Abstract
Objective
To investigate the efficacy and safety of angiogenesis inhibitors in the treatment of recurrent ovarian cancer (OC).
Methods
Electronic databases including PubMed, Web of Science, and the Cochrane Library were searched to find eligible studies until August 10, 2021. The data on overall survival (OS), progression-free survival (PFS), and objective response rate (ORR) were pooled. Furthermore, grade ≥ 3 adverse events (AEs) were investigated.
Results
A total of 13 studies with 3953 patients were included. Compared with control group, angiogenesis inhibitors resulted in significant improvement in PFS (hazard ratio (HR) = 0.61, 95%CI, 0.54–0.69), OS (HR = 0.88, 95%CI, 0.81–0.95), and ORR (odds ratio (OR) = 2.15, 95% CI, 1.74–2.65). However, angiogenesis inhibitors were associated with a higher risk of grade ≥ 3 AEs (relative risk (RR), 1.20, 95% CI, 1.04–1.38).
Conclusion
Angiogenesis inhibitors can improve ORR, PFS, and OS in patients with recurrent OC, but they can increase the incidence of AEs ≥ 3.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13048-022-01028-7.
Keywords: Recurrent ovarian cancer, Angiogenesis inhibitors, Overall survival, Progression-free survival, Objective response rate
Introduction
Ovarian cancer (OC) is the primary cause of death from gynecological cancers [1]. Since OC is not easy to find in the early stage, most patients are usually diagnosed in the advance stage, resulting in a low 5-year relative survival rate [2]. The mainstay of treatment for OC is cytoreductive surgery followed by platinum-based chemotherapy. Despite complete remission with the best treatment, approximately 70% of patients will relapse within 5 years [3, 4]. Therefore, OC still threatens the health of women worldwide.
Anti-angiogenic drugs have become a promising class of drugs for patients with OC. Anti-angiogenic drugs disrupt tumor vascularization and inhibit tumor cells from acquiring nutrition by damaging existing tumor blood vessels and preventing the development of new ones [5, 6]. Angiogenesis inhibitors have been shown in numerous clinical trials to benefit OC patients [7, 8]. As one of the angiogenesis inhibitors, bevacizumab has been shown to significantly improve PFS and ORR in recurrent OC patients. In addition, previous studies have shown that angiogenesis inhibitors are beneficial for the treatment of OC, but there is no systematic report on the treatment of recurrent OC with angiogenesis inhibitors [9]. Therefore, this study conducted a systematic review and meta-analysis of randomized clinical trials (RCTs) to study the efficacy and safety of angiogenesis inhibitors in patients with recurrent OC.
Methods
The Preferred Reporting Items for the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used to conduct the meta-analysis (Table S1).
Search strategy
The literature search is conducted through PubMed, Web of Science and Cochrane Library databases, and the search date is up to August 10, 2021. The following combined text and MeSH terms are used: "ovarian cancer", "ovarian tumor", "angiogenesis inhibitor", "Bevacizumab", "Aflibercept", "Avastin", "Sorafenib", "Sunitinib", "Imatinib", "vandetanib", "Nexavar", "Trebananib" and "Perifosine".
Study selection
Studies that met the following criteria were chosen: (1) Adult women with OC confirmed by histology; (2) these studies were clinical trials conducted to evaluate the efficacy and safety of angiogenesis inhibitors in patients with recurrent OC. (3) types of outcome measures are overall survival (OS), progression-free survival (PFS), objective response rate (ORR) and toxicity. (4) When the study derived from the same patients, the most complete and latest report of the trial was chosen. Duplicate articles, reviews, case reports, animal or cell experiments, single arm study and trials with insufficient data were all removed.
Data extraction and quality assessment
Two investigators (ZCM and ZWC) conducted the study selection process independently based on the inclusion and exclusion criteria. Extract the following data from each study: first author's name, publication year, trial design, patient status, age (years), sample size, follow-up time, etc. The main results were PFS, OS, ORR and grade 3 or higher adverse events (grade ≥ 3 AEs). Disagreements were resolved through debate and consensus during the research selection and data extraction processes. Cochrane Collaboration’s tool was used to assess the risk of bias.
Statistical analysis
The Review Manager 5.3 software (Cochrane Library, Oxford, UK) and STATA 14.0 (Stata Corp., College Station, TX) software were used for all statistical analysis. A generic inverse variance method was used to calculate the estimated pooled Hazard ratio (HR) for OS and PFS. The Mantel–Haenszel method was used to calculate the estimated pooled odds ratio (OR) and risk ratio (RR) with 95% confidence interval (CI) for pooled ORR and grade ≥ 3 AEs. The I2 statistics were used to assess the statistical heterogeneity between studies. When I2 > 50%, indicating that there is significant heterogeneity between the studies, and the random effects model was used; otherwise, the fixed effects model was used. In addition, Egger's test and funnel plot were used to assess the publication bias of the included studies.
