Abstract
Background and objectives
Although previous reports have shown intraocular pressure changes during robotic-assisted laparoscopic prostatectomy, they did not discuss the time course of changes or the timing of the largest change. We conducted this study to quantify pressure changes over time in patients assuming the steep Trendelenburg position during robotic-assisted laparoscopic prostatectomy.
Methods
Twenty-one men were enrolled. Intraocular pressure was measured before anesthesia induction in the supine position (T0); 30 (T1), 90 (T2), and 150 minutes after assuming the Trendelenburg position (T3); and 30 minutes after reassuming the supine position (T4). End-tidal carbon dioxide and blood pressure were also recorded. To compare intraocular pressure between the time points, we performed repeated-measures analysis of variance. A mixed-effects multivariate regression analysis was conducted to adjust for confounding factors.
Results
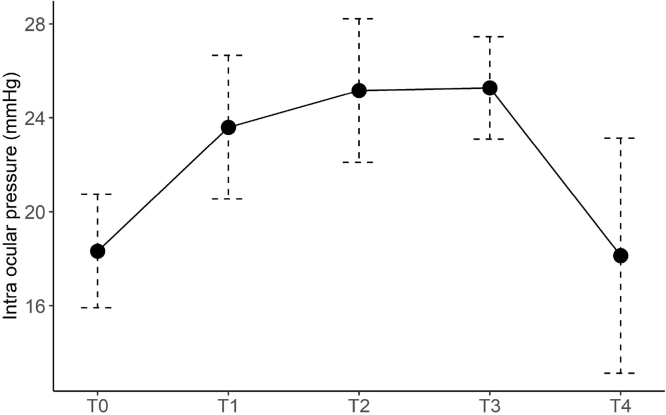
The mean (standard deviation) intraocular pressure was 18.3 (2.4), 23.6 (3.0), 25.1 (3.1), 25.3 (2.2), and 18.1 (5.0) mmHg at T0, T1, T2, T3, and T4, respectively. The mean intraocular pressure was higher at T1, T2, and T3 than at T0 (p < 0.0001 for all). There was no significant difference between T0 and T4, and between T3 and T2 (p > 0.99 for both).
Conclusions
The Trendelenburg position during robotic-assisted laparoscopic prostatectomy increased intraocular pressure. The increase was moderate at 90 minutes after the position was assumed, with the value being approximately 7 mmHg higher than the baseline value. The baseline intraocular pressure was restored at 30 minutes after the supine position was reassumed.
Trial registration
UMIN ID 000014973
Date of registration
August 27, 2014
Keywords: Robotic surgical procedures, Prostatectomy, Intraocular pressure, Trendelenburg position
Introduction
Robotic-assisted laparoscopic prostatectomy (RALP) is one of the newest and most technically advanced surgical procedures. The technique is increasingly performed worldwide due to the advantages in decreasing postoperative pain and blood loss.1 The steep Trendelenburg position required for RALP may cause high intraocular pressure (IOP), which leads to low ocular perfusion pressure. It may cause central retinal artery occlusion, and several ocular complications have been reported.2
Some studies have reported that the Trendelenburg position increases IOP.3, 4, 5 These reports showed IOP elevated during RALP but did not discuss the time course of IOP change or the timing of the largest IOP change. Awad et al reported that IOP was higher at the end of the duration of the steep Trendelenburg position than in the supine position. Although they measured IOP at seven time points, they did not measure it during the surgery (i.e., between the start and the end of the Trendelenburg position).3 Matsuyama et al measured IOP between the start and the end of the Trendelenburg position; specifically, measurements were made at 15, 60, and 90 minutes after the position was assumed. They showed that IOP was higher in the Trendelenburg position than in the supine position and increased from 15 minutes to 90 minutes in the Trendelenburg position. However, they did not measure IOP after 90 minutes.4 Hoshikawa et al. measured IOP every hour after the Trendelenburg position was assumed and compared the value at the start of the position with those after the position was assumed. They found that IOP was higher at each time point during the Trendelenburg position than at the start,5 although the time at which the increase was the largest was not determined. Taken together, although these reports showed changes in IOP during RALP, they did not discuss the time course of IOP changes or the timing of the largest IOP change.
