Abstract
Aim: Recently, the atherogenic index of plasma (AIP) has been proposed as a novel, reliable plasma atherogenicity marker. This study aimed to investigate the association of AIP with the risk of in-stent restenosis (ISR) in patients with acute coronary syndrome (ACS).
Methods: This study retrospectively enrolled patients with ACS followed by angiography within 6 to 18 months after successful percutaneous coronary intervention (PCI) with a drug-eluting stent (DES). And the participants were divided into ISR or non-ISR groups based on the angiographic follow-up results. AIP was defined as the base 10 logarithm of the ratio of serum triglyceride (mmol/L) to high-density lipoprotein cholesterol (mmol/L).
Results: This study recruited 1319 patients with ACS, 199 of which had ISR. Compared with the non-ISR group, patients in the ISR group had higher level of AIP (0.199±0.290 vs 0.131±0.282,p=0.002). In the multiple logistic regression analysis, AIP was an independent risk factor for DES-ISR (OR=2.100, 95% CI 1.134 to 3.891,p=0.018). When we modulated AIP as a categorical variable, the risk of DES-ISR increased in quartile 4 compared to quartile 1 (OR=1.713, 95% CI 1.040 to 2.822,p=0.034). Furthermore, this association remains stable in various subgroups. Unexpectedly, the subgroup analysis suggested AIP and DES-ISR had a stronger positive association in individuals with low-density lipoprotein cholesterol (LDL-C) <1.8 mmol/L.
Conclusions: AIP and the risk of DES-ISR were positively and independently correlated in patients with ACS, especially in those with an LDL-C <1.8 mmol/L.
Keywords: Atherogenic index of plasma, Drug-eluting stent, In-stent restenosis, Acute coronary syndrome, Low-density lipoprotein cholesterol
Introduction
Despite advances in the primary and secondary prevention of cardiovascular disease in recent years, the morbidity and mortality of atherosclerotic cardiovascular diseases (ASCVDs) remain high in contemporary society 1 , 2) . Atherogenic dyslipidemia, characterized by reduced concentration of high-density lipoprotein cholesterol (HDL-C), increased circulating triglyceride (TG), and high small dense low-density lipoprotein (sdLDL) with normal low-density lipoprotein cholesterol (LDL-C), was considered a substantial residual risk for ASCVDs and may account for it 3 - 6) .
The atherogenic index of plasma (AIP), determined with formula log10 (TG/HDL-C) and associated with lipoprotein particle size, could reflect the main atherogenic dyslipidemia characteristics and regarded as a plasma atherogenicity marker 7 - 9) . According to previous studies, AIP was associated with various risk factors for ASCVD, such as insulin resistance, abdominal obesity, diabetes mellitus (DM), and metabolic syndrome 10 - 13) . Meanwhile, mounting evidence associated AIP with the prevalence and severity of coronary artery disease (CAD) 10 , 14) . AIP was an independent risk factor for the rapid progress of coronary atherosclerosis 15 , 16) . Furthermore, recent evidence also demonstrated AIP was associated with a worse prognosis and could serve as a biomarker to predict long-term prognoses in patients with acute coronary syndrome (ACS) 17 , 18) .
However, to the best of our knowledge, no prior studies have been designed to evaluate the association between AIP and in-stent restenosis (ISR), which remains as the primary cause of percutaneous coronary intervention (PCI) failure even in the era of drug-eluting stents (DES) 19) .
Aim
To fill this knowledge gap and provide a novel promising way to prevent ISR, we aim to assess the correlation of AIP with ISR in patients with ACS after DES implantation.
Methods
Study Population
In this single-center, retrospective, and observational study, we consecutively screened the patients with ACS admitted for PCI in Beijing AnZhen Hospital, Capital Medical University, from January 2018 to August 2020. The main inclusion criteria were: (1) culprit lesions in native coronary artery treated successfully by PCI (successful PCI was defined as the recanalization of lesion with thrombolysis in myocardial infarction grade 3 and residual stenosis <10%); (2) patients followed by coronary angiography within 6 to 18 months after successful PCI. We also excluded patients according to the following criteria: (1) age <18 years; (2) previous history of coronary artery bypass grafting (CABG); (3) target lesion only treated by PCI with a bare metal stent or percutaneous transluminal coronary angioplasty (PTCA); (4) those with severe dysfunction in liver, renal, or thyroid; (5) cancer, acute/chronic inflammatory disease, and patients without completed baseline data. The Ethics Committee of Beijing AnZhen Hospital, Capital Medical University approved this study, which strictly complied with the Declaration of Helsinki. Meanwhile, all the patients provided informed consent.
Procedure and Periprocedural Management
Diagnostic coronary angiography, PCI, and periprocedural management were all implemented according to current guidelines 20) . All patients enrolled received a loading dose of aspirin plus clopidogrel or ticagrelor before the intervention. Additionally, unfractionated heparin (70–100 U/Kg, with additional boluses if required) was also administered at the beginning/during the PCI to maintain the activated clotting time >250 s. Most importantly, the PCI was performed by experienced interventional cardiologists using 6F or 7 F (1F=0.33 mm) guiding catheter mainly with radial access. And the application of intracoronary imaging techniques, the type of DES, and the size of stents were all left to the operator’s discretion.
Data Collection and Definitions
A trained physician, blinded to the study objective, collected the participants’ demographic and clinical characteristics, including age, gender, height, weight, admission blood pressure, medical history, cardiac function, laboratory parameters, procedural details, and medication used at discharge, with a dedicated case report form. Noteworthy, venous blood samples were obtained after patients fasted overnight (>8 h). Common hematology and biochemical parameters, including high sensitivity C-reactive protein (Hs-CRP), glycosylated hemoglobin A1c (HbA1c), estimated glomerular filtration rate (eGFR), and lipid profile, were all determined in center laboratory of Beijing AnZhen Hospital.
