Abstract
Metal complexes have been widely used for applications in the chemical and physical sciences due to their unique electronic and stereochemical properties. For decades the use of metal complexes for medicinal applications has been postulated and demonstrated. The distinct characteristics of metal complexes, including their molecular geometries (that are not readily accessed by organic molecules), as well as their ligand exchange, redox, catalytic, and photophysical reactions, give these compounds the potential to interact and react with biomolecules in unique ways and by distinct mechanisms of action. Herein, the potential of metal complexes to act as components bioactive therapeutic compounds is discussed.
Keywords: Bioinorganic Chemistry, Enzyme Inhibitors, Chemical Biology, Coordination Compounds, Metals in Medicine
Metal complexes - attractive alternatives
Modern medicinal chemistry and chemical biology are based on the identification of compounds with well-defined biological effects. While typically the realm of organic molecules, metal complexes can offer distinctive properties and can employ alternative modes of action. While this might be surprising for some readers, metal ions and their coordination complexes have been used since ancient times for medical applications. Although mixtures of compounds have been employed for centuries, the use of the arsenic-containing organometallic complex Arsphenamine, also referred to as Salvarsan, or Compound 606, was the earliest example of a structurally defined metal complex to be isolated and biologically evaluated near the beginning of the 20th century. Upon testing of hundreds of arsenic-containing complexes, and developing a rudimentary structure-activity relationship (see Glossary), Ehrlich and co-workers began their search for the “magic bullet”, a compound with anti-microbial activity that did not exhibit toxicity in humans [1, 2]. Capitalizing on these efforts, they identified the aforementioned Arsphenamine, which was utilized as an effective treatment against syphilis until the discovery of penicillin. While the authors isolated a single compound, the correct structure of the active compound was not determined for nearly another century [3]. Decades later, the serendipitous discovery of cis-diamminodichloroplatinum(II) (commonly referred to as cisplatin) by Rosenberg and co-workers in the late 1960s, presented a milestone in the development of metal complexes for medicinal applications [4, 5]. Today, the platinum-based drugs cisplatin, carboplatin, and oxaliplatin are widely used and are mong the most prominent examples of the use of metals in medicine. A recent study suggested that in one hospital setting, approximately 50% of chemotherapeutic treatments involved a platinum anticancer drug [6, 7].
Since these early discoveries, various kinds of metal-containing compounds have been investigated for their applications in therapy and diagnostics. While a number of these compounds have proven to be effective, metal complexes are generally still, often erroneously, associated with high toxicity and poor pharmacological properties, which impedes their development in modern medicine. The biophysical properties and activities of metal-containing compounds is dependent on many factors, including the choice and oxidation state of the metal, coordination geometry, and the type of coordinated ligands. As has been well established during the last century, elemental mercury and many of its complexes are highly toxic and must be handled with caution. Conversely, this metal found application in dental amalgams, as a preservative for vaccines (the organometallic compound Thiomersal/Merthiolate), and as a topical antiseptic agent with the compound Merbromin/Mercurochrome/Asceptichrome [8].
Interest in the application of metal complexes for diagnostic and therapeutic uses has remained, despite these clinical reservations (and often misperceptions). To date, metal complexes based on the elements Li, Mg, Al, K, Ca, Fe, Co, Ga, As, Sr, Y, Zr, Pd, Ag, Sb, Sm, Lu, Pt, Au, Hg, Bi, and Ra have already been clinically approved in the US and/or the EU for a wide range of medicinal uses [9, 10]. In these compounds, the metal typically exerts a function which cannot be readily achieved by an organic molecule. In this short review, the properties and potential advantages of metal complexes (relative to organic molecules) for their application as therapeutic agents is discussed.
Structural scaffolds beyond organic molecules
Organic molecules are typically constructed from linear, planar, or tetrahedral building blocks, primarily driven by the hybridization, valency, and geometry of carbon atoms (Figure 1a). Metal centers offer the opportunity to utilize building blocks with increased valency and more varied geometries (Figure 1b). For example, an octahedral metal center with six different substituents is capable of forming 30 stereoisomers, compared to the two possible isomers formed by chiral carbon centers [11]. It has been demonstrated that binding selectivity towards biomolecules correlates with shape and stereochemical complexity [12] and the simple, yet well-defined, rigid shapes that can be found in some metal complexes may possess the complexity necessary to achieve high protein-binding specificity [13]. A number of different metal-based enzyme inhibitors can been developed using square planar, trigonal bipyramidal, square pyramidal, octahedral, sandwich, and half sandwich geometries (Figure 1b) [14, 15]. These coordination compounds can serve as useful scaffolds in the creation of unique shapes and geometries that can selectively engage enzyme targets.
Figure 1.
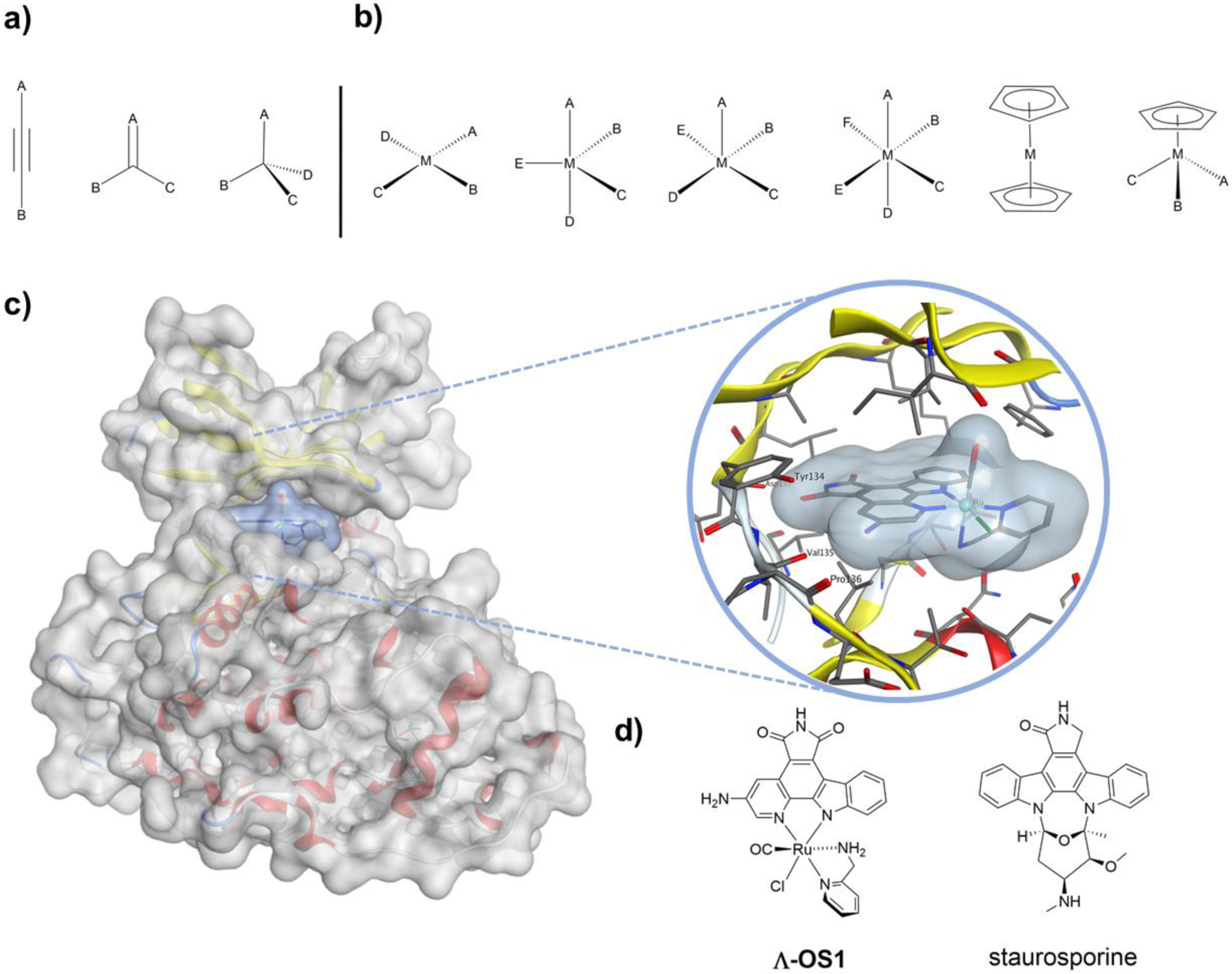
Geometries of carbon and metal containing complexes. a) Linear, planar, and tetrahedral geometry of carbon centers; b) Square planar, trigonal bipyramidal, square pyramidal, octahedral, sandwich and half sandwich geometries of metal complexes; c) X-ray crystal structure of Λ-OS1 bound to GSK3β. Λ-OS1 shows a highly complementary molecular surface that is able to form a novel interaction with the glycine rich loop via an induced fit binding mode; d) Structures of Λ-OS1 and staurosporine. PDB accession codes 3PUP and 1Q3D for protein-bound Λ-OS1 and staurosporine, respectively [19].
