Abstract
The B-box proteins (BBXs) are a family of zinc-finger transcription factors with one/two B-Box domain(s) and play important roles in plant growth and development as well as stress responses. Wolfberry (Lycium barbarum L.) is an important traditional medicinal and food supplement in China, and its genome has recently been released. However, comprehensive studies of BBX genes in Lycium species are lacking. In this study, 28 LbaBBX genes were identified and classified into five clades by a phylogeny analysis with BBX proteins from Arabidopsis thaliana and the LbaBBXs have similar protein motifs and gene structures. Promoter cis-regulatory element prediction revealed that LbaBBXs might be highly responsive to light, phytohormone, and stress conditions. A synteny analysis indicated that 23, 20, 8, and 5 LbaBBX genes were orthologous to Solanum lycopersicum, Solanum melongena, Capsicum annuum, and Arabidopsis thaliana, respectively. The gene pairs encoding LbaBBX proteins evolved under strong purifying selection. In addition, the carotenoid content and expression patterns of selected LbaBBX genes were analyzed. LbaBBX2 and LbaBBX4 might play key roles in the regulation of zeaxanthin and antheraxanthin biosynthesis. Overall, this study improves our understanding of LbaBBX gene family characteristics and identifies genes involved in the regulation of carotenoid biosynthesis in wolfberry.
Keywords: Lycium barbarum, LbaBBX gene family, LbaBBX gene expression, carotenoid biosynthesis, protein subcellular localization
1. Introduction
Zinc finger transcription factors (TFs) are some of the most abundant TFs in plants and play a vital regulatory role in the regulation of transcription and various biological functions [1,2]. B-Box (BBX) proteins are a class of zinc-finger TFs possessing one or two B-box domains (CX2CX8CX7CX2CX4HX8H) in the N-terminus; some have an additional CCT (CONSTANS, CO-like, and TOC1) conserved domain or VP (valine–proline) motifs in the C-terminus. The B-box domains can be classified into two types: B-box1(B1) and B-box2 (B2). Two B-box conserved domains are recognized based on their consensus sequence and the distance between the zinc-binding residues [3]. Potential segmented duplication and deletion events result in differences in the consensus sequences and space between the zinc-binding residues in the two B-box domains [3,4]. In addition, the highly conserved CCT domain is comprised of 42–43 amino acids and is important for the regulation of transcription and nuclear protein transport [5,6]. According to the existence of BBX and CCT domains, 32 BBX proteins have been identified and classified into five subgroups in Arabidopsis [7]. Therefore, members of BBX proteins are divided into five categories depending on the presence of B-Box domains along with the CCT domain and have been reported in multiple species [4].
Subsequently, many studies have shown that plant BBX proteins play important roles in diverse physiological and biochemical processes, such as flowering time regulation, photomorphogenesis, shade avoidance, secondary metabolism, and biotic and abiotic stress responses [8,9,10,11,12]. The first BBX gene (CONSTANS (CO), known as AtBBX1) was identified and characterized in Arabidopsis; it can activate FLOWERING LOCUS T (FT) by binding to its promoter under a long day length [13]. Other BBX genes were subsequently discovered and functionally characterized, including BBX4, BBX6, BBX7, and BBX32, with roles in the regulation of flowering time [14,15,16]. Recently, BBX proteins have been reported to regulate secondary metabolism in fruits, especially anthocyanin and carotenoid biosynthesis. In Arabidopsis, BBX21/22/23 are positive regulators of anthocyanin accumulation [10], while BBX24/25/31 negatively regulate anthocyanin biosynthesis in response to several environmental factors. The overexpression of VvBBX44 decreased the expression of VvHY5 and VvUFGT and reduced the anthocyanin content in grape calli [17]. In pear, PpBBX16, a homolog of AtBBX22, is a positive regulator of light-induced anthocyanin accumulation [18]. In apple, MdBBX1/20/21/22 and MdBBX33 promote anthocyanin biosynthesis by light-induced anthocyanin accumulation, whereas MdBBX37 is a negative regulator of anthocyanin accumulation via light signaling. In tomatoes, SlBBX20 regulates the synthesis of carotenoids by directly binding to the promoter of the gene encoding the carotenoid biosynthesis enzyme PHYTOENESYNTHASE 1 [11]. However, studies of BBX genes in wolfberry are rare.
Wolfberry (Lycium barbarum L.; 2n = 2x = 24), a fruit tree in the family of Solanaceae, is an important medicinal and edible plant in China. L. barbarum is a rich source of carotenoid esters, which are mainly composed of zeaxanthin dipalmitate, lutein palmitate, antheraxanthin, and β-cryptoxanthin. Therefore, carotenoids are responsible for the orange, yellow, and red colors of L. barbarum fruits [19,20]. During the past few decades, many studies have identified TFs families with important roles in the regulation of carotenoid biosyntheses, such as SlMYB72, SlWRKY35, MdAP2, and SlBBX20 [11,21,22,23]. Two R2R3-MYB family members, LbaMYB26(Lba02g01219) and LbaMYB123 (Lba11g01830), are candidate genes involved in the regulation of carotenoid biosynthesis in L. barbarum fruits [24]. The BBX gene family has been identified and evaluated in many plant species, such as Solanum lycopersicum [25], Capsicum annuum [26], Iris germanica [27], Prunus avium [28], Vitis Vinifera [29], and Arabidopsis thaliana [30], and has diverse functions. Our understanding of the functions of the BBX gene family, such as roles in responses to biotic and abiotic stresses and secondary metabolite biosynthesis, has advanced. However, the mechanism by which BBX genes contribute to the regulation of carotenoid biosynthesis is still unclear.
Comprehensive studies of BBX genes in wolfberry have not been reported to date. The recent completion of the L. barbarum genome provides a basis for investigating the BBX gene family in the species at the genome level [31]. To further characterize the BBX gene family in Lycium, we performed systematic genome-wide identification and analyses of the BBX gene family, bridging the research gap in BBX gene family studies. Analyses of physical and chemical characteristics, collinearity analysis, phylogenetic and evolutionary relationships, conserved domains, gene structures, cis-regulatory networks, subcellular localizations, and expression patterns of LbaBBX genes were performed. This study lays a foundation for further analyses of the roles of LbaBBX genes in carotenoid biosynthesis and fruit development in wolfberry.
2. Results
2.1. Identification and Characteristics of LbaBBX Genes
To identify BBX genes in the wolfberry genome, hidden Markov model (HMM) searches with the B-box domain HMM profile (PF00643) and BLSATP using 32 BBX protein sequences from Arabidopsis thaliana as queries were performed. The candidate BBX protein sequences were used to detect the presence of B-box conserved domains by the Simple Modular Architecture Research Tool (SMART) and the National Center for Biotechnology Information (NCBI) batch CD-Search. A total of 28 putative LbaBBX genes were identified (Table 1). These BBX genes were named LbaBBX1 to LbaBBX28 according to their location on the L. barbarum chromosomes. The coding sequences (CDS) of BBX genes ranged from 330 bp to 1374 bp. They encoded proteins that were 109 to 457 amino acids (AA) in length, with predicted putative molecular weights ranging from 12.49 kDa to 51.73 KDa. The grand average of hydropathicity (GRAVY) values for all BBXs were negative, indicating that the BBX proteins were hydrophilic. The subcellular localization results showed that most of the LbaBBX proteins were found in the nucleus.
Table 1.
Information on the BBX gene family in wolfberry.
