Abstract
To feed the continuously expanding world’s population, new crop varieties have been generated, which significantly contribute to the world’s food security. However, the growth of these improved plant varieties relies primarily on synthetic fertilizers, which negatively affect the environment and human health; therefore, continuous improvement is needed for sustainable agriculture. Several plants, including cereal crops, have the adaptive capability to combat adverse environmental changes by altering physiological and molecular mechanisms and modifying their root system to improve nutrient uptake efficiency. These plants operate distinct pathways at various developmental stages to optimally establish their root system. These processes include changes in the expression profile of genes, changes in phytohormone level, and microbiome-induced root system architecture (RSA) modification. Several studies have been performed to understand microbial colonization and their involvement in RSA improvement through changes in phytohormone and transcriptomic levels. This review highlights the impact of genes, phytohormones, and particularly root microbiota in influencing RSA and provides new insights resulting from recent studies on rice root as a model system and summarizes the current knowledge about biochemical and central molecular mechanisms.
Keywords: Rice, Root system architecture, Root growth, Genetic regulation, Phytohormone, Microbiota
Introduction
Continuously increasing adverse climatic pressure and anthropogenic activities pose a serious threat to crop productivity and sustainable agriculture. Therefore, it is essential to improve plant tolerance to produce crops in increasingly hostile conditions and existing agricultural resources to feed the ever-increasing world population. Plants require water, specific micronutrients, and macronutrients to maintain their normal growth and development. They acquire these essential nutrients mainly through the root system, from the soil surrounded by active roots, i.e., the rhizosphere (Zhang et al. 2020a). As a result, the low surface area of active roots accounts for insufficient uptake of nutrients. This condition is more detrimental under low nutrient availability and negatively affects the overall crop development and yield. Therefore, increasing the active root surface area may improve resource exploration and uptake. The increased active root surface area can be attained by enhancing the three-dimensional root configuration or root system architecture (RSA). Thus, RSA modification plays a significant role in improving plant growth by enhancing soil resource utilization and is considered to be more productive under adverse conditions such as low nutrient availability (Paez-Garcia et al. 2015). The RSA primarily improves soil exploration and facilitates enhanced nutrient and water uptake that affects overall plant productivity and stress tolerance. Rice is one of the most widely cultivated and consumed staple crops that feed nearly half of the world's population. Improving rice productivity may contribute to global food security at a greater extent. Therefore, understanding the mechanistic details of RSA modification in rice would play an essential role in enhancing rice productivity.
As a model plant, rice offers a functional experimental system to explore underlying molecular mechanisms of the fibrous root system of monocot plants (Coudert et al. 2010). Moreover, studying the adventitious roots system of Arabidopsis is insufficient to comprehend the genetic regulatory network for root development in monocot crops like rice, maize, and wheat. Consequently, understanding the hormonal and genetic regulation of rice root development will benefit in deciphering the regulatory network of root development in monocots. In addition, a detailed understanding of the critical genes/loci involved in RSA modification in rice would allow breeders and researchers to use genetic modification or marker-assisted selection to produce high-yield cultivars.
Several anatomical and developmental differences exist in dicots, and monocot roots. Embryonically derived primary or ‘taproot’ system is a typical feature of dicots, such as Arabidopsis root system (Bellini et al. 2014). In contrast, overall root architecture is more complex in the ‘fibrous’ root system of monocots. The monocot root system mostly constitutes seminal roots and shoot-borne crown and brace roots. These crown roots (CRs) in monocots also referred as adventitious roots emerged from root primordia during the developmental program (Hochholdinger et al. 2004). The CRs emerge 5–10 days post-germination from below-ground stem nodes, while brace roots emerge around 6 weeks after germination from above-ground stem nodes. For example, in maize, primary roots (PRs) were visible after 2–3 days of germination and seminal roots approximately 7 days after germination. Seminal roots are formed embryonically and emerge from the scutellar node in a similar manner as emergence of PRs (Singh et al. 2010). In cereals, lateral root (LR) initiation is also different as endodermal cells and pericycle both proceed to LR cells and create at the phloem poles than the xylem poles. Arabidopsis roots possess two xylem poles only, and LRs are initiated in an alternating left–right pattern at each pole. On the other hand, in cereal roots, there are ten or more phloem poles, resulting in a radial branching pattern around the parent root compared to that in Arabidopsis roots (Smith and De Smet 2012). Thus, in contrast to Arabidopsis, rice has a relatively complex root system, consisting of the embryonic PRs, seminal roots and post-embryonic adventitious roots. Primary and seminal roots have vital functions throughout the seedling stage, while adventitious roots form the functional root system (Steffens and Rasmussen 2016). Different rice cultivars have evolved adaptive root systems that adapt to various environmental stresses, including water and nutrient stress (Pedersen et al. 2021; Verma et al. 2021). For example, plants develop a deeper root system during water deficit conditions to obtain water from the subsoil portion to combat drought stress conditions (Lynch and Wojciechowski 2015). In addition to water uptake, a deeper root system would also be favorable for the rapid acquisition of nitrogen from the subsoil (Plett et al. 2020). In general, a thicker, branched, and deeper root system with a high root-to-shoot relationship can increase plant resistance to drought and nutrient stress (Klein et al. 2020). Therefore, studying the adaptive genetic variation (variation resulting from natural selection) in root system development and RSA modification would be more informative and beneficial for developing stress tolerance cultivars. Earlier research has shown that numerous factors can affect the root system development and alter the RSA through a complex regulatory network, as well as genetic, phytohormone and environmental stress factors. Genetic and phytohormones are two most prevalent factors affecting root development as well as shaping the RSA. Auxin and cytokinin (CK) are the most essential phytohormones required for the de novo genesis of root primordia. Other phytohormones such as abscisic acid (ABA) ethylene, gibberellic acid (GA) and salicylic acid (SA) are also involved in root formation, growth and modification of RSA (Qin et al. 2022). The crosstalk between these phytohormones and genetic factors affects a specific set of downstream regulators that lead to changes in gene expression, signal transduction, and metabolic conversions. For instance, CK induces the expression of auxin transporter, PIN1 and PIN7 via CYTOKININ RESPONSE FACTOR 2 (CRF2) and CRF6 which leads to modification of RSA (Šimášková et al. 2015). However, these interactions have been mainly characterized in dicot plants such as Arabidopsis, and translating this information into economically important monocot crop plants will open new research possibilities for increasing stress resilience and crop productivity by RSA modification.
Several microbes can affect root system development and RSA modification by altering hormonal, genetic and regulatory mechanisms (Valette et al. 2020; Batista et al. 2021). Thus, it is crucial to discover hormonal and genetic determinants governing root system development and RSA modification to decode the underlying mechanisms. The current review presents an overview focused on the genetic, hormonal, and microbial effects on root system development and RSA modification in rice and its relationship to the sustainable improvement of agricultural productivity.
Rice root system development
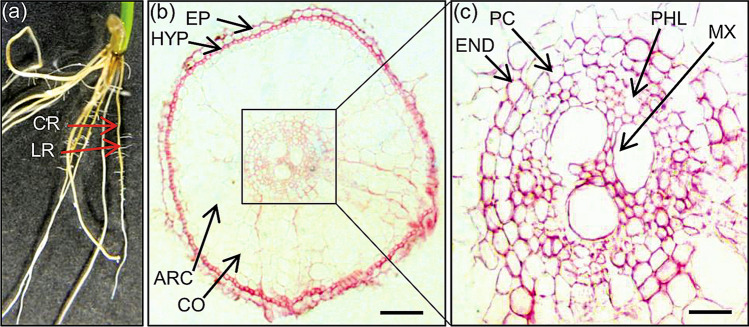
Rice forms a thick fibrous root system consisting of seminal, nodal and lateral roots (LRs) and post-embryonic adventitious roots (nodal roots or CRs) emerge from the basal nodes of the stem (Wang et al. 2020a). As soon as germination starts, the radicle finds its way out through the coleorrhiza and enveloping glume which forms the seminal root. Lateral roots emerge from the seminal roots, while adventitious roots emerge from the basal nodes with the advent of seedling growth. When seedlings emerge, most of the roots snapped off, but fresh roots develop very soon. The consecutive post-embryonic nodal roots or CRs differentiated from the nodal regions of stems and tillers. The bifurcation of the radicle and CRs results in longer LRs and shorter LRs. Longer LRs show positive geotropism and indeterminate growth, while shorter LRs are short, ageotropic and show determinate growth. Moreover, longer LRs emerge intermittently and carry expandable long or short LRs until the fifth branching order. Simultaneously, shorter LRs are larger in number and never develop a higher branching order. However, CRs differentiate from the stem but have similar characteristic features like root tissue (Mai et al. 2014). The radial structure of these CRs encompasses the stele (phloem and xylem), pericycle, endoderm, cortex, aerenchyma, sclerenchyma, exodermis, and epidermis locating from the center to periphery (Fig. 1). This radial structure reflects the capacity of rice roots to grow in both aerobic and anaerobic (flooding) environments (Maurel and Nacry 2020). Notably, the aerenchyma helps gas exchange with the shoot in anaerobic conditions (Pedersen et al. 2021). Additionally, pericycle and endodermal root cells proliferate to differentiate LRs and help the plant’s nutrients and water uptake (Jing and Strader 2019). Overall, the rice root system is highly diverse between the different varieties and is affected by several edaphic and environmental conditions such as nutrient availability, organic content, and soil type.
Fig. 1.
Root architecture and the anatomy of rice. a Seedling root system (cultivar Nipponbare) after 15 days of germination, b transverse section of rice root stained with Safranin, c enlarge view of root stele. ARC, aerenchyma; CO, cortex; CR, crown root; END, endodermis; EP, epidermis; HYP, hypodermis; LR, lateral root; MX, metaxylem; PC, pericycle; PHL, phloem. Scale bars: b 50 µm, c 20 µm
Root system architecture (RSA) and RSA modification
The RSA is a three-dimensional spatial configuration of the root system in the supporting medium. It determines the distribution of different root types across the soil profile (Ogura et al. 2019). The RSA refers to the root system surface topography described in the medium, particularly the root axis geometry precision. A primary RSA can be determined by numbers, patterns, lengths, orientation, angle, and diameter of primary and secondary branches just like any other phenotypic signifier. In addition, these components may be evolutionarily governed by complex and interactive genetic pathways that regulate growth and development to address environmental challenges. Hence, modifying RSA is an important strategy to enhance nutrient acquisition, especially under nutrient-deficient and water-stress conditions (Li et al. 2016).
Rice domestication started about 10,000 years ago, several rice varieties have grown in different ecosystems, including upland, rainfed lowland, flood-prone, and under different growing regimes ranging from conventional low-input to intensive high-input systems. A Gene bank at The International Rice Research Institute (IRRI), Philippines, contains over 100,000 rice genotypes (Wing et al. 2018). Many genotypes with variability in RSA establish a significant connection for identifying new genes involved in modifying RSA and root development in rice.. Several studies on "intrinsic pathway" or extrinsic "environmental response pathways" are significantly linked to the relationship between genetic and environmental factors listed in Table 1.
Table 1.
Effects of extrinsic factors in modulating root system architecture
| Condition | Impact | Genes | Interactions | References |
|---|---|---|---|---|
| Root system submergence | CRP development | SUB1 | GA, ethylene | Xu et al. (2006) and Fukao et al. (2011) |
| Drought | LR emergence | OsABA8ox2 | ABA | Zhang et al. (2020b) |
| Low phosphate availability | Root foraging: increased LR initiation | OsPTF1, Pho2, mir 399 | Auxin, CK, Ethylene, GA | Yi et al. (2005) and Bari et al. 2006 |
| High Al3+ | Inhibition of LR initiation | ART1 | Auxin, ethylene | Yamaji et al. (2009) |
Genetic regulation of RSA
To decode the mechanism for RSA regulation by genes, the direct genetic approach was traditionally used earlier before the advent of reverse genetic approaches. The quantitative trait locus (QTL) identification in mapping populations was the starting point for the direct genetic approach. In 1995, Champoux et al. (1995) published the first study on rice root system architecture modification. In 2008, Kamoshita et al. (2008) reviewed a QTL associated with primary or secondary or integrated traits of RSA parameters correlated with drought stress in 15 mapping rice populations, including CT9993/IR62266 for RSA modification. They suggested genotype responses for stress tolerance such as deep and thick roots and maintaining water status were of immense importance for higher yield under stress. In advancement, Khowaja et al. (2009) identified the QTL by meta-QTL analysis in the Bala/Azucena population associated with drought stress. They evaluated RSA-related traits like root number, length, thickness, roots depths, and root-to-shoot ratio. The advancement of high-throughput root phenotyping and 3D imaging regain interest in QTL identification in mapping populations. However, the large population size of the confidence interval is the fundamental restriction of QTL detection in mapping populations. In comparison to mapping populations, genome-wide association studies (GWAS) in natural populations with quick linkage disequilibrium decay have recently emerged as powerful methods for improving the resolution in localizing QTLs. The first GWAS findings studying root characteristics in rice were released by Clark et al. 2013; Courtois et al. 2013. Even though the resolution of QTL location in GWAS is far better than in mapping populations, the number of potential candidate genes is still vast, necessitating further evidence to identify the functional genes. Several QTLs have been identified in diversified rice populations that affect the root system and grain yield. Comprehensive knowledge directly impacted rice irrigation and fertilizer efficiency in the molecular breeding of RSA (Zhou et al. 2016). For example, the characterization of qRDWN6XB resulted in a different genetic resource for breeding rice cultivars and a reference point for strengthened grain yields and improved RSA under low nitrogen availability (Anis et al. 2019). Apart from identifying and characterizing RSA-related QTLs, a number of genes involved in manipulating RSA to address nutrient and water imbalance were identified and characterized (Table 2).
Table 2.