In line with previous studies 9) , AIP in the present study was calculated with the formula Log10(TG/HDL-C). Body mass index (BMI) was computed as weight (Kg) divided by height squared (m). Hypertension and DM were ascertained based on self-report of physician’s diagnosis, which complied with criteria from current guidelines 21 , 22) , and/or active therapy with medications. Hypercholesterolemia was defined as LDL-C >4.14 mmol/L, and/or total cholesterol (TC) >6. 22 mmol/L, or active therapy with lipid-lowering drugs. The SYNTAX and Gensini scores, which evaluate coronary disease severity, were calculated by 2 independent intervention cardiologists blinded to the participant’s information.
Follow-Up Angiography and Evaluation of ISR
The patients recruited were followed up by coronary angiography at 6 to 18 months after DES implantation. The ISR was defined as a lumen diameter stenosis ≥ 50% occurred at the segment inside the stent or involving its 5 mm-edges. The 2 independent intervention cardiologists were responsible for interpreting follow-up angiography and further dividing the participants into ISR or non-ISR groups. The disagreements encountered were resolved by a discussion with senior researcher.
Statistical Analysis
Continuous variables with a normal distribution were expressed as mean±standard deviation, and those with skewed distribution were summarized as median with interquartile ranges. The difference in continuous variables between the ISR group and non-ISR group was examined by the Student’s t test or Mann–Whitney U-test, which depends on normality of data distribution. Meanwhile, categorical variables were presented as numbers (percentage) and compared by the Pearson chi-square test or Fisher’s exact test. We also performed the Pearson correlation test and Spearman’s rank correlation test to evaluate the correlation between AIP and traditional cardiometabolic risk factors.
Subsequently, the determinants of DES-ISR were explored by us using univariate logistic regression analysis. Variables showed p<0.1 in univariate analysis and those judged to be of clinical significance were purposefully introduced into the following multivariate logistic regression analysis models. Model 1 included age, sex, and BMI. Model 2 consisted of variables in Model 1 and systolic blood pressure, hypertension, previous PCI, Hs-CRP, platelet count, HbA1c, TC, and left ventricular ejection fraction. Model 3 was used to adjust confounders in Model 2 and SYNTAX score, target in multivessel, target in the left anterior descending artery (LAD), target in right coronary artery, multiple stents (≥ 2), DES-sirolimus, minimal stent diameter ≤ 3 mm, intracoronary imaging technique, and follow-up time. Model 4, as the fully adjusted model, was established to adjust variables in Model 3 and the medical therapy with angiotensin-converting enzyme inhibitor/angiotensin receptor blocker (ACEI/ARB), insulin, and other hypoglycemic agents.
All the statistical analyses involved in the present study were performed using SPSS 20.0 (IBM, Armonk, New York), and a two-sided p<0.05 was regarded as statistically significant.
Results
Baseline Characteristics
A total of 1319 patients (84.7% were diagnosed as unstable angina) who were followed up by coronary angiography at 11.11±3.04 months after successful PCI were enrolled for the final analysis. As shown in Table 1 , the average age of the study population was 58.40±9.47 years, and 77.7% of participants were men. The percentages of current smokers, hypertension, DM, and previous PCI were 35.3%, 65.1%, 36.1%, and 17.5%, respectively. Meanwhile, more than half of the participants underwent PCI for LAD and implanted multiple stents (≥ 2).
Table 1. Baseline characteristics of the study population.
| Variables | Total (n = 1319) | ISR group (n = 199) | Non-ISR group (n = 1120) | P-value |
|---|---|---|---|---|
| Demographics | ||||
| Age, years | 58.40±9.47 | 58.96±10.00 | 58.30±9.37 | 0.368 |
| Male, n (%) | 1025 (77.7) | 158 (79.4) | 867 (77.4) | 0.535 |
| BMI, Kg/m2 | 25.92±3.24 | 26.05±2.84 | 25.90±3.30 | 0.552 |
| SBP, mmHg | 129.13±17.34 | 131.11±17.83 | 128.78±17.23 | 0.081 |
| DBP, mmHg | 76.42±11.20 | 75.63±11.87 | 76.56±11.08 | 0.281 |
| Diagnosis, n (%) | 0.792 | |||
| UA | 1117 (84.7) | 167 (83.9) | 950 (84.8) | |
| NSTEMI | 98 (7.4) | 14 (7.0) | 84 (7.5) | |
| STEMI | 104 (7.9) | 18 (9.0) | 86 (7.7) | |
| Medical History, n (%) | ||||
| Current smoker | 466 (35.3) | 74 (37.2) | 392 (350) | 0.552 |
| Hypertension | 859 (65.1) | 142 (71.4) | 717 (64.0) | 0.045 |
| Diabetes mellitus | 476 (36.1) | 97 (48.7) | 379 (33.8) | <0.001 |
| Hypercholesterolemia | 514 (39.0) | 77 (38.7) | 437 (39.0) | 0.931 |
| Stroke | 107 (8.1) | 20 (10.1) | 87 (7.8) | 0.277 |
| Previous PCI | 231 (17.5) | 55 (27.6) | 176 (15.7) | <0.001 |
