Abstract
England has experienced a heavy burden of COVID-19, with multiple waves of SARS-CoV-2 transmission since early 2020 and high infection levels following the emergence and spread of Omicron variants since late 2021. In response to rising Omicron cases, booster vaccinations were accelerated and offered to all adults in England. Using a model fitted to more than 2 years of epidemiological data, we project potential dynamics of SARS-CoV-2 infections, hospital admissions and deaths in England to December 2022. We consider key uncertainties including future behavioural change and waning immunity and assess the effectiveness of booster vaccinations in mitigating SARS-CoV-2 disease burden between October 2021 and December 2022. If no new variants emerge, SARS-CoV-2 transmission is expected to decline, with low levels remaining in the coming months. The extent to which projected SARS-CoV-2 transmission resurges later in 2022 depends largely on assumptions around waning immunity and to some extent, behaviour, and seasonality.
Subject terms: Population dynamics, Bayesian inference, Dynamical systems, Complexity, Epidemiology
This mathematical modelling study projects the dynamics of SARS-CoV-2 in England until the end of 2022 assuming that the Omicron BA.2 sublineage remains dominant. They show that booster vaccination was highly effective in mitigating severe outcomes and that future dynamics will depend greatly on assumptions about waning immunity.
Introduction
Over two years into the COVID-19 pandemic, more than 500 million confirmed cases and 6 million deaths have been attributed to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) worldwide1. In England as of 12th May 2022, cumulative confirmed COVID-19 cases exceed 18 million, with more than 700,000 hospitalisations and 150,000 deaths within 28 days of a positive test being recorded, respectively2. Different variants of SARS-CoV-2 have emerged, with five (Alpha, Beta, Gamma, Delta and Omicron) currently designated as variants of concern (VOC) associated with either increased transmissibility, severity, or changes in immunity by the World Health Organisation3.
England saw the emergence and fixation of the Alpha B.1.1.7 variant in late 2020, which was subsequently overtaken by the Delta B.1.617.2 variant in Spring 2021, followed by the Omicron B.1.1.529 variant in late 2021 and the Omicron BA.2 sublineage in early 2022. Various public health and social measures (PHSMs) have been implemented and relaxed at different times to control SARS-CoV-2 transmission in England, including national lockdowns, staged relaxations of lockdowns, tiered regional restrictions4, and so-called ‘Plan B’ measures which were introduced in response to Omicron’s emergence. Following the large wave of Omicron transmission beginning in late 2021, all legal restrictions in England were lifted on 24th February 20225.
Safe and effective COVID-19 vaccines have been developed at unprecedented speed, with six currently approved for use by the UK’s Medicines and Healthcare products Regulatory Agency6. The COVID-19 vaccine rollout in England began on the 8th of December 2020 and to date more than 44 million people have received at least their first COVID-19 vaccine dose2. The rollout of vaccines followed guidance issued by the UK’s Joint Committee on Vaccination and Immunisation (JCVI)7, with vaccines being targeted to health and social care workers and those in the highest risk categories first. Vaccines were then offered to sequentially younger age groups of adults (18 years and above). In August 2021, children aged 16 and 17 years old and clinically vulnerable children aged 12–15 were offered COVID-19 vaccines8. In September 2021, healthy 12–15-year-olds in England were offered their first COVID-19 vaccination and from April 2022 vaccination has been extended to 5–11-year-olds9.
A COVID-19 booster vaccination programme began in England in September 2021, initially targeting the same highest-risk priority groups that were first vaccinated. A full dose of the Pfizer/BioNTech or a half dose of the Moderna vaccine are recommended as a booster dose, regardless of what vaccine was received previously, to those at least 6 months after their primary course of vaccination. On 15th November 2021, the JCVI issued advice recommending that the widespread COVID-19 booster vaccination programme be extended to individuals aged 40–49 years10. However, following the World Health Organization (WHO) designating the Omicron SARS-CoV-2 variant as a variant of concern in late November 202111, with Omicron cases being detected in England12 and with two vaccine doses found to offer little protection against Omicron a few months after vaccination13, the JCVI announced an extension and acceleration of the booster vaccination campaign14. This announcement recommended that 18–39 year olds should be offered booster vaccinations in descending age order, that the minimum gap between the primary vaccination course and the booster vaccine should be reduced from 6 to 3 months, and that children aged 12–15 years of age should receive their second vaccine dose15. The JCVI also recommended that children aged 16 and 17 years of age could receive booster vaccinations at least 3 months after completion of their primary vaccination course16. Reduced dose COVID-19 vaccinations were offered to the most vulnerable 5–11 year old children from the end of January 202217, and in February 2022 the JCVI recommended a non-urgent extension of this rollout to children aged 5–11 who are not in a clinical risk group18. Given the high levels of Omicron transmission in England since late 2021, emergence of the BA.2 sublineage, the accelerated booster vaccination campaign, and evidence suggesting that vaccine protection wanes over time19,20, it is important to assess the likely medium-term dynamics of SARS-CoV-2 transmission as the Omicron wave subsides and as, in the absence of legal restrictions, behaviour returns to a (potentially new) baseline.
Using an age-structured deterministic compartmental model of SARS-CoV-2 community transmission, we explore the consequences of key uncertainties on projected COVID-19 cases, hospitalisations, and deaths to December 202221. We fit the model to regional data up to May 2022 on COVID-19 deaths, hospitalisations, and hospital bed occupancy, as well as PCR prevalence, seroprevalence, and the emergence of the Alpha B.1.1.7, Delta B.1.617.2, and Omicron B.1.1.529 VOCs. We also integrate data on: the number of vaccinations delivered at the level of NHS England regions and by age group over time, historic school attendance and community mobility data to inform behavioural changes over time since the start of the COVID-19 pandemic, and genomic sequencing data to inform the proportion of Omicron cases attributable to the BA.1 and BA.2 sublineages over time. We consider the effects of future behaviour change, waning immunity, seasonality, and vaccination of children and adolescents at different levels of uptake on potential future dynamics of SARS-CoV-2 transmission in England to December 2022. We also assess the effectiveness of COVID-19 booster vaccinations by exploring counterfactual booster vaccination scenarios and their effect on SARS-CoV-2 transmission in England between October 2021 and December 2022.
Results
Model fitting & uncertainty
Our compartmental model (Fig. 1) fits the observed dynamics of SARS-CoV-2 community transmission during the COVID-19 epidemic in England between March 2020 and May 2022 (Figs. 2, S1A, B), reproducing NHS England region-specific observed deaths, hospitalisations, hospital and intensive care unit (ICU) bed occupancy, PCR prevalence, and seropositivity. Following the initial differential evolution Markov chain Monte Carlo (DE-MCMC) fitting, the model applies continuous-time multipliers to the infection fatality, hospital admissions, hospital bed occupancy and the ICU bed occupancy rates for each region in order to match real-world measured outcomes (Fig. S1C). There are notable peaks in the fatality rate adjustment corresponding to the initial SARS-CoV-2 wave in early 2020 and to the Alpha B.1.1.7 wave in the winter of 2020–21 (Fig. S1C). All four rate adjustments have reached low levels in early 2022; we carry forward the last adjustment for forward projections (Fig. S1C).
Fig. 1. Compartmental model diagram.
A three-variant deterministic dynamic susceptible-exposed-infected-recovered (SEIR) compartmental model with clinical and sub-clinical infectious pathways and with vaccination describes SARS-CoV-2 transmission in England. We model seven NHS England regions66 separately, with each divided into 16 five-year age groups: 0–4 years up to 70–74 years, and 75 years and older. The model incorporates COVID-19 vaccination with two vaccine products (corresponding to the viral-vector (Va) and mRNA-based (Vb) vaccines in use in England), each with first- (Va1, Vb1) and second-dose (Va2, Vb2) protection, and each with lower levels of protection for individuals who received their primary vaccinations but no booster vaccine and have waned (Va2w, Vb2w). A detailed description is provided within the Supplementary Information.
Fig. 2. Comparison between aggregated model fits and epidemiological data from England between March 2020 and May 2022.
Black lines show reported data, with black ribbons showing 95% confidence intervals for PCR prevalence. Coloured lines and shaded areas show medians, 50% and 90% interquantile ranges from the fitted model. The original model fitting is done independently for each NHS England region (see Fig. S1A, B), with the aggregated model output for the whole of England shown here. a COVID-19 deaths over time, where data was provided by the UK Health Security Agency (UKHSA). b COVID-19 hospital admissions over time, where data was provided by NHS England. c COVID-19 hospital bed occupancy over time, where data was provided by NHS England. d COVID-19 ICU bed occupancy over time, where data was provided by NHS England. e COVID-19 PCR prevalence over time, where publicly-available PCR prevalence data was obtained from the Office for National Statistics’ COVID-19 Infection Survey (ONS-CIS)53. The data sources for COVID-19 deaths, hospital admissions, hospital and ICU bed occupancy are unpublished and not publicly available but are closely aligned with the UK Government’s COVID-19 dashboard2. ICU intensive care unit. NHS National Health Service.
The model also captures the emergence and spread of the Alpha B.1.1.7, Delta B.1.617.2, and Omicron B.1.1.529 VOCs in late 2020, early 2021, and late 2021, fitting to the prevalence of S gene target failure (Fig. S2a, c) as a proxy for the proportion of cases attributable to the Alpha and Omicron variants, and to the proportion of Delta sequenced cases (Fig. S2b). Model estimates for increased transmissibility of Alpha relative to previously circulating SARS-CoV-2 variants, of Delta relative to Alpha, and of the earlier Omicron BA.1 sublineage relative to Delta, as well as the overall estimated increases in transmissibility between early SARS-CoV-2 variants and each VOC and Omicron sublineage are given in Tables S1A, B, respectively. Note that we assume that the Omicron BA.2 sublineage confers a 50% increase in transmissibility compared to the previous Omicron BA.1 sublineage.