Results
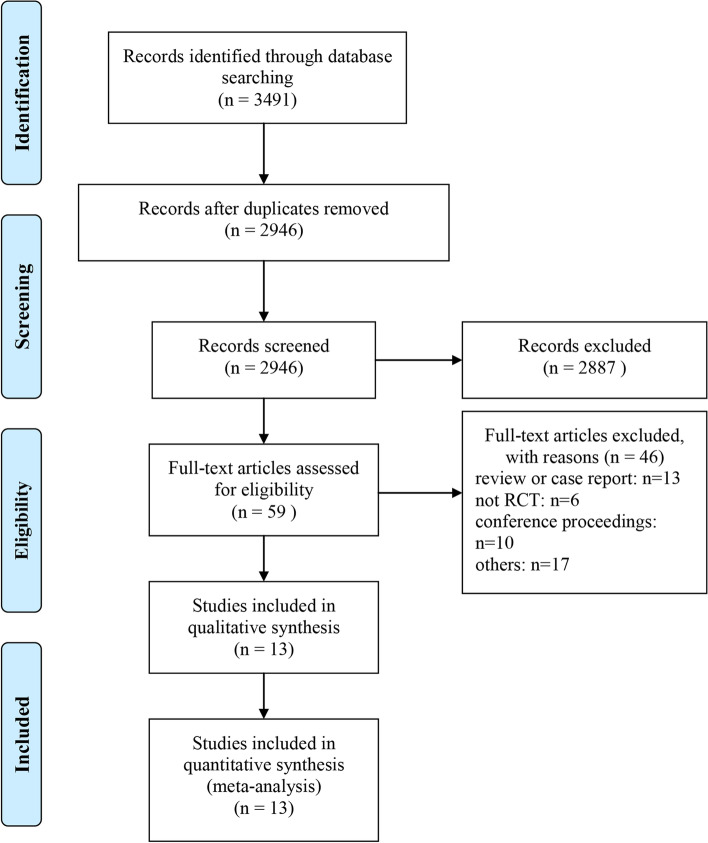
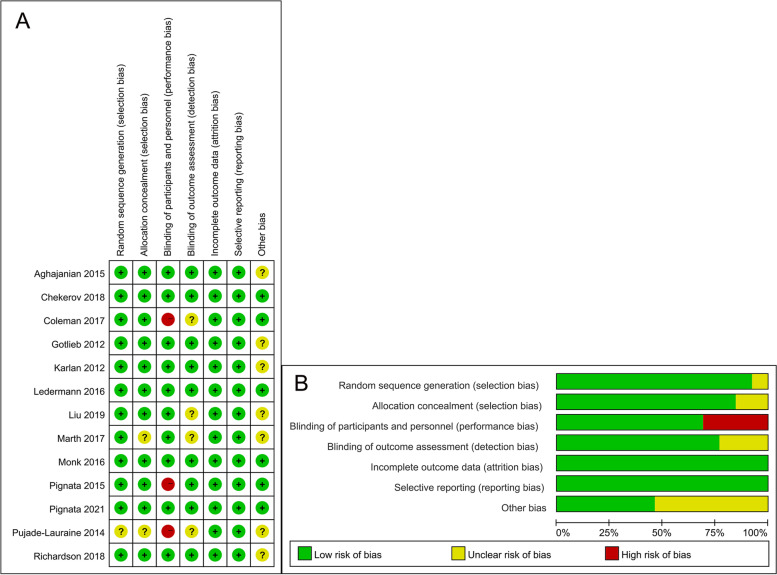
3491 articles were detected from all retrieved databases, with 2946 articles remaining after deduplication. Then, 2887 articles that did not meet the inclusion criteria were excluded through the title and abstract. Finally, after reading the full text, a total of 13 studies with 3953 patients were included [7–19] (Fig. 1). These 13 studies were published between 2012 and 2021 and involved six different angiogenesis inhibitors: Aflibercept (1 trial), Trebananib (3 trials), Bevacizumab (4 trials), Pazopanib (2 trials), Cediranib (2 trials), sorafenib (1 trial). The baseline characteristics of the included studies are shown in Table 1, and the risk of bias assessment is shown in Fig. 2.
Fig. 1.
The PRISMA Flow Diagram of Study Selection. The PRISMA diagram included searches of databases, registers, and other sources and the various reasons for the excluded articles
Table 1.
