Abstract
The treponemal fla operon is comprised of numerous motility-related genes; however, the initial gene of this operon, tap1, has no known function. A recently developed system to generate specific mutants in Treponema denticola was utilized to determine if Tap1 was essential for motility. T. denticola tap1 and flanking DNA were identified, cloned, and sequenced, and a suicide plasmid that contained tap1 interrupted with an erythromycin resistance cassette (ermF and ermAM) was constructed. Because of potential polar effects from this cassette, a second plasmid that contained tap1 interrupted with a modified erythromycin resistance cassette that lacked the putative ermF transcription terminator was constructed. Electroporation-mediated allelic exchange incorporated the interrupted tap1 genes into the T. denticola chromosome, creating Tap1-deficient mutants. Reverse transcriptase PCR revealed that the erythromycin resistance cassette within tap1 did not terminate fla operon transcription in either mutant. Moreover, the phenotypes of the two mutants were indistinguishable. These mutants lacked motion in liquid culture, were unable to spread on agar plates, and lacked flagellar filaments as determined by electron microscopy. Immunoblots revealed a marked reduction in detectable FlaB flagellar filament protein compared to that of wild type; however, flaB RNA was easily detectable, and transcription levels did not appear to be altered. The basis for the lack of filament protein expression is unknown. Immunoblotting also showed that the flagellar hook protein (FlgE) was synthesized in the Tap1-deficient mutant; however, electron microscopy revealed that the mutant possessed unusual elongated hooks of variable lengths. We propose that treponemal Tap1 is analogous to FliK, which is involved in monitoring the flagellar hook length of Salmonella typhimurium.
Treponema denticola is an anaerobic spirochete that is associated with periodontal disease (32, 33). As with all spirochetes, the structure and motility of T. denticola are unusual and enable movement in highly viscous environments (17, 18). A unique feature of T. denticola (and all spirochetes) is the location of the flagellar filaments within the periplasmic space, although under certain conditions they may protrude from the cell (3, 15). The T. denticola flagellar filaments are complex, consisting of three core proteins (FlaB1, FlaB2, and FlaB3) and a major sheath protein, FlaA (29). However, the general structure of the flagellum appears similar to that of other bacteria, since it consists of a basal body, rod, flagellar hook, and filament (25, 29). In contrast to the structural similarities, DNA sequencing and transcription studies have clearly shown that the organization and expression of motility genes are unique in the spirochetes (7–10, 12, 13, 22).
Motility of spirochetes is complex and likely requires approximately 50 genes, many of which are organized into several large operons (7–10, 12, 13, 22). The extensive treponemal fla operon in Treponema phagedenis and Treponema pallidum comprises genes directly associated with motility, including the flagellar hook gene (flgE) and flagellar motor genes (motA and motB), in addition to genes of unknown function (tap1) (12, 22). Previous studies have shown that T. phagedenis Tap1 is a soluble protein that partitions in the aqueous phase during Triton X-114 extraction and is associated with motility, because tap1 is the first gene of the fla motility operon (22). However, there has been no direct evidence for a role of tap1 in the motility of treponemes.
Genetic analysis of T. denticola, like that of other spirochetes, has been hampered by a paucity of genetic tools for analysis. Recently, Li et al. showed that a gene cassette that includes ermF and ermAM in tandem (ermF-ermAM) conferred erythromycin resistance in T. denticola (ermF) and also in Escherichia coli (ermAM) (19). This cassette was inserted into flgE, and a specific mutant was created by electroporation-mediated allelic exchange of the insertionally inactivated flgE. This nonmotile mutant lacked flagellar hooks and filaments, as determined by electron microscopy. Using immunoblotting, Ruby et al. revealed that this mutant was also deficient in flagellar filament proteins (29). Specifically, FlaB polypeptides were not detected and minimal amounts of FlaA were detected in the flgE-deficient mutant. These results demonstrated the utility of the erythromycin resistance cassette in generating specific mutants for analysis.
The ermF-ermAM cassette used by Li et al. was postulated by Fletcher et al. to have a potential transcription terminator located between ermF and ermAM (6). This terminator-like sequence would potentially cause polar effects when inserted within an operon, but it is not known if the terminator is functional in T. denticola. For analysis of single genes of the fla operon, it is critical to have a nonpolar cassette available for gene interruptions. Conceivably, removal of this putative terminator would permit the erythromycin resistance cassette to be used to study single genes located within operons without affecting downstream genes.
Our goal was to identify the fla operon of T. denticola and interrupt the T. denticola tap1 gene with a nonpolar erythromycin resistance cassette to determine whether tap1 is involved in motility. We found that the organization of the T. denticola fla operon is similar to that for other treponemes, and interruption of T. denticola tap1 with a nonpolar cassette resulted in a nonmotile cell. The Tap1-deficient mutants possessed unusually long and variable-length flagellar hooks, suggesting that Tap1 may be involved in monitoring hook length, analogously to FliK of Salmonella typhimurium (14).
MATERIALS AND METHODS
Culture media and conditions, strains, and molecular methods.
T. denticola (ATCC 33520) was grown in new oral spirochete medium (NOS) with 10% heat-inactivated rabbit serum and 10 μg of cocarboxylase per ml at 36°C in an anaerobic chamber (Coy Laboratory Products, Inc., Grass Lake, Mich.) with an atmosphere of 85% nitrogen, 5% carbon dioxide, and 10% hydrogen. For growth on semisolid media, 0.5% agarose was included with NOS.
E. coli DH5α (Gibco BRL, Grand Island, N.Y.) was used for most transformations and was grown in Luria-Bertani (LB) broth at 37°C with shaking. Nonmethylated plasmid DNA was prepared in E. coli SCS110 (Stratagene Corp., La Jolla, Calif.) for electroporation of T. denticola.
The PCR and reverse transcriptase PCR (RT-PCR) were performed with reagents and thermal cyclers available from Perkin-Elmer (Foster City, Calif.). Plasmid pCRII-TOPO was purchased from Invitrogen Corp. (Carlsbad, Calif.), and pUC19CAT, which contains a chloramphenicol resistance gene in pUC19, was a lab stock. Lambda ZAP Express was obtained from Stratagene. Ligations were performed either via rapid ligations (Boehringer Mannheim Corp., Indianapolis, Ind.) or traditional 16-h ligations with T4 DNA ligase (New England BioLabs, Beverly, Mass.) or directly by the TOPO TA cloning system (Invitrogen). Total T. denticola RNA was isolated as previously described for T. phagedenis with materials available through Amresco (Solon, Ohio) (24). Plasmid minipreparation DNA was isolated by standard techniques (26), and Qiagen Midi columns (Qiagen Corp., Chatsworth, Calif.) were used for purification of larger amounts of DNA for transformation of T. denticola. Radioisotopes were obtained from Amersham Life Sciences, Inc. (Arlington Heights, Ill.).
The ermF-ermAM cassette and a plasmid containing the T. denticola flgE gene interrupted with this cassette were kindly provided by Howard Kuramitsu (State University of New York, Buffalo).