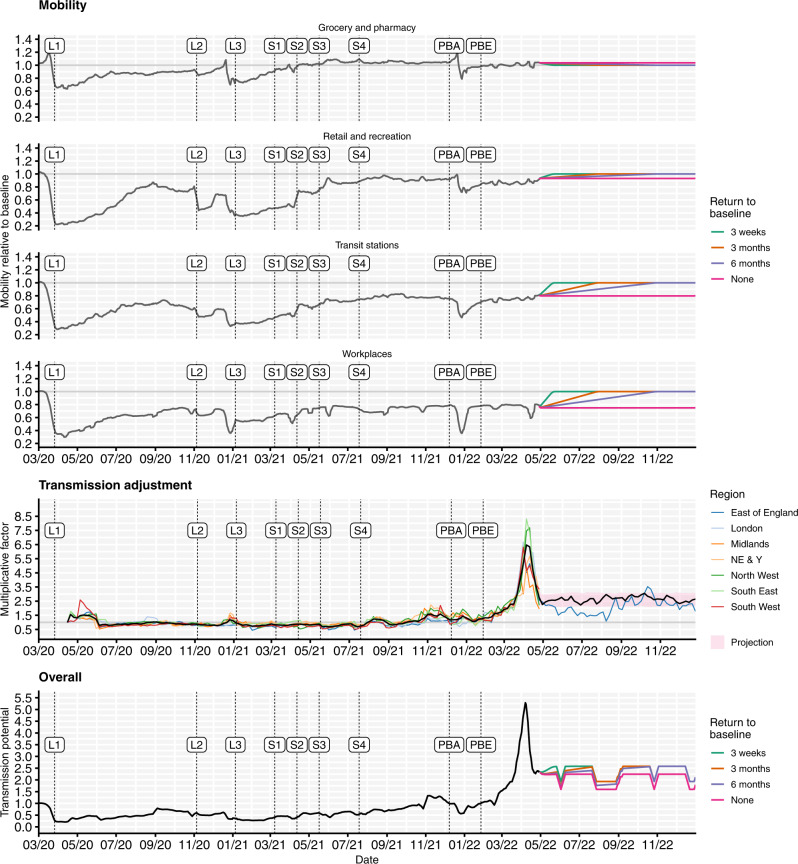
To capture historic behavioural changes, the model uses Google Community Mobility indices22 over time to derive contact rates for each NHS England region modelled, based upon a measured relationship between Google Mobility indices and age-specific contact rates as measured by the CoMix study23, and in combination with school attendance data24 and assumptions about school terms (Fig. 3). The model also fits a time-varying transmission adjustment component for each NHS England region in order to capture additional variability in transmission that is not explained by the mobility data (Fig. 3, middle row; Methods). To project behavioural changes forwards from May to December 2022, we combine various assumptions on future mobility changes (Fig. 3) with simulated trajectories for future transmission adjustments based on the historic fitted transmission adjustment (full details are given in the Methods section).
Fig. 3. Mobility scenarios, transmission adjustments and overall transmission potential for the fitted model, shown from March 2020 to December 2022.
Top: Historic Google Community Mobility data22 (grey) and assumed future mobility in England for no change (pink), a 3-week return to pre-pandemic baseline levels (green), a 3-month return to pre-pandemic baseline levels (orange) and a 6-month return to pre-pandemic baseline levels (purple) scenarios used for model projections. Mobility indices are measured relative to baseline mobility levels recorded during early 2020, prior to the COVID-19 pandemic. The beginning of each lockdown and each roadmap Step4 is marked with a vertical dashed line and ‘L’ and ‘S’ labels, respectively. Vertical dashed lines with ‘PBA’ and ‘PBE’ labels correspond to the announcement of ‘Plan B’ measures for England on 8th December 2021 and the ending of these measures on 27th January 202267. Middle: Fitted transmission adjustments between April 2020 and May 2022 by NHS England region (coloured lines) and the average across regions (black line), example projection between May and December 2022 for East of England (blue line) and mean (black line) and interquartile range (red shaded) for projected transmission adjustments between May and December 2022 across NHS England regions. Bottom: The overall transmission potential captures the combined impact of mobility and transmission adjustments on the time-varying potential for effective transmission, ignoring the impact of immunity and novel variants, though including the impact of school vacation periods. NE & Y North East & Yorkshire. NHS National Health Service.
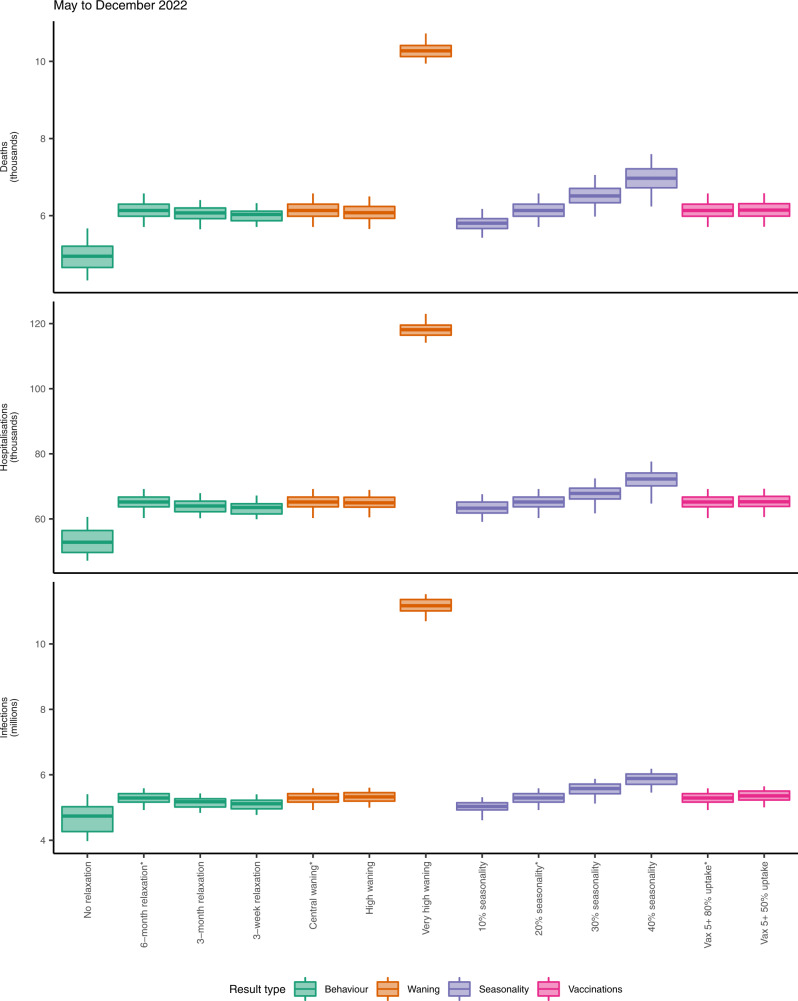
We present the majority of results here by plotting the median and interquantile ranges of a number of simulated future trajectories of SARS-CoV-2 transmission in England, but it is important to note that individual epidemic trajectories can fall outside of the model’s projection intervals (Fig. 4c). A comparison of the projected cumulative number of SARS-CoV-2 infections, hospital admissions and deaths between May and December 2022 across all scenarios considering future uncertainties (namely, behaviour, waning immunity, seasonality, and vaccination uptake for children aged 5 and above) is shown in Fig. 5. Detailed results related to each type of uncertainty considered are given in the Supplementary Information (Figs. S3, S4A, B, S5–S8). A complete list of scenarios considered and key assumptions for each scenario is given in Table 1.
Fig. 4. Summary of basecase model fits and projections and key results on uncertainty, behaviour, booster vaccinations and waning immunity.
a The number of COVID-19 hospital admissions in England, for the basecase scenario, between March 2020 and December 2022. Black lines show reported data, provided by UKHSA. Coloured lines and shaded areas show medians, 50% and 90% interquantile ranges from the fitted model and from the model projection. b PCR prevalence in England, for the basecase scenario, between March 2020 and December 2022. The black ribbons show 95% confidence intervals for PCR prevalence data. Coloured lines and shaded areas show medians, 50% and 90% interquantile ranges from the fitted model and from the model projection. c The fitted and projected number of COVID-19 deaths in England between March and December 2022, shown for the very high waning scenario (see Table S4). The black line shows the median trajectory of COVID-19 deaths in England over time, with the shaded areas showing the 50% and 90% interquantile ranges. Individual model trajectories are plotted in coloured lines. d The effect of future behaviour on COVID-19 deaths and cumulative deaths (thousands) over time is shown with four scenarios for future mobility: a 3-week, a 3-month and a 6-month return to baseline levels, and a no change scenario (see Table 1). Coloured lines and shaded areas show medians, 50% and 90% interquantile ranges from the fitted model and from the model projection. e The effect of booster vaccination policy on cumulative infections and deaths since October 2021 is shown with four scenarios for booster policies (Table 1). Coloured lines and shaded areas show medians, 50% and 90% interquantile ranges from the fitted model and from the model projection. f The effect of waning immunity on cumulative infections and deaths between March and December 2022 is shown with three scenarios for waning (Tables 1, S4). Coloured lines and shaded areas show medians, 50% and 90% interquantile ranges from the fitted model and from the model projection. The basecase scenarios (shown in a, b) and scenarios marked with an asterisk (*) are equivalent.
Fig. 5. Summary of projected cumulative numbers (log-scale) of COVID-19 deaths (thousands), hospital admissions (thousands) and infections (millions) in England between May and December 2022, across behavioural, waning, seasonality and vaccination scenarios considered.
Each box plot shows the projected median (central line), 5th (minima), 25th (lower bound of box), 75th (upper bound of box) and 95th (maxima) percentile values across 100 simulations for each relevant scenario, calculated between May and December 2022. Scenarios are coloured according to the result type (from left to right: behaviour, waning immunity, seasonality, and vaccination policies for children aged 5 years and older). A full list of scenarios and relevant modelling assumptions is given in Table 1. Scenarios marked with an asterisk (*) are equivalent and correspond to the basecase scenario. N.B. The y-axes are plotted on a log scale and are truncated and do not extend to zero.
