Abstract
Aims
Osteoarthritis (OA) is a common degenerative joint disease characterized by chronic inflammatory articular cartilage degradation. Long noncoding RNAs (lncRNAs) have been previously indicated to play an important role in inflammation-related diseases. Herein, the current study set out to explore the involvement of lncRNA H19 in OA.
Methods
Firstly, OA mouse models and interleukin (IL)-1β-induced mouse chondrocytes were established. Expression patterns of IL-38 were determined in the synovial fluid and cartilage tissues from OA patients. Furthermore, the targeting relationship between lncRNA H19, tumour protein p53 (TP53), and IL-38 was determined by means of dual-luciferase reporter gene, chromatin immunoprecipitation, and RNA immunoprecipitation assays. Subsequent to gain- and loss-of-function assays, the levels of cartilage damage and proinflammatory factors were further detected using safranin O-fast green staining and enzyme-linked immunosorbent assay (ELISA) in vivo, respectively, while chondrocyte apoptosis was measured using Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling (TUNEL) in vitro.
Results
IL-38 was highly expressed in lentivirus vector-mediated OA mice. Meanwhile, injection of exogenous IL-38 to OA mice alleviated the cartilage damage, and reduced the levels of proinflammatory factors and chondrocyte apoptosis. TP53 was responsible for lncRNA H19-mediated upregulation of IL-38. Furthermore, it was found that the anti-inflammatory effects of IL-38 were achieved by its binding with the IL-36 receptor (IL-36R). Overexpression of H19 reduced the expression of inflammatory factors and chondrocyte apoptosis, which was abrogated by knockdown of IL-38 or TP53.
Conclusion
Collectively, our findings evidenced that upregulation of lncRNA H19 attenuates inflammation and ameliorates cartilage damage and chondrocyte apoptosis in OA by upregulating TP53, IL-38, and by activating IL-36R.
Cite this article: Bone Joint Res 2022;11(8):594–607.
Keywords: Osteoarthritis, Long noncoding RNA H19, Interleukin-38, Tumour protein p53, Interleukin-36 receptor, Inflammatory response
Article focus
The current study was conducted to investigate the interaction between long noncoding RNA (lncRNA) H19, tumour protein p53 (TP53), and interleukin (IL)-38 in osteoarthritis (OA), employing the mouse model and the cellular model of OA in order to provide insights to understand the pathogenesis of OA and find a novel treatment regimen for OA.
Key messages
LncRNA H19 is highly expressed in OA and upregulates IL-38 by recruiting TP53.
IL-38 is highly expressed in OA and exerts anti-inflammatory properties.
IL-38 exerts its anti-inflammatory action by binding to IL-36R.
Strengths and limitations
The lncRNA H19/IL-38 axis alleviates and may be a therapeutic target for OA.
The lack of pre-existing data on the potential role of lncRNA H19 or IL-38 in OA progression made it difficult to provide further details on this topic.
Different stages of OA (early, intermediate, established OA) cannot be fully addressed in this study.
All issues (more in vivo and in vitro experiments needed to confirm our conclusion) raised by the reviewers are also limitations of this study; these issues could not be fully addressed by additional experiments.
Introduction
Osteoarthritis (OA) is one of the leading degenerative joint diseases affecting the diarthrodial joints, such as those of the hand, knee, and hip joints. 1 Patients plagued by OA commonly present with joint dysfunction, deformity, and pain, while the known risk factors of OA include sex, age, obesity, and heredity. 2,3 Although treatment approaches such as intra-articular therapy, exercise, and weight loss have allowed cost-effective care of OA, it is still imperative to develop methods to decrease the overall incidence rate of OA and attenuate the development of OA. 4 Clinically, the heterogeneous nature of OA remains challenging for the development of effective treatment of the disease. 5 Nevertheless, advances in this field of research have identified the involvement of chronic low-grade inflammatory reactions and innate and adaptive immune systems in the pathogenesis of OA. 6 Therefore, it would be prudent to explore the underlying mechanism of inflammatory response seen in OA, in order to advance the development of effective therapeutic options that would minimize destructive changes and permanent functional impairment in OA patients. 7
Long noncoding RNAs (lncRNAs) are well-established as versatile molecules that interact with RNA, proteins, or DNA to regulate numerous biological functions, while also exerting protective roles against excessive inflammatory responses. 8,9 For example, a prior study revealed that MALAT1 knockdown inhibits the inflammation and proliferation of synovial fibroblasts from OA patients. 10 Meanwhile, Pearson et al 11 identified several cartilage/chondrocyte inflammation-associated lncRNAs, including H19. Inherently, H19 serves as a maternally imprinted gene encoding a lncRNA, and changes in the expression of genes in the H19-insulin-like growth factor 2 (IGF2) imprint locus are associated with both Beckwith-Wiedemann syndrome and Russell-Silver syndrome. 12 Moreover, prior studies have also shown the association of lncRNA H19 with inflammatory conditions in intestinal epithelial regeneration. 13 Further owing to its involvement in the pathogenesis of psoriasis arthritis (PA), two particular studies highlighted lncRNA H19 as a potential molecular marker for OA. 14,15 Furthermore, lncRNA H19 possesses the ability to regulate tumour protein p53 (TP53) activation in bladder cancer, which holds particular therapeutic significance. 16 Meanwhile, TP53, also known as p53, is known to serve as a critical regulator of cellular activities, including senescence, cell cycle arrest, and apoptosis during the development of diseases. 17,18 Besides, a previous study came across upregulation of TP53 in knee OA and its involvement in OA pathogenesis. 19 The somatic mutations and dysfunction of TP53 are known to exert tumour-promoting effects on rheumatoid arthritis (RA) hyperplasia of fibroblast-like synoviocytes. 20 Moreover, another study established that mutant TP53 promotes inflammation by repressing secreted interleukin (IL)-1 receptor antagonist. 21 Additionally, IL-38, belonging to the IL-1 family, functions as a crucial mediator of innate immunity and inflammation. 22 In addition, IL-38 was previously found to suppress excessive inflammation in RA, which highlights its potential as a diagnostic marker for RA. 23 It is also noteworthy that IL-38 possesses certain anti-inflammatory properties. 24 For instance, IL-38 overexpression can inhibit inflammation in mouse models of arthritis and macrophages in vitro. 25 Furthermore, the anti-inflammatory effects of IL-38 have also been highlighted in regard to experimental arthritis. 26 Additionally, IL-36 receptor (IL-36R) is also a member of the group of IL-1 cytokines with wide expression in stromal cells, wherein IL-36 expression levels were documented in patients suffering from inflammatory arthritis. 27 In lieu of the aforementioned evidence, the current study set out to investigate the interaction between lncRNA H19, TP53, and IL-38 in OA employing mouse models and cellular models of OA, aiming to provide novel insight in the pathogenesis of OA.
Methods
Ethics
The current study was approved by the Ethics Committee of our hospital and was conducted in accordance with the Declaration of Helsinki issued in 1975. Signed written informed consents were obtained from each participant prior to the commencement of the study.
Animal experiments were performed based on the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health and approved by the Institutional Animal Care and Use Committee of our hospital. Extensive efforts were made to minimize both the number and suffering of the included animals.
We have included an ARRIVE checklist to show that we have conformed to the ARRIVE guidelines.
Collection of human cartilage specimens
Firstly, synovial fluid and cartilage tissues were collected from 37 OA patients (15 males and 22 females; mean calculated age 62.84 years (standard deviation (SD) 8.46)) during total knee arthroplasty and regarded as the OA group. Additionally, samples of synovial fluid and cartilage tissues of the knee joint were collected from 24 healthy people (10 males and 14 females; mean calculated age 60.67 years (SD 9.73)) while undergoing operations for traffic accidents at our hospital and regarded as the control group. None of the patients in the control group presented with a previous history of arthritis. Furthermore, patients with OA induced by degenerative articular cartilage were also included in our study. Patients presenting with other serious diseases, such as malignancies or diabetes in the past five years were excluded. After collection, the cartilage tissues from the two groups were graded. In accordance with the evaluation criteria of the modified Mankin scoring method, two independent observers (JL and FX) evaluated the cartilage tissues in a double-blind manner, and recorded and calculated means for the results. The mean Mankin score of the control group was calculated to be 0.625 (SD 0.495), and that of the OA group was 9.081 (SD 2.861).
Induction of mouse model of OA
75 C57BL/6 J mice (sham group n = 15, OA group n = 60), weighing between 11.55 and 17.50 g, aged four weeks, were procured from the Laboratory Animal Centre of Charles River (China) and housed in a specific pathogen-free animal facility. OA mouse models were induced in the inner half-moon board (destabilization of the medial meniscus (DMM)) of randomly selected C57BL/6 J mice (n = 10 to 15). Subsequently, the OA mice were subjected to dissection of right medial collateral and anterior cruciate ligaments, followed by transection of the right medial knee meniscus and total closure of articular cavity. Meanwhile, in the sham-operated mice, the skin on the right knee joint was resected, sutured layer by layer, and cut, followed by disinfection. After awakening, the mice were reared in cages with free activity, without any fixations of the operative limbs. All mice were euthanized in the fifth week, and then subjected to a knee surgery to collect the knee joint synovial fluid and cartilage tissues for subsequent experimentation.