Antiserum against T. pallidum recombinant FlgE was previously generated by mouse immunizations in our laboratory (20, 23). Rabbit antisera generated with T. phagedenis FlaB and FlaA polypeptides were kindly provided by Nyles Charon (West Virginia University, Morgantown) (20).
Identification of the T. denticola fla operon.
The 5′ end of the T. denticola fla operon was identified by screening a Lambda ZAP Express library prepared by partial Sau3AI digestion of T. denticola chromosomal DNA by previously described methods (24). The probe was a segment of flgE made by PCR of T. denticola with a DNA sequence available from Li et al. (19) (GenBank accession no. L75953). DNA sequencing at the Wadsworth Center Molecular Genetics Core facility was done with the Perkin-Elmer ABI Prism 377 and ABI 373A sequencers. In addition, approximately 78 bp at the 3′ end of flgE of T. denticola was previously determined by amplification of this region by degenerate primer PCR and DNA sequencing (4, 5). Sequences were assembled and analyzed with the Wisconsin package, version 9.1 (Genetics Computer Group, Madison, Wis.).
Primer extension.
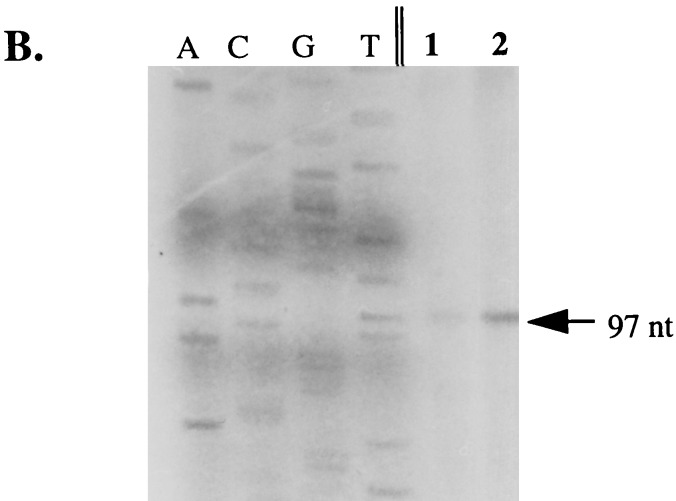
To identify the start site of transcription of the T. denticola fla operon, primer extension was performed. TDW13 (5′-CAGAGTCTTCTCTTTCCG-3′) was labeled with 32P by T4 polynucleotide kinase, and the primer extension reaction was performed with the Primer Extension System (Promega Corp., Madison, Wis.). In the extension reaction, 1 pmol of labeled TDW13 was used. For determining the length of the primer extension products, a 35S-labeled DNA ladder was generated by PCR with the control sequence from the AmpliCycle sequence kit (Perkin-Elmer) and the forward M13 primer. The Burst-Pak sequencing gels were used (Owl Scientific, Inc., Woburn, Mass.) to prepare 6% polyacrylamide gels for electrophoresis. Autoradiography was performed with XAR-2 film (Eastman Kodak, New Haven, Conn.) at −70°C with an intensifying screen.
Construction of two plasmids containing T. denticola tap1 interrupted with modified erythromycin resistance cassettes.
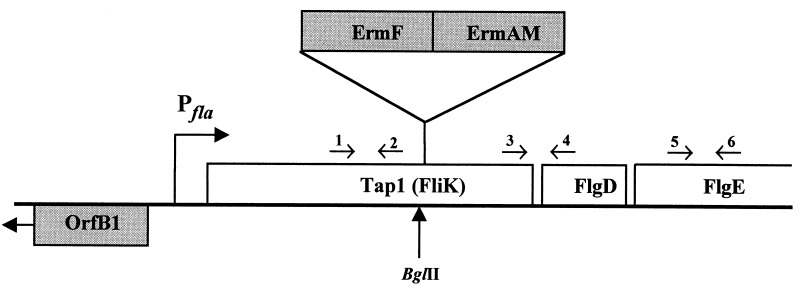
A DNA fragment containing T. denticola tap1 was prepared by PCR of T. denticola chromosomal DNA with primers TDW8 (5′-CTGATGAAGCCCGATTTG-3′) and TDW5 (5′-GTTTACCTGCATAAACTACCTC-3′) and ligated to pCRII-TOPO. A clone harboring the 1,230-bp insert was selected, the plasmid DNA was digested with EcoRI, and the insert DNA was ligated to pUC19CAT. A clone containing the 1,230-bp insert of tap1 DNA was digested with BglII, which cut at a site 706 bp downstream from the start of the tap1 gene (see Fig. 1). Tap1 was then inactivated by cloning the two erythromycin resistance cassettes into the BglII site as indicated below.
FIG. 1.
Diagram of the fla operon organization of T. denticola and 5′ upstream region. Pfla indicates the approximate location of the fla operon promoter. The ermF-ermAM cassette is indicated above the operon and is shown where it is inserted in the BglII site for creating Tap1-deficient mutants. The locations of primer pairs used for RT-PCR are indicated by arrows and are represented as follows: 1, TDW8; 2, TDW9; 3, TDW12; 4, TDW5; 5, TDWFLGEF; 6, TDWFLGER. Sequences of the primers are given in Materials and Methods.
Two different erythromycin resistance cassettes were constructed and used for insertional inactivation of T. denticola tap1. The first utilized the ermF-ermAM cassette described by Li et al. (19). To facilitate genetic manipulations, this cassette was amplified from pHfLE (19) with the primers ERMBGLF (5′-TATAAGATCTCCGATAGCTTCCGCTATTGC-3′) and ERMBGLR (5′-TATAAGATCTGAAGCTGTCAGTAGTATACC-3′), which both contain synthetic BglII sites, and then cloned into pCRII-TOPO. This plasmid was digested with BglII, and the 2.1-kb DNA product (ermF-ermAM cassette) was gel purified, phenol extracted, and ethanol precipitated. The ermF-ermAM cassette was then ligated into the BglII-digested pUC19CAT plasmid that contained T. denticola tap1, transformed into E. coli DH5α, and plated on LB plates containing 300 μg of erythromycin per ml. Erythromycin-resistant E. coli colonies were picked and grown in LB broth, and plasmid DNA was isolated. Orientation of the tap1 gene and ermF-ermAM cassette was determined by PCR and DNA sequencing; only those clones containing the ermF-ermAM cassette oriented in the same direction as tap1 were chosen for the insertional inactivation in T. denticola.