In a series of seminal studies, Meggers and coworkers demonstrated how organometallic complexes are capable of acting as highly selective protein kinase inhibitors [16–18]. Considering the vast complexity and diversity of biomolecules, it was hypothesized that the activity of organic molecules could be augmented by metal coordination to increase structural diversity and hence enhance protein binding affinity and selectivity. In an effort to show the utility of coordination compounds, several tailored octahedral metal complexes were prepared as specific inhibitors of select protein kinases [19]. These Ru(II) and Ir(III) pyridocarbazole metal complexes were derived from the natural product staurosporine, which is a relatively nonspecific protein kinase inhibitor [20, 21]. The resulting metal complexes, termed octasporines (OS), contained an organic component related to staurosporine that could also function as a bidentate ligand to a metal center. The staurosporine derivatives were bound to different metal complexes, which allowed for the introduction of new ligands and molecular geometries/shapes. These inert metal complexes presented well-defined molecular surfaces that aided in the identification of new interactions that could contribute to binding strength and selectivity toward kinases. Some compounds were used as enantiomeric mixtures, while others were isolated as single enantiomers. For example compound (Λ-OS1, Figure 1d), an octahedral Ru(II) complex, showed highly potent and selective inhibition of the α-isoform of glycogen synthase kinase 3 (GSK3α), with a half maximal inhibitory concentration (IC50) value of 0.9 nM, compared to staurosporine, which had an IC50 value of 50 nM [16] (it should be noted that the IC50 values are strongly dependent on assay conditions) [22]. Additionally, Λ-OS1 demonstrated 15- to >111,000-fold higher selectivity when evaluated against five other protein kinases. Interestingly, Λ-OS1 also showed ~7-fold selectivity for the α-isoform over the β-isoform of GSK3, though their catalytic domains share 98% homology [23] and the residues responsible for binding Λ-OS1 are identical. The authors suggest that it could be possible that the rigidity of the octahedral complex allows for Λ-OS1 to select between isoforms due to dynamics caused by sequence identity differences in distant, nonbinding residues. Additional selectivity was demonstrated by screening Λ-OS1 against a suite of 102 protein kinases at equal or lesser concentration, of which only four showed >25% reduction in activity (including both GSK3 isoforms). As shown in Figure 1c, Λ-OS1 demonstrates a highly globular and complimentary molecular surface to the active site of the β-isoform of glycogen synthase kinase 3 (GSK3β). Λ-OS1 occupies the ATP-binding site, similar to staurosporine (Figure 1d) and other organic inhibitors [24, 25]. In addition to a range of well-defined hydrogen bonds, hydrophobic interactions, and edge-to-face stacking, the CO ligand of the metal complex interacts with a flexible glycine-rich loop caused by a conformational change, which is not possible with the natural product staurosporine. This example highlights how metal complexes with diverse geometries can occupy regions of chemical and topological space that cannot be readily accessed by conventional organic compounds [15, 26], and demonstrates the potential of octahedral metal complexes to act as enzyme inhibitors with highly complementary binding surfaces.
Three-dimensionality
Biomolecular recognition is often driven by protein-ligand interactions. Accordingly, biological macromolecules will bind to molecules (including their native binding partners) with complementary three-dimensional (3D) binding surfaces [27]. Thus, many research efforts have been devoted towards molecules with high structural and topological diversity [27, 28]. It has been demonstrated that increasing the ‘3-dimensionality’ of molecules can lead to an increased likelihood of clinical success [29]. For example, 3D molecules demonstrate higher solubility due to increased solvation and diminished solid state crystal lattice packing [30]. Additionally, crucial properties of preclinical compounds such as absorption, distribution, metabolism, excretion, and toxicity (ADMET) have been shown to be influenced by molecular topology [31]. Other 3D measurements, such as the fraction of sp3 hybridized carbons have also been studied as methods of improving drug-like properties (Box 1) [32, 33]. Current drug discovery efforts typically rely on high-throughput screening (HTS) or fragment-based drug discovery (FBDD) to identify bioactive compounds [34]. Despite the advantages of highly 3D molecules, modern drug discovery relies heavily on two-dimensional (2D), and planar structures for both fragment and lead-like molecules [30, 32, 35]. Although there have been many recent efforts to design libraries that are composed of highly 3D molecules for drug discovery purposes [28, 33, 36–38], most drug-like molecules trend toward planarity [32].
Box 1. Drug-likeness in modern drug discovery.
There are many guidelines that practitioners of modern drug discovery employ to lower drug candidate attrition. One of the most common guidelines is Lipinski’s ‘rule of five’ (Ro5) [91]. The Ro5 suggests that molecules containing more than 5 H-bond donors, 10 H-bond acceptors, a molecular weight of more than 500 Da, and a calculated Log P (cLogP) greater than 5 will have poor oral availability. The Ro5 has often been used as a broader proxy for drug-likeness. An analogous ‘rule of three’ (Ro3) has been suggested for fragment-based drug discovery [92], which states that fragment molecules should have a molecular weight of less than 300 Da, no more than 3 H-bond donors, no more than 3 H-bond acceptors, and a cLogP of less than 3. Adherence to the Ro3 should, in principle, allow for the identification of fragment molecules that can be grown into suitable drug-like molecules that still obey the Ro5. Readers may have noticed that some of the metal complexes presented here as viable drug-like molecules do not strictly adhere to the Ro5. Though most obey the criteria of H-bond donors and acceptors and cLogP, the higher atomic weights of metal ions will sometimes result in molecular weights of greater than 500 Da. However, it has been demonstrated that though transition metal complexes often have much higher weights, they are not necessarily proportionally larger, and that the molecular weight parameter of these rules can be redefined as a molecular volume [36].
Increasing attention has been devoted towards the application of metal complexes that can act as 3D scaffolds for inhibitors. No longer bound by the linear, planar, or tetrahedral geometries imposed by carbon centers, metal complexes present a wide variety of geometries and topologies that can be easily accessed (Figure 1b). Indeed, in the case of stable octahedral complexes, the metal can be considered as a ‘hypervalent carbon center’, providing access to regions of chemical space not readily attainable by small organic molecules [39]. Recent work has highlighted the ability of metal complexes to be used in FBDD to occupy highly 3D space [36]. Figure 2a demonstrates the topological diversity of a collection of sandwich, half sandwich, and octahedral metal complexes when compared with all Food and Drug Administration (FDA) approved drugs (Figure 2b) on a normalized principle moment of inertia (PMI) plot [36, 40]. This chart demonstrates the three-dimensionality of a molecule by graphically comparing it to molecular standards that are intrinsically one, two, and three-dimensional (octa-2,4,6-triyne, benzene, and adamantane, respectively). For each molecule, the resistance to angular acceleration is calculated along each primary axis (I1, I2, and I3). The ratios (I1/I3 and I2/I3) are then plotted, demonstrating a normalized representation of relative chemical space. FDA approved drugs cluster along the linear/planar region of the chart while metal complexes reach more deeply into 3D space. In addition to populating 3D topologies, providing a more uniform sampling of overall chemical space, and being synthetically accessible, these metal complexes have been shown to have some modest inhibitory activity against viral, bacterial, and cancer targets.
Figure 2.