| Cardiac Function | ||||
| LVEF, % | 62.14±6.91 | 61.28±7.91 | 62.29±6.71 | 0.090 |
| Laboratory Results | ||||
| Hs-CRP, mg/L | 1.45 (0.57,4.12) | 2.23 (0.72, 5.26) | 1.33 (0.55, 3.89) | <0.001 |
| Platelet, 109/L | 223.99±56.17 | 231.13±62.71 | 222.72±54.87 | 0.077 |
| Hemoglobin, g/L | 144.24±14.88 | 145.47±15.24 | 144.03±14.81 | 0.209 |
| Uric acid, umol/L | 345.36±84.40 | 339.84±75.49 | 346.34±85.88 | 0.275 |
| Triglyceride, mmol/L | 1.40 (1.03, 1.96) | 1.52 (1.14, 2.05) | 1.38 (1.01, 1.94) | 0.011 |
| LDL-C, mmol/L | 2.45±0.89 | 2.52±0.97 | 2.44±0.88 | 0.241 |
| HDL-C, mmol/L | 1.07±0.24 | 1.03±0.23 | 1.07±0.24 | 0.017 |
| TC, mmol/L | 4.12±1.16 | 4.26±1.45 | 4.10±1.09 | 0.140 |
| HbA1c, % | 6.55±1.35 | 7.14±1.63 | 6.44±1.27 | <0.001 |
| eGFR, ml/min/1.73m2 | 96.21±14.88 | 96.20±15.31 | 96.21±14.81 | 0.991 |
| Intervention, n (%) | ||||
| Gensini score | 40.00 (25.00,62.00) | 47.00 (29.00,72.00) | 40.00 (24.00,60.00) | 0.001 |
| SYNTAX score | 13.00 (8.50, 18.00) | 13.00 (9.00,21.00) | 12.00 (8.00,18.00) | 0.024 |
| No. of target vessel | 0.002 | |||
| One | 989 (75.0) | 132 (66.3) | 857 (76.5) | |
| Multivessel | 330 (25.0) | 67 (33.7) | 263 (23.5) | |
| Target in LM | 66 (5.0) | 11 (5.5) | 55 (4.9) | 0.713 |
| Target in LAD | 743 (56.3) | 123 (61.8) | 620 (55.4) | 0.091 |
| Target in LCX | 356 (27.0) | 54 (27.1) | 302 (27.0) | 0.960 |
| Target in RCA | 544 (41.2) | 100(50.3) | 444 (39.6) | 0.005 |
| DES-sirolimus | 707 (53.6) | 121 (60.8) | 586 (52.3) | 0.027 |
| DES-zotarolimus | 285 (21.6) | 46 (23.1) | 239 (21.3) | 0.575 |
| DES-everolimus | 517 (39.2) | 75 (37.7) | 442 (39.5) | 0.636 |
| Intracoronary imagine | 88 (6.7) | 7 (3.5) | 81 (7.2) | 0.053 |
| Multiple Stents (≥ 2) | 667 (50.6) | 118 (59.3) | 549 (49.0) | 0.008 |
| Minimal stent diameter ≤ 3 mm | 1057 (80.1) | 170 (85.4) | 887 (79.2) | 0.042 |
| Follow up time, months | 11.11±3.04 | 10.62±3.02 | 11.19±3.04 | 0.015 |
| Medication used after discharge, n (%) | ||||
| Aspirin | 1319 (100.0) | 199 (100.0) | 1120 (100.0) | > 0.999 |
| Clopidogrel/Ticagrelor | 1319 (100.0) | 199 (100.0) | 1120 (100.0) | > 0.999 |
| Statin | 1316 (99.8) | 199 (100.0) | 1117 (99.7) | 0.465 |
| β-block | 924 (70.1) | 143 (71.9) | 781 (69.7) | 0.546 |
| ACEI/ARB | 614 (46.6) | 107 (53.8) | 507 (45.3) | 0.027 |
| Insulin | 150 (11.4) | 38 (19.1) | 112 (10.0) | <0.001 |
| Other hypoglycemic agents | 377 (28.6) | 78 (39.2) | 299 (26.7) | <0.001 |
ISR, in-stent restenosis; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; UA, unstable angina; NSTEMI, non- ST-segment elevation myocardial infarction; STEMI, ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention; LVEF, left ventricular ejection fraction; Hs-CRP, high sensitivity C-reactive protein; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TC, total cholesterol; HbA1c, glycosylated hemoglobin A1c; eGFR, estimated glomerular filtration rate; LM, left main artery; LAD, left anterior descending artery; LCX, left circumflex artery; RCA, right coronary artery; DES, drug-eluting stents; ACEI/ARB, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker.
Later, the study population was divided into ISR and non-ISR groups. As demonstrated in Table 1 , the prevalence of hypertension, DM, and previous PCI; serum Hs-CRP, TG, and HbA1c; SYNTAX score and Gensini score were all higher in the ISR group than the non-ISR group. Furthermore, the patients in the ISR group were more likely to undergo PCI for multivessel and implant multiple stents. However, the patients in the ISR group had lower serum HDL-C and were less likely to take intracoronary imaging techniques.
Correlation between AIP and Cardiometabolic Risk Factors
The distribution of AIP is normal, and the mean AIP of the study population was 0.14±0.28. As shown in Table 2 , AIP correlated positively with various cardiometabolic risk factors, including BMI, DBP, DM, current smoker, Hs-CRP, uric acid, TG, LDL-C, TC, and HbA1c.However, AIP has negative association with age (r=−0.232, p<0.001) and HDL-C (r=−0.606, p<0.001).
Table 2. Association between AIP and cardiometabolic risk factors.
| Variables | Correlation coefficient (r) | P-value |
|---|---|---|
| Age | -0.232 | <0.001 |
| BMI | 0.224 | <0.001 |
| DBP | 0.090 | 0.001 |
| DM | 0.078 | 0.004 |
| Current smoker | 0.146 | <0.001 |
| Hs-CRP | 0.124 | <0.001 |
| Hemoglobin | 0.182 | <0.001 |
| Uric acid | 0.242 | <0.001 |
| Triglyceride | 0.933 | <0.001 |
| LDL-C | 0.093 | 0.001 |
| HDL-C | -0.606 | <0.001 |
| TC | 0.233 | <0.001 |
| HbA1c | 0.120 | <0.001 |
AIP, atherogenic index of plasma; BMI, body mass index; DBP, diastolic blood pressure; DM, diabetes mellitus; Hs-CRP, high sensitivity-C reactive protein; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TC, total cholesterol; HbA1c, glycosylated hemoglobin A1c.