The basic characteristics of the included studies
| study | Agent type | Treatment arms | Dosage of angiogenesis inhibitors | Patients' status | Sample size | Median age | Median duration of follow-up (mo) |
|---|---|---|---|---|---|---|---|
| Gotlieb 2012 | VEGF inhibitor | Aflibercept vs. Placebo | 4 mg/kg every 2 weeks | Advanced chemoresistant ovarian cancer and recurrent symp tomatic malignant ascites; ECOG performance status ≤ 2 | 29/26 | 60.0/53.5 | / |
| Karlan 2012 | angiopoietin inhibitor | Trebananib + paclitaxel VS. placebo + paclitaxel | 10 mg/kg QW | Recurrent epithelial ovarian (FIGO stage II to IV), fallopian tube, or primary epithelial peritoneal cancer; ECOG performance status 0–1 | 53/55 | 62/59 | 5.5/5.4 |
| Pujade-Lauraine 2014 | VEGF inhibitor | Bevacizumab + Chemotherapy vs. Chemotherapy Alone | 10 mg/kg every 2 weeks or 15 mg/kg every 3 weeks | Platinum-resistant recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer; ECOG performance status 0–2 | 179/182 | 62/61 | 13.0/13.9 |
| Aghajanian 2015 | VEGF inhibitor | gemcitabine + carboplatin + bevacizumab vs. gemcitabine + carboplatin + placebo | 15 mg/kg every 3 weeks | Platinum-sensitive recurrent ovarian cancer (ie, epithelial ovarian, fallopian tube, or primary peritoneal carcinoma); ECOG performance status 0–1 | 242/242 | 60/61 | 9.6/8.4 |
| Pignata 2015 | VEGFR inhibitor | Paclitaxel + pazopanib vs. Paclitaxel only | 800 mg daily | Platinum-resistant epithelial ovarian, fallopian tube, or peritoneal cancer, stage IC–IV according to FIGO criteria; ECOG performance status 0–1 | 37/36 | 56/58 | 16.3/16.1 |
| Ledermann 2016 | VEGFR inhibitor | Platinum-based chemotherapy + Cediranib vs. Platinum-based chemotherapy + Placebo | 20 mg once-daily | Platinum-sensitive recurrent ovarian, fallopian tube, or primary peritoneal cancer after first-line platinumbased chemotherapy; ECOG performance status 0–1 | 164/118 | 62/62 | 19.5/19.5 |
| Monk 2016 | angiopoietin inhibitor | Paclitaxel + Trebananib vs. Paclitaxel + Placebo | 15 mg/kg once weekly | Recurrent partially platinum- sensitive or -resistant epithelial ovarian, primary peritoneal or fallopian tube cancer; GOG performance status ≤ 1 | 461/458 | 60/59 | 18/17.5 |
| Coleman 2017 | VEGF inhibitor | chemotherapy plus bevacizumab vs. chemotherapy | 15 mg/kg every 3 weeks | Platinum-sensitive, recurrent clinically evident epithelial ovarian, primary peritoneal, or fallopian tube cancer; COG performance status 0–2 | 337/337 | 59.5/60.6 | 49.6/49.6 |
| Marth 2017 | angiopoietin inhibitor | pegylated liposomal doxorubicin + Trebananib vs. pegylated liposomal doxorubicin + Placebo | 15 mg/kg every week | Platinum-resistant epithelial ovarian, peritoneal or fallopian tube cancer; ECOG performance status 0–2 | 114/109 | 61/60 | 12.4/12.4 |
| Chekerov 2018 | VEGFR inhibitor | Topotecan + sorafenib vs. Topotecan + placebo | 400 mg twice daily on days 6–15, repeated every 21 days | Platinum-resistant ovarian, peritoneal, or fallopian tube cancers; ECOG performance status 0–2 | 83/89 | 59/58 | 11.3/8.7 |
| Richardson 2018 | VEGFR inhibitor | Paclitaxel + pazopanib vs. Paclitaxel + Placebo | 800 mg orally daily | Recurrent or persistent epithelial ovarian, fallopian tube, or primary peritoneal cancer; COG performance status 0–1 | 52/54 | 61/61 | 17.7/17.7 |
| Liu 2019 | VEGFR inhibitor | Cediranib + olaparib vs. olaparib | 30 mg daily | relapsed high-grade serous or high-grade endometrioid ovarian cancer or a high-grade histology with a known germline BRCA mutation (gBRCAm); platinum-sensitive disease | 44/46 | 58.1/57.8 | 46/46 |
| Pignata 2021 | VEGF inhibitor | carboplatin-based doublet plus bevacizumab vs. carboplatin-based doublet intravenously | 10 mg/kg intravenous every 14 days | FIGO stage IIIB–IV platinum-sensitive ovarian cancer, fallopian tube carcinoma, or peritoneal carcinoma; ECOG performance status 0–2 | 203/203 | 61/60 | 20.1/20.1 |
Fig. 2.