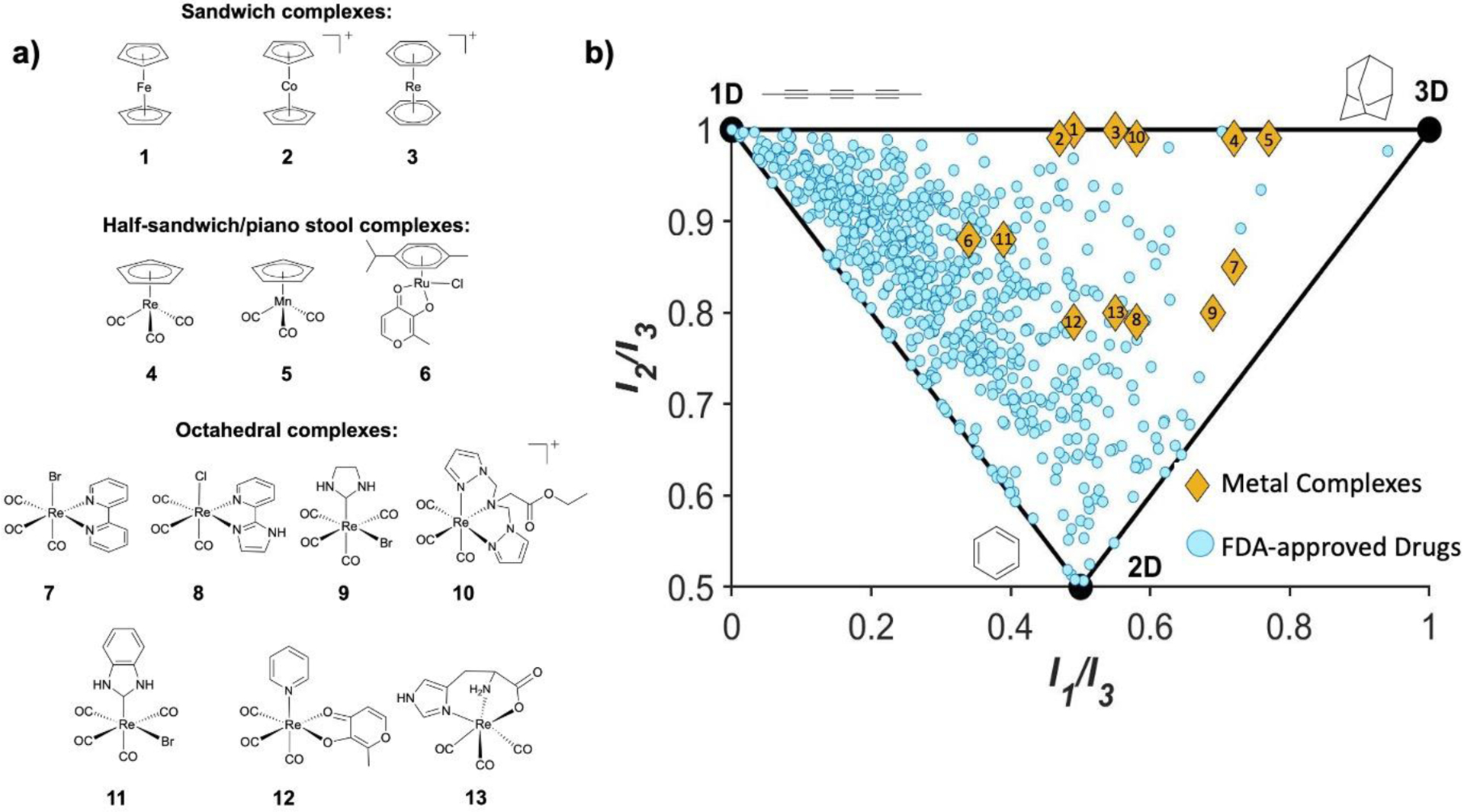
Representation of diverse chemical space occupied by metal complexes. a) Structures of plotted sandwich, half sandwich, and octahedral metal complexes. b) Principle moment of inertia plot showing FDA approved drugs (blue circles) and metal complexes (orange diamonds) from part ‘a’ of the figure. Adapted with permission from [36].
Ligand exchange
In contrast to the kinetically inert metal complexes described in the sections above, many metal complexes readily undergo ligand exchange reactions that can allow for reactivity with biomolecular targets [41]. The most famous example of a metal complex with this mechanism is the clinically approved anticancer drug cisplatin. Cisplatin enters the cell by passive diffusion or through the copper membrane transporter CTR1 pathway [42]. With respect to CTR1 transport, studies suggest that cisplatin may lose its amine ligands when transported by the methionine rich motive in CTR1 [43]. While cisplatin stays intact in the bloodstream due to high chloride concentrations (~100 mM), these chloride ligands dissociate inside cells, where the chloride concentration is significantly lower (~4–12 mM) forming the monoaqua complex [Pt(NH3)2Cl(H2O)]+ [44]. Based on the electrophilic nature of the monoaqua complex, this intermediate can rapidly react with nucleophiles such as the nucleobases of DNA. The coordinated water molecule can be displaced by purine bases in the N7 position of guanine and adenosine, generating platinum-DNA adducts (Figure 3a) [45]. In proximity to other nucleobases, the remaining chloride ligand is released, and the metal center can bind to a second purine base, generating a cross-linked adduct. These cross-linked purine bases significantly hamper DNA replication and transcription, causing cell cycle arrest and ultimately triggering cell death by apoptosis [46–48].
Figure 3.
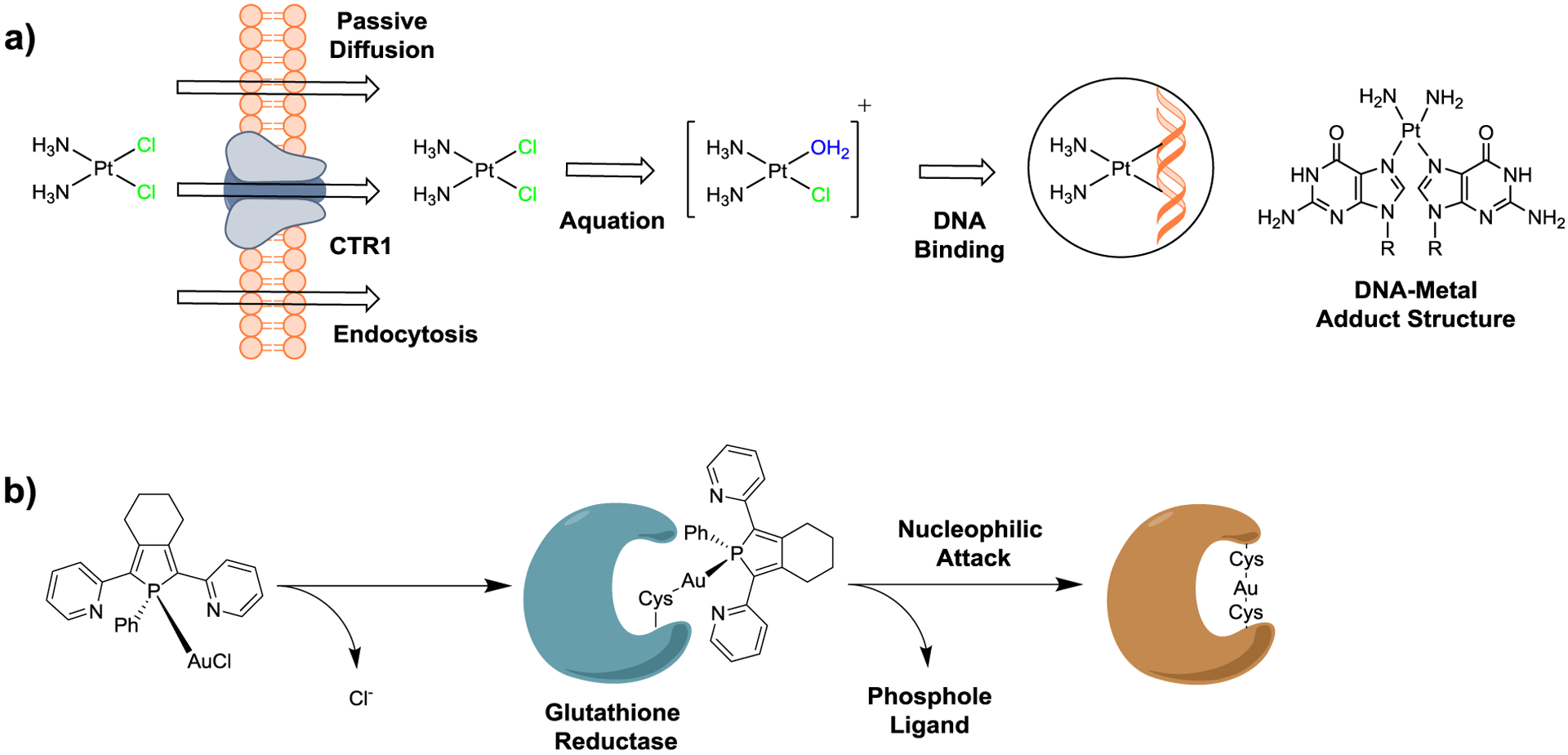
Ligand exchange activity of metal complexes. Mechanism of action of: a) the clinically approved anticancer drug cisplatin, and b) a coordinationally covalent bound Au(I) complex as an enzyme inhibitor.
Next to the targeting of nucleic acids, metal complexes with labile ligands can also interact with proteins. Fricker and coworkers demonstrated the ability of Au(III), Pd(II), and Re(V) complexes as covalent coordination-driven inhibitors against mammalian and parasitic cysteine proteases [49, 50]. Of particular interest, Au(I) containing compounds are receiving increased attention as glutathione reductase and thioredoxin reductase inhibitors for anticancer applications. Typically, these metal complexes are able to release one or more labile ligands from the metal center and then form coordinate covalent bonds to cysteine residues in the active site (Figure 3b) [51–53].
