Abstract
Nucleotide-binding leucine-rich repeat-containing proteins, or NOD-like receptors (NLRs), are intracellular innate immune sensors that can regulate several signaling pathways, including MyD88- and TRIF-dependent pathways. In addition to these regulatory roles, some NLRs can assemble into multimeric protein complexes known as inflammasomes. NLRP12 is a member of the NLR family that contains an N-terminal pyrin domain, a central nucleotide-binding domain, and a C-terminal leucine-rich repeat. It has been shown to play a role in forming an inflammasome in response to specific infections, and it can also function as a regulator of inflammatory signaling. During Yersinia pestis or Plasmodium chabaudi, infection, NLRP12 induces the release of the inflammasome-dependent cytokines IL-1β and IL-18. These NLRP12-dependent cytokines confer protection against severe infections caused by these pathogens. Conversely, during infection with Salmonella enterica serovar Typhimurium, vesicular stomatitis virus, Klebsiella pneumoniae, or Mycobacterium tuberculosis, and in colonic tumorigenesis, NLRP12 acts as a negative regulator of the NFκB and MAPK signaling pathways. NLRP12 also negatively regulates canonical and non-canonical signaling in T cells and causes exacerbated autoimmune diseases. Furthermore, NLRP12 acts as a central component in maintaining intestinal inflammation and gut homeostasis. Therefore, the ability of NLRP12 to function as an inflammasome or as a negative regulator is context-dependent. In this review, we provide an overview of the NLR family members and summarize recent insights into the roles of NLRP12 as an inflammasome and as a negative regulator.
Keywords: NLRP12, ASC, Caspase-1, IL-1β, IL-18, Gasdermin, Pyroptosis, Inflammasome, Inflammation, Infection, Cell death, Pyrin domain, Cancer, Negative regulator
1. Introduction
Innate immunity acts as the first line of defense against pathogenic and sterile insults. Innate immune cells like monocytes, macrophages, dendritic cells, endothelial cells, and neutrophils, use fixed, germline-encoded pattern recognition receptors (PRRs) to recognize activating stimuli in the host system (Newton and Dixit, 2012; Takeuchi and Akira, 2010; Tuncer et al., 2014). PRRs can detect pathogen-associated molecular patterns (PAMPs) and endogenous damage-associated molecular patterns (DAMPs) (Kesavardhana and Kanneganti, 2017; Malik and Kanneganti, 2017). Based on their localization, PRRs are categorized as membrane-bound or cytoplasmic. Toll-like receptors (TLRs) and C-type lectin receptors (CLRs), which are able to sense extracellular pathogens, are membrane-bound and are localized to the plasma membrane and endosomes. Retinoic acid inducible gene I (RIG-I)-like receptors (RLRs), nucleotide-binding leucine-rich repeat-containing proteins (or NOD-like receptors, NLRs), absent in melanoma 2 (AIM2)-like receptors (ALRs), and Pyrin are localized in the cytoplasm of the cell and act as surveillance molecules for intracellular microbes, PAMPs, and DAMPs (Fritz et al., 2006; Kanneganti et al., 2006; Meylan et al., 2006; Proell et al., 2008; Tuncer et al., 2014; Werts et al., 2006). These intracellular sensors can be activated when they recognize bacterial components (cell wall molecules, flagellin, inner rod or proteins from the bacterial secretion system), viral proteins, microbial nucleic acid, or microbial toxins. In addition to detecting microbial components, these sensors can also detect cellular components (e.g., ATP), released due to host cell damage or a disbalance in the potassium efflux, and the presence of endosome danger signals, such as uric acid crystals (Bauernfeind et al., 2009; Broz and Dixit, 2016; Franchi et al., 2009a; Kesavardhana and Kanneganti, 2017; Man et al., 2017; Muñoz-Planillo et al., 2013). Following the recognition of these various ligands and cellular components, the PRRs are activated and induce a cascade of signaling pathways that can lead to the upregulation or suppression of inflammatory cytokines.
Upon activation, a subset of the PRRs can form a multiprotein complex known as the inflammasome to drive cytokine secretion and cell death. Though inflammasome activation is important in eliminating pathogens from the host, dysregulation leads to diseases such as atherosclerosis, type 2 diabetes, neurodegenerative diseases, cancer, and auto-inflammatory diseases. Thus, it is very important to fine tune the activation and inhibition of inflammasomes to avoid any accidental damage to the host tissue. Several NLR family members that form inflammasome complexes in response to the PAMPs and DAMPs have been well described, with the most well characterized being NLRP1, NLRP3, and NLRC4. In recent years, additional NLRs, such as NLRC1, NLRC2, NLRC3, NLRC5, NLRP6, NLRP12, and NLRX1, have been shown to play a role as positive or negative regulators of immune signaling. Studies have shown that NLRP6 and NLRP12 have dual functions, and each can function as an inflammasome or as a negative regulator of immune signaling. In this review, we will present an overview of the NLRs and focus on NLRP12 and its functions as an inflammasome and a negative regulator of signaling pathways.
2. The NLRs
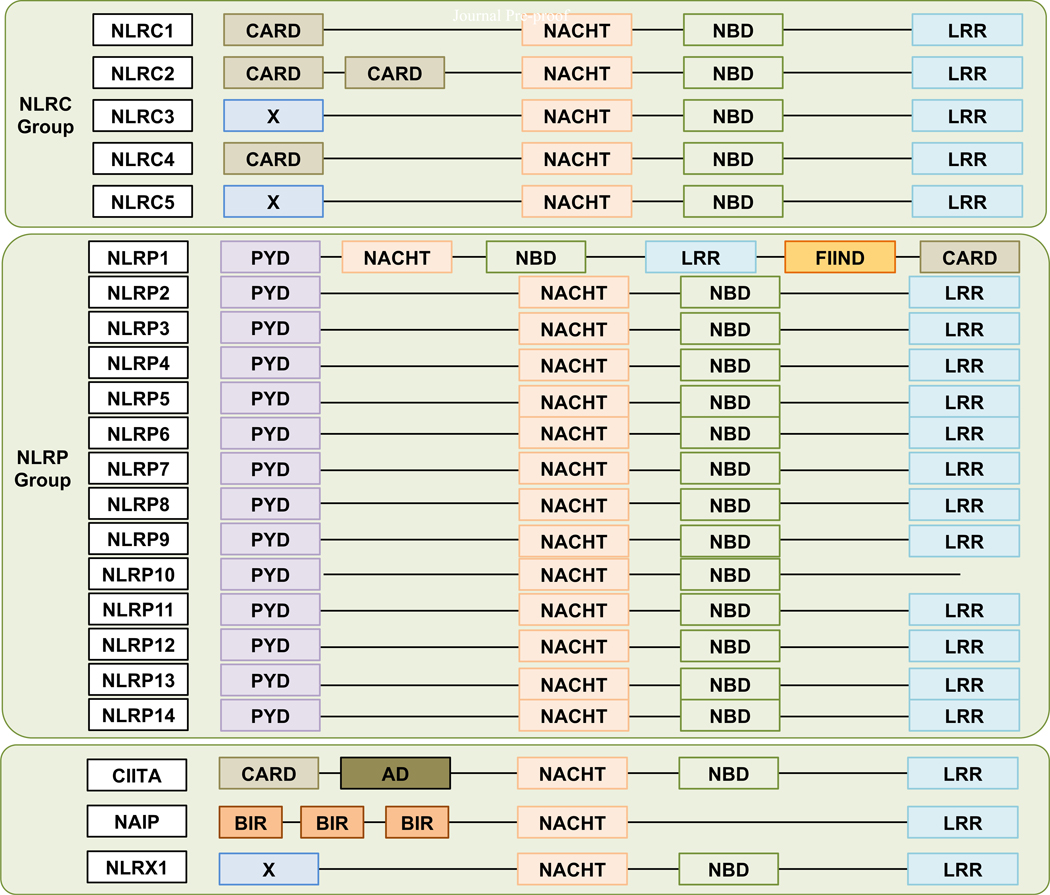
The NLR family consists of more than 20 members, and these proteins play diverse roles in innate immunity and inflammation (Figure 1) (Carneiro et al., 2008; Clay et al., 2014; Harton et al., 2002; Inohara and Nuñez, 2003; Martinon et al., 2009; Proell et al., 2008). NLRs are multi-domain proteins with tripartite architecture which share common structural motifs: they consist of a C-terminal LRR motif, a central NATCH domain that is responsible for ATP-dependent oligomerization, and an N-terminal effector domain that is either a pyrin domain (PYD), a caspase recruitment domain (CARD), or baculovirus inhibitor of apoptosis protein repeat (BIR) domain that links the NLR to downstream signaling molecules (Figure 1) (Clay et al., 2014; Pinheiro et al., 2011; Proell et al., 2008). However, NLRs are distinct in their domain architecture and function (Cridland et al., 2012; Harton et al., 2002). Several NLRs can undergo a conformational change allowing them to oligomerize and recruit the adaptor protein apoptosis-associated speck-like protein (ASC), which contains a PYD and CARD, to form a large multiprotein complex known as the inflammasome (Karki and Kanneganti, 2019; Kesavardhana and Kanneganti, 2017; Lu et al., 2014; Man et al., 2017; Rathinam and Fitzgerald, 2016; Shaw et al., 2011). Recruitment of ASC and pro–caspase-1 to these sensors then allows autoproteolytic cleavage to form the catalytically active protease caspase-1, which mediates the proteolytic processing of proinflammatory cytokines IL-1β and IL-18 and gasdermin D (GSDMD) to their active forms. Cleavage of GSDMD leads to the execution of a type of inflammatory cell death called pyroptosis (Broz et al., 2010; Broz and Dixit, 2016; Ding et al., 2016; He et al., 2015; Liu et al., 2016; Shi et al., 2015).
Figure 1:
Schematic representation of NLR family members, including members of the NLRC and NLRP groups. The arrow indicates that NLRP2 to NLRP9 and NLRP11 to NLRP14 share the same domains.
AD, activation domain; BIR, baculovirus inhibitor of apoptosis protein repeat; CARD, caspase recruitment domain; CIITA, class II major histocompatibility complex transactivator; FIIND, function to find domain; LRR, leucine-rich repeats; NACHT domain, NAIP, CIITA, HET-E, and TP1-containing domain; NBD, nucleotide-binding domain; PYD, pyrin domain.
NAIP-NLRC4 inflammasomes assemble in response to bacterial flagellin or rod or needle subunits of the bacterial type III secretion system (T3SS) (Franchi et al., 2009b; Miao et al., 2010, 2006; Poyet et al., 2001; Reyes Ruiz et al., 2017; Zhao et al., 2011). The bacterial ligands do not directly interact with NLRC4; NLRC4 uses NAIPs as sensors upstream of its inflammasome assembly. Both the NAIPs and NLRC4 are transcriptionally regulated by IRF9 (Karki et al., 2018). NAIP1 and NAIP2 sense rod and needle from the T3SS, and NAIP5 and NAIP6 bind to flagellin (Kofoed and Vance, 2011; Lightfield et al., 2008; Suzuki et al., 2014; Yang et al., 2013; Zhao et al., 2011).