The second strategy involved construction of an erythromycin resistance cassette that did not contain putative transcription terminator or ermF promoter sequences. The ermF gene possesses sequences resembling a transcription terminator following the end of the coding region (6). This terminator-like region, as well as DNA preceding the ermF region (including the ermF promoter), and some nonessential DNA flanking the ermAM gene were removed by the following procedure. An oligonucleotide primer that annealed just upstream of the ribosome binding site of ermF and contained a synthetic BglII site was synthesized (ERFNEWF, 5′-TATAAGATCTATTATCCGCACCCAAAAAG-3′) together with a second primer that anneals to the complementary strand at the stop codon of ermF (ERFNEWR, 5′-TATACCCGGGCAACCACCCGACTTTGAACTA-3′). A synthetic SmaI site was engineered into the second primer. After amplification from the pHfLE template DNA, this ermF gene was cloned into pCRII-TOPO and verified by DNA sequencing. This vector containing ermF was digested with SmaI to open a blunt-end cloning site just downstream of ermF. Next, ermAM was amplified with primers that anneal just upstream of the regulatory region (ERAMNEWF, 5′-GAAGCAAACTTAAGAGTGTG-3′) and also near the stop codon of ermAM (ERAMNEWR, 5′-TATAAGATCTGAAGCTGTCAGTAGTATACC-3′). A synthetic BglII site was incorporated into ERAMNEWR. This fragment was blunt end ligated to the SmaI site of the pCRII-TOPO/ermF plasmid, transformed into E. coli, and selected with 300 μg of erythromycin per ml. Proper orientation of the insert DNA was confirmed by PCR and DNA sequencing. The resulting clone had ermF and ermAM in tandem and no putative transcription terminator. This erythromycin resistance cassette was removed from the plasmid by digestion with BglII and cloned into the BglII site of tap1 as indicated above for the original ermF-ermAM cassette. This cassette that lacked the ermF promoter and the putative transcription terminator was designated ermF-ermAMnp.
Insertional inactivation of T. denticola tap1 with the ermF-ermAM and ermF-ermAMnp cassettes.
The protocol developed by Li et al. was used with some minor modifications for insertional inactivation of T. denticola (19). Briefly, 100 ml of T. denticola was grown to an optical density of 0.3 at 600 nm. Cells were washed three times with cold 10% glycerol in water and resuspended in a final volume of 2 ml on ice. Electroporation was done with 10 μg of linearized DNA with a Bio-Rad Gene Pulser at 1.8 kV, 200 Ω, with 25 μF and a 0.1-cm cuvette. Time constants of 4.1 to 4.6 were considered optimal. After overnight incubation in 10 ml of NOS broth without erythromycin, plating was done on NOS containing 0.5% agarose and 20 μg of erythromycin per ml. Colonies usually were visible within 7 days. PCR and Southern blotting were used to determine that the wild-type tap1 gene was replaced with the insertionally inactivated tap1 gene by allelic exchange (21, 31).
Identification of T. denticola flaB.
T. denticola contains three flaB genes (29). To obtain a partial sequence from one of these genes, primers from conserved regions of T. phagedenis flaB were used in a PCR amplification of T. denticola (24). The primers FLAB2 (5′-GTGGTTCCATATCGGGGCC-3′) and FLAB4 (5′-CCTGCAAAAAGTTTAGCGC-3′) amplified a 620-bp fragment of T. denticola DNA which was cloned into the pCRII-TOPO cloning vector and sequenced to confirm a flaB identity.
Isolation of flagellar hooks and electron microscopy.
Flagellar hooks were isolated by a modification of previously described methods (23). Approximately 109 cells of T. denticola were washed twice with Tris-buffered saline, the outer sheath was removed with 2% Triton X-100, and the cells were washed once with Tris-buffered saline. The final pellet was resuspended in 10 mM sodium phosphate buffer and sonicated on ice for 1 min at a 1-s cycle time and 50% duty cycle. DNase, RNase, and lysozyme (20 mg each) were added to the sonicate along with final concentrations of 0.05% Triton X-100, 0.02 mM EDTA, 2.5% glycerol, and 25 mM MgSO4, and the mixture was incubated overnight at 4°C. Next, Triton X-100 was added to a final concentration of 2% and the incubation was continued for 1 h at 20°C. The lysate was centrifuged at 41,000 × g in an SW-28 rotor for 30 min through a gradient containing 100, 75, 50, and 25% glycerol. The viscous material at the 75%–100% glycerol interface was collected and dialyzed overnight against water at 4°C. This fraction was enriched for flagellar hooks; some material was saved for electron microscopy, and the remainder was treated with pH 2.2 glycine buffer to remove any flagellar filaments as previously described (23).
For electron microscopic visualization of T. denticola cells, 1 ml of logarithmic-phase culture was centrifuged for 1 min at 10,000 × g. The pellet was resuspended in the same amount of water containing 1% Triton X-100 reduced, a chemically modified form of Triton X-100 (Aldrich Chemical Co., Milwaukee, Wis.), and incubated overnight at 4°C. The sample was centrifuged for 1 min at 10,000 × g, and the pellet was resuspended in 100 μl of water.
Negative staining was used to visualize cells or purified hooks. Drops (40 μl) of the sample were placed on dental wax. Formvar-coated copper grids were floated on the drops for 2 to 4 min; excess liquid was removed by wicking with filter paper, and the grids were immediately washed by floating them on 2 drops of double-distilled water. After the final wash, excess water was removed, the grids were briefly floated on 2% sodium phosphotungstate (pH 7.0), liquid was removed by wicking, and the samples were viewed in a Zeiss (LEO) 910 transmission electron microscope operating at 80 keV.
Transcription analysis.
RT-PCR was used to determine whether genes of the fla operon were transcribed (22). Northern blots are ineffective because the fla operon transcript is very large and the RNA transcript is significantly degraded. For analysis of transcription by RT-PCR (see Fig. 1), primers for upstream of the ermF-ermAM cassette insertion within tap1 were TDW8 (5′-CTGATGAAGCCCGATTTG-3′) and TDW9 (5′-TTTAGCAAATCCTTGGGC-3′), and primers for genes immediately downstream of the ermF-ermAM cassette were TDW12 (5′-CGGGAACAGTAACTGTTGCCTC-3′) and TDW5 (5′-GTTTACCTGCATAAACTACCTC-3′). Primers located within flgE were TDWFLGEF (5′-CGTTGCCAACGTAAATAC-3′) and TDWFLGER (5′-AAATTCGGTAGACCAAGTAG-3′). Oligonucleotide primers for detection of flaB RNA were TDWFLABF (5′-CTGCAAATAGGAGCATTGGAAC-3′) and TDWFLABR (5′-GCTATTGTTATTAGCCTGAGCGAG-3′).
Production of Tap1-MBP fusion protein, antiserum production, and immunoblotting.
T. denticola tap1 was amplified with the primers TDWTAPF (5′-TATAGAATTCCAGGCTTTGCCGGTAAAAGAGC-3′) and TDWTAPR (5′-TATACTGCAGGTTTACCTGCATAAACTACCTC-3′), which contains synthetic EcoRI and PstI sites, respectively. TDWTAPF anneals 21 nucleotides (nt) downstream of the ATG start codon of tap1, and TDWTAPR includes the native stop codon. After amplification, the 1,372-bp product was cloned into pCRII-TOPO. This plasmid was then digested with EcoRI and PstI to liberate the fragment containing tap1, which was ligated into pMAL-c2 (New England BioLabs). The ligation mixture was transformed into CaCl2-competent E. coli DH5α, heat shocked, and plated on LB agar plates containing X-Gal (5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside) and IPTG (isopropyl-β-d-thiogalactopyranoside) as previously described (24). White colonies were picked, and clones with the proper insert were identified by restriction endonuclease digestion followed by sequencing of the plasmid-insert junctions. Tap1–maltose-binding protein (MBP) fusion protein was induced with IPTG and purified over amylose resin by previously described methods (23). Immunization of rabbits with the Tap1-MBP fusion protein and collection of antisera were done by Biodesign Inc. (Kennebunkport, Maine).