| Gene ID | Gene Name | CDS | AA | pI | MW (kDa) | GRAVY | Subcellular Localization | Domains | Structure |
|---|---|---|---|---|---|---|---|---|---|
| Lba01g02500 | LbaBBX1 | 705 | 234 | 4.91 | 25.99 | −0.393 | nucleus | 2BBX | Ⅳ |
| Lba02g02688 | LbaBBX2 | 903 | 300 | 6.20 | 33.42 | −0.522 | nucleus | 2BBX | Ⅳ |
| Lba03g02797 | LbaBBX3 | 1224 | 407 | 5.23 | 46.60 | −0.86 | nucleus | 1BBX + CCT | Ⅲ |
| Lba04g02191 | LbaBBX4 | 900 | 299 | 5.13 | 32.15 | −0.272 | nucleus | 2BBX | Ⅳ |
| Lba04g02506 | LbaBBX5 | 666 | 221 | 5.03 | 24.38 | −0.582 | nucleus | 1BBX | Ⅴ |
| Lba04g02507 | LbaBBX6 | 591 | 196 | 5.10 | 21.65 | −0.683 | chloroplast | 1BBX | Ⅴ |
| Lba04g02527 | LbaBBX7 | 369 | 122 | 7.52 | 13.83 | −0.241 | nucleus | 1BBX | Ⅴ |
| Lba04g02528 | LbaBBX8 | 1269 | 422 | 6.08 | 46.01 | −0.564 | nucleus | 2BBX + CCT | Ⅱ |
| Lba04g02630 | LbaBBX9 | 1239 | 412 | 5.13 | 45.55 | −0.656 | nucleus | 1BBX + CCT | Ⅱ |
| Lba05g00735 | LbaBBX10 | 1272 | 423 | 5.24 | 46.57 | −0.508 | nucleus | 1BBX + CCT | Ⅱ |
| Lba05g00905 | LbaBBX11 | 693 | 230 | 5.63 | 25.67 | −0.343 | nucleus | 2BBX | Ⅳ |
| Lba05g01291 | LbaBBX12 | 1227 | 408 | 5.64 | 44.95 | −0.601 | nucleus | 1BBX + CCT | Ⅱ |
| Lba05g01679 | LbaBBX13 | 1338 | 445 | 7.05 | 48.87 | −0.603 | nucleus | 2BBX + CCT | Ⅱ |
| Lba05g02193 | LbaBBX14 | 1374 | 457 | 5.24 | 51.73 | −0.741 | nucleus | 1BBX + CCT | Ⅲ |
| Lba06g03364 | LbaBBX15 | 600 | 199 | 5.88 | 21.86 | −0.518 | nucleus | 2BBX | Ⅳ |
| Lba06g03380 | LbaBBX16 | 585 | 194 | 5.88 | 21.54 | −0.573 | chloroplast | 2BBX | Ⅳ |
| Lba07g00041 | LbaBBX17 | 1140 | 379 | 6.70 | 41.94 | −0.35 | chloroplast | 2BBX + CCT | Ⅰ |
| Lba07g01710 | LbaBBX18 | 960 | 319 | 8.40 | 35.23 | −0.479 | nucleus | 2BBX | Ⅳ |
| Lba07g01848 | LbaBBX19 | 1068 | 355 | 5.96 | 39.31 | −0.561 | chloroplast | 2BBX + CCT | Ⅰ |
| Lba09g00845 | LbaBBX20 | 1140 | 379 | 5.27 | 42.66 | −0.728 | nucleus | 2BBX + CCT | Ⅱ |
| Lba09g01983 | LbaBBX21 | 966 | 321 | 5.69 | 35.91 | −0.592 | cytoplasmic | 2BBX + CCT | Ⅰ |
| Lba10g01709 | LbaBBX22 | 1263 | 420 | 5.37 | 48.09 | −0.824 | nucleus | 1BBX + CCT | Ⅲ |
| Lba10g01753 | LbaBBX23 | 1257 | 418 | 4.94 | 46.75 | −0.506 | nucleus | 2BBX | Ⅱ |
| Lba11g00500 | LbaBBX24 | 882 | 293 | 4.98 | 31.56 | −0.404 | nucleus | 2BBX | Ⅳ |
| Lba11g00948 | LbaBBX25 | 624 | 207 | 4.71 | 23.43 | −0.916 | nucleus | 1BBX | Ⅴ |
| Lba11g00982 | LbaBBX26 | 345 | 114 | 8.56 | 12.96 | −0.421 | cytoplasmic | 1BBX | Ⅴ |
| Lba11g01258 | LbaBBX27 | 981 | 326 | 4.65 | 35.69 | −0.423 | nucleus | 2BBX + CCT | Ⅱ |
| Lba12g01725 | LbaBBX28 | 330 | 109 | 9.21 | 12.49 | −0.446 | cytoplasmic | 1BBX | Ⅴ |
2.2. Protein Domains and Phylogenetic Analysis of LbaBBX Proteins
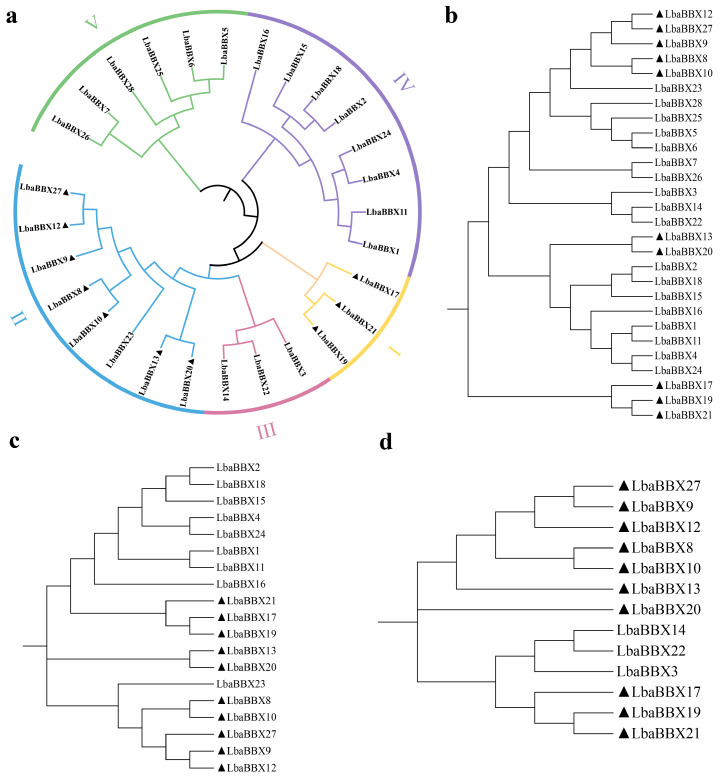
The conserved sequences of B-Box domains (B-Box1 and B-Box2), CCT domain, and VP motif in wolfberry BBX proteins were identified, and sequence logos are shown in Figure S1. Out of 28 LbaBBXs, ten contained two B-box domains and a conserved CCT domain, whereas three members had a valine–proline (VP) motif. Three and six members contained one B-box domain plus a CCT domain and only one B-Box domain, respectively, and the remaining nine members contained two B-Box domains. Conserved structures of LbaBBX members were found with B-box1 sequence (C-X2-C-X8-C-X2-D-X4-C-X2-C-D-X3-H-X8-H-X-R-X, X represents any amino acid) and B-box2 (C-X2-X8-C-X8-C-C-X3-X9-H-X-R-X4). Additionally, the CCT domain was highly conserved. Multiple sequence alignments of B-box1, B-box2, CCT domain, and VP motif for all LbaBBX proteins were also generated (Figure S2). Based on the alignments, some absolutely conserved amino acid residues were found, such as the Cysteine (C) and Histidine (H) residues in the B-box domain, Arginine (R) and Lysine (K) residues in the CCT domain, and Valine (V) and Proline (P) residues in the VP motif.
The full-length amino acid sequences were used to construct a phylogenetic tree by the maximum likelihood (ML) method using IQ-TREE. As shown in Figure 1a, the LbaBBX family was divided into five subgroups, consistent with previous studies of the gene family in tomato, pepper, and Arabidopsis [25,26,30]. We found that LbaBBX proteins assigned to the same group possess similar domain organizations. For example, in subgroup I, three LbaBBXs contained two B-Box domains, an additional CCT domain, and a VP domain (Figure 1a). In order to confirm the subfamily clustering of BBX members in wolfberry, a phylogenetic tree of LbaBBX together with SlBBX, CaBBX, StBBX, SmBBX, IcBBX, and AtBBX was also constructed by using the ML method (Figure S3 and Table S1). All BBX proteins were also divided into five subfamilies. Furthermore, the sequences of B-box 1 (Figure 1b), B-box 2 (Figure 1c), and CCT (Figure 1d) domains were also evaluated. The members of subgroups I and II contained both B-Box and CCT domains, except for LbaBBX23, which harbored only two B-Box domains. Members of subgroup III had one B-box domain and one CCT domain. Members of subgroups IV and V had no CCT domain and only two or one B-Box domain(s), respectively.
Figure 1.
Phylogenetic tree analysis of BBX proteins in wolfberry. (a) The trees shown were based on the alignments of the protein sequences of the full length, and the phylogenetic tree was constructed using maximum likelihood method with 1000 bootstrap replicates by IQ-TREE. (b–d) The tree shown were based on the alignments of the protein’s sequences of the B-box 1 domain, B-box 2 domain and CCT domain, respectively. The members marked in black triangle contain two B-Box and one CCT domains.
2.3. Gene Structure and Motif Composition of the LbaBBX Gene Family
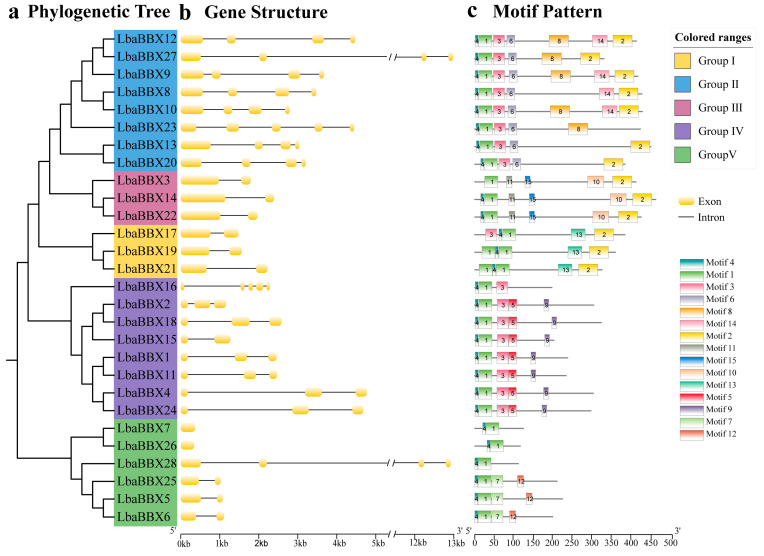
The exon–intron structures and conserved motifs were examined to gain insight into the structural diversity of LbaBBX genes. As shown in Figure 2b, the number of exons ranged from one to five, with an average of 2.9. Additionally, wolfberry BBX genes in clades I, II, III, and Ⅳ exhibited highly similar gene structures; however, LbaBBX genes in clade V showed highly variable structures. For example, most of the LbaBBX genes in clades I, II, III, and Ⅳ possessed two, four (except LbaBBX23), two, and four (except LbaBBX16) genes, respectively (Figure 2a). These results suggested that exon losses or gains occurred during the evolution of the gene family and resulted in functional divergence among closely related LbaBBXs.
Figure 2.
Phylogenetic relationships and motif composition of the LbaBBX proteins, and gene structure of the LbaBBX genes. (a) The phylogenetic tree was constructed based on the full-length sequences using IQ-TREE software by maximum likelihood (ML) method and 1000 bootstrap replicates. (b) Exon/intron structures of BBX genes from wolfberry. The exons and introns are represented by yellow boxes and black lines, respectively. The sizes of exons and introns can be estimated using the scale below. (c) The conserved motifs of wolfberry proteins were elucidated by MEME. The 15 motifs were displayed by the different colored rectangles. The sequence information for each motif is provided in Table S2. The length of protein can be estimated using the scale at the bottom.
To further examine the structural features of LbaBBX proteins, the conserved motif compositions were analyzed using MEME. Fifteen conserved motifs were predicted and named motifs 1–15 (Figure 2c). Motifs one and four were found in all LbaBBX proteins except LbaBBX3. Most of the LbaBBX genes assigned to the same group shared similar motif compositions and arrangements, which further validated the classification results. For example, motifs 6 and 13 were detected only in groups II and I, respectively. Three LbaBBX members (LbaBBX9, LbaBBX10, and LbaBBX12) from group II possessed maximum motifs, containing motifs 1, 2, 3, 4, 6, 8, and 14. Except for LbaBBX7, LbaBBX26 and LbaBBX28 harbored only two motifs (motif one and motif four) in group V. The detailed sequence information for these 15 motifs is shown in Table S2.