The Association between DES-ISR and AIP
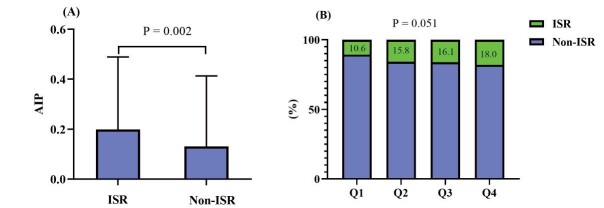
First of all, we compared the difference in AIP between the ISR and non-ISR groups. As presented in Fig.1A , serum AIP level was significantly higher in patients with ISR than those without (0.199±0.290 vs. 0.131±0.282, p=0.002). Subsequently, we stratified the patients recruited into 4 groups according to the quartile (Q) of AIP (Q1(AIP ≤ −0.042), Q2(−0.042< AIP ≤ 0.136), Q3(0.136< AIP ≤ 0.305), Q4(AIP >0.305)) and investigated the incidence of DES-ISR according to the quartiles of AIP. As presented in Fig.1B , the incidences of DES-ISR according to the quartile of baseline AIP were 10.6%, 15.8%, 16.1%, and 18.0%, respectively. And the trend was marginally significant (p=0.051).
Fig.1.
Comparison in the level of AIP between ISR and non-ISR group (A), and the incidence of ISR according to the quartiles (Q) of AIP (B). AIP, atherogenic index of plasma; ISR, in-stent restenosis.
Association of AIP with the Risk of DES-ISR in Logistic Regression Analysis
As a continuous variable, AIP was associated with the increased risk of DES-ISR (OR=2.284, 95% CI 1.355 to 3.849, p=0.002) in the univariate logistic regression analysis.
Multivariate logistic regression analysis models were established to avoid the interference of confounding risk factors ( Table 3 ) . As presented in Table 3 , AIP remained independently associated with the elevated risk of DES-ISR (OR=2.100, 95% CI 1.134 to 3.891, p=0.018) after fully adjusted the confounding risk factors with Model 4. And the optimal AIP cutoff point for DES-ISR was −0.037 (sensitivity: 81.9%, specificity:27.5%, area under curve: 0.557 (0.516 to 0.599), p=0.0072).
Table 3. Multiple logistic regression analysis Models for evaluating the association between AIP and the risk of DES-ISR.
| Models | OR | 95% CI | P-value |
|---|---|---|---|
| Model 1 | 2.498 | 1.449 to 4.309 | 0.001 |
| Model 2 | 1.958 | 1.079 to 3.555 | 0.027 |
| Model 3 | 2.064 | 1.117 to 3.815 | 0.021 |
| Model 4 | 2.100 | 1.134 to 3.891 | 0.018 |
Model 1: adjusted for age, sex, BMI;
Model 2: adjusted for confounders in Model 1 plus SBP, hypertension, previous PCI, Hs-CRP, platelet count, HbA1c, TC, and LVEF;
Model 3: adjusted for confounders in Model 2 plus SYNTAX score, target in multivessel, target in LAD, target in RCA, multiple stents (≥ 2), DES- sirolimus, minimal stent diameter ≤ 3mm, intracoronary imaging technique, and follow-up time;
Model 4: adjusted for confounders in Model 3 plus therapy with ACEI/ARB, insulin, and other hypoglycemic agents.
AIP, atherogenic index of plasma; DES, drug-eluting stent; ISR, in-stent restenosis; BMI, body mass index; SBP, systolic blood pressure; PCI, percutaneous coronary intervention; Hs-CRP, high sensitivity-C reactive protein; HbA1c, glycosylated hemoglobin A1c; TC, total cholesterol; LVEF, left ventricular ejection fraction; LAD, left anterior descending artery; RCA, right coronary artery; ACEI/ARB; angiotensin-converting enzyme inhibitor/angiotensin receptor blocker.
To further evaluate the association between AIP and DES-ISR risk in patients with ACS, we also modulated AIP as a categorical variable. In univariate analyses ( Fig.2A ) , the ORs (95% CI) for Q2, Q3, and Q4 were 1.582 (1.000, 2.503), 1.618 (1.024, 2.556), and 1.855(1.183, 2.908), respectively. After fully adjusting for traditional clinical risks with a multiple logistic regression analysis model, the incidence of DES-ISR increased significantly in Q4 compared to the reference Q1(OR=1.713, 95 % CI 1.040 to 2.822, p=0.034, Fig.2B ).
Fig.2.
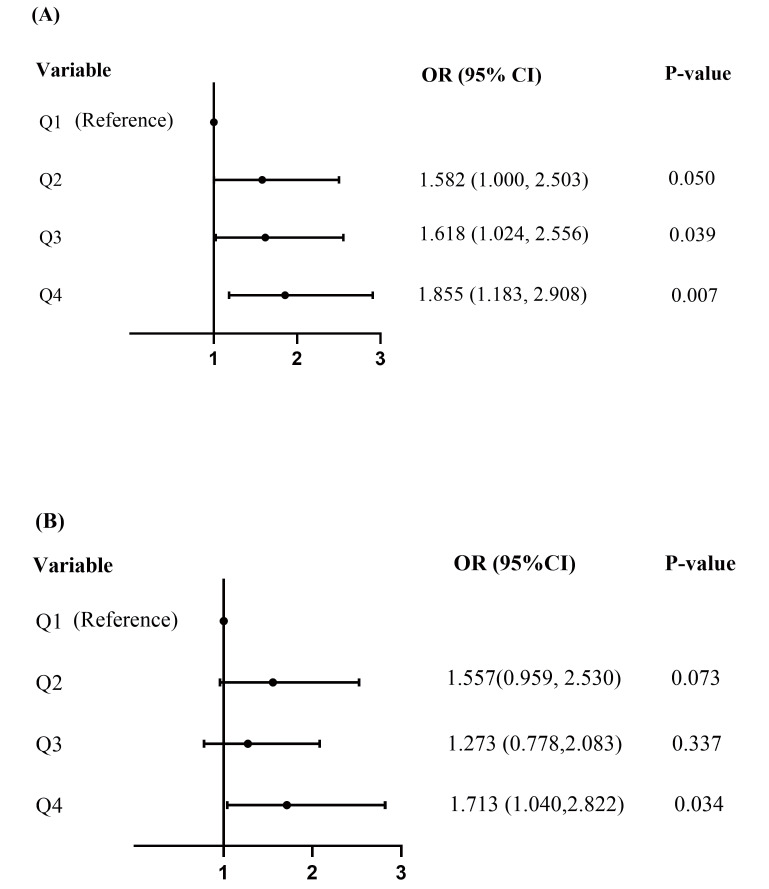
Forest plot of the risk of ISR according to quartiles (Q) of AIP in unadjusted Model (A), and the fully adjusted Model (B). ISR, in-stent restenosis; AIP, atherogenic index of plasma.