Genes and their involvement in root growth and development
| Gene | Function | References |
|---|---|---|
| DRO1 | Regulating the root growth angle | Uga et al. (2013), Singh et al. (2021) and Kitomi et al. (2020) |
| OsARF12 | Controlling PR length | Qi et al. (2012) |
| OsEXPA8 | Lengthening PR, boosting LRs and root hair counts | Ma et al. (2013) |
| OsEXPB2 | Modulating RSA and plant height by inhibiting cell growth | Zou et al. (2015) |
| OsWOX4 | Primary root elongation by regulating auxin transport | Chen et al. (2020) |
| OsMYB2P-1 | Regulation of root development under both phosphate sufficient and deficient conditions | Dai et al. (2012) |
| OsMYB4P | Regulation root development under phosphate deficient conditions | Yang et al. (2014) |
| OsRPK1 | Negatively controls the development of rice roots | Zou et al. (2014) |
| SOR1 | Modify RSA by altering gravitropic root response | Hanzawa et al. (2013) |
| OsARF16 | RSA modification by regulating auxin transport | Shen et al. (2015) |
| OsSIZ1 | RSA manipulation | Wang et al. (2015) |
| OsWRKYP74 | RSA modification | Dai et al. (2016) |
| OsRab6a | The architecture of the root system in response to Fe-deficient medium | Yang and Zhang (2016) |
| OSPIN2 | Root gravitropic response by affecting polar auxin transport at the root tip | Wang et al. (2018a) |
| RCc3 | Expanded growth of PR, adventitious and LRs | Li et al. (2018a) |
| OsWRKY28 | Influenced the root growth at the seedling stage | Wang et al. (2018b) |
| OsACS1 and OsACS2 | Controlling the modification of RSA | Lee et al. (2019) |
| OsJAZ9 | JA-mediated modulation of RSA | Singh et al. (2020) |
| OsGrx_C7 | Improving root growth in response to arsenic | Verma et al. (2020) |
| WEG1 | Increasing number, length and thickness of LRs | Lucob‐Agustin et al. (2020) |
| qSOR1 | Shallower root growth angle | Kitomi et al. (2020) |
| qRDWN6XB | Improving RSA under low nitrogen availability | Anis et al. (2019) |
| OsMED25 | JA-mediated root development | Suzuki et al. (2021) |
| OsEIL1 | Inhibition of primary root growth by activation of auxin, ABA biosynthesis and GA catabolism | Kin et al. (2022) |
| OsHDAC1 | OsHDAC1-GSK2/OsBZR1 module controls LR formation in rice | Hou et al. (2022) |
| OUR1/OsbZIP1 | Promoting root development through repressing auxin signaling | Hasegawa et al. (2021) |
| OsMED25 | JA-mediated root development | Suzuki et al. (2021) |
| OsFPFL4 | Modulating the root by affecting auxin and ROS homeostasis | Guo et al. (2020) |
| MHZ1/OsHK1 | Root growth regulation by inhibiting the ethylene receptors | Zhao et al. (2020) |
| OsZHD2 | Promotes root meristem activity in rice by inducing ethylene biosynthesis | Yoon et al. (2020) |
| MAL | Maintaining cell viability in the meristem after the initiation of root primordial formation, mediated by cytokinin signaling and reactive oxygen species (ROS) | Jiang et al. (2020) |
| OsVST1 | Influence meristem size through broad modulation of cell–cell communication or through specific signaling complexes | Shao et al. (2021) |
| CIPK9 | NH4+ dependent root growth | Xuan et al. (2019) |
| OsMADS57 | Modulating root growth by regulating nitrate translocation from root to shoot | Huang et al. (2019) |
| LAZY1 | Controlling shoot gravitropism and tiller angle by regulating the expression of auxin transporters | Zhu et al. (2020) |
| OsNAC2 | Modulating rice root development by integrating auxin and cytokinin pathways | Chanjuan et al. (2020), Mao et al. (2020) |
| WOX11 | YUCCA-Auxin-WOX11 module controls CR development in rice | Zhang et al. (2018a, b) |
| OsMADS25 | Regulating root system development via auxin signaling in rice | Zhang et al. (2018a, b) |
| OsAUX1 | Auxin influx carriers involved in primary root and root hair elongation | Yu et al. (2015) and Wang et al. (2019) |
Several studies were conducted to identify genetic regulators involved in modifying RSA. Auxin is the most important phytohormone involved in the regulation of RSA thus by modifying auxin transport it is possible to modify RSA. For instance, by influencing polar auxin transport at the root tip, OSPIN2 plays a vital role in root gravitropic reactions and deciding RSA in rice (Wang et al. 2018a). Similarly, a leucine-rich receptor-like kinase OsRPK1 negatively controls rice roots development by controlling polar auxin transport (Zou et al. 2014). OsARF16 also regulates RSA by regulating auxin transport and Fe homeostasis (Shen et al. 2015). OsARF12 is an auxin response factor that regulates auxin synthesis and polar auxin transport through OsYUCCAs, OsPINs, and OsPGPs, resulting in shortened length in rice plants (Qi et al. 2012). A transcription factor OsWOX4 played a pivotal role in PR elongation by regulating auxin transport, indicating its importance in RSA modification of the rice root system (Chen et al. 2020). The root growth angle (RGA) is an essential component of RSA, and it is used as a plant breeding parameter to determine variety. RGA can be regulated by modifying the expression of genes such as, DEEPER ROOTING 1 (DRO1), qSOR1 (quantitative trait locus for SOIL SURFACE ROOTING 1), and SOR1 (SOIL SURFACE ROOTING 1) involved in RSA modification by controlling root growth angle. DRO1 triggers unidirectional root growth and lowers root bending in response to gravity by causing cell elongation at the root tip, and it is negatively regulated by auxin (Uga et al. 2013; Singh et al. 2021). Another gene, qSOR1 (quantitative trait locus for SOIL SURFACE ROOTING 1), a DRO1 homolog, regulates the root growth angle. The qSOR1 gene primarily expresses in root columella cells and engages in root gravitropic responses. Alternatively, qSOR1 is negatively regulated by auxin (Kitomi et al. 2020). Another gene, SOR1 (SOIL SURFACE ROOTING 1), modified RSA by altering the gravitropic root response (Hanzawa et al. 2013). OsSIZ1 was concerned with RSA manipulation, as the ossiz1 mutant had short PRs and adventitious roots compared to wild plants (Wang et al. 2015). The LR development and root hair emergence are of immense importance to increase active root surface area. For example, A root-specific α-expansin gene, OsEXPA8, improve RSA by elongating PRs, boosting LRs and root hair counts, and improving rice root development (Ma et al. 2013). Another expansin gene, OsEXPB2, also modifies RSA by LR elongation and shoot growth (Zou et al. 2015).
Transcription factors are master regulator involved in RSA modification by controlling the expression of RSA-related genes. Many transcription factors were identified and characterized to understand the molecular networking involved in RSA modification. For instance, RSA was substantially modified by the overexpression of OsMYB2P-1 (Dai et al. 2012) and OsMYB4P transcription factors in rice (Yang et al. 2014). OsWRKYP74 conferred RSA modification as the transgenic plants better tolerate low Pi stress through activating genes triggered by Pi starvation and modulating RSA (Dai et al. 2016). A small GTPase, OsRab6a, plays a critical role in manipulating Fe+2 absorption in rice plants by regulating the physiological functions associated with the acquisition of Fe and RSA in response to Fe-deficiency (Yang and Zhang 2016). Furthermore, in RCC3 overexpression lines, local auxin biosynthesis and polar auxin transport increased auxin accumulation in the root. RCC3 generates pleiotropic phenotypes of reinforced RSA, such as expanded growth of PRs adventitious and LRs at the seedling stage (Li et al. 2018a). Another root growth controlling transcription factor, OsWRKY28, influenced root growth at the seedling stage and fertility at the reproductive stage, which likely affects jasmonic acid (JA) homeostasis (Wang et al. 2018b). The study showed that exogenous JA treatments mimicked oswrky28 mutant phenotypes with inhibited root elongation. Another study showed that osmed25 mutants had longer PRs than WT and exhibited JA-insensitive phenotypes. Also, S-type LRs density was lower while L-type LRs density was higher in osmed25 mutants than WT. This result suggested that OsMED25 participates in JA-mediated root development in rice (Suzuki et al. 2021).
Further studies related to root growth suggested that OsACS1 and OsACS2 are concerned with regulating RSA modification, transcription regulation of genes induced by Pi starvation, and cellular phosphorus homeostasis. The study has shown that OsACS mutants do not encourage LR growth in Pi-deficient condition, displaying the role of ethylene in LR production under Pi-deficiency (Lee et al. 2019). An additional study by Singh et al. (2020) stated that OsJAZ9 modulates RSA in response to potassium deficiency, as OsJAZ9 overexpressed plants showed shorter seminal roots with longer LRs. Moreover, LRs play an essential role in RSA, allowing plants to efficiently search for water and nutrients from the soil. However, in monocotyledonous plants, the mechanisms that regulate the evolution of LRs are poorly understood. According to a recent study, rice gene wavy root elongation growth 1 (WEG1) is involved in the formation of more extended and thicker LRs (L-type LRs) via asymmetric cell growth in the elongation region (Lucob-Agustin et al. 2020). In this regard, further study is still needed to identify the major genetic determinants governing RSA modification to engineer more branched root systems. In the current scenario, adverse environmental as well as edaphic factors affect the plant health and productivity. Studies discussed here provide a base for future studies to generate a robust root system. The root system with improved RSA can be generated by overexpressing the above discussed genes in root by advanced molecular biology tools or by plant breeding. These improved roots can be used for grafting with elite cultivar without disrupting the crop quality.
Hormonal regulation of RSA
The key biochemical and molecular elements of the intrinsic pathways include hormones, receptors, signaling components, and transcription factors (TFs). Interconnected networks of environmental stimuli receptors, downstream signal transduction, and TFs are extrinsic response pathways. In addition, many ecological perception and response network components are inter-regulated or shared between, intrinsically and hormonally regulated to respond to external signals (Table 3).
Table 3.
Hormones and their involvement in root growth and development
| Phytohormone | Function | References |
|---|---|---|
| Auxin (IAA) NAA | Increases CRP initiation and outgrowth | Sreevidya et al. (2010) |
| 2,4-D | Increases CRP initiation through cell division (but does not promote cell elongation and root outgrowth) | Sreevidya et al. (2010) and Debi et al. (2005) |
| Kinetin, trans-zeatin | Stimulates elongation of LRs | Sreevidya et al. (2010), Ashikari et al. (2005), Jain et al. (2006), Kurakawa et al. (2007) and Debi et al. (2005) |
| Kinetin, trans-zeatin | Stimulates CRP formation | Debi et al. (2005), Hirose et al. (2007) and Zhao et al. (2009) |
| Gibberellins (GA3) | Interacts with ethylene to promote CRP outgrowth and elongation | Steffens et al. (2006) |
| Gibberellins | Controlling root development by regulating local auxin biosynthesis and polar auxin transport by modulating the expression of OsYUCCA6 and PIN | Li et al. (2020a, b) |
| Ethylene | Promotes CR formation at submerged nodes–internodes | Lorbiecke and Sauter (1999) |
| Ethylene | Promotes CR emergence at submerged nodes through induction of epidermal cell death | Mergemann and Sauter (2000) |
| Strigolactones (SLs) | Promote CR elongation by inducing meristematic cell division | Arite et al. (2012) |
| Abscisic acid | Promotes root hair elongation by regulating auxin homeostasis in root tips | Wang et al. (2017) |
| Auxin and melatonin | Regulates root architecture by modulating auxin response in rice | Liang et al. (2017) |
Phytohormone regulation of primary root initiation, elongation, and development
In monocots, PR is derived from the radicle which established during embryogenetic development and forms the first CRs-based fibrous root system. Auxin is one of the critical phytohormones involved in regulating root system development and RSA modification. Changes in auxin levels can control the different sets of genes involved in root development and growth through the action of auxin/indole acetic acid (AUX/IAA) and Auxin Response Factor (ARF) modules (Guilfoyle and Hagen 2007). ARFs recognize and bind to auxin-responsive elements (AREs) in target gene promoters, activating or suppressing transcription. AUX/IAA proteins negatively regulate auxin response genes under no auxin or low auxin levels by binding or inactivating ARF activity (Abel et al. 1994). AUX/IAA proteins are intended for destruction by SCFTIR E3 ubiquitin ligase complex at higher auxin concentration (Gray et al. 2001). Jing and co-workers investigated a LATERAL ROOTLESS 2 (LRT2), which acts on AUX/IAA proteins. LRT2 is a cyclophilin that efficiently controls cis/trans isomerization of peptidylprolyl. LRT2 catalyzes the isomerization of the tandem proline residues of AUX/IAA necessary for recognition by OsTIR1. Also, the association of LRT2 with OsTIR1:OsIAA11 complex is increased by auxin and required to efficiently degrade AUX/IAAs (Jing et al. 2015). Perhaps, other modules of ARF regulation include miRNAs. For instance, the miR160 family plays a considerable role in improving PR and LR growth by improving ARF TFs, ARF10, and ARF16 functionally repetitive and required for root cap cell formation and maintenance. The overexpression of miR160 in rice also led to severe root cap defects, implying equivalent regulatory pathways in monocots (Wang et al. 2005). Moreover, the rice gene SLENDER RICE (SLR) and homolog SLENDER1 (SLN1) in barley are negative GA-mediated root growth regulators that tend to be influenced by auxin. When auxin is present, DELLA TFs are ubiquitinated and destroyed, allowing root cell division and elongation (Ikeda et al. 2001). A recent study reported that the mutation of OUR1/OsbZIP1, a member of the leucine zipper TF family, promotes root development by suppressing auxin signaling in rice (Hasegawa et al. 2021). As a result, rice is a suitable model for studying how the auxin-dependent gene regulatory network has developed to control post-embryonic root growth.
