri D.K., Quini C.C., Wolfe T., et al. Therapeutic Delivery of miR-200c Enhances Radiosensitivity in Lung Cancer. Mol. Ther. J. Am. Soc. Gene Ther. 2014;22:1494–1503. doi: 10.1038/mt.2014.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.De Antonellis P., Liguori L., Falanga A., Carotenuto M., Ferrucci V., Andolfo I., Marinaro F., Scognamiglio I., Virgilio A., De Rosa G., et al. MicroRNA 199b-5p delivery through stable nucleic acid lipid particles (SNALPs) in tumorigenic cell lines. Naunyn-Schmiedebergs Arch. fur Exp. Pathol. und Pharmakol. 2013;386:287–302. doi: 10.1007/s00210-013-0837-4. [DOI] [PubMed] [Google Scholar]
- 125.Labatut A.E., Mattheolabakis G. Non-viral based miR delivery and recent developments. Eur. J. Pharm. Biopharm. 2018;128:82–90. doi: 10.1016/j.ejpb.2018.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Sokolova V., Epple M. Inorganic Nanoparticles as Carriers of Nucleic Acids into Cells. Angew. Chem. Int. Ed. 2008;47:1382–1395. doi: 10.1002/anie.200703039. [DOI] [PubMed] [Google Scholar]
- 127.Morimoto Y., Mizushima T., Wu X., Okuzaki D., Yokoyama Y., Inoue A., Hata T., Hirose H., Qian Y., Wang J., et al. miR-4711-5p regulates cancer stemness and cell cycle progression via KLF5, MDM2 and TFDP1 in colon cancer cells. Br. J. Cancer. 2020;122:1037–1049. doi: 10.1038/s41416-020-0758-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Hiraki M., Nishimura J., Takahashi H., Wu X., Takahashi Y., Miyo M., Nishida N., Uemura M., Hata T., Takemasa I., et al. Concurrent Targeting of KRAS and AKT by MiR-4689 Is a Novel Treatment Against Mutant KRAS Colorectal Cancer. Mol. Ther.-Nucleic Acids. 2015;4:e231. doi: 10.1038/mtna.2015.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Inoue A., Mizushima T., Wu X., Okuzaki D., Kambara N., Ishikawa S., Wang J., Qian Y., Hirose H., Yokoyama Y., et al. A miR-29b Byproduct Sequence Exhibits Potent Tumor-Suppressive Activities via Inhibition of NF-κB Signaling in KRAS-Mutant Colon Cancer Cells. Mol. Cancer Ther. 2018;17:977–987. doi: 10.1158/1535-7163.MCT-17-0850. [DOI] [PubMed] [Google Scholar]
- 130.Mamaeva V., Sahlgren C., Lindén M. Mesoporous silica nanoparticles in medicine—Recent advances. Adv. Drug Deliv. Rev. 2013;65:689–702. doi: 10.1016/j.addr.2012.07.018. [DOI] [PubMed] [Google Scholar]
- 131.Slowing I.I., Vivero-Escoto J.L., Wu C.-W., Lin V.S.-Y. Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev. 2008;60:1278–1288. doi: 10.1016/j.addr.2008.03.012. [DOI] [PubMed] [Google Scholar]
- 132.Bertucci A., Prasetyanto E.A., Septiadi D., Manicardi A., Brognara E., Gambari R., Corradini R., De Cola L. Combined Delivery of Temozolomide and Anti-miR221 PNA Using Mesoporous Silica Nanoparticles Induces Apoptosis in Resistant Glioma Cells. Small. 2015;11:5687–5695. doi: 10.1002/smll.201500540. [DOI] [PubMed] [Google Scholar]
- 133.Jiang C., Chen L., Zhang J., Han L., Zhang A., Zhang C., Zheng Y., Jiang T., Pu P., Kang C. Downregulation of miR-221/222 sensitizes glioma cells to temozolomide by regulating apoptosis independently of p53 status. Oncol. Rep. 2011;27:854–860. doi: 10.3892/or.2011.1535. [DOI] [PubMed] [Google Scholar]
- 134.Bowman M.-C., Ballard T.E., Ackerson C.J., Feldheim D.L., Margolis D.M., Melander C. Inhibition of HIV Fusion with Multivalent Gold Nanoparticles. J. Am. Chem. Soc. 2008;130:6896–6897. doi: 10.1021/ja710321g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Ryan J.A., Overton K.W., Speight M.E., Oldenburg C.N., Loo L., Robarge W., Franzen S., Feldheim D.L. Cellular Uptake of Gold Nanoparticles Passivated with BSA−SV40 Large T Antigen Conjugates. Anal. Chem. 2007;79:9150–9159. doi: 10.1021/ac0715524. [DOI] [PubMed] [Google Scholar]