Intra-articular injection of recombinant plasmid in OA mice
According to the published gene sequences of IL-38 (NM-153077.2) in the GenBank database, a plasmid PCDH-MSC-T2A-copGFP-MSCV was constructed to form the recombinant plasmid PCDH-SSTR1-T2A-copGFP-MSCV. Lentivirus was packed with correctly sequenced recombinant plasmid (Hanbio, China). Subsequently, the recombinant plasmid and pPACK packaged plasmid were cotransfected into the HEK293T cells (CRL-3216; American Type Culture Collection, USA). The HEK293T cells were then cultured in Dulbecco’s Modified Eagle Medium (DMEM) (10,566,016; Gibco, USA) containing 10% (vol:vol) fetal bovine serum (FBS; 10,270,160; Gibco) and 1% glutamine (25,030,081; Gibco) at 37°C in a humidified incubator (5% CO2). Afterwards, the cells were collected and concentrated. The virus titer of the cells was determined for subsequent experiments.
A total of 10 μl lentivirus (4 × 108 TU/ml) was injected into the knee joints of OA mice and sham-operated mice on day 7 and day 14 after the initial surgery. After five weeks, the mice were euthanized by means of CO2 inhalation, and knee joint tissues and synovial fluid were harvested. The OA mice were injected with lentivirus vector (LV)-negative control (NC) (lentivirus with empty vector), LV-IL-38 (lentivirus overexpressing IL-38), LV-overexpressed (oe)-H19, both LV-oe-H19 and LV-sh-IL-38, both LV-oe-H19 and LV-sh-TP53, or both LV-oe-H19 and LV-sh-IL-36R. Simultaneously, the sham-operated mice were injected with LV-NC or LV-IL-38. To further determine the expression patterns of IL-38 in OA mice at different periods, ten sham-operated mice and ten OA mice were randomly selected after three days, seven days, and 35 days of modelling to detect the concentration of IL-38 in knee joints.
Construction of cellular model of OA
Mouse chondrocytes (American Type Culture Collection) were cultured in DMEM (10,566,016; Gibco) containing 10% (vol:vol) FBS (10,270,160; Gibco) and 1% glutamine (25,030,081; Gibco) at 37°C in a humidified incubator (5% CO2). Subsequently, the chondrocytes were incubated with IL-1β (Sigma-Aldrich, USA) (10 ng/ml final concentration) at 37°C with 5% CO2 in air for 72 hours. Cells without IL-1β treatment served as control. Following incubation, a microscope was adopted to observe the presence of cytoplasmic retraction and intracellular vacuoles, in order to validate the successful model establishment, with the success rate for model establishment calculated to be 100%. Afterwards, the chondrocytes at the logarithmic period of growth were dispersed with trypsin into a cell suspension (5 × 104 cells/ml), which was then seeded in a six-well plate at a density of 2 ml/well. Next, the cells were incubated at 37°C overnight, and then infected with the following viruses (4 × 108 TU/ml): oe-NC, oe-H19, sh-NC, sh-H19, oe-NC + sh NC, oe-H19 + sh NC, oe-H19 + sh-TP53, oe-H19 + sh-IL-38, or oe-H19 + sh-IL-36R. Subsequent to 48 hours of infection, the cells were treated with IL-1β to induce cellular models of OA.
Enzyme-linked immunosorbent assay
Levels of inflammation-related factors IL-6 (Cat # BMS603-2, Invitrogen, Thermo Fisher Scientific, USA), IL-8 (Cat # KHC0081, Invitrogen), IL-17 (Cat # BMS6001, Invitrogen), IL-22 (Cat # BMS2047, Invitrogen), tumour necrosis factor (TNF)-α (Cat # BMS223HS, Invitrogen), interferon (IFN)-γ (Cat # abs552811-96T, Absin, China), and cartilage oligomeric matrix protein (COMP; Cat # AF5085, R&D Systems, USA) were determined in the joint synovial fluid of mice or the supernatant of culture medium in accordance with the manufacturer’s instructions provided in the ELISA kits (R&D Systems, USA). The experiment was conducted in triplicates to obtain the mean value.
Safranin O-fast green staining
Paraffin-embedded sections of mouse cartilage tissues were stained with fast green dye liquor for a five minutes, and then washed with 1% acetic acid for 15 seconds. Subsequently, the samples were stained with 0.1% safranin O dye liquor for five minutes, and dehydrated using 95% ethanol, absolute ethanol, and xylene. Images of the sections were obtained using the Zeiss Axiocam microscope camera (Zeiss, Germany). The tibial plateau was scored as per the Osteoarthritis Research Society International (OARSI) score (the middle of tibial plateau was used for the score statistics, and the mean value was obtained). Higher scores were indicative of more severe cartilage damage of OA in a score-dependent manner. 28 The experiment was conducted in triplicates to obtain the mean value.
Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling assay
Apoptosis in mouse cartilage tissue sections or mouse chondrocytes was detected using the one-step method in accordance with the instructions of the Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling (TUNEL) kit-fluorescein isothiocyanate kits (CA1040-50; Solarbio, China). Briefly, the cryostat tissue sections or cell smears were fixed with 4% paraformaldehyde for one hour, and incubated with 1% Triton-X 100 (Solarbio) on ice for a duration of two minutes. Subsequently, the cells were incubated with freshly prepared TUNEL detection buffer solution for one hour at 37°C under conditions devoid of light. Afterwards, the nucleus was stained with 4',6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich). The number of apoptotic cells was counted in randomly selected fields under a fluorescence microscope (AX70, Olympus, Japan). The experiment was conducted three times to obtain mean value.
RNA isolation and quantitation
Total RNA content was extracted from mouse cartilage tissues or mouse chondrocytes using the TRIzol reagent (15596026, Invitrogen). The obtained RNA was then reverse-transcribed into complementary DNA (cDNA) following the instructions provided on the reverse transcription kit (RR047A, Takara, Japan). Subsequently, SYBR Premix EX Taq kits (RR420A, Takara) were employed for sample loading. Quantitative reverse transcription polymerase chain reaction (RT-qPCR) analysis was performed with the help of an ABI7500 qPCR system (Applied Biosystems (ABI), USA), and all samples were tested in triplicate. All primers were synthesized by Shanghai Biotech (China) (Supplementary Table i). The results of the experiment were expressed as fold changes in gene expression by means of relative quantification (the 2-ΔΔCt method), with glyceraldehyde 3-phosphate dehydrogenase (GAPDH) serving as the internal control. The experiment was conducted in triplicates to obtain the mean value.
Western blot analysis
Total protein content was extracted from mouse cartilage tissues or mouse chondrocytes using the high-efficiency radioimmunoprecipitation assay (RIPA) lysis (R0010, Solarbio) in strict accordance with the manufacturer’s instructions. Subsequent to cell lysing at 4°C for 15 minutes, the cells were centrifuged at 15,000 r/min for a duration of 15 minutes. Next, the supernatant was extracted, and the protein concentration was determined using bicinchoninic acid (BCA) kits (20,201ES76, Yeasen Bio Technologies, China). Following sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE), the proteins were transferred onto a polyvinylidene fluoride membrane using the wet-transfer method, and further blocked with 5% bovine serum albumin (BSA) at room temperature for one hour. The membrane was probed with the diluted primary rabbit polyclonal IL-10 antibody (ab180898, dilution ratio of 1:100, Abcam, UK) in a shaker overnight at 4°C. Afterwards, the membrane was re-probed with the diluted horseradish peroxidase-labelled secondary antibody for one hour at room temperature. Following this, the membrane was developed with VILBER FUSION FX5 (VILBER LOURMAT, France). The ImageJ 1.48 u software (National Institutes of Health, USA) was adopted for protein quantitation, which was indicated by the ratio of relative gray value of the corresponding protein bands to that of the internal reference protein bands GAPDH (ab9485, 1:500, Abcam). Each experiment was repeated three times to obtain the mean value.
Immunofluorescence
The slides covered with mouse chondrocytes in the culture dish were rinsed thrice with PBS, for three minutes each time. Next, the slides were fixed with 4% paraformaldehyde for the duration of 15 minutes, and further permeated with 0.5% Triton X-100 (Sangon, China) at room temperature for 20 minutes. Subsequently, the slices were added with normal goat serum (Solarbio) and blocked at room temperature for 30 minutes before the blocking solution was absorbed. Following this, the slides were added with the primary anti-IL-38 antibodies (dilution ratio of 1:100; ab180898, Abcam) and incubated in a humid box overnight at 4°C. The following day, the slides were immersed in PBST thrice (each time for three minutes) prior to the excess liquid being absorbed with absorbent papers. Afterwards, the slides were incubated with diluted Alexa Fluor 488-labelled secondary antibody working solution dropwise in a humid box at 37°C for one hour. Finally, the slides were sealed with mounting solution containing anti-fluorescence quencher, and observed under a fluorescence microscope (IX73, Olympus).
RNA binding protein immunoprecipitation
Enrichment of lncRNA H19 by TP53 was detected with the help of RNA binding protein immunoprecipitation (RIP) kits (Millipore, USA). Briefly, OA mouse chondrocytes were first lysed with equal volume of lysis buffer on an ice bath for five minutes. The supernatant was then extracted following centrifugation at 14,000× g and 4°C for ten minutes. Part of the cell extract was used as the Input, while the remaining was probed with the TP53 antibody (2524 T, 1:500, Cell Signaling Technology, USA) for coprecipitation. The cells were further probed with an immunoglobulin G (IgG) antibody (ab190475, dilution ratio of 1:100, Abcam), which served as the NC. The RNA content was extracted from the samples and Input after protease K detachment for RT-qPCR analyses. The experiment was conducted in triplicate.
Dual-luciferase reporter gene assay
A dual-luciferase reporter gene assay was adopted to detect the activity of IL-38 promoter affected by lncRNA H19 following the co-transfection of oe-NC, oe-H19, sh-NC, or sh-H19 with luciferase reporter plasmid into OA cells. The cells were harvested and lysed 48 hours after transfection. Afterwards, the luciferase activity was measured using dual-luciferase detection kits (K801-200, BioVision, USA) with a dual-luciferase reporter gene analysis system (Promega, USA). Herein, the Renilla luciferase was used as an internal reference. The results of the analysis were expressed as the ratio of relative light unit (RLU) of firefly luciferase normalized to that of Renilla luciferase. This relative luciferase activity was indicative of target reporter gene activity. The experiment was conducted in triplicate to obtain the mean value.