Table 1.
List of modelling scenarios and key assumptions
| Scenario | Result type | Assumptions | ||||
|---|---|---|---|---|---|---|
| Mobility | Waning | Booster uptake | Seasonality | Vaccine policy | ||
| Basecase* | - | 6-month | Central | Actual | 20% | 5+ 80% uptake |
| No return | Behaviour | No change | Central | Actual | 20% | 5+ 80% uptake |
| 6-month return* | 6-month | Central | Actual | 20% | 5+ 80% uptake | |
| 3-month return | 3-month | Central | Actual | 20% | 5+ 80% uptake | |
| 3-week return | 3-week | Central | Actual | 20% | 5+ 80% uptake | |
| No boosters | Boosters eligibility/ uptake (relative to second dose levels) | 6-month | Central | None | 20% | 5+ 80% uptake |
| 50+ boosters | 6-month | Central | 95% in 50+ only | 20% | 5+ 80% uptake | |
| Actual boosters* | 6-month | Central | Age-specific measured uptake as of 14th April 2022†. Individuals aged 50 and above have at least 86.1% and at most 96.7% uptake. Individuals aged 15-49 have at least 40% and at most 80.9% uptake, relative to second dose levels of uptake. | 20% | 5+ 80% uptake | |
| High uptake | 6-month | Central | 90% in <50 s; 98% in 50+ | 20% | 5+ 80% uptake | |
| Central waning* | Waning | 6-month | Central | Actual | 20% | 5+ 80% uptake |
| High waning | 6-month | High | Actual | 20% | 5+ 80% uptake | |
| Very high waning | 6-month | Very high | Actual | 20% | 5+ 80% uptake | |
| 10% | Seasonality | 6-month | Central | Actual | 10% | 5+ 80% uptake |
| 20%* | 6-month | Central | Actual | 20% | 5+ 80% uptake | |
| 30% | 6-month | Central | Actual | 30% | 5+ 80% uptake | |
| 40% | 6-month | Central | Actual | 40% | 5+ 80% uptake | |
| 5+, 80%* | Vaccine uptake for children | 6-month | Central | Actual | 20% | 5+ 80% uptake |
| 5+, 50% | 6-month | Central | Actual | 20% | 5+ 50% uptake | |
Scenarios marked with an asterisk (*) are equivalent. Mobility scenarios are shown in Fig. 3. Assumptions for waning scenarios are given in Table S4. Assumptions for vaccine effectiveness against different SARS-CoV-2 variants are given in Table S2. Assumptions for cross-protection against different SARS-CoV-2 variants given prior infection are given in Table S3.
†The detailed age-specific probability of receiving a booster vaccine for this scenario is listed in Table S5B in the Supplementary Information.
Basecase scenario
Following the Omicron BA.1 and BA.2 waves of transmission beginning in late 2021 and early 2022, our basecase scenario projects a continued reduction in transmission until August 2022, with a resurgence in transmission from August 2022 onwards (Fig. 4a, b). This period of low infection incidence results from very high levels of immunity derived from both vaccination and prior infection within the population in early 2022 following the Omicron waves of infection (Fig. S8) and assumes that no novel variants of SARS-CoV-2 emerge and outcompete the current Omicron BA.2 sublineage.
Future behaviour
Assumptions about future levels of mobility have a small effect on projected dynamics of SARS-CoV-2 transmission in 2022 (Figs. 4d, 5 and S3). We consider four scenarios for future mobility following measured levels on 29th April 2022 (no change versus a 3-week, 3-month or 6-month return to pre-pandemic baseline levels, shown in Fig. 3). All behavioural change scenarios considered project a reduction in SARS-CoV-2 transmission to very low levels during the summer of 2022 (Fig. S3). However, the timing and extent of the projected resurgence later in 2022 depend on the assumed changes in future mobility (Fig. S3), with a later resurgence occurring if mobility remains at current levels. The timing and speed of the return to baseline mobility interacts with school terms and other modelling assumptions such as waning and seasonality. A 3-week return to baseline mobility results in the earliest projected resurgence, while a 6-month return to baseline mobility results in a later, but steeper resurgence in transmission towards December 2022 (Fig. S3).
Booster vaccinations
An influential factor considered here is the availability and uptake of COVID-19 booster vaccinations (Figs. 4e and S4A, B). Here, we consider a number of scenarios exploring the effects of different booster vaccination rollouts on SARS-CoV-2 transmission in England between October 2021 and December 2022 (Table 1). A counterfactual scenario in which no COVID-19 booster programme was deployed has a large effect, with an additional 23,400 deaths and 108,000 hospital admissions between October 2021 and December 2022, compared to our basecase scenario using levels of booster vaccination uptake measured in April 2022 (Fig. S4A Tables).
There are minimal differences among the remaining three booster vaccination scenarios which consider age-specific booster uptake as measured on the 14th of April 2022, booster vaccines being administered to 95% of individuals aged 50 years and above only, and booster vaccines being administered to 90% of individuals aged 15–49 and 98% of individuals aged 50 years and above, representing higher booster uptake than was measured to date (Table 1). Burdens are similar across these three scenarios, with the scenario where booster vaccinations were only offered to individuals aged 50 and above resulting in higher levels of infections and deaths during the first wave of Omicron transmission in late 2021 to early 2022 (Figs. 4e and S4A, B).
Waning immunity
The extent to which vaccine-induced and natural immunity wanes over time is a key driver of projected transmission dynamics in 2022 (Figs. 4f, 5 and S5). We consider three waning immunity scenarios which apply to vaccinated individuals and to individuals who have recovered from a prior infection: basecase, high and very high waning (Table S4). All three of these scenarios assume booster vaccination uptake as measured in April 202225. The basecase waning scenario is projected to result in a total of 5.29 million infections, 65,200 hospital admissions and 6130 deaths between May and December 2022 (Fig. S5 Tables). The high waning scenario is projected to result in slightly more infections but slightly fewer hospital admissions and deaths, compared to the central waning scenario, over this time period. The very high waning scenario results in an earlier resurgence beginning in June 2022, with an additional 6 million infections, 52,800 hospital admissions and 4170 deaths projected between May and December 2022, compared to the basecase waning scenario (Fig. S5 Tables).
Seasonality
Different assumptions related to the extent of seasonality also influence the projected dynamics of SARS-CoV-2 transmission (Figs. 5 and S6). Scenarios assuming the most extreme seasonal effects result in the largest cumulative burdens between May and December 2022 (Figs. 5, S6). However, scenarios assuming the least extreme seasonal effects result in resurgences in transmission earlier in 2022 than scenarios with greater seasonality (Fig. S6). To fully explore projected SARS-CoV-2 dynamics with different seasonal effects requires consideration of much longer time horizons than those considered in this study.
Vaccination of children
We consider two scenarios for the vaccination of children aged 5 years and above between May and December 2022 (Fig. 5 and S7). Of all the uncertainties considered here, using different assumptions related to the vaccination of children had the smallest overall effect on projected SARS-CoV-2 infection and disease until December 2022 (Fig. 5). The scenario assuming higher uptake of COVID-19 vaccinations in children aged 5 and above is projected to result in small reductions in infections, hospital admissions and deaths between May and December 2022 (Fig. S7 Table), but not to significantly alter projected transmission dynamics (Fig. S7). This is due to high levels of infection in the model in these age groups during the autumn of 2021 and winter 2021–22, leading to naturally acquired immunity (Fig. S8).
Discussion
We have fitted a deterministic compartmental model of SARS-CoV-2 transmission to data between March 2020 and May 2022 on COVID-19 deaths, hospital admissions, hospital bed and ICU bed occupancy, PCR prevalence, seroprevalence, and the emergence and spread of the Alpha B.1.1.7, Delta B.1.617.2 and Omicron B.1.1.529 variants of concern, including the BA.1 and BA.2 sublineages of Omicron, and incorporating additional data on vaccination coverage and behaviour over time21. Projecting SARS-CoV-2 transmission in England forwards to December 2022, we have considered a number of future uncertainties around behaviour, waning immunity, seasonality, and vaccination of children aged 5 years and above, as well as retrospectively assessing the effectiveness of booster vaccinations.
Modelled levels of immunity in England as a result of the extensive COVID-19 primary and booster vaccination campaigns and following the recent waves of Omicron infections (Fig. S8) suggest that in the absence of further new variants or Omicron sublineages outcompeting the currently-dominant Omicron BA.2 sublineage, SARS-CoV-2 transmission will continue to fall in the next few months, remaining at low levels during the summer months of 2022. Our modelling suggests that higher SARS-CoV-2 transmission may resurge later in 2022, with the timing and extent of this resurgence largely dependent on our modelling assumptions related to waning immunity and, to some extent, future behaviour, and seasonality. There remains significant uncertainty around both the extent and the timescale to which immunity wanes, as well as future behaviour and the speed and level that any return towards pre-pandemic baseline behaviours will reach.
Our modelling suggests that the COVID-19 booster vaccination campaign was highly effective in reducing transmission and in particular mitigating severe outcomes during recent months, with a counterfactual scenario without any booster vaccinations projected to result in a large peak of hospital admissions in England exceeding the peak levels recorded throughout the COVID-19 pandemic to date (Fig. S4A). Importantly, the model shows that this effect mostly derives from boosting immunity in those over 50 years of age, with little additional benefit being derived from boosting younger age groups. We also show that the projected dynamics of SARS-CoV-2 transmission depend on modelling assumptions about the extent of seasonal effects and that achieving higher levels of vaccination coverage for children aged 5 years and older will not significantly influence the epidemiological dynamics of SARS-CoV-2 in England until December 2022, primarily due to the late initiation of paediatric vaccination, high rates of natural immunity that has already accumulated (Fig. S8), the lower risk of severe disease, and lower inherent susceptibility to infection for this age group26.