Phytohormone regulation of initiation and elongation of CRs
CRs are adventitious roots and specific to monocotyledons. CRs are also designated as nodal roots or roots that grow from shoots and account for most of its fibrous root system. CRs can be divided into adventitious roots and post-embryonic CRs. As soon as seedlings begin to grow, adventitious roots evolve from the coleoptile node of PR (radicle). When seedlings are pulled out, most of the roots get torn off, but very soon fresh post-embryonic CRs evolve and persist for whole plant life (Hochholdinger and Zimmermann 2008). All CRs (embryonic and post-embryonic) and seminal PR may be primary order roots because they emerge from the central plant stem and not from another root like LRs. However, most of the studies regarding root development focused on PR and LR in dicots like Arabidopsis. Until now most of the studies regarding genetic control of CRs development have been derived from research on Arabidopsis roots. Recent studies on rice and maize mutants suggest that the initiation and progression of CRs are regulated by auxin-mediated signaling, equivalent to PR and LR development in Arabidopsis (Rebouillat et al. 2009). However, the role of specific genes in developmental pathways can differ in the rice root system. Like Arabidopsis root development auxin signals are necessary for the equitable and asymmetric division of parenchyma cells which lead to formation of CRs. On the other hand, CK played a secondary role in promoting CR formation by antagonizing auxin-based signaling pathways. For example, WUSCHEL-RELATED HOMEOBOX11 (WOX11), a rice gene that encodes auxin and CK-induced transcription factor, expressed in the early Crown Root Primordia (CRP) and actively divided areas of the apical meristem (Jain et al. 2006). CR growth was inhibited in WOX11 knock-out mutants, while its overexpression increased CR cell division rates, resulting in CR development and growth. The transcription of CK and auxin-responsive genes has also been modified, indicating that WOX11 could have a crucial role in incorporating auxin and CK signaling to regulate cell division rates in CRP. Another gene RR2 operate as a negative CK signaling regulator that represses CR emergence by repressing meristem cellular proliferation (Zhao et al. 2009).
Studies on genetic and physiological processes also suggest the regulation of CRP formation and the production of CRs in stem nodes in response to environmental impacts (Mergemann and Sauter 2000). For instance, submergence-induced ethylene accumulation kills epidermal cells above CRP in deepwater rice accessions, allowing CRs to emerge through the epidermis of submerged nodal branches. CRP development is regulated by auxin as a study suggested that OsPIN1 RNAi-knockdown resulted in the discontinued development of CRP in rice (Xu et al. 2005). Recent studies suggest that GA is involved in CRP emergence and elongation with ethylene as a synergistic regulator. Furthermore, ABA acts as an inhibitor of GA and ethylene signaling pathways (Steffens and Sauter 2005). Similarly, strigolactones play a role in the positive regulation of CR elongation, potentially through modulation of auxin translocation and promoting meristematic cell division in the root (Arite et al. 2012). Rice dwarf mutants had a short CR phenotype due to an apparent decrease in cell division, resulting in a narrower meristematic region, for genes involved in SL biosynthesis (SL-deficient rice mutants max3/rms5/d17, max4/rms1/d10, d27) or SL (SL-insensible rice mutants max2/rms4/d3 and d14). This reduced cell distribution could be due to local auxin levels in SL-insensible rice mutants where SL modulation affects the numbers of the meristem as seen in PRs for homologous Arabidopsis genes (Arite et al. 2012). Even though numerous genes have been studied involving CR development in rice, our understanding of this process remains disorganized, and understanding its molecular mechanisms requires significant elucidation.
Role of jasmonic acid, salicylic acid, and brassinosteroids in RSA modification
Jasmonic acid and salicylic acid are also involved in regulation of root growth. JA increases auxin levels by inducing the expression of auxin biosynthetic gene ASA1, which leads to root growth inhibition (Sun et al. 2009). The potential interaction between JA and auxin signaling also occurs via AUXIN RESISTANT 1 (AXR1). In addition to its role in auxin signaling, AXR1 also shows JA-mediated root growth inhibition (Tiryaki and Staswick 2002). Taken together, the above reports suggest that JA signaling is linked to auxin homeostasis, leading to root growth inhibition. Similarly, SA can also modulate root growth by regulating root apical meristem (RAM) activity. As the 30 µM SA reduces the number of cells expressing cell division marker CYCLIN B1;1 (CYCB1;1) in the proximal meristem which causes the decreased root length (Pasternak et al. 2019). A treatment with higher SA concentrations (150 µM) results in enlarged proximal meristem cells without any CYCB1;1 signal in more than a half of treated roots (Pasternak et al. 2019). SA regulates root growth in interaction with plant hormones. Concentration-dependent effects on root growth were also shown for auxin (indole-3-acetic acid, IAA), ABA, and brassinosteroids (Bagautdinova et al. 2022). SA also cross-talks with CK for root growth regulation (Argueso et al. 2012). For instance, CK, benzyl adenine (BA), inhibits PR growth in the wild type Arabidopsis at 50 nM, whereas in SA biosynthesis mutant (eds16), this occurs at a lower (5 nM) concentration. In addition, endogenous auxin controls apoplast acidification and alkalization, which are required for the activation or repression of root cell elongation, respectively (Barbez et al. 2017). For ethylene, CK, and JA only inhibitory effects on root growth were demonstrated, while for gibberellins only stimulating effects on root growth were shown in various species (Kudo et al. 2012). Brassinosteroids (BRs) are steroidal hormones that play an essential role in many aspects of plant growth. BRs, in low concentrations, promote root growth through the control of root meristem size and cell elongation (Lv et al. 2018 and promote LR initiation (Bao et al. 2004). Moreover, treatments of rice seeds with BRs may also affect the root length as well as the LR density in rice (Vázquez-Glaría et al. 2021).
Root microbiota affect root system development and RSA modification
The soil contains an exceptionally diverse microbiome, susceptible to nutrient availability that is influenced by soil properties, including moisture, pH and nutrient content. Recent deep sequencing techniques suggest soil type significantly impacts rhizosphere microbial communities compared to plant genotype (Xu et al. 2020). These findings suggested that soil properties are essential in shaping the microbial communities. In addition to the soil type, the nature and composition of root exudate by plants roots are also likely to influence microbial communities (Dennis et al. 2010). Plants exudate contain polysaccharides, sugars, aromatic acids, amino acids, fatty acids and aliphatic acids and help in plant–microbe interaction by attracting and sustaining microorganisms (Hu et al. 2018; Venturi and Keel 2016; Vives-Peris et al. 2019). These microbes reinforce plant growth by nutrient solubilization, humification of recalcitrant organic matter and improving nutrient uptake and distribution; (Tfaily et al. 2014); averting pathogens’ entry and colonizing in plants’ roots (Enebe and Babalola 2019); harmonizing host immunity via induced systemic resistance (ISR); (Bruno et al. 2020) and regulating signaling pathways (Van Wees et al. 2008). Several microbes produce extracellular enzymes like cellulases, proteases, chitinases, lipases, and β-1–3 glucanases that hydrolyze a wide range of cell wall compounds, such as cellulose and chitin hemicelluloses and proteins (Lugtenberg et al. 2017). These enzymes improve soil fertility by releasing organic nutrients like N, P, and K into the plant rhizosphere (Doni et al. 2019).
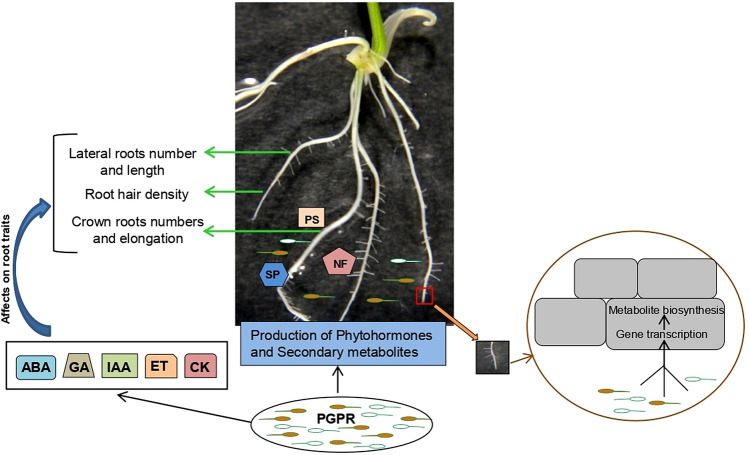
Apart from this, several rhizospheric microorganisms are able to modify RSA to improve nutrient and water exploration through distinct mechanisms (Vacheron et al. 2013). The central mechanism of microbial modulation of RSA encompasses the altering hormonal balance in plant roots either by secreting plant hormones or producing secondary metabolites interfering with hormonal pathways concerned with root development such as CK, auxin, ethylene, GA, and ABA (Ghosh et al. 2019). For instance, Azospirillum brasilense, Klebsiella pneumoniae and Bacillus altitudinis induces IAA production which resulted in increased root length. Microbes such as Phomopsis liquidambari B3 (Zhu et al. 2021), Azospirillum brasilense (Zaheer et al. 2022) and Rhizophagus irregularis (previously known as Glomus intraradices) (Mitra et al. 2021) affect the CK level which resulted in more LRs. As discussed above, these chemical signals alternatively affect the expression of root-related genes in plants. In addition, microbes have been shown to influence post-embryonic root development by modifying cell division and differentiation in PR and root hair emergence and LR formation (Zhang et al. 2017). The most characteristic root phenotype of plant tolerance in nutrient stress is PR growth inhibition. The proliferation of LRs and root hairs results in increased nutrient assimilation and improve shoot biomass. Another phenotype is an increase in shoot biomass accompanied by an increase in PR growth or deep root system. Both effects are dependent on nutrient type and availability. Plant root growth is also affected by microbial density and the distance between the bacteria and plant roots (Ortíz-Castro et al. 2009). One of the critical mechanisms reported by a study is that several microbes affect root development by cell division and differentiation mainly at two sites, i.e., meristem zone and LR formation zone (Wu et al. 2018). These changes affect the overall RSA of plants, usually accompanied by differences in endogenous plant responses. Because several rhizospheric microbes have been identified as producers of these hormones, it is tempting to speculate that microbial hormones may directly induce root system changes. The different studies are summarized in Table 4, and the impact of root microbiota on RSA in rice is illustrated in Fig. 2.
Table 4.
Effects of microbes in modulating root system architecture
| Microbes | Function | Effects | References |
|---|---|---|---|
| Azospirillum brasilense | IAA production | Increased in root elongation, root surface area, root dry matter and development of LRs | El-Khawas and Adachi (1999) |
| Azospirillum brasilense Sp245 | Enhances the expression of ethylene receptors | Increasing numbers of LRs | Vargas et al. (2012) |
| Azospirillum brasilense CA-10 | IAA production | Enhanced PG (polygalacturonase) activity in roots, better root morphogenesis | Sekar et al. (2000) |
| Azospirillum lipoferum 4B | Alters rice secondary metabolite profiles | Increased the number of roots, total root length and root surface | Chamam et al. (2013) |
| Azospirillum B510 | |||
| Azospirillum irakense | Induces polygalacturonase expression | Modification of root tissue structural properties | Sekar et al. (2000) and Dobbelaere et al. (2002) |
| Acanthamoeba castellanii (Protozoa) and Betaproteobacteria | Nutrient solubilization | Elongated (L-type) laterals, branched root systems | Kreuzer et al. (2006) |
| Bacillus altitudinis strain FD48 | IAA production | Modifies the early events of root architecture | Ambreetha et al. (2018) |
| Bacillus altitudinis (strain FD48) | Modulates the expression of AUX/IAA family genes | Modifies the root architecture | Ambreetha et al. (2018) |
| Bacillus amyloliquefaciens RWL-1 | ABA | LR development | Shahzad et al. (2017) |
| Bacillus amyloliquefaciens | Induces the expression of auxin-responsive OsASR6 | Root improvement | Agarwal et al. (2019) |
| Burkholderia pyrrocinia (R-46) | Auxins, siderophore production | Improves root length and diameter | Ferreira Rêgo et al. (2014) |
| Herbaspirillum seropedicae | Improves the expression of auxin and ethylene-responsive genes | LR initiation | Brusamarello-Santos et al. (2012) |
| Klebsiella pneumoniae | IAA production | Increased in root elongation, root surface area, root dry matter and development of LRs | El-Khawas and Adachi (1999) |
| Phomopsis liquidambari B3 | Enhanced auxin, cytokinin, and ethylene level | Nitrogen-mediated growth of LRs | Li et al. (2018b) |
| Pseudomonas fluorescens | Stress-related induction | Increased root length | Kandasamy et al. (2009) |
| Pseudomonas putida REN5, Pseudomonas fluorescens REN1 | IAA, ACC deaminase, siderophore production | Increase length, fresh weight and dry weight, branching | Etesami and Alikhani (2016) |
| Pseudomonas fluorescens (R-55) | Auxins, siderophore production | Improves root length and diameter | Ferreira Rêgo et al. (2014) |
| Glomus intraradices (Rhizophagus irregularis) | Affects the expression of OsCYCLOPS1 | Increases in root mass, thickness, length, and LR number, induces the formation of long LRs | Yano et al. (2008) and Gutjahr et al. (2009) |
| OsLRT1 and also alters auxin, ABA, CK, ethylene concentration | |||
| Rhizophagus irregularis | Perception of chitin oligomers | Increases growth of LRs | Chiu et al. (2018) |
| Rhizosphere isolates PGG2 and PGB4 | IAA Production | Increases root length | Ashrafuzzaman et al. (2009) |
| Trichoderma koningiopsis NBRI-PR5 and T. asperellum NBRI-K14 | Altered lignification patterns and packing of the sclerenchyma and exodermis cells | Stimulated the root growth | Anshu et al. (2022) |
Fig. 2.