2.4. Chromosomal Location and Duplication of LbaBBX Genes
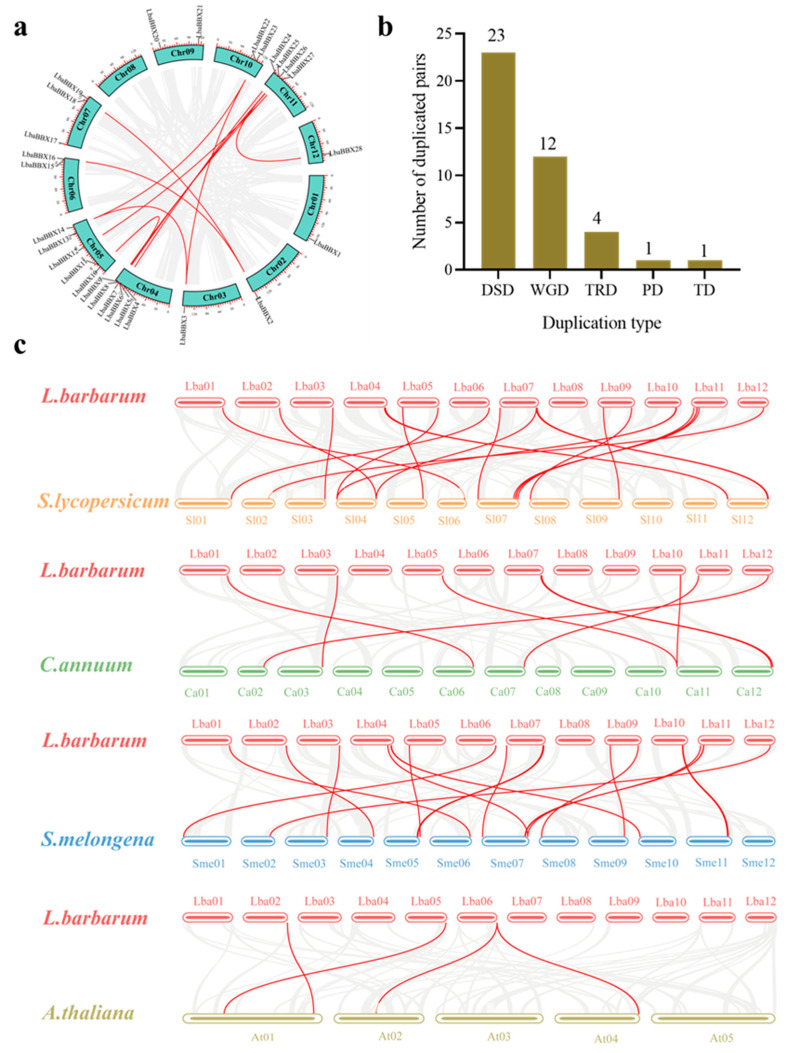
We plotted the LbaBBX genes on the chromosomes of the wolfberry genome (Figure 3a). A total of 28 LbaBBX genes were evenly distributed on 11 of 12 wolfberry chromosomes, and the number of LbaBBX genes on each chromosome was not related to the chromosome size (Figure S4). Each LbaBBX gene name corresponds to its physical position from the top to the bottom of L. barbarum chromosome 1 to chromosome 12. Chromosome 4 contained the largest number of LbaBBX genes (6 genes, ~21.4%), followed by chromosome 5 (5 genes, ~17.9%) and chromosome 11 (5 genes, ~17.9%). Only one LbaBBX gene was located on each of chromosomes 1, 2, 3, and 12, and only two were detected on chromosomes 6, 9, and 10. No LbaBBX genes were located on chromosome 8.
Figure 3.
Chromosomal location and duplicated genes among LbaBBX genes. (a) Intraspecific collinearity analysis. A total of 28 LbaBBX genes were mapped onto the chromosomes based on their physical location. The red lines indicate duplicated LbaBBX gene pairs. (b) Different models of gene duplication in LbaBBX family. The x-axis represents the duplication type. The y-axis represents the number of duplicated gene pairs. Whole genome duplication (WGD), tandem duplication (TD), proximal duplication (PD), transposed duplication (TRD), dispersed duplication (DSD). (c) Analysis of collinearity between two different species. The gray lines indicated duplicated blocks, while the red lines indicated the syntenic BBX gene pairs. Chromosome numbers are indicated above or below each chromosome.
Different patterns of gene duplication contributed to the evolution of the BBX gene family, including whole-genome duplication (WGD) as well as segmental duplication, tandem duplication (TD), proximal duplication (PD), transposed duplication (TRD), and dispersed duplication (DSD). We used DupGen_finder [32] to detect duplicated BBX family gene pairs in wolfberry. The numbers of DSD, WGD, TRD, PD, and TD duplication events in wolfberry were 23, 12, 4, 1, and 1, respectively (Figure 3b). These results indicated that DSDs and WGDs explained the majority of gene duplication events in the LbaBBX gene family.
In addition, collinearity was analyzed among homologous regions in other species, including Solanum lycopersicum, Capsicum annuum, Solanum melongena, and Arabidopsis thaliana. The interspecific collinearity results revealed 56 orthologous pairs (Figure 3c). Orthologous relationships were detected between LbaBBX genes and genes in four species belonging to Solanaceae and A. thaliana, including L. barbarum–S. lycopersicum (23 pairs), L. barbarum–S. melongena (20 pairs), L. barbarum–C. annuum (8 pairs), and L. barbarum–A. thaliana (5 pairs) (Table S3). The numbers of orthologous events of LbaBBX-SlBBX, LbaBBX-SmeBBX, and LbaBBX-CaBBX were greater than that of LbaBBX-AtBBX. These results indicated that wolfberry was closely related to the other three species in Solanaceae. The high numbers of orthologous events of LbaBBX-SlBBX identified in our study suggest that LbaBBX genes in wolfberry share a similar structure and function to those of SlBBX genes in tomato.
2.5. Nonsynonymous (Ka) and Synonymous (Ks) Substitutions per Site, and Ka/Ks Analysis of BBX Family Genes
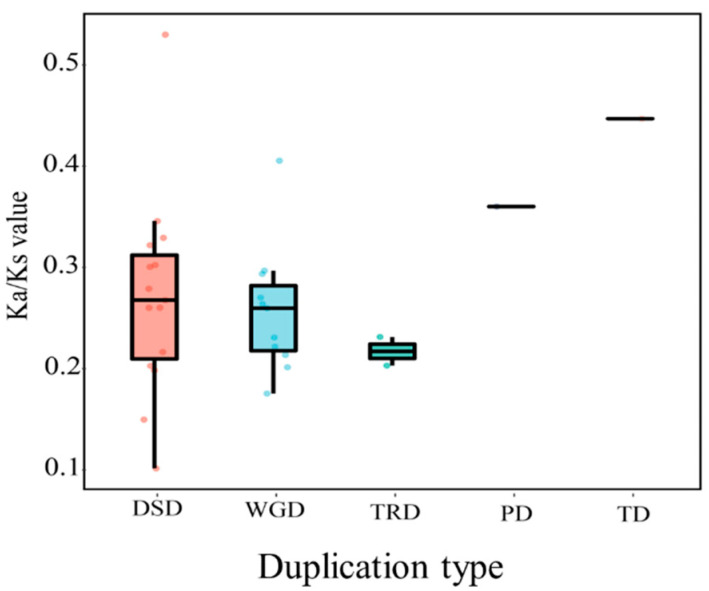
We estimated rates of synonymous (Ks) and nonsynonymous (Ka) substitutions for 56 duplicated gene pairs. As illustrated in Figure 4, the Ka/Ks values for WGD-derived gene pairs in wolfberry ranged from 0.172 to 0.403, and the Ka/Ks values for gene pairs derived from DSD, TRD, PD, and TD were 0.098–0.527, 0.199–0.227, 0.357–0.357, and 0.444–0.444, respectively (Figure 4 and Table S6). In general, Ka/Ks value greater than 1.0 provide evidence for positive selection, values less than 1.0 suggest purifying selection, and values equal to 1.0 suggest neutral evolution. In our study, all LbaBBX gene pairs had Ka/Ks values less than 1, indicating that these genes primarily underwent strong purifying selection.
Figure 4.
Different models of gene duplication in the LbaBBX family. The x-axis represents the duplication type. The colorful dots represent duplicated gene pairs. Whole genome duplication (WGD), tandem duplication (TD), proximal duplication (PD), transposed duplication (TRD), and dispersed duplication (DSD).
2.6. Cis-Regulatory Elements in the Promoters of LbaBBX Genes
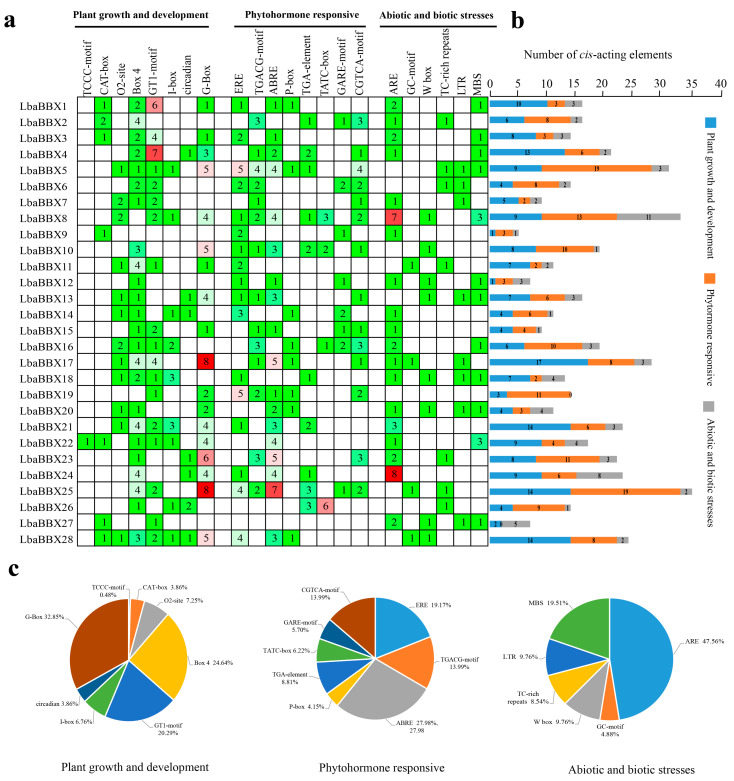
The 1500 bp upstream sequences of the 28 LbaBBX genes were extracted for analyses of the cis-regulatory elements in the promoter regions. In total, 482 cis-acting elements were identified and classified into three basic categories, including plant growth and development, phytohormone, and stress responses (abiotic/biotic) (Figure 5 and Table S4). In the first subgroup (i.e., plant growth and development), the majority (84.5%) of elements were light-responsive elements, such as GT1-motif, Box 4, G-box, and I-box, which are widespread in plants (Figure 5c). The second subgroup included elements involved in phytohormone responses; the ABREs for abscisic acid (ABA) responsiveness were the most common elements, appearing 55 times in 28 LbaBBXs, accounting for 27.9% of the hormone-responsive cis-regulatory elements (Figure 5c). The others were the CGTCA-motif and TGACG-motif for MeJA-responsiveness elements, TATC-box and P-box for gibberellin-responsive elements, and TGA-element for auxin-responsive elements, suggesting that LbaBBXs are regulated by various hormones (Figure 5c). The last subgroup included elements related to different stress responses. A cis-acting regulatory element essential for anaerobic induction (ARE) was identified in 19 LbaBBX gene promoters (Table S4), suggesting that these genes might be induced by low oxygen levels. W box (wounding and pathogen responsiveness), LTR (low-temperature responsiveness), and MBS (MYB binding site involved in drought-inducibility) were also found. Furthermore, MYC was found in 24 LbaBBX genes, suggesting that LbaBBXs contribute to the response to abiotic stress (Table S4).