We hypothesized that IOP does not constantly increase throughout the Trendelenburg position and aimed to determine the pattern of IOP elevation during surgery. Accordingly, we quantified the IOP changes over time in patients assuming the steep Trendelenburg position during RALP.
Methods
This was a single-center, prospective, observational study. The study protocol was registered at the University Hospital Medical Information Network (Registration No.: UMIN ID 000014973) before patient recruitment. After approval by the Research Ethics Committee of our institution (Approval No.: 26-33), written informed consent was obtained. Inclusion criteria were as follows: patients scheduled to undergo RALP, age >18 years), and an American Society Anesthesiologists (ASA) physical status of I–II. Recruitment was performed at our institution between August 2014 and March 2015. The exclusion criterion was the presence of glaucoma. All patients consulted an ophthalmologist before surgery. We did not exclude patients with any other ophthalmic disease.
General anesthesia was induced with propofol, rocuronium, and fentanyl and maintained with sevoflurane and remifentanil. All patients received tracheal intubation, and their radial artery was catheterized for continuous monitoring of arterial blood pressure. Mechanical ventilation was provided at the discretion of each anesthesiologist. Trendelenburg position was standardized at 30° from horizontal.
The primary endpoint of this study was the IOP during RALP. Secondary endpoints included any ocular complications such as blindness, narrowing of the visual field, or impaired visual perception, which were assessed at discharge and at 1 and 3 months after surgery.
Measurement and instrumentation
IOP measurements were performed with the rebound tonometer Icare PRO® (M.E. Technica, Tokyo, Japan). The IOP was measured at the following five time points. T0: before induction of anesthesia in the supine position, T1: 30 minutes after assuming the Trendelenburg position, T2: 90 minutes after assuming the Trendelenburg position, T3: 150 minutes after assuming the Trendelenburg position, and T4: 30 minutes after reassuming the supine position. End-tidal carbon dioxide and blood pressure were also recorded at each time point.
Sample size calculation
Assuming a 5-mmHg difference in IOP with a standard deviation of 5 mmHg,4, 5 sample size estimation analysis suggested that 18 patients were required. The power was set at 80%. To adjust for the ten time-point comparisons (i.e., every pair at the time points T0, T1, T2, T3, and T4), the type I error was set at 0.005 (0.05/10). We recruited 21 patients considering a 10% dropout rate.
Statistical analysis
The normality of the data was visually assessed using a normal Q-Q plot and tested using the Kolmogorov–Smirnov test. Data normality was not rejected in the normal Q-Q plot and the Kolmogorov–Smirnov test. All continuous values are shown as mean (standard deviation). To compare the IOP at each time point (longitudinal data), repeated-measures analysis of variance with Student’s t-test and Holm’s correction was performed for multiple comparison adjustments. We conducted Mauchly’s Test of Sphericity and adopted the Huynh-Feldt correction if sphericity was not confirmed. In addition, we used a mixed-effects multivariate regression model to adjust for confounding factors. In the mixed-effects model, the individual patient was set as a random effect, while the systolic blood pressure, end-tidal carbon dioxide, and time points were set as fixed effects. We did not include the IOP measured at T0 for the mixed-effects analysis because end-tidal carbon dioxide data were unavailable at this time point. P-values < 0.05 were considered statistically significant. Statistical analyses were performed using the R statistical software package, version 3.3.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Twenty-one male patients were included in the analyses. The demographics of the study participants are summarized in Table 1. As shown in Figure 1, the mean (standard deviation) IOP was 18.3 (2.4) mmHg before induction of anesthesia, 23.6 (3.0) mmHg at T1, 25.1 (3.1) mmHg at T2, 25.3 (2.2) mmHg at T3, and 18.1 (5.0) mmHg at T4. We have reasonably strong evidence that the mean IOP was higher at T1, T2 and T3 than at T0 (p < 0.0001 for all). There was no significant difference in IOP between T0 and T4 (p > 0.99). The IOP was higher at T3 than at T1 (p = 0.045), but there was no significant difference in IOP between T1 and T2 (p = 0.05) and between T2 and T3 (p > 0.99). The IOP was higher than 30 mmHg in three patients (3/21 or 14.3%), but lower than 35 mmHg in all patients during surgery. The mixed-effects multivariate regression analysis indicated that the results did not change when adjusted for the influence of the systolic blood pressure and end-tidal carbon dioxide (Table 2). No ocular complications such as blindness, narrowing of the visual field, or impaired visual perception were reported in the first three months postoperatively.
