Abstract
Introduction
Hepatic ischemia-reperfusion injury is a common pathophysiological process in liver surgery. Whether Propofol can reduce myocardial ischemia-reperfusion injury induced by hepatic ischemia-reperfusion injury in rats, together with related mechanisms, still needs further studies.
Objective
To investigate if propofol would protect the myocardial cells from apoptosis with hepatic ischemia-reperfusion injury.
Methods
Male Sprague-Dawley rats (n = 18) were randomly allocated into three groups: Sham Group (Group S, n = 6), Hepatic Ischemia-reperfusion Injury Group (Group IR, n = 6) and Propofol Group (Group P, n = 6). Group S was only subjected to laparotomy. Group IR was attained by ischemia for 30 min and reperfusion for 4 h. Group P was subjected identical insult as in Group IR with the administration of propofol started 10 min before ischemia with 120 mg.kg−1, following by continuous infusion at 20 mg.kg−1.h−1. Cell apoptosis was examined by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling assay. Endoplasmic reticulum Ca2+-ATPase2 (SERCA2) and cysteine-containing aspartic acid cleaved-caspase3 (cleaved-caspase3) were assayed by western blot and Altimeter polymerase chain reaction.
Results
Apoptosis rate was increased, with mRNA and protein of SERCA2 down-regulated and cleaved-caspase3 up-regulated in Group IR compared with Group S (p < 0.01). Apoptosis rate was decreased, with mRNA and protein of SERCA2 up-regulated and cleaved-caspase3 down-regulated in Group P compared with Group IR (p < 0.01).
Conclusions
Propofol can reduce hepatic ischemia-reperfusion injury-induced myocardial cell apoptosis, meanwhile, can up-regulate mRNA and protein of SERCA2 in rats.
Keywords: Propofol, Liver, Reperfusion injury, Myocardium, Endoplasmic reticulum Ca2+-ATPase2
Resumo
Introdução
A lesão hepática por isquemia-reperfusão é um processo fisiopatológico comum em cirurgias hepáticas. Mais estudos ainda são necessários para avaliar se o propofol pode reduzir a lesão de isquemia-reperfusão miocárdica induzida pela lesão de isquemia-reperfusão hepática em ratos, juntamente com os mecanismos que estão relacionados.
Objetivo
Investigar se propofol protege as células do miocárdio da apoptose com a lesão hepática por isquemia-reperfusão.
Métodos
Ratos machos da raça Sprague-dawley (n = 18) foram alocados aleatoriamente em três grupos: Grupo Sham (Grupo S, n = 6), Grupo Lesão Hepática por Isquemia-reperfusão (Grupo IR, n = 6) e Grupo Propofol (Grupo P, n = 6). O Grupo S foi submetido apenas à laparotomia. O grupo IR foi submetido à isquemia por 30 min e reperfusão por 4 h. O grupo P foi submetido à mesma isquemia do grupo IR, com a administração de 120 mg.kg-1 de propofol iniciada 10min antes da isquemia, seguida de infusão contínua a 20 mg.kg-1.h-1. A apoptose celular foi examinada por meio do ensaio de marcação de terminações dUTP pela deoxinucleotidil transferase. Retículo endoplasmático Ca2+-ATPase2 (SERCA2) e caspase-3 do ácido aspártico contendo cisteína (caspase-3 clivada) foram avaliadas usando o ensaio western blot e reação em cadeia da polimerase.
Resultados
A taxa de apoptose foi maior com mRNA e proteína de SERCA2 regulados para baixo e caspase-3 clivada suprarregulada no Grupo IR, em comparação com o Grupo S (p < 0,01). A taxa de apoptose foi menor com mRNA e proteína de SERCA2 suprarregulada e caspase-3 clivada subrregulada no Grupo P, em comparação com o Grupo IR (p < 0,01).
Conclusões
O propofol pode reduzir a apoptose de células miocárdicas induzida por lesão hepática por isquemia-reperfusão, entretanto, pode suprarregular o mRNA e a proteína de SERCA2 em ratos.