Chromatin immunoprecipitation (ChIP) assay
OA mouse chondrocytes were fixed in formaldehyde for a duration of ten minutes for DNA-protein crosslinking. Chromatin fragments were obtained by means of ultrasonification (ten seconds on and ten seconds off in each cycle for 15 cycles). The supernatant was collected followed by centrifugation at 12,000× g for ten minutes at 4°C. The obtained supernatant was separated into two tubes, and then incubated with anti-mouse TP53 antibody (2524 T, dilution ratio of 1:500, Cell Signaling Technology) or NC IgG (ab190475, dilution ratio of 1:100, Abcam) at 4°C overnight. The following day, the DNA-protein complex was precipitated using protein agarose/Sepharose, followed by centrifugation at 12,000× g for five minutes. The resultant supernatant was discarded and the non-specific complex was washed, followed by de-crosslinking at 65°C overnight. Afterwards, the resultant DNA fragments were purified and extracted by phenol/chloroform. Finally, the binding of IL-38 promoter was detected using RT-qPCR analyses. The experiment was conducted in triplicate.
Receptor-binding assay
The binding of IL-38 to a group of soluble members of the IL-1 receptor family was further explored. Briefly, the extracellular domains of IL-1R type I, IL-1R accessory protein-Fc, IL-18 receptor α chain-Fc, and IL-36R-Fc (R&D Systems) were coated in a 96-well ELISA plate at 4°C overnight. Subsequently, the plate was blocked with PBS containing 1% BSA at 37°C for a duration of two hours. Further, recombinant mouse IL-38 (R&D Systems) was diluted with PBS containing 0.5% BSA and 0.05% Tween-20, and then incubated in a coated/blocked ELISA plate at 4°C overnight. The following day, the binding of IL-38 was detected using a biotin-labelled affinity purified goat anti-mouse IL-38 antibody (R&D Systems) and hydroxytryptophan-conjugated streptavidin. The experiment was conducted in triplicate to obtain the mean value.
Co-immunoprecipitation assay
OA mouse chondrocytes were lysed by means of a radioimmunoprecipitation assay (Thermo Fisher Scientific) with ultrasound at 4°C for 30 minutes. Subsequently, the cells were subjected to centrifugation at 13,000× g at 4°C for 30 minutes. The subsequent cell lysate was removed with the addition of Protein-A/G-agarose beads (40 μl/ml sample, Cat #IP05; Calbiochem, MilliporeSigma, USA), followed by 30 minutes of incubation at 4°C. The Protein-A/G-agarose beads were removed through centrifugation. Afterwards, the specific antibody was incubated with the previously obtained supernatant at 4°C overnight. Later, the Protein-A/G-agarose beads were added to the lysate and the mixture was incubated at 4°C for four hours with gentle shaking. The beads were mixed with loading buffer, followed by odium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and western blot analysis.
Electrophoretic mobility shift assay
Gel detection analyses were performed as per the instructions provided with the electrophoretic mobility shift assay (EMSA)/Gel-shift kits (GS002; Beyotime Biotechnology, China). Briefly, the extracted protein samples of anti-TP53 and control anti-IgG were purified, and then specifically incubated with probe-labelled IL-38 promoter-DNA to form specific protein complexes, followed by gel detection analysis. Subsequently, 6.5% non-denatured polyacrylamide gel was prepared for Super-Shift EMSA. The specific binding of the target protein and probe-labelled IL-38 promoter-DNA could delay the migration of the complexes. Super-Shift EMSA functioned in the reaction system, wherein the antibody and DNA/protein complex produces a reaction to form a complex to enlarge the complex, and the slow movement in the non-denaturing gel is capable of distinguishing the DNA/protein complex. All samples contained 8.7 × 10–7 M DNA, while the binding buffer was composed of 10 mM Tris (pH 7.6 at 20°C), 50 mM KCl, 1 mM dithiothreitol, and 10 μg/ml BSA. Samples were resolved on a 10% weight/volume (w/v) polyacrylamide gel cast and run in 40 mM Tris-Acetate, 2 mM EDTA, 50 mM KCl.
Isolation and purification of peripheral blood mononuclear cells
Following mice eyeball removal, blood samples were obtained from the optic vein, and added with heparin to prevent clotting, followed by Ficoll density gradient centrifugation. Subsequently, the blood samples were mixed with equal amounts of culture medium and double amounts of lymphocyte separation medium. Afterwards, the mixture was subjected to centrifugation at 400 to 600× g for 30 minutes to obtain a mononuclear cell layer. Next, the cells were rinsed twice with the culture medium and re-suspended in the complete medium at 4 × 106 cells/ml. The resultant cells were seeded in a 24-well plate at 2 × 106 cells/well, and then incubated at 37°C with 5% CO2. Following the removal of the supernatant, the wells were rinsed thrice with the medium to remove non-adherent cells. The adherent cells obtained were regarded as the purified peripheral blood mononuclear cells (PBMCs).
Statistical analysis
The SPSS 21.0 software (IBM, USA) was adopted for data analyses. Measurement data were summarized by mean (SD). All data were tested for normal distribution and equal variance. Comparisons between two groups were conducted using the independent-samples t-test, while comparisons among multiple groups were carried out by one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test. Correlation between lncRNA H19 and IL-38 in knee joint cartilage tissues in OA mice was analyzed using the Pearson correlation coefficient. Furthermore, data comparison among cell viability at different timepoints was conducted by two-way repeated measures ANOVA, followed by Bonferroni correction. A value of p < 0.05 was considered statistically significant.
Results
IL-38 is highly-expressed in OA
Firstly, mice models of OA were established by surgical destabilization of the medial meniscus. The degree of destabilization was subsequently evaluated by means of safranin O-fast green staining, the results of which illustrated that positive green staining was significantly increased in cartilages of OA mice relative to that in the sham-operated mice (p < 0.05, independent-samples t-test), which was indicative of substantial cartilage damage in OA mice (Figures 1a and 1b), consequently validating the successful induction of OA mouse models. Furthermore, the results of ELISA demonstrated that IL-38 levels in the synovial fluid were significantly increased in the OA mice in comparison to the sham-operated mice (Figure 1c). Similarly, IL-38 levels were observed to be elevated in chondrocytes subsequent to IL-1β exposure (Figure 1d). Additionally, consistent results were documented by RT-qPCR and western blot analysis in cartilage tissues (Figure 1e, Supplementary Figure aa), such that messenger RNA (mRNA) and protein expression levels of IL-38 were significantly increased in OA mice (both p < 0.05, independent-samples t-test).
Fig. 1.
Expression of interleukin (IL)-38 is upregulated in osteoarthritis (OA). a) Representative images of cartilage damage in OA mice assessed by safranin O-fast green staining (n = 10; 200×); b) Osteoarthritis Research Society International (OARSI) scores of cartilage damage in OA mice, *p < 0.05 versus sham-operated mice (n = 10). c) IL-38 level in synovial fluid from OA mice detected by enzyme-linked immunosorbent assay (ELISA). *p < 0.05 versus sham-operated mice at different timepoints (n = 5). d) IL-38 level in the supernatant of chondrocytes after IL-1β treatment detected by ELISA. *p < 0.05 versus control chondrocytes. e) Messenger RNA (mRNA) and protein expressions of IL-38 in cartilage tissues from OA mice (n = 10). *p < 0.05 versus sham-operated mice (n = 10). f) and g) mRNA (f) and protein (g) expression of IL-38 in chondrocytes after IL-1β treatment measured by quantitative reverse transcription polymerase chain reaction and western blot analysis. h) The expression of IL-38 in chondrocytes determined via immunofluorescence (400×). *p < 0.05 versus control chondrocytes. The measurement data were expressed as mean (standard deviation). Comparison between two groups was conducted by independent-samples t-test. The cell experiment was repeated three times independently. Ctr, control.
Additionally, we explored the expression patterns of IL-38 in the chondrocytes of the OA cell models using RT-qPCR, western blot analysis, and immunofluorescence analysis, the results of which showed that compared to the control chondrocytes, IL-38 expression levels were markedly increased in the OA chondrocytes (Figures 1f to 1h, Supplementary Figures ac and be). Altogether, these findings indicate that IL-38 was highly expressed in the articular cartilage of mice and chondrocytes with OA.
Exogenous IL-38 alleviates inflammatory response of OA mice
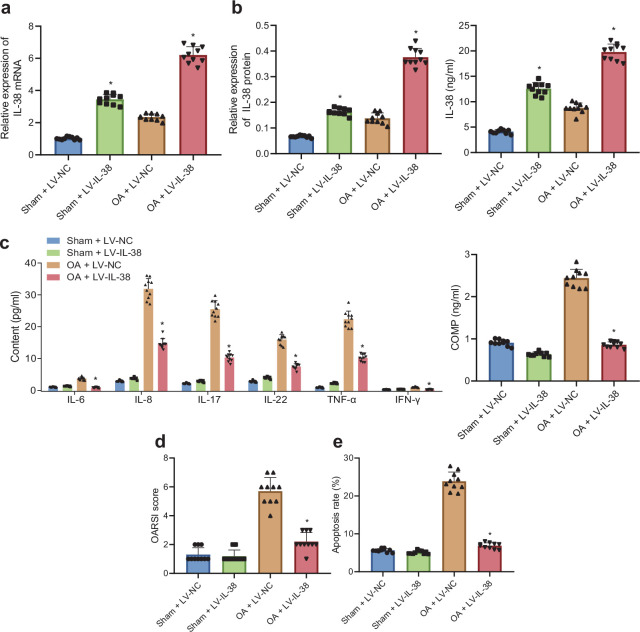
In order to further elucidate the effects of IL-38 on the inflammatory response of OA mice, OA mice and sham-operated mice were both injected with LV-IL-38 or LV-NC. Subsequent results of RT-qPCR and western blot analysis revealed a significant increase in IL-38 mRNA and protein levels in the cartilage tissues of OA mice and sham-operated mice in response to LV-IL-38 compared to LV-NC (all p < 0.05, independent-samples t-test, Figures 2a and 2b, Supplementary Figure ab). In addition, the results of ELISA illustrated that IL-38 levels in the synovial fluid were also augmented in OA mice and sham-operated mice following LV-IL-38 treatment relative to LV-NC treatment (Figure 2b). These cumulative results confirmed the overexpression of IL-38 following treatment with LV-IL-38.