Table 1.
Patient demographics and operative variables.
| Age (years) | 65.5 ± 6.7 |
| Height (cm) | 168.5 ± 5.14 |
| Weight (kg) | 68.9 ± 7.6 |
| BMI (kg.m-2) | 24.3 ± 2.6 |
| Intravenous fluid (mL) | 2230 ± 460 |
| Duration of operation (min) | 263 ± 61 |
| Duration of pneumoperitoneum (min) | 225 ± 62 |
BMI, body mass index.
All variables are presented as mean ± standard deviation.
Figure 1.
Intraocular pressure (IOP) at each time point. Line graph showing the mean and standard deviation of the IOP. IOP is higher at T3 than at T0 and T1. There is no significant difference in IOP between T2 and T3. IOP is lower at T4 than at T3.
Table 2.
IOP after adjustment of SBP and EtCO2.
| MD | 95% CI | p-value | |
|---|---|---|---|
| Time (T1 vs T3) | -2.1 | -4 to -0.29 | 0.027 |
| Time (T2 vs T3) | -0.32 | -2.09 to 1.45 | 0.72 |
| Time (T4 vs T3) | -7.7 | -9.66 to -5.75 | < 0.001 |
| EtCO2 | -0.15 | -0.33 to 0.03 | 0.098 |
| SBP | -0.006 | -0.06 to 0.05 | 0.82 |
T1, 30 minutes after Trendelenburg position; T2, 90 minutes after Trendelenburg position; T3, 150 minutes after Trendelenburg position; T4, 30 minutes after assuming supine position; MD, mean difference; CI, confidence interval; EtCO2, end-tidal CO2; SBP, systolic blood pressure.
Discussion
The study findings showed that IOP increased approximately by 5 mmHg 30 minutes after assuming the Trendelenburg position. This IOP increase leveled off 90 minutes after the Trendelenburg position was assumed, at a value of approximately 7 mmHg higher than that at baseline. The IOP at 30 minutes after reassuming the supine position was at the same level as before anesthesia induction.
Previous studies have showed that IOP increases time-dependently after Trendelenburg positioning.4, 5 The present study revealed a similar result when comparing T0 with other time points, but there was no significant difference in IOP between 90 minutes and 150 minutes after assuming the Trendelenburg position. Although IOP was higher at 150 minutes than at 30 minutes after Trendelenburg positioning, the IOP difference between the two timepoints was small. These findings suggest that the magnitude of the IOP increase is the largest in the first 30 minutes and plateaus after 90 minutes. To avoid ocular complications, it may be effective to pay attention to the IOP increase, especially up to 90 minutes after the Trendelenburg position.