Impact of root microbiota on RSA in rice. Rhizospheric microbes can modulate root growth and development via the production of secondary metabolites and phytohormones. Microbes can influence plant nutrition by phosphorus solubilization, nitrogen fixation and siderophore production, and alter gene transcription and metabolite biosynthesis in plant cells that affect root physiology. CK, cytokinin; ET, ethylene; GA, gibberellic acid; IAA, indole acetic acid; NF, nitrogen fixation; PS, phosphate solubilization; SP, siderophore production
Impact of microbial phytohormone on RSA improvement
Rhizospheric microbes can modify RSA and the structure of root tissues mainly through their ability to interfere with plant hormonal balance. Changes in RSA may result from interference of microbes with the main hormonal pathways involved in regulating plant root development: auxin, CK, ethylene, gibberellins, and ABA. In addition, phytohormone production and secretion, such as indole-3-acetic acid (IAA) by rhizospheric microbes, becomes a significant player in RSA reconfiguration. Finally, microbes can also alter the signals that control plant root morphogenesis and modulate RSA (Ortíz-Castro et al. 2009). Thus, using rhizospheric microbes promises to be a novel and environmentally friendly method of improving RSA and ultimately ensuring a long-term strategy for improving crop quality and yields.
Impact of microbial auxin and cytokinin
The potent regulator of plant organogenesis is the balance of auxin and CK, which regulate the root development and shapes RSA (Jing and Strader 2019). The endogenous ratio of auxin to CK in plants may be influenced by auxin and CK secretion by rhizospheric microbes and microbial metabolites that may interact with these hormonal pathways. To date, IAA is the well-studied auxin produced by a large number of rhizospheric microbes (Mohite 2013). Exogenous IAA regulates a broad range of plant development and root growth; for example, low IAA levels can promote the elongation of PR, while higher IAA levels encourage LR formation, enhance root hair formation, and reduce PR growth (Zhang et al. 2018a, b). Microbial metabolites like 2,4-diacetyl phloroglucinol (DAPG) and nitric oxide can also stimulate RSA modification in plants by interfering with the auxin synthesis pathway. DAPG is a well-known natural phenolic compound found in some specific strains of gram-negative bacteria and is responsible for the antimicrobial properties of Pseudomonas fluorescens (Weller et al. 2007). DAPG serves as a signal molecule for plants at low concentrations, stimulates root exudation (Phillips et al. 2004) and improves root branching (Brazelton et al. 2008). An auxin-dependent signaling pathway can be interfered with by DAPG, consequently modifying RSA (Brazelton et al. 2008).
The rice endophyte Phomopsis liquidambari B3 induces auxin and CK production as the endophyte's inoculation in rice root significantly enhanced auxin, CK, and ethylene levels in plant under varying nitrogen levels (Li et al. 2018b). Additionally, Azospirillum brasilense has nitrite reductase activity and produces nitric oxide during root colonization involved in regulating LR development by modulating the auxin signaling pathway (Rondina et al. 2020).
CK is the next most crucial phytohormone that regulates RSA in rice by CK signaling and CK interaction with auxin signaling (Neogy et al. 2020). Plant growth has been stimulated by inoculating plants with rhizospheric microbes that produce CK (Li et al. 2020a; Kieber and Schaller 2018). CK production has been reported in various microbes like Arthrobactergia comelloi, Azospirillum brasilense, Paenibacillus polymyxa, Bradyrhizobium japonicum, Pseudomonas fluorescens and Bacillus licheniformis (Cacciari et al. 1989; Perrig et al. 2007; García de Salamone et al. 2001; Timmusk et al. 1999). However, the role of CK synthesis by PGPR in RSA alterations is yet to be speculative.
Ethylene
Another important phytohormone that inhibits root elongation is ethylene, which induces senescence, abscission of different organs, and contributes to fruit ripening (Iqbal et al. 2017). Apart from senescence, abscission and fruit ripening, ethylene also modulates plant defence pathways (Yang et al. 2017). Rhizospheric microbes affect ethylene levels in plants by degrading ethylene precursors. These microbes secrete 1-aminocyclopropane-1-carboxylic acid deaminase (ACCd) coded by a gene acdS, which degrades ACC (immediate precursor of ethylene) into α-ketobutyrate and ammonium. Microbes use ammonium as a carbon and nitrogen source (Kim et al. 2020). Thus, microbial ACCd activity is thought to reduce root ethylene production by reducing the abundance of ethylene precursor ACC (Gamalero and Glick 2015), thereby decreasing the repressive effect of ethylene on root growth. Meanwhile, minimal impacts on RSA were seen when plants were infected with an acdS bacterial mutant or when plants impaired their ethylene signaling pathway (Contesto et al. 2008; Galland et al. 2012). Thus, it implies that ethylene is involved in RSA modification but not as a significant participant. Overall, the physiological significance of bacterial ACCd function requires further investigation.
Abscisic acid (ABA) and gibberellic acid (GA)
Several reports have revealed that rhizospheric microbes produce ABA and GA, or compounds that control the concentration of these phytohormones in plants (Wang et al. 2020b). It is well-studied that ABA plays a significant role in drought tolerance; however, it also plays a distinct role during LR development. An endophytic bacterium, Bacillus amyloliquefaciens RWL-1, produces ABA under saline conditions, and thus inoculation with this microbe improves salinity stress tolerance in rice (Shahzad et al. 2017). Gibberellins, particularly GA3, influence rice root elongation and modulate local auxin production and polar auxin transport (Li et al. 2020b). Gibberellins are produced by many microbes, including Achromobacter xylosoxidans, Acinetobacter spp., Azospirillum spp., Bacillus spp., Herbaspirillum seropedicae, Gluconobacter diazotrophicus, and Gelhizobia (Dodd et al. 2010). The application of GA in rice at an amount comparable to that produced by Azospirillum endorses root growth (Bottini et al. 2004). In addition to their involvement in RSA modification in plants, these hormones have also shown engagement in plant defensive systems. However, even though PGPR-mediated hormone production has been well studied, the genetic variables involved in their biosynthesis are mostly unknown. As a result, bacterial origin hormones in RSA modulation by interaction or hormonal balance in plants have not been fully understood.
Role of polyamine and GABA in plant–microbe interaction and RSA modification
Plant growth substance like GABA, and polyamines (PAs) also affect the plant–microbe association which in turns leads to the RSA modification. Studies suggest that PAs enhance nodule tolerance to the oxidative stress caused by the establishment of the symbiotic interaction. Research on Lotus japonicus showed that the expression of PA biosynthesis genes LjSPDS and LjSPMS were higher in early stage of nodulation (Efrose et al. 2008). In addition, metabolites derived from PAs catabolism also have important role in nodulation. For instance, GABA (γ-Aminobutyric acid) produced from 4-amino butanal has a dual role, acting as signaling molecule during plant-bacteria communication and functioning as an amino acid precursor in bacterial metabolism (Sulieman and Schulze 2010). In this trend, feeding with GABA into the phloem sap of Medicago truncatula plants causes a short-term increase in nodule activity and a remarkably increment in the amino acid and organic acid content in nodules (Sulieman and Schulze 2010). The mycorrhizal-plant root symbiosis is also affected by PAs and GABA in the same manner. For example, PAs increases the mycorrhizal symbiosis by increasing total free PA pools in Lotus glaber following colonization by Rhizophagus irregularis (Sannazzaro et al. 2007). In turn Rhizophagus irregularis modulate the expression of OsCYCLOPS1, OsLRT1 and alter the phytohormone such as auxin, CK, ABA and ethylene concentration. This altered phytohormone concentration increases root mass, thickness, length, and LR number. Thus, PAs and GABA increase the microbial colonization which leads to the modification of RSA.
Root microbiota impact on modification of root tissue structural properties in rice
Changes in plant gene expression by microbes occur via cell wall expansion or loosening of cell wall which is caused by changes in the ultrastructure of the root cell wall. For example, Bacillus subtilis GB03 enhances Arabidopsis development by generating volatile organic compounds found to influence the expression of 38 genes linked with the cell wall structure, in which 30 genes were responsible for expanding or loosening cell walls (Zhang et al. 2007). The endophytic microbe Azospirillum irakense induces polygalacturonase expression in inoculated rice roots (Sekar et al. 2000). Another study found that exogenous auxin application improves the role of induced polygalacturonase found in rice roots inoculated with Azospirillum irakense (Dobbelaere et al. 2002). PGPRs also play a significant role in changes in the chemical composition of the root cell wall. El Zemrany et al. (2007) conducted a study and observed that maize root cell walls had lower lignin content when inoculated with Azospirillum lipoferum CRT1 than uninoculated ones. Consequently, lower lignin content may promote cell elongation, which leads to root elongation. However, the impact of microbes on gene expression involving cell wall expansion and lignin deposition in rice root has not been well studied and needs further clarification.
Root microbiota impact on plant transcriptome in rice
The rice RSA could be improved by changing the hormonal level or expression of the root architecture-related genes discussed above. For instance, the up-regulation of DRO1 in shallow-rooted rice cultivars correlates to root enhancement due to improved root growth angle, with even more downward root growth and high yield under drought conditions (Singh et al. 2021; Uga et al. 2013). Furthermore, inoculation with Rhizophagus irregularis and Acanthamoeba castellanii increased the number of LRs (Kreuzer et al. 2006). Azospirillum lipoferum inoculation increased the root numbers, root surface area, and total root length by altering the expression of root-related genes (Chamam et al. 2013). Moreover, a low level of IAA induces PR elongation, whereas high concentration promotes LR development, increases root hair formation, and decreases PR length (Perrig et al. 2007; Remans et al. 2008).
Microbial strains may either directly supply IAA to the host plant or modify auxin pathways in the plant by controlling the expression of auxin-responsive genes. For example, Klebsiella and Azospirillum transformed tryptophan into IAA, which accounted for altering rice root growth (El-Khawas and Adachi 1999). Similarly, microbial isolates from rice rhizospheric soil produce IAA and influence rice seedling's root length (Ashrafuzzaman et al. 2009). A recent study showed that the inoculation of Bacillus altitudinis strain FD48 could modify root architecture by increasing root thickness, raising LRs while reducing root length. The IAA modulation in rice root caused by Bacillus altitudinis strain FD48 is due to variation in the expression level of the AUX/IAA gene family. An early study suggested the association of Azospirillum with the expression of plant genes, as the inoculation of Azospirillum brasilense Sp245 enhances the expression of ethylene receptors in two rice cultivars with contrasting nitrogen acquisition capacities (Vargas et al. 2012). All ethylene receptors need to be accumulated to create a positive relationship between the plant and bacteria. In terms of endophytes, Azoarcus was found to have differential rice root colonization (Miché et al. 2006). A mild defensive response occurred in a less compatible interaction followed by the stimulation of proteins linked to pathogenesis and proteins sharing domains with receptors such as pathogen-induced kinases, which were also stimulated by jasmonate (Miché et al. 2006). Rice roots inoculation with endophytic microbes Herbaspirillum seropedicae caused the expression of auxin and ethylene-responsive genes and the repression of PBZ1 proteins and thionin associated with defence (Brusamarello-Santos et al. 2012). These findings suggest that plant defence responses can be modulated during the colonization of endophytes and rhizospheric microbes. Furthermore, Plant-associated Bacillus spp. able to regulate auxin-responsive genes' expression level, modulating auxin concentration in the root and thus changing the early stages of root system architecture in rice seedlings. In addition to bacterial species, an AM fungus, Rhizophagus irregularis, causes the host to develop more LRs (Chen et al. 2018), which is dependent on receptor-like kinase OsCERK1, implying that the recognition of chitin oligomers is critical in AM fungal-mediated induction of LR growth in rice. Furthermore, another study reported that R. irregularis triggers plant signaling responsible for the induction of LR formation, involving a receptor kinase CHITIN ELICITOR RECEPTOR KINASE 1 (CERK1) in rice (Chiu and Paszkowski 2020; Chiu et al. 2018).
Furthermore, microbes can alter the composition and volume of metabolites and influence root exudation. For example, when inoculated with Herbaspirillum seropedicae, rice plants showed a higher malate content in shoot tissues (Curzi et al. 2008). The early effect of numerous Azospirillum strains on rice root and shoot secondary metabolite profiles has been investigated in recent studies. Secondary metabolite profiling of two rice cultivars inoculated with two different strains of Azospirillum has shown that secondary metabolite profiles have been modified with phenolic compounds like flavonoids hydroxyl cinnamic derivatives (Chamam et al. 2013). In addition, a relatively high concentration of quaternary compounds, glycine betaine, was observed in rice infected with Pseudomonas pseudoalcali (Jha et al. 2011). Thus, establishing beneficial associations requires mutual recognition and substantial coordination of plant and microbial responses in controlling RSA and improving crop production and quality.
Concluding remarks and future perspectives
Significant progress has been made to improve understanding of the genetic, hormonal and microbial regulation of root development and RSA modification in rice. Detailed research on genes controlling root traits will open the door for additional in-depth molecular and genetic studies of rice and other cereal root system development. Despite the identification of numerous genes involved in root growth, our understanding of molecular mechanisms behind CR growth, root elongation, LR development and root hair formation is still unsatisfactory. Most investigations focus on identifying through mutants or QTL analyses of particular genes that restrict our overall understanding of root growth development mechanisms. Integrating functional genomics, proteomics, transcriptomics, and phenomics will significantly expand our knowledge of the molecular mechanisms underlying RSA modification. However, several genes involved in RSA have been identified, still a limited data is available about how these genes could be utilized to strengthen the root system through breeding or genetic engineering techniques. Cloning genes from QTL analysis is a viable method for identifying potential candidates for molecular breeding; nevertheless, identifying QTLs with minor effects. Another primary strategy for improving crops is genetic manipulation. It offers the chance to modify specific genes for particular requirements. The CRISPR/Cas9 method allows for more focused genome editing of RSA-related alleles to improve locus performance. Additionally, using novel technologies to examine RSA in natural soils and complex environments, such as magnetic resonance imaging and X-ray micro-computed tomography, will allow for a more accurate assessment of root traits and identification of associated genes.
It is not surprising that rhizospheric microbes influence rice root development. The latest evidence on the impacts of rhizospheric microbes on root development is summarized in this review. It will encourage the use of naturally growing soil microbes to facilitate plant growth and health while minimizing herbicides and synthetic fertilizers in the field. Future research combining plant developmental biology and plant–microbe interactions will illuminate how soil microbes influence root development. This review will assist in better understanding these complex cross-kingdom interactions, root developmental biology, and microbial signaling. Finally, the knowledge will aid in developing sustainable plant growth-promoting technologies, which can significantly improve crop yield and food security.