Figure 5.
Identification of cis-elements in promoter regions of LbaBBXs. (a) Three categories of cis-acting elements in the LbaBBXs. Different numbers and color of the gird representing the number of different elements in these LbaBBXs. (b) Histogram of the cis-acting elements in each LbaBBX gene. The blue rectangle represents plant growth and development responsive cis-elements, the orange rectangle represents phytohormone responsive cis-elements, and the gray rectangle represents abiotic and biotic stress responsive cis-elements. (c) Pie charts of different sizes indicated the ration of each promoter element in each category, respectively.
2.7. Expression Patterns of LbaBBX Genes in Different Tissues
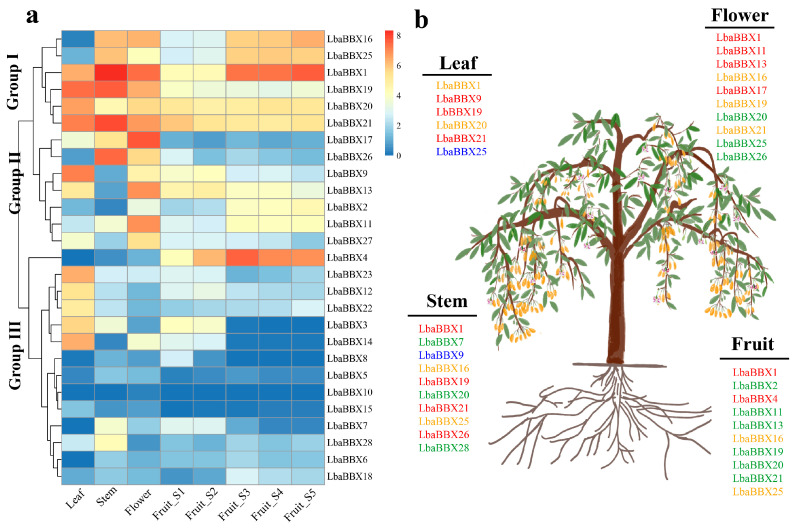
To further understand the dynamic gene expression patterns of BBX gene family members in L. barbarum, we evaluated expression profiles in four tissues (leaf, stem, flower, and fruit) with RNA-seq analysis. The LbaBBX genes exhibited tissue-specific expression and were further divided into three groups (Figure 6 and Table S5). In Group I, six genes (LbaBBX16, LbaBBX25, LbaBBX1, LbaBBX19, LbaBBX20, and LbaBBX21) presented high overall expression levels in all four organs, suggesting that these LbaBBX genes play important roles in the formation of these tissues, except two genes (LbaBBX15 and LbaBBX25) in leaves with relatively low expression. Of 28 genes, seven BBXs were assigned to Group II. Remarkably, not all homologous gene pairs exhibited similar patterns of expression; for example, LbaBBX9 had the highest transcript abundance in the leaf, and LbaBBX26 expression was highest in the stem (Figure 6b). In Group III, the remaining 14 genes shared similar low expression levels in these tissues, except LbaBBX4 (which was highly expressed in fruits). Additionally, several genes that were highly expressed in the fruit were identified, including LbaBBX1, LbaBBX4, LbaBBX16, and LbaBBX25 (Figure 6b).
Figure 6.
Expression pattern of LbaBBX genes. (a) Tissue-specific expression pattern of LbaBBX genes in four tissues: leaf, stem, flower, and fruits, including five development stages. Blue and red color indicated lower and higher transcript abundance, respectively. (b) Identification of highly expressed BBX genes in L. barbarum. Blue, green, orange, red indicated low (1–7.3 FPKM), mid-low (7.3–48 FPKM), mid-high (48–114 FPKM), and high (114–317 FPKM) expression, respectively.
2.8. Identification LbaBBX Genes Related to Carotenoid Contents
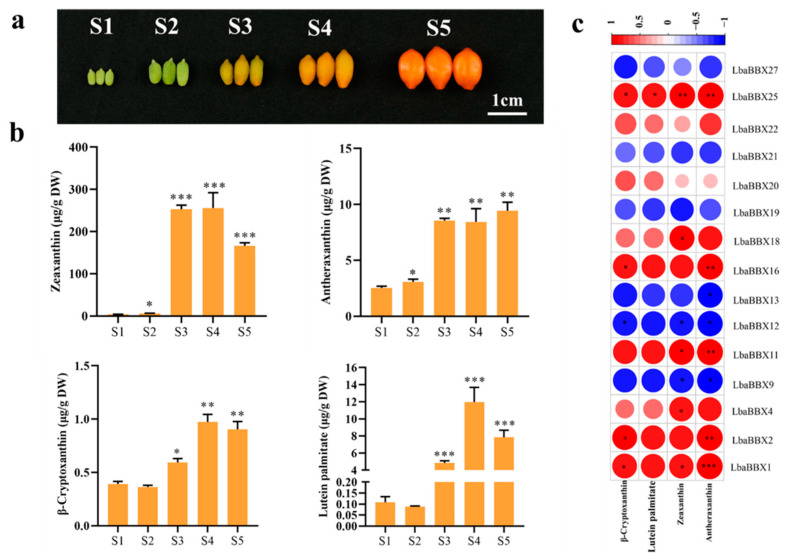
In order to ascertain how LbaBBX gene expression (FPKM values) may be predictive of carotenoid accumulation in mature wolfberry fruit, a Pearson correlation analysis was performed based on estimates at the mature (S5) stages. First, the carotenoid content was analyzed by HPLC at five developmental stages in L. barbarum var. auranticarpum (Figure 7a). During fruit development, carotenoid contents (zeaxanthin, antheraxanthin, β-cryptoxanthin, and lutein palmitate) increased sharply as maturation progressed (Figure 7b). Among the four types of carotenoid metabolites, zeaxanthin was the most abundant in all stages of fruit development. Correlation tests were performed to evaluate relationships between abundances of various carotenoids (zeaxanthin, antheraxanthin, β-cryptoxanthin, and lutein palmitate) and transcript abundances of LbaBBX genes. As shown in Figure 7c, we observed a positive correlation between LbaBBX25 with zeaxanthin (r = 0.967, p < 0.05) (Table S7). Strong positive correlations were observed between transcript levels of LbaBBX1 and LbaBBX2 with antheraxanthin contents (r = 0.993, p < 0.001; r = 0.989, p < 0.001, respectively). Significant correlations were also observed between LbaBBX11 (r = 0.985), LbaBBX16 (r = 0.981), and LbaBBX25 (r = 0.985) expression levels and antheraxanthin accumulation (all p < 0.05). Significant correlations were also observed between LbaBBX11, LbaBBX16, and LbaBBX25 expression levels and antheraxanthin accumulation (r = 0.985, p < 0.05; r = 0.981, p < 0.05; r = 0.985, p < 0.05, respectively). However, weak negative correlations of LaBBX9, LbaBBX12, and LbaBBX13 expression levels with antheraxanthin contents were observed. These correlations indicate that carotenoid accumulation in wolfberry fruits is correlated with the expression patterns of LbaBBXs.
Figure 7.
Identification LbaBBX genes related to carotenoid biosynthesis. (a) Fruits of Lycium barbarum var. auranticarpum at different stages of development. S1, S2, S3, S4, and S5 period represent 12, 19, 25, 30, and 37 days after full bloom (DAF), respectively. Scale bars represent 1 cm. (b) Trends in carotenoids (zeaxanthin, antheraxanthin, β–cryptoxanthin and lutein palmitate) at five developmental stages. The data contain the averages and standard deviations of three individual replicates. Asterisks indicate a significant difference (* p < 0.05, ** p < 0.01, *** p < 0.001) compared with S1 at the different time points during development. (c) Correlation analysis was constructed using the expression levels of LbaBBX genes and carotenoids content in five different developmental stages. The blue color means negative correlation, the red color means positive correlation. Asterisks indicate a significant difference (* p < 0.05, ** p < 0.01, *** p < 0.001).
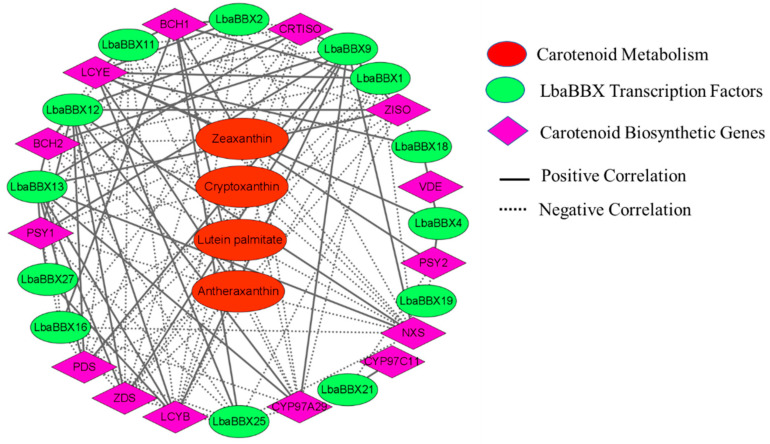
To further investigate the regulation of LbaBBX genes in wolfberry, a correlation network was constructed combining four metabolites, 14 structural genes, and 13 LbaBBX TFs related to carotenoid biosynthesis. Only the pairs with a Pearson correlation coefficient >0.8 were included in this analysis (Figure 8). The network (visualized using Cytoscape) included 31 nodes connected by 123 edges. The pairwise correlations between genes (FPKM values) and between gene and metabolite levels revealed that 74 and 49 pairs of nodes, respectively, showed positive and negative correlations. As shown in Figure 8, all nine carotenoid biosynthesis genes exhibited positive correlations with carotenoid contents, with LbaCYP97A29 showing the highest positive correlation (Table S8). For the 13 LbaBBX TFs, the transcript changes in LbaBBX1, LbaBBX2, LbaBBX4, LbaBBX11, LbaBBX16, LbaBBX18, and LbaBBX25 showed positive correlations, while LbaBBX9, LbaBBX12, and LbaBBX13 showed negative correlations (Table S7). For relationships between levels of carotenoid biosynthesis genes and BBX TFs, the highest positive correlation was observed between LbaBBX2 and LbaPDS, followed by LbaBBX1 and LbaCRTISO, while the highest negative correlation was found between LbaBBX11 and LbaLCYE (Table S9). It is worth noting that LbaBBX1, LbaBBX2, LbaBBX11, and LbaBBX16 levels showed strong positive correlations with levels of nine carotenoid biosynthesis genes each (Table S9). These results indicated that these five LbaBBXs (LbaBBX1, LbaBBX2, LbaBBX4, LbaBBX11, and LbaBBX16) might be involved in the regulation of carotenoid biosynthesis.