Palavras-chave: Propofol, Fígado, Lesão de reperfusão, Miocárdio, Retículo endoplasmático Ca2+-ATPase2
Introduction
Total hepatic ischemia/reperfusion is commonly found in the central macrosection of the liver tumor or the liver area, liver caudate lobe tumor, and liver resection and orthotopie liver transplantation. In recent years, studies reported that hepatic ischemia-reperfusion injury (HIRI, I/RI) has general reaction and can influence function of liver and generate damage for far organs.1, 2, 3, 4 The main mechanisms may be derived from increased oxygen radical and calcium overload in cells, and the former is the most important factor.5, 6, 7 How to prevent and cure post-HIRI liver injury and secondary injuries of distant organs are major issues to be solved.
Reperfusion after liver ischemia from hepatic vein to right atrium, heart is the first stop to receive blood perfusion, and the early stage may generate damage. Calcium overload, generated oxygen radical and caused cardiac cell death are main reasons to lead to myocardium I/RI.8, 9 The early stage of hepatic ischemia reperfusion injury can generate a great amount of oxygen radical,7 which can arrive in cardiac muscle tissues via blood reperfusion. Oxygen radical may activate peroxide in membrane lipid and lead to rising of the myocardial membrane permeability and direct act on endoplasmic Reticulum Ca2+-ATPase2 (SERCA2) in sarcoplasmic reticulum. The process can reduce SERCA2 function or inhibit its activity, further reduce Ca2+ ingestion and lead to calcium overload.10, 11, 12 A great amount of activated caspase3 may lead to myocardial apoptosis.13, 14
Propofol has been widely applied in current clinical anesthesia, and it is the common drug to study protection of ischemia reperfusion injury. Propofol can protect far organs in liver ischemia reperfusion in rats,15, 16 and this study aimed to estimate the effect of propofol on myocardial apoptosis and its mechanism.
Materials and methods
Animals
Male Sprague-Dawley male rats, clean grade, weighing 200–230 g, were purchased from the Experimental Animal Center (license number: SCXK–Jun 2012-0004); experiments were started after 1 week of adaptive feeding with adequate food and water. This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Shanxi Medical University.
Grouping
The rats (n = 18) were randomly divided into three groups: Group S (n = 6), Group IR (n = 6), and Group P (n = 6).
Preparation of rat HIRI model
According to previous studies,16, 17 after anesthetic induction through intraperitoneal injectiom of 25% urethane (0.4 mL.100 mg−1), the rat was supinely secured to the operating table, the abdominal fur was shaved, the animal was disinfected, and the abdominal cavity was opened layer by layer. One micro non-invasive vascular clamp was then used to clamp the liver pedicle to prepare the total hepatic ischemia model; 30 min. later, the perfusion was recovered. For rats in Group S, the abdomen was only opened to the porta hepatis, but was not ligated. Group IR was prepared as with the total HIRI model. For Group P, a loading dose of propofol at 20 mg.kg−1 was slowly injected via the tail vein 10 min. before model preparation, and then continuously infused at a speed of 20 mg.kg−1.h−1, until execution. The remaining procedures were the same as that of IR group.
Specimen collection
The rats in Groups IR and P were euthanized 4 h after reperfusion, and the rats in Group S were sacrificed 4 h after dissected the porta hepatis. The thoracic cavity was then cut open and the heart was exposed; after rinsing with saline, the left and right ventricles were cut along the interventricular septum, and the myocardial tissues of the left ventricular wall were quickly cut, and stored at −80 °C for subsequent detection of apoptosis in cardiomyocytes and examination of related proteins. The left lateral lobe tissues were then sampled for Hematoxylin-Eosin (HE) staining, and histopathological observations were performed using a light microscope (at a magnification of 200×).