Acknowledgements
P.K.V. and N.P. thankfully acknowledged University of Lucknow and University Grants Commission (UGC), India for DSKPDF.
Funding
This research is funded by University Grants Commission (UGC), India (No. F.4-2/2006(BSR)/BL/17-18/0140).
Declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
The manuscript does not involve any animal study.
Contributor Information
Pankaj Kumar Verma, Email: pankajverma66@gmail.com.
Nalini Pandey, Email: nalini_pandey@rediffmail.com.
References
- Abel S, Oeller PW, Theologis A. Early auxin-induced genes encode short-lived nuclear proteins. Proc Natl Acad Sci. 1994;91:326–330. doi: 10.1073/pnas.91.1.326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agarwal P, Singh PC, Chaudhry V, Shirke PA, Chakrabarty D, Farooqui A, Nautiyal CS, Sane AP, Sane VA. PGPR-induced OsASR6 improves plant growth and yield by altering root auxin sensitivity and the xylem structure in transgenic Arabidopsis thaliana. J Plant Physiol. 2019;240:153010. doi: 10.1016/j.jplph.2019.153010. [DOI] [PubMed] [Google Scholar]
- Ambreetha S, Chinnadurai C, Marimuthu P, Balachandar D. Plant-associated Bacillus modulates the expression of auxin-responsive genes of rice and modifies the root architecture. Rhizosphere. 2018;5:57–66. doi: 10.1016/j.rhisph.2017.12.001. [DOI] [Google Scholar]
- Anis GB, Zhang Y, Islam A, Zhang Y, Cao Y, Wu W, Cao L, Cheng S. RDWN6 XB, a major quantitative trait locus positively enhances root system architecture under nitrogen deficiency in rice. BMC Plant Biol. 2019;19:1–13. doi: 10.1186/s12870-018-1620-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anshu A, Agarwal P, Mishra K, Yadav U, Verma I, Chauhan S, Srivastava PK, Singh PC. Synergistic action of Trichoderma koningiopsis and T. asperellum mitigates salt stress in paddy. Physiol Mol Biol Plants. 2022;28:987–1004. doi: 10.1007/s12298-022-01192-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Argueso CT, Ferreira FJ, Epple P, To JP, Hutchison CE, Schaller GE, Dangl JL, Kieber JJ. Two-component elements mediate interactions between cytokinin and salicylic acid in plant immunity. PLoS Genet. 2012;8:e1002448. doi: 10.1371/journal.pgen.1002448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arite T, Kameoka H, Kyozuka J. Strigolactone positively controls crown root elongation in rice. J Plant Growth Regul. 2012;31:165–172. doi: 10.1007/s00344-011-9228-6. [DOI] [Google Scholar]
- Ashikari M, Sakakibara H, Lin S, Yamamoto T, Takashi T, Nishimura A, Angeles ER, Qian Q, Kitano H, Matsuoka M. Cytokinin oxidase regulates rice grain production. Science. 2005;309:741–745. doi: 10.1126/science.1113373. [DOI] [PubMed] [Google Scholar]
- Ashrafuzzaman M, Hossen FA, Ismail MR, Hoque A, Islam MZ, Shahidullah S, Meon S. Efficiency of plant growth-promoting rhizobacteria (PGPR) for the enhancement of rice growth. Afr J Biotech. 2009;8:1247–1252. [Google Scholar]
- Bagautdinova ZZ, Omelyanchuk N, Tyapkin AV, Kovrizhnykh VV, Lavrekha VV, Zemlyanskaya EV. Salicylic acid in root growth and development. Int J Mol Sci. 2022;23:1–26. doi: 10.3390/ijms23042228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bao F, Shen J, Brady SR, Muday GK, Asami T, Yang Z. Brassinosteroids interact with auxin to promote lateral root development in Arabidopsis. Plant Physiol. 2004;4:1624–1631. doi: 10.1104/pp.103.036897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbez E, Dünser K, Gaidora A, Lendl T, Busch W. Auxin steers root cell expansion via apoplastic pH regulation in Arabidopsis thaliana. Proc Natl Acad Sci. 2017;114:E4884–E4893. doi: 10.1073/pnas.1613499114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bari R, Pant BD, Stitt M, Scheible W-R. PHO2, microRNA399, and PHR1 define a phosphate-signaling pathway in plants. Plant Physiol. 2006;141:988–999. doi: 10.1104/pp.106.079707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Batista BD, Dourado MN, Figueredo EF, Hortencio RO, Marques JP, Piotto FA, Bonatelli ML, Settles ML, Azevedo JL, Quecine MC. The auxin-producing Bacillus thuringiensis RZ2MS9 promotes the growth and modifies the root architecture of tomato (Solanum lycopersicum cv. Micro-Tom) Arch Microbiol. 2021;19:1–4. doi: 10.1007/s00203-021-02361-z. [DOI] [PubMed] [Google Scholar]
- Bellini C, Pacurar DI, Perrone I. Adventitious roots and lateral roots: similarities and differences. Annu Rev Plant Biol. 2014;65:639–666. doi: 10.1146/annurev-arplant-050213-035645. [DOI] [PubMed] [Google Scholar]
- Bottini R, Cassán F, Piccoli P. Gibberellin production by bacteria and its involvement in plant growth promotion and yield increase. Appl Microbiol Biotechnol. 2004;65:497–503. doi: 10.1007/s00253-004-1696-1. [DOI] [PubMed] [Google Scholar]
- Brazelton JN, Pfeufer EE, Sweat TA, Gardener BBM, Coenen C (2008) 2,4-Diacetylphloroglucinol alters plant root development. Mol Plant Microbe Interact® 21:1349–1358. 10.1094/MPMI-21-10-1349 [DOI] [PubMed]
- Bruno M, Kersten S, Bain JM, Jaeger M, Rosati D, Kruppa MD, Lowman DW, Rice PJ, Graves B, Ma Z. Transcriptional and functional insights into the host immune response against the emerging fungal pathogen Candida auris. Nat Microbiol. 2020;5:1516–1531. doi: 10.1038/s41564-020-0780-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brusamarello-Santos L, Pacheco F, Aljanabi S, Monteiro R, Cruz L, Baura V, Pedrosa F, Souza E, Wassem R. Differential gene expression of rice roots inoculated with the diazotroph Herbaspirillum seropedicae. Plant Soil. 2012;356:113–125. doi: 10.1007/s11104-011-1044-z. [DOI] [Google Scholar]
- Cacciari I, Lippi D, Pietrosanti T, Pietrosanti W. Phytohormone-like substances produced by single and mixed diazotrophic cultures of Azospirillum and Arthrobacter. Plant Soil. 1989;115:151–153. doi: 10.1007/BF02220706. [DOI] [Google Scholar]
- Chamam A, Sanguin H, Bellvert F, Meiffren G, Comte G, Wisniewski-Dyé F, Bertrand C, Prigent-Combaret C. Plant secondary metabolite profiling evidences strain-dependent effect in the Azospirillum-Oryza sativa association. Phytochemistry. 2013;87:65–77. doi: 10.1016/j.phytochem.2012.11.009. [DOI] [PubMed] [Google Scholar]
- Champoux M, Wang G, Sarkarung S, Mackill DJ, O’Toole JC, Huang N, McCouch SR. Locating genes associated with root morphology and drought avoidance in rice via linkage to molecular marker. Theor Appl Genet. 1995;90:969–981. doi: 10.1007/BF00222910. [DOI] [PubMed] [Google Scholar]
- Chen EC, Morin E, Beaudet D, Noel J, Yildirir G, Ndikumana S, Charron P, St-Onge C, Giorgi J, Krüger M. High intraspecific genome diversity in the model arbuscular mycorrhizal symbiont Rhizophagus irregularis. New Phytol. 2018;220:1161–1171. doi: 10.1111/nph.15472. [DOI] [PubMed] [Google Scholar]
- Chen R, Xu N, Yu B, Wu Q, Li X, Wang G, Huang J. The WUSCHEL-related homeobox transcription factor OsWOX4 controls the primary root elongation by activating OsAUX1 in rice. Plant Sci. 2020;298(110575):1–13. doi: 10.1016/j.plantsci.2020.110575. [DOI] [PubMed] [Google Scholar]
- Chiu CH, Paszkowski U. Receptor-like kinases sustain symbiotic scrutiny. Plant Physiol. 2020;182:1597–1612. doi: 10.1104/pp.19.01341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu CH, Choi J, Paszkowski U. Independent signalling cues underpin arbuscular mycorrhizal symbiosis and large lateral root induction in rice. New Phytol. 2018;217:552–557. doi: 10.1111/nph.14936. [DOI] [PubMed] [Google Scholar]
- Clark RT, Famoso AN, Zhao K, Shaff JE, Craft EJ, Bustamante CD, McCouch SR, Aneshansley DJ, Kochian LV. High-throughput two-dimensional root system phenotyping platform facilitates genetic analysis of root growth and development. Plant Cell Environ. 2013;36:454–466. doi: 10.1111/j.1365-3040.2012.02587. [DOI] [PubMed] [Google Scholar]
- Contesto C, Desbrosses G, Lefoulon C, Béna G, Borel F, Galland M, Gamet L, Varoquaux F, Touraine B. Effects of rhizobacterial ACC deaminase activity on Arabidopsis indicate that ethylene mediates local root responses to plant growth-promoting rhizobacteria. Plant Sci. 2008;175(1–2):178–189. doi: 10.1016/j.plantsci.2008.01.020. [DOI] [Google Scholar]
- Coudert Y, Périn C, Courtois B, Khong NG, Gantet P. Genetic control of root development in rice, the model cereal. Trends Plant Sci. 2010;15:219–226. doi: 10.1016/j.tplants.2010.01.008. [DOI] [PubMed] [Google Scholar]
- Courtois B, Audebert A, Dardou A, Roques S, Ghneim-Herrera T, Droc G, Frouin J, Rouan L, Goze E, Kilian A, Ahmadi N, Dingkuhn M. Genome-wide association mapping of root traits in a japonica rice panel. PLoS ONE. 2013;8(11):1–18. doi: 10.1371/journal.pone.0078037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curzi M, Ribaudo C, Trinchero G, Curá J, Pagano E. Changes in the content of organic and amino acids and ethylene production of rice plants in response to the inoculation with Herbaspirillum seropedicae. J Plant Interact. 2008;3:163–173. doi: 10.1080/17429140802255167. [DOI] [Google Scholar]
- Dai X, Wang Y, Yang A, Zhang W-H. OsMYB2P-1, an R2R3 MYB transcription factor, is involved in the regulation of phosphate-starvation responses and root architecture in rice. Plant Physiol. 2012;159:169–183. doi: 10.1104/pp.112.194217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai X, Wang Y, Zhang W-H. OsWRKY74, a WRKY transcription factor, modulates tolerance to phosphate starvation in rice. J Exp Bot. 2016;67:947–960. doi: 10.1093/jxb/erv515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debi BR, Chhun T, Taketa S, Tsurumi S, Xia K, Miyao A, Hirochika H, Ichii M. Defects in root development and gravity response in the aem1 mutant of rice are associated with reduced auxin efflux. J Plant Physiol. 2005;162:678–685. doi: 10.1016/j.jplph.2004.09.012. [DOI] [PubMed] [Google Scholar]
- Dennis PG, Miller AJ, Hirsch PR. Are root exudates more important than other sources of rhizodeposits in structuring rhizosphere bacterial communities? FEMS Microbiol Ecol. 2010;72:313–327. doi: 10.1111/j.1574-6941.2010.00860.x. [DOI] [PubMed] [Google Scholar]
- Dobbelaere S, Croonenborghs A, Thys A, Ptacek D, Okon Y, Vanderleyden J. Effect of inoculation with wild type Azospirillum brasilense and A. irakense strains on development and nitrogen uptake of spring wheat and grain maize. Biol Fertil Soils. 2002;36:284–297. doi: 10.1007/s00374-002-0534-9. [DOI] [Google Scholar]
- Dodd I, Zinovkina N, Safronova V, Belimov A. Rhizobacterial mediation of plant hormone status. Ann Appl Biol. 2010;157:361–379. doi: 10.1111/j.1744-7348.2010.00439.x. [DOI] [Google Scholar]
- Doni F, Fathurrahman F, Mispan MS, Suhaimi NSM, Yusoff WMW, Uphoff N. Transcriptomic profiling of rice seedlings inoculated with the symbiotic fungus Trichoderma asperellum SL2. J Plant Growth Regul. 2019;38:1507–1515. doi: 10.1007/s00344-019-09952-7. [DOI] [Google Scholar]
- El Zemrany H, Czarnes S, Hallett PD, Alamercery S, Bally R, Jocteur-Monrozier L. Early changes in root characteristics of maize (Zea mays) following seed inoculation with the PGPR Azospirillum lipoferum CRT1. Plant Soil. 2007;291:109–118. doi: 10.1007/s11104-006-9178-0. [DOI] [Google Scholar]
- El-Khawas H, Adachi K. Identification and quantification of auxins in culture media of Azospirillum and Klebsiella and their effect on rice roots. Biol Fertil Soils. 1999;28:377–381. doi: 10.1007/s003740050507. [DOI] [Google Scholar]
- Enebe MC, Babalola OO. The impact of microbes in the orchestration of plants resistance to biotic stress: a disease management approach. Appl Microbiol Biotechnol. 2019;103:9–25. doi: 10.1007/s00253-018-9433-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferreira Rêgo MC, Ilkiu-Borges F, Filippi MCC, de Gonçalves LA, Silva GB. Morphoanatomical and biochemical changes in the roots of rice plants induced by plant growth-promoting microorganisms. J Bot. 2014;2014:818797. doi: 10.1155/2014/818797. [DOI] [Google Scholar]