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis and immunoblotting were performed as previously described, and the blots were developed with alkaline phosphatase-labeled secondary antibody (20). About 50 μg of each cell lysate was loaded per lane, and primary antisera were used at a 1:50 or 1:100 dilution.
Nucleotide sequence accession number.
The GenBank nucleotide sequence accession number for T. denticola tap1, flgD, and partial flgE is AF049342 and for partial flaB is AF072133.
RESULTS
Identification and molecular characterization of the T. denticola fla operon.
After screening of the T. denticola Lambda ZAP Express library, one clone that possessed a 3,902-bp insert was obtained. This cloned DNA was sequenced in both directions with synthetic oligonucleotides and revealed the 5′ region of the T. denticola fla operon (Fig. 1). As noted with other treponemal fla operons, T. denticola fla begins with tap1 (457 amino acids), followed by flgD, which is involved in flagellar hook assembly (169 amino acids) (28), and flgE, the flagellar hook protein (the first 237 amino acids were deduced) (23). In addition, there is an open reading frame upstream of tap1 that is transcribed in the opposite direction from the fla operon; this is similar to the gene arrangement in T. phagedenis and T. pallidum (8, 22).
Preceding tap1 is a sigma 28-like sequence (Fig. 2A) that is likely involved in transcription of the fla operon. Primer extension analysis revealed that the start site of transcription was located 9 nt downstream of this promoter and 32 nt upstream from the ATG translational start site (Fig. 2B). The promoter sequence was very similar to the Pfla and sigma 28 sequences in other treponemes (Fig. 3). RT-PCR also revealed that transcription proceeded from Pfla through flgD and flgE, which confirmed the organization of these genes into an operon (data not shown).
FIG. 2.
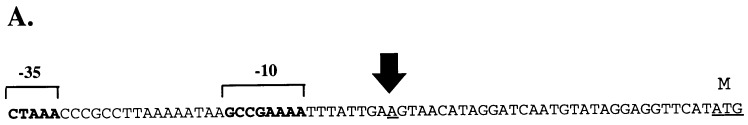
Identification of the fla operon promoter. (A) DNA sequence including the proposed −10 and −35 regions of Pfla. The large arrow indicates the start site of transcription determined by primer extension. M indicates the first amino acid of the Tap1 polypeptide. (B) Primer extension assay to determine the start site of transcription of Pfla. A, C, G, and T indicate nucleotides used to generate a size ladder with unrelated DNA. Lanes 1 and 2 contain the primer extension reaction products of 2 and 5 μl, respectively.
FIG. 3.
Comparison of the T. denticola fla promoter sequence with promoter sequences from various spirochete motility genes and consensus sigma 28 sequences.
The deduced amino acid sequence of T. denticola Tap1 was aligned with those of other known treponemal Tap1 homologs (Fig. 4A). This alignment revealed extensive amino acid sequence identity within a 90-amino-acid region near the C terminus with less identity in the remainder of the polypeptide (Fig. 4A). The overall T. denticola Tap1 identity as determined with the GAP program was 32% with T. phagedenis and 21% with T. pallidum. Within the conserved 90-amino-acid region from F-324 to G-394, T. denticola has 30% amino acid sequence identity with FliK of S. typhimurium (16) as shown in Fig. 4B. BLAST 2.0 searches (2) with the 90-amino-acid conserved region also revealed a similar sequence identity with FliK proteins of Bacillus subtilis and Rhodobacter sphaeroides (references 1 and 11 and data not shown).
FIG. 4.
Alignment of Tap1 amino acid sequences. (A) Identical amino acids from three treponemes are boxed and shaded with SHADYBOX, which reveals the conserved region near the carboxyl terminus. The dark inverted triangle indicates the location of the point of insertion for the erythromycin resistance cassette into the T. denticola tap1 gene to generate a Tap1-deficient mutant. (B) Alignment of the conserved C-terminal region of T. denticola Tap1 with FliK of S. typhimurium (16). Identical amino acids are boxed and shaded as described above.
Identification of a T. denticola flaB gene.
PCR of T. denticola DNA with primers located in a conserved region of treponemal FlaB resulted in a 620-bp amplicon. Sequence analysis revealed the highest amino acid sequence identity (76%) with FlaB1 of T. pallidum. However, a definitive identity was not assigned to this T. denticola flaB gene because the 5′ end of the gene was not sequenced. That sequence could have been used for comparison with the published N-terminal amino acid sequences of T. denticola FlaB1, FlaB2, and FlaB3 (29). The T. denticola flaB DNA sequence was essential for designing oligonucleotides for use in RT-PCR to determine whether a flaB gene was transcribed. Because of the extensive identity among flaB genes, we cannot exclude the possibility that the primers used in RT-PCR have amplified more than one flaB gene.
Interruption of tap1 with antibiotic resistance cassettes.
To create a specific mutant that was deficient in Tap1, a suicide plasmid that contained an ermF-ermAM cassette inserted within T. denticola tap1 was constructed (Fig. 1 and 4A). Electroporation-mediated allelic exchange replaced the wild-type tap1 with the interrupted version in the T. denticola chromosome. Although the yields of double-crossover recombinants are sometimes quite low (<10 colonies per electroporation), this transformation with the ermF-ermAM cassette produced 533 colonies with linear DNA and 71 colonies with uncut plasmid DNA after 1 week of incubation following electroporation. Although nonmethylated plasmid DNA was used for these transformations, we have subsequently found that E. coli-methylated DNA may also be used. Several colonies were selected for further analysis, but additional testing suggested that these were indistinguishable clones that had undergone a similar double-crossover recombination event to inactivate tap1 (data not shown). Therefore, one clone was used for all subsequent analysis and is termed JS97. Southern blotting and PCR revealed that JS97 had incorporated the erythromycin resistance cassette within tap1 via double-crossover recombination (data not shown).
A second tap1-deficient mutant was generated with the modified ermF-ermAMnp cassette. This construct contained the ermF-ermAMnp cassette, which lacked the putative terminator and nonessential DNA upstream of ermF. The transformation of T. denticola with the ermF-ermAMnp cassette produced another mutant, termed AS98. As indicated below, there was no major difference in transcription between the T. denticola mutants containing ermF-ermAM or ermF-ermAMnp cassettes within tap1. Therefore, except as indicated, the data presented below were obtained from the tap1 mutant containing the original ermF-ermAM cassette (JS97). The MIC of erythromycin is at least 300 μg/ml for both JS97 and AS98.
Analysis of the Tap1-deficient mutant JS97 of T. denticola. (i) Colony and cell morphology and motility.