Detection of apoptosis in myocardial cells by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling assay (TUNE)
An appropriate amount of myocardial tissue was used for the detection of apoptosis using the TUNEL apoptosis detection kit (Beyotime Biotechnology Co. Ltd., Shanghai, China), in accordance with the manufacturer's instructions, under a light microscope (at a magnification of 200×).
Detection of SERCA2 and cleaved-caspase3 proteins in myocardial tissues by western blot
Total tissue proteins were extracted from 100 mg of myocardium tissues; Bovine Serum Albumin (BSA) (Beyotime Biotechnology Co. Ltd., Shanghai, China) was used as the protein standard. The protein contents were then determined using the Coomassie brilliant blue method. An equal amount of protein sample (50 μg) was then separated using 10% sodium dodecyl sulfate (SDS) polyacrylamide gel electrophoresis, and transferred onto a nitrocellulose membrane using the semi-dry transfer method. After blocking with 3% BSA for 2 hours, diluted SERCA2 antibody (1:1000; Abcam Inc., USA) and cleaved-caspase3 antibody (1:1000; Abcam Inc., USA) were added to membranes and incubated overnight at 4 °C. After washing with TBST (Tris-HCl, NaCl, Tween 20) (5 min × 3 times), goat anti-rabbit IgG (H + L) secondary antibody (1:5000) was added to the membrane and incubated at 37 °C for 2 h. This was followed by TBST washing (5 min × 3 times) and development with the BeyoECL Plus Chemiluminescence Kit. The ChemiDoc™ XRS gel imaging system (Bio-Rad, USA) was used for scanning, and Quantity One analysis software was used for detection and analysis of SERCA2 and caspase3 expression.
Detection of SERCA2 and cleaved-caspase3 mRNA in myocardial tissues by real time polymerase chain reaction (PCR)
Total RNA was extracted in accordance with the instructions of the trizol kit (Invitrogen). RNA was then reverse transcribed into cDNA using the RT-PCR kit and one-step method for further PCR amplification; the PCR amplification products were then analyzed by gel electrophoresis. The reverse transcription system was utilized with 2 μL of the total RNA template (1 μg), 2 μL of dNTP mixture, 2 μL of MgCl2, plus EDPC and distilled water to 25 μL. Reaction parameters were as follows: predenaturation at 95 °C for 5 min, denaturation at 95 °C for 30s, annealing at 58 °C for 30s, and extension 72 °C for 30s, for 40 cycles. The primers used are shown in Table 1 (Sangon Biotech (Shanghai) Co., Ltd., China).
Table 1.
RT-PCR primers of SERCA, caspase3 and GADPH.
| Gene | Primer (5′–3′) |
|---|---|
| SERCA | For: GAGATCAGCTAGGTCAGCG |
| Rev: GCATTGGTTACGCTGCTAG | |
| Caspase3 | For: GGCATGGAGAACACTGAAAC |
| Rev: GCGAATCTGTTTCTTTGCATG | |
| GADPH | For: AGCCACATCGCTCAGACA |
| Rev: TGGACTCCACGACGTACT |
SERCA mean endoplasmic reticulum Ca2+-ATPase2. GADPH means Glyceraldehyde-3-phosphate dehydrogenase. GADPH is used as an internal reference. Primer (5′–3′) means the sequence from the 5′ end to the 3′ end.
Data analysis
SPSS 13.0 software was used for statistical analysis. Data were of normal distribution and expressed as mean ± standard deviation, and intergroup comparisons were performed using an ANOVA, with p < 0.05 considered as statistically significant.
Results
SHE staining
The liver tissues of Group S showed no infiltration of neutrophils, and the liver structures were clear and complete. The liver tissues in Groups IR and P showed a narrowed hepatic sinusoid, obvious edema, and vacuolar degeneration after 4 h reperfusion. The liver cell stripes were disordered with local slabby necrosis. The liver cell injuries in Group P were significantly reduced compared to those of Group IR, and the liver morphology was close to normal, with congestion in the sinusoid and occasional infiltration of a small amount of inflammatory cells.