Figure 8.
Correlation network analysis for structural genes, LbaBBX transcription factors and carotenoid content. The red ellipse boxes indicated that carotenoid metabolism, the green ellipse boxes indicated that LbaBBX transcription factors, and the purple diamond boxes indicated carotenoid biosynthetic genes, respectively. The black solid lines indicated positive regulation, while the dot lines indicated negative regulation, respectively. The edges are drawn when the linear correlation coefficient is >0.8 with p-value < 0.05. The related correlation coefficients were shown in Table S8.
2.9. Gene Expression Analyses by qRT-PCR
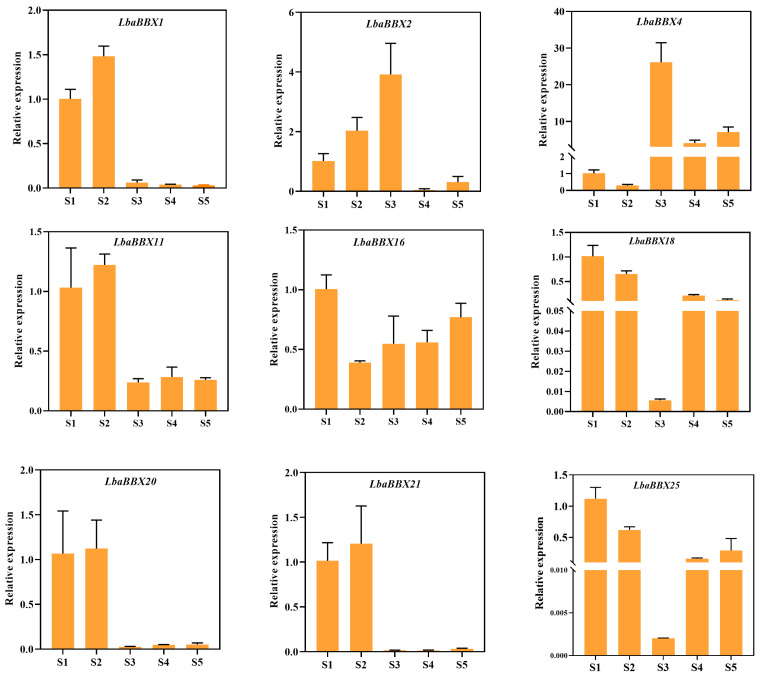
Nine potential LbaBBXs that showed strong correlations with the carotenoid content during fruit development we further evaluated by qRT-PCR. The expression patterns of several individual genes were highly correlated with the carotenoid content during wolfberry fruit development. Our results indicated that the expression levels of LbaBBX2 and LbaBBX4 increased sharply from S1 (12 DAF) to S3 (25 DAF) and reached peak values (Figure 9). The trends in the expression levels of these genes were consistent with trends in zeaxanthin content. Taken together, LbaBBX2 and LbaBBX4 were identified as important candidate genes involved in carotenoid biosynthesis and should be the focus of further functional research.
Figure 9.
The relative expression levels of nine LbaBBX genes at different fruit developmental stages. Actin gene was used as reference gene to measure expression levels in each period. The x-axis indicates the five distinct fruit developmental stages (12 DAF, 19 DAF, 25 DAF, 30 DAF and 37 DAF). The y-axis indicates the relative expression. Data represent the means ± SDs (n = 3).
2.10. Subcellular Localization of LbaBBX2 and LbaBBX4
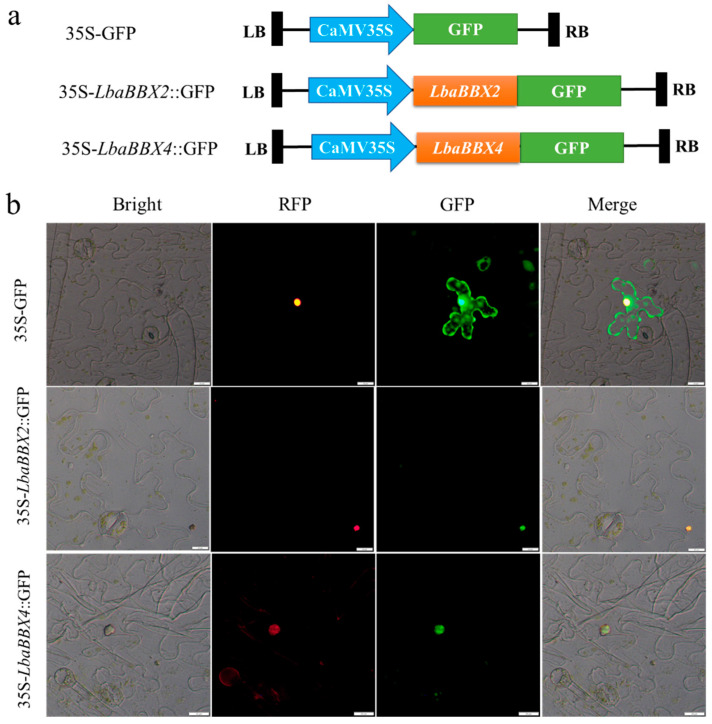
The candidate carotenoid-related genes LbaBBX2 and LbaBBX4 were selected for further analyses of subcellular localization. Their proteins were predicted to be located in the nucleus. To observe the subcellular localization of LbaBBX2 and LbaBBX4, 35S-LbaBBX2::GFP and 35S-LbaBBX4:GFP were constructed and transiently expressed in tobacco leaves, and 35S-GFP was used as a negative control. As determined by fluorescence microscopy, the 35S-LbaBBX2::GFP and 35S-LbaBBX4::GFP fusion proteins were located exclusively in the nucleus, whereas the 35S-GFP control was distributed in the tobacco leaf protoplasts (Figure 10). These results indicate that both LbaBBX2 and LbaBBX4 encode nuclear-localized proteins.
Figure 10.
Subcellular localization of LbaBBX4 protein. (a) Schematic diagram of the 35S-GFP, 35S-LbaBBX2::GFP, and 35S-LbaBBX4::GFP fusion protein constructs used for transient expression. (b) The LbaBBX2-GFP and LbaBBX4-GFP fusion proteins were transiently expressed in N. benthamiana leaves and observed by fluorescence microscopy 48 h later. The 35S-GFP was used as positive control. From left to right, bright field, red fluorescent protein (RFP) (nuclear localization signal (NLS)-RFP), green fluorescent protein (GFP), and merge image of RFP and GFP image. Scale bars =20 μm.
3. Discussion
The BBX gene family has recently been identified in many higher plants, such as Arabidopsis, tomato, and pepper [7,25,26]. The quantity of BBX genes varies among species. For example, 32 BBX genes were identified in Arabidopsis [7], 31 in tomato [25], 24 in pepper [26], and 30 in potato [33]. In this study, 28 LbaBBX genes were identified in the wolfberry genome. The number of BBX genes in the four species in the family Solanaceae (tomato, pepper, potato, and wolfberry) was relatively conserved. However, there are 64 BBX members in apple [34]. Of note, the wolfberry genome (1.67 Gb) [31] is larger than those of Arabidopsis (134 Mb) [35] and tomato (900 Mb) [36], although it was smaller than the pepper genome (3.48 Gb) [37]. These results indicated that the number of BBX gene family members might not be directly related to the plant genome size. Furthermore, the composition of the BBX genes in different subclades also differed among species (Figure S3). In wolfberry, the numbers of BBX members with two tandem B-boxes plus the CCT domain, two tandem B-boxes, box 1 plus CCT, and B-box 1 were only 7, 9, 6, and 6, respectively. The corresponding counts were 13, 8, 4, and 7 in Arabidopsis [30] and 8, 11, 5, and 7 in tomatoes [25]. These results suggested that BBX genes shared a common ancestor and underwent an independent expansion after the divergence between monocots and dicots [38].
Previous phylogenetic analyses have verified that most plant BBX genes can be divided into five subgroups (Ⅰ–IV) [4,38]. In this present study, a phylogenetic tree based on BBX protein sequences from Arabidopsis, tomato, pepper, potato, eggplant, Iochroma cyaneum, and wolfberry also supported their clustering into five subfamilies (Figure S3), consistent with previous results [38]. On the other hand, BBX proteins were grouped into five groups based on structure, depending on the presence of at least one B-box domain and a CCT domain. For example, 32 Arabidopsis BBXs were divided into five subclades according to a combination of conserved domains. The conserved domain-based classification of BBX proteins in L. barbarum was rather complex. As shown in Table 1, LbaBBX17, LbaBBX19, and LbaBBX21 were classified into group I, which had two B-boxes and a CCT plus a VP domain. Eight BBX members were classified into group II, including three LbaBBXs (LbaBBX9, LbaBBX10, and LbaBBX12) with one B-box plus a CCT domain, four LbaBBXs (LbaBBX8, LbaBBX13, LbaBBX20, and LbaBBX27) with two B-boxes and a CCT domain, and one LbaBBX (LbaBBX23) with two B-boxes. Group five contained only one B-box. A sequence alignment of LbaBBXs revealed a high degree of conservation of the B-Box1 domain among LbaBBX1 to LbaBBX28 (Figure S2). Thus, the clustering results were similar to those based on B-box 1. These results revealed that some LbaBBX proteins lost the B-box2 domain during evolution.
Gene duplication is one of the key factors responsible for the generation of novel genes, including WGD, TD, PD, TRD, and DSD, contributing to the expansion of gene family members in many species [32]. WGD, TD, and DSD are the main events in eukaryotic genome evolution and drove the development of new functions and genetic evolutionary systems [39]. Gene families, such as R2R3-MYB and BAHD acyltransferase families, expanded primarily through WGD and DSD [24,40]. WGD and TD are the main duplication events in the PMEI gene family [41]. In this study, DSD and WGD were the main factors driving BBX expansion in wolfberry, with relatively minor contributions from other replication modes.