Beside the release of ligands to generate a biomolecule-metal adducts that exerts a cytotoxic effect (as with cisplatin), the released ligand itself can also result in a therapeutic effect. Carbon monoxide (CO) releasing molecules (CORMs) are an example of this strategy. Despite its reputation as the ‘silent killer’ through strong binding to hemoglobin, CO is also an endogenous small signaling molecule in the human body. Capitalizing on this function, the triggered release of CO can be used as a therapy for a variety of conditions, including bacterial infections, inflammatory diseases, sepsis, obesity, diabetes, and cancer [54, 55]. Both organic and inorganic CORMs have been described, with metal carbonyl complexes serving as an example of metal-based CORMs. Modification of the coordination environment of the central metal ion can modulate the binding of CO to the metal center to generate a slower or faster CO-releasing metal complex [56, 57]. Recently, Wilson and coworkers have demonstrated the potential of persulfide bridged Ru complexes, which are activated upon reduction of the Ru(III) center to Ru(II) in hypoxic cells, to release cytoprotective hydrogen sulfide (H2S) [58]. This is an example of redox mediated (see below) release of a bioactive ligand.
Redox activity
While electron transfer reactions of metal complexes have been used for various applications in chemistry, these processes have also found utilization for medicinal purposes. One example of introducing redox active groups is the replacement of phenyl substituents in an organic compound with a metallocene moiety. A number of examples utilizing this strategy have been reported with compounds such as Ferrocerone, Ferrocifen, or Ferroquine, which have been or are currently investigated for clinical development (Figure 4a) [59, 60]. Ferrocifen, which was developed by Jaouen and co-worker, is an isostere analog of tamoxifen, a selective estrogen receptor α (ERα) modulator which is able to repress estradiol-mediated DNA transcription inside a tumor. Ferrocifen has been investigated for the prevention and treatment of breast cancer. Due to the high selectivity of tamoxifen, it is only active in patients with an ERα overexpression. Surprisingly, Ferrocifen showed an equal activity on various kinds of breast cancer including ERα negative ones, which comprise about one third of all breast cancer patients, thereby overcoming a significant limitation of tamoxifen. Further studies have identified that the mechanism of action is based on the reversible redox properties of the ferrocene moiety of the Fe(II)/Fe(III) couple [61–63]. It is important to note that the ruthenocene derivative, which is isostructural to ferrocene but not redox active, was found to be active in ERα positive, but not active in ERα negative breast cancer models, similar to tamoxifen.[64] The proposed mechanism of Ferrocifen involves an electron transfer reaction to the metallocene moiety which is delocalized over the whole compound, followed by proton abstraction from water or biomolecules. After the release of two electrons and two protons, a reactive quinone methide intermediate is formed, which acts as a Michael acceptor that can quickly react with nucleophiles like glutathione, thioredoxin reductases or nucleobases, triggering cell death (Figure 4b) [65, 66].
Figure 4.
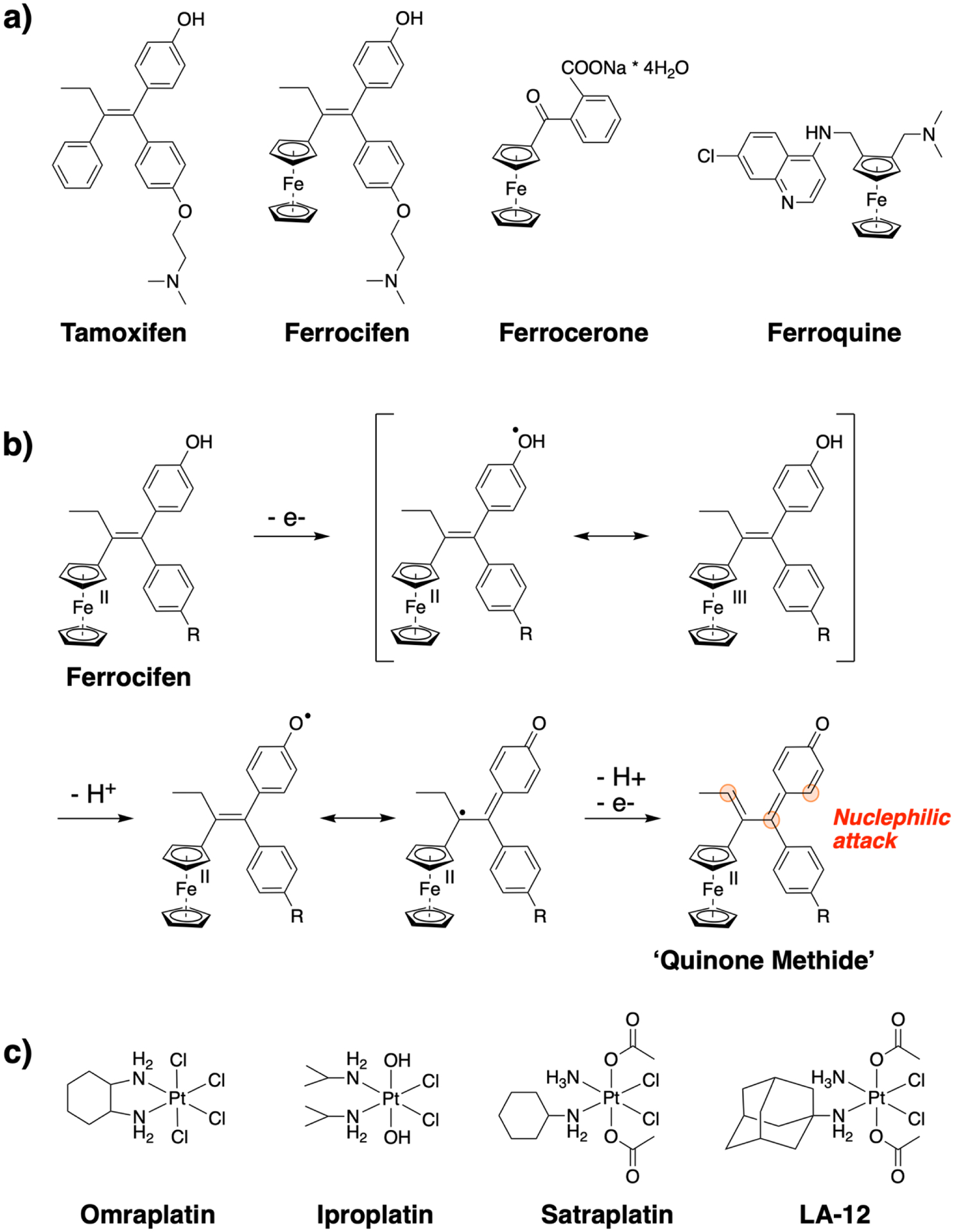
Redox bioactive of metal complexes. a) Chemical structures of Tamoxifen, Ferrocifen, Ferrocerone, and Ferroquine; b) Redox activation mechanism of Ferrocifen; c) chemical structures of redox active Pt(IV) complexes that have entered clinical trials.
A different redox initiated mechanism is the reduction of the metal center to release cytotoxic species or activate the bioactivity of the compound. This strategy is commonly used in the design of potential anticancer agents commonly based on Pt(IV), Ru(III), or Co(III) [67]. Hypoxic environments are often found around cancerous tumors, which can facilitate reduction and prevent re-oxidation [68]. The most prominent examples of this redox trigger are the Pt(IV) complexes ormaplatin, iproplatin, LA-12, and satraplatin, which have entered clinical trials (Figure 4c). Generally, octahedral Pt(IV) complexes are kinetically inert relative to Pt(II) compounds. The reduced reactivity of Pt(IV) compounds can help minimize unwanted side reactions with off-target biomolecules. In the presence of glutathione, nicotinamide adenine dinucleotide/ nicotinamide adenine dinucleotide phosphate, ascorbate, or cysteine containing biomolecules, the Pt(IV) center can be reduced to Pt(II) (Figure 4d), releasing the axial ligands and triggering greater bioactivity. As described in the previous section, the Pt(II) complex can undergo ligand exchange and ultimately bind to DNA and trigger cell death of cancerous cells [69–72]. Prior to release, the axial ligands can be used to fine tune the biological properties of the Pt(IV) complex, including lipophilicity, redox stability, cellular uptake, and cancer cell targeting, or upon release can exert a complementary cytotoxic effect. To date, due to undesired side effects and an overall low therapeutic efficacy, no Pt(IV) complexes have yet been clinically approved [73].
Catalytic activity
Nearly all current drugs act in a stoichiometric (or sub-stoichiometric) manner to achieve the desired therapeutic effect. By contrast, catalytic drugs could require far less agent to elicit a desired therapeutic effect and provides a novel, and possibly more effective, mechanism of action. By acting catalytically, and reducing the required dose, catalytic drugs could help reduce side effects and off-target toxicity [74]. Recent developments in the field of catalytic drugs have demonstrated the potential of metal complexes to act in this capacity [75]. It is important to highlight that metal complexes have been used as catalysts for centuries, but only recently have efforts to exploit their catalytic properties in biological systems been described. Most efforts have focused on metal complexes based on ruthenium, iridium, or osmium that have redox mechanism which can be activated in a biological environment [76–78].