Detection of myocardial apoptosis by TUNEL
Compared with that of Group S, the rate of apoptosis in Group IR was increased significantly (all p < 0.01). Compared with that of the Group IR, the apoptotic rate in Group P was decreased significantly (all p < 0.01). As demonstrated in Fig. 1 and Table 2.
Figure 1.
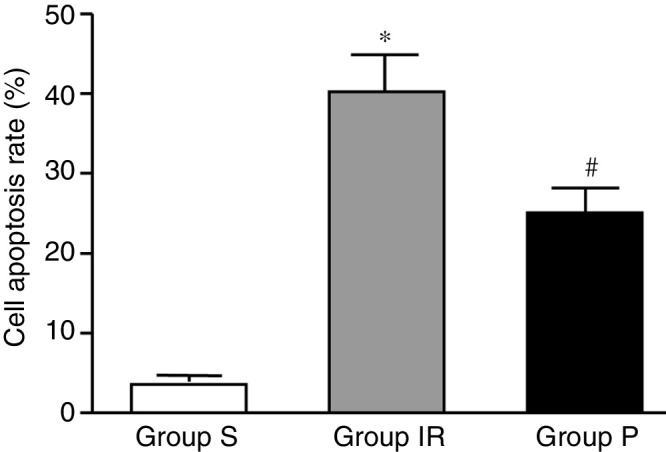
Detection of myocardial apoptosis. Group IR means hepatic ischemia-reperfusion injury group, and was attained by ischemia for 30 min. and reperfusion for 4 h. Group P means propofol group, and was subjected identical insult as in Group IR with the administration of propofol started 10 min. before ischemia with 20 mg.kg−1, following by continuous infusion at 20 mg.kg−1.h−1. S, Sham group; IR, ischemia-reperfusion group; P, propofol group.
*p < 0.01: compared with group S; #p < 0.01: compared with group IR.
Table 2.
Content detection of myocardial cell apoptosis rate in each group.
| Group | Myocardial cell apoptosis rate |
|---|---|
| Group S (n = 6) | 2.06 ± 0.89 |
| Group IR (n = 6) | 33.06 ± 3.09a |
| Group P (n = 6) | 19.26 ± 1.62b |
p < 0.01 compared with Group S.
p < 0.01 compared with Group IR.
Data are presented as mean ± standard deviation.
Detection of SERCA2 and cleaved-caspase3 proteins
Compared with those of Group S, SERCA2 protein expression was significantly down-regulated in Group IR, but cleaved-caspase3 protein was significantly up-regulated (all p < 0.01). Compared with those of the Group IR, SERCA2 was significantly up-regulated in Groups P but cleaved-caspase3 was significantly down-regulated in Group P (all p < 0.01). As demonstrated in Fig. 2 and Table 3.
Figure 2.
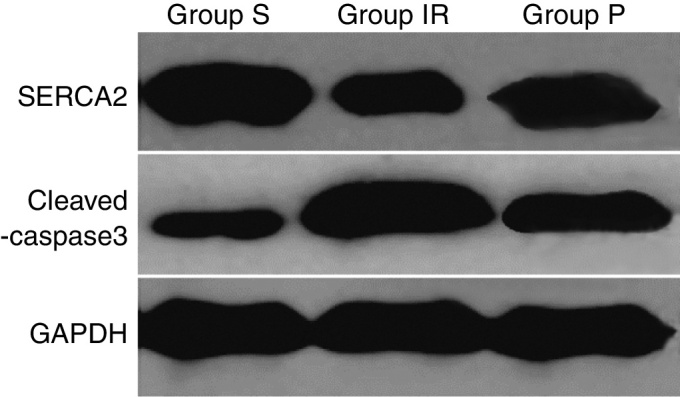
Detection of SERCA2 and cleaved-caspase3 proteins in each group. Group IR means hepatic ischemia-reperfusion injury group, and was attained by ischemia for 30 min. and reperfusion for 4 h. Group P means propofol group, and was subjected identical insult as in group IR with the administration of propofol started 10 min. before ischemia with 20 mg.kg−1, following by continuous infusion at 20 mg.kg−1.h−1. SERCA2, endoplasmic reticulum Ca2+-ATPase2; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; S, Sham group; IR, ischemia-reperfusion group; P, propofol group; SERCA2, endoplasmic reticulum Ca2+-ATPase2; GAPDH, glyceraldehyde-3-phosphate dehydrogenase. GADPH is used as an internal reference.