Although a number of our modelling scenarios project resurgences in SARS-CoV-2 transmission occurring later in 2022, none of these are expected to reach the peak levels of infections, hospital admissions or deaths recorded so far during the COVID-19 pandemic in England, at least during the time periods we are considering. This is largely due to very high levels of vaccine and natural protection that have built up in the population, particularly following the recent Omicron BA.1 and BA.2 waves of infection, and which under our central waning immunity scenarios remain high through December 2022 (Fig. S8). However, it is important to note that we do not consider any projected SARS-CoV-2 dynamics past December 2022. Since many of the scenarios considered here appear to be resurging towards the end of the simulation time period, comparing overall burdens across these scenarios must therefore be done with caution. Moreover, although we consider differential contact-making across the 5-year age groups in the model, we do not explore any differences in contact-making behaviour within age-groups, such as for individuals in vulnerable or at-risk groups. These mixing assumptions may not be sufficient to capture all heterogeneities in behaviour that could lead to some individuals avoiding SARS-CoV-2 infection.
Furthermore, these modelling scenarios do not consider the introduction of any additional SARS-CoV-2 variants, or sublineages of Omicron aside from BA.1 and BA.2, which may possess characteristics such as increased transmissibility, immune evasion, or increased pathogenicity relative to existing variants and sublineages circulating in the population. As we have seen in England, novel VOCs with transmission advantages can spread to dominance very quickly (Fig. S2), and in particular the introduction of a new VOC which evades existing immune protection can significantly alter the dynamics of an epidemic, even in a previously highly immune population (Fig. S8), as we have seen with the Omicron BA.1 sublineage. Since the earliest SARS-CoV-2 variants, we have observed both increases and reductions in the intrinsic pathogenicity of the virus as different VOCs have emerged and spread to dominance globally27–30. The currently-dominant Omicron VOC has reduced pathogenicity compared to the previously-circulating Delta VOC29, and these reduced levels of severity are carried forward in our modelling projections. We do not explore the effects of any future change in intrinsic pathogenicity in this work.
In addition to future emergence of VOCs, it is impossible to predict future policy or behavioural changes with any certainty. It remains unclear whether mobility and behaviour will return to the same levels seen prior to the COVID-19 pandemic. We have considered one scenario where behaviour remains largely unchanged, and three scenarios in which mobility increases to pre-pandemic levels in 3 weeks, 3 months, or 6 months, to capture this uncertainty. We do not consider scenarios where behaviour increases above the baseline levels recorded prior to the COVID-19 pandemic, which may increase the extent and/or change the timing of any future resurgences if such an extreme behavioural change were to occur. In addition to longer-term changes, we are not able to forecast sudden shorter-term behavioural changes that have been observed in the data, e.g., around Christmas holidays (Fig. 3), although our 3-week return to baseline mobility scenario is intended to explore the consequences of a sudden increase in mobility (Fig. S3). Behavioural changes can also occur in response to awareness of a growing or declining epidemic31,32, which is something that we do not capture in any forward projections.
There are a number of other limitations in this work which are important to consider. The fitted model does not accurately capture the dynamics of PCR prevalence in England from March 2022 onwards, during Omicron BA.2’s dominance (Figs. 2, S1A, B). The modelled transmission adjustment during this time increases, temporarily, to an extremely high level (Fig. 3). This suggests that we may not have captured some properties of the Omicron BA.2 sublineage, e.g., if BA.2’s transmission advantage over BA.1 is larger than the assumed 50%, or if BA.2 has a shorter serial interval33 or longer infectious period. Additionally or alternatively, it may be that behaviours that are not captured within mobility data—such as mask wearing, physical distancing, and testing—have changed substantially in recent months following the removal of all legal restrictions in February 20225,34. Further, our modelled estimates of the proportion of the population in England who have some form of immunity against SARS-CoV-2 are high (Fig. S8), which may explain the poor fitting to PCR prevalence during the Omicron BA.2 era, particularly if we are overestimating the level of protection conferred by vaccines and/or underestimating the rate at which immune protection from SARS-CoV-2 infection and from COVID-19 vaccination is lost.
The extent to which immunity from a prior infection and/or from COVID-19 vaccination and boosting wanes is uncertain. Individuals may retain very long-term protection against severe outcomes but have faster rates of waning against less severe outcomes such as mild or asymptomatic infection. In our basecase scenario, we model waning vaccine protection for individuals who do not receive a booster vaccination by moving them into a vaccinated and waned disease state where their vaccine protection against different SARS-CoV-2 outcomes is reduced to different extents (see assumptions in Tables S2, S5B). Vaccinated and waned individuals and individuals who have recovered following a SARS-CoV-2 infection can then further wane back to a completely susceptible disease state (based on assumed rates of waning shown in Table S4). For our basecase scenario, we have parameterised these waning rates in relation to measured reductions in protection against severe outcomes, but our assumption that individuals return to being completely naive to infection may still be overly pessimistic. On the other hand, our basecase scenario assumes that individuals who receive booster vaccinations do not wane back to complete susceptibility and that the protection afforded by their vaccinations remains at least at second-dose assumed levels throughout the time periods considered here (see vaccine effectiveness assumptions in Table S2).
Over the long term, individuals in the model who receive their first booster vaccination will eventually reach the end of their second-dose duration, where the same probability of boosting remains; therefore, some individuals in the model will continually receive boosters and retain second-dose levels of protection, whilst some individuals are not boosted by their second or third decision point and move into lower levels of protection. Given our assumed second-dose duration (see Table S5B) and given that boosters were first administered in England in September 2021, the earliest that individuals in the model will reach their second booster decision point is April 2022. Given that the JCVI in England has recommended a spring 2022 COVID-19 booster vaccine for the most vulnerable individuals who received their first boosters in September and October 202135, ahead of a wider autumn 2022 booster programme, we think that it is reasonable to assume (i.e., as we have done in our central basecase scenario) that the majority of individuals will continue to receive booster vaccines. However, the modelling scenarios with higher rates of waning (high and very high waning, see Table S4) can be considered as a sensitivity analysis looking at a situation where fewer people continue to be boosted.
Throughout this work we have also assumed identical levels of protection and rates of waning across age groups, and we do not differentiate across risk groups. It is likely that protection will differ from person to person and that more clinically vulnerable individuals may have lower levels of protection or faster reductions in immunological protection following a previous infection or a vaccination. Future work needs to consider differential waning over time whereby higher protection against severe outcomes can be maintained but protection against infection is reduced, as well as of more complex immune dynamics induced by the numerous possible orders and timings of infections, vaccinations, reinfections and booster vaccinations and heterogeneity in individual’s immune responses.
We have assumed that following an infection with Omicron, individuals have complete cross protection against reinfection with Omicron or any other pre-existing variant (see cross protection assumptions in Table S3) whilst they remain in the recovered disease state. If reinfections following early Omicron infections are occuring36,37, particularly since the BA.2 Omicron sublineage has become dominant in England33 and other sublineages are emerging worldwide, we may see a slower reduction in SARS-CoV-2 transmission in England than our modelling projects, and the potential for higher levels of transmission throughout the time period under consideration.
Following the Omicron BA.1 and BA.2 waves of SARS-CoV-2 transmission, and an extensive vaccination and booster vaccination campaign, our work suggests that levels of immunity in England are sufficiently high to lead to a sustained reduction in transmission in the coming months, assuming no new variants of concern emerge. The extent to which SARS-CoV-2 transmission resurges later in 2022 depends upon a combination of factors, including behaviour, the rate that immunity wanes, and seasonality. It is clear that the extent to which immunity, both derived from natural infection and from COVID-19 vaccination, wanes will become extremely important in the medium to long term. It is crucial to improve our current understanding of the dynamics of SARS-CoV-2 immunity and to consider the consequences of this on community transmission in the context of varying levels of PHSMs and vaccination coverage, both in England and worldwide.
Methods
Epidemiological model
We use an age-structured and region-specific deterministic dynamic compartmental model of SARS-CoV-2 transmission (Fig. 1), building on previous work23,38–42. Geographic structure is by NHS England region (of which there are seven in England) and age groups are divided into 5-year age bands from 0–4 to 70–74 years, with an additional age group comprising individuals aged 75 years and over. The model tracks three variants of SARS-CoV-2 which enables us to capture wild-type, Alpha B.1.1.7, Delta B.1.617.2, and Omicron B.1.1.529 variants separately. The second and third modelled variants track Alpha B.1.1.7 and Delta B.1.617.2 respectively; the first modelled variant initially describes wildtype and other variants circulating prior to the emergence of Alpha B.1.1.7, before switching to describe the Omicron B.1.1.529 variant on 22nd September 2021. The date of 22nd September 2021 is chosen to be sufficiently before the emergence of Omicron in England and well after wildtype and other variants circulating prior to Alpha B.1.1.7 have been competitively excluded. On this date, we update all of the variant-specific parameters and assumptions for the first modelled variant to represent the Omicron variant and ensure that any individuals remaining in the recovered disease state for the first variant are moved into the recovered disease state for the third variant, Delta B.1.617.2. To capture the effect of the BA.2 Omicron sublineage taking over from the previously-dominant BA.1 Omicron sublineage33,36, we use sequencing data from the Wellcome Sanger Institute43 to inform the proportion of modelled Omicron infections attributable to BA.2 over time, assuming that BA.2 confers a 50% increase in transmissibility relative to BA.133,36,44–47. We assume the same levels of vaccine protection and rates of waning immunity for both Omicron BA.1 and BA.2 sublineages48.
When individuals have recovered following a SARS-CoV-2 infection with one variant, they move into a recovered disease compartment where (depending on parameter assumptions, see Table S3) we allow for re-infections and cross-infections with other variants to occur. We model COVID-19 vaccination with separate compartments for two vaccine products—for each of the viral vector and mRNA-based COVID-19 vaccines in use in England—and with each vaccine product having three compartments for three levels of protection (one dose, two doses, and waned from two doses). Initially, vaccinated individuals move from the susceptible compartment to a first-dose vaccinated compartment (Va1/Vb1 in Fig. 1) 28-days following receipt of their vaccination.