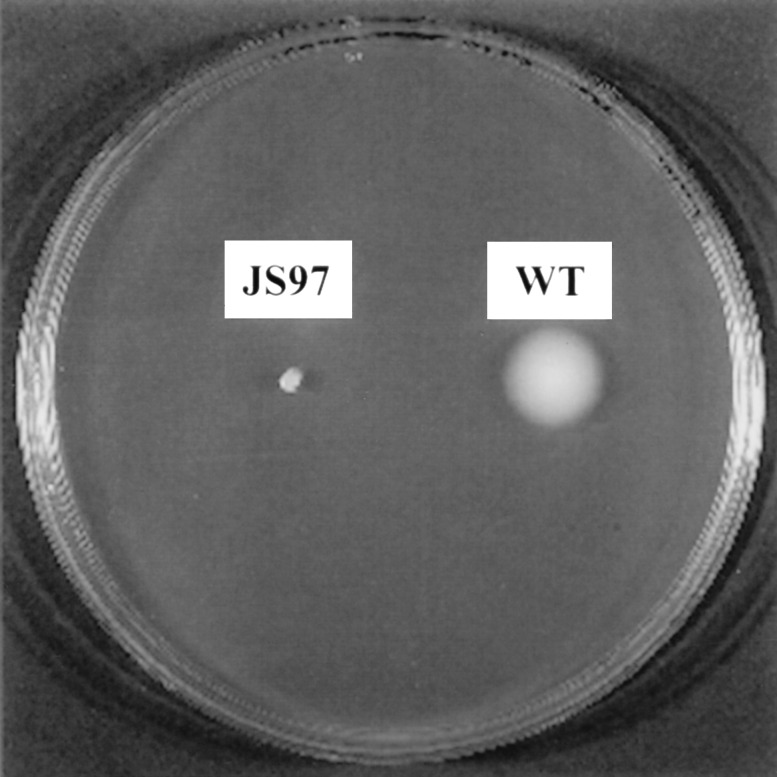
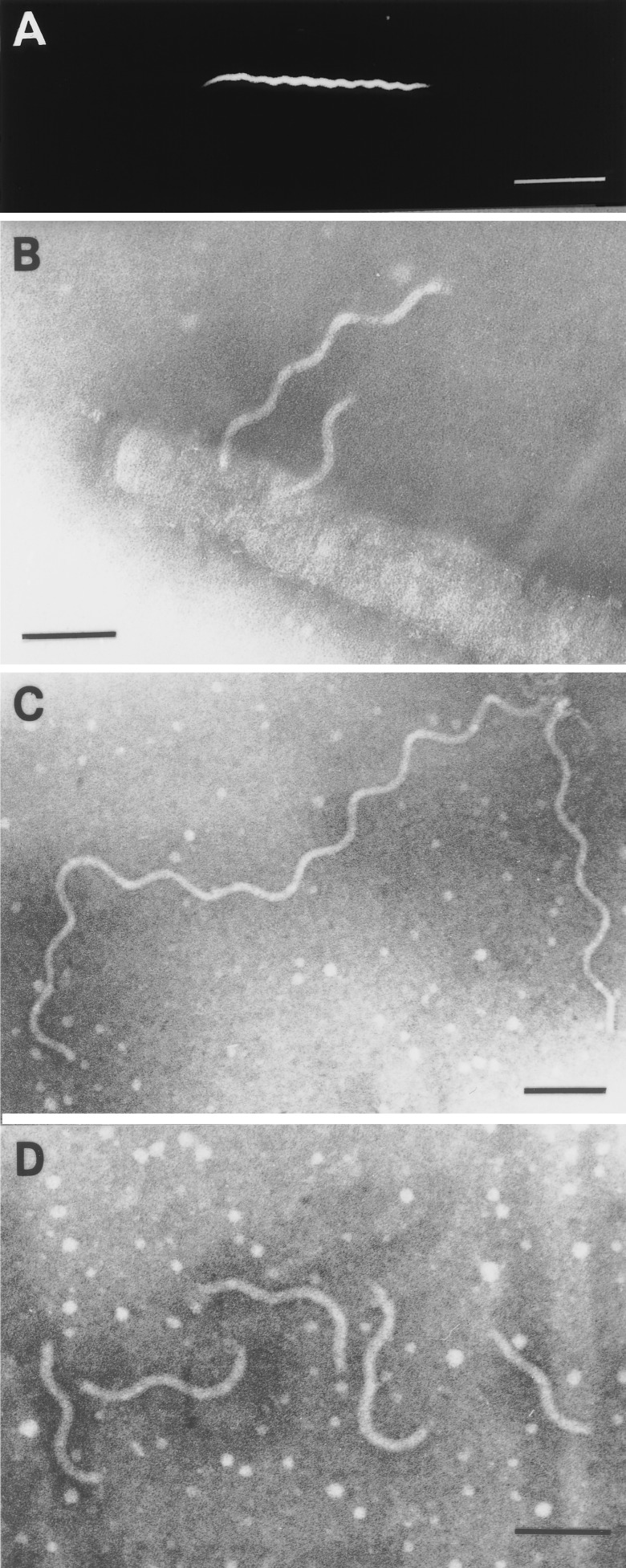
The tap1 mutant JS97 produced greyish white raised colonies on 0.5% NOS-agarose with a colony diameter of 1 to 2 mm after 7 days of incubation. There was no subsurface migration of JS97 in agar plates even after 3 weeks of incubation (data not shown). In contrast, the wild-type colony spread beneath the agar surface to about 9 mm in diameter during this time. Figure 5 shows the relationship of spreading of the wild-type T. denticola to that of JS97. In NOS broth, the tap1 mutant JS97 grew more slowly and to a lower density than the wild type (data not shown). Dark-field microscopic observations revealed that individual cells of JS97 were generally longer than the wild type and tapered at the cell ends (Fig. 6A). The cells exhibited no movement in liquid or viscous media. Electron microscopic analysis of the mutant JS97 showed a lack of flagellar filaments; however, the mutant did possess elongated flagellar hooks of variable length with a wavelike morphology (Fig. 6B, C, and D). To confirm the identity of the flagellar hooks, a cell lysate was enriched for hooks by a modification of procedures described earlier (23). The final step of this procedure was to treat the samples with a low-pH (2.2) glycine buffer, which disrupts flagellar filaments and basal bodies but leaves the flagellar hooks intact. As shown in Fig. 6C and D, electron microscopic analysis revealed that the long wavelike hooks remained after acid treatment, indicating that these structures were not of flagellar filament origin. This sample reacted strongly on immunoblots with antibodies raised against recombinant T. pallidum FlgE (data not shown and reference 23).
FIG. 5.
T. denticola wild type (WT) and Tap1-deficient mutant JS97 after growth for 7 days on NOS plates containing 0.5% agarose. Approximately 0.1 μl was placed on the plate and incubated at 36°C in an anaerobic chamber.
FIG. 6.
Microscopic analysis of the Tap1-deficient mutant JS97. (A) Dark-field micrograph. Bar, 5 μm. (B) Electron micrograph of a JS97 cell showing elongated hooks that do not possess flagellar filaments. The outer sheath was removed by treatment with 1% Triton X-100 reduced as indicated in the text. Bar, 100 nm. (C and D) Electron micrographs of preparations of enriched hooks from JS97. Bars, 100 nm.