The diversity of the biochemical functions of BBX genes has been identified in different species, including roles in plant photomorphogenesis, growth, development, metabolism, and responses to biotic or abiotic stresses [30]. For example, a number of BBXs, such as AtBBX21, AtBBX22, and AtBBX25, are involved in photomorphogenesis in Arabidopsis [42]. In apple, MdBBX37 is a negative regulator of anthocyanin accumulation via light signaling [43]. In tomatoes, SlBBX17 is a positive regulator of heat stress tolerance [12]. Despite the diverse functions of BBX gene family members, we focus on their roles in carotenoid biosynthesis. Few studies have reported that BBX genes are involved in the regulation of carotenoid metabolism. In tomatoes, SlBBX20 enhances carotenoid accumulation by activating SlPSY1 promoter activity [11]. The 28 LbaBBX genes identified in this study showed variation in expression levels across the five stages of wolfberry fruit development. Based on transcriptome expression profiles combined with a correlation network analysis, expression levels of two candidate genes (LbaBBX2 and LbaBBX4) belonging to clade Ⅳ were strongly correlated with carotenoid contents during fruit ripening. qRT-PCR analyses of LbaBBX2 and LbaBBX4 expression yielded consistent results, supporting the validity of the RNA-seq data. In addition, phylogenetic analyses indicated that LbaBBX2 and LbaBBX4 share high protein sequence homology with SlBBX20 (Figure S3). Together, we speculate that LbaBBX2 and LbaBBX4 are involved in the regulation of carotenoid biosynthesis in wolfberry. A limitation of this study is that a genetic transformation system for wolfberry plants has not been established. Therefore, the mechanism underlying LbaBBX gene expression dynamics in wolfberry plants still needs to be fully elucidated.
4. Materials and Methods
4.1. Plant Materials
Fruits of Lycium barbarum var. auranticarpum (with yellow fruit) were picked from the wolfberry germplasm of the National Wolfberry Engineering Research Center located at Yinchuan in Ningxia, China (38°38′49″ N, 106°9′10″ E). Fruit samples were harvested at five developmental stages (12, 19, 25, 30, and 37 days after full bloom, DAF). These fruits were immediately ground in liquid nitrogen and stored at −80 °C until further analysis.
4.2. Identification and Characterization of LbaBBX Genes in the L. barbarum Genome
In order to identify all possible BBX TFs in wolfberry, two strategies were used. In the first strategy, 31 BBX genes in the Arabidopsis thaliana genome were downloaded from the Arabidopsis Information Resource (TAIR, https://www.arabidopsis.org/, accessed on 4 January 2022) and were used as queries to search for potential BBXs in the L. barbarum genome database [31] by BLSATP with e value cutoff set at 1 × 10−5. In the second strategy, the HMMs of the B-box domain (PF00643) were downloaded from the Pfam database (https://pfam.xfam.org/, accessed on 8 January 2022), and HMMER 3.2 was used to identify BBX genes from the BLASTP alignments with default parameters. Subsequently, the presence of the BBX domain in each of the putative gene family members was further verified using the Pfam database (https://pfam.xfam.org/, accessed on 8 January 2022) [44], SMART database [45] (http://smart.embl-heidelberg.de/, accessed on 8 January 2022), and Conserved Domain Database (https://www.ncbi.nlm.nih.gov/cdd, accessed on 8 January 2022) [46]. Genes encoding proteins containing B-box domains were identified as BBX genes. The chemical properties, including the number of amino acids (aa), isoelectric point (pI), molecular weight (MW), and grand average of hydropathicity (GRAVY), were obtained from the ExPasy website (https://web.expasy.org/protparam/, accessed on 15 January 2022) [47]. The subcellular localizations of LbaBBX genes were predicted using WoLF PSORT (https://www.genscript.com/wolf-psort.html?src=leftbar, accessed on 15 January 2022) [48].
4.3. Multiple Sequence Alignment and Phylogenetic Analysis of LbaBBX Proteins
The BBX sequences for five species in Solanaceae, including tomato, pepper, eggplant, potato, and Iochroma cyaneum, were obtained from the Solanaceae Genomics Network (https://solgenomics.net/, accessed on 20 January 2022). First, full-length BBX protein sequences for the six species in Solanaceae and Arabidopsis were aligned by using Muscle v3.8 [49]. The deduced amino acid sequences in the B-box1, B-box2, and CCT domains were then adjusted manually using GeneDoc [50]. IQ-TREE [51] was used to construct a maximum likelihood (ML) phylogenetic tree based on all 194 full-length protein sequences. The best-fit substitution model, JTT+G, was determined using MEGA 6.06 [52]. The number of bootstrap replicates was 1000. The phylogenetic trees were visualized using iTOL v5 (https://itol.embl.de/, accessed on 20 January 2022) [53].
4.4. Gene Structure and Motif Composition of the LbaBBX Gene Family
The BBX genomic sequence and corresponding coding regions retrieved from the wolfberry genome were sent to the Gene Structure Display Server (http://gsds.gao-lab.org/, accessed on 25 January 2022) [54] to investigate exon–intron structures. MEME (https://meme-suite.org/meme/tools/meme, accessed on 25 January 2022) [55] was used to predict conserved motifs with a maximum number of motifs of 15 and optimum width of 3 to 50 bp.
4.5. Chromosomal Location and Gene Duplication Analysis of LbaBBX Genes
The chromosomal distribution of LbaBBX genes was visualized using TBtools [56]. To examine duplication events for LbaBBX genes in wolfberry and other plants, including A. thaliana, S. lycopersicum, and C. annuum, TBtools were used. The gene duplication pairs were visualized in Tbtools [56]. The whole-genome sequences of three species in Solanaceae, including L. barbarum, S. lycopersicum, C. annuum, and A. thaliana, were used to analyze collinearity. The detected syntenic blocks were visualized using Tbtools [56]. Furthermore, Ka and Ks substitution rates were calculated for each syntenic pair using KaKs_Calculator 3.0 [57].
4.6. Cis-Regulatory Elements in the Promoters of LbaBBX Genes
The 1500 bp genomic DNA sequences upstream of the start codon (ATG) of LbaBBX genes were extracted from the wolfberry genome database using Tbtools [56]. The cis-regulatory elements in these LbaBBX gene promoters were predicted by using PlantCARE (https://bioinformatics.psb.ugent.be/webtools/plantcare/html/, accessed on 3 March 2022).
4.7. RNA Isolation, cDNA Library Construction, and RNA-Seq Analysis
Total RNA was extracted independently from different wolfberry tissues using an RNA Kit (Tiangen, Beijing, China), according to the manufacturer’s instructions. RNA purity, concentration, and integrity were measured using a Nanodrop spectrophotometer (Thermo Scientific, Waltham, MA, USA) and an Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA). High-quality RNAs were used to construct a cDNA library. First- and second-strand cDNAs were synthesized using Superscript II reverse transcriptase and random hexamer primers. Double-strand cDNA was fragmented by nebulization and used to generate RNA-seq libraries, as described previously [58]. Three biological replicates of cDNA libraries were sequenced using the Illumina HiSeq 4000 platform (Illumina Inc., San Diego, CA, USA) with a paired-end read length of 150 bp.
4.8. Expression Profiles of LbaBBXs
In order to study the expression profiles of LbaBBX genes, RNA-Seq data were downloaded from the NCBI database (PRJNA845109), including data for various tissues (stems, leaves, flowers, and fruits). The estimated expression levels of the BBX genes were represented and normalized in the form of fragments per kilobase of transcript per million mapped (FPKM). The heatmap for LbaBBX genes was visualized using Tbtools [56].
4.9. Quantitative Real-Time PCR (qRT-PCR) Analysis
RNA extraction and qRT-PCR were performed as previously described [24]. The primers for LbaBBX genes were designed using Primer Premier 5 and are listed in Table S10. The wolfberry Actin gene was used as an internal control [20]. Three independent biological replications were conducted.
4.10. Carotenoid Extraction and HPLC Analysis
The extraction steps were as follows. Freeze-dried fruits were homogenized (30 Hz, 1.5 min) to a powder with a grinder (MM 400; Retsch, Haan, Germany). A mixture of n-hexane: acetone: ethanol (1:1:1, v/v/v) was prepared as an extraction solution, and then 0.01% BHT (g/mL) and 50 mg of power were mixed with an appropriate amount of extraction solution and internal standard. The extract was vortexed for 20 min at room temperature. The mixture was then centrifuged at 12,000 rpm/min for 5 min at 4 °C, and the supernatant was removed. The residue was re-extracted by repeating Steps under the same conditions. The supernatants were combined and evaporated to dryness. A mixture of methanol and methyl tert butyl ether was prepared; the sample was resuspended with an appropriate amount of the solution, vortexed thoroughly until it was fully dissolved, and centrifuged. The solution was filtered through a 0.22 μm membrane filter for further LC-MS/MS analysis [59]. Carotenoid contents were detected using the AB Sciex WTRAP 6500 LC-MS/MS platform by MetWare (Wuhan, China).
4.11. Correlation Network Construction
Expression patterns were explored based on RNA-seq data for five stages. The correlation coefficients for relationships between gene pairs and the carotenoid content were measured based on Pearson’s correlation coefficients (PCC). These values were screened using Excel (threshold > 0.8). A network including carotenoid contents, LbaBBX TFs, and structural genes was constructed and visualized using Cytoscape [60].
4.12. Subcellular Localization
For subcellular location assays, the 35S-LbaBBX2::GFP, 35S-LbaBBX4::GFP, and 35S::NLS-RFP (control) constructs were introduced into tobacco (Nicotiana benthamiana) epidermal cells via Agrobacterium infiltrated tobacco leaves. Samples transformed with 35-GFP were used as controls. After 2 days, GFP and RFP signals from the tobacco leaves inoculated with A. tumefaciens were detected by fluorescence microscopy (Olympus, BX63; Tokyo, Japan). Three independent experiments were performed for each gene.
4.13. Statistical Analyses
The data are presented as means ± SD of at least three independent experiments. Differences were evaluated by the Student’s t-test (* p < 0.05, ** p < 0.01, *** p < 0.001).
5. Conclusions
Our study provided the first genome-wide analysis of the BBX gene family in L. barbarum. A total of 28 LbaBBXs were identified and were unevenly distributed across the whole genome. A systematic and comprehensive analysis of the LbaBBX gene family was performed, including analyses of phylogenetic relationships, conserved domains, gene structure, motif composition, chromosome location, gene duplication, cis-acting elements, and expression patterns. Many cis-acting elements were found in the LbaBBX promoter sequences, indicating that LbaBBX genes are involved in complex regulatory networks controlling development. Correlation and qRT-PCR analyses revealed that LbaBBX genes might be involved in the regulation of carotenoid synthesis. Therefore, our genome-wide analysis of the BBX family provides a foundation for further studies of the molecular mechanisms underlying carotenoid synthesis in wolfberry.