Association of AIP with the Risk of DES-ISR in Different Subgroups
The independent association of AIP with the incidence of DES-ISR was further assessed by us in the different subgroups of study population. As presented in Fig.3 , the association was still persisted in the subgroups of male (OR=2.382, 95% CI 1.192 to 4.761, p=0.014), age <65 years (OR=2.292, 95% CI 1.130 to 4.649, p=0.022), BMI<25 kg/m2 (OR=3.012, 95%CI 1.162 to 7.811, p=0.023), DM (OR=3.115, 95%CI 1.271 to 7.636, p=0.013), and LDL-C <1.8 mmol/L (OR=3.116, 95% CI 1.157 to 8.396, p=0.025) after fully adjusting the confounding variables. Furthermore, this trend was also observed by us in the subgroup of current smoker as well as non-current smoker (p<0.1).
Fig.3.
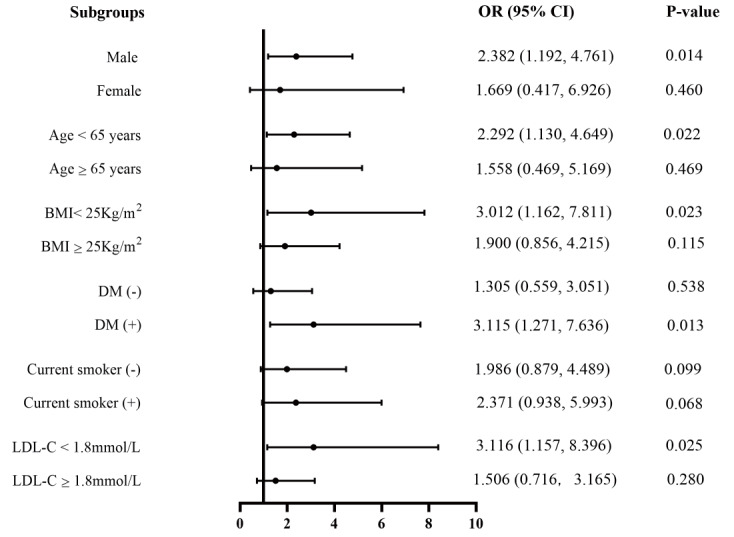
Forest plot for the subgroup analysis of the effect size of AIP on the incidence of ISR in our cohort study. AIP, atherogenic index of plasma; ISR, in-stent restenosis; BMI, body mass index; DM, diabetes mellitus; LDL-C, low-density lipoprotein cholesterol.
Discussion
In the present study, we evaluated the association between AIP and ISR incidence after successful DES-based PCI for the first time. And the major findings were listed as follows: (1) AIP, as a comprehensive lipid parameter, was associated with various cardiometabolic risk factors; (2) AIP, either as a continuous or categorical variable, was an independent predictor of DES-ISR in patients with ACS; (3) Furthermore, the independent predictive effect of AIP on the risk of DES-ISR was also reflected in the subgroups of male, age <65 years, BMI <25 kg/m2, DM, and LDL-C <1.8 mmol/L.
ASCVDs remains the leading cause of morbidity and mortality for patients worldwide, despite great advances in management strategies to lessen ASCVDs risk factors 1 , 2) . A major reason is atherogenic dyslipidemia, consisting of reduced serum HDL-C and increased TG-rich lipoproteins as well as sdLDL 5 , 6) . Notably, therapy with statins alone fails to correct it adequately, and a single lipid parameter commonly used in the clinical practice at present cannot fully reflect this specific dyslipidemia 23) . The triglyceride-based indices AIP, calculated with the formula log10 (TG/HDL-C) and could act as a surrogate marker of sdLDL, may serve as a more useful and comprehensive parameter to reflect plasma atherogenicity 7 - 9) . A higher AIP level was also associated with an increasingly atherogenic lipid phenotype and cholesterol esterification rete 8 , 9) . Moreover, our present study and prior studies suggested AIP was relevant to various cardiometabolic risk factors 10 - 12) .