- 136.Kim S.T., Saha K., Kim C., Rotello V.M. The Role of Surface Functionality in Determining Nanoparticle Cytotoxicity. Accounts Chem. Res. 2013;46:681–691. doi: 10.1021/ar3000647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Chompoosor A., Saha K., Ghosh P.S., Macarthy D.J., Miranda O.R., Zhu Z.-J., Arcaro K.F., Rotello V.M. The Role of Surface Functionality on Acute Cytotoxicity, ROS Generation and DNA Damage by Cationic Gold Nanoparticles. Small. 2010;6:2246–2249. doi: 10.1002/smll.201000463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.De Jong W.H., Hagens W.I., Krystek P., Burger M.C., Sips A.J., Geertsma R.E. Particle size-dependent organ distribution of gold nanoparticles after intravenous administration. Biomaterials. 2008;29:1912–1919. doi: 10.1016/j.biomaterials.2007.12.037. [DOI] [PubMed] [Google Scholar]
- 139.Zhu M., Perrett S., Nie G. Understanding the Particokinetics of Engineered Nanomaterials for Safe and Effective Therapeutic Applications. Small. 2012;9:1619–1634. doi: 10.1002/smll.201201630. [DOI] [PubMed] [Google Scholar]
- 140.Daniel M.-C., Astruc D. Gold Nanoparticles: Assembly, Supramolecular Chemistry, Quantum-Size-Related Properties, and Applications toward Biology, Catalysis, and Nanotechnology. Chem. Rev. 2003;104:293–346. doi: 10.1021/cr030698+. [DOI] [PubMed] [Google Scholar]
- 141.Xue H.-Y., Liu Y., Liao J.-Z., Lin J.-S., Li B., Yuan W.-G., Lee R.J., Li L., Xu C.-R., He X.-X. Gold nanoparticles delivered miR-375 for treatment of hepatocellular carcinoma. Oncotarget. 2016;7:86675–86686. doi: 10.18632/oncotarget.13431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Ghosh R., Singh L.C., Shohet J.M., Gunaratne P.H. A gold nanoparticle platform for the delivery of functional microRNAs into cancer cells. Biomaterials. 2012;34:807–816. doi: 10.1016/j.biomaterials.2012.10.023. [DOI] [PubMed] [Google Scholar]
- 143.Gilam A., Conde J., Weissglas-Volkov D., Oliva N., Friedman E., Artzi N., Shomron N. Local microRNA delivery targets Palladin and prevents metastatic breast cancer. Nat. Commun. 2016;7:12868. doi: 10.1038/ncomms12868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Tkach M., Théry C. Communication by Extracellular Vesicles: Where We Are and Where We Need to Go. Cell. 2016;164:1226–1232. doi: 10.1016/j.cell.2016.01.043. [DOI] [PubMed] [Google Scholar]
- 145.Zheng D., Huo M., Li B., Wang W., Piao H., Wang Y., Zhu Z., Li D., Wang T., Liu K. The Role of Exosomes and Exosomal MicroRNA in Cardiovascular Disease. Front. Cell Dev. Biol. 2021;8 doi: 10.3389/fcell.2020.616161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Valadi H., Ekström K., Bossios A., Sjöstrand M., Lee J.J., Lötvall J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007;9:654–659. doi: 10.1038/ncb1596. [DOI] [PubMed] [Google Scholar]
- 147.De Jong O.G., Kooijmans S.A.A., Murphy D.E., Jiang L., Evers M.J.W., Sluijter J.P.G., Vader P., Schiffelers R.M. Drug Delivery with Extracellular Vesicles: From Imagination to Innovation. Acc. Chem. Res. 2019;52:1761–1770. doi: 10.1021/acs.accounts.9b00109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Théry C., Ostrowski M., Segura E. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 2009;9:581–593. doi: 10.1038/nri2567. [DOI] [PubMed] [Google Scholar]
- 149.Cocucci E., Racchetti G., Meldolesi J. Shedding microvesicles: Artefacts no more. Trends Cell Biol. 2009;19:43–51. doi: 10.1016/j.tcb.2008.11.003. [DOI] [PubMed] [Google Scholar]