Abbreviations
| TFs | Transcription factors |
| HMM | Hidden Markov model |
| CDS | Coding sequence |
| GRAVY | Grand average of hydropathicity |
| M W | Molecular weight |
| FPKM | Fragments per Kilobase Million |
| Ks | Synonymous substitutions |
| Ka | Nonsynonymous substitutions |
| GFP | Green Fluorescent Protein |
| RFP | Red Fluorescent Protein |
| NCBI | National Center for Biotechnology Information |
| qRT-PCR | Quantitative Real-Time PCR |
| WGD | Whole genome duplication |
| TD | Tandem duplication |
| PD | Proximal duplication |
| TRD | Transposed duplication |
| DSD | Dispersed duplication |
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms23158440/s1.
Author Contributions
Conceptualization, Y.Y. and H.S.; methodology, J.M.; software, Y.Y.; validation, D.Z. and C.G.; investigation, X.H. and X.Q.; resources, J.Z. (Jianhua Zhao) and W.A.; data curation, Y.Y. and J.M.; Writing—original draft preparation, Y.Y. and Y.C.; visualization, Y.C.; supervision and manuscript revisions, Y.C., J.Z. (Jianhua Zhu) and X.Z. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The wolfberry genome datasets used during the current study are available in NCBI database (https://www.ncbi.nlm.nih.gov/bioproject/PRJNA640228, accessed on 20 December 2021). The tomato, pepper, eggplant, and potato genome sequences were downloaded from the Genome Database for the Solanaceae (https://solgenomics.net/, accessed on 20 December 2021). The sequence of Arabidopsis was downloaded from the Arabidopsis Information Resource (https://www.arabidopsis.org/, accessed on 20 December 2021). The raw data of the transcriptome analysis used in this study were submitted to the Sequence Read Archive (SRA) at a NCBI database (PRJNA845109).
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research was funded by the Key Research and Development projects of Ningxia Hui Autonomous Region (No.2022BBF02008), the Natural Foundation of Ningxia (No.2020AAC03284), and the Employee Innovation Project of All-China Federation of Trade Unions (No.2018300002).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Takatsuji H. Zinc-finger transcription factors in plants. Cell. Mol. Life Sci. CMLS. 1998;54:582–596. doi: 10.1007/s000180050186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kielbowicz-Matuk A. Involvement of plant C2H2-type zinc finger transcription factors in stress responses. Plant Sci. 2012;185–186:78–85. doi: 10.1016/j.plantsci.2011.11.015. [DOI] [PubMed] [Google Scholar]
- 3.Crocco C.D., Botto J.F. BBX proteins in green plants: Insights into their evolution, structure, feature and functional diversification. Gene. 2013;531:44–52. doi: 10.1016/j.gene.2013.08.037. [DOI] [PubMed] [Google Scholar]
- 4.Yu L., Lyu Z., Liu H., Zhang G., He C., Zhang J. Insights into the evolutionary origin and expansion of the BBX gene family. Plant Biotechnol. Rep. 2022;16:205–214. doi: 10.1007/s11816-022-00745-1. [DOI] [Google Scholar]
- 5.Jang S., Marchal V., Panigrahi K.C., Wenkel S., Soppe W., Deng X.W., Valverde F., Coupland G. Arabidopsis COP1 shapes the temporal pattern of CO accumulation conferring a photoperiodic flowering response. EMBO J. 2008;27:1277–1288. doi: 10.1038/emboj.2008.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Robson F., Costa M.M.R., Hepworth S.R., Vizir I., Piñeiro M., Reeves P.H., Putterill J., Coupland G. Functional importance of conserved domains in the flowering-time gene CONSTANS demonstrated by analysis of mutant alleles and transgenic plants. Plant J. 2001;28:619–631. doi: 10.1046/j.1365-313x.2001.01163.x. [DOI] [PubMed] [Google Scholar]
- 7.Khanna R., Kronmiller B., Maszle D.R., Coupland G., Holm M., Mizuno T., Wu S.-H. The Arabidopsis B-Box Zinc Finger Family. Plant Cell. 2009;21:3416–3420. doi: 10.1105/tpc.109.069088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang M.J., Ding L., Liu X.H., Liu J.X. Two B-box domain proteins, BBX28 and BBX29, regulate flowering time at low ambient temperature in Arabidopsis. Plant Mol. Biol. 2021;106:21–32. doi: 10.1007/s11103-021-01123-1. [DOI] [PubMed] [Google Scholar]
- 9.Heng Y., Lin F., Jiang Y., Ding M., Yan T., Lan H., Zhou H., Zhao X., Xu D., Deng X.W. B-Box Containing Proteins BBX30 and BBX31, Acting Downstream of HY5, Negatively Regulate Photomorphogenesis in Arabidopsis. Plant Physiol. 2019;180:497–508. doi: 10.1104/pp.18.01244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Crocco C.D., Holm M., Yanovsky M.J., Botto J.F. AtBBX21 and COP1 genetically interact in the regulation of shade avoidance. Plant J. 2010;64:551–562. doi: 10.1111/j.1365-313X.2010.04360.x. [DOI] [PubMed] [Google Scholar]
- 11.Xiong C., Luo D., Lin A., Zhang C., Shan L., He P., Li B., Zhang Q., Hua B., Yuan Z., et al. A tomato B-box protein SlBBX20 modulates carotenoid biosynthesis by directly activating PHYTOENE SYNTHASE 1, and is targeted for 26S proteasome-mediated degradation. New Phytol. 2019;221:279–294. doi: 10.1111/nph.15373. [DOI] [PubMed] [Google Scholar]
- 12.Xu X., Wang Q., Li W., Hu T., Wang Q., Yin Y., Liu X., He S., Zhang M., Liang Y., et al. Overexpression of SlBBX17 affects plant growth and enhances heat tolerance in tomato. Int. J. Biol. Macromol. 2022;206:799–811. doi: 10.1016/j.ijbiomac.2022.03.080. [DOI] [PubMed] [Google Scholar]
- 13.An H., Roussot C., Suarez-Lopez P., Corbesier L., Vincent C., Pineiro M., Hepworth S., Mouradov A., Justin S., Turnbull C., et al. CONSTANS acts in the phloem to regulate a systemic signal that induces photoperiodic flowering of Arabidopsis. Development. 2004;131:3615–3626. doi: 10.1242/dev.01231. [DOI] [PubMed] [Google Scholar]
- 14.Datta S., Hettiarachchi G.H., Deng X.W., Holm M. Arabidopsis CONSTANS-LIKE3 is a positive regulator of red light signaling and root growth. Plant Cell. 2006;18:70–84. doi: 10.1105/tpc.105.038182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hassidim M., Harir Y., Yakir E., Kron I., Green R.M. Over-expression of CONSTANS-LIKE 5 can induce flowering in short-day grown Arabidopsis. Planta. 2009;230:481–491. doi: 10.1007/s00425-009-0958-7. [DOI] [PubMed] [Google Scholar]
- 16.Cheng X.F., Wang Z.Y. Overexpression of COL9, a CONSTANS-LIKE gene, delays flowering by reducing expression of CO and FT in Arabidopsis thaliana. Plant J. 2005;43:758–768. doi: 10.1111/j.1365-313X.2005.02491.x. [DOI] [PubMed] [Google Scholar]
- 17.Liu W., Tang R., Zhang Y., Liu X., Gao Y., Dai Z., Li S., Wu B., Wang L. Genome-wide identification of B-box proteins and VvBBX44 involved in light-induced anthocyanin biosynthesis in grape (Vitis vinifera L.) Planta. 2021;253:114. doi: 10.1007/s00425-021-03618-z. [DOI] [PubMed] [Google Scholar]
- 18.Bai S., Tao R., Tang Y., Yin L., Ma Y., Ni J., Yan X., Yang Q., Wu Z., Zeng Y., et al. BBX16, a B-box protein, positively regulates light-induced anthocyanin accumulation by activating MYB10 in red pear. Plant Biotechnol. J. 2019;17:1985–1997. doi: 10.1111/pbi.13114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Long J.T., Fan H.X., Zhou Z.Q., Sun W.Y., Li Q.W., Wang Y., Ma M., Gao H., Zhi H. The major zeaxanthin dipalmitate derivatives from wolfberry. J. Asian Nat. Prod. Res. 2019;22:746–753. doi: 10.1080/10286020.2019.1621855. [DOI] [PubMed] [Google Scholar]
- 20.Liu Y., Zeng S., Sun W., Wu M., Hu W., Shen X., Wang Y. Comparative analysis of carotenoid accumulation in two goji (Lycium barbarum L. and L. ruthenicum Murr.) fruits. BMC Plant Biol. 2014;14:269. doi: 10.1186/s12870-014-0269-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu M., Xu X., Hu X., Liu Y., Cao H., Chan H., Gong Z., Yuan Y., Luo Y., Feng B., et al. SlMYB72 Regulates the Metabolism of Chlorophylls, Carotenoids, and Flavonoids in Tomato Fruit. Plant Physiol. 2020;183:854–868. doi: 10.1104/pp.20.00156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yuan Y., Ren S., Liu X., Su L., Wu Y., Zhang W., Li Y., Jiang Y., Wang H., Fu R., et al. SlWRKY35 positively regulates carotenoid biosynthesis by activating the MEP pathway in tomato fruit. New Phytol. 2022;234:164–178. doi: 10.1111/nph.17977. [DOI] [PubMed] [Google Scholar]
- 23.Dang Q., Sha H., Nie J., Wang Y., Yuan Y., Jia D. An apple (Malus domestica) AP2/ERF transcription factor modulates carotenoid accumulation. Hortic. Res. 2021;8:223. doi: 10.1038/s41438-021-00694-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yin Y., Guo C., Shi H., Zhao J., Ma F., An W., He X., Luo Q., Cao Y., Zhan X. Genome-Wide Comparative Analysis of the R2R3-MYB Gene Family in Five Solanaceae Species and Identification of Members Regulating Carotenoid Biosynthesis in Wolfberry. Int. J. Mol. Sci. 2022;23:2259. doi: 10.3390/ijms23042259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bu X., Wang X., Yan J., Zhang Y., Zhou S., Sun X., Yang Y., Ahammed G.J., Liu Y., Qi M., et al. Genome-Wide Characterization of B-Box Gene Family and Its Roles in Responses to Light Quality and Cold Stress in Tomato. Front. Plant Sci. 2021;12:698525. doi: 10.3389/fpls.2021.698525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ma J., Dai J.X., Liu X.W., Lin D. Genome-wide and expression analysis of B-box gene family in pepper. BMC Genom. 2021;22:883. doi: 10.1186/s12864-021-08186-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang Y., Zhang Y., Liu Q., Zhang T., Chong X., Yuan H. Genome-Wide Identification and Expression Analysis of BBX Transcription Factors in Iris germanica L. Int. J. Mol. Sci. 2021;22:8793. doi: 10.3390/ijms22168793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang Y., Zhai Z., Sun Y., Feng C., Peng X., Zhang X., Xiao Y., Zhou X., Wang W., Jiao J., et al. Genome-Wide Identification of the B-BOX Genes that Respond to Multiple Ripening Related Signals in Sweet Cherry Fruit. Int. J. Mol. Sci. 2021;22:1622. doi: 10.3390/ijms22041622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wei H., Wang P., Chen J., Li C., Wang Y., Yuan Y., Fang J., Leng X. Genome-wide identification and analysis of B-BOX gene family in grapevine reveal its potential functions in berry development. BMC Plant Biol. 2020;20:72. doi: 10.1186/s12870-020-2239-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gangappa S.N., Botto J.F. The BBX family of plant transcription factors. Trends Plant Sci. 2014;19:460–470. doi: 10.1016/j.tplants.2014.01.010. [DOI] [PubMed] [Google Scholar]