- Fukao T, Yeung E, Bailey-Serres J. The submergence tolerance regulator SUB1A mediates crosstalk between submergence and drought tolerance in rice. Plant Cell. 2011;23:412–427. doi: 10.1105/tpc.110.080325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galland M, Gamet L, Varoquaux F, Touraine B, Desbrosses G. The ethylene pathway contributes to root hair elongation induced by the beneficial bacteria Phyllobacterium brassicacearum STM196. Plant Sci. 2012;190:74–81. doi: 10.1016/j.plantsci.2012.03.008. [DOI] [PubMed] [Google Scholar]
- Gamalero E, Glick BR. Bacterial modulation of plant ethylene levels. Plant Physiol. 2015;169:13–22. doi: 10.1104/pp.15.00284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García de Salamone IE, Hynes RK, Nelson LM. Cytokinin production by plant growth promoting rhizobacteria and selected mutants. Can J Microbiol. 2001;47:404–411. doi: 10.1139/w01-029. [DOI] [PubMed] [Google Scholar]
- Ghosh D, Gupta A, Mohapatra S. Dynamics of endogenous hormone regulation in plants by phytohormone secreting rhizobacteria under water-stress. Symbiosis. 2019;77:265–278. doi: 10.1007/s13199-018-00589-w. [DOI] [Google Scholar]
- Gray WM, Kepinski S, Rouse D, Leyser O, Estelle M. Auxin regulates SCF TIR1-dependent degradation of AUX/IAA proteins. Nature. 2001;414:271–276. doi: 10.1038/35104500. [DOI] [PubMed] [Google Scholar]
- Guilfoyle TJ, Hagen G. Auxin response factors. Curr Opin Plant Biol. 2007;10:453–460. doi: 10.1016/j.pbi.2007.08.014. [DOI] [PubMed] [Google Scholar]
- Guo Y, Wu Q, Xie Z, Yu B, Zeng R, Min Q, Huang J. OsFPFL4 is involved in the root and flower development by affecting auxin levels and ROS accumulation in rice (Oryza sativa) Rice. 2020;13:1–15. doi: 10.1186/s12284-019-0364-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutjahr C, Casieri L, Paszkowski U. Glomus intraradices induces changes in root system architecture of rice independently of common symbiosis signaling. New Phytol. 2009;182:829–837. doi: 10.1111/j.1469-8137.2009.02839.x. [DOI] [PubMed] [Google Scholar]
- Hanzawa E, Sasaki K, Nagai S, Obara M, Fukuta Y, Uga Y, Miyao A, Hirochika H, Higashitani A, Maekawa M. Isolation of a novel mutant gene for soil-surface rooting in rice (Oryza sativa L.) Rice. 2013;6:1–11. doi: 10.1186/1939-8433-6-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasegawa T, Lucob-Agustin N, Yasufuku K, Kojima T, Nishiuchi S, Ogawa A, Takahashi-Nosaka M, Kano-Nakata M, Inari-Ikeda M, Sato M, Tsuji H. Mutation of OUR1/OsbZIP1, which encodes a member of the basic leucine zipper transcription factor family, promotes root development in rice through repressing auxin signaling. Plant Sci. 2021;306:110861. doi: 10.1016/j.plantsci.2021.110861. [DOI] [PubMed] [Google Scholar]
- Hirose N, Makita N, Kojima M, Kamada-Nobusada T, Sakakibara H. Overexpression of a type-A response regulator alters rice morphology and cytokinin metabolism. Plant Cell Physiol. 2007;48:523–539. doi: 10.1093/pcp/pcm022. [DOI] [PubMed] [Google Scholar]
- Hochholdinger F, Zimmermann R. Conserved and diverse mechanisms in root development. Curr Opin Plant Biol. 2008;11:70–74. doi: 10.1016/j.pbi.2007.10.002. [DOI] [PubMed] [Google Scholar]
- Hochholdinger F, Park WJ, Sauer M, Woll K. From weeds to crops: genetic analysis of root development in cereals. Trends Plant Sci. 2004;9:42–48. doi: 10.1016/j.tplants.2003.11.003. [DOI] [PubMed] [Google Scholar]
- Hou J, Zheng X, Ren R, Shi Q, Xiao H, Chen Z, Yue M, Wu Y, Hou H, Li L. The histone deacetylase 1/GSK3/SHAGGY-like kinase 2/BRASSINAZOLE-RESISTANT 1 module controls lateral root formation in rice. Plant Physiol. 2022;189:858–873. doi: 10.1093/plphys/kiac015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu L, Robert CA, Cadot S, Zhang X, Ye M, Li B, Manzo D, Chervet N, Steinger T, Van Der Heijden MG. Root exudate metabolites drive plant-soil feedbacks on growth and defense by shaping the rhizosphere microbiota. Nat Commun. 2018;9:1–13. doi: 10.1038/s41467-018-05122-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang S, Liang Z, Chen S, Sun H, Fan X, Wang C, Xu G, Zhang Y. A transcription factor, OsMADS57, regulates long-distance nitrate transport and root elongation. Plant Physiol. 2019;180:882–895. doi: 10.1104/pp.19.00142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda A, Ueguchi-Tanaka M, Sonoda Y, Kitano H, Koshioka M, Futsuhara Y, Matsuoka M, Yamaguchi J. slender rice, a constitutive gibberellin response mutant, is caused by a null mutation of the SLR1 gene, an ortholog of the height-regulating gene GAI/RGA/RHT/D8. Plant Cell. 2001;13:999–1010. doi: 10.1105/tpc.13.5.999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iqbal N, Khan NA, Ferrante A, Trivellini A, Francini A, Khan M. Ethylene role in plant growth, development and senescence: interaction with other phytohormones. Front Plant Sci. 2017;8:475. doi: 10.3389/fpls.2017.00475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain M, Tyagi AK, Khurana JP. Molecular characterization and differential expression of cytokinin-responsive type-A response regulators in rice (Oryza sativa) BMC Plant Biol. 2006;6:1–11. doi: 10.1186/1471-2229-6-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jha Y, Subramanian R, Patel S. Combination of endophytic and rhizospheric plant growth promoting rhizobacteria in Oryza sativa shows higher accumulation of osmoprotectant against saline stress. Acta Physiol Plant. 2011;33:797–802. doi: 10.1007/s11738-010-0604-9. [DOI] [Google Scholar]
- Jiang W, Zhou S, Huang H, Song H, Zhang Q, Zhao Y. MERISTEM ACTIVITYLESS (MAL) is involved in root development through maintenance of meristem size in rice. Plant Mol Biol. 2020;104:499–511. doi: 10.1007/s11103-020-01053-4. [DOI] [PubMed] [Google Scholar]
- Jing H, Strader LC. Interplay of auxin and cytokinin in lateral root development. Int J Mol Sci. 2019;20:1–12. doi: 10.3390/ijms20030486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jing H, Yang X, Zhang J, Liu X, Zheng H, Dong G, Nian J, Feng J, Xia B, Qian Q. Peptidyl-prolyl isomerization targets rice Aux/IAAs for proteasomal degradation during auxin signalling. Nat Commun. 2015;6:1–10. doi: 10.1038/ncomms8395. [DOI] [PubMed] [Google Scholar]
- Kamoshita A, Chandra Babu R, Boopathi MN, Fukai S. Phenotypic and genotypic analysis of drought-resistance traits for development of rice cultivars adapted to rainfed environments. Field Crop Res. 2008;109:1–23. doi: 10.1016/j.fcr.2008.06.010. [DOI] [Google Scholar]
- Kandasamy S, Loganathan K, Muthuraj R, Duraisamy S, Seetharaman S, Thiruvengadam R, Ponnusamy B, Ramasamy S. Understanding the molecular basis of plant growth promotional effect of Pseudomonas fluorescens on rice through protein profiling. Prot Sci. 2009;7:1–8. doi: 10.1186/1477-5956-7-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khowaja FS, Norton GJ, Courtois B, Price AH. Improved resolution in the position of drought-related QTLs in a single mapping population of rice by meta-analysis. BMC Genomics. 2009;10:1–14. doi: 10.1186/1471-2164-10-276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kieber JJ, Schaller GE. Cytokinin signaling in plant development. Development. 2018;145:1–7. doi: 10.1242/dev.149344. [DOI] [PubMed] [Google Scholar]
- Kim J-Y, Kim HS, Lee SM, Park H-J, Lee S-H, Jang JS, Lee MH. Plant growth promoting and disease controlling activities of Pseudomonas geniculata ANG3, Exiguobacterium acetylicum ANG40 and Burkholderia stabilis ANG51 isolated from soil. Microbiol Biotechnol Lett. 2020;48:38–47. doi: 10.4014/mbl.1906.06002. [DOI] [Google Scholar]
- Kitomi Y, Hanzawa E, Kuya N, Inoue H, Hara N, Kawai S, Kanno N, Endo M, Sugimoto K, Yamazaki T, Sakamoto S. Root angle modifications by the DRO1 homolog improve rice yields in saline paddy fields. Proc Natl Acad Sci. 2020;117:21242–21250. doi: 10.1073/pnas.2005911117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein SP, Schneider HM, Perkins AC, Brown KM, Lynch JP. Multiple integrated root phenotypes are associated with improved drought tolerance. Plant Physiol. 2020;183:1011–1025. doi: 10.1104/pp.20.00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreuzer K, Adamczyk J, Iijima M, Wagner M, Scheu S, Bonkowski M. Grazing of a common species of soil protozoa (Acanthamoeba castellanii) affects rhizosphere bacterial community composition and root architecture of rice (Oryza sativa L.) Soil Biol Biochem. 2006;38:1665–1672. doi: 10.1016/j.soilbio.2005.11.027. [DOI] [Google Scholar]
- Kudo T, Makita N, Kojima M, Tokunaga H, Sakakibara H. Cytokinin activity of cis-zeatin and phenotypic alterations induced by overexpression of putative cis-zeatin-O-glucosyltransferase in rice. Plant Physiol. 2012;160:319–331. doi: 10.1104/pp.112.196733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurakawa T, Ueda N, Maekawa M, Kobayashi K, Kojima M, Nagato Y, Sakakibara H, Kyozuka J. Direct control of shoot meristem activity by a cytokinin-activating enzyme. Nature. 2007;445:652–655. doi: 10.1038/nature05504. [DOI] [PubMed] [Google Scholar]
- Lee HY, Chen Z, Zhang C, Yoon GM. Editing of the OsACS locus alters phosphate deficiency-induced adaptive responses in rice seedlings. J Exp Bot. 2019;70:1927–1940. doi: 10.1093/jxb/erz074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Zeng R, Liao H. Improving crop nutrient efficiency through root architecture modifications. J Integr Plant Biol. 2016;58:193–202. doi: 10.1111/jipb.12434. [DOI] [PubMed] [Google Scholar]
- Li X, Chen R, Chu Y, Huang J, Jin L, Wang G, Huang J. Overexpression of RCc3 improves root system architecture and enhances salt tolerance in rice. Plant Physiol Biochem. 2018;130:566–576. doi: 10.1016/j.plaphy.2018.08.008. [DOI] [PubMed] [Google Scholar]
- Li X, Zhou J, Xu R-S, Meng M-Y, Yu X, Dai C-C. Auxin, cytokinin, and ethylene involved in rice N availability improvement caused by endophyte Phomopsis liquidambari. J Plant Growth Regul. 2018;37:128–143. doi: 10.1007/s00344-017-9712-8. [DOI] [Google Scholar]
- Li S-M, Zheng H-X, Zhang X-S, Sui N. Cytokinins as central regulators during plant growth and stress response. Plant Cell Rep. 2020;2:271–282. doi: 10.1007/s00299-020-02612-1. [DOI] [PubMed] [Google Scholar]
- Li J, Yang Y, Chai M, Ren M, Yuan J, Yang W, Dong Y, Liu B, Jian Q, Wang S. Gibberellins modulate local auxin biosynthesis and polar auxin transport by negatively affecting flavonoid biosynthesis in the root tips of rice. Plant Sci. 2020;298:1–10. doi: 10.1016/j.plantsci.2020.110545. [DOI] [PubMed] [Google Scholar]
- Liang C, Li A, Yu H, Li W, Liang C, Guo S, Zhang R, Chu C. Melatonin regulates root architecture by modulating auxin response in rice. Front Plant Sci. 2017;8:134. doi: 10.3389/fpls.2017.00134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorbiecke R, Sauter M. Adventitious root growth and cell-cycle induction in deepwater rice. Plant Physiol. 1999;119:21–30. doi: 10.1104/pp.119.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucob-Agustin N, Kawai T, Takahashi-Nosaka M, Kano-Nakata M, Wainaina CM, Hasegawa T, Inari-Ikeda M, Sato M, Tsuji H, Yamauchi A, Inukai Y. WEG1, which encodes a cell wall hydroxyproline-rich glycoprotein, is essential for parental root elongation controlling lateral root formation in rice. Physiol Plant. 2020;169:214–227. doi: 10.1111/ppl.13063. [DOI] [PubMed] [Google Scholar]