Risk of bias assessment. A risk of bias graph for all the included RCTs. The items are scored ( +) low risk; (-) high risk; (?) unclear risk of bias. B risk of bias summary
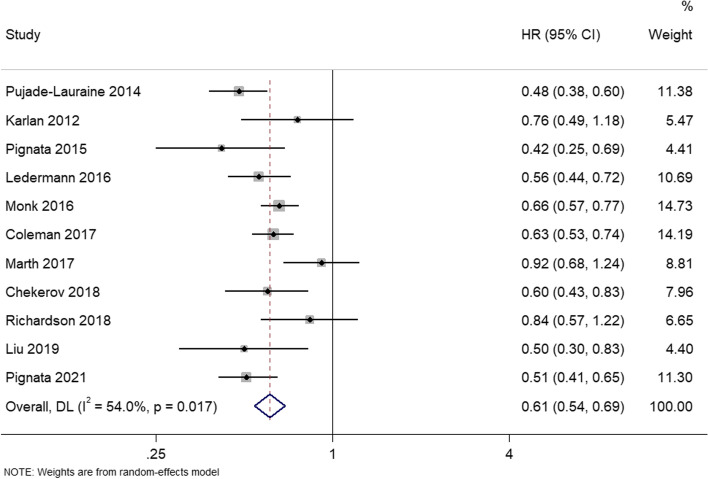
PFS
PFS was reported in 11 studies. There was heterogeneity between the studies (I2 = 54.0%; P = 0.017), so a random effects model was used for meta-analysis. Analysis showed that the angiogenesis inhibitors group had significant advantages in improving PFS, as compared to the control group (HR = 0.61, 95%CI, 0.54–0.69) (Fig. 3).
Fig. 3.
Forest plots of the meta-analysis on the effects of antiangiogenic drugs on progression free survival (PFS). Compared with the control group, angiogenesis inhibitor group can significantly improve PFS
OS
A total of 13 studies were integrated to analyze the OS. There was no heterogeneity between the studies (I2 = 0%; P = 0.597), so a fixed effects model was used for meta-analysis. The pooled result showed that angiogenesis inhibitors were significantly correlated with longer OS than control group (HR = 0.88, 95%CI, 0.81–0.95) (Fig. 4).
Fig. 4.
Forest plots of the meta-analysis on the effects of antiangiogenic drugs on overall survival (OS). Compared with the control group, angiogenesis inhibitor group can significantly improve OS
ORR
Eight studies reported reported ORR. There was no statistical heterogeneity between studies, and a fixed effects model was used for meta-analysis (I2 = 34.8%; P = 0.15). The meta-analysis showed that patients receiving angiogenesis inhibitors had higher ORRs compared to the control group (OR = 2.15, 95% CI, 1.74–2.65) (Fig. 5).
Fig. 5.
Forest plots of the meta-analysis on the effects of antiangiogenic drugs on objective response rate (ORR). Angiogenesis inhibitors had higher ORR compared to the control group
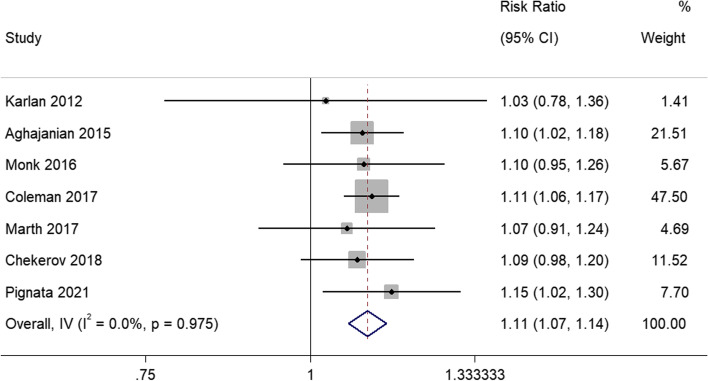
Grade ≥ 3 AEs
Seven studies reported the incidence of grade ≥ 3 AEs. Due to the results demonstrated heterogeneity between studies (I2 = 0%; P = 0.975), the meta-analysis was conducted using a random effects model. The pooled RR of grade ≥ 3 AEs showed that the angiogenesis inhibitors group had a greater incidence of grade ≥ 3 AEs than the control group (RR = 1.11, 95% CI, 1.07–1.14) (Fig. 6).
Fig. 6.