Individuals remain in the first-dose vaccinated states for an assumed duration (see dVa1/dVb1 in Table S5B), before transitioning into the second-dose vaccinated compartments (Va2/Vb2 in Fig. 1). The first-dose duration assumptions are based on measured delays between first and second doses in UKHSA vaccination data, separated into two periods (before and after the JCVI issued guidance on widening the dosing gap from 3 weeks to a maximum of 12 weeks), see Table S5B for details. Upon leaving the first-dose vaccine state, individuals transition into the second-dose vaccine state with increased levels of protection (Table S2). Another assumed distribution governs the duration that individuals remain in the second-dose state (see dVa2/dVb2 in Table S5B). The distribution governing the duration that individuals have second-dose levels of vaccine protection is chosen to match the time between the start of the COVID-19 vaccination rollout (8th December 2020) and the start of the COVID-19 booster dose rollout (September 2021) less the assumed average duration of first-dose protection.
When individuals reach the end of their second-dose duration, they either receive a booster vaccination (using an age-specific probability of receiving a booster vaccination, see Table S5B) and return to the start of the mRNA second-dose vaccine compartment (Vb2 in Fig. 1), or do not receive a booster and transition into vaccine product-specific vaccinated and waned compartments with lower levels of protection (Va2w/Vb2w in Fig. 1). We assume that all boosted individuals move into the mRNA two-dose vaccinated (Vb2 in Fig. 1) compartment to reflect the fact that all booster vaccinations in England are either the Pfizer/BioNTech or Moderna mRNA vaccines, regardless of which vaccine product was received for the primary COVID-19 vaccination course, and evidence finding higher immunogenicity for individuals receiving Pfizer/BioNTech following Oxford-AstraZeneca, compared with individuals receiving both Oxford-AstraZeneca vaccine doses49. In addition to individuals who receive a booster vaccination remaining in the second-dose mRNA vaccine compartment, for first booster vaccinations administered in late 2021/early 2022, we increase the level of protection within the two doses compartment to account for an additional booster effect (see Booster vaccinations). The model also captures eventual full waning of immunity that has been derived from a previous infection and/or vaccination. A full description of assumptions related to vaccine protection against different outcomes, cross protection and waning immunity is provided in the Supplementary Information in Tables S2–S4 and Tables S6, S7.
Hospital admissions and occupancy data were provided by NHS England and deaths, immunisations and variant data were provided by the UK Health Security Agency (UKHSA). These data sources are unpublished and not public, but are closely aligned with healthcare, deaths, and vaccinations data on the UK Government COVID-19 Dashboard2. Seroprevalence data were obtained from the UK Biobank50 and the REACT-2 study51, and seroprevalence and PCR positivity data were obtained from the Office for National Statistics COVID-19 Infection Survey (ONS-CIS)52,53.
The age-specific susceptibility to infection and age-specific probability of clinical symptoms for SARS-CoV-2 are adopted from a study using data from 6 countries26. This study found that susceptibility to SARS-CoV-2 infection for 0–19-year-olds was roughly half that for >20-year-olds, and that the probability of clinical symptoms also increased with age26. The age-specific probability of hospital admission, ICU admission, and death given infection are fitted to data from England, with the relative rates by age group based on data collected by a large meta-analysis of the COVID-19 infection fatality rate54 and based on data collected by ISARIC (the CO-CIN study) for England23, then adjusted over time to better match observed hospitalisations and deaths in England (see Model fitting). Each of these age-specific probabilities of severe outcomes is allowed to vary over the course of the epidemic in England and vary between pre-existing variants and Alpha B.1.1.7. For the third variant Delta B.1.617.2, we assume that the probability of severe outcomes is twice that of Alpha B.1.1.7, in line with estimates from Public Health Scotland and UKHSA28. For the fourth variant Omicron B.1.1.529 (and both modelled sublineages BA.1 and BA.2), we assume that the probability of severe outcomes is half that of Delta B.1.617.2, in line with estimates from the UK33,55,56. The model fitted adjustments to the infection fatality, hospital admission, hospital bed occupancy and ICU bed occupancy rates over time is shown in Fig. S1C. A full description of fitted and assumed parameters is provided in Tables S5A, S5B.
The model uses age-specific contact rates as measured by the POLYMOD study57 in the UK as a baseline contact matrix representing pre-pandemic age-specific mixing rates. The POLYMOD study estimates age-specific mixing rates for contacts made at home, work, school, and other settings, such that the baseline contact rate between age group i and age group j is , where and are coefficients on the four component matrices. For the pre-pandemic baseline matrix, then, , but these coefficients are allowed to vary over time according to mobility data, school schedules and school attendance data as follows. The model uses Google Community Mobility data22 to capture mobility in various settings: workplaces, retail & recreation venues, transit stations, and grocery & pharmacy locations. In turn, the relationship between mobility data and social contact rates23 is derived from the historical relationship between Google Community Mobility indices and social contact rates as measured by the CoMix study in 2020. This relationship furnishes how and vary over time as a function of mobility patterns.
School openings and closings are accounted for in contacts among school-aged children, university-aged young adults, and school/university staff. We assume that schools in England follow their traditional schedules (i.e., are closed during holiday periods), and we combine these assumptions with school attendance data in England published on the 4th of May 202224. To reflect the introduction of mass testing within educational facilities in the Spring of 2021, we have assumed an additional 30% reduction in transmission related to educational settings between the reopening of schools on 8th March 2021 and school closures in July 2021. This reduction in transmission is reflected in the model with a 30% reduction in school-related contacts. Seasonality is modelled as a sinusoidally-varying multiplier on transmission with the peak occurring on January 1st and the trough on July 1st of each year. By default, we assume the amplitude of the seasonal component is 20% from trough to peak and is introduced from 1st April 2021.
Model fitting
The model is fitted using a two-stage process. In the first stage, the model parameters are fitted by Bayesian inference using Markov chain Monte Carlo (MCMC) to reported regional data on hospital admissions, hospital and ICU bed occupancy, seroprevalence, PCR positivity, and deaths within 28 days of a patient’s first positive SARS-CoV-2 test, as well as to data tracking the emergence and spread of the Alpha B.1.1.7 and Omicron B.1.1.529 variants (using the frequency of S-gene target failure in PCR tests) in late 2020 and late 2021 respectively, and of the Delta B.1.617.2 variant (using the frequency of Delta in genomic sequencing data) in 2021. We use the DE-MCMC algorithm58 implemented in C++ (see analysis code).
The introduction time and relative transmissibility of both the Alpha, Delta, and Omicron variants are fitted for each geographic region in the model. We use data recording the number of first COVID-19 vaccine doses delivered by age, geography, and vaccine product from 8th December 2020 to 2nd May 2022 to inform the fraction of first-dose vaccinated individuals in each age group, NHS England region and by vaccine type over time. Additionally, in this initial stage, 18 additional parameters are fitted to define a transmission multiplier function, with each of the 18 parameters defining a stepwise change in transmission occurring at fixed six-week intervals beginning 12th April 2020 and ending 8th May 2022. This transmission multiplier function allows the modelled epidemic trajectory to better capture changes in SARS-CoV-2 transmission over time and reflects residual changes in transmission that are not captured by mobility data alone, e.g., as resulting from changes in personal protective behaviours such as mask-wearing or from changes in social behaviour such as during holidays.
In the second stage of model fitting, a particle filtering algorithm59 is used to refine the rough transmission multiplier function, while holding other fitted model parameters constant, to achieve a more fine-scaled function with stepwise changes to the transmission multiplier every 5 days, instead of the cruder 6-week increment used in the initial stage of fitting. In the second stage, the transmission multiplier is fitted as a random walk on a logarithmic scale, that is, multiplicative increments to the transmission multiplier are proposed rather than absolute levels.
The same likelihood is used for Bayesian inference in both stages of the model fitting process. In particular, the likelihood allows the infection fatality rate (IFR), infection hospitalisation rate (IHR), and infection critical-illness rate (ICR) to vary over the course of the epidemic, to reflect changes in treatment success, admissions criteria, and availability of hospital resources over time. To achieve this, the model’s initial output of deaths and hospital burdens, which are based upon a fixed IFR, IHR, and ICR (except as modified by variant-specific characteristics and by vaccine protection), are treated as the prior expectation for deaths and hospital burdens on each day. This expectation is used as the mean of a gamma distribution, with standard deviation set to 0.3 times the mean, which, in turn, is taken as the prior distribution for the mean of a Poisson distribution from which the observed burden for a given day is assumed to be drawn. This process allows the IFR/IHR/ICR to change over time, while not straying too far from the expected burden based upon the underlying fixed IFR/IHR/ICR.
Model assumptions
Vaccine effectiveness
We model vaccine protection against five separate outcomes for each SARS-CoV-2 variant: infection, disease (i.e., symptomatic infection), hospitalisation, mortality and onward transmission following a breakthrough infection (i.e., when an individual who has vaccine protection becomes infected). We assume the same vaccine effectiveness for the first two SARS-CoV-2 variants considered in the model (pre-Alpha B.1.1.7 and Alpha B.1.1.7), and separate specific vaccine effectiveness estimates for the Delta B.1.617.2 and Omicron B.1.1.529 variants, shown in Table S2. Throughout, we assume identical vaccine effectiveness across all age groups in the model. These may be subject to change in future work, as new evidence emerges. We treat individuals who have been and will be vaccinated with Moderna vaccines the same as individuals receiving Pfizer vaccines. We model individuals who have received different vaccine products (e.g., viral vector AstraZeneca (Va compartments in Fig. 1) and mRNA Pfizer/Moderna (Vb compartments in Fig. 1)) and one or two vaccine doses separately, assuming separate efficacy estimates for each category (Table S2). We additionally consider individuals who have received two vaccine doses but no booster dose as having reduced levels of protection in the vaccinated and waned state (Fig. 1, Table S2).