Sadler and coworkers demonstrated that optimizing the choice of metal ion, oxidation state, and ligands of organometallic complexes can lead to catalytic biological activity [76]. A Ru(II) arene azopyridine complex was shown to be cytotoxic by disrupting cellular redox metabolism. This was accomplished by generating reactive oxygen species, which proved cytotoxic to cancerous human ovarian carcinoma or adenocarcinomic human alveolar basal epithelial cells. Additionally, these “piano-stool” compounds catalyzed the oxidation of glutathione (GSH), an important antioxidant, to glutathione disulfide (GSSH), which further contributed to oxidative stress (Figure 5a). Another recent study by Sadler and coworkers described the synthesis and evaluation of catalytically active metal complexes that are capable of the enantioselective reduction of pyruvate for the selective treatment of ovarian cancer cells [79]. In cancerous environments glycolysis is upregulated and lactate dehydrogenase is overexpressed, which catalyzes the interconversion of pyruvate and l-lactate [80, 81]. The authors note that the addition of a catalyst that can convert pyruvate to lactate can inhibit the ability of cancer cells to utilize metabolites such as NAD+. Half-sandwich Os(II) arene sulfonyl diamine complexes are able to perturb cancer cell metabolism by generating lactate in high enantiomeric excess (e.e.), which depletes cells of pyruvate. The low-spin 5d6 Os(II) complex is more kinetically stable than the analogous second row Ru(II) complex, and shows no degradation or deactivation in cell-culture media over 24 hours [82]. By synthesizing the R,R enantiomer of the Os(II) catalyst, pyruvate could be converted to d-lactate in 83% e.e. (Figure 5b). Additionally, the preparation of the S,S enantiomer resulted in the conversion of pyruvate to l-lactate with similar enantioselectivity. Administration of the Os(II) catalyst with a hydride source resulted in the selective killing of cancerous human ovarian carcinoma cells over noncancerous fibroblasts in low micromolar concentrations. This work demonstrates that the use of metal complexes to catalytically modify biomolecules that interact with enzymes could be an exciting and viable treatment method not easily achievable with organic molecules [79].
Figure 5.
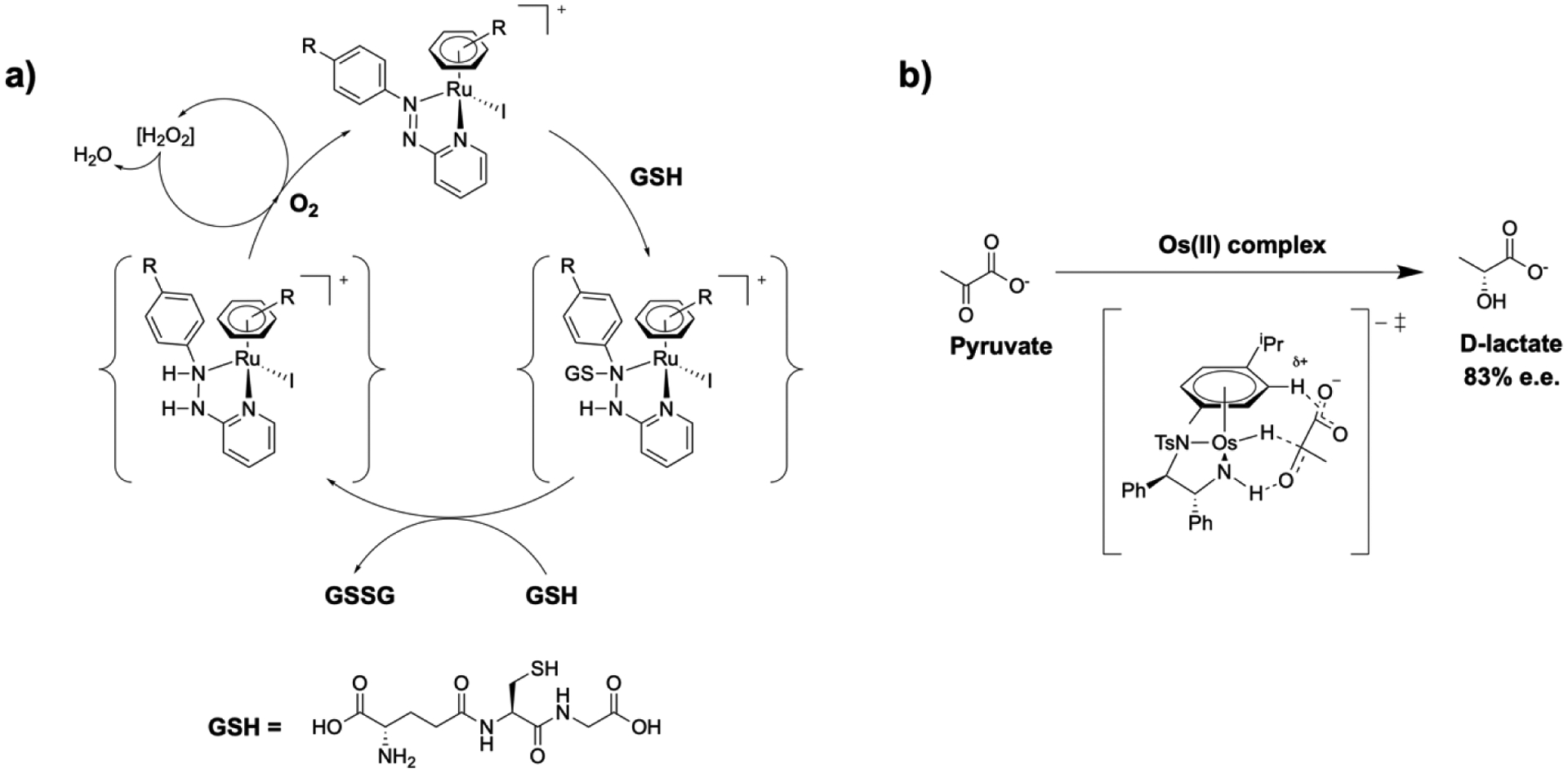
Catalytic metal complexes. a) Proposed catalytic cycle for the oxidation of GSH to GSSH; b) Enantioselective conversion of pyruvate to d-lactate with the proposed transitions state of the chiral Os(II) complex.
Photophysical activity
A number of metal complexes have photophysically accessible excited states that can be used for therapeutic applications. Upon exposure to light, an electron is excited from the ground state to a singlet state from which it can undergo an intersystem crossing process to access a triplet state [83, 84]. This excited state can interact with biomolecules or molecular oxygen to generate oxidative stress and therefore ultimately cellular damage. Kodadek and coworkers have reported the dual activity of combining a peptoid enzyme inhibitor with affinity in the micromolar range (IC50 = 49 μM) that binds in the active site of the vascular endothelial growth factor[85] with a Ru(II) polypyridine complex that can act as a photosensitizer (Figure 6a) [84]. Upon irradiation, the Ru(II) polypyridine complex catalytically generates singlet oxygen, which causes oxidative damage near the protein active site and therefore deactivates the protein (Figure 6b). Based on the dual activity of the conjugate, the activity improves significantly, with a more than 800-fold increase in activity (IC50 = 59 nM) [86].
Figure 6.