Forest plots of the meta-analysis on the effects of antiangiogenic drugs on grade ≥ 3 adverse effects (AEs). The angiogenesis inhibitors group had a greater incidence of grade ≥ 3 AEs than the control group
Subgroup analysis
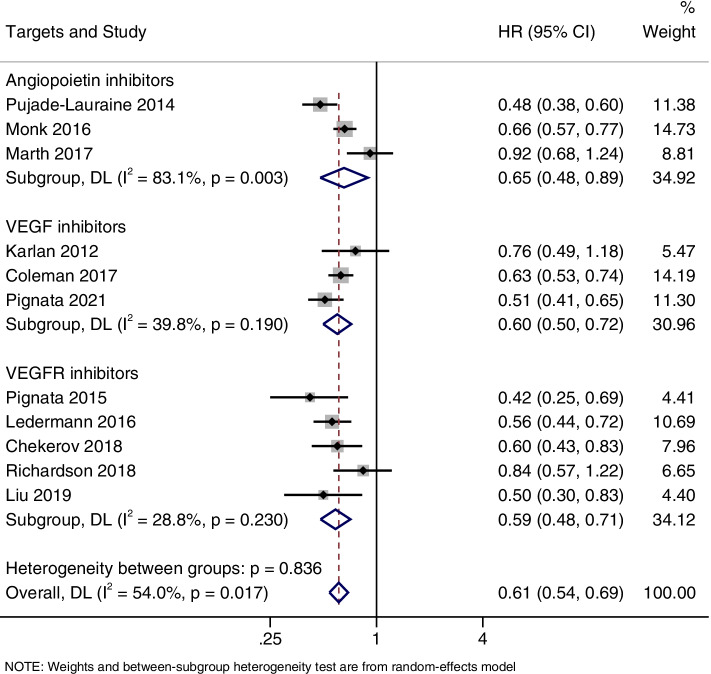
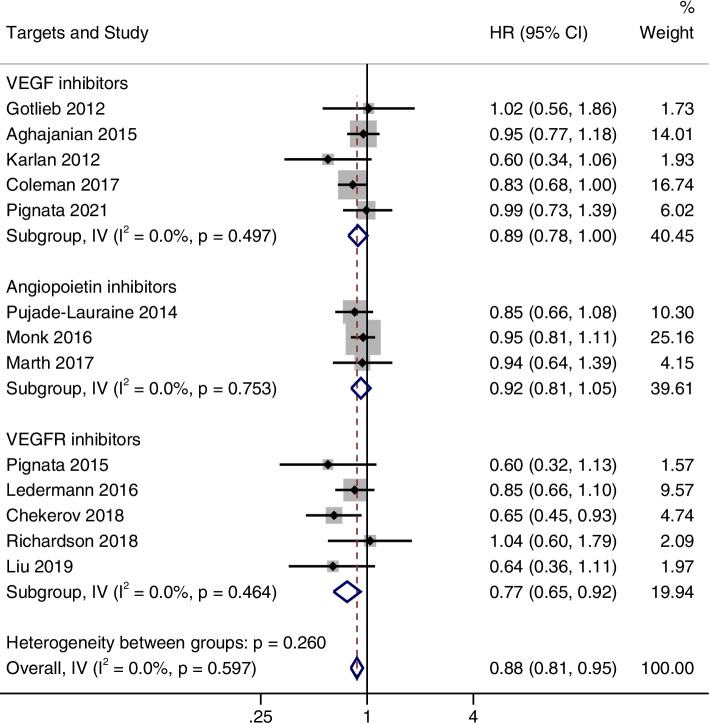
According to the drug target (vascular endothelial growth factor (VEGF) inhibitors include bevacizumab and aflibercept, VEGF receptor (VEGFR) inhibitors include pazopanib, cediranib, nintedanib, sorafenib, and angiopoietin inhibitors include trebananib), PFS, OS and ORR were subgroup analyzed. As shown in Fig. 7, the PFS improved significantly in all three subgroups (HR = 0.65, 95% CI, 0.48–0.89 for the angiopoietin inhibitor group; HR = 0.60, 95% CI, 0.50–0.72 for the VEGF inhibitors group; and HR = 0.59, 95% CI, 0.48–0.71 for the VEGFR inhibitors group). However, OS improvement was only observed in the VEGFR inhibitors group (HR = 0.77, 95% CI, 0.65–0.92), and there was no significant difference in OS between the two groups in angiopoietin inhibitor group (HR = 0.92, 95% CI, 0.81–1.05) and VEGF inhibitors group (HR, 0.89, 95% CI, 0.78–1.00) (Fig. 8). Furthermore, it was also found that ORR was significantly improved in all three subgroups (OR = 3.0, 95% CI, 1.92–4.68 for the angiopoietin inhibitor group; OR = 1.85, 95% CI, 1.41–2.42 for the VEGF inhibitors group; and OR = 2.36, 95% CI, 1.42–3.94 for the VEGFR inhibitors group) (Fig. 9).