- 150.Escrevente C., Keller S., Altevogt P., Costa J. Interaction and uptake of exosomes by ovarian cancer cells. BMC Cancer. 2011;11:108. doi: 10.1186/1471-2407-11-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Akao Y., Iio A., Itoh T., Noguchi S., Itoh Y., Ohtsuki Y., Naoe T. Microvesicle-mediated RNA Molecule Delivery System Using Monocytes/Macrophages. Mol. Ther. 2011;19:395–399. doi: 10.1038/mt.2010.254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Katakowski M., Buller B., Zheng X., Lu Y., Rogers T., Osobamiro O., Shu W., Jiang F., Chopp M. Exosomes from marrow stromal cells expressing miR-146b inhibit glioma growth. Cancer Lett. 2013;335:201–204. doi: 10.1016/j.canlet.2013.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Vázquez-Ríos A.J., Molina-Crespo Á., Bouzo B.L., López-López R., Moreno-Bueno G., De La Fuente M. Exosome-mimetic nanoplatforms for targeted cancer drug delivery. J. Nanobiotechnol. 2019;17:85. doi: 10.1186/s12951-019-0517-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Lou G., Song X., Yang F., Wu S., Wang J., Chen Z., Liu Y. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J. Hematol. Oncol. 2015;8:122. doi: 10.1186/s13045-015-0220-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kobayashi M., Sawada K., Miyamoto M., Shimizu A., Yamamoto M., Kinose Y., Nakamura K., Kawano M., Kodama M., Hashimoto K., et al. Exploring the potential of engineered exosomes as delivery systems for tumor-suppressor microRNA replacement therapy in ovarian cancer. Biochem. Biophys. Res. Commun. 2020;527:153–161. doi: 10.1016/j.bbrc.2020.04.076. [DOI] [PubMed] [Google Scholar]
- 156.Ohno S.-I., Takanashi M., Sudo K., Ueda S., Ishikawa A., Matsuyama N., Fujita K., Mizutani T., Ohgi T., Ochiya T., et al. Systemically Injected Exosomes Targeted to EGFR Deliver Antitumor MicroRNA to Breast Cancer Cells. Mol. Ther. 2013;21:185–191. doi: 10.1038/mt.2012.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Langel Ü. Cell-Penetrating Peptides. Volume 1324 Springer; New York, NY, USA: 2015. [Google Scholar]
- 158.Ramsey J.D., Flynn N.H. Cell-penetrating peptides transport therapeutics into cells. Pharmacol. Ther. 2015;154:78–86. doi: 10.1016/j.pharmthera.2015.07.003. [DOI] [PubMed] [Google Scholar]
- 159.Kauffman W.B., Fuselier T., He J., Wimley W.C. Mechanism Matters: A Taxonomy of Cell Penetrating Peptides. Trends Biochem. Sci. 2015;40:749–764. doi: 10.1016/j.tibs.2015.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Cummings J.C., Zhang H., Jakymiw A. Peptide carriers to the rescue: Overcoming the barriers to siRNA delivery for cancer treatment. Transl. Res. 2019;214:92–104. doi: 10.1016/j.trsl.2019.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Meric-Bernstam F., Saleh M.N., Infante J.R., Goel S., Falchook G.S., Shapiro G., Chung K.Y., Conry R.M., Hong D.S., Wang J.S.-Z., et al. Phase I trial of a novel stapled peptide ALRN-6924 disrupting MDMX- and MDM2-mediated inhibition of WT p53 in patients with solid tumors and lymphomas. J. Clin. Oncol. 2017;35:2505. doi: 10.1200/JCO.2017.35.15_suppl.2505. [DOI] [Google Scholar]
- 162.Søgaard C.K., Blindheim A., Røst L.M., Petrović V., Nepal A., Bachke S., Liabakk N.-B., Gederaas O.A., Viset T., Arum C.-J., et al. “Two hits - one stone”; increased efficacy of cisplatin-based therapies by targeting PCNA’s role in both DNA repair and cellular signaling. Oncotarget. 2018;9:32448–32465. doi: 10.18632/oncotarget.25963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Arrouss I., Nemati F., Roncal F., Wislez M., Dorgham K., Vallerand D., Rabbe N., Karboul N., Carlotti F., Bravo J., et al. Specific Targeting of Caspase-9/PP2A Interaction as Potential New Anti-Cancer Therapy. PLoS ONE. 2013;8:e60816. doi: 10.1371/journal.pone.0060816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Lulla R.R., Goldman S., Yamada T., Beattie C.W., Bressler L., Pacini M., Pollack I.F., Fisher P.G., Packer R.J., Dunkel I.J., et al. Phase I trial of p28 (NSC745104), a non-HDM2-mediated peptide inhibitor of p53 ubiquitination in pediatric patients with recurrent or progressive central nervous system tumors: A Pediatric Brain Tumor Consortium Study. Neuro-Oncology. 