- 31.Cao Y.L., Li Y.L., Fan Y.F., Li Z., Yoshida K., Wang J.Y., Ma X.K., Wang N., Mitsuda N., Kotake T., et al. Wolfberry genomes and the evolution of Lycium (Solanaceae) Commun. Biol. 2021;4:671. doi: 10.1038/s42003-021-02152-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Qiao X., Li Q., Yin H., Qi K., Li L., Wang R., Zhang S., Paterson A.H. Gene duplication and evolution in recurring polyploidization-diploidization cycles in plants. Genome Biol. 2019;20:38. doi: 10.1186/s13059-019-1650-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Talar U., Kielbowicz-Matuk A., Czarnecka J., Rorat T. Genome-wide survey of B-box proteins in potato (Solanum tuberosum)-Identification, characterization and expression patterns during diurnal cycle, etiolation and de-etiolation. PLoS ONE. 2017;12:e0177471. doi: 10.1371/journal.pone.0177471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu X., Li R., Dai Y., Chen X., Wang X. Genome-wide identification and expression analysis of the B-box gene family in the Apple (Malus domestica Borkh.) genome. Mol. Genet. Genom. 2018;293:303–315. doi: 10.1007/s00438-017-1386-1. [DOI] [PubMed] [Google Scholar]
- 35.Wang B., Yang X., Jia Y., Xu Y., Jia P., Dang N., Wang S., Xu T., Zhao X., Gao S., et al. High-quality Arabidopsis thaliana Genome Assembly with Nanopore and HiFi Long Reads. Genom. Proteom. Bioinform. 2021. in press . [DOI] [PMC free article] [PubMed]
- 36.Sato S., Tabata S., Hirakawa H., Asamizu E., Shirasawa K., Isobe S., Kaneko T., Nakamura Y., Shibata D., Aoki K., et al. The tomato genome sequence provides insights into fleshy fruit evolution. Nature. 2012;485:635–641. doi: 10.1038/nature11119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Qin C., Yu C., Shen Y., Fang X., Chen L., Min J., Cheng J., Zhao S., Xu M., Luo Y., et al. Whole-genome sequencing of cultivated and wild peppers provides insights into Capsicum domestication and specialization. Proc. Natl. Acad. Sci. USA. 2014;111:5135–5140. doi: 10.1073/pnas.1400975111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cao Y., Han Y., Meng D., Li D., Jiao C., Jin Q., Lin Y., Cai Y. B-BOX genes: Genome-wide identification, evolution and their contribution to pollen growth in pear (Pyrus bretschneideri Rehd.) BMC Plant Biol. 2017;17:156. doi: 10.1186/s12870-017-1105-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Friedman R., Hughes A.L. Pattern and timing of gene duplication in animal genomes. Genome Res. 2001;11:1842–1847. doi: 10.1101/gr.200601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu C., Qiao X., Li Q., Zeng W., Wei S., Wang X., Chen Y., Wu X., Wu J., Yin H., et al. Genome-wide comparative analysis of the BAHD superfamily in seven Rosaceae species and expression analysis in pear (Pyrus bretschneideri) BMC Plant Biol. 2020;20:14. doi: 10.1186/s12870-019-2230-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhu X., Tang C., Li Q., Qiao X., Li X., Cai Y., Wang P., Sun Y., Zhang H., Zhang S., et al. Characterization of the pectin methylesterase inhibitor gene family in Rosaceae and role of PbrPMEI23/39/41 in methylesterified pectin distribution in pear pollen tube. Planta. 2021;253:118. doi: 10.1007/s00425-021-03638-9. [DOI] [PubMed] [Google Scholar]
- 42.Yadav A., Ravindran N., Singh D., Rahul P.V., Datta S. Role of Arabidopsis BBX proteins in light signaling. J. Plant Biochem. Biotechnol. 2020;29:623–635. doi: 10.1007/s13562-020-00597-2. [DOI] [Google Scholar]
- 43.An J.P., Wang X.F., Espley R.V., Lin-Wang K., Bi S.Q., You C.X., Hao Y.J. An Apple B-Box Protein MdBBX37 Modulates Anthocyanin Biosynthesis and Hypocotyl Elongation Synergistically with MdMYBs and MdHY5. Plant Cell Physiol. 2020;61:130–143. doi: 10.1093/pcp/pcz185. [DOI] [PubMed] [Google Scholar]
- 44.Mistry J., Chuguransky S., Williams L., Qureshi M., Salazar G.A., Sonnhammer E.L.L., Tosatto S.C.E., Paladin L., Raj S., Richardson L.J., et al. Pfam: The protein families database in 2021. Nucleic Acids Res. 2021;49:D412–D419. doi: 10.1093/nar/gkaa913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Letunic I., Khedkar S., Bork P. SMART: Recent updates, new developments and status in 2020. Nucleic Acids Res. 2021;49:D458–D460. doi: 10.1093/nar/gkaa937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lu S., Wang J., Chitsaz F., Derbyshire M.K., Geer R.C., Gonzales N.R., Gwadz M., Hurwitz D.I., Marchler G.H., Song J.S., et al. CDD/SPARCLE: The conserved domain database in 2020. Nucleic Acids Res. 2020;48:D265–D268. doi: 10.1093/nar/gkz991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wilkins M.R., Gasteiger E., Bairoch A., Sanchez J.C., Williams K.L., Appel R.D., Hochstrasser D.F. Protein identification and analysis tools in the ExPASy server. Methods Mol. Biol. 1999;112:531–552. doi: 10.1385/1-59259-584-7:531. [DOI] [PubMed] [Google Scholar]
- 48.Horton P., Park K.J., Obayashi T., Fujita N., Harada H., Adams-Collier C.J., Nakai K. WoLF PSORT: Protein localization predictor. Nucleic Acids Res. 2007;35:W585–W587. doi: 10.1093/nar/gkm259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Edgar R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004;32:1792–1797. doi: 10.1093/nar/gkh340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nicholas K.B., Nicholas H.B., Deerfield D.W. GeneDoc: Analysis and visualization of genetic variation. Embnew. News. 1997;4:1–22. [Google Scholar]
- 51.Minh B.Q., Schmidt H.A., Chernomor O., Schrempf D., Woodhams M.D., von Haeseler A., Lanfear R. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol. Biol. Evol. 2020;37:1530–1534. doi: 10.1093/molbev/msaa015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tamura K., Stecher G., Peterson D., Filipski A., Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 2013;30:2725–2729. doi: 10.1093/molbev/mst197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Letunic I., Bork P. Interactive Tree Of Life (iTOL) v5: An online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021;49:W293–W296. doi: 10.1093/nar/gkab301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hu B., Jin J., Guo A.Y., Zhang H., Luo J., Gao G. GSDS 2.0: An upgraded gene feature visualization server. Bioinformatics. 2015;31:1296–1297. doi: 10.1093/bioinformatics/btu817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bailey T.L., Johnson J., Grant C.E., Noble W.S. The MEME Suite. Nucleic Acids Res. 2015;43:W39–W49. doi: 10.1093/nar/gkv416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chen C., Chen H., Zhang Y., Thomas H.R., Frank M.H., He Y., Xia R. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant. 2020;13:1194–1202. doi: 10.1016/j.molp.2020.06.009. [DOI] [PubMed] [Google Scholar]
- 57.Zhang Z. KaKs_calculator 3.0: Calculating selective pressure on coding and non-coding sequences. Genom. Proteom. Bioinform. 2022. in press . [DOI] [PMC free article] [PubMed]
- 58.Zhang L., Miao L., He J., Li H., Li M. The Transcriptome and Metabolome Reveal the Potential Mechanism of Lodging Resistance in Intergeneric Hybrids between Brassica napus and Capsella bursa-pastoris. Int. J. Mol. Sci. 2022;23:4481. doi: 10.3390/ijms23094481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhou W., Niu Y., Ding X., Zhao S., Li Y., Fan G., Zhang S., Liao K. Analysis of carotenoid content and diversity in apricots (Prunus armeniaca L.) grown in China. Food Chem. 2020;330:127223. doi: 10.1016/j.foodchem.2020.127223. [DOI] [PubMed] [Google Scholar]
- 60.Otasek D., Morris J.H., Boucas J., Pico A.R., Demchak B. Cytoscape Automation: Empowering workflow-based network analysis. Genome Biol. 2019;20:185. doi: 10.1186/s13059-019-1758-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The wolfberry genome datasets used during the current study are available in NCBI database (https://www.ncbi.nlm.nih.gov/bioproject/PRJNA640228, accessed on 20 December 2021). The tomato, pepper, eggplant, and potato genome sequences were downloaded from the Genome Database for the Solanaceae (https://solgenomics.net/, accessed on 20 December 2021). The sequence of Arabidopsis was downloaded from the Arabidopsis Information Resource (https://www.arabidopsis.org/, accessed on 20 December 2021). The raw data of the transcriptome analysis used in this study were submitted to the Sequence Read Archive (SRA) at a NCBI database (PRJNA845109).