Fig. 7.
Forest plots of the subgroup analysis on the effects of antiangiogenic drugs on PFS. VEGF: vascular endothelial growth factor, VEGFR: vascular endothelial growth factor receptor
Fig. 8.
Forest plots of the subgroup analysis on the effects of antiangiogenic drugs on OS. VEGF: vascular endothelial growth factor, VEGFR: vascular endothelial growth factor receptor
Fig. 9.
Forest plots of the subgroup analysis on the effects of antiangiogenic drugs on ORR. VEGF: vascular endothelial growth factor, VEGFR: vascular endothelial growth factor receptor
In addition, subgroup analyses were performed according to the treatment modality of angiogenesis inhibitors (monotherapy and combination therapy). Due to data limitations, we only performed a subgroup analysis of OS. It was found that the combination therapy of angiogenesis inhibitors can significantly improve OS compared with the control group (HR = 0.87, 95%CI, 0.57–0.66) (Figure S1). However, monotherapy with angiogenesis inhibitors was not significantly different from the control group (HR = 1.02, 95%CI, 0.56–1.86).
Publication bias
Visual inspection of the funnel plots were roughly symmetric (Figure S2). Egger’s test was used to further test the asymmetry of the funnel plots (Figure S3), and the results also showed that there was no publication bias in the study.
Discussion
Oncologists continue to face a formidable challenge in treating OC. Recurrent OC is almost always incurable, even when patients receive multiple lines of platinum and non-platinum therapy for advanced disease [20]. A promising novel therapeutic aimed at the tumor microenvironment has been proposed. Neovascularization is required for tumor growth and spread, and several antiangiogenic medicines have since been developed [21, 22]. The results of this meta-analysis showed that angiogenesis inhibitor therapy can significantly improve PFS, OS, and ORR in recurrent OC patients while increasing the risk of common AEs of grade ≥ 3.
According to the current results, angiogenesis inhibitor can significantly improve OS and PFS of the recurrent OC patients compared with the control group, which showed the similar results with the literature reported before [23]. Besides, an interesting finding of this study is that angiogenesis inhibitors can also significantly improve the ORR of patients with recurrent OC compared with the control group, which further proved the efficiency of the angiogenesis inhibitor in the treatment of recurrent OC.
It is reported that VEGF plays an important role in the formation of new blood vessels [24]. VEGF communicates with VEGFRs and activates downstream signaling pathways [25]. Another pathway makes use of angiopoietin, a tumor angiogenesis regulator [26]. According to the targets of drugs, we divided the studies into three groups for subgroup analysis. In this study, PFS in the VEGF inhibitors group, VEGFR inhibitors group and angiopoietin inhibitors group can significantly improve recurrent OC. However, only an improvement in OS was observed in the VEGFR inhibitors group. This is inconsistent with the previous meta-analysis results [23]. It may be because this study classified bevacizumab and aflibercept as the VEGF inhibitors group, while the previous study did not include aflibercept. In addition, this study also found that the combination of angiogenesis inhibitors and other drugs can significantly improve OS, but the monotherapy of angiogenesis inhibitors has no significant difference with the control group. Since only one of the included studies was monotherapy, more follow-up studies with larger sample sizes are needed to verify.
In addition, this study found that angiogenesis inhibitors are associated with a higher incidence of grade ≥ 3 AEs. This is consistent with previous research reports, which may be related to the mechanism of angiogenesis inhibitors [27, 28]. Angiogenesis inhibitors may cause vasodilation by increasing nitric oxide production in endothelial cells [29]. Therefore, angiogenesis inhibitors suppression may result in vasoconstriction and increased peripheral vascular resistance. Therefore, the usage of angiogenesis inhibitors might result in vascular abnormalities, which are the primary cause for the AEs of these drugs. To minimize the risks, it is necessary to monitor and manage these AEs during antiangiogenics therapy.
This study has some limitations. First, heterogeneity among studies reporting PFS may be related to differences in statistical quality, follow-up period, treatment modality, treatment duration, and ethnicity among patients receiving angiogenesis inhibitors. Secondly, despite the fact that the majority of the included studies were published in high-impact journals, there were study factors that could lead to bias, such as pharmaceutical industry sponsorship. Finally, this is a trial-level meta-analysis that is based on studies rather than individual patient data. Subgroup analyses based on cumulative high-, mid-, and low-dose inhibitors were not performed due to data limitations.
Conclusion
Treatment with angiogenesis inhibitors for recurrent OC patients was associated with significant improvements in PFS, OS, and ORR, but also with a higher incidence of grade ≥ 3 AEs. Our results clearly support the use of angiogenesis inhibitors in the clinical management of recurrent OC patients.