Currently, the link between AIP and ASCVD has been well established in previous epidemiological studies as well. Zhou et al. Wang et al. and Shanker et al. reported AIP was an independent and powerful predictor for the presence of CAD, and adding it with family history to traditional risk factors models could improve predictive accuracy for CAD 10 , 14 , 24) . Meanwhile, recent evidence also confirmed that AIP was independently and strongly associated with the risk of CAD in special populations such as very young adults and postmenopausal women, who usually had the unique risk factor profile 25 , 26) . Furthermore, mounting evidence suggests AIP may serve as an independent predictor for arterial stiffness, coronary artery calcification progression, and the rapid progression of coronary plaque, all of which were powerful predictors of adverse cardiac events 15 , 27 , 28) . Most importantly, Qin et al. and Ma et al. demonstrated that increased AIP upon admission correlated positively with the worse long-term prognosis in patients after PCI. The effects were independent of LDL-C level 17 , 18) . Extending those prior studies, our present study indicated the AIP is strongly related to the prevalence of ISR beyond the traditional risk factors in patients with ACS after DES-based PCI. This result suggests optimizing the lipid management according to AIP may be a promising way to prevent ISR in the future. However, the predictive ability of AIP for DES-ISR was mild, with the cutoff point −0.037 in this study. Considering this study’s limitations, future studies are required to identify a more accurate reference value of AIP for clinical guidance.
According to previous studies, gender-specific associations existed between AIP and cardiovascular disorder 12 , 25) . Consistent with those studies, our subgroup analysis also confirmed the gender difference and revealed the incidence of ISR increased with AIP elevated in men while not in women. Unexpectedly, our subgroup analysis also revealed that AIP was more closely associated with the DES-ISR in patients LDL-C <1.8 mmol/L, which prior studies have not reported. Although the exact reasons underlying this phenomenon remain unclear, further strengthening lipid management based on AIP value in patients with LDL-C <1.8 mmol/l may substantially reduce the residual risk for DES-ISR.
Although the potential mechanisms underlying AIP and DES-ISR were unknown at present, several possible explanations were listed according to previous studies. First, this study and prior studies all suggested AIP correlates positively with various metabolic disorders such as insulin resistance, DM, hyperglycemia, and hyperuricemia 10 , 11) , all of which were well-established risk factors for ISR 29 - 31) . Second, remnant-lipoprotein cholesterol, as a risk factor for the occurrence and development of ISR 32 , 33) , is usually increased with increasing AIP levels 8 , 10) . Last but not least, AIP is a surrogate marker of sdLDL, which is one of the LDL subfractions with increased proinflammatory and proatherogenic action 9 , 34) . The clinical trial also reported an association between sdLDL and ISR incidence based on population 35) .
Limitations
First, this retrospective cohort study fails to control all of the potential confounding factors and cannot provide a definite causal relationship between AIP and DES-ISR. Second, the present study only enrolled subjects who took follow-up angiography within 6 to 18 months after PCI, which may cause select bias. Third, this study was mainly based on the Chinese population, which means our present findings may not generalize to other ethnic groups. Finally, our present study only assessed the association of baseline AIP with the risk of ISR, failing to evaluate the effect of consecutive change in AIP during follow-up on ISR incidence. Therefore, future prospective, multicenter studies are needed to determine the association between them.
Conclusions
Based on our cohort study’s findings, the increased AIP level was independently associated higher risk of ISR in patients with ACS during the intermediate-term follow-up. And this association was much stronger in those with LDL-C <1.8 mmol/L. These results suggest optimizing the lipid management based on AIP may be a promising way to prevent ISR. To further confirm our findings, multicenter, randomized, and prospective studies were necessary.
List of Abbreviation
ASCVDs, atherosclerotic cardiovascular diseases; HDL-C, high-density lipoprotein cholesterol; TG, triglyceride; sdLDL, small dense low-density lipoprotein; LDL-C, low-density lipoprotein cholesterol; AIP, atherogenic index of plasma; DM, diabetes mellitus; CAD, coronary artery disease; ACS, acute coronary syndrome; ISR, in-stent restenosis; PCI, percutaneous coronary intervention; DES, drug-eluting stent; TIMI, thrombolysis in myocardial infarction; CABG, coronary artery bypass grafting; PTCA, percutaneous transluminal coronary angioplasty; Hs-CRP, high sensitivity-C reactive protein; HbA1c, glycosylated hemoglobin A1c; eGFR, estimated glomerular filtration rate; BMI, body mass index; TC, total cholesterol; SBP, systolic blood pressure; LVEF, left ventricular ejection fraction; LAD, left anterior descending artery; RCA, right coronary artery; ACEI/ARB, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker; TRLs, TG-rich lipoproteins.
Declaration Ethics approval and Consent to Participate
The present study was approved by the Ethics Committee of Beijing AnZhen Hospital, Capital Medical University and was performed in line with Declaration of Helsinki. Meanwhile, all the patients included were informed and consent to participate.
Consent for Publication
Not applicable.
Availability of Data and Materials
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing Interests
The authors declare that they have no competing interests.
Funding
This work was supported by the grant from National Key Research and Development Program of China (2017YFC0908800), the “Beijing Municipal Administration of Hospitals” Ascent Plan (DFL20150601) and Mission plan (SML20180601), and Beijing Municipal Health Commission “Project of Science and Technology Innovation Center” (PXM2019_026272_000006) (PXM2019_026272_000005).
Authors’ Contribution
YZ was responsible for the data analysis and manuscript writing; MLC, KSL, AG, XYK, and YL contributed to the data collection; HYH and HL reviewed and revised this manuscript carefully.
Acknowledgements
Not applicable.