We base our vaccine effectiveness assumptions for the pre-Alpha, Alpha, and Delta variants on a number of studies considering vaccine efficacy and effectiveness (Tables S2, S6, S7). For the Omicron B.1.1.529 variant (comprising both BA.1 and BA.2 sublineages), we assume a 5.5-fold reduction in neutralisation between Delta and Omicron41, resulting in approximately 45% lower vaccine protection against infection. We use the resulting vaccine protection against infection with Omicron to generate assumptions for vaccine protection against Omicron disease, hospitalisation, and mortality. For protection against disease, we assume the same conditional protection against disease given infection as for the Delta B.1.617.2 variant. For protection against hospitalisation and mortality, we use Khoury et al.’s modelled relationship between efficacy against any infection and efficacy against severe infection60 to scale up our assumptions for protection against infection with Omicron to higher levels of protection against these two severe outcomes. For protection against onward transmission, we use the same assumptions as for the Delta B.1.617.2 variant (Table S2).
Our assumptions about the levels of vaccine protection for individuals who have received two vaccine doses but no booster dose, and their protection has waned, are shown in Table S2. We have based our assumptions for vaccine protection against infection, disease and onward transmission on scaling down assumed levels of protection for dose-two vaccinated individuals using vaccine product-specific measured percentage reductions in vaccine protection over time from Andrews et al19. For protection against infection and disease, we referred to the measured product-specific percentage reductions in protection against symptomatic infection for individuals aged 16 years and older after 20+ weeks, compared to measured protection in week 1. For reductions in protection against onward transmission, we calculate the average product-specific measured percentage reduction in protection across the measured percentage changes in protection against symptomatic infection and hospitalisation measured at 20+ weeks compared to week 1 and the percentage change in protection against death measured at 20+ weeks compared to weeks 2–9. Since we model an individual’s average duration in the second dose compartment as 29.3 weeks (Table S5B), before they are either boosted and remain in the two-dose vaccinated compartment or are not boosted and wane, we have chosen to further reduce our scaled vaccine effectiveness estimates against infection and onward transmission resulting from using Andrews et al19. 20+ week estimates by an additional 25% reduction on top of the measured percentage reductions (Table S2). To balance this assumption, we have not included an additional 25% reduction in protection for the assumed level of vaccine protection against disease for vaccinated and waned individuals (Table S2). To arrive at assumptions for vaccine protection against severe outcomes (hospitalisation and mortality) for individuals in the vaccinated and waned state, we use Khoury et al.’s modelled relationship between efficacy against any infection and efficacy against severe infection60 to increase our assumed levels of protection against infection up to higher levels (Table S2).
Booster vaccinations
We model the effect of booster vaccinations by boosting individuals in two categories: those who remain in the second-dose vaccinated compartment following receiving a booster as well as individuals who are in the recovered disease state but would also have received a primary course of COVID-19 vaccination followed by a booster vaccination. Boosted individuals remain in their current disease state, but we model an additional increase in their level of vaccine protection and cross protection against reinfection following their first booster vaccine in late 2021/early 2022 as follows. We firstly assume that the Pfizer-BioNTech and Moderna booster vaccinations being administered in England result in a 2.5-fold increase in neutralisation titres61, and we use Khoury et al.’s modelled relationship between neutralisation titres and protective efficacy60 to increase our two-dose vaccine effectiveness and cross protection assumptions against infection to boosted levels (boosted vaccine effectiveness assumptions are shown in Table S2). For the boosted levels of cross-protection against infection in the recovered disease state, we use the same increase in protection, but this is applied only to the proportion of recovered individuals who would have received a primary vaccination course multiplied by the age-specific probability of receiving a booster vaccine (see Table S5B). We scale up the boosted protection against infection to equivalent levels of protection against hospitalisation and mortality using Khoury et al.’s modelled relationship between efficacy against any infection and efficacy against severe infection60.
We refer to data on the number of COVID-19 booster vaccinations being delivered over time on the UK Government’s COVID-19 dashboard2 to inform the time at which the additional booster effect is applied to each 5-year age group, from individuals aged 75 years and above to individuals aged 15 years and above, in sequentially younger age groups over time. Between the start of the COVID-19 booster vaccination rollout (September 2021) and the 15th of December 2021, we assume a daily supply of booster vaccines of 229,000. We assume that booster supply increases to 1 million doses per day from the 15th of December 2021, following the JCVI announcement of an acceleration and widening of the booster vaccination programme14. Using these daily supply levels allows us to approximate the effects of the booster vaccination campaign by introducing the assumed booster effect on vaccine protection and cross protection at specific times for each 5-year age group in the model, from the oldest to the youngest eligible group. We assume that this additional booster effect lasts for 180 days, before vaccine protection returns to assumed two-dose levels (Table S2) and cross protection returns to previous levels (Table S3).
Waning immunity
We model waning immune protection against SARS-CoV-2 developed from a prior SARS-CoV-2 infection and/or vaccination. For waning immune protection following a previous infection, we assume identical rates of waning for all virus variants and for all age groups (Table S4). Once individuals who have recovered from a prior infection wane, they return to a susceptible disease state; thus, we model waning of so-called natural immunity against different endpoints (infection, disease, hospitalisation, deaths, and onward transmission) at the same rate. For our central waning assumptions, we do not allow individuals in the two-dose vaccinated compartments to wane directly back to being susceptible. To account for booster vaccinations, upon reaching the end of the assumed duration within the second dose state (Table S5B), individuals either receive a booster vaccine and return to the start of the second-dose compartment (with additional levels of protection afforded for the first booster vaccination in late 2021/early 2022, given an assumed booster duration of 180 days, see description above), or move into a third state with reduced levels of vaccine protection across different outcomes (see Table S2). This third state corresponds to individuals who have received two vaccine doses and no booster dose, leading to waning of their vaccine protection (see Vaccine effectiveness above). Once individuals have moved into this waned state with reduced levels of vaccine protection, they are also allowed to wane back to being susceptible, with different rates considered for each vaccine product (Table S4). The assumed percentage loss in reduction for the central waning scenario is based on measured percentage changes in vaccine protection against hospitalisation after 20+ weeks for each vaccine product in Andrews et al.19.
Mobility and vaccination schedules
To produce forward projections, the model requires information about future contact rates and vaccination rates. We base our assumptions on how social contact rates might be expected to change by referring to historical mobility data22 and making assumptions about future mobility until December 2022 (Fig. 3). Mobility levels have generally been gradually increasing since early 2020 but have still not returned to pre-pandemic levels. Hence, we consider four scenarios: we project current levels of mobility forwards (i.e., no change), and we consider a return to pre-pandemic baseline levels of mobility within periods of 3 weeks, 3 months, and 6 months. These assumed future changes in mobility are combined with future school term schedules to generate an overall schedule for future contact rates (Fig. 3).
The time-varying transmission multiplier also needs to be projected forwards for model projections. This is done by using the forecast R package62 to fit a second-order moving-average model to the fitted transmission multiplier function, then using random forecasts of the resulting moving-average model for each new projection. This process of using different realisations of the projected transmission multiplier is what produces most of the variability among different runs of each scenario (Fig. 4c). For forward projections, we combine the assumed future contact rates schedule with the projected transmission multiplier (Fig. 3) to produce an overall time-varying transmission potential until the end of the simulation period.
For future first-dose vaccinations, we generate vaccine schedules according to assumed future vaccine supply (i.e., number of doses available per week) and uptake limits per 5-year age group, in combination with the historic first-dose vaccine schedules generated using UKHSA data on vaccines delivered up to the 2nd of May 2022. Future first-dose vaccine supply is assumed to be 150,000 doses per week for England throughout the remainder of the projection period (i.e., until December 2022). We assume that first-dose vaccination uptake limits in individuals aged 15 years and above have already been reached, so no future first doses are delivered to these age groups. Our basecase scenario assumes that uptake is limited at 80% for individuals aged 5–14, delivering future first doses first to the 10–14 age group up to the uptake limit, followed by 5–9-year-olds. This assumption of 80% uptake for 5–14-year-olds is chosen based on the first-dose uptake measured by age across England; uptake is lower for younger age groups, with uptake reaching at least 80% as of May 2022 for the youngest eligible adults, who have been eligible to receive their first COVID-19 vaccinations in England since the summer of 2021. We also consider the impact of an alternative vaccination schedule generated assuming lower uptake levels for children aged 5–14 years (Fig. S7).
The assumed future supply of first doses per week is firstly divided into daily supply levels. Supply levels for each day are distributed into the seven NHS England regions according to the population size of each region. The allocated number of future first doses per day in each region are initially allocated to the oldest age groups which have not yet reached their age-specific uptake limits (i.e., since we assume maximum uptake has been reached for individuals aged 15 and above, 10–14-year-olds are prioritised first up to their 80% uptake limit, followed by 5–9-year-olds up to their uptake limit). The allocated number of first doses per day, per region and per age group are divided into specified proportions of vaccine products relevant to each age group (75% Pfizer and 25% Moderna for <40 year olds, and 60% AstraZeneca, 30% Pfizer and 10% Moderna for 40+ year olds; since May 2021 the JCVI has advised a preference for individuals under 40 years of age to receive an alternative to the viral-vector Oxford-AstraZeneca vaccine63). Any doses remaining after this process are carried over to the next age group down (up to the relevant uptake limit and reallocated according to our assumptions on vaccine product mix), the next NHS England region, or the next day, or are left unallocated in the schedule and recorded as leftover doses.
