Abstract
We generated random mutations in Synechococcus sp. strain PCC 7942 to look for genes of output pathways in the cyanobacterial circadian system. A derivative of transposon Tn5 was introduced into the chromosomes of reporter strains in which cyanobacterial promoters drive the Vibrio harveyi luxAB genes and produce an oscillation of bioluminescence as a function of circadian gene expression. Among low-amplitude mutants, one mutant, tnp6, had an insertion in a 780-bp open reading frame. The tnp6 mutation produced an altered circadian phasing phenotype in the expression rhythms of psbAI::luxAB, psbAII::luxAB, and kaiA::luxAB but had no or little effect on those of psbAIII::luxAB, purF::luxAB, kaiB::luxAB, rpoD2::luxAB, ndhD::luxAB, and conII::luxAB. This suggests that the interrupted gene in tnp6, named cpmA (circadian phase modifier), is part of a circadian output pathway that regulates the expression rhythms of psbAI, psbAII, and kaiA.
Circadian (daily) rhythms are ubiquitous regulatory functions, found in organisms from bacteria to mammals, that control various biological activities including behavior, metabolism, and gene expression (2, 12, 27, 32). The mechanism of the circadian clock has not been clarified. However, clock function can be modeled as having three common constituents: input pathways, a central oscillator, and output pathways (16). Light and temperature are the environmental stimuli recognized most commonly to act through input pathways to synchronize the phase of the circadian rhythm with environmental cycles. The central oscillator generates and sustains an oscillation that has an approximately 24-h period. The endogenous circadian oscillation is coupled to clock-controlled processes through output pathways.
Several genes that are involved in circadian clock function have been identified in the cyanobacterium Synechococcus sp. strain PCC 7942. Complementation of altered period and arrhythmic mutants and sequence analysis of the relevant loci revealed that all of the mutants carried a point (missense) mutation in one of three genes, designated kaiA, kaiB, and kaiC, which are clustered in the Synechococcus genome (15). Gene disruption and overexpression experiments suggest that the kai genes encode central oscillator components. Another gene, pex, was isolated by suppression of the short-period mutant SP22 (21). Disruption of pex shortens the circadian period and overexpression lengthens the period. The pex product is thought to modulate function of the central clock oscillator. A mutation in the rpoD2 gene, which encodes a group 2 ς70-like transcription factor, causes a low-amplitude phenotype in the psbAI gene’s expression rhythm (34). A similar phenotype was observed for one other gene, ndhD, but the expression rhythms of several other genes were not affected by the mutation. The specificity of the mutation for a subset of genes indicates that the rpoD2 gene is a component of an output pathway in the Synechococcus circadian clock.
Random insertion of promoterless luxAB genes, which encode luciferase from Vibrio harveyi, into the Synechococcus genome revealed robust gene expression rhythms as an output of the circadian clock. The approximately 800 insertion strains tested showed circadian oscillation of luciferase activity, measured as bioluminescence, and none was clearly arrhythmic (23). The genes were grouped into five categories ranging from class 1 to class 5 according to the waveforms of the bioluminescence rhythms: class 1 to class 3 rhythms have symmetrical waveforms but peak in different phases, whereas the others have asymmetric waveforms which are saw-toothed (class 4) or show bimodal peaks per cycle (class 5). These results suggest that there are several pathways of output from the circadian oscillator in this cyanobacterium, each of which generates a customized pattern of circadian expression for its target subset of genes.
Growth competition experiments have suggested that the different phase relationships of the expression of various genes may be important for optimal fitness (26). The relative phasing of circadian oscillations is described by the phase angle of each, or the time between the last synchronizing light-dark cycle and the peak or trough of the endogenous rhythm (27). One goal of our research is to map the output pathways responsible for the different phase angles of the expression rhythms in the Synechococcus genome.
We isolated a transposon-generated mutant of Synechococcus sp. strain PCC 7942, tnp6, which is affected in both amplitude and phasing of the psbAI::luxAB circadian expression rhythm. We examined the mutation’s effect on circadian expression rhythms for several other genes and assigned a function of the tnp6 gene, renamed cpmA (circadian phase modifier), to an output pathway of the cyanobacterial circadian clock. Surprisingly, inactivation of cpmA dramatically changed the circadian phasing of promoter activity for one of the genes thought to encode a central clock component (kaiA::luxAB), but it had little or no effect on that which drives the other two clock genes (kaiB::luxAB). The net effect is a dramatic phase angle difference between these two gene expression oscillations. These data suggest that the coordinated expression of the kai genes is not essential for circadian timekeeping in Synechococcus.
MATERIALS AND METHODS
Strains and culture conditions.
The bacterial strains and plasmids used in this study are listed in Table 1. All reporter strains were derived from Synechococcus sp. strain PCC 7942 by introducing luxAB fusions into the chromosome at neutral site I (NSI [6]) or, for purF::luxAB and in AMC393, at neutral site II (NSII [1]). ndhD::luxAB was introduced as a duplication at the native ndhD locus. All reporter genes are transcriptional fusions except two: kaiA and psbAII are translationally fused to luxA in AMC509 and AMC520, respectively. All Synechococcus strains were grown on modified BG-11 medium (BG-11M [5]), in liquid cultures or on agar plates (11), under continuous light (LL) at 30°C. Chloramphenicol (7.5 and 17 μg/ml) and/or spectinomycin (5 μg/ml and 20 μg/ml) was added to select for NSI and NSII targeting vectors in Synechococcus and in Escherichia coli, respectively. The tnp6 mutants were selected in the presence of either kanamycin (5 μg/ml for the psbAI::luxAB, psbAII::luxAB, psbAIII::luxAB, kaiA::luxAB, kaiB::luxAB, rpoD2::luxAB, and conII::luxAB strains) or spectinomycin (10 μg/ml for the ndhD::luxAB strain).
TABLE 1.
Strains and plasmids
| Strain or plasmid | Characteristics or genotype |
|---|---|
| E. coli strains | |
| AM1452 | HB101 containing helper plasmid pRL528a and transposon plasmid pAM1037 |
| AM1460 | HB101 containing plasmid pRK2013b to provide conjugal transfer functions |
| DH10B | Host for plasmids |
| Synechococcus strainsc | |
| PCC 7942 | Wild-type Synechococcus |
| AMC149 | psbAI::luxAB reporter gene fusion inserted into NSId |
| AMC393 | psbAI::luxAB reporter gene fusion inserted into NSIIe (Cmr) psbAI::luxCDE fusion inserted into NSI (Spr) |
| AMC408 | purF::luxAB reporter gene fusion inserted into NSIIe (Cmr) psbAI::luxCDE fusion inserted into NSI (Spr) |
| AMC412 | Derivative of AMC149 with psbAI::luxCDE fusion inserted into NSII (from pAM1706) |
| AMC412(cpmA::Km) | Derivative of AMC412 in which cpmA has been inactivated by recombination with pAM2087 (Kmr) |
| AMC462 | kaiBC::luxAB reporter gene fusion inserted into NSI |
| AMC509 | kaiA::luxAB reporter gene fusion (translational) inserted into NSI |
| AMC520 | psbAII::luxAB reporter gene fusion (translational) inserted into NSI |
| AMC537 | psbAIII::luxAB reporter gene fusion inserted into NSI |
| AMC538 | rpoD2::luxAB reporter gene fusion inserted into NSI |
| AMC539 | conII::luxAB reporter gene fusion inserted into NSI |
| AMC540 | Derivative of AMC149 with psbAI::luxCDE fusion and an ectopic copy of cpmA inserted into NSII (from pAM2089) |
| AMC540(cpmA::Km) | Derivative of AMC540 in which cpmA has been inactivated by recombination with pAM2087 (Kmr) |
| AMC543 | psbAI::luxAB reporter gene fusion inserted at BstEII site (Kmr selection) and psbAI::luxCDE inserted at BglII site (Cmr selection) of NSII |
| AMC550 | Derivative of AMC543 in which an IPTG-inducible ectopic copy of cpmA is inserted at NSI (from pAM1850) |
| LUX6 | ndhD::luxAB reporter gene fusion; a 3.0-kb genomic DNA fragment including ndhD is integrated into the ndhD locus by single recombination |
| Plasmids | |
| pAM1037e | Tn5 plasmid derivative pRL1058, further modified by insertion of a 0.7-kb fragment in the XbaI site to add outward-reading promoters from the Anabaena sp. strain PCC 7120 glnA and rbcL genes |
| pAM1706 | Vector that transfers psbAI::luxCDE fusion to NSII (subsite NS2.1, BstEII) of the Synechococcus chromosome |
| pAM1825 | NSI vector that carries lacIq, IPTG-inducible trc promoter, and ribosome-binding site upstream of unique NcoI and SmaI cloning sites |
| pAM1850 | Vector that transfers psbAI::luxCDE fusion to NSII (subsite NS2.2, BglII) of the Synechococcus chromosome |
| pAM2086 | pUC18 containing 0.9-kb EcoRI fragment including cpmA ORF in the same orientation as the lacZ promoter |
| pAM2087 | 46-bp NruI fragment of pAM2086 was replaced by a Kmr cassette |
| pAM2088 | 46-bp NruI fragment of pAM2086 was replaced by an Spr-Smr cassette |
| pAM2089 | Derivative of pAM1706 that carries a PlacZ::cpmA fusion downstream of psbAI::luxCDE |
| pAM2107 | Entire cpmA ORF inserted into pBR322 at the EcoRV site |
| pAM2108 | 0.9-kb EcoRI fragment from pAM2107 (with ends modified as described in Materials and Methods) inserted into pAM1825 for IPTG-inducible expression of cpmA |
Reference 8.
Reference 7.
All Synechococcus strains carry a psbAI::luxCDE fusion in NSII except PCC 7942, AMC149, AMC393, and AMC408; all except PCC 7942 and AMC149 are bioluminescent without exogenously added aldehyde substrate. All insertions into NSI were selected by Spr; all insertions into NSII were selected by Cmr, except as otherwise noted for AMC543 and ACM550.
Reference 19.
Reference 1.
Transposon mutagenesis.
pAM1037, which is a derivative of pRL1058 (36), was introduced into Synechococcus by conjugal transfer from E. coli (1). AMC149, a psbAI::luxAB reporter strain of Synechococcus (19), was grown to an optical density at 750 nm of 0.5. Cells from a 100-ml sample were harvested by centrifugation at 2,000 × g and resuspended in 10 ml of fresh BG-11M. Ten-milliliter overnight cultures of E. coli AM1452, which carries the helper plasmid pRL528 and the transposon plasmid pAM1037, and of E. coli AM1460, which carries the conjugal plasmid pRK2013, were each washed twice with 10 ml of fresh Luria-Bertani (LB) medium and resuspended in 10 ml of sterile water. Ten milliliters of Synechococcus cell suspension and 5 ml of each E. coli cell suspension were mixed, and 400-μl aliquots of the mixture were spread onto plates containing BG-11M plus 5% LB medium (vol/vol) (1). The plates were kept under dim light (10 to 30 μE · m−2 · s−1) at 30°C for 24 h. Kanamycin was underlaid beneath the agar (11) for a final concentration of 50 μg/ml, and the plates were incubated at 30°C under 250 μE · m−2 · s−1 for 4 weeks. Km-resistant (Kmr) transformants were streaked onto fresh BG-11M–kanamycin (50 μg/ml) plates, incubated for an additional 2 weeks, and then inoculated into 250 μl of BG-11 liquid medium containing 5 μg of kanamycin/ml in 96-well microtiter plates.
Cloning and sequencing of the tnp6 region.
Restriction enzymes and most modifying enzymes were purchased from Promega and used as directed by the manufacturer. Recovery of the transposon is described in the Results section. One cosmid clone, P2-C3, was isolated from a cosmid library of Synechococcus sp. strain PCC 7942 by DNA hybridization with a probe made from the flanking region of the recovered transposon (3). An EcoRI fragment of 0.9 kb which hybridized to the probe was cloned into the EcoRI site of pUC18 to produce pAM2086. We used this plasmid as a template for sequencing the tnp6 locus. Insert DNA was sequenced on both strands by using the cycle sequencing method (dye terminator cycle sequencing ready reaction, ABI PRISM; PE Applied Biosystems, Foster City, Calif.).
Construction of plasmids for disruption and complementation of cpmA.
We digested pAM2086 with NruI and inserted a 2.0-kb HincII fragment from pSKS101, including the Kmr gene, to produce pAM2087 (see Fig. 2B). We used pAM2087 to create the cpmA null mutation in psbAI::luxAB, psbAII::luxAB, psbAIII::luxAB, kaiA::luxAB, kaiB::luxAB, purF::luxAB, rpoD2::luxAB, and conII::luxAB reporter strains. We digested pAM2086 with NruI and inserted a 2.1-kb SmaI fragment from pHP45Ω (28), which carries the spectinomycin-streptomycin resistance Ω fragment (Spr fragment), to produce pAM2088. We used this plasmid to introduce the cpmA mutation into the ndhD::luxAB reporter strain, which already encodes Kmr (34).
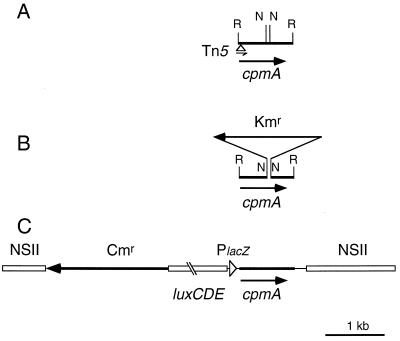
FIG. 2.
Chromosomal maps of specific loci from recombinant cyanobacterial strains. Shown are physical maps of original tnp6 mutant of Synechococcus (A), the inactivated cpmA locus in which an NruI fragment was replaced by a Kmr gene cartridge (B), and the NSII loci in AMC540 and AMC540(cpmA::Kmr) (C).
For complementation of the cpmA mutation, a 1.4-kb PvuII fragment in which the cpmA gene is preceded by the lacZ promoter, in the same orientation, was excised from pAM2086, ligated with the KpnI linker 5′-GGGTACCC-3′, and cloned into the KpnI site of pAM1706 to produce pAM2089. In this plasmid the cpmA gene was inserted downstream of the luxCDE genes in the same orientation (see Fig. 2C). We transformed wild-type AMC149 with pAM2089 to create AMC540 and then transformed this strain with pAM2087. Transformants that carried the cpmA::Kmr mutation in the original cpmA locus, and an ectopic copy of cpmA in NSI, were identified by genomic Southern hybridization (3) and assayed for their circadian phenotypes. One such clone was designated AMC540(cpmA::Kmr).
Overexpression of cpmA.
A DNA fragment which includes the initiation codon and N-terminal coding portion of the cpmA gene was amplified by PCR with the primer set 5′-GTGCTCATGATTGATTCACAG-3′ (AMO282) and 5′-CCTGAAGTCGGGGAATGCG-3′ (AMO283). This 288-bp fragment was made blunt ended by T4 DNA polymerase and cloned into the EcoRV site of pBR322; a 0.3-kb BanII fragment of this plasmid was then replaced by the 1.1-kb BanII fragment of pAM2086 to complete the gene and produce pAM2107. A 0.9-kb EcoRI fragment was removed from pAM2107, blunted with T4 DNA polymerase, digested with BspHI, and cloned into NcoI-SmaI sites of pAM1825 to produce pAM2108. pAM1825 is an NSI targeting vector that includes the lacIq gene, the isopropyl-β-d-thiogalactopyranoside (IPTG)-inducible trc promoter, and a Shine-Dalgarno ribosome-binding sequence (30). pAM2108 was introduced into the psbAI::luxAB reporter strain AMC543 to produce AMC550. For induction of cpmA expression, samples were inoculated onto BG-11M agar pads containing 1 mM IPTG (Research Products International Corp., Mt. Prospect, Ill.), incubated in LL for 24 h, and entrained by a 12-h dark pulse before bioluminescence measurement began.
Assay of bioluminescence rhythms.
The original tnp6 mutant was identified by continuous bioluminescence monitoring of Tn5-generated mutants by using a turntable apparatus described previously (20). The genetic background, AMC149, carried psbAI::luxAB; decanal was added exogenously in the vapor phase as a substrate for luciferase (20). When the mutation was recreated in AMC149 and other reporter strains, long-chain aldehyde production was engineered in vivo by insertion of the luxCDE genes of Xenorhabdus luminescens, driven by the psbAI promoter, at NSII (1). Cultures grown on BG-11M agar in LL were inoculated onto BG-11M agar in 96-well microtiter plates. An antibiotic (kanamycin or spectinomycin, as appropriate) was included in the agar for monitoring of cpmA-inactivated strains. These sample plates were incubated for 24 h under standard LL conditions and then subjected to a 12-h dark interval to synchronize the clocks of all cells in the population. Bioluminescence rhythms were measured by using a Packard TopCount luminometer (1). Measurements of bioluminescence from firefly luciferase fusions were performed similarly, except that the reporter strains did not carry luxCDE genes for aldehyde production; luciferin (Biosynth International, Naperville, Ill.) was added to the top of the agar in each well for a final concentration of 190 nM.
Nucleotide sequence accession number.
The cpmA gene sequence was entered into the GenBank database (accession no. AF117208).
RESULTS
Isolation of the mutant tnp6.
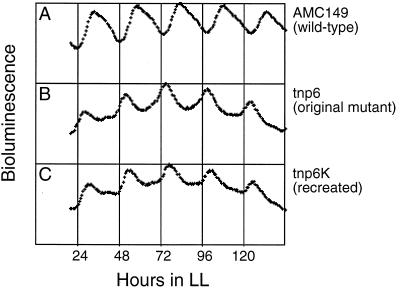
We generated random transposon mutations in a bioluminescent reporter strain of Synechococcus sp. strain PCC 7942 (AMC149) to isolate mutants affected in circadian clock function. The transposon was delivered by conjugal transfer from E. coli of pAM1037, which carries a Tn5 derivative to which the tandem rbcL and glnA promoters of Anabaena sp. strain PCC 7120 have been added at one end of the transposon (1). In addition to simple gene disruption, the modified Tn5 can, theoretically, cause the over- or underexpression of adjacent genes by the activities of these strong outward-reading promoters. The modified Tn5 also bears a p15A replication origin to facilitate recovery in E. coli after genomic insertion (36). AMC149 carries a reporter gene in which the promoter region of psbAI, which encodes form I of the photosystem II D1 protein, is fused to promoterless luxAB genes of V. harveyi (19). AMC149 shows a circadian bioluminescence rhythm that has peaks at subjective dusk and troughs at subjective dawn (Fig. 1A). Among approximately 3,000 independent Kmr exconjugants, we identified a mutant, tnp6, which showed both a low-amplitude oscillation and an early-phase-angle phenotype (Fig. 1B).
FIG. 1.
Bioluminescence traces from the tnp6 mutant of Synechococcus AMC149. Bioluminescence was measured from streaks on agar plates of wild-type AMC149 (A); original tnp6 mutant (B); and recreated tnp6 mutant, tnp6K (C). Time shown on the x axis refers to hours in LL after a synchronizing 12-h dark incubation. The y axis indicates bioluminescence (counts/3-min bin) from baseline of 0 to maxima of 101,000 (panel A), 57,300 (panel B), and 64,100 (panel C) as detected by a cooled charge-coupled device camera (20).
Genomic DNA was extracted from tnp6 and digested by StuI, which does not cut within the transposon. Ligated genomic DNA was used to transform E. coli. The transposon, flanked by approximately 24 kb of Synechococcus genomic DNA, was recovered as a Kmr plasmid. We transformed wild-type AMC149 with the recovered Kmr plasmid, which had been linearized by digestion with StuI, to recreate the tnp6 mutant. The recreated mutant, tnp6K, showed the same phenotype as the original tnp6 mutant (Fig. 1C), confirming that the phase change resulted from the transposon insertion rather than from an unrelated secondary mutation. In addition to the circadian phenotype, tnp6 and tnp6K had a paler green color and slower growth on BG-11M agar medium than the wild type under light at 100 to 150 μE · m−2 · s−1. Under stronger light conditions (250 to 300 μE · m−2 · s−1) or in liquid media, revertants that showed wild-type color and growth rate and normal phasing and amplitude of circadian bioluminescence oscillation were readily obtained. We routinely maintained tnp6 mutant strains by streaking on BG-11M agar medium with selective antibiotics under light at 100 to 150 μE · m−2 · s−1, and we did not observe reversion of any of the mutant phenotypes under these conditions.
Sequencing the tnp6 locus and determining the insertion site of the transposon.
We determined the insertion site of Tn5 from the recovered transposon-based plasmid using a primer that is complementary to one end of the transposon and reads outward into the flanking Synechococcus DNA. The wild-type locus was isolated from a cosmid library by using the recovered tnp6 plasmid as a probe. Southern hybridization against DNA from the cosmid clone indicated that the transposon inserted into the chromosome in a region, of which we subsequently determined the nucleotide sequence, bounded by EcoRI sites 0.9 kb apart (Fig. 2A). The EcoRI fragment included a 780-bp open reading frame (ORF). Comparison with the sequence we obtained from the recovered transposon showed that the insertion occurred at nucleotide 34 of this ORF. The direction of transcription of the tandem glnA and rbcL promoters on the transposon was the same as that of the 780-bp ORF. We designated this ORF the cpmA gene (circadian phase modifier).
Database searches showed that the deduced CpmA protein has extensive sequence similarity to predicted proteins of unknown function that have been identified through total genome sequence determinations (Fig. 3). CpmA has the following degrees of identity to hypothetical proteins: 53.8% identity to sll1489 of Synechocystis sp. strain PCC 6803 (17), 53.2% identity to MJ0165 of Methanococcus jannaschii (4), 42.0% identity to the phosphoribosylaminoimidazole carboxylase-related protein of Methanobacterium thermoautotrophicum (31), and 40.6% identity to AF1275 of Archaeoglobus fulgidus (18). The degrees of identity among these amino acid sequences, along their entire lengths, and striking blocks of identical residues, suggest that they are CpmA homologs. The putative CpmA protein also shows some sequence similarity to the purE product from diverse prokaryotes (Fig. 3). purE encodes the catalytic subunit of the de novo purine biosynthesis enzyme 5′-phosphoribosyl-5-amino-4-imidazole (AIR) carboxylase, which catalyzes the carboxylation of AIR to 5′-phosphoribosyl-5-aminoimidazole-4-carboxylic acid (33, 35). The hydrophobicity profile of CpmA indicates that there are two hydrophobic regions (amino acids 185 to 205 and 214 to 234 in the sequence of Synechococcus CpmA) (Fig. 3) in the part of the protein that is similar to PurE. These hydrophobic regions exist in the putative cyanobacterial and archaeal CpmA homologs as well.
FIG. 3.
Comparison of amino acid sequences including that deduced from the cpmA gene of Synechococcus sp. strain PCC 7942 (Synco-CpmA), putative cpmA homologs from Synechocystis sp. strain PCC 6803 (Syncy-CpmA), M. jannaschii (Metco-CpmA), M. thermoautotrophicum (Metba-CpmA), and A. fulgidus (Arcog-CpmA), and the sequences of PurE proteins of Synechocystis sp. strain PCC 6803 (Syncy-PurE), M. jannaschii (Metco-PurE), Mycobacterium leprae (Mycba-PurE), and E. coli (Esche-PurE). Residues identical among all CpmA (but not all PurE) sequences are boxed; those common to all aligned sequences are shown in boldface.
Regeneration and complementation of the tnp6 mutation.
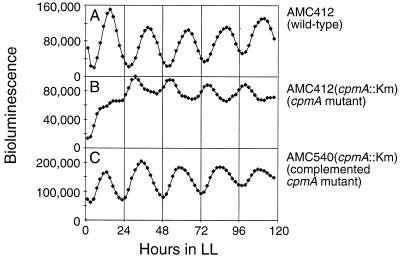
The transposon inserted close to the initiation codon (position 12 in the amino acid sequence) of cpmA and the direction of transcription from the strong glnA-rbcL promoter on the transposon were the same as those of cpmA (Fig. 2A). Therefore, it was considered possible that the phenotype of tnp6 was caused either by overexpression of a portion of cpmA or by interference with the transcription of a downstream gene. To exclude these possibilities, we generated a cpmA null mutation by replacement of the internal NruI fragment with a Kmr cartridge in the psbAI::luxAB reporter strain; the orientation of the Kmr gene was reversed relative to that of cpmA (Fig. 2B). This mutant (Fig. 4B) showed the same circadian phenotype as the original tnp6 mutant (Fig. 1B). This result confirmed that disruption of expression, rather than overexpression, can cause the phenotype.
FIG. 4.
Loss of cpmA function causes low amplitude and early phase angle in the psbAI::luxAB bioluminescence rhythm. Bioluminescence traces were obtained from AMC412, the wild-type strain (A), AMC412(cpmA::Kmr), the mutant in which the cpmA gene was inactivated by the replacement of an internal NruI fragment with a Kmr gene cartridge (B), and AMC540(cpmA::Km), the cpmA mutant that carries an ectopic wild-type allele of cpmA in trans (C).
We provided the cpmA gene in trans to determine whether loss of function of this gene alone was responsible for the mutant phenotype. Sequence data showed that cpmA is located 21 bp downstream of an ORF that is similar to the oligopeptide transport permease gene of Synechocystis sp. strain PCC 6803 (sll1699; data not shown) and transcribed in the same direction. In addition, a 1.0-kb EcoRI fragment immediately upstream of cpmA did not drive a luxAB reporter gene in Synechococcus above the basal level of bioluminescence (data not shown). Therefore, we considered it likely that cpmA is part of an operon together with this upstream ORF and does not have its own promoter. We removed cpmA from pAM2086 in such a way as to provide the lacZ promoter from the pUC18 vector and cloned it into an NSII vector (pAM1706) downstream of the luxCDE genes (Fig. 2C), which direct the synthesis of the luciferase long-chain aldehyde substrate in vivo. We transformed AMC149 with this plasmid (pAM2089) to create AMC540 (which carries both native and ectopic copies of cpmA) and in parallel with pAM1706 (to create the autonomously bioluminescent wild-type reporter strain AMC412). The cpmA null mutation was then introduced into both genetic backgrounds to create strains AMC540(cpmA::Km) and AMC412(cpmA::Km), respectively. Southern blot analysis was performed to distinguish whether the disrupted cpmA gene replaced the native or ectopic cpmA copy in the AMC540 background. The mutation was created after addition of the ectopic copy because the reversion frequency of the cpmA mutant in liquid culture precluded transformation of that strain.
Figure 4 shows bioluminescence traces of the wild-type AMC412 (Fig. 4A), the cpmA mutant AMC412(cpmA::Km) (Fig. 4B), and the complemented strain AMC540(cpmA::Km) (Fig. 4C). The phase and amplitude of the bioluminescence rhythm in the complemented strain were almost identical to those of the wild-type strain (Fig. 4A). The complemented strain also had wild-type pigmentation and growth rate. These data showed that the loss of function of the cpmA gene was responsible for the phenotypes of the tnp6 mutant.
Three mechanisms could account for the high frequency of reversion observed for the cpmA phenotypes when cells were exposed to higher light intensities. (i) The transposon (or antibiotic resistance cassette) might be lost somehow. (ii) Wild-type chromosomes may persist in the cells in addition to cpmA-inactivated genomes. In either case, degradation of the selecting antibiotics during incubation might allow wild-type (or reverted) segregants to overgrow the population. Alternatively, (iii) mutation at a second locus in the presence of a fully-segregated inactive cpmA gene (pseudoreversion) might be responsible for loss of the mutant phenotypes.
We confirmed the complete segregation of the cpmA mutation in AMC412(cpmA::Km) by genomic Southern hybridization with the 0.9-kb EcoRI fragment (cpmA) used as a probe (data not shown). We extracted genomic DNA after several subcultures on BG-11M–kanamycin plates and digested it with EcoRV. We detected a 4.5-kb band in the wild-type strain and a 6.5-kb band, larger than the wild-type band by the size of the Kmr gene cartridge, in the cpmA mutant. No wild-type band was detectable in the mutant (data not shown).
We also removed selective pressure to determine whether wild-type genomes, which could segregate in the absence of antibiotics, persisted, at a level not detectable by Southern analysis. We streaked AMC412(cpmA::Km) onto BG-11M plates that lacked antibiotics to obtain single colonies and then transferred 100 colonies to a plate containing BG-11M and kanamycin to assess antibiotic resistance. All colonies grew in the presence of kanamycin, indicating that the cpmA-disrupting insertion was intact and supporting the Southern analysis data that showed complete segregation of the mutant allele. However, all colonies had normal pigmentation and growth rate, and the circadian phenotypes of the six clones tested were wild type. Southern analysis of these clones again confirmed the absence of a wild-type cpmA locus (data not shown). Thus, the Kmr clones that show wild-type phenotypes are pseudorevertants rather than true revertants of the original mutation. We concluded that the gene is not essential for viability of Synechococcus, that cpmA disruption instead causes a severe growth defect, and that there is a strong selection for a second mutation that reverts the phenotype under all conditions that expose cells to high light intensity. Plating for single colonies, like suspension in liquid culture or incubation under strong light, seems to increase the amount of irradiation received by individual cells. The nature of the selective pressure is not yet obvious.
Effect of cpmA inactivation on the circadian expression rhythms of other genes.
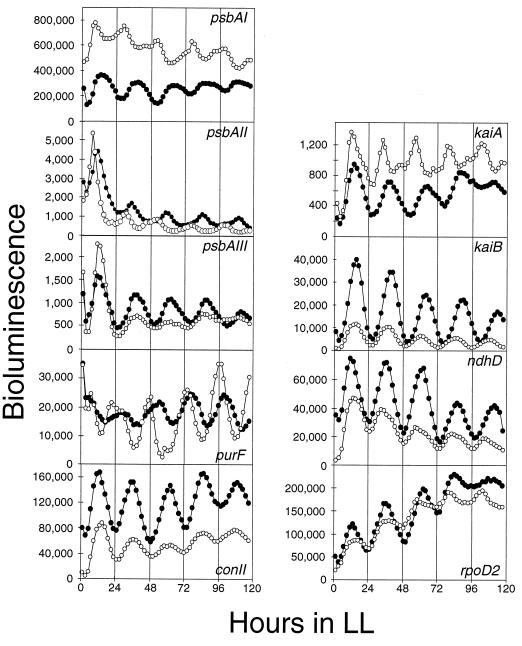
Most, if not all, genes in Synechococcus show a circadian rhythm of expression, and the majority of these rhythms are in the same phase, having a peak at subjective dusk and a trough at subjective dawn (class 1 rhythm) (23). Exceptionally, the expression rhythm of purF, which encodes a regulatory enzyme in the de novo purine synthetic pathway, glutamine phosphoribosyl pyrophosphate amidotransferase, shows a rhythm with an opposite phase (class 2) (22). We were interested in whether mutation of cpmA affects the expression rhythms of different genes in Synechococcus in the same way. We introduced the cpmA disruption into several reporter strains in which luxAB is driven by the following promoters: psbAII and psbAIII, which encode form II of the photosystem II reaction center D1 protein (10, 29); purF; kaiA and kaiB, which encode circadian clock component proteins (15); ndhD, which encodes subunit D of NADH dehydrogenase (34); rpoD2, which encodes a group 2 ς70-like transcription factor that has been shown to influence the expression rhythm of psbAI (34); and the artificial promoter, conII, whose sequence matches the E. coli −35 and −10 elements (9).
Figure 5 shows traces of the bioluminescence rhythms from each reporter strain. The cpmA mutation changed the phase angles in psbAII and kaiA expression rhythms by about 10 h, as was seen for the psbAI expression rhythm (Fig. 4A and 5). The mutation had little or no effect on the phases of the expression rhythms of psbAIII, purF, kaiB, ndhD, rpoD2, and conII (Fig. 5). Compared with the effect on phase, the effect on amplitude was less clear. The cpmA mutation reproducibly lowered the expression rhythm amplitude for psbAI, kaiA, and kaiB and increased the amplitude for purF (Fig. 5). The effect of cpmA disruption on amplitude was not consistent for the expression rhythms of the other promoters.
FIG. 5.
Effect of cpmA inactivation on the expression rhythms of various reporter genes. Closed circles, wild-type traces; open circles, cpmA mutant traces. The x axis is labeled as for Fig. 1. The y axis values are counts per second. A representative trace is shown for each genotype, and the identity of the Synechococcus gene fused to luxAB is indicated for each panel. The fraction of independent experiments and the fraction of total independent colonies for which marked phase-change phenotypes (several hours’ phase angle difference) were observed, respectively, follow in parentheses for each reporter strain: psbAI::luxAB (9/10; 37/48), psbAII::luxAB (6/10; 22/46), psbAIII::luxAB (1/6; 4/31), purF::luxAB (0/9; 0/41), kaiA::luxAB (8/9; 38/42), kaiB::luxAB (0/10; 0/47), rpoD2::luxAB (3/9; 12/31), ndhD::luxAB (1/9; 1/37), and conII::luxAB (2/7; 10/35) reporter fusions.
The product of the luxAB genes, bacterial luciferase, requires reduced flavin mononucleotide (FMNH2) and a long-chain fatty aldehyde as substrates for bioluminescence (13). FMNH2 is produced in the cyanobacterium by photosynthesis, and synthesis of the aldehyde is directed by the luxCDE genes which we introduced into the reporter strain at a neutral site independent of the luxAB reporter fusion (1). Therefore, it was considered possible that the phenotype observed in cpmA mutants was caused by an effect on the synthesis of the luciferase substrates. We tested whether the cpmA disruption caused the same phenotype when the psbAI expression rhythm was measured by a different reporter. Firefly luciferase is another real-time reporter that has a half-life sufficiently short for visualization of circadian troughs (25). Firefly luciferase, like bacterial luciferase, is an oxidase, but its other substrate requirements are different, being ATP and a molecule known as a luciferin (added exogenously). We introduced the cpmA disruption into strains in which the psbAI or purF promoter is fused to a promoterless firefly luciferase gene (psbAI::luc and purF::luc, respectively). The results for the luc reporter strains were consistent with those for luxAB reporter strains: early phase angle of the expression rhythm of psbAI and little or no effect on the phase angle of the rhythm of purF (data not shown). We concluded that the effect of cpmA inactivation is a genuine change in the phasing of psbAI expression rather than a modification of bioluminescence through alteration of substrate levels.
Overexpression of cpmA.
An IPTG-inducible promoter was used to drive expression of an ectopic copy of cpmA in an otherwise wild-type psbAI::luxAB reporter strain. No alteration of the wild-type circadian bioluminescence pattern was observed in either the presence or absence of IPTG (data not shown).
DISCUSSION
We concluded that cpmA is involved in an output pathway of the circadian clock, rather than an input pathway or the central oscillator itself, from the following observations. Mutation of cpmA altered the phasing of the circadian rhythm for a subset of genes; it changed the phase angles of the psbAI, psbAII, and kaiA expression rhythms dramatically, but it had little effect on those of psbAIII, purF, rpoD2, ndhD, and conII. Theoretically, a change in the central oscillator should be reflected in all downstream behaviors. Also, cpmA inactivation did not affect the phase of the expression rhythm of the kaiB promoter, which directs a dicistronic message that encodes KaiB and KaiC. It has been suggested that KaiC functions as a central clock component in this organism because disruption of kaiC eliminates the circadian rhythm, missense mutations in kaiC change the periods of all gene expression rhythms in the cell, and pulsed overexpression of kaiC can shift the phase of the circadian rhythm (15).
It is surprising that the cpmA mutation dramatically changed the phase of the kaiA expression rhythm (phase angle difference of approximately 10 h) but had a very minor effect on the phase of the kaiB expression rhythm. Disruption of kaiA eliminates the circadian rhythm of kaiBC expression, and overexpression of kaiA leads to high-level and low-amplitude expression of kaiBC, suggesting that kaiA functions as an activator of the kaiBC promoter (15). Furthermore, some period-altering mutations map to kaiA, which is one criterion for its role as a central clock component; if it is such a component, the relative concentrations of KaiA and KaiC at different times during the circadian cycle should be important for sustaining the rhythm. In the cpmA mutant background, the kaiA and kaiB promoters are out of phase by 8 h or more. Our results indicate that synchronization of the phase of the circadian expression rhythms of kaiA and kaiBC may not be necessary for normal clock function.
It is not clear that the early-phase-angle and low-amplitude phenotypes are closely related. Changes in amplitude resulting from cpmA inactivation were less reproducible than the early-phase-angle phenotype. Both phenotypes were always observed in the psbAI reporter strain. However, the low-amplitude phenotype was not reproducibly observed in the psbAII and kaiA rhythms, even though their phasing was affected by the mutation. Furthermore, sometimes the low-amplitude phenotype was observed in the expression rhythms of psbAIII, kaiB, and conII, whose phases were not markedly affected by the mutation. For the purF rhythm, this mutation reliably increased the amplitude of the oscillation.
In this organism the expression of the conII promoter fragment, an entirely heterologous sequence that includes the consensus elements recognized by the E. coli ς70 subunit, shows circadian oscillation with class 1 phase and waveform. This result suggests that the class 1 rhythm is generated through the basic transcription machinery as an original mode of the circadian expression rhythm and that other components in specific output pathways modify this rhythm to generate the different waveforms represented by the different classes (23). Mutation of cpmA dramatically affected the phase relationships of a subset of class 1 gene expression rhythms, those of psbAI, psbAII and kaiA, but had only a modest effect on the phase angles of the conII expression rhythm or other class 1 rhythms, such as those of psbAIII, kaiB, ndhD, and rpoD2. This indicates that the category of class 1, identified as constituting up to 85% of the expression patterns of the genome, actually encompasses two or more subclasses that are controlled by different output pathways. Mutation of cpmA did not affect the phase of the class 2 purF expression rhythm, which is opposite in phase to class 1. This confirms the separation of class 2 from class 1 as being controlled by independent output pathways. In all, our results indicate that cpmA functions in an output pathway that modulates the phasing (and amplitude) of a subset of class 1 rhythms.
Another Synechococcus gene, rpoD2, was previously identified as a component of an output pathway of the circadian clock (34). Disruption of rpoD2 decreases the amplitude of the psbAI expression rhythm. The rpoD2 sequence predicts a product that is a group 2 ς factor of RNA polymerase. Generally, cyanobacteria have, in addition to a group 1 ς factor which is essential for exponential growth, multiple group 2 sigmas that are dispensable but have extensive sequence similarity to the group 1 ς factor. Tsinoremas et al. suggested a model in which temporal incorporation of the RpoD2 sigma factor into RNA polymerase decreases the expression of psbAI during subjective night and generates the high-amplitude circadian oscillation of psbAI expression (34). Inactivation of rpoD2 decreases the amplitude of the expression rhythm of ndhD as well as that of psbAI but does not affect the purF expression rhythm (34). The effect of the cpmA mutation was different in that it affected phase as well as amplitude of the psbAI rhythm and separated ndhD and psbAI into separate subclasses.
Constitutive high expression of cpmA did not affect the phase or amplitude of the psbAI expression rhythm. This result suggests that temporal oscillations in the amount of the cpmA product are not necessary to determine the correct amplitude and phase. This contrasts with the case of components that are involved in circadian output pathways in other organisms. The expression of the Atgrp7 gene, encoding a glycine-rich RNA-binding protein of Arabidopsis thaliana, shows circadian oscillation in its mRNA and protein levels; overexpression of AtGRP7 greatly suppresses and eliminates the circadian oscillation of Atgrp7 expression (14). In another example, the lark gene of Drosophila melanogaster encodes an RNA-binding protein that is thought to function as a negative repressor of the circadian eclosion rhythm (24). lark/+ heterozygotes, which have a decreased amount of Lark protein, have an early-eclosion phenotype, and an increase in gene dosage of lark causes a late-eclosion phenotype. In both cases the overexpression of the output component disrupts the normal circadian output pathway.
Database searches revealed that a cyanobacterium, Synechocystis sp. strain PCC 6803, and three species of Archaea, M. janaschii, M. thermoautotrophicum, and A. fulgidus, have a gene that is very similar to cpmA. It is interesting that all of these organisms have a putative kaiC homolog. Because KaiC may function as a central clock component in the circadian system of Synechococcus, it is possible that the kaiC genes in other prokaryotes also encode a clock component and that cpmA is a circadian output factor in those organisms as well. Because all of these organisms also carry purE homologs, we expect that CpmA will be found to function distinctly from PurE; however, it is likely to have a similar biochemical activity. The nature of this activity may be more apparent when the locus or loci that allow pseudoreversion of the cpmA phenotypes are identified.
Mutation of cpmA caused a severe growth defect in the cyanobacterium. One possible explanation is that its product is involved in basic cell metabolism. CpmA may have a carboxylase activity similar to that of PurE, such that its loss causes disruption of an unidentified metabolic pathway. Another possibility is that the detrimental effect on growth is a more direct result of a circadian role for CpmA because loss of this function causes a conflict among gene expression rhythms of different phases in this organism. In unicellular cyanobacteria some processes that are incompatible are known to be separated temporally. For example, in unicellular diazotrophic cyanobacteria, the oxygen-sensitive enzyme nitrogenase, which is irreversibly inactivated by exposure to oxygen, is expressed in the night, in phase opposite to those of expression rhythms of (oxygen-evolving) photosynthesis-related genes (12). In Synechococcus the expression rhythm of purF, which encodes the oxygen-sensitive enzyme glutamine phosphoribosyl pyrophosphate amidotransferase, is expressed in phase (class 2 phase) opposite to that of psbAI, which encodes the photosystem II reaction center D1 protein. Furthermore, bioluminescence patterns following random insertion of promoterless luxAB genes suggest that this organism has several genes expressed with a class 2 phase (23). It is possible that the activities of the enzymes which are specifically expressed in the night, like purF, are inhibited by the approach of the peak of a subset of class 1 rhythms, including the rhythms of the photosynthesis genes psbAI and psbAII, into their phase.
ACKNOWLEDGMENTS
We thank Yao Ouyang and Carl Johnson for the purF::luc and psbAI::luc reporter plasmids and Hiroshi Yamamoto and Norio Murata for the IPTG-inducible NSI-targeting vector entered into our strain collection and designated here as pAM1825. We also thank Vincent Cassone, Carl Johnson, and Stanly Williams for suggestions regarding the manuscript.
This work was supported by grants from the National Science Foundation to S.S.G. (MCB-9513367) and from the International Human Frontier Science Program to S.S.G. and T.K. (RG0385/1996-M with co-PIs C.H. Johnson and M. Ishiura) and by a JSPS fellowship to M.K.
REFERENCES
- 1.Andersson, C. A., N. F. Tsinoremas, J. Shelton, N. V. Lebedeva, J. Yarrow, H. Min, and S. S. Golden. Application of bioluminescence to the study of circadian rhythms in cyanobacteria. Methods Enzymol., in press. [DOI] [PubMed]
- 2.Aschoff J. Temporal orientation: circadian clocks in animals and humans. Anim Behav. 1989;37:881–896. [Google Scholar]
- 3.Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K. Current protocols in molecular biology. New York, N.Y: Greene Publishing Associates and Wiley-Interscience; 1987. [Google Scholar]
- 4.Bult C J, White O, Olsen G J, Zhou L, Fleischmann R D, Sutton G G, Blake J A, FitzGerald L M, Clayton R A, Gocayne J D, Kerlavage A R, Dougherty B A, Tomb J F, Adams M D, Reich C I, Overbeek R, Kirkness E F, Weinstock K G, Merrick J M, Glodek A, Scott J L, Geoghagen N S M, Venter J C. Complete genome sequence of the methanogenic archaeon, Methanococcus jannaschii. Science. 1996;273:1058–1073. doi: 10.1126/science.273.5278.1058. [DOI] [PubMed] [Google Scholar]
- 5.Bustos S A, Golden S S. Expression of the psbDII gene in Synechococcus sp. strain PCC 7942 requires sequences downstream of the transcription start site. J Bacteriol. 1991;173:7525–7533. doi: 10.1128/jb.173.23.7525-7533.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bustos S A, Golden S S. Light-regulated expression of the psbD gene family in Synechococcus sp. strain PCC 7942: evidence for the role of duplicated psbD genes in cyanobacteria. Mol Gen Genet. 1992;232:221–230. doi: 10.1007/BF00280000. [DOI] [PubMed] [Google Scholar]
- 7.Cohen M F, Wallis J G, Campbell E L, Meeks J C. Transposon mutagenesis of Nostoc sp. strain ATCC 29133, a filamentous cyanobacterium with multiple cellular differentiation alternatives. Microbiology. 1994;140:3233–3240. doi: 10.1099/13500872-140-12-3233. [DOI] [PubMed] [Google Scholar]
- 8.Elhai J, Wolk C P. Conjugal transfer of DNA to cyanobacteria. Methods Enzymol. 1988;167:747–754. doi: 10.1016/0076-6879(88)67086-8. [DOI] [PubMed] [Google Scholar]
- 9.Elledge S J, Sugiono P, Guarente L, Davis R W. Genetic selection for genes encoding sequence-specific DNA-binding proteins. Proc Natl Acad Sci USA. 1989;86:3689–3693. doi: 10.1073/pnas.86.10.3689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Golden S S, Brusslan J, Haselkorn R. Expression of a family of psbA genes encoding a photosystem II polypeptide in the cyanobacterium Anacystis nidulans R2. EMBO J. 1986;5:2789–2798. doi: 10.1002/j.1460-2075.1986.tb04569.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Golden S S, Brusslan J, Haselkorn R. Genetic engineering of the cyanobacterial chromosome. Methods Enzymol. 1987;153:215–231. doi: 10.1016/0076-6879(87)53055-5. [DOI] [PubMed] [Google Scholar]
- 12.Golden S S, Ishiura M, Johnson C H, Kondo T. Cyanobacterial circadian rhythms. Annu Rev Plant Physiol Plant Mol Biol. 1997;48:327–354. doi: 10.1146/annurev.arplant.48.1.327. [DOI] [PubMed] [Google Scholar]
- 13.Hastings J W, Presswood R P. Bacterial luciferase: FMNH2-aldehyde oxidase. Methods Enzymol. 1978;53:558–570. doi: 10.1016/s0076-6879(78)53057-7. [DOI] [PubMed] [Google Scholar]
- 14.Heintzen C, Nater M, Apel K, Staiger D. AtGRP7, a nuclear RNA-binding protein as a component of a circadian-regulated negative feedback loop in Arabidopsis thaliana. Proc Natl Acad Sci USA. 1997;94:8515–8520. doi: 10.1073/pnas.94.16.8515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ishiura M, Kutsuna S, Aoki S, Iwasaki H, Andersson C R, Tanabe A, Golden S S, Johnson C J, Kondo T. Expression of a clock gene cluster kaiABC as a circadian feedback process in cyanobacteria. Science. 1998;281:1519–1523. doi: 10.1126/science.281.5382.1519. [DOI] [PubMed] [Google Scholar]
- 16.Johnson C H, Hastings J W. The elusive mechanism of the circadian clock. Am Sci. 1986;74:29–36. [Google Scholar]
- 17.Kaneko T, Sato S, Kotani H, Tanaka A, Asamizu E, Nakamura Y, Miyajima N, Hirosawa M, Sugiura M, Sasamoto S, Kimura T, Hosouchi T, Matsuno A, Muraki A, Nakazaki N, Naruo K, Okumura S, Shimpo S, Takeuchi C, Wada T, Watanabe A Y, Yamada M, Yasuda M, Tabata S. Sequence analysis of the genome of the unicellular cyanobacterium Synechocystis sp. strain PCC6803. II. Sequence determination of the entire genome and assignment of potential protein-coding regions. DNA Res. 1996;3:109–136. doi: 10.1093/dnares/3.3.109. [DOI] [PubMed] [Google Scholar]
- 18.Klenk H P, Clayton R A, Tomb J F, White O, Nelson K E, Ketchum K A, Dodson R J, Gwinn M, Hickey E K, Peterson J D, Richardson D L, Kerlavage A R, Graham D E, Kyrpides N C, Fleischmann R D, Quackenbush J, Lee N H, Sutton G G, Gill S, Kirkness E F, Dougherty B A, McKenney K, Adams M D, Loftus B, Venter J C, et al. The complete genome sequence of the hyperthermophilic, sulphate-reducing archaeon Archaeoglobus fulgidus. Nature. 1997;390:364–370. doi: 10.1038/37052. [DOI] [PubMed] [Google Scholar]
- 19.Kondo T, Strayer C A, Kulkarni R D, Taylor W, Ishiura M, Golden S S, Johnson C H. Circadian rhythms in prokaryotes: luciferase as a reporter of circadian gene expression in cyanobacteria. Proc Natl Acad Sci USA. 1993;90:5672–5676. doi: 10.1073/pnas.90.12.5672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kondo T, Tsinoremas N F, Golden S S, Johnson C H, Kutsuna S, Ishiura M. Circadian clock mutants of cyanobacteria. Science. 1994;266:1233–1236. doi: 10.1126/science.7973706. [DOI] [PubMed] [Google Scholar]
- 21.Kutsuna S, Kondo T, Aoki S, Ishiura M. A period-extender gene, pex, that extends the period of the circadian clock in the cyanobacterium Synechococcus sp. strain PCC 7942. J Bacteriol. 1998;180:2167–2174. doi: 10.1128/jb.180.8.2167-2174.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu Y, Tsinoremas N F, Golden S S, Kondo T, Johnson C H. Circadian expression of genes involved in the purine biosynthetic pathway of the cyanobacterium Synechococcus sp. strain PCC 7942. Mol Microbiol. 1996;20:1071–1081. doi: 10.1111/j.1365-2958.1996.tb02547.x. [DOI] [PubMed] [Google Scholar]
- 23.Liu Y, Tsinoremas N F, Johnson C H, Lebedeva N V, Golden S S, Ishiura M, Kondo T. Circadian orchestration of gene expression in cyanobacteria. Genes Dev. 1995;9:1469–1478. doi: 10.1101/gad.9.12.1469. [DOI] [PubMed] [Google Scholar]
- 24.McNeil G P, Zhang X, Genova G, Jackson F R. A molecular rhythm mediating circadian clock output in Drosophila. Neuron. 1998;20:297–303. doi: 10.1016/s0896-6273(00)80457-2. [DOI] [PubMed] [Google Scholar]
- 25.Millar A J, Short S R, Chua N H, Kay S A. A novel circadian phenotype based on firefly luciferase expression in transgenic plants. Plant Cell. 1992;4:1075–1087. doi: 10.1105/tpc.4.9.1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ouyang Y, Andersson C R, Kondo T, Golden S S, Johnson C H. Resonating circadian clocks enhance fitness in cyanobacteria. Proc Natl Acad Sci USA. 1998;95:8660–8664. doi: 10.1073/pnas.95.15.8660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pittendrigh C S. Circadian systems: general perspective and entrainment. 1981. pp. 57–80. and p. 95–124. In J. Aschoff (ed.), Handbook of behavioral neurobiology: biological rhythms. Plenum Press, New York, N.Y. [Google Scholar]
- 28.Prentki P, Krisch H M. In vitro insertional mutagenesis with a selectable DNA fragment. Gene. 1984;29:303–313. doi: 10.1016/0378-1119(84)90059-3. [DOI] [PubMed] [Google Scholar]
- 29.Schaefer M R, Golden S S. Light availability influences the ratio of two forms of D1 in cyanobacterial thylakoids. J Biol Chem. 1989;264:7412–7417. [PubMed] [Google Scholar]
- 30.Shine J, Dalgarno L. The 3′-terminal sequence of Escherichia coli 16S ribosomal RNA: complementarity to nonsense triplets and ribosome binding sites. Proc Natl Acad Sci USA. 1974;71:1342–1346. doi: 10.1073/pnas.71.4.1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Smith D R, Doucette-Stamm L A, Deloughery C, Lee H, Dubois J, Aldredge T, Bashirzadeh R, Blakely D, Cook R, Gilbert K, Harrison D, Hoang L, Keagle P, Lumm W, Pothier B, Qiu D, Spadafora R, Vicaire R, Wang Y, Wierzbowski J, Gibson R, Jiwani N, Caruso A, Bush D, Safer H, Patwell D, Prabhakar S, McDougall S, Shimer G, Goyal A, Pietrokovski S, Church G M, Daniels C J, Mao J-I, Rice P, Nölling J, Reeve J N. Complete genome sequence of Methanobacterium thermoautotrophicum ΔH: functional analysis and comparative genomics. J Bacteriol. 1997;179:7135–7155. doi: 10.1128/jb.179.22.7135-7155.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sweeney B M. Rhythmic phenomena in plants. San Diego, Calif: Academic Press; 1987. [Google Scholar]
- 33.Tiedeman A A, Keyhani J, Kamholz J, Daum III H A, Gots J S, Smith J M. Nucleotide sequence analysis of the purEK operon encoding 5′-phosphoribosyl-5-aminoimidazole carboxylase of Escherichia coli K-12. J Bacteriol. 1989;171:205–212. doi: 10.1128/jb.171.1.205-212.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tsinoremas N F, Ishiura M, Kondo T, Andersson C R, Tanaka K, Takahashi H, Johnson C H, Golden S S. A sigma factor that modifies the circadian expression of a subset of genes in cyanobacteria. EMBO J. 1996;15:2488–2495. [PMC free article] [PubMed] [Google Scholar]
- 35.Watanabe W, Sampei G, Aiba A, Mizobuchi K. Identification and sequence analysis of Escherichia coli purE and purK genes encoding 5′-phosphoribosyl-5-amino-4-imidazole carboxylase for de novo purine biosynthesis. J Bacteriol. 1989;171:198–204. doi: 10.1128/jb.171.1.198-204.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wolk C P, Cai Y, Panoff J-M. Use of a transposon with luciferase as a reporter to identify environmentally responsive genes in a cyanobacterium. Proc Natl Acad Sci USA. 1991;88:5355–5359. doi: 10.1073/pnas.88.12.5355. [DOI] [PMC free article] [PubMed] [Google Scholar]