NLRP1b recognizes anthrax lethal toxin, which is secreted by Bacillus anthracis. The lethal toxin cleaves at the N-terminal region of NLRP1b, causing autoproteolytic processing that leads to the activation and assembly of the NLRP1b inflammasome (Boyden and Dietrich, 2006; Chavarría-Smith et al., 2016; Chavarría-Smith and Vance, 2013; Chui et al., 2019; Eldeeb et al., 2019; Levinsohn et al., 2012; Nour et al., 2009; Sandstrom et al., 2019).
NLRP3 can be activated by a diverse array of DAMPs, such as uric acid, silica, alum, asbestos, and endogenous stimuli like ATP, newly synthesized mitochondrial DNA and PAMPs derived from viral, bacterial, fungal, and parasitic pathogens (Franchi et al., 2009a; Kanneganti et al., 2007, 2006; Lamkanfi and Kanneganti, 2010; Man and Kanneganti, 2015; Muñoz-Planillo et al., 2013; Zhong et al., 2018). However, no unified mechanism for NLRP3 inflammasome activation has been identified. It was recently discovered that the DEAD-box family member and helicase, DDX3X, is required for the formation of stress granules and the NLRP3 inflammasome, acting as a molecular switch between cell survival and cell death in response to certain stress signals (Samir et al., 2019). Furthermore, several other mediators like Z-DNA-binding protein 1/DNA-dependent activator of IFN-regulatory factors (ZBP1/DAI), TGF-β-activated kinase 1 (TAK1), Fas-associated death domain (FADD), and Toll or interleukin-1 receptor domain-containing adaptor inducing IFN-β (TRIF) have been shown to regulate NLRP3 inflammasome activation (Gurung et al., 2016, 2014, 2012; Kuriakose et al., 2016; Malireddi et al., 2018)
In addition to these canonical inflammasome-forming NLRs, studies have shown that other NLRs can function as either positive or negative regulators of crucial inflammatory signaling pathways such as NFκB, mitogen-activated protein kinase (MAPK), type I IFN, autophagy, and the generation of reactive oxygen species (ROS) (Anand et al., 2012; Chamaillard et al., 2003; Chen et al., 2019; Cui et al., 2010; Franchi et al., 2009b; Ghimire et al., 2018; Kanneganti et al., 2007; Kobayashi et al., 2005; Meissner et al., 2010; Park et al., 2007; Schneider et al., 2012; Xia et al., 2011; Zhang et al., 2014).
NLRC1 is expressed in various leukocytes and epithelial cells (Strober et al., 2006), whereas NLRC2 is expressed in macrophages, dendritic cells, Paneth cells, keratinocytes, epithelial cells in the lungs, and in the intestine and oral cavity (Franchi et al., 2009b; Ogura et al., 2003, 2001; Tada et al., 2005) . These multi-domain proteins consist of one or two CARDs, a centrally located NOD, and several C-terminal LRRs. NLRC1 recognizes meso-diaminopimelic acid (meso-DAP)-containing peptidoglycan (PGN) fragments produced by all Gram-negative bacteria such as Shigella flexneri, Escherichia coli, Chlamydia, Pseudomonas aeruginosa and Helicobacter pylori and some Gram-positive bacteria; alternatively, NLRC2 recognizes muramyl dipeptide (MDP) found in the PGN of both Gram-negative and Gram-positive microbes (Chamaillard et al., 2003; Girardin et al., 2001; Kim et al., 2004; Opitz et al., 2005; Travassos et al., 2005; Watanabe et al., 2011). Upon recognition of bacterial components, NLRC1 and NLRC2 are activated and can directly recruit serine-threonine kinase receptor-interacting protein-like interacting CLARP kinase (RICK) via CARD-CARD interactions (Ogura et al., 2001). RICK binds to the NFκB essential modulator (NEMO) and promotes activation of IKKα and IKKβ, which leads to degradation of IκB and the release and translocation of NFκB to the nucleus (Abbott et al., 2004; Hasegawa et al., 2008; Kim et al., 2008; Kobayashi et al., 2005; Park et al., 2007).
NLRC3 is highly expressed in immune cells and consists of a central NOD and a C-terminal LRR domain (Schneider et al., 2012). NLRC3 acts as a negative regulator of signaling pathways that are activated by TLRs and the DNA sensor stimulator of IFN-inducible genes (STING) in response to PAMPs or viral infection (Karki et al., 2016; Li et al., 2019; Schneider et al., 2012; Zhang et al., 2014). During colorectal cancer, NLRC3 mediates protection by associating with PI3K and blocking the activation of AKT kinase, inhibiting mTOR pathways in the epithelial cells of the colon (Karki et al., 2016). Furthermore, NLRC3 regulates cellular proliferation and apoptosis by suppressing c-Myc expression and activating PI3K-AKT targets like FoxO3a and FoxO1 (Karki et al., 2017). NLRC3 has also been shown to be a negative regulator or CD4+ T cell as it limits the activation, proliferation, and downstream IFN-γ and TNF expression in T cells by interfering with NFκB signaling (Hu et al., 2018; Uchimura et al., 2018).
NLRC5 is mostly expressed in bone marrow, lymph nodes, spleen, and in mucosal surfaces like lung, small intestine, colon, and uterus (Benko et al., 2010; Cui et al., 2010; Davis et al., 2011; Kuenzel et al., 2010; Lamkanfi and Kanneganti, 2012). NLRC5 contains a CARD-like domain, a central NOD domain, and a C-terminal LRR region (Cui et al., 2010; Dowds et al., 2003). The role of NLRC5 as a regulator of innate and adaptive immune responses during host defense is controversial. NLRC5 dampens production of proinflammatory cytokines and IFNs in response to viral infection (Benko et al., 2010; Cui et al., 2010). However, other studies have shown that NLRC5 promotes production of type I IFN and proinflammatory cytokines in fibroblasts and in primary human cells during cytomegalovirus or Sendai virus infection (Kuenzel et al., 2010; Neerincx et al., 2010).
NLRX1 is ubiquitously expressed in immune cells and is the only member of the NLR family that has a mitochondrial targeting sequence in its N-terminus (Shaw et al., 2010). NLRX1 inhibits type I IFN production by interacting with mitochondrial antiviral signaling protein (MAVS), RIG-I, and melanoma differentiation-associated protein (MDA5) signaling (Allen et al., 2011; Qin et al., 2017; Xia et al., 2011). Studies have shown that NLRX1 dissociates from TRAF6 and interacts with the IKK complex to downregulate NFκB signaling (Allen et al., 2011; Xia et al., 2011). Furthermore, during Group A Streptococcus (GAS) infection, NLRX1 functions as a negative regulator by interacting with the Beclin 1-UVRAG complex, which regulates invasion and autophagy, via its NATCH domain. This interaction inactivates the complex (Aikawa et al., 2018). Although studies have shown NLRX1 to be a negative regulator of innate immunity, in human hepatocytes NLRX1 acts as a positive regulator and is shown to enhance antiviral responses by restricting the replication of Hepatitis A virus and Hepatitis C virus (Feng et al., 2017). Additionally, during Sendai virus infection, NLRX1 positively regulates interferon regulatory factor 1 (IRF1) signaling but suppresses IRF3 dimerization, showing that NLRX1 can act both as a positive and negative regulator; the functional consequences can be dependent on the cell type or pathogen involved (Feng et al., 2017).
NLRP6 is highly expressed in liver, lung, kidney, and small and large intestine (Chen et al., 2011; Elinav et al., 2011; Normand et al., 2011). Though the nature of the NLRP6 inflammasome and its agonist remain unclear, NLRP6 has been shown to recognize bacterial metabolites, bacterial lipoteichoic acid (LTA), and viral RNA (Hara et al., 2018; Levy et al., 2015). Recently it was found that LPS and ATP stimulation can activate NLRP6, causing it to form a homodimer and self-assemble into a linear platform (Leng et al., 2020). LTA binding activates NLRP6, leading to recruitment and processing of caspase-11 using ASC as an adaptor (Hara et al., 2018). Using filamentous and crystal structures, it was recently discovered that pyrin filaments from NLRP6 are critical for recruiting the PYD of ASC (Shen et al., 2019). It is thought that NLRP6 activation then leads to the polymerization and activation of ASC, which acts as a platform for the recruitment and activation of caspase-1 (Shen et al., 2019).
NLRP6 is also known to play a role in regulating TLR-induced canonical NFκB and MAPK signaling. The phosphorylation of extracellular signal-regulated kinases 1 and 2 (ERK1/2) and IκB is significantly increased in Nlrp6−/− macrophages upon infection with Listeria monocytogenes (Anand et al., 2012). Furthermore, levels of NFκB and MAPK-dependent cytokines and chemokines such as TNF, IL-6, and KC are significantly increased, NFκB effector p105 is highly phosphorylated, and translocation of p65 to the nucleus is increased in Nlrp6−/− macrophages upon infection with L. monocytogenes (Anand et al., 2012). During Staphylococcus aureus infection, NLRP6 serves as a negative regulator of neutrophil-mediated host defense (Ghimire et al., 2018).
3. NLRP12 as a negative regulator
NLRP12 (also known as NALP12, MONARCH-1, and PYPAF-7) is an intracellular protein is expressed in bone marrow neutrophils, granulocytes, macrophages and dendritic cells (Williams et al., 2003). The role of NLRP12 remains controversial, as it has been shown to function as both a negative regulator of inflammation (Table 1) and as an inflammasome (Table 2) (Pinheiro et al., 2011; Tuncer et al., 2014; Wang et al., 2002).