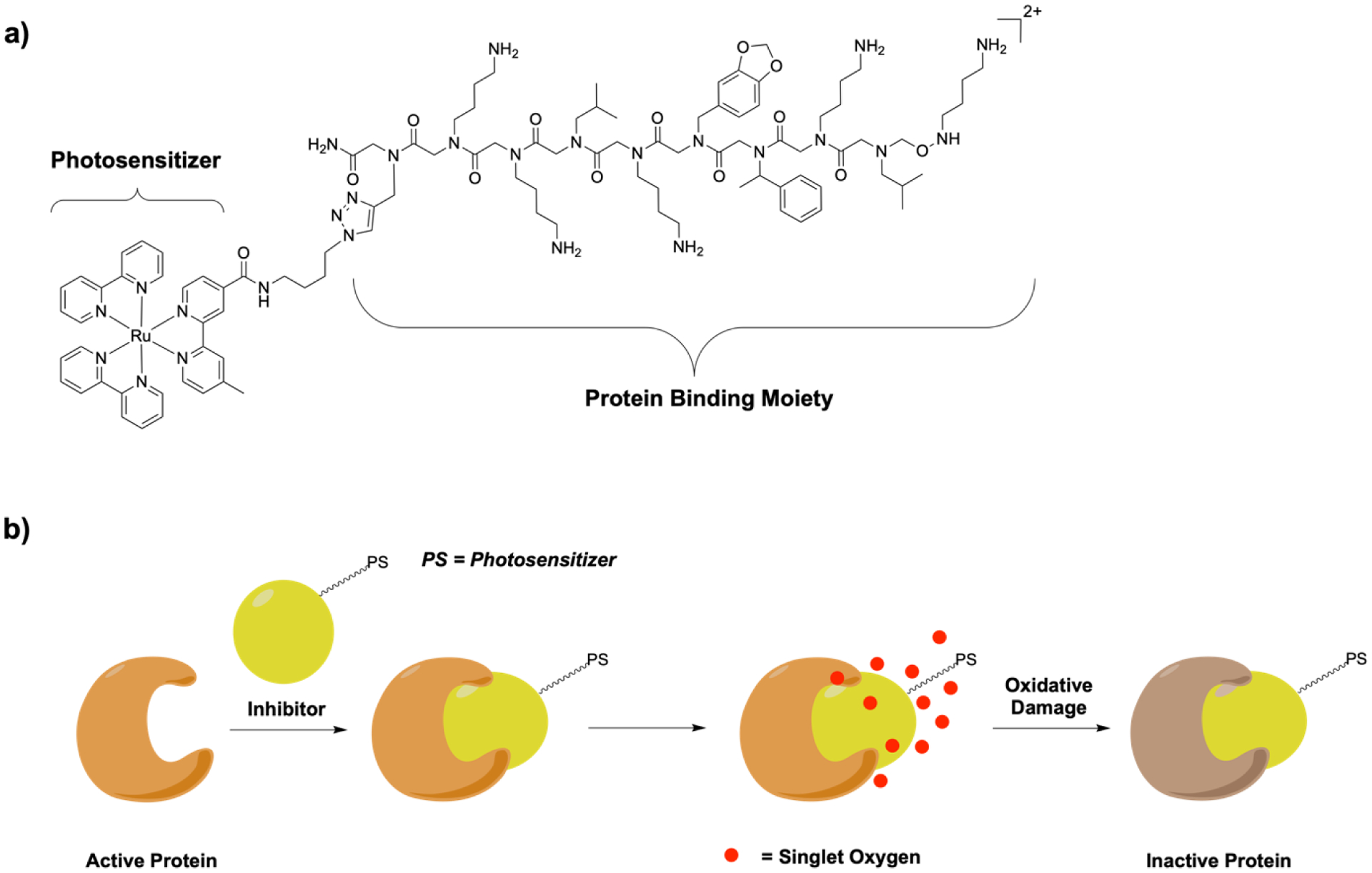
Photophysical activity of metal complexes. a) Structure of a peptoid-Ru(II) polypyridine complex conjugate; b) Dual mechanism of action of the peptoid as an enzyme inhibitor and a Ru(II) polypyridine complex as a photosensitizer for light activated protein inactivation.
Concluding remarks
Over the last several decades, increasing attention has been devoted towards the application of metal complexes for medicinal applications. Based on their unique electronic properties, reactivity, and stereochemical features, metal-containing compounds have demonstrated the ability to interact with biomolecules and biological systems through mechanisms of action which often cannot be readily accessed by organic molecules. Their broad structural diversity, three-dimensionality, ligand exchange reactions, as well as their redox, catalytic, and photophysical activity all contribute to the attractive features available with coordination complexes for use in medicine and biotechnology. Importantly, the ability of metal complexes to promote novel mechanisms of action may lead to the discovery of new treatments and therapies. These intriguing properties allow metal complexes to complement the existing drug landscape, which is composed of mostly organic molecules. Capitalizing on these advantages, a variety of metal containing compounds have already been clinically approved for a wide range of therapeutic applications. Increasing efforts to not only identify bioactive metal complexes [41], but deeply understand their mechanism of action [87–90] will be an important next step in their advancement and greater acceptance in the medical community (see Outstanding Questions). Further investigation into the therapeutic applications of metal complexes will yield novel compounds with increased potency and selectivity and may even lead to the discovery of entirely new therapeutic strategies. With these and other advancements, the use of metal complexes for medicinal applications is expected to expand and increase in the coming decades.
Acknowledgments
This work was supported by National Institute of Health grant R01 AI149444 and R21 AI138934. R.W.S. was supported, in part, by the Graduate Research Fellowship Program (GRFP) from the National Science Foundation (DGE-1650112).
Glossary
- Fragment-based drug discovery (FBDD)
A technique to discover bioactive molecules for therapeutic use. In FBDD, libraries of low molecular weight molecules are screened against a biological target of interest. The ‘hits’ from the screen are then elaborated into lead molecules with higher affinity and specificity for the target
- Half maximal inhibitory concentration (IC50)
A measure of the activity of a compound that corresponds to the concentration required to inhibit 50% of a biological process in vitro
- Ligand exchange reaction
A ligand exchange reaction involves the substitution of one or more ligands in a complex ion (coordination compound) with one or more different ligands
- High-throughput screening (HTS)
A common strategy to rapidly identify bioactive molecules for pharmaceutical use. Screening libraries of both natural and synthetic compounds are often very large, containing up to 106 compounds
- Isostere
A molecule or functional group with similar shape or electronic properties. In drug discovery, (bio)iosteres are often used to perform similar functionality as the parent molecule but mitigate pharmacokinetic liabilities
- Photosensitizer
A molecule that generates a chemical modification in another molecule during a photophysical process
- Principle moment of inertia (PMI)
Describes the resistance to angular acceleration along the principal axes (I1, I2, and I3). Normalization allows comparison of molecules with different sizes
- Structure-activity relationship
The relationship between the structure of a molecule and its ability to evoke a specific biological response
Footnotes
Declaration of Interests
There are no interests to declare.
References
- 1.Kaufmann SHE (2008) Paul Ehrlich: Founder Of Chemotherapy. Nat. Rev. Drug Discov 7 (5), 373–373. [DOI] [PubMed] [Google Scholar]
- 2.Winau F et al. (2004) Paul Ehrlich — In Search Of The Magic Bullet. Microbes Infect. 6 (8), 786–789. [DOI] [PubMed] [Google Scholar]
- 3.Lloyd NC et al. (2005) The Composition Of Ehrlich’s Salvarsan: Resolution Of A Century-Old Debate. Angew. Chem. Int. Ed 44 (6), 941–944. [DOI] [PubMed] [Google Scholar]
- 4.Rosenberg B et al. (1965) Inhibition Of Cell Division In Escherichia Coli By Electrolysis Products From A Platinum Electrode. Nature 205 (4972), 698–699. [DOI] [PubMed] [Google Scholar]
- 5.Rosenberg B et al. (1969) Platinum Compounds: A New Class Of Potent Antitumour Agents. Nature 222 (5191), 385–386. [DOI] [PubMed] [Google Scholar]
- 6.Gasser G (2015) Metal Complexes And Medicine: A Successful Combination. Chimia 7, 442–446. [DOI] [PubMed] [Google Scholar]
- 7.Armstrong-Gordon E et al. (2018) Patterns Of Platinum Drug Use In An Acute Care Setting: A Retrospective Study. J. Cancer Res. Clin. Oncol 144 (8), 1561–1568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Karges J (2020) Combining Inorganic Chemistry and Biology: The Underestimated Potential of Metal Complexes in Medicine. ChemBioChem 21 (21), 3044–3046. [DOI] [PubMed] [Google Scholar]
- 9.Anthony EJ et al. (2020) Metallodrugs Are Unique: Opportunities And Challenges Of Discovery And Development. Chem. Sci, accepted, doi: 10.1039/D0SC04082G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gasser G and Metzler-Nolte N (2012) The Potential Of Organometallic Complexes In Medicinal Chemistry. Curr. Opin. Chem. Biol 16 (1), 84–91. [DOI] [PubMed] [Google Scholar]
- 11.Meggers E (2009) Targeting Proteins With Metal Complexes. Chem. Commun (9), 1001–1010. [DOI] [PubMed] [Google Scholar]
- 12.Clemons PA et al. (2010) Small Molecules Of Different Origins Have Distinct Distributions Of Structural Complexity That Correlate With Protein-Binding Profiles. Proc. Natl. Acad. Sci 107 (44), 18787–18792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang L et al. (2004) Ruthenium Complexes As Protein Kinase Inhibitors. Org. Lett 6 (4), 521–523. [DOI] [PubMed] [Google Scholar]
- 14.Mjos KD and Orvig C (2014) Metallodrugs In Medicinal Inorganic Chemistry. Chem. Rev 114 (8), 4540–4563. [DOI] [PubMed] [Google Scholar]
- 15.Meggers E (2007) Exploring Biologically Relevant Chemical Space With Metal Complexes. Curr. Opin. Chem. Biol 11 (3), 287–292. [DOI] [PubMed] [Google Scholar]
- 16.Bregman H et al. (2004) An Organometallic Inhibitor For Glycogen Synthase Kinase 3. J. Am. Chem. Soc 126 (42), 13594–13595. [DOI] [PubMed] [Google Scholar]
- 17.Bregman H et al. (2006) Rapid Access To Unexplored Chemical Space By Ligand Scanning Around A Ruthenium Center: Discovery Of Potent And Selective Protein Kinase Inhibitors. J. Am. Chem. Soc 128 (3), 877–884. [DOI] [PubMed] [Google Scholar]
- 18.Atilla-Gokcumen GE et al. (2006) Organometallic Compounds With Biological Activity: A Very Selective And Highly Potent Cellular Inhibitor For Glycogen Synthase Kinase 3. ChemBioChem 7 (9), 1443–1450. [DOI] [PubMed] [Google Scholar]