(ii) Transcription analysis.
RT-PCR was used to analyze transcription in the tap1 mutants containing ermF-ermAM (JS97) and ermF-ermAMnp (AS98). Because the tap1 gene is located in an operon, there was concern that potential polar effects from the ermF-ermAM cassette could result in lack of transcription of all downstream genes. Previous studies suggested that a putative transcription terminator was located between ermF and ermAM (6). By RT-PCR, transcription was analyzed upstream of the ermF-ermAM cassette (within tap1) and downstream of the ermF-ermAM cassette (within flgD and flgE) (Fig. 1). Results indicate that transcription of these genes was not significantly altered and that the levels of RNA were comparable to those of the wild type (Fig. 7). In addition, flaB RNA was easily detectable by RT-PCR (Fig. 7) as were fliG, ermF, and ermAM RNA (data not shown).
FIG. 7.
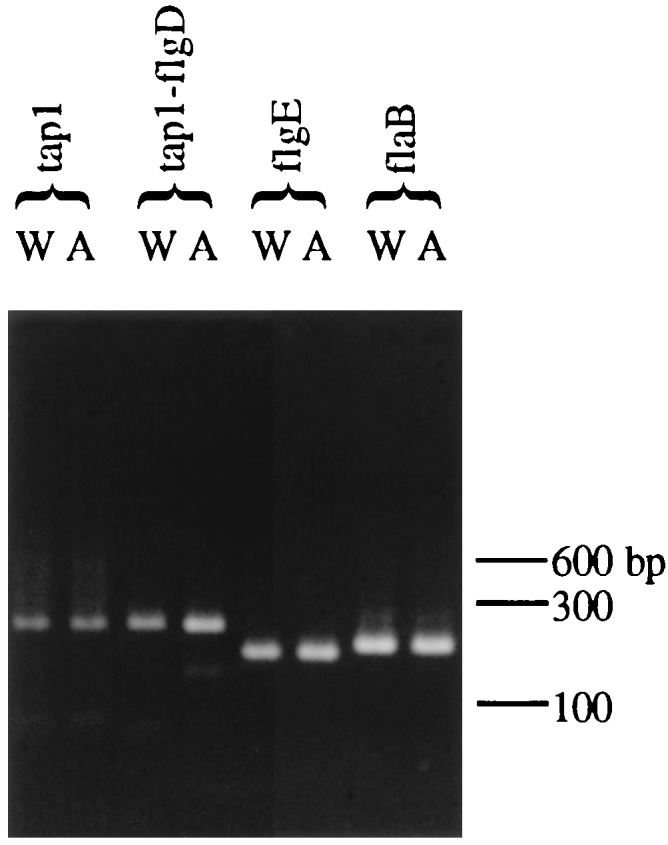
RT-PCR products after agarose gel electrophoresis and staining with ethidium bromide. W, wild-type T. denticola; A, Tap1-deficient mutant JS97; tap1, RT-PCR with primers 1 and 2 within tap1; tap1-flgD, RT-PCR with primers 3 and 4 downstream of the BglII site; flgE, RT-PCR with primers 5 and 6 (see Fig. 1 for the location of these primers); flaB, RT-PCR of the flagellar filament flaB gene with primers as described in Materials and Methods. Note that levels of RT-PCR products in the wild type and in JS97 are similar. Numbers at right are the molecular size markers in base pairs. Control reactions without RT did not show any bands (data not shown).
(iii) Protein expression determined by immunoblotting.
Immunoblotting was done to assess expression of proteins within the fla operon (Tap1 and FlgE) and outside of the fla operon (FlaB) in the Tap1-deficient mutant. The T. denticola Tap1 antisera reacted with Tap1 (Fig. 8A) and also with at least one other polypeptide (data not shown) in wild-type T. denticola. The basis for this cross-reaction is unknown. As shown in Fig. 8A, the Tap1-deficient mutants JS97 and AS98 produced no detectable level of Tap1. In contrast, these mutants synthesized FlgE at approximately wild-type levels (Fig. 8B). Surprisingly, the Tap1-deficient mutants expressed only trace amounts of FlaB polypeptide compared to wild-type T. denticola (Fig. 8C). This result was unexpected because these mutants possess a level of flaB RNA comparable to that of the wild type. Using immunoblotting, we determined that the flagellar sheath polypeptide, FlaA, was also present in only trace amounts (data not shown).
FIG. 8.

Western blots with T. denticola Tap1 antiserum (A), T. pallidum FlgE antiserum (B), and T. phagedenis FlaB antiserum (C). Lanes 1, JS97; lanes 2, AS98; lanes 3, wild type. The arrow in panel A indicates the Tap1 polypeptide band. In panel B, the polypeptide ladder is a typical pattern found in treponemal hook polypeptides that may be due to cross-linking. The significance of the minor band missing in lane 3 is unknown. Numbers at right of each panel represent molecular masses in kilodaltons.
DISCUSSION
The genetic analysis of spirochetes has been hindered by the inability to generate specific mutations. In S. typhimurium and other better-defined systems, specific mutations have helped to ascertain the function and regulation of genes involved in bacterial motility. Although spirochetes and enteric bacteria have many homologs that are presumed to be involved in motility, the functions in spirochetes are inferred from nucleotide and amino acid sequence identities. Comparative sequence analysis, although quite useful, has limitations for ascertaining the function of genes that have no homologs in other bacteria. The recent discovery by Li et al. that an erythromycin resistance cassette could be used to inactivate T. denticola genes has proven to be a valuable tool for genetic manipulation (19).
This work was designed primarily to identify, characterize, and then determine whether T. denticola tap1, a gene of unknown function, was essential for treponemal motility. T. denticola tap1 was identified as the first gene of the fla motility operon, an arrangement found in T. phagedenis and T. pallidum as well, which suggested a role of Tap1 in motility. Alignment of the amino acid sequence of T. denticola Tap1 with other treponemal Tap1 sequences revealed a well-conserved 90-amino-acid region near the C terminus and little identity in the remainder of the polypeptide. Moreover, the amino acid sequence of the conserved 90-amino-acid C-terminal region of T. denticola Tap1 possessed a modest identity with FliK proteins of R. sphaeroides, B. subtilis, and S. typhimurium (1, 11, 16). These comparisons suggest that the C-terminal region provides a fundamental conserved region necessary for the structure or function of tap1. However, since there is limited amino acid sequence identity to known genes, genetic evidence was required to determine the role of Tap1 in T. denticola motility.