We showed that IOP at 30 minutes after reassuming the supine position returned to the same level as that before anesthesia induction. This finding suggests that when the IOP is too high, a return to the supine position may be effective in avoiding further increases in IOP and reducing ocular complications. A previous study showed that the administration of continuous infusion of dexmedetomidine to a patient undergoing laparoscopic surgery in the Trendelenburg position is a valuable method for attenuating the increase in IOP.6 Another report showed that continuous deep neuromuscular blockade may improve surgical conditions and facilitate RALP at a low intra-abdominal pressure, resulting in significant attenuation of IOP increase.7 However, these studies were designed to administer drugs from the beginning, and it is unknown whether the same effect can be obtained if the drugs were used after elevation in IOP. Additional research on improving IOP during surgery is required. In the other study, dorzolamide hydrochloride-timolol maleate drops significantly reduce elevated IOP of patients who undergo laparoscopic, robotic surgery in the Trendelenburg position.8 The medication reduces IOP by decreasing the production of aqueous humor by inhibition of carbonic anhydrase II in the ciliary processes and by direct action on β2 adrenergic receptors in the ciliary processes.9 It may be useful in the case of high intraoperative IOP, but it cannot be used for patients with bronchial asthma and chronic obstructive pulmonary disease due to beta-blocking action.
IOP is expressed as a division of aqueous humor production by the facility of outflow plus episcleral vein pressure.10, 11 The most influential factor contributing to IOP is the capacity for outflow from the vein. The increase in episcleral vein pressure does not increase IOP as much as it reduces the outflow capacity. In patients with glaucoma, the capacity for outflow is reduced, and in some patients, it approaches zero. In these cases, the IOP increases to a high degree.10 However, in cases with normal vein outflow, it is considered rare for IOP to exceed 35 mmHg even if the upper scleral venous pressure increases to 20 mmHg. In the present study, patients with glaucoma were excluded, and it was assumed that patients had a normal facility for outflow; none of the patients showed an IOP of 35 mmHg or higher.
We believe that the main cause of IOP increase during RALP is the increase in central venous pressure that is related to episcleral vein pressure3 and reduction in venous drainage.12 The increase in airway pressure caused by the Trendelenburg position, positive end expiratory pressure, and pneumoperitoneum pressure leads to an increase in central venous pressure in theory, but this increase is expected to be limited. A previous study showed that central venous pressure increased in the first 5 minutes after assuming Trendelenburg position, and in the next hour it decreased modestly.13
The present study has several limitations. First, there was a relatively high IOP recorded at baseline (18.3 mmHg). The mean (standard deviation) intraocular pressure in the Japanese population is reported to be 14.5 (2.5) mmHg.14 The exact reason for the relatively high IOP was unclear, but it may be due to the nature of the tonometer used (i.e., Icare PRO®). Previous research showed that the IOP measured was higher when using the Icare PRO® tonometer than when using the Goldmann applanation tonometer, although there was a strong correlation between the IOP measured by the two tonometers.15 Second, there is no evidence of a direct causal relationship between high IOP and ocular complications. It is unknown how much IOP influences the optic nerve and how long a patient can withstand the Trendelenburg position. Additional research on this subject is therefore required. Third, several studies reporting IOP changes during RALP have been published, and the novelty of our findings may be limited. Nevertheless, we believe that our findings are useful for confirming the reproducibility of previous findings in other populations. Future studies should now focus on measures to prevent or mitigate the IOP increase during RALP.
Conclusions
The Trendelenburg position during RALP leads to high IOP. The IOP increased after assuming the Trendelenburg position, and the magnitude of IOP increase became moderate 90 minutes after assuming the Trendelenburg position. The IOP at 30 minutes after reassuming the supine position was at the same level as that before the induction of anesthesia.
Conflict of interests
The authors declare no conflicts of interest.
Acknowledgments
We thank Editage (www.editage.jp) for English language editing.