Fig. 2.
Exogenous overexpression of interleukin (IL)-38 attenuates inflammatory response of osteoarthritis (OA) mice. a) Messenger RNA (mRNA) expression of IL-38 in knee joint cartilage tissues from OA mice and sham-operated mice in response to lentivirus vector (LV)-IL-38 or LV-negative control (NC), as determined by quantitative reverse transcription polymerase chain reaction (RT-qPCR). *p < 0.05 versus sham-operated mice injected with LV-NC; #p < 0.05 versus OA mice injected with LV-NC. b) IL-38 protein level in the knee joint cartilage tissues (left) and synovial fluid (right) from OA mice and sham-operated mice in response to LV-IL-38 or LV-NC as measured using western blot analysis and enzyme-linked immunosorbent assay (ELISA). *p < 0.05 versus sham-operated mice injected with LV-NC; #p < 0.05 versus OA mice injected with LV-NC. c) Levels of inflammation-related factors (IL-6, IL-8, IL-17, IL-22, tumour necrosis factor (TNF)-α, interferon (IFN)-γ, and cartilage oligomeric matrix protein (COMP)) in synovial fluid from OA mice and sham-operated mice in response to LV-IL-38 or LV-NC, tested using ELISA. d) The Osteoarthritis Research Society International (OARSI) score of cartilage damage in OA mice and sham-operated mice treated with LV-IL-38 or LV-NC identified by safranin O-fast green staining. e) Chondrocyte apoptosis in OA mice and sham-operated mice treated with LV-IL-38 or LV-NC detected using Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling (TUNEL) assay. In panels c) to f), *p < 0.05 versus OA mice with injection of LV-NC. The measurement data were expressed as mean (standard deviation). The cell experiment was repeated three times independently. Comparison between two groups was conducted by independent-samples t-test; n = 10.
Existing evidence suggests that inflammatory responses are heightened during OA, 29 hence we further determined the levels of inflammation-related factors (IL-6, IL-8, IL-17, IL-22, TNF-α, IFN-γ, and COMP) by means of ELISA. It was found that the expressions of inflammation-related factors in the OA mice injected with LV-NC were all increased in comparison to sham-operated mice injected with LV-NC; meanwhile, the levels of inflammation-related factors in the OA mice were significantly reduced following LV-IL-38 treatment compared to those with LV-NC treatment; whereas, no significant differences were observed in the expressions of inflammation-related factors between the sham-operated mice treated with LV-IL-38 and sham-operated mice treated with LV-NC (Figure 2c).
Additionally, the results of safranin O-fast green staining illustrated that LV-IL-38 treatment brought about improvement of cartilage damage in OA mice; the cartilage tissues in knee joints were observed to be normal in sham-operated mice injected with LV-38 or LV-NC; whereas, no significant differences were found among the sham-operated mice. The OARSI score of cartilage damage by safranin O-fast green staining is shown in Figure 2d, which revealed that injection with LV-38 reduced the score of cartilage damage. Moreover, the results of TUNEL assay demonstrated that in contrast to the sham-operated mice transduced with LV-NC, the OA mice transduced with LV-NC presented with an elevated number of apoptotic chondrocytes in the knee joint, whereas the number of apoptotic chondrocytes in the knee joint were reduced in the OA mice following the LV-IL-38 treatment compared to those treated with LV-NC (Figure 2e). Overall, these cumulative findings suggested that exogenous IL-38 could potentially alleviate the inflammatory response in OA mice.
lncRNA H19 binds to TP53 and upregulates IL-38
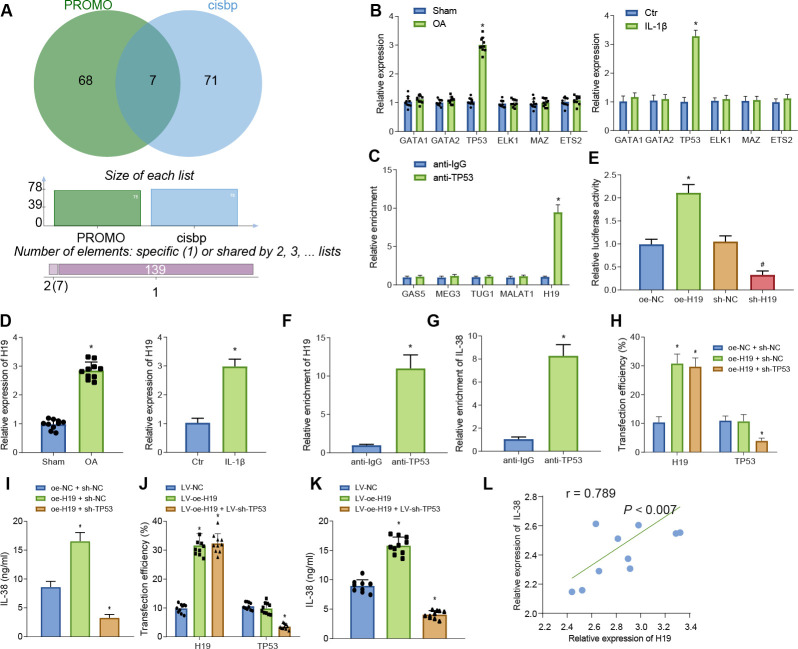
The PROMO 30 and CIS-BP 31 databases were adopted to predict the potential transcription factors of IL-38 and seven transcription factors, including ETS1, TP53, GATA2, GATA1, ELK1, MAZ, and ETS2 (Figure 3a). Further investigation revealed that among the seven transcription factors, only the mRNA expression of TP53 was differentially expressed in OA mice and cell models (Figure 3b). Current evidence further suggests that lncRNAs are capable of regulating gene functions via transcription factors; 32 therefore, the current study adopted the RNA-Society website 33 to screen lncRNAs that could bind to TP53, 34 the results of which revealed the following five potential lncRNAs: GAS5, MEG3, TUG1, MALAT1, and H19. Additionally, a RIP assay was performed to analyze the binding of these five potential lncRNAs to TP53, and it was found that lncRNA H19 induced the highest TP53 enrichment ( all p < 0.05, independent-samples t-test, Figure 3c). Consequently, the potential interaction was predicted among lncRNA H19, TP53, and IL-38.
Fig. 3.
Long noncoding RNA (lncRNA) H19 increases interleukin (IL)-38 expression through transcription factor TP53. a) Prediction of potential transcription factors regulating IL-38 by PROMO and CIS-BP databases. b) Messenger RNA (mRNA) expression of ETS1, TP53, GATA2, GATA1, ELK1, MAZ, and ETS2 in osteoarthritis (OA) mice and chondrocytes. *p < 0.05 versus sham-operated mice or control chondrocytes. c) TP53 enrichment of lncRNAs GAS5, MEG3, TUG1, MALAT1, and H19 determined by RNA binding protein immunoprecipitation (RIP) assay. Results are normalized to immunoglobulin G (IgG). *p < 0.05 versus anti-IgG. d) mRNA expression of lncRNA H19 in OA mice (n = 10) and chondrocytes. *p < 0.05 versus sham-operated mice or control chondrocytes. e) Relative luciferase activity of IL-38 promoter in OA chondrocytes after various treatments. *p < 0.05 versus OA chondrocytes treated with oe-negative control (NC), #p < 0.05 versus OA chondrocytes treated with sh-NC. f) Relative enrichment of lncRNA H19 by TP53 determined by RIP assay. *p < 0.05 versus anti-IgG. g) Relative enrichment of IL-38 by TP53 in OA chondrocytes determined by chromatin immunoprecipitation (ChIP) assay. *p < 0.05 versus anti-IgG. h) Transfection efficiency in OA chondrocytes detected by quantitative reverse transcription polymerase chain reaction (RT-qPCR). *p < 0.05 versus OA chondrocytes treated with both oe-NC and sh-NC. i) IL-38 level in OA chondrocytes after various treatments. *p < 0.05 versus OA chondrocytes co-treated with oe-NC and sh-NC. j) Transfection efficiency in OA mice detected by RT-qPCR. *p < 0.05 versus OA mice treated with lentivirus vector (LV)-NC. k) IL-38 levels in OA mice after various treatments. *p < 0.05 versus OA mice treated with LV-NC. l) Pearson analysis for the expression of lncRNA H19 and IL-38 in knee joint cartilage tissues from OA mice. The measurement data were expressed as mean (standard deviation). Comparison between two groups was conducted by independent-samples t-test. Comparison among multiple groups was conducted by one-way analysis of variance, followed by Tukey’s post hoc test; n = 10. The cell experiment was repeated three times independently. Ctr, control.
