Abstract
Environmental insults impair human health around the world. Contaminated air, water, soil and food, occupational and household settings expose humans of all ages to a plethora of chemicals and environmental stressors. We propose eight hallmarks of environmental insults which jointly underpin the damaging impact of environmental exposures during the life-span. Specifically, they include oxidative stress and inflammation, genomic alterations and mutations, epigenetic alterations, mitochondrial dysfunction, endocrine disruption, altered intercellular communication, altered microbiome communities, and impaired nervous system function. They provide a framework to understand why complex mixtures of environmental exposures induce severe health effects even at relatively modest concentrations.
Introduction
The proportion of older individuals is increasing worldwide. Today one in eleven people in the world is over 65 years of age (UN, 2019). The World Health Organization has emphasized that aging is characterized by the accumulation of modifiable risks for chronic disease (WHO, 2005). Environmental exposures contribute significantly to these modifiable risk factors jointly with life-style related risk factors such as unhealthy diet, sedentary life-style, tobacco smoking and alcohol drinking. Recent burden of disease studies estimate that preventable deaths due to environmental exposures range between 9 million (Landrigan et al., 2018) and 12.6 million (Prüss-Ustün, 2016). These estimates quantify the health impacts from a number of environmental exposures including particulate matter air pollution and ozone, water pollution, occupational exposures to carcinogens and particulate matter, contaminated soil by heavy metals, chemicals and lead. Ambient air pollution, chemical pollution, and soil pollution are on the rise, with the most marked increases in rapidly developing and industrializing low and middle-income countries (Landrigan et al., 2018). Since 1950 more than 140 000 new chemicals and pesticides have been synthesized and often their impact on health is largely unknown. Environmental pollution as described above is typically higher in urban than in rural areas. Moreover, urban areas may expose their inhabitants to heat, noise and light pollution (Nieuwenhuijsen, 2020).
Environmental exposures impact the human body throughout life, from conception to old age. They govern gene expression, train and shape the immune system, trigger many physiological responses and, most importantly, determine wellbeing and disease. Barrier organs, such as the lung, the skin or the gut are directly impacted by environmental exposures and have evolved over time to cope with potential insults. Immune function in these barrier organs is in many instances the first line of defense. The microbiome of the barrier organs is considered an integral part of the barrier function, but can also be impacted by exogenous influences. Our sensory systems receive environmental exposures and elicit neurological responses to adapt to changing environmental conditions. However, their impacts are not limited to the immediate and local responses. Chemicals and nanoparticles can potentially impact organs without a direct barrier function. In addition, local reactions may induce secondary, systemic responses, activate the immune system beyond the immediate location of the insult, activate metabolic functions, alter organ-to-organ signaling and autonomic nervous system control. Most of these responses are geared at maintaining the homeostasis of cell and organ functions. Most environmental exposures are toxic at low and high levels. However, some environmental chemicals such as for example essential trace elements are toxic only at high doses.
Define the hallmarks of environmental insults
We propose eight hallmarks of environmental insults (Figure 1) which describe the cellular and molecular processes involved in essential cellular mechanisms and activities that are capable of linking environmental exposures to chronic diseases such as cancer, respiratory, cardiovascular and metabolic diseases and diseases of the nervous system. Ideally, a hallmark shall fulfil the following criteria: (1) it should be clearly altered in response to environmental exposures in human studies, (2) it should be experimentally verified that environmental exposures have the capacity to alter these characteristics and (3) there should be evidence from human studies that clearly link the alterations to environmentally induced diseases.
Figure 1: Hallmarks of environmental insults.
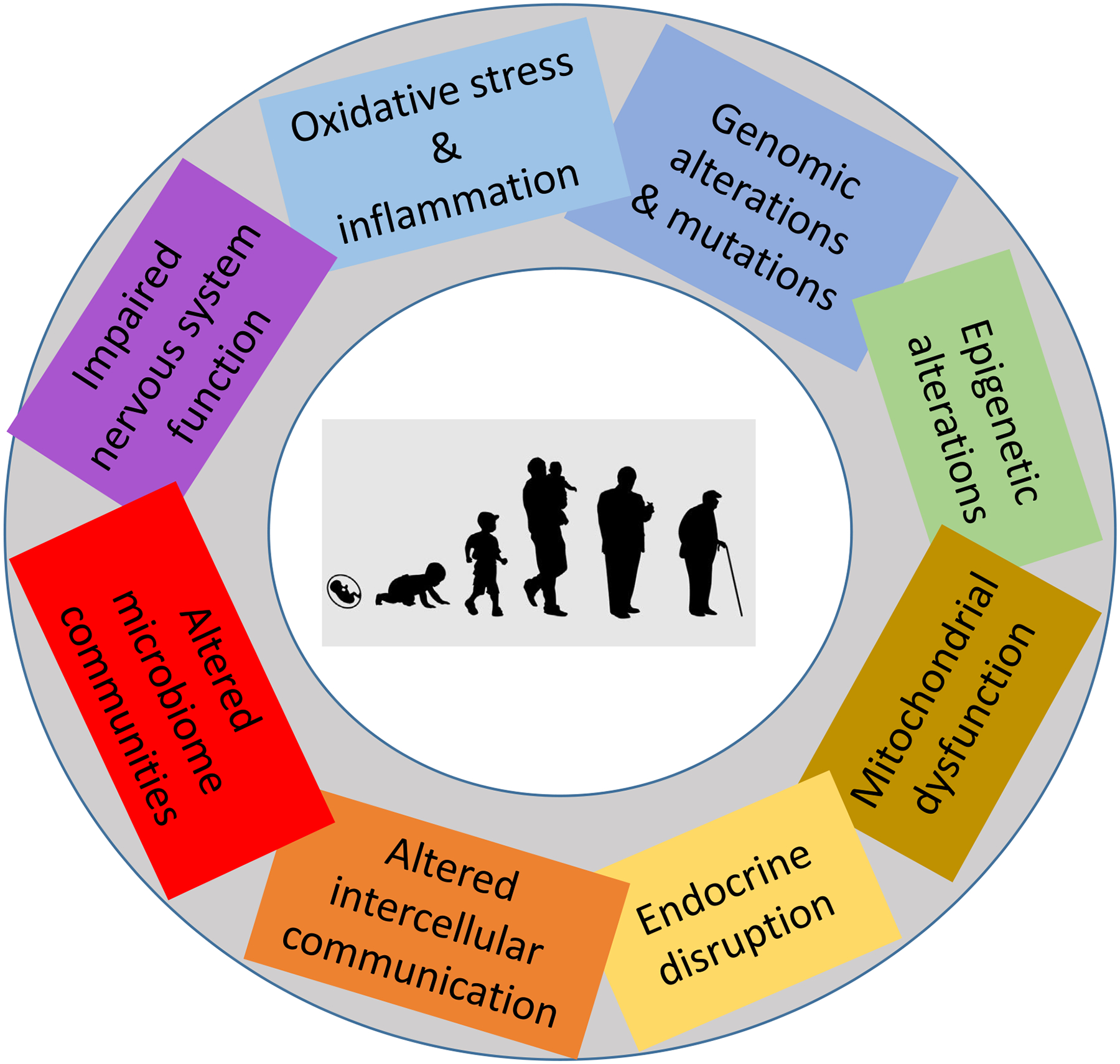
The scheme enumerates the hallmarks of environmental insults described in this review: Oxidative stress and inflammation, genomic alterations and mutations, epigenetic alterations, mitochondrial dysfunction, endocrine disruption, altered intercellular communication, altered microbiome communities, and impaired nervous system function.
In the following section we highlight the role of the eight hallmarks, to provide a framework which addresses the complexity of the issue and enables understanding of interactions of environmental insults on a cellular and organ level.
Oxidative stress and inflammation
Reactive oxygen species (ROS) are part of the normal aerobic life. They have a central role in redox signaling and at physiological levels they are essential for maintain cellular function (Sies and Jones, 2020). The ability to respond to oxidative stress has been a central determinant of ageing and longevity (Finkel and Holbrook, 2000) and is implicated in many diseases in humans including cancer, atherosclerosis and related cardiovascular diseases, respiratory diseases and neurological diseases (Sies and Jones, 2020).
At physiological levels, hydrogen peroxide (H2O2) and superoxide anion radicals are generated under the control of growth factors and more than 40 cytokines. Endogenous ROS activate signaling cascades which engage in metabolic regulation and stress responses that support cellular adaptation to changing environments. In order to maintain these highly sophisticated regulatory pathways, cells have developed enzyme systems to scavenge any excess ROS inside and outside the cell. H2O2 concentrations above 100 nM lead to unspecific oxidation of proteins and altered response patterns and cause reversible and irreversible damage of biomolecules (Sies and Jones, 2020). Many proteins are highly sensitive to oxidation and transmit signals activating defense mechanisms. Especially the amino acid residues cysteine and methionine of proteins are highly sensitive to oxidation and their regulation plays a key role in aging (Stadtman et al., 2005). Furthermore, an oxidative stress induced, pro-inflammatory phenotype progressively increases with age in humans and underlies the risks of a range of age-related chronic disease (Salminen et al., 2012).