Ethics
Ethical approval for this research was given by the London School of Hygiene & Tropical Medicine Ethics Committee, project ID: 22828.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Supplementary information
Acknowledgements
The following funding sources are acknowledged as providing funding for the named authors. This project has received funding from the European Union’s Horizon 2020 research and innovation programme - project EpiPose (Grant agreement number 101003688: R.C.B., M.J., W.J.E.). This work reflects only the authors’ view. The European Commission is not responsible for any use that may be made of the information it contains. This project has also received funding from the UK Medical Research Council (MC_PC_19065: N.G.D., W.J.E.), was partly funded by the National Institute for Health and Care Research (NIHR) (Health Protection Research Unit in Immunisation NIHR200929: N.G.D., M.J.; Health Protection Research Unit in Modelling and Health Economics NIHR200908: M.J., W.J.E.; PR-OD-1017-20002: W.J.E.). Funding statements for CMMID COVID-19 working group members are listed in the Supplementary Information. The authors would like to thank Stephen J. Rivers, Lloyd A. C. Chapman and Ciara V. McCarthy for helpful discussions which have contributed towards completion of this work.
Author contributions
R.C.B. and N.G.D. accessed and verified the data and conducted the analyses. R.C.B., N.G.D., M.J., and W.J.E. contributed to study design and drafting of the manuscript. Centre for Mathematical Modelling of Infectious Diseases COVID-19 working group members contributed in processing, cleaning and interpretation of data, interpreted findings, contributed to the manuscript, and approved the work for publication.
Peer review
Peer review information
Nature Communications thanks Jodie McVernon and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Data availability
Data sources used for these analyses are either publicly available or were provided to the named authors as members of the UK’s Scientific Pandemic Influenza Modelling (SPI-M) group, which provides expert advice to the UK’s Department of Health and Social Care and wider UK government on scientific matters. Specific permissions have been sought to use SPI-M data for this publication. The SPI-M datasets used for model fitting are unpublished and not publicly available, but are closely aligned with the UK Government’s COVID-19 dashboard2 and other publicly available sources such as the Wellcome Sanger Institute’s COVID-19 genomic surveillance data64. The following public datasets have been used in this work: school attendance data65, PCR prevalence data53, seroprevalence data50–52, mobility data22, COVID-19 vaccination statistics25, and data on SARS-CoV-2 lineages in England43.
Code availability
Analysis and code21 is available at: https://github.com/rosannaclairebarnard/covidm-mtd-Omi
Competing interests
The authors declare the following competing interests: R.C.B., N.G.D., M.J. and W.J.E. are participants of the UK’s Scientific Pandemic Influenza Group on Modelling. W.J.E. attends the UK’s Scientific Advisory Group for Emergencies.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A list of authors and their affiliations appears at the end of the paper.
Contributor Information
Rosanna C. Barnard, Email: rosanna.barnard@lshtm.ac.uk
Centre for Mathematical Modelling of Infectious Diseases COVID-19 working group:
James D. Munday, Rachel Lowe, Gwenan M. Knight, Quentin J. Leclerc, Damien C. Tully, David Hodgson, Rachael Pung, Joel Hellewell, Mihaly Koltai, David Simons, Kaja Abbas, Adam J. Kucharski, Simon R. Procter, Frank G. Sandmann, Carl A. B. Pearson, Kiesha Prem, Alicia Showering, Sophie R. Meakin, Kathleen O’Reilly, Ciara V. McCarthy, Matthew Quaife, Kerry L. M. Wong, Yalda Jafari, Arminder K. Deol, Rein M. G. J. Houben, Charlie Diamond, Thibaut Jombart, C. Julian Villabona-Arenas, William Waites, Rosalind M. Eggo, Akira Endo, Hamish P. Gibbs, Petra Klepac, Jack Williams, Billy J. Quilty, Oliver Brady, Jon C. Emery, Katherine E. Atkins, Lloyd A. C. Chapman, Katharine Sherratt, Sam Abbott, Nikos I. Bosse, Paul Mee, Sebastian Funk, Jiayao Lei, Yang Liu, Stefan Flasche, James W. Rudge, Fiona Yueqian Sun, Graham Medley, Timothy W. Russell, Amy Gimma, Stéphane Hué, Christopher I. Jarvis, Emilie Finch, and Samuel Clifford
Supplementary information
The online version contains supplementary material available at 10.1038/s41467-022-32404-y.
References
- 1.COVID-19 Data Explorer. Our World in Datahttps://ourworldindata.org/coronavirus-data-explorer.
- 2.UK Summary. https://coronavirus.data.gov.uk.
- 3.Tracking SARS-CoV-2 variants. https://www.who.int/activities/tracking-SARS-CoV-2-variants.
- 4.Website. https://www.instituteforgovernment.org.uk/charts/uk-government-coronavirus-lockdowns.
- 5.Prime Minister’s Office,. PM statement on living with COVID: 21 February 2022. GOV.UKhttps://www.gov.uk/government/speeches/pm-statement-on-living-with-covid-21-february-2022 (2022).
- 6.Medicines and Healthcare products Regulatory Agency. Valneva COVID-19 vaccine approved by MHRA. GOV.UKhttps://www.gov.uk/government/news/valneva-covid-19-vaccine-approved-by-mhra (2022).
- 7.Joint Committee on Vaccination and Immunisation: advice on priority groups for COVID-19 vaccination, 30 December 2020. https://www.gov.uk/government/publications/priority-groups-for-coronavirus-covid-19-vaccination-advice-from-the-jcvi-30-december-2020/joint-committee-on-vaccination-and-immunisation-advice-on-priority-groups-for-covid-19-vaccination-30-december-2020.
- 8.Department of Health and Social Care. All young people aged 16 and 17 in England to be offered vaccine by next week. GOV.UKhttps://www.gov.uk/government/news/all-young-people-aged-16-and-17-in-england-to-be-offered-vaccine-by-next-week (2021).
- 9.England, N. H. S. NHS England » NHS rolls out COVID vaccine to five million 5 to 11 year olds. https://www.england.nhs.uk/2022/04/nhs-rolls-out-covid-vaccine-to-five-million-5-to-11-year-olds/.
- 10.UK Health Security Agency. JCVI issues advice on COVID-19 booster vaccines for those aged 40 to 49 and second doses for 16 to 17 year olds. GOV.UKhttps://www.gov.uk/government/news/jcvi-issues-advice-on-covid-19-booster-vaccines-for-those-aged-40-to-49-and-second-doses-for-16-to-17-year-olds (2021).
- 11.Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern.
- 12.Department of Health and Social Care. First UK cases of Omicron variant identified. GOV.UKhttps://www.gov.uk/government/news/first-uk-cases-of-omicron-variant-identified (2021).
- 13.Andrews, N. et al. Covid-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 386, (2022). [DOI] [PMC free article] [PubMed]
- 14.Department of Health and Social Care. JCVI advice on the UK vaccine response to the Omicron variant. GOV.UKhttps://www.gov.uk/government/publications/uk-vaccine-response-to-the-omicron-variant-jcvi-advice/jcvi-advice-on-the-uk-vaccine-response-to-the-omicron-variant (2021).
- 15.JCVI statement on the adult COVID-19 booster vaccination programme and the Omicron variant: 7 January 2022. GOV.UKhttps://www.gov.uk/government/publications/jcvi-statement-on-the-adult-covid-19-booster-vaccination-programme-and-the-omicron-variant/jcvi-statement-on-the-adult-covid-19-booster-vaccination-programme-and-the-omicron-variant-7-january-2022.
- 16.JCVI statement on COVID-19 vaccination of children and young people: 22 December 2021. GOV.UKhttps://www.gov.uk/government/publications/jcvi-update-on-advice-for-covid-19-vaccination-of-children-and-young-people/jcvi-statement-on-covid-19-vaccination-of-children-and-young-people-22-december-2021.
- 17.England, N. H. S. NHS England » NHS expands COVID vaccinations to the most vulnerable 5 to 11 year olds. https://www.england.nhs.uk/2022/01/nhs-expands-covid-vaccinations-to-the-most-vulnerable-5-to-11-year-olds/.
- 18.JCVI statement on vaccination of children aged 5 to 11 years old. GOV.UKhttps://www.gov.uk/government/publications/jcvi-update-on-advice-for-covid-19-vaccination-of-children-aged-5-to-11/jcvi-statement-on-vaccination-of-children-aged-5-to-11-years-old.
- 19.Andrews, N. et al. Duration of Protection against Mild and Severe Disease by Covid-19 Vaccines. N. Engl. J. Med. 386, 340–350 (2022). [DOI] [PMC free article] [PubMed]
- 20.UK Health Security Agency. COVID-19 vaccine weekly surveillance reports (weeks 39 to 42). https://www.gov.uk/government/publications/covid-19-vaccine-weekly-surveillance-reports (2021).
- 21.Barnard, R. rosannaclairebarnard/covidm-mtd-Omi: First release associated with preprint. 10.5281/zenodo.6806637. https://zenodo.org/record/6806637 (2022).
- 22.Google. COVID-19 Community Mobility Reports. https://www.google.com/covid19/mobility/.
- 23.Davies NG, et al. Association of tiered restrictions and a second lockdown with COVID-19 deaths and hospital admissions in England: a modelling study. Lancet Infect. Dis. 2021;21:482–492. doi: 10.1016/S1473-3099(20)30984-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Attendance in education and early years settings during the coronavirus (COVID-19) pandemic. https://explore-education-statistics.service.gov.uk/find-statistics/attendance-in-education-and-early-years-settings-during-the-coronavirus-covid-19-outbreak/2022-week-16.
- 25.Statistics. Statistics » COVID-19 Vaccinations. https://www.england.nhs.uk/statistics/statistical-work-areas/covid-19-vaccinations/.
- 26.Davies NG, et al. Age-dependent effects in the transmission and control of COVID-19 epidemics. Nat. Med. 2020;26:1205–1211. doi: 10.1038/s41591-020-0962-9. [DOI] [PubMed] [Google Scholar]
- 27.Davies NG, et al. Increased mortality in community-tested cases of SARS-CoV-2 lineage B.1.1.7. Nature. 2021;593:270–274. doi: 10.1038/s41586-021-03426-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Public Health England. Investigation of SARS-CoV-2 variants of concern: technical briefings. https://www.gov.uk/government/publications/investigation-of-novel-sars-cov-2-variant-variant-of-concern-20201201 (2020).