Furthermore, the results of RT-qPCR illustrated that the expression of lncRNA H19 was significantly higher in OA mice and chondrocytes (Figure 3d). Moreover, as revealed by dual-luciferase reporter gene assay, the luciferase activity was significantly higher in oe-H19 compared to that in oe-NC, whereas luciferase activity was lower in sh-H19 in comparison to sh-NC, which suggested that lncRNA H19 increased IL-38 promoter ( all p < 0.05, independent-samples t-test, Figure 3e). In addition, the RIP assay revealed that the enrichment of lncRNA H19 in anti-TP53 was much higher than that of anti-IgG (all p < 0.05, independent-samples t-test, Figure 3f). In addition, the results of ChIP assay showed that the enrichment of IL-38 in anti-TP53 was much higher in comparison to anti-IgG ( all p < 0.05, independent-samples t-test, Figure 3g).
To further determine the combined effect of H19 and TP53 on IL-38, we transduced cells with both oe-NC and sh-NC, both oe-H19 and sh-NC, or both oe-H19 and sh-TP53 in order to explore the combined effect of lncRNA H19 and TP53 on IL-38. Transduction efficiency in all groups was satisfactory for subsequent experiments (Figure 3h). IL-38 levels were significantly higher in response to the co-treatment with oe-H19 and sh-NC, while being markedly declined following co-treatment with oe-H19 and sh-TP53 compared to cells undergoing co-treatment with oe-NC and sh-NC (all p < 0.05, independent-samples t-test, Figure 3i). Additionally, further in vivo experiments were conducted, during which the OA mice were injected with LV-NC, LV-oe-H19, and both LV-oe-H19 and LV-sh-TP5, and the injection efficiency in all groups was verified (Figure 3j). Subsequent results demonstrated that IL-38 levels were significantly increased in the OA mice injected with LV-oe-H19, whereas diminished IL-38 levels were found in the OA mice injected with both LV-oe-H19 and LV-sh-TP53 compared to those injected with LV-NC (Figure 3k), and these findings were consistent with the findings previously obtained during in vitro experimentation.
Furthermore, the Pearson correlation coefficient demonstrated the presence of a positive correlation between the expression of lncRNA H19 and IL-38 in knee joint cartilage tissues obtained from the OA mice (Figure 3l). Meanwhile, the results of RT-qPCR illustrated that the expression levels of H19 and TP58 were both markedly upregulated in cartilage samples of OA patients compared to those in healthy subjects (Supplementary Figures ba and bb), highlighting the possible combined effect of H19 and TP58. Moreover, IL-38 levels in the synovial fluid obtained from OA patients were found to be higher in comparison to healthy subjects (all p < 0.05, independent-samples t-test, Supplementary Figure bc). Furthermore, IL-38 mRNA expression levels in the cartilage tissues were also established to be elevated in OA patients compared to their healthy counterparts ( all p < 0.05, independent-samples t-test, Supplementary Figure bd; Supplementary Table ii).
Altogether, the above-mentioned findings indicate that lncRNA H19 binds to TP53, leading to an increase in IL-38 expression in OA.
LncRNA H19 inhibits inflammation in OA by upregulating IL-38, TP53, and IL-36R
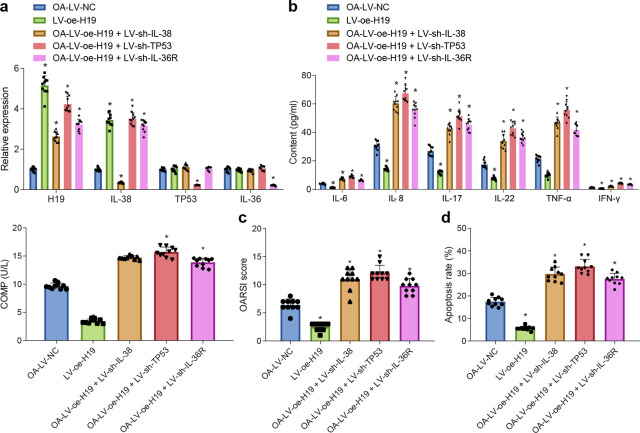
Prior studies have reported that IL-38 can specifically bind to IL-36R in human PBMCs, wherein IL-38 exhibits receptor antagonistic activity similar to IL-36R antagonist to inhibit the binding of IL-36 and IL-36R, thereby inhibiting inflammation. 35 Accordingly, the current study further investigated whether lncRNA H19 affects the inflammatory response of OA by regulating the expression of IL-38, and also explored whether the role of IL-38 in OA mice was associated with IL-36R. Subsequently, LV-NC, LV-oe-H19, combined LV-oe-H19 and LV-sh-IL-38, combined LV-oe-H19 and LV-sh-TP53, or combined LV-oe-H19 and LV-sh-IL-36R were transfected into the OA mice, and transduction efficiency was validated by means of RT-qPCR for further experimentation (Figure 4a).
Fig. 4.
Elevated long noncoding RNA (lncRNA) H19 exerts anti-inflammation effects on osteoarthritis (OA) by promoting interleukin (IL)-38, tumour protein p53 (TP53), and IL-36 receptor (IL-36R). a) Transfection efficiency in knee joint cartilage tissues from OA mice detected by quantitative reverse transcription polymerase chain reaction (RT-qPCR). b) Levels of inflammation-related factors (IL-6, IL-8, IL-17, IL-22, TNF-α, interferon (IFN)-γ, and cartilage oligomeric matrix protein (COMP)) in synovial fluid from OA mice with various treatments. c) The Osteoarthritis Research Society International (OARSI) score of cartilage damage in OA mice with various treatments identified by safranin O-fast green staining. d) Chondrocyte apoptosis after various treatments determined by Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling (TUNEL) assay. *p < 0.05 versus OA mice treated with lentivirus vector (LV)-negative control (NC); #p < 0.05 versus OA mice treated with LV-oe-H19. The cell experiment was repeated three times independently. The measurement data were expressed as mean (standard deviation). One-way analysis of variance was used for data comparison among multiple groups, followed by Tukey’s post hoc test; n = 10.
The results of the analysis illustrated that the levels of IL-6, IL-8, IL-17, IL-22, TNF-α, IFN-γ, and COMP were all reduced by LV-oe-H19, while being increased by the combinations of LV-oe-H19 and LV-sh-IL-38, LV-oe-H19 and LV-sh-TP53, as well as LV-oe-H19 and LV-sh-IL-36R in comparison to LV-NC in synovial fluid in OA mice ( all p < 0.05, independent-samples t-test, Figure 4b). Furthermore, it was observed that cartilage damage and chondrocyte apoptosis were reduced by LV-oe-H19 and increased the combinations of LV-oe-H19 and LV-sh-IL-38, LV-oe-H19 and LV-sh-TP53, as well as LV-oe-H19 and LV-sh-IL-36R relative to LV-NC in the OA mice ( all p < 0.05, independent-samples t-test, Figures 4c and 4d). Altogether, these findings suggest that the anti-inflammatory effect of lncRNA H19 in OA was achieved through the upregulation of IL-38, TP53, and IL-36R.
LncRNA H19/IL-38 axis exerts anti-inflammatory effects in PBMCs isolated from OA mice by binding to IL-36R in vitro
To further explore the binding sites of IL-38 and IL-36R, the receptor-binding (Figure 5a) and Co-IP (Figure 5b) assays revealed that IL-38 bound to IL-36R. Subsequently, OA chondrocytes were treated with both oe-NC and sh-NC, both oe-H19 and sh-NC, both oe-H19 and sh-IL-38, or both oe-H19 and sh-IL-36R for a duration of 48 hours. Transduction efficiency was found to be sufficient for subsequent experiments (Figure 5c). The results of ELISA illustrated that the levels of IL-17, IL-22, and IFN-γ were reduced following co-treatment with oe-H19 and sh-NC in comparison to treatment with combined oe-NC and sh-NC, whereas IL-17, IL-22, and IFN-γ levels were found to be increased by combined oe-H19 and sh-IL-38 and combined oe-H19 and sh-IL-36R when compared to combined oe-H19 and sh-NC (p = 0.012, independent-samples t-test, Figure 5d).
Fig. 5.
Long noncoding RNA (lncRNA) H19/interleukin (IL)-38 axis involving IL-36 receptor (IL-36R) harbours the anti-inflammatory property in osteoarthritis (OA) in vitro. a) Binding between mouse IL-38 and IL-36R in peripheral blood mononuclear cells (PBMCs) analyzed by receptor-binding assay. b) Binding between IL-38 and IL-36R in PBMCs detected by co-immunoprecipitation assay. c) Transfection efficiency detected by quantitative reverse transcription polymerase chain reaction. d) Levels of pro-inflammatory factors in PBMCs after various treatments. In panels c) and d), *p < 0.05 versus co-treatment of oe-negative control (NC) and sh-NC; #p < 0.05 versus co-treatment of oe-H19 and sh-NC. The measurement data were expressed as mean (standard deviation). One-way analysis of variance was used for data comparison among multiple groups, followed by Tukey’s post hoc test. Comparison of IL-38 expression among multiple groups was analyzed by two-way analysis of variance, followed by Bonferroni correction. The cell experiment was repeated three times independently. GFP, green fluorescence protein; IFN, interferon; OD, optical density.
To further validate the combination of TP53 and IL-38 promoter sites, we adopted gel electrophoresis mobility shift assay (EMSA), the results of which demonstrated that anti-TP53 and IL-38 promoter-DNA presented with significant combination between TP53 and IL-38 promoter-DNA, forming a large protein-DNA complex; relatively large bands were observed in gel displacement analysis (all p < 0.05, independent-samples t-test), compared with IL-38 promoter-DNA in the presence or absence of anti-IgG. However, it was found that IL-38 promoter-DNA in the presence or absence of anti-IgG exhibited no combined protein, and only IL-38 promoter-DNA molecular weight bands were observed in the displacement result (Supplementary Figure c). Altogether, the aforementioned results indicate that inflammation in OA is suppressed following the binding of lncRNA H19/IL-38 axis to IL-36R in vitro.