Table 1:
Negative regulatory role of NLRP12
| Model | NLRP12 as a negative regulator of inflammation | ||
|---|---|---|---|
| In vitro | In vivo | Reference | |
| Salmonella infection | • Increased expression of IL-6, KC, and TNF mRNA transcripts and proteins in murine Nlrp12−/− bone marrow-derived macrophages (BMDMs) infected with Salmonella. • Increased phosphorylation of IκBα and ERK in murine Nlrp12−/− BMDMs infected with Salmonella. |
• Increased phosphorylation of IκBα and ERK in the liver of Nlrp12−/− mice infected with Salmonella. |
Zaki et al; 2014 |
| DSS | NA | • Increased IL-6 and TNF levels in CD11b+ colonic lamina propria myeloid cells obtained from AOM/DSS-treated Nlrp12−/− mice. • Increased production of proinflammatory cytokines such as IL-1β, IL-6, TNF, IL-17, and IL-15 and chemokines such as GCSF, eotaxin, KC, IP-10, MIP-1α, MIP-1β, and MIP2 in the colon of AOM/DSS-treated Nlrp12−/− mice. • Increased IκBα ERK, and STAT3 activation in Nlrp12−/− mice treated with AOM/DSS. • Increased NIK activation in the colon of Nlrp12−/− mice treated with AOM/DSS. • Nlrp12−/− mice are more susceptible to DSS-induced colitis than WT mice. Nlrp12−/− mice raised under GF conditions are indistinguishable from GF WT mice during experimental colitis. |
Zaki et al; 2012 Allen et al; 2012 Chen et al; 2017 |
| HFD-induced obesity | NA | • Nlrp12−/− mice gained weight. • Impaired glucose tolerance and elevated levels of adipose tissue inflammation Nlrp12−/− mice. |
Truax et al; 2018 |
| DEN-induced HCC | NA | • Increased tumor burden and tumor size in livers of DEN-treated Nlrp12−/− mice. • Increased expression of proinflammatory cytokines such as IL-6, and TNF, and chemokines such as KC, MIP2, MCP1, and COX2 in the liver of DEN-treated Nlrp12−/− mice. • Increased production of proinflammatory cytokines such as IL-6, and TNF, and the chemokine KC in the liver of DEN-treated Nlrp12−/− mice. • Increased JNK activation in the liver of DEN-treated Nlrp12−/− mice. • Increased expression of pro-proliferative molecules such as Myc, Ccnd1, Ccnb1, Survivin, and MKi67 in the liver of DEN-treated Nlrp12−/− mice. • Increased expression of pro-proliferative molecules such as Myc, Ccnd1, Ccnb1, Survivin, and MKi67 in the liver of DEN-treated Nlrp12−/− mice. |
Udden et al; 2019 |
| TNF | • Increased activation of NIK in murine Nlrp12−/− dendritic cells stimulated with TNF. | NA | Allen et al; 2012 |
| TLR ligands | • Increased activation of IκBα and ERK in murine Nlrp12−/− dendritic cells stimulated with Pam3Cys4. • Increased activation of IκBα and ERK in murine Nlrp12−/− BMDMs stimulated with LPS, Pam3Cys4, and poly(I:C) in Nlrp12−/− BMDMs. • Increased mRNA expression of IL-6, KC, TNF, MIP2, COX2, and NOS2 transcripts in murine Nlrp12−/− BMDMs stimulated with LPS. • Increased activation of JNK in murine Nlrp12−/− hepatocytes stimulated with LPS. • Increased expression of Ccl2, Cxcl1, Cxcl2, Ccnd1 and Myc in murine Nlrp12−/− hepatocytes stimulated with LPS. |
• Decreased neutrophil recruitment to the site of inflammation (lungs) in Nlrp12−/− mice challenged with LPS. • Decreased vascular leakage into the lungs of Nlrp12−/− mice challenged with LPS. |
Lich et al;2007
Allen et al; 2012 Udden et al; 2019 Ulland et al; 2016 |
| Leishmania major | • No difference in NFκB, and ERK activation in murine WT and Nlrp12−/− bone marrow neutrophils. • Decreased neutrophil migration toward the neutrophil chemokine CXCL1/KC and L. major by murine Nlrp12−/− bone marrow neutrophils. |
NA | Zamoshnikova et al; 2016 |
| VSV infection | • Increased production of TNF and IFN-β in murine Nlrp12−/− dendritic cells infected with VSV. • Increased activation of TBK1 and IRF3 in murine Nlrp12−/− dendritic cells infected with VSV. |
• Increased production of IFN-β in serum and cerebral spinal fluid of Nlrp12−/− mice infected with VSV. |
Chen et al; 2019 |
| Klebsiella pneumoniae infection | • Increased production of TNF and IL-6 in murine Nlrp12−/− dendritic cells infected with K. pneumoniae. | NA | Allen et al; 2013 |
| Mycobacterium tuberculosis infection | • Increased production of TNF and IL-6 in murine Nlrp12−/− dendritic cells infected with M. tuberculosis. | NA | Allen et al; 2013 |
| Brucella abortus infection | • Increased production of IL-12 in murine Nlrp12−/− BMDMs infected with B. abortus. • Increased activation of p65, JNK, and p38 in murine Nlrp12−/− BMDMs infected with B. abortus. |
• Increased circulating IL-1β and IFN-γ in Nlrp12−/− mice infected with B. abortus S2308. |
Silveira et al; 2017 |
| Porphyromonas gingivalis LPS | • Increased expression of iNOS in Nlrp12 silenced RAW264.7 murine macrophages stimulated with P. gingivalis LPS. • Increased production of TNF in Nlrp12 silenced RAW264.7 murine macrophages stimulated with P. gingivalis LPS. |
NA |
Pudla et al; 2019 |
| Francisella tularensis LVS infection | NA | • Decreased neutrophil recruitment at the site of infection in Nlrp12−/− mice. • Increased F. tularensis LVS burden in Nlrp12−/− mice. |
Ulland et al; 2016 |
| Staphylococcus aureus infection | NA | • Decreased neutrophil recruitment at the site of infection in Nlrp12−/− mice. • Increased S. aureus burden in Nlrp12−/− mice. |
Ulland et al; 2016 |
| Pseudomonas aeruginosa | NA | • Decreased neutrophil recruitment at the site of infection in Nlrp12−/− mice. • Increased P. aeruginosa burden in Nlrp12−/− mice. |
Ulland et al; 2016 |
| Influenza A virus (IAV) | NA | • Decreased neutrophil chemoattractant CXCL1 in Nlrp12−/− mice infected with IAV. • Decreased pulmonary neutrophils in Nlrp12−/− mice infected with IAV. • Decreased neutrophil recruitment at the site of infection in Nlrp12−/− mice in response to IAV • Decreased vascular permeability in Nlrp12−/− mice during IAV infection. |
Hornick et al; 2018 |
| Experimental autoimmune encephalomyelitis (EAE) |
NA | • Nlrp12−/− mice have hyperinflammatory myelin-specific T-cell responses. • Nlrp12−/− T cells produce more IFN-γ and IL-17 per cell. • Nlrp12−/− T cells produce large amounts of Th2-associated cytokines like IL-4, IL-5, and IL-13, which ultimately contributes to the development of intestinal inflammation. • Enhanced phosphorylation of IκBα and processing of the NFκ p100 to p52 in Nlrp12−/− T cells. |
Lukens et al., 2015 |
Table 2:
NLRP12 as an inflammasome
| Triggers | Inflammasome activation | ||
|---|---|---|---|
| In vitro | In vivo | Reference | |
|
Yersinia pestis
Y. enterocolitica Y. pseudotuberculosis |
• Reduced IL-1β and IL-18 production in murine Nlrp12−/− peritoneal macrophages and BMDMs upon infection with virulent and attenuated Y. pestis strains. • Decreased caspase-1 and IL-1β cleavage in murine Nlrp12−/− BMDMs upon infection with attenuated Y. pestis. • Reduced IL-1β production in murine Nlrp12−/− BMDMs upon infection with Y. enterocolitica and Y. pseudotuberculosis. |
• Decreased expression of active caspase-1 in bone marrow cells, and Ly6G- or F4/80positive splenocytes obtained from Nlrp12−/− mice infected with virulent and attenuated Y. pestis strains. • Decreased levels of IL-1β and IL-18 in serum and spleen of Nlrp12−/− mice infected with virulent Y. pestis. • Decreased survival and increased bacterial load in the spleen of Nlrp12−/− mice upon infection with virulent and attenuated strains of Y. pestis strains. |
Valdimer et al; 2012 |
| Plasmodium chabaudi | NA | • Decreased expression of active caspase-1 and cell death in splenic macrophages and neutrophils obtained from Nlrp12−/− mice infected with P. chabaudi. • Decreased caspase-1 cleavage in splenocytes of Nlrp12−/− mice infected with P. chabaudi. • Decreased serum IL-1β levels in Nlrp12−/− mice infected with P. chabaudi followed by LPS challenge. • Decreased expression of active caspase-1 in splenocytes and decreased IL-1β levels in serum of Nlrp12−/− mice infected with parasitized red blood cells followed by LPS challenge. • Reduced mortality in Nlrp12−/− mice infected with parasitized red blood cells followed by LPS challenge. |
Ataide et al; 2014 |
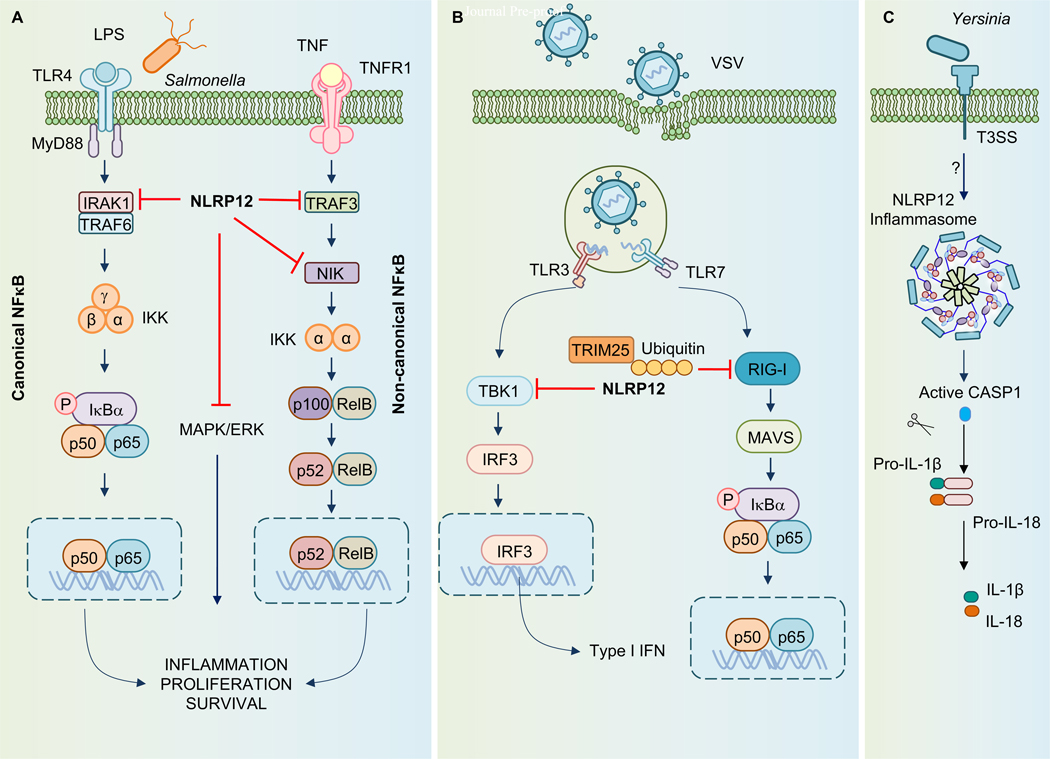
Studies have shown that NLRP12 functions as a negative regulator of both canonical and non-canonical NFκB signaling pathways (Figure 2A). Attenuation of canonical NFκB signaling occurs when NLRP12 interacts with and inhibits the accumulation of hyperphosphorylated IRAK1, downstream of TLR signaling. Additionally, an association of NLRP12 with NFκB-inducing kinase (NIK) leads to rapid proteasomal degradation of NIK in non-canonical NFκB signaling (Lich et al., 2007; Williams et al., 2005). NLRP12 has also been shown to interact with TRAF3, which is involved in NIK degradation (Allen et al., 2012). Physiologically, the inhibition of NFκB signaling by NLRP12 suppresses colon inflammation (Allen et al., 2012; Zaki et al., 2011). During azoxymethane (AOM)/dextran sodium sulfate (DSS)-induced colitis, Nlrp12−/− mice are highly susceptible to inflammation in the colon and show significantly increased levels of proinflammatory cytokines and chemokines due to the activation of canonical NFκB and ERK signaling pathways (Zaki et al., 2011). In contrast, another study showed that the susceptibility of Nlrp12−/− mice to colitis and colitis-associated colon cancer is caused by the activation of non-canonical NFκB, ERK, and AKT signaling pathways. The discrepancy between studies could be attributed to differences in the NFκB complex components and cell types analyzed, the stimuli used, and the mouse colony and housing conditions. Despite the discrepancy, both studies suggest that NLRP12 serves as a pivotal checkpoint for NFκB signaling.