Using the gene interruption technique of Li et al. (19), we specifically interrupted the carboxy-terminal region of T. denticola Tap1. The resulting mutant, JS97, was completely nonmotile and lacked periplasmic flagellar filaments but possessed flagellar hooks. Thus, genetic evidence showed that Tap1 was essential for T. denticola motility. Interestingly, these hooks were elongated and of variable length compared with wild-type hooks. Our hypothesis is that Tap1 is involved in monitoring the length of the flagellar hook in T. denticola, a function that is performed by FliK in enteric bacteria (14, 16, 27, 34). It has been postulated previously that the N terminus of FliK might function as a hook length measurement domain and that the C terminus is responsible for flagellar export specificity in S. typhimurium (16). Frameshift mutants disrupted in the C-terminal region of S. typhimurium FliK have a hook length control defect together with a flagellar filament-negative phenotype (34). The variable hook length of T. denticola JS97 together with a lack of flagellar filaments is similar to the phenotype of these FliK mutants of S. typhimurium (16, 34). Moreover, isolated hooks from JS97 clearly demonstrated variable length, suggesting a lack of control on the hook length assembly. The morphology of these polyhooks is similar to that of known FliK-deficient mutants of S. typhimurium (34). On the amino acid level, the modest identity of the conserved C-terminal region of Tap1 with S. typhimurium FliK suggests that these polypeptides are homologs. Therefore, on the basis of the remarkable similarity of the phenotype of the Tap1-deficient T. denticola to that of the FliK mutants of S. typhimurium, we propose that Tap1 be designated T. denticola FliK.
One unexpected finding was that the Tap1 (FliK)-deficient mutant JS97 synthesized abundant levels of flaB flagellar filament RNA and yet lacked flagellar filaments and FlaB polypeptides. Presumably, either the flagellar filament proteins are synthesized and then rapidly degraded or there is an unknown form of translational control of filament protein synthesis. Enteric mutants with C-terminal deletions of FliK also fail to assemble flagellar filaments (14, 34); however, whether flagellar filament RNA is transcribed is not clear. This phenotype has been noted for other spirochete motility mutants as well. For example, we reconstructed the T. denticola flgE mutant HL51 (29) and found wild-type levels of flaB RNA. However, HL51 does not possess flagellar filament structures (21, 29). In the FlgE-deficient mutant HL51, the lack of a flagellar hook structure should prevent the filament from assembling. In contrast to the FlgE mutant, the Tap1 (FliK)-deficient mutant JS97 synthesizes an intact hook structure, albeit one that is abnormally long; however, the flagellar filament may be unable to be assembled because the FliK deficiency renders the cell unable to switch to filament export (34). Both of these T. denticola mutants, HL51 and JS97, together with a previously described chemically derived mutant of T. phagedenis (20) and a spontaneous mutant of Borrelia burgdorferi (30) have a flagellar filament structure-deficient phenotype despite adequate filament RNA expression, suggesting a common spirochete response to mutations within certain motility genes. The basis for this unusual regulation of spirochete flagellar filament protein synthesis is unknown.
The ermF-ermAM construct utilized by Li et al. possessed a putative transcription terminator (6). Consequently, we were concerned that the tap1 mutant JS97 was nonmotile because of polar effects on downstream genes. However, when the ermF-ermAM cassette was analyzed with the TERMINATOR or RNAFOLD program in the Wisconsin package no significant transcription terminator was identified. To show that transcription was not terminated, we removed the putative terminator and some noncritical DNA to create a second erythromycin resistance cassette. Both of these cassettes were incorporated into T. denticola tap1 and showed similar levels of fla operon RNA that were comparable to that of the wild type. Although quantitative RT-PCR or RNase protection studies were not performed, it is clear that levels of RNA downstream of the ermF-ermAM cassette were comparable to those of the wild type and were sufficient for synthesis and assembly of flagellar components such as FlgE. On the basis of the good transcription and translation of flgE in JS97, we believe that this cassette is useful for studying genes both within and outside operons.
In summary, using specific gene inactivation, we have shown that Tap1 is involved in the motility of T. denticola, perhaps by perturbation of the ability of the cell to monitor flagellar hook length or export. Various lines of evidence suggest that Tap1 performs a function analogous to that of FliK in other bacteria. Because the motility of spirochetes contributes to their pathogenesis, these studies could prove useful in creating specific avirulent or less-virulent strains as potential vaccine candidates.
ACKNOWLEDGMENTS
We acknowledge Mary Beth Kinoshita and Andrea Knaggs, Wadsworth Center Molecular Genetics and Electron Microscopy Core Facilities, Photography Unit, for technical assistance and Howard Kuramitsu and Hong Li for providing strains and advice on electroporation of spirochetes.
J.I. was supported in part by a basic research grant from Health Research Incorporated. This work was supported by Public Health Research Service grant AI34354 from the National Institutes of Health.
REFERENCES
- 1.Albertini A M, Caramori T, Crabb W D, Scoffone F, Galizzi A. The flaA locus of Bacillus subtilisis part of a large operon coding for flagellar structures, motility functions, and an ATPase-like polypeptide. J Bacteriol. 1991;173:3573–3579. doi: 10.1128/jb.173.11.3573-3579.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Altschul S F, Madden T L, Schaffer A A, Zhang J, Zhang Z, Miller W, Lipman D J. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 1997;25:3389–3402. doi: 10.1093/nar/25.17.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Charon N W, Goldstein S F, Block S M, Kurci K, Ruby J D, Kreiling J A, Limberger R J. Morphology and dynamics of protruding spirochete periplasmic flagella. J Bacteriol. 1992;174:832–840. doi: 10.1128/jb.174.3.832-840.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Compton T. Degenerate primers for DNA amplification. In: Innis M A, Gelfand D H, Sninsky J J, White T J, editors. PCR protocols: a guide to methods and applications. New York, N.Y: Academic Press, Inc.; 1990. pp. 39–45. [Google Scholar]
- 5.Dantuono L A. Characterization of the fla motility operon of Treponema phagedenis. M.S. thesis. Albany, N.Y: State University of New York; 1996. [Google Scholar]