Discussion
OA, the most prevalent joint disease, is characterized by the progressive degeneration and loss of articular cartilage. 36 Currently, treatment strategies against OA focus on pain relief and disability management, and are plagued by limited efficiency and numerous side effects. 37 The diligent work of our peers has shown that several lncRNAs, including lncRNA H19, are implicated in the pathogenesis of OA. 38 Therefore, the current study set out to obtain a deeper understanding of the underlying mechanisms involved in the progression of OA and to provide further insights that can advance the development of potential novel therapeutic targets against OA. Ultimately, the obtained findings showed that lncRNA H19 upregulated IL-38, which bound to IL-36R and brought about suppression of knee joint inflammation in OA.
Firstly, our findings illustrate that lncRNA H19 was highly expressed in OA. H19, an imprinted gene, is well known for its extensive roles in the process of cellular differentiation. 39 Additionally, a prior study highlighted lncRNA H19 as an oncogenic gene in various cancers, wherein overexpression of H19 was closely associated with tumorigenesis and cancer progression. 40 Moreover, our findings are in line with a previous study which documented upregulation of lncRNA H19 in OA tissues relative to normal tissues. 15 Similarly, elevated lncRNA H19 was previously observed in OA cartilage, and further suggested to serve as a metabolic link in cultured chondrocytes and cartilage. 14 Together, our findings and existing evidence cumulatively provide strong evidence that lncRNA H19 is highly expressed in OA, and might be potentially implicated in the pathogenesis of OA. Different from these studies, subsequent experiments in the current study also revealed that the lncRNA H19 promoted inflammation response in OA via interactions between TP53, IL-38, and IL-36R.
Subsequent experimentation in our study further revealed that IL-38 was highly expressed in OA, such that lncRNA H19 resulted in the upregulation of IL-38. Unsurprisingly, a number of studies have uncovered the involvement of IL-38 in several autoimmune diseases. 41 For instance, a prior study came across elevated IL-38 expression levels in mice with arthritis induced by collagen as well as in patients with RA, 42 and further suggested that IL-38 exerts anti-protective effects on cartilage and bone destruction in collagen-induced arthritis. Importantly, increased expression levels of IL-38 were previously reported in patients with RA. 23 Furthermore, overexpression of IL-38 is known to exert anti-inflammatory properties in a mouse model of arthritis through articular injection of adeno-associated virus 2/8 encoding IL-38. 25 Nevertheless, there has been no report on the activation of IL-38 in OA. Expanding our existing knowledge on the same, our findings further evidenced the anti-inflammatory properties of IL-38 and H19, owing to the fact that IL-38 brought about suppressed levels of IL-6, IL-8, IL-17, IL-22, TNF-α, IFN-γ, and COMP. Additionally, there is a plethora of evidence that highlights the essential role of inflammation in OA pathology. For example, a positive link between COMP expression and the risk of OA has been established by a previous study. 43 Furthermore, regional upregulation of ILs, TNF-α, and IFN-γ is established to serve as inflammatory mediators in OA. 44 Elaborating on the same, one study has also shown that lower levels of pro-inflammatory cytokines such as IL-6, IL-8, TNF-α, and IFN-γ in OA chondrocytes lead to alleviation of OA symptoms. 45 Additionally, a growing volume of studies have documented the anti-inflammatory role of IL-38 in various conditions. 46-48 In particular, one prior study validated that IL-38 exerts its anti-inflammatory effects via downregulation of IL-8, IL-17, IL-22, IL-6, and TNF-α, which is in accordance with our findings. 24,25 Also, Mora et al 49 observed that loss of IL-38 in apoptotic cells can precipitate increased levels of IL-6 and IL-8 in co-cultured human primary macrophages.
Additionally, we further explored the mechanisms by which lncRNA and IL-38 are involved in OA as part of the current study, and uncovered the involvement of the transcription regulator TP53 in OA. According to a previous study, TP53 possesses the ability to cause cell apoptosis or cell growth arrest in response to various stress signals. 50 Meanwhile, the regulatory function of TP53 on gene expression is influenced by the interaction between TP53 and long intergenic noncoding RNAs. 51 It is also noteworthy that TP53 was previously revealed to be involved in the protective effect of lncRNA H19 in subarachnoid haemorrhage. 52 Moreover, mutant TP53 is capable of promoting inflammation by repressing secreted IL-1 receptor antagonist, which includes IL-38. 21,22 In addition, our findings displayed that TP53 was upregulated in OA mouse models and OA cell models relative to sham-operated mice or control cells. Interestingly, a previous study has further shown that p53 levels are significantly higher in synovial tissues of patients with early and long-term rheumatoid arthritis as compared to those of patients with reactive arthritis and OA (all p < 0.05, independent-samples t-test). 53 Together, these findings are indicative of the various roles of p53 in different types of arthritis. Moreover, another investigation demonstrated that loss of p53 in cancer cells induces WNT ligand secretion, which further stimulates IL-1β production of tumour-associated macrophages, thus promoting systemic inflammation. 54 However, inhibition of WNT secretion by p53-deleted cancer cells through pharmacological and genetic means can reverse IL-1β production of macrophages and inhibit subsequent neutrophil inflammation, thereby attenuating metastasis formation. 54 Also, an investigation focusing on atherosclerosis uncovered that IL-37 (IL-1 family member 7) bound to the IL-18Ra chain to downregulate p53 through intracellular mechanisms, which orchestrated inflammatory cytokines mediating atherosclerosis. 55 Furthermore, the IL-1 family factors are also involved in the pathogenesis of numerous chronic diseases, including inflammatory bowel disease, RA, and various autoimmune and autoinflammatory diseases. 56,57 Elaborating on existing knowledge, our findings revealed that lncRNA H19 activated IL-38 by binding to TP53, to further promote IL-38 binding to IL-36R. All of the aforementioned findings were concurrent with our results suggesting that TP53 was implicated in lncRNA H19-mediated anti-inflammatory effects in OA. However, at present, the specific interaction between p53 and IL-1 family members in OA remains elusive, and will be explored in future studies. Furthermore, we observed that IL-38 bound to IL-36R in PBMCs, and through this mechanism it exerts its anti-inflammatory effects. This finding was consistent with a previous study, proving that IL-38 binds to IL-36R and plays a functional role as an IL-36R antagonist, thereby suppressing the binding between IL-36 and IL-36R. 35 Meanwhile, it is also known that IL-36 could be targeted by blockage of the proinflammatory agonists or enhancement of the physiological inhibitory feedback on the antagonists IL-37 and IL-38-mediated inflammation. 58 Moreover, skin inflammation can be inhibited by blocking the IL-36/IL-36R auto-stimulatory loop. 59 Additionally, it has been documented that TGF-β type 2 receptor-orchestrated IL-36 family may serve as a therapeutic target for OA. 60
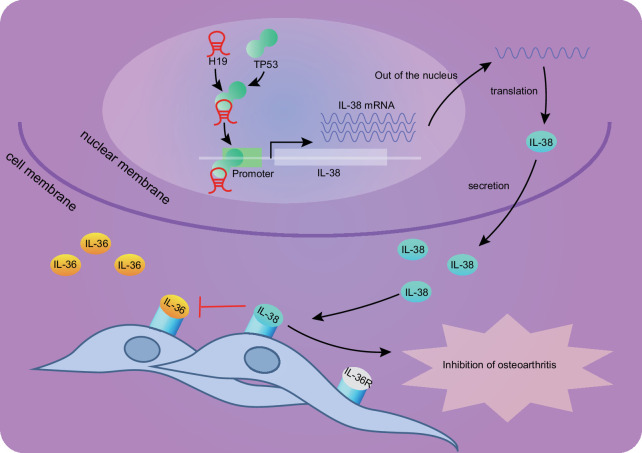
In conclusion, the current study provides evidence highlighting the protective role of lncRNA H19 against inflammatory response and chondrocyte apoptosis in OA (Figure 6), which was achieved through the upregulation of TP53, IL-38, and activation of IL-36R. Our findings could potentially serve as the foundation basis for the development of novel therapeutic options for OA, which is an area that requires further investigation. Nonetheless, the lack of pre-existing data on the potential role of lncRNA H19 or IL-38 in OA progression makes it difficult to provide further details on this topic, which therefore requires more investigations in the future.
Fig. 6.
The regulatory mechanism of the long noncoding RNA (lncRNA) H19/tumour protein p53 (TP53)/interleukin (IL)-38 axis involved in osteoarthritis (OA). lncRNA H19 was upregulated in OA. H19 upregulated the expression of IL-38 by recruiting TP53 to the IL-38 promoter region, which promoted binding of IL-38 with IL-36 receptor and inhibited the inflammatory response in OA. IL-36R, interleukin-36 receptor; mRNA, messenger RNA.
Author contributions
Y. Zhou: Writing – original draft, Methodology.
J. Li: Data curation, Formal Analysis, Methodology.
F. Xu: Conceptualization, Formal Analysis, Methodology.
E. Ji: Data curation.
C. Wang: Data curation.
Z. Pan: Conceptualization, Writing – review & editing.
Funding statement
The authors disclose receipt of the following financial or material support for the research, authorship, and/or publication of this article: funding by Zhejiang Medicine and Health Science and Technology Plan Project (2021422050) and Wenzhou Science and Technology Plan Funded Project (Y20180310).
ICMJE COI statement
The authors declare no conflict of interest.
Data sharing
The data that support the findings of this study are available on request from the corresponding author.
Acknowledgements
Many thanks to the Division of Laboratory Animal Research at Wenzhou Medical University for their patience and collaboration in the successful completion of this study. The authors gratefully acknowledge all authors whose work contributed to this review.