Figure 2:
NLRP12 acts in innate immune signaling in a pathogen-specific manner. (A) NLRP12 negatively regulates inflammatory signaling by suppressing canonical and non-canonical NFκB signaling and the MAPK/ERK signaling pathway in bone marrow-derived macrophages (BMDMs)002E (B) NLRP12 associates with TRIM25 to reduce polyubiquitination and inhibit the RIG-I-mediated IFN response during VSV infection. (C) The NLRP12 inflammasome can drive caspase-1 activation and IL-1β and IL-18 release in BMDMs during infection with Yersinia pestis and Plasmodium chabaudi.
Additionally, NLRP12 plays a critical role in regulating the gut microbiota. While Nlrp12−/− mice are more susceptible to DSS-induced colitis than WT mice are, Nlrp12−/− mice raised under germ-free (GF) conditions are indistinguishable from GF WT mice during experimental colitis, indicating that the interaction between host cells and resident bacteria are important for the susceptibility of Nlrp12−/− mice to colitis (Chen et al., 2017). Overall this study found that NLRP12 functions during inflammatory bowel disease (IBD) to regulate the excessive production of inflammatory cytokines and maintain the commensal diversity of the gut microbiota to limit intestinal inflammation (Chen et al., 2017).
The colonic inflammation and microbiome dysbiosis that occur as a result of the loss of NLRP12 can cause obesity. Truax et al. evaluated the role of NLRP12 in high fat diet (HFD)-induced obesity and showed that Nlrp12−/− mice gained weight and had impaired glucose tolerance and elevated levels of adipose tissue inflammation compared with WT mice (Truax et al., 2018). By using antibiotics to treat Nlrp12−/− mice and cohousing WT and Nlrp12−/− mice, the authors also showed that HFD-induced weight gain in Nlrp12−/− mice was due to the gut microbiota (Truax et al., 2018). Together, these data strongly suggest that the microbiota contribute to inflammation and obesity in Nlrp12−/− mice and that NLRP12 restricts HFD-induced obesity and maintains the gut microbiota that dampens the inflammatory response.
The loss of NLRP12 has also been associated with increased susceptibility to hepatocellular carcinoma (HCC). In the mouse model of HCC induced by single administration of diethylnitrosamine (DEN), Nlrp12−/− mice had higher tumor burden and increased tumor size due to an increase in the expression of pro-proliferative molecules like Myc, Ccnd1, Ccnb1, MKi67, and Survivin (Udden et al., 2019). During DEN-induced HCC, Nlrp12−/− mice show significantly increased levels of proinflammatory cytokines and chemokines, and activation of the JNK signaling pathways, suggesting that NLRP12 also regulates the JNK signaling pathway. Also, Nlrp12−/− hepatocytes stimulated with LPS show increased activation of JNK and expression of Ccl2, Cxcl1, Cxcl2, Ccnd1, and Myc (Udden et al., 2019).
Furthermore, NLRP12 has also been implicated in the immune response to various pathogens. During infection with Mycobacterium tuberculosis or Klebsiella pneumonia, bone marrow-derived dendritic cells (BMDCs) lacking NLRP12 induce production of TNF and IL-6 but do not confer inflammasome-mediated protection against these bacteria (Allen et al., 2013). During Salmonella infection, NLRP12 mediates NFκB and ERK inhibition by targeting the phosphorylation of IκBα and ERK, which results in reduced production of proinflammatory cytokines (Zaki et al., 2014). Though the activator of NLRP12 is currently not known, Salmonella LPS alone can induce NLRP12-mediated inhibition of NFκB activation (Zaki et al., 2014). This suggests that the activator of NLRP12 could be upregulated upon LPS stimulation. Also, NLRP12 regulates NFκB and MAPK signaling by inhibiting the phosphorylation of p65, JNK, and p38 in response to Brucella abortus infection (Silveira et al., 2017). When RAW264.7 macrophages that have NLRP12 silenced are stimulated with Porphyromonas gingivalis LPS leads to increased expression of iNOS and higher production of TNF, suggesting that NLRP12 negatively regulates iNOS and TNF during bacterial infection (Pudla et al., 2019). During viral infection, TRIM25 is required for Lys63-linked polyubiquitination and activation of RIG-I, which then activates the mitochondrial antiviral signaling adaptor MAVS. MAVS associates with the adaptor protein TRAF3 and TRAF family member-associated NFκB activator (TANK) to trigger the activation of TANK-binding kinase 1 (TBK1) and IκB kinase, leading to the activation of IRFs and IFN. During vesicular stomatits virus (VSV) infection, NLRP12 interacts with the E3 ubiquitin ligase TRIM25 to disrupt its association with RIG-I, resulting in reduced type I IFN production (Figure 2B) (Chen et al., 2019).
Along with regulating the canonical and non-canonical NFκB pathways, NLRP12 can also impact immune cell localization and recruitment. NLRP12 negatively regulates neutrophil migration and neutrophil recruitment to the site of infection during infection with viral, bacterial, and parasitic pathogens such as influenza A virus (IAV), Francisella tularensis LVS, P. aeruginosa, and S. aureus, and Leishmania major (Hornick et al., 2018; Ulland et al., 2016; Zamoshnikova et al., 2016). Due to the lack of neutrophil recruitment to the site of infection Nlrp12−/− mice were unable to control replication of various bacterial and viral pathogens (Ulland et al., 2016; Zamoshnikova et al., 2016). CXCL1 and CXCL2 expression in perivascular macrophages in response to various stimuli triggers both the adhesion and transmigration of neutrophils out of the blood stream. In the absence of NLRP12, the macrophages are defective in producing the chemoattractant chemokine CXCL1 which results in defective neutrophil recruitment in response to a variety of pathogens and TLR agonist (Ulland et al., 2016). During IAV infection, Hornick et al. showed that NLRP12 contributes to the severity of disease through its effect on CXCL1 production, which ultimately affects neutrophil recruitment and vascular permeability. Though it is not clear how NLRP12 regulates CXCL1 expression, Ulland et al. showed that failure to recruit neutrophils in response to a range of inflammatory stimuli is associated with a missense mutation in Nlrp12 in C57BL/6J mice (Ulland et al., 2016).
NLRP12 has also been shown to negatively regulate T cells in responses to experimental autoimmune encephalomyelitis (EAE) (Gurung and Kanneganti, 2015; Lukens et al., 2015). Nlrp12−/− mice have hyperinflammatory myelin-specific T-cell responses that result in exacerbated demyelinating disease. Using adoptive transfer studies, Lukens et al., showed that NLRP12 is an intrinsic negative regulator of T-cell responses; Nlrp12−/− T cells produced more IFN-γ and IL-17 per cell than do WT T-cells. Furthermore, Nlrp12−/− T cells were shown to produce large amounts of Th2-associated cytokines like IL-4, IL-5, and IL-13, which ultimately contributes to the development of intestinal inflammation and atopic dermatitis in Nlrp12−/− mice. Additionally, adoptive transfer of Nlrp12−/− T cells causes enhanced phosphorylation of IκBα and processing of the NFκ p100 to p52, showing that NLRP12 negatively regulates canonical and non-canonical NFκB signaling in T-cells (Lukens et al., 2015).
Overall, regulation of the immune response by NLRP12 can be multifaceted and pathogen-specific. Regulation of the NFκB signaling pathway by NLRP12 upon infection with various pathogens could contribute to pathogen survival and persistence in the host. Also, NLRP12 plays an important role in gut homeostasis, where it serves as a negative regulator that inhibits proinflammatory cytokines and chemokines (Chen et al., 2017; Lau and Dombrowski, 2018; Truax et al., 2018). These regulatory functions are important for the host, as NLRP12 influences key pathways to prevent overt damage to the tissues.
4. NLRP12 as an inflammasome
The role of NLRP12 as a negative regulator of immune signaling has been established during infection and colon cancer, IBD, and tumorigenesis. There are limited studies that have elucidated the role of NLRP12 as a component of the inflammasome (Table 2). During Yersinia pestis infection, NLRP12 has been shown to be an inflammasome component that becomes activated through an unknown mechanism and mediates Y. pestis-dependent release of IL-1β and IL-18 (Figure 2C). Caspase-1 activation and IL-1β and IL-18 release are decreased in the spleen and bone marrow of Nlrp12−/− mice upon infection with Yersinia. Additionally, Nlrp12−/− mice succumb to Yersinia infection, while WT mice do not (Vladimer et al., 2012). Although the ligand that activates NLRP12 is not known, this study showed that the generation of ligands for NLRP12 activation during Yersinia infection requires the virulence-associated T3SS. This further suggests that activation of NLRP12 may be associated with the sensing of damage that is associated with the T3SS, the effector proteins and other molecules that are secreted and channeled by the T3SS, or the host proteins that are modified. In the context of a parasitic infection, macrophages and dendritic cells in the spleen activate caspase-1 upon infection with Plasmodium chabaudi. This caspase-1 activation is ASC-dependent. During P. chabaudi infection, both NLRP3 and NLRP12 are required for caspase-1 activation, the systemic production of IL-1β, and pyroptosis, as Nlrp12−/− mice have decreased caspase-1 activation and IL-1β release upon Plasmodium infection (Ataide et al., 2014). While Nlrp12−/− mice infected with Yersinia or Plasmodium exhibit decreased caspase-1 activation and IL-1β release, infection with B. abortus results in increased IL-1β circulation in Nlrp12−/− mice (Silveira et al., 2017), complicating the potential role of NLRP12 as an inflammasome component.