Supplementary Information
Acknowledgements
We would like to thank all authors who provided published data for our meta-analysis.
Abbreviations
- OC
Ovarian cancer
- RCTs
Randomized clinical trials
- PRISMA
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- OS
Overall survival
- PFS
Progression-free survival
- ORR
Objective response rate
- AEs
Adverse events
- OR
Odds ratio
- RR
Relative risk
- HR
Hazard ratio
Authors’ Contributions
ZCM and ZWC contributed to the conception, design, data acquisition, data analysis, and editing the manuscript. ZCM and ZWC together completed all analyses for this study. All authors read and approved the final manuscript.
Funding
This work was supported by the internal fund of Shengjing Hospital of China Medical University (Grant No. M0797).
Availability of data and materials
All data is available in this paper.
Declarations
Ethics approval and consent to participate
This study does not contain any studies with human participants or animals performed.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 2.Wang Z, Huang Y, Long L, et al. Apatinib treatment efficiently delays biochemical-only recurrent ovarian cancer progression. J Ovarian Res. 2021;14(1):91. doi: 10.1186/s13048-021-00843-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McGee J, Bookman M, Harter P, et al. Fifth Ovarian Cancer Consensus Conference: individualized therapy and patient factors. Ann Oncol. 2017;28(4):702–710. doi: 10.1093/annonc/mdx010. [DOI] [PubMed] [Google Scholar]
- 4.Hope JM, Blank SV. Current status of maintenance therapy for advanced ovarian cancer. Int J Womens Health. 2010;1:173–180. doi: 10.2147/ijwh.s4661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jaszai J, Schmidt MHH. Trends and Challenges in Tumor Anti-Angiogenic Therapies. Cells. 2019;8(9):1102. doi: 10.3390/cells8091102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abdalla AME, Xiao L, Ullah MW, Yu M, Ouyang C, Yang G. Current Challenges of Cancer Anti-angiogenic Therapy and the Promise of Nanotherapeutics. Theranostics. 2018;8(2):533–548. doi: 10.7150/thno.21674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ledermann JA, Embleton AC, Raja F, et al. Cediranib in patients with relapsed platinum-sensitive ovarian cancer (ICON6): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2016;387(10023):1066–1074. doi: 10.1016/S0140-6736(15)01167-8. [DOI] [PubMed] [Google Scholar]
- 8.Monk BJ, Poveda A, Vergote I, et al. Final results of a phase 3 study of trebananib plus weekly paclitaxel in recurrent ovarian cancer (TRINOVA-1): Long-term survival, impact of ascites, and progression-free survival-2. Gynecol Oncol. 2016;143(1):27–34. doi: 10.1016/j.ygyno.2016.07.112. [DOI] [PubMed] [Google Scholar]
- 9.Pujade-Lauraine E, Hilpert F, Weber B, et al. Bevacizumab combined with chemotherapy for platinum-resistant recurrent ovarian cancer: The AURELIA open-label randomized phase III Trial. Obstet Gynecol Surv. 2014;69(7):402–404. doi: 10.1097/01.ogx.0000452705.82050.e4. [DOI] [PubMed] [Google Scholar]
- 10.Aghajanian C, Goff B, Nycum LR, Wang YV, Husain A, Blank SV. Final overall survival and safety analysis of OCEANS, a phase 3 trial of chemotherapy with or without bevacizumab in patients with platinum-sensitive recurrent ovarian cancer. Gynecol Oncol. 2015;139(1):10–16. doi: 10.1016/j.ygyno.2015.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chekerov R, Hilpert F, Mahner S, et al. Sorafenib plus topotecan versus placebo plus topotecan for platinum-resistant ovarian cancer (TRIAS): a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncology. 2018;19(9):1247–1258. doi: 10.1016/S1470-2045(18)30372-3. [DOI] [PubMed] [Google Scholar]
- 12.Coleman RL, Brady MF, Herzog TJ, et al. Bevacizumab and paclitaxel-carboplatin chemotherapy and secondary cytoreduction in recurrent, platinum-sensitive ovarian cancer (NRG Oncology/Gynecologic Oncology Group study GOG-0213): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18(6):779–791. doi: 10.1016/S1470-2045(17)30279-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gotlieb WH, Amant F, Advani S, et al. Intravenous aflibercept for treatment of recurrent symptomatic malignant ascites in patients with advanced ovarian cancer: A phase 2, randomised, double-blind, placebo-controlled study. Lancet Oncol. 2012;13(2):154–162. doi: 10.1016/S1470-2045(11)70338-2. [DOI] [PubMed] [Google Scholar]