Ethical review statement
The current study was approved by the Ethics Committee of the First Affiliated Hospital of Wenzhou Medical University and was conducted in accordance with the Declaration of Helsinki issued in 1975. Signed written informed consents were obtained from each participant prior to the commencement of the study.
Open access funding
The authors report that they received open access funding for their manuscript from Zhejiang Medicine and Health Science and Technology Plan Project (2021422050) and Wenzhou Science and Technology Plan Funded Project (Y20180310).
Supplementary material
Figures showing: representative western blot images of interleukin (IL)-38 protein; the expression of long noncoding RNA H19, tumour protein p53, and IL-38 in osteoarthritis patients and healthy subjects; and IL-38 promoter-DNA with mouse tumour protein p53 determined by gel electrophoresis mobility shift assay. Tables showing primer sequences for and original data of quantitative reverse transcription polymerase chain reaction.
© 2022 Author(s) et al. This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial No Derivatives (CC BY-NC-ND 4.0) licence, which permits the copying and redistribution of the work only, and provided the original author and source are credited. See https://creativecommons.org/licenses/by-nc-nd/4.0/
Contributor Information
Yeli Zhou, Email: YYeli2019@163.com.
Jing Li, Email: JLingli11@163.com.
Feng Xu, Email: drxffengxu@163.com.
Encheng Ji, Email: JEEncheng@yeah.net.
Chenglong Wang, Email: wcl_chenglong@126.com.
Zheer Pan, Email: drZheer@126.com.
References
- 1. Martel-Pelletier J , Barr AJ , Cicuttini FM , et al. . Osteoarthritis . Nat Rev Dis Primers . 2016. ; 2 : 16072 . 10.1038/nrdp.2016.72 [DOI] [PubMed] [Google Scholar]
- 2. Robinson WH , Lepus CM , Wang Q , et al. . Low-grade inflammation as a key mediator of the pathogenesis of osteoarthritis . Nat Rev Rheumatol . 2016. ; 12 ( 10 ): 580 – 592 . 10.1038/nrrheum.2016.136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Sansone V , Applefield RC , De Luca P , et al. . Does a high-fat diet affect the development and progression of osteoarthritis in mice?: A systematic review . Bone Joint Res . 2019. ; 8 ( 12 ): 582 – 592 . 10.1302/2046-3758.812.BJR-2019-0038.R1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Nelson AE . Osteoarthritis year in review 2017: clinical . Osteoarthr Cartil . 2018. ; 26 ( 3 ): 319 – 325 . 10.1016/j.joca.2017.11.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Thysen S , Luyten FP , Lories RJU . Targets, models and challenges in osteoarthritis research . Dis Model Mech . 2015. ; 8 ( 1 ): 17 – 30 . 10.1242/dmm.016881 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Scanzello CR . Role of low-grade inflammation in osteoarthritis . Curr Opin Rheumatol . 2017. ; 29 ( 1 ): 79 – 85 . 10.1097/BOR.0000000000000353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Liu-Bryan R , Terkeltaub R . Emerging regulators of the inflammatory process in osteoarthritis . Nat Rev Rheumatol . 2015. ; 11 ( 1 ): 35 – 44 . 10.1038/nrrheum.2014.162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Atianand MK , Caffrey DR , Fitzgerald KA . Immunobiology of long noncoding RNAs . Annu Rev Immunol . 2017. ; 35 : 177 – 198 . 10.1146/annurev-immunol-041015-055459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Chen YG , Satpathy AT , Chang HY . Gene regulation in the immune system by long noncoding RNAs . Nat Immunol . 2017. ; 18 ( 9 ): 962 – 972 . 10.1038/ni.3771 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Nanus DE , Wijesinghe SN , Pearson MJ , et al. . Regulation of the inflammatory synovial fibroblast phenotype by metastasis-associated lung adenocarcinoma transcript 1 long noncoding RNA in obese patients with osteoarthritis . Arthritis Rheumatol . 2020. ; 72 ( 4 ): 609 – 619 . 10.1002/art.41158 [DOI] [PubMed] [Google Scholar]
- 11. Pearson MJ , Philp AM , Heward JA , et al. . Long intergenic noncoding RNAs mediate the human chondrocyte inflammatory response and are differentially expressed in osteoarthritis cartilage . Arthritis Rheumatol . 2016. ; 68 ( 4 ): 845 – 856 . 10.1002/art.39520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Azzi S , Abi Habib W , Netchine I . Beckwith-Wiedemann and Russell-Silver Syndromes: from new molecular insights to the comprehension of imprinting regulation . Curr Opin Endocrinol Diabetes Obes . 2014. ; 21 ( 1 ): 30 – 38 . 10.1097/MED.0000000000000037 [DOI] [PubMed] [Google Scholar]
- 13. Geng H , Bu HF , Liu F , et al. . In inflamed intestinal tissues and epithelial cells, interleukin 22 signaling increases expression of H19 long noncoding RNA, which promotes mucosal regeneration . Gastroenterology . 2018. ; 155 ( 1 ): 144 – 155 . 10.1053/j.gastro.2018.03.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Steck E , Boeuf S , Gabler J , et al. . Regulation of H19 and its encoded microRNA-675 in osteoarthritis and under anabolic and catabolic in vitro conditions . J Mol Med (Berl) . 2012. ; 90 ( 10 ): 1185 – 1195 . 10.1007/s00109-012-0895-y [DOI] [PubMed] [Google Scholar]
- 15. Xing D , Liang J , Li Y , et al. . Identification of long noncoding RNA associated with osteoarthritis in humans . Orthop Surg . 2014. ; 6 ( 4 ): 288 – 293 . 10.1111/os.12147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Liu C , Chen Z , Fang J , Xu A , Zhang W , Wang Z . H19-derived miR-675 contributes to bladder cancer cell proliferation by regulating p53 activation . Tumour Biol . 2016. ; 37 ( 1 ): 263 – 270 . 10.1007/s13277-015-3779-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Chen Z , Guo J , Zhang K , Guo Y . TP53 mutations and survival in osteosarcoma patients: a meta-analysis of published data . Dis Markers . 2016. ; 2016 : 4639575 . 10.1155/2016/4639575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Eun K , Park MG , Jeong YW , et al. . Establishment of TP53-knockout canine cells using optimized CRIPSR/Cas9 vector system for canine cancer research . BMC Biotechnol . 2019. ; 19 ( 1 ): 1 . 10.1186/s12896-018-0491-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Zhu X , Yang S , Lin W , et al. . Roles of cell cyle regulators cyclin D1, CDK4, and p53 in knee osteoarthritis . Genet Test Mol Biomarkers . 2016. ; 20 ( 9 ): 529 – 534 . 10.1089/gtmb.2016.0020 [DOI] [PubMed] [Google Scholar]
- 20. Igarashi H , Hashimoto J , Tomita T , Yoshikawa H , Ishihara K . TP53 mutations coincide with the ectopic expression of activation-induced cytidine deaminase in the fibroblast-like synoviocytes derived from a fraction of patients with rheumatoid arthritis . Clin Exp Immunol . 2010. ; 161 ( 1 ): 71 – 80 . 10.1111/j.1365-2249.2010.04163.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ubertini V , Norelli G , D’Arcangelo D , et al. . Mutant p53 gains new function in promoting inflammatory signals by repression of the secreted interleukin-1 receptor antagonist . Oncogene . 2015. ; 34 ( 19 ): 2493 – 2504 . 10.1038/onc.2014.191 [DOI] [PubMed] [Google Scholar]
- 22. Garlanda C , Dinarello CA , Mantovani A . The interleukin-1 family: back to the future . Immunity . 2013. ; 39 ( 6 ): 1003 – 1018 . 10.1016/j.immuni.2013.11.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Xu WD , Su LC , He CS , Huang AF . Plasma interleukin-38 in patients with rheumatoid arthritis . Int Immunopharmacol . 2018. ; 65 : 1 – 7 . 10.1016/j.intimp.2018.09.028 [DOI] [PubMed] [Google Scholar]
- 24. Yuan X , Peng X , Li Y , Li M . Role of IL-38 and its related cytokines in inflammation . Mediators Inflamm . 2015. ; 2015 : 807976 . 10.1155/2015/807976 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Boutet MA , Najm A , Bart G , et al. . IL-38 overexpression induces anti-inflammatory effects in mice arthritis models and in human macrophages in vitro . Ann Rheum Dis . 2017. ; 76 ( 7 ): 1304 – 1312 . 10.1136/annrheumdis-2016-210630 [DOI] [PubMed] [Google Scholar]
- 26. Ummarino D . Experimental arthritis: IL-38 promotes anti-inflammatory effects . Nat Rev Rheumatol . 2017. ; 13 ( 5 ): 260 . 10.1038/nrrheum.2017.55 [DOI] [PubMed] [Google Scholar]
- 27. Frey S , Derer A , Messbacher ME , et al. . The novel cytokine interleukin-36α is expressed in psoriatic and rheumatoid arthritis synovium . Ann Rheum Dis . 2013. ; 72 ( 9 ): 1569 – 1574 . 10.1136/annrheumdis-2012-202264 [DOI] [PubMed] [Google Scholar]
- 28. Glasson SS , Chambers MG , Van Den Berg WB , Little CB . The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the mouse . Osteoarthr Cartil . 2010. ; 18 Suppl 3 : S17 - 23 . 10.1016/j.joca.2010.05.025 [DOI] [PubMed] [Google Scholar]