Although NLRP12 can recognize Yersinia infection and contributes to in vivo resistance, Nlrp12−/− mice infected with Yersinia do not completely lose their ability to release IL-1β and IL-18. Additionally, both Yersinia and Plasmodium infections also require NLRP3 for host defense. Thus, this suggests that redundancy in the activation of various NLRs contributes to optimal protection in the host.
6. Summary and Future Directions
NLRP12 plays a multifaceted role in innate immunity, and its functions in inflammasome formation and as a negative regulator are context-dependent. During Yersinia and Plasmodium infection, NLRP12 activation leads to caspase-1 activation and the release of IL-1β and IL-18, suggesting it can contribute to inflammasome formation and proinflammatory signaling. Conversely, during experimental colitis, NLRP12 regulates canonical and non-canonical NFκB signaling pathways, suggesting that it has anti-inflammatory and anti-tumorigenic functions. NLRP12 has also been shown to play a role in gut homeostasis and maintaining the microbiota in the gut. Furthermore, NLRP12 regulates T cells and neutrophil migration. Lack of NLRP12 in T cells can cause T cells to become hyper-inflammatory, resulting in the development of autoimmune diseases.
Detailed studies of the structural components and assembly of inflammasomes and mechanisms of pyroptosis have provided tremendous advancement in our understanding of inflammasome formation, activation, and function. Though the role of NLRP12 in resisting microbial infections has been shown, it is still not clear how NLRP12 interacts with other components of the inflammasome or what molecules directly trigger NLRP12 inflammasome assembly. Further studies are required to fully understand what conditions cause NLRP12 to act as a negative regulator or as an inflammasome.
Overall, the conflicting roles of NLRP12 as a negative regulator or an inflammasome are dependent on the cell type analyzed and, most importantly are pathogen-specific. Based on its critical function in disease, NLRP12 may serve as therapeutic target to control infections and for diseases such as colitis and IBD.
Acknowledgements
Research studies from our laboratory are supported by the U.S. National Institutes of Health (AR056296, CA163507, AI124346, and AI101935 to T.-D.K.), and the American Lebanese Syrian Associated Charities (to T.-D.K.). We apologize to our colleagues whose work was not cited due to space limitations.
Footnotes
Conflicts of Interest
The authors have no conflicts of interest to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Abbott DW, Wilkins A, Asara JM, Cantley LC, 2004. The Crohn’s disease protein, NOD2, requires RIP2 in order to induce ubiquitinylation of a novel site on NEMO. Curr. Biol. CB 14, 2217–2227. 10.1016/j.cub.2004.12.032 [DOI] [PubMed] [Google Scholar]
- Aikawa C, Nakajima S, Karimine M, Nozawa T, Minowa-Nozawa A, Toh H, Yamada S, Nakagawa I, 2018. NLRX1 Negatively Regulates Group A Streptococcus Invasion and Autophagy Induction by Interacting With the Beclin 1-UVRAG Complex. Front. Cell. Infect. Microbiol. 8, 403. 10.3389/fcimb.2018.00403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen IC, McElvania-TeKippe E, Wilson JE, Lich JD, Arthur JC, Sullivan JT, Braunstein M, Ting JPY, 2013. Characterization of NLRP12 during the in vivo host immune response to Klebsiella pneumoniae and Mycobacterium tuberculosis. PloS One 8, e60842. 10.1371/journal.pone.0060842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen IC, Moore CB, Schneider M, Lei Y, Davis BK, Scull MA, Gris D, Roney KE, Zimmermann AG, Bowzard JB, Ranjan P, Monroe KM, Pickles RJ, Sambhara S, Ting JPY, 2011. NLRX1 protein attenuates inflammatory responses to infection by interfering with the RIG-I-MAVS and TRAF6-NF-κB signaling pathways. Immunity 34, 854–865. 10.1016/j.immuni.2011.03.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen IC, Wilson JE, Schneider M, Lich JD, Roberts RA, Arthur JC, Woodford R-MT, Davis BK, Uronis JM, Herfarth HH, Jobin C, Rogers AB, Ting JP-Y, 2012. NLRP12 suppresses colon inflammation and tumorigenesis through the negative regulation of noncanonical NF-κB signaling. Immunity 36, 742–754. 10.1016/j.immuni.2012.03.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anand PK, Malireddi RKS, Lukens JR, Vogel P, Bertin J, Lamkanfi M, Kanneganti T-D, 2012. NLRP6 negatively regulates innate immunity and host defence against bacterial pathogens. Nature 488, 389–393. 10.1038/nature11250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ataide MA, Andrade WA, Zamboni DS, Wang D, Souza M. do C., Franklin BS, Elian S, Martins FS, Pereira D, Reed G, Fitzgerald KA, Golenbock DT, Gazzinelli RT, 2014. Malaria-Induced NLRP12/NLRP3-Dependent Caspase-1 Activation Mediates Inflammation and Hypersensitivity to Bacterial Superinfection. PLoS Pathog. 10. 10.1371/journal.ppat.1003885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauernfeind FG, Horvath G, Stutz A, Alnemri ES, MacDonald K, Speert D, Fernandes-Alnemri T, Wu J, Monks BG, Fitzgerald KA, Hornung V, Latz E, 2009. Cutting edge: NF-kappaB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J. Immunol. Baltim. Md 1950 183, 787–791. 10.4049/jimmunol.0901363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benko S, Magalhaes JG, Philpott DJ, Girardin SE, 2010. NLRC5 limits the activation of inflammatory pathways. J. Immunol. Baltim. Md 1950 185, 1681–1691. 10.4049/jimmunol.0903900 [DOI] [PubMed] [Google Scholar]
- Boyden ED, Dietrich WF, 2006. Nalp1b controls mouse macrophage susceptibility to anthrax lethal toxin. Nat. Genet. 38, 240–244. 10.1038/ng1724 [DOI] [PubMed] [Google Scholar]
- Broz P, Dixit VM, 2016. Inflammasomes: mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 16, 407–420. 10.1038/nri.2016.58 [DOI] [PubMed] [Google Scholar]
- Broz P, von Moltke J, Jones JW, Vance RE, Monack DM, 2010. Differential requirement for Caspase-1 autoproteolysis in pathogen-induced cell death and cytokine processing. Cell Host Microbe 8, 471–483. 10.1016/j.chom.2010.11.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carneiro L. a. M., Magalhaes JG, Tattoli I, Philpott DJ, Travassos LH, 2008. Nod-like proteins in inflammation and disease. J. Pathol. 214, 136–148. 10.1002/path.2271 [DOI] [PubMed] [Google Scholar]
- Chamaillard M, Hashimoto M, Horie Y, Masumoto J, Qiu S, Saab L, Ogura Y, Kawasaki A, Fukase K, Kusumoto S, Valvano MA, Foster SJ, Mak TW, Nuñez G, Inohara N, 2003. An essential role for NOD1 in host recognition of bacterial peptidoglycan containing diaminopimelic acid. Nat. Immunol. 4, 702–707. 10.1038/ni945 [DOI] [PubMed] [Google Scholar]
- Chavarría-Smith J, Mitchell PS, Ho AM, Daugherty MD, Vance RE, 2016. Functional and Evolutionary Analyses Identify Proteolysis as a General Mechanism for NLRP1 Inflammasome Activation. PLoS Pathog. 12, e1006052. 10.1371/journal.ppat.1006052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chavarría-Smith J, Vance RE, 2013. Direct proteolytic cleavage of NLRP1B is necessary and sufficient for inflammasome activation by anthrax lethal factor. PLoS Pathog. 9, e1003452. 10.1371/journal.ppat.1003452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen GY, Liu M, Wang F, Bertin J, Núñez G, 2011. A Functional Role for Nlrp6 in Intestinal Inflammation and Tumorigenesis. J. Immunol. 186, 7187–7194. 10.4049/jimmunol.1100412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L, Wilson JE, Koenigsknecht MJ, Chou W-C, Montgomery SA, Truax AD, Brickey WJ, Packey CD, Maharshak N, Matsushima GK, Plevy SE, Young VB, Sartor RB, Ting JP-Y, 2017. NLRP12 attenuates colon inflammation by maintaining colonic microbial diversity and promoting protective commensal bacterial growth. Nat. Immunol. 18, 541–551. 10.1038/ni.3690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S-T, Chen L, Lin DS-C, Chen S-Y, Tsao Y-P, Guo H, Li F-J, Tseng W-T, Tam JW, Chao C-W, Brickey WJ, Dzhagalov I, Song M-J, Kang H-R, Jung JU, Ting JP-Y, 2019. NLRP12 Regulates Anti-viral RIG-I Activation via Interaction with TRIM25. Cell Host Microbe 25, 602–616.e7. 10.1016/j.chom.2019.02.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chui AJ, Okondo MC, Rao SD, Gai K, Griswold AR, Johnson DC, Ball DP, Taabazuing CY, Orth EL, Vittimberga BA, Bachovchin DA, 2019. N-terminal degradation activates the NLRP1B inflammasome. Science 364, 82–85. 10.1126/science.aau1208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clay GM, Sutterwala FS, Wilson ME, 2014. NLR proteins and parasitic disease. Immunol. Res. 59, 142–152. 10.1007/s12026-014-8544-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cridland JA, Curley EZ, Wykes MN, Schroder K, Sweet MJ, Roberts TL, Ragan MA, Kassahn KS, Stacey KJ, 2012. The mammalian PYHIN gene family: phylogeny, evolution and expression. BMC Evol. Biol. 12, 140. 10.1186/1471-2148-12-140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui J, Zhu L, Xia X, Wang HY, Legras X, Hong J, Ji J, Shen P, Zheng S, Chen ZJ, Wang R-F, 2010. NLRC5 negatively regulates the NF-kappaB and type I interferon signaling pathways. Cell 141, 483–496. 10.1016/j.cell.2010.03.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis BK, Roberts RA, Huang MT, Willingham SB, Conti BJ, Brickey WJ, Barker BR, Kwan M, Taxman DJ, Accavitti-Loper M-A, Duncan JA, Ting JP-Y, 2011. Cutting edge: NLRC5-dependent activation of the inflammasome. J. Immunol. Baltim. Md 1950 186, 1333–1337. 10.4049/jimmunol.1003111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding J, Wang K, Liu W, She Y, Sun Q, Shi J, Sun H, Wang D-C, Shao F, 2016. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 535, 111–116. 10.1038/nature18590 [DOI] [PubMed] [Google Scholar]