Environmental exposures deplete antioxidant defense and induce inflammation and cell death
Many environmental exposures can induce oxidative stress resulting in inflammation. One of the key transcription factors responsible for mounting an antioxidant defense in response to environmental exposures is the nuclear factor erythroid 2-related factor 2 (NRF2). In the inactive state, NRF2 is bound to the Kelch-like ECH-associated protein 1 (KEAP1) in the cytoplasm. KEAP1 harbors several cysteine residues that can be oxidized by oxidative and electrophilic stressors resulting in conformational changes (Yamamoto et al., 2018). As a consequence of the oxidation of the residues, the stability of NRF2 is increased and the transition to the nucleus is enabled. NRF2 binds to the antioxidants’ response element and thereby is a transcription factor with a truly pleiotropic function (Rojo de la Vega et al., 2018). Important target genes under NRF2 regulation are part of the xenobiotic metabolism and excretion, glutathione synthesis, antioxidant systems, iron regulation, autophagy, proteasome activity, DNA repair, fatty acid oxidation and mitochondrial physiology (Rojo de la Vega et al., 2018). Thereby, an effective defense against ROS induced by heavy metals such as lead and arsenic, transition metals such as copper and nickel or detoxification of poly-aromatic hydrocarbons such as benz(a)pyren is mounted. It is important to note that through NRF2 activation also tumor-promoting effects are elicited (Rojo de la Vega et al., 2018). The transcriptional factor κB (NF-κB) serves as a master switch of inflammation in response to extensive oxidative stress (Sies and Jones, 2020). Transcription of cytokines, chemokines, antimicrobial peptides and anti-apoptotic proteins is induced (Mudway et al., 2020). Similarly, the NF-κB pathways play a major role in activating and controlling the function of immune cells essential for innate immune responses such as macrophages (Dorrington and Fraser, 2019). Taken together, Figure 2 summarizes the continuum from tolerance of ROS to cell death. Tox21, a toxicology screening program that has tested biological responses to more than 9,000 chemicals, showed that numerous organic chemicals, including phthalates, bisphenols, polycyclic aromatic hydrocarbons (PAHs) and flame retardants, alter molecular signaling pathways related to inflammation and inflammation-related diseases (Kleinstreuer et al., 2014). Continued and persistent environmental exposures are considered to induce apoptosis and necroptosis (Ryter et al., 2007). Fine particulate matter, which is a mixture of transition metals, polyaromatic hydrocarbons, soot and other oxygen radical producing substances (Ghio et al., 2012) contributes through these pathways to the loss of a functional lung epithelium and subsequent chronic obstructive lung disease (Mudway et al., 2020). Similarly, chemicals or nanoparticles reaching the blood stream may enhance ROS-induced formation of atherosclerotic plaques (Brook et al., 2010). Given that environmental contaminants are also capable of lipid peroxidation, the newly discovered mechanism of cell death induction, called ferroptosis (Conrad and Pratt, 2019) may also add to cell death and organ damage as a response to environmental insults.
Figure 2: Role of reactive oxygen species as agents for exhibiting environmental insults.
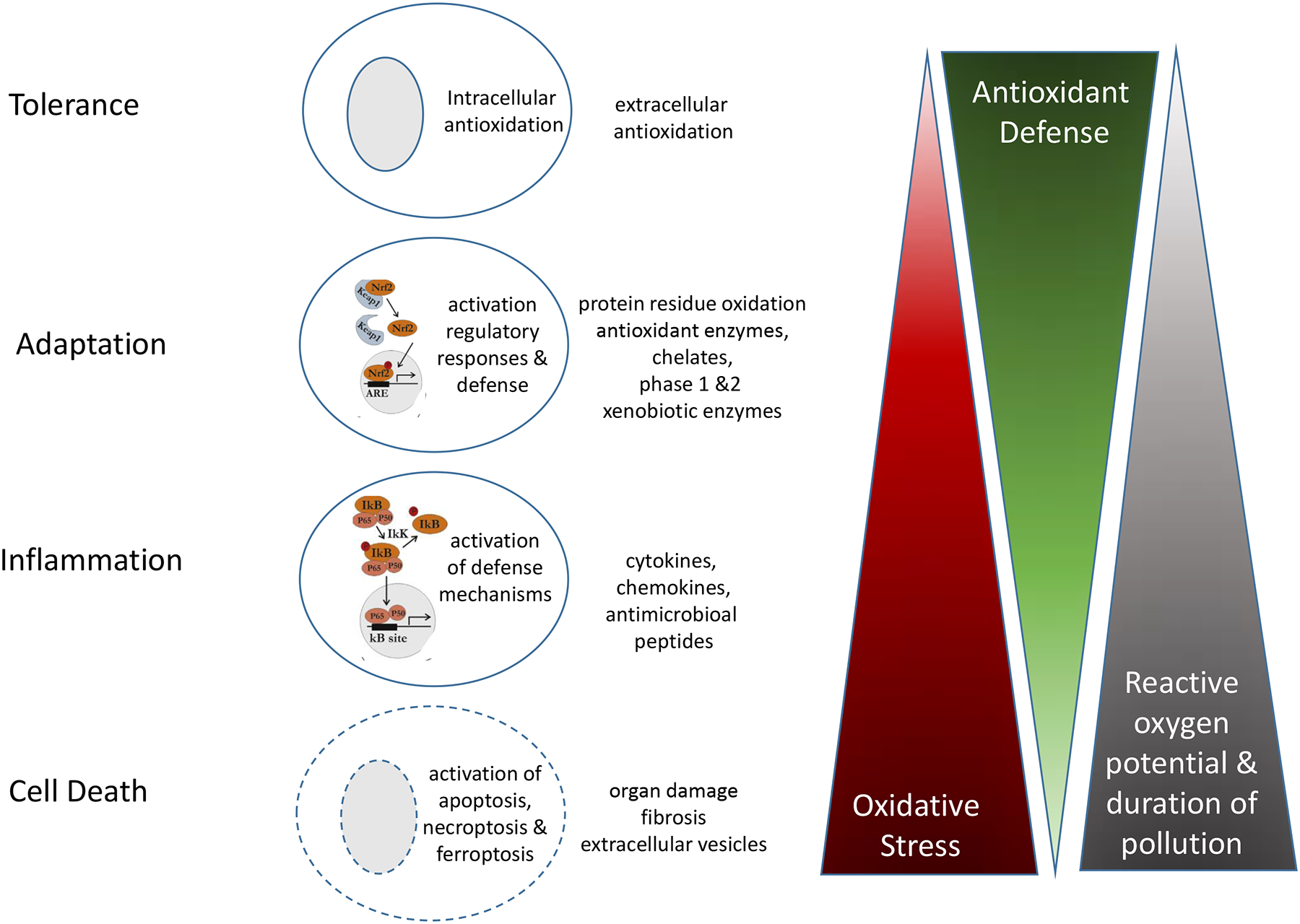
Environmental exposures challenge the cells based on their reactive oxygen potential, their concentrations, and the duration of exposures. While the cells are able to tolerate certain exposures, others elicit adaptation or inflammatory responses. Overwhelming oxidative stress results in cell death and organ damage. The activation of the NRF2 and NF-κB pathways activated by particulate matter air pollution in lung epithelial cells (Mudway et al., 2020) are depicted as examples.
In summary, ROS are generated by many environmental exposures. They link them to diseases at the barrier organs and mediate the systemic impacts of environmental exposures. As part of physiological signaling cascades, intracellular and extracellular ROS are tightly regulated. In response to environmental exposures, a continuum is exhibited ranging from tolerance towards ROS via inflammation to ROS-induced cell death and organ damage.
Genomic alterations and mutations
Somatic mutations, defined as change in the DNA sequence of a somatic cell after conception accumulate with age. These mutations are typically considered harmful and their increased load is seen as a deterioration of the genetic material linked to aging and age-related diseases (Balmain, 2020). An example is the accumulation of somatic mutations in hematopoietic stem cells connecting aging and inflammation to cardiovascular disease (Jaiswal and Libby, 2020). Next-generation sequencing has shown that the accumulation of age-related somatic mutations is more prevalent in normal human tissues than previously assumed. Normal cells in the esophageal epithelium already carry several hundred mutations per cell in younger individuals, and this number may rise to more than 2,000 mutations per cell by 60 years of age (Martincorena et al., 2018). These somatic mutations may persist and accumulate during our lifetime because a full repair of DNA lesions may be an energetically unfavorable investment for human cells, which may hence tolerate the accumulation of somatic mutations in aging tissues (Balmain, 2020).
Somatic mutations induced by environmental exposures affect diseases beyond carcinogenesis
Mutagenesis studies have long identified a host of environmental chemicals that are mutagens. Recently, an extensive catalog of mutational signatures from pluripotent stem cells exposed to 79 known or suspected environmental mutagens identified a variety of DNA sequence alterations, including signatures similar to those found in human tumors, double-substitution signatures, and short insertion / deletion signatures (Kucab et al., 2019). This study suggested that DNA damage induced by environmental mutagens can be resolved by various repair and/or replicative pathways, resulting in an assortment of signatures even for a single chemical. However, the extent to which environmental exposures contribute to generating mutations through human aging and whether such mutations mediate the effects of environmental exposures among older individuals is only partially understood. Indeed, few studies have examined the effects of environmental exposures in large-scale human populations. Far fewer have examined them within large, diverse populations with repeated measures of environmental exposures and DNA sequencing over time. The growing availability of next-generation sequencing data on large scale cohorts provides untapped opportunities for characterizing somatic mutations in relation to environmental exposures and their potential roles for disease development throughout the lifespan.
In summary, somatic mutations become more frequent with age and deteriorations of the genetic material are associated with a variety of diseases. Environmental exposures contribute to this burden and support the link of environmental exposures with aging and chronic disease development.
Epigenetic alterations
Epigenetics studies heritable changes in gene expression that occur without changes in DNA sequence. Epigenetics include flexible genomic marks that regulate gene expression and can change genome function under exogenous influence, but also provide mechanisms that allow for stable propagation of gene activity states from one generation of cells to the next. As such, the human epigenome is inherently related to the programming of cellular functions and of key regulators of biological aging.
Environmental exposures induce age-related epigenomic changes
Studies across species and cell types have consistently shown age-associated changes of different layers of the human epigenome, including DNA methylation, histone modifications, and chromatin remodeling. Functional studies in model organisms demonstrated that these epigenomic changes impact both healthy and pathological aging. Human epigenomic aging studies, as well as investigations of the impact of the environment on the aging epigenome, have followed two interrelated paths: the identification of epigenetic changes associated with aging and the development of epigenomic biomarkers that quantify aging. Significant chromatin structure changes occur during aging and senescence, including loss of heterochromatin regions, global histone loss, and chromatin spatial interaction changes (Sun et al., 2018). These changes are associated with age-related changes in histone modifications, including tissue-dependent decreases of global H3K27me3 and H3K9me3 levels and gains of H3K4me3 and H3K36me3. Histone acetylation at multiple histone tail residues also decreases with age. DNA methylation within genes shows both gains and losses in DNA methylation and those changes have been often associated with loss of appropriate gene regulation. The result of epigenetic changes during aging is altered local accessibility to the genetic material, leading to aberrant gene expression, reactivation of transposable elements, and genomic instability. Environmental exposures have been repeatedly associated with changes in DNA methylation that resemble epigenetic aging. For instance, exposure to air pollution has been associated with methylation loss in DNA methylation of repeated elements in multiple studies. Also, epigenome-wide association studies have shown both increases and decrease in DNA methylation associated with environmental exposures, including air pollution, metals, and organic chemicals. In vitro and animal experiments demonstrated that several toxic metals induce changes in histone modifications, including loss of acetylation and alteration of histone methylation at several histone tail modifications (Ijomone et al., 2020). The mechanisms that induce these epigenomic changes have been actively investigated across a variety of exposures. For instance, arsenic is metabolized into a methylated form using the methyl donor S-adenosyl methionine, a biotransformation reaction that has been suggested to deplete the methyl donor pool and cause the arsenic-induced hypomethylation demonstrated in several in vitro studies (Ryu et al., 2015). However, this hypothesis is not sufficient to account for the array of influences that arsenic has on the epigenome (Ijomone et al., 2020). Acute cadmium exposure has been shown to decrease DNA methylation through noncompetitive inhibition of the activity of DNA methyltransferases (DNMTs). On the contrary, chronic cadmium exposure may result in a global increase of DNA methylation due to increased DNMT activity. Cadmium influences on the epigenome are also not limited to DNA methylation as decreases in histone modifications such as H3K4me3, H3K27me3, H3K9me3 and H3 phosphorylation have been linked to this metal, possibly due to cadmium-induced inhibition of histone modifying enzymes (Ryu et al., 2015). Other metals can alter the function of the Ten-eleven translocation (TET) family proteins, which are Fe(II)- and 2-oxoglutarate-dependent dioxygenases that induces demethylation by catalyzing the oxidation of 5-methylcytosine. For instance, nickel binds to the Fe(II)-chelating motif of the TET proteins with an affinity of 7.5-fold as high as Fe(II) (Yin et al., 2017). Hence, nickel can displace the cofactor Fe(II) of TET dioxygenases and inhibit TET-mediated oxidation of 5-methyl-cytosine. Cobalt, which also induces hypomethylation in experimental conditions, has also been shown to affect TET activity. Many epigenetic changes induced by environmental exposures may be part of coordinated responses to the exposures that are adaptive in nature. The epigenome is a main mechanism through which our cells deploy an array of adaptive responses to help our body to live in an ever-changing environment, including responses to environmental pollution. Finally, many epigenetic associations with environmental exposures may be accounted for by changes in the cellular mix in a given organ or tissue that are induced by the exposure, rather than by actual changes in epigenetic markings within cells (Lappalainen and Greally, 2017). A recent analysis of older individuals in the Greater Boston area showed that the levels of different ambient air pollutants, ambient temperature, and relative humidity were associated with the proportions of subtypes of circulating blood leukocytes (Gao et al., 2019).
Environmental exposures accelerate epigenetic aging clocks
Robust patterns of age-related genome-wide DNA methylation (DNAm) changes have been observed in blood and across many human tissue types, and may underlie age-related changes to the transcriptome (Nwanaji-Enwerem et al., 2018). Several groups have leveraged these age-related changes in DNA methylation to develop clocks trained to predict chronological age based on DNA methylation data, including the widely used Horvath and Hannum epigenetic age measures (Hannum et al., 2013; Horvath, 2013). These DNA methylation clocks were built using machine learning techniques to select an ensemble of DNA methylation CpG sites that together can accurately estimate the chronological age of the individual. While these age-trained clocks were not necessarily expected to reflect biological aging, a series of studies have shown that the difference between DNA methylation age and chronological age, often referred to as “delta epigenetic age” or “age acceleration”, is associated with shorter average life expectancy and increased risk of age-related disease (Horvath and Raj, 2018; Marioni et al., 2015). Environmental health studies have repeatedly indicated that adverse environmental exposures hasten the epigenetic clocks (Dhingra et al., 2018). For instance, individuals living in areas with higher ambient fine particulate matter (PM2.5) exhibited older DNA methylation age in at least two independent studies (Nwanaji-Enwerem et al., 2016; Ward-Caviness et al., 2016). Also, plasma levels of the pesticide biomarkers 2,2-bis (4-chlorophenyl)-1,1-dichloroethene (p,p’-DDE) and transnonachlor (TNC) were associated in a recent study with older DNA methylation age (Lind et al., 2018).
In summary, epigenetics is a prime hallmark of environmental insults and persuasive evidence exists that environmental exposures cause alterations in gene regulation via alteration in DNA methylation and histone modifications and promote aging-related epigenetic changes including the acceleration of epigenetic clocks.
Mitochondrial dysfunction
Mitochondria are subcellular organelles that provide the large majority of the energy required for cellular functions and play an important role in the cellular response to environmental stressors. They might act as a plausible mechanistic link, or act as a biosensor, between environmental exposure and health outcome.
Environmental exposures induce mitochondrial dysfunction
Alterations in mitochondrial DNA (mtDNA) content (total amount of mtDNA copies) is an established marker of mitochondrial damage and function (Sahin et al., 2011) and has been identified as an etiological determinant in a variety of human diseases. The importance of placental mitochondrial function during in utero life on fetal brain development with long-lasting consequences on the IQ has recently been reported (Bijnens et al., 2019). Also, mitochondrial quality and activity decline as we age and lower mtDNA content of white blood cells has been shown in type-2 diabetes and breast cancer (Wallace, 2010).
The key role for mitochondria in the toxicity of environmental exposures, is due to the central role that oxidative stress plays in environmental toxicants. Exposure to air pollution such as particulate matter, benzene, PAHs, and toxic metals are involved in mitochondrial dysfunction in the blood (Breton et al., 2019; Byun et al., 2013; Janssen et al., 2012). Besides, a molecular circuit directly links telomere damage to compromised mitochondrial biogenesis via the tumor suppressor gene TP53 (Sahin et al., 2011). Growing evidence suggests mitochondrial epigenetics as a novel mechanism to understand the pathophysiology of diseases with a mitochondrial dysfunction involvement (Shaughnessy et al., 2014).
Environmental exposures impact the mitochondrial - telomere aging axis
The telomere-mitochondrial aging hypothesis builds on findings of a direct link between mitochondria and telomeres (Sahin et al., 2011). Telomeres are highly sensitive to ROS, and environmental exposures such as air pollution have shown to increase levels of ROS (Figure 2) which leads to shortened and potentially dysfunctional telomeres. Telomere dysfunction in mice resulted in suppression of peroxisome proliferator-activated receptor gamma genes and consequently a decrease in mitochondrial biogenesis and function, subsequently leading to an impaired adenosine three phosphate generation and increased ROS production. Furthermore, DNA damage at telomeres activates several signaling pathways and reduces Sirt1 gene expression. All these markers of the core axis of aging are associated with long-term exposure to air pollution even at low concentrations (Pieters et al., 2016). It is important to note that prenatal air pollution exposure is one of the main predictors of telomere length at birth (Martens et al., 2019).
In summary, mitochondrial dysfunction is involved in many chronic diseases and aging. Environmental exposures have been shown to induce alterations in mtDNA content, nucleus-mitochondria crosstalk and mitochondrial epigenetics. Highlighted is recent evidence showing that environmental exposures jointly affect the telomere-mitochondrial aging axis.
Endocrine disruption
The endocrine system controls a countless number of organism functions through fine-tuned hormonal signals released by endocrine glands into the circulation. During normal aging, at least three hormonal systems show decreasing circulating hormone concentrations, including (i) the sex hormones estrogens (after menopause) and androgens; (ii) the adrenal hormones dehydroepiandrosterone and its sulphate; (iii) and the growth hormone/insulin-like growth factor I axis (Lamberts et al., 1997). Clinical manifestation related to endocrine alterations also increases during aging; for instance, the number of patients with impaired glucose tolerance and type 2 diabetes mellitus increase exponentially with age and age-related thyroid dysfunction is more common in older individuals.
Endocrine disrupting chemicals
Endocrine Disrupting Chemicals (EDCs) are defined as “exogenous chemicals, or mixture of chemicals, that can interfere with any aspect of hormone action” (Zoeller et al., 2012). EDCs are found in our environment, food and consumer products and can bind to the body’s endocrine receptors to activate or block their actions or alter the synthesis, release, transport, or metabolism of endogenous hormones (Baccarelli et al., 2000). While mapping EDCs may be challenging, it is estimated that nearly 1,500 chemicals have endocrine effects today and this number grows as new EDCs are detected among the thousands of chemicals entering the marketplace every year. EDCs show plausible links with reproductive, neurodevelopmental, metabolic effects, bone alterations, immune disorders, cancers, and other conditions (WHO, 2012). The best characterized EDCs include bisphenols, phthalates, polybrominated diphenyl ethers (PBDEs), per- and polyfluorinated chemicals (PFAs), diethylstilbestrol (DES), dioxins, and other chlorinated hydrocarbons such as dichlorodiphenyltrichloroethane (DDT) and polychlorinated biphenyls (PCBs).
Because of the critical role of endocrine signaling in reproduction and prenatal and postnatal development, a large amount of research to date has focused on the reproductive and developmental effects of EDCs. Nonetheless, EDCs as well as other environmental exposures including air pollution and metals, have been linked with a variety of age-related outcomes that may be mediated by accelerated endocrine aging, including metabolic and cardiovascular disease, accelerated bone loss and osteoporosis, and accelerated cognitive aging and dementia. However, evidence indicating that environmental exposures affect endocrine pathways in older individuals and that such endocrine alterations mediate those health effects is still limited. One specific area of active EDC research has revolved around their potential transgenerational and epigenetic effects. Some chemicals, including EDCs such as pesticides and persistent organic pollutants have been reported to cause health-related effects in the offspring of exposed animals and that those effects, purportedly due to epigenetic inheritance, can propagate themselves across multiple generations among descendants not directly exposed (Messerlian et al., 2017). If the initial animal experiments supporting transgenerational inheritance of chemical effects were to be confirmed in independent experimental studies as well as in human investigations, this knowledge would open fundamental questions about the influences of EDCs on human aging. Healthy and unhealthy aging hinges on lifetime trajectories that can be accelerated or delayed by exposures occurring as early as in utero and early postnatal life. However, this new type of inheritance would extend the susceptible windows to previous generations, create a broader concept of environmental responsibility, and open novel opportunities for prevention with possible beneficial effects far beyond those that typically projected today.
In summary, alterations of the endocrine system are pervasive, responsible for many physiological changes during child development and normal aging and in chronic diseases. Understanding the impact of especially the EDCs is a critical area for future investigations of the impact of environmental exposures on human health over the life-course.
Altered intercellular communication
Aging is characterized by changes in intercellular communication that modify the varied set of the body’s coordinated functions and responses (Lopez-Otin et al., 2013). In aging organisms, the amount and type of secreted cellular factors undergo extensive changes that affect the function of neighboring and distant cells. Specifically, senescent cells have been shown to communicate with neighboring cells via various means of intercellular communication, a finding that has been conceptualized as the senescence-associated secretory phenotype (SASP) (Fafian-Labora and O’Loghlen, 2020). SASP encompasses multiple families of soluble and insoluble factors that affect surrounding cells by activating various cell-surface receptors and corresponding signal transduction pathways. Classical SASP factors include soluble signaling factors (interleukins, chemokines, and growth factors), secreted proteases, and secreted insoluble proteins/extracellular matrix (ECM) components. Other emerging SASP factors have been recently identified, including Extracellular Vesicles (EVs). EVs are nano-sized membrane-bound vesicles released by human cells into the bloodstream that deliver messages between cells, in both healthy and pathological processes, by transporting cargo from one tissue to another throughout the body (Panagiotou et al., 2018). Messaging occurs via the EV cargo, such as such as short noncoding RNAs (e.g., miRNAs), proteins, and metabolites, which varies in response to internal and external stimuli (including environmental pollutants) and acts on distal cells. Particularly, EV-associated miRNAs are powerful regulators that can control gene expression in recipient cells, thereby modifying the recipient cell’s biology.
Environmental exposures alter intracellular communication
Increasing evidence shows that SASP signaling is affected by environmental exposures. In addition to alterations of interleukin and chemokines receptors (See “Oxidative Stress and Inflammation” section), several environmental exposures can impact the levels and function of other classical SASP factors. For instance, ozone have been shown to dysregulate epidermal growth factor signaling in the lung and proteases, for instance metalloproteases in the lung and systemically (Feng et al., 2016).
Several studies showed that exposure to air pollution induces the release of large quantities of EVs in the bloodstream that can operate as novel proinflammatory modulators (Alkoussa et al., 2020). For example, in a coordinated set of human and in-vitro studies, Bollati and colleagues identified miR-128 packaged in EVs (EV-miR-128), a miRNA that can activate inflammatory pathways, as a primary response to acute air pollution exposure (Bollati et al., 2015). Additional research has demonstrated that EVs are also modified by other environmental exposures, including bisphenol A, phthalates and metals (Tumolo et al., 2020).
In summary, environmental exposures activate and/or disrupt intercellular communications that are key to biological aging, including the emerging EV-mediated signaling system.
Altered microbiome communities
The microbiome is an emerging key player in aging and longevity. Not only does the human microbiome at multiple locations vary as we age, but microbiome research has also established a causal relationship with host aging (Kim and Benayoun, 2020). Interventions that increase longevity in animal models have been shown to have rejuvenating effects on the microbiome, and changes in microbiome composition have been linked to increased predisposition to age-related diseases, including obesity, cardiovascular disease, cancer, and neurodegeneration. The microbiome of the lung, the skin and the gut has been implicated to play a major role in environmentally determined diseases. The symbiosis between the microbiota and its mammalian host encompasses multiple relationships, including mutualistic, parasitic, and commensal (Belkaid and Hand, 2014). The capacity of a given microbe, including those composing the microbiota, to trigger or promote disease is highly contextual, and some microbes can shift from mutualist to commensal to parasite according to the state of activation of the host, co-infection, or localization (Chen et al., 2018).
Environmental exposures can protect against or enhance allergies
The microbiome of the barrier organs in particular can be considered as linked to communities of bacteria, viruses and other microorganisms from the environment. However, the conditions under which environmental exposures contribute to disease development are complex. For example, exposures to environmental airborne biological and chemical agents have been demonstrated to be highly variable in space and time (Jiang et al., 2018). The microbiome at the barrier organs and the modulation of the immune system has proven to be essential for understanding the environmental determinants of childhood asthma and allergies and the interplay between environmental biological agents and pollution (Figure 3). The presence of harmful factors such as air pollution and pathogenic bacteria, and a gut microbiome with low diversity is associated with an increased risk of virus-induced wheeze, asthma and allergies (Smits et al., 2016). These harmful factors are accompanied by increased inflammation, T-cell activation and a reduced antimicrobial response. In contrast, microbial exposure from traditional farm environments or worm infections during an early-life window show protective effects against asthma and allergies. In these settings, increased antimicrobial responses and regulatory responses by the immune system via T-regulatory cells or dendritic cells are observed (Nowarski et al., 2017). This leads to a higher variability both in gut and lung microbiome. Immune modulating short chain fatty acids potentially foster the gut-lung-microbiome interactions.
Figure 3: Environmental insults and protective factors for the lung and the gut microbiome.
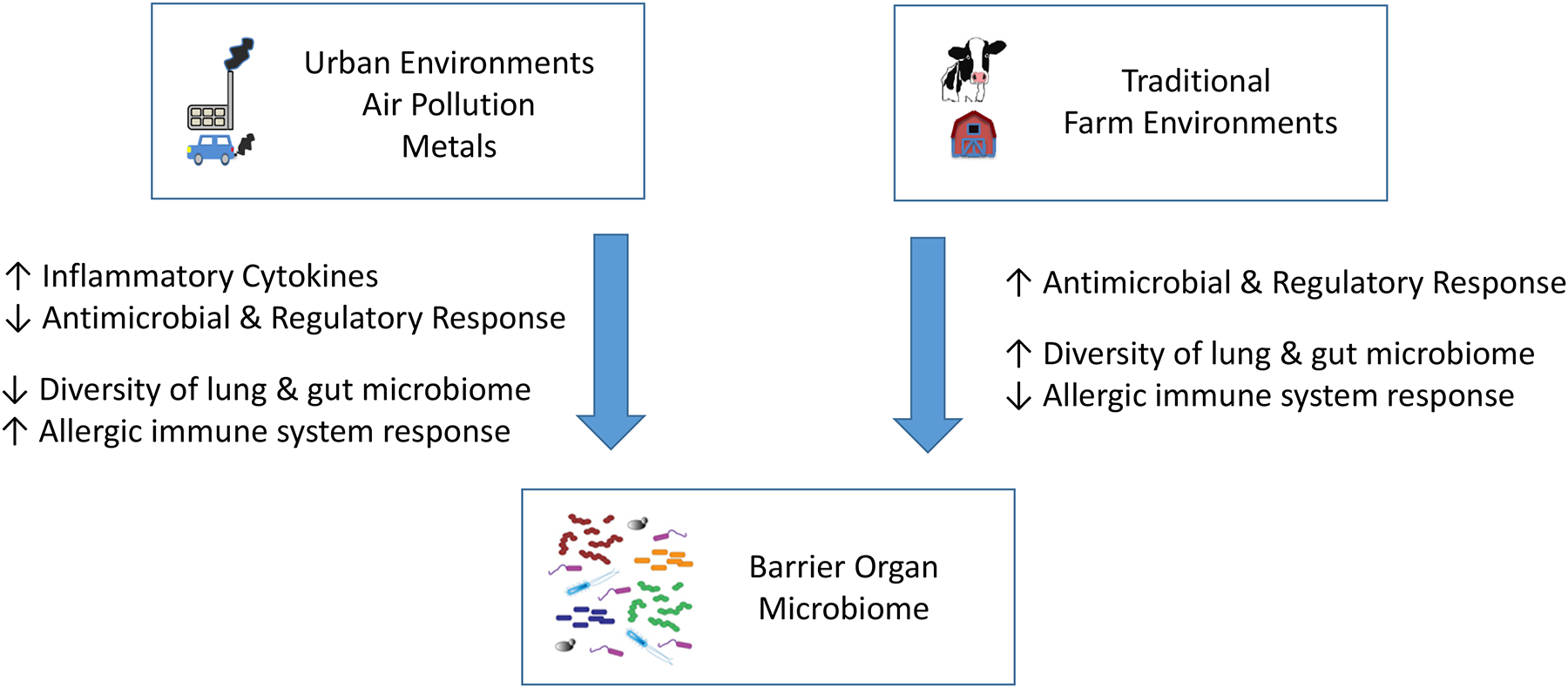
Complex urban and traditional farm environments alter the microbiome variability and the immune function at the barrier organs and have been implicated to either promote or protect against allergies.
Similarly, the skin microbiome has been recognized as an essential component in maintaining the barrier function and modulating immune responses (Belkaid and Segre, 2014; Chen et al., 2018). It is deregulated in skin diseases. Environmental exposures such as ultraviolet radiation and allergens have been implicated to alter the skin microbiota directly by activation of immune responses and indirectly by their induction of oxidative stress (Stefanovic et al., 2020; Suwanpradid et al., 2017).
The composition of the gut microbiome has been associated with a large number of diseases and research on their specific role and the potential to treat diseases by modulating and altering gut microbiota composition is still ongoing. Environmental exposures as for example arsenic have been put forward to not only induce oxidative stress but to also have substantial impact on the gut microbiome (Chi et al., 2018). Similarly, the protective properties of the farm environments with respect to allergies may be mediated in parts through the gut microbiota composition (Smits et al., 2016).
Environmental exposures promote viral infections at barrier organs
The interaction of the microbiome and the virome at barrier organs is likely to play an important role for maintaining function and abating disease development. Air pollution has been shown experimentally to reactivate dormant herpes viruses (Sattler et al., 2017). This evidence proposes a novel paradigm and implies different mechanisms by which nanoparticles could induce inflammatory responses independent of the classical acute phase responses. One can rightly expect that this research will be sparked by the ongoing COVID-19 pandemic (Barouki et al., 2020). It has been hypothesized that for example ambient air pollution promotes COVID-19 disease severity, which is supported by recent evidence that in intensive care patients hospitalized for various underlying pathologies, pre-admission air pollution exposure prolongs the duration of artificial-ventilation (De Weerdt et al., 2020).
In summary, the microbiome of the barrier organs is essential for their function and its misbalance and reduced diversity plays most likely an important role in chronic disease development. Environmental insults induce these imbalances, but environmental exposures have also been identified to protect against allergies, to foster in particular microbiome diversity and contribute significantly to barrier organ functioning.
Impaired nervous system function
The central nervous system jointly with the autonomous nervous system is responsible for the functioning of nearly all physiological processes. They play a major role in neurodevelopmental and neurodegenerative diseases but also impair cardiovascular, metabolic and lung diseases. The brain and other components of the nervous system experience complex age-related changes. Those include loss of brain volume, alterations in neurons and synapses, as well as biochemical, oxidative, and inflammatory changes. Also, the aging nervous system shows less pronounced and slower autonomic responses, lower cognitive functioning, and higher vulnerability to degenerative and vascular disease.
Environmental exposures affect nervous system functions
Environmental exposures such as noise, heat and light directly activate the nervous system via sensory organs. Noise has been studied extensively and has been linked to hearing loss, increases in blood pressure and onset and progression of cardiovascular disease (Babisch, 2011; Basner et al., 2014). Specifically, the direct impact manifests itself through interaction between the acoustic nerves and the central nervous system. Indirect effects include the cognitive perception of sound, cortical activation and emotional responses. Both, direct and indirect effects promote stress responses in various brain regions including the modulation of the autonomous nervous system with activation of the hypothalamus – pituitary – adrenal axis and the sympathetic – adrenal – medulla axis (Munzel et al., 2017). This physiological response to external stimuli increases the heart rate and the release of the stress hormones cortisol, adrenalin, and noradrenalin. While these responses enable us to react quickly to perceived dangers, continuous upregulation is also linked to oxidative stress and inflammatory responses both locally at the site of sound perception (Basner et al., 2014) systematically associating it to cardiometabolic diseases (Munzel et al., 2017). Also, air pollution has been implied to activate the autonomous nervous system via irritant receptors in the lung (Peters et al., 2006). The impact of particulate matter air pollution on the brain is further supported by the observation that ambient traffic-derived nanoparticles reach the brain via the olfactory nerve and via systemic translocation (Stone et al., 2017) (Figure 4). Consequently, it is to be considered that noise and air pollution exhibit biologically independent, but augmenting environmental insults if these exposure co-occur in urban environments (Munzel et al., 2017).
Figure 4: Illustration of organ-specific impacts based on the hallmarks of environmental insults.
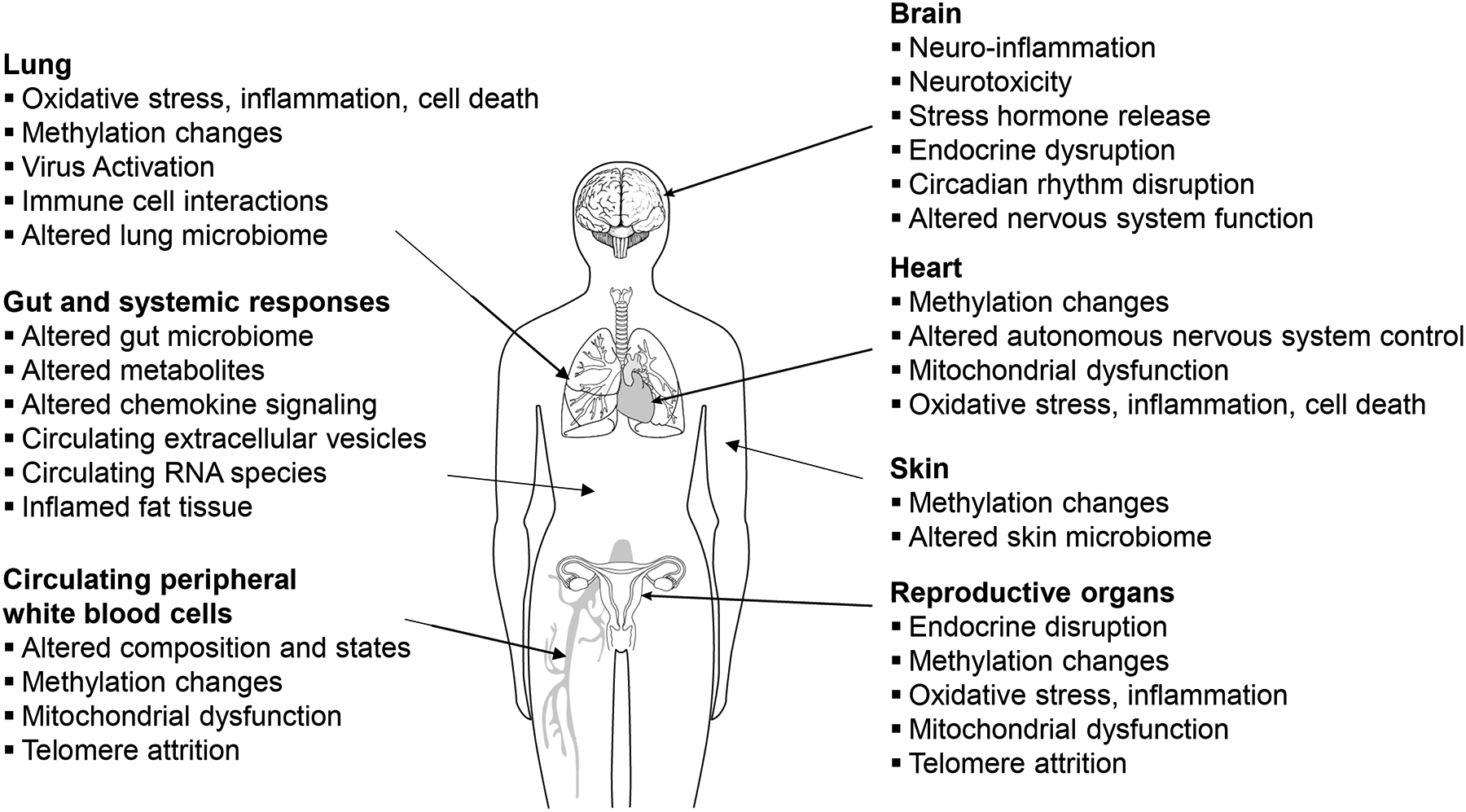
These pathophysiological changes summarize examples of how environmental exposures impact organs locally as well as exhibit their insults systemically.
The central nervous system controls the core body temperature and responds to outer temperatures on the skin sensing cold and warmth and the visceral fat sensing warmth (Madden and Morrison, 2019). Environmental heat exposures due to excessive exercise on hot days, heat waves and urban heat islands can result in heat exhaustion and heat stroke (Schneider et al., 2017). Heat exposure becomes more and more relevant as climate change progresses (see textbox).
Textbox: Ambient particulate matter air pollution and climate change in light of the hallmarks of environmental insults.
Air Pollution
Clean air is a basic requirement of human health and well-being, but unfortunately, air pollution is still an important contributor to morbidity and mortality (Peters et al., 2019). Exposure to fine particles (particulate matter smaller than 2.5 μm in aerodynamic diameter (PM2.5)) alone is responsible for about 4 million deaths world-wide (Cohen et al., 2017). After smoking, air pollution is therefore one of the most important risk factors, in the same order of magnitude as overweight or lack of physical activity. Experimental studies have demonstrated that ultrafine particles smaller than 100 nm can translocate from the lung into the systemic circulation and reach other organs including the brain and the placenta (Stone et al., 2017).
Ambient air pollution has been linked to all hallmarks of environmental as highlighted by throughout the review, namely, oxidative stress and inflammation (Mudway et al., 2020), mitochondrial dysfunction (Breton et al., 2019), genomic alterations (Yauk et al., 2008), epigenetic alterations (Neven et al., 2018; Ward-Caviness et al., 2016), endocrine disruption (Janssen et al., 2017), altered intercellular communication (Bollati et al., 2015), altered gut microbiome (Mutlu et al., 2018) and impaired autonomous nervous system (Peters et al., 2006). There is ample evidence that air pollution is affecting all organs highlighted in Figure 4 (Thurston et al., 2017). Importantly, air pollution impairs all these hallmarks at low concentrations, well below for example the current European standards of annually 25 μg/m3 PM2.5.
Climate Change
Global warming and higher weather variability will substantially increase the heat-related mortality in most regions of the world (Gasparrini et al., 2015). Exposures to extreme temperatures, including both acute heat and cold exposures, have been associated with increased cerebrovascular, cardiovascular, and respiratory morbidity and mortality rates (Watts et al., 2019). The underlying mechanisms for the adverse health effects include enhanced sympathetic reactivity followed by activation of the sympathetic nervous system, renin-angiotensin system, as well as dehydration and a systemic inflammatory response (Liu et al., 2015). At extreme warm temperatures, hemoconcentrations, hyper viscosity, systemic inflammation, consumptive coagulation, and microvascular thrombosis are observed (Schneider et al., 2017). Recent evidence suggests that also in utero, exposures to both cold and heat are associated with adverse birth outcomes, such as a trigger for preterm births (Cox et al., 2016). Generally, newborns, infants, aging individuals and those with underlying cardiovascular or neurological diseases as well as mental impairments are among the most vulnerable. Evidence for impacts of environment temperature on global DNA methylation in old age (Bind et al., 2014) and newborns (Abraham et al., 2018) were observed. Telomere length was found to be shorted by high ambient temperature during gestation (Martens et al., 2019). Taken together, there is the suggestion that ambient temperatures impair health through the hallmarks of environmental insults to a far greater extent than currently known.
Light at night has been linked to cardiometabolic diseases, cancer and mental health (Lunn et al., 2017). Light disruption and circadian disruption have been studied extensively in experimental and occupational settings which led to an in depth understanding of the regulation of circadian rhythms. At the eye level, photosensitive retinal ganglion cells in the inner layer of the retina control biological rhythms by light, the sleep-wake cycle, alertness and pupillary constriction. These are sensitive to the blue band (400–500 nm) of the light spectrum, whose role is fundamental in circadian system function and the entrainment of the internal clock. Light emitting diodes (LEDs) are thought to disrupt the circadian rhythm and exhibit downstream dysregulation of cortisol and melatonin concentrations and impair sleep (Touitou and Point, 2020). It is to be expected that personal blue light exposures will further increase with digitalization. Therefore, circadian rhythm disruption due to a combination of personal and environmental light pollution is of concern.
The brain is particularly vulnerable to neurotoxic chemicals throughout the life-course which impair development and programming of the brain, hinder functional maturation and trigger adult neurological diseases and neurodegeneration (Grandjean and Landrigan, 2014). Industrial pollutants induce a substantial health burden via reduced intelligence quotient points, altered behavior and induction of neurodegenerative diseases later in life. For at least eleven chemicals neurotoxicity is clearly documented: lead, methylmercury, polychlorinated biphenyls, arsenic, toluene, manganese, fluoride, chlorpyrifos, dichlorodiphenyltrichloroethane, tetrachloroethylene, and the polybrominated diphenyl ethers (Grandjean and Landrigan, 2014). Methylmercury, for example, exhibits neural stem cell toxicity in vitro and induces altered gene expression through epigenetic changes (Ijomone et al., 2020). Furthermore, endogenous H2O2-levels control neuronal stem cell growth and regeneration (Sies and Jones, 2020), which provides evidence that ROS-generating chemicals may interfere with brain function during child development as well as in old age.
Beneficial effects of nature exposure
In contrast to environmental insults, exposure to nature has been reported to have beneficial impacts on mental health (Bratman et al., 2019; van den Bosch and Meyer-Lindenberg, 2019). Specifically, urban green exposure resulted in reduced lateral prefrontal activation (Tost et al., 2019) and increased the cingulate cortex activity, a brain area that may be less developed in citizens of urban areas (van den Bosch and Meyer-Lindenberg, 2019). These observations provide a direct link between the sensory reception of urban green and health. Less consistent associations were observed for indirect, mechanistically more questionable links such as for example cardiovascular diseases or mortality (Nieuwenhuijsen, 2018).
In summary, sensory organs are key recipients of environmental cues and insults. Noise, heat, and artificial light can modulate and impair the autonomous nervous system, induce stress responses, and a number of downstream responses specific to activated nervous system functions and control mechanisms. Brain development and neurodegenerative diseases are substantially impacted by neurotoxic chemicals.
Discussion
This article proposes eight hallmarks of environmental insults. It is important to note that due to the vast dimensional space of the chemical and physical properties of environmental exposures, this review can by no means be complete. However, it is the ambition of the review to create a framework which allows to delineate why complex mixtures of environmental exposures are capable of inducing severe health effects even at relatively modest concentrations from a toxicological standpoint. Especially environmental exposures that are able to impair multiple of these essential hallmarks on a cellular level lead to local and systemic organ damage and induce chronic diseases at the barrier organs as well as cardiovascular or neurological diseases or cancer.
Traditionally, environmental insults are characterized in toxicological frameworks. Mechanism of toxicity for single chemicals or chemical classes are established and their impact is mainly exhibited through one or few defined pathways. However, most of the time the challenge is to understand the impacts of complex mixtures that impose the insults through a variety of mechanisms related to their chemical and physical properties and through multiple impacts on cellular and organ levels (Figure 4). Air pollution is a prominent example as highlighted in the textbox. To result in notable burden of disease, a number of essential cell and organ function need to be impaired. Indeed, for these complex mixtures, the relative risk may even be higher at low concentrations than at high concentrations due to the fact that in relatively clean environments the negative impacts on the hallmarks of environmental insults are not downregulated through feedback mechanisms. To match the complexity of the environment and to make full use of the sweep of biomedical technology that is available today, the idea of the exposome research as to complement the genome research has been put forward. These approaches will in particular generate tools, data and knowledge to better capture and analyze the complexity of environmental exposures, to characterize their relationship to the internal exposome, to link them to pathophysiological responses and to quantify their impact on chronic and infectious disease burden (Vermeulen et al., 2020).
The hallmarks of environmental insults have relevant overlaps with the hallmarks of aging (Lopez-Otin et al., 2013). This indeed stresses the interlinkages between processes of aging and the external environmental drivers of health and disease. We consider it important to emphasize the role of underpinning cellular mechanisms that impair health from conception to old age. We bring forward important novel aspects by highlighting the impact of environmental insults on oxidative stress and inflammation, endocrine disruption, microbiome communities, and nervous system function. There are certain areas which have emerged when considering aging (Lopez-Otin et al., 2013), but have not been prominently featured here. These include for example the area of insults of environmental exposures on stem cells, which have been mentioned briefly as part of the section on mutagenesis, intracellular communication and nervous system function. Also, it is very likely that environmental exposures contribute to impairment of protein translation, post-translational protein modification and also protein folding. However, research in these areas considering environmental exposures is currently just emerging (Calderon-Garciduenas et al., 2019).
Environmental exposures are highly variable in time and space which increases the challenge of delineating their impact on the cellular, organ and organism level. Environmental exposures may trigger or contribute significantly to the development of manifest chronic diseases. For a trigger, an immediate response disturbs homeostasis in a hallmark in vulnerable individuals. Environmental insults that persist over longer time periods and have multiple impacts may induce diseases chronically and their impact may be in part reversible if exposures cease. However, some environmental exposures may also induce long-lasting epigenetic modifications with transgenerational impacts that would even persist after the exposures are eliminated.
Conclusion
Environmental insults are omnipresent and highly complex. Nevertheless, they impair essential cellular functions thereby leading to a plethora of diseases. Eight hallmarks of environmental insults underpin most, if not all, observed associations between environmental exposures and disease. Specifically, they comprise oxidative stress and inflammation, genomic alterations and mutations, epigenetic alterations, mitochondrial dysfunction, endocrine disruption, altered intercellular communication, altered microbiome communities, and impaired nervous system function. Taken together, they provide a framework for understanding how environmental insults, even at relatively low concentrations, can lead from health to manifest chronic diseases. The hallmarks shall also aid to jointly assess the intersection of environmental exposures and life-style related behaviors such as for example physical activity, exposure to urban green and air pollution.
These hallmarks call for novel experimental research to expand our knowledge of disease mechanisms going beyond genetics. Integrated approaches combining observational and experimental research seem to be mandated for further refining the concept put forward here. Advances in biomedical technologies and data science will allow to delineated the complex interplay of environmental insults down to the single cell level. There is an important potential to leverage the understanding of linkages between environmental insults and health for proposing individualized prevention and treatment strategies. However, the most important environmental challenges of today and tomorrow such as for example air pollution and climate change can only be partially addressed or abated on the individual level. Strong local, national and inter-governmental policies needed to ensure healthy environments for healthy citizens.
Acknowledgment
The authors thank Dr. Regina Pickford, Helmholtz Zentrum München for her comments to the initial and revised manuscript. Tim Nawrot is currently funded by the Flemish Research Fund (G048420N).
Footnotes
Declaration of Interest
The authors declare no conflict of interest.
References
- Abraham E, Rousseaux S, Agier L, Giorgis-Allemand L, Tost J, Galineau J, Hulin A, Siroux V, Vaiman D, Charles MA, et al. (2018). Pregnancy exposure to atmospheric pollution and meteorological conditions and placental DNA methylation. Environ Int 118, 334–347. [DOI] [PubMed] [Google Scholar]
- Alkoussa S, Hulo S, Courcot D, Billet S, and Martin PJ (2020). Extracellular vesicles as actors in the air pollution related cardiopulmonary diseases. Crit Rev Toxicol 50, 402–423. [DOI] [PubMed] [Google Scholar]
- Babisch W (2011). Cardiovascular effects of noise. Noise Health 13, 201–204. [DOI] [PubMed] [Google Scholar]
- Baccarelli A, Pesatori AC, and Bertazzi PA (2000). Occupational and environmental agents as endocrine disruptors: experimental and human evidence. J Endocrinol Invest 23, 771–781. [DOI] [PubMed] [Google Scholar]
- Balmain A (2020). The critical roles of somatic mutations and environmental tumor-promoting agents in cancer risk. Nat Genet 52, 1139–1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barouki R, Kogevinas M, Audouze K, Belesova K, Bergman A, Birnbaum L, Boekhold S, Denys S, Desseille C, Drakvik E, et al. (2020). The COVID-19 pandemic and global environmental change: Emerging research needs. Environ Int 146, 106272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basner M, Babisch W, Davis A, Brink M, Clark C, Janssen S, and Stansfeld S (2014). Auditory and non-auditory effects of noise on health. Lancet 383, 1325–1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belkaid Y, and Hand TW (2014). Role of the microbiota in immunity and inflammation. Cell 157, 121–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belkaid Y, and Segre JA (2014). Dialogue between skin microbiota and immunity. Science 346, 954–959. [DOI] [PubMed] [Google Scholar]
- Bijnens EM, Derom C, Weyers S, Janssen BG, Thiery E, and Nawrot TS (2019). Placental mitochondrial DNA content is associated with childhood intelligence. J Transl Med 17, 361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bind MA, Zanobetti A, Gasparrini A, Peters A, Coull B, Baccarelli A, Tarantini L, Koutrakis P, Vokonas P, and Schwartz J (2014). Effects of temperature and relative humidity on DNA methylation. Epidemiology 25, 561–569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bollati V, Angelici L, Rizzo G, Pergoli L, Rota F, Hoxha M, Nordio F, Bonzini M, Tarantini L, Cantone L, et al. (2015). Microvesicle-associated microRNA expression is altered upon particulate matter exposure in healthy workers and in A549 cells. J Appl Toxicol 35, 59–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bratman GN, Anderson CB, Berman MG, Cochran B, de Vries S, Flanders J, Folke C, Frumkin H, Gross JJ, Hartig T, et al. (2019). Nature and mental health: An ecosystem service perspective. Sci Adv 5, eaax0903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breton CV, Song AY, Xiao J, Kim SJ, Mehta HH, Wan J, Yen K, Sioutas C, Lurmann F, Xue S, et al. (2019). Effects of air pollution on mitochondrial function, mitochondrial DNA methylation, and mitochondrial peptide expression. Mitochondrion 46, 22–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brook RD, Rajagopalan S, Pope CA III, Brook JR, Bhatnagar A, Diez-Roux AV, Holguin F, Hong Y, Luepker RV, Mittleman MA, et al. (2010). Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation 121, 2331–2378. [DOI] [PubMed] [Google Scholar]
- Byun HM, Panni T, Motta V, Hou L, Nordio F, Apostoli P, Bertazzi PA, and Baccarelli AA (2013). Effects of airborne pollutants on mitochondrial DNA methylation. Part Fibre Toxicol 10, 18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderon-Garciduenas L, Gonzalez-Maciel A, Kulesza RJ, Gonzalez-Gonzalez LO, Reynoso-Robles R, Mukherjee PS, and Torres-Jardon R (2019). Air Pollution, Combustion and Friction Derived Nanoparticles, and Alzheimer’s Disease in Urban Children and Young Adults. J Alzheimers Dis 70, 343–360. [DOI] [PubMed] [Google Scholar]
- Chen YE, Fischbach MA, and Belkaid Y (2018). Skin microbiota-host interactions. Nature 553, 427–436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chi L, Gao B, Tu P, Liu CW, Xue J, Lai Y, Ru H, and Lu K (2018). Individual susceptibility to arsenic-induced diseases: the role of host genetics, nutritional status, and the gut microbiome. Mamm Genome 29, 63–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen AJ, Brauer M, Burnett R, Anderson HR, Frostad J, Estep K, Balakrishnan K, Brunekreef B, Dandona L, Dandona R, et al. (2017). Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: an analysis of data from the Global Burden of Diseases Study 2015. Lancet 389, 1907–1918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conrad M, and Pratt DA (2019). The chemical basis of ferroptosis. Nat Chem Biol 15, 1137–1147. [DOI] [PubMed] [Google Scholar]
- Cox B, Vicedo-Cabrera AM, Gasparrini A, Roels HA, Martens E, Vangronsveld J, Forsberg B, and Nawrot TS (2016). Ambient temperature as a trigger of preterm delivery in a temperate climate. J Epidemiol Community Health 70, 1191–1199. [DOI] [PubMed] [Google Scholar]
- De Weerdt A, Janssen BG, Cox B, Bijnens EM, Vanpoucke C, Lefebvre W, El Salawi O, Jans M, Verbrugghe W, Nawrot TS, et al. (2020). Pre-admission air pollution exposure prolongs the duration of ventilation in intensive care patients. Intensive Care Med 46, 1204–1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhingra R, Nwanaji-Enwerem JC, Samet M, and Ward-Caviness CK (2018). DNA Methylation Age-Environmental Influences, Health Impacts, and Its Role in Environmental Epidemiology. Curr Environ Health Rep 5, 317–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorrington MG, and Fraser IDC (2019). NF-kappaB Signaling in Macrophages: Dynamics, Crosstalk, and Signal Integration. Front Immunol 10, 705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fafian-Labora JA, and O’Loghlen A (2020). Classical and Nonclassical Intercellular Communication in Senescence and Ageing. Trends Cell Biol 30, 628–639. [DOI] [PubMed] [Google Scholar]
- Feng F, Jin Y, Duan L, Yan Z, Wang S, Li F, Liu Y, Samet JM, and Wu W (2016). Regulation of ozone-induced lung inflammation by the epidermal growth factor receptor in mice. Environ Toxicol 31, 2016–2027. [DOI] [PubMed] [Google Scholar]
- Finkel T, and Holbrook NJ (2000). Oxidants, oxidative stress and the biology of ageing. Nature 408, 239–247. [DOI] [PubMed] [Google Scholar]
- Gao X, Colicino E, Shen J, Kioumourtzoglou MA, Just AC, Nwanaji-Enwerem JC, Coull B, Lin X, Vokonas P, Zheng Y, et al. (2019). Impacts of air pollution, temperature, and relative humidity on leukocyte distribution: An epigenetic perspective. Environ Int 126, 395–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasparrini A, Guo Y, Hashizume M, Lavigne E, Zanobetti A, Schwartz J, Tobias A, Tong S, Rocklov J, Forsberg B, et al. (2015). Mortality risk attributable to high and low ambient temperature: a multicountry observational study. Lancet 386, 369–375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghio AJ, Carraway MS, and Madden MC (2012). Composition of air pollution particles and oxidative stress in cells, tissues, and living systems. J Toxicol Environ Health B Crit Rev 15, 1–21. [DOI] [PubMed] [Google Scholar]
- Grandjean P, and Landrigan PJ (2014). Neurobehavioural effects of developmental toxicity. Lancet Neurol 13, 330–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hannum G, Guinney J, Zhao L, Zhang L, Hughes G, Sadda S, Klotzle B, Bibikova M, Fan JB, Gao Y, et al. (2013). Genome-wide methylation profiles reveal quantitative views of human aging rates. Mol Cell 49, 359–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horvath S (2013). DNA methylation age of human tissues and cell types. Genome biology 14, R115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horvath S, and Raj K (2018). DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat Rev Genet 19, 371–384. [DOI] [PubMed] [Google Scholar]
- Ijomone OM, Ijomone OK, Iroegbu JD, Ifenatuoha CW, Olung NF, and Aschner M (2020). Epigenetic influence of environmentally neurotoxic metals. Neurotoxicology 81, 51–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaiswal S, and Libby P (2020). Clonal haematopoiesis: connecting ageing and inflammation in cardiovascular disease. Nat Rev Cardiol 17, 137–144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssen BG, Munters E, Pieters N, Smeets K, Cox B, Cuypers A, Fierens F, Penders J, Vangronsveld J, Gyselaers W, et al. (2012). Placental mitochondrial DNA content and particulate air pollution during in utero life. Environ Health Perspect 120, 1346–1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssen BG, Saenen ND, Roels HA, Madhloum N, Gyselaers W, Lefebvre W, Penders J, Vanpoucke C, Vrijens K, and Nawrot TS (2017). Fetal Thyroid Function, Birth Weight, and in Utero Exposure to Fine Particle Air Pollution: A Birth Cohort Study. Environ Health Perspect 125, 699–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang C, Wang X, Li X, Inlora J, Wang T, Liu Q, and Snyder M (2018). Dynamic Human Environmental Exposome Revealed by Longitudinal Personal Monitoring. Cell 175, 277–291 e231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim M, and Benayoun BA (2020). The microbiome: an emerging key player in aging and longevity. Transl Med Aging 4, 103–116. [PMC free article] [PubMed] [Google Scholar]
- Kleinstreuer NC, Yang J, Berg EL, Knudsen TB, Richard AM, Martin MT, Reif DM, Judson RS, Polokoff M, Dix DJ, et al. (2014). Phenotypic screening of the ToxCast chemical library to classify toxic and therapeutic mechanisms. Nat Biotechnol 32, 583–591. [DOI] [PubMed] [Google Scholar]
- Kucab JE, Zou X, Morganella S, Joel M, Nanda AS, Nagy E, Gomez C, Degasperi A, Harris R, Jackson SP, et al. (2019). A Compendium of Mutational Signatures of Environmental Agents. Cell 177, 821–836 e816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamberts SW, van den Beld AW, and van der Lely AJ (1997). The endocrinology of aging. Science 278, 419–424. [DOI] [PubMed] [Google Scholar]
- Landrigan PJ, Fuller R, Acosta NJR, Adeyi O, Arnold R, Basu NN, Balde AB, Bertollini R, Bose-O’Reilly S, Boufford JI, et al. (2018). The Lancet Commission on pollution and health. Lancet 391, 462–512. [DOI] [PubMed] [Google Scholar]
- Lappalainen T, and Greally JM (2017). Associating cellular epigenetic models with human phenotypes. Nat Rev Genet 18, 441–451. [DOI] [PubMed] [Google Scholar]
- Lind PM, Salihovic S, and Lind L (2018). High plasma organochlorine pesticide levels are related to increased biological age as calculated by DNA methylation analysis. Environ Int 113, 109–113. [DOI] [PubMed] [Google Scholar]
- Liu C, Yavar Z, and Sun Q (2015). Cardiovascular response to thermoregulatory challenges. Am J Physiol Heart Circ Physiol 309, H1793–1812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Otin C, Blasco MA, Partridge L, Serrano M, and Kroemer G (2013). The hallmarks of aging. Cell 153, 1194–1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lunn RM, Blask DE, Coogan AN, Figueiro MG, Gorman MR, Hall JE, Hansen J, Nelson RJ, Panda S, Smolensky MH, et al. (2017). Health consequences of electric lighting practices in the modern world: A report on the National Toxicology Program’s workshop on shift work at night, artificial light at night, and circadian disruption. Sci Total Environ 607–608, 1073–1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madden CJ, and Morrison SF (2019). Central nervous system circuits that control body temperature. Neurosci Lett 696, 225–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marioni RE, Shah S, McRae AF, Chen BH, Colicino E, Harris SE, Gibson J, Henders AK, Redmond P, Cox SR, et al. (2015). DNA methylation age of blood predicts all-cause mortality in later life. Genome biology 16, 25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martens DS, Plusquin M, Cox B, and Nawrot TS (2019). Early Biological Aging and Fetal Exposure to High and Low Ambient Temperature: A Birth Cohort Study. Environ Health Perspect 127, 117001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martincorena I, Fowler JC, Wabik A, Lawson ARJ, Abascal F, Hall MWJ, Cagan A, Murai K, Mahbubani K, Stratton MR, et al. (2018). Somatic mutant clones colonize the human esophagus with age. Science 362, 911–917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messerlian C, Martinez RM, Hauser R, and Baccarelli AA (2017). ‘Omics’ and endocrine-disrupting chemicals - new paths forward. Nat Rev Endocrinol 13, 740–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mudway IS, Kelly FJ, and Holgate ST (2020). Oxidative stress in air pollution research. Free Radic Biol Med 151, 2–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munzel T, Sorensen M, Gori T, Schmidt FP, Rao X, Brook FR, Chen LC, Brook RD, and Rajagopalan S (2017). Environmental stressors and cardio-metabolic disease: part II-mechanistic insights. Eur Heart J 38, 557–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mutlu EA, Comba IY, Cho T, Engen PA, Yazici C, Soberanes S, Hamanaka RB, Nigdelioglu R, Meliton AY, Ghio AJ, et al. (2018). Inhalational exposure to particulate matter air pollution alters the composition of the gut microbiome. Environ Pollut 240, 817–830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neven KY, Saenen ND, Tarantini L, Janssen BG, Lefebvre W, Vanpoucke C, Bollati V, and Nawrot TS (2018). Placental promoter methylation of DNA repair genes and prenatal exposure to particulate air pollution: an ENVIRONAGE cohort study. Lancet Planet Health 2, e174–e183. [DOI] [PubMed] [Google Scholar]
- Nieuwenhuijsen MJ (2018). Influence of urban and transport planning and the city environment on cardiovascular disease. Nature reviews Cardiology 15, 432–438. [DOI] [PubMed] [Google Scholar]
- Nieuwenhuijsen MJ (2020). Urban and transport planning pathways to carbon neutral, liveable and healthy cities; A review of the current evidence. Environ Int 140, 105661. [DOI] [PubMed] [Google Scholar]
- Nowarski R, Jackson R, and Flavell RA (2017). The Stromal Intervention: Regulation of Immunity and Inflammation at the Epithelial-Mesenchymal Barrier. Cell 168, 362–375. [DOI] [PubMed] [Google Scholar]
- Nwanaji-Enwerem JC, Colicino E, Trevisi L, Kloog I, Just AC, Shen J, Brennan K, Dereix A, Hou L, Vokonas P, et al. (2016). Long-term ambient particle exposures and blood DNA methylation age: findings from the VA normative aging study. Environ Epigenet 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nwanaji-Enwerem JC, Weisskopf MG, and Baccarelli AA (2018). Multi-tissue DNA methylation age: Molecular relationships and perspectives for advancing biomarker utility. Ageing Res Rev 45, 15–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panagiotou N, Neytchev O, Selman C, and Shiels PG (2018). Extracellular Vesicles, Ageing, and Therapeutic Interventions. Cells 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters A, Kunzli N, Forastiere F, and Hoffmann B (2019). Promoting clean air: combating fake news and denial. Lancet Respir Med 7, 650–652. [DOI] [PubMed] [Google Scholar]
- Peters A, Veronesi B, Calderon-Garciduenas L, Gehr P, Chen LC, Geiser M, Reed W, Rothen-Rutishauser B, Schurch S, and Schulz H (2006). Translocation and potential neurological effects of fine and ultrafine particles a critical update. Part FibreToxicol 3, 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieters N, Janssen BG, Dewitte H, Cox B, Cuypers A, Lefebvre W, Smeets K, Vanpoucke C, Plusquin M, and Nawrot TS (2016). Biomolecular Markers within the Core Axis of Aging and Particulate Air Pollution Exposure in the Elderly: A Cross-Sectional Study. Environ Health Perspect 124, 943–950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prüss-Ustün A,; Wolf J; Corvalán C; Bos R; Neira M (2016). Preventing disease through healthy environments: a global assessment of the burden of disease from environmental risks (Geneva, Switzerland: ). [Google Scholar]
- Rojo de la Vega M, Chapman E, and Zhang DD (2018). NRF2 and the Hallmarks of Cancer. Cancer Cell 34, 21–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryter SW, Kim HP, Hoetzel A, Park JW, Nakahira K, Wang X, and Choi AM (2007). Mechanisms of cell death in oxidative stress. Antioxid Redox Signal 9, 49–89. [DOI] [PubMed] [Google Scholar]
- Ryu HW, Lee DH, Won HR, Kim KH, Seong YJ, and Kwon SH (2015). Influence of toxicologically relevant metals on human epigenetic regulation. Toxicol Res 31, 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin E, Colla S, Liesa M, Moslehi J, Muller FL, Guo M, Cooper M, Kotton D, Fabian AJ, Walkey C, et al. (2011). Telomere dysfunction induces metabolic and mitochondrial compromise. Nature 470, 359–365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salminen A, Kaarniranta K, and Kauppinen A (2012). Inflammaging: disturbed interplay between autophagy and inflammasomes. Aging 4, 166–175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sattler C, Moritz F, Chen S, Steer B, Kutschke D, Irmler M, Beckers J, Eickelberg O, Schmitt-Kopplin P, Adler H, et al. (2017). Nanoparticle exposure reactivates latent herpesvirus and restores a signature of acute infection. Part Fibre Toxicol 14, 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider A, Ruckerl R, Breitner S, Wolf K, and Peters A (2017). Thermal Control, Weather, and Aging. Curr Environ Health Rep 4, 21–29. [DOI] [PubMed] [Google Scholar]
- Shaughnessy DT, McAllister K, Worth L, Haugen AC, Meyer JN, Domann FE, Van Houten B, Mostoslavsky R, Bultman SJ, Baccarelli AA, et al. (2014). Mitochondria, Energetics, Epigenetics, and Cellular Responses to Stress. Environ Health Perspect 122, 1271–1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sies H, and Jones DP (2020). Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol 21, 363–383. [DOI] [PubMed] [Google Scholar]
- Smits HH, van der Vlugt LE, von Mutius E, and Hiemstra PS (2016). Childhood allergies and asthma: New insights on environmental exposures and local immunity at the lung barrier. Curr Opin Immunol 42, 41–47. [DOI] [PubMed] [Google Scholar]
- Stadtman ER, Van Remmen H, Richardson A, Wehr NB, and Levine RL (2005). Methionine oxidation and aging. Biochim Biophys Acta 1703, 135–140. [DOI] [PubMed] [Google Scholar]
- Stefanovic N, Flohr C, and Irvine AD (2020). The exposome in atopic dermatitis. Allergy 75, 63–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stone V, Miller MR, Clift MJD, Elder A, Mills NL, Moller P, Schins RPF, Vogel U, Kreyling WG, Alstrup Jensen K, et al. (2017). Nanomaterials Versus Ambient Ultrafine Particles: An Opportunity to Exchange Toxicology Knowledge. Environ Health Perspect 125, 106002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun L, Yu R, and Dang W (2018). Chromatin Architectural Changes during Cellular Senescence and Aging. Genes (Basel) 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suwanpradid J, Holcomb ZE, and MacLeod AS (2017). Emerging Skin T-Cell Functions in Response to Environmental Insults. J Invest Dermatol 137, 288–294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thurston GD, Kipen H, Annesi-Maesano I, Balmes J, Brook RD, Cromar K, De Matteis S, Forastiere F, Forsberg B, Frampton MW, et al. (2017). A joint ERS/ATS policy statement: what constitutes an adverse health effect of air pollution? An analytical framework. Eur Respir J 49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tost H, Reichert M, Braun U, Reinhard I, Peters R, Lautenbach S, Hoell A, Schwarz E, Ebner-Priemer U, Zipf A, et al. (2019). Neural correlates of individual differences in affective benefit of real-life urban green space exposure. Nat Neurosci 22, 1389–1393. [DOI] [PubMed] [Google Scholar]
- Touitou Y, and Point S (2020). Effects and mechanisms of action of light-emitting diodes on the human retina and internal clock. Environ Res 190, 109942. [DOI] [PubMed] [Google Scholar]
- Tumolo MR, Panico A, De Donno A, Mincarone P, Leo CG, Guarino R, Bagordo F, Serio F, Idolo A, Grassi T, et al. (2020). The expression of microRNAs and exposure to environmental contaminants related to human health: a review. Int J Environ Health Res, 1–23. [DOI] [PubMed] [Google Scholar]
- UN (2019). World Population Ageing 2019: Highlights (New York City, USA: United Nations; ). [Google Scholar]
- van den Bosch M, and Meyer-Lindenberg A (2019). Environmental Exposures and Depression: Biological Mechanisms and Epidemiological Evidence. Annu Rev Public Health 40, 239–259. [DOI] [PubMed] [Google Scholar]
- Vermeulen R, Schymanski EL, Barabasi AL, and Miller GW (2020). The exposome and health: Where chemistry meets biology. Science 367, 392–396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace DC (2010). Mitochondrial DNA mutations in disease and aging. Environmental and Molecular Mutagenesis 51, 440–450. [DOI] [PubMed] [Google Scholar]
- Ward-Caviness CK, Nwanaji-Enwerem JC, Wolf K, Wahl S, Colicino E, Trevisi L, Kloog I, Just AC, Vokonas P, Cyrys J, et al. (2016). Long-term exposure to air pollution is associated with biological aging. Oncotarget 7, 74510–74525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watts N, Amann M, Arnell N, Ayeb-Karlsson S, Belesova K, Boykoff M, Byass P, Cai W, Campbell-Lendrum D, Capstick S, et al. (2019). The 2019 report of The Lancet Countdown on health and climate change: ensuring that the health of a child born today is not defined by a changing climate. Lancet 394, 1836–1878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO (2005). Preventing chronic disease - a vital investment (Geneva, Switzerland: ). [Google Scholar]
- WHO (2012). State of the Science of Endocrine Disrupting Chemicals (Geneva, Switzerland:). [Google Scholar]
- Yamamoto M, Kensler TW, and Motohashi H (2018). The KEAP1-NRF2 System: a Thiol-Based Sensor-Effector Apparatus for Maintaining Redox Homeostasis. Physiol Rev 98, 1169–1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yauk C, Polyzos A, Rowan-Carroll A, Somers CM, Godschalk RW, Van Schooten FJ, Berndt ML, Pogribny IP, Koturbash I, Williams A, et al. (2008). Germ-line mutations, DNA damage, and global hypermethylation in mice exposed to particulate air pollution in an urban/industrial location. Proc Natl Acad Sci U S A 105, 605–610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin R, Mo J, Dai J, and Wang H (2017). Nickel(II) Inhibits Tet-Mediated 5-Methylcytosine Oxidation by High Affinity Displacement of the Cofactor Iron(II). ACS Chem Biol 12, 1494–1498. [DOI] [PubMed] [Google Scholar]
- Zoeller RT, Brown TR, Doan LL, Gore AC, Skakkebaek NE, Soto AM, Woodruff TJ, and Vom Saal FS (2012). Endocrine-disrupting chemicals and public health protection: a statement of principles from The Endocrine Society. Endocrinology 153, 4097–4110. [DOI] [PMC free article] [PubMed] [Google Scholar]