- Lugtenberg B, Rozen DE, Kamilova F. Wars between microbes on roots and fruits. F1000Research. 2017;6:343–343. doi: 10.12688/f1000research.10696.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lv B, Tian H, Zhang F, Liu J, Lu S, Bai M, Li C, Ding Z. Brassinosteroids regulate root growth by controlling reactive oxygen species homeostasis and dual effect on ethylene synthesis in Arabidopsis. PLoS Genet. 2018;14:e1007144. doi: 10.1371/journal.pgen.1007144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch JP, Wojciechowski T. Opportunities and challenges in the subsoil: pathways to deeper rooted crops. J Exp Bot. 2015;66:2199–2210. doi: 10.1093/jxb/eru508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma N, Wang Y, Qiu S, Kang Z, Che S, Wang G, Huang J. Overexpression of OsEXPA8, a root-specific gene, improves rice growth and root system architecture by facilitating cell extension. PLoS ONE. 2013;8:1–10. doi: 10.1371/journal.pone.0075997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mai CD, Phung NT, To HT, Gonin M, Hoang GT, Nguyen KL, Do VN, Courtois B, Gantet P. Genes controlling root development in rice. Rice. 2014;7:1–11. doi: 10.1186/s12284-014-0030-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao C, He J, Liu L, Deng Q, Yao X, Liu C, Qiao Y, Li P, Ming F. OsNAC2 integrates auxin and cytokinin pathways to modulate rice root development. Plant Biotechnol J. 2020;18:429–442. doi: 10.1111/pbi.13209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurel C, Nacry P. Root architecture and hydraulics converge for acclimation to changing water availability. Nat Plants. 2020;6:744–749. doi: 10.1038/s41477-020-0684-5. [DOI] [PubMed] [Google Scholar]
- Mergemann H, Sauter M. Ethylene induces epidermal cell death at the site of adventitious root emergence in rice. Plant Physiol. 2000;124:609–614. doi: 10.1104/pp.124.2.609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miché L, Battistoni F, Gemmer S, Belghazi M, Reinhold-Hurek B. Upregulation of jasmonate-inducible defense proteins and differential colonization of roots of Oryza sativa cultivars with the endophyte Azoarcus sp. Mol Plant Microbe Interact. 2006;19:502–511. doi: 10.1094/MPMI-19-0502. [DOI] [PubMed] [Google Scholar]
- Mitra D, Be GS, Khoshru B, De Los SV, Belz C, Chaudhary P, Shahri FN, Djebaili R, Adeyemi NO, El-Ballat EM, El-Esawi MA. Impacts of arbuscular mycorrhizal fungi on rice growth, development, and stress management with a particular emphasis on strigolactone effects on root development. Commun Soil Sci Plant Anal. 2021;52:1591–1621. doi: 10.1080/00103624.2021.1892728. [DOI] [Google Scholar]
- Mohite B. Isolation and characterization of indole acetic acid (IAA) producing bacteria from rhizospheric soil and its effect on plant growth. J Soil Sci Plant Nutr. 2013;13:638–649. doi: 10.4067/S0718-95162013005000051. [DOI] [Google Scholar]
- Neogy A, Singh Z, Mushahary KKK, Yadav SR. Dynamic cytokinin signaling and function of auxin in cytokinin responsive domains during rice crown root development. Plant Cell Rep. 2020;12:1–9. doi: 10.1007/s00299-020-02618-9. [DOI] [PubMed] [Google Scholar]
- Ogura T, Goeschl C, Filiault D, Mirea M, Slovak R, Wolhrab B, Satbhai SB, Busch W. Root system depth in Arabidopsis is shaped by EXOCYST70A3 via the dynamic modulation of auxin transport. Cell. 2019;178:400–412. doi: 10.1016/j.cell.2019.06.021. [DOI] [PubMed] [Google Scholar]
- Ortíz-Castro R, Contreras-Cornejo HA, Macías-Rodríguez L, López-Bucio J (2009) The role of microbial signals in plant growth and development. Plant Signal Behav 4:701–712. 10.4161/psb.4.8.9047 [DOI] [PMC free article] [PubMed]
- Paez-Garcia A, Motes CM, Scheible WR, Chen R, Blancaflor EB, Monteros MJ. Root traits and phenotyping strategies for plant improvement. Plants. 2015;4:334–355. doi: 10.3390/plants4020334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasternak T, Groot EP, Kazantsev FV, Teale W, Omelyanchuk N, Kovrizhnykh V, Palme K, Mironova VV. Salicylic acid affects root meristem patterning via auxin distribution in a concentration-dependent manner. Plant Physiol. 2019;180:1725–1739. doi: 10.1104/pp.19.00130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen O, Sauter M, Colmer TD, Nakazono M. Regulation of root adaptive anatomical and morphological traits during low soil oxygen. New Phytol. 2021;229:42–49. doi: 10.1111/nph.16375. [DOI] [PubMed] [Google Scholar]
- Perrig D, Boiero M, Masciarelli O, Penna C, Ruiz O, Cassán F, Luna M. Plant-growth-promoting compounds produced by two agronomically important strains of Azospirillum brasilense, and implications for inoculant formulation. Appl Microbiol Biotechnol. 2007;75:1143–1150. doi: 10.1007/s00253-007-0909-9. [DOI] [PubMed] [Google Scholar]
- Phillips DA, Fox TC, King MD, Bhuvaneswari T, Teuber LR. Microbial products trigger amino acid exudation from plant roots. Plant Physiol. 2004;136:2887–2894. doi: 10.1104/pp.104.044222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plett DC, Ranathunge K, Melino VJ, Kuya N, Uga Y, Kronzucker HJ. The intersection of nitrogen nutrition and water use in plants: new paths toward improved crop productivity. J Exp Bot. 2020;71:4452–4468. doi: 10.1093/jxb/eraa049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qi Y, Wang S, Shen C, Zhang S, Chen Y, Xu Y, Liu Y, Wu Y, Jiang D. OsARF12, a transcription activator on auxin response gene, regulates root elongation and affects iron accumulation in rice (Oryza sativa) New Phytol. 2012;193:109–120. doi: 10.1111/j.1469-8137.2011.03910.x. [DOI] [PubMed] [Google Scholar]
- Qin H, Pandey BK, Li Y, Huang G, Wang J, Quan R, Zhou J, Zhou Y, Miao Y, Zhang D, Bennett MJ. Orchestration of ethylene and gibberellin signals determines primary root elongation in rice. Plant Cell. 2022;34:1273–1288. doi: 10.1093/plcell/koac008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rebouillat J, Dievart A, Verdeil J-L, Escoute J, Giese G, Breitler J-C, Gantet P, Espeout S, Guiderdoni E, Périn C. Molecular genetics of rice root development. Rice. 2009;2:15–34. doi: 10.1007/s12284-008-9016-5. [DOI] [Google Scholar]
- Remans R, Ramaekers L, Schelkens S, Hernandez G, Garcia A, Reyes JL, Mendez N, Toscano V, Mulling M, Galvez L. Effect of Rhizobium-Azospirillum coinoculation on nitrogen fixation and yield of two contrasting Phaseolus vulgaris L. genotypes cultivated across different environments in Cuba. Plant Soil. 2008;312:25–37. doi: 10.1186/s40068-017-0091-8. [DOI] [Google Scholar]
- Rondina ABL, dos Santos Sanzovo AW, Guimarães GS, Wendling JR, Nogueira MA, Hungria M. Changes in root morphological traits in soybean co-inoculated with Bradyrhizobium spp. and Azospirillum brasilense or treated with A. brasilense exudates. Biol Fertil Soils. 2020;56:537–549. doi: 10.1007/s00374-020-01453-0. [DOI] [Google Scholar]
- Sannazzaro AI, Echeverría M, Albertó EO, Ruiz OA, Menéndez AB. Modulation of polyamine balance in Lotus glaber by salinity and arbuscular mycorrhiza. Plant Physiol Biochem. 2007;45:39–46. doi: 10.1016/j.plaphy.2006.12.008. [DOI] [PubMed] [Google Scholar]
- Sekar C, Prasad N, Sundaram M (2000) Enhancement of polygalacturonase activity during auxin induced para nodulation and endorhizosphere colonization of Azospirillum in rice roots. Indian J Exp Bot 38:80–83. http://nopr.niscair.res.in/handle/123456789/23880 [PubMed]
- Shahzad R, Khan AL, Bilal S, Waqas M, Kang S-M, Lee I-J. Inoculation of abscisic acid-producing endophytic bacteria enhances salinity stress tolerance in Oryza sativa. Environ Exp Bot. 2017;136:68–77. doi: 10.1016/j.envexpbot.2017.01.010. [DOI] [Google Scholar]
- Shao Y, Lehner KR, Zhou H, Taylor I, Zhu M, Mao C, Benfey PN. VAP-RELATED SUPPRESSORS OF TOO MANY MOUTHS (VST) family proteins are regulators of root system architecture. Plant Physiol. 2021;185:457–468. doi: 10.1093/plphys/kiaa036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen C, Yue R, Sun T, Zhang L, Yang Y, Wang H. OsARF16, a transcription factor regulating auxin redistribution, is required for iron deficiency response in rice (Oryza sativa L.) Plant Sci. 2015;231:148–158. doi: 10.1016/j.plantsci.2014.12.003. [DOI] [PubMed] [Google Scholar]
- Šimášková M, O’Brien JA, Khan M, Van Noorden G, Ötvös K, Vieten A, De Clercq I, Van Haperen JMA, Cuesta C, Hoyerová K, Vanneste S. Cytokinin response factors regulate PIN-FORMED auxin transporters. Nat Commun. 2015;6:1–11. doi: 10.1038/ncomms9717. [DOI] [PubMed] [Google Scholar]
- Singh V, van Oosterom EJ, Jordan DR, Messina CD, Cooper M, Hammer GL. Morphological and architectural development of root systems in sorghum and maize. Plant Soil. 2010;333:287–299. doi: 10.1007/s11104-010-0343-0. [DOI] [Google Scholar]
- Singh AP, Pandey BK, Mehra P, Heitz T, Giri J. OsJAZ9 overexpression modulates jasmonic acid biosynthesis and potassium deficiency responses in rice. Plant Mol Biol. 2020;104:397–410. doi: 10.1007/s11103-020-01047-2. [DOI] [PubMed] [Google Scholar]
- Singh BK, Ramkumar MK, Dalal M, Singh A, Solanke AU, Singh NK, Sevanthi AM. Allele mining for a drought responsive gene DRO1 determining root growth angle in donors of drought tolerance in rice (Oryza sativa L.) Physiol Mol Biol Plants. 2021;27:523–534. doi: 10.1007/s12298-021-00950-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith S, De Smet I. Root system architecture: insights from Arabidopsis and cereal crops. Philos Trans R Soc B Biol Sci. 2012;367:1441–1452. doi: 10.1098/rstb.2011.0234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sreevidya V, Hernandez-Oane RJ, Gyaneshwar P, Lara-Flores M, Ladha JK, Reddy PM. Changes in auxin distribution patterns during lateral root development in rice. Plant Sci. 2010;178:531–538. doi: 10.1016/j.plantsci.2010.03.004. [DOI] [Google Scholar]
- Steffens B, Rasmussen A. The physiology of adventitious roots. Plant Physiol. 2016;170:603–617. doi: 10.1104/pp.15.01360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steffens B, Sauter M. Epidermal cell death in rice is regulated by ethylene, gibberellin, and abscisic acid. Plant Physiol. 2005;139:713–721. doi: 10.1104/pp.105.064469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steffens B, Wang J, Sauter M. Interactions between ethylene, gibberellin and abscisic acid regulate emergence and growth rate of adventitious roots in deepwater rice. Planta. 2006;223:604–612. doi: 10.1007/s00425-005-0111-1. [DOI] [PubMed] [Google Scholar]
- Sulieman S, Schulze J. Phloem-derived γ-aminobutyric acid (GABA) is involved in upregulating nodule N2 fixation efficiency in the model legume Medicago truncatula. Plant Cell Environ. 2010;33:2162–2172. doi: 10.1111/j.1365-3040.2010.02214.x. [DOI] [PubMed] [Google Scholar]
- Sun J, Xu Y, Ye S, Jiang H, Chen Q, Liu F, Zhou W, Chen R, Li X, Tietz O, Wu X. Arabidopsis ASA1 is important for jasmonate-mediated regulation of auxin biosynthesis and transport during lateral root formation. Plant Cell. 2009;21:1495–1511. doi: 10.1105/tpc.108.064303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki G, Lucob-Agustin N, Kashihara K, Fujii Y, Inukai Y, Gomi K. Rice MEDIATOR25, OsMED25, is an essential subunit for jasmonate-mediated root development and OsMYC2-mediated leaf senescence. Plant Sci. 2021;306:110853. doi: 10.1016/j.plantsci.2021.110853. [DOI] [PubMed] [Google Scholar]
- Tfaily MM, Cooper WT, Kostka JE, Chanton PR, Schadt CW, Hanson PJ, Iversen CM, Chanton JP. Organic matter transformation in the peat column at Marcell Experimental Forest: humification and vertical stratification. J Geophys Res Biogeosci. 2014;119:661–675. doi: 10.1002/2013JG002492. [DOI] [Google Scholar]
- Timmusk S, Nicander B, Granhall U, Tillberg E. Cytokinin production by Paenibacillus polymyxa. Soil Biol Biochem. 1999;31:1847–1852. doi: 10.1016/s0038-0717(99)00113-3. [DOI] [Google Scholar]
- Tiryaki I, Staswick PE. An Arabidopsis mutant defective in jasmonate response is allelic to the auxin-signaling mutant axr1. Plant Physiol. 2002;130:887–894. doi: 10.1104/pp.005272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uga Y, Sugimoto K, Ogawa S, Rane J, Ishitani M, Hara N, Kitomi Y, Inukai Y, Ono K, Kanno N. Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nat Genet. 2013;45:1097–1102. doi: 10.1038/ng.2725. [DOI] [PubMed] [Google Scholar]
- Vacheron J, Desbrosses G, Bouffaud M-L, Touraine B, Moënne-Loccoz Y, Muller D, Legendre L, Wisniewski-Dyé F, Prigent-Combaret C. Plant growth-promoting rhizobacteria and root system functioning. Front Plant Sci. 2013;4:19. doi: 10.3389/fpls.2013.00356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valette M, Rey M, Doré J, Gerin F, Wisniewski-Dyé F. Identification of a small set of genes commonly regulated in rice roots in response to beneficial rhizobacteria. Physiol Mol Biol Plants. 2020;26:2537–2551. doi: 10.1007/s12298-020-00911-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Wees SC, Van der Ent S, Pieterse CM. Plant immune responses triggered by beneficial microbes. Curr Opin Plant Biol. 2008;11:443–448. doi: 10.1016/j.pbi.2008.05.005. [DOI] [PubMed] [Google Scholar]