References
- 1).Roth GA, Forouzanfar MH, Moran AE, Barber R, Nguyen G, Feigin VL, Naghavi M, Mensah GA, and Murray CJL: Demographic and Epidemiologic Drivers of Global Cardiovascular Mortality. N Engl J Med, 2015; 372: 1333-1341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2).Timmis A, Townsend N, Gale C, Grobbee R, Maniadakis N, Flather M, Wilkins E, Wright L,Vos R, Bax J, Blum M, Pinto F, and Vardas P: European Society of Cardiology: Cardiovascular Disease Statistics 2017. Eur Heart J, 2018; 39: 508-579 [DOI] [PubMed] [Google Scholar]
- 3).Xiao C, Dash S, Morgantini C, Hegele RA, and Lewis GF: Pharmacological Targeting of the Atherogenic Dyslipidemia Complex: The Next Frontier in CVD Prevention Beyond Lowering LDL Cholesterol. Diabetes, 2016; 65: 1767-1778 [DOI] [PubMed] [Google Scholar]
- 4).Chapman MJ, Ginsberg HN, Amarenco P, Andreotti F, Borén J, Catapano AL, Descamps OS, Fisher E, Kovanen PT, Kuivenhoven JA, Lesnik P, Masana L, Nordestgaard BG, Ray KK, Reiner Z, Taskinen MR, Tokgözoglu L, Tybjærg-Hansen A, and Watts GF: Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: evidence and guidance for management. Eur Heart J, 2011; 32: 1345-1361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5).Castañer O, Pintó X, Subirana I, Amor AJ, Ros E, Hernáez Á, Martínez-González MÁ, Corella D, Salas-Salvadó J, Estruch R, Lapetra J, Gómez-Gracia E, Alonso-Gomez AM, Fiol M, Serra-Majem L, Corbella E, Benaiges D, Sorli JV, Ruiz-Canela M, Babió N, Sierra LT, Ortega E, and Fitó M: Remnant Cholesterol, Not LDL Cholesterol, Is Associated with Incident Cardiovascular Disease. J Am Coll Cardiol, 2020; 76: 2712-2724 [DOI] [PubMed] [Google Scholar]
- 6).Duran EK, Aday AW, Cook NR, Buring JE, Ridker PM, and Pradhan AD: Triglyceride-Rich Lipoprotein Cholesterol, Small Dense LDL Cholesterol, and Incident Cardiovascular Disease. J Am Coll Cardiol, 2020; 75: 2122-2135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7).Dobiášová M, Frohlich J, Šedová M, Cheung MC, and Brown BG: Cholesterol esterification and atherogenic index of plasma correlate with lipoprotein size and findings on coronary angiography. J Lipid Res, 2011; 52: 566-571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8).Quispe R, Manalac RJ, Faridi KF, Blaha MJ, Toth PP, Kulkarni KR, Nasir K, Virani SS, Banach M, Blumenthal RS, Martin SS, and Jones SR: Relationship of the triglyceride to high-density lipoprotein cholesterol (TG/HDL-C) ratio to the remainder of the lipid profile: The Very Large Database of Lipids-4 (VLDL-4) study. Atherosclerosis, 2015; 242: 243-250 [DOI] [PubMed] [Google Scholar]
- 9).Dobiasova M, and Frohlich J: The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem, 2001; 34: 583-588 [DOI] [PubMed] [Google Scholar]
- 10).Zhou K, Qin Z, Tian J, Cui K, Yan Y, and Lyu S: The Atherogenic Index of Plasma: A Powerful and Reliable Predictor for Coronary Artery Disease in Patients with Type 2 Diabetes. Angiology, 2021: doi: 10.1177/00033197211012129 [DOI] [PubMed] [Google Scholar]
- 11).Zhu X, Deng F, and Lei S: Meta-analysis of Atherogenic Index of Plasma and other lipid parameters in relation to risk of type 2 diabetes mellitus. Prim Care Diabetes, 2015; 9: 60-67 [DOI] [PubMed] [Google Scholar]
- 12).Zhang X, Zhang X, Li X, Feng J, and Chen X: Association of metabolic syndrome with atherogenic index of plasma in an urban Chinese population: A 15-year prospective study. Nutr Metab Cardiovasc Dis, 2019; 29: 1214-1219 [DOI] [PubMed] [Google Scholar]
- 13).Shen S, Lu Y, Li F, Yang C, Feng Y, Li H, Yao W, and Shen Z: Atherogenic index of plasma is an effective index for estimating abdominal obesity. Lipides Health Dis, 2018; 17: 11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14).Wang L, Chen F, Xiaoqi C, Yujun C, and Zijie L: Atherogenic Index of Plasma Is an Independent Risk Factor for Coronary Artery Disease and a Higher SYNTAX Score. Angiology, 2021; 72: 181-186 [DOI] [PubMed] [Google Scholar]
- 15).Won KB, Heo R, Park HB, Lee BK, Lin FY, Hadamitzky M, Kim YJ, Sung JM, Conte E, Andreini D, Pontone G, Budoff MJ, Gottlieb I, Chun EJ, Cademartiri F, Maffei E, Maffei E, de Araújo Gonçalves P, Leipsic JA, Lee SE, Shin S, Choi JH, Virmani R, Samady H, Chinnaiyan K, Berman DS, Narula J, Shaw LJ, Bax JJ, Min JK, and Chang HJ: Atherogenic index of plasma and the risk of rapid progression of coronary atherosclerosis beyond traditional risk factors. Atherosclerosis, 2021; 324: 46-51 [DOI] [PubMed] [Google Scholar]
- 16).Won KB, Jang MH, Park EJ, Park HB, Heo R, Hang D, and Chang HJ: Atherogenic index of plasma and the risk of advanced subclinical coronary artery disease beyond traditional risk factors: An observational cohort study. Clin Cardiol, 2020; 43: 1398-1404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17).Ma X, Sun Y, Cheng Y, Shen H, Gao F, Qi J, Yang L, Wang Z, Shi D, Liu Y, Liu XL, and Zhou Yj: Prognostic impact of the atherogenic index of plasma in type 2 diabetes mellitus patients with acute coronary syndrome undergoing percutaneous coronary intervention. Lipids Health Dis, 2020; 19: 240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18).Qin Z, Zhou K, Li Y, Cheng W, Wang Z, Wang J, Gao F, Yang L, Xu Y, Wu Y, He H, Zhou YJ: The atherogenic index of plasma plays an important role in predicting the prognosis of type 2 diabetic subjects undergoing percutaneous coronary intervention: results from an observational cohort study in China. Cardiovasc Diabetol, 2020; 19: 23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19).Giacoppo D, Alfonso F, Xu B, Claessen BEPM, Adriaenssens T, Jensen C, Pérez-Vizcayno MJ, Kang D, Degenhardt R, Pleva L, Baan J, Cuesta J, Park DW, Kukla P, Jiménez-Quevedo P, Unverdorben M, Gao R, Naber CK, Park SJ, Henriques JPS, Kastrati A, and Byrne RA: Drug-Coated Balloon Angioplasty Versus Drug-Eluting Stent Implantation in Patients with Coronary Stent Restenosis. J Am Coll Cardiol, 2020; 75: 2664-2678 [DOI] [PubMed] [Google Scholar]