References
- 1.Rassweilera Jens, Hruzaa Marcel, Tebera Dogu, et al. Laparoscopic and robotic assisted radical prostatectomy - critical analysis of the results. Eur Urol. 2006;49:612–614. doi: 10.1016/j.eururo.2005.12.054. [DOI] [PubMed] [Google Scholar]
- 2.Weber E.D., Colyer M.H., Lesser R.L., et al. Posterior ischemic optic neuropathy after minimally invasive prostatectomy. J Neuroophthalmol. 2007;27:285–287. doi: 10.1097/WNO.0b013e31815b9f67. [DOI] [PubMed] [Google Scholar]
- 3.Awad H., Santilli S., Ohr M., et al. The effects of steep Trendelenburg positioning on intraocular pressure during robotic radical prostatectomy. Anesth Analg. 2009;109:473–478. doi: 10.1213/ane.0b013e3181a9098f. [DOI] [PubMed] [Google Scholar]
- 4.Matsuyama K., Fujinaka W., Takatori M. Intra-ocular pressure during robotic-assisted laparoscopic radical prostectomy (in Japanese with English abstract) Jpn J Anesthesiol. 2014;63:1366–1368. [PubMed] [Google Scholar]
- 5.Hoshikawa Y., Tsutsumi N., Ohkoshi K., et al. The effect of steep Trendelenburg positioning on intraocular pressure and visual function during robotic-assisted radical prostatectomy. Br J Ophthalmol. 2014;98:305–308. doi: 10.1136/bjophthalmol-2013-303536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Joo J., Koh H., Lee K., et al. Effects of systemic administration of dexmedetomidine on intraocular pressure and ocular perfusion pressure during laparoscopic surgery in a steep Trendelenburg position: prospective, randomized, double-blinded study. J Kor Med Sci. 2016;31:989–996. doi: 10.3346/jkms.2016.31.6.989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yoo Y.-C., Kim N.Y., Shin S., et al. The intraocular pressure under deep versus moderate neuromuscular blockade during low-pressure robot assisted laparoscopic radical prostatectomy in a randomized trial. PLoS One. 2015;10 doi: 10.1371/journal.pone.0135412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Molloy B., Cong X. Perioperative dorzolamide-timolol intervention for rising intraocular pressure during steep Trendelenburg positioned surgery. AANA J. 2014;82:203–211. [PubMed] [Google Scholar]
- 9.Yeh J., Kravitz D., Francis B. Rational use of the fixed combination of dorzolamide – timolol in the management of raised intraocular pressure and glaucoma. Clin Ophthalmol. 2008;2:389–399. doi: 10.2147/opth.s1813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Suzuki R. Intraocular smooth muscle physiology relating to pathogenesis and therapy of glaucoma. J Smooth Muscle Res. 1996;32:175–194. doi: 10.1540/jsmr.32.175. [DOI] [PubMed] [Google Scholar]
- 11.Goel M., Picciani R.G., Lee R.K., et al. Aqueous humor dynamics: a review. Open Ophthalmol J. 2010;4:52–59. doi: 10.2174/1874364101004010052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kelly D.J., Farrell S.M. Physiology and role of intraocular pressure in contemporary anesthesia. Anesth Analg. 2018;126:1551–1562. doi: 10.1213/ANE.0000000000002544. [DOI] [PubMed] [Google Scholar]
- 13.Kalmar A.F., Foubert L., Hendrickx J.F.A., et al. Influence of steep Trendelenburg position and CO(2) pneumoperitoneum on cardiovascular, cerebrovascular, and respiratory homeostasis during robotic prostatectomy. Br J Anaesth. 2010;104:433–439. doi: 10.1093/bja/aeq018. [DOI] [PubMed] [Google Scholar]
- 14.Iwase A., Suzuki Y., Araie M., et al. The prevalence of primary open-angle glaucoma in Japanese: the Tajimi Study. Ophthalmology. 2004;111:1641–1648. doi: 10.1016/j.ophtha.2004.03.029. [DOI] [PubMed] [Google Scholar]
- 15.Tsumura T. Clinical evaluation of the new rebound tonometer Icare PRO in the sitting and supine positions. Atarashiiganka. 2015;32:1022–1026. [Google Scholar]