- 19.Feng L et al. (2011) Structurally Sophisticated Octahedral Metal Complexes As Highly Selective Protein Kinase Inhibitors. J. Am. Chem. Soc 133 (15), 5976–5986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tamaoki T et al. (1986) Staurosporine, A Potent Inhibitor Of PhospholipidCa++ Dependent Protein Kinase. Biochem. Biophys. Res. Commun 135 (2), 397–402. [DOI] [PubMed] [Google Scholar]
- 21.Caravatti G et al. (1994) Inhibitory Activity And Selectivity Of Staurosporine Derivatives Towards Protein Kinase C. Bioorg. Med. Chem. Lett 4 (3), 399–404. [Google Scholar]
- 22.Kramer T et al. (2012) Small-Molecule Inhibitors Of GSK-3: Structural Insights And Their Application To Alzheimer’s Disease Models. Int. J. Alzheimers Dis 2012, 381029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Eldar-Finkelman H (2002) Glycogen Synthase Kinase 3: An Emerging Therapeutic Target. Trends Mol. Med 8 (3), 126–132. [DOI] [PubMed] [Google Scholar]
- 24.Bertrand JA et al. (2003) Structural Characterization Of The GSK-3β Active Site Using Selective And Non-Selective ATP-Mimetic Inhibitors. J. Mol. Biol 333 (2), 393–407. [DOI] [PubMed] [Google Scholar]
- 25.Meijer L et al. (2003) GSK-3-Selective Inhibitors Derived From Tyrian Purple Indirubins. Chem. Biol 10 (12), 1255–1266. [DOI] [PubMed] [Google Scholar]
- 26.Davies CL et al. (2009) Supramolecular Interactions Between Functional Metal Complexes And Proteins. Dalton Trans. (46), 10141–10154. [DOI] [PubMed] [Google Scholar]
- 27.Galloway WRJD et al. (2010) Diversity-Oriented Synthesis As A Tool For The Discovery Of Novel Biologically Active Small Molecules. Nat. Commun 1 (1), 80. [DOI] [PubMed] [Google Scholar]
- 28.Hung AW et al. (2011) Route To Three-Dimensional Fragments Using Diversity-Oriented Synthesis. Proc. Natl. Acad. Sci 108 (17), 6799–6804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kombo DC et al. (2013) 3D Molecular Descriptors Important For Clinical Success. J. Chem. Inf. Model 53 (2), 327–342. [DOI] [PubMed] [Google Scholar]
- 30.Firth NC et al. (2012) Plane Of Best Fit: A Novel Method To Characterize The Three-Dimensionality Of Molecules. J. Chem. Inf. Model 52 (10), 2516–2525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yang Y et al. (2012) Beyond Size, Ionization State, And Lipophilicity: Influence Of Molecular Topology On Absorption, Distribution, Metabolism, Excretion, And Toxicity For Druglike Compounds. J. Med. Chem 55 (8), 3667–3677. [DOI] [PubMed] [Google Scholar]
- 32.Meyers J et al. (2016) On The Origins Of Three-Dimensionality In Drug-Like Molecules. Future Med. Chem 8 (14), 1753–1767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lovering F et al. (2009) Escape From Flatland: Increasing Saturation As An Approach To Improving Clinical Success. J. Med. Chem 52 (21), 6752–6756. [DOI] [PubMed] [Google Scholar]
- 34.Scott DE et al. (2012) Fragment-Based Approaches In Drug Discovery And Chemical Biology. Biochemistry 51 (25), 4990–5003. [DOI] [PubMed] [Google Scholar]
- 35.Prosser KE et al. (2020) Evaluation Of 3-Dimensionality In Approved And Experimental Drug Space. ACS Med. Chem. Lett 11 (6), 1292–1298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Morrison CN et al. (2020) Expanding Medicinal Chemistry Into 3D space: Metallofragments As 3D Scaffolds For Fragment-Based Drug Discovery. Chem. Sci 11 (5), 1216–1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Leach AR et al. (2010) Three-Dimensional Pharmacophore Methods In Drug Discovery. J. Med. Chem 53 (2), 539–558. [DOI] [PubMed] [Google Scholar]
- 38.Twigg DG et al. (2016) Partially Saturated Bicyclic Heteroaromatics As An Sp3-Enriched Fragment Collection. Angew. Chem. Int. Ed 55 (40), 12479–12483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kilpin KJ and Dyson PJ (2013) Enzyme Inhibition By Metal Complexes: Concepts, Strategies And Applications. Chem. Sci 4 (4), 1410–1419. [Google Scholar]
- 40.Sauer WHB and Schwarz MK (2003) Molecular Shape Diversity Of Combinatorial Libraries: A Prerequisite For Broad Bioactivity. J. Chem. Inf. Model 43 (3), 987–1003. [DOI] [PubMed] [Google Scholar]
- 41.Meier-Menches SM et al. (2018) Structure–Activity Relationships For Ruthenium And Osmium Anticancer Agents – Towards Clinical Development. Chem. Soc. Rev 47 (3), 909–928. [DOI] [PubMed] [Google Scholar]
- 42.Holzer AK et al. (2006) Contribution Of The Major Copper Influx Transporter CTR1 To The Cellular Accumulation Of Cisplatin, Carboplatin, And Oxaliplatin. Mol. Pharmacol 70 (4), 1390–1394. [DOI] [PubMed] [Google Scholar]
- 43.Alessia L et al. (2018) Monitoring Interactions Inside Cells By Advanced Spectroscopies: Overview Of Copper Transporters And Cisplatin. Curr. Med. Chem 25 (4), 462–477. [DOI] [PubMed] [Google Scholar]
- 44.Jung Y and Lippard SJ (2007) Direct Cellular Responses To Platinum-Induced DNA Damage. Chem. Rev 107 (5), 1387–1407. [DOI] [PubMed] [Google Scholar]
- 45.Eastman A (1987) The Formation, Isolation And Characterization Of DNA Adducts Produced By Anticancer Platinum Complexes. Pharmacol. Ther 34 (2), 155–166. [DOI] [PubMed] [Google Scholar]
- 46.Basu A and Krishnamurthy S (2010) Cellular Responses To Cisplatin-Induced DNA Damage. J. Nucleic Acids 2010, 201367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Browning RJ et al. (2017) Drug Delivery Strategies For Platinum-Based Chemotherapy. ACS Nano 11 (9), 8560–8578. [DOI] [PubMed] [Google Scholar]
- 48.Todd RC and Lippard SJ (2009) Inhibition Of Transcription By Platinum Antitumor Compounds. Metallomics 1 (4), 280–291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fricker SP et al. (2008) Metal Compounds For The Treatment Of Parasitic Diseases. J. Inorg. Biochem 102 (10), 1839–1845. [DOI] [PubMed] [Google Scholar]
- 50.Fricker SP (2010) Cysteine Proteases As Targets For Metal-Based Drugs. Metallomics 2 (6), 366–377. [DOI] [PubMed] [Google Scholar]
- 51.Bindoli A et al. (2009) Thioredoxin Reductase: A Target For Gold Compounds Acting As Potential Anticancer Drugs. Coord. Chem. Rev 253 (11), 1692–1707. [Google Scholar]
- 52.Urig S et al. (2006) Undressing Of Phosphine Gold(I) Complexes As Irreversible Inhibitors Of Human Disulfide Reductases. Angew. Chem. Int. Ed 45 (12), 1881–1886. [DOI] [PubMed] [Google Scholar]
- 53.Ott I (2009) On The Medicinal Chemistry Of Gold Complexes As Anticancer Drugs. Coord. Chem. Rev 253 (11), 1670–1681. [Google Scholar]
- 54.Faizan M et al. (2019) CO-Releasing Materials: An Emphasis On Therapeutic Implications, As Release And Subsequent Cytotoxicity Are The Part Of Therapy. Materials 12 (10), 1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ismailova A et al. (2018) An Overview Of The Potential Therapeutic Applications Of CO-Releasing Molecules. Bioinorg. Chem. Appl 2018, 8547364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yan H et al. (2019) Emerging Delivery Strategies Of Carbon Monoxide For Therapeutic Applications: From CO Gas To CO Releasing Nanomaterials. Small 15 (49), 1904382. [DOI] [PubMed] [Google Scholar]