Table 3.
Content detection of SERCA2 and cleaved-caspase3 proteins in each group.
| Group | SERCA2/GAPDH | Cleaved-caspase3/GAPDH |
|---|---|---|
| Group S (n = 6) | 1.12 ± 0.058 | 0.33 ± 0.032 |
| Group IR (n = 6) | 0.54 ± 0.010a | 0.99 ± 0.058a |
| Group P (n = 6) | 0.93 ± 027b | 0.70 ± 0.053b |
p < 0.01 compared with Group S.
p < 0.01 compared with Group IR.
Data are presented as mean ± standard deviation.
Detection of SERCA2 and cleaved-caspase3 mRNA
Compared with those of Group S, SERCA2 mRNA was significantly down-regulated in the Group IR, but cleaved-caspase3 mRNA was significantly up-regulated (all p < 0.01). Compared with those of the IR group, SERCA2 mRNA was significantly up-regulated in subgroups P but cleaved-caspase3 was significantly down-regulated in Group P (all p < 0.01), as demonstrated in Table 4.
Table 4.
Content detection of SERCA2 and cleaved-caspase3 mRNA in each group.
| Group | SERCA2/GAPDH | Cleaved-caspase3/GAPDH |
|---|---|---|
| Group S (n = 6) | 1.10 ± 0.11 | 0.46 ± 0.033 |
| Group IR (n = 6) | 0.57 ± 0.052a | 0.98 ± 0.063a |
| Group P (n = 6) | 0.95 ± 0.92b | 0.65 ± 0.056b |
p < 0.01 compared with Group S.
p < 0.01 compared with Group IR.
Data are presented as mean ± standard deviation.
Discussion
Caspase3 is an important member in apoptosis execution enzyme in Caspase family, and it is necessary way in apoptotic protease cascade response. Once it is activate, apoptosis will be not avoided, and it has been maker enzyme and key protease in apoptosis process as sensitive index and main effectiveness factor in cell apoptosis. Therefore, expression level of caspase3 can be used to estimate cell apoptosis situation. Based on established I/R rat model according.
Goldhaber et al.11 this study aimed to detect cardiac myocyte apoptosis rate using TUNEL method and understand cleaved-caspase3 expression using western blot and PCR methods. We found that cardiac myocyte apoptosis rate is increased in IR model group than that in Group S, and cleaved-caspase3 mRNA and protein expressions are obviously up-regulated, indicating that the I/R model of myocardial injury is successfully constructed. In primary experiment, the highest apoptosis rate is detected at 4 h reperfusion with the serious pathological damage. Therefore, we selected the relevant indexes at 4 h. According to designed drug administration time and dose.16 We found that apoptosis rates in Group P is reduced than that in Group IR, cleaved-caspase3 mRNA and protein expressions are reduced using HE staining, TUNEL method, western blot and PCR methods. These results implicate that propofol can reduce apoptosis, myocardial damage in liver I/R rat, which are similar to Gao et al.16 and Jin et al.17
Intracellular calcium overload is an important role in leading to myocardium I/RI and improve cardiac myocyte apoptosis rate,18, 19, 20 and inhibiting calcium overload can significantly reduce myocardial damage caused by I/R.21 Sarcoplasmic reticulum is the main place to ingest, conserve and release Ca2+, SERCA2 can maintain the low level of Ca2+ by transmitting Ca2+ from intracytoplasm to sarcoplasmic reticulum. Talukder et al.22 indicated that higher SERCA2 expression can increase ingestion of Ca2+, reduce calcium overload and then reduce myocardial I/R injury. Propofol can adjust intracellular calcium homeostasis and reduce myocardial apoptosis.23 SERCA2 expression can reflect indirectly concentration of intracellular Ca2+ by detecting Ca2+ in cardiac muscle tissue. Compared with Group S, SERCA2 expression in I/R Group is reduced, and apoptosis rate increased; compared with I/R Group, SERCA2 in Group P is increased and apoptosis rate reduced. All of these results indicate that propofol can inhibit calcium overload by increasing expression of SERCA2, and then reduce myocardial apoptosis caused by I/R.