- Dowds TA, Masumoto J, Chen FF, Ogura Y, Inohara N, Núñez G, 2003. Regulation of cryopyrin/Pypaf1 signaling by pyrin, the familial Mediterranean fever gene product. Biochem. Biophys. Res. Commun. 302, 575–580. 10.1016/s0006-291x(03)00221-3 [DOI] [PubMed] [Google Scholar]
- Eldeeb MA, Fahlman RP, Esmaili M, Fon EA, 2019. When Degradation Elicits the Alarm: N-Terminal Degradation of NLRP1B Unleashes Its Inflammasome Activity. Mol. Cell 74, 637–639. 10.1016/j.molcel.2019.04.032 [DOI] [PubMed] [Google Scholar]
- Elinav E, Strowig T, Kau AL, Henao-Mejia J, Thaiss CA, Booth CJ, Peaper DR, Bertin J, Eisenbarth SC, Gordon JI, Flavell RA, 2011. NLRP6 Inflammasome Regulates Colonic Microbial Ecology and Risk for Colitis. Cell 145, 745–757. 10.1016/j.cell.2011.04.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng H, Lenarcic EM, Yamane D, Wauthier E, Mo J, Guo H, McGivern DR, González-López O, Misumi I, Reid LM, Whitmire JK, Ting JP-Y, Duncan JA, Moorman NJ, Lemon SM, 2017. NLRX1 promotes immediate IRF1-directed antiviral responses by limiting dsRNA-activated translational inhibition mediated by PKR. Nat. Immunol. 18, 1299–1309. 10.1038/ni.3853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franchi L, Eigenbrod T, Núñez G, 2009a. Cutting Edge: TNF-α Mediates Sensitization to ATP and Silica via the NLRP3 Inflammasome in the Absence of Microbial Stimulation. J. Immunol. 183, 792–796. 10.4049/jimmunol.0900173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franchi L, Warner N, Viani K, Nuñez G, 2009b. Function of Nod-like receptors in microbial recognition and host defense. Immunol. Rev. 227, 106–128. 10.1111/j.1600-065X.2008.00734.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritz JH, Ferrero RL, Philpott DJ, Girardin SE, 2006. Nod-like proteins in immunity, inflammation and disease. Nat. Immunol. 7, 1250–1257. 10.1038/ni1412 [DOI] [PubMed] [Google Scholar]
- Ghimire L, Paudel S, Jin L, Baral P, Cai S, Jeyaseelan S, 2018. NLRP6 negatively regulates pulmonary host defense in Gram-positive bacterial infection through modulating neutrophil recruitment and function. PLoS Pathog. 14, e1007308. 10.1371/journal.ppat.1007308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Girardin SE, Tournebize R, Mavris M, Page AL, Li X, Stark GR, Bertin J, DiStefano PS, Yaniv M, Sansonetti PJ, Philpott DJ, 2001. CARD4/Nod1 mediates NF-kappaB and JNK activation by invasive Shigella flexneri. EMBO Rep. 2, 736–742. 10.1093/embo-reports/kve155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurung P, Kanneganti T-D, 2015. NLRP12 in autoimmune diseases. Oncotarget 6, 19950–19951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurung P, Anand PK, Malireddi RKS, Vande Walle L, Van Opdenbosch N, Dillon CP, Weinlich R, Green DR, Lamkanfi M, Kanneganti T-D, 2014. FADD and caspase-8 mediate priming and activation of the canonical and noncanonical Nlrp3 inflammasomes. J. Immunol. Baltim. Md 1950 192, 1835–1846. 10.4049/jimmunol.1302839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurung P, Burton A, Kanneganti T-D, 2016. NLRP3 inflammasome plays a redundant role with caspase 8 to promote IL-1β–mediated osteomyelitis. Proc. Natl. Acad. Sci. 113, 4452–4457. 10.1073/pnas.1601636113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurung P, Malireddi RKS, Anand PK, Demon D, Vande Walle L, Liu Z, Vogel P, Lamkanfi M, Kanneganti T-D, 2012. Toll or interleukin-1 receptor (TIR) domain-containing adaptor inducing interferon-β (TRIF)-mediated caspase-11 protease production integrates Toll-like receptor 4 (TLR4) protein- and Nlrp3 inflammasome-mediated host defense against enteropathogens. J. Biol. Chem. 287, 34474–34483. 10.1074/jbc.M112.401406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara H, Seregin SS, Yang D, Fukase K, Chamaillard M, Alnemri ES, Inohara N, Chen GY, Núñez G, 2018. The NLRP6 Inflammasome Recognizes Lipoteichoic Acid and Regulates Gram-Positive Pathogen Infection. Cell 175, 1651–1664.e14. 10.1016/j.cell.2018.09.047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harton JA, Linhoff MW, Zhang J, Ting JP-Y, 2002. Cutting edge: CATERPILLER: a large family of mammalian genes containing CARD, pyrin, nucleotide-binding, and leucine-rich repeat domains. J. Immunol. Baltim. Md 1950 169, 4088–4093. 10.4049/jimmunol.169.8.4088 [DOI] [PubMed] [Google Scholar]
- Hasegawa M, Fujimoto Y, Lucas PC, Nakano H, Fukase K, Núñez G, Inohara N, 2008. A critical role of RICK/RIP2 polyubiquitination in Nod-induced NF-kappaB activation. EMBO J. 27, 373–383. 10.1038/sj.emboj.7601962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- He W, Wan H, Hu L, Chen P, Wang X, Huang Z, Yang Z-H, Zhong C-Q, Han J, 2015. Gasdermin D is an executor of pyroptosis and required for interleukin-1β secretion. Cell Res. 25, 1285–1298. 10.1038/cr.2015.139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hornick EE, Banoth B, Miller AM, Zacharias ZR, Jain N, Wilson ME, Gibson-Corley KN, Legge KL, Bishop GA, Sutterwala FS, Cassel SL, 2018. Nlrp12 Mediates Adverse Neutrophil Recruitment during Influenza Virus Infection. J. Immunol. Baltim. Md 1950 200, 1188–1197. 10.4049/jimmunol.1700999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu S, Du X, Huang Y, Fu Y, Yang Y, Zhan X, He W, Wen Q, Zhou X, Zhou C, Zhong X-P, Yang J, Xiong W, Wang R, Gao Y, Ma L, 2018. NLRC3 negatively regulates CD4+ T cells and impacts protective immunity during Mycobacterium tuberculosis infection. PLoS Pathog. 14, e1007266. 10.1371/journal.ppat.1007266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inohara N, Nuñez G, 2003. NODs: intracellular proteins involved in inflammation and apoptosis. Nat. Rev. Immunol. 3, 371–382. 10.1038/nri1086 [DOI] [PubMed] [Google Scholar]
- Kanneganti T-D, Lamkanfi M, Núñez G, 2007. Intracellular NOD-like receptors in host defense and disease. Immunity 27, 549–559. 10.1016/j.immuni.2007.10.002 [DOI] [PubMed] [Google Scholar]
- Kanneganti T-D, Ozören N, Body-Malapel M, Amer A, Park J-H, Franchi L, Whitfield J, Barchet W, Colonna M, Vandenabeele P, Bertin J, Coyle A, Grant EP, Akira S, Núñez G, 2006. Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature 440, 233–236. 10.1038/nature04517 [DOI] [PubMed] [Google Scholar]
- Karki R, Kanneganti T-D, 2019. Diverging inflammasome signals in tumorigenesis and potential targeting. Nat. Rev. Cancer. 10.1038/s41568-019-0123-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karki R, Lee E, Place D, Samir P, Mavuluri J, Sharma BR, Balakrishnan A, Malireddi RKS, Geiger R, Zhu Q, Neale G, Kanneganti T-D, 2018. IRF8 Regulates Transcription of Naips for NLRC4 Inflammasome Activation. Cell 173, 920–933.e13. 10.1016/j.cell.2018.02.055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karki R, Malireddi RKS, Zhu Q, Kanneganti T-D, 2017. NLRC3 regulates cellular proliferation and apoptosis to attenuate the development of colorectal cancer. Cell Cycle Georget. Tex 16, 1243–1251. 10.1080/15384101.2017.1317414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karki R, Man SM, Malireddi RKS, Kesavardhana S, Zhu Q, Burton AR, Sharma BR, Qi X, Pelletier S, Vogel P, Rosenstiel P, Kanneganti T-D, 2016. NLRC3 is an inhibitory sensor of PI3K-mTOR pathways in cancer. Nature 540, 583–587. 10.1038/nature20597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kesavardhana S, Kanneganti T-D, 2017. Mechanisms governing inflammasome activation, assembly and pyroptosis induction. Int. Immunol. 29, 201–210. 10.1093/intimm/dxx018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JG, Lee SJ, Kagnoff MF, 2004. Nod1 is an essential signal transducer in intestinal epithelial cells infected with bacteria that avoid recognition by toll-like receptors. Infect. Immun. 72, 1487–1495. 10.1128/iai.72.3.1487-1495.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J-Y, Omori E, Matsumoto K, Núñez G, Ninomiya-Tsuji J, 2008. TAK1 is a central mediator of NOD2 signaling in epidermal cells. J. Biol. Chem. 283, 137–144. 10.1074/jbc.M704746200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi KS, Chamaillard M, Ogura Y, Henegariu O, Inohara N, Nuñez G, Flavell RA, 2005. Nod2-dependent regulation of innate and adaptive immunity in the intestinal tract. Science 307, 731–734. 10.1126/science.1104911 [DOI] [PubMed] [Google Scholar]
- Kofoed EM, Vance RE, 2011. Innate immune recognition of bacterial ligands by NAIPs determines inflammasome specificity. Nature 477, 592–595. 10.1038/nature10394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuenzel S, Till A, Winkler M, Häsler R, Lipinski S, Jung S, Grötzinger J, Fickenscher H, Schreiber S, Rosenstiel P, 2010. The nucleotide-binding oligomerization domain-like receptor NLRC5 is involved in IFN-dependent antiviral immune responses. J. Immunol. Baltim. Md 1950 184, 1990–2000. 10.4049/jimmunol.0900557 [DOI] [PubMed] [Google Scholar]