- 29. Shen J , Abu-Amer Y , O’Keefe RJ , McAlinden A . Inflammation and epigenetic regulation in osteoarthritis . Connect Tissue Res . 2017. ; 58 ( 1 ): 49 – 63 . 10.1080/03008207.2016.1208655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Messeguer X , Escudero R , Farré D , Núñez O , Martínez J , Albà MM . PROMO: detection of known transcription regulatory elements using species-tailored searches . Bioinformatics . 2002. ; 18 ( 2 ): 333 – 334 . 10.1093/bioinformatics/18.2.333 [DOI] [PubMed] [Google Scholar]
- 31. Weirauch MT , Yang A , Albu M , et al. . Determination and inference of eukaryotic transcription factor sequence specificity . Cell . 2014. ; 158 ( 6 ): 1431 – 1443 . 10.1016/j.cell.2014.08.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Wang Y , He L , Du Y , et al. . The long noncoding RNA lncTCF7 promotes self-renewal of human liver cancer stem cells through activation of Wnt signaling . Cell Stem Cell . 2015. ; 16 ( 4 ): 413 – 425 . 10.1016/j.stem.2015.03.003 [DOI] [PubMed] [Google Scholar]
- 33. Ning L , Cui T , Zheng B , et al. . MNDR v3.0: mammal ncRNA-disease repository with increased coverage and annotation . Nucleic Acids Res . 2021. ; 49 ( D1 ): D160 – D164 . 10.1093/nar/gkaa707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Yi Y , Zhao Y , Li C , et al. . RAID v2.0: an updated resource of RNA-associated interactions across organisms . Nucleic Acids Res . 2017. ; 45 ( D1 ): D115 – D118 . 10.1093/nar/gkw1052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. van de Veerdonk FL , Stoeckman AK , Wu G , et al. . IL-38 binds to the IL-36 receptor and has biological effects on immune cells similar to IL-36 receptor antagonist . Proc Natl Acad Sci U S A . 2012. ; 109 ( 8 ): 3001 – 3005 . 10.1073/pnas.1121534109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Mobasheri A , Bay-Jensen A-C , van Spil WE , Larkin J , Levesque MC . Osteoarthritis Year in Review 2016: biomarkers (biochemical markers) . Osteoarthr Cartil . 2017. ; 25 ( 2 ): 199 – 208 . 10.1016/j.joca.2016.12.016 [DOI] [PubMed] [Google Scholar]
- 37. Hausmann LRM , Youk A , Kwoh CK , et al. . Testing a positive psychological intervention for osteoarthritis . Pain Med . 2017. ; 18 ( 10 ): 1908 – 1920 . 10.1093/pm/pnx141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Tan F , Wang D , Yuan Z . The fibroblast-like synoviocyte derived exosomal long non-coding RNA H19 alleviates osteoarthritis progression through the miR-106b-5p/TIMP2 axis . Inflammation . 2020. ; 43 ( 4 ): 1498 – 1509 . 10.1007/s10753-020-01227-8 [DOI] [PubMed] [Google Scholar]
- 39. Runge S , Nielsen FC , Nielsen J , Lykke-Andersen J , Wewer UM , Christiansen J . H19 RNA binds four molecules of insulin-like growth factor II mRNA-binding protein . J Biol Chem . 2000. ; 275 ( 38 ): 29562 – 29569 . 10.1074/jbc.M001156200 [DOI] [PubMed] [Google Scholar]
- 40. Liang WC , Fu WM , Wong CW , et al. . The lncRNA H19 promotes epithelial to mesenchymal transition by functioning as miRNA sponges in colorectal cancer . Oncotarget . 2015. ; 6 ( 26 ): 22513 – 22525 . 10.18632/oncotarget.4154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Xie L , Huang Z , Li H , Liu X , Zheng S , Su W . IL-38: a new player in inflammatory autoimmune disorders . Biomolecules . 2019. ; 9 ( 8 ): E345 . 10.3390/biom9080345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Boutet MA , Bart G , Penhoat M , et al. . Distinct expression of interleukin (IL)-36α, β and γ, their antagonist IL-36Ra and IL-38 in psoriasis, rheumatoid arthritis and Crohn’s disease . Clin Exp Immunol . 2016. ; 184 ( 2 ): 159 – 173 . 10.1111/cei.12761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Saberi Hosnijeh F , Siebuhr AS , Uitterlinden AG , et al. . Association between biomarkers of tissue inflammation and progression of osteoarthritis: evidence from the Rotterdam study cohort . Arthritis Res Ther . 2016. ; 18 : 81 . 10.1186/s13075-016-0976-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Zhou S , Thornhill TS , Meng F , Xie L , Wright J , Glowacki J . Influence of osteoarthritis grade on molecular signature of human cartilage . J Orthop Res . 2016. ; 34 ( 3 ): 454 – 462 . 10.1002/jor.23043 [DOI] [PubMed] [Google Scholar]
- 45. Samavedi S , Diaz-Rodriguez P , Erndt-Marino JD , Hahn MS . A three-dimensional chondrocyte-macrophage coculture system to probe inflammation in experimental osteoarthritis . Tissue Eng Part A . 2017. ; 23 ( 3–4 ): 101 – 114 . 10.1089/ten.TEA.2016.0007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Akdis M , Aab A , Altunbulakli C , et al. . Interleukins (from IL-1 to IL-38), interferons, transforming growth factor β, and TNF-α: Receptors, functions, and roles in diseases . J Allergy Clin Immunol . 2016. ; 138 ( 4 ): 984 – 1010 . 10.1016/j.jaci.2016.06.033 [DOI] [PubMed] [Google Scholar]
- 47. Li J , Liu L , Rui W , et al. . New interleukins in psoriasis and psoriatic arthritis patients: the possible roles of interleukin-33 to interleukin-38 in disease activities and bone erosions . Dermatology . 2017. ; 233 ( 1 ): 37 – 46 . 10.1159/000471798 [DOI] [PubMed] [Google Scholar]
- 48. Garraud T , Harel M , Boutet MA , Le Goff B , Blanchard F . The enigmatic role of IL-38 in inflammatory diseases . Cytokine Growth Factor Rev . 2018. ; 39 : 26 – 35 . 10.1016/j.cytogfr.2018.01.001 [DOI] [PubMed] [Google Scholar]
- 49. Mora J , Schlemmer A , Wittig I , et al. . Interleukin-38 is released from apoptotic cells to limit inflammatory macrophage responses . J Mol Cell Biol . 2016. ; 8 ( 5 ): 426 – 438 . 10.1093/jmcb/mjw006 [DOI] [PubMed] [Google Scholar]
- 50. Su P , Wang F , Qi B , Wang T , Zhang S . P53 regulation-association long non-coding RNA (LncRNA PRAL) inhibits cell proliferation by regulation of P53 in human lung cancer . Med Sci Monit . 2017. ; 23 : 1751 – 1758 . 10.12659/msm.900205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Melo CA , Léveillé N , Rooijers K , et al. . A p53-bound enhancer region controls a long intergenic noncoding RNA required for p53 stress response . Oncogene . 2016. ; 35 ( 33 ): 4399 – 4406 . 10.1038/onc.2015.502 [DOI] [PubMed] [Google Scholar]
- 52. Yang S , Tang W , He Y , Wen L , Sun B , Li S . Long non-coding RNA and microRNA-675/let-7a mediates the protective effect of melatonin against early brain injury after subarachnoid hemorrhage via targeting TP53 and neural growth factor . Cell Death Dis . 2018. ; 9 ( 2 ): 99 . 10.1038/s41419-017-0155-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Tak PP , Smeets TJ , Boyle DL , et al. . p53 overexpression in synovial tissue from patients with early and longstanding rheumatoid arthritis compared with patients with reactive arthritis and osteoarthritis . Arthritis Rheum . 1999. ; 42 ( 5 ): 948 – 953 . [DOI] [PubMed] [Google Scholar]
- 54. Wellenstein MD , Coffelt SB , Duits DEM , et al. . Loss of p53 triggers WNT-dependent systemic inflammation to drive breast cancer metastasis . Nature . 2019. ; 572 ( 7770 ): 538 – 542 . 10.1038/s41586-019-1450-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Conti P , Lessiani G , Kritas SK , Ronconi G , Caraffa A , Theoharides TC . Mast cells emerge as mediators of atherosclerosis: Special emphasis on IL-37 inhibition . Tissue Cell . 2017. ; 49 ( 3 ): 393 – 400 . 10.1016/j.tice.2017.04.002 [DOI] [PubMed] [Google Scholar]
- 56. Dinarello CA . Overview of the IL-1 family in innate inflammation and acquired immunity . Immunol Rev . 2018. ; 281 ( 1 ): 8 – 27 . 10.1111/imr.12621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Sims JE , Smith DE . The IL-1 family: regulators of immunity . Nat Rev Immunol . 2010. ; 10 ( 2 ): 89 – 102 . 10.1038/nri2691 [DOI] [PubMed] [Google Scholar]
- 58. Boutet MA , Nerviani A , Pitzalis C . IL-36, IL-37, and IL-38 cytokines in skin and joint inflammation: a comprehensive review of their therapeutic potential . Int J Mol Sci . 2019. ; 20 ( 6 ): 1257 . 10.3390/ijms20061257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Takaishi M , Satoh T , Akira S , Sano S . Regnase-1, an immunomodulator, limits the IL-36/IL-36R autostimulatory loop in keratinocytes to suppress skin inflammation . J Invest Dermatol . 2018. ; 138 ( 6 ): 1439 – 1442 . 10.1016/j.jid.2017.12.033 [DOI] [PubMed] [Google Scholar]
- 60. Li T , Chubinskaya S , Esposito A , et al. . TGF-β type 2 receptor-mediated modulation of the IL-36 family can be therapeutically targeted in osteoarthritis . Sci Transl Med . 2019. ; 11 ( 491 ): eaan2585 . 10.1126/scitranslmed.aan2585 [DOI] [PMC free article] [PubMed] [Google Scholar]