- 29.Nyberg, T. et al. Comparative analysis of the risks of hospitalisation and death associated with SARS-CoV-2 omicron (B.1.1.529) and delta (B.1.617.2) variants in England: a cohort study. Lancet399, 1303–1312 (2022). [DOI] [PMC free article] [PubMed]
- 30.Sigal A, Milo R, Jassat W. Estimating disease severity of Omicron and Delta SARS-CoV-2 infections. Nat. Rev. Immunol. 2022;22:267–269. doi: 10.1038/s41577-022-00720-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Teslya A, et al. Impact of self-imposed prevention measures and short-term government-imposed social distancing on mitigating and delaying a COVID-19 epidemic: A modelling study. PLoS Med. 2020;17:e1003166. doi: 10.1371/journal.pmed.1003166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Weitz JS, Park SW, Eksin C, Dushoff J. Awareness-driven behavior changes can shift the shape of epidemics away from peaks and toward plateaus, shoulders, and oscillations. Proc. Natl Acad. Sci. U. S. A. 2020;117:32764–32771. doi: 10.1073/pnas.2009911117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.UK Health Security Agency. Investigation of SARS-CoV-2 variants: technical briefings. GOV.UKhttps://www.gov.uk/government/publications/investigation-of-sars-cov-2-variants-technical-briefings (2021).
- 34.Bonnie Lewis, Ed Pyle, Matt Dennes, Tim Vizard. Coronavirus and the social impacts on Great Britain - Office for National Statistics. https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/healthandwellbeing/bulletins/coronavirusandthesocialimpactsongreatbritain/1april2022 (2022).
- 35.UK Health Security Agency. JCVI advises a spring COVID-19 vaccine dose for the most vulnerable. GOV.UKhttps://www.gov.uk/government/news/jcvi-advises-a-spring-covid-19-vaccine-dose-for-the-most-vulnerable (2022).
- 36.Lyngse, F. P. et al. Transmission of SARS-CoV-2 Omicron VOC subvariants BA.1 and BA.2: Evidence from Danish Households. medRxiv 2022.01.28.22270044 (2022).
- 37.Stegger, M. et al. Occurrence and significance of Omicron BA.1 infection followed by BA.2 reinfection. medRxiv 2022.02.19.22271112 (2022).
- 38.Davies, N. G. et al. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science372, eabg3055 (2021). [DOI] [PMC free article] [PubMed]
- 39.Scientific Advisory Group for Emergencies. LSHTM: Updated roadmap assessment – prior to delayed Step 4, 7 July 2021. https://www.gov.uk/government/publications/lshtm-updated-roadmap-assessment-prior-to-delayed-step-4-7-july-2021 (2021).
- 40.Scientific Advisory Group for Emergencies. LSHTM: Interim roadmap assessment – prior to steps 3 and 4, 5 May 2021. GOV.UKhttps://www.gov.uk/government/publications/lshtm-interim-roadmap-assessment-prior-to-steps-3-and-4-5-may-2021 (2021).
- 41.Barnard, R. C., Davies, N. G., Pearson, C. A. B., Jit, M. & John Edmunds, W. Projected epidemiological consequences of the Omicron SARS-CoV-2 variant in England, December 2021 to April 2022. medRxiv 2021.12.15.21267858 (2021).
- 42.Scientific Advisory Group for Emergencies. LSHTM: autumn and winter scenarios 2021 to 2022, 13 October 2021. GOV.UKhttps://www.gov.uk/government/publications/lshtm-autumn-and-winter-scenarios-2021-to-2022-13-october-2021 (2021).
- 43.COVID-19 Genomic Surveillance – Wellcome Sanger Institute. https://covid19.sanger.ac.uk/lineages/raw.
- 44.Ito, K., Piantham, C. & Nishiura, H. Estimating relative generation times and relative reproduction numbers of Omicron BA.1 and BA.2 with respect to Delta in Denmark. medRxiv 2022.03.02.22271767 (2022). [DOI] [PubMed]
- 45.Yamasoba, D. et al. Virological characteristics of SARS-CoV-2 BA.2 variant. bioRxiv 2022.02.14.480335 10.1101/2022.02.14.480335 (2022).
- 46.Lentini, A., Pereira, A., Winqvist, O. & Reinius, B. Monitoring of the SARS-CoV-2 Omicron BA.1/BA.2 variant transition in the Swedish population reveals higher viral quantity in BA.2 cases. medRxiv 2022.03.26.22272984 (2022). [DOI] [PMC free article] [PubMed]
- 47.Statement on Omicron sublineage BA.2. https://www.who.int/news/item/22-02-2022-statement-on-omicron-sublineage-ba.2.
- 48.Kirsebom, F. C. M. et al. COVID-19 Vaccine Effectiveness against the Omicron BA.2 variant in England. medRxiv 2022.03.22.22272691 (2022). [DOI] [PMC free article] [PubMed]
- 49.Liu X, et al. Safety and immunogenicity of heterologous versus homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine (Com-COV): a single-blind, randomised, non-inferiority trial. Lancet. 2021;398:856–869. doi: 10.1016/S0140-6736(21)01694-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.COVID-19 hub. https://www.ukbiobank.ac.uk/learn-more-about-uk-biobank/covid-19-hub.
- 51.Real-time Assessment of Community Transmission findings. https://www.imperial.ac.uk/medicine/research-and-impact/groups/react-study/real-time-assessment-of-community-transmission-findings/.
- 52.Hill, R. Y. A. Coronavirus (COVID-19) Infection Survey, antibody and vaccination data, UK. https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/datasets/coronaviruscovid19antibodydatafortheuk (2021).
- 53.Yapp, R. & Willis, Z. Coronavirus (COVID-19) Infection Survey: England. https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/datasets/coronaviruscovid19infectionsurveydata (2022).
- 54.Levin, A. T. et al. Assessing the Age Specificity of Infection Fatality Rates for COVID-19: Systematic Review, Meta-Analysis, and Public Policy Implications. medRxiv 2020.07.23.20160895 (2020). [DOI] [PMC free article] [PubMed]
- 55.Scientific Advisory Group for Emergencies. Imperial College London: Omicron severity and vaccine effectiveness, 5 January 2022. GOV.UKhttps://www.gov.uk/government/publications/imperial-college-london-omicron-severity-and-vaccine-effectiveness-5-january-2022 (2022).
- 56.Report 50 - Hospitalisation risk for Omicron cases in England. Imperial College Londonhttp://www.imperial.ac.uk/medicine/departments/school-public-health/infectious-disease-epidemiology/mrc-global-infectious-disease-analysis/covid-19/report-50-severity-omicron/.
- 57.Mossong J, et al. Social contacts and mixing patterns relevant to the spread of infectious diseases. PLoS Med. 2008;5:e74. doi: 10.1371/journal.pmed.0050074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Braak CJFT. A Markov Chain Monte Carlo version of the genetic algorithm Differential Evolution: easy Bayesian computing for real parameter spaces. Stat. Comput. 2006;16:239–249. doi: 10.1007/s11222-006-8769-1. [DOI] [Google Scholar]
- 59.Endo A, van Leeuwen E, Baguelin M. Introduction to particle Markov-chain Monte Carlo for disease dynamics modellers. Epidemics. 2019;29:100363. doi: 10.1016/j.epidem.2019.100363. [DOI] [PubMed] [Google Scholar]
- 60.Khoury DS, et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021;27:1205–1211. doi: 10.1038/s41591-021-01377-8. [DOI] [PubMed] [Google Scholar]
- 61.Cromer, D. et al. Neutralising antibody titres as predictors of protection against SARS-CoV-2 variants and the impact of boosting: a meta-analysis. Lancet. Microbe3, e52–e61 (2022). [DOI] [PMC free article] [PubMed]
- 62.Hyndman RJ, Khandakar Y. Automatic Time Series Forecasting: The forecast Package for R. J. Stat. Softw. 2008;27:1–22. doi: 10.18637/jss.v027.i03. [DOI] [Google Scholar]
- 63.Public Health England. JCVI advises on COVID-19 vaccine for people aged under 40. GOV.UKhttps://www.gov.uk/government/news/jcvi-advises-on-covid-19-vaccine-for-people-aged-under-40 (2021).
- 64.COVID-19 Genomic Surveillance – Wellcome Sanger Institute. https://covid19.sanger.ac.uk/downloads.
- 65.Attendance in education and early years settings during the coronavirus (COVID-19) pandemic. https://explore-education-statistics.service.gov.uk/find-statistics/attendance-in-education-and-early-years-settings-during-the-coronavirus-covid-19-outbreak.
- 66.England, N. H. S. NHS England » Regional teams. https://www.england.nhs.uk/about/regional-area-teams/.
- 67.Department of Health and Social Care. England to return to Plan A following the success of the booster programme. GOV.UKhttps://www.gov.uk/government/news/england-to-return-to-plan-a-following-the-success-of-the-booster-programme (2022).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data sources used for these analyses are either publicly available or were provided to the named authors as members of the UK’s Scientific Pandemic Influenza Modelling (SPI-M) group, which provides expert advice to the UK’s Department of Health and Social Care and wider UK government on scientific matters. Specific permissions have been sought to use SPI-M data for this publication. The SPI-M datasets used for model fitting are unpublished and not publicly available, but are closely aligned with the UK Government’s COVID-19 dashboard2 and other publicly available sources such as the Wellcome Sanger Institute’s COVID-19 genomic surveillance data64. The following public datasets have been used in this work: school attendance data65, PCR prevalence data53, seroprevalence data50–52, mobility data22, COVID-19 vaccination statistics25, and data on SARS-CoV-2 lineages in England43.
Analysis and code21 is available at: https://github.com/rosannaclairebarnard/covidm-mtd-Omi