- Vargas L, de Carvalho TLG, Ferreira PCG, Baldani VLD, Baldani JI, Hemerly AS. Early responses of rice (Oryza sativa L.) seedlings to inoculation with beneficial diazotrophic bacteria are dependent on plant and bacterial genotypes. Plant Soil. 2012;356:127–137. doi: 10.1007/s11104-012-1274-8. [DOI] [Google Scholar]
- Vázquez-Glaría A, Eichler-Löbermann B, Loiret FG, Ortega E, Kavka M. Root-system architectures of two Cuban rice cultivars with salt stress at early development stages. Plants. 2021;10:1194. doi: 10.3390/plants10061194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venturi V, Keel C. Signaling in the rhizosphere. Trends Plant Sci. 2016;21:187–198. doi: 10.1016/j.tplants.2016.01.005. [DOI] [PubMed] [Google Scholar]
- Verma PK, Verma S, Tripathi RD, Chakrabarty D. A rice glutaredoxin regulate the expression of aquaporin genes and modulate root responses to provide arsenic tolerance. Ecotoxicol Environ Saf. 2020;195:1–11. doi: 10.1016/j.ecoenv.2020.110471. [DOI] [PubMed] [Google Scholar]
- Verma PK, Verma S, Tripathi RD, Pandey N, Chakrabarty D. CC-type glutaredoxin, OsGrx_C7 plays a crucial role in enhancing protection against salt stress in rice. J Biotechnol. 2021;329:192–203. doi: 10.1016/j.jbiotec.2021.02.008. [DOI] [PubMed] [Google Scholar]
- Vives-Peris V, de Ollas C, Gómez-Cadenas A, Pérez-Clemente RM. Root exudates: from plant to rhizosphere and beyond. Plant Cell Rep. 2019;39:3–17. doi: 10.1007/s00299-019-02447-5. [DOI] [PubMed] [Google Scholar]
- Wang JW, Wang LJ, Mao YB, Cai WJ, Xue HW, Chen XY. Control of root cap formation by microRNA-targeted auxin response factors in Arabidopsis. Plant Cell. 2005;17:2204–2216. doi: 10.1105/tpc.105.033076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Sun R, Cao Y, Pei W, Sun Y, Zhou H, Wu X, Zhang F, Luo L, Shen Q. OsSIZ1, a SUMO E3 ligase gene, is involved in the regulation of the responses to phosphate and nitrogen in rice. Plant Cell Physiol. 2015;56:2381–2395. doi: 10.1093/pcp/pcv162. [DOI] [PubMed] [Google Scholar]
- Wang T, Li C, Wu Z, Jia Y, Wang H, Sun S, Mao C, Wang X. Abscisic acid regulates auxin homeostasis in rice root tips to promote root hair elongation. Front Plant Sci. 2017;8:1121. doi: 10.3389/fpls.2017.01121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Guo M, Li Y, Ruan W, Mo X, Wu Z, Sturrock CJ, Yu H, Lu C, Peng J. LARGE ROOT ANGLE1, encoding OsPIN2, is involved in root system architecture in rice. J Exp Bot. 2018;69:385–397. doi: 10.1093/jxb/erx427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang P, Xu X, Tang Z, Zhang W, Huang XY, Zhao F-J. OsWRKY28 regulates phosphate and arsenate accumulation, root system architecture and fertility in rice. Front Plant Sci. 2018;9:1–13. doi: 10.3389/fpls.2018.01330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M, Qiao J, Yu C, Chen H, Sun C, Huang L, Li C, Geisler M, Qian Q, Jiang DA, Qi Y. The auxin influx carrier, OsAUX3, regulates rice root development and responses to aluminium stress. Plant, Cell Environ. 2019;42:1125–1138. doi: 10.1111/pce.13478. [DOI] [PubMed] [Google Scholar]
- Wang C, Li X, Caragea D, Bheemanahallia R, Jagadish SK. Root anatomy based on root cross-section image analysis with deep learning. Comput Electron Agric. 2020;175:1–13. doi: 10.1016/j.compag.2020.105549. [DOI] [Google Scholar]
- Wang R, Wang H-L, Tang R-P, Sun M-Y, Chen T-M, Duan X-C, Lu X-F, Liu D, Shi X-C, Laborda P. Pseudomonas putida represses JA-and SA-mediated defense pathways in rice and promotes an alternative defense mechanism possibly through ABA signaling. Plants. 2020;9:1–14. doi: 10.3390/plants9121641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weller DM, Landa BB, Mavrodi OV, Schroeder KL, De La Fuente L, Blouin Bankhead S, Allende Molar R, Bonsall RF, Mavrodi DV, Thomashow LS. Role of 2,4-diacetylphloroglucinol-producing fluorescent Pseudomonas spp. in the defense of plant roots. Plant Biol. 2007;9:4–20. doi: 10.1055/s-2006-924473. [DOI] [PubMed] [Google Scholar]
- Wing RA, Purugganan MD, Zhang Q. The rice genome revolution: from an ancient grain to Green Super Rice. Nat Rev Genet. 2018;19:505–517. doi: 10.1038/s41576-018-0024-z. [DOI] [PubMed] [Google Scholar]
- Wu Q, Peng X, Yang M, Zhang W, Dazzo FB, Uphoff N, Jing Y, Shen S. Rhizobia promote the growth of rice shoots by targeting cell signaling, division and expansion. Plant Mol Biol. 2018;97:507–523. doi: 10.1007/s11103-018-0756-3. [DOI] [PubMed] [Google Scholar]
- Xu M, Zhu L, Shou H, Wu P. A PIN1 family gene, OsPIN1, involved in auxin-dependent adventitious root emergence and tillering in rice. Plant Cell Physiol. 2005;46:1674–1681. doi: 10.1093/pcp/pci183. [DOI] [PubMed] [Google Scholar]
- Xu K, Xu X, Fukao T, Canlas P, Maghirang-Rodriguez R, Heuer S, Ismail AM, Bailey-Serres J, Ronald PC, Mackill DJ. Sub1A is an ethylene-response-factor-like gene that confers submergence tolerance to rice. Nature. 2006;442:705–708. doi: 10.1038/nature04920. [DOI] [PubMed] [Google Scholar]
- Xu Y, Ge Y, Song J, Rensing C. Assembly of root-associated microbial community of typical rice cultivars in different soil types. Biol Fertil Soils. 2020;56:249–260. doi: 10.1007/s00374-019-01406-2. [DOI] [Google Scholar]
- Xuan YH, Kumar V, Han X, Kim SH, Jeong JH, Kim CM, Gao Y, Han CD (2019) CBL-INTERACTING PROTEIN KINASE 9 regulates ammonium-dependent root growth downstream of IDD10 in rice (Oryza sativa). Ann Bot 124:947–960. 10.1093/aob/mcy242 [DOI] [PMC free article] [PubMed]
- Yamaji N, Huang CF, Nagao S, Yano M, Sato Y, Nagamura Y, Ma JF. A zinc finger transcription factor ART1 regulates multiple genes implicated in aluminum tolerance in rice. Plant Cell. 2009;21:3339–3349. doi: 10.1093/pcp/pcw073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang A, Zhang W-H. A small GTPase, OsRab6a, is involved in the regulation of iron homeostasis in rice. Plant Cell Physiol. 2016;57:1271–1280. doi: 10.1093/pcp/pcw073. [DOI] [PubMed] [Google Scholar]
- Yang WT, Baek D, Yun D-J, Hwang WH, Park DS, Nam MH, Chung ES, Chung YS, Yi YB, Kim DH. Overexpression of OsMYB4P, an R2R3-type MYB transcriptional activator, increases phosphate acquisition in rice. Plant Physiol Biochem. 2014;80:259–267. doi: 10.1016/j.plaphy.2014.02.024. [DOI] [PubMed] [Google Scholar]
- Yang C, Li W, Cao J, Meng F, Yu Y, Huang J, Jiang L, Liu M, Zhang Z, Chen X. Activation of ethylene signaling pathways enhances disease resistance by regulating ROS and phytoalexin production in rice. Plant J. 2017;89:338–353. doi: 10.1111/tpj.13388. [DOI] [PubMed] [Google Scholar]
- Yano K, Yoshida S, Müller J, Singh S, Banba M, Vickers K, Markmann K, White C, Schuller B, Sato S. CYCLOPS, a mediator of symbiotic intracellular accommodation. Proc Natl Acad Sci. 2008;105:20540–20545. doi: 10.1073/pnas.0806858105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi K, Wu Z, Zhou J, Du L, Guo L, Wu Y, Wu P. OsPTF1, a novel transcription factor involved in tolerance to phosphate starvation in rice. Plant Physiol. 2005;138:2087–2096. doi: 10.1104/pp.105.063115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon J, Cho LH, Yang W, Pasriga R, Wu Y, Hong WJ, Bureau C, Wi SJ, Zhang T, Wang R, Zhang D. Homeobox transcription factor OsZHD2 promotes root meristem activity in rice by inducing ethylene biosynthesis. J Exp Bot. 2020;71:5348–5364. doi: 10.1093/jxb/eraa209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu C, Sun C, Shen C, Wang S, Liu F, Liu Y, Chen Y, Li C, Qian Q, Aryal B, Geisler M. The auxin transporter, Os AUX 1, is involved in primary root and root hair elongation and in Cd stress responses in rice (Oryza sativa L.) Plant J. 2015;83:818–830. doi: 10.1111/tpj.12929. [DOI] [PubMed] [Google Scholar]
- Zaheer MS, Ali HH, Iqbal MA, Erinle KO, Javed T, Iqbal J, Hashmi MIU, Mumtaz MZ, Salama EA, Kalaji HM, Wróbel J. Cytokinin production by Azospirillum brasilense contributes to increase in growth, yield, antioxidant, and physiological systems of wheat (Triticum aestivum L.) Front Microbiol. 2022;13:886041. doi: 10.3389/fmicb.2022.886041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, Kim MS, Krishnamachari V, Payton P, Sun Y, Grimson M, Farag MA, Ryu CM, Allen R, Melo IS, Paré PW. Rhizobacterial volatile emissions regulate auxin homeostasis and cell expansion in Arabidopsis. Planta. 2007;226:839–851. doi: 10.1007/s00425-007-0530-2. [DOI] [PubMed] [Google Scholar]
- Zhang R, Vivanco JM, Shen Q. The unseen rhizosphere root-soil-microbe interactions for crop production. Curr Opin Microbiol. 2017;37:8–14. doi: 10.1016/j.mib.2017.03.008. [DOI] [PubMed] [Google Scholar]
- Zhang G, Xu N, Chen H, Wang G, Huang J. OsMADS25 regulates root system development via auxin signalling in rice. Plant J. 2018;95:1004–1022. doi: 10.1111/tpj.14007. [DOI] [PubMed] [Google Scholar]
- Zhang T, Li R, Xing J, Yan L, Wang R, Zhao Y. The YUCCA-auxin-WOX11 module controls crown root development in rice. Front Plant Sci. 2018;9:523. doi: 10.3389/fpls.2018.00523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D, Lyu Y, Li H, Tang X, Hu R, Rengel Z, Zhang F, Whalley WR, Davies WJ, Cahill JF., Jr Neighbouring plants modify maize root foraging for phosphorus: coupling nutrients and neighbours for improved nutrient use efficiency. New Phytol. 2020;226:244–253. doi: 10.1111/nph.16206. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Wang X, Luo Y, Zhang L, Yao Y, Han L, Chen Z, Wang L, Li Y. OsABA8ox2, an ABA catabolic gene, suppress root elongation of rice seedlings and contributes to drought response. Crop J. 2020;8:480–491. doi: 10.1016/j.cj.2019.08.006. [DOI] [Google Scholar]
- Zhao Y, Hu Y, Dai M, Huang L, Zhou D-X. The WUSCHEL-related homeobox gene WOX11 is required to activate shoot-borne crown root development in rice. Plant Cell. 2009;21:736–748. doi: 10.1105/tpc.108.061655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H, Duan KX, Ma B, Yin CC, Hu Y, Tao JJ, Huang YH, Cao WQ, Chen H, Yang C, Zhang ZG. Histidine kinase MHZ1/OsHK1 interacts with ethylene receptors to regulate root growth in rice. Nat Commun. 2020;11:1–13. doi: 10.1038/s41467-020-14313-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y, Dong G, Tao Y, Chen C, Yang B, Wu Y, Yang Z, Liang G, Wang B, Wang Y. Mapping quantitative trait loci associated with toot traits using sequencing-based genotyping chromosome segment substitution lines derived from 9311 and Nipponbare in Rice (Oryza sativa L.) PLoS ONE. 2016;11:1–13. doi: 10.1371/journal.pone.0151796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu M, Hu Y, Tong A, Yan B, Lv Y, Wang S, Ma W, Cui Z, Wang X. LAZY1 controls tiller angle and shoot gravitropism by regulating the expression of auxin transporters and signaling factors in rice. Plant Cell Physiol. 2020;61:2111–2125. doi: 10.1093/pcp/pcaa131. [DOI] [PubMed] [Google Scholar]
- Zhu Q, Tang MJ, Yang Y, Sun K, Tian LS, Lu F, Hao AY, Dai CC. Endophytic fungus Phomopsis liquidambaris B3 induces rice resistance to RSRD caused by Fusarium proliferatum and promotes plant growth. J Sci Food Agric. 2021;101:4059–4075. doi: 10.1002/jsfa.11042. [DOI] [PubMed] [Google Scholar]
- Zou Y, Liu X, Wang Q, Chen Y, Liu C, Qiu Y, Zhang W. OsRPK1, a novel leucine-rich repeat receptor-like kinase, negatively regulates polar auxin transport and root development in rice. Biochim Biophys Acta (BBA) Gen Subj. 2014;1840:1676–1685. doi: 10.1016/j.bbagen.2014.01.003. [DOI] [PubMed] [Google Scholar]
- Zou H, Wenwen Y, Zang G, Kang Z, Zhang Z, Huang J, Wang G. OsEXPB2, a β-expansin gene, is involved in rice root system architecture. Mol Breed. 2015;35:1–14. doi: 10.1007/s11032-015-0203-y. [DOI] [Google Scholar]