- 20).Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, Byrne RA, Collet JP, Falk V, Head SJ, Jüni P, Kastrati A, Koller A, Kristensen SD, Niebauer J, Richter DJ, Seferovic PM, Sibbing D, Stefanini GG, Windecker S, Yadav R, and Zembala MO: 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J, 2019; 40: 87-165 [Google Scholar]
- 21).American Diabetes Association: 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2020. Diabetes Care, 2019; 43(Supplement 1): S14-S31 [DOI] [PubMed] [Google Scholar]
- 22).Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M, Clement DL, Coca A, Simone GD, Dominiczak A, Kahan T, Mahfoud F, Redon J, Ruilope L, Zanchetti A, Kerins M, Kjeldsen SE, Kreutz R, Laurent S, Lip GYH, McManus R, Narkiewicz K, Ruschitzka F, Schmieder RE, Shlyakhto E, Tsioufis C, Aboyans V, and Desormais I: 2018 ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. J Hypertens, 2018; 36: 1953-2041 [Google Scholar]
- 23).Taskinen M, and Borén J: New insights into the pathophysiology of dyslipidemia in type 2 diabetes. Atherosclerosis, 2015; 239: 483-495 [DOI] [PubMed] [Google Scholar]
- 24).Shanker J, and Kakkar VV: Contribution of classical and emerging risk factors to coronary artery disease in Asian Indians. Int J Cardiol, 2016; 214: 97-106 [DOI] [PubMed] [Google Scholar]
- 25).Cai G, Liu W, Lv S, Wang X, Guo Y, Yan Z, Du Y, and Zhou Y: Gender-specific associations between atherogenic index of plasma and the presence and severity of acute coronary syndrome in very young adults: a hospital-based observational study. Lipids Health Dis, 2019; 18: 99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26).Wu T, Gao Y, Zheng Y, Ma Y, and Xie X: Atherogenic index of plasma (AIP): a novel predictive indicator for the coronary artery disease in postmenopausal women. Lipids Health Dis, 2018; 17: 197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27).Yin J, Li M, Yu L, Hu F, Yu Y, Hu L, Bao H, and Cheng X: The relationship between the atherogenic index of plasma and arterial stiffness in essential hypertensive patients from China: a cross-sectional study. BMC Cardiovasc Disord, 2021; 21: 245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28).Won K, Han D, Lee JH, Choi S, Chun EJ, Park SH, Han H, Sung J, Jung HO, and Chang H: Atherogenic index of plasma and coronary artery calcification progression beyond traditional risk factors according to baseline coronary artery calcium score. Sci Rep, 2020; 10: 21324. doi: 10.1038/s41598-020-78350-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29).Yang CD, Shen Y, Lu L, Yang ZK, Hu J, Zhang RY, Shen WF, Ding FH, and Wang XQ: Visit-to-visit HbA1c variability is associated with in-stent restenosis in patients with type 2 diabetes after percutaneous coronary intervention. Cardiovasc Diabetol, 2020; 19: 133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30).Turak O, Canpolat U, Özcan F, Mendi MA, Öksüz F, İşleyen A, Gürel ÖM, Çay S, Aras D, and Aydoğdu S: Usefulness of Preprocedural Serum Uric Acid Level to Predict Restenosis of Bare Metal Stents. Am J Cardiol, 2014; 113: 197-202 [DOI] [PubMed] [Google Scholar]
- 31).Zhao L, Xu W, Wang L, Li H, Shao C, Gu H, Chan S, Xu H, and Yang X: Influence of insulin resistance on in-stent restenosis in patients undergoing coronary drug-eluting stent implantation after long-term angiographic follow-up. Coronary Artery Dis, 2015; 26: 5-10 [DOI] [PubMed] [Google Scholar]
- 32).Xu X, Pandit RU, Han L, Li Y, and Guo X: Remnant Lipoprotein Cholesterol Independently Associates with In-Stent Restenosis After Drug-Eluting Stenting for Coronary Artery Disease. Angiology, 2019; 70: 853-859 [DOI] [PubMed] [Google Scholar]
- 33).Qin Z, Zhou K, Li Y, Wang J, Cheng W, Hu C, Shi C, He H, and Zhou Y: Remnant lipoproteins play an important role of in-stent restenosis in type 2 diabetes undergoing percutaneous coronary intervention: a single-centre observational cohort study. Cardiovasc Diabetol, 2019; 18: 11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34).Jin J, Zhang H, Cao Y, Liu H, Hua Q, Li Y, Zhang Y, Wu N, Zhu C, Xu R, Gao Y, Li XL, Cui CJ, Liu G, Sun J, Dong Q, Guo YL, and Li JJ: Association of small dense low-density lipoprotein with cardiovascular outcome in patients with coronary artery disease and diabetes: a prospective, observational cohort study. Cardiovasc Diabetol, 2020; 19: 45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35).Kim J, Kim M, Lee BK, Rim S, Min P, Yoon S, Kim J, Rhee J, Yoon YW, Hong BK, Kwon HM, and Kim C: Effects of Increasing Particle Size of Low-Density Lipoprotein on Restenosis After Coronary Stent Implantation. Circ J, 2008; 72: 1059-1064 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.