Taken together, total hepatic ischemia/reperfusion can lead to myocardial apoptosis in rat, propofol can reduce induced myocardial apoptosis by hepatic ischemia/reperfusion, and the main mechanism may be up-regulation of SERCA2 expression inhibit calcium overload. The further detailed mechanism should be further studied by measuring intracellular Ca2+ concentration.
Conflicts of interest
The authors declare no conflicts of interest.
Acknowledgments
This study was supported by Science and Technology Projects of Shanxi Health Planning Commission (n° 201-201-055).
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The animal use protocol has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Shanxi Medical University.
References
- 1.Miranda L.E., Capellini V.K., Reis G.S., et al. Effects of partial liver ischemia followed by global liver reperfusion on the remote tissue expression of nitric oxide synthase: lungs and kidneys. Transplant Proc. 2010;42:1557–1562. doi: 10.1016/j.transproceed.2010.02.097. [DOI] [PubMed] [Google Scholar]
- 2.Yang J.C., Wang Z.W., Li C.L., et al. Multiple organ injury at early stage of intestinal and hepatic ischemia-reperfusion in rats. J First Mil Med Univ. 2004;24:198–201. [PubMed] [Google Scholar]
- 3.Magan A.A., Khalil A.A., Ahmed M.H. Terlipressin and hepatorenal syndrome: what is important for nephrologists and hepatologists. World J Gastroenterol. 2010;16:5139–5147. doi: 10.3748/wjg.v16.i41.5139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cash W.J., McConville P., McDermott E., et al. Current concepts in the assessment and treatment of hepatic encephalopathy. QJM. 2010;103:9–16. doi: 10.1093/qjmed/hcp152. [DOI] [PubMed] [Google Scholar]
- 5.Weinbroun A.A., Hochhauser E., Rudick V., et al. Multiple oxygen dysfunction after remote circulatory arrest; common pathway of radical oxygen species. J Trauma. 1999;47:691–698. doi: 10.1097/00005373-199910000-00013. [DOI] [PubMed] [Google Scholar]
- 6.Omar M.A., Wang L., Clanachan A.S. Cardioprotection by GSK-3 inhibition: role of enhanced glycogen synthesis and attenuation of calcium overload. Cardiovasc Res. 2010;86:478–486. doi: 10.1093/cvr/cvp421. [DOI] [PubMed] [Google Scholar]
- 7.Arii S., Teramoto K., Kawamura T. Current progress in the understanding of and therapeutic strategies for ischetmia and reperfusion injury of the liver. J Hepatobiliary Pancreat Surg. 2003;10:189–194. doi: 10.1007/s00534-002-0720-z. [DOI] [PubMed] [Google Scholar]
- 8.Chen H., Lei Z.L., Han S.J., et al. Protective effects of propofol on renal injury during liver ischemia/reperfusion. Med J Chin PAPF. 2009;20:15–17. [Google Scholar]
- 9.Lotz C., Ping P., Kehl F. Letter by Lotz regarding article, “Reactive oxygen species-induced stimulation of 5′ AMP-activated protein kinase mediates sevoflurane-induced cardioprotection”. Circulation. 2010;121:e399–e400. doi: 10.1161/CIR.0b013e3181df9134. [DOI] [PubMed] [Google Scholar]
- 10.Kendzari D.E., Tcheng J.E. Double negatives. Am Heart J. 2003;145:9–11. doi: 10.1067/mhj.2003.33. [DOI] [PubMed] [Google Scholar]