- Kuriakose T, Man SM, Malireddi RKS, Karki R, Kesavardhana S, Place DE, Neale G, Vogel P, Kanneganti T-D, 2016. ZBP1/DAI is an innate sensor of influenza virus triggering the NLRP3 inflammasome and programmed cell death pathways. Sci. Immunol. 1, aag2045–aag2045. 10.1126/sciimmunol.aag2045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamkanfi M, Kanneganti T-D, 2012. Regulation of immune pathways by the NOD-like receptor NLRC5. Immunobiology 217, 13–16. 10.1016/j.imbio.2011.08.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamkanfi M, Kanneganti T-D, 2010. Nlrp3: an immune sensor of cellular stress and infection. Int. J. Biochem. Cell Biol. 42, 792–795. 10.1016/j.biocel.2010.01.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lau JM-F, Dombrowski Y, 2018. The innate immune receptor NLRP12 maintains intestinal homeostasis by regulating microbiome diversity. Cell. Mol. Immunol. 15, 193–195. 10.1038/cmi.2017.61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leng F, Yin H, Qin S, Zhang K, Guan Y, Fang R, Wang H, Li G, Jiang Z, Sun F, Wang D-C, Xie C, 2020. NLRP6 self-assembles into a linear molecular platform following LPS binding and ATP stimulation. Sci. Rep. 10, 198. 10.1038/s41598-019-57043-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levinsohn JL, Newman ZL, Hellmich KA, Fattah R, Getz MA, Liu S, Sastalla I, Leppla SH, Moayeri M, 2012. Anthrax lethal factor cleavage of Nlrp1 is required for activation of the inflammasome. PLoS Pathog. 8, e1002638. 10.1371/journal.ppat.1002638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy M, Thaiss CA, Zeevi D, Dohnalová L, Zilberman-Schapira G, Mahdi JA, David E, Savidor A, Korem T, Herzig Y, Pevsner-Fischer M, Shapiro H, Christ A, Harmelin A, Halpern Z, Latz E, Flavell RA, Amit I, Segal E, Elinav E, 2015. Microbiota-Modulated Metabolites Shape the Intestinal Microenvironment by Regulating NLRP6 Inflammasome Signaling. Cell 163, 1428–1443. 10.1016/j.cell.2015.10.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Deng M, Petrucelli AS, Zhu C, Mo J, Zhang L, Tam JW, Ariel P, Zhao B, Zhang S, Ke H, Li P, Dokholyan NV, Duncan JA, Ting JP-Y, 2019. Viral DNA Binding to NLRC3, an Inhibitory Nucleic Acid Sensor, Unleashes STING, a Cyclic Dinucleotide Receptor that Activates Type I Interferon. Immunity 50, 591–599.e6. 10.1016/j.immuni.2019.02.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lich JD, Williams KL, Moore CB, Arthur JC, Davis BK, Taxman DJ, Ting JP-Y, 2007. Cutting Edge: Monarch-1 Suppresses Non-Canonical NF-κB Activation and p52-Dependent Chemokine Expression in Monocytes. J. Immunol. 178, 1256–1260. 10.4049/jimmunol.178.3.1256 [DOI] [PubMed] [Google Scholar]
- Lightfield KL, Persson J, Brubaker SW, Witte CE, von Moltke J, Dunipace EA, Henry T, Sun Y-H, Cado D, Dietrich WF, Monack DM, Tsolis RM, Vance RE, 2008. Critical function for Naip5 in inflammasome activation by a conserved carboxy-terminal domain of flagellin. Nat. Immunol. 9, 1171–1178. 10.1038/ni.1646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Zhang Z, Ruan J, Pan Y, Magupalli VG, Wu H, Lieberman J, 2016. Inflammasome-activated gasdermin D causes pyroptosis by forming membrane pores. Nature 535, 153–158. 10.1038/nature18629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu A, Magupalli VG, Ruan J, Yin Q, Atianand MK, Vos MR, Schröder GF, Fitzgerald KA, Wu H, Egelman EH, 2014. Unified polymerization mechanism for the assembly of ASC-dependent inflammasomes. Cell 156, 1193–1206. 10.1016/j.cell.2014.02.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lukens JR, Gurung P, Shaw PJ, Barr MJ, Zaki Md.H., Brown SA, Vogel P, Chi H, Kanneganti T-D, 2015. The NLRP12 Sensor Negatively Regulates Autoinflammatory Disease by Modulating Interleukin-4 Production in T Cells. Immunity 42, 654–664. 10.1016/j.immuni.2015.03.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik A, Kanneganti T-D, 2017. Inflammasome activation and assembly at a glance. J. Cell Sci. 130, 3955–3963. 10.1242/jcs.207365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malireddi RKS, Gurung P, Mavuluri J, Dasari TK, Klco JM, Chi H, Kanneganti T-D, 2018. TAK1 restricts spontaneous NLRP3 activation and cell death to control myeloid proliferation. J. Exp. Med. 215, 1023–1034. 10.1084/jem.20171922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Man SM, Kanneganti T-D, 2015. Regulation of inflammasome activation. Immunol. Rev. 265, 6–21. 10.1111/imr.12296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Man SM, Karki R, Kanneganti T-D, 2017. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol. Rev. 277, 61–75. 10.1111/imr.12534 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinon F, Mayor A, Tschopp J, 2009. The inflammasomes: guardians of the body. Annu. Rev. Immunol. 27, 229–265. 10.1146/annurev.immunol.021908.132715 [DOI] [PubMed] [Google Scholar]
- Meissner TB, Li A, Biswas A, Lee K-H, Liu Y-J, Bayir E, Iliopoulos D, van den Elsen PJ, Kobayashi KS, 2010. NLR family member NLRC5 is a transcriptional regulator of MHC class I genes. Proc. Natl. Acad. Sci. U. S. A. 107, 13794–13799. 10.1073/pnas.1008684107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meylan E, Tschopp J, Karin M, 2006. Intracellular pattern recognition receptors in the host response. Nature 442, 39–44. 10.1038/nature04946 [DOI] [PubMed] [Google Scholar]
- Miao EA, Alpuche-Aranda CM, Dors M, Clark AE, Bader MW, Miller SI, Aderem A, 2006. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1beta via Ipaf. Nat. Immunol. 7, 569–575. 10.1038/ni1344 [DOI] [PubMed] [Google Scholar]
- Miao EA, Mao DP, Yudkovsky N, Bonneau R, Lorang CG, Warren SE, Leaf IA, Aderem A, 2010. Innate immune detection of the type III secretion apparatus through the NLRC4 inflammasome. Proc. Natl. Acad. Sci. 107, 3076–3080. 10.1073/pnas.0913087107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muñoz-Planillo R, Kuffa P, Martínez-Colón G, Smith BL, Rajendiran TM, Núñez G, 2013. K+ efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter. Immunity 38, 1142–1153. 10.1016/j.immuni.2013.05.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neerincx A, Lautz K, Menning M, Kremmer E, Zigrino P, Hösel M, Büning H, Schwarzenbacher R, Kufer TA, 2010. A role for the human nucleotide-binding domain, leucine-rich repeat-containing family member NLRC5 in antiviral responses. J. Biol. Chem. 285, 26223–26232. 10.1074/jbc.M110.109736 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newton K, Dixit VM, 2012. Signaling in innate immunity and inflammation. Cold Spring Harb. Perspect. Biol. 4. 10.1101/cshperspect.a006049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Normand S, Delanoye-Crespin A, Bressenot A, Huot L, Grandjean T, Peyrin-Biroulet L, Lemoine Y, Hot D, Chamaillard M, 2011. Nod-like receptor pyrin domain-containing protein 6 (NLRP6) controls epithelial self-renewal and colorectal carcinogenesis upon injury. Proc. Natl. Acad. Sci. 108, 9601–9606. 10.1073/pnas.1100981108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nour AM, Yeung Y-G, Santambrogio L, Boyden ED, Stanley ER, Brojatsch J, 2009. Anthrax Lethal Toxin Triggers the Formation of a Membrane-Associated Inflammasome Complex in Murine Macrophages. Infect. Immun. 77, 1262–1271. 10.1128/IAI.01032-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogura Y, Inohara N, Benito A, Chen FF, Yamaoka S, Nunez G, 2001. Nod2, a Nod1/Apaf-1 family member that is restricted to monocytes and activates NF-kappaB. J. Biol. Chem. 276, 4812–4818. 10.1074/jbc.M008072200 [DOI] [PubMed] [Google Scholar]
- Ogura Y, Lala S, Xin W, Smith E, Dowds TA, Chen FF, Zimmermann E, Tretiakova M, Cho JH, Hart J, Greenson JK, Keshav S, Nuñez G, 2003. Expression of NOD2 in Paneth cells: a possible link to Crohn’s ileitis. Gut 52, 1591–1597. 10.1136/gut.52.11.1591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Opitz B, Förster S, Hocke AC, Maass M, Schmeck B, Hippenstiel S, Suttorp N, Krüll M, 2005. Nod1-mediated endothelial cell activation by Chlamydophila pneumoniae. Circ. Res. 96, 319–326. 10.1161/01.RES.0000155721.83594.2c [DOI] [PubMed] [Google Scholar]
- Park J-H, Kim Y-G, McDonald C, Kanneganti T-D, Hasegawa M, Body-Malapel M, Inohara N, Núñez G, 2007. RICK/RIP2 mediates innate immune responses induced through Nod1 and Nod2 but not TLRs. J. Immunol. Baltim. Md 1950 178, 2380–2386. 10.4049/jimmunol.178.4.2380 [DOI] [PubMed] [Google Scholar]
- Pinheiro AS, Eibl C, Ekman-Vural Z, Schwarzenbacher R, Peti W, 2011. The NLRP12 Pyrin Domain: Structure, Dynamics, and Functional Insights. J. Mol. Biol. 413, 790–803. 10.1016/j.jmb.2011.09.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poyet JL, Srinivasula SM, Tnani M, Razmara M, Fernandes-Alnemri T, Alnemri ES, 2001. Identification of Ipaf, a human caspase-1-activating protein related to Apaf-1. J. Biol. Chem. 276, 28309–28313. 10.1074/jbc.C100250200 [DOI] [PubMed] [Google Scholar]
- Proell M, Riedl SJ, Fritz JH, Rojas AM, Schwarzenbacher R, 2008. The Nod-Like Receptor (NLR) Family: A Tale of Similarities and Differences. PLOS ONE 3, e2119. 10.1371/journal.pone.0002119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pudla M, Srisaowakarn C, Utaisincharoen P, 2019. NLRP12 negatively modulates inducible nitric oxide synthase (iNOS) expression and tumor necrosis factor-α production in Porphyromonas gingivalis LPS-treated mouse macrophage cell line (RAW264.7). Inflamm. Res. 68, 841–844. 10.1007/s00011-019-01267-3 [DOI] [PubMed] [Google Scholar]
- Qin Y, Xue B, Liu C, Wang X, Tian R, Xie Q, Guo M, Li G, Yang D, Zhu H, 2017. NLRX1 Mediates MAVS Degradation To Attenuate the Hepatitis C Virus-Induced Innate Immune Response through PCBP2. J. Virol. 91. 10.1128/JVI.01264-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rathinam VAK, Fitzgerald KA, 2016. Inflammasome Complexes: Emerging Mechanisms and Effector Functions. Cell 165, 792–800. 10.1016/j.cell.2016.03.046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyes Ruiz VM, Ramirez J, Naseer N, Palacio NM, Siddarthan IJ, Yan BM, Boyer MA, Pensinger DA, Sauer J-D, Shin S, 2017. Broad detection of bacterial type III secretion system and flagellin proteins by the human NAIP/NLRC4 inflammasome. Proc. Natl. Acad. Sci. U. S. A. 114, 13242–13247. 10.1073/pnas.1710433114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samir P, Kesavardhana S, Patmore DM, Gingras S, Malireddi RKS, Karki R, Guy CS, Briard B, Place DE, Bhattacharya A, Sharma BR, Nourse A, King SV, Pitre A, Burton AR, Pelletier S, Gilbertson RJ, Kanneganti T-D, 2019. DDX3X acts as a live-or-die checkpoint in stressed cells by regulating NLRP3 inflammasome. Nature 573, 590–594. 10.1038/s41586-019-1551-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandstrom A, Mitchell PS, Goers L, Mu EW, Lesser CF, Vance RE, 2019. Functional degradation: A mechanism of NLRP1 inflammasome activation by diverse pathogen enzymes. Science 364. 10.1126/science.aau1330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider M, Zimmermann AG, Roberts RA, Zhang L, Swanson KV, Wen H, Davis BK, Allen IC, Holl EK, Ye Z, Rahman AH, Conti BJ, Eitas TK, Koller BH, Ting JP-Y, 2012. The innate immune sensor NLRC3 attenuates Toll-like receptor signaling via modification of the signaling adaptor TRAF6 and transcription factor NF-κB. Nat. Immunol. 13, 823–831. 10.1038/ni.2378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw PJ, Lamkanfi M, Kanneganti T-D, 2010. NOD-like receptor (NLR) signaling beyond the inflammasome. Eur. J. Immunol. 40, 624–627. 10.1002/eji.200940211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw PJ, McDermott MF, Kanneganti T-D, 2011. Inflammasomes and autoimmunity. Trends Mol. Med. 17, 57–64. 10.1016/j.molmed.2010.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen C, Lu A, Xie WJ, Ruan J, Negro R, Egelman EH, Fu T-M, Wu H, 2019. Molecular mechanism for NLRP6 inflammasome assembly and activation. Proc. Natl. Acad. Sci. 116, 2052–2057. 10.1073/pnas.1817221116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi J, Zhao Y, Wang K, Shi X, Wang Y, Huang H, Zhuang Y, Cai T, Wang F, Shao F, 2015. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 526, 660–665. 10.1038/nature15514 [DOI] [PubMed] [Google Scholar]
- Silveira TN, Gomes MTR, Oliveira LS, Campos PC, Machado GG, Oliveira SC, 2017. NLRP12 negatively regulates proinflammatory cytokine production and host defense against Brucella abortus. Eur. J. Immunol. 47, 51–59. 10.1002/eji.201646502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strober W, Murray PJ, Kitani A, Watanabe T, 2006. Signalling pathways and molecular interactions of NOD1 and NOD2. Nat. Rev. Immunol. 6, 9–20. 10.1038/nri1747 [DOI] [PubMed] [Google Scholar]
- Suzuki S, Franchi L, He Y, Muñoz-Planillo R, Mimuro H, Suzuki T, Sasakawa C, Núñez G, 2014. Shigella type III secretion protein MxiI is recognized by Naip2 to induce Nlrc4 inflammasome activation independently of Pkcδ. PLoS Pathog. 10, e1003926. 10.1371/journal.ppat.1003926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tada H, Aiba S, Shibata K-I, Ohteki T, Takada H, 2005. Synergistic effect of Nod1 and Nod2 agonists with toll-like receptor agonists on human dendritic cells to generate interleukin-12 and T helper type 1 cells. Infect. Immun. 73, 7967–7976. 10.1128/IAI.73.12.7967-7976.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeuchi O, Akira S, 2010. Pattern Recognition Receptors and Inflammation. Cell 140, 805– 820. 10.1016/j.cell.2010.01.022 [DOI] [PubMed] [Google Scholar]
- Travassos LH, Carneiro LAM, Girardin SE, Boneca IG, Lemos R, Bozza MT, Domingues RCP, Coyle AJ, Bertin J, Philpott DJ, Plotkowski MC, 2005. Nod1 participates in the innate immune response to Pseudomonas aeruginosa. J. Biol. Chem. 280, 36714–36718. 10.1074/jbc.M501649200 [DOI] [PubMed] [Google Scholar]
- Truax AD, Chen L, Tam JW, Cheng N, Guo H, Koblansky AA, Chou W-C, Wilson JE, Brickey WJ, Petrucelli A, Liu R, Cooper DE, Koenigsknecht MJ, Young VB, Netea MG, Stienstra R, Sartor RB, Montgomery SA, Coleman RA, Ting JP-Y, 2018. The Inhibitory Innate Immune Sensor NLRP12 Maintains a Threshold against Obesity by Regulating Gut Microbiota Homeostasis. Cell Host Microbe 24, 364–378.e6. 10.1016/j.chom.2018.08.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuncer S, Fiorillo MT, Sorrentino R, 2014. The multifaceted nature of NLRP12. J. Leukoc. Biol. 96, 991–1000. 10.1189/jlb.3RU0514-265RR [DOI] [PubMed] [Google Scholar]
- Uchimura T, Oyama Y, Deng M, Guo H, Wilson JE, Rampanelli E, Cook KD, Misumi I, Tan X, Chen L, Johnson B, Tam J, Chou W-C, Brickey WJ, Petrucelli A, Whitmire JK, Ting JPY, 2018. The Innate Immune Sensor NLRC3 Acts as a Rheostat that Fine-Tunes T Cell Responses in Infection and Autoimmunity. Immunity 49, 1049–1061.e6. 10.1016/j.immuni.2018.10.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Udden SN, Kwak Y-T, Godfrey V, Khan MAW, Khan S, Loof N, Peng L, Zhu H, Zaki H, 2019. NLRP12 suppresses hepatocellular carcinoma via downregulation of cJun N-terminal kinase activation in the hepatocyte. eLife 8. 10.7554/eLife.40396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulland TK, Jain N, Hornick EE, Elliott EI, Clay GM, Sadler JJ, Mills KAM, Janowski AM, Volk APD, Wang K, Legge KL, Gakhar L, Bourdi M, Ferguson PJ, Wilson ME, Cassel SL, Sutterwala FS, 2016. Nlrp12 mutation causes C57BL/6J strain-specific defect in neutrophil recruitment. Nat. Commun. 7, 13180. 10.1038/ncomms13180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vladimer GI, Weng D, Paquette SWM, Vanaja SK, Rathinam VAK, Aune MH, Conlon JE, Burbage JJ, Proulx MK, Liu Q, Reed G, Mecsas JC, Iwakura Y, Bertin J, Goguen JD, Fitzgerald KA, Lien E, 2012. The NLRP12 Inflammasome Recognizes Yersinia pestis. Immunity 37, 96–107. 10.1016/j.immuni.2012.07.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Manji GA, Grenier JM, Al-Garawi A, Merriam S, Lora JM, Geddes BJ, Briskin M, DiStefano PS, Bertin J, 2002. PYPAF7, a Novel PYRIN-containing Apaf1like Protein That Regulates Activation of NF-κB and Caspase-1-dependent Cytokine Processing. J. Biol. Chem. 277, 29874–29880. 10.1074/jbc.M203915200 [DOI] [PubMed] [Google Scholar]
- Watanabe T, Asano N, Kitani A, Fuss IJ, Chiba T, Strober W, 2011. Activation of type I IFN signaling by NOD1 mediates mucosal host defense against Helicobacter pylori infection. Gut Microbes 2, 61–65. 10.4161/gmic.2.1.15162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werts C, Girardin SE, Philpott DJ, 2006. TIR, CARD and PYRIN: three domains for an antimicrobial triad. Cell Death Differ. 13, 798–815. 10.1038/sj.cdd.4401890 [DOI] [PubMed] [Google Scholar]
- Williams KL, Lich JD, Duncan JA, Reed W, Rallabhandi P, Moore C, Kurtz S, Coffield VM, Accavitti-Loper MA, Su L, Vogel SN, Braunstein M, Ting JP-Y, 2005. The CATERPILLER Protein Monarch-1 Is an Antagonist of Toll-like Receptor-, Tumor Necrosis Factor α-, and Mycobacterium tuberculosis -induced Pro-inflammatory Signals. J. Biol. Chem. 280, 39914–39924. 10.1074/jbc.M502820200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams KL, Taxman DJ, Linhoff MW, Reed W, Ting JP-Y, 2003. Cutting edge: Monarch-1: a pyrin/nucleotide-binding domain/leucine-rich repeat protein that controls classical and nonclassical MHC class I genes. J. Immunol. Baltim. Md 1950 170, 5354–5358. 10.4049/jimmunol.170.11.5354 [DOI] [PubMed] [Google Scholar]
- Xia X, Cui J, Wang HY, Zhu L, Matsueda S, Wang Q, Yang X, Hong J, Songyang Z, Chen ZJ, Wang R-F, 2011. NLRX1 negatively regulates TLR-induced NF-κB signaling by targeting TRAF6 and IKK. Immunity 34, 843–853. 10.1016/j.immuni.2011.02.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J, Zhao Y, Shi J, Shao F, 2013. Human NAIP and mouse NAIP1 recognize bacterial type III secretion needle protein for inflammasome activation. Proc. Natl. Acad. Sci. 110, 14408–14413. 10.1073/pnas.1306376110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaki MH, Man SM, Vogel P, Lamkanfi M, Kanneganti T-D, 2014. Salmonella exploits NLRP12-dependent innate immune signaling to suppress host defenses during infection. Proc. Natl. Acad. Sci. U. S. A. 111, 385–390. 10.1073/pnas.1317643111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaki MH, Vogel P, Malireddi RKS, Body-Malapel M, Anand PK, Bertin J, Green DR, Lamkanfi M, Kanneganti T-D, 2011. The NOD-like receptor NLRP12 attenuates colon inflammation and tumorigenesis. Cancer Cell 20, 649–660. 10.1016/j.ccr.2011.10.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamoshnikova A, Groß CJ, Schuster S, Chen KW, Wilson A, Tacchini-Cottier F, Schroder K, 2016. NLRP12 is a neutrophil-specific, negative regulator of in vitro cell migration but does not modulate LPS- or infection-induced NF-κB or ERK signalling. Immunobiology 221, 341–346. 10.1016/j.imbio.2015.10.001 [DOI] [PubMed] [Google Scholar]
- Zhang L, Mo J, Swanson KV, Wen H, Petrucelli A, Gregory SM, Zhang Z, Schneider M, Jiang Y, Fitzgerald KA, Ouyang S, Liu Z-J, Damania BA, Shu H-B, Duncan JA, Ting JP-Y, 2014. NLRC3, a member of the NLR family of proteins, is a negative regulator of innate immune signaling induced by the DNA sensor STING. Immunity 40, 329–341. 10.1016/j.immuni.2014.01.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Yang J, Shi J, Gong Y-N, Lu Q, Xu H, Liu L, Shao F, 2011. The NLRC4 inflammasome receptors for bacterial flagellin and type III secretion apparatus. Nature 477, 596–600. 10.1038/nature10510 [DOI] [PubMed] [Google Scholar]
- Zhong Z, Liang S, Sanchez-Lopez E, He F, Shalapour S, Lin X-J, Wong J, Ding S, Seki E, Schnabl B, Hevener AL, Greenberg HB, Kisseleva T, Karin M, 2018. New mitochondrial DNA synthesis enables NLRP3 inflammasome activation. Nature 560, 198–203. 10.1038/s41586-018-0372-z [DOI] [PMC free article] [PubMed] [Google Scholar]