Abstract
Background:
Infection is a frequent and serious complication after combat-related trauma. The Sequential Organ Failure Assessment (SOFA) score has been shown to have predictive value for outcomes, including sepsis and mortality, among various populations. We evaluated the prognostic ability of SOFA score in a combat-related trauma population.
Methods:
Combat casualties (2009–2014) admitted to Landstuhl Regional Medical Center (LRMC; Germany) intensive care unit (ICU) within 4 days post-injury followed by transition to ICUs in military hospitals in the United States were included. Multivariate logistic regression was used to determine predictive effect of selected variables and receiver operating characteristic (ROC) curve analysis was used to evaluate overall accuracy of SOFA score for infection prediction.
Results:
Of the 748 patients who met inclusion criteria, 436 (58%) were diagnosed with an infection (32% bloodstream, 63% skin and soft tissue, and 40% pulmonary) and were predominantly young (median 24 years) males. Penetrating trauma accounted for 95% and 86% of injuries among those with and without infections, respectively (p<0.001). Median LRMC admission SOFA score was 7 (interquartile range [IQR]: 4–9) in patients with infections versus 4 (IQR: 2–6) in patients without infections (p<0.001). Thirty-day mortality was 2% in both groups. On multivariate regression, LRMC SOFA score was independently associated with infection development (odds ratio: 1.2; 95% confidence interval: 1.1–1.3). The ROC curve analysis revealed an area under the curve of 0.69 for infection prediction, and 0.80 for mortality prediction.
Conclusions:
The SOFA scores obtained up to 4 days post-injury predict late onset infection occurrence. This study revealed that for every 1 point increase in LRMC SOFA score, the odds of having an infection increases by a factor of 1.2, controlling for other predictors. The use of SOFA score in admission assessments may assist clinicians with identifying those at higher risk of infection following combat-related trauma.
Keywords: Infection, infectious disease, trauma, severity score systems, SOFA score, combat trauma
BACKGROUND
Infectious complications following both civilian and combat-related trauma have remained a persistent threat throughout history, and have been associated with increased mortality, prolonged hospital lengths of stay, worsened functional outcomes, and increased utilization of healthcare resources that may continue for several months post-injury.1–9 Infection rates after traumatic injury vary between 2–38%, depending on the definitions used and population studied.6–12 Among combat casualties, intensive care unit (ICU) admission, blood transfusions in the first 24 hours following injury, Injury Severity Score (ISS), shock index, sustaining an open fracture or amputation, and blast-injuries have been identified as infection risk factors.3,12–15 Additionally, among civilian trauma patients, preexisting medical comorbidities (i.e. cardiac disease, diabetes, and immune deficiency) are well-associated with subsequent infection.7 The ability to identify those patients at increased risk of infection remains important for the development of practices and procedures that aim to prevent such complications among critically injured combat casualties.
The Society of Critical Care Medicine and the European Society of Intensive Care Medicine published an updated definition for sepsis in February 2016,16 which moved away from the systemic inflammatory response syndrome (SIRS) criteria and defined sepsis as a dysregulated immune response to infection and suggested use of the Sequential Organ Failure Assessment (SOFA) score to identify those with sepsis. In the 2021 update,17 it was recommended against using quick SOFA (qSOFA) based on low sensitivity compared with SIRS, the National Early Warning Score, or the Modified Early Warning Score as a single screening tool for sepsis or septic shock. While the SOFA score has been shown to be a better predictor of mortality among critically-ill patients with an infection diagnosis compared to SIRS criteria,18 there are few studies evaluating the application of the SOFA score to trauma patients. In particular, SIRS criteria have been shown to be nonspecific in the identification of mortality or infection among trauma patients, most likely related to the robust host inflammatory response to injury that creates a clinical picture largely indistinguishable from sepsis.5,19 Furthermore, the evaluation of SOFA scores among civilian trauma populations have had mixed results. In a study of 532 patients admitted to a trauma ICU, there was no correlation between SIRS criteria, SOFA score, or qSOFA score and the development of a new infection over a 90-day period on multivariable regression modeling.20 Alternatively, in another study using 30-day mortality as the outcome, it was demonstrated that the total maximum SOFA score obtained during admission for 506 trauma patients maintained the best discriminative power as compared to other general and trauma scoring systems.21
The combat-injured trauma population differs from civilian trauma in terms of injury type and severity, as well as patient demographics. Military personnel often lack medical comorbidities present among civilian trauma patients, and while civilian personnel are more commonly injured in motor vehicle collisions or falls resulting in blunt injures, the military population frequently sustain multiple penetrating injuries, often via a blast mechanism.22 While studies have examined ISS among combat casualties, SOFA scores have not been fully evaluated. Thus, we sought to evaluate the prognostic ability of the SOFA score for outcomes among critically-ill military personnel with combat-related injuries. For this study herein, we utilized the recommendations from the February 2016 sepsis guidelines16 as the comprehensive full SOFA provides significantly more information and was worth exploring with regard to use with combat casualties.
METHODS
Study Population and Design
Data were obtained from the U.S. Department of Defense (DoD) — Department of Veterans Affairs, Trauma Infectious Disease Outcomes Study (TIDOS), which is a multicenter, observational study designed to assess short- and long-term infectious disease outcomes associated with deployment-related traumatic injuries.23,24 Patients were eligible for inclusion in TIDOS if they sustained deployment-related trauma (June 2009 – December 2014) and required medical evacuation to Landstuhl Regional Medical Center (LRMC) in Germany, followed by a transfer to a participating military hospital in the United States (i.e., Walter Reed National Military Medical Center [previously Walter Reed Army Medical Center and National Naval Medical Center] and Brooke Army Medical Center). Since prior reported validation studies evaluating the predictive ability of SOFA score on outcomes focused on critical care units,25–27 an additional criterion for this study was admission to the LRMC ICU and subsequent admission to the U.S. hospital ICU. Exclusion criteria included missing LRMC admission SOFA scores (any of its components), delayed admission to LRMC (>4 days post-injury), infection diagnosed prior to LRMC admission, or non-combat injury as the reason for evacuation. This study was approved by the Institutional Review Board of the Uniformed Services University of the Health Sciences in Bethesda, Maryland.
Data related to trauma history, patterns and severity of injury, and surgical interventions were obtained from the DoD Trauma Registry.28 Traumatic injuries were categorized according to the Abbreviated Injury Scale (AIS), which classifies injury severity according to anatomic region and magnitude of the injury in that region.22 These scores are then used to determine a composite ISS, which provides a better estimate of mortality risk.29,30 Combat-related injuries were defined as traumatic injuries directly related to combat operations and included the following mechanisms: blast, burn, and gunshot wound. Shock index was obtained at the initial support hospital in the theater of operation and is defined as the quotient of heart rate divided by systolic blood pressure. Patients missing data related to blood transfusion were analyzed in combination with those receiving zero units of blood, as a prior TIDOS analysis showed a correlation between lack of data regarding blood transfusion and low ISS, suggesting a reduced likelihood of these patients having received blood products.12
Infection-related data were captured through the TIDOS infectious disease module of the DoD Trauma Registry and classified using a combination of clinical findings and ancillary data obtained through medical record review based on standardized definitions for nosocomial infections by the National Healthcare Safety Network.23,31 Infections were also counted if clinically diagnosed by a physician and associated with the initiation of targeted antimicrobial treatment with continuation for ≥5 days (≥21 days for osteomyelitis). Infections were excluded if an alternate diagnosis was made and antimicrobials were discontinued. Data on infections were collected over the entire period of hospitalization. Of patients with multiple infections diagnosed during their hospitalization, assessment of location (i.e. hospital) and timing was restricted to the first infection diagnosed. The SOFA scores were collected upon admission to LRMC and participating U.S. hospitals.