2016;18:1319–1325. doi: 10.1093/neuonc/now047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Bennett G., Lutz R., Park P., Harrison H., Lee K. Abstract 1167: Development of BT1718, a novel Bicycle Drug Conjugate for the treatment of lung cancer. Cancer Res. 2017;77:1167. doi: 10.1158/1538-7445.AM2017-1167. [DOI] [Google Scholar]
- 166.Kurrikoff K., Vunk B., Langel Ü. Status update in the use of cell-penetrating peptides for the delivery of macromolecular therapeutics. Expert Opin. Biol. Ther. 2020;21:361–370. doi: 10.1080/14712598.2021.1823368. [DOI] [PubMed] [Google Scholar]
- 167.Srimanee A., Arvanitidou M., Kim K., Hällbrink M., Langel Ü. Cell-penetrating peptides for siRNA delivery to glioblastomas. Peptides. 2018;104:62–69. doi: 10.1016/j.peptides.2018.04.015. [DOI] [PubMed] [Google Scholar]
- 168.Lehto T., Ezzat K., Wood M.J.A., EL Andaloussi S. Peptides for nucleic acid delivery. Adv. Drug Deliv. Rev. 2016;106:172–182. doi: 10.1016/j.addr.2016.06.008. [DOI] [PubMed] [Google Scholar]
- 169.Roberts T.C., Langer R., Wood M.J.A. Advances in oligonucleotide drug delivery. Nat. Rev. Drug Discov. 2020;19:673–694. doi: 10.1038/s41573-020-0075-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Guidotti G., Brambilla L., Rossi D. Cell-Penetrating Peptides: From Basic Research to Clinics. Trends Pharmacol. Sci. 2017;38:406–424. doi: 10.1016/j.tips.2017.01.003. [DOI] [PubMed] [Google Scholar]
- 171.Xiao X., Wang X., Wang Y., Yu T., Huang L., Chen L., Li J., Zhang C., Zhang Y. Multi-Functional Peptide-MicroRNA Nanocomplex for Targeted MicroRNA Delivery and Function Imaging. Chem.–A Eur. J. 2018;24:2277–2285. doi: 10.1002/chem.201705695. [DOI] [PubMed] [Google Scholar]
- 172.Yang G., Zhao Y., Gong A., Miao W., Yan L., Nie P., Wang Z. Improved Cellular Delivery of Antisense Oligonucleotide for miRNA-21 Imaging In Vivo Using Cell-Penetrating Peptide-Based Nanoprobes. Mol. Pharm. 2021;18:787–795. doi: 10.1021/acs.molpharmaceut.0c00160. [DOI] [PubMed] [Google Scholar]
- 173.Suh J.S., Lee J.Y., Choi Y.S., Chong P.C., Park Y.J. Peptide-mediated intracellular delivery of miRNA-29b for osteogenic stem cell differentiation. Biomaterials. 2013;34:4347–4359. doi: 10.1016/j.biomaterials.2013.02.039. [DOI] [PubMed] [Google Scholar]
- 174.Urgard E., Lorents A., Klaas M., Padari K., Viil J., Runnel T., Langel K., Kingo K., Tkaczyk E., Langel Ü., et al. Pre-administration of PepFect6-microRNA-146a nanocomplexes inhibits inflammatory responses in keratinocytes and in a mouse model of irritant contact dermatitis. J. Control. Release. 2016;235:195–204. doi: 10.1016/j.jconrel.2016.06.006. [DOI] [PubMed] [Google Scholar]
- 175.Cantini L., Attaway C.C., Butler B., Andino L.M., Sokolosky M.L., Jakymiw A. Fusogenic-Oligoarginine Peptide-Mediated Delivery of siRNAs Targeting the CIP2A Oncogene into Oral Cancer Cells. PLoS ONE. 2013;8:e73348. doi: 10.1371/journal.pone.0073348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Alexander-Bryant A.A., Zhang H., Attaway C.C., Pugh W., Eggart L., Sansevere R.M., Andino L.M., Dinh L., Cantini L.P., Jakymiw A. Dual peptide-mediated targeted delivery of bioactive siRNAs to oral cancer cells in vivo. Oral Oncol. 2017;72:123–131. doi: 10.1016/j.oraloncology.2017.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Holjencin C.E., Feinberg C.R., Hedrick T., Halsey G., Williams R.D., Patel P.V., Biles E., Cummings J.C., Wagner C., Vyavahare N., et al. Advancing peptide siRNA-carrier designs through L/D-amino acid stereochemical modifications to enhance gene silencing. Mol. Ther.