- 57.Abeyrathna N et al. (2017) Nonmetallic Carbon Monoxide Releasing Molecules (CORMs). Org. Biomol. Chem 15 (41), 8692–8699. [DOI] [PubMed] [Google Scholar]
- 58.Woods JJ and Wilson JJ (2021) A Dinuclear Persulfide-Bridged Ruthenium Compound Is A Hypoxia-Selective Hydrogen Sulfide (H2S) Donor. Angew. Chem. Int. Ed 60 (3), 1588–1592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ong YC and Gasser G (2019) Organometallic Compounds In Drug Discovery: Past, Present And Future. Drug Discov. Today Technol [DOI] [PubMed] [Google Scholar]
- 60.McCarthy JS et al. (2016) A Phase II Pilot Trial To Evaluate Safety And Efficacy Of Ferroquine Against Early Plasmodium Falciparum In An Induced Blood-Stage Malaria Infection Study. Malar. J 15 (1), 469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hillard E et al. (2006) Ferrocene-Mediated Proton-Coupled Electron Transfer In A Series Of Ferrocifen-Type Breast-Cancer Drug Candidates. Angew. Chem. Int. Ed 45 (2), 285–290. [DOI] [PubMed] [Google Scholar]
- 62.Vessières A et al. (2005) Modification Of The Estrogenic Properties Of Diphenols By The Incorporation Of Ferrocene. Generation Of Antiproliferative Effects In Vitro. J. Med. Chem 48 (12), 3937–3940. [DOI] [PubMed] [Google Scholar]
- 63.Gerard J et al. (2004) The First Organometallic Selective Estrogen Receptor Modulators (SERMs) And Their Relevance to Breast Cancer. Curr. Med. Chem 11 (18), 2505–2517. [DOI] [PubMed] [Google Scholar]
- 64.Pigeon P et al. (2005) Selective Estrogen Receptor Modulators In The Ruthenocene Series. Synthesis And Biological Behavior. J. Med. Chem 48 (8), 2814–2821. [DOI] [PubMed] [Google Scholar]
- 65.Hamels D et al. (2009) Ferrocenyl Quinone Methides As Strong Antiproliferative Agents: Formation By Metabolic And Chemical Oxidation Of Ferrocenyl Phenols. Angew. Chem. Int. Ed 48 (48), 9124–9126. [DOI] [PubMed] [Google Scholar]
- 66.Jaouen G et al. (2015) Ferrocifen Type Anti Cancer Drugs. Chem. Soc. Rev 44 (24), 8802–8817. [DOI] [PubMed] [Google Scholar]
- 67.Graf N and Lippard SJ (2012) Redox Activation Of Metal-Based Prodrugs As A Strategy For Drug Delivery. Adv. Drug Deliv. Rev 64 (11), 993–1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Brown JM (1999) The Hypoxic Cell. Cancer Res 59 (23), 5863–5870. [PubMed] [Google Scholar]
- 69.Johnstone TC et al. (2016) The Next Generation Of Platinum Drugs: Targeted Pt(II) Agents, Nanoparticle Delivery, And Pt(IV) Prodrugs. Chem. Rev 116 (5), 3436–3486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Karges J et al. (2020) A Multi-action And Multi-target RuII–PtIV Conjugate Combining Cancer-Activated Chemotherapy And Photodynamic Therapy To Overcome Drug Resistant Cancers. Angew. Chem. Int. Ed 59 (18), 7069–7075. [DOI] [PubMed] [Google Scholar]
- 71.Han X et al. (2015) Recent Advances In Platinum (IV) Complex-Based Delivery Systems To Improve Platinum (II) Anticancer Therapy. Med. Res. Rev 35 (6), 1268–1299. [DOI] [PubMed] [Google Scholar]
- 72.Muhammad N and Guo Z (2014) Metal-Based Anticancer Chemotherapeutic Agents. Curr. Opin. Chem. Biol 19, 144–153. [DOI] [PubMed] [Google Scholar]
- 73.Gibson D (2019) Multi-Action Pt(IV) Anticancer Agents; Do We Understand How They Work? J. Inorg. Biochem 191, 77–84. [DOI] [PubMed] [Google Scholar]
- 74.Soldevila-Barreda JJ and Metzler-Nolte N (2019) Intracellular Catalysis With Selected Metal Complexes And Metallic Nanoparticles: Advances Toward The Development of Catalytic Metallodrugs. Chem. Rev 119 (2), 829–869. [DOI] [PubMed] [Google Scholar]
- 75.Yu Z and Cowan JA (2017) Catalytic Metallodrugs: Substrate-Selective Metal Catalysts As Therapeutics. Chem. Eur. J 23 (57), 14113–14127. [DOI] [PubMed] [Google Scholar]
- 76.Dougan SJ et al. (2008) Catalytic Organometallic Anticancer Complexes. Proc. Natl. Acad. Sci 105 (33), 11628–11633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Liu Z et al. (2014) The Potent Oxidant Anticancer Activity Of Organoiridium Catalysts. Angew. Chem. Int. Ed 53 (15), 3941–3946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Fu Y et al. (2012) The Contrasting Chemical Reactivity Of Potent Isoelectronic Iminopyridine And Azopyridine Osmium(ii) Arene Anticancer Complexes. Chem. Sci 3 (8), 2485–2494. [Google Scholar]
- 79.Coverdale JPC et al. (2018) Asymmetric Transfer Hydrogenation By Synthetic Catalysts In Cancer Cells. Nat. Chem 10 (3), 347–354. [DOI] [PubMed] [Google Scholar]
- 80.Hirschhaeuser F et al. (2011) Lactate: A Metabolic Key Player In Cancer. Cancer Res 71 (22), 6921–6925. [DOI] [PubMed] [Google Scholar]
- 81.Doherty JR and Cleveland JL (2013) Targeting Lactate Metabolism For Cancer Therapeutics. J. Clin. Invest 123 (9), 3685–3692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Peacock AFA et al. (2006) Tuning The Reactivity Of Osmium(II) And Ruthenium(II) Arene Complexes Under Physiological Conditions. J. Am. Chem. Soc 128 (5), 1739–1748. [DOI] [PubMed] [Google Scholar]
- 83.Vogler A and Kunkely H (2001) Luminescent Metal Complexes: Diversity Of Excited States. In Transition Metal and Rare Earth Compounds: Excited States, Transitions, Interactions I (Yersin Hed), pp. 143–182, Springer Berlin; Heidelberg. [Google Scholar]
- 84.Heinemann F et al. (2017) Critical Overview of the Use of Ru(II) Polypyridyl Complexes As Photosensitizers In One-Photon and Two-Photon Photodynamic Therapy. Acc. Chem. Res 50 (11), 2727–2736. [DOI] [PubMed] [Google Scholar]
- 85.Udugamasooriya DG et al. (2008) A Peptoid “Antibody Surrogate” That Antagonizes VEGF Receptor 2 Activity. J. Am. Chem. Soc 130 (17), 5744–5752. [DOI] [PubMed] [Google Scholar]
- 86.Lee J et al. (2010) Potent And Selective Photo-Inactivation Of Proteins With Peptoid-Ruthenium Conjugates. Nat. Chem. Biol 6 (4), 258–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.King AP and Wilson JJ (2020) Endoplasmic reticulum stress: an arising target for metal-based anticancer agents. Chem. Soc. Rev 49, 8113–8136. [DOI] [PubMed] [Google Scholar]
- 88.Marker SC et al. (2020) Exploring Ovarian Cancer Cell Resistance to Rhenium Anticancer Complexes. Angew. Chem. Int. Ed 59, 13391–13400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Woods JJ et al. (2020) Redox Stability Controls the Cellular Uptake and Activity of Ruthenium-Based Inhibitors of the Mitochondrial Calcium Uniporter (MCU). Angew. Chem. Int. Ed 59, 6482–6491. [DOI] [PubMed] [Google Scholar]
- 90.Suntharalingam K et al. (2013) Bidentate Ligands on Osmium(VI) Nitrido Complexes Control Intracellular Targeting and Cell Death Pathways. Journal of the American Chemical Society 135 (38), 14060–14063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lipinski CA et al. (2001) Experimental And computational Approaches To Estimate Solubility And Permeability In Drug Discovery And Development Settings. Adv. Drug Deliv. Rev 46 (1), 3–26. [DOI] [PubMed] [Google Scholar]
- 92.Congreve M et al. (2003) A ‘Rule of Three’ For Fragment-Based Lead Discovery? Drug Discov. Today 8 (19), 876–877. [DOI] [PubMed] [Google Scholar]