- 11.Goldhaber J.I., Qayyum M.S. Oxygen free radicals and excitation-contraction coupling. Antioxid Redox Signal. 2000;2:55–64. doi: 10.1089/ars.2000.2.1-55. [DOI] [PubMed] [Google Scholar]
- 12.Yoshiki U., Yoshiyuki A., Kosuke M., et al. Endothelin-1 prolongs intracellular calcium transient decay in neonatal rat cardiac myocytes. Heart Vessels. 2012;27:98–105. doi: 10.1007/s00380-011-0133-5. [DOI] [PubMed] [Google Scholar]
- 13.Sun G.B., Sun H., Meng X.B., et al. Aconitine-induced Ca2+ overload causes arrhythmia and triggers apoptosis through p38 MAPK signaling pathway in rats. Toxicol Appl Pharmacol. 2014;279:8–22. doi: 10.1016/j.taap.2014.05.005. [DOI] [PubMed] [Google Scholar]
- 14.Chang G., Zhang D., Liu J., et al. Exenatide protects against hypoxia/reoxygenation-induced apoptosis by improving mitochondrial function in H9c2 cells. Exp Biol Med (Maywood) 2014;239:414–422. doi: 10.1177/1535370214522177. [DOI] [PubMed] [Google Scholar]
- 15.Lu B., Zhao J.L., Niu J.C., et al. Effect of propofol on myocardial injury induced by hepatic ischemla/reperfusion in rats and the role of PI3K/Akt signaling pathway. Chin J Anesthesiol. 2010;30:1250–1253. [Google Scholar]
- 16.Gao W.Z., Dan L., Tian Z.D., et al. Effects of propofol on lung injury and PI3K/Akt pathway in rats after liver ischemia and reperfusion. Chin J Pathol. 2013;29:488–492. [Google Scholar]
- 17.Jin Y.C., Kim W., Ha Y.M., et al. Propofol limits rat myocardial ischemia and reperfusion injury with an associated reduction in apoptotic cell death in vivo. Vascul Pharmacol. 2009;50:71–77. doi: 10.1016/j.vph.2008.10.002. [DOI] [PubMed] [Google Scholar]
- 18.Cheng Y.S., Chen Y., Li D. The mechanism and research status of calcium overload in myocardial ischemia/reperfusion injury. Chinese Med Innov. 2013;10:150–152. [Google Scholar]
- 19.Zhu N., Yao R.L., Liu J.S., et al. The regulation of the apoptosis and oxidative stress of myocardial apoptosis and oxidative stress in rat limb ischemia/reperfusion. Chin J Gerontol. 2015;35:6666–6668. [Google Scholar]
- 20.Talukder M.A., Yang F., Nishijima Y., et al. Reduced SERCA2a converts sub-lethal myocardial injury to infarction and affects postischemic functional recovery. J Mol Cell Cardiol. 2009;46:285–287. doi: 10.1016/j.yjmcc.2008.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang M.H., Yin Y.Q., He H.Y., et al. Effects of dioscin on calcium channels in rat ventricular cardiomyocytes. Pharmacol Clin Chin Mater Med. 2011;27:23–26. [Google Scholar]
- 22.Talukder M.A., Kalyanasundaram A., Zhao X., et al. Expression of SERCA isoform with faster Ca2+ transport properties improves postischemic cardiac function and Ca2+ handling and decreases myocardial infarction. Am J Physiol Heart Circ Physiol. 2007;293:H2418–H2428. doi: 10.1152/ajpheart.00663.2007. [DOI] [PubMed] [Google Scholar]
- 23.Kim H.S., Chang W.C., Hwang K.C., et al. Effect of propofol on calcium homeostasis in hypoxia-reoxygenated neonatal rat cardiomyocytes. Eur J Pharmacol. 2008;594:139–145. doi: 10.1016/j.ejphar.2008.07.027. [DOI] [PubMed] [Google Scholar]