-Nucleic Acids. 2021;24:462–476. doi: 10.1016/j.omtn.2021.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Heusermann W., Hean J., Trojer D., Steib E., Von Bueren S., Graff-Meyer A., Genoud C., Martin K., Pizzato N., Voshol J., et al. Exosomes surf on filopodia to enter cells at endocytic hot spots, traffic within endosomes, and are targeted to the ER. J. Cell Biol. 2016;213:173–184. doi: 10.1083/jcb.201506084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Lehmann M.J., Sherer N.M., Marks C.B., Pypaert M., Mothes W. Actin- and myosin-driven movement of viruses along filopodia precedes their entry into cells. J. Cell Biol. 2005;170:317–325. doi: 10.1083/jcb.200503059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Lidke D.S., Lidke K.A., Rieger B., Jovin T.M., Arndt-Jovin D.J. Reaching out for signals. J. Cell Biol. 2005;170:619–626. doi: 10.1083/jcb.200503140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Rehman Z.U., Sjollema K.A., Kuipers J., Hoekstra D., Zuhorn I.S. Nonviral Gene Delivery Vectors Use Syndecan-Dependent Transport Mechanisms in Filopodia To Reach the Cell Surface. ACS Nano. 2012;6:7521–7532. doi: 10.1021/nn3028562. [DOI] [PubMed] [Google Scholar]
- 182.Romero S., Grompone G., Carayol N., Mounier J., Guadagnini S., Prevost M.-C., Sansonetti P.J., Van Nhieu G.T. ATP-Mediated Erk1/2 Activation Stimulates Bacterial Capture by Filopodia, which Precedes Shigella Invasion of Epithelial Cells. Cell Host Microbe. 2011;9:508–519. doi: 10.1016/j.chom.2011.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Rodriguez-Dorantes M., Romero-Cordoba S.L., Peralta-Zaragoza O., Salido-Guadarrama I., Hidalgo-Miranda A. MicroRNAs transported by exosomes in body fluids as mediators of intercellular communication in cancer. OncoTargets Ther. 2014;7:1327–1338. doi: 10.2147/OTT.S61562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Stalder L., Heusermann W., Sokol L., Trojer D., Wirz J., Hean J., Fritzsche A., Aeschimann F., Pfanzagl V., Basselet P., et al. The rough endoplasmatic reticulum is a central nucleation site of siRNA-mediated RNA silencing. EMBO J. 2013;32:1115–1127. doi: 10.1038/emboj.2013.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Li S., Liu L., Zhuang X., Yu Y., Liu X., Cui X., Ji L., Pan Z., Cao X., Mo B., et al. MicroRNAs Inhibit the Translation of Target mRNAs on the Endoplasmic Reticulum in Arabidopsis. Cell. 2013;153:562–574. doi: 10.1016/j.cell.2013.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Reid G., Kao S.C., Pavlakis N., Brahmbhatt H., MacDiarmid J., Clarke S., Boyer M., van Zandwijk N. Clinical development of TargomiRs, a miRNA mimic-based treatment for patients with recurrent thoracic cancer. Epigenomics. 2016;8:1079–1085. doi: 10.2217/epi-2016-0035. [DOI] [PubMed] [Google Scholar]
- 187.Kao S.C., Fulham M., Wong K., Cooper W., Brahmbhatt H., MacDiarmid J., Pattison S., Sagong J.O., Huynh Y., Leslie F., et al. A Significant Metabolic and Radiological Response after a Novel Targeted MicroRNA-based Treatment Approach in Malignant Pleural Mesothelioma. Am. J. Respir. Crit. Care Med. 2015;191:1467–1469. doi: 10.1164/rccm.201503-0461LE. [DOI] [PubMed] [Google Scholar]
- 188.van Zandwijk N., Pavlakis N., Kao S.C., Linton A., Boyer M.J., Clarke S., Huynh Y., Chrzanowska A., Fulham M.J., Bailey D.L., et al. Safety and activity of microRNA-loaded minicells in patients with recurrent malignant pleural mesothelioma: A first-in-man, phase 1, open-label, dose-escalation study. Lancet Oncol. 2017;18:1386–1396. doi: 10.1016/S1470-2045(17)30621-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.