- 14.Karlan BY, Oza AM, Richardson GE, et al. Randomized, double-blind, placebo-controlled phase II study of AMG 386 combined with weekly paclitaxel in patients with recurrent ovarian cancer. J Clin Oncol. 2012;30(4):362–371. doi: 10.1200/JCO.2010.34.3178. [DOI] [PubMed] [Google Scholar]
- 15.Liu JF, Barry WT, Birrer M, et al. Overall survival and updated progression-free survival outcomes in a randomized phase II study of combination cediranib and olaparib versus olaparib in relapsed platinum-sensitive ovarian cancer. Ann Oncol. 2019;30(4):551–557. doi: 10.1093/annonc/mdz018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Marth C, Vergote I, Scambia G, et al. ENGOT-ov-6/TRINOVA-2: Randomised, double-blind, phase 3 study of pegylated liposomal doxorubicin plus trebananib or placebo in women with recurrent partially platinum-sensitive or resistant ovarian cancer. Eur J Cancer. 2017;70:111–121. doi: 10.1016/j.ejca.2016.09.004. [DOI] [PubMed] [Google Scholar]
- 17.Pignata S, Lorusso D, Joly F, et al. Carboplatin-based doublet plus bevacizumab beyond progression versus carboplatin-based doublet alone in patients with platinum-sensitive ovarian cancer: a randomised, phase 3 trial. Lancet Oncol. 2021;22(2):267–276. doi: 10.1016/S1470-2045(20)30637-9. [DOI] [PubMed] [Google Scholar]
- 18.Pignata S, Lorusso D, Scambia G, et al. Pazopanib plus weekly paclitaxel versus weekly paclitaxel alone for platinum-resistant or platinum-refractory advanced ovarian cancer (MITO 11): a randomised, open-label, phase 2 trial. Lancet Oncol. 2015;16(5):561–568. doi: 10.1016/S1470-2045(15)70115-4. [DOI] [PubMed] [Google Scholar]
- 19.Richardson DL, Sill MW, Coleman RL, et al. Paclitaxel With and Without Pazopanib for Persistent or Recurrent Ovarian Cancer: A Randomized Clinical Trial. JAMA Oncol. 2018;4(2):196–202. doi: 10.1001/jamaoncol.2017.4218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ding T, Tang D, Xi M. The survival outcome and complication of secondary cytoreductive surgery plus chemotherapy in recurrent ovarian cancer: a systematic review and meta-analysis. J Ovarian Res. 2021;14(1):93. doi: 10.1186/s13048-021-00842-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Coleman RL, Monk BJ, Sood AK, Herzog TJ. Latest research and treatment of advanced-stage epithelial ovarian cancer. Nat Rev Clin Oncol. 2013;10(4):211–224. doi: 10.1038/nrclinonc.2013.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cummings M, Freer C, Orsi NM. Targeting the tumour microenvironment in platinum-resistant ovarian cancer. Semin Cancer Biol. 2021;77:3–28. doi: 10.1016/j.semcancer.2021.02.007. [DOI] [PubMed] [Google Scholar]
- 23.Yi S, Zeng L, Kuang Y, et al. Antiangiogenic drugs used with chemotherapy for patients with recurrent ovarian cancer: a meta-analysis. Onco Targets Ther. 2017;10:973–984. doi: 10.2147/OTT.S119879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shibuya M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis: A Crucial Target for Anti- and Pro-Angiogenic Therapies. Genes Cancer. 2011;2(12):1097–1105. doi: 10.1177/1947601911423031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Abhinand CS, Raju R, Soumya SJ, Arya PS, Sudhakaran PR. VEGF-A/VEGFR2 signaling network in endothelial cells relevant to angiogenesis. J Cell Commun Signal. 2016;10(4):347–354. doi: 10.1007/s12079-016-0352-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Thurston G, Daly C. The complex role of angiopoietin-2 in the angiopoietin-tie signaling pathway. Cold Spring Harb Perspect Med. 2012;2(9):a006550. doi: 10.1101/cshperspect.a006650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Monk BJ, Minion LE, Coleman RL. Anti-angiogenic agents in ovarian cancer: past, present, and future. Ann Oncol. 2016;27(Suppl 1):i33–i39. doi: 10.1093/annonc/mdw093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Randall LM, Monk BJ. Bevacizumab toxicities and their management in ovarian cancer. Gynecol Oncol. 2010;117(3):497–504. doi: 10.1016/j.ygyno.2010.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Steeghs N, Gelderblom H, Roodt JO, et al. Hypertension and rarefaction during treatment with telatinib, a small molecule angiogenesis inhibitor. Clin Cancer Res. 2008;14(11):3470–3476. doi: 10.1158/1078-0432.CCR-07-5050. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data is available in this paper.