Statistical Analysis
Tests of association for categorical variables were performed using chi-squared or Fisher’s exact test where applicable; continuous variables were compared using Mann-Whitney U test. Predictive factors for infection were evaluated using logistic regression. Injury characteristics in the univariate analysis that were significantly associated with an increased risk of infection were examined in the multivariate model. A backward selection method was used to choose the final predictive factors for development of infection. Receiver operating characteristic (ROC) curve analysis was performed to further evaluate the predictive ability of SOFA score for the development of infection. Statistical analysis was conducted using SAS® version 9.3 (SAS, Cary, NC). Data are expressed as odds ratios (OR) with 95% confidence intervals (CI). Statistical significance was defined as p<0.05.
RESULTS
Study Population and Injury Patterns
Among 5,845 wounded military personnel, 4,271 (73%) sustained a combat-related traumatic injury with 882 (15%) requiring ICU admission at both LRMC and U.S. hospitals and 748 (13%) meeting the remaining inclusion criteria (Figure 1). The majority of patients were male (>98%), enlisted (91%) and serving in the U.S. Army (62%) or U.S. Marines (31%) (Table 1). The median age was 24 years (interquartile range [IQR]: 21–29), and personnel were primarily injured in Afghanistan (95%). More than 90% of the study population had penetrating wounds and the most frequent mechanism of injury was blast (79%). Of those sustaining a blast injury, improvised explosive devices accounted for 84%.
Figure 1.
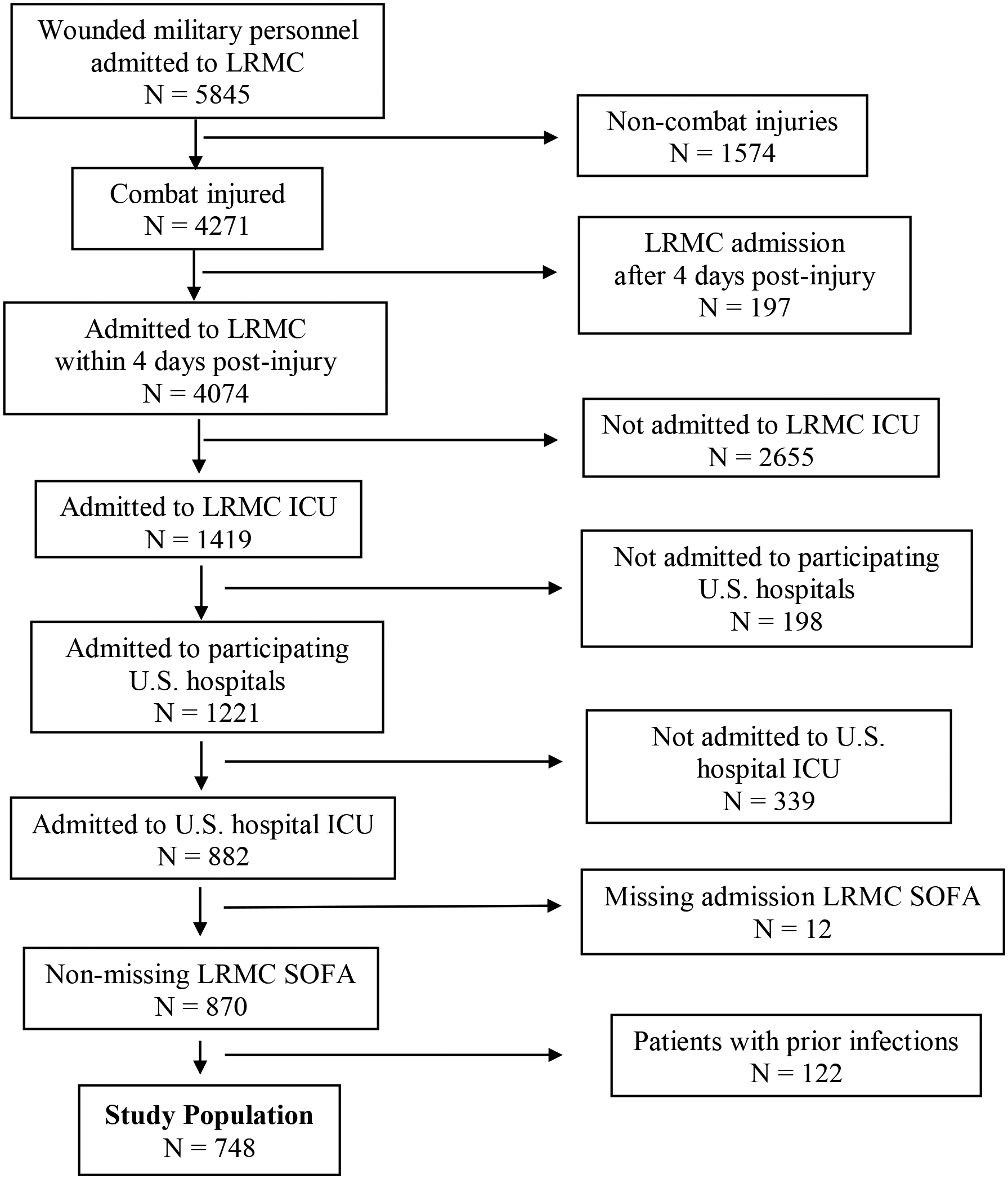
Study Population Flow Diagram. LRMC – Landstuhl Regional Medical Center; ICU – intensive care unit; SOFA – sequential organ failure assessment
Table 1.
Characteristics and Outcomes of U.S. Combat Casualties Admitted to the Intensive Care Unit (June 2009 – December 2014)
| Characteristics | Overall Population (N = 748) |
Patients Who Developed Any Infection (N = 436) |
Patients Who Did Not Develop an Infection (N = 312) |
p-valuea |
|---|---|---|---|---|
| Demographics, No. (%) | ||||
| Age, median (IQR) | 24 (21–29) | 24 (21–29) | 24 (22–28) | 0.370 |
| Male | 739 (98.8) | 433 (99.3) | 306 (98.1) | 0.175b |
| Enlisted | 681 (91.0) | 400 (91.7) | 281 (90.1) | 0.332c |
| Branch of Service | 0.008c | |||
| Army | 464 (62.0) | 247 (56.7) | 217 (69.6) | |
| Marine | 235 (31.4) | 159 (36.5) | 76 (24.4) | |
| Air Force | 18 (2.4) | 11 (2.5) | 7 (2.2) | |
| Navy | 22 (2.9) | 13 (2.9) | 9 (2.9) | |
| Injured in Afghanistan | 710 (94.9) | 414 (95.0) | 296 (94.9) | 0.960c |
| Injury Type, No. (%) | ||||
| Blunt | 11 (1.5) | 5 (1.1) | 6 (1.9) | 0.540b |
| Penetrating | 682 (91.2) | 415 (95.2) | 267 (85.6) | <0.0001c |
| Burn | 94 (12.6) | 60 (13.8) | 34 (10.9) | 0.244c |
| Injury Mechanism, No. (%) | ||||
| Blast | 588 (78.6) | 352 (80.7) | 236 (75.6) | 0.094c |
| IEDd | 494 (84.0) | 314 (89.2) | 180 (76.3) | 0.094c |
| Non-blast | 160 (21.3) | 84 (19.3) | 132 (42.3) | 0.094c |
| GSWd | 125 (78.1) | 65 (77.4) | 60 (79.0) | 0.811c |
| Severity Score, median (IQR) | ||||
| ISS | 30 (22.5–43) | 33 (27–45) | 27 (19–37.5) | <0.0001 |
| Shock Indexe | 0.87 (0.67–1.17) | 0.98 (0.74–1.32) | 0.76 (0.61–0.97) | <0.0001 |
| SOFA | 5 (3–8) | 7 (4–9) | 4 (2–6) | <0.0001 |
| Blood transfusions within 24 hrs post-injury, median units (IQR)f | 11 (5–19) | 14 (6–22) | 6 (3–12) | <0.0001 |
| Types of Infections, No. (%) | ||||
| Bloodstream | 139 (18.6) | 139 (31.9) | - | NA |
| Osteomyelitis | 53 (7.1) | 53 (2.2) | - | NA |
| SSTI | 275 (36.8) | 275 (63.1) | - | NA |
| CNS | 26 (3.5) | 26 (6.9) | - | NA |
| Pulmonary / Intrathoracic | 174 (23.3) | 174 (39.9) | - | NA |
| Intraabdominal | 23 (3.1) | 23 (5.3) | - | NA |
| Sepsis | 35 (4.7) | 35 (8.0) | - | NA |
| Other | 112 (15.0) | 112 (25.7) | - | NA |
| Time to First Infection, median days (IQR) g | ||||
| Bloodstream | 7 (5–16) | 7 (5–16) | - | NA |
| Osteomyelitis | 16 (7–26) | 16 (7–26) | - | NA |
| SSTI | 8 (6–15) | 8 (6–15) | - | NA |
| CNS | 12.5 (6–21) | 12.5 (6–21) | - | NA |
| Pulmonary / Intrathoracic | 6 (4–10) | 6 (4–10) | - | NA |
| Intraabdominal | 14 (8–17) | 14 (8–17) | - | NA |
| Sepsis | 12 (5–22) | 12 (5–22) | - | NA |
| Other | 16 (8–34) | 16 (8–34) | - | NA |
| Location of First Infection, No. (%)g | ||||
| LRMC | 76 (10.2) | 76 (17.4) | - | NA |
| Outcomes | ||||
| Total Days, median (IQR) | ||||
| Hospital LOS | 34 (21–53) | 45 (31.5–65) | 21 (15–31) | <0.0001 |
| Ventilator | 2 (0–5) | 3 (1–7) | 1 (0–3) | <0.0001 |
| ICU LOS | 6 (4–10) | 7 (4–14) | 5 (4–7) | <0.0001 |
| 30-Day Mortality, No. (%) | 15 (2) | 9 (2.1) | 6 (1.9) | 0.892c |
CNS – central nervous system; GSW – gunshot wound; ICU – intensive care unit; IED – improvised explosive device; ISS – injury severity score; IQR – interquartile range; LOS – length of stay; LRMC – Landstuhl Regional Medical Center; OEF – Operation Enduring Freedom; SOFA – sequential organ failure assessment; SSTI – skin and soft-tissue infection
Unless otherwise noted, p-values calculated with Mann-Whitney U
p-value calculated with Pearson Chi square
p-value calculated with Fisher’s exact test
Proportions calculated using total for that type of injury mechanism (blast or non-blast)
Shock index missing for 93 patients (53 with infections and 40 without infections)
Blood transfusion data were not available for 161 patients, so these patients were analyzed in combination with those receiving zero units of blood
Among patients with multiple infections, data from the first diagnosed infection was assessed
Infection Characteristics and Outcomes
At least one infection developed among 436 (58%) patients. Baseline demographics of patients who developed an infection were comparable to those who did not have an infection except there was a significant difference in the distribution of the branch of service categories (p=0.008; Table 1). As an example, 37% and 56% of those with an infection were Marines and Army, respectively, versus 24% and 70% among those without infections. Patients with an infection had a higher proportion of penetrating trauma (95% versus 86%; p<0.0001), as well as greater injury severity (median ISS 33 versus 27; p<0.0001) and LRMC admission SOFA scores (7 versus 4; p<0.0001) compared to patients without infections. In addition, patients who developed infections had larger blood transfusion requirements during the first 24 hours following injury (median units 14 versus 6; p<0.0001).
The most common type of infection observed was skin and soft-tissue infections (63% of 436 patients), followed by pulmonary and intrathoracic infections (40%) and bloodstream infections (32%). Seventy-six patients (17%) developed their first infection at LRMC, while the remainder (83%) did not have their infection diagnosed until after transfer to U.S. hospitals. The median time to infection ranged from 6 days (IQR: 4–10 days) with pulmonary and intrathoracic infections to 16 days (IQR: 7–26 days) with osteomyelitis. Patients who developed an infection had longer ICU stays (median 7 days; IQR: 4–14 versus 5 days; IQR: 4–7; p<0.0001) and longer total hospital lengths of stay (median 45 days; IQR: 31.5–65 versus 21 days; IQR: 15–31; p<0.0001) compared to patients without an infection. Thirty-day mortality rate was 2% and did not differ among groups.
Risk Factor Analysis
Composite ISS, penetrating injury, blast injury type, blood transfusion requirements within 24 hours of injury, shock index, LRMC admission SOFA score, ICU length of stay, ventilator days, and total hospital length of stay were significantly associated with the development of infection in the univariate model (Table 2). In addition, ISS, blood transfusion requirements, and shock index were evaluated as continuous variables and remained significant predictors of infection in the univariate analysis (data not shown).
Table 2.
Unadjusted Logistic Regression Analysis of Potential Factors Associated with the Risk of Infection among Combat Casualties Admitted to the Intensive Care Unit
| Potential Risk Factor | Univariate Odds Ratio (95% CI) | p-value |
|---|---|---|
| Injury Severity Score (reference: 1–9) | ||
| 10–15 | 2.3 (0.71–7.44) | 0.164 |
| 16–25 | 4.03 (1.46–11.16) | 0.007 |
| ≥26 | 8.77 (3.28–23.45) | <0.0001 |
| Injury type | ||
| Penetrating (reference: non-penetrating injury) | 3.33 (1.94–5.72) | <0.0001 |
| Blast type (reference: non-blast injury) | ||
| IED | 1.58 (1.10–2.26) | 0.013 |
| Non-IED | 0.61 (0.37–1.03) | 0.064 |
| PRBC transfusions within 24 hours of post-injury (reference: zero or missing units) | ||
| 1–9 units | 2.61 (1.73–3.94) | <0.0001 |
| 10–20 units | 6.46 (4.12–10.12) | <0.0001 |
| >20 units | 15.22 (8.41–27.55) | <0.0001 |
| Shock index (reference: <0.8) | ||
| ≥0.8 | 2.89 (2.09–3.99) | <0.0001 |
| LRMC Admission SOFA score | 1.26 (1.20–1.33) | <0.0001 |
| Total ICU days | 1.10 (1.07–1.13) | <0.0001 |
| Total ventilator days | 1.22 (1.16–1.29) | <0.0001 |
| Total hospital LOS | 1.08 (1.06–1.09) | <0.0001 |
CI – confidence interval; ICU – intensive care unit; IED – improvised explosive device; LOS – length of stay; LRMC – Landstuhl Regional Medical Center; SOFA – sequential organ failure assessment
In the multivariate model, receipt of >20 units of blood within 24 hours of injury conferred the greatest risk for infection (OR: 9.01; 95% CI: 4.83–16.78; p<0.0001; Table 3). Additionally, LRMC admission SOFA score remained an independent predictor of infection, though yielding only a modestly increased risk (OR: 1.19; 95% CI: 1.13–1.26; p<0.0001). Shock index and blast injury type were not retained in the model. Although ISS remained in the model, it was not a significant predictor of infection, even among the highest scores (ISS ≥26 versus 1–9; p=0.075). The ROC curve analysis revealed an area under the curve (AUC) of infection prediction for SOFA of 0.69 (95% CI: 0.66–0.73; p<0.0001; Figure 2). Further evaluation of the optimal cut-off point showed that SOFA score ≥5 yielded a sensitivity of 72% and specificity of 58%. Regarding mortality predication of SOFA, the AUC was 0.80 (95% CI: 0.70–0.89; p<0.001).
Table 3.
Adjusted Logistic Regression Analysis of Factors Associated with Risk of Infection among Combat Casualties Admitted to the Intensive Care Unit
| Potential Risk Factor | Multivariate Odds Ratio (95% CI) | p-value |
|---|---|---|
| Injury Severity Score (reference: 1–9) | ||
| 10–15 | 1.27 (0.37–4.36) | 0.707 |
| 16–25 | 1.62 (0.56–4.71) | 0.374 |
| ≥26 | 2.55 (0.91–7.13) | 0.075 |
| LRMC Admission SOFA score | 1.19 (1.13–1.26) | <0.0001 |
| Blood transfusions within 24 hours post-injury (reference: zero or missing units) | ||
| 1–9 units | 2.24 (1.45–3.47) | <0.001 |
| 10–20 units | 4.86 (3.02–7.82) | <0.0001 |
| >20 units | 9.01 (4.83–16.78) | <0.0001 |
CI – confidence interval; LRMC – Landstuhl Reginal Medical Center; SOFA – sequential organ failure assessment
Figure 2.
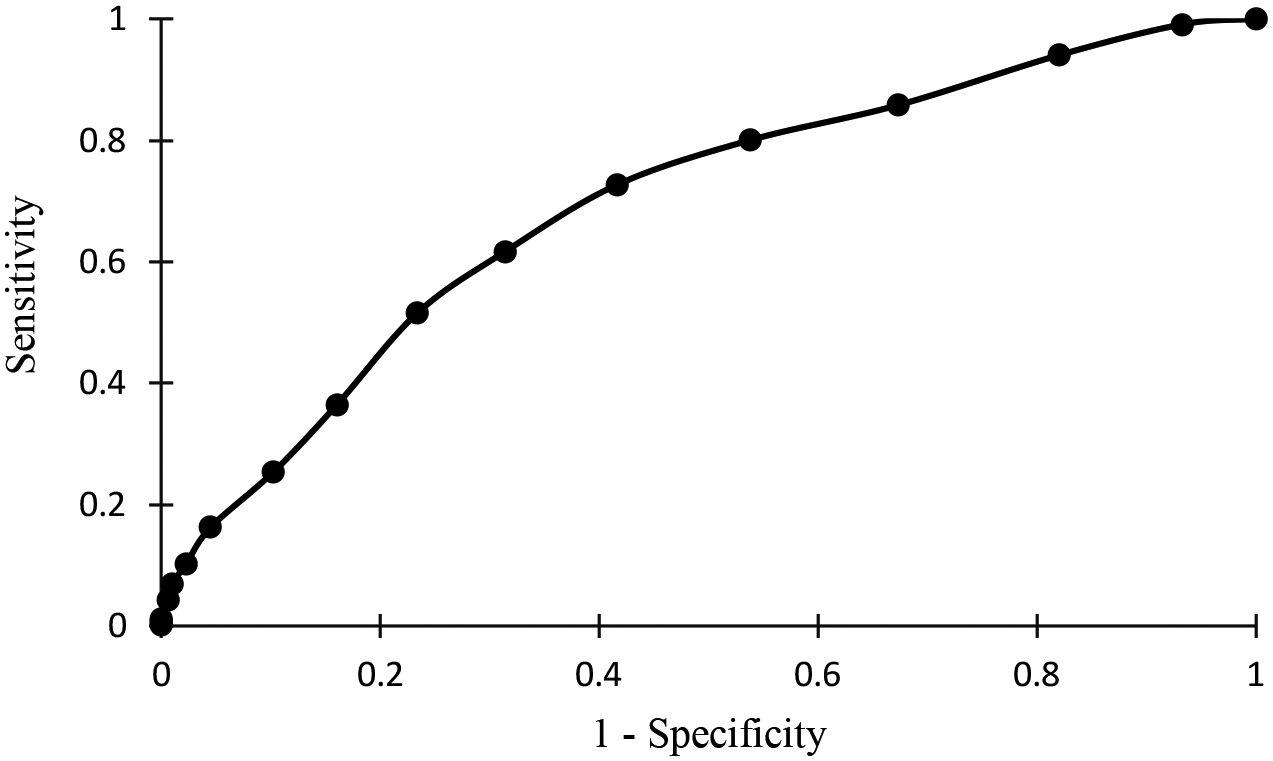
Receiver-Operating Characteristic Curve Analysis of Infection Predictions Using the Admission Sequential Organ Failure Assessment Score. The area under the curve is 0.694 (94% confidence interval: 0.656–0.732; p<0.0001).
When bloodstream infection was used as the dependent variable, the multivariate model showed results similar to the model evaluating any infection. Both blood transfusion and LRMC admission SOFA score remained significant predictors. Transfusion of >20 units of blood within 24 hours of injury was associated with the greatest risk for bloodstream infection (OR 3.6, 95% CI: 1.82–7.11; p=0.0002), whereas a rising SOFA score conferred similar risk as when any infection was used as the outcome measure (OR 1.23, 95% CI: 1.13–1.26; p<0.0001).
When hospital length of stay was used as the dependent variable in multivariate analysis, both blood transfusion and admission SOFA score continued to be significant predictors. Receipt of >20 units of blood within 24 hours of injury was associated with an increase of 32 days length of stay (95% CI: 25–39; p<0.0005), compared to an 11-day increase for those who received 1–9 units (95% CI: 5–17; p<0.0001). The LRMC admission SOFA score had a less prominent effect, though still significant, predicting a 2-day increase in length of stay for every 1-point rise in SOFA score (p<0.0001).
DISCUSSION
Infectious complications following combat-related trauma continue to occur at high rates, particularly among service members requiring ICU admission; however, early predictors of who may go on to develop infections are not clearly defined. Among those admitted to the ICU at LRMC and also at participating U.S. hospitals, 58% were diagnosed with an infection and the LRMC admission SOFA score did show predictive ability for the subsequent development of infection during hospitalization. Specifically, our results showed that for every 1-point increase in LRMC admission SOFA score, the odds of developing an infection increase by a factor of 1.2. This prognostic finding is consistent with a previously published multicenter study of patients admitted to civilian critical care units. In a study of 1,449 surgical ICU patients, the increasing numbers of organ failures, defined as SOFA score ≥3 within a particular organ system, on day of ICU admission was associated with an increased risk of infection (17% of patients with no organ failure developed infection, whereas 74% of patients with ≥4 organ failures were diagnosed with infection).32
The ROC curve analysis was utilized as an additional measure of the predictive ability of SOFA score for any infection. Our study found the AUC was 0.69, indicating fair discrimination. The optimal cut-off point is a score ≥5, which yields a sensitivity of 72% and specificity of 58%. Choosing a higher value of ≥8 leads to improved specificity of 84%, while dropping sensitivity to 36%. These values are similar to those obtained among studies involving medical and surgical critical care patients when mortality is utilized as the outcome, with AUCs ranging from 69–80%.18,26,27,33,34
Although multiple variables other than LRMC admission SOFA score were shown to have predictive ability for infection among the univariate model, to include composite ISS, injury type and mechanism, shock index, ventilator days, and ICU and overall hospital length of stay, only blood transfusion requirement remained a significant predictor on the multivariate analysis. The association between blood transfusion and infection has been shown among both civilian and combat-related trauma populations.35–38 This finding remains independent of ISS, which argues against the notion of the importance of large-volume blood transfusions being only a marker of injury severity. The immunomodulatory effects of blood transfusion after trauma have been described, though notably the hypothesis that immune dysregulation is due in large part to the blood transfusions themselves, as opposed to the inciting trauma and blood-loss anemia has not been definitively shown.39 In fact, among healthy surgical patients without underlying comorbidities or significantly immunocompromising conditions, blood transfusion has not been shown to significantly affect cellular or humoral immunological function.40 It is likely a combination of factors that results in the increased risk of infection seen in those suffering traumatic injuries and requiring large amounts of blood products during initial resuscitation.
We also evaluated outcomes of bloodstream infection and overall hospital length of stay. Multivariate analysis showed that LRMC admission SOFA score remains an independent predictor for bloodstream infection, with an OR near the same value as when any infections are used as the dependent variable (OR of 1.23 with bloodstream infection and 1.19 with any infection). This finding is similar to a single-center study including 1,316 patients admitted to a multidisciplinary ICU, which found that admission SOFA score was the only variable independently related to bacteremia (OR 1.2, p<0.001).26
One unique finding of our study is that ISS did not remain a significant predictor of any infection, nor of bloodstream infection. Multiple prior studies reported an association between ISS and subsequent infection risk among both civilian and combat-associated trauma populations.3,7,9,12,14,41 One possible explanation is the differing methods used to define infection and sepsis. Examination of infection risk factors from more than 16,000 casualties from Iraq and Afghanistan between 2003–2009 identified ISS as a significant predictor of infection on multivariate analysis.3 Infections in this study; however, were defined by ICD-9 codes with a reported infection rate of 5.5%. The authors noted inadequate U.S.-based hospital and long-term data regarding infectious complications leading to an underrepresentation of infections in their analysis. As described earlier, our study utilizes the TIDOS population, which uses standardized definitions to more accurately capture infectious complications. Data from a prior TIDOS analysis noted a much higher proportion of infections (34%); however, it should be noted that patients evacuated to TIDOS-participating U.S. hospitals are more severely injured than those who transitioned to non-participating hospitals (15% with ISS ≥25 versus 2%).12 More accurate identification of those truly infected may be in part responsible for the discrepancy seen between studies regarding the association between ISS and infection.
Another explanation could be related to the timing of infection after injury. In our study, index infections largely occurred at day 6 or later after injury (59%), and we excluded those who had any infection occurring prior to LRMC admission. As ISS is obtained at the time of injury, its relevance in early infections may be more pronounced than in late-onset infections. This lack of association between ISS and late infections has been shown in studies of both combat and noncombat populations. Examination of combat-injured veterans with orthopedic injuries found no difference in median ISS among those who sustained a recurrent episode of osteomyelitis compared to those who did not.42 Additionally, a single-center study demonstrated that ISS was not a significant risk factor for subsequent Acinetobacter baumannii infection among a civilian trauma population in which a majority of the infections identified had occurred beyond 72 hours of admission.43
Another factor to consider is differences among study populations. A prior study involving the TIDOS population (3-year study versus 5.5 years in our present study) evaluated risk factors for infection among patients admitted to all wards, not just the ICU as in our present study.12 The timing of infections was similar to our findings, with >50% occurring beyond a median of nine days. Although there was an association between ISS and infection, it was only the highest scores that showed significance. Specifically, ISS ≥25 was shown to have an adjusted hazard ratio for infectious complications of 1.79 (95% CI 1.29–2.49); however, only 15% of the entire population had scores in this life-threatening range.12 In our study, 70% of participants had ISS ≥26, which highlights the challenge of generalizing findings across severities of illness. When evaluating a population with severe injuries and an overall increased rate of infection, scoring systems that focus on more granular data in terms of organ damage, such as SOFA, may be necessary to more accurately predict those at risk for infectious complications. Even with the increased granularity compared to SIRS; however, extrapolation of our findings to civilian trauma populations must be done with caution. Analysis of 1,942 patient-days from trauma and surgical ICUs found no association between the risk of infection and daily SOFA scores (median of 6 vs 7). This may be related to the overall higher SOFA scores experienced by patients in this civilian population.20 Specifically, 98% of the civilian patients had a SOFA score ≥2,20 whereas only 89% of our study population had admission SOFA scores ≥2.
Of the 436 patients who developed an infection, 59% were diagnosed with their first infection more than six days following their injury. Since all patients in this population were admitted to LRMC no later than four days post-injury, the infections diagnosed ≥6 days are, by definition, nosocomial. This count does not include all infection types, as many patients in this study had multiple infections and only the time to the first infection was assessed; thus, is an underrepresentation of the total number of nosocomial infections sustained. The significance of nosocomial infections among combat-injured patients from recent conflicts has been well-described, with multiple studies corroborating the transmission of multidrug-resistant organisms throughout the combat casualty healthcare system.44–48 As such, the importance of consistent adherence to infection prevention and control practices and antimicrobial stewardship in the deployed environment was recognized and led to updates to the Joint Trauma System DoD clinical practice guidelines for the prevention of combat trauma-related infections.49 Although existing evidence does not suggest those identified as being at higher risk of infection should receive altered duration or spectrum of post-injury antimicrobials, they may benefit from closer monitoring for infectious complications and represent a population that will benefit from strict adherence to infection control practices throughout all levels of care.49,50
While our study represents the first evaluation of the predictive ability of SOFA score among the combat-injured patients from recent conflicts, there are limitations that should be noted. As SOFA scores are typically only calculated in critically-ill patients, only those who were admitted to the ICUs at both LRMC and U.S. TIDOS-participating hospitals were included, representing a more severely injured population. Therefore, the infection rates and findings of this analysis may not be generalizable to all combat casualties (i.e. those with mild to moderate injuries). We were unable to assess mortality as an outcome measure in this study given such low incidence at 2% overall. This is likely due to the fact that a majority of combat trauma-related deaths occur shortly after injury, prior to admission into higher echelons of care.51 Previous studies have shown associations between both the maximum SOFA value obtained during a hospitalization or the delta SOFA with mortality and infection;26,34,52 however, only admission SOFA scores were available for our review, so we were unable to assess these variables. It should also be noted that military personnel included in our study were healthy prior to sustaining battlefield trauma, so their admission SOFA scores would be lower than civilian trauma patients who have comorbidities (e.g., cardiovascular disease or diabetes). In particular, a SOFA score of 7 is generally correlated with an approximately 15% risk of mortality in civilian trauma,25 while it was associated with infection risk in our population.
In conclusion, this analysis shows that the application of SOFA scores to the combat-injured population has similar predictive ability for infection as in the civilian trauma literature despite the differences amongst these two populations. Although ISS remains the primary scoring system used to assess the severity of initial traumatic injuries, it was unable to independently predict subsequent infection in our study, whereas SOFA did. Incorporation of the SOFA score into initial assessments may help triage patients who may benefit from more frequent wound monitoring and earlier debridement, as wound infections (i.e., skin and soft-tissue infections) are the most common type of infection among combat casualties. This may be particularly helpful for patients requiring transfer following initial trauma stabilization, as there may be incomplete records of initial resuscitation and injury patterns, but the SOFA calculated on transfer remains independently associated with infection risk. Pneumonia and bloodstream infections were also frequent in our analysis, consistent with several previous studies involving combat-injured trauma populations, and infection control measures and ICU bundles proven to decrease these infections should be emphasized in patients identified as higher risk. Such measures include avoidance of mechanical ventilation when possible, daily sedation holidays, and spontaneous breathing trials among those intubated patients, as well as central venous catheter insertion and maintenance bundles along with daily assessment of the continued need for central venous access.53 It has been emphasized that an ongoing commitment to proper education, training, research and implementation of infection control practices in the deployed setting are necessary for the reduction of infectious complications seen after combat-related traumatic injury.46 Incorporation of the SOFA score into combat-injured patient assessments upon ICU admission may assist clinicians in identifying those individuals with higher risk of subsequent infection and allow for earlier triage and the application of appropriate mitigation strategies.
ACKNOWLEDGMENTS
We are indebted to the Infectious Disease Clinical Research Program TIDOS study team of clinical coordinators, microbiology technicians, data managers, clinical site managers, and administrative support personnel for their tireless hours to ensure the success of this project. We specifically would like to thank Joseph Petfield, DO, Timothy Whitman, MD, Teresa Merritt, and M. Leigh Carson for their efforts on this project.
Funding:
Support for this work (IDCRP-024) was provided by the Infectious Disease Clinical Research Program (IDCRP), a Department of Defense (DoD) program executed by the Uniformed Services University of the Health Sciences (USU) through a cooperative agreement with The Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc. (HJF). This project has been supported with federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIH), under Inter-Agency Agreement Y1-AI-5072, the Defense Health Program, U.S. DoD, under award HU0001190002, and the Department of the Navy under the Wounded, Ill, and Injured Program under award HU0001-10-1-0014.
Footnotes
Declaration of Conflicting Interest: The authors have no conflict of interest to declare.
A portion of this data was presented at the 2019 Society for Critical Care Medicine Annual Meeting, February 17–20, 2019, San Diego, CA.
Publisher's Disclaimer: Disclaimer: The view(s) expressed herein are those of the author(s) and do not reflect the official policy or position of Uniformed Services University of the Health Sciences, Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc., National Institutes of Health or the Department of Health and Human Services, Brooke Army Medical Center, the U.S. Army Medical Department, the U.S. Army Office of the Surgeon General, the Department of the Air Force, Navy, and Army, or the Department of Defense or the U.S. Government. Mention of trade names, commercial products, or organization does not imply endorsement by the U.S. Government. Some of the co-authors are military service members (or employees of the U.S. Government). This work was prepared as part of their official duties. Title 17 U.S.C. §105 provides that ‘Copyright protection under this title is not available for any work of the United States Government.’ Title 17 U.S.C. §101 defines a U.S. Government work as a work prepared by a military service member or employee of the U.S. Government as part of that person’s official duties.
REFERENCES:
- 1.Murray CK, Hinkle MK, Yun HC. History of infections associated with combat-related injuries. J Trauma. 2008;64(3 Suppl):S221–S231. [DOI] [PubMed] [Google Scholar]
- 2.Blyth DM, Yun HC, Tribble DR, Murray CK. Lessons of war: Combat-related injury infections during the Vietnam War and Operation Iraqi and Enduring Freedom. J Trauma Acute Care Surg. 2015;79(4 Suppl 2 Proceedings of 2014 Military Health System Research Symposium):S227–S235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Murray CK, Wilkins K, Molter NC, Li F, Yu L, Spott MA, Eastridge B, Blackbourne LH, Hospenthal DR. Infections complicating the care of combat casualties during operations Iraqi Freedom and Enduring Freedom. J Trauma. 2011;71(1 Suppl):S62–S73. [DOI] [PubMed] [Google Scholar]
- 4.Yun HC, Blackbourne LH, Jones JA, Holcomb JB, Hospenthal DR, Wolf SE, Renz EM, Murray CK. Infectious complications of noncombat trauma patients provided care at a military trauma center. Mil Med. 2010;175(5):317–323. [DOI] [PubMed] [Google Scholar]
- 5.Muckart DJ, Bhagwanjee S. American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference definitions of the systemic inflammatory response syndrome and allied disorders in relation to critically injured patients. Crit Care Med. 1997;25(11):1789–1795. [DOI] [PubMed] [Google Scholar]
- 6.Cook A, Norwood S, Berne J. Ventilator-associated pneumonia is more common and of less consequence in trauma patients compared with other critically ill patients. J Trauma. 2010;69(5):1083–1091. [DOI] [PubMed] [Google Scholar]
- 7.Osborn TM, Tracy JK, Dunne JR, Pasquale M, Napolitano LM. Epidemiology of sepsis in patients with traumatic injury. Crit Care Med. 2004;32(11):2234–2240. [DOI] [PubMed] [Google Scholar]
- 8.McDonald JR, Liang SY, Li P, Maalouf S, Murray CK, Weintrob AC, Schnaubelt ER, Kuhn J, Ganesan A, Bradley W, Tribble DR, Infectious Disease Clinical Research Program Trauma Infectious Disease Outcomes Study Group. Infectious complications after deployment trauma: following wounded United States military personnel into Veterans Affairs care. Clin Infect Dis. 2018;67(8):1205–1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Czaja AS, Rivara FP, Wang J, Koepsell T, Nathens AB, Jurkovich GJ, Mackenzie E. Late outcomes of trauma patients with infections during index hospitalization. J Trauma. 2009;67(4):805–814. [DOI] [PubMed] [Google Scholar]
- 10.Hyllienmark P, Brattstrom O, Larsson E, Martling CR, Petersson J, Oldner A. High incidence of post-injury pneumonia in intensive care-treated trauma patients. Acta Anaesthesiol Scand. 2013;57(7):848–854. [DOI] [PubMed] [Google Scholar]
- 11.Tribble DR, Krauss M, Murray CK, Warkentien TE, Lloyd BA, Ganesan A, Greenberg L, Xu J, Li P, Carson ML, Bradley W, Weintrob AC. Epidemiology of trauma-related infections among a combat casualty cohort after initial hospitalization: the Trauma Infectious Disease Outcomes Study. Surg Infect (Larchmt). 2018;19(5):494–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Weintrob AC, Murray CK, Xu J, Krauss M, Bradley W, Warkentien TE, Lloyd BA, Tribble DR. Early infections complicating the care of combat casualties from Iraq and Afghanistan. Surg Infect (Larchmt). 2018;19(3):286–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stewart L, Shaikh F, Bradley W, Lu D, Blyth DM, Petfield JL, Whitman TJ, Krauss M, Greenberg L, Tribble DR. Combat-related extremity wounds: injury factors predicting early onset infections. Mil Med. 2019;184(suppl 1):83–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Petersen K, Riddle MS, Danko JR, Blazes DL, Hayden R, Tasker SA, Dunne JR. Trauma-related infections in battlefield casualties from Iraq. Ann Surg. 2007;245(5):803–811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tribble DR, Li P, Warkentien TE, Lloyd BA, Schnaubelt ER, Ganesan A, Bradley W, Aggarwal D, Carson ML, Weintrob AC, Murray CK. Impact of operational theater on combat and noncombat trauma-related infections. Mil Med. 2016;181(10):1258–1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent JL, Angus DC. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Evans L, Rhodes A, Alhazzani M, Coopersmith CM, French C, Machado FR, McIntyre L, Ostermann M, Prescott HC, Schorr C, Simpson S, Wiersinga WJ, Alshamsi F, Angus DC, Arabi Y, Azevedo L, Beale R, Beilman G, Belley-Cote E, Burry L, Cecconi M, Centofanti J, Coz Yataco A, De Waele J, Dellinger RP, Doi K, Du B, Estenssoro E, Ferrer R, Gomersall C, Hodgson C, Hylander Møller M, Iwashyna T, Jacob S, Kleinpell R, Klompas M, Koh Y, Kumar A, Kwizera A, Lobo S, Masur H, McGloughlin S, Mehta S, Mehta Y, Mer M, Nunnally M, Oczkowski S, Osborn T, Papathanassoglou E, Perner A, Puskarich M, Roberts J, Schweickert W, Seckel M, Sevransky J, Sprung CL, Welte T, Zimmerman J, Levy M. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit Care Med. 2021;49(11):e1063–e1143. [DOI] [PubMed] [Google Scholar]
- 18.Seymour CW, Liu VX, Iwashyna TJ, Brunkhorst FM, Rea TD, Scherag A, Rubenfeld G, Kahn JM, Shankar-Hari M, Singer M, Deutschman CS, Escobar GJ, Angus DC. Assessment of clinical criteria for sepsis: for the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):762–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Giannoudis PV. Current concepts of the inflammatory response after major trauma: an update. Injury. 2003;34(6):397–404. [DOI] [PubMed] [Google Scholar]
- 20.Krebs ED, Hassinger TE, Guidry CA, Berry PS, Elwood NR, Sawyer RG. Non-utility of sepsis scores for identifying infection in surgical intensive care unit patients. Am J Surg. 2019;218(2):243–247. [DOI] [PubMed] [Google Scholar]
- 21.Fueglistaler P, Amsler F, Schuepp M, Fueglistaler-Montali I, Attenberger C, Pargger H, Jacob AL, Gross T. Prognostic value of Sequential Organ Failure Assessment and Simplified Acute Physiology II Score compared with trauma scores in the outcome of multiple-trauma patients. Am J Surg. 2010;200(2):204–214. [DOI] [PubMed] [Google Scholar]
- 22.Champion HR, Holcomb JB, Lawnick MM, Kelliher T, Spott MA, Galarneau MR, Jenkins DH, West SA, Dye J, Wade CE, Eastridge BJ, Blackbourne LH, Shair EK. Improved characterization of combat injury. J Trauma. 2010;68(5):1139–1150. [DOI] [PubMed] [Google Scholar]
- 23.Tribble DR, Conger NG, Fraser S, Gleeson TD, Wilkins K, Antonille T, Weintrob A, Ganesan A, Gaskins LJ, Li P, Grandits G, Landrum ML, Hospenthal DR, Millar EV, Blackbourne LH, Dunne JR, Craft D, Mende K, Wortmann GW, Herlihy R, McDonald J, Murray CK. Infection-associated clinical outcomes in hospitalized medical evacuees after traumatic injury: Trauma Infectious Disease Outcome Study. J Trauma. 2011;71(1 Suppl):S33–S42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tribble DR, Murray CK, Lloyd BA, Ganesan A, Mende K, Blyth DM, Petfield JL, McDonald JR. After the battlefield: infectious complications among wounded warriors in the Trauma Infectious Disease Outcomes Study. Mil Med. 2019;184(Suppl 2):18–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ferreira FL, Bota DP, Bross A, Melot C, Vincent JL. Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA. 2001;286(14):1754–1758. [DOI] [PubMed] [Google Scholar]
- 26.Routsi C, Pratikaki M, Sotiropoulou C, Platsouka E, Markaki V, Paniara O, Vincent JL, Roussoss C. Application of the sequential organ failure assessment (SOFA) score to bacteremic ICU patients. Infection. 2007;35(4):240–244. [DOI] [PubMed] [Google Scholar]
- 27.Raith EP, Udy AA, Bailey M, McGloughlin S, MacIsaac C, Bellomo R, Pilcher DV, Australian, New Zealand Intensive Care Society Centre for Outcomes and Resource Evaluation. Prognostic accuracy of the SOFA Score, SIRS criteria, and qSOFA score for In-Hospital mortality among adults with suspected infection admitted to the intensive care unit. JAMA. 2017;317(3):290–300. [DOI] [PubMed] [Google Scholar]
- 28.Eastridge BJ, Wade CE, Spott MA, Costanzo G, Dunne J, Flaherty S, Holcomb JB, West S, Apodaca A, Blackbourne L, Casscells SW. Utilizing a trauma systems approach to benchmark and improve combat casualty care. J Trauma. 2010;69(Suppl 1):S5–S9. [DOI] [PubMed] [Google Scholar]
- 29.Baker SP, O’Neill B, Haddon W Jr., Long WB. The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma. 1974;14(3):187–196. [PubMed] [Google Scholar]
- 30.Songer T Measuring injury severity: a brief introduction. University of Pittsburgh; 2008. Available at http://www.pitt.edu/~epi2670/severity/severity.pdf; accessed 15 April 2021. [Google Scholar]
- 31.Centers for Disease Control and Prevention. CDC/NHSN Surveillance Definitions for Specific Types of Infections. Available at https://www.cdc.gov/nhsn/pdfs/pscmanual/17pscnosinfdef_current.pdf; accessed 27 May 2021.
- 32.Vincent JL, de Mendonca A, Cantraine F, Moreno R, Takala J, Suter PM, Sprung CL, Colardyn F, Blecher S. Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: results of a multicenter, prospective study. Working group on “sepsis-related problems” of the European Society of Intensive Care Medicine. Crit Care Med. 1998;26(11):1793–1800. [DOI] [PubMed] [Google Scholar]
- 33.Cheng B, Li Z, Wang J, Xie G, Liu X, Xu Z, Chu L, Zhao J, Yao Y, Fang X. Comparison of the performance between sepsis-1 and sepsis-3 in ICUs in China: a retrospective multicenter study. Shock. 2017;48(3):301–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ladhani HA, Sajankila N, Zosa BM, He JC, Yowler CJ, Brandt C, Claridge JA, Khandelwal AK. Utility of Sequential Organ Failure Assessment score in predicting bacteremia in critically ill burn patients. Am J Surg. 2018;215(3):478–481. [DOI] [PubMed] [Google Scholar]
- 35.Edna TH, Bjerkeset T. Association between blood transfusion and infection in injured patients. J Trauma. 1992;33(5):659–661. [DOI] [PubMed] [Google Scholar]
- 36.Papia G, McLellan BA, El-Helou P, Louie M, Rachlis A, Szalai JP, Simor AE. Infection in hospitalized trauma patients: incidence, risk factors, and complications. J Trauma. 1999;47(5):923–927. [DOI] [PubMed] [Google Scholar]
- 37.Claridge JA, Sawyer RG, Schulman AM, McLemore EC, Young JS. Blood transfusions correlate with infections in trauma patients in a dose-dependent manner. Am Surg. 2002;68(7):566–572. [PubMed] [Google Scholar]
- 38.Dunne JR, Riddle MS, Danko J, Hayden R, Petersen K. Blood transfusion is associated with infection and increased resource utilization in combat casualties. Am Surg. 2006;72(7):619–625; discussion 625–626. [PubMed] [Google Scholar]
- 39.Jackman RP, Utter GH, Muench MO, Heitman JW, Munz MM, Jackman RW, Biswas HH, Rivers RM, Tobler LH, Busch MP, Norris PJ. Distinct roles of trauma and transfusion in induction of immune modulation after injury. Transfusion. 2012;52(12):2533–2550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Suksompong S, Tassaneetrithep B, Ariyawatkul T, Sirivanasandha B, Wilartratsami S, Wongsa A, von Bormann B. Allogeneic red cell transfusion and its influence on relevant humoral and cellular immunological parameters: A prospective observational trial. Eur J Anaesthesiol. 2019;36(11):814–824. [DOI] [PubMed] [Google Scholar]
- 41.Wafaisade A, Lefering R, Bouillon B, Sakka SG, Thamm OC, Paffrath T, Neugebauer E, Maegele M, Trauma Registry of the German Society for Trauma Surgery. Epidemiology and risk factors of sepsis after multiple trauma: an analysis of 29,829 patients from the Trauma Registry of the German Society for Trauma Surgery. Crit Care Med. 2011;39(4):621–628. [DOI] [PubMed] [Google Scholar]
- 42.Yun HC, Branstetter JG, Murray CK. Osteomyelitis in military personnel wounded in Iraq and Afghanistan. J Trauma. 2008;64(2 Suppl):S163–S168. [DOI] [PubMed] [Google Scholar]
- 43.Caricato A, Montini L, Bello G, Michetti V, Maviglia R, Bocci MG, Mercurio G, Maggiore SM, Antonelli M. Risk factors and outcome of Acinetobacter baumanii infection in severe trauma patients. Intensive Care Med. 2009;35(11):1964–1969. [DOI] [PubMed] [Google Scholar]
- 44.Griffith ME, Lazarus DR, Mann PB, Boger JA, Hospenthal DR, Murray CK. Acinetobacter skin carriage among US army soldiers deployed in Iraq. Infect Control Hosp Epidemiol. 2007;28(6):720–722. [DOI] [PubMed] [Google Scholar]
- 45.Scott P, Deye G, Srinivasan A, Murray C, Moran K, Hulten E, Fishbain J, Craft D, Riddell S, Lindler L, Mancuso J, Milstrey E, Bautista CT, Patel J, Ewell A, Hamilton T, Gaddy C, Tenney M, Christopher G, Petersen K, Endy T, Petruccelli B. An outbreak of multidrug-resistant Acinetobacter baumannii-calcoaceticus complex infection in the US military health care system associated with military operations in Iraq. Clin Infect Dis. 2007;44(12):1577–1584. [DOI] [PubMed] [Google Scholar]
- 46.Yun HC, Murray CK. Infection Prevention in the Deployed Environment. US Army Med Dep J. 2016(2–16):114–118. [PubMed] [Google Scholar]
- 47.Campbell WR, Li P, Whitman TJ, Blyth DM, Schnaubelt ER, Mende K, Tribble DR, The Infectious Disease Clinical Research Program Trauma Infectious Disease Outcomes Study Group. Multi-drug–resistant Gram-negative infections in deployment-related trauma patients. Surg Infect. 2017;18(3):357–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sutter DE, Bradshaw LU, Simkins LH, Summers AM, Atha M, Elwood RL, Robertson JL, Murray CK, Wortmann GW, Hospenthal DR. High incidence of multidrug-resistant gram-negative bacteria recovered from Afghan patients at a deployed US military hospital. Infect Control Hosp Epidemiol. 2011;32(9):854–860. [DOI] [PubMed] [Google Scholar]
- 49.Hospenthal DR, Murray CK, Andersen RC, Bell RB, Calhoun JH, Cancio LC, Cho JM, Chung KK, Clasper JC, Colyer MH, Conger NG, Costanzo GP, Crouch HK, Curry TK, D’Avignon LC, Dorlac WC, Dunne JR, Eastridge BJ, Ficke JR, Fleming ME, Forgione MA, Green AD, Hale RG, Hayes DK, Holcomb JB, Hsu JR, Kester KE, Martin GJ, Moores LE, Obremskey WT, Petersen K, Renz EM, Saffle JR, Solomkin JS, Sutter DE, Tribble DR, Wenke JC, Whitman TJ, Wiesen AR, Wortmann GW, Infectious Diseases Society of America, Surgical Infection Society. Guidelines for the prevention of infections associated with combat-related injuries: 2011 update: endorsed by the Infectious Diseases Society of America and the Surgical Infection Society. J Trauma. 2011;71(2 Suppl 2):S210–S234. [DOI] [PubMed] [Google Scholar]
- 50.Saeed O, Tribble DR, Biever KA, Crouch HK, Kavanaugh M. Infection prevention in combat-related injuries. Mil Med. 2018;183(suppl 2):137–141. [DOI] [PubMed] [Google Scholar]
- 51.United States Department of Defense. OIF/OEF Casualty Status. Available at http://www.defense.gov/casualty.pdf; accessed 14 June 2021.
- 52.de Grooth HJ, Geenen IL, Girbes AR, Vincent JL, Parienti JJ, Oudemans-van Straaten HM. SOFA and mortality endpoints in randomized controlled trials: a systematic review and meta-regression analysis. Crit Care. 2017;21(1):38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yokoe DS, Anderson DJ, Berenholtz SM, Calfee DP, Dubberke ER, Ellingson KD, Gerding DN, Haas JP, Kaye KS, Klompas M, Lo E, Marschall J, Mermel LA, Nicolle LE, Salgado CD, Bryant K, Classen D, Crist K, Deloney VM, Fishman NO, Foster N, Goldmann DA, Humphreys E, Jernigan JA, Padberg J, Perl TM, Podgorny K, Septimus EJ, VanAmringe M, Weaver T, Weinstein RA, Wise R, Maragakis LL, Society for Healthcare Epidemiology of America. A compendium of strategies to prevent healthcare-associated infections in acute care hospitals: 2014 updates. Infect Control Hosp Epidemiol. 2014;35(8):967–977. [DOI] [PMC free article] [PubMed] [Google Scholar]