- 6.Fletcher H M, Schenkein H A, Morgan R M, Bailey K A, Berry C R, Macrina F L. Virulence of a Porphyromonas gingivalis W83 mutant defective in the prtHgene. Infect Immun. 1995;63:1521–1528. doi: 10.1128/iai.63.4.1521-1528.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fraser C M, Casjens S, Huang W M, Sutton G G, Clayton R, Lathigra R, White O, Ketchum K A, Dodson R, Hickey E K, Gwinn M, Dougherty B, Tomb J F, Fleischmann R D, Richardson D, Peterson J, Kerlavage A R, Quackenbush J, Salzberg S, Hanson M, van Vugt R, Palmer N, Adams M D, Gocayne J, Weidman J, Utterback T, Watthey L, McDonald L, Artiach P, Bowman C, Garland S, Fujii C, Cotton M, Horst K, Roberts K, Hatch B, Smith H O, Venter J C. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature. 1997;390:580–586. doi: 10.1038/37551. [DOI] [PubMed] [Google Scholar]
- 8.Fraser C M, Norris S J, Weinstock G M, White O, Sutton G G, Dodson R, Gwinn M, Hickey E K, Clayton R, Ketchum K A, Sodergren E, Hardham J M, McLeod M P, Salzberg S, Peterson J, Khalak H, Richardson D, Howell J K, Chidambaram M, Utterback T, McDonald L, Artiach P, Bowman C, Cotton M D, Fujii C, Garland S, Hatch B, Horst K, Roberts K, Sandusky M, Weidman J, Smith H O, Venter J C. Complete genome sequence of Treponema pallidum, the syphilis spirochete. Science. 1998;281:375–388. doi: 10.1126/science.281.5375.375. [DOI] [PubMed] [Google Scholar]
- 9.Ge Y, Charon N W. Molecular characterization of a flagellar/chemotaxis operon in the spirochete Borrelia burgdorferi. FEMS Microbiol Lett. 1997;153:425–431. doi: 10.1111/j.1574-6968.1997.tb12606.x. [DOI] [PubMed] [Google Scholar]
- 10.Ge Y, Old I G, Saint Girons I, Charon N W. Molecular characterization of a large Borrelia burgdorferi motility operon which is initiated by a consensus ς70promoter. J Bacteriol. 1997;179:2289–2299. doi: 10.1128/jb.179.7.2289-2299.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gonzalez-Pedrajo B, Ballado T, Campos A, Sockett R E, Camarena L, Dreyfus G. Structural and genetic analysis of a mutant of Rhodobacter sphaeroidesWS8 deficient in hook length control. J Bacteriol. 1997;179:6581–6588. doi: 10.1128/jb.179.21.6581-6588.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hardham J M, Frye J G, Stamm L V. Identification and sequences of the Treponema pallidum fliM′, fliY, fliP, fliQ, fliR, and flhB′ genes. Gene. 1995;166:57–64. doi: 10.1016/0378-1119(95)00583-x. [DOI] [PubMed] [Google Scholar]
- 13.Heinzerling H F, Olivares M, Burne R A. Genetic and transcriptional analysis of flgB flagellar operon constituents in the oral spirochete Treponema denticolaand their heterologous expression in enteric bacteria. Infect Immun. 1997;65:2041–2051. doi: 10.1128/iai.65.6.2041-2051.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hirano T, Yamaguchi S, Oosawa K, Aizawa S. Roles of FliK and FlhB in determination of flagellar hook length in Salmonella typhimurium. J Bacteriol. 1994;176:5439–5449. doi: 10.1128/jb.176.17.5439-5449.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Holt S C. Anatomy and chemistry of spirochetes. Microbiol Rev. 1978;42:114–160. doi: 10.1128/mr.42.1.114-160.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kawagishi I, Homma M, Williams A W, Macnab R M. Characterization of the flagellar hook length control protein fliK of Salmonella typhimurium and Escherichia coli. J Bacteriol. 1996;178:2954–2959. doi: 10.1128/jb.178.10.2954-2959.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kimsey R B, Spielman A. Motility of Lyme disease spirochetes in fluids as viscous as the extracellular matrix. J Infect Dis. 1990;162:1205–1208. doi: 10.1093/infdis/162.5.1205. [DOI] [PubMed] [Google Scholar]
- 18.Klitorinos A, Noble P, Siboo R, Chan E C. Viscosity-dependent locomotion of oral spirochetes. Oral Microbiol Immunol. 1993;8:242–244. doi: 10.1111/j.1399-302x.1993.tb00567.x. [DOI] [PubMed] [Google Scholar]
- 19.Li H, Ruby J, Charon N, Kuramitsu H. Gene inactivation in the oral spirochete Treponema denticola: construction of an flgEmutant. J Bacteriol. 1996;178:3664–3667. doi: 10.1128/jb.178.12.3664-3667.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Limberger R J, Charon N W. Treponema phagedenishas at least two proteins residing together on its periplasmic flagella. J Bacteriol. 1986;166:105–112. doi: 10.1128/jb.166.1.105-112.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Limberger, R. J., and L. L. Slivienski. 1998. Unpublished observations.
- 22.Limberger R J, Slivienski L L, El-Afandi M C, Dantuono L A. Organization, transcription, and expression of the 5′ region of the fla operon of Treponema phagedenis and Treponema pallidum. J Bacteriol. 1996;178:4628–4634. doi: 10.1128/jb.178.15.4628-4634.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Limberger R J, Slivienski L L, Samsonoff W A. Genetic and biochemical analysis of the flagellar hook of Treponema phagedenis. J Bacteriol. 1994;176:3631–3637. doi: 10.1128/jb.176.12.3631-3637.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Limberger R J, Slivienski L L, Yelton D B, Charon N W. Molecular genetic analysis of a class B periplasmic flagellum gene of Treponema phagedenis. J Bacteriol. 1992;174:6404–6410. doi: 10.1128/jb.174.20.6404-6410.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Macnab R M. Genetics and biogenesis of bacterial flagella. Annu Rev Genet. 1992;26:131–158. doi: 10.1146/annurev.ge.26.120192.001023. [DOI] [PubMed] [Google Scholar]
- 26.Maniatis T, Fritsch E F, Sambrook J. Molecular cloning: a laboratory manual. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1982. [Google Scholar]
- 27.Muramoto K, Makishima S, Aizawa S I, Macnab R M. Effect of cellular level of FliK on flagellar hook and filament assembly in Salmonella typhimurium. J Mol Biol. 1998;277:871–882. doi: 10.1006/jmbi.1998.1659. [DOI] [PubMed] [Google Scholar]
- 28.Ohnishi K, Ohto Y, Aizawa S, Macnab R M, Iino T. FlgD is a scaffolding protein needed for flagellar hook assembly in Salmonella typhimurium. J Bacteriol. 1994;176:2272–2281. doi: 10.1128/jb.176.8.2272-2281.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ruby J D, Li H, Kuramitsu H, Norris S J, Goldstein S F, Buttle K F, Charon N W. Relationship of Treponema denticolaperiplasmic flagella to irregular cell morphology. J Bacteriol. 1997;179:1628–1635. doi: 10.1128/jb.179.5.1628-1635.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sadziene A, Thomas D D, Bundoc V G, Holt S C, Barbour A G. A flagella-less mutant of Borrelia burgdorferi. Structural, molecular and in vitrofunctional characterization. J Clin Investig. 1991;88:82–92. doi: 10.1172/JCI115308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schleicher & Schuell. Transfer and immobilization of nucleic acids to S&S solid supports. Keene, N.H.: Schleicher & Schuell, Inc.; 1987. [Google Scholar]
- 32.Simonson L G, Goodman C H, Bial J J, Morton H E. Quantitative relationship of Treponema denticolato severity of periodontal disease. Infect Immun. 1988;56:726–728. doi: 10.1128/iai.56.4.726-728.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Smibert R M. Genus III. Treponema Schaudin 1905, 1728AL. In: Krieg N R, Holt J G, editors. Bergey’s manual of systematic bacteriology. Vol. 1. Baltimore, Md: The Williams and Wilkins Co.; 1984. pp. 49–57. [Google Scholar]
- 34.Williams A W, Yamaguchi S, Togashi F, Aizawa S I, Kawagishi I, Macnab R M. Mutations in fliK and flhB affecting flagellar hook and filament assembly in Salmonella typhimurium. J Bacteriol. 1996;178:2960–2970. doi: 10.1128/jb.178